Feb 18, 2026
Some researchers see dramatic weight loss within weeks. Others spend months chasing results that never come. And increasingly, the difference has nothing to do with the compound itself. It has everything to do with how it enters the body.
Tirzepatide has become the most talked-about weight management peptide in recent memory. The SURMOUNT clinical trials demonstrated average weight loss of 20.2% over 72 weeks at the highest dose, outperforming every other GLP-1 receptor agonist on the market. Those numbers generated massive demand. And with that demand came a question researchers keep asking: can I take tirzepatide as a tablet instead of an injection?
The answer is more complicated than most sources admit. Products marketed as "oral tirzepatide" have appeared from compounding pharmacies, online vendors, and gray-market suppliers. Some come as sublingual drops. Others as orally disintegrating tablets. A few claim to be capsules with absorption enhancers. The problem? None of them are FDA-approved. None have clinical trial data supporting their efficacy. And some have triggered adverse event reports that should concern every serious researcher.
This guide breaks down the complete science behind tirzepatide delivery methods. You will learn exactly why oral tirzepatide faces fundamental biological barriers, what the clinical data actually shows for injectable versus oral delivery, which future oral alternatives are approaching FDA approval, and how to make informed decisions about your tirzepatide protocol. SeekPeptides has compiled the most current research, regulatory updates, and practical guidance to help you navigate this rapidly evolving landscape.
Why tirzepatide only works as an injection right now
Understanding why tirzepatide requires injection means understanding what happens to peptides inside your digestive system. It is not a matter of preference or convenience. Biology dictates the delivery method.
Tirzepatide is a 39-amino-acid peptide. That is a relatively large molecule. When you swallow any peptide of this size, your stomach acid immediately begins breaking it apart. Pepsin, the primary digestive enzyme in your stomach, exists specifically to chop proteins and peptides into smaller fragments. Your body cannot distinguish between a therapeutic peptide and the protein in your lunch. Both get digested.
This is not a minor reduction in potency. Studies on peptide bioavailability show that oral administration of unprotected peptides typically results in less than 1% of the active compound reaching the bloodstream. For context, injectable tirzepatide has approximately 80% bioavailability. The difference between 80% and less than 1% is not something you can overcome by simply taking a higher dose.
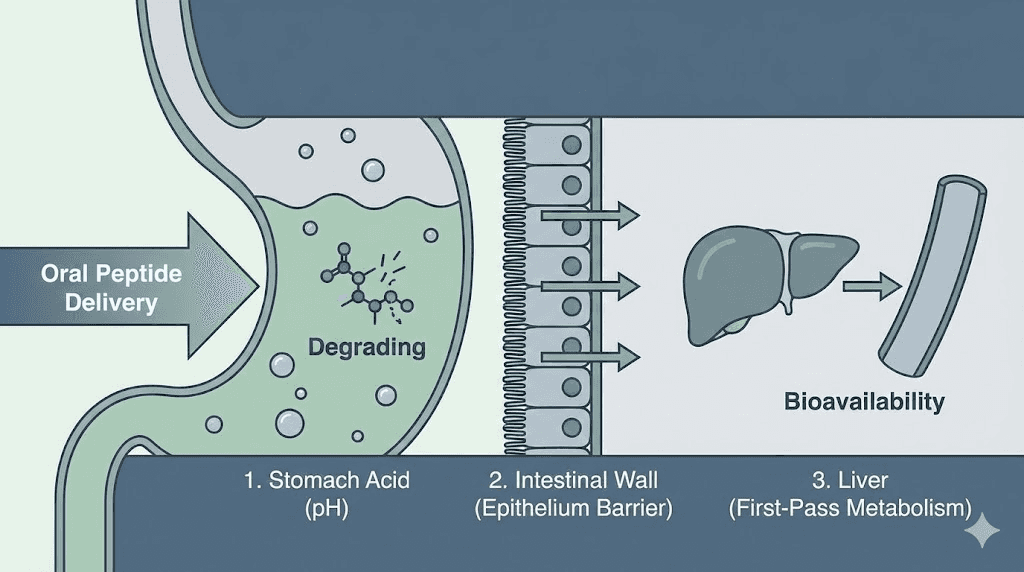
The three barriers to oral peptide delivery
Every peptide faces three distinct biological barriers when taken orally, and tirzepatide drops or tablets face all three simultaneously.
Barrier one: enzymatic degradation. Your gastrointestinal tract contains dozens of proteolytic enzymes. Pepsin in the stomach. Trypsin and chymotrypsin in the small intestine. Carboxypeptidases. Aminopeptidases. Each one targets different bonds within peptide chains. A 39-amino-acid peptide like tirzepatide offers numerous cleavage sites for these enzymes. By the time it reaches the intestinal wall where absorption could theoretically occur, most of the active compound has been reduced to inactive fragments.
Barrier two: the mucosal layer. Even if a peptide survives enzymatic attack, it still needs to cross the intestinal epithelium. This barrier consists of a thick mucus layer, a single layer of epithelial cells connected by tight junctions, and the lamina propria beneath. Tight junctions between epithelial cells have gaps of approximately 0.3 to 1.0 nanometers. Tirzepatide, as a peptide with a molecular weight around 4,810 daltons, is far too large to pass through these gaps by passive diffusion.
Barrier three: first-pass metabolism. Even if a small fraction of tirzepatide somehow survived digestion and crossed the intestinal wall, it would enter the portal vein and travel directly to the liver before reaching systemic circulation. The liver contains additional enzymes that would further degrade any remaining peptide. This hepatic first-pass effect eliminates most of whatever survived the gastrointestinal tract.
These are not theoretical concerns. They are fundamental biological realities that apply to virtually every therapeutic peptide. The challenge of oral peptide delivery has occupied pharmaceutical researchers for decades, and only a handful of compounds have ever overcome these barriers, each requiring specialized formulation technology that does not currently exist for tirzepatide.
What happened with oral semaglutide and why tirzepatide is different
If you are wondering why oral semaglutide exists but oral tirzepatide does not, the answer reveals just how difficult oral peptide delivery truly is.
Rybelsus, the oral form of semaglutide, uses a specialized absorption enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate). This compound was developed specifically to help semaglutide survive the stomach environment and cross the gastric epithelium. SNAC works through multiple mechanisms: it raises the local pH around the tablet, protecting the peptide from acid degradation. It inhibits pepsin activity in the immediate vicinity. And it temporarily increases the permeability of the stomach lining, creating a brief window for absorption.
Even with all of this technology, oral semaglutide has a bioavailability of approximately 0.4% to 1%. That means for every 14 mg of semaglutide in a Rybelsus tablet, only about 0.056 to 0.14 mg actually reaches the bloodstream. The injectable version delivers the full dose. This massive gap in bioavailability explains why oral semaglutide requires strict dosing conditions: patients must take it on an empty stomach, with no more than 4 ounces of water, and then wait at least 30 minutes before eating, drinking, or taking other medications.
The weight loss results reflect this bioavailability difference. In the SURMOUNT-5 trial comparing tirzepatide versus semaglutide, injectable tirzepatide produced 20.2% weight loss at 72 weeks compared to injectable semaglutide at 13.7%. Oral semaglutide produces even less weight loss than its injectable counterpart, typically around 11.4% in clinical trials. The delivery method gap compounds the efficacy gap.
Why SNAC does not work for tirzepatide
SNAC technology was specifically engineered for semaglutide. It is not a universal solution for all peptides. Tirzepatide presents unique formulation challenges that SNAC cannot address.
First, tirzepatide is a larger molecule. Semaglutide has a molecular weight of approximately 4,114 daltons. Tirzepatide weighs approximately 4,810 daltons. That additional mass makes transepithelial transport even more difficult. Second, tirzepatide has a different amino acid sequence with different susceptibility patterns to digestive enzymes. SNAC inhibition of pepsin may not provide sufficient protection for tirzepatide specifically. Third, tirzepatide is a dual GIP/GLP-1 receptor agonist. Its unique mechanism depends on both receptor interactions remaining intact after absorption, and even partial degradation could alter its pharmacological profile in ways that reduce or eliminate the GIP receptor component.
Eli Lilly, the manufacturer of tirzepatide (branded as Mounjaro for diabetes and Zepbound for weight management), has not announced any oral tirzepatide formulation in development. Instead, the company has invested in orforglipron, a completely different molecule designed from the ground up as an oral GLP-1 therapy.
The truth about compounded oral tirzepatide products
Despite the biological impossibility of effective oral tirzepatide without breakthrough formulation technology, products marketed as oral tirzepatide have flooded certain market channels. Understanding what these products actually are, and why they pose significant risks, is essential for any researcher considering them.
Types of compounded oral products
Several categories of non-FDA-approved oral tirzepatide products have appeared.
Sublingual drops claim to deliver tirzepatide through the mucous membranes under the tongue. While sublingual absorption works for some small molecules like nitroglycerin, peptides of tirzepatide size face the same enzymatic and permeability challenges in the oral mucosa as in the gastrointestinal tract. Salivary enzymes begin degrading the peptide immediately. And the sublingual epithelium, while more permeable than the intestinal wall, still presents a significant barrier to molecules of this size. Our detailed review of tirzepatide drops covers the specific concerns with this formulation.
Orally disintegrating tablets (ODTs) dissolve in the mouth and claim to deliver tirzepatide through buccal absorption. Empower Pharmacy notably offered a tirzepatide ODT product during the shortage period. However, these tablets face identical absorption challenges to sublingual drops. The oral mucosa is not designed to absorb large peptides, and no clinical trial has demonstrated that tirzepatide ODTs achieve therapeutic blood levels.
Capsules with absorption enhancers attempt to mimic the SNAC approach used in oral semaglutide. As discussed above, SNAC was specifically designed for semaglutide and has not been validated for tirzepatide. Using unproven absorption enhancer combinations with tirzepatide introduces unknown risks without established efficacy data.
FDA regulatory action
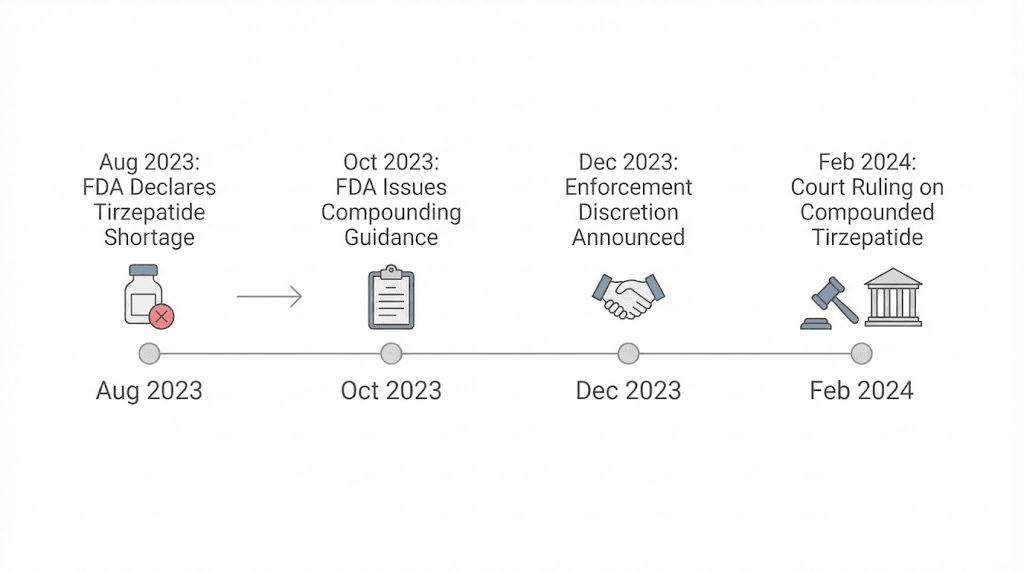
The regulatory landscape for compounded tirzepatide has shifted dramatically. In December 2024, the FDA removed tirzepatide from the drug shortage list. This decision had immediate legal consequences.
Under the Federal Food, Drug, and Cosmetic Act, compounding pharmacies operating under Section 503A could only produce copies of commercially available drugs when those drugs were in shortage. With tirzepatide off the shortage list, the legal basis for compounding evaporated. The FDA set a 60-day transition period for 503A pharmacies (ending February 2025) and a 90-day period for 503B outsourcing facilities (ending March 2025).
In May 2025, the US District Court for the Northern District of Texas upheld the FDA decision, ruling that the agency acted within its statutory authority. This court ruling effectively closed the door on legal compounded tirzepatide in the United States.
As of now, compounded tirzepatide, whether injectable or oral, cannot be legally produced except in very narrow circumstances: when a patient has a documented medical need that cannot be met by the commercially available product, such as an allergy to a specific excipient. Cost, convenience, and preference for oral administration are not valid reasons for compounding.
The FDA has received more than 215 adverse event reports related to compounded tirzepatide products. These reports include contamination, incorrect dosing, and reactions to unknown ingredients. For researchers who previously relied on compounded oral tirzepatide, these regulatory changes mean the products are no longer available through legitimate channels.
Injectable tirzepatide: what the clinical data actually shows
With oral delivery off the table, understanding the full clinical picture for injectable tirzepatide becomes critical. The SURMOUNT trial program represents one of the most comprehensive clinical development programs for any weight management compound.
SURMOUNT-1: the foundational trial
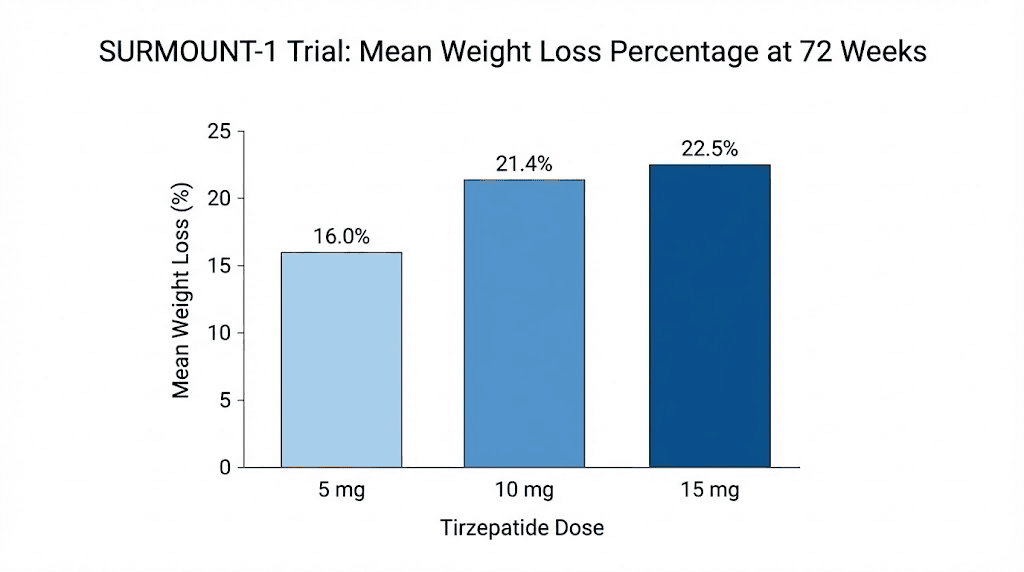
The SURMOUNT-1 trial enrolled 2,539 adults with obesity or overweight (BMI of 30 or higher, or 27 or higher with at least one weight-related complication). Participants received weekly subcutaneous injections of tirzepatide at 5 mg, 10 mg, or 15 mg, or placebo, for 72 weeks alongside lifestyle intervention.
Results at the 15 mg dose were remarkable. Average weight loss reached 20.9%. But the percentage-based milestones tell an even more compelling story. Of participants on the highest dose, 92.7% lost at least 5% of their body weight. That number alone would be impressive for any weight management intervention. Then 84.4% lost at least 10%. And 73.6% achieved 15% or more. More than half, 56.1%, lost 20% or greater. And 38.7% reached the 25% threshold.
These response rates are unprecedented. Previous GLP-1 receptor agonists typically showed 5% weight loss thresholds being met by 70-80% of participants. Tirzepatide pushed those numbers dramatically higher across every milestone.
SURMOUNT-3: adding lifestyle intervention first
SURMOUNT-3 took a different approach. Participants first completed an intensive 12-week lifestyle intervention, losing an average of 6.9% of their body weight. They were then randomized to tirzepatide or placebo for an additional 72 weeks.
The tirzepatide group lost an additional 18.4% from randomization. The placebo group gained 2.5% back. The estimated treatment difference was a staggering 20.8 percentage points. This trial demonstrated that tirzepatide works synergistically with lifestyle modification, each approach amplifying the other.
SURMOUNT-4: the maintenance question
Perhaps the most important trial for long-term researchers was SURMOUNT-4. This study asked a question that keeps people up at night: what happens when you stop tirzepatide?
All participants received tirzepatide for 36 weeks, losing an average of 20.9% of their body weight. They were then randomized to continue tirzepatide or switch to placebo for another 52 weeks.
Those who continued tirzepatide lost an additional 5.5% from week 36 to week 88. Those switched to placebo regained 14.0%. The difference was 19.4 percentage points. But here is the critical finding: 89.5% of participants who continued tirzepatide maintained at least 80% of their initial weight loss through week 88. Only 16.6% of those on placebo achieved the same.
This data has profound implications for researchers evaluating tirzepatide protocols. Weight management with tirzepatide appears to require ongoing treatment. Discontinuation leads to significant regain in most cases. Understanding this reality should inform how anyone approaches their dosing schedule.
SURMOUNT-5: head-to-head against semaglutide
The SURMOUNT-5 trial directly compared tirzepatide against semaglutide, both at their maximum approved doses. Over 72 weeks, tirzepatide produced 20.2% weight loss versus semaglutide at 13.7%. That 6.5 percentage point difference was statistically significant.
This trial matters for researchers deciding between the two compounds. While both produce clinically meaningful weight loss, tirzepatide dual receptor agonism appears to provide a measurable advantage. Our comprehensive semaglutide versus tirzepatide comparison breaks down the full differences between these compounds, and our detailed analysis of side effect profiles helps researchers weigh the tradeoffs.
How tirzepatide injections work in practice
For researchers who have established that injectable tirzepatide is the evidence-based delivery method, understanding the practical aspects of administration is essential. The injection process is straightforward, but proper technique affects both comfort and efficacy.
Injection sites and rotation
Tirzepatide is administered subcutaneously, meaning the needle goes into the fatty tissue just beneath the skin. Three primary injection sites are approved: the abdomen (at least 2 inches from the navel), the front of the thigh, and the back of the upper arm. Each site offers different advantages.
The abdomen typically provides the most consistent absorption. It has reliable subcutaneous fat depth in most individuals and is the easiest site for self-injection. The thigh offers a large surface area for rotation. The upper arm requires assistance from another person for most researchers, making it less practical for solo administration. Our guide on where to inject GLP-1 medications covers site selection in detail, and the broader injection technique guide walks through each step.
Rotation matters more than most researchers realize. Using the same spot repeatedly can cause lipodystrophy, localized changes in fat tissue that affect both appearance and absorption. Maintain at least 1 inch between consecutive injection sites. Some researchers use a systematic rotation pattern: right abdomen, left abdomen, right thigh, left thigh, cycling through a grid pattern within each area.
Proper injection technique
The injection itself follows a simple sequence. Allow the medication to reach room temperature by removing it from the refrigerator approximately 30 minutes before injection. Cold medication causes more discomfort and may affect absorption rates. Clean the injection site with an alcohol swab and allow it to dry completely. Pinch the skin gently to create a fold of subcutaneous tissue. Insert the needle at a 90-degree angle. For prefilled pens, press and hold the injection button until the dose counter shows zero, typically requiring about 10 seconds. Release the skin fold and remove the needle.
Before injecting, always inspect the solution. It should be clear and colorless to slightly yellow. Do not use it if it appears cloudy, discolored, or contains visible particles. Our guide on how to reconstitute tirzepatide covers preparation from lyophilized powder, and understanding proper storage requirements ensures your compound remains viable.
Dosing schedule and titration
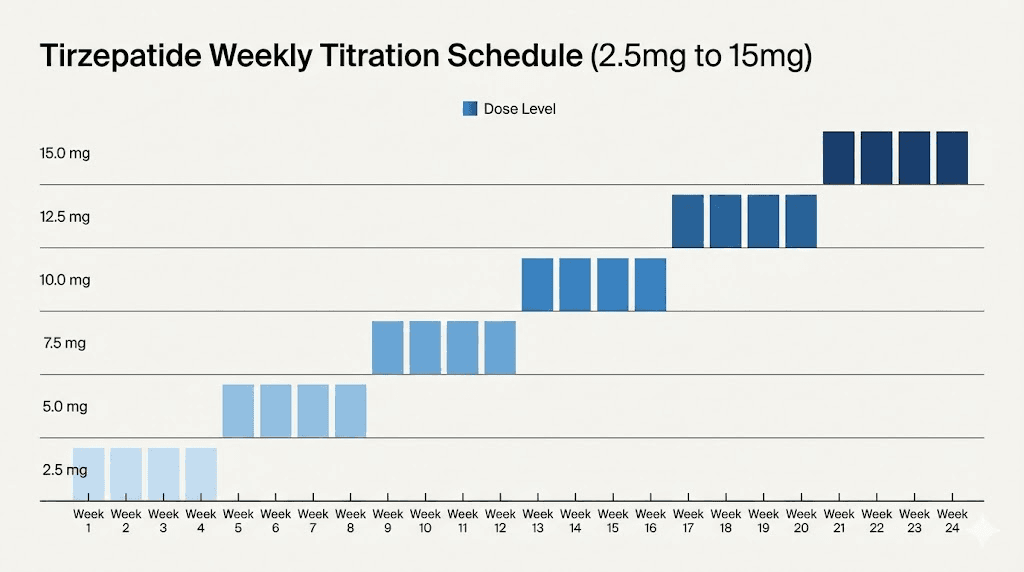
Tirzepatide follows a titration schedule designed to minimize gastrointestinal side effects. The standard progression starts at 2.5 mg weekly for the first four weeks. This is not a therapeutic dose. It exists solely to allow your body to adjust to the medication.
After four weeks, the dose increases to 5 mg weekly. This is the first therapeutic dose level, and some researchers see meaningful results here. After at least four more weeks, the dose can increase to 7.5 mg, then 10 mg, then 12.5 mg, and finally 15 mg, each step maintaining a minimum of four weeks at the current dose.
Not everyone needs to reach 15 mg. The optimal dose is the one that produces satisfactory results with manageable side effects. Our tirzepatide dose chart provides visual reference for the complete titration schedule, and the microdosing chart offers guidance for researchers who prefer a more gradual approach. Understanding the dosage in units is important for anyone using compounded preparations rather than prefilled pens, and our compounded tirzepatide dosage calculator helps with those conversions.
Managing injection side effects
The most common reason researchers ask about tablets is a desire to avoid injection-related side effects. Understanding what these side effects actually involve, and how to manage them, often eliminates the perceived need for oral alternatives.
Injection site reactions
Injection site reactions occur in approximately 1.9% to 4.5% of tirzepatide users. These typically manifest as redness, swelling, or mild itching at the injection site. Most reactions are mild and resolve within hours to days without intervention.
Several strategies minimize injection site reactions. Allowing the medication to reach room temperature before injection reduces discomfort significantly. Using proper technique, particularly inserting at a 90-degree angle with adequate skin pinch, ensures the medication reaches subcutaneous tissue rather than intradermal or intramuscular layers. Rotating sites prevents tissue irritation from repeated injections in the same area. After 3-4 weeks of consistent treatment, most users report that injection site reactions become less frequent and less severe as the body adapts.
Researchers experiencing persistent injection site issues should review our guide on tirzepatide side effects including muscle pain for additional management strategies.
Gastrointestinal side effects
The side effects that concern most researchers are gastrointestinal, not injection-related. Nausea affects approximately 20% of tirzepatide users. Diarrhea occurs in about 16%. Constipation, decreased appetite, and dyspepsia round out the most common complaints. These side effects would occur regardless of delivery method because they result from tirzepatide mechanism of action, not the injection itself.
This point deserves emphasis. Switching to a tablet would not eliminate nausea, diarrhea, or constipation. These effects occur because tirzepatide slows gastric emptying and alters gut hormone signaling. An oral formulation, if one existed, would produce identical gastrointestinal side effects. In fact, oral GLP-1 medications may produce more gastrointestinal side effects because the drug contacts the GI mucosa directly before entering the bloodstream.
Effective management strategies include eating smaller, more frequent meals rather than large ones. Avoiding fatty, greasy, and spicy foods during the titration period. Staying well hydrated. Not lying down immediately after eating. And not eating close to bedtime. Our comprehensive guides on tirzepatide constipation treatment and tirzepatide headache management provide detailed protocols for these specific side effects. For dietary guidance during treatment, our foods to avoid on tirzepatide guide and what to eat on tirzepatide resource offer practical meal strategies.
The tirzepatide diet plan integrates all of these dietary recommendations into a structured framework, and our downloadable meal plan provides ready-to-use templates.
Anxiety and mood effects
Some researchers report mood changes on tirzepatide. Our article on whether tirzepatide can cause anxiety examines the evidence for this reported side effect. GLP-1 receptors exist in the brain, and modulation of these receptors can influence mood, appetite-related anxiety, and stress responses. For most users, these effects are mild and transient. For researchers who experience persistent mood changes, dose adjustment or slower titration often helps.
Tirzepatide tablets that actually exist: what is being sold and why it does not work
Despite everything discussed above, you can still find products sold as tirzepatide tablets online. Understanding what these products actually contain and why they cannot deliver on their promises protects both your health and your investment.
Sublingual tirzepatide products
Sublingual products claim that holding tirzepatide solution under the tongue allows absorption through the sublingual mucosa. This route bypasses the stomach acid problem but faces its own barriers. The sublingual epithelium is thinner than the intestinal wall, which theoretically helps. But salivary enzymes still degrade peptides. The available surface area under the tongue is tiny compared to the gastrointestinal tract. And the contact time before swallowing occurs is brief, usually 30-60 seconds.
For small, lipophilic molecules like nitroglycerin (molecular weight 227 daltons), sublingual delivery works well. For a 4,810-dalton peptide like tirzepatide, the physics simply do not support meaningful absorption. Even optimistic estimates suggest sublingual bioavailability for peptides this size would be well below 1%, potentially yielding no therapeutic effect whatsoever.
Orally disintegrating tablets
ODTs dissolve in the mouth, creating a solution that contacts the oral and buccal mucosa. The theory is similar to sublingual delivery: bypass the stomach by absorbing the drug through the mouth lining. The same limitations apply. Oral mucosal absorption is effective for small molecules but inadequate for large peptides.
During the tirzepatide shortage period, some compounding pharmacies did offer ODT products. However, no clinical data supported their efficacy, and the FDA has never approved any oral tirzepatide formulation. With the end of the shortage and the subsequent regulatory crackdown, these products are no longer legally available from licensed pharmacies.
Troches and lozenges
Troches are medicated lozenges designed to dissolve slowly in the mouth over 15-30 minutes, providing extended contact with the oral mucosa. While this longer contact time theoretically allows more absorption than rapid sublingual administration, the fundamental limitation remains the same. The oral mucosa cannot efficiently absorb peptides of tirzepatide size.
Additionally, a significant portion of any troche formulation is inevitably swallowed, subjecting it to the full gastrointestinal degradation pathway. The small amount that might be absorbed through the oral mucosa is unlikely to reach therapeutic blood levels.
Products sold online without FDA oversight
The most concerning category includes products sold through unregulated online channels. These products may or may not actually contain tirzepatide. Without FDA oversight, there is no guarantee of identity, purity, potency, or sterility. The FDA has specifically warned consumers about unapproved GLP-1 products, noting concerns about contamination, incorrect dosing, and the use of unknown ingredients.
For researchers who value their health and their results, the calculus is straightforward. No oral tirzepatide product has demonstrated efficacy in clinical trials. No oral formulation has FDA approval. The biological barriers to oral peptide delivery are well-established and severe. The risk-reward profile of any currently available oral tirzepatide product is unfavorable by every measure.
The future of oral GLP-1 therapies
The desire for oral weight management medications is legitimate and widely shared. Pharmaceutical companies recognize this demand and are investing billions in developing oral alternatives. The most advanced candidate is not oral tirzepatide. It is orforglipron.
Orforglipron: the oral GLP-1 that might actually work
Orforglipron is a small-molecule GLP-1 receptor agonist developed by Eli Lilly, the same company that makes tirzepatide. But orforglipron is fundamentally different from tirzepatide in a crucial way: it is not a peptide.
Small-molecule drugs are much smaller than peptides. They are typically more resistant to enzymatic degradation. They can cross epithelial barriers more effectively. And they can be formulated as conventional oral tablets without specialized absorption enhancement technology. Orforglipron was designed from the ground up as an oral medication, rather than attempting to force an injectable peptide into an oral format.
ATTAIN trial results
The Phase 3 ATTAIN-1 trial evaluated orforglipron in adults with obesity or overweight. At the highest dose (36 mg daily), orforglipron produced an average of 12.4% weight loss over 72 weeks, compared to 0.9% with placebo. The percentage of participants achieving meaningful weight loss milestones was encouraging: 59.6% lost at least 10% of their body weight, and 39.6% lost at least 15%.
The ATTAIN-2 trial focused on patients with type 2 diabetes and obesity. Results showed clinically meaningful weight loss and improvement in glycemic control, supporting orforglipron use in this population as well.
The ATTAIN-MAINTAIN trial addressed a unique question: can patients switch from injectable GLP-1 therapies (semaglutide or tirzepatide) to oral orforglipron and maintain their weight loss? Participants who had achieved significant weight loss on injectable semaglutide maintained all but 0.9 kg after switching to orforglipron over 52 weeks. Those switching from injectable tirzepatide showed a slightly larger gap, gaining back an average of 5.0 kg, reflecting the greater efficacy of tirzepatide compared to a single-target GLP-1 agonist.
How orforglipron compares to injectable tirzepatide
The comparison is important for researchers weighing convenience against efficacy.
Injectable tirzepatide at maximum dose produces approximately 20% weight loss at 72 weeks. Orforglipron at maximum dose produces approximately 12.4% at the same timepoint. That is a substantial difference. Tirzepatide dual GIP/GLP-1 mechanism provides additional metabolic benefits that a GLP-1-only agonist cannot match.
However, orforglipron offers daily oral dosing versus weekly injection. For researchers who cannot or will not use injections, orforglipron represents a meaningful alternative, even if the absolute weight loss is lower. The convenience factor is real: no needles, no injection site rotation, no refrigeration requirements, no reconstitution.
Eli Lilly has submitted a new drug application to the FDA for orforglipron. Approval for obesity could come by late 2026, with a diabetes indication potentially following. Researchers interested in the broader landscape of oral versus injectable delivery can review our comparison of injectable versus oral peptides for foundational principles that apply beyond tirzepatide specifically.

Cost and accessibility considerations
The tablets-versus-injections conversation often starts with cost concerns. Understanding the financial landscape helps researchers make practical decisions.
Brand-name injectable tirzepatide (Mounjaro or Zepbound) carries a significant price tag without insurance coverage. This cost was one of the primary drivers pushing researchers toward compounded alternatives, including oral formulations. With the FDA ending the compounding pathway for tirzepatide, researchers face a narrower set of options.
Insurance coverage for tirzepatide varies dramatically. Some plans cover Mounjaro for type 2 diabetes but not Zepbound for weight management. Others cover neither. Manufacturer savings programs exist but have eligibility requirements. Our guide on affordable tirzepatide options explores the available cost-reduction strategies in detail.
For researchers considering alternatives, the cost calculus extends beyond the price of the medication itself. Oral products that do not work represent a complete waste of money, regardless of their price point. An injection that costs more but produces documented results is ultimately more cost-effective than a tablet that produces none. And the potential medical costs from adverse events caused by unregulated products must factor into the equation.
Researchers switching from tirzepatide to semaglutide for cost reasons should understand the conversion process and expected difference in outcomes. Our conversion chart provides dose equivalence guidance.
Storage differences: tablets versus injectables
If oral tirzepatide existed and worked, one genuine advantage would be storage simplicity. Tablets generally do not require refrigeration. Injectable tirzepatide has specific storage requirements that researchers must follow to maintain potency.
Unopened tirzepatide pens or vials should be stored in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Once in use, prefilled pens can be stored at room temperature (up to 86 degrees Fahrenheit / 30 degrees Celsius) for up to 21 days. After that, the medication should be discarded.
Our detailed guide on how long tirzepatide lasts in the fridge covers specific shelf life data, and the article on how long compounded tirzepatide can be out of the fridge addresses the handling considerations for reconstituted preparations. Understanding tirzepatide expiration is crucial for maintaining effective protocols. For researchers handling lyophilized powder, our reconstitution guide provides step-by-step instructions.
Supplement and combination strategies while using injectable tirzepatide
Rather than seeking an oral tirzepatide alternative, many researchers optimize their injectable protocols through strategic supplementation. Several compounds have shown particular value alongside tirzepatide.
B12 and tirzepatide
Vitamin B12 supplementation addresses one of the common nutritional concerns with GLP-1 agonist therapy. Reduced food intake from appetite suppression can lead to micronutrient deficiencies over time. B12 is particularly important because tirzepatide may reduce the absorption of nutrients from food by slowing gastric emptying and reducing overall food volume. Our complete guide on tirzepatide with B12 covers dosing, timing, and the specific rationale for this combination.
Glycine supplementation
Glycine has emerged as a popular complementary supplement for tirzepatide users. As the simplest amino acid, glycine supports multiple processes relevant to weight management: sleep quality, insulin sensitivity, and anti-inflammatory effects. Some compounding formulations have included glycine directly with tirzepatide, though this practice is now limited by regulatory changes. Our guide on tirzepatide with glycine and the specific tirzepatide glycine B12 compound guide explain the rationale and evidence for these combinations.
Niacinamide combinations
Niacinamide (vitamin B3) is another compound frequently paired with tirzepatide. It supports cellular energy metabolism and may help protect against some of the metabolic stress associated with rapid weight loss. Our tirzepatide niacinamide guide details the evidence and practical protocols for this combination.
General supplement strategy
Beyond specific combinations, researchers on tirzepatide benefit from comprehensive nutritional support. A high-quality multivitamin, adequate protein intake (to preserve lean mass during weight loss), and specific supplements based on individual needs form the foundation of a sound approach. Our supplements to take with tirzepatide guide provides a complete framework for nutritional optimization during GLP-1 therapy.
Who should consider injectable tirzepatide versus waiting for oral alternatives
The decision between starting injectable tirzepatide now and waiting for oral options like orforglipron depends on individual circumstances, goals, and timeline.
Strong candidates for injectable tirzepatide now
Researchers with significant weight-related health concerns should not delay treatment for the sake of convenience. Tirzepatide clinical trials demonstrated improvements in blood pressure, lipid profiles, inflammatory markers, and insulin sensitivity that begin early in treatment. Waiting 12-18 months or more for an oral alternative that produces approximately 40% less weight loss means 12-18 months of continued metabolic stress.
Researchers comfortable with injections, or willing to learn proper technique, should recognize that weekly subcutaneous injection is a minor inconvenience compared to the magnitude of benefit. The injection itself takes less than 30 seconds. The entire preparation-to-completion process, including alcohol swab, skin pinch, and injection, requires under two minutes once per week. Many researchers report that injection anxiety disappears entirely after the first few weeks.
Our resource on tirzepatide before and after results shows what real users have achieved, and the men before and after tirzepatide page focuses on male-specific outcomes.
Potential candidates for waiting
Researchers with a genuine needle phobia, a clinical condition characterized by vasovagal responses to needle exposure, may legitimately need to wait for oral alternatives. This is different from mild injection anxiety, which typically resolves with experience.
Researchers who are not in urgent need of weight management and who prefer the convenience of daily pills over weekly injections may choose to wait for orforglipron approval, understanding the trade-off in efficacy.
Researchers who have already tried injectable tirzepatide and experienced intolerable fatigue or other side effects may want to see whether an oral GLP-1 agonist produces a more tolerable side effect profile, though gastrointestinal effects are likely to be similar regardless of delivery route.
Practical comparison: tablets versus injections at a glance
The following comparison summarizes the key differences between hypothetical oral tirzepatide and proven injectable tirzepatide for researchers making delivery method decisions.
Factor | Oral tirzepatide (not FDA-approved) | Injectable tirzepatide (FDA-approved) |
|---|---|---|
FDA status | Not approved, not legal | Approved (Mounjaro, Zepbound) |
Clinical trial data | None | Extensive (SURMOUNT 1-5) |
Bioavailability | Estimated less than 1% | Approximately 80% |
Weight loss evidence | No data | 15-21% at 72 weeks |
Administration | Daily (theoretical) | Once weekly |
Storage | Room temperature (theoretical) | Refrigerated, room temp up to 21 days |
Dosing precision | Uncertain (absorption variable) | Precise (full dose delivered) |
Safety data | 215+ adverse event reports (compounded) | Extensive safety profile from trials |
Cost | Variable, no insurance coverage | Insurance may cover for diabetes |
Availability | Not legally available | Widely available by prescription |
The table tells a clear story. Every measurable factor favors injectable tirzepatide. The only potential advantages of an oral formulation, convenience and needle avoidance, are irrelevant when the oral product does not actually work.
Making the switch from compounded oral to injectable
For researchers who previously used compounded oral tirzepatide products and are transitioning to injectable administration, the switch requires careful consideration of dosing and expectations.
If you were taking a compounded oral product, it is likely that you were receiving subtherapeutic blood levels of tirzepatide (if any). Starting injectable tirzepatide from scratch, following the standard titration beginning at 2.5 mg, is the safest approach. Do not attempt to estimate your previous oral dose and convert it to an injectable equivalent. The bioavailability difference makes any such calculation meaningless.
Expect to experience the full range of side effects that are typical during titration. GI effects like nausea, changes in appetite, and altered bowel habits are common as your body adjusts to therapeutic blood levels that you may not have experienced with oral products. These effects typically diminish within the first few weeks at each dose level.
Track your results carefully. Our timeline for tirzepatide results provides realistic expectations for when various outcomes typically appear. And if you experience a plateau early in treatment, our troubleshooting guides can help identify common issues.
Understanding tirzepatide pharmacokinetics: why weekly injection works
One reason some researchers seek daily oral options is a misunderstanding of tirzepatide pharmacokinetics. Understanding why weekly dosing is sufficient, and even preferable, can reshape how you think about delivery.
Tirzepatide has a half-life of approximately five days (roughly 113 hours). After a single subcutaneous injection, plasma concentrations rise over 8-72 hours, reaching peak levels typically within 24-72 hours. The concentration then declines gradually over the following days, but the five-day half-life means that significant drug levels persist throughout the week.
After several weeks of weekly dosing, tirzepatide reaches a steady state where the amount entering the body each week approximately equals the amount being eliminated. This creates a relatively stable therapeutic level with moderate peaks and troughs. The peaks are not dramatically higher than the troughs because the long half-life smooths out the concentration curve.
This pharmacokinetic profile is actually an advantage of injectable delivery. A daily oral dose would create more pronounced peaks and troughs in blood concentration, potentially increasing side effects (from higher peaks) and reducing efficacy between doses (from lower troughs). The weekly injection delivers a steadier therapeutic effect with less dosing complexity.
For researchers interested in the detailed unit-by-unit calculations, our guides on how many units is 2.5 mg, 5 mg in units, 7.5 mg in units, 20 units to mg, 40 units to mg, and 50 units to mg provide precise conversion references.
What about other oral peptide delivery technologies in development
Beyond orforglipron, several emerging technologies could eventually make oral tirzepatide (or similar dual-agonist peptides) feasible. Researchers monitoring the field should be aware of these developments, even though none are close to market for tirzepatide specifically.
Permeation enhancers beyond SNAC
Researchers are exploring new classes of permeation enhancers that may work for larger peptides. These include medium-chain fatty acid derivatives, bile salt conjugates, and synthetic surfactants that temporarily disrupt tight junctions in the intestinal epithelium. Early-stage research has shown improved bioavailability for various peptides, but nothing has reached clinical trials for tirzepatide-class molecules.
Nanoparticle delivery systems
Nanoparticle encapsulation protects peptides from enzymatic degradation while facilitating transport across the intestinal barrier. Various nanoparticle materials, including chitosan, PLGA (poly lactic-co-glycolic acid), and lipid-based systems, have shown promise in preclinical models. The challenge is scaling these technologies to produce consistent, commercially viable oral peptide products.
Enteric-coated formulations with site-specific release
Rather than attempting stomach absorption like SNAC, some approaches target specific regions of the small intestine where enzymatic activity is lower and permeability may be higher. These formulations use pH-sensitive coatings that dissolve only in the appropriate intestinal environment. While conceptually promising, achieving reliable absorption of large peptides in the intestine remains technically challenging.
Mucoadhesive technologies
Mucoadhesive formulations stick to the intestinal mucosa, prolonging contact time and potentially improving absorption. Combined with permeation enhancers and enzyme inhibitors, mucoadhesive systems could theoretically deliver therapeutic levels of large peptides. This remains an area of active research rather than imminent commercial application.
The honest assessment: effective oral delivery of tirzepatide-size peptides is likely years away. Orforglipron sidesteps the problem entirely by using a small molecule rather than a peptide. Researchers who need GLP-1 therapy now should not wait for theoretical oral tirzepatide formulations that may never materialize.
Comparing delivery methods across the GLP-1 class
Placing tirzepatide in the broader context of GLP-1 therapies helps researchers understand where different delivery methods stand.
Compound | Delivery | Weight loss (72 weeks) | Mechanism | Status |
|---|---|---|---|---|
Tirzepatide | Weekly injection | ~20% | Dual GIP/GLP-1 | FDA-approved |
Semaglutide (injectable) | Weekly injection | ~14% | GLP-1 only | FDA-approved |
Semaglutide (oral) | Daily tablet | ~11% | GLP-1 only | FDA-approved |
Orforglipron | Daily tablet | ~12% | GLP-1 only | FDA review |
Retatrutide | Weekly injection | ~24% | Triple agonist | Phase 3 trials |
The pattern is clear. Injectable delivery consistently outperforms oral delivery within the same drug class. And dual or triple agonist mechanisms outperform single-target approaches. Tirzepatide sits in the sweet spot of proven efficacy and regulatory approval, making it the strongest evidence-based choice for researchers right now.
For those interested in the even more potent retatrutide, which adds glucagon receptor agonism to the GIP/GLP-1 mechanism, our guides on retatrutide availability, dosage charts, and the retatrutide versus semaglutide comparison provide comprehensive coverage. For those comparing current options, mazdutide versus tirzepatide examines another emerging dual agonist. And our comparison of phentermine versus semaglutide covers how older weight management drugs stack up against the new generation.
Researchers wondering about combining phentermine with semaglutide or considering a switch between tirzepatide and semaglutide will find detailed protocols in those respective guides. Understanding the dosage differences between tirzepatide and semaglutide is essential for anyone comparing these two leading compounds.
What researchers who want oral convenience should actually do
If you are drawn to oral delivery for practical reasons, here are evidence-based approaches that actually work.
Approach one: start with injectable tirzepatide now, switch later. Begin the standard tirzepatide titration schedule. Achieve your weight management goals using the most effective tool available. When orforglipron receives FDA approval, the ATTAIN-MAINTAIN trial data suggests you can transition to oral therapy while maintaining most of your results. This approach maximizes initial weight loss and then transitions to oral maintenance.
Approach two: use oral semaglutide (Rybelsus) if injectable is not an option. Oral semaglutide is the only FDA-approved oral GLP-1 therapy currently available. It produces less weight loss than injectable tirzepatide (approximately 11% versus 20%), but it is a legitimate, evidence-based option for researchers who cannot use injections. Our guide on oral semaglutide provides detailed information on this option.
Approach three: address injection anxiety directly. Most injection anxiety resolves with proper preparation and technique. Using the smallest gauge needle appropriate for subcutaneous injection (typically 30-31 gauge), allowing medication to reach room temperature, and practicing on a practice pad (available from medical supply stores) can transform the experience. After 2-3 injections, most researchers report minimal or no anxiety. The step-by-step injection guide covers everything from preparation to aftercare.
Approach four: optimize your protocol to minimize injection frequency concerns. Tirzepatide is already a once-weekly injection. That is 52 injections per year. Compared to daily oral semaglutide at 365 doses per year, the weekly schedule is actually simpler. Researchers who organize their protocol around a consistent injection day (same day, same time each week) find that it becomes routine rapidly. Tools like our dose chart and the semaglutide dosage calculator help researchers stay organized with their protocols.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members access personalized protocol builders, detailed dosing guides, and expert-reviewed resources that account for the individual factors most generic sources completely ignore.
Frequently asked questions
Can you take tirzepatide as a pill?
No. Tirzepatide is only FDA-approved as a subcutaneous injection (Mounjaro for diabetes, Zepbound for weight management). No oral tirzepatide formulation has been approved or shown effective in clinical trials. Products sold as oral tirzepatide are not FDA-approved and lack safety and efficacy data. Learn more about the oral versus injection comparison for tirzepatide.
Is oral tirzepatide as effective as the injection?
No products currently marketed as oral tirzepatide have demonstrated clinical efficacy. The biological barriers to oral peptide delivery, including stomach acid degradation and poor intestinal absorption, mean that oral tirzepatide cannot achieve the therapeutic blood levels that injectable tirzepatide delivers with approximately 80% bioavailability. For detailed efficacy data, see our guide on how quickly tirzepatide works.
When will oral tirzepatide be available?
There is no oral tirzepatide formulation in development. Eli Lilly is instead developing orforglipron, a small-molecule oral GLP-1 agonist that has completed Phase 3 trials showing 12.4% weight loss. Orforglipron could receive FDA approval by late 2026. It is a GLP-1-only agonist, not a dual GIP/GLP-1 agonist like tirzepatide, so efficacy will be somewhat lower than injectable tirzepatide.
Are compounded tirzepatide tablets safe?
Compounded tirzepatide tablets lack clinical safety data and are no longer legally available in the United States following the FDA removal of tirzepatide from the drug shortage list. The FDA received over 215 adverse event reports related to compounded tirzepatide products. For safe, evidence-based options, stick with FDA-approved injectable tirzepatide from licensed pharmacies. Read about compounded tirzepatide safety considerations.
Can I switch from compounded oral tirzepatide to the injection?
Yes, and this is recommended. Start the standard injectable titration at 2.5 mg weekly regardless of your previous oral dose, because the bioavailability of compounded oral products is unknown and likely minimal. Follow the standard dosing schedule and titrate upward every four weeks as tolerated.
What is the difference between orforglipron and tirzepatide?
Orforglipron is a small-molecule GLP-1 receptor agonist taken as a daily oral pill. Tirzepatide is a peptide-based dual GIP/GLP-1 receptor agonist given as a weekly injection. Tirzepatide produces approximately 20% weight loss at 72 weeks versus approximately 12.4% for orforglipron. Tirzepatide dual mechanism provides additional metabolic benefits. Orforglipron advantage is oral convenience. See our detailed comparison of GLP-1 therapies.
Why does tirzepatide need to be injected?
Tirzepatide is a 39-amino-acid peptide that is rapidly destroyed by stomach acid and digestive enzymes when taken orally. Subcutaneous injection bypasses the digestive system entirely, delivering approximately 80% of the dose directly into the bloodstream. This is why injectable tirzepatide produces dramatically better results than any oral peptide formulation. Our injection guide covers proper technique.
Does tirzepatide injection hurt?
Most users describe tirzepatide injections as causing minimal discomfort, less painful than a typical blood draw. Using a fine-gauge needle (30-31 gauge), allowing the medication to reach room temperature, and rotating injection sites all reduce discomfort. Injection site reactions (redness, mild swelling) occur in under 5% of users and typically resolve within hours.
External resources
New England Journal of Medicine: SURMOUNT-1 tirzepatide trial results
National Institutes of Health: SNAC absorption enhancer technology review
In case I do not see you, good afternoon, good evening, and good night. May your injections stay painless, your protocols stay effective, and your results stay consistent.