Feb 20, 2026

Some researchers use every last drop before day 14. Others discover half a vial sitting in the fridge on day 35. One person sees full results. The other wonders why appetite suppression faded, why the scale stopped moving, why the medication seems weaker than it was three weeks ago. The difference is not the semaglutide itself. It is what happens to that semaglutide after the vial or pen is opened for the first time.
The 28-day question comes up constantly. And it deserves more than a yes-or-no answer, because the reality is far more nuanced than most sources suggest. Compounded semaglutide, Ozempic, and Wegovy each follow different rules. The concentrations differ. The preservative systems differ. The pen designs differ. And the actual shelf life of semaglutide after opening depends on which version you are using, how you store it, and what standards your compounding pharmacy follows.
This guide breaks down exactly what happens to compounded semaglutide and brand-name formulations after that first puncture or injection. You will learn the science behind peptide degradation, the real-world storage rules that matter, how to spot a compromised vial, and precisely when to throw it away versus when you still have safe, effective medication. SeekPeptides has compiled the most comprehensive breakdown available, covering every formulation, every scenario, and every common mistake researchers make with semaglutide storage.
The 28-day rule explained (and why it exists)
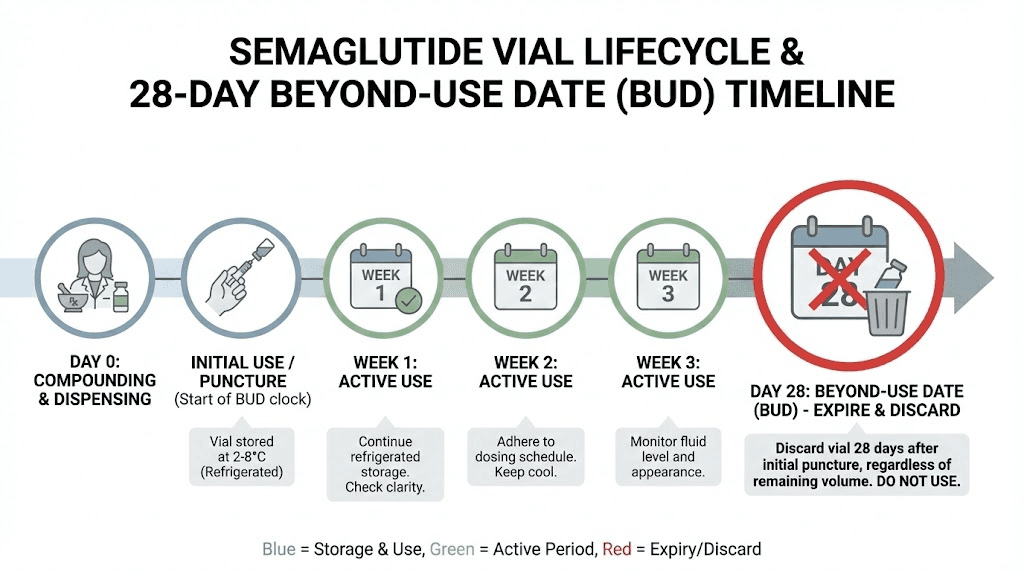
The number 28 is not arbitrary. It did not come from a marketing department or a rough guess. It comes from USP 797, a set of standards published by the United States Pharmacopeia that governs sterile compounding practices. These standards exist to protect patients from two specific dangers: microbial contamination and chemical degradation.
When a compounding pharmacy prepares a compounded semaglutide vial, they assign a beyond-use date. This BUD represents the last day the pharmacy can guarantee the medication meets its labeled potency and sterility standards. For most compounded sterile preparations stored under refrigeration, USP 797 sets a default BUD of 28 days unless the pharmacy has conducted extended stability testing that proves the formulation lasts longer.
Here is the critical distinction most people miss. A beyond-use date is not the same as an expiration date. An expiration date appears on FDA-approved medications and reflects extensive stability testing conducted over months or years under controlled conditions. A BUD is a more conservative estimate based on compounding standards. It tells you how long the pharmacy can guarantee full potency and sterility under their specific compounding conditions.
Think of it this way. An expiration date says the manufacturer tested this product for 36 months and confirmed it stays effective. A BUD says the compounding pharmacy followed standard protocols that support a 28-day window of guaranteed quality. The actual medication might remain effective beyond that window, but the pharmacy cannot make that promise.
Every time a needle punctures a vial, it creates a potential entry point for microorganisms. Even with proper technique, even with alcohol swabs, even with bacteriostatic water containing preservatives, each puncture introduces a small risk. Over 28 days of repeated access, that cumulative risk reaches a threshold where USP 797 says the guarantee expires.
The second factor is chemical stability. Peptides are proteins, and proteins degrade over time. Temperature fluctuations, light exposure, and repeated handling all accelerate this process. After 28 days in use, the active semaglutide molecules may have broken down enough to reduce the effective dose you receive with each injection. Not necessarily to zero, but potentially below the therapeutic threshold needed for consistent results.
Different formulations, different timelines
Not all semaglutide follows the 28-day rule. This is where confusion multiplies, because three different products contain the same active ingredient but come with dramatically different storage windows.
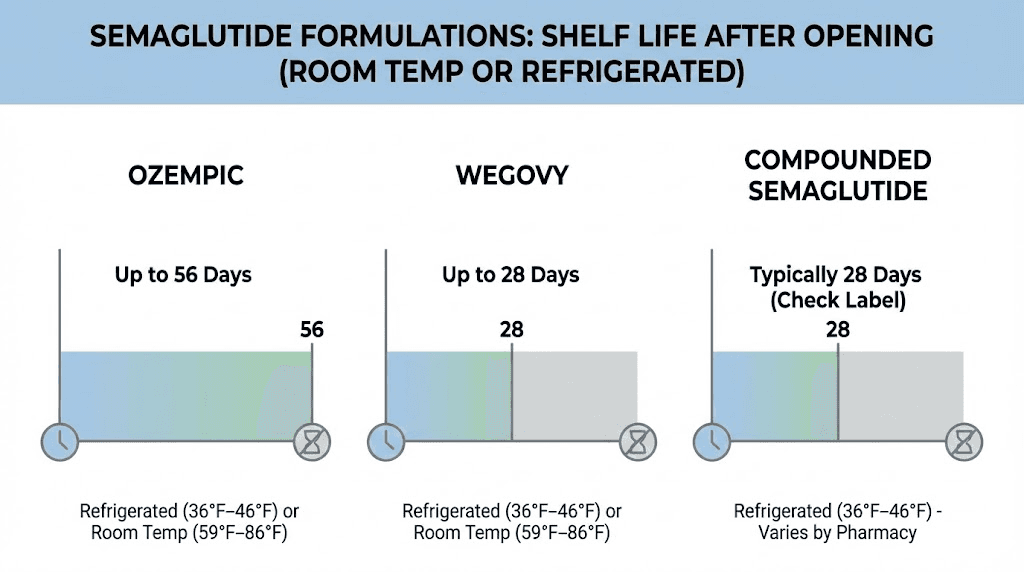
Ozempic pens: 56 days after first use
Ozempic is the brand-name semaglutide pen approved for type 2 diabetes management. Once you perform the first injection from an Ozempic pen, you have 56 days to use the remaining doses. That is double the 28-day window that applies to compounded versions. The pen can be stored at room temperature (59 to 86 degrees Fahrenheit) or kept refrigerated during this period.
Why 56 days instead of 28? Several factors converge. Ozempic pens use lower concentrations of semaglutide compared to many compounded formulations, and lower concentrations tend to be more stable. The pen delivery system is a sealed, single-patient device designed by Novo Nordisk with specific preservative formulations validated through extensive stability testing. The company submitted data to the FDA showing the medication remains within specification for 56 days after first use under real-world conditions. These tests evaluated potency, sterility, and physical characteristics at multiple time points throughout the 56-day window.
Understanding semaglutide dosage in units matters here because Ozempic pens contain multiple doses. A single pen typically holds four weekly doses, meaning you will use the pen over approximately one month. The 56-day window provides a generous buffer beyond that typical usage period. If you follow the standard semaglutide dosage chart and inject weekly, you should finish the pen well within that timeframe.
Wegovy pens: 28 days at room temperature
Wegovy is the brand-name semaglutide pen approved specifically for weight management. Despite containing the same active ingredient as Ozempic, Wegovy follows a 28-day rule for room temperature storage. Here is where it gets interesting. Wegovy pens are designed as single-dose, single-use devices. Each pen contains exactly one dose, and you are supposed to discard the pen after that single injection even if medication remains visible inside.
The 28-day figure for Wegovy applies to how long an unrefrigerated pen remains viable, not how many days you should draw from it. Since each pen is one injection, the practical relevance of this number relates to storage before use rather than in-use stability. If you take a Wegovy pen out of the refrigerator, you have 28 days to use it before it should be discarded.
Unopened Wegovy pens stored in the refrigerator remain good until the printed expiration date, which can extend well beyond 28 days. The distinction matters. Refrigerated and unopened is one timeline. Room temperature or opened is another.
Compounded semaglutide vials: 28 days after first puncture
This is where most of the confusion lives. Compounded semaglutide comes in multi-dose vials that you draw from repeatedly over the course of your dosing protocol. Once the vial seal is punctured for the first time, the 28-day countdown begins. After 28 days, the BUD has passed, and the pharmacy can no longer guarantee potency or sterility.
Some compounding pharmacies assign longer BUDs based on their own stability testing. You might receive a vial with a 30-day, 60-day, or even 90-day BUD. This is allowed under USP 797 if the pharmacy has conducted or has access to appropriate stability data supporting the extended date. Always check the label on your specific vial rather than assuming the default 28-day window applies.
The type of bacteriostatic water used in compounding also affects the timeline. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which inhibits microbial growth. Vials reconstituted with bacteriostatic water generally have longer viable windows compared to those prepared with sterile water, which contains no preservative. If you are reconstituting semaglutide yourself for research purposes, the choice of diluent directly impacts how long the solution remains safe.
Why the timelines differ
Three main factors explain the variation between formulations.
Concentration. Ozempic uses relatively low semaglutide concentrations. Lower concentrations tend to be more chemically stable because there are fewer peptide molecules available to aggregate or interact with each other. Research published in the Journal of Peptide Science found that pH, buffer composition, and concentration all significantly influence semaglutide degradation rates. Compounded vials often use higher concentrations to reduce injection volume, which may trade convenience for reduced stability.
Delivery system. Ozempic and Wegovy use sealed pen systems designed to minimize air exposure and contamination with each use. The pen needle is attached immediately before injection and removed afterward, limiting the time the medication contacts external air. Multi-dose compounded vials, by contrast, require repeated needle insertions through a rubber stopper, creating cumulative contamination risk over the 28-day use period.
Preservative systems. Brand-name formulations undergo proprietary preservative optimization validated through FDA-required stability testing. Compounded formulations rely on standard preservative approaches that may or may not match the stability profile of the branded products. This is not a quality judgment. It simply reflects different levels of testing and validation.
If you are tracking your semaglutide dosage in ml from a compounded vial, calculate whether your total prescribed doses will be completed within the BUD window. If the math does not work out, you may need to discuss vial sizing with your provider to avoid wasting medication.
What actually happens to semaglutide after 28 days
Day 29 does not trigger a magic switch that turns effective medication into water. Degradation is a gradual process that begins the moment a vial is opened and accelerates over time. Understanding the mechanisms helps you make informed decisions about your medication.
Peptide degradation mechanisms
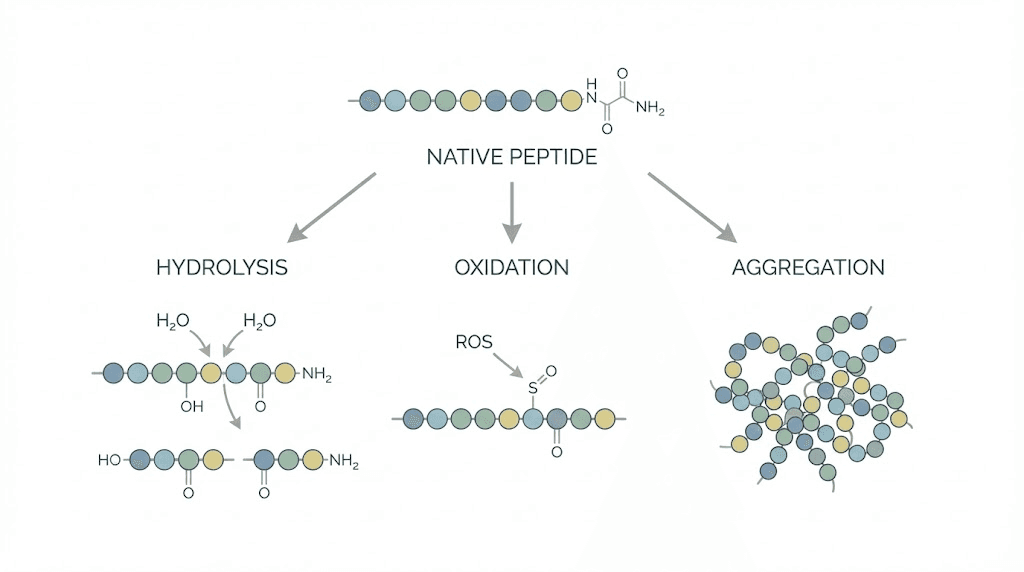
Semaglutide is a modified GLP-1 receptor agonist, a peptide with a fatty acid side chain that extends its half-life in the body. Like all peptides, it degrades through several chemical pathways. Hydrolysis breaks peptide bonds when water molecules attack the amino acid chain. Oxidation damages specific amino acid residues, particularly methionine and tryptophan. Deamidation converts asparagine residues to aspartate, altering the molecule structure.
A preformulation study published in the Journal of Peptide Science examined semaglutide stability across different temperatures and pH conditions. The researchers found that degradation accelerated significantly at higher temperatures and at pH values near semaglutide isoelectric point of 5.4. The practical takeaway is that proper refrigeration at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit) substantially slows these degradation processes.
At refrigerator temperatures, degradation happens slowly enough that the medication remains within acceptable potency ranges for weeks. At room temperature, the rate increases. At elevated temperatures above 86 degrees Fahrenheit, degradation can become clinically significant within days rather than weeks. This is why temperature excursions matter so much for semaglutide storage.
Aggregation and fibril formation
Research published in Biomacromolecules revealed something fascinating about semaglutide behavior in solution. The fatty acid chain that makes semaglutide long-acting in the body also causes it to self-assemble into structures called micelles at higher concentrations. Think of micelles as clusters of semaglutide molecules that form spontaneously when the concentration is high enough.
At lower concentrations, semaglutide forms dimers and trimers, small clusters of two or three molecules. At higher concentrations, it forms spherical micelles. A small population of needle-shaped fibril aggregates was also observed. While the researchers noted no evidence of chemical degradation from this aggregation, the formation of larger aggregates could potentially affect how the medication is absorbed after injection.
This aggregation tendency is one reason why concentration matters for stability. Higher-concentration compounded vials may be more prone to aggregation over time compared to lower-concentration branded formulations. Proper storage minimizes this effect, but it cannot eliminate it entirely.
Bacterial contamination risks
Every time you insert a needle through a vial stopper, you create a pathway for bacteria. Good aseptic technique reduces this risk dramatically, but it does not eliminate it completely. Over 28 days of repeated access, even with careful handling, the cumulative contamination probability increases.
The bacteriostatic water preservative system provides a critical safety margin. The benzyl alcohol in bacteriostatic water actively kills many common bacteria, but it is not effective against all organisms. Certain molds and resistant bacterial species can survive in preserved solutions, particularly if the vial has been exposed to environmental contaminants.
Signs of bacterial contamination include cloudiness, visible particles floating in the solution, and discoloration. However, some contamination is invisible to the naked eye. This is why the 28-day BUD exists as a precautionary measure, even when the solution looks perfectly clear.
Potency loss over time
The most practical concern for most researchers is potency loss. If you are using semaglutide for appetite suppression or weight management, reduced potency means reduced effectiveness. The medication may still contain active semaglutide after 28 days, but the amount per injection might be lower than the labeled dose.
Exact potency loss percentages after 28 days are not publicly available for most compounded formulations. Pharmaceutical manufacturers conduct extensive stability testing but rarely publish granular degradation curves. What we know is that USP standards require a minimum of 90% of labeled potency for the medication to be considered within specification. If a pharmacy assigns a 28-day BUD, they are saying the medication should maintain at least 90% potency through day 28 under proper storage conditions.
After day 28, potency could drop below that 90% threshold. It might not. But the pharmacy can no longer guarantee it. This matters most when you are on a carefully titrated dosing protocol where precise dosing determines your results. If you are following a 5mg semaglutide dosage chart and expecting specific results at specific dose levels, even a 10% potency reduction changes the effective dose you are receiving.
Signs your semaglutide has gone bad
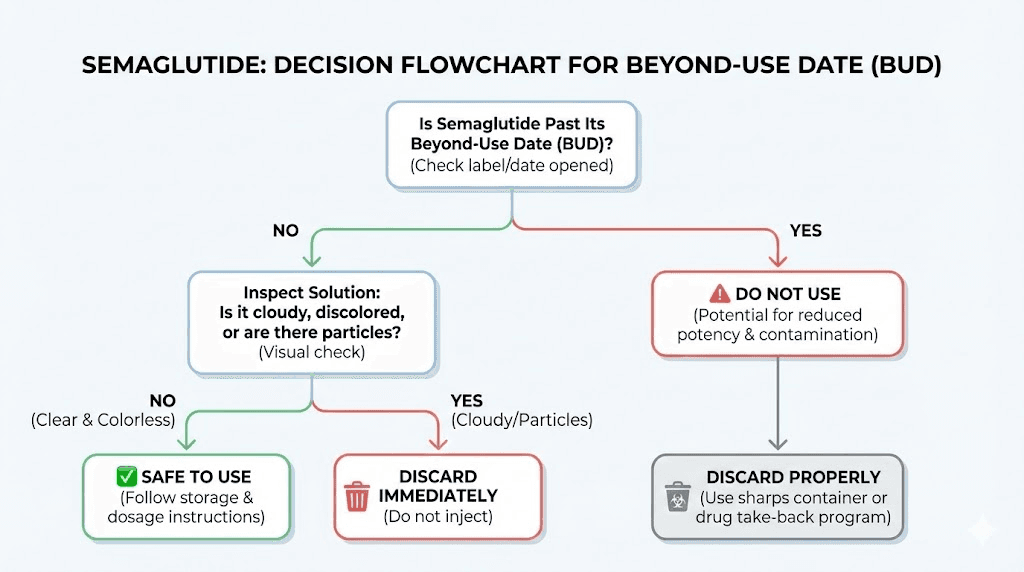
Visual inspection is your first line of defense. Before every injection, take five seconds to examine your medication. This habit could save you from injecting a compromised product.
What normal semaglutide looks like
Properly stored semaglutide solution should be completely clear and colorless. Hold the vial up to a light source and look through it. The liquid should look like water, with no visible particles, no cloudiness, and no color whatsoever. Compounded semaglutide with vitamin B12 may have a pink or reddish tint from the cyanocobalamin, which is normal for that specific formulation.
The rubber stopper on a multi-dose vial should show no signs of coring, where small pieces of rubber break off into the solution from repeated needle punctures. If you see tiny rubber fragments floating in the liquid, the vial is compromised regardless of the date on the label.
Warning signs that mean immediate disposal
Cloudiness or turbidity. If the solution appears hazy, foggy, or opaque rather than crystal clear, something has changed. Cloudiness can indicate bacterial growth, protein aggregation, or chemical precipitation. None of these are acceptable.
Visible particles. Any floating specks, fibers, or sediment disqualify the medication from use. Even a single visible particle raises concerns about the overall sterility and stability of the solution.
Color changes. Standard semaglutide should be colorless. Any yellowing, browning, or other discoloration suggests chemical degradation. Again, formulations containing methylcobalamin or cyanocobalamin will have a characteristic color from the B12 component, but any change from the original color warrants caution.
Unusual odor. Semaglutide solution should have little to no odor. A strong or unpleasant smell could indicate bacterial contamination or chemical breakdown of the preservative system.
Frozen or previously frozen solution. If your semaglutide has frozen at any point, discard it. Freezing causes peptide molecules to denature and aggregate in ways that cannot be reversed by thawing. The solution may look normal after thawing but could contain structurally damaged semaglutide that no longer functions properly.
The pre-injection inspection checklist
Build this into your routine. Every single time.
Check the date. Is the vial within its BUD or expiration date? Check the appearance. Is the solution clear, colorless, and free of particles? Check the storage. Has it been properly refrigerated? Check the seal. Is the vial stopper intact with no signs of damage? If any answer raises doubt, do not inject. A fresh vial is always safer than a questionable one. Understanding peptide safety starts with these basic inspection practices.
Proper storage to maximize every day of shelf life
Storage is not complicated. But it is unforgiving. Small mistakes accumulate over days and weeks, and the consequences show up as reduced potency or compromised sterility long before the medication looks any different.
Temperature requirements
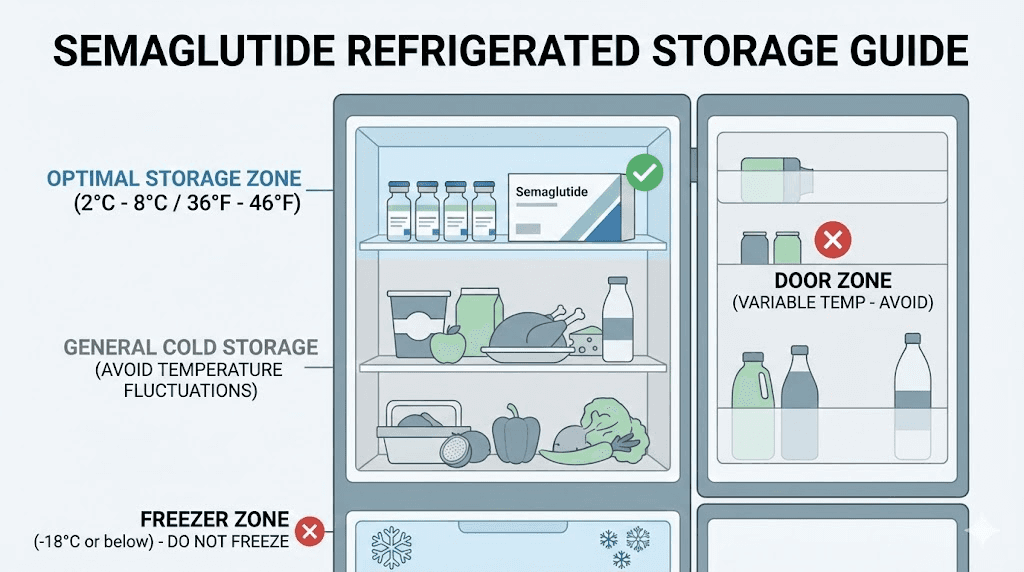
The ideal storage temperature for semaglutide is 2 to 8 degrees Celsius, which translates to 36 to 46 degrees Fahrenheit. This is standard refrigerator temperature. Your medication should live on a shelf inside the refrigerator, not in the door where temperatures fluctuate every time you open it, and not near the back where items can freeze against the cooling element.
If you want to confirm your refrigerator maintains the right range, consider placing a simple thermometer inside. Refrigerators can have surprising temperature variations between zones, and the difference between 4 degrees Celsius and minus 2 degrees Celsius could mean the difference between perfectly stored medication and a ruined vial. This principle applies equally to all reconstituted peptides, not just semaglutide.
Room temperature windows
Brand-name Ozempic pens can tolerate room temperature storage (59 to 86 degrees Fahrenheit) for up to 56 days after first use. Wegovy tolerates room temperature for up to 28 days. Compounded semaglutide generally should remain refrigerated at all times once opened, though brief room-temperature exposure during injection preparation is expected and acceptable.
Brief means minutes, not hours. Taking your vial out to prepare and administer an injection, then returning it to the refrigerator within 15 to 20 minutes, falls well within acceptable parameters. Leaving a compounded vial on your bathroom counter all day does not. Each hour at room temperature accelerates degradation processes that are nearly dormant at refrigerator temperatures.
If you have questions about how temperature affects semaglutide that has gotten warm, the general guideline is that short exposures to room temperature are unlikely to cause significant degradation, but extended exposure or exposure to heat above 86 degrees Fahrenheit should be taken seriously.
Freezing: the absolute no
Never freeze semaglutide. Not to extend its shelf life. Not because you think colder is better. Not accidentally by pushing it too far back in the refrigerator. Freezing destroys peptide structure through a process called freeze-thaw denaturation. Ice crystals that form during freezing physically damage protein molecules, causing irreversible aggregation and loss of biological activity.
The damage from freezing is invisible. A thawed vial of semaglutide may look perfectly clear and normal. But the molecules inside have been scrambled. You could inject the full labeled dose and receive a fraction of the expected biological activity. If there is any chance your medication has frozen, even briefly, discard it and start fresh.
Light exposure
Semaglutide is sensitive to light, particularly ultraviolet light. Store vials in their original packaging or in a location that protects them from direct light. A refrigerator interior is generally dark enough, but if you store medication in a transparent container or on an open shelf, light exposure could accelerate degradation.
The brand-name pens come in opaque packaging for this reason. If you transfer compounded vials to a different container for organization, ensure the container is opaque or kept in a consistently dark environment.
Vial handling best practices
When drawing from a multi-dose vial, always clean the stopper with an alcohol swab before inserting the needle. Use a new, sterile needle for each access. Never touch the stopper surface with your fingers after cleaning it. Draw the medication smoothly without excessive agitation. Do not shake the vial vigorously, as this can promote aggregation and introduce air bubbles.
If you are learning proper injection technique, our guide on where to inject GLP-1 covers site selection, while the GLP-1 injection guide walks through the full process step by step.
Compounded vs brand-name semaglutide storage: the real differences
The gap between compounded and brand-name semaglutide storage requirements is not about quality. It is about testing, infrastructure, and regulatory frameworks. Understanding these differences helps you set appropriate expectations for each product type.
Manufacturing and quality control
Brand-name manufacturers like Novo Nordisk operate under Current Good Manufacturing Practice (cGMP) regulations enforced by the FDA. Every batch undergoes extensive testing, and the company maintains stability data spanning years. When Novo Nordisk says Ozempic lasts 56 days after first use, that claim is backed by thousands of data points from controlled stability studies.
Compounding pharmacies operate under different regulations, primarily state boards of pharmacy and USP standards. They produce smaller batches with less extensive stability testing. This does not mean compounded semaglutide is inferior, but it does mean the safety margins built into their BUDs tend to be more conservative. A 28-day BUD might actually reflect a product that remains stable for 45 or 60 days, but the pharmacy cannot prove it without conducting additional testing.
The Empower Pharmacy and Olympia Pharmacy are among the larger compounding pharmacies that produce semaglutide. Larger operations often have more resources for stability testing and may assign extended BUDs based on their testing data. Smaller pharmacies may default to the USP 797 standard 28-day BUD.
Preservative differences
Brand-name formulations use proprietary preservative systems optimized specifically for their product. These preservatives are validated through FDA-required studies that demonstrate effectiveness against a panel of microorganisms over the entire labeled shelf life.
Compounded formulations typically rely on bacteriostatic water with 0.9% benzyl alcohol. This is an effective preservative for many applications, but it has limitations. Benzyl alcohol is most effective against gram-positive bacteria and less effective against gram-negative organisms and molds. The preservative concentration can also diminish over time through chemical interaction with the rubber stopper or the peptide itself.
For researchers who reconstitute semaglutide themselves, always use bacteriostatic water rather than sterile water for multi-dose vials. Sterile water contains no preservative, meaning bacterial contamination from repeated access has nothing to prevent it from multiplying. A vial reconstituted with sterile water should ideally be used in a single session or discarded within 24 hours.
Multi-dose vial puncture considerations
This is a practical concern that brand-name pen users never face. Every time you push a needle through a compounded vial stopper, you are introducing a potential contamination pathway. After 10, 15, or 20 punctures over 28 days, the rubber stopper may develop micro-channels that increase contamination risk.
Good technique minimizes this risk. Use the thinnest needle that allows comfortable withdrawal of the medication, typically 27 to 30 gauge. Insert the needle at a slight angle to reduce coring. Do not use dull or previously used needles, as these are more likely to damage the stopper. If you are drawing doses for research, review the proper reconstitution ratios for 5mg vials or 10mg vials to ensure you are using the correct diluent volume.
What to ask your compounding pharmacy
Before your first injection, get clear answers to these questions. What is the beyond-use date for this specific vial? Was the BUD based on stability testing or the default USP 797 standard? What preservative system was used? What storage temperature do you recommend? What should I do if the medication was left out of the refrigerator for several hours?
A good compounding pharmacy will answer these questions without hesitation. If the staff cannot tell you the basis for their BUD assignment, that is a red flag worth noting.
Traveling with semaglutide and keeping it safe
Travel introduces every storage variable that degrades semaglutide: temperature swings, movement, time out of refrigeration, and disrupted routines. Planning ahead prevents these factors from compromising your medication.
Short trips (1 to 3 days)
For brief travel, an insulated cooler bag with a gel pack provides adequate temperature control. Place the gel pack in the bag at least two hours before travel so it reaches the proper temperature. Position your medication so it does not directly contact the frozen gel pack (which could cause localized freezing). A small towel or cloth barrier between the gel pack and the vial works well.
Brand-name Ozempic pens are more forgiving for short trips since they tolerate room temperature for extended periods. If you are traveling for a day or two with an Ozempic pen, it can stay at room temperature (below 86 degrees Fahrenheit) without concern. Compounded vials need refrigeration, so a cooler bag is essential even for overnight trips.
Air travel
Always carry semaglutide in your carry-on luggage, never in checked bags. Cargo holds can experience extreme temperature fluctuations, including freezing conditions at altitude. TSA allows injectable medications in carry-on bags. Bring your prescription label or pharmacy documentation in case questions arise at security.
For a deeper dive into travel logistics, our complete travel guide for semaglutide covers everything from international travel considerations to emergency backup plans.
Hotel and accommodation storage
Most hotel rooms have a mini-fridge. Check the temperature with a thermometer if possible, as hotel mini-fridges often run warmer than standard refrigerators. If the fridge seems unreliable, ask the front desk if they can store your medication in a proper refrigerator, perhaps in the hotel kitchen or a staff area.
Never leave semaglutide in a hot car, on a windowsill, or anywhere exposed to direct sunlight. Temperatures inside a parked car can exceed 150 degrees Fahrenheit in summer, which would degrade the medication rapidly. If you need to leave the medication during activities, the insulated cooler bag with a fresh gel pack is your best option.
What to do if you used semaglutide past 28 days
It happens. You forgot to check the date. You miscounted. You squeezed one more dose out of a vial that was a few days past its BUD. Take a breath. The practical risk depends on several factors.
Realistic risk assessment
Using semaglutide a few days past the 28-day BUD under proper refrigeration is unlikely to cause harm. The BUD is a conservative threshold, not a cliff edge. Day 29 compounded semaglutide that has been properly refrigerated is probably not meaningfully different from day 28 semaglutide.
The risks increase the further you go past the BUD and the worse the storage conditions have been. A vial on day 35 that has been consistently refrigerated carries less risk than a vial on day 30 that sat at room temperature for a full day. Time and temperature are the two variables that matter most.
The primary risk from using post-BUD semaglutide is not a dangerous adverse reaction. It is reduced potency. If the semaglutide has degraded, you may simply receive less active medication per injection. This shows up as reduced appetite suppression, slower progress, or a sense that the medication is not working as well as it used to.
Symptoms to watch for
If you suspect contamination rather than simple degradation, watch for injection site reactions that seem more pronounced than usual: increased redness, swelling, warmth, or pain at the injection site. Fever or flu-like symptoms within 24 to 48 hours of injection could also indicate an issue. These would be signs of a potential infection rather than a semaglutide-specific problem.
Contaminated medication does not always cause immediate symptoms. Some bacterial contamination produces effects days later. If you develop unexpected symptoms after using medication past its BUD, mention the expired medication to your healthcare provider so they can assess appropriately.
How to get back on track
Discard the expired vial. Start a fresh one. Resume your normal dosing schedule. There is no need to adjust your dose up to compensate for potentially reduced potency from the expired vial. Simply return to your standard protocol.
If you experienced a noticeable reduction in effectiveness during the period you were using expired medication, you may notice results improving once you switch to fresh medication. This can actually serve as a useful indicator. If your results noticeably improve after switching vials, it suggests the previous vial had indeed lost potency, which confirms the importance of tracking your BUD going forward.
For researchers tracking their progress over time, our guide on how fast semaglutide works provides realistic timelines for what to expect at each stage. And if you notice a plateau in results, expired medication is one factor worth investigating before assuming you need a dose adjustment.
Maximizing your vial and reducing waste before day 28
Wasted medication is wasted money and wasted progress. With proper planning, you can use every dose in a vial before the BUD expires.
Matching vial size to your dosing schedule
The most common source of waste is a mismatch between vial size and dosing frequency. If you have a 5mg vial and you are taking 0.25mg per week, that vial contains 20 doses, enough for 20 weeks. But the vial expires in 28 days. The math does not work.
Work with your provider to choose a vial size that matches your dosing schedule within the BUD window. At 0.25mg per week, a vial should contain no more than about 1mg to be fully used within 28 days (four weekly doses of 0.25mg). At 1mg per week, a vial containing up to 4mg works within the window. At higher maintenance doses, larger vials become practical.
Use the SeekPeptides semaglutide dosage calculator to determine exactly how much medication you need per injection and per vial. The peptide reconstitution calculator can then help you determine the right amount of bacteriostatic water to add for your target concentration.
Reconstitution math
Getting the concentration right at reconstitution time is critical for avoiding waste. If you reconstitute a 5mg vial with too much water, each injection requires drawing a larger volume, which can be impractical and may mean you cannot fit all planned doses within a single vial.
For example, reconstituting 5mg of semaglutide in 2ml of bacteriostatic water gives you a concentration of 2.5mg/ml. At a dose of 0.5mg, you would draw 0.2ml per injection (20 units on an insulin syringe). The vial would contain 10 doses at 0.5mg each, requiring 10 weeks to use completely, which exceeds the 28-day BUD.
A better approach might be reconstituting 5mg in 1ml, giving a 5mg/ml concentration. At 0.5mg per dose, each injection is 0.1ml (10 units), and the vial still contains 10 doses. The concentration per injection is the same, but the total vial volume is smaller and easier to manage. Still, 10 doses at weekly intervals takes 10 weeks, which still exceeds the BUD.
The real solution is matching the vial to your protocol period. If you inject weekly and your BUD is 28 days, you get four injections per vial. At 0.5mg per injection, you need a 2mg vial. At 1mg per injection, you need a 4mg vial. Discuss this with your compounding pharmacy or provider.
Our semaglutide reconstitution chart and the 5mg reconstitution chart provide ready-made reference tables for common vial sizes and concentrations. The semaglutide mixing chart offers additional guidance for getting your ratios right.
Dose tracking and dating
Write the date of first puncture on the vial with a permanent marker. Do this immediately, not later, not when you remember. It takes two seconds and eliminates all uncertainty about when the 28-day clock started.
Some researchers also track the number of doses drawn from each vial using tally marks on a piece of tape attached to the vial. This confirms whether the vial contains enough medication for remaining doses and helps identify if doses have been missed or miscounted.
Common myths about semaglutide expiration
Misinformation about medication expiration is everywhere. Some myths lead to unnecessary waste. Others lead to using compromised medication. Neither outcome is acceptable.
Myth 1: semaglutide goes bad at exactly 28 days
The BUD is not a timer that triggers instant degradation. Day 28 and day 29 are chemically indistinguishable. The 28-day mark represents the point beyond which the pharmacy can no longer guarantee the medication meets full specifications. Actual degradation is a gradual curve, not a step function.
That said, treating the BUD as a hard deadline is the safest approach. The fact that day 29 is probably fine does not mean day 42 is probably fine. The further you go beyond the BUD, the less certainty you have about potency and sterility. The date exists as a bright line for a reason.
Myth 2: refrigeration makes semaglutide last forever
Refrigeration slows degradation dramatically. It does not stop it. Even at optimal refrigerator temperatures, peptide bonds slowly break, oxidation reactions proceed, and aggregation occurs. The rate is low enough to maintain potency for weeks or months in an unopened vial, but refrigeration alone cannot override the BUD for an opened multi-dose vial.
The contamination risk from repeated vial access is independent of temperature. Bacteria introduced through the stopper can grow slowly even at refrigerator temperatures, though the cold inhibits rapid multiplication. The BUD accounts for both degradation and contamination risks together, and refrigeration only addresses one of these.
Myth 3: if it looks clear, it is safe to use
Visual inspection catches the most obvious problems. Cloudiness, particles, and color changes are definitive reasons to discard medication. But clear, colorless semaglutide is not automatically safe. Chemical degradation that reduces potency does not necessarily change the solution appearance. Bacterial contamination in early stages may not produce visible cloudiness.
Use visual inspection as a necessary first check, not as a sufficient one. If the solution looks bad, definitely discard it. But a clear solution past its BUD does not mean the medication is fine. The two criteria, appearance and date, both matter independently. Understanding peptide storage principles helps put this in context for all research peptides, not just semaglutide.
Myth 4: brand-name and compounded semaglutide last the same time after opening
They do not. As detailed above, Ozempic lasts 56 days after first use. Compounded semaglutide typically has a 28-day BUD. Using brand-name timelines for compounded medication could mean injecting compromised medication for nearly a month past its actual BUD.
Always follow the specific guidance for your specific product. If you are switching between compounded semaglutide and a brand-name pen, adjust your storage expectations accordingly.
Myth 5: you can freeze semaglutide to extend its shelf life
Freezing destroys semaglutide. Full stop. Unlike many medications that tolerate or even benefit from frozen storage, peptide-based drugs like semaglutide are irreversibly damaged by ice crystal formation. The molecules unfold and aggregate in ways that eliminate biological activity.
This myth likely originates from the correct observation that lyophilized (freeze-dried) peptides can be stored frozen or at room temperature for extended periods. But lyophilization is a controlled industrial process that removes water before freezing, protecting the peptide structure. Freezing a liquid semaglutide solution in your home freezer is nothing like lyophilization, and the results are destructive. For more on how peptide form affects storage, see our guide on how long peptides last in powder form versus reconstituted solutions.
The science behind semaglutide stability
For researchers who want to understand the molecular details, the science of semaglutide stability is genuinely fascinating.
Semaglutide molecular structure and vulnerability
Semaglutide is not a simple small-molecule drug. It is a 31-amino-acid peptide analog of human GLP-1 with several modifications designed to extend its half-life. The most significant modification is a C18 fatty acid chain attached via a linker to a lysine residue at position 26. This fatty acid chain enables semaglutide to bind to albumin in the bloodstream, shielding it from rapid degradation and extending its activity from minutes (natural GLP-1) to approximately one week.
But this same modification creates interesting stability challenges in the vial. The fatty acid chain is hydrophobic, meaning it naturally avoids water. In solution, semaglutide molecules arrange themselves so their fatty acid tails cluster together, forming micellar structures. At higher concentrations, these micelles become more abundant and can evolve into larger aggregates over time.
The amino acid sequence itself contains several residues vulnerable to chemical modification. Asparagine residues can undergo deamidation, a reaction where a nitrogen-containing group is replaced by oxygen. Methionine residues are susceptible to oxidation. These reactions proceed slowly at refrigerator temperatures but accelerate with heat, light exposure, or unfavorable pH conditions.
pH matters more than you think
The preformulation study published in the Journal of Peptide Science found that semaglutide stability is highly pH-dependent. The most degradation occurred between pH 4.5 and 5.5, near the isoelectric point of semaglutide (pH 5.4). At its isoelectric point, a peptide has zero net charge, which means molecules are less repelled by each other and more likely to aggregate.
Commercial semaglutide formulations are buffered at pH values above 7.0, away from the isoelectric point, to maximize stability. Compounded formulations should follow the same principle, but the specific buffer system and pH can vary between pharmacies. This is another variable that affects how long your medication remains stable after opening.
Temperature stress studies
Research data shows semaglutide demonstrated reasonable thermal stability at elevated temperatures, remaining stable for 3 hours at 80 degrees Celsius (176 degrees Fahrenheit). However, extended exposure to even moderate heat significantly increases degradation. Stress stability studies conducted at 25, 40, and 60 degrees Celsius for 28 days showed increasing degradation at each temperature step.
The practical implication is clear. Brief temperature excursions, like the 15 minutes your vial sits on the counter during injection prep, cause negligible degradation. Extended exposure to temperatures above refrigeration, like leaving a vial in a warm car for hours, causes measurable damage that accumulates irreversibly. Every degree matters. Every hour counts. This is why proper refrigeration is non-negotiable for compounded formulations.
Semaglutide shelf life by formulation: quick reference
Here is the information distilled into a single reference table.
Formulation | Unopened shelf life | After opening | Storage temperature | Key notes |
|---|---|---|---|---|
Ozempic pen | Until printed expiration (refrigerated) | 56 days | 36-86F (2-30C) | Can store at room temp after opening |
Wegovy pen | Until printed expiration (refrigerated) | 28 days at room temp | 36-86F (2-30C) | Single-use pen, discard after one injection |
Compounded vial | Check pharmacy BUD | 28 days typical (check label) | 36-46F (2-8C) | Must stay refrigerated, check specific BUD |
Compounded with B12 | Check pharmacy BUD | 28 days typical | 36-46F (2-8C) | Pink/red color is normal from B12 |
This table covers the most common formulations, but always defer to the specific guidance provided with your medication. Compounding pharmacy BUDs can vary based on their stability data, and your specific vial may have a different timeline than the defaults listed here.
For complete dosing guidance alongside storage information, our semaglutide dosage chart in units and dosing chart provide the reference tables you need. If you are working with a specific vial size, the 5mg vial dosage chart or 10mg vial dosage chart breaks down injection volumes by dose.
How semaglutide storage compares to other GLP-1 medications
Semaglutide is not the only GLP-1 receptor agonist with specific storage requirements. Understanding how it compares to similar medications helps put the 28-day rule in broader context.
Tirzepatide storage comparison
Tirzepatide (brand names Mounjaro and Zepbound) follows similar storage principles but with its own specific timelines. Brand-name tirzepatide pens can be stored at room temperature for up to 21 days. If you are also researching whether tirzepatide expires, the same general principles apply: peptide degradation, contamination risk, and temperature sensitivity all factor into the shelf life equation.
Compounded tirzepatide storage in the fridge follows similar BUD guidelines to compounded semaglutide, typically 28 days after first puncture. The refrigeration requirements are comparable, and the same precautions about freezing, light exposure, and aseptic technique apply.
For researchers using both medications, whether switching from tirzepatide to semaglutide or comparing their side effect profiles, storage requirements are one area where the two medications behave very similarly. The same cooler bag, the same refrigerator shelf, and the same inspection habits work for both.
General peptide storage context
The 28-day BUD for compounded semaglutide is consistent with storage guidelines for most compounded injectable peptides. Whether you are working with BPC-157, TB-500, or other research peptides, the same storage fundamentals apply: refrigerate after reconstitution, use within the assigned BUD, inspect before each use, and never freeze reconstituted solutions.
Our comprehensive peptide storage guide covers these principles across all common research peptides. For specific information about how long peptides last in the fridge and at room temperature, those dedicated guides provide detailed breakdowns by peptide type.
Planning your semaglutide protocol around the 28-day window
Smart protocol design accounts for the BUD from the start. Waiting until you are staring at an almost-expired vial to figure out the math is a recipe for waste and frustration.
For new users starting at low doses
Most semaglutide protocols start at 0.25mg per week for the first four weeks. At this dose, a standard 5mg vial contains 20 weeks worth of medication, far more than the 28-day window allows. This means you will inevitably discard unused medication from each vial during the titration phase unless your pharmacy offers smaller vial sizes.
Ask your compounding pharmacy whether they offer 1mg or 2mg vials for the initial titration period. At 0.25mg per week, a 1mg vial provides exactly four doses, perfectly aligning with a 28-day window. At 0.5mg per week (the typical second month dose), a 2mg vial again provides four doses within the window.
This approach costs more per milligram of semaglutide because you are using more vials, but it eliminates waste and ensures every injection comes from fresh, fully potent medication. The compounded dosage calculator can help you work through the math for your specific situation, and the peptide dosage calculation guide covers the principles applicable to any peptide.
For maintenance dose users
Once you reach your maintenance dose, the math becomes simpler. At 1mg per week, a 4mg vial provides four doses within 28 days. At 2mg per week, an 8mg vial works. At 2.4mg per week (the maximum Wegovy dose), a 10mg vial provides just over four doses within the window.
At higher maintenance doses, larger vials become practical and cost-effective. The key is ensuring the total medication in the vial equals approximately four times your weekly dose (four weeks within the 28-day BUD). A small buffer is fine, but a vial containing three months worth of medication at your current dose creates guaranteed waste.
Dose escalation planning
Most semaglutide protocols involve gradual dose increases every four weeks. Each dose change represents an opportunity to re-evaluate your vial size. During the escalation phase, your weekly dose is changing, which means the optimal vial size changes too.
Map out your entire escalation schedule before starting. Know what dose you will be taking during each four-week block. Order vials sized to match each block. This level of planning might seem excessive, but it prevents the common scenario where researchers end up with mismatched vials and either waste medication or stretch vials past their BUD.
If you are following a standard 5mg/ml dosage chart for compounded semaglutide, the chart typically outlines the volume per injection at each dose level. Cross-reference these volumes with your vial size to confirm how many doses each vial contains.
The role of your compounding pharmacy
Not all compounding pharmacies are equal when it comes to semaglutide stability and quality. The pharmacy you choose directly impacts the shelf life and reliability of your medication.
Questions that matter
Beyond the basic BUD questions mentioned earlier, ask about the pharmacy accreditation. PCAB (Pharmacy Compounding Accreditation Board) accredited pharmacies undergo voluntary inspection and must meet standards that exceed state requirements. Accredited pharmacies typically have more robust quality systems, better stability testing, and more reliable BUD assignments.
Ask about potency testing. Does the pharmacy test each batch for potency before dispensing? Not all pharmacies do. Potency testing confirms that the semaglutide concentration in your vial matches what the label claims. Without testing, you are relying on the accuracy of the compounding process alone.
Ask about sterility testing. While testing every batch is impractical for most pharmacies, understanding their contamination prevention protocols provides insight into the reliability of their products. Environmental monitoring, personnel training, and cleanroom specifications all contribute to the sterility assurance of your medication.
Red flags to watch for
Vials without clear BUD labels. Pharmacies that cannot explain their stability data. Unusually long BUDs without published supporting data. Packaging that arrives without temperature control during shipping. Pharmacies that refuse to answer questions about their compounding process. Any of these suggest quality systems that may not fully protect the medication you depend on.
For researchers exploring different pharmacy options, our reviews of Empower Pharmacy, Olympia Pharmacy, and Direct Meds provide detailed breakdowns of what each major compounding pharmacy offers for semaglutide.
Additional factors that affect semaglutide longevity
Beyond the main variables of formulation, temperature, and time, several other factors influence how long your semaglutide remains effective.
Altitude and air pressure
Air pressure changes during air travel or at high altitude can affect the seal integrity of vial stoppers. Reduced atmospheric pressure at altitude can cause slight expansion of air inside the vial, potentially weakening the stopper seal over time. While this is a minor concern for typical use, researchers who travel frequently by air or live at high altitude should inspect vial seals more carefully.
Vibration and agitation
Excessive shaking or vibration can promote peptide aggregation. This is relevant during travel, where luggage handling can be rough. Pack medication in a padded, insulated case that minimizes movement. At home, avoid storing vials where they might be subject to vibration from appliances, traffic, or other sources.
Needle gauge and technique
The gauge of needle used to access the vial affects how much damage accumulates on the rubber stopper. Thinner needles (higher gauge numbers) create smaller punctures and less stopper damage over the course of a 28-day use period. Using 29 or 30 gauge needles for vial access minimizes the cumulative damage and reduces the risk of rubber coring or micro-channel formation.
Proper injection technique matters for both the vial and for your own safety. Our guides on GLP-1 injection sites and injection technique cover the practical aspects of subcutaneous injection for any GLP-1 medication.
Concurrent medications in the same syringe
Some researchers combine semaglutide with other compounds in the same syringe. Mixing medications introduces compatibility concerns that can affect stability. Semaglutide with B12 is a common combination available pre-mixed from pharmacies, but mixing in the syringe at the time of injection is different from pharmaceutical co-formulation.
Never combine medications in a syringe unless your pharmacy or provider has specifically confirmed compatibility. Incompatible mixtures can cause precipitation, pH shifts, or chemical interactions that degrade one or both compounds before they reach your body.
Frequently asked questions
Can I use compounded semaglutide on day 30 if it looks clear?
Visual clarity alone does not guarantee safety or potency past the beyond-use date. While a clear solution on day 30 has not undergone visible degradation, chemical potency loss and microbial contamination are not always visible. The safest approach is to follow the BUD on your label. If you do use it slightly past the BUD, understand that you may receive a reduced dose of active medication. For a deeper look at this question, see our guide on using expired semaglutide.
Does the 28-day rule apply to Ozempic?
No. Ozempic pens have a 56-day in-use period after the first injection, not 28 days. The 28-day rule applies specifically to most compounded semaglutide vials and to Wegovy pens stored at room temperature. Always check the specific product labeling for your formulation. Our comparison of GLP-1 and Ozempic explains the relationships between these different products.
What if my compounding pharmacy gave me a 60-day BUD?
Some pharmacies assign extended BUDs based on their own stability testing data. If your pharmacy has conducted testing that supports a 60-day or 90-day BUD, their assigned date takes precedence over the default 28-day standard. Ask them to confirm the testing basis for their BUD if you want additional reassurance.
Should I bring semaglutide to room temperature before injecting?
Allowing the medication to reach room temperature for a few minutes before injection can reduce injection site discomfort. Cold medication can sting slightly. However, limit room temperature exposure to 15 to 20 minutes maximum for compounded formulations. Brand-name pens are more tolerant of room temperature, but there is no benefit to extended warming. Read more about optimizing your injection experience in our best time to take semaglutide guide.
Can I transfer semaglutide to a different vial to extend its life?
Transferring medication between vials introduces contamination risk and does not reset the BUD clock. The degradation and contamination that have occurred over 28 days transfer with the medication. Additionally, each transfer involves needle punctures, air exposure, and handling that can introduce new contamination. This practice is not recommended.
Does semaglutide with B12 expire faster than plain semaglutide?
The addition of vitamin B12 (cyanocobalamin or methylcobalamin) to compounded semaglutide does not significantly change the storage timeline. The BUD assigned by the pharmacy accounts for the complete formulation, including any added ingredients. Follow the same BUD and storage guidelines as plain compounded semaglutide.
What is the shelf life of unopened compounded semaglutide?
Unopened compounded semaglutide stored under proper refrigeration typically has a BUD of 3 to 6 months from the date of compounding, though this varies by pharmacy. The BUD on the label applies from the compounding date, not from the date you receive it. Check the printed date carefully, because a vial that sat in transit or at the pharmacy for weeks before reaching you may have less time remaining than you expect.
How do I know if my refrigerator is cold enough?
Place a simple refrigerator thermometer on the same shelf where you store your medication. Check it after the door has been closed for at least two hours. The reading should be between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). If it consistently reads above 46 degrees Fahrenheit, adjust your refrigerator settings. If it reads at or near 32 degrees Fahrenheit, move the medication away from the cooling element to prevent accidental freezing.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based storage guides, proven protocols, dosing calculators, and a community of thousands who have navigated these exact questions. SeekPeptides members access detailed protocol builders, weight-based calculators, and expert-reviewed guidance that accounts for the nuances most resources overlook.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your refrigerator stay cold, and your protocols stay consistent.