Feb 9, 2026

You are wasting peptide. Right now, probably. A study of compounded medication errors found that concentration miscalculations account for a significant percentage of dosing failures, and compounded tirzepatide sits right at the center of this problem. The vial says 10mg. The syringe reads in units. The chart your provider gave you assumes a concentration your pharmacy did not use. And somewhere between milligrams, milliliters, and units, the math breaks down. The result is not just wasted product. It is wasted time, wasted money, and wasted potential for the weight loss results that brought you to tirzepatide in the first place.
Here is the uncomfortable truth. Compounded tirzepatide arrives in concentrations that vary wildly between pharmacies. One sends a 5mg/mL solution. Another ships 10mg/mL. A third compounds at 20mg/mL. The dosage in units changes completely depending on which concentration you received, and using the wrong chart for the wrong concentration means every single injection is off target.
This guide eliminates that confusion. Every concentration. Every dose. Every conversion from milligrams to milliliters to syringe units, mapped out in charts you can actually use. Whether you are getting started with compounded tirzepatide or switching between concentrations mid-protocol, the numbers are here. No guesswork. No estimation. No wasted peptide. SeekPeptides built this reference because accurate dosing is not optional when the compound sitting in your refrigerator represents a significant investment in your fat loss goals.
Understanding compounded tirzepatide concentrations
Before touching a syringe, you need to understand one concept that determines everything else about your dosing. Concentration. It is the single most important number on your vial, and getting it wrong cascades into every dose you draw.
Concentration tells you how many milligrams of tirzepatide are dissolved in each milliliter of solution. A vial labeled 10mg/mL contains 10 milligrams of active tirzepatide per milliliter of liquid. A 20mg/mL vial contains twice as much medication in the same volume. This means that drawing the same number of units from a 20mg/mL vial delivers double the dose compared to a 10mg/mL vial. Double. That is not a rounding error or a minor variation. That is the difference between a therapeutic dose and a potentially problematic one.
Compounding pharmacies produce tirzepatide in several standard concentrations. The three most common are 5mg/mL, 10mg/mL, and 20mg/mL. Some pharmacies also compound at non-standard concentrations like 8mg/mL or 12.5mg/mL, which makes accurate calculation even more critical. Your provider should always specify which concentration was prescribed. If your vial arrives without clear labeling, contact the pharmacy before using it.
The concentration also determines how many doses each vial contains. A 30mg vial reconstituted to 10mg/mL yields 3mL of solution, enough for several weeks of lower doses. That same 30mg vial at 5mg/mL yields 6mL, twice the volume but the same total medication. Understanding this relationship helps you plan your protocol timeline and know exactly when you will need a refill.
Why concentration matters more than total vial size
Many people fixate on the total milligrams in their vial. They see "30mg" and assume all 30mg vials are identical. They are not. A 30mg vial at 10mg/mL contains 3mL of solution. A 30mg vial at 5mg/mL contains 6mL. The total tirzepatide is the same, but the volume and the number of units per dose are completely different.
This distinction trips up even experienced users. Someone switching pharmacies might receive the same "30mg vial" but at a different concentration. If they continue drawing the same number of units per dose, they will either underdose or overdose, depending on which direction the concentration changed. The most common mistake in compounded tirzepatide dosing is applying one concentration chart to a different concentration vial.
How compounded differs from pre-filled brand pens
Brand name tirzepatide products like Mounjaro and Zepbound come in pre-filled injection pens. Each pen delivers a fixed dose. No math required. No syringes to fill. No concentration to calculate. The pen does the work.
Compounded tirzepatide is fundamentally different. It arrives as either a pre-mixed liquid in a multi-dose vial or as lyophilized powder that requires reconstitution before use. Either way, you must draw the correct volume using a syringe, and that correct volume depends entirely on the concentration. This manual process introduces the possibility of error at every step, from reading the concentration label to selecting the right syringe to drawing the correct number of units. The standard dose chart for brand products does not apply to compounded formulations unless the concentration matches exactly.
Compounded formulations also lack the extensive clinical trial data backing brand products. The SURMOUNT-1 trial showed that FDA-approved tirzepatide produced average weight loss of 15% at 5mg, 19.5% at 10mg, and 20.9% at 15mg over 72 weeks. While the active ingredient is the same, compounded versions have not been through equivalent testing for safety and consistency. Understanding exactly what you are taking, and exactly how much, becomes even more important.
Complete compounded tirzepatide dosage chart by concentration
These charts convert your prescribed milligram dose into the exact number of syringe units you need to draw, based on your specific vial concentration. Find your concentration first. Then find your dose. The units column tells you exactly where to fill your insulin syringe. These numbers assume a standard U-100 insulin syringe where 100 units equals 1mL.
5mg/mL concentration dosage chart
The 5mg/mL concentration is the most dilute common formulation. It requires the largest injection volumes, which means you may need a larger syringe for higher doses. The advantage is that small dose adjustments are easier to measure because each unit represents a smaller amount of medication.
Weekly dose (mg) | Volume (mL) | Insulin syringe units |
|---|---|---|
2.5 mg | 0.50 mL | 50 units |
5 mg | 1.00 mL | 100 units |
7.5 mg | 1.50 mL | 150 units |
10 mg | 2.00 mL | 200 units |
12.5 mg | 2.50 mL | 250 units |
15 mg | 3.00 mL | 300 units |
Notice that at 5mg/mL, even the starting dose of 2.5mg requires 50 units. By the time you reach higher doses, you exceed the capacity of a standard 1mL insulin syringe. Doses above 5mg at this concentration require either a larger syringe or splitting the injection into two draws. Many users find this concentration inconvenient for maintenance dosing, which is one reason higher concentrations exist. If you are new to peptide injections, the larger volumes can actually be easier to measure accurately at the cost of a slightly larger injection.
10mg/mL concentration dosage chart
The 10mg/mL concentration hits the sweet spot for most users. All standard doses from 2.5mg through 10mg fit within a single 1mL insulin syringe, and the unit markings are clean, round numbers that are easy to read. This is the concentration most compounding pharmacies default to, and most dosing instructions you find online assume this concentration unless stated otherwise.
Weekly dose (mg) | Volume (mL) | Insulin syringe units |
|---|---|---|
2.5 mg | 0.25 mL | 25 units |
5 mg | 0.50 mL | 50 units |
7.5 mg | 0.75 mL | 75 units |
10 mg | 1.00 mL | 100 units |
12.5 mg | 1.25 mL | 125 units |
15 mg | 1.50 mL | 150 units |
At 10mg/mL, the math is straightforward. Each unit on your insulin syringe delivers 0.1mg of tirzepatide. The relationship between units and milligrams becomes simple multiplication. Twenty units equals 2mg. Fifty units equals 5mg. This clean math makes dose adjustments and microdosing protocols much simpler to implement.
For the standard dose escalation, you start at 25 units (2.5mg) and increase by 25 units every four weeks. The progression goes 25, 50, 75, 100 units, each step representing an additional 2.5mg. This predictable pattern is one of the reasons the 10mg/mL concentration is so popular for compounded tirzepatide.
20mg/mL concentration dosage chart
The 20mg/mL concentration packs twice the medication into each milliliter. This means smaller injection volumes, which some users prefer. But it also means that small measurement errors translate to larger dosing mistakes. If you accidentally draw 5 extra units at 20mg/mL, you have added 1mg to your dose. The same error at 10mg/mL only adds 0.5mg. Precision matters more at higher concentrations.
Weekly dose (mg) | Volume (mL) | Insulin syringe units |
|---|---|---|
2.5 mg | 0.125 mL | 12.5 units |
5 mg | 0.25 mL | 25 units |
7.5 mg | 0.375 mL | 37.5 units |
10 mg | 0.50 mL | 50 units |
12.5 mg | 0.625 mL | 62.5 units |
15 mg | 0.75 mL | 75 units |
The half-unit measurements at 20mg/mL create a practical challenge. Standard insulin syringes mark whole units, so hitting exactly 12.5 or 37.5 units requires careful estimation between lines. Some users choose syringes with half-unit markings for this reason. Others round to the nearest whole unit, accepting a slight variation. If your pharmacy compounds at 20mg/mL and you switched from a different peptide concentration, take extra care with the conversion.
The main advantage of 20mg/mL is efficiency. Every dose through the maximum 15mg fits easily within a 1mL syringe. You use less syringe volume per dose, and each vial lasts longer in terms of injection count. For users on maintenance doses who have stabilized their protocol, this concentration minimizes injection volume while maximizing vial longevity.
How to reconstitute compounded tirzepatide
Some compounded tirzepatide arrives as a pre-mixed liquid, ready to draw and inject. Others arrive as lyophilized powder, a freeze-dried form that requires mixing with bacteriostatic water before use. If your vial contains powder, reconstitution is the first step, and it determines the concentration you will be working with for every dose thereafter.
Getting reconstitution right matters. Add too little water and the concentration becomes dangerously high. Add too much and you dilute the solution beyond what standard syringes can accurately measure for your target dose. The goal is a concentration that allows precise, convenient dosing throughout your entire escalation schedule.
Reconstitution ratios by vial size
The general guideline follows a simple ratio. For a 10mg/mL final concentration, add 1mL of bacteriostatic water per 10mg of tirzepatide powder. For a 5mg/mL concentration, double the water. Here are the specific ratios for common vial sizes, targeting the most practical concentrations.
Vial size | Bacteriostatic water added | Final concentration | Total solution volume |
|---|---|---|---|
5 mg | 0.5 mL | 10 mg/mL | 0.5 mL |
5 mg | 1.0 mL | 5 mg/mL | 1.0 mL |
10 mg | 1.0 mL | 10 mg/mL | 1.0 mL |
10 mg | 2.0 mL | 5 mg/mL | 2.0 mL |
15 mg | 1.5 mL | 10 mg/mL | 1.5 mL |
30 mg | 3.0 mL | 10 mg/mL | 3.0 mL |
30 mg | 1.5 mL | 20 mg/mL | 1.5 mL |
60 mg | 6.0 mL | 10 mg/mL | 6.0 mL |
60 mg | 3.0 mL | 20 mg/mL | 3.0 mL |
The peptide reconstitution calculator can handle these calculations for any vial size and desired concentration. Simply enter your total peptide amount and target concentration, and it returns the exact water volume needed. For users who want to verify the math manually, the formula is straightforward. Divide the total milligrams by your desired concentration in mg/mL. The result is the volume of bacteriostatic water to add. A 30mg vial at a target of 10mg/mL requires 30 divided by 10, which equals 3mL of water.
Step by step reconstitution process
Gather your supplies first. You need the tirzepatide vial, a vial of bacteriostatic water, alcohol swabs, and a sterile syringe with needle for drawing and transferring the water. A separate syringe from your injection syringes works best for this step.
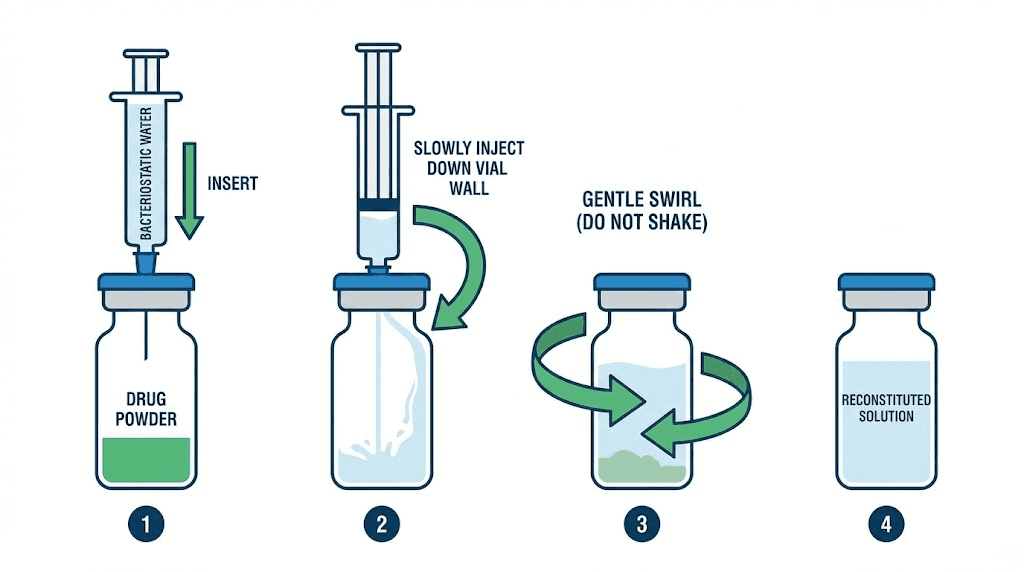
Clean both vial stoppers with alcohol swabs and let them air dry for thirty seconds. Draw the calculated amount of bacteriostatic water into your syringe. Insert the needle through the tirzepatide vial stopper at a slight angle. Here is the critical part. Inject the water slowly down the inside wall of the vial. Do not spray it directly onto the powder. Direct contact with a forceful stream causes foaming and can damage the peptide. Let the water run gently down the glass.
Remove the needle. Gently swirl the vial between your fingers. Do not shake it. Shaking creates air bubbles and can denature the tirzepatide through physical agitation. A gentle rolling motion, tilting the vial slowly back and forth, dissolves the powder within one to two minutes. The solution should become clear and colorless. If particles remain, continue gentle swirling. Do not use the solution if it remains cloudy after five minutes of gentle mixing, as this may indicate degradation.
Once dissolved, the reconstituted solution is ready for use. Label the vial with the date, concentration, and total volume. This simple step prevents confusion later, especially if you have multiple vials or use other peptides alongside tirzepatide. Proper reconstitution technique preserves potency and ensures every dose delivers what you expect.
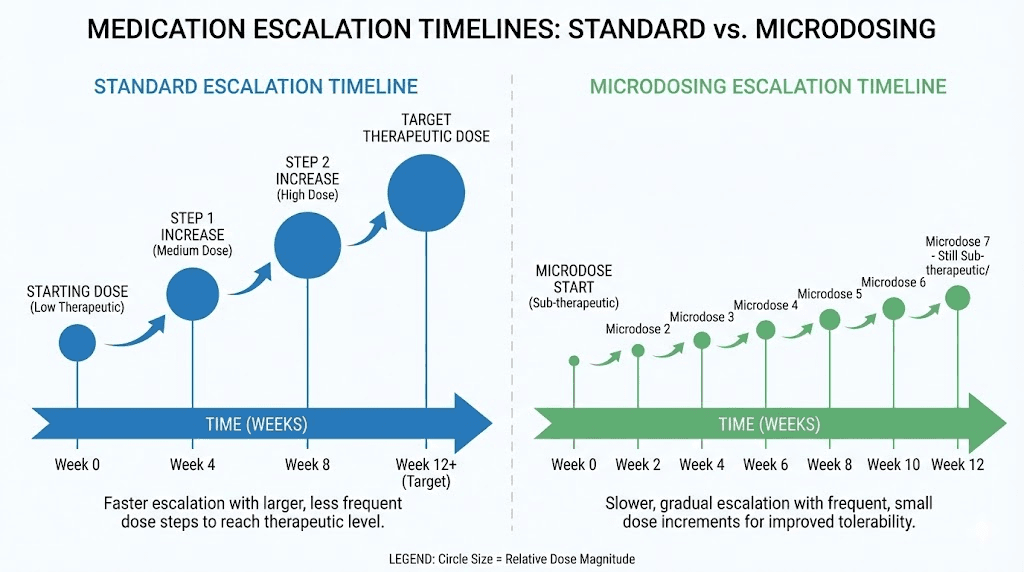
The standard dose escalation schedule
Tirzepatide works through gradual dose escalation. You do not start at your target dose. The body needs time to adapt to the dual GIP and GLP-1 receptor activation, and jumping to a higher dose invites unnecessary side effects. The FDA-approved escalation schedule, which serves as the baseline for compounded protocols, moves in 2.5mg increments every four weeks.
This slow build exists for good reason. Clinical trials demonstrated that patients who followed gradual escalation experienced significantly fewer gastrointestinal side effects compared to those on faster schedules. The body adapts to the appetite-suppressing and gastric-slowing effects over time, and each four-week period allows that adaptation to occur before the next increase.
Week by week escalation timeline
Phase | Weeks | Weekly dose | Units at 10mg/mL | Purpose |
|---|---|---|---|---|
Starting | 1-4 | 2.5 mg | 25 units | Assess tolerance, begin GI adaptation |
Escalation 1 | 5-8 | 5 mg | 50 units | First therapeutic dose for most |
Escalation 2 | 9-12 | 7.5 mg | 75 units | Intermediate dose, monitor response |
Escalation 3 | 13-16 | 10 mg | 100 units | Higher therapeutic range |
Escalation 4 | 17-20 | 12.5 mg | 125 units | Near-maximum dose |
Maximum | 21+ | 15 mg | 150 units | Maximum approved dose |
Not everyone needs to reach 15mg. The SURMOUNT-1 trial showed meaningful weight loss at every dose level, with 5mg producing an average 15% body weight reduction over 72 weeks. Some users find their optimal balance of results and tolerability at 7.5mg or 10mg and maintain there long-term. The escalation schedule provides a framework, not a mandate. Your response, your rate of progress, and your side effect profile should guide when and whether to increase.
The numbers from clinical data tell a compelling story. At the 5mg dose, 85% of participants lost at least 5% of their body weight. At 10mg, that number rose to 89%. At 15mg, 91% achieved that threshold, and 57% lost 20% or more. Perhaps most encouraging, 90% of participants who were slow responders, those who had not lost 5% by week 12, went on to achieve clinically meaningful weight loss by week 72. Patience with the escalation process pays off.
When to slow down or hold a dose
The four-week intervals between dose increases are minimums, not deadlines. If you are experiencing significant fatigue, persistent nausea, or other side effects that have not resolved before your scheduled increase, staying at your current dose for an additional two to four weeks is both safe and sensible.
Signs you should hold your current dose include nausea lasting more than three days after injection, vomiting more than once per week, inability to maintain adequate hydration, or significant disruption to daily activities. These signals mean your body needs more time to adapt at the current level. Pushing through with a dose increase typically makes symptoms worse, not better, and can lead to treatment discontinuation, which defeats the entire purpose of the graduated approach.
Conversely, if you are tolerating your current dose well and seeing steady results, there may be no reason to increase at all. The goal is finding the lowest effective dose that produces meaningful progress. Lower doses mean fewer side effects, lower cost, and a longer vial lifespan. A researcher seeing consistent weekly weight loss at 5mg does not need to escalate to 10mg simply because the schedule says to. Monitor your progress and results at each level before deciding whether an increase is warranted.
Insulin syringe vs tuberculin syringe for compounded tirzepatide
This is where dosing errors get dangerous. There are two types of syringes commonly used for subcutaneous injections, and they measure liquid differently. Using the wrong one, or reading the wrong scale, can result in dramatically incorrect doses.
An insulin syringe uses a U-100 scale where 100 units equals 1mL. The markings on the barrel read in units, typically going from 0 to 100 in increments of 1 or 2. When your dosing chart says "draw 50 units," you fill to the 50-unit line on an insulin syringe.
A tuberculin syringe measures in milliliters. It typically holds 1mL total, marked in increments of 0.01mL. There are no "unit" markings. When your dosing chart says "draw 0.5mL," you fill to the 0.5mL line on a tuberculin syringe.
Here is the critical point. The numbers on these two syringes are not interchangeable. Drawing to the "50" line on an insulin syringe gives you 0.5mL. Drawing to the "50" line on a tuberculin syringe, if one existed at that scale, would mean something completely different. If your dosing instructions are written in units, use an insulin syringe. If they are written in milliliters, use a tuberculin syringe. Mixing these up is one of the most common and serious dosing mistakes with compounded medications.
For compounded tirzepatide, most providers and pharmacies give instructions in units, assuming a U-100 insulin syringe. All of the charts in this guide use the same assumption. If your provider has given you instructions in milliliters instead, use the mL column in the dosage charts above and a tuberculin syringe. Either approach is valid as long as the syringe type matches the measurement system. The fundamentals of peptide injection apply regardless of syringe type, but the measurement system must be consistent.
When in doubt, confirm with your provider. Ask specifically which syringe type they recommend for your concentration and which measurement system their dosing instructions use. This single clarification prevents the most common source of compounded tirzepatide dosing errors.
Converting between concentrations
Pharmacy changes, supply issues, or provider adjustments sometimes mean switching from one tirzepatide concentration to another mid-protocol. This requires recalculating your dose in units, because the same milligram dose requires a different volume at each concentration. The formula is simple, but failing to apply it leads to either underdosing or overdosing.
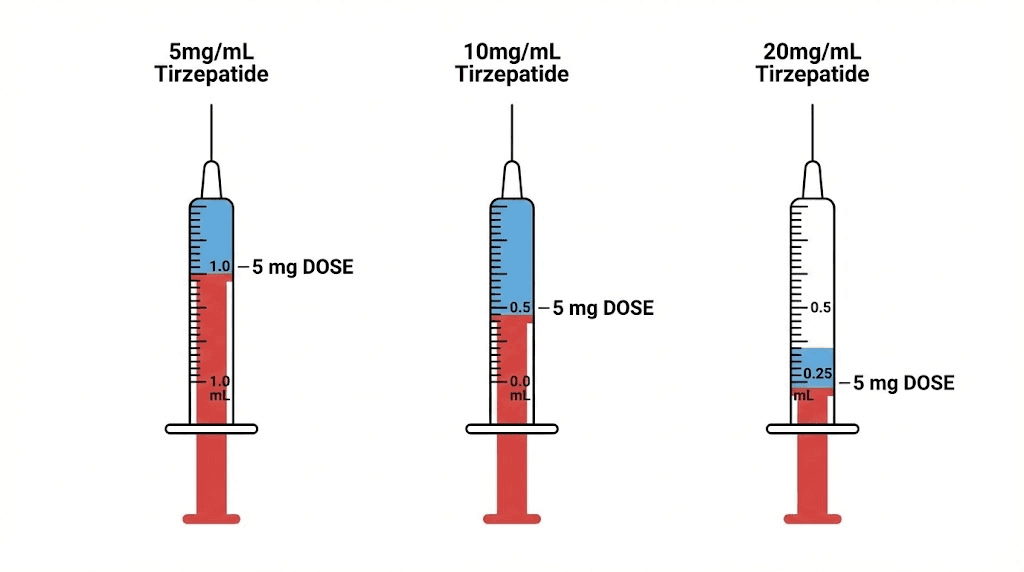
The conversion formula works like this. Divide your prescribed dose in milligrams by your new concentration in mg/mL. Multiply the result by 100 to convert to insulin syringe units. For example, a 5mg dose from a 20mg/mL vial requires 5 divided by 20, which equals 0.25mL, times 100, which equals 25 units. The same 5mg dose from a 10mg/mL vial requires 5 divided by 10, which equals 0.5mL, times 100, which equals 50 units.
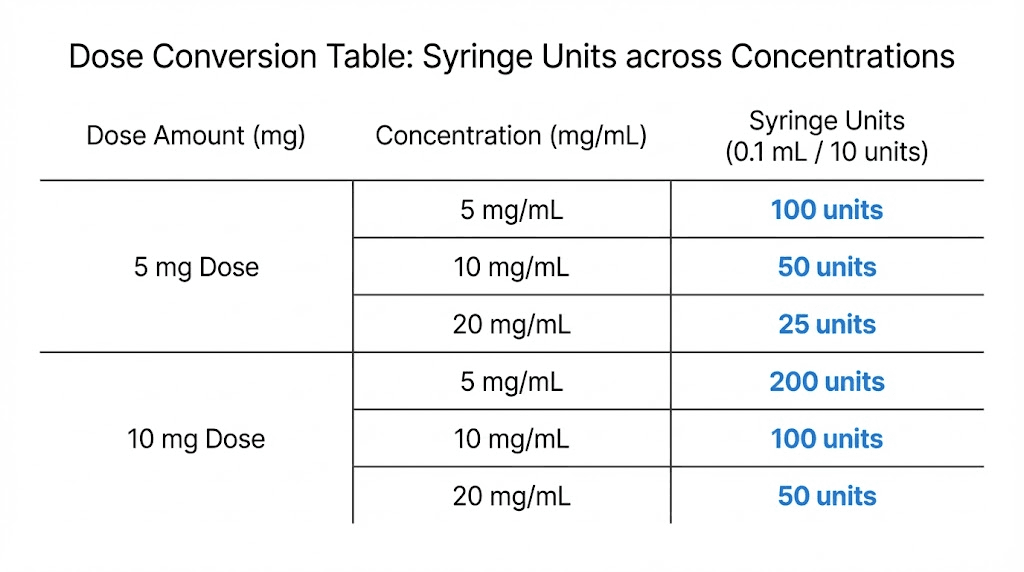
Dose (mg) | Units at 5mg/mL | Units at 10mg/mL | Units at 20mg/mL |
|---|---|---|---|
2.5 mg | 50 units | 25 units | 12.5 units |
5 mg | 100 units | 50 units | 25 units |
7.5 mg | 150 units | 75 units | 37.5 units |
10 mg | 200 units | 100 units | 50 units |
12.5 mg | 250 units | 125 units | 62.5 units |
15 mg | 300 units | 150 units | 75 units |
This cross-reference table belongs on your refrigerator, next to your vials. If you ever switch concentrations, a quick glance confirms your new unit count without recalculating. The peptide calculator at SeekPeptides can also perform these conversions instantly for any concentration, including non-standard ones that some pharmacies use.
One critical rule when switching. Always verify the concentration on your new vial before drawing any dose. Do not assume it matches your previous vial. Even the same pharmacy can switch concentrations between batches. Read the label every single time. This takes two seconds and prevents the kind of dosing error that can disrupt weeks of careful progress, undermine your dosing protocol, and create unnecessary side effects.
Storage and stability after reconstitution
Proper storage is not a suggestion. It is a requirement that directly affects the potency and safety of every dose you draw from that vial. Tirzepatide is a peptide, and peptides degrade when exposed to heat, light, or bacterial contamination. Your storage protocol determines whether the last dose from a vial is as effective as the first.
Reconstituted tirzepatide should be refrigerated at 2-8 degrees Celsius, which corresponds to 36-46 degrees Fahrenheit. The standard recommendation is to use the solution within 28 days of reconstitution. After 28 days, potency may decline even under ideal storage conditions. Some sources suggest shorter windows for certain compounded formulations, so check with your pharmacy for their specific guidance.
Do not freeze reconstituted tirzepatide. Freezing can cause the peptide to denature, which means it unfolds from its active three-dimensional structure into a non-functional form. Once denatured, no amount of thawing or re-mixing will restore activity. A vial that has been accidentally frozen should be discarded. If you are curious about how temperature affects other peptide preparations, our guide on tirzepatide refrigeration timelines covers the specifics.
Unreconstituted lyophilized powder has different requirements. Store it frozen at minus 20 degrees Celsius (minus 4 degrees Fahrenheit) in a dry, dark location. Lyophilized peptides are significantly more stable than their reconstituted counterparts, which is why this form exists. If you receive a lyophilized vial and do not plan to use it immediately, leaving it in powder form until needed preserves potency far longer than reconstituting and refrigerating. Understanding post-reconstitution storage prevents wasteful degradation.
Practical storage tips
Store vials upright in the middle of your refrigerator, not in the door. Door shelves experience temperature fluctuations every time the refrigerator opens, while the middle shelves maintain the most consistent temperature. A small designated container or bag keeps your vials organized and prevents accidental contact with food items.
Keep vials away from light. Even refrigerator lights can contribute to degradation over time with repeated exposure. Wrapping vials in aluminum foil or storing them in an opaque container addresses this concern with minimal effort. These small precautions extend the usable life of your compound and maintain the potency throughout the vial lifespan.
If your vial must be out of refrigeration temporarily, such as during travel, keep it cool using an insulated bag with an ice pack. Tirzepatide can tolerate brief periods at room temperature, but extended exposure above 8 degrees Celsius accelerates degradation. Our detailed guide on peptide stability at room temperature outlines exactly how long various peptides remain viable outside refrigeration. For compounded tirzepatide specifically, aim to keep room temperature exposure under two hours when possible.
Managing side effects during dose escalation
Side effects are part of the tirzepatide experience for most users, particularly during dose escalation. Clinical trials reported nausea in 12-29% of participants, making it the single most common adverse event. The good news is that these effects are overwhelmingly transient, peaking during the first few weeks at each new dose level and subsiding as the body adapts. The approach to managing them can make the difference between tolerating the escalation process and abandoning it.
GI side effect management
Nausea, the hallmark tirzepatide side effect, stems from the peptide slowing gastric emptying. Food stays in the stomach longer, and the usual signals that prompt hunger and digestion are suppressed. This is actually part of the therapeutic mechanism, since delayed gastric emptying contributes to appetite suppression and reduced caloric intake. But when the effect is too pronounced, nausea follows.
Dietary modifications help significantly. Eat smaller, more frequent meals instead of two or three large ones. Reduce fatty, greasy, and heavily spiced foods, which are harder to digest when gastric motility is already slowed. Stay well hydrated, but avoid drinking large volumes during meals, as this compounds the fullness sensation. Some users find that sipping ginger tea or taking ginger supplements provides mild anti-nausea relief without pharmaceutical intervention.
Over-the-counter options include bismuth subsalicylate and dimenhydrinate. Prescription anti-nausea medications like ondansetron are available for more severe cases and can be discussed with a healthcare provider. These medications manage symptoms without interfering with tirzepatide mechanism of action.
The most important management strategy is patience. Studies show that GI side effects in tirzepatide trials were predominantly mild to moderate and concentrated during the dose escalation period. For most participants, symptoms resolved or became manageable within the first two to three weeks at each dose level. Knowing that nausea is temporary and expected helps frame the experience correctly. It is not a sign that something is wrong. It is a sign that the peptide is active and the body is adjusting.
Constipation affects some users and responds well to increased fiber intake, adequate hydration, and gentle movement like walking. Diarrhea, paradoxically, also occurs in a subset of users and typically resolves on its own. If GI symptoms are severe or persistent beyond three weeks at any given dose, consult your healthcare provider rather than pushing to the next escalation level. Your body is telling you something, and listening produces better long-term outcomes than ignoring it.
Microdosing as an alternative approach
The microdosing approach takes a different path to the same destination. Instead of jumping in 2.5mg increments, microdosing uses smaller steps, often 1mg or 1.25mg, over longer intervals. The logic is simple. Smaller dose increases mean smaller disruptions to the GI system, which translates to fewer and milder side effects.
A typical microdosing schedule might look like this. Start at 1.25mg for two weeks. Increase to 2.5mg for two weeks. Then move to 3.75mg. Then 5mg. Each step represents half the standard increment, given twice as frequently or at the same four-week interval. The total time to reach therapeutic doses is longer, but the journey is smoother.
This approach works particularly well for individuals who have experienced severe GI side effects on previous GLP-1 medications, those with pre-existing digestive conditions, or anyone who prioritizes tolerability over speed of escalation. The compounded tirzepatide format actually facilitates microdosing better than pre-filled pens, since you can draw precisely whatever volume corresponds to your micro-dose. A 10mg/mL concentration allows 1.25mg doses at just 12.5 units, easy to measure on a standard insulin syringe.
Whether you follow the standard escalation or a microdosing protocol, tracking your response at each level helps optimize your approach. SeekPeptides members gain access to protocol tracking tools and detailed escalation guides that account for individual variation, making it easier to find the dose that delivers results without unnecessary discomfort.
Common dosing mistakes and how to avoid them
Compounded tirzepatide introduces more variables than pre-filled pens, and each variable creates an opportunity for error. Knowing the most frequent mistakes before they happen is worth far more than correcting them afterward. Some of these errors waste product. Others waste weeks of progress. A few can create genuine safety concerns.
Mistake 1: using the wrong concentration chart
This is the number one error, and it bears repeating. A dosing chart designed for 10mg/mL concentration will overdose by 100% if applied to a 20mg/mL vial, or underdose by 50% if applied to a 5mg/mL vial. Always, always verify your vial concentration before consulting any chart. If you have bookmarked a chart from the internet, confirm that it matches your specific concentration. The charts in this guide cover all three common concentrations precisely because this mistake is so prevalent.
Mistake 2: confusing syringe types
Drawing tirzepatide with a tuberculin syringe while following instructions written for an insulin syringe, or vice versa, produces incorrect doses. The injection guide covers syringe selection in detail. The simple rule is that units go with insulin syringes and milliliters go with tuberculin syringes. If your instructions say "units," use insulin. If they say "mL," use tuberculin. Never mix these.
Mistake 3: skipping dose escalation steps
Impatience leads some users to jump from 2.5mg directly to 7.5mg or higher, reasoning that faster escalation means faster results. The clinical data tells a different story. Rapid escalation correlates with more severe GI side effects, higher discontinuation rates, and no improvement in long-term outcomes. Users who follow the gradual schedule are more likely to reach and maintain their target dose successfully. The appetite suppression timeline begins at the starting dose and builds progressively, so skipping steps does not meaningfully accelerate results.
Mistake 4: poor reconstitution technique
Shooting bacteriostatic water directly onto lyophilized powder, shaking the vial vigorously, or using too little water all compromise the final product. Damaged peptide means reduced potency means reduced results, and you will not know the damage has occurred until doses stop working as expected. Follow the reconstitution process described above precisely. Slow, gentle, and sterile. These three principles preserve the peptide and protect your investment. Detailed bacteriostatic water ratios and techniques apply across all reconstituted peptides.
Mistake 5: inconsistent injection timing
Tirzepatide is a once-weekly injection, and maintaining a consistent day and approximate time each week optimizes blood levels. Spacing doses unevenly, such as five days one week and nine days the next, creates peaks and valleys that increase side effects and reduce efficacy. Pick a day. Set a reminder. Stay consistent. This applies whether you are on compounded or brand tirzepatide, and it becomes particularly important during the dose escalation phase when your body is already adapting to increasing amounts. If your results have plateaued, inconsistent timing is one of the first variables to examine.
Mistake 6: not accounting for air bubbles
Small air bubbles in your syringe displace medication. If you draw 50 units but 5 units of that volume is actually air, you are only injecting 45 units of tirzepatide. Over time, these small shortfalls add up and may partially explain inconsistent results. After drawing your dose, hold the syringe needle-up and tap it gently to move bubbles to the top. Push the plunger slightly to expel the air, then verify your fill level. This basic technique is standard for any subcutaneous injection and takes only a few seconds.

Compounded vs brand name tirzepatide
Understanding the differences between compounded tirzepatide and FDA-approved brand products helps contextualize why dosing precision matters even more with compounded formulations.
Brand name tirzepatide, sold as Mounjaro for type 2 diabetes and Zepbound for weight management, comes in pre-filled pens with fixed doses. The manufacturing process is regulated by the FDA, each batch undergoes extensive quality testing, and the pens eliminate dosing calculations entirely. The clinical trial data, including the SURMOUNT series showing average weight loss of 15-21% depending on dose, comes from these FDA-approved formulations.
Compounded tirzepatide is produced by compounding pharmacies using the same active ingredient but without the same regulatory oversight. The FDA has noted that compounded formulations have not undergone equivalent safety and efficacy testing. Quality can vary between pharmacies, concentrations may differ from batch to batch, and the responsibility for accurate dosing shifts from the manufacturer to the user. This is precisely why a reliable dosage reference is essential for anyone using compounded formulations.
The regulatory landscape around compounded tirzepatide has evolved. Compounding was initially permitted under drug shortage provisions. As availability of brand products has changed, so have the rules governing compounded alternatives. Regardless of regulatory status, the pharmacological action of tirzepatide remains the same. It activates both GIP and GLP-1 receptors, producing the dual-agonist effects that differentiate it from semaglutide and other GLP-1-only medications.
For those using compounded tirzepatide, several precautions apply. Source from a licensed, reputable compounding pharmacy. Verify that the product matches the description, clear and colorless for liquid preparations. Check the concentration label matches what was prescribed. And follow the dosage charts in this guide, or use a verified dosage calculator, for every single dose. The extra diligence required with compounded formulations is the trade-off for accessibility and cost considerations that lead many to this route. For a comprehensive look at sourcing, our guide to compounded tirzepatide options covers what to look for and what to avoid.
Tracking your progress on compounded tirzepatide
Dosing charts tell you what to inject. Progress tracking tells you whether it is working. Without systematic monitoring, you are guessing about whether your current dose is appropriate, whether an escalation is warranted, and whether your protocol is producing the outcomes you set out to achieve.
Weekly weigh-ins provide the most obvious metric, but they are not the only one that matters. Body weight fluctuates daily based on hydration, food volume, exercise, and hormonal cycles. A single weekly measurement captures one point in that fluctuation. More useful is a rolling average, weighing daily at the same time and calculating a weekly average to smooth out the noise. The trend line of weekly averages reveals whether your dose is producing consistent progress.
Body measurements, specifically waist circumference, add information that scale weight misses. Users on tirzepatide sometimes maintain or even gain weight while losing inches, particularly if they are simultaneously building muscle through resistance training. A tape measure around the narrowest part of the waist, taken at the same time and tension each week, catches these changes. The before and after documentation from other users illustrates how diverse progress patterns can be.
Appetite tracking matters too. One of the primary ways tirzepatide produces weight loss is through appetite reduction. Logging how hungry you feel throughout the day, how much food satisfies you, and whether cravings have changed provides qualitative data about whether the peptide is working at a mechanistic level. If appetite suppression fades significantly between doses or fails to appear at a given dose level, that information helps guide escalation decisions. Our guide on appetite suppression timelines provides a framework for what to expect at each dose.
Side effect logging rounds out the picture. Note the day and severity of any GI symptoms, fatigue, injection site reactions, or other effects. Over time, patterns emerge. Maybe nausea always hits on day two and resolves by day four. Maybe constipation appears only at higher doses. This data helps your healthcare provider make informed adjustments to your protocol, whether that means slowing escalation, adding supportive interventions, or confirming that your current dose is well-tolerated.
Energy levels, sleep quality, and mood deserve tracking as well. Fatigue on tirzepatide is reported by some users, particularly during dose escalation. Others report improved energy as weight decreases and metabolic function improves. Your individual pattern informs protocol decisions that a generic chart cannot capture.
Combining tirzepatide with other compounds
Some users explore combining compounded tirzepatide with other peptides or supplements to enhance results. While this guide focuses specifically on tirzepatide dosing, understanding common combinations provides context for how the dosage chart fits within a broader protocol.
Tirzepatide with B12 is one of the most common additions. Vitamin B12 supplementation supports energy levels that can dip during caloric restriction, and some compounding pharmacies offer tirzepatide pre-mixed with B12. If your compound includes B12, the dosing charts in this guide still apply for the tirzepatide component, as the B12 addition does not change the tirzepatide concentration calculations.
Niacinamide combinations represent another popular pairing. Niacinamide, a form of vitamin B3, supports metabolic health and skin appearance during weight loss. Again, the addition of niacinamide to a tirzepatide compound does not alter the tirzepatide dosing calculations, but always confirm the tirzepatide concentration from your pharmacy to ensure your charts remain accurate.
For those interested in combining tirzepatide with other GLP-1 agents or weight loss peptides, the cagrilintide and tirzepatide combination guide covers one of the more researched pairings. Other peptide stacking approaches can be explored through the peptide stack calculator and the comprehensive stacking guide. When combining multiple compounds, accurate dosing of each individual component becomes even more critical, and the charts in this guide serve as the foundation for the tirzepatide portion of any combination protocol.
It is worth noting that combining tirzepatide with other appetite-suppressing or GI-active compounds may intensify side effects. Start each new compound individually before combining, so you can identify which substance causes which effects. The guidance on simultaneous peptide use outlines the principles for safe combination protocols.
Understanding the math behind the charts
Some users prefer to understand the underlying calculation rather than relying solely on reference charts. If you can do the math yourself, you can verify any chart, handle non-standard concentrations, and troubleshoot discrepancies between different sources. The formula is not complicated.
Three numbers define every dose. The prescribed dose in milligrams, the vial concentration in mg/mL, and the syringe scale factor. For U-100 insulin syringes, the scale factor is 100 (because 100 units equals 1mL). The formula is:
Units to draw = (prescribed dose in mg / concentration in mg/mL) x 100
For a 7.5mg dose from a 10mg/mL vial: 7.5 / 10 = 0.75mL x 100 = 75 units.
For a 7.5mg dose from a 20mg/mL vial: 7.5 / 20 = 0.375mL x 100 = 37.5 units.
For a 7.5mg dose from a 5mg/mL vial: 7.5 / 5 = 1.5mL x 100 = 150 units.
Same dose. Three different unit counts. The concentration is the variable that changes everything. Once you internalize this formula, you can calculate the correct dose for any concentration, including the non-standard ones that occasionally appear from certain pharmacies. The peptide dosage calculation guide covers this math in even greater detail, along with calculations for body-weight-based dosing and multi-peptide protocols.
If math is not your strength, that is perfectly fine. That is why the charts exist. But checking your chart against the formula at least once, with your specific vial, builds confidence that your doses are correct. It takes thirty seconds. It is worth the effort.
What to do if you miss a dose
Life happens. Missed doses on tirzepatide are not ideal, but they are not catastrophic either. The appropriate response depends on how much time has passed since your scheduled injection.
If fewer than four days have passed since your missed dose, take it as soon as you remember and resume your regular schedule the following week. If more than four days have passed, skip the missed dose entirely and take your next scheduled dose on time. Do not double up. Taking two doses close together concentrates the GI-slowing effects and dramatically increases the risk of nausea, vomiting, and other side effects.
Frequent missed doses disrupt the steady-state blood levels that produce consistent appetite suppression and metabolic effects. If you are regularly missing doses, consider adjusting your injection day to one that better fits your routine, or set multiple reminders. Consistency, more than any other single factor, determines whether a tirzepatide protocol delivers the results shown in clinical trials.
After a prolonged break from tirzepatide, say two or more weeks, do not resume at the dose you left off at. Drop back by at least one escalation step and work up again. Your body loses its adaptation during the break, and jumping back to a higher dose reintroduces the side effects you spent weeks building tolerance to. This restart approach is standard for all peptide dosing protocols and protects against unnecessary adverse effects.
When compounded tirzepatide is not working
Sometimes the scale does not move despite following the dosage chart correctly. Before assuming the compound is ineffective, systematically eliminate the most common culprits.
Verify your concentration and dosing math first. This sounds basic, but it catches a surprising number of stalled protocols. Recalculate your dose using the formula above and cross-reference it against the charts in this guide. One person recounting their experience discovered they had been drawing from a 20mg/mL vial using a 10mg/mL chart, effectively halving their dose for weeks. The fix was immediate.
Check your storage conditions. If your reconstituted vial has been stored improperly, the peptide may have degraded. Signs of degradation include cloudiness, particles, color change, or unusual odor. Even without visible signs, peptides stored above recommended temperatures or for longer than 28 days post-reconstitution may have lost significant potency. A fresh vial, properly stored, often resolves unexplained plateaus. The refrigeration requirements are non-negotiable for maintaining activity.
Evaluate whether your dose is actually therapeutic for your body weight and composition. The starting dose of 2.5mg is explicitly a tolerance-building dose, not a therapeutic one. Meaningful weight loss typically begins at 5mg and becomes more pronounced at higher doses. If you are still at 2.5mg or 5mg, escalation according to the schedule may be the answer. If you have already escalated and results have stalled, the comprehensive troubleshooting guide walks through dietary, activity, and protocol factors that influence outcomes.
Consider the source of your compounded tirzepatide. Not all pharmacies maintain the same quality standards, and purity varies. If you suspect a quality issue, discussing alternative sources with your provider is warranted. Our overview of tirzepatide sourcing options and the broader research vs pharmaceutical peptide comparison can inform that conversation.
Transitioning between tirzepatide and other GLP-1 medications
Users sometimes switch between tirzepatide and semaglutide or other GLP-1 receptor agonists. These transitions require careful dose management because the medications, while related, have different potency profiles and receptor activity patterns.
The tirzepatide to semaglutide transition guide covers the specific protocols for moving between these two medications. The general principle is that direct dose equivalence does not exist. Tirzepatide at 5mg is not equivalent to semaglutide at 0.5mg, even though both are starting-range doses. The dual GIP/GLP-1 mechanism of tirzepatide produces different receptor activation patterns compared to semaglutide, which is GLP-1-only.
When transitioning from semaglutide to compounded tirzepatide, most protocols suggest starting tirzepatide at 2.5mg regardless of the previous semaglutide dose. This allows assessment of how the individual responds to the dual-agonist mechanism before escalating. The detailed comparison between these two medications helps contextualize why a fresh escalation is safer than attempting dose equivalence. Semaglutide users can review the side-by-side dosage chart for reference.
For those coming from compounded semaglutide, the semaglutide dosage calculator helps verify your current semaglutide dose before planning the transition. Understanding where you stand on one medication informs the approach to starting another, and having both semaglutide dosage charts and tirzepatide dosage charts accessible makes the planning process clearer.
Frequently asked questions
How many units is 2.5mg of compounded tirzepatide?
It depends entirely on your vial concentration. At 10mg/mL, 2.5mg equals 25 units. At 5mg/mL, it equals 50 units. At 20mg/mL, it equals 12.5 units. Always check your concentration label before drawing any dose. Our detailed breakdown of 2.5mg unit conversions covers every common concentration.
Can I use the same dosing chart for all compounded tirzepatide vials?
No. Dosing charts are concentration-specific. A chart for 10mg/mL produces dangerously incorrect doses if applied to a 20mg/mL or 5mg/mL vial. This guide provides separate charts for each common concentration. Always match your chart to your specific vial concentration.
How long does compounded tirzepatide last after reconstitution?
Reconstituted tirzepatide remains stable for up to 28 days when properly refrigerated at 2-8 degrees Celsius (36-46 degrees Fahrenheit). Do not freeze reconstituted solution. For detailed storage timeline information, including what affects shelf life, see our dedicated guide.
What syringe should I use for compounded tirzepatide?
Use a U-100 insulin syringe if your dosing instructions are in units, which is the standard. Use a tuberculin syringe only if your instructions are in milliliters. Never mix unit-based instructions with a milliliter syringe or vice versa. A 1mL insulin syringe handles all doses through 10mg at 10mg/mL concentration. For complete injection guidance, see our injection technique guide.
Is compounded tirzepatide as effective as Mounjaro or Zepbound?
The active ingredient is the same tirzepatide molecule. However, compounded formulations have not undergone the same clinical trials as FDA-approved products. Efficacy may vary based on compounding quality, concentration accuracy, and proper handling practices. Accurate dosing from a reputable pharmacy maximizes the likelihood of results comparable to brand products.
Can I switch from 10mg/mL to 20mg/mL mid-protocol?
Yes, but you must recalculate your dose in units. If you were taking 50 units at 10mg/mL (5mg dose), you would draw only 25 units from a 20mg/mL vial for the same 5mg dose. Use the conversion table in this guide or the peptide calculator to ensure accuracy when switching concentrations.
What happens if I draw too many units accidentally?
If you notice the error before injecting, simply push the excess back into the vial and re-draw the correct amount. If you have already injected an excessive dose, monitor for increased GI side effects including nausea, vomiting, and diarrhea. Contact your healthcare provider for guidance, especially if the overage was significant. Tirzepatide has a long half-life, so effects from a single overdose may persist for several days. Document the error and use it as motivation to double-check every future draw.
Do I need to factor in the dead space of the syringe?
Standard insulin syringes account for dead space in their calibration, so the marked units reflect the actual amount delivered. However, some needle-syringe combinations have additional dead space, meaning a small amount of medication remains in the hub after injection. For most users, this loss is negligible. If precision is critical, such as with high-concentration formulations where small volume differences mean larger dose differences, low dead space syringes are available and eliminate this variable.
External resources
SURMOUNT-1 clinical trial results (New England Journal of Medicine)
Mayo Clinic tirzepatide information
For researchers committed to getting the most from their compounded tirzepatide protocol, SeekPeptides provides the most comprehensive resource available. Members access personalized dosing calculators, protocol tracking tools, detailed escalation guides tailored to individual goals, and a community of thousands navigating the same journey. Accurate dosing is where results begin, and SeekPeptides makes sure every number, every unit, every calculation is right.
In case I do not see you, good afternoon, good evening, and good night. May your concentrations stay accurate, your doses stay consistent, and your results stay on track.