Feb 22, 2026

Experienced researchers know something about bulk retatrutide that first-time buyers almost always learn the hard way. The peptide itself is only half the equation. Sourcing, purity verification, proper storage, and protocol design matter just as much as the compound sitting inside the vial. Maybe more.
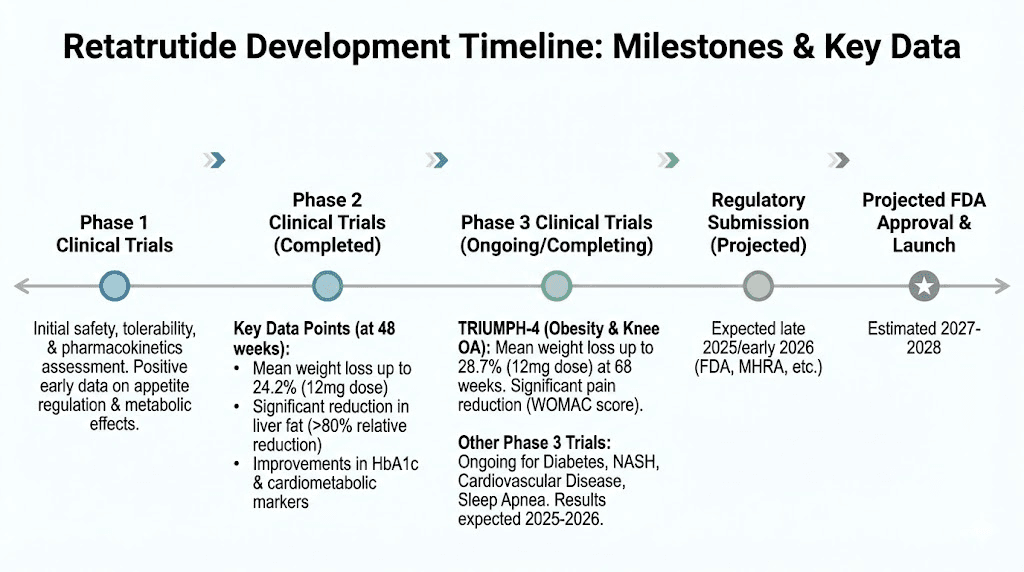
Retatrutide has become the most discussed research peptide in the weight management space, and for good reason. As a triple receptor agonist targeting GLP-1, GIP, and glucagon pathways simultaneously, it produced weight reduction numbers in clinical trials that made even seasoned researchers pause. Phase 2 data showed 24.2% mean body weight reduction at the 12mg dose over 48 weeks. Phase 3 results pushed even further, with the TRIUMPH-4 trial reporting 28.7% mean weight loss at 12mg. Those numbers changed the entire conversation around peptides for fat loss.
But here is the problem. Demand for retatrutide has exploded far faster than the supply chain can reliably support. That means bulk buyers face a minefield of quality concerns, pricing inconsistencies, and outright scams that did not exist even a year ago. Navigating this landscape requires more than a credit card and a search engine. It requires understanding what separates genuine research-grade retatrutide from the questionable material flooding the market.
This guide covers everything a serious researcher needs to know about purchasing retatrutide in bulk quantities. From purity testing and supplier evaluation to storage protocols and cost analysis, every detail that matters when spending significant capital on peptide research is addressed here. SeekPeptides has compiled this resource specifically for researchers who refuse to compromise on quality, because in peptide research, cutting corners on sourcing is the fastest way to invalidate months of work.
What is retatrutide and why it dominates the research landscape
Before diving into bulk purchasing strategies, understanding what makes retatrutide unique is essential. This is not just another incretin mimetic. It represents a fundamental shift in how researchers approach metabolic peptide protocols.
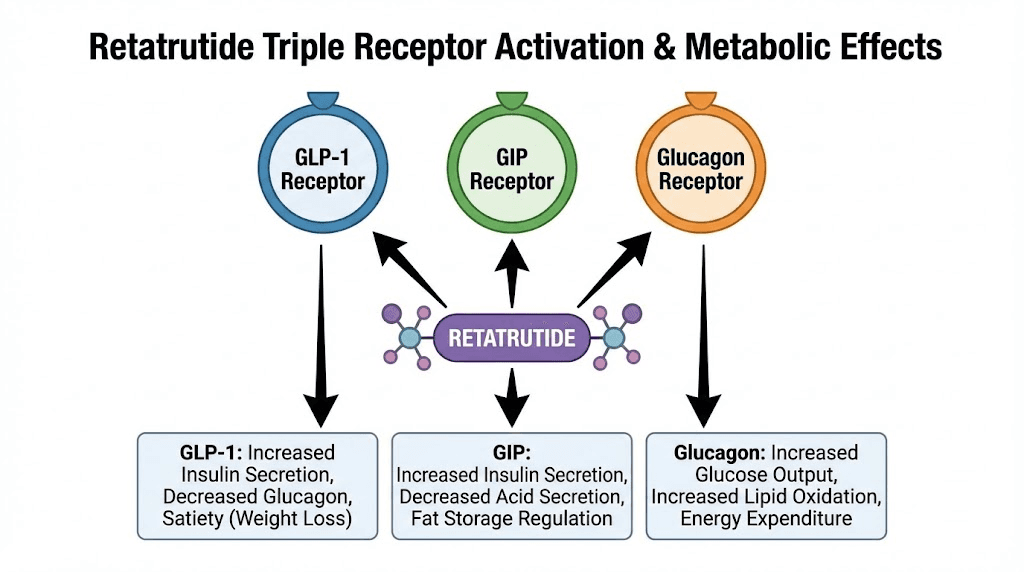
Retatrutide, also known by its development code LY3437943, is a 39-amino-acid synthetic peptide engineered by Eli Lilly. What sets it apart from predecessors like semaglutide and tirzepatide is its mechanism of action. While semaglutide activates only the GLP-1 receptor and tirzepatide targets both GLP-1 and GIP receptors, retatrutide activates all three metabolic hormone receptors: GLP-1, GIP, and glucagon.
That third receptor, glucagon, is the game changer.
Glucagon receptor activation drives energy expenditure through thermogenesis, promotes hepatic fat oxidation, and enhances lipolysis in ways that GLP-1 activation alone simply cannot achieve. The result is a compound that reduces appetite through GLP-1, enhances insulin sensitivity through GIP, and actively burns stored fat through glucagon receptor engagement. Three mechanisms working in concert, each amplifying the others.
Clinical trial results that changed the conversation
The numbers from clinical trials tell a compelling story. In the Phase 2 trial published in the New England Journal of Medicine, participants receiving the highest dose of retatrutide (12mg weekly) achieved a mean body weight reduction of 24.2% over 48 weeks. More than 90% of participants lost at least 10% of their baseline weight. Nearly two-thirds lost 20% or more. And roughly one quarter lost 30% or more of their starting body weight.
Those results surpassed anything previously documented with semaglutide protocols or tirzepatide protocols.
The Phase 3 TRIUMPH program, enrolling more than 5,800 participants across four global trials, continued to validate these findings. The TRIUMPH-4 trial reported percent body weight changes of -26.4% at the 9mg dose and -28.7% at the 12mg dose, compared to just -2.1% with placebo. Beyond weight reduction, researchers documented improvements in BMI, waist circumference, fasting plasma glucose, HbA1c, blood pressure, and liver fat content. More than 85% of participants on the 12mg dose achieved normal levels of liver fat after 48 weeks.
This broad spectrum of metabolic benefits is precisely why demand for retatrutide has surged among research institutions worldwide.
The molecular design that enables weekly dosing
Retatrutide incorporates a C20 fatty diacid moiety that binds to serum albumin in the bloodstream. This albumin binding dramatically extends the peptide half-life to approximately six days, which supports once-weekly subcutaneous administration. For researchers designing long-term protocols, this pharmacokinetic profile offers significant practical advantages over compounds requiring more frequent dosing.
The peptide backbone is engineered from a GIP sequence, modified to achieve balanced activation across all three target receptors. Compared to native glucagon and GLP-1, retatrutide shows lower potency at GLP-1 and glucagon receptors but higher potency at the GIP receptor. This carefully calibrated receptor selectivity profile contributes to the favorable side effect profile observed in clinical studies, where gastrointestinal adverse events were predominantly mild and occurred mainly during dose escalation phases.
What "bulk retatrutide" actually means for researchers
The term "bulk" in the peptide world does not have a single universal definition. For some researchers, bulk means purchasing ten vials instead of one. For others, it means ordering gram-scale quantities for extended protocol series. Understanding the different tiers of bulk purchasing helps researchers make smarter decisions about how much to order and when.
Volume tiers in the research peptide market
Most suppliers structure their bulk pricing across several tiers. The exact breakpoints vary, but a typical structure looks something like this:
Tier | Typical Quantity | Discount Range | Best For |
|---|---|---|---|
Single purchase | 1-3 vials (10-30mg) | None | Initial testing |
Small bulk | 5-10 vials (50-100mg) | 10-15% off | Short protocols |
Medium bulk | 20-50 vials (200-500mg) | 20-30% off | Extended research |
Large bulk | 100+ vials or gram quantities | 30-50% off | Institutional programs |
The sweet spot for most independent researchers falls in the small to medium bulk range. Ordering 5-10 vials provides enough material for a complete retatrutide dosage protocol while qualifying for meaningful volume discounts. Institutional buyers and research facilities typically operate at the medium to large bulk level, where per-milligram costs drop substantially.
One critical consideration that many first-time bulk buyers overlook: shelf life. Lyophilized retatrutide stored properly can remain stable for 12-24 months. But once reconstituted, the clock starts ticking much faster. Buying more than you can use within the stability window of your reconstituted solution is not saving money. It is wasting it.
Who actually buys retatrutide in bulk
Bulk retatrutide purchasing spans several distinct buyer profiles, each with different needs and priorities.
Academic research laboratories running controlled studies need consistent supply from a single batch to minimize variability across experimental conditions. These buyers prioritize batch consistency and comprehensive documentation above all else. They typically order medium to large bulk quantities and require full certificates of analysis for every batch.
Clinical research organizations conducting investigator-initiated trials often require substantial quantities to support multi-subject protocols running 24-48 weeks. Their purchasing decisions involve institutional review, vendor qualification processes, and stringent quality documentation requirements that go well beyond what individual researchers typically need.
Independent researchers exploring peptide protocols for personal investigation represent the largest segment of bulk buyers by transaction count, though not by volume. These researchers usually purchase in the small bulk range, motivated primarily by cost savings and supply security. For this group, understanding retatrutide pricing structures and quality verification methods is especially important, because they typically lack the institutional quality assurance infrastructure that larger organizations maintain.
Why serious researchers buy retatrutide in bulk
Buying retatrutide in bulk is not just about getting a lower price per vial. There are several strategic reasons why experienced researchers prefer larger orders, and understanding these reasons helps first-time buyers make better decisions.
Cost efficiency at scale
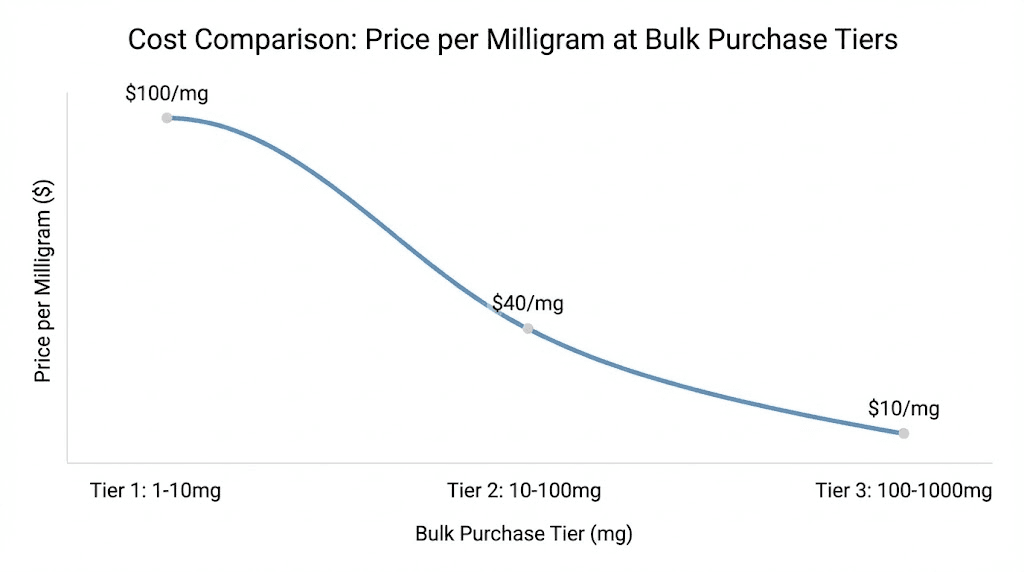
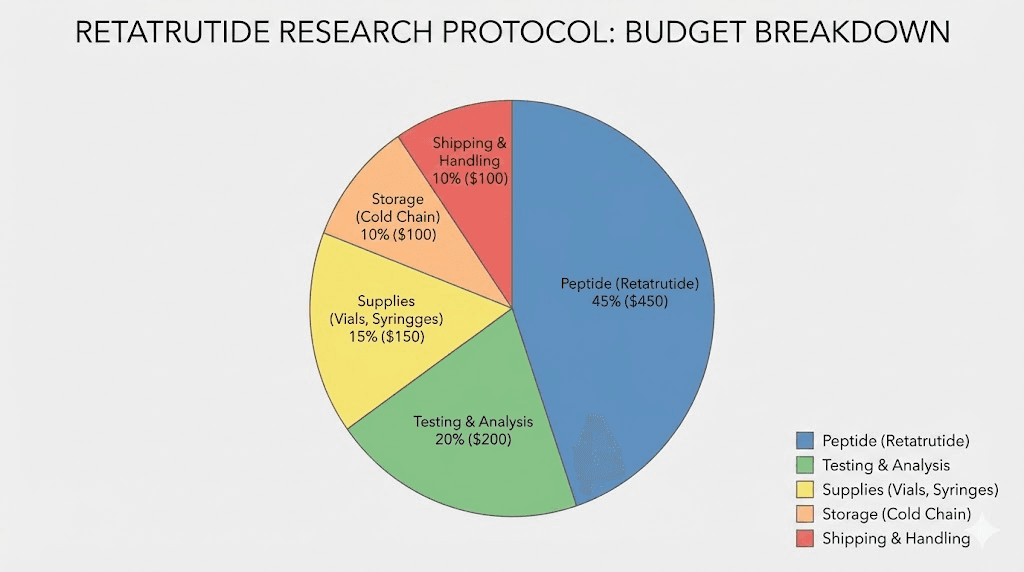
The most obvious advantage is price. Bulk purchasing reduces the per-milligram cost of retatrutide significantly. A single 10mg vial might cost $80-150 depending on the supplier. But ordering 10 vials often brings the per-vial cost down to $55-100, and ordering 50+ vials can push prices below $40-60 per vial. Over the course of a 48-week protocol using the retatrutide dosage calculator to plan administration, those savings compound into hundreds or even thousands of dollars.
Use the peptide cost calculator to model the actual cost difference between single-vial and bulk purchasing for your specific protocol requirements. The numbers often surprise researchers who have been ordering one vial at a time.
Batch consistency across protocol duration
This is the reason that separates experienced researchers from beginners. Every batch of synthesized peptide has slightly different characteristics, even from the same manufacturer. Purity might be 99.2% in one batch and 98.7% in the next. Trace impurity profiles differ. Reconstitution behavior can vary subtly.
When you run a 48-week protocol and your peptide comes from four different batches ordered at different times, you introduce variability that has nothing to do with the compound itself. Bulk purchasing from a single batch eliminates this variable entirely. One batch, one purity profile, one set of characteristics across the entire protocol duration.
For researchers publishing data or tracking precise outcomes, batch consistency is not optional. It is fundamental to research-grade methodology.
Supply chain security
The peptide supply chain is not always reliable. Suppliers run out of stock. Manufacturing delays happen. Shipping disruptions occur. Regulatory changes can temporarily restrict certain compounds. Having a sufficient supply on hand means your protocol does not get interrupted by factors completely outside your control.
Researchers who have experienced a mid-protocol supply disruption, where they could not obtain their peptide for two or three weeks mid-study, understand this viscerally. That interruption does not just delay results. It can invalidate weeks of prior data collection. Buying bulk upfront is insurance against supply chain volatility, and in the current retatrutide market where availability fluctuates, that insurance has real value.
How to evaluate retatrutide quality before committing to a bulk purchase
Quality evaluation is the single most important skill a bulk peptide buyer can develop. The difference between genuine research-grade retatrutide and substandard material is not always visible to the naked eye. Both arrive as white lyophilized powder in sealed vials. Both dissolve in bacteriostatic water. But only one will produce reliable, reproducible results in your research.
Certificate of analysis: the first line of defense
Every reputable peptide supplier provides a Certificate of Analysis (COA) with each batch. This document should contain, at minimum:
HPLC purity analysis showing purity percentage (look for 98% or higher, preferably 99%+)
Mass spectrometry data confirming molecular identity matches retatrutide (molecular weight approximately 4,113 Da)
Amino acid analysis verifying the correct 39-amino-acid sequence
Residual solvent testing showing levels below ICH guidelines
Water content by Karl Fischer analysis (typically below 5%)
Peptide content showing actual peptide mass versus total vial contents
A COA missing any of these core analyses should raise immediate concerns. If a supplier cannot provide mass spectrometry data confirming molecular identity, walk away. Full stop.
Third-party testing: trust but verify
Here is an uncomfortable truth about the peptide industry. Independent verification studies have found that 15-20% of supplier-provided certificates of analysis contain significant discrepancies when checked by third-party laboratories. The most common issue is purity overstatement, where a supplier claims 99% purity but independent testing reveals the actual figure is closer to 88-94%.
For bulk purchases where you are committing significant capital, third-party verification is not paranoia. It is basic due diligence. Several independent laboratories offer peptide testing services:
HPLC purity testing typically costs $150-300 per sample
Identity plus purity combination testing runs $400-600
Full characterization panels including residual solvents and heavy metals cost $800-1,200
For a bulk order worth several thousand dollars, spending $300-600 on independent verification is a small price for certainty. Look for testing facilities with ISO/IEC 17025 accreditation, dedicated HPLC and mass spectrometry equipment, and documented experience with peptide analysis.
Researchers who understand lyophilized versus liquid peptide quality markers are better positioned to evaluate test results meaningfully.
Supplier evaluation criteria that actually matter
Not all suppliers are created equal, and the cheapest option is rarely the best value. When evaluating suppliers for bulk retatrutide, prioritize these factors:
Transparency. Does the supplier publish batch-specific COAs? Can you request a COA before purchasing? Do they name their testing laboratory? Suppliers who obscure their quality documentation are hiding something.
Manufacturing standards. GMP (Good Manufacturing Practice) certification indicates a higher level of quality control. While not all research peptide suppliers operate under full pharmaceutical GMP, those that follow GMP-adjacent protocols for peptide synthesis typically produce more consistent, higher-quality material.
Track record. How long has the supplier operated? What do independent reviews say? Are there third-party test reports from customers confirming stated purity? A supplier with years of verified quality history is a lower risk than one that appeared last month with the lowest prices in the market.
Communication. Can you reach a knowledgeable person who answers technical questions about their product? Legitimate suppliers employ people who understand peptide chemistry. If customer service cannot explain the difference between HPLC purity and net peptide content, consider that a warning sign.
For researchers exploring where to buy retatrutide, these evaluation criteria separate reliable sources from risky ones.
Understanding purity testing methods for retatrutide
If you are spending serious money on bulk retatrutide, understanding the testing methods that verify quality is not optional. You do not need a chemistry degree. But you do need to know what these tests measure, what the numbers mean, and what constitutes a passing result versus a concerning one.
HPLC purity analysis explained
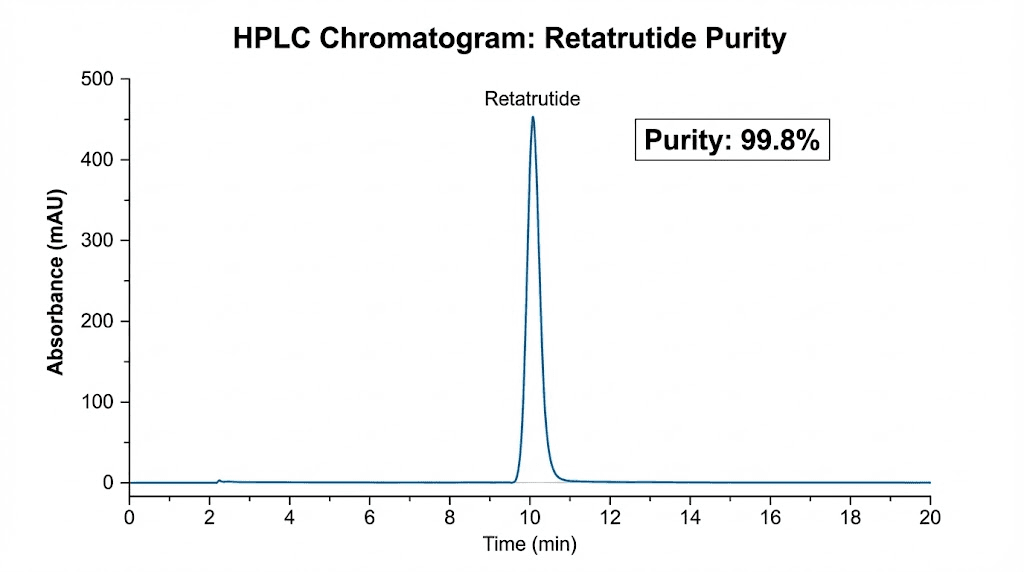
High-Performance Liquid Chromatography, or HPLC, is the gold standard method for peptide purity determination. The technique works by dissolving the peptide sample and passing it through a column packed with specialized material. Different molecules travel through the column at different speeds based on their chemical properties. A detector at the end measures what comes through and when.
The result is a chromatogram, essentially a graph showing peaks. The main peak represents your target peptide (retatrutide). Smaller peaks represent impurities, degradation products, truncated sequences, or other contaminants. Purity is calculated as the percentage of total peak area represented by the main retatrutide peak.
For research-grade retatrutide, you want to see HPLC purity of 98% or higher. Premium suppliers achieve 99%+ consistently. Anything below 95% should be rejected for serious research applications, as the cumulative effect of impurities over a multi-week protocol introduces unacceptable variability.
Mass spectrometry for identity confirmation
HPLC tells you how pure your sample is. Mass spectrometry tells you what your sample actually is. This distinction matters enormously in the current market, where mislabeled peptides occur more frequently with high-demand compounds like retatrutide.
Mass spectrometry measures the molecular weight of the peptide with extreme precision. Retatrutide has a known molecular weight of approximately 4,113.58 Da. If the mass spec reading matches this value within the expected margin of error, you have confirmation that the vial actually contains retatrutide and not a cheaper substitute or completely different peptide.
Without mass spectrometry confirmation, you are trusting the label. In a market where independent studies show significant discrepancy rates in supplier documentation, trusting the label is a gamble that experienced researchers refuse to take.
Additional quality markers worth checking
Beyond HPLC and mass spec, several additional tests provide useful quality information:
Karl Fischer water content analysis measures residual moisture in the lyophilized powder. Water content above 5% accelerates peptide degradation during storage and can affect reconstitution behavior. Well-manufactured lyophilized retatrutide typically shows water content between 1-3%.
Amino acid analysis verifies that the peptide contains the correct amino acid residues in the correct proportions. For a 39-amino-acid peptide like retatrutide, even small sequence errors can dramatically alter biological activity.
Endotoxin testing checks for bacterial contamination, which is particularly important for peptides intended for injection in research subjects. Endotoxin levels should be below 5 EU/mg for research-grade material.
Heavy metal analysis screens for toxic metal contamination that can occur during peptide synthesis or purification. Acceptable limits follow ICH Q3D guidelines for elemental impurities.
Understanding these quality markers helps researchers make informed decisions when evaluating bulk suppliers and comparing products from different sources. The difference between research-grade and pharmaceutical-grade peptides often comes down to the stringency of these very testing protocols.
Red flags and scam prevention in the bulk peptide market
The retatrutide market attracts both legitimate suppliers and bad actors looking to profit from surging demand. Recognizing red flags before you hand over money for a bulk order can save you thousands of dollars and months of wasted research time.
Pricing that defies reality
Retatrutide is a complex 39-amino-acid peptide with a C20 fatty diacid modification. Synthesizing it at high purity requires sophisticated equipment, skilled chemists, and expensive raw materials. If a supplier offers prices dramatically below market rates, ask yourself how they are achieving that.
Sometimes the answer is volume manufacturing in regions with lower labor costs. That is legitimate and can produce quality product. But sometimes the answer is that the product is not what it claims to be. Purity might be far below stated levels. The peptide might be cut with fillers. Or the vial might contain a completely different, cheaper peptide mislabeled as retatrutide.
Compare pricing across at least five established suppliers before making a bulk purchase. If one supplier is 60-70% cheaper than the market average, proceed with extreme caution and demand independent third-party testing before committing to a large order.
Missing or suspicious documentation
Legitimate suppliers provide batch-specific COAs without hesitation. They name their testing laboratories. They provide lot numbers that can be traced to specific production runs. They welcome questions about their manufacturing and quality control processes.
Red flags in documentation include:
Generic COAs that do not reference a specific batch or lot number
COAs with identical data across different batches (suggesting the document is fabricated)
Refusal to provide COA before purchase
Testing laboratory names that cannot be verified as real entities
HPLC chromatograms that appear altered or inconsistent with stated purity values
Missing mass spectrometry data
If you cannot verify the authenticity of a supplier COA, send a sample to an independent lab before ordering in bulk. The $300 investment in verification testing can prevent a $3,000 loss on fraudulent product.
Website and communication red flags
Pay attention to the details that suggest whether a supplier is a legitimate operation or a fly-by-night scam.
Newly registered domains (less than six months old) selling high-demand peptides at steep discounts are high-risk. Websites with no verifiable physical address, no phone number, and only email contact should trigger caution. Suppliers who cannot answer basic technical questions about their products, such as the synthesis method, purification approach, or storage recommendations, likely do not manufacture or even properly source what they sell.
Professional presentation alone does not guarantee legitimacy, but a lack of professional basics almost always signals problems. Researchers who have read our guide on buying retatrutide online will recognize many of these warning patterns.
Payment method restrictions
Be cautious of suppliers who only accept cryptocurrency or wire transfers with no buyer protection. While some legitimate peptide suppliers accept crypto for privacy reasons, the inability to pay with methods that offer buyer recourse (credit cards, PayPal) can indicate a supplier who does not plan to resolve quality disputes.
For large bulk orders, establishing a purchase agreement that includes quality specifications and remedies for non-conforming product is standard practice among professional buyers. If a supplier will not agree to basic quality terms in writing, find a different supplier.
Proper storage protocols for bulk retatrutide
Buying retatrutide in bulk only makes financial sense if you can store it properly. Peptide degradation is real, measurable, and irreversible. Once a peptide degrades, no amount of careful reconstitution or protocol adjustment can restore its original potency. Proper storage is not optional. It is the difference between usable material and expensive waste.
Lyophilized storage: your primary defense
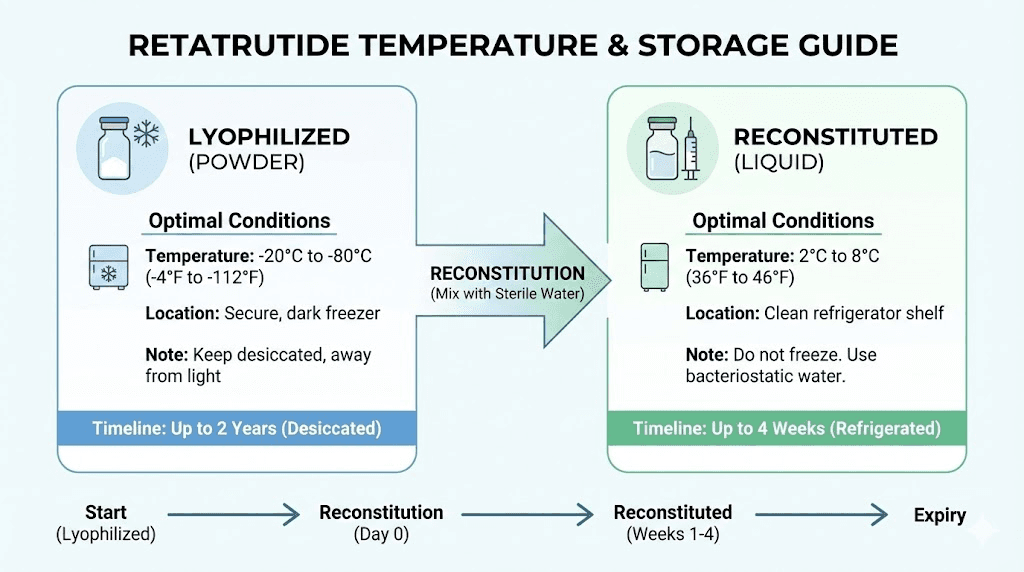
Unreconstituted, lyophilized retatrutide should be stored at -20 degrees Celsius (-4 degrees Fahrenheit) for long-term preservation. At this temperature, properly manufactured lyophilized peptide can maintain stability for 24 months or longer. Some researchers report acceptable stability at 2-8 degrees Celsius (standard refrigerator temperature) for periods up to 12 months, but -20 degrees Celsius provides the widest safety margin for bulk quantities intended for extended use.
Key storage principles for lyophilized retatrutide:
Store in original sealed vials until ready for reconstitution
Protect from light exposure using opaque containers or amber vials
Minimize moisture exposure by storing in airtight containers with desiccant packs
Avoid repeated freeze-thaw cycles, as thermal cycling accelerates degradation
Keep vials upright to prevent powder from contacting rubber stoppers
Label each vial with lot number, date received, and storage temperature
For researchers with significant bulk quantities, consider dividing stock across multiple storage locations as a risk mitigation strategy. A single freezer failure should not destroy your entire supply. This is basic laboratory risk management that applies to all valuable reagents, not just peptides.
Reconstituted storage: the ticking clock
Once you add bacteriostatic water to your retatrutide, the stability profile changes dramatically. Reconstituted retatrutide should be stored at 2-8 degrees Celsius (refrigerated) and used within 2-4 weeks for optimal results.
Some researchers push reconstituted storage to 4-6 weeks, but peptide degradation accelerates with each passing day in solution. HPLC analysis of reconstituted peptide solutions typically shows measurable purity decline beginning around week three, with more significant degradation after week four. For protocols requiring precise dosing, reconstituting only what you can use within a two-week window provides the best balance between convenience and potency.
This is why buying bulk lyophilized material and reconstituting individual vials as needed is far superior to reconstituting everything at once. Your bulk supply stays stable in the freezer at -20 degrees while you work through one reconstituted vial at a time.
Researchers working with temperature-sensitive peptides will find that the same storage principles apply across most research compounds in this class.
Environmental threats to peptide stability
Two factors destroy peptide stability faster than anything else: light and moisture.
UV light breaks peptide bonds and triggers oxidative degradation. Even ambient room light, over extended periods, can reduce potency. Store all retatrutide, whether lyophilized or reconstituted, away from direct light. Amber vials provide some protection, but supplementing with opaque outer containers is better.
Moisture is equally destructive to lyophilized peptide. Even small amounts of absorbed water can initiate hydrolysis reactions that fragment the peptide chain. This is why proper lyophilization technique during manufacturing matters so much, and why storage with desiccant packs is essential for long-term stability.
Temperature fluctuations pose a more subtle threat. Each cycle from cold to warm and back again stresses the peptide structure. If your freezer cycles through defrost periods, the resulting temperature swings can gradually degrade stored peptide. Frost-free freezers are particularly problematic because they achieve frost prevention through periodic warming cycles. A manual defrost laboratory freezer held at a steady -20 degrees Celsius is the ideal storage environment for bulk peptide stock.
Reconstitution protocols for bulk retatrutide
Proper reconstitution is the bridge between a vial of lyophilized powder and a usable research solution. Get it right, and you preserve all the potency and purity you paid for. Get it wrong, and you degrade the peptide before the first administration.
Essential equipment and materials
Before reconstituting any retatrutide, gather these materials:
Bacteriostatic water (BAC water) preserved with 0.9% benzyl alcohol
Insulin syringes (1mL with 29-31 gauge needles) for precise volume measurement
Alcohol swabs for sterilizing vial stoppers
Clean workspace free from dust, direct light, and temperature extremes
Do not use sterile water or normal saline for reconstitution unless you plan to use the entire vial within 24-48 hours. Bacteriostatic water contains a preservative that inhibits microbial growth, extending the usable life of your reconstituted solution to 2-4 weeks when refrigerated. Sterile water without preservative becomes a potential bacterial growth medium within days.
Standard reconstitution procedure
The process is straightforward but demands attention to detail.
Clean the rubber stopper of your retatrutide vial and your BAC water vial with alcohol swabs. Allow them to dry completely. Draw your desired volume of BAC water into a clean insulin syringe. For a standard 10mg vial, adding 2mL of BAC water creates a concentration of 5mg/mL. This concentration works well for the common dose ranges used in retatrutide research.
Insert the needle through the rubber stopper and direct the BAC water stream against the inside wall of the vial, not directly onto the lyophilized cake. Let the water run slowly down the glass wall and into the powder. This gentle approach minimizes physical stress on the peptide molecules.
Do not shake the vial. Ever. Shaking creates foam, and foam means air-liquid interfaces where peptide molecules are subjected to surface tension forces that cause denaturation. Instead, gently swirl the vial in circular motions. Or better yet, simply let it sit. Most properly lyophilized retatrutide will dissolve within 5-10 minutes without any agitation at all.
Once the solution is clear with no visible particles, your retatrutide is ready for use. If particles remain after 30 minutes of gentle swirling, the peptide may have degraded or been improperly lyophilized. Do not use a solution with persistent cloudiness or particulates.
Researchers who want exact reconstitution volumes for different vial sizes can reference our peptide reconstitution calculator or check the general peptide reconstitution guide that applies similar principles across all lyophilized peptides.
Reconstitution math for common vial sizes
Vial Size | BAC Water Added | Concentration | Units per 0.1mg |
|---|---|---|---|
5mg | 1.0 mL | 5.0 mg/mL | 2 units (0.02mL) |
10mg | 2.0 mL | 5.0 mg/mL | 2 units (0.02mL) |
15mg | 3.0 mL | 5.0 mg/mL | 2 units (0.02mL) |
20mg | 2.0 mL | 10.0 mg/mL | 1 unit (0.01mL) |
30mg | 3.0 mL | 10.0 mg/mL | 1 unit (0.01mL) |
Choose a concentration that allows you to measure your target dose accurately with the syringes you have available. Higher concentrations reduce injection volume but require more precise measurement. Lower concentrations are more forgiving of small measurement errors but require larger injection volumes. For most researchers working with standard insulin syringes, a concentration of 5mg/mL provides the best balance.
Common reconstitution mistakes that destroy potency
Even experienced researchers sometimes make errors that compromise their reconstituted peptide. Avoid these common pitfalls:
Injecting BAC water directly onto the lyophilized cake. The force of the liquid stream can physically damage peptide structures. Always direct the stream against the vial wall.
Shaking the vial to dissolve faster. Patience costs nothing. Shaking costs potency. Let the peptide dissolve at its own pace.
Using non-sterile equipment. Contamination introduces bacteria that metabolize peptides and produce endotoxins. Every piece of equipment that contacts your solution must be sterile.
Reconstituting too far in advance. Only reconstitute what you will use within 2-4 weeks. Your bulk supply remains more stable as lyophilized powder than as a solution. Reconstitute one vial at a time.
Storing reconstituted solution at room temperature. Even brief exposure to room temperature accelerates degradation. Reconstituted retatrutide should go straight into the refrigerator at 2-8 degrees Celsius.
Cost analysis: bulk purchasing economics for retatrutide
Money matters. Understanding the economics of bulk retatrutide purchasing helps researchers maximize their budgets without sacrificing quality. The goal is not to find the cheapest retatrutide. The goal is to find the best value, which means the highest quality material at the lowest per-milligram cost.
Price per milligram: the metric that matters
When comparing suppliers or evaluating bulk deals, always convert to price per milligram. Vial sizes vary (5mg, 10mg, 15mg, 20mg, 30mg), pricing structures differ, and promotional discounts muddy direct comparison. Price per milligram cuts through the noise.
As of early 2026, research-grade retatrutide from established U.S. suppliers typically falls in these ranges:
Purchase Type | Price Per mg | Quality Level | Testing Standard |
|---|---|---|---|
Single vial (premium) | $8-15/mg | 99%+ HPLC, full COA | Third-party verified |
Small bulk (5-10 units) | $5-10/mg | 98-99%+ HPLC | In-house + COA |
Medium bulk (20-50 units) | $3-7/mg | 98%+ HPLC | In-house COA |
Large bulk (100+ units) | $2-5/mg | 97-99% HPLC | Varies |
Prices below $2/mg should prompt careful quality verification. While legitimate manufacturers in certain regions can produce at these price points, the risk of compromised quality increases significantly at the bottom of the market.
Total cost of a research protocol
Let us calculate the real cost of a standard retatrutide research protocol to illustrate how bulk pricing impacts total investment.
A typical 48-week protocol following the Phase 2 trial escalation schedule requires approximately 432mg of retatrutide total (starting at 2mg weekly and escalating to 12mg weekly maintenance). Here is how the numbers break down at different purchasing tiers:
Approach | Estimated Cost | Cost Per Week |
|---|---|---|
Single vials as needed | $3,456-6,480 | $72-135 |
Small bulk purchase | $2,160-4,320 | $45-90 |
Medium bulk purchase | $1,296-3,024 | $27-63 |
Large bulk purchase | $864-2,160 | $18-45 |
The difference between buying single vials and making a medium bulk purchase can save $1,500-3,500 over the course of a complete protocol. That is a significant sum, and it explains why cost-conscious researchers consistently choose bulk purchasing once they understand the economics.
Compare these figures using the peptide cost calculator to model your specific protocol parameters, including vial size, desired concentration, and protocol duration.
Hidden costs to factor into your budget
The sticker price of the peptide itself is not the complete picture. Smart bulk buyers account for several additional costs:
Shipping and cold chain. Bulk peptide orders often require cold chain shipping to maintain stability during transit. Overnight or priority shipping with ice packs adds $30-100 per order depending on volume and destination. Some suppliers include shipping in bulk pricing. Others do not.
Storage equipment. If you do not already own a laboratory-grade freezer capable of maintaining -20 degrees Celsius, factor that investment into your budget. A reliable manual-defrost laboratory freezer costs $500-2,000 depending on size and features. This is a one-time investment that pays for itself across multiple protocols.
Testing costs. Budget $300-600 for initial third-party verification testing of your first bulk order from a new supplier. This cost decreases in relevance for subsequent orders from the same supplier and batch, but the initial verification is a smart investment.
Reconstitution supplies. Bacteriostatic water, insulin syringes, alcohol swabs, and other consumables represent a minor but ongoing cost. Budget approximately $20-40 per month for basic reconstitution supplies.
Waste factor. Some product loss during reconstitution and administration is inevitable. Dead volume in syringes, residual material left in vials, and small measurement errors typically result in 3-8% product waste. Factor this into your quantity calculations when placing bulk orders.
Retatrutide dosage protocols from clinical research
Understanding the dosage protocols used in published clinical trials provides essential context for researchers designing their own protocols. While individual research applications may vary, the Phase 2 and Phase 3 trial data establishes the evidence base from which most protocols are derived.
The standard escalation protocol
The Phase 2 trial published in the New England Journal of Medicine established a dose escalation framework that has become the reference standard for retatrutide research. The protocol follows a methodical four-week escalation schedule designed to minimize gastrointestinal side effects while progressing toward therapeutic dose levels.
The starting dose is 2mg administered once weekly by subcutaneous injection. After four weeks at 2mg, the dose increases to 4mg weekly. Four weeks later, it escalates to 8mg weekly. And after another four weeks, the target maintenance dose of 12mg weekly is reached. Total escalation period: 12 weeks from initiation to full maintenance dose.
Weeks | Weekly Dose | Purpose |
|---|---|---|
1-4 | 2 mg | Initial tolerance assessment |
5-8 | 4 mg | First escalation, GI adaptation |
9-12 | 8 mg | Second escalation |
13-48 | 12 mg | Full maintenance dose |
The four-week interval between dose increases is not arbitrary. Retatrutide reaches pharmacokinetic steady state in approximately 4-5 weeks of consistent dosing at any given level. This means each escalation step allows the compound to reach equilibrium before the researcher assesses tolerance and decides whether to proceed to the next dose level.
For researchers who need precise dosing calculations based on vial size and concentration, the retatrutide dosage calculator provides exact unit measurements for each escalation step. Cross-reference with the comprehensive retatrutide dosage chart for visual protocol planning.
Target dose levels and their clinical outcomes
Clinical trials evaluated three primary maintenance dose levels: 4mg, 8mg, and 12mg weekly. The results show a clear dose-response relationship:
At 4mg weekly, participants in the Phase 2 trial achieved approximately 17.5% mean body weight reduction over 48 weeks. This dose produces meaningful results while minimizing side effect burden. It may be appropriate for researchers studying metabolic effects in subjects who are sensitive to gastrointestinal side effects or who have lower baseline body weight.
At 8mg weekly, mean weight reduction reached approximately 22.1% over the same period. This intermediate dose offers a strong balance between efficacy and tolerability, making it a popular choice for many research applications.
At 12mg weekly, the maximum studied dose, mean weight reduction hit 24.2% in Phase 2 and 28.7% in Phase 3 (TRIUMPH-4). This dose produces the most dramatic results but also carries the highest incidence of gastrointestinal side effects, particularly during the escalation phase. Nausea, vomiting, diarrhea, and constipation were all more common at higher doses, though these effects typically diminished after the body adapted to each new dose level.
Understanding the dose-response relationship helps researchers, particularly bulk buyers, calculate exactly how much material they need for their planned protocol duration and target dose. A researcher running a 48-week protocol at 4mg weekly needs significantly less total material than one targeting 12mg maintenance.
Administration and injection technique
Retatrutide is administered by subcutaneous injection once weekly. The injection site should rotate among the abdomen, thigh, and upper arm to prevent lipodystrophy at any single site. Our guide on where to inject retatrutide covers injection site selection and technique in complete detail.
Administration timing is flexible, but consistency matters. Injecting at approximately the same time each week maintains the steadiest blood levels. Researchers who understand GLP-1 injection techniques will find the process virtually identical for retatrutide, with the same emphasis on proper angle, depth, and rotation.
Retatrutide compared to other GLP-1 peptides: why bulk buyers choose it
Researchers who buy retatrutide in bulk have typically done their homework comparing available options. Understanding where retatrutide fits in the GLP-1 peptide landscape explains why it commands premium pricing and why demand continues to outstrip supply.
Retatrutide vs semaglutide
The comparison between retatrutide and semaglutide highlights a fundamental difference in mechanism. Semaglutide is a GLP-1 receptor agonist only. It reduces appetite and slows gastric emptying through a single receptor pathway. Clinical data shows semaglutide produces approximately 15-17% body weight reduction at maximum doses over similar timeframes.
Retatrutide, targeting all three metabolic receptors, consistently outperforms semaglutide in head-to-head comparisons within trial populations. The 24-29% weight reduction seen with retatrutide at 12mg represents a 50-70% improvement over semaglutide efficacy, depending on the specific semaglutide dose and study population being compared.
For researchers studying metabolic mechanisms, this triple-agonist approach also opens up research avenues that semaglutide cannot, particularly around glucagon-mediated energy expenditure and hepatic fat metabolism. The liver fat reduction data from retatrutide trials, where more than 85% of participants achieved normal liver fat levels, has no equivalent in semaglutide literature.
Researchers interested in semaglutide efficacy timelines or semaglutide onset of action will find that retatrutide generally shows faster initial response, though direct comparison studies are limited.
Retatrutide vs tirzepatide
The comparison with tirzepatide is more nuanced because tirzepatide already targets two receptors (GLP-1 and GIP). Tirzepatide produces approximately 20-22% body weight reduction at its highest approved dose of 15mg weekly, which is closer to retatrutide performance than semaglutide is.
The key differentiator remains the glucagon receptor. Retatrutide activates glucagon receptors in addition to GLP-1 and GIP, driving thermogenesis and fat oxidation mechanisms that tirzepatide does not directly engage. This additional metabolic pathway likely accounts for the incremental weight loss difference between the two compounds.
From a bulk purchasing perspective, tirzepatide is currently more readily available than retatrutide through both pharmaceutical and research channels, since tirzepatide already has FDA approval (as Mounjaro for type 2 diabetes and Zepbound for weight management). Retatrutide remains investigational, which means supply chains are less established and pricing can be more volatile.
For detailed comparisons between these compounds, researchers can consult the tirzepatide comparison guides and ozempic versus retatrutide analysis available through SeekPeptides.
Why bulk buyers specifically choose retatrutide
Despite higher pricing and less established supply chains, researchers continue choosing retatrutide for bulk purchase for several compelling reasons.
The efficacy ceiling is highest with retatrutide among available GLP-1 receptor agonists. For researchers pursuing maximum metabolic outcomes, no other currently available compound matches its clinical performance data. The triple-agonist mechanism provides unique research value that cannot be replicated by combining other single or dual-agonist compounds.
Metabolic versatility is another factor. Beyond weight management, retatrutide shows promise for liver fat reduction, glycemic control in type 2 diabetes, and potentially cardiovascular risk reduction. Bulk buyers working across multiple research areas find that a single compound addressing multiple metabolic pathways simplifies protocol design and reduces overall peptide inventory requirements.
And then there is the investment thesis. Researchers who believe retatrutide will eventually receive FDA approval (currently projected no earlier than 2027) see bulk purchasing at current research peptide prices as an opportunity to work with the compound before commercial availability reshapes pricing and access.
Legal and regulatory considerations for bulk peptide purchases
Navigating the legal landscape around bulk peptide purchasing is essential for researchers who want to avoid regulatory complications. The rules are not always intuitive, and they vary by jurisdiction.
Research use only: what it means in practice
Research-grade retatrutide is sold exclusively for laboratory and research use. It is not approved for human or veterinary therapeutic use. This "research use only" designation carries specific implications:
Researchers are expected to use the material in legitimate research contexts, whether academic studies, independent investigation, or institutional research programs. Commercial vendors operating legally will include research-use-only disclaimers on their products and may require purchasers to acknowledge these terms.
Documentation of research intent is increasingly important. Some suppliers request information about intended use, institutional affiliation, or research objectives as part of their compliance process. Maintaining clear records of purchase intent, storage conditions, and usage logs is good practice for any researcher buying bulk quantities.
FDA regulatory status
Retatrutide is currently classified as an investigational new drug (IND) by the FDA. It has not received approval for any clinical indication. Eli Lilly, the compound developer, is running the Phase 3 TRIUMPH clinical trial program, and FDA approval is not expected before 2027 at the earliest.
This investigational status means that the only legitimate pathway for obtaining pharmaceutical-grade retatrutide for clinical use is through participation in authorized clinical trials. Research-grade retatrutide available from peptide suppliers is synthesized by third-party manufacturers and is not the same material used in Eli Lilly clinical trials.
Understanding the distinction between research and pharmaceutical peptides is important context for bulk buyers navigating this regulatory landscape.
Import and shipping regulations
Bulk peptide orders crossing international borders face additional regulatory scrutiny. Customs agencies in many countries may inspect, delay, or seize large peptide shipments, particularly for compounds that are associated with weight loss or performance enhancement.
Domestic purchasing from suppliers within your own country generally avoids import complications. For international orders, research the specific customs regulations in both the origin and destination countries. Some researchers have found that orders above certain quantity thresholds trigger additional documentation requirements or inspection procedures.
Working with suppliers who have experience shipping to your jurisdiction and who provide proper customs documentation reduces the risk of shipment delays or seizures. This is another advantage of establishing relationships with reputable, experienced suppliers rather than chasing the lowest price from unknown sources.
Setting up a proper research protocol with bulk retatrutide
Having bulk retatrutide in your freezer is only the beginning. Translating raw material into a well-designed research protocol requires planning, documentation, and systematic execution. Here is a framework for researchers ready to move from purchasing to protocol implementation.
Pre-protocol planning
Before the first injection, establish these foundational elements:
Define your research objectives. What specific outcomes are you measuring? Weight change? Metabolic markers? Body composition? Appetite metrics? Clear objectives drive everything from dose selection to measurement frequency.
Select your dose protocol. Based on clinical trial data, determine whether you are targeting 4mg, 8mg, or 12mg maintenance with the standard escalation schedule. Use the retatrutide dosage calculator to calculate exact material requirements for your planned duration.
Calculate total material needs. A 48-week protocol at 12mg maintenance requires approximately 432mg total. Add 5-10% for waste and reconstitution losses. Round up to the nearest bulk purchasing tier that covers your needs plus a small buffer.
Establish baseline measurements. Document starting weight, body composition, metabolic markers, and any other relevant parameters before beginning the protocol. Without rigorous baseline data, results lack meaningful context.
Documentation and tracking
Maintain detailed records throughout the protocol. At minimum, document:
Date, time, and dose of each administration
Injection site rotation schedule
Batch and lot number of peptide used for each administration
Storage temperature logs (daily minimum and maximum)
Reconstitution dates and volumes for each vial
Subjective observations (appetite changes, energy levels, GI effects)
Objective measurements on a defined schedule (weekly, biweekly, or monthly depending on parameter)
Good documentation transforms anecdotal experience into usable data. It also helps identify issues early, such as a batch that behaves differently from previous ones, or a storage temperature excursion that might have compromised product quality.
The GLP-1 plotter tool can help track progress visually over the course of a long protocol.
Safety monitoring and side effect management
Retatrutide clinical trials documented a generally favorable safety profile, but gastrointestinal side effects were common, especially during dose escalation. Nausea, vomiting, diarrhea, and constipation were the most frequently reported adverse events.
Most GI side effects occurred during dose escalation phases and diminished as the body adapted to each new dose level. The protocol design, with its four-week intervals between escalations, intentionally allows time for this adaptation. Researchers should resist the temptation to escalate faster than the standard protocol, as doing so increases the likelihood and severity of GI events.
Monitoring should also include attention to less common effects documented in trials: potential hair thinning, fatigue patterns, and injection site reactions. SeekPeptides provides comprehensive safety guides and side effect comparison resources that help researchers anticipate and manage common adverse events across the GLP-1 peptide class.
Nutritional considerations are also important during extended protocols. Significant appetite suppression can lead to inadequate protein intake, micronutrient deficiencies, and muscle mass loss if dietary composition is not actively managed. Researchers using retatrutide should reference nutritional guidelines similar to those recommended for semaglutide dietary protocols and tirzepatide meal planning.

Maximizing the value of your bulk retatrutide investment
Bulk purchasing is a financial commitment. Maximizing the return on that investment means extracting full value from every milligram through careful handling, precise dosing, and efficient protocol management.
Minimizing waste during reconstitution and administration
Product loss during reconstitution and administration is inevitable, but it can be minimized with attention to technique.
Dead volume in syringes is the biggest source of waste for most researchers. Standard insulin syringes retain approximately 0.02-0.05mL of solution in the needle hub after the plunger is fully depressed. Over the course of a 48-week protocol with weekly injections, that dead volume waste adds up to 1-2.5mL of wasted solution. Low dead volume syringes, designed specifically to minimize this retention, cost slightly more but pay for themselves in preserved peptide.
Vial retention is another loss point. After withdrawing the final dose from a vial, some solution inevitably remains trapped in the vial and cannot be reached by the needle. Using the vial inverted, with the needle touching the lowest point of the rubber stopper, maximizes the amount of solution you can extract. Expect to lose approximately 0.03-0.05mL per vial.
Measurement precision matters more at higher concentrations. If your reconstituted solution is 10mg/mL and your target dose is 2mg (0.2mL), a measurement error of just 0.02mL represents a 10% dose error. At 5mg/mL concentration, the same 0.02mL error on a 0.4mL draw represents only 5% error. Choosing reconstitution concentrations that match your measurement precision reduces both waste and dosing variability.
Supplemental strategies for enhanced protocols
Researchers running retatrutide protocols often combine the compound with supportive interventions that enhance outcomes. While the peptide itself drives the primary metabolic effects, several supplemental approaches can improve overall protocol results:
Adequate protein intake prevents lean mass loss during periods of significant caloric deficit. Target a minimum of 1.2-1.6g of protein per kilogram of body weight daily, adjusted based on activity level and body composition goals. Researchers studying similar compounds have documented the importance of nutritional support alongside GLP-1 agonist protocols.
Resistance training preserves muscle mass during weight loss phases and improves body composition outcomes. The combination of peptide-driven fat loss with exercise-driven muscle preservation produces more favorable metabolic profiles than either intervention alone. Peptides for muscle growth and athletic performance resources provide additional context for researchers interested in the exercise-peptide interaction.
Hydration monitoring becomes more important during protocols that significantly reduce food intake, as a substantial portion of daily fluid intake typically comes from food. Dehydration can exacerbate gastrointestinal side effects and affect the subjective experience of the protocol.
When to reorder: timing your bulk purchases
Do not wait until you are running low. Plan reorder timing to account for:
Supplier lead time (typically 3-7 business days for domestic orders, 2-4 weeks for international)
Testing time if you verify each new batch (add 1-2 weeks for third-party testing)
Buffer stock (maintain enough supply to cover 4-6 weeks of protocol even if a reorder is delayed)
A practical reorder trigger for most researchers is when remaining stock drops below 8 weeks of protocol supply. This provides sufficient runway to place an order, receive it, and verify quality without any risk of protocol interruption.
Establishing a relationship with a reliable supplier means faster processing times, consistent pricing, and sometimes advance notification of batch availability or stock changes. SeekPeptides members often share supplier experiences and reorder strategies in the community, helping fellow researchers navigate the sometimes unpredictable peptide supply landscape.
Building a quality-first approach to bulk peptide sourcing
The researchers who get the best results from bulk retatrutide protocols share a common trait: they never compromise on quality to save on price. This quality-first mindset informs every decision from supplier selection to storage protocol design.
Establishing supplier qualification criteria
Before placing your first bulk order, develop a written set of supplier qualification criteria. This does not need to be a formal pharmaceutical quality management document. A simple checklist works:
Does the supplier provide batch-specific COAs for every product?
Can the supplier provide COA before purchase upon request?
Does the COA include HPLC purity, mass spectrometry, and water content at minimum?
Has the supplier operated for more than 12 months with verifiable track record?
Can you find independent test reports or reviews confirming stated quality?
Does the supplier offer a clear returns or quality dispute resolution policy?
Is customer service responsive and technically knowledgeable?
Suppliers who meet all seven criteria represent the lower-risk tier of the market. Those who fail on multiple points should be avoided for bulk purchases, regardless of pricing.
Sample testing before bulk commitment
A smart strategy for new supplier relationships is the "sample-then-scale" approach. Purchase a single vial or small quantity first. Submit a sample for third-party testing. If the results match the supplier COA within acceptable tolerance (typically 1-2% for purity), proceed with confidence to a bulk order from the same batch.
This approach costs a bit more than jumping straight to a bulk purchase, but it provides empirical quality validation before committing significant capital. The cost of a single-vial test order plus third-party testing ($250-500 total) is a tiny fraction of a bulk purchase that could run into the thousands.
Long-term supplier relationship benefits
Researchers who work with the same supplier over multiple orders often receive benefits that one-time buyers do not:
Priority access to limited-stock batches, especially valuable for high-demand compounds like retatrutide. Consistent pricing structures that are not subject to the spot-market volatility that affects new customers. Personalized support for technical questions about product specifications, storage, and protocol design. And in some cases, early notification of upcoming batch releases that allows advanced planning for bulk purchases.
Building these relationships takes time but pays dividends in supply reliability, quality consistency, and overall research program efficiency. The peptide market, despite its challenges, rewards loyalty and professional engagement.
The future of retatrutide: what bulk buyers should know
Retatrutide is on a trajectory from investigational compound to potential blockbuster pharmaceutical. Understanding this trajectory helps bulk buyers make informed decisions about timing, quantity, and long-term planning.
Clinical development timeline
Eli Lilly Phase 3 TRIUMPH program is the most extensive clinical development program for retatrutide. With more than 5,800 participants across four global trials, the data generated will form the basis of any future regulatory submission. Initial Phase 3 results have been highly positive, with weight loss numbers meeting or exceeding Phase 2 performance.
FDA approval, if granted, is not expected before 2027. This timeline is based on the normal pace of Phase 3 data analysis, regulatory submission preparation, and FDA review processes. Any complications in the trial program or regulatory requirements could push this timeline further out.
What FDA approval would mean for bulk buyers
Once a peptide receives FDA approval for a specific indication, the commercial pharmaceutical version typically becomes available through pharmacies and medical providers. This has historically led to changes in the research peptide market:
Research-grade availability may decrease as manufacturers shift toward pharmaceutical supply chains. Pricing in the research market may actually decrease initially as the compound becomes more widely manufactured, but could also become more restricted as regulatory attention increases.
For researchers who want to work with retatrutide as a research compound, the current pre-approval period represents a window of relatively broad access and competitive pricing. Bulk buying during this period, while supply is available and pricing is market-driven rather than pharmacy-driven, may be strategically advantageous.
Researchers interested in how this trajectory has played out with similar compounds can look at the history of compounded semaglutide availability as a reference case.
Frequently asked questions
How much retatrutide do I need to buy in bulk for a complete protocol?
A standard 48-week retatrutide protocol at the 12mg maintenance dose requires approximately 432mg total, accounting for the escalation phase (2mg, 4mg, 8mg, then 12mg weekly). Add 5-10% for waste, bringing the total to approximately 450-475mg. This typically translates to 45-48 vials of 10mg product. For lower maintenance doses, proportionally less material is needed.
What purity level should I look for when buying bulk retatrutide?
Target 98% HPLC purity or higher for research applications. Premium suppliers consistently achieve 99%+ purity. Always request a batch-specific Certificate of Analysis showing HPLC chromatography, mass spectrometry identity confirmation, and water content analysis. For large bulk purchases, consider submitting a sample for independent third-party verification testing before committing to the full order.
How should I store bulk retatrutide long-term?
Store lyophilized (unreconstituted) retatrutide at -20 degrees Celsius in sealed original vials, protected from light and moisture. Under these conditions, lyophilized retatrutide maintains stability for 24+ months. Only reconstitute individual vials as needed. Reconstituted retatrutide should be refrigerated at 2-8 degrees Celsius and used within 2-4 weeks. Never freeze reconstituted solution.
Is it legal to buy retatrutide in bulk for research purposes?
Research-grade retatrutide is sold legally for laboratory and research use in most jurisdictions. It is not approved for human therapeutic use by the FDA or other regulatory agencies. Purchasers should maintain documentation of research intent and comply with local regulations regarding peptide procurement. Import regulations vary by country, so domestic purchasing is generally simpler for regulatory compliance.
What is the best vial size for bulk retatrutide purchases?
The 10mg vial is the most versatile for bulk purchasing. It provides enough material for approximately one week of maintenance-dose administration (assuming 12mg target, with a reconstitution concentration that allows accurate measurement). Larger vials (15mg, 20mg, 30mg) offer slightly better per-mg pricing but require reconstitution volumes that may exceed optimal stability windows if not used quickly enough.
How do I know if my bulk retatrutide has degraded?
Signs of degradation in lyophilized retatrutide include: color change from white to yellow or brown, collapse or liquefaction of the lyophilized cake, unusual odor upon opening the vial, and failure to dissolve clearly during reconstitution. In reconstituted solution, look for cloudiness, particulate formation, color change, or unusual viscosity. When in doubt, do not use the product. The cost of one wasted vial is far less than the cost of using degraded material that produces unreliable results.
Can I combine retatrutide with other research peptides in a stack?
Combining retatrutide with other compounds is an active area of research interest. However, stacking introduces additional variables that complicate interpretation of results. For researchers interested in peptide combinations, the peptide stack calculator provides a framework for planning multi-compound protocols. Researchers should approach stacking methodically, establishing baseline responses to each compound individually before combining them.
What happens if my bulk retatrutide shipment is delayed or seized by customs?
For domestic orders, shipping delays rarely exceed 1-2 weeks. For international orders, customs holds can last 2-6 weeks or result in seizure. To mitigate this risk: order from domestic suppliers when possible, maintain buffer stock to cover delays, request proper customs documentation from your supplier, and avoid ordering quantities that might trigger enhanced scrutiny. If a shipment is seized, work with the supplier to understand their reshipping or refund policy.
External resources
New England Journal of Medicine - Retatrutide Phase 2 Trial Results
PubMed Central - Retatrutide Systematic Review and Meta-Analysis
For researchers serious about optimizing their peptide research protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, dosage calculators, and a community of thousands of researchers who have navigated these exact sourcing and quality questions.
In case I do not see you, good afternoon, good evening, and good night. May your peptides stay pure, your storage stay cold, and your research stay rigorous.