Feb 15, 2026

At the molecular level, semaglutide triggers something most people never think about. It slows gastric emptying. It reduces stomach acid production. It changes the entire environment your gut uses to absorb nutrients. And one nutrient takes a particularly hard hit: vitamin B12.
That is not speculation. A study tracking patients on semaglutide for 12 months found mean vitamin B12 levels dropped from 567 to 494 micrograms per deciliter, a significant decline that happened quietly while patients focused on their shrinking waistlines. Another two-year study of 500 type 2 diabetes patients showed 15% experienced decreased B12 levels, with 5% developing outright deficiency.
This is where methylcobalamin enters the conversation.
Compounding pharmacies now combine semaglutide with methylcobalamin, the bioactive form of vitamin B12, into a single formulation. The idea is straightforward: if GLP-1 therapy depletes B12, why not deliver both in one dose? But the reality is more complicated than that pitch suggests. Not all B12 forms are equal. Not all delivery methods work the same way. And whether you actually need this combination depends on factors most prescribers never bother to check. This guide covers every angle of the semaglutide methylcobalamin combination, from the science behind B12 depletion during GLP-1 therapy to the specific formulations available, the dosing protocols clinicians use, and the honest assessment of whether this compounded product delivers on its promises. SeekPeptides members get access to detailed protocol comparisons, and here we break down the essential information every researcher needs.
Why semaglutide depletes vitamin B12 in the first place
Understanding the semaglutide methylcobalamin combination requires understanding the problem it attempts to solve. B12 absorption is not a passive process. It is one of the most complex nutrient absorption pathways in the human body, and semaglutide interferes with it at multiple points simultaneously.
Here is how B12 absorption normally works. You eat food containing B12. Stomach acid and pepsin separate the B12 from food proteins. The free B12 binds to a protein called R-binder (haptocorrin) in the stomach. In the small intestine, pancreatic enzymes release B12 from R-binder. The freed B12 then binds to intrinsic factor, a glycoprotein produced by parietal cells in the stomach lining. This B12-intrinsic factor complex travels to the terminal ileum, where specialized receptors absorb it into the bloodstream.
Every single step requires specific conditions. Adequate stomach acid. Sufficient intrinsic factor production. Normal gastric emptying speed. Healthy intestinal transit time.
Semaglutide disrupts at least three of those conditions.
First, GLP-1 receptor agonists suppress gastric acid secretion. Less acid means less B12 gets liberated from food proteins in the first place. Second, semaglutide dramatically slows gastric emptying, which changes the timing and efficiency of the entire absorption cascade. Food sits in the stomach longer, but that does not mean more B12 gets absorbed. The altered transit actually disrupts the carefully timed handoff between R-binder in the stomach and intrinsic factor in the duodenum. Third, the reduced caloric intake that comes with appetite suppression means less dietary B12 enters the system to begin with.
The result is a triple threat to B12 status. Less B12 consumed. Less B12 liberated from food. Less B12 properly absorbed.
The clinical evidence for B12 depletion on GLP-1 therapy
The data is consistent, if not yet overwhelming. Multiple studies confirm that semaglutide treatment correlates with declining B12 levels over time.
The Japanese study mentioned earlier tracked patients who received once-weekly semaglutide after sleeve gastrectomy. Along with significant weight loss, they observed meaningful decreases in serum albumin, vitamin B12, and zinc. The B12 decline from 567 to 494 mcg/dL represented a 13% drop in 12 months.
A larger analysis of 500 type 2 diabetes patients over two years found more granular results. Fifteen percent experienced measurable B12 decline. Five percent crossed into clinical deficiency territory. The patients at highest risk were those on higher doses, those with pre-existing borderline B12 levels, those with a history of metformin use (which independently depletes B12), and those over age 60.
Now here is the nuance that most articles miss. A 13% decline in B12 levels does not automatically mean deficiency. Many people start with levels well above the minimum threshold. A drop from 567 to 494 still leaves you in the normal range. The people who run into trouble are those who started closer to the lower end of normal, around 200-300 pg/mL, where even a modest decline pushes them into deficiency.
The symptoms of B12 deficiency overlap uncomfortably with common semaglutide side effects. Fatigue. Brain fog. Nausea. Tingling in the extremities. Mood changes. This overlap makes B12 deficiency easy to miss during GLP-1 therapy, because both the patient and the prescriber attribute these symptoms to the medication itself rather than investigating a correctable nutritional deficit.
Who is most at risk for B12 depletion on semaglutide
Not everyone on semaglutide needs to worry about B12 equally. Certain populations face substantially higher risk.
People over 60 absorb B12 less efficiently even without medication. Add semaglutide to the mix and the decline accelerates. Metformin users, common among type 2 diabetes patients who then add semaglutide, face a double hit since metformin independently reduces B12 absorption by 10-30% over long-term use. Patients with a history of gastrointestinal issues including gastritis, acid reflux treated with PPIs, or any condition affecting stomach lining integrity already have compromised B12 absorption pathways.
Vegetarians and vegans on semaglutide face the highest risk. Their dietary B12 intake is already limited to fortified foods and supplements. When semaglutide further impairs absorption, the math gets ugly fast.
People who have undergone bariatric surgery before starting semaglutide also need close monitoring. Procedures like gastric bypass or sleeve gastrectomy permanently alter the stomach anatomy, reducing intrinsic factor production and the surface area available for B12 absorption.

What methylcobalamin actually is and why it differs from other B12 forms
Vitamin B12 exists in four chemical forms. Cyanocobalamin. Methylcobalamin. Hydroxocobalamin. Adenosylcobalamin. Each has different properties, different bioavailability, and different roles in the body. The distinction matters when choosing a semaglutide B12 combination product.
Cyanocobalamin is synthetic. It does not exist in nature. Your body must convert it into one of the active forms before it can use it. That conversion happens in the liver and requires specific enzymes, energy, and cofactors. The process works for most people, but not all. Some individuals, particularly those with MTHFR gene variants, convert cyanocobalamin poorly.
Methylcobalamin is the bioactive form. Your body uses it directly without conversion. It serves as a cofactor for the enzyme methionine synthase, which converts homocysteine to methionine. This reaction is critical for DNA synthesis, red blood cell formation, and nervous system function. Methylcobalamin participates directly in methylation reactions throughout the body, influencing gene expression, neurotransmitter production, and detoxification pathways.
The difference matters clinically. Research shows that methylcobalamin has higher retention in the body compared to cyanocobalamin. While cyanocobalamin may have slightly better initial absorption rates, more of it gets excreted rather than utilized. Methylcobalamin stays in tissues longer and reaches the nervous system more effectively.
For semaglutide users specifically, the methylcobalamin advantage is about bypassing a compromised absorption system. Since semaglutide already impairs the normal B12 absorption pathway, using a form that requires fewer metabolic steps to become active reduces the total burden on an already stressed system.
Methylcobalamin vs cyanocobalamin in semaglutide formulations
Compounding pharmacies use both forms. The choice between them is not arbitrary, but the reasoning differs depending on the delivery method.
For injectable semaglutide formulations, most compounding pharmacies pair semaglutide with cyanocobalamin (vitamin B12). The typical concentration is 1 mg cyanocobalamin per mL of compounded semaglutide solution. Cyanocobalamin is more stable in solution and withstands the compounding process better. When injected subcutaneously, the absorption bypass makes the conversion issue less relevant since the B12 enters the bloodstream directly, bypassing the gut entirely.
For oral formulations, the picture changes. Empower Pharmacy specifically chose methylcobalamin for their semaglutide orally disintegrating tablet (ODT). The reasoning: when the tablet dissolves on the tongue, some absorption happens through the oral mucosa (buccal absorption). Methylcobalamin, being the active form, can be utilized by tissues immediately after buccal absorption without needing liver conversion. This is a meaningful advantage for oral delivery because it partially sidesteps the very gut absorption problems that semaglutide creates.
The practical comparison:
Factor | Methylcobalamin | Cyanocobalamin |
|---|---|---|
Bioactive form | Yes, used directly | No, requires conversion |
Tissue retention | Higher | Lower, more excreted |
Nervous system access | Superior | Adequate after conversion |
Stability in solution | Lower, light-sensitive | Higher, more stable |
Best delivery method | Oral, sublingual, ODT | Injectable, oral |
MTHFR variant friendly | Yes | May be problematic |
Cost | Higher | Lower |
Neither form is universally superior. The best choice depends on the delivery method, individual genetics, and clinical goals. For the specific semaglutide methylcobalamin ODT formulation, using methylcobalamin makes pharmacological sense given the buccal absorption route.
Available semaglutide methylcobalamin formulations
The semaglutide methylcobalamin market is smaller and more specific than the broader compounded semaglutide market. While dozens of pharmacies compound semaglutide with cyanocobalamin injections, the methylcobalamin combination is primarily available as an oral formulation.
Empower Pharmacy semaglutide methylcobalamin ODT
Empower Pharmacy offers the most well-known semaglutide methylcobalamin product: an orally disintegrating tablet (ODT) available through their 503A compounding pharmacy with a patient-specific prescription.
Three strength combinations are available:
2 mg semaglutide / 0.1 mg methylcobalamin (starting dose)
5 mg semaglutide / 0.1 mg methylcobalamin (intermediate dose)
12 mg semaglutide / 0.1 mg methylcobalamin (higher dose)
The methylcobalamin dose remains constant at 0.1 mg (100 mcg) across all three strengths. This provides approximately 4,167% of the daily recommended intake of B12, which is deliberately supraphysiological to compensate for the reduced absorption caused by semaglutide.
Administration is once daily. Place the tablet on the tongue and let it dissolve completely, which takes 30 to 60 seconds. Do not chew, crush, or swallow whole. Avoid eating or drinking for at least 30 minutes after the tablet dissolves. This last point is critical because food and beverages can interfere with the mucosal absorption of semaglutide, significantly reducing bioavailability.
How the ODT formulation works
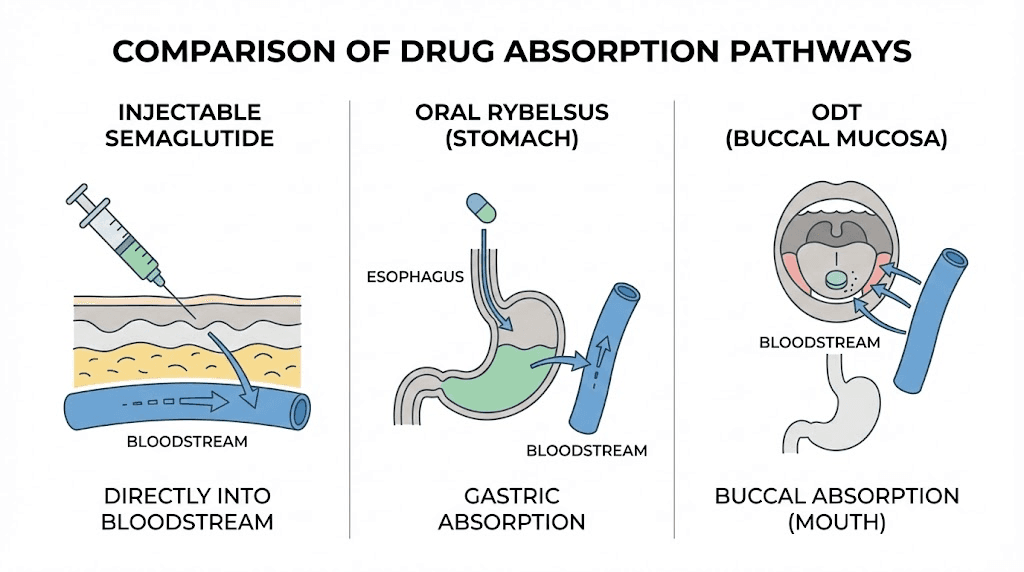
The orally disintegrating tablet approach is fundamentally different from both the standard injectable semaglutide and the FDA-approved oral semaglutide (Rybelsus).
Rybelsus uses a SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate) absorption enhancer that creates a localized pH change in the stomach, allowing semaglutide to cross the gastric mucosa. It requires taking on a completely empty stomach with no more than 4 ounces of water, then waiting 30 minutes before eating or drinking anything else.
The compounded ODT takes a different approach. By dissolving on the tongue, it aims for buccal and sublingual absorption, where the semaglutide and methylcobalamin cross the oral mucosa directly into the venous drainage leading to the superior vena cava. This bypasses first-pass metabolism in the liver and avoids the stomach entirely.
Research on sublingual semaglutide absorption is limited but promising. A rat study found that at equivalent doses, sublingual semaglutide achieved significantly higher area under the curve (AUC) than oral administration: 82.53 vs 15.08 ng*h/mL. Human data on compounded ODT formulations is still emerging.
The absorption pathway for the methylcobalamin component is arguably more important. Buccal absorption of methylcobalamin bypasses the entire gastric absorption pathway that semaglutide disrupts. No need for stomach acid. No dependence on intrinsic factor. No reliance on terminal ileum receptors. The B12 goes directly from the oral mucosa into the bloodstream in its active form.
Semaglutide methylcobalamin dosing protocols
Dosing semaglutide methylcobalamin ODT requires a different approach than injectable semaglutide. The bioavailability is lower, the absorption is more variable, and the daily dosing schedule replaces the once-weekly injection rhythm most users are familiar with.
Standard titration protocol
The typical titration mirrors the general semaglutide dosing approach but with oral-specific adjustments:
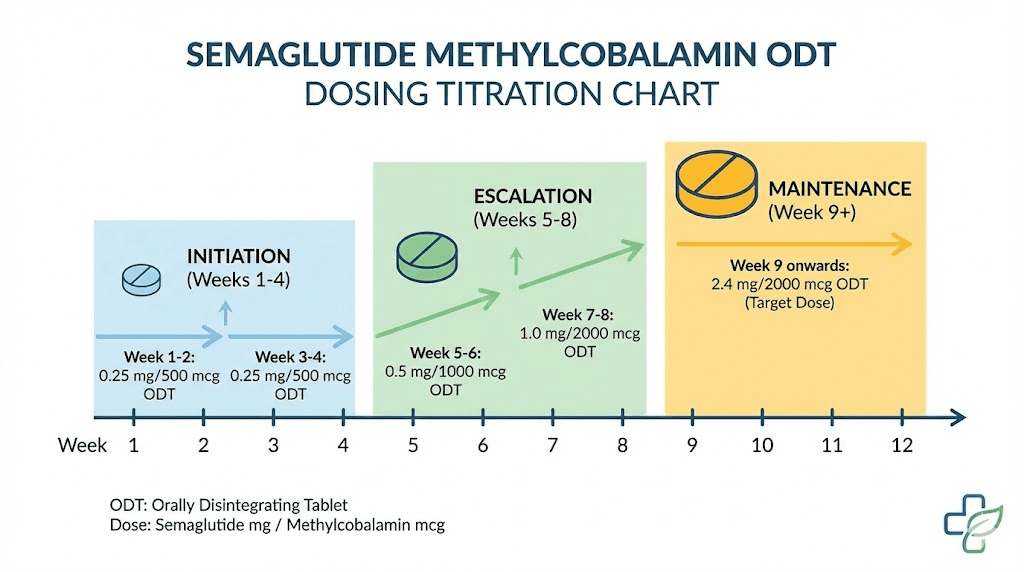
Phase 1 (Weeks 1 through 4): Start with the 2 mg/0.1 mg tablet once daily. This allows the body to adjust to the GLP-1 effects through a low dose that minimizes gastrointestinal side effects. The methylcobalamin begins providing B12 supplementation immediately.
Phase 2 (Weeks 5 through 8): If tolerated, increase to the 5 mg/0.1 mg tablet once daily. Most patients notice meaningful appetite suppression at this level. Nausea, if present, typically peaks during this transition and improves within 1 to 2 weeks.
Phase 3 (Week 9 and beyond): For patients who need additional effect, the 12 mg/0.1 mg tablet is available. Not all patients will require or tolerate this dose. The decision to escalate should be based on weight loss trajectory, appetite control, and side effect profile.
The methylcobalamin component stays constant across all doses. This means you get the same 100 mcg of active B12 regardless of the semaglutide dose. For most people, this amount is adequate. For those in higher-risk categories (over 60, metformin users, vegans), additional B12 supplementation beyond the formulation may be warranted.
Timing and administration details
The timing matters more than most people realize.
Take the ODT first thing in the morning on an empty stomach. Place it on the tongue. Let it dissolve completely without moving it around or chewing. Wait at least 30 minutes before consuming any food, beverages, or other medications. This fasting window is essential for adequate buccal absorption of the semaglutide component.
Some clinicians recommend keeping the dissolved tablet residue in the mouth for an additional 2 to 3 minutes after the tablet has physically dissolved, as residual semaglutide may continue absorbing through the oral mucosa during this period.
Store the tablets at room temperature, between 68 and 77 degrees Fahrenheit. Protect from moisture and light. Keep in the original packaging until use. Methylcobalamin is light-sensitive and degrades with UV exposure, so never store these tablets in a clear container on a windowsill or under bright lights.
Switching from injectable semaglutide to methylcobalamin ODT
Patients switching from injectable semaglutide to the methylcobalamin ODT formulation face a dosing conversion challenge. The bioavailability of oral semaglutide is dramatically lower than injectable. FDA-approved Rybelsus requires 14 mg orally to approximate the effect of about 1 mg injected. The compounded ODT may have different bioavailability characteristics since the absorption pathway differs from Rybelsus.
There is no validated conversion chart between injectable and ODT semaglutide doses. Clinicians typically start patients at the middle ODT dose (5 mg) when switching from low-dose injectables (0.25 to 0.5 mg weekly) and titrate based on response. The transition often involves a period of reduced efficacy as the body adjusts to the new delivery method and absorption profile.
For those interested in conversion between different GLP-1 formulations, the general principle is to prioritize symptom management over dose equivalence. If appetite suppression and weight loss continue at a satisfactory rate, the dose is adequate regardless of what the injectable equivalent might be.
Injectable semaglutide with B12 vs methylcobalamin ODT: choosing the right formulation
The decision between injectable semaglutide with B12 (typically cyanocobalamin) and the semaglutide methylcobalamin ODT involves trade-offs across multiple dimensions.
Efficacy comparison
Injectable semaglutide has the strongest evidence base. The STEP trials, which led to FDA approval of Wegovy, all used subcutaneous injection. Patients lost an average of 12 to 15% of body weight over 68 weeks. The bioavailability of injected semaglutide is near 89%, meaning almost all the administered dose reaches systemic circulation.
The ODT formulation does not have equivalent clinical trial data. Oral semaglutide bioavailability is significantly lower, estimated at 0.4 to 1% for standard oral formulations. Sublingual and buccal routes may improve this, but published data on compounded ODT bioavailability in humans is limited. A real-world comparison study found that oral semaglutide users lost an average of 3.64 kg in 6 months versus 5.26 kg for injectable users, though the difference was not statistically significant.
Practically, this means the ODT requires higher doses to achieve comparable effects, and individual responses will vary more than with injections.
Side effect profiles
The most notable difference: injection site reactions do not apply to the ODT. For people who develop persistent redness, itching, or nodules at injection sites, this is a meaningful advantage.
However, the ODT may produce more gastrointestinal side effects. One study found that adverse events occurred more frequently with oral semaglutide than with injectable (16.7% vs 4.9%). The oral route exposes the GI tract to higher local concentrations of semaglutide, which can trigger more nausea, vomiting, and diarrhea.
The methylcobalamin component may partially offset some GI side effects. B12 supplementation has been reported to help with fatigue and nausea that semaglutide users commonly experience, though the evidence for this is largely anecdotal rather than clinical.
Convenience and compliance
This is where the ODT wins decisively for some patients.
Injectable semaglutide requires weekly subcutaneous injection, proper reconstitution technique (for compounded vials), refrigerated storage, needle disposal, and comfort with self-injection. Some people never get comfortable with needles. Period.
The ODT is a tablet that dissolves on your tongue. No needles. No reconstitution. No refrigeration (room temperature storage). No sharps disposal. For needle-averse patients, the difference between continuing therapy and dropping out can hinge entirely on the delivery method.
The daily dosing schedule of the ODT versus weekly injection is a trade-off. Some prefer the once-weekly simplicity. Others find a daily oral habit easier to maintain consistently.
Factor | Injectable semaglutide + B12 | Semaglutide methylcobalamin ODT |
|---|---|---|
Bioavailability | ~89% (high) | Variable (lower) |
Evidence base | Strong (clinical trials) | Limited (emerging) |
B12 form | Usually cyanocobalamin | Methylcobalamin (bioactive) |
Administration | Weekly subcutaneous injection | Daily oral dissolving tablet |
Needle required | Yes | No |
Storage | Refrigerated | Room temperature |
GI side effects | Lower incidence | Higher incidence |
Injection site reactions | Possible | Not applicable |
Cost | Varies by pharmacy | Varies by pharmacy |
The science behind combining B12 with semaglutide
The rationale for adding methylcobalamin to semaglutide sounds logical. But does the science actually support it? The honest answer is: partially. The theoretical basis is sound. The clinical proof is thin.
What we know
We know semaglutide reduces B12 absorption. Multiple studies confirm this. We know methylcobalamin is the bioactive form of B12 and does not require hepatic conversion. We know buccal absorption of methylcobalamin bypasses the gut, which is relevant when the gut is compromised by GLP-1 therapy. We know B12 deficiency causes symptoms that overlap with and compound GLP-1 side effects, potentially making patients feel worse than they should on therapy alone.
What we do not know
We do not have randomized controlled trials comparing semaglutide alone versus semaglutide with methylcobalamin. There are no head-to-head studies showing that adding methylcobalamin to semaglutide improves weight loss outcomes, reduces side effects, or meaningfully prevents B12 depletion better than taking a separate B12 supplement.
We do not know the optimal dose of methylcobalamin when combined with semaglutide. The 0.1 mg (100 mcg) dose used in the Empower ODT formulation was not determined through dose-finding studies specific to semaglutide users. It was selected based on general B12 supplementation guidelines and pharmacological convention.
We do not know whether the buccal absorption of methylcobalamin from an ODT is sufficient to fully compensate for the B12 absorption impairment caused by semaglutide. The amount absorbed buccally versus the amount swallowed and subjected to the impaired GI pathway has not been quantified in published research.
The honest assessment
Is combining semaglutide with methylcobalamin harmful? Almost certainly not. Methylcobalamin has an excellent safety profile with no established upper limit for toxicity. Water-soluble B vitamins are excreted when consumed in excess.
Is it beneficial? Probably, especially for higher-risk groups. B12 supplementation during GLP-1 therapy makes physiological sense. Using the bioactive form adds a marginal advantage. Delivering it via buccal route when the gut is compromised is logical.
Is it necessary for everyone on semaglutide? No. Many people maintain adequate B12 levels throughout their semaglutide treatment, especially those who start with robust B12 stores, eat a varied diet including animal products, and are not taking other B12-depleting medications.
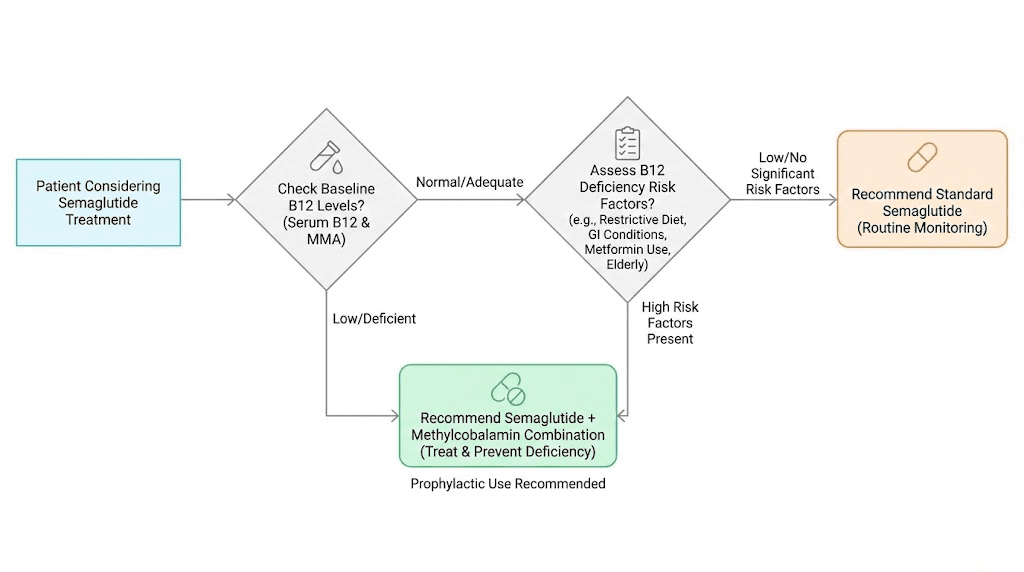
The most evidence-based approach: get your B12 levels tested before starting semaglutide. Test again at 3 months and 6 months. If levels are declining, supplementation is warranted. If levels are stable, additional B12 beyond dietary intake is unnecessary.
Side effects of semaglutide methylcobalamin
The side effect profile of semaglutide methylcobalamin is dominated by the semaglutide component. Methylcobalamin at the doses used (100 mcg) is extremely well tolerated and rarely causes adverse effects on its own.
Gastrointestinal effects
Nausea is the most commonly reported side effect and typically peaks during dose escalation. Most patients find it improves after 4 to 8 weeks at a stable dose. The nausea from oral semaglutide tends to be more frequent than from the injectable form because of higher local concentrations in the GI tract.
Vomiting, diarrhea, and constipation are also reported, usually in the first few weeks of treatment or after dose increases. These follow the same pattern seen with all GLP-1 receptor agonist medications.
Decreased appetite is technically a therapeutic effect, not a side effect, but it can feel distressing for patients who experience complete food aversion rather than gentle appetite reduction. If eating becomes impossible rather than simply reduced, the dose is likely too high.
Fatigue and energy changes
Some patients report initial fatigue when starting semaglutide, which can be confusing given that one of the supposed benefits of adding methylcobalamin is improved energy. The semaglutide-induced fatigue stems from caloric deficit, blood sugar fluctuations, and metabolic recalibration, not from B12 deficiency. The methylcobalamin may help if B12 depletion is contributing to the fatigue, but it will not eliminate the other mechanisms.
Over time, many patients report that their energy stabilizes and improves, particularly around weeks 6 to 10 when the body has adjusted to the new caloric intake and metabolic state. Patients with confirmed B12 deficiency who start the methylcobalamin formulation often notice the most dramatic energy improvements.
Cardiovascular considerations
Semaglutide can cause minor heart rate increases of 2 to 4 beats per minute on average. This is a class effect of GLP-1 receptor agonists. The methylcobalamin does not influence this effect. Patients with pre-existing cardiac conditions should be monitored regardless of which semaglutide formulation they use.
Contraindications
Semaglutide methylcobalamin ODT should not be used by individuals with:
Personal or family history of medullary thyroid carcinoma (MTC)
Multiple endocrine neoplasia syndrome type 2 (MEN 2)
Known hypersensitivity to semaglutide or any formulation component
History of severe pancreatitis
Severe gastrointestinal disease that may be worsened by delayed gastric emptying
Severe renal impairment
Pregnancy or planned pregnancy
The methylcobalamin component has no meaningful contraindications at the doses used. There is no known toxicity level for methylcobalamin supplementation.
Semaglutide methylcobalamin and weight loss: what to expect
Let us set realistic expectations based on available data.
Weight loss timeline
Clinical trials of semaglutide show a predictable weight loss trajectory. The pattern applies to the methylcobalamin formulation as well, though exact numbers may vary with the ODT delivery method.
Weeks 1 through 4: Most patients notice reduced appetite. Weight loss is modest, typically 1 to 3 pounds, as the dose is at the starting level. The body is adjusting to GLP-1 receptor activation. Some patients experience no visible weight change during this phase.
Weeks 5 through 12: As the dose increases, appetite suppression becomes more pronounced. Weight loss accelerates. Most patients lose 3 to 8% of their starting body weight by week 12. This is the phase where the medication is doing most of the heavy lifting.
Weeks 13 through 28: Weight loss continues but the rate gradually slows as the body adjusts. Total weight loss of 8 to 12% is typical by this point for injectable semaglutide. The ODT formulation may produce slightly less dramatic results due to lower bioavailability, though individual responses vary considerably.
Weeks 29 through 68: Weight loss plateaus for many patients. The clinical trials show average total weight loss of 12 to 15% at 68 weeks. Maintaining this loss requires continued medication. Stopping semaglutide typically leads to weight regain.
Does methylcobalamin improve weight loss results
There is no evidence that adding methylcobalamin directly enhances the weight loss effects of semaglutide. B12 does not have weight loss properties. The marketing claim that B12 "boosts metabolism" is technically true in the context of correcting a deficiency, since B12 is essential for energy metabolism, but only applies if you are actually deficient. If your B12 levels are normal, extra B12 does not speed up metabolism or fat burning.
Where methylcobalamin may indirectly support weight loss outcomes is through improved energy and well-being. Patients who feel less fatigued are more likely to exercise, maintain their daily routines, and stay compliant with their treatment protocol. A patient who feels terrible is a patient who eventually stops their medication. If methylcobalamin prevents the cumulative drag of B12 depletion on energy levels, it supports adherence, which supports results.
Other common additives in compounded semaglutide formulations
Methylcobalamin is not the only ingredient pharmacies add to semaglutide. Understanding the broader landscape of semaglutide additives helps put the methylcobalamin combination in context.
Glycine
Glycine is an amino acid added to some compounded semaglutide formulations. It enhances absorption and stability of the semaglutide molecule. Some formulations, like those from Strive Pharmacy, combine semaglutide, glycine, and B12 together. Glycine also plays a role in preserving lean muscle mass during weight loss, which is a legitimate concern during rapid caloric deficit.
Niacinamide (vitamin B3)
Some formulations include niacinamide to support cellular energy production and metabolism. Niacinamide helps with NAD+ synthesis, which is important for energy pathways. It also has anti-inflammatory properties and may support skin health during rapid weight loss.
L-Carnitine
L-Carnitine is occasionally added to promote fat utilization for energy. It transports fatty acids into mitochondria for oxidation. The evidence for supplemental L-Carnitine improving weight loss in non-deficient individuals is weak, but it has a reasonable safety profile.
NAD+
Some premium compounded formulations include NAD+ precursors to support cellular energy production. NAD+ is involved in hundreds of metabolic reactions and declines with age. The addition to semaglutide formulations is more about general wellness optimization than direct weight loss support.
Each additive comes with its own evidence profile, cost implications, and potential interactions. SeekPeptides provides detailed comparison tools for members evaluating different compounded formulations.
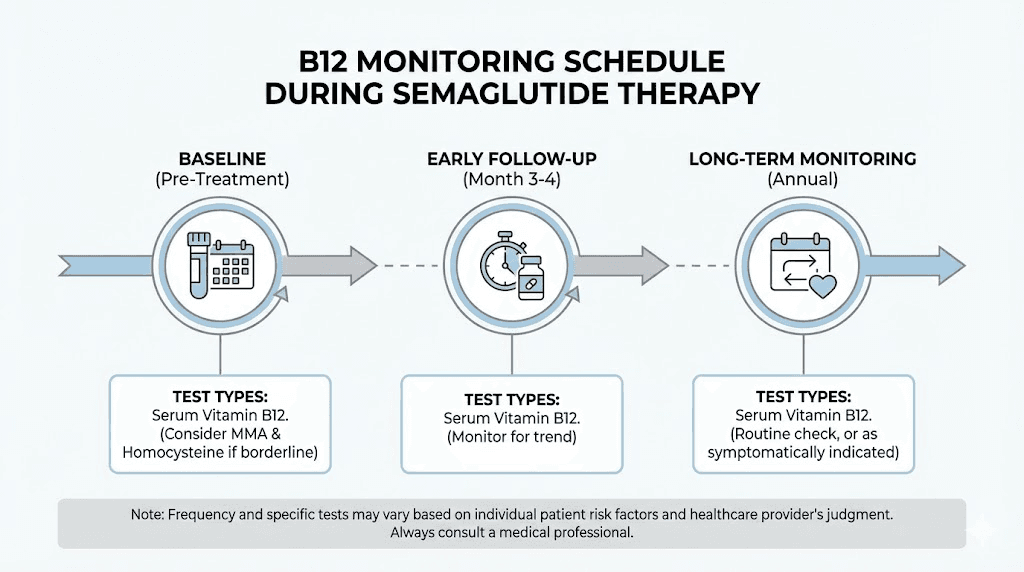
How to monitor B12 levels during semaglutide therapy
Whether you choose the semaglutide methylcobalamin combination or take semaglutide separately with B12 supplementation, monitoring is the most important piece of the puzzle.
Baseline testing before starting semaglutide
Get a complete B12 panel before your first dose. This includes:
Serum B12: The standard test. Normal range is 200 to 900 pg/mL. However, serum B12 is a crude measure and can be normal even in the presence of functional deficiency. If your level is below 400 pg/mL before starting semaglutide, supplementation is strongly recommended.
Methylmalonic acid (MMA): A more sensitive marker of B12 function. Elevated MMA indicates functional B12 deficiency even when serum B12 looks normal. This test catches early deficiency before symptoms develop.
Homocysteine: Elevated levels indicate both B12 and folate insufficiency. It is less specific than MMA but provides useful context for overall methylation status, which is directly relevant to methylcobalamin function.
Monitoring schedule
Healthcare providers who specialize in GLP-1 therapy typically recommend this monitoring cadence:
Baseline: Full B12 panel before starting semaglutide
3 months: Repeat serum B12 and MMA
6 months: Full panel reassessment
Annually: Once stable, annual monitoring is usually sufficient
For high-risk patients (metformin users, vegans, those over 60, post-bariatric surgery), the 3-month check is particularly important. A significant decline at 3 months predicts continued depletion if left unaddressed.
What to do if B12 drops despite supplementation
If you are taking semaglutide methylcobalamin and your B12 levels still decline, several options exist:
Switch to injectable B12 supplementation. B12 injections (typically cyanocobalamin or hydroxocobalamin shots) bypass the GI tract entirely and are the gold standard for correcting B12 deficiency. Monthly injections of 1000 mcg are a common maintenance protocol.
Add a high-dose sublingual methylcobalamin supplement. Sublingual B12 at doses of 1000 to 5000 mcg daily provides buccal absorption that does not depend on the gut. This can supplement the 100 mcg in the ODT formulation.
Evaluate other contributing factors. Check for concurrent metformin use. Assess dietary B12 intake. Test for pernicious anemia (anti-intrinsic factor antibodies). Sometimes the solution is not more B12 but addressing what else is depleting it.
Common questions about semaglutide methylcobalamin
Real questions from patients and researchers, answered directly.
Can I just take a separate B12 supplement instead of the compounded version?
Yes. There is no pharmacological reason why the methylcobalamin must be combined with semaglutide in a single tablet. Taking a separate sublingual methylcobalamin supplement of 1000 mcg or more daily will provide significantly more B12 than the 100 mcg in the compounded ODT. The advantage of the combination product is convenience, not superior B12 delivery.
Is the semaglutide in ODT form as effective as injectable?
The effectiveness per milligram is lower because oral bioavailability is significantly less than injectable. However, the higher oral doses compensate for this difference. Real-world data suggests comparable weight loss outcomes between oral and injectable semaglutide when doses are optimized, though individual results vary more with oral formulations. Refer to our oral versus injectable comparison for a broader perspective.
Does methylcobalamin actually reduce semaglutide nausea?
There is anecdotal but not clinical evidence for this. B12 supplementation has been reported by some clinicians and patients to help with GLP-1 nausea, but no controlled studies have confirmed this effect. The mechanism is plausible, as B12 plays a role in neurological function that could influence nausea pathways, but it remains unproven. If nausea is your primary concern, timing your dose correctly and following a slow titration schedule are better supported strategies.
What happens to B12 levels after stopping semaglutide?
When semaglutide is discontinued, gastric acid production, gastric emptying speed, and appetite all return toward baseline over several weeks. B12 absorption should recover accordingly. However, if a deficiency developed during treatment, it will not self-correct just because the medication stopped. The body does not retroactively make up for months of poor absorption. Active repletion through supplementation or injections is needed.
Is compounded semaglutide methylcobalamin FDA approved?
No. Compounded medications are not FDA approved. The FDA does not review compounded medications for safety or efficacy. This applies to all compounded semaglutide products, not just the methylcobalamin combination. The FDA-approved semaglutide products are Ozempic (injection for diabetes), Wegovy (injection for weight management), and Rybelsus (oral tablet for diabetes). None of these contain methylcobalamin.
Can I switch from semaglutide cyanocobalamin injections to the methylcobalamin ODT?
Yes, with prescriber guidance. The switch involves two changes simultaneously: the B12 form (cyanocobalamin to methylcobalamin) and the delivery method (injection to oral). There is no established dose conversion between injectable semaglutide with cyanocobalamin and oral semaglutide with methylcobalamin. Most clinicians recommend starting the ODT at a moderate dose and titrating based on response rather than attempting a direct dose translation. Refer to our switching between GLP-1 formulations guide for additional context.
How long should I take semaglutide methylcobalamin?
The duration of semaglutide therapy is generally long-term. Current evidence shows that stopping semaglutide leads to weight regain in most patients. The methylcobalamin component does not change this calculus. As long as you are taking semaglutide, B12 supplementation is advisable. If you stop semaglutide, the need for B12 supplementation should be reassessed based on your current levels and dietary intake.
Comparing semaglutide methylcobalamin to tirzepatide with B12
Some patients considering semaglutide methylcobalamin may also be evaluating tirzepatide with B12. While this guide focuses on the semaglutide formulation, a brief comparison is useful.
Tirzepatide is a dual GIP/GLP-1 receptor agonist that generally produces greater weight loss than semaglutide in head-to-head trials. In the SURMOUNT-1 trial, tirzepatide at the highest dose (15 mg) produced average weight loss of 22.5% versus approximately 15% for semaglutide in comparable trials.
Tirzepatide also affects B12 absorption through similar mechanisms: reduced gastric acid and delayed emptying. The same rationale for B12 supplementation applies. Compounded tirzepatide formulations commonly include B12 (both cyanocobalamin and methylcobalamin forms), glycine, and sometimes niacinamide.
The choice between semaglutide and tirzepatide depends on factors beyond B12 formulation: insurance coverage, weight loss goals, side effect tolerance, and whether oral or injectable delivery is preferred. The side effect profile differs between the two medications, and individual response varies significantly.
For a detailed head-to-head analysis including dosing charts, efficacy data, and practical recommendations, see our dedicated comparison guides.
Practical tips for optimizing semaglutide methylcobalamin results
Beyond dosing and monitoring, several practical considerations affect outcomes.
Diet during semaglutide methylcobalamin therapy
Eating a nutrient-dense diet becomes even more important when your appetite is suppressed. With fewer total calories coming in, every meal needs to count nutritionally. Focus on foods rich in B12 even though you have the methylcobalamin supplement: eggs, fish, poultry, dairy, and fortified foods provide additional B12 through dietary pathways that, while impaired, are not completely shut down.
Protein intake matters enormously during GLP-1 therapy. Aim for at least 0.7 to 1 gram per pound of lean body mass daily. Insufficient protein during rapid weight loss leads to muscle wasting, which worsens fatigue, reduces metabolic rate, and produces the "skinny fat" outcome nobody wants. The semaglutide diet plan should prioritize protein at every meal.
Stay hydrated. Semaglutide slows gastric emptying and can reduce fluid intake along with food. Dehydration mimics and amplifies fatigue, making it harder to distinguish B12-related tiredness from simple fluid deficit.
Exercise considerations
Resistance training is non-negotiable during any GLP-1 therapy. The caloric deficit created by appetite suppression does not selectively burn fat. Without the stimulus of resistance exercise, the body catabolizes muscle along with fat. This is true for all semaglutide formulations, methylcobalamin or not.
Timing exercise around the ODT dose requires minimal adjustment. Take your tablet first thing in the morning. Wait 30 minutes. Then exercise, eat, or proceed with your day. The medication will be absorbed before any vigorous activity.
Supplement stack to complement semaglutide methylcobalamin
Beyond the methylcobalamin already in the formulation, several supplements warrant consideration during GLP-1 therapy:
Electrolytes: Reduced food and fluid intake can deplete sodium, potassium, and magnesium. An electrolyte supplement prevents the cramping, dizziness, and fatigue that electrolyte imbalance causes.
Vitamin D: Weight loss mobilizes vitamin D stored in fat tissue, which can paradoxically cause fluctuations in serum levels. Testing and supplementing as needed supports bone health and immune function.
Iron: Reduced stomach acid from semaglutide can impair iron absorption similarly to B12. Women of reproductive age are at particular risk. Monitor ferritin levels and supplement if needed.
Zinc: The Japanese semaglutide study that found B12 decline also documented significant zinc decreases. Zinc is essential for immune function, wound healing, and taste perception, the last of which is already affected by the altered appetite signals from GLP-1 therapy.
Omega-3 fatty acids: Support cardiovascular health and reduce inflammation. Particularly relevant for patients using semaglutide for type 2 diabetes management alongside weight loss.
The regulatory landscape for compounded semaglutide methylcobalamin
Understanding the regulatory context is essential for making informed decisions about any compounded medication.
FDA position on compounded semaglutide
The FDA has been increasingly vocal about concerns with compounded semaglutide products. In their view, compounded versions are not the same as the FDA-approved products (Ozempic, Wegovy, Rybelsus) and have not undergone the same rigorous testing for safety, efficacy, and manufacturing quality.
Compounding pharmacies operate under either 503A (patient-specific prescriptions) or 503B (outsourcing facilities) federal exemptions. The semaglutide methylcobalamin ODT from Empower Pharmacy falls under 503A, meaning each order requires a patient-specific prescription and is compounded based on the individual order.
Eli Lilly, manufacturer of the competing GLP-1 medication tirzepatide (Mounjaro, Zepbound), has filed lawsuits against some compounding pharmacies. While these primarily target tirzepatide compounding, the legal landscape affects the entire compounded GLP-1 market, including semaglutide products.
Quality considerations
Not all compounding pharmacies are equal. When choosing a semaglutide methylcobalamin product, verify that the pharmacy holds proper state licenses, has current USP 795 and USP 797 compliance (for sterile compounding), conducts potency and sterility testing on their products, and carries appropriate accreditations.
The Empower Pharmacy product specifically includes methylcobalamin because, according to their documentation, the physician may determine that it provides a significant clinical difference from commercially available products. This language matters because 503A compounding requires that the compound contain a meaningful clinical difference from an available FDA-approved product to justify its existence.
Frequently asked questions
What is the difference between semaglutide methylcobalamin and semaglutide cyanocobalamin?
The core difference is the form of vitamin B12. Methylcobalamin is the bioactive form your body uses directly. Cyanocobalamin is synthetic and requires liver conversion. Semaglutide methylcobalamin formulations are typically oral (ODT tablets). Semaglutide cyanocobalamin formulations are typically injectable. Both provide B12 to counteract depletion from GLP-1 therapy.
How much B12 is in semaglutide methylcobalamin ODT?
Each tablet contains 0.1 mg (100 mcg) of methylcobalamin across all three strength levels. This is approximately 4,167% of the daily recommended intake, which is intentionally high to compensate for reduced absorption during GLP-1 therapy.
Do I still need to take a separate B12 supplement with semaglutide methylcobalamin?
For most patients, the 100 mcg of methylcobalamin in each tablet is adequate. However, patients in high-risk groups (over 60, concurrent metformin use, vegan diet, post-bariatric surgery) may benefit from additional supplementation. Your B12 lab results at the 3-month check should guide this decision.
Can semaglutide methylcobalamin ODT replace my semaglutide injection?
It can serve as an alternative delivery method, but it is not a direct replacement. The bioavailability differs, the dosing schedule changes from weekly to daily, and the evidence base is less robust. Discuss the switch with your prescriber and expect a titration period while finding the effective oral dose. Use our semaglutide dosage calculator as a starting reference.
Is semaglutide methylcobalamin safe during pregnancy?
No. Semaglutide is contraindicated during pregnancy regardless of the formulation. It should be discontinued at least 2 months before planned conception due to its long half-life. This applies to the methylcobalamin ODT just as it applies to injectable formulations.
What does semaglutide methylcobalamin cost?
Costs vary significantly by pharmacy, strength, and quantity. Compounded medications are typically not covered by insurance. Contact the prescribing pharmacy directly for current pricing. Costs for all compounded GLP-1 products have been fluctuating as the regulatory and legal landscape evolves.
Can I take semaglutide methylcobalamin with other medications?
Semaglutide slows gastric emptying, which can affect the absorption of other oral medications. Take other medications either at least 1 hour before or several hours after the semaglutide methylcobalamin ODT. Specific interactions with phentermine, oral contraceptives, and thyroid medications are documented and should be discussed with your prescriber.
How long does semaglutide methylcobalamin ODT take to work?
Initial appetite suppression typically begins within the first week of treatment. Meaningful weight loss usually becomes apparent by weeks 4 to 8. The methylcobalamin component begins working immediately to support B12 levels, though the impact on energy and well-being may take 2 to 4 weeks to become noticeable, depending on your baseline B12 status.
External resources
For researchers serious about optimizing their GLP-1 protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members get access to detailed formulation comparisons, B12 monitoring templates, and personalized protocol recommendations that account for individual risk factors most resources simply ignore.
In case I do not see you, good afternoon, good evening, and good night. May your B12 levels stay robust, your GLP-1 therapy stay effective, and your protocols stay evidence-based.