Feb 12, 2026

Oral tirzepatide and injectable tirzepatide look like the same molecule on paper. Same amino acid sequence. Same dual GIP and GLP-1 receptor activity. Same promise of appetite suppression and metabolic improvement. But the outcomes? They could not be more different.
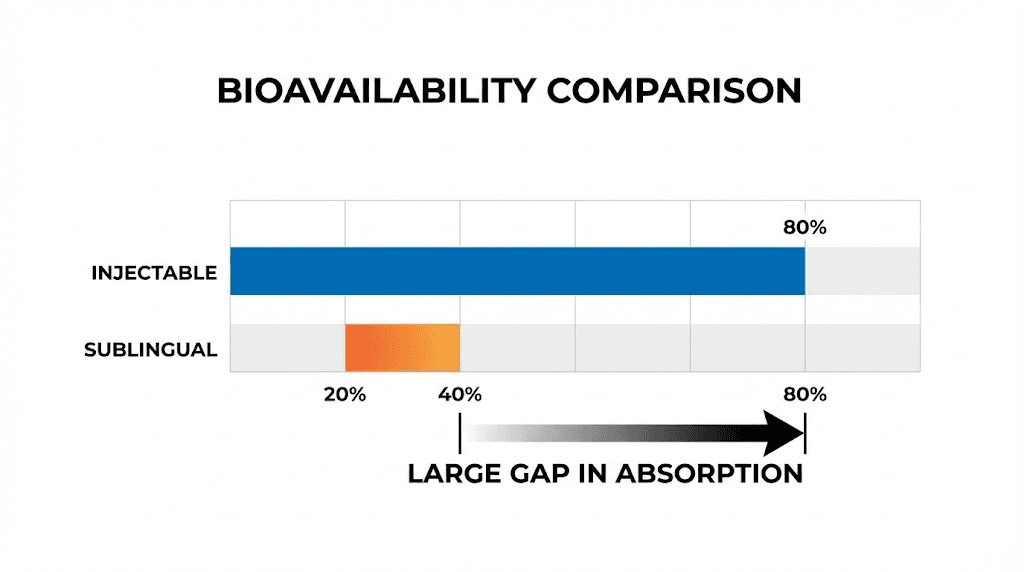
That gap between identical molecules and wildly different results comes down to one thing. Delivery. How tirzepatide enters your bloodstream determines how much of it actually reaches the receptors that matter, how consistently it works from dose to dose, and whether you get the weight loss and blood sugar control the clinical trials promised. Injectable tirzepatide delivers roughly 80% of its dose into circulation. Sublingual drops and oral formulations? Estimates range from 20% to 40%, and that is a generous estimate that varies dramatically from person to person.
This matters more than most people realize. Choosing between oral and injectable tirzepatide is not just a preference question about needles versus drops. It is a decision that affects your results, your timeline, your dosing protocol, and your budget. Get it wrong and you spend months wondering why the peptide is not working when the real problem was never the molecule itself.
This guide breaks down everything researchers need to know about both delivery methods. You will find the pharmacokinetic data, the absorption science, the real-world protocol differences, and a clear framework for deciding which approach matches your goals. Whether you are considering tirzepatide drops for convenience or sticking with the proven injectable route, the data here will help you make an informed decision rather than an expensive guess.
How tirzepatide works regardless of delivery method
Before comparing delivery methods, it helps to understand what tirzepatide actually does once it reaches your bloodstream. The molecule is a 39-amino-acid peptide that acts as a dual agonist, activating both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor simultaneously. This dual mechanism is what separates tirzepatide from single-receptor drugs like semaglutide, which only targets GLP-1.
GIP receptor activation enhances insulin sensitivity and promotes fat metabolism. GLP-1 receptor activation slows gastric emptying, reduces appetite, and improves glucose-dependent insulin secretion. Together, these two pathways create a synergistic effect that neither pathway achieves alone.
Clinical trials demonstrate this clearly. At the 15mg dose, injectable tirzepatide produced median weight loss of 15% of body weight. That is significantly more than semaglutide alone typically achieves. The dual mechanism is the reason.
Here is what matters for the oral versus injection debate. None of this works unless enough tirzepatide reaches your bloodstream in active form. The molecule must survive digestion, cross biological membranes, avoid liver metabolism, and arrive at GIP and GLP-1 receptors in sufficient concentration. How well each delivery method accomplishes this determines everything.
Think of it like this. You can have the most powerful engine in the world, but if the fuel line only delivers half the gasoline, you are not getting full performance. Delivery method is the fuel line. The molecule is the engine.
Injectable tirzepatide: the clinical gold standard
Injectable tirzepatide is the only FDA-approved form of this molecule. It is available as Mounjaro for type 2 diabetes and Zepbound for weight management. Both are administered once weekly via subcutaneous injection.
The pharmacokinetic profile is well-established through extensive clinical trials. Subcutaneous tirzepatide has an absolute bioavailability of approximately 80%. That means for every milligram you inject, roughly 0.8mg reaches systemic circulation in active form. The time to peak plasma concentration ranges from 8 to 72 hours after injection, with most people reaching peak levels around 24 to 48 hours.
The half-life sits at approximately 5 days. This long half-life is what makes once-weekly dosing possible. After four weeks of consistent weekly injections, you reach steady-state plasma concentrations, meaning the amount of tirzepatide in your system stabilizes at a predictable level.
FDA-approved dosing protocol
The standard tirzepatide dosing protocol follows a gradual escalation schedule designed to minimize gastrointestinal side effects while building toward therapeutic levels.
You start at 2.5mg weekly for four weeks. This is a tolerability dose, not a therapeutic dose. Most people do not see significant weight loss during this phase. Then you increase to 5mg weekly for at least four weeks. This is where therapeutic effects typically begin. From there, your provider may increase the dose in 2.5mg increments every four weeks based on response and tolerability. The maximum approved dose is 15mg weekly.
Each dose level has a corresponding dosage chart that accounts for vial concentration and injection volume. If you are using compounded tirzepatide, the compound dosage charts differ based on concentration. A 5mg/mL vial requires different injection volumes than a 20mg/mL formulation.
For researchers working with specific vial sizes, you can reference the 10mg dosage chart or the 30mg dosage chart depending on what you have available. The math matters. Getting the conversion from milligrams to units correct prevents underdosing and overdosing.
Injection sites and technique
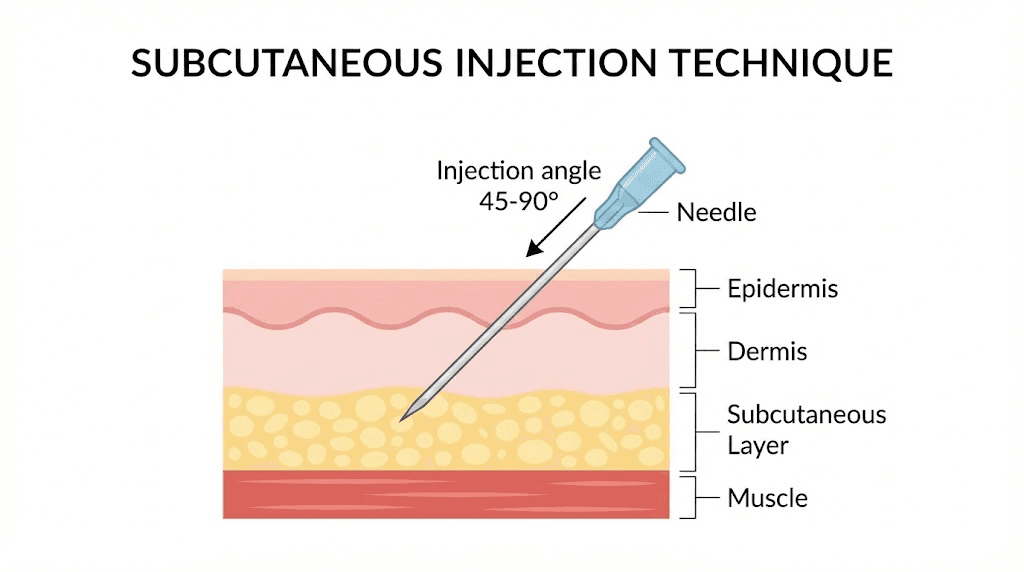
Subcutaneous injection means the needle goes into the fatty tissue just beneath the skin. The three recommended injection sites are the abdomen (at least two inches from the belly button), the front of the thigh, and the upper arm. Research shows similar absorption across all three sites, though many people find abdominal injections slightly less painful.
Proper technique matters. You pinch the skin to create a fold of fatty tissue, insert the needle at a 90-degree angle, and hold it in place for about 10 seconds after injecting. Rotate your injection sites each week to prevent irritation and lipodystrophy. If you are new to peptide injections, our complete injection guide covers the process step by step.
The needle itself is small. We are talking about a 29 to 31 gauge needle, roughly the width of a human hair. Most people describe the sensation as a quick pinch, nothing more. The injection pen devices used with brand-name tirzepatide make the process even simpler since the needle is hidden and the dose is pre-measured.
Storage and stability of injectable tirzepatide
Injectable tirzepatide requires refrigeration. Unopened vials and pens should be stored between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). Once you start using a pen, it can stay at room temperature (up to 86 degrees Fahrenheit or 30 degrees Celsius) for up to 21 days.
Proper storage directly affects potency. If you are wondering about specifics, our guides on tirzepatide refrigeration requirements and how long tirzepatide lasts in the fridge cover every scenario. Compounded formulations have their own stability profiles, and knowing how long compounded tirzepatide can be out of the fridge prevents accidental degradation.
Oral tirzepatide: what exists today and what does not
Here is the critical distinction that many sources blur. There is no FDA-approved oral tirzepatide formulation. None. Not tablets, not drops, not troches, not sublingual strips. The only FDA-approved tirzepatide products are injectable Mounjaro and Zepbound.
What does exist is compounded oral tirzepatide. Compounding pharmacies create sublingual drops, orally dissolving tablets (ODTs), and troches that contain tirzepatide. These are not FDA-approved products. They have not been through the rigorous clinical trial process that injectable tirzepatide completed. This distinction matters enormously.
Sublingual drops and how they work
Sublingual delivery places the medication under the tongue, where it dissolves and absorbs through the mucous membrane directly into the bloodstream. This route bypasses the digestive system, which is critical for a peptide like tirzepatide that would be destroyed by stomach acid and digestive enzymes if swallowed.
The area under the tongue has a dense network of blood vessels, making it one of the most permeable mucous membranes in the body. Medications absorbed sublingually bypass first-pass liver metabolism, meaning more of the active compound reaches systemic circulation compared to something swallowed and processed through the gut and liver.
For a deeper look at how sublingual peptide delivery works across different molecules, our sublingual peptides guide covers the science in detail. The tirzepatide drops guide focuses specifically on this formulation.
In theory, sublingual tirzepatide should work. The molecule reaches the bloodstream and activates the same GIP and GLP-1 receptors.
In practice? The results are inconsistent.
The bioavailability problem
This is where the oral versus injection comparison gets uncomfortable for oral advocates. Injectable tirzepatide delivers approximately 80% bioavailability. Sublingual tirzepatide? Estimates range from 20% to 40%, and those are generous figures that vary wildly between individuals.
Several factors cause this variability. Saliva dilutes the concentration under the tongue. If you swallow even a small amount during the absorption window, that portion gets destroyed by digestive enzymes. The thickness of the sublingual mucosa varies between people. Food, drink, smoking, and even pH levels in the mouth affect absorption.
What this means practically is that two people taking the exact same sublingual tirzepatide dose can have dramatically different blood levels. One might absorb 35% and get meaningful appetite suppression. Another might absorb 15% and feel nothing. This inconsistency is the fundamental weakness of oral delivery for peptides.
Orally dissolving tablets and troches
Some compounding pharmacies offer tirzepatide as orally dissolving tablets (ODTs) or troches. These are designed to dissolve in the mouth and absorb through the oral mucosa, similar to sublingual drops but in solid form.
Empower Pharmacy, Strive Pharmacy, and others have developed proprietary formulations designed to enhance sublingual absorption. Strive Pharmacy, for example, markets their tirzepatide sublingual drops under the name SubMagna, claiming enhanced absorption technology.
Despite marketing claims, no published clinical data demonstrates that any compounded oral tirzepatide formulation achieves bioavailability comparable to injection. The absorption challenge is inherent to the molecule itself, not just the delivery format. Tirzepatide is a 39-amino-acid peptide with a molecular weight of approximately 4,810 daltons. Molecules this large face fundamental barriers to transmucosal absorption regardless of the formulation technology used.
Dosing differences between oral and injectable
Because of lower bioavailability, oral tirzepatide typically requires higher doses than injectable to achieve similar blood levels. While injectable dosing starts at 2.5mg weekly, oral formulations often use daily dosing at varied concentrations.
This creates a compounding problem. Literally. Higher doses mean more raw material per prescription, which increases cost. Daily dosing means more frequent administration, which introduces more opportunities for inconsistent absorption. And without standardized clinical data, finding the right oral dose often becomes a trial-and-error process.
If you are working with compounded formulations and need help with the math, the tirzepatide dosage chart in units and the dosage chart in milliliters can help you convert between measurement systems. The reconstitution chart is essential if you are working with lyophilized powder that needs to be mixed before use.
Head-to-head comparison: oral vs injectable tirzepatide
Let us put the key differences side by side so you can see exactly where each delivery method stands.
Factor | Injectable tirzepatide | Oral/sublingual tirzepatide |
|---|---|---|
FDA approval | Yes (Mounjaro, Zepbound) | No |
Bioavailability | ~80% | Estimated 20-40% |
Dosing frequency | Once weekly | Daily (typically) |
Dose consistency | Highly consistent | Variable between doses |
Clinical trial data | Extensive (SURPASS, SURMOUNT) | None published |
Starting dose | 2.5mg weekly | Varies by pharmacy |
Maximum dose | 15mg weekly | Not standardized |
Weight loss evidence | Up to 15% body weight (15mg) | Anecdotal only |
Needle required | Yes | No |
Storage | Refrigeration required | Varies by formulation |
Cost | Higher (brand) / Moderate (compounded) | Often lower per dose |
The table makes it clear. Injectable tirzepatide wins on every clinical metric. But oral tirzepatide wins on convenience and needle avoidance. The question is which factors matter most for your specific situation.
Effectiveness and weight loss outcomes
Injectable tirzepatide has mountains of clinical data behind it. The SURPASS trials demonstrated significant HbA1c reduction in type 2 diabetes patients. The SURMOUNT trials showed weight loss of up to 22.5% of body weight at the highest doses in people with obesity.
Oral tirzepatide has no published clinical trial data. Zero. Every effectiveness claim for oral tirzepatide is either extrapolated from the injectable data (assuming comparable blood levels) or based on individual anecdotal reports.
This does not mean oral tirzepatide does not work. Some people report meaningful appetite suppression and weight loss with sublingual formulations. But some people also report no effect whatsoever. Without controlled trials, we cannot separate the responders from the non-responders in any statistically meaningful way. We cannot know the true response rate.
For context on what typical weight loss timelines look like with the proven injectable form, see our guides on how fast tirzepatide works and how long tirzepatide takes to produce results. These timelines are based on injection data. Oral timelines remain undefined.
Side effect profiles
Both delivery methods produce similar gastrointestinal side effects because the side effects come from the mechanism of action, not the delivery route. When tirzepatide activates GLP-1 receptors, it slows gastric emptying. This causes nausea, diarrhea, constipation, and abdominal discomfort regardless of whether the molecule entered through a needle or through sublingual absorption.
The most common side effects include nausea (occurring in up to 30% of users at higher doses), diarrhea, constipation, decreased appetite, vomiting, and abdominal pain. These effects are typically worst during the first few weeks at a new dose level and tend to improve over time.
Injectable tirzepatide adds the possibility of injection-site reactions, including redness, itching, or mild pain at the injection site. These are generally mild and transient.
Oral tirzepatide, particularly sublingual formulations, can cause a bitter or metallic taste in the mouth. Some people experience mild oral irritation from holding the drops under the tongue for the recommended absorption period.
If side effects are a concern, understanding what to expect helps you manage them. Our guides cover specific issues like tirzepatide and fatigue, tirzepatide and anxiety, and troubleshooting when tirzepatide is not producing results.
Convenience and lifestyle factors
This is where oral tirzepatide has its strongest argument. No needles. No injection supplies. No refrigeration concerns with some formulations. No need to find a private spot to inject. For people with genuine needle phobia, the convenience factor is significant.
Daily dosing is both a pro and a con. Some people prefer daily consistency over remembering a weekly injection. Others find daily dosing inconvenient, especially the sublingual requirement to hold drops under the tongue for 60 to 90 seconds without swallowing.
Travel with oral tirzepatide is simpler. No syringes to explain at airport security. No cold chain to maintain. No sharps disposal to worry about. For frequent travelers, this convenience is real and meaningful.
But convenience means nothing if the medication is not working. And the inconsistent bioavailability of oral formulations means some percentage of users are paying for convenience while getting subtherapeutic blood levels.
The science of peptide absorption: why delivery method matters so much
To understand why oral tirzepatide faces such steep challenges, you need to understand what happens to a peptide molecule in the human body.
Peptides are chains of amino acids held together by peptide bonds. Your body has evolved incredibly efficient systems for breaking peptide bonds. This is digestion. Your stomach produces hydrochloric acid with a pH between 1.5 and 3.5, specifically designed to denature proteins and peptides. Your small intestine releases proteolytic enzymes, including trypsin, chymotrypsin, and carboxypeptidase, that systematically cleave peptide bonds into individual amino acids for absorption.
This is excellent for nutrition. It is terrible for peptide drug delivery.
The gastrointestinal destruction pathway
If you swallowed tirzepatide as a regular pill, here is what would happen. The stomach acid would begin denaturing the three-dimensional structure of the molecule within minutes. In the small intestine, proteolytic enzymes would cleave the 39-amino-acid chain into fragments too small to activate GIP or GLP-1 receptors. Very little, if any, intact tirzepatide would reach the intestinal wall. And even the tiny amount that survived would face the intestinal epithelial barrier, a wall of tightly packed cells that peptides of this molecular weight cannot easily cross.
Then comes first-pass metabolism. Anything that does cross the intestinal wall enters the portal vein and goes directly to the liver before reaching systemic circulation. The liver metabolizes a significant percentage of most drugs during this first pass.
The result? Oral bioavailability for an unprotected peptide of tirzepatide size would likely be less than 1%. Essentially zero therapeutic value.
How sublingual delivery tries to solve this
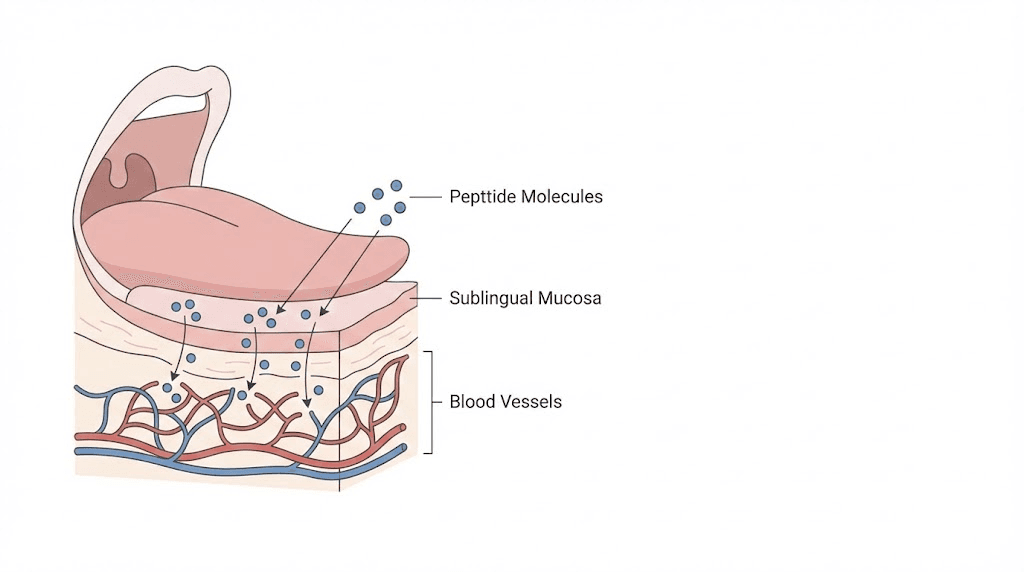
Sublingual delivery bypasses the stomach entirely. The medication absorbs through the mucous membrane under the tongue directly into the sublingual vein, which drains into the internal jugular vein and then into systemic circulation. No stomach acid. No digestive enzymes. No first-pass liver metabolism.
Sounds perfect. But there are limits.
The sublingual mucosa is not designed for large molecule absorption. Small molecules like nitroglycerin (molecular weight 227 daltons) absorb sublingually with excellent efficiency. Tirzepatide, at approximately 4,810 daltons, is more than 20 times larger. The mucous membrane acts as a size filter, and larger molecules pass through much less efficiently.
Absorption also requires time. The drops must stay under the tongue long enough for significant quantities to cross the membrane. Swallowing, talking, or moving the tongue disrupts the process. Even the act of saliva production dilutes the concentration and reduces absorption efficiency.
This is why the bioavailability estimates for sublingual tirzepatide range so widely. Under perfect conditions with a cooperative patient and ideal formulation, you might achieve 30 to 40% absorption. Under real-world conditions? Often much less.
Emerging delivery technologies
Research is actively pursuing better oral peptide delivery methods. One promising approach uses milk-derived small extracellular vesicles as delivery vehicles. These natural nanoparticles can encapsulate tirzepatide and protect it during transit through the gastrointestinal tract. Early preclinical data suggests this could dramatically improve oral bioavailability, though human trials are years away.
Another approach comes from Eli Lilly themselves. Rather than trying to deliver tirzepatide orally, they developed an entirely different molecule. Orforglipron is a small-molecule GLP-1 receptor agonist designed from the ground up for oral delivery. Unlike tirzepatide, orforglipron is not a peptide. It is a small molecule with a molecular weight that allows efficient oral absorption. Phase 3 trials showed weight loss of up to 11.2% at the highest dose over 72 weeks. The FDA is currently reviewing the drug, with approval potentially coming soon.
However, orforglipron only activates GLP-1 receptors. It does not have the dual GIP/GLP-1 activity that makes tirzepatide uniquely effective. Understanding this distinction is important, and our comparison of GLP-1 medications and their differences provides additional context. The oral tirzepatide complete guide also covers the pipeline of upcoming oral formulations in detail.
Dosing protocols: injectable versus oral
Getting the dose right is everything. Too little tirzepatide and you get no therapeutic benefit. Too much and the side effects become unbearable. The dosing protocol you follow depends entirely on which delivery method you choose.
Injectable dosing: the proven escalation
The FDA-approved injectable dosing schedule has been validated across thousands of clinical trial participants.
Weeks 1-4: 2.5mg once weekly. This is a tolerability phase. Your body adjusts to the GIP and GLP-1 receptor activation. Expect mild nausea. Do not expect significant weight loss.
Weeks 5-8: 5mg once weekly. Therapeutic effects begin. Appetite suppression becomes noticeable. Most people start losing weight during this phase.
Weeks 9-12: 7.5mg once weekly (if needed). Increased effect on both appetite and blood sugar control.
Weeks 13-16: 10mg once weekly (if needed). Significant appetite suppression. Most people report fundamentally changed relationships with food at this dose.
Weeks 17+: 12.5mg or 15mg once weekly (maximum). Reserved for those who need additional effect. Side effects are more common at these higher doses.
The key principle is slow escalation. Each dose level needs at least four weeks before increasing. This gives your GI system time to adapt to the slowed gastric emptying. Rushing the escalation is the most common mistake researchers make, and it leads to severe nausea that could have been avoided.
For detailed escalation charts, the tirzepatide dosing for weight loss in units guide provides exact unit conversions at every dose level. If you are using a specific pharmacy formulation, charts for Empower, Southend, Olympia, and Mochi pharmacies are available.
Oral dosing: the uncharted territory
No standardized oral tirzepatide dosing protocol exists because no clinical trials have established one. What exists are compounding pharmacy guidelines and practitioner experience, neither of which has been validated through controlled research.
Most compounded sublingual protocols use daily dosing rather than weekly. The logic is straightforward: lower bioavailability means less medication reaches the bloodstream per dose, so more frequent dosing compensates. But this compensation is imprecise.
A typical compounded sublingual protocol might look like this. Start with a low daily dose. Hold the drops under the tongue for 60 to 90 seconds without swallowing. Take on an empty stomach, ideally first thing in the morning. Wait at least 30 minutes before eating or drinking. Gradually increase the dose based on response and tolerability.
The challenge? Without knowing your individual bioavailability, you cannot know if you are getting a therapeutic dose. Some people respond well at moderate oral doses. Others need to increase significantly before seeing any effect. And some never achieve consistent enough absorption for meaningful results.
The tirzepatide sublingual dosage chart provides a reference point for common compounded formulations, though individual results will vary substantially.
Microdosing approaches
Some researchers use microdosing tirzepatide protocols with either delivery method. Microdosing involves using sub-therapeutic doses, often to assess individual sensitivity before escalating. This approach can help identify whether oral absorption is adequate before committing to a full protocol.
With injectable microdosing, the absorbed dose is predictable. You know exactly how much tirzepatide is reaching your system. With oral microdosing, the absorbed dose is a guess. A useful guess, but a guess nonetheless.
Who should choose injectable tirzepatide
Injectable tirzepatide is the right choice for most people. Not because needles are fun, but because the evidence is overwhelming and the results are predictable.
Choose injectable if you want proven results
If your primary goal is maximum weight loss with the highest confidence of success, injectable tirzepatide is the answer. The clinical trial data is extensive and clear. At 5mg weekly, average weight loss reaches approximately 7.5% of body weight. At 10mg, it is around 10%. At 15mg, it is up to 15% or more.
These are not ceiling numbers. Many individual participants in the SURMOUNT trials lost significantly more than the averages suggest. Some lost over 25% of their starting body weight. The key point is that these results are documented, reproducible, and statistically significant.
Real-world results often mirror the trials. Our tirzepatide before and after guide and the detailed weight loss results compilation show what researchers are achieving outside of clinical settings. For gender-specific outcomes, the men before and after tirzepatide guide offers additional perspective.
Choose injectable if you want dose precision
With injectable tirzepatide, what you inject is essentially what you get (minus the 20% that the body does not absorb). The unit conversions are exact. Whether you are measuring 5mg in units, 7.5mg in units, or converting from units back to milligrams, the math is reliable.
This precision matters for titration. When you increase from 5mg to 7.5mg, you know that your actual absorbed dose increased by approximately 2mg (accounting for the 80% bioavailability). You can correlate dose changes with effect changes and adjust accordingly.
With oral tirzepatide, the same dose can produce different blood levels on different days. Tuesday absorption might be 30%. Friday absorption might be 18%. This unpredictability makes systematic dose optimization nearly impossible.
Choose injectable if you have metabolic conditions
For type 2 diabetes management specifically, injectable tirzepatide is the only responsible choice. The HbA1c reductions demonstrated in the SURPASS trials (up to 2.4% reduction) were achieved with injectable administration under controlled conditions. Using an unproven oral formulation for blood sugar management introduces unnecessary risk.
The same applies if you are switching between GLP-1 medications. Dose equivalence calculations between semaglutide and tirzepatide are based entirely on injectable pharmacokinetics. Adding oral bioavailability uncertainty to a medication switch creates too many unknown variables.
Who might consider oral tirzepatide
Despite the clear advantages of injectable delivery, some situations exist where oral tirzepatide becomes a reasonable consideration.
Severe needle phobia
True needle phobia (trypanophobia) is a recognized medical condition that affects approximately 10% of adults. Not just discomfort with needles, but genuine anxiety or panic responses that prevent injection entirely. If needle phobia is blocking you from using a medication that could significantly improve your health, an imperfect oral alternative may be better than no treatment at all.
That said, most people who think they have severe needle phobia can overcome it with proper desensitization techniques and the right injection supplies. Modern injection pens hide the needle entirely. You press the pen against your skin, push a button, and the injection happens without you ever seeing the needle. Many people who were needle-averse report that the anticipation was far worse than the reality.
Inability to self-inject
Some people physically cannot self-inject due to conditions like severe arthritis, tremor, limited dexterity, or visual impairment. If no one is available to assist with weekly injections, oral delivery provides an accessible alternative.
Transition tool
Some practitioners use oral tirzepatide as a bridge. A patient starts with oral drops to experience the appetite suppression and confirm they tolerate the medication. Once they see benefits and gain confidence, they transition to injectable for better results. This approach is not evidence-based, but it can help anxious patients take the first step.
Cost considerations
Brand-name injectable tirzepatide (Mounjaro, Zepbound) costs over $1,000 per month without insurance. Compounded injectable tirzepatide costs significantly less but still represents a substantial monthly expense. Some compounded oral formulations cost even less than compounded injectables, making them attractive for budget-conscious researchers.
But cheaper means nothing if it does not work. A $300 monthly oral formulation that delivers inconsistent results costs more than a $500 monthly injectable that produces reliable outcomes. Cost per result matters more than cost per dose.
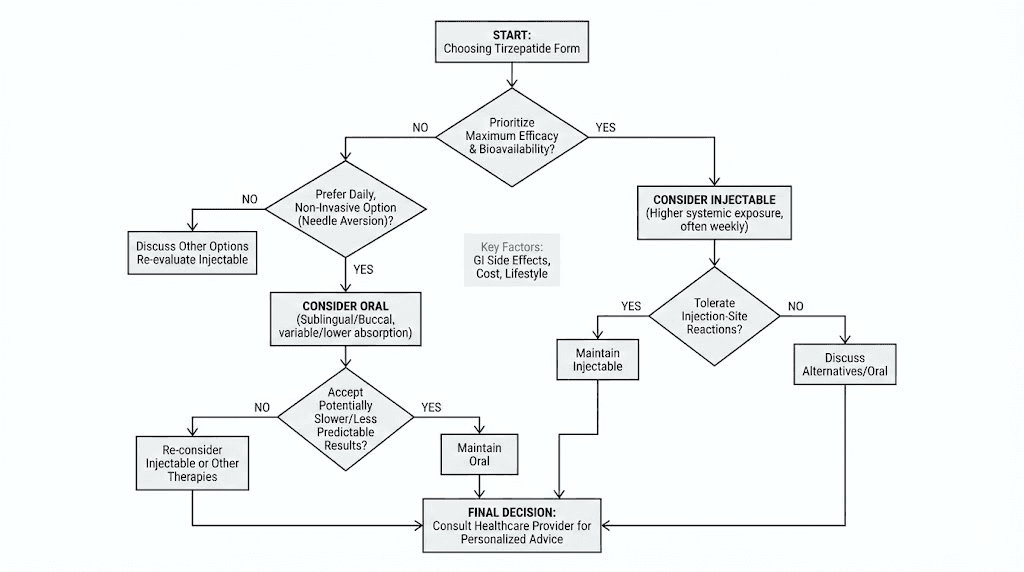
Reconstitution and preparation differences
How you prepare each form of tirzepatide affects both the experience and the effectiveness.
Preparing injectable tirzepatide
Brand-name tirzepatide (Mounjaro, Zepbound) comes in pre-filled injection pens. No preparation required. You select your dose, attach the needle, inject, and dispose of the needle. Simple.
Compounded injectable tirzepatide typically comes as lyophilized (freeze-dried) powder that requires reconstitution with bacteriostatic water. The reconstitution process is straightforward but must be done correctly. You add a precise volume of bacteriostatic water to the vial, gently swirl (never shake) until dissolved, and then draw up your dose with an insulin syringe.
The amount of bacteriostatic water you add determines the concentration, which determines how many units equal your target dose. The reconstituting tirzepatide chart and reconstitution chart PDF make these calculations easy. If you are new to peptide reconstitution in general, the peptide reconstitution calculator handles the math for you.
Preparing oral tirzepatide
Compounded sublingual drops typically come ready to use. The pharmacy provides a bottle with a measured dropper. You draw up the prescribed volume, place it under your tongue, and hold for the absorption period.
ODTs and troches are also ready to use. You place the tablet under your tongue and allow it to dissolve completely without chewing or swallowing.
The preparation is simpler for oral forms. No reconstitution. No syringes. No needles. No sharps disposal. This simplicity is genuinely appealing.
But simpler preparation does not mean better results. It means easier process. These are different things.
Combining oral and injectable tirzepatide: a bad idea
Some people wonder about using both delivery methods, perhaps injecting on some days and using sublingual drops on others. This is not recommended by any medical authority and creates significant dosing uncertainty.
The half-life of injectable tirzepatide is approximately 5 days. If you inject on Monday and then take sublingual drops on Thursday, the injected tirzepatide is still very much active in your system. Adding an oral dose with unpredictable absorption on top of residual injectable tirzepatide creates uncontrollable blood levels.
Stick with one delivery method. If you choose injectable, stay injectable. If you choose oral, stay oral. Mixing introduces variables that make dose management impossible.
What the research pipeline looks like
The oral GLP-1 space is evolving rapidly. Understanding what is coming helps contextualize the current oral versus injection debate.
Orforglipron: Eli Lilly oral GLP-1
The most significant development is orforglipron, Eli Lilly oral GLP-1 receptor agonist. Unlike compounded oral tirzepatide, orforglipron was designed from the ground up as an oral medication. It is not a peptide. It is a small molecule that fits naturally into an oral delivery format.
Phase 3 results from the ATTAIN trials show meaningful weight loss. At the 36mg dose, participants lost an average of 11.2% body weight over 72 weeks. About 18% of participants lost 20% or more. Side effects were consistent with the GLP-1 class, primarily mild to moderate nausea and diarrhea.
The FDA is currently reviewing the new drug application. If approved, orforglipron would be the first oral medication that directly competes with injectable GLP-1 therapies on efficacy.
However, orforglipron is GLP-1 only. It does not have the GIP receptor activity that gives tirzepatide its edge. So it may not match injectable tirzepatide results, even as a purpose-built oral medication.
Danuglipron: the discontinued competitor
Pfizer was developing danuglipron, another small-molecule oral GLP-1 agonist. Despite initially promising results, Pfizer discontinued development due to unacceptable side effect profiles at effective doses. This illustrates how difficult it is to create an oral medication that matches injectable GLP-1 performance.
Advanced delivery technologies
Research into novel peptide delivery systems continues across the pharmaceutical industry. Milk-derived extracellular vesicles, nanoparticle encapsulation, absorption enhancers, and enteric coating technologies all show promise in preclinical models.
But promise in the lab and performance in humans are separated by years of development and billions of dollars in investment. For the foreseeable future, injectable tirzepatide remains the most effective delivery method for this specific molecule.
The GLP-1 patches represent another alternative delivery approach worth watching. Transdermal delivery could potentially combine the convenience of needleless administration with more consistent absorption than sublingual delivery.
Practical considerations that affect your decision
Beyond the science, real-world factors influence which delivery method works best for your life.
Travel logistics
Traveling with injectable tirzepatide requires a few extra steps. You need a cooler or insulated bag for refrigerated vials. You need syringes, alcohol swabs, and a sharps container. TSA allows insulin syringes and prescription medications, but having your prescription label visible prevents delays.
Oral tirzepatide travels in a small bottle. No temperature concerns (for most formulations). No sharps. No explanations needed. For frequent travelers, this convenience is substantial.
Privacy and stigma
Unfortunately, weight loss medications still carry social stigma. Some people prefer the discretion of oral drops over injectable preparations. Drops under the tongue look like any supplement. Syringes raise questions.
While this should not be a factor in medical decisions, it is a reality for many people. If privacy concerns are preventing you from starting treatment at all, an oral option might lower that barrier.
Lifestyle compatibility
Weekly injections require minimal lifestyle adjustment. Pick a day. Inject. Done for the week.
Daily sublingual dosing requires more routine integration. Empty stomach each morning. Hold drops for 60 to 90 seconds. Wait before eating. For people with irregular schedules, this daily requirement can be harder to maintain consistently than a once-weekly injection.
Diet and nutrition interactions
Regardless of delivery method, tirzepatide affects appetite and digestion. Following an appropriate tirzepatide diet plan maximizes results and minimizes side effects. Knowing which foods to avoid on tirzepatide prevents unnecessary GI distress.
Oral tirzepatide adds an additional dietary restriction. You must take it on an empty stomach and wait before eating. This fasting window complicates morning routines for people who prefer early breakfasts or who take other medications with food.
Supplementation with tirzepatide: oral and injectable
Many researchers combine tirzepatide with supportive supplements to enhance results or offset side effects. The delivery method of tirzepatide does not change which supplements are beneficial, but it does affect timing.
B12 and tirzepatide
GLP-1 receptor agonists can reduce vitamin B12 absorption over time. Supplementing with B12 is common and well-supported. Tirzepatide with B12 is one of the most popular combinations. Some compounding pharmacies even offer tirzepatide pre-mixed with B12.
With injectable tirzepatide, B12 can be taken at any time since there is no fasting requirement. With oral tirzepatide, B12 supplements should be taken separately from the sublingual dose to avoid interfering with absorption.
Glycine and tirzepatide
Glycine supplementation with tirzepatide has gained attention for its potential to support lean mass preservation during rapid weight loss. Glycine is an amino acid that supports collagen synthesis, muscle maintenance, and sleep quality.
Niacinamide and tirzepatide
Niacinamide (vitamin B3) with tirzepatide is another combination being explored. Niacinamide supports NAD+ production, which plays roles in cellular energy metabolism and may complement the metabolic effects of tirzepatide.
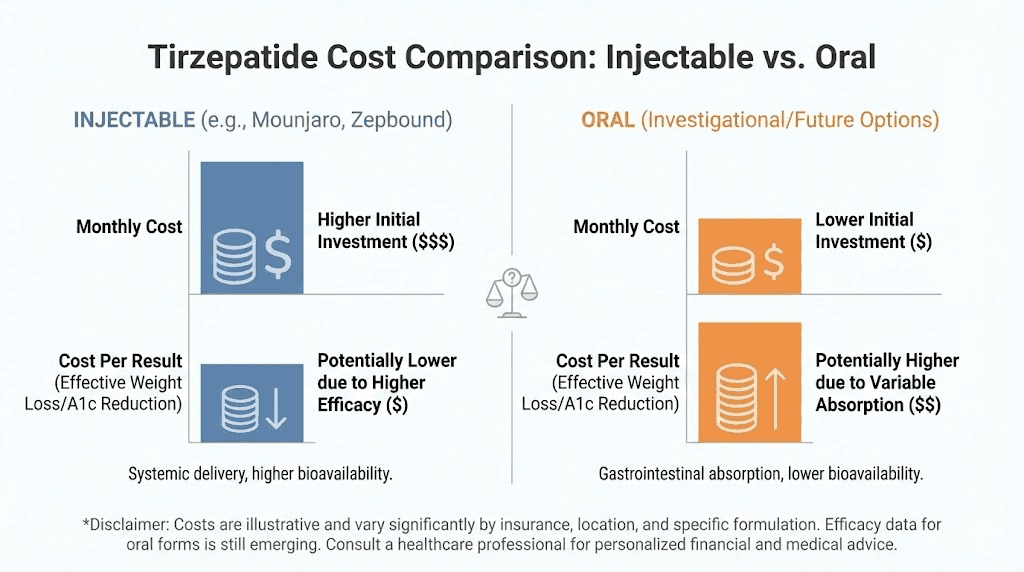
Cost analysis: oral versus injectable
Understanding the true cost requires looking beyond the sticker price.
Brand-name injectable costs
Mounjaro and Zepbound carry list prices exceeding $1,000 per month. With insurance or manufacturer savings cards, the cost can drop significantly. Without any coverage, brand-name injectable tirzepatide is prohibitively expensive for most people.
Compounded injectable costs
Compounded injectable tirzepatide through a compounding pharmacy typically costs between $200 and $600 per month depending on dose and pharmacy. This requires a valid prescription and is subject to compounding regulations that vary by state.
For researchers looking for cost-effective compounded options, our guide on affordable compounded tirzepatide covers strategies for reducing costs without compromising quality. Multiple pharmacy options exist, including Peptide Sciences and Lavender Sky.
Compounded oral costs
Compounded oral tirzepatide often costs less than compounded injectables, typically ranging from $150 to $400 per month. However, if higher doses are needed to compensate for lower bioavailability, the per-milligram cost advantage can shrink or disappear entirely.
The real cost calculation
Cost per month is the wrong metric. Cost per result is what matters.
If injectable tirzepatide costs $400/month and produces 2% body weight loss per month, that is $200 per percentage point lost. If oral tirzepatide costs $250/month but only produces 0.5% body weight loss per month (due to lower bioavailability), that is $500 per percentage point lost.
The cheaper option by sticker price became the more expensive option by outcomes. Always calculate the cost per result, not just the cost per dose.
Safety considerations specific to each delivery method
Both delivery methods carry the inherent safety profile of tirzepatide as a molecule, but each route adds its own specific considerations.
Injectable safety specifics
Injection site reactions occur in a small percentage of users. These include redness, swelling, itching, or mild pain at the injection site. Proper rotation of injection sites minimizes this risk.
Sterility is critical. Using contaminated needles or failing to clean the injection site can introduce bacteria. Always use new, sterile needles for each injection. Clean the injection site with alcohol before injecting. Never share needles or vials.
Needle stick injuries, while rare with proper technique, are possible. Proper sharps disposal in a designated container prevents accidental sticks after injection.
The peptide safety and risks guide covers injection safety in comprehensive detail. For storage-related safety, understanding how to store peptides after reconstitution prevents degradation that could affect both safety and efficacy.
Oral safety specifics
Compounded oral tirzepatide carries unique safety concerns. The compounding process itself is less regulated than FDA-approved manufacturing. Potency, purity, and sterility can vary between batches and between pharmacies.
The FDA has issued warnings about compounded tirzepatide products, noting that some may lack proper quality controls. When choosing a compounding pharmacy, verify that they follow USP compounding standards and undergo regular third-party testing.
Sublingual absorption variability creates a hidden safety issue. If you normally absorb 20% of your oral dose and then one day absorb 40% (perhaps due to oral pH changes, a small cut under the tongue, or a different holding technique), you effectively received double your usual dose. This unpredictable dose variability introduces risks that injectable delivery does not have.
Making the switch: oral to injectable or vice versa
If you have been using one delivery method and want to switch to the other, the transition requires careful planning.
Switching from oral to injectable
This is the more common switch. People start with oral for convenience, find their results underwhelming, and transition to injectable for better outcomes.
There is no established equivalence between oral and injectable tirzepatide doses. You cannot simply take your oral daily dose, multiply by seven, and use that as your weekly injectable dose. The bioavailability difference means your actual absorbed oral dose is unknown.
The safest approach is to start the injectable escalation protocol from the beginning. 2.5mg weekly for four weeks, then escalate. Yes, this means potentially repeating a dose level you have technically already been on (orally). But starting conservatively with the injectable ensures you tolerate the higher and more consistent blood levels that injection provides.
Switching from injectable to oral
Less common, but it happens. Some people achieve their weight loss goals with injectable tirzepatide and want to switch to oral for maintenance convenience.
This transition carries risk. Your maintenance dose on injectable was producing reliable blood levels. Switching to oral with variable absorption means those blood levels become unpredictable. You might maintain your results. You might regain weight. There is no way to know in advance.
If making this switch, monitor your weight closely. If you see upward trends within the first four to six weeks of oral dosing, the oral formulation may not be delivering adequate blood levels for maintenance. The dose conversion charts available for injectable-to-injectable switches do not apply to oral transitions because bioavailability cannot be assumed.
What SeekPeptides members use most
Based on community data, the overwhelming majority of SeekPeptides members use injectable tirzepatide. The reasons align with everything discussed in this guide: better evidence, more consistent results, predictable dosing, and proven clinical trial backing.
That said, a meaningful minority of members use sublingual formulations, primarily those with needle phobia or travel-related constraints. Members who use oral tirzepatide report more variable results, which is consistent with the absorption science.
SeekPeptides provides resources for both delivery methods, including dosing calculators, protocol guides, and community discussions where members share real-world experiences with each approach. The peptide calculator helps with dose calculations regardless of which delivery method you choose.
How tirzepatide compares to other GLP-1 medications by delivery method
Tirzepatide is not the only GLP-1 receptor agonist available. Understanding how the oral versus injectable debate plays out across the class provides useful context.
Semaglutide: the oral comparison point
Semaglutide is the only GLP-1 receptor agonist with an FDA-approved oral formulation (Rybelsus). Oral semaglutide uses a specialized absorption enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate) that creates a temporary, localized pH change in the stomach, allowing semaglutide to absorb through the gastric lining.
Even with this specialized technology, oral semaglutide achieves lower blood levels than injectable semaglutide. In head-to-head comparisons, patients on injectable semaglutide or injectable tirzepatide lost significantly more weight than those on oral semaglutide.
This is instructive for the tirzepatide debate. If a pharmaceutical company with billions in R&D could not make oral semaglutide match injectable semaglutide, the likelihood that a compounding pharmacy can achieve that with tirzepatide is low.
For detailed comparisons between the two molecules, our semaglutide vs tirzepatide guide and the dosage comparison chart provide comprehensive analysis. You can also explore the oral semaglutide drops guide for parallel information on that molecule delivery debate.
GLP-1 medication class overview
The broader GLP-1 landscape includes medications that are only available as injections (dulaglutide, liraglutide) and the emerging oral small molecules (orforglipron). Within this landscape, tirzepatide stands out because of its dual GIP/GLP-1 mechanism, which produces superior weight loss compared to GLP-1-only medications.
Our guide on next-generation incretin peptides covers where the science is heading. And for those considering related weight loss approaches, the comparison between injectable versus oral peptides broadly applies the same bioavailability principles discussed here.
Troubleshooting common issues with each delivery method
Things do not always go perfectly. Here is how to address the most common problems with each approach.
Injectable troubleshooting
Problem: Medication not producing results. First verify your dose is correct. Use the dosage chart in units to double-check your calculations. If the dose is correct, ensure your tirzepatide has been stored properly. Degraded peptide looks the same but works less. If storage and dosing are confirmed correct, you may need to escalate to the next dose level.
Problem: Severe nausea. You likely escalated too quickly. Consider stepping back to your previous dose for an additional two to four weeks before trying to increase again. The weight loss dosing conversion chart helps you identify intermediate dose steps between standard levels.
Problem: Injection site reactions. Rotate sites more aggressively. Allow at least one inch between injection sites. Let the alcohol dry completely before injecting. Bring the medication to room temperature before injection to reduce pain.
Problem: Weight loss plateau. Plateaus are normal and expected, particularly at lower doses. The guide on why you are not losing weight on tirzepatide covers every common cause and solution.
Oral troubleshooting
Problem: No appetite suppression at all. This likely indicates inadequate absorption. Try extending the sublingual hold time to two minutes. Ensure you are truly on an empty stomach with no food or drink for at least 30 minutes before and after dosing. If these adjustments do not help, oral delivery may simply not achieve therapeutic levels for you.
Problem: Inconsistent results day to day. Variable absorption is the likely cause. Monitor factors that might affect absorption: oral pH, hydration level, time of day, position of drops under tongue. Keeping conditions as consistent as possible from day to day can help.
Problem: Bitter taste and oral irritation. This is common with sublingual tirzepatide. Rinsing your mouth with plain water after the absorption period can help. Do not rinse during the hold time as this dilutes the medication.
Long-term considerations for each delivery method
Weight management is a long-term commitment. Your chosen delivery method needs to be sustainable for months or years.
Injectable long-term use
Studies on injectable tirzepatide extend to 72 weeks (about 16 months) with consistent efficacy. Anecdotal evidence from early adopters extends even further. The weekly injection becomes routine quickly. Most long-term users report it takes less than two minutes of their week.
Long-term injectable use requires ongoing prescription management, pharmacy access, and proper storage infrastructure. For researchers asking how long to stay on GLP-1 medications, the answer depends on individual goals and ongoing consultation with a healthcare provider.
Oral long-term use
No long-term data exists for oral tirzepatide formulations. The daily dosing requirement creates more opportunities for missed doses over time. Absorption variability may compound over months, leading to less predictable long-term outcomes.
That said, if oral delivery works for you and produces consistent results, there is no specific reason it cannot be sustained long-term. The molecule is the same. Only the delivery differs. Monitor your progress closely and be willing to switch to injectable if oral results plateau or become inconsistent.
The verdict: which delivery method should you choose
After analyzing every angle, the evidence points clearly in one direction.
Choose injectable tirzepatide if:
You want the highest probability of significant weight loss
You value dose consistency and predictable results
You have type 2 diabetes or other metabolic conditions
You are comfortable with (or can learn to tolerate) weekly injections
You want to use an FDA-approved, clinically validated approach
Consider oral tirzepatide if:
You have genuine, severe needle phobia that cannot be overcome
Physical limitations prevent self-injection
Travel requirements make injectable logistics impossible
You want to test tolerance before committing to injections
You understand and accept the lower and variable bioavailability
For most researchers, injectable tirzepatide is the right choice. Not because it is perfect, but because it is proven. The 80% bioavailability, the extensive clinical trial data, the standardized dosing protocols, and the predictable outcomes make it the gold standard against which all alternatives are measured.
Oral tirzepatide is not the wrong choice in the right circumstances. But it should be chosen with clear eyes. You are accepting lower bioavailability, inconsistent absorption, no clinical trial validation, and potentially diminished results in exchange for convenience. For some people, that trade-off makes sense. For most, it does not.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, detailed dosing calculators, and a community of thousands who have navigated these exact questions. Whether you choose oral or injectable tirzepatide, having access to accurate information and experienced guidance makes the difference between guessing and succeeding.
Frequently asked questions
Is oral tirzepatide FDA-approved?
No. There is no FDA-approved oral tirzepatide formulation. Only injectable tirzepatide is FDA-approved, under the brand names Mounjaro (for type 2 diabetes) and Zepbound (for weight management). Oral and sublingual tirzepatide products are compounded formulations that have not been evaluated by the FDA for safety or effectiveness.
How much less effective is oral tirzepatide compared to injections?
Injectable tirzepatide has approximately 80% bioavailability, while sublingual formulations are estimated at 20 to 40%. This means the oral route may deliver less than half the active medication per dose. However, individual absorption varies significantly, and no clinical trials have directly compared the two delivery methods. Some people report good results with oral tirzepatide, while others experience minimal effect.
Can I switch from oral tirzepatide to injectable?
Yes, but start the injectable escalation protocol from the beginning (2.5mg weekly). Do not attempt to calculate an equivalent injectable dose based on your oral dose because the variable bioavailability of oral delivery makes equivalence calculations unreliable. Our tirzepatide dosing guide covers the standard escalation protocol.
Why do oral tirzepatide drops need to be held under the tongue?
Sublingual (under the tongue) delivery allows tirzepatide to absorb directly into the bloodstream through the mucous membrane, bypassing the digestive system. If swallowed, tirzepatide would be destroyed by stomach acid and digestive enzymes. The 60 to 90 second hold time maximizes the amount of medication that absorbs through the sublingual tissue before saliva dilutes it.
Is oral tirzepatide cheaper than injectable?
Often, yes, by sticker price. Compounded oral formulations typically cost $150 to $400 per month versus $200 to $600 for compounded injectables. However, if lower bioavailability means weaker results, the cost per result (weight lost per dollar spent) may actually favor injectable delivery. Always compare outcomes, not just price tags.
Will an FDA-approved oral tirzepatide ever be available?
Eli Lilly has not announced plans for an oral tirzepatide formulation. However, they are developing orforglipron, an oral small-molecule GLP-1 receptor agonist that does not require injection. Orforglipron is currently under FDA review. Unlike tirzepatide, orforglipron only activates GLP-1 receptors, not both GIP and GLP-1.
Do oral and injectable tirzepatide have the same side effects?
Mostly yes. Both delivery methods produce similar gastrointestinal side effects (nausea, diarrhea, constipation) because these result from the mechanism of action, not the delivery route. Injectable tirzepatide can additionally cause injection site reactions. Oral tirzepatide may cause bitter taste and mild oral irritation from the sublingual drops.
How do I know if oral tirzepatide is working for me?
Track appetite changes, weight trends, and overall well-being. With injectable tirzepatide, most people notice appetite suppression within the first two weeks at a therapeutic dose. If you have been on oral tirzepatide for four or more weeks at a reasonable dose and notice no change in appetite, hunger patterns, or weight, the oral formulation is likely not achieving therapeutic blood levels for you.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your delivery method stay effective, your bioavailability stay high, and your results stay consistent.