Feb 20, 2026

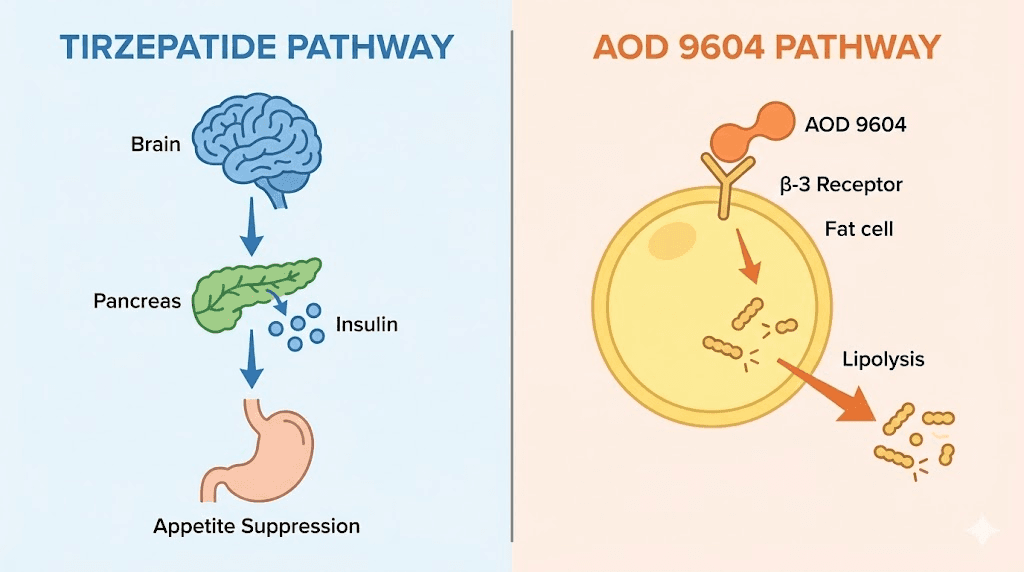
At the cellular level, AOD 9604 and tirzepatide trigger two completely separate cascades. One activates beta-3 adrenergic receptors in fat tissue. The other mimics gut hormones that control hunger and insulin. They never compete. They never interfere. And when researchers combine them, the result is a metabolic environment where fat breaks down faster while appetite drops simultaneously.
That dual action is what makes this combination so interesting.
Most fat loss protocols rely on a single mechanism. Eat less. Burn more. Suppress appetite. Block absorption. But the AOD 9604 and tirzepatide stack attacks the problem from two entirely different directions, creating what some clinicians describe as a synergistic metabolic advantage that neither compound achieves alone. The science behind this pairing is not complicated once you understand how each compound works independently, and this guide breaks down everything you need to know about combining them safely, timing your doses correctly, and understanding what realistic results actually look like.
SeekPeptides has tracked the growing interest in this combination as more researchers explore multi-pathway approaches to fat loss protocols. Whether you are already using tirzepatide and considering adding AOD 9604, or you are researching this stack from scratch, this guide covers the mechanisms, the protocols, the safety data, and the practical details that most resources skip entirely.
What AOD 9604 actually does inside fat cells
AOD 9604 is not a random peptide with vague fat loss claims. It is a specific 16-amino acid fragment of human growth hormone, corresponding to the region between amino acids 176 and 191. This fragment was isolated precisely because it contains the lipolytic (fat-breaking) properties of HGH without the growth-promoting effects that cause problems like insulin resistance, joint swelling, and organ enlargement.
The distinction matters enormously.
Full-length growth hormone stimulates fat breakdown, yes. But it also raises IGF-1, disrupts glucose metabolism, and can accelerate unwanted tissue growth. AOD 9604 strips away all of those side effects while preserving the fat-specific activity. Researchers confirmed this through knockout studies in mice, where animals lacking beta-3 adrenergic receptors showed zero response to AOD 9604, proving the peptide works specifically through this receptor pathway in adipose tissue.
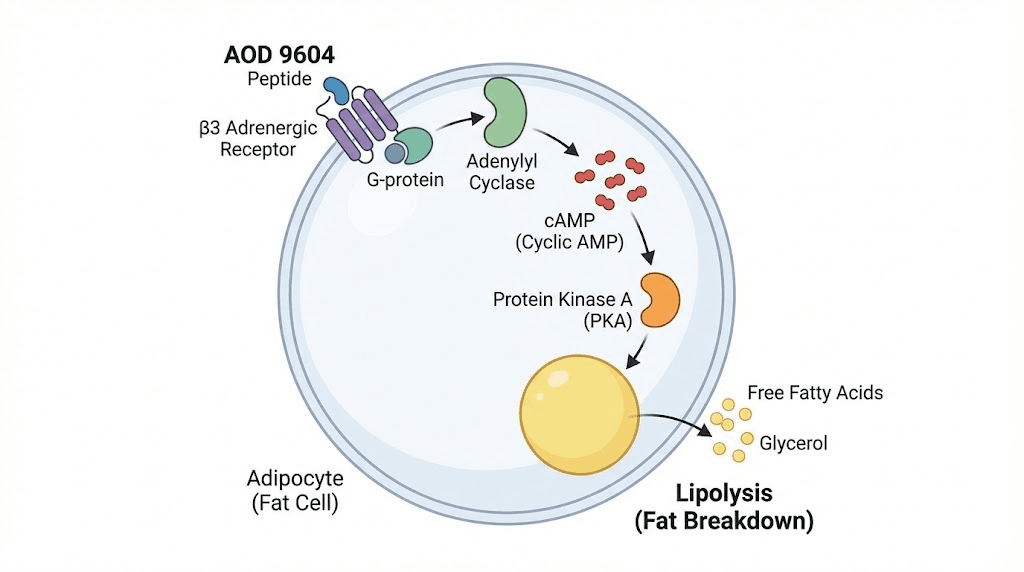
Here is the mechanism in practical terms. When AOD 9604 binds to beta-3 adrenergic receptors on white fat cells, it triggers a cascade that starts with adenylyl cyclase activation. That enzyme increases intracellular cyclic AMP levels. Higher cAMP activates protein kinase A. And protein kinase A phosphorylates hormone-sensitive lipase, the enzyme that physically breaks triglycerides apart into free fatty acids and glycerol. Those free fatty acids then enter the bloodstream and become available for oxidation, which means your body burns them for energy.
But AOD 9604 does something else that most guides ignore. It simultaneously inhibits lipogenesis, the process of converting excess calories into new fat deposits. This dual action, increasing fat breakdown while blocking fat formation, creates a metabolic environment that favors net fat loss even when caloric intake is not dramatically reduced. Understanding this complete AOD 9604 mechanism is essential before combining it with anything.
Clinical trials involving over 900 human subjects across six randomized, double-blind, placebo-controlled studies confirmed the safety profile. No serious adverse events. No changes in IGF-1 levels. No negative effects on carbohydrate metabolism. The most commonly reported side effect was mild headache, occurring at rates comparable to placebo. The full AOD 9604 side effects profile shows a peptide that is remarkably well-tolerated in human subjects.
One important limitation: the clinical weight loss results were modest. In a 23-week trial, subjects taking 1mg daily lost an average of 2.8kg compared to 0.8kg for placebo. That 2kg difference is real but not dramatic. Development actually halted in 2007 because results across 536 subjects over 24 weeks did not reach statistical significance for the primary endpoint. This is crucial context because it explains why AOD 9604 works better as part of a combination strategy rather than as a standalone fat loss solution.
How tirzepatide works through completely different pathways
Tirzepatide operates in a different universe from AOD 9604. Where AOD 9604 directly attacks fat cells through adrenergic receptors, tirzepatide works upstream by modifying appetite signaling, insulin response, and gastric emptying. It is a dual GLP-1 and GIP receptor agonist, meaning it activates two incretin hormone pathways simultaneously.
GLP-1 receptor activation does several things. It slows gastric emptying so food stays in the stomach longer. It enhances insulin secretion in response to meals. It suppresses glucagon release. And critically, it acts on the hypothalamus to reduce appetite, creating powerful satiety signals that make people eat significantly less without feeling deprived. Understanding how quickly tirzepatide works helps set realistic expectations.
GIP receptor activation adds something unique. GIP (glucose-dependent insulinotropic polypeptide) further enhances insulin sensitivity and may directly influence fat metabolism in adipose tissue. The combination of GLP-1 and GIP activation is what separates tirzepatide from pure GLP-1 agonists like semaglutide, and clinical data suggests the dual mechanism produces superior weight loss outcomes.
The numbers are staggering.
In the SURMOUNT-1 trial, tirzepatide produced average weight loss of 15-22.5% of body weight over 72 weeks depending on dose. At the 15mg dose, participants lost an average of 52 pounds. Those results dwarf what any single peptide, supplement, or lifestyle modification achieves in isolation. The tirzepatide before and after results demonstrate why this compound has generated so much attention.
Body composition data from the SURMOUNT-1 DXA substudy showed approximately 75% of weight lost came from fat mass and 25% from lean mass. While losing some lean mass during aggressive weight loss is expected, this ratio is favorable compared to diet-only approaches where lean mass losses can reach 30-40%. The SURPASS-3 MRI substudy added another layer, showing tirzepatide actually reduced muscle fat infiltration across all doses, indicating improved muscle quality even as total muscle volume decreased.
But here is the limitation that makes the AOD 9604 combination appealing. Tirzepatide does not directly stimulate fat cell breakdown. It works primarily through appetite suppression and metabolic improvements. You eat less, your insulin works better, and your body gradually draws on fat stores. There is no direct, targeted mechanism hitting the fat cells themselves the way AOD 9604 dosing protocols achieve through beta-3 receptor activation.
Why combining them creates a synergistic advantage
The case for combining AOD 9604 with tirzepatide comes down to one principle: they target completely non-overlapping biological pathways. There is no redundancy. No competition. No interference. Each compound does something the other cannot.
Tirzepatide handles the demand side. It reduces how much energy comes in by suppressing appetite, slowing digestion, and improving how the body handles the food that does get consumed. Think of it as turning down the flow of incoming calories.
AOD 9604 handles the supply side. It accelerates the breakdown of existing fat stores and prevents new fat from forming, regardless of caloric intake. Think of it as opening the drainage valves on stored body fat.
When both mechanisms operate simultaneously, you create a metabolic state where the body is consuming fewer calories while simultaneously breaking down fat reserves faster. The theoretical advantage is significant. Tirzepatide creates the caloric deficit needed for weight loss, and AOD 9604 ensures that the stored fat cells are actively being mobilized for energy rather than stubbornly holding onto their contents.
Researchers at wellness clinics who administer this combination report that patients experience enhanced results compared to tirzepatide alone, particularly in stubborn fat deposits that resist GLP-1 therapy. Abdominal visceral fat, which responds well to beta-3 adrenergic stimulation, appears particularly susceptible to the combination approach. For those targeting specific problem areas, understanding peptides for visceral fat loss provides additional context.
There is an additional benefit worth noting. One of the concerns with GLP-1 therapy is muscle loss. While tirzepatide preserves lean mass better than diet alone, some researchers look for ways to further protect muscle during aggressive weight loss. AOD 9604 does not stimulate muscle growth, but it does not promote muscle breakdown either. By selectively targeting fat cells without affecting muscle tissue, it shifts the weight loss equation toward preferential fat loss. Researchers exploring peptides for weight loss and muscle gain often look at this balance carefully.
The safety argument is equally compelling. AOD 9604 and tirzepatide have no known pharmacological interactions. They bind to different receptors. They activate different second messenger systems. They metabolize through different pathways. Clinicians administering the combination have not reported any adverse interaction effects beyond the individual side effects of each compound. This clean safety profile is what distinguishes this stack from other combination approaches that carry interaction risks.
AOD 9604 with tirzepatide dosing protocols
Dosing this combination correctly requires understanding each compound independently before combining them. Mistakes in either direction, too much AOD 9604 or too rapid tirzepatide escalation, create unnecessary side effects without additional benefit.
AOD 9604 dosing fundamentals
Standard AOD 9604 protocols use 250-500mcg daily via subcutaneous injection. Clinical studies tested doses ranging from 250mcg to 1mg daily, with 300mcg showing effective fat reduction at the lowest side effect profile. Most experienced researchers settle on 300-500mcg as the optimal daily dose.
The complete AOD 9604 dosing guide covers the specifics, but for combination purposes, here is what matters. Start at the lower end. Begin with 250mcg daily for the first week to assess tolerance. If no adverse effects occur, increase to 300-500mcg for the remainder of the cycle. There is no evidence that doses above 500mcg produce proportionally better results, and clinical trials using 1mg daily did not dramatically outperform lower doses.
Cycle length runs 12-16 weeks. After a full cycle, take a 4-week break before resuming. This cycling approach prevents receptor desensitization and maintains effectiveness across multiple cycles.
Tirzepatide dosing fundamentals
Tirzepatide follows a structured escalation protocol regardless of whether you are combining it with other compounds. The standard approach starts at 2.5mg weekly for four weeks, then escalates in 2.5mg increments every four weeks based on tolerance and response.
For those new to tirzepatide dosing in units, the escalation typically looks like this: 2.5mg for weeks 1-4, then 5mg for weeks 5-8, then 7.5mg for weeks 9-12, with optional increases to 10mg and 15mg for those who tolerate lower doses well but need additional appetite suppression. Not everyone needs to reach the highest doses. Many researchers find their optimal response at 5-10mg weekly.
Understanding the tirzepatide dose chart is essential for proper escalation. The compounded tirzepatide dosage calculator can help determine exact unit measurements based on your specific vial concentration.
Combining the two: the practical protocol
Here is where the combination protocol differs from running either compound alone.
Phase 1: Establish tirzepatide baseline (weeks 1-4)
Start tirzepatide alone at 2.5mg weekly. Do not introduce AOD 9604 yet. This phase lets you assess tirzepatide tolerance, identify any GI side effects, and establish a baseline response. Common early side effects include constipation, nausea, and reduced appetite. These typically resolve within 2-3 weeks.
Phase 2: Introduce AOD 9604 (weeks 5-8)
Once tirzepatide is tolerated at the initial dose, add AOD 9604 at 250mcg daily. Continue tirzepatide at 5mg weekly (first escalation). This staggered introduction allows you to identify which compound causes any new side effects rather than guessing.
Phase 3: Optimize both (weeks 9-16)
Increase AOD 9604 to 300-500mcg daily based on tolerance. Continue escalating tirzepatide as needed per standard protocols. This is the phase where the synergistic effects become most apparent. Fat loss typically accelerates, body composition shifts become visible, and stubborn areas begin responding.
Phase 4: AOD 9604 cycle break (week 17+)
After 12-16 weeks of AOD 9604, discontinue it for a 4-week break while continuing tirzepatide. This prevents beta-3 receptor desensitization while maintaining the appetite suppression and metabolic benefits of tirzepatide. Resume AOD 9604 after the break if desired. Understanding peptide cycle planning helps you structure these phases correctly.
The timing question: when to inject each compound
Timing matters more for AOD 9604 than for tirzepatide, and getting this wrong can significantly reduce AOD 9604 effectiveness while having almost no impact on tirzepatide response.
AOD 9604 timing
AOD 9604 must be administered in a fasted state. Period. No exceptions. The reason is physiological, not arbitrary. When you eat, insulin levels rise. Insulin activates fat storage pathways that directly oppose AOD 9604 lipolytic signaling. Injecting AOD 9604 after eating is like pressing the gas and brake simultaneously. You waste the peptide.
The optimal protocol is to inject first thing in the morning, after an overnight fast of at least 8 hours. Then wait 30-60 minutes before consuming any food or caloric beverages. Water, black coffee, and plain tea are fine during this window. Many researchers combine this fasting window with light morning cardio, as the elevated free fatty acid levels from AOD 9604 provide readily available fuel for aerobic exercise.
For those following intermittent fasting protocols, AOD 9604 integrates seamlessly. Inject during the fasting window, not during the eating window.
Tirzepatide timing
Tirzepatide is far more forgiving. It is injected once weekly, and the timing relative to meals does not significantly impact its effectiveness. Most researchers pick one day per week and inject at the same time for consistency. Some prefer morning injections, others evening. The half-life of tirzepatide is approximately 5 days, so the effects are essentially continuous throughout the week regardless of when you inject. Knowing how long tirzepatide takes to work helps manage expectations during the initial weeks.
Common injection sites include the abdomen, thigh, and upper arm. The tirzepatide stomach injection guide covers proper technique. Rotate sites to prevent lipohypertrophy (lumps at injection sites).
Can you inject both on the same day?
Yes. Since AOD 9604 is daily and tirzepatide is weekly, one day per week they will overlap. This is not a problem. Inject them at different sites (for example, AOD 9604 in the abdomen and tirzepatide in the thigh) and at different times if you prefer. Some researchers inject AOD 9604 in the morning fasted and tirzepatide in the evening. Others inject both in the morning at different sites. Neither approach shows a meaningful difference in outcomes. The peptide injection guide covers general best practices for subcutaneous administration.
Optimizing the daily AOD 9604 schedule
For maximum fat oxidation, some advanced protocols split the daily AOD 9604 dose into two injections: 150-250mcg in the morning fasted and 150-250mcg before bed (at least 3 hours after the last meal). This approach maintains more consistent beta-3 receptor stimulation throughout the 24-hour period. However, the convenience of a single morning dose means most researchers prefer once-daily administration, and the clinical data supporting split dosing specifically for AOD 9604 is limited.
Body composition: what actually changes and when
Understanding the timeline of body composition changes helps set realistic expectations and prevents premature protocol abandonment. The two compounds produce results on different timescales, and the combined effect unfolds in stages.
Weeks 1-4: tirzepatide only phase
During the tirzepatide-only phase, the primary change is appetite reduction. Weight loss begins but may be modest, averaging 2-4 pounds depending on starting weight and dose. Some of this initial loss is water weight from reduced glycogen stores. Body composition changes are not yet visible. Patience matters here.
Weeks 5-8: combination introduction
When AOD 9604 enters the picture alongside the first tirzepatide dose escalation, fat loss typically accelerates noticeably. The combination of reduced caloric intake (from tirzepatide appetite suppression) and enhanced fat breakdown (from AOD 9604 lipolysis) creates a steeper downward trajectory. Researchers often report that this is when they notice the first visible changes in problem areas, particularly abdominal fat. Those tracking tirzepatide before and after progress often see the most dramatic shifts during this phase.
Weeks 9-16: optimization phase
With both compounds at optimal doses, the body composition changes become significant. Fat loss continues at an accelerated rate. Lean mass preservation remains relatively good because AOD 9604 selectively targets adipose tissue while tirzepatide maintains favorable lean-to-fat loss ratios. The SURMOUNT-1 data showed 75% fat loss and 25% lean mass loss with tirzepatide alone. Adding AOD 9604 targeted fat breakdown may shift this ratio even further toward preferential fat loss, though controlled studies of the combination are limited.
This phase is also where visceral fat reduction becomes measurable. Deep abdominal fat, which responds well to beta-3 adrenergic stimulation, appears particularly susceptible during combination therapy. Researchers using DEXA scans or waist circumference measurements often see disproportionate reductions in trunk fat compared to peripheral fat during this phase.
Protecting muscle during aggressive fat loss
Muscle preservation during any weight loss protocol requires deliberate effort. Neither AOD 9604 nor tirzepatide is a muscle-building compound, and relying solely on pharmaceuticals to protect lean mass is a mistake. The following strategies are essential during combination therapy.
Resistance training at least three times per week is non-negotiable. Research consistently shows that resistance exercise is the single most effective intervention for preserving lean mass during caloric restriction. You do not need elaborate programs. Compound movements (squats, deadlifts, presses, rows) at moderate intensity preserve muscle effectively. For those looking at peptides that support muscle growth, additional compounds may be worth researching alongside this fat loss stack.
Protein intake should reach 1.0-1.2 grams per pound of body weight daily. This is challenging when tirzepatide suppresses appetite, so dense protein sources become important. Whey protein shakes, Greek yogurt, chicken breast, and fish deliver high protein per calorie. The tirzepatide food guide covers optimal dietary strategies. Some researchers also find protein shakes formulated for GLP-1 users particularly helpful for maintaining adequate intake.
Sleep quality affects body composition outcomes significantly. Growth hormone release occurs primarily during deep sleep, and HGH is the body primary muscle-preserving hormone during caloric deficit. Aim for 7-9 hours of quality sleep. Tirzepatide does not typically disrupt sleep, though some researchers report tirzepatide-related sleep changes that may need management.
Safety profile of the combination
Safety is the most critical consideration with any combination protocol, and the AOD 9604 plus tirzepatide stack has a favorable profile compared to other multi-compound approaches.
AOD 9604 safety data
Six randomized, double-blind, placebo-controlled trials enrolling 925 adult subjects established AOD 9604 safety. Key findings include no serious adverse events related to AOD 9604. No withdrawals due to adverse effects. No changes in serum IGF-1 levels. No negative effects on carbohydrate metabolism. No anti-AOD9604 antibodies detected. The safety profile was statistically indistinguishable from placebo across all trials. This is remarkable for any pharmaceutical compound and reflects AOD 9604 highly targeted mechanism that affects fat cells without interfering with other physiological systems.
Understanding the general peptide safety landscape helps put these results in context. AOD 9604 stands out as one of the best-tolerated peptides in clinical research.
Tirzepatide safety data
Tirzepatide safety data comes from the SURPASS and SURMOUNT trial programs involving thousands of subjects. The most common adverse events are gastrointestinal: nausea (occurring in 12-33% of subjects depending on dose), diarrhea, constipation, and decreased appetite. These effects are typically mild to moderate, dose-dependent, and tend to diminish with continued use and proper dose escalation.
More serious but rare adverse events include pancreatitis, gallbladder events, and hypoglycemia (primarily in patients using insulin simultaneously). The tirzepatide vs semaglutide side effects comparison shows tirzepatide generally has a comparable safety profile to other GLP-1 class medications.
Combination safety considerations
The combination of AOD 9604 and tirzepatide has not been studied in formal clinical trials. This is important to acknowledge. However, several factors support its safety profile. The compounds bind to entirely different receptors (beta-3 adrenergic vs GLP-1/GIP). They activate different intracellular signaling cascades (cAMP/PKA pathway in fat cells vs incretin signaling in gut and brain). They metabolize through different hepatic pathways. And they have no known pharmacological interactions.
Clinical practitioners who administer this combination report side effect profiles consistent with each compound used individually. The most commonly reported adverse effects are the GI symptoms associated with tirzepatide (nausea, reduced appetite) plus the mild headache occasionally seen with AOD 9604. No synergistic adverse effects have been reported in clinical practice.
That said, responsible use requires medical supervision. Do not combine these compounds without baseline bloodwork and regular monitoring. At minimum, check metabolic panels, liver function, lipid profiles, and fasting glucose before starting and every 8-12 weeks during combination therapy. The common peptide mistakes guide covers other errors to avoid.
Common side effects and how to manage them
Knowing what to expect and how to handle side effects prevents unnecessary protocol abandonment. Most side effects in this combination come from tirzepatide, not AOD 9604.
Nausea from tirzepatide
Nausea is the most common complaint, affecting up to one-third of tirzepatide users at higher doses. It typically peaks during dose escalation and resolves within 1-2 weeks at each new dose level. Management strategies include eating smaller, more frequent meals. Avoiding high-fat and greasy foods during the first week at each new dose. Staying hydrated. Taking ginger supplements or drinking ginger tea. If nausea persists beyond two weeks, your dose may be too high. Drop back to the previous dose and attempt escalation again after an additional 2-4 weeks.
Understanding which foods to avoid on tirzepatide significantly reduces GI discomfort. A well-planned tirzepatide diet plan makes the transition much smoother.
Constipation
Tirzepatide slows gastric motility, which frequently causes constipation. This affects approximately 6-11% of users across clinical trials. Management approaches include increasing fiber intake gradually (too much fiber at once worsens bloating), drinking at least 64 ounces of water daily, taking magnesium citrate (400-600mg at bedtime), and increasing physical activity. The complete tirzepatide constipation treatment guide covers additional strategies including osmotic laxatives for persistent cases.
Headache from AOD 9604
Mild to moderate headache is the most commonly reported AOD 9604 side effect, occurring in roughly 70% of subjects in clinical trials at rates comparable to placebo. These headaches are typically transient, lasting a few hours, and respond to standard over-the-counter pain relief. Ensuring adequate hydration before and after AOD 9604 injection reduces headache frequency in most cases.
Fatigue and energy changes
Some researchers report transient fatigue when first combining the two compounds. This makes sense physiologically. Reduced caloric intake from tirzepatide plus enhanced fat mobilization from AOD 9604 creates a significant metabolic shift that the body needs time to adapt to. Fatigue typically resolves within 1-2 weeks as the body becomes more efficient at utilizing free fatty acids for energy. The GLP-1 fatigue guide covers this phenomenon in detail.
Interestingly, many researchers report increased energy levels after the initial adaptation period. Once the body adapts to running on mobilized fat stores rather than relying solely on dietary glucose, energy levels often stabilize at or above baseline. Some users specifically report that tirzepatide provides an energy boost after the initial adjustment phase.
Injection site reactions
Minor redness, swelling, or itching at injection sites occurs occasionally with both compounds. Rotating injection sites prevents this from becoming problematic. Use at least 3-4 different sites and do not inject in the same spot more than once per week. The GLP-1 injection site guide covers optimal locations and rotation strategies.
Hair thinning
Some GLP-1 users report hair thinning during aggressive weight loss phases. This is not specific to tirzepatide or AOD 9604 but rather a consequence of rapid weight loss itself, known as telogen effluvium. It typically reverses once weight stabilizes. Adequate protein intake, biotin supplementation, and avoiding excessively rapid weight loss (more than 2-3 pounds per week) reduce the risk. The GLP-1 hair loss guide provides comprehensive management strategies.
What the clinical research actually shows
Separating marketing claims from actual research data is essential for anyone considering this combination. Here is what the evidence says, clearly and without exaggeration.
AOD 9604 clinical evidence
The most rigorous AOD 9604 data comes from the Metabolic Pharmaceuticals clinical program conducted between 2001 and 2007. Six trials enrolled 925 subjects testing intravenous and oral formulations at various doses. The results showed statistically significant but clinically modest weight loss (2kg average advantage over placebo at the 1mg oral dose over 23 weeks). Development was discontinued because the Phase IIb trial of 536 subjects over 24 weeks failed to meet its primary efficacy endpoint.
This does not mean AOD 9604 does not work. It means that as a standalone oral therapy at the tested doses, it did not produce the magnitude of weight loss that pharmaceutical companies require for commercial viability. Subcutaneous administration (which bypasses first-pass metabolism and delivers more peptide to target tissues) was not extensively tested in human trials, and this route is what most current research protocols use.
Animal data is more robust. Chronic treatment with AOD 9604 in obese mice produced significant weight reduction and changes in fat pad weight. The beta-3 adrenergic receptor knockout study definitively proved the mechanism. And safety data across all human trials was excellent. These factors support continued research interest despite the disappointing oral efficacy results.
Tirzepatide clinical evidence
Tirzepatide evidence is vastly more extensive and impressive. The SURPASS program (for type 2 diabetes) and SURMOUNT program (for obesity) together enrolled over 20,000 participants across multiple Phase III trials. Weight loss results were consistently superior to placebo and to comparator medications including semaglutide.
Key findings from SURMOUNT-1 include a mean weight loss of 15.0% at the 5mg dose, 19.5% at the 10mg dose, and 20.9% at the 15mg dose over 72 weeks. In comparison, placebo produced only 3.1% weight loss. The proportion of participants achieving 20% or greater weight loss was 36% at the 10mg dose and 40% at the 15mg dose, results approaching what was previously achievable only with bariatric surgery.
Combination evidence
Direct clinical trial evidence for the AOD 9604 plus tirzepatide combination does not exist. This is an important caveat. The rationale for combining them is based on mechanistic complementarity (non-overlapping pathways), individual safety profiles, and clinical observations from practitioners administering the combination. Real-world data from clinics offering this combination protocol suggests enhanced fat loss compared to tirzepatide alone, but these observations come from uncontrolled clinical settings and cannot substitute for randomized controlled trials.
Researchers exploring the peptide research landscape should interpret combination protocols as rational but unproven, guided by biological plausibility rather than definitive clinical evidence.
Who should and should not consider this stack
Not every researcher or patient benefits equally from this combination. Understanding who responds best and who should avoid it prevents wasted resources and potential health risks.
Good candidates for the combination
Researchers already using tirzepatide who have plateaued represent the strongest candidates. If tirzepatide has stopped working or weight loss has stalled despite dose escalation, adding AOD 9604 introduces a new mechanism that can restart progress. The plateau-breaking potential is one of the primary reasons clinicians combine these compounds.
Individuals with significant stubborn fat deposits, particularly visceral abdominal fat, may benefit disproportionately. The beta-3 adrenergic pathway that AOD 9604 activates is highly expressed in visceral adipose tissue, making it a targeted tool for belly fat reduction.
People concerned about muscle loss during GLP-1 therapy may find the combination appealing. While AOD 9604 is not a muscle-building peptide, its selective fat-targeting mechanism means more of the weight lost comes from fat rather than lean tissue. Researchers comparing weight loss peptide stacks often include AOD 9604 for this reason.
Who should avoid this combination
Individuals with a history of thyroid cancer or MEN 2 syndrome should not use tirzepatide, and therefore should not use this combination. This is a black box warning for the entire GLP-1 class based on thyroid C-cell tumor findings in rodent studies.
People with active pancreatitis or a history of pancreatitis should approach tirzepatide with extreme caution and only under close medical supervision.
Pregnant or breastfeeding women should avoid both compounds. There is insufficient safety data for either compound during pregnancy, and animal studies with GLP-1 agonists showed adverse fetal effects. Those researching peptide use during this period should consult the GLP-1 and breastfeeding guide.
Type 1 diabetics should not use tirzepatide. And anyone with severe renal impairment should discuss both compounds with their physician before proceeding.
Finally, anyone not under medical supervision should not combine multiple injectable compounds. Self-administering peptide combinations without bloodwork monitoring and clinical oversight is not responsible research practice. Understanding peptide legality in your jurisdiction is also important before proceeding.
Practical tips for combining AOD 9604 and tirzepatide
Storage and handling
Both compounds require refrigeration after reconstitution. AOD 9604 lyophilized powder should be stored frozen at -20 degrees C until ready for reconstitution. After reconstituting with bacteriostatic water, store at 2-8 degrees C (standard refrigerator temperature) and use within 28-30 days. The peptide storage guide covers optimal conditions for maintaining potency.
Tirzepatide follows similar cold storage requirements. Compounded tirzepatide should be refrigerated consistently and used before the expiration date on the vial. Understanding tirzepatide fridge shelf life prevents using degraded product.
Keep both compounds in separate, clearly labeled vials. Never mix AOD 9604 and tirzepatide in the same vial. Use separate syringes for each injection.
Reconstitution specifics
AOD 9604 typically comes in 5mg vials. Adding 2ml of bacteriostatic water creates a concentration of 2.5mg/ml (2500mcg/ml). At this concentration, a 300mcg dose equals 12 units (0.12ml) on an insulin syringe. The peptide reconstitution guide covers the technique in detail.
Tirzepatide reconstitution depends on the vial concentration. Common formulations include 5mg, 10mg, 15mg, and 30mg vials. Use the tirzepatide reconstitution guide for step-by-step instructions specific to your vial size. The peptide reconstitution calculator eliminates dosing math errors.
Always use slow, gentle injection when adding bacteriostatic water to lyophilized peptide powder. Direct the stream against the vial wall, not directly onto the powder cake. Swirl gently to dissolve. Never shake. Aggressive mixing can denature peptide chains and reduce potency. Understanding how to mix peptides with bacteriostatic water properly is a foundational skill.
Tracking your results
Objective tracking separates productive research from guesswork. For this combination, track the following metrics weekly.
Body weight, first thing in the morning after using the bathroom, same scale every time. Weight fluctuates daily due to water retention, food volume, and hormonal cycles, so weekly averages provide better data than daily numbers.
Waist circumference at the navel. This is the single best proxy for visceral fat changes and responds particularly well to the AOD 9604 mechanism. Take three measurements and average them.
Body composition measurements via DEXA scan or bioimpedance scale every 4-8 weeks. These provide objective data on fat mass versus lean mass changes that the scale alone cannot reveal.
Progress photos every two weeks in consistent lighting, clothing, and positioning. Visual changes often precede scale changes, especially when fat loss is accompanied by lean mass preservation.
Side effect diary noting any GI symptoms, energy changes, sleep quality, or injection site reactions. This documentation is invaluable for protocol adjustments. Reviewing peptide before and after examples from others can help calibrate expectations.
Supplements that support the combination
Certain supplements enhance the effectiveness and tolerability of this stack. Magnesium citrate (400-600mg daily) addresses constipation from tirzepatide and supports hundreds of enzymatic reactions including fat metabolism. Electrolytes (sodium, potassium, magnesium) prevent dehydration-related symptoms during reduced food intake. A comprehensive tirzepatide supplement guide covers the full list.
B12 supplementation deserves special mention. Some compounded tirzepatide formulations include B12 (methylcobalamin), and researchers report improved energy levels with this addition. The tirzepatide with B12 guide explains the rationale. Glycine is another common addition, with the tirzepatide glycine combination showing potential for enhanced nausea management and metabolic support.
Omega-3 fatty acids (2-4g daily) support cardiovascular health during metabolic changes and may enhance insulin sensitivity. Vitamin D (if deficient) plays a role in fat metabolism and should be optimized before starting any fat loss protocol.
Comparing this stack to alternatives
The AOD 9604 plus tirzepatide combination is not the only multi-compound fat loss approach. Understanding how it compares to alternatives helps you determine if it is the right choice for your specific situation.
Tirzepatide alone vs. tirzepatide plus AOD 9604
Tirzepatide alone produces exceptional results. The question is whether adding AOD 9604 justifies the additional cost, injection frequency, and complexity. For researchers who are achieving strong results with tirzepatide alone and losing weight steadily without plateau, adding AOD 9604 is unnecessary. Save it for when you actually need it.
However, if you have hit a plateau on tirzepatide, if you have stubborn visceral fat that is not responding, or if you want to maximize the fat-to-lean-mass loss ratio, the addition of AOD 9604 provides a mechanistically sound enhancement. Think of it as a tool you add when the primary tool needs reinforcement, not something you deploy preemptively.
Semaglutide plus AOD 9604
This combination works similarly in principle but uses a single GLP-1 agonist instead of the dual GLP-1/GIP agonist. Semaglutide vs tirzepatide comparisons consistently show tirzepatide producing greater weight loss. If you have access to tirzepatide, it is the superior GLP-1 class option for combination with AOD 9604. If you are currently on compounded semaglutide, the same principles of combination with AOD 9604 apply, just with somewhat lower expected results from the GLP-1 component.
Tirzepatide plus cagrilintide
Cagrilintide is an amylin analog that provides complementary appetite suppression through a different pathway than GLP-1. The cagrilintide with tirzepatide combination targets appetite and satiety through multiple mechanisms but does not include the direct fat-cell targeting that AOD 9604 provides. This stack maximizes appetite suppression rather than combining appetite control with targeted lipolysis.
The triple stack: tirzepatide plus AOD 9604 plus additional peptides
Some aggressive protocols add additional compounds like MOTS-c (mitochondrial peptide for metabolic optimization) or 5-amino-1MQ (fat metabolism enhancer). While mechanistically interesting, stacking three or more compounds dramatically increases complexity, cost, and potential for unexpected interactions. The guide on combining multiple peptides covers the considerations. For most researchers, the dual AOD 9604 plus tirzepatide combination provides sufficient metabolic coverage without the risks of more complex stacks. The MOTS-c complete guide and 5-amino-1MQ guide cover these compounds independently for those interested.
Comparison table: fat loss peptide combinations
Combination | Primary mechanism | Weight loss potential | Muscle preservation | Complexity | Best for |
|---|---|---|---|---|---|
Tirzepatide alone | Appetite suppression + insulin | 15-22% body weight | Good (75:25 ratio) | Low | Most researchers |
Tirzepatide + AOD 9604 | Appetite + direct fat breakdown | Enhanced beyond tirzepatide | Very good | Moderate | Plateau breakers, stubborn fat |
Semaglutide + AOD 9604 | Appetite + direct fat breakdown | Good | Very good | Moderate | Semaglutide users |
Tirzepatide + cagrilintide | Dual appetite suppression | Very high | Good | Moderate | Maximum appetite control |
Triple stacks | Multiple pathways | Highest theoretical | Variable | High | Advanced researchers only |
Cost considerations and practical budgeting
Running this combination involves ongoing costs that researchers should budget for before committing to a multi-month protocol.
AOD 9604 is relatively affordable in the peptide world. A 5mg vial, which provides approximately 10-16 days of supply at 300-500mcg daily, typically costs significantly less than GLP-1 medications. Monthly AOD 9604 costs vary by source and concentration but remain manageable for most researchers. The peptide cost calculator helps estimate monthly expenses based on your specific dosing protocol.
Tirzepatide represents the larger expense. Compounded tirzepatide is considerably less expensive than brand-name Mounjaro, but still represents a significant monthly investment. Comparing sources for affordable tirzepatide options and compounded tirzepatide pricing can reduce costs substantially.
Additional costs include bacteriostatic water, insulin syringes, alcohol swabs, and periodic bloodwork. These add relatively small amounts to the monthly total but should be factored into the overall budget. Using our peptide dosage calculation guide ensures you order the right quantities and avoid waste.
Reconstitution and preparation checklist
Getting the preparation right is fundamental. Errors during reconstitution or storage directly impact effectiveness and safety.
What you need
Before starting, gather the following supplies. AOD 9604 lyophilized powder (5mg vials). Tirzepatide (your prescribed concentration and quantity). Bacteriostatic water (ensure it contains 0.9% benzyl alcohol). Insulin syringes (29-31 gauge, 0.5-1ml capacity). Alcohol swabs for injection site and vial top preparation. A sharps container for used needle disposal. Refrigerator space for reconstituted vials.
For detailed preparation guidance, the bacteriostatic water measurement guide prevents dosing errors during reconstitution.
Step by step AOD 9604 reconstitution
Clean the vial top with an alcohol swab. Draw 2ml of bacteriostatic water into your syringe. Insert the needle into the AOD 9604 vial at an angle, directing the stream against the glass wall. Inject the water slowly. Do not squirt it directly onto the powder. Remove the syringe and gently swirl the vial until the powder dissolves completely. Label the vial with the date, concentration (2500mcg/ml), and expiration date (28 days from reconstitution). Store immediately in the refrigerator.
This process is identical to standard peptide reconstitution but the concentration math is specific to AOD 9604 vial sizes. The reconstitution calculator eliminates guesswork.
Measuring your AOD 9604 dose
With 2ml of bac water in a 5mg vial (2500mcg/ml concentration), here are common doses in insulin syringe units. For 250mcg: draw to the 10 unit mark (0.10ml). For 300mcg: draw to the 12 unit mark (0.12ml). For 500mcg: draw to the 20 unit mark (0.20ml). Always double-check calculations before injecting. The peptide dosage chart provides a quick reference.
What to eat during AOD 9604 and tirzepatide combination therapy
Nutrition strategy becomes critical during combination therapy. Tirzepatide suppresses appetite so dramatically that some researchers struggle to eat enough, while the fasting requirement for AOD 9604 adds an additional timing constraint.
The daily eating framework
Morning: Inject AOD 9604 fasted. Wait 30-60 minutes. Break the fast with a protein-rich meal (30-40g protein). This first meal is critical because appetite may diminish as tirzepatide effects persist throughout the day. Front-loading protein ensures adequate intake before satiety makes eating difficult later.
Midday: A moderate meal focusing on protein and vegetables. If appetite is low, prioritize protein density. Chicken, fish, eggs, and Greek yogurt deliver maximum protein per calorie. The tirzepatide meal plan provides structured daily templates.
Evening: A lighter meal or snack. Many researchers on tirzepatide find that dinner becomes the most challenging meal to finish. This is normal. Focus on nutrient density rather than volume. Do not force-feed.
Macronutrient targets
Protein: 1.0-1.2 grams per pound of body weight daily. This is the single most important dietary target for body composition during this stack. Non-negotiable. If you are not hitting this target, muscle loss will accelerate regardless of what peptides you are using.
Fat: 0.3-0.5 grams per pound of body weight. Essential fatty acids support hormone production, cell membrane integrity, and fat-soluble vitamin absorption. Do not go below 0.3g/lb even during aggressive caloric restriction.
Carbohydrates: Fill remaining calories after protein and fat targets are met. For most people on this combination, carbohydrate intake naturally decreases as overall caloric intake drops. Prioritize complex carbohydrates (vegetables, whole grains, legumes) over simple sugars.
Understanding optimal food choices during GLP-1 therapy applies equally to tirzepatide-based protocols. The principles of nutrient density, protein prioritization, and eating slowly remain consistent across the GLP-1 class.
Hydration
Dehydration is an underappreciated risk during this combination. Reduced food intake means less water from food sources. AOD 9604 fasting windows temporarily restrict fluid intake options. And tirzepatide GI effects can cause fluid loss. Target at least 80 ounces of water daily, more during exercise or in warm climates. Add electrolytes if you experience lightheadedness, muscle cramps, or headaches.
Exercise optimization during combination therapy
The right exercise approach amplifies the effects of both compounds while protecting lean mass. The wrong approach can accelerate muscle loss and reduce adherence.
Resistance training: the non-negotiable foundation
Strength training at least three times per week preserves muscle mass during caloric deficit more effectively than any supplement, peptide, or dietary strategy. This is supported by decades of research across multiple weight loss contexts. During AOD 9604 and tirzepatide combination therapy, resistance training becomes even more important because the aggressive fat loss rate means the body will readily catabolize muscle if it is not receiving the stimulus to maintain it.
Focus on compound movements. Squats, deadlifts, bench press, overhead press, rows, and pull-ups/lat pulldowns engage the most muscle mass per exercise and produce the strongest muscle preservation signal. Three to four sets of 6-12 repetitions at a challenging weight provides sufficient stimulus. You do not need to train like a competitive bodybuilder. You need to convince your body that its muscle is necessary and should not be broken down for energy. Researching muscle-supporting peptides can complement this training approach for those looking at additional options.
Cardiovascular exercise: strategic use
Morning fasted cardio after AOD 9604 injection takes advantage of elevated free fatty acid levels. Low to moderate intensity cardio (walking, cycling, light jogging at conversational pace) performed during the 30-60 minute fasting window maximizes fat oxidation from mobilized fatty acids. This is one of the most effective synergies in the entire protocol.
High-intensity cardio is less important during this combination. The caloric deficit from tirzepatide combined with AOD 9604 lipolysis provides ample fat loss stimulus without exhausting glycogen reserves through intense cardio. Two to three sessions of 20-40 minute moderate-intensity cardio per week is sufficient. More aggressive cardio schedules risk muscle catabolism, fatigue, and reduced training performance. For researchers interested in the peptide performance optimization angle, balancing recovery with training volume matters.
Recovery considerations
Recovery takes longer during caloric restriction. Period. With both compounds reducing energy intake (tirzepatide through appetite) and mobilizing stored energy (AOD 9604 through lipolysis), the body is working harder to maintain homeostasis. Reduce training volume by 20-30% compared to maintenance-calorie periods. Prioritize sleep (7-9 hours nightly). Allow 48-72 hours between sessions targeting the same muscle groups.
If you experience unusual fatigue, persistent muscle soreness beyond 72 hours, or declining training performance over 2-3 weeks, reduce either training volume or one of the compound doses. Pushing through excessive fatigue during aggressive fat loss protocols leads to overtraining syndrome, which actually increases muscle catabolism and slows fat loss. The tirzepatide body aches guide helps distinguish normal adjustment from overtraining symptoms.
Transitioning off the combination
How you end the combination protocol matters as much as how you start it. Abrupt cessation without a transition plan leads to rebound weight gain, metabolic disruption, and frustration.
Discontinuing AOD 9604
AOD 9604 can be stopped without tapering. Since it does not affect hormone levels, insulin sensitivity, or appetite regulation, there is no rebound effect from discontinuation. Simply stop injecting at the end of your cycle and begin the 4-week break. If you plan to resume, restart at the full dose rather than titrating back up, since the break period allows receptor resensitization.
Reducing tirzepatide
Tirzepatide discontinuation requires more careful planning. Abruptly stopping tirzepatide after months of use leads to rapid appetite return and weight regain in most people. The GLP-1 withdrawal symptom guide covers what to expect. Clinical data shows that most participants in tirzepatide trials regained significant weight within one year of stopping treatment.
If you plan to discontinue tirzepatide, consider a gradual dose reduction over 8-12 weeks. Drop one dose level every 4 weeks (for example, from 10mg to 7.5mg to 5mg to 2.5mg). This allows the body to gradually readjust appetite regulation systems. During this taper, implement aggressive lifestyle strategies: establish consistent meal patterns, maintain protein intake, continue resistance training, and develop hunger management strategies that do not rely on pharmaceutical appetite suppression.
Some researchers transition to maintenance-dose tirzepatide (typically 2.5-5mg weekly) indefinitely rather than stopping completely. This approach mirrors how anti-obesity medications are used in clinical practice, where long-term maintenance therapy prevents weight regain.
Maintaining results long-term
The metabolic benefits of fat loss persist only if the behavioral changes persist. This means the habits established during combination therapy need to become permanent features of your lifestyle. Continued resistance training, adequate protein intake, regular physical activity, and mindful eating are the actual determinants of long-term success. Peptides accelerate the process, but they cannot substitute for sustainable lifestyle changes.
SeekPeptides members access detailed maintenance protocols and long-term strategy guides that help researchers transition from active fat loss phases into sustainable maintenance phases without losing their results.
Frequently asked questions
Can I inject AOD 9604 and tirzepatide at the same site?
No. Always use different injection sites for each compound. This prevents potential local interactions and makes it easier to identify the source of any injection site reactions. Common rotation patterns include AOD 9604 in the lower abdomen and tirzepatide in the upper thigh, alternating sides weekly. The injection technique guide covers site selection in detail.
How long does it take to see results from this combination?
Most researchers notice appetite reduction from tirzepatide within the first week. Scale changes typically appear within 2-3 weeks. Visible body composition changes from the combination usually become apparent at weeks 6-8, when AOD 9604 has been active for 2-4 weeks alongside escalating tirzepatide doses. Maximum results occur during weeks 10-16 when both compounds are at optimized doses. The peptide timeline expectations guide provides detailed week-by-week projections.
Does AOD 9604 affect blood sugar or insulin?
No. This is one of the key advantages of AOD 9604 over full-length growth hormone. Clinical trials confirmed no negative effects on carbohydrate metabolism. AOD 9604 does not raise blood sugar, does not cause insulin resistance, and does not interfere with tirzepatide insulin-sensitizing effects. This metabolic neutrality is precisely why they combine well.
Can I use oral AOD 9604 instead of injectable?
Oral AOD 9604 formulations exist but have significantly lower bioavailability than subcutaneous injection. The clinical trials that showed disappointing results used oral administration. Subcutaneous injection bypasses first-pass hepatic metabolism, delivering more active peptide to target tissues. For combination protocols where maximizing AOD 9604 effectiveness matters, injectable is strongly preferred. Compare injectable vs oral peptide delivery to understand the bioavailability differences.
What if tirzepatide side effects are too severe to continue?
If GI side effects from tirzepatide prevent dose escalation, you have several options. Drop back to the last tolerated dose and stay there for 6-8 weeks before attempting escalation again. Switch to a different GLP-1 agonist like semaglutide, which some people tolerate better. Continue at a lower tirzepatide dose while relying more heavily on AOD 9604 for fat loss. Or explore alternative tirzepatide delivery methods that may reduce GI side effects.
Should I take AOD 9604 on rest days?
Yes. AOD 9604 works by stimulating fat cell receptor pathways, not by enhancing exercise performance. Take it daily regardless of whether you exercise that day. Consistent daily dosing maintains steady beta-3 adrenergic receptor stimulation and produces better cumulative results than intermittent dosing.
Can I drink alcohol while on this combination?
Alcohol is not strictly contraindicated with either compound, but it undermines the protocol in multiple ways. Alcohol inhibits fat oxidation (the body prioritizes metabolizing alcohol over burning fat). It provides empty calories that counteract the caloric deficit from tirzepatide. And GLP-1 agonists change how the body processes alcohol, leading to increased sensitivity and faster intoxication in many users. The alcohol and tirzepatide guide covers the details. If you choose to drink, keep consumption minimal and infrequent.
Is this combination legal?
Tirzepatide is FDA-approved for type 2 diabetes (as Mounjaro) and for obesity (as Zepbound). AOD 9604 is not FDA-approved and is sold as a research compound. Legality varies by jurisdiction and intended use. Understanding peptide legal status in your area is essential before beginning any protocol. Work with a licensed healthcare provider for proper medical oversight.
External resources
PubMed: AOD9604 lipolytic fragment effects on lipid metabolism
Journal of Endocrinology and Metabolism: AOD9604 safety and metabolism
The Lancet: Tirzepatide and muscle composition changes (SURPASS-3 MRI)
For researchers serious about optimizing their fat loss protocols with evidence-based guidance, SeekPeptides offers the most comprehensive resource available, with detailed protocol builders, dosing calculators, stacking guides, and a community of thousands who have navigated these exact questions about combining compounds safely and effectively.
In case I do not see you, good afternoon, good evening, and good night. May your fat cells stay responsive, your lean mass stay protected, and your protocols stay evidence-based.