Feb 21, 2026

Ozempic is the name everyone knows. It dominates conversations, headlines, and pharmacy shelves. Retatrutide is the name clinical researchers cannot stop talking about. One is available right now at your local pharmacy. The other produced nearly double the weight loss in clinical trials but remains years away from FDA approval.
That gap between what is available and what is coming matters more than most people realize. Ozempic, powered by the single-receptor GLP-1 agonist semaglutide, helped reshape how the medical world thinks about obesity treatment. But retatrutide, a triple-receptor agonist targeting GLP-1, GIP, and glucagon simultaneously, delivered weight loss numbers in Phase 3 trials that made researchers rethink what pharmaceutical intervention could achieve. We are talking about 28.7% average body weight reduction at the highest dose. For a 250-pound person, that translates to roughly 71 pounds lost.
So which one actually deserves your attention? The answer depends on where you stand right now, what your goals are, and how much patience you have. This guide breaks down every meaningful difference between Ozempic and retatrutide, from molecular mechanisms to clinical trial data to real-world side effects, so you can make an informed decision about GLP-1 based therapies and what the future holds for weight management.
How Ozempic and retatrutide work at the molecular level
Understanding why these two medications produce such different results requires looking beneath the surface. Both fall under the GLP-1 umbrella. Both are injectable peptides administered once weekly. But the similarity ends there, because their mechanisms of action diverge in fundamental ways that directly impact fat loss outcomes, metabolic health, and long-term body composition.
The single-receptor approach: how Ozempic targets GLP-1
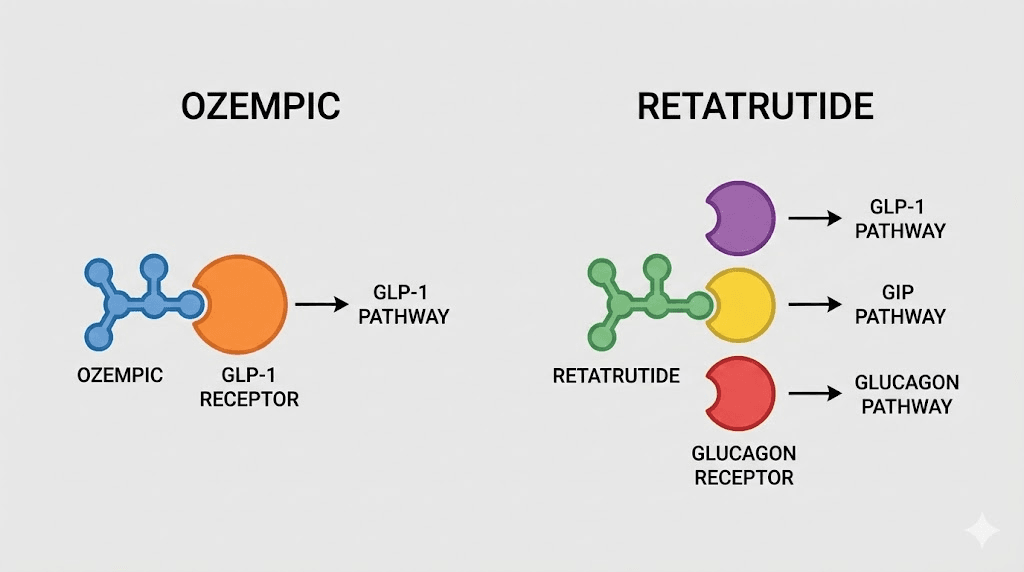
Ozempic contains semaglutide, a synthetic analog of the human glucagon-like peptide-1 hormone. When you inject semaglutide, it binds exclusively to GLP-1 receptors throughout your body. These receptors sit in the pancreas, the brain, the gut, and several other tissues. Activation triggers a cascade of metabolic effects.
The pancreas responds by increasing insulin secretion when blood sugar rises. That matters for glucose control.
But the real weight loss magic happens in the brain. Semaglutide crosses the blood-brain barrier and acts on appetite centers in the hypothalamus and brainstem. It reduces hunger signals, increases satiety after meals, and changes food reward pathways. People on Ozempic frequently report that food simply becomes less interesting. The constant background noise of cravings goes quiet. For many, that experience alone transforms their relationship with eating. You can read more about how long semaglutide takes to suppress appetite in our detailed timeline guide.
Semaglutide also slows gastric emptying, meaning food stays in your stomach longer. You feel full sooner during meals and stay satisfied for hours afterward. This combination of reduced appetite, enhanced satiety, and delayed digestion creates a powerful caloric deficit without the white-knuckle willpower that traditional dieting demands. For people tracking their semaglutide dosage, these effects typically strengthen as the dose increases through the titration schedule.
One receptor. Multiple downstream effects. That is the elegance of the semaglutide approach.
The triple-receptor approach: how retatrutide targets GLP-1, GIP, and glucagon
Retatrutide takes a fundamentally different approach. Instead of targeting one receptor, it activates three simultaneously: GLP-1, glucose-dependent insulinotropic polypeptide (GIP), and the glucagon receptor. This triple-agonist design was not accidental. Eli Lilly engineered retatrutide with specific potency ratios at each receptor to create a synergistic metabolic effect that no single-receptor or even dual-receptor drug can match.
The GLP-1 component works similarly to Ozempic. Appetite suppression, improved insulin sensitivity, slowed gastric emptying. You get those baseline benefits. The GIP component adds another layer. GIP receptors exist in fat tissue, bone, and the brain. Activation enhances insulin release, supports fat metabolism, and may improve how efficiently your body processes and stores nutrients. Research suggests GIP agonism also amplifies the weight loss effects of GLP-1, which explains why dual-agonists like tirzepatide outperform single-agonists like semaglutide. For a comparison of those two, see our semaglutide vs tirzepatide breakdown.
But the glucagon receptor is where retatrutide truly separates itself.
Why the glucagon receptor changes everything
Glucagon is traditionally viewed as a blood sugar raising hormone, the opposite of insulin. When blood sugar drops, glucagon tells your liver to release stored glucose. That is its textbook function. But glucagon does far more than manage blood sugar.
Glucagon activation increases energy expenditure. Your body burns more calories at rest. It promotes fat oxidation, meaning your body preferentially burns stored fat for fuel rather than relying solely on reduced caloric intake. It also appears to help preserve lean muscle mass during weight loss, a critical advantage that most weight loss medications lack.
Think about what this means practically. Ozempic works primarily by making you eat less. Retatrutide makes you eat less AND tells your body to burn more. That dual mechanism, reduced input plus increased output, explains the dramatic differences in clinical trial results. The retatrutide dosage chart reflects how these effects scale with increasing doses through the titration protocol.
Structural analysis published in Cell Discovery reveals that retatrutide achieves its triple agonism through a single peptide chain that adopts different conformations when binding each receptor type. It is most potent at the GIP receptor (EC50 of 0.0643 nM), moderately potent at GLP-1 (EC50 of 0.775 nM), and least potent at the glucagon receptor (EC50 of 5.79 nM). This potency hierarchy was deliberately designed. Too much glucagon activation could cause excessive blood sugar elevation. Too little would negate the metabolic benefits. The ratio Lilly settled on appears to hit a sweet spot that maximizes fat loss while maintaining metabolic safety.
Weight loss results compared: what the clinical data actually shows
Numbers do not lie. And the numbers from clinical trials paint a clear picture of how these two medications compare when it comes to the outcome most people care about: how much weight you actually lose. Both Ozempic and retatrutide have been tested in large, well-designed, placebo-controlled trials. Both have produced statistically significant results. But the magnitude of those results differs substantially. Understanding how fast semaglutide works versus the trajectory of retatrutide requires examining the data from each trial program separately before comparing them directly.
Ozempic weight loss data from the STEP trials
The STEP (Semaglutide Treatment Effect in People with Obesity) clinical trial program is one of the most comprehensive weight loss drug studies ever conducted. Multiple trials, thousands of participants, and consistent results across different populations.
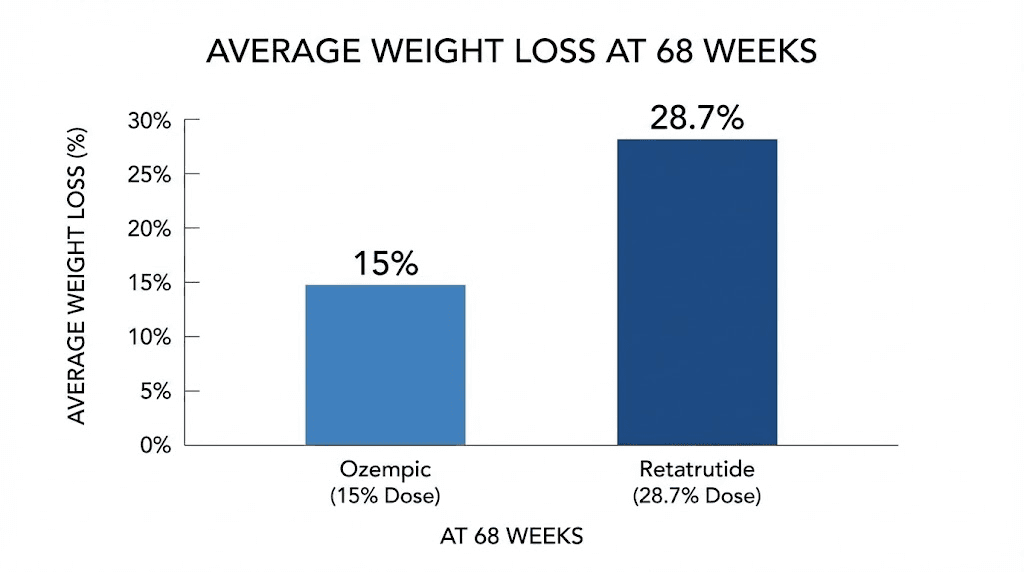
STEP 1 enrolled 1,961 adults with obesity or overweight with at least one weight-related comorbidity. After 68 weeks of treatment with semaglutide 2.4 mg (the Wegovy dose, identical to the molecule in Ozempic), participants lost an average of 14.9% of their body weight. The placebo group lost 2.4%. That is a treatment difference of roughly 12.5 percentage points. Approximately 86% of semaglutide-treated participants achieved at least 5% weight loss, and about 69% to 79% achieved 10% or more across the STEP trials.
STEP 3 combined semaglutide with intensive behavioral therapy and an initial low-calorie diet phase. Results improved to 16.0% average weight loss at 68 weeks. STEP 8 compared semaglutide directly against liraglutide and showed clear superiority for semaglutide.
Across all STEP trials, the consistent finding was 14.9% to 17.4% average body weight reduction at 68 weeks with the 2.4 mg dose. For a 220-pound person, that translates to roughly 33 to 38 pounds lost. Meaningful. Significant. Life-changing for many people who had struggled with obesity for years. If you are four weeks in and not seeing results yet, our guide on 4 weeks on semaglutide with no weight loss explains what could be happening and when to expect changes.
But is 15% the ceiling? For semaglutide, the answer appears to be yes, at least at currently approved doses. Recent data from the STEP UP trial exploring a higher 7.2 mg dose suggests additional weight loss is possible with dose escalation, but the standard 2.4 mg remains the benchmark.
Retatrutide weight loss from Phase 2 and TRIUMPH-4
Retatrutide entered the clinical picture with a Phase 2 trial published in the New England Journal of Medicine. The results were, frankly, stunning. At the highest dose of 12 mg, participants lost an average of 24.2% of their body weight at 48 weeks. The 8 mg group lost 22.8%. Even the 4 mg group achieved 17.1%, roughly matching what semaglutide achieves at its highest studied doses.
Then came TRIUMPH-4. This Phase 3 trial, completed in December of the previous year, enrolled participants with obesity and knee osteoarthritis. At 68 weeks, the 12 mg retatrutide group lost an average of 28.7% of their body weight. That is approximately 71 pounds for the average participant. The 9 mg group lost 26.4%. Placebo managed just 2.1%.
Read those numbers again. Nearly 29% average body weight reduction. For context, bariatric surgery typically produces 25% to 35% weight loss. Retatrutide is approaching surgical results with a weekly injection. The retatrutide dose guide covers the full protocol for how these results were achieved, including the gradual titration that minimizes side effects while building toward maximum efficacy.
What makes these numbers even more remarkable is the consistency. Not just a few exceptional responders pulling up the average, but broad, reliable weight loss across the study population. The percentage of participants achieving clinically meaningful thresholds (5%, 10%, 15%, 20%+ weight loss) exceeded anything previously seen with GLP-1 based therapies.
Head to head weight loss comparison
Metric | Ozempic (semaglutide 2.4 mg) | Retatrutide (12 mg) |
|---|---|---|
Average weight loss (68 weeks) | 14.9% to 17.4% | 28.7% |
Pounds lost (220 lb person) | 33 to 38 lbs | 63 lbs |
Pounds lost (250 lb person) | 37 to 44 lbs | 72 lbs |
Participants losing 10%+ | 69% to 79% | Not yet published (Phase 2 data: approximately 93%) |
Participants losing 15%+ | 50% to 56% | Not yet published (Phase 2 data: approximately 83%) |
Trial program | STEP trials (Phase 3, completed) | Phase 2 (NEJM) + TRIUMPH-4 (Phase 3) |
Trial duration | 68 weeks | 48 weeks (Phase 2) / 68 weeks (TRIUMPH-4) |
The difference is not subtle. Retatrutide nearly doubles the weight loss seen with semaglutide at the same treatment duration. Whether these results hold across all Phase 3 TRIUMPH trials remains to be seen, but the consistency between Phase 2 and TRIUMPH-4 data builds confidence. For researchers comparing options across the retatrutide vs semaglutide landscape, the weight loss advantage is the most striking differentiator.
Side effects and safety: what to expect from each
Weight loss numbers mean nothing if the side effects make a medication intolerable. Both Ozempic and retatrutide share a common foundation of gastrointestinal side effects, which is expected given that they both activate GLP-1 receptors. But the differences in their side effect profiles are worth understanding before making decisions. People managing existing side effects should review strategies like semaglutide constipation treatment and dietary adjustments from our foods to avoid on semaglutide guide.
Common side effects of Ozempic
Semaglutide has been on the market long enough that its side effect profile is well characterized. Years of real-world data supplement the clinical trial findings. The most commonly reported issues are gastrointestinal in nature.
Nausea affects roughly 20% to 44% of patients depending on the study and dose. It is typically most pronounced during dose escalation and tends to diminish as the body adapts over 4 to 8 weeks. Diarrhea and constipation each affect approximately 15% to 30% of users. Vomiting occurs in about 5% to 15%. Decreased appetite is technically a side effect but also the primary mechanism through which weight loss occurs, so most people consider it a feature rather than a bug.
Less common but noteworthy side effects include fatigue, dizziness, headaches, and abdominal pain. Some people report hair thinning, which appears related to rapid weight loss rather than the drug itself. Rare but serious concerns include pancreatitis, gallbladder problems, and potential thyroid C-cell tumors (observed in rodent studies, not confirmed in humans).
The SELECT trial, which followed over 17,000 participants, showed that 16.6% of semaglutide-treated patients discontinued due to adverse events compared to 8.2% on placebo. Most discontinuations were GI-related. For practical management of these issues, our guides on the semaglutide diet plan and foods to eat while on semaglutide can help minimize discomfort.
Common side effects of retatrutide
Retatrutide shares the standard GLP-1 gastrointestinal side effect profile. Nausea, diarrhea, vomiting, and constipation are the most frequently reported issues. In the Phase 2 trial, GI side effects were dose-dependent and generally mild to moderate in severity. Most resolved without treatment discontinuation.
However, retatrutide introduced a few notable differences. The TRIUMPH-4 Phase 3 data revealed a new safety signal: dysesthesia. This manifests as skin sensitivity, tingling, or tenderness to the touch. Approximately 20.9% of participants on the highest 12 mg dose reported this symptom. While not dangerous, it was unexpected and is something researchers will continue monitoring in ongoing trials. The mechanism may relate to glucagon receptor activation in nerve tissue, though the exact pathway remains under investigation.
Heart rate increases were also noted at higher doses of retatrutide, another effect potentially linked to glucagon-mediated increases in metabolic rate. Whether this carries clinical significance requires longer-term cardiovascular outcome data that is not yet available.
The Phase 2 trial did show that gradual titration significantly reduces GI side effects. When participants were started directly at 8 mg instead of titrating from 1 mg, GI symptom rates nearly doubled. This finding reinforced the importance of the slow retatrutide dose schedule that begins at 1 mg and increases every four weeks.
Long-term safety considerations
This is where Ozempic holds a significant advantage. Semaglutide has been in clinical use since 2017 for diabetes and since 2021 for weight management. We have years of post-marketing surveillance data. The SELECT trial followed patients for over three years and demonstrated cardiovascular safety, even showing a 20% reduction in major adverse cardiovascular events.
Retatrutide, by contrast, has no post-marketing data. The longest completed trial is 68 weeks. Phase 3 trials are still underway, and the drug has never been used outside of controlled clinical settings. Its long-term effects on thyroid function, pancreatic health, cardiovascular outcomes, and bone density remain unknown. This is not necessarily alarming, but it represents a real gap in knowledge. Managing withdrawal symptoms after discontinuation is another area where Ozempic has more documented guidance, while retatrutide data on this topic does not yet exist.
Side effect | Ozempic | Retatrutide |
|---|---|---|
Nausea | 20% to 44% | Similar rates, dose-dependent |
Diarrhea | 15% to 30% | Similar rates |
Constipation | 15% to 30% | Similar rates |
Vomiting | 5% to 15% | Similar rates |
Fatigue | Reported | Reported |
Hair thinning | Related to weight loss | Related to weight loss |
Dysesthesia (skin tingling) | Not reported | Up to 20.9% at 12 mg |
Heart rate increase | Mild | More pronounced at higher doses |
Long-term safety data | Years of data (safe) | Limited to clinical trials |
Cardiovascular outcomes | 20% MACE reduction (SELECT) | Not yet studied |
Beyond the scale: metabolic benefits compared
Weight loss is the headline. But for both Ozempic and retatrutide, the metabolic benefits extend well beyond the number on the scale. These medications reshape cardiovascular risk profiles, liver health, blood sugar regulation, and kidney function in ways that impact overall health outcomes for years to come. Understanding these broader effects is essential for anyone considering how long to stay on GLP-1 therapy and what outcomes to prioritize beyond weight.
Cardiovascular protection
Ozempic has proven cardiovascular benefits supported by two major trials. SUSTAIN-6, conducted in patients with type 2 diabetes, showed a 26% reduction in major adverse cardiovascular events (heart attack, stroke, or cardiovascular death) with semaglutide compared to placebo. The hazard ratio was 0.74, meaning roughly one-quarter fewer serious cardiac events in the semaglutide group.
The SELECT trial extended this finding to people with obesity but without diabetes. Over a mean follow-up of approximately 33 months, semaglutide 2.4 mg reduced MACE by 20% (hazard ratio 0.80). This was a landmark finding. It established that semaglutide reduces cardiovascular risk independent of blood sugar control. The mechanism likely involves anti-inflammatory effects, improved endothelial function, and direct vascular benefits beyond what weight loss alone would provide.
Retatrutide does not yet have dedicated cardiovascular outcome trial data. TRIUMPH-4 did show improvements in several cardiovascular risk markers: reductions in non-HDL cholesterol, triglycerides, high-sensitivity C-reactive protein (hsCRP), and systolic blood pressure.
These are encouraging surrogate markers, but they are not the same as proven cardiovascular event reduction. Dedicated TRIUMPH cardiovascular outcome trials are planned but have not reported results.
Liver fat reduction
This is where retatrutide truly shines. A Phase 2a trial specifically examining liver fat showed extraordinary results. At 48 weeks, 93% of participants taking 12 mg retatrutide achieved normal liver fat levels (below 5%), compared to 0% in the placebo group. The mean relative reduction in liver fat was 86% at the 12 mg dose. Even the 8 mg dose produced an 89% normalization rate.
These results are unprecedented for any pharmaceutical intervention targeting metabolic dysfunction-associated steatotic liver disease (MASLD, formerly called NAFLD). No other GLP-1 medication has come close to this level of liver fat clearance. The glucagon component of retatrutide appears to drive this effect, as glucagon promotes hepatic fat oxidation, essentially telling the liver to burn its own stored fat.
Ozempic does reduce liver fat to some degree. Studies show modest improvements in hepatic steatosis with semaglutide treatment. But the magnitude does not approach what retatrutide achieves. For the estimated 100 million Americans with fatty liver disease, this difference could be clinically transformative.
Blood sugar and insulin sensitivity
Both medications improve glycemic control, but through slightly different pathways. Ozempic is FDA approved for type 2 diabetes management and has extensive data supporting its ability to reduce HbA1c by 1.0% to 1.8% depending on baseline levels and dose. It accomplishes this through enhanced insulin secretion, reduced glucagon release (paradoxically, since it only hits GLP-1 receptors), and improved beta-cell function.
Retatrutide also demonstrated impressive glycemic improvements in its Phase 2 diabetes trial published in The Lancet. At 36 weeks, 63% of adults with overweight or obesity and type 2 diabetes treated with retatrutide lost at least 15% of their body weight, compared to 40% achieving that level with 15 mg tirzepatide at 40 weeks. HbA1c reductions were substantial across all dose groups.
The addition of glucagon agonism creates a complex glycemic picture. Glucagon raises blood sugar, which might seem counterproductive for diabetes management. However, the simultaneous GLP-1 and GIP agonism appears to counterbalance this effect while still allowing glucagon to drive its metabolic benefits. Clinical data supports this, no concerning hyperglycemia patterns emerged in retatrutide trials.
Kidney function
The SELECT trial produced an important secondary finding: semaglutide reduced the 5-component kidney composite endpoint by 22% compared to placebo. This suggests renal protective effects that extend beyond blood sugar control. For the millions of people with obesity-related kidney disease, this finding adds significant value to the semaglutide profile.
Retatrutide has not been specifically studied for kidney outcomes. Any kidney benefits would need to be demonstrated in dedicated trials before claims could be made. This remains an open question in the retatrutide research program.
Dosing protocols and administration
Both Ozempic and retatrutide are once-weekly subcutaneous injections. That shared convenience factor makes either option relatively easy to incorporate into a routine. But the specifics of dosing, titration speed, and available dose strengths differ in ways that impact the practical experience of using each medication. Anyone managing GLP-1 injection sites will find the administration technique identical, as the subcutaneous injection method is the same for both.
Ozempic dosing schedule
Ozempic follows a relatively straightforward titration. You start at 0.25 mg once weekly for four weeks, primarily to assess tolerability. Then you increase to 0.5 mg for another four weeks. If more glycemic control or weight loss is needed, the dose can increase to 1 mg and then to the maximum of 2 mg. The full titration from starting dose to maximum takes approximately 16 weeks.
For weight loss specifically, Wegovy (the obesity-indication brand of semaglutide) follows a similar but slightly different schedule, titrating up to 2.4 mg. The principle is the same: slow, gradual dose escalation to minimize GI side effects. Our semaglutide dosage in units guide breaks down the exact unit measurements for each dose level, and the semaglutide 5mg/ml dosage chart provides the visual reference many people find helpful.
Pre-filled pens make dosing simple. Select the dose, inject, done. No mixing required. No calculations needed.
Retatrutide titration protocol
Retatrutide requires a longer and more gradual titration. Based on Phase 2 trial protocols, the standard approach looks like this:
Weeks 1 to 4: 1 mg once weekly
Weeks 5 to 8: 2 mg once weekly
Weeks 9 to 12: 4 mg once weekly
Weeks 13 to 16: 8 mg once weekly
Weeks 17 onward: 12 mg once weekly (if tolerated)
That is a minimum 16 to 20 week ramp-up to reach the maximum dose, compared to a similar timeline for Ozempic. The key difference is the number of dose steps: five for retatrutide versus three to four for Ozempic. Each step is critical. The Phase 2 trial demonstrated that skipping steps or accelerating the titration nearly doubles the rate of gastrointestinal side effects. Patience during titration directly translates to better tolerability. For detailed protocols, the retatrutide dosage chart provides the complete week-by-week schedule, and the retatrutide dosage calculator can help with precise measurements.
Researchers preparing their own solutions will need to understand the reconstitution process. The bacteriostatic water mixing guide for retatrutide covers the specifics of preparation, while the retatrutide injection site guide explains optimal administration locations.
Parameter | Ozempic | Retatrutide |
|---|---|---|
Route | Subcutaneous injection | Subcutaneous injection |
Frequency | Once weekly | Once weekly |
Starting dose | 0.25 mg | 1 mg |
Maximum dose | 2 mg (Ozempic) / 2.4 mg (Wegovy) | 12 mg |
Titration steps | 3 to 4 | 5 |
Time to max dose | 16 weeks | 16 to 20 weeks |
Preparation | Pre-filled pen (no mixing) | Clinical trial: pre-filled / Research: requires reconstitution |
Availability, approval status, and timeline
The single biggest practical difference between Ozempic and retatrutide right now has nothing to do with efficacy or safety. It is availability. One medication exists as a commercially available product that you can get with a prescription. The other does not yet exist outside clinical trials and research settings.
Ozempic: available now
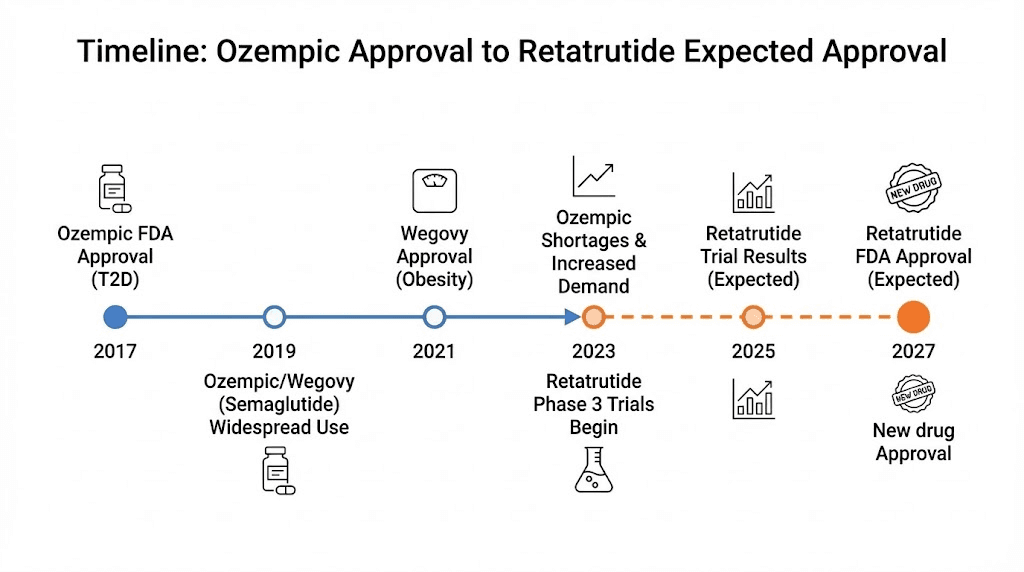
Ozempic (semaglutide 0.5 mg, 1 mg, 2 mg) has been FDA approved for type 2 diabetes since 2017. Wegovy (semaglutide 2.4 mg), the weight management formulation, received FDA approval in 2021. Both are manufactured by Novo Nordisk and available through standard pharmacy channels with a prescription.
Supply has been a challenge. Demand has outstripped manufacturing capacity at various points, leading to periodic shortages. But the situation has improved significantly, and most patients can now access semaglutide with reasonable wait times. Insurance coverage varies widely, from full coverage with certain plans to complete exclusion under others. Some people turn to compounded semaglutide as an alternative when brand-name versions are unavailable or cost-prohibitive. Others explore oral semaglutide options for needle-free administration.
The bottom line: you can start Ozempic this week if your doctor agrees it is appropriate.
Retatrutide: the timeline to your medicine cabinet
Retatrutide is in Phase 3 clinical trials under the TRIUMPH program. Eli Lilly is running multiple trials simultaneously, each examining different patient populations and indications. TRIUMPH-4 (obesity with knee osteoarthritis) reported positive results in late last year. Seven additional Phase 3 readouts are expected this year, including the pivotal TRIUMPH-1 (general obesity) and TRIUMPH-2 (type 2 diabetes) trials.
Assuming all goes well, Lilly is expected to submit a New Drug Application (NDA) to the FDA in late this year or early next year. The FDA review process typically takes 10 months, though Priority Review could shorten that to 6 months. The most realistic timeline places FDA approval in late next year, with commercial launch projected for early the year after.
That means if you are reading this right now, retatrutide is likely 18 to 24 months away from being something you can actually obtain with a prescription. It is important to note that retatrutide availability is evolving, and our regularly updated guide tracks the latest developments. For those who want to understand the expected cost of retatrutide, we maintain a comprehensive pricing analysis based on market projections.
Some researchers access retatrutide through research chemical suppliers. Our guides on buying retatrutide and obtaining retatrutide online cover what is currently available in the research market, though this route carries different risks and considerations compared to FDA-approved medications.
Cost landscape and what to expect
Ozempic currently carries a list price of approximately $900 to $1,000 per month without insurance. With insurance coverage (where available), copays can range from $25 to several hundred dollars depending on the plan. Manufacturer savings programs and coupons can reduce out-of-pocket costs for commercially insured patients. For those exploring alternatives, compounded options in the GLP-1 space can significantly reduce costs, and our peptide cost calculator helps compare pricing across different sources and formulations.
Retatrutide pricing has not been established since the drug is not yet commercially available. However, Eli Lilly has shown with tirzepatide (Mounjaro/Zepbound) a willingness to price competitively in the GLP-1 market. Industry analysts project retatrutide will likely fall in a similar range to tirzepatide, possibly with a premium given its enhanced efficacy profile. That could mean $1,000 to $1,200+ per month at list price.
The cost conversation is evolving rapidly. Generic semaglutide is expected in the coming years as patents expire, which could dramatically reduce Ozempic pricing. Meanwhile, retatrutide will enter the market as a premium branded product. For cost-sensitive individuals, this timing dynamic matters: cheaper semaglutide options may arrive around the same time retatrutide launches at full price.
Who should consider each option
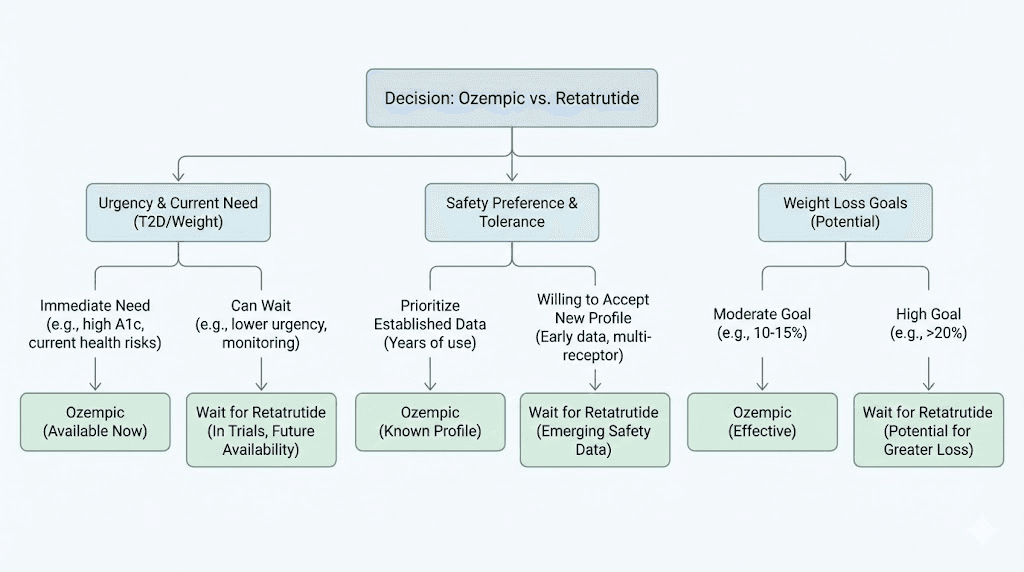
Choosing between Ozempic and retatrutide is not just about which produces bigger weight loss numbers. It is about matching the right medication to your specific situation, timeline, and health priorities. SeekPeptides members frequently navigate this exact decision, and the answer almost always depends on individual circumstances rather than blanket recommendations.
When Ozempic makes more sense
Ozempic is the stronger choice when you need treatment now. If obesity or type 2 diabetes is actively impacting your health, waiting two years for a potentially better option is not a reasonable strategy. Every month of uncontrolled weight and blood sugar carries real cardiovascular, metabolic, and psychological consequences.
It also makes sense when you value proven long-term safety. Years of post-marketing data, cardiovascular outcome trials showing actual event reduction, kidney benefits, and extensive real-world experience provide a level of certainty that retatrutide simply cannot match yet. If you are risk-averse and want the most well-characterized option available, Ozempic wins on safety data alone.
For people whose primary goal involves cardiovascular risk reduction beyond weight loss, the SELECT trial data gives Ozempic a clear evidence-based advantage. No other GLP-1 medication has the same level of cardiovascular outcome data in people without diabetes.
People who have found success with other GLP-1 medications may also prefer staying within the familiar semaglutide framework rather than waiting for an entirely new drug class. If you are considering alternatives like phentermine, our comparison guides help you evaluate every available option. And if you have questions about optimizing your semaglutide timing, small adjustments can make a measurable difference in both efficacy and tolerability.
When retatrutide might be worth the wait
If your weight loss goals are ambitious and currently available medications have fallen short, retatrutide represents the most promising next-generation option. People who have plateaued on semaglutide or tirzepatide, losing 10% to 15% but needing to lose significantly more, may find retatrutide closes that gap.
Anyone with fatty liver disease should pay close attention. The liver fat reduction data from retatrutide is unlike anything seen with other medications. If MASLD/NASH is a primary health concern alongside obesity, retatrutide could offer dual benefits that no current medication can match. Understanding the supplement strategies that complement GLP-1 therapy can help bridge the gap while waiting for newer options.
Research-oriented individuals who have access to retatrutide through peptide suppliers may choose to start earlier than the formal FDA approval timeline. This route requires careful consideration of sourcing quality, proper reconstitution protocols, and appropriate medical oversight. SeekPeptides provides comprehensive guidance on navigating these decisions safely, with evidence-based protocols and a community of experienced researchers.
Switching from Ozempic to retatrutide: what to know
For people currently taking Ozempic who are eyeing retatrutide as a future upgrade, the transition question is natural. Can you switch? What would that look like? Is there a washout period needed?
No formal switching protocols exist yet because retatrutide is not commercially available. However, the clinical precedent from switching between other GLP-1 medications provides some guidance. Generally, transitions between GLP-1 agonists can be done without a washout period, though starting the new medication at a lower dose and titrating up is standard practice regardless of what you were taking before.
The consideration unique to switching from Ozempic to retatrutide is the additional receptor activity. Your body will be adapted to GLP-1 agonism but not to GIP and glucagon agonism. Starting retatrutide at 1 mg and following the standard titration would allow your body to adjust to these new receptor activations gradually. Skipping ahead based on your semaglutide dose history would likely increase side effect risk.
The semaglutide to tirzepatide conversion experience offers useful parallels, since that transition also introduces GIP agonism. Most clinicians recommend completing the full titration schedule of the new medication regardless of prior GLP-1 experience. For those tracking their semaglutide timeline, the switch point typically makes more sense once you have reached your maximum tolerated dose and assessed whether the results meet your goals.
One practical consideration: if you are currently losing weight on Ozempic and are satisfied with your trajectory, there may be no compelling reason to switch. Retatrutide produces more weight loss on average, but individual responses vary. Some people achieve exceptional results on semaglutide. The need to switch is most relevant for people who have hit a plateau or whose weight loss has not been sufficient with current therapy. Our guide on why you are not losing weight on semaglutide can help determine whether the issue is the medication itself or modifiable factors in your protocol.
What the research community is saying
The research and clinical community has responded to retatrutide data with cautious excitement. The Phase 2 results published in the New England Journal of Medicine generated significant buzz, and the TRIUMPH-4 Phase 3 confirmation elevated that interest further.
Key perspectives from the medical community center on several themes. First, the magnitude of weight loss approaching bariatric surgery results without surgery has shifted conversations about what pharmacotherapy can achieve. Second, the liver fat data has implications beyond obesity treatment, potentially offering a first-line pharmaceutical option for MASLD/NASH. Third, the triple-receptor approach raises questions about whether more receptors always mean better outcomes, or whether there is a point of diminishing returns with added complexity.
Skeptics rightfully note the limited long-term data. A 68-week trial, no matter how impressive the results, does not replace years of real-world experience. The dysesthesia signal needs monitoring. The cardiovascular effects need dedicated outcome trials. And the question of weight regain after discontinuation, a significant issue documented with semaglutide, remains completely unanswered for retatrutide.
What most researchers agree on: retatrutide represents a genuine step forward in obesity pharmacotherapy, not just an incremental improvement. Whether it lives up to its Phase 2 and Phase 3 promise across the full TRIUMPH program will determine whether it becomes the new standard of care or remains one option among several. For context on the broader landscape, understanding how mazdutide compares to tirzepatide and how cagrilintide fits in shows just how active the GLP-1 research space has become.
The tirzepatide factor: the middle ground
No comparison of Ozempic and retatrutide is complete without acknowledging the medication that sits between them. Tirzepatide (Mounjaro/Zepbound), also made by Eli Lilly, is a dual GLP-1/GIP agonist. It targets two of the three receptors that retatrutide hits, and its clinical results fall between semaglutide and retatrutide.
Tirzepatide produces average weight loss of approximately 20% to 22.5% at its highest approved dose of 15 mg over 72 weeks. That is substantially more than Ozempic and within striking distance of retatrutide. Importantly, tirzepatide is FDA approved and commercially available right now.
For someone unsatisfied with Ozempic results, switching to tirzepatide represents a middle path: better weight loss than semaglutide, available now, with more data than retatrutide though less than semaglutide. The tirzepatide before and after results demonstrate what the dual-agonist approach can achieve in practice, and the tirzepatide dose chart shows the stepwise protocol. For a head-to-head breakdown, our semaglutide vs tirzepatide comparison page lays out every meaningful difference.
The progression from single-agonist (semaglutide) to dual-agonist (tirzepatide) to triple-agonist (retatrutide) tells a clear story about where obesity treatment is heading. Each additional receptor target appears to unlock additional efficacy. Whether the jump from dual to triple proves as significant in large-scale Phase 3 data as the jump from single to dual remains one of the most important questions in the field.
Storage, handling, and practical considerations
The practical side of using any injectable medication matters more than most people expect. Both Ozempic and retatrutide (in research settings) require refrigerated storage. Understanding how to handle these peptides properly ensures they maintain their potency throughout the treatment course.
Ozempic pens should be stored in the refrigerator at 36 to 46 degrees Fahrenheit before first use. After first use, pens can be kept at room temperature (below 86 degrees Fahrenheit) for up to 56 days. They should never be frozen. For people navigating similar storage questions with related medications, our guides on semaglutide refrigeration requirements and semaglutide shelf life in the fridge cover the specifics. Questions about expired semaglutide and 28-day expiration concerns are among the most common we encounter.
Retatrutide in research settings typically arrives as lyophilized powder requiring reconstitution with bacteriostatic water. Proper reconstitution technique is critical for maintaining peptide integrity. The peptide reconstitution calculator takes the guesswork out of this process, and detailed guides on reconstitution for semaglutide and other peptides apply the same principles. Once reconstituted, retatrutide solutions should be refrigerated and used within a timeframe that depends on the specific preparation method.
Injection technique is identical for both medications: subcutaneous injection into the abdomen, thigh, or upper arm. Rotating injection sites prevents lipodystrophy. Our GLP-1 injection guide walks through proper technique step by step.
Diet and lifestyle optimization with either medication
Neither Ozempic nor retatrutide works in isolation. The medications create a metabolic environment where weight loss becomes dramatically easier, but dietary choices and physical activity still influence outcomes significantly. The difference between average results and exceptional results often comes down to what you do alongside the medication.
Protein intake becomes especially important during rapid weight loss. Both medications can lead to loss of lean muscle mass alongside fat, and adequate protein (0.7 to 1.0 grams per pound of body weight daily) helps preserve muscle. This is particularly relevant with retatrutide, where the pace of weight loss is faster and the risk of excessive lean mass loss is proportionally higher, though the glucagon component may offer some muscle-sparing benefit.
Dietary strategies tailored to GLP-1 therapy can reduce side effects and improve overall outcomes. Our semaglutide diet plan and tirzepatide diet plan offer detailed frameworks. Understanding which foods to avoid and what to eat on GLP-1 therapy makes a meaningful difference in both tolerability and results. Even simple adjustments like understanding alcohol interactions and the role of B12 supplementation can optimize the treatment experience.
Exercise during GLP-1 therapy should emphasize resistance training to preserve muscle mass. Cardiovascular exercise supports the metabolic benefits both medications provide. The combination of pharmaceutical intervention plus structured exercise plus optimized nutrition produces results that exceed any single approach alone. For those who prefer a lower-effort approach, our analysis of losing weight on semaglutide without exercise examines what is realistically achievable with medication alone.
Compounding and the research peptide landscape
The GLP-1 market has spawned a parallel ecosystem of compounded and research-grade peptides. Understanding how Ozempic and retatrutide fit into this landscape helps contextualize the options available today.
Compounded semaglutide, produced by 503B pharmacies, has become widely available as an alternative to brand-name Ozempic and Wegovy. These formulations contain the same active ingredient but are manufactured differently and are not FDA-approved products. They typically cost significantly less than brand-name options. Pharmacies like Empower and others have become major players in this space. For those exploring compounded options, understanding reconstitution of 5mg semaglutide and the proper mixing ratios is essential for safe use.
Retatrutide exists in the research peptide market, available from various suppliers for research purposes. Quality varies significantly between suppliers. Our retatrutide purchasing guide and reviews of specific suppliers like Paradigm Peptides help researchers evaluate their options. The research peptide route requires more knowledge about dosage calculation, reconstitution, storage, and injection technique than using a pre-filled pharmaceutical pen.
Enhanced semaglutide formulations have also emerged, combining semaglutide with additives like glycine, methylcobalamin, or other compounds intended to reduce side effects or enhance efficacy. Whether these additions provide meaningful benefits remains debated, but they represent an evolving area of the compounded GLP-1 market.
Common misconceptions about Ozempic vs retatrutide
The conversation around these medications is filled with half-truths and oversimplifications. Clearing up the most persistent misconceptions helps set realistic expectations.
Misconception: Retatrutide is just a stronger version of Ozempic. Not exactly. While both contain GLP-1 activity, retatrutide is a fundamentally different molecule with a different mechanism. It is not simply semaglutide with the dial turned up. The additional GIP and glucagon receptor activity creates qualitatively different metabolic effects, not just quantitatively more of the same thing. Understanding whether GLP-1 is the same as Ozempic helps clarify why these distinctions matter.
Misconception: Retatrutide will make Ozempic obsolete. Unlikely. Different patients have different needs. Some will prefer the proven safety profile and simpler mechanism of semaglutide. Others will benefit from the enhanced efficacy of retatrutide. The weight loss medication market will likely support both, just as both tirzepatide and semaglutide coexist today. Even within the current landscape, some people respond better to one medication over another based on their individual side effect profile.
Misconception: You should wait for retatrutide instead of starting Ozempic. For most people, no. If you need treatment now, starting Ozempic (or tirzepatide) provides real benefits immediately. Waiting two years for a theoretical advantage means two years of untreated obesity and its associated health risks. The best medication is the one you can access and tolerate today.
Misconception: Retatrutide has no side effects because it is newer. Wrong. It has all the same GI side effects as other GLP-1 medications, plus a few unique ones like dysesthesia. Newer does not mean cleaner. It means less characterized.
Misconception: Ozempic results are disappointing compared to retatrutide. Perspective matters. A 15% average weight loss was considered extraordinary before retatrutide data emerged. It still represents meaningful, life-improving change for millions of people. Comparing everything to the best possible alternative makes every other option look inadequate, which is neither fair nor helpful for decision-making.
The future of weight loss pharmacotherapy
Ozempic and retatrutide represent two points on a trajectory that is moving fast. The GLP-1 revolution, which started with relatively modest single-agonist drugs, has progressed to dual-agonists, then triple-agonists, and the pipeline continues to expand.
Amycretin, a dual GLP-1/amylin agonist from Novo Nordisk, showed 22% weight loss at just 36 weeks in early trials. Oral formulations that match injectable efficacy are in development. Combination approaches pairing GLP-1 drugs with myostatin inhibitors (to preserve muscle) or with other metabolic targets are being explored. GLP-1 patches and novel delivery systems aim to make administration even easier.
Within the next five to ten years, the treatment landscape will look dramatically different from today. Medications capable of producing 30%+ weight loss with minimal side effects and muscle preservation are not science fiction. They are in clinical trials right now. The GLP-1 plotter can help visualize projected outcomes across different medication options as the field evolves.
For those navigating this landscape, the key is making the best decision with the information and options available today, while staying informed about what is coming. SeekPeptides tracks these developments in real time, providing members with updated protocols, emerging research summaries, and practical guidance on both currently available and pipeline therapies.
Frequently asked questions
Is retatrutide better than Ozempic for weight loss?
Based on clinical trial data, retatrutide produces significantly more weight loss than Ozempic. The TRIUMPH-4 Phase 3 trial showed 28.7% average weight loss with retatrutide 12 mg at 68 weeks, compared to 14.9% to 17.4% with semaglutide 2.4 mg across STEP trials. However, retatrutide is not yet FDA approved and lacks the long-term safety data that semaglutide has. The retatrutide vs semaglutide comparison explores this question in greater detail.
When will retatrutide be available?
FDA approval for retatrutide is projected for late next year, with commercial launch expected in early the year after. Multiple Phase 3 TRIUMPH trials are expected to report results this year. For the most current information, our retatrutide availability tracker is updated regularly as new developments emerge.
Can I switch from Ozempic to retatrutide?
When retatrutide becomes commercially available, switching from Ozempic will likely be possible under medical supervision. The standard approach would involve starting retatrutide at its lowest dose (1 mg) and following the full titration schedule, regardless of your previous semaglutide dose. This allows your body to adapt to the additional GIP and glucagon receptor activation. The retatrutide dose schedule outlines the expected titration timeline.
Do Ozempic and retatrutide have the same side effects?
They share common gastrointestinal side effects including nausea, diarrhea, constipation, and vomiting. However, retatrutide has shown a unique side effect called dysesthesia (skin tingling/sensitivity) in approximately 20.9% of patients at the highest dose. Retatrutide may also cause more pronounced heart rate increases. For managing GI side effects common to both, our constipation treatment guide and related strategies apply broadly to GLP-1 therapy.
Is retatrutide the same as tirzepatide?
No. Both are made by Eli Lilly, but tirzepatide is a dual-agonist (GLP-1 + GIP) while retatrutide is a triple-agonist (GLP-1 + GIP + glucagon). The additional glucagon receptor activity in retatrutide produces greater weight loss and more dramatic liver fat reduction. Tirzepatide is FDA approved and available now, while retatrutide is still in clinical trials. Our comparison guides help differentiate all the options in this space.
Does Ozempic have cardiovascular benefits that retatrutide does not?
Currently, yes. The SELECT trial demonstrated a 20% reduction in major adverse cardiovascular events with semaglutide in people with obesity without diabetes. Retatrutide has not yet been studied in a dedicated cardiovascular outcome trial. Early data shows improvements in cardiovascular risk markers (cholesterol, blood pressure, inflammation), but these are surrogate endpoints, not proven event reduction.
How does retatrutide help with fatty liver disease?
Retatrutide demonstrated remarkable liver fat reduction in a Phase 2a trial. At 48 weeks, 93% of participants taking 12 mg achieved normal liver fat levels (below 5%), with an average 86% reduction in liver fat from baseline. The glucagon component appears to drive this effect by promoting hepatic fat oxidation. No other GLP-1 medication has produced comparable liver results.
Should I start Ozempic now or wait for retatrutide?
For most people needing weight loss treatment, starting now with an available medication is the better choice. Untreated obesity carries ongoing health risks. You can always transition to retatrutide later if it proves advantageous. Waiting two or more years for a drug that may or may not receive approval means losing valuable treatment time. Consult with your healthcare provider about the best option for your specific situation. If you do start with semaglutide, understanding how quickly appetite suppression begins helps set expectations for the early weeks of treatment.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Whether you are evaluating Ozempic, exploring retatrutide, or comparing every option in between, SeekPeptides members access the detailed analysis and personalized guidance that makes confident decisions possible.
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your protocols stay optimized, and your results stay consistent.