Feb 5, 2026

Before you skip your next dose, stop. What happens after you discontinue semaglutide is not what most people expect. It is not a simple return to normal. It is not just gaining a few pounds back. The changes that unfold over the weeks and months following your last injection touch every system in your body, from the hormones that control your hunger to the neurotransmitters that regulate your mood. And the timeline matters far more than most guides will tell you.
The reality is stark. Clinical trial data from the STEP 1 extension study shows that participants regained two-thirds of their lost weight within one year of stopping semaglutide. Two-thirds. That means someone who lost 30 pounds might find 20 of those pounds returning, along with the cardiovascular risk factors, blood sugar elevations, and metabolic disruptions that came with the original weight. But weight regain is only part of the story.
The return of appetite, the surge in food cravings, the digestive changes, and the psychological shifts all follow a predictable pattern that you can prepare for if you understand the science behind it.
This guide covers every aspect of semaglutide withdrawal. You will learn the complete timeline of symptoms, the hormonal mechanisms driving those symptoms, evidence-based strategies for maintaining your progress, safe tapering protocols, and when it might make sense to restart treatment. Whether you are planning to stop semaglutide by choice or because of supply issues, insurance changes, or side effects, understanding what comes next is the single most important thing you can do to protect your results. SeekPeptides has compiled the research, the clinical data, and the practical strategies into one comprehensive resource so you can navigate this transition with confidence rather than confusion.
What happens when you stop semaglutide
Semaglutide does not cause classical withdrawal in the way that opioids, benzodiazepines, or alcohol do. There is no physical dependence. There are no seizure risks. There is no acute danger from stopping abruptly. What happens instead is something equally challenging but fundamentally different. Your body begins reverting to its pre-treatment state, and the biological systems that semaglutide was suppressing, modulating, and regulating start returning to their baseline function.
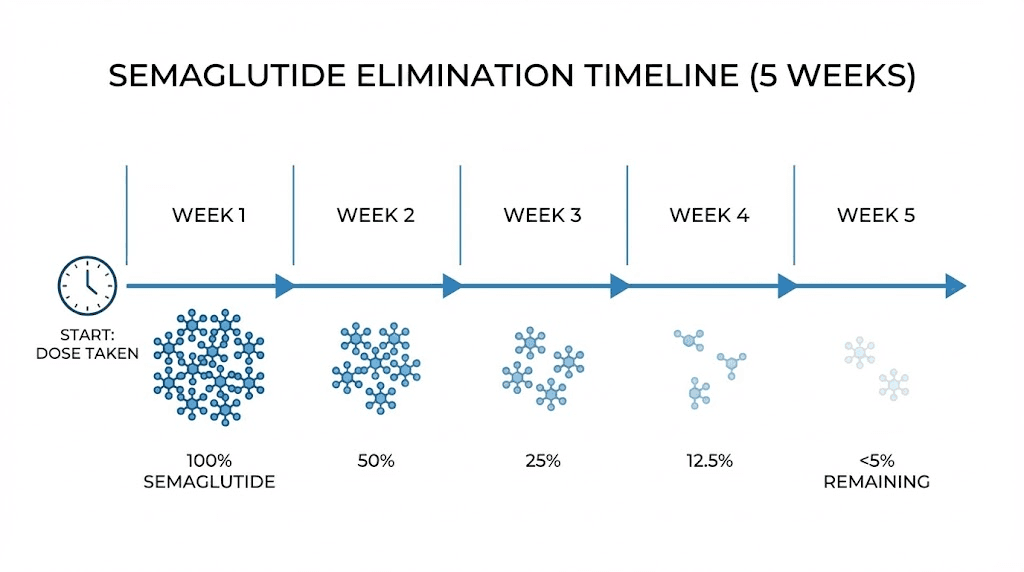
Think of it this way. Semaglutide was holding multiple physiological doors closed. Appetite suppression. Slowed gastric emptying. Enhanced insulin secretion. Reduced glucagon production. Dampened reward signaling in the brain. When you stop the medication, those doors do not fly open all at once. They open gradually, one by one, over a period of roughly five weeks as the drug clears your system. But they do open. Every single one of them.
The changes affect multiple body systems simultaneously.
Your endocrine system begins shifting as GLP-1 receptor stimulation fades. Your digestive system speeds up as gastric emptying normalizes. Your brain, particularly the hypothalamus and reward centers, loses the signal that was keeping food noise quiet and cravings at bay. Your pancreas adjusts its insulin and glucagon output. Your adipose tissue begins responding to shifting leptin levels.
Not everyone experiences every symptom. Some people transition off semaglutide with minimal disruption. Others find the return of appetite and cravings overwhelming. The severity depends on several factors, including how long you were on the medication, what dose you reached, how much weight you lost, your underlying metabolic health, and whether you built sustainable lifestyle habits during treatment.
Here is what makes semaglutide discontinuation particularly challenging. Obesity is a chronic condition. The STEP 1 trial investigators concluded explicitly that their findings "confirm the chronicity of obesity and suggest ongoing treatment is required to maintain improvements in weight and health." This is not a moral failing. It is biology. The same adaptive mechanisms that helped our ancestors survive famine now work against modern weight management efforts, and semaglutide was keeping those mechanisms in check.
Complete timeline of semaglutide withdrawal symptoms
Understanding the timeline gives you power. When you know exactly what to expect and when to expect it, the experience becomes manageable rather than alarming. The following timeline is based on semaglutide pharmacokinetics, clinical trial data, and real-world patient reports. Your individual experience may vary, but this framework covers what most people encounter.
Days 1 through 6 after your last dose
Very little changes during this first week. Semaglutide has a half-life of approximately 7 days, which means that after one week, roughly 50% of the medication remains active in your system. You are still experiencing significant appetite suppression. Blood sugar regulation remains largely intact. Gastric emptying is still delayed. Most people feel completely normal during this period and may wonder what all the concern was about.
This quiet period is deceptive.
Use these days wisely. Stock your kitchen with high-protein, high-fiber foods. Establish or strengthen your exercise routine. Set up meal planning systems. Build the infrastructure you will need when the medication effects begin fading. If you have been using semaglutide dosage tracking tools, now is the time to shift your tracking focus toward nutrition and activity metrics instead.
Days 7 through 14, the first real shift
This is when most people notice the first changes. With approximately 25% of the original dose remaining in your system by day 14, the appetite suppression that felt so effortless begins to weaken. Cravings return. Not all at once, but in waves. You might find yourself thinking about food more often, noticing snack commercials, lingering in the kitchen after meals. The phenomenon that many people describe as "food noise," that constant background chatter about what to eat next, begins re-emerging.
Specific cravings tend to follow a pattern. Sweet and carbohydrate-rich foods typically return first. This makes biological sense because ghrelin, the hunger hormone that semaglutide was suppressing, has a particularly strong effect on cravings for calorie-dense foods. Your brain reward circuitry is waking up to food stimuli again, and it tends to reach for the most calorically rewarding options first.
Blood sugar may begin fluctuating during this period, particularly if you have type 2 diabetes or prediabetes. Research suggests blood glucose can rise 20 to 30 mg/dL within the first two weeks after the final dose. This is not dangerous for most people, but it is noticeable. You might feel slightly more fatigued, experience mild headaches, or notice changes in your energy levels throughout the day. Monitoring your health markers during this transition becomes essential.
Weeks 3 through 4, medication clearing
By week three, the vast majority of semaglutide has left your system. By week five, it takes approximately 35 days for complete elimination, only trace amounts remain. This is the period where the full impact of discontinuation becomes apparent.
Appetite has largely returned to pre-treatment levels. The feeling of fullness that came easily with smaller portions is noticeably diminished. Portion sizes that felt perfectly satisfying two months ago now feel inadequate. Gastric emptying has normalized, meaning food moves through your stomach faster, and you feel hungry sooner after meals.
Digestive changes become apparent during this window. Some people experience bloating, increased gas, or changes in bowel habits as their gastrointestinal system readjusts. If semaglutide had been causing nausea or constipation, those side effects resolve. But the rapid normalization of gastric motility can create its own temporary discomfort. Understanding how peptides affect gut health provides context for these digestive shifts.
Metabolically, this is a critical period. Blood sugar regulation is now fully dependent on your own insulin and glucagon production. For people with diabetes, this transition requires close medical monitoring and potentially adjustments to other medications. For those without diabetes, you may notice subtle changes in energy levels, concentration, and how your body handles carbohydrate-heavy meals.
Months 2 through 3, weight regain begins
Here is where the data becomes sobering. Weight regain typically becomes measurable and consistent during months two and three. Research indicates an average weight regain rate of approximately 0.8 kg, about 1.8 pounds, per month after stopping semaglutide. This rate can be higher for individuals who lost more weight during treatment or who did not establish strong lifestyle habits alongside the medication.
The weight does not return uniformly. Visceral fat, the metabolically dangerous fat around internal organs, tends to return more readily than subcutaneous fat. This means the health risks associated with weight regain may be disproportionate to the number on the scale. Monitoring waist circumference alongside weight provides a more complete picture. For context on visceral fat management, research on peptides for visceral fat offers additional perspective on how different compounds target this specific fat deposit.
Psychologically, this period can be the most difficult. Seeing the scale move in the wrong direction after months of progress triggers frustration, disappointment, and sometimes hopelessness. These emotional responses are normal and valid. They are also predictable, which means you can prepare for them.
Months 6 through 18, the long-term trajectory
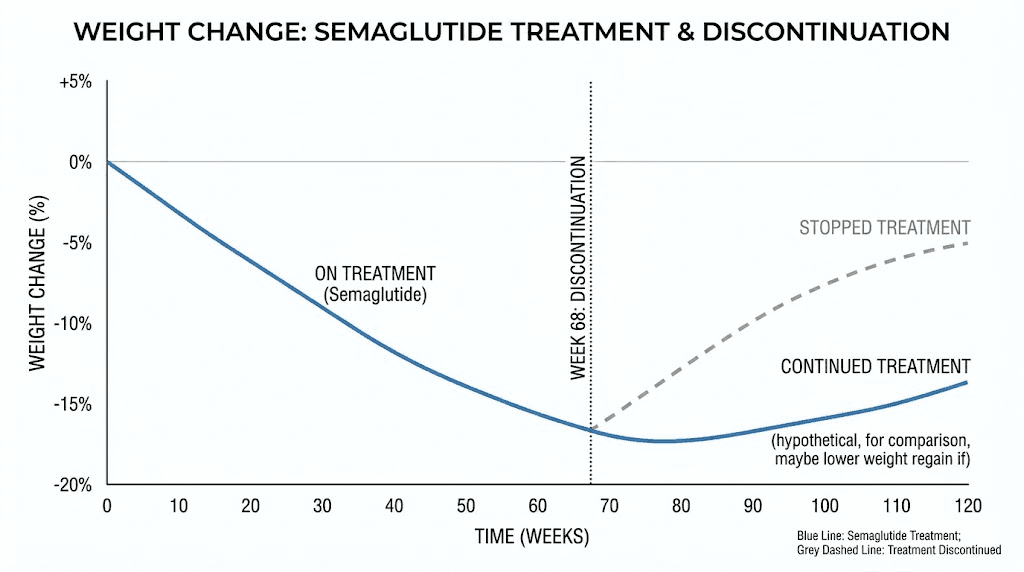
The STEP 1 extension trial provides the clearest picture of long-term outcomes. Participants who had lost an average of 17.3% of their body weight during 68 weeks of treatment regained 11.6 percentage points of that loss by week 120, one year after stopping. The net weight loss from baseline was only 5.6% at week 120, compared to 17.3% at the end of treatment.
Put differently, two-thirds of the weight returned within a year.
Real-world data from the STEP-10 trial shows even more rapid regain in some populations, with over 40% of lost weight returning within just 28 weeks. However, real-world observational data also reveals important nuances. A study using electronic health records found that 17.7% of patients regained all their weight or more, but more than half maintained at least some weight loss after one year. The outcomes vary enormously based on individual behavior, lifestyle modifications, and ongoing support.
The cardiometabolic improvements also reverse. Blood pressure, lipid profiles, inflammatory markers, and blood sugar control all trend back toward pre-treatment levels. The STEP 1 extension documented similar patterns of reversal across all measured cardiovascular risk factors. This underscores the medical community position that obesity is a chronic disease requiring chronic management, whether through medication, lifestyle intervention, or both.
The science behind semaglutide withdrawal
Understanding the biology makes the experience less frightening and more predictable. Semaglutide is a GLP-1 receptor agonist. It mimics the hormone glucagon-like peptide-1, which your body naturally produces in the intestines after eating. But semaglutide is not identical to natural GLP-1. It has been engineered with specific structural modifications that give it dramatically different pharmacokinetic properties.
Why semaglutide lasts so long in the body
Natural GLP-1 has a half-life of approximately 2 minutes. Semaglutide has a half-life of approximately 7 days. That is a 5,040-fold increase. Three structural modifications make this possible. First, a single amino acid substitution at position 8 protects the molecule from degradation by the enzyme DPP-4. Second, a fatty acid chain attached to the molecule allows it to bind tightly to albumin, the most abundant protein in blood plasma. This albumin binding is the principal mechanism of protraction, reducing renal clearance and protecting the molecule from metabolic degradation. Third, additional amino acid modifications further stabilize the molecule.
When you stop injecting semaglutide, the existing molecules in your bloodstream continue working while they are slowly metabolized. The primary elimination pathway involves proteolytic cleavage of the peptide backbone and sequential beta-oxidation of the fatty acid sidechain. Approximately 75% is eliminated through urine and stool. The process takes roughly five half-lives, about 35 days, to reduce circulating levels to less than 3% of peak concentration.
This gradual clearance is actually a blessing. Unlike medications with shorter half-lives that can produce abrupt withdrawal effects, semaglutide provides a built-in tapering period as it naturally leaves your system. The transition is gradual rather than sudden. Understanding how peptides function at the molecular level helps explain why this clearance pattern matters.
GLP-1 receptor mechanisms and the rebound effect
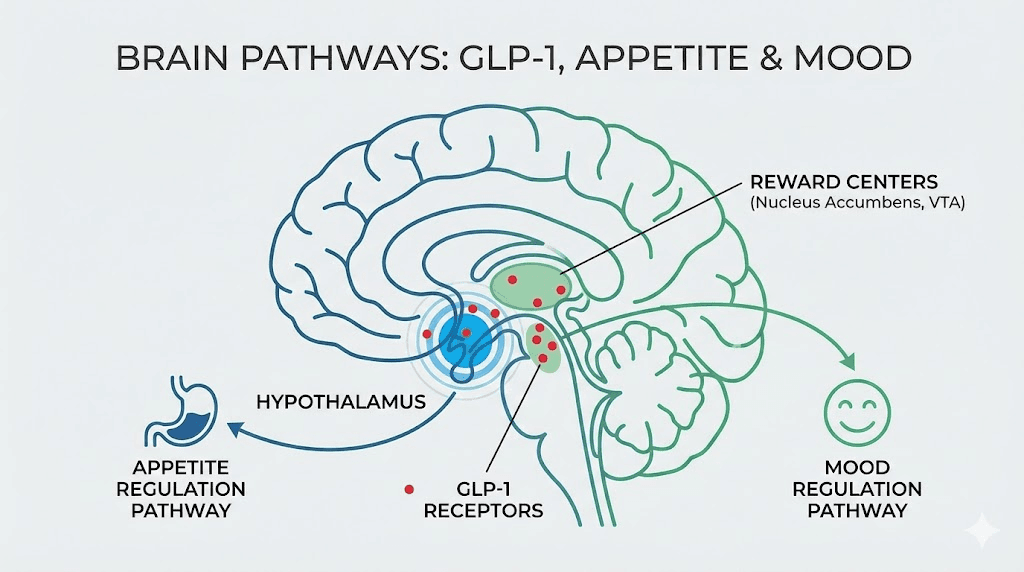
GLP-1 receptors are found throughout the body. They are concentrated in the pancreas, where they stimulate insulin secretion and suppress glucagon release. They are present in the stomach, where they slow gastric emptying. They are abundant in the brain, particularly the hypothalamus and brainstem regions that control appetite, satiety, and food reward processing. They even appear in the heart, kidneys, and other organs.
During semaglutide treatment, these receptors receive continuous stimulation far beyond what natural GLP-1 provides. When treatment stops, the receptors lose that stimulation. But the situation may be worse than simply returning to baseline. Some research suggests that chronic supraphysiological GLP-1 receptor stimulation may lead to receptor desensitization or downregulation, meaning your body might temporarily have a reduced response to even its own natural GLP-1 after stopping the medication.
This potential rebound effect explains why some people report feeling hungrier after stopping semaglutide than they were before starting it. The hunger is not imagined. It reflects genuine neurochemical changes in appetite regulation circuits.
The hunger hormone cascade
Weight loss, regardless of how it is achieved, triggers a cascade of hormonal adaptations designed to restore body weight to its previous set point. These are not psychological weaknesses. They are evolutionary survival mechanisms encoded in our biology over millions of years.
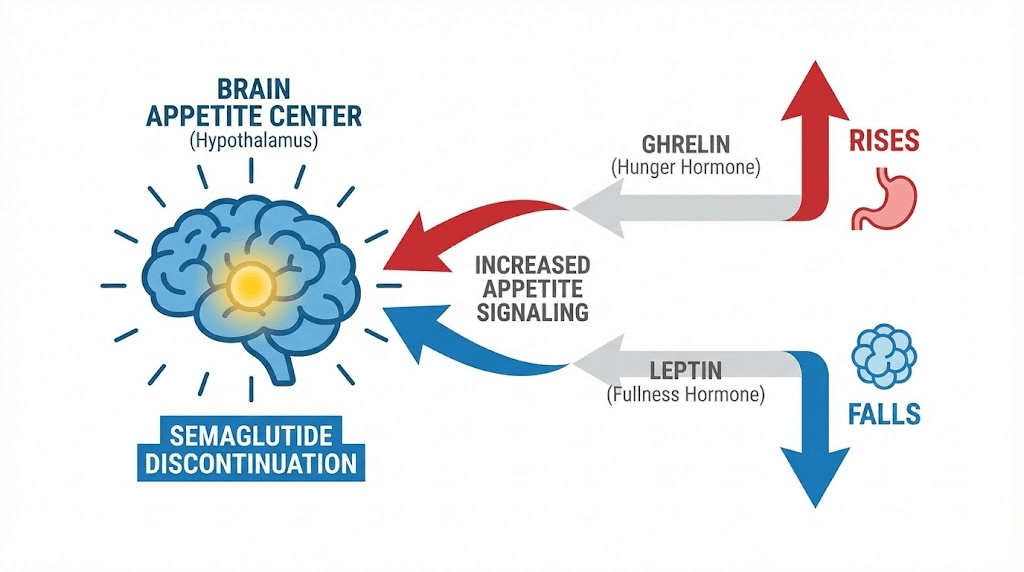
Ghrelin rises. Ghrelin is the primary hunger hormone, produced mainly in the stomach, and it increases after weight loss to stimulate appetite and promote food-seeking behavior. Semaglutide suppresses ghrelin signaling. When semaglutide stops, ghrelin levels rebound, often to levels higher than pre-treatment baseline.
Leptin falls. Leptin is produced by fat cells and signals satiety to the brain. When you lose fat, you produce less leptin, and your brain interprets this as a starvation signal. This reduced leptin drives increased appetite and decreased metabolic rate. The combination is particularly difficult because you are simultaneously hungrier and burning fewer calories.
Peptide YY decreases. This satiety hormone, produced in the gut after eating, tells your brain you are full. Levels drop following weight loss, meaning the fullness signal after meals becomes weaker. Other hormones shift as well, including cholecystokinin, amylin, and pancreatic polypeptide, all nudging the system toward increased food intake and energy conservation. For deeper understanding of these hormonal mechanisms, research on amylin receptor agonists reveals how related pathways interact with GLP-1 signaling.
The net result is a powerful biological drive to regain weight. It is the same mechanism that makes traditional dieting so difficult to sustain long-term. Semaglutide was overriding these signals. Without it, they return in full force.
Weight regain after stopping semaglutide
Weight regain is the most studied and most discussed consequence of semaglutide discontinuation. The data is extensive, coming from randomized controlled trials, extension studies, and real-world observational research. Let us examine what the evidence actually shows.
STEP 1 trial extension data
The STEP 1 trial is the landmark study for understanding post-discontinuation weight regain. During the 68-week treatment phase, participants receiving semaglutide 2.4 mg weekly plus lifestyle intervention lost an average of 17.3% of body weight. The placebo group lost 2.0%.
After treatment was stopped at week 68, participants were followed for an additional 52 weeks. The results were clear and consistent. Semaglutide participants regained an average of 11.6 percentage points of their lost weight. By week 120, the net weight loss from baseline was only 5.6%, down from 17.3% at the end of treatment. That represents a loss of approximately two-thirds of the weight loss achieved during treatment.
The placebo group showed a parallel but smaller rebound. They regained 1.9 percentage points, ending at only 0.1% net loss from baseline at week 120.
Consider what this looks like in practical terms. A person weighing 220 pounds at baseline who lost 17.3% of body weight during treatment would have reached approximately 182 pounds. After one year off the medication, that person would likely weigh around 208 pounds, having regained about 26 of the 38 pounds lost. Understanding how peptides support weight management provides broader context for why sustained intervention matters.
STEP-10 and other trial data
The STEP-10 trial documented even more rapid weight regain in some scenarios, with over 40% of lost weight returning within just 28 weeks, roughly six months, of discontinuation. This faster regain may reflect differences in study populations, lifestyle support, or other factors, but it underscores the variability in outcomes.
A systematic review and meta-analysis published in eClinicalMedicine confirmed the pattern across multiple studies. The pooled data showed a mean weight regain of approximately 9.69 kg among semaglutide and tirzepatide users after discontinuation, with near-complete reversal of prior improvements within 52 weeks. For comparison, liraglutide users showed a smaller pooled regain of 2.20 kg, likely reflecting the lower weight loss achieved during treatment.
Real-world data tells a more nuanced story
Clinical trials control variables tightly. Real-world data reveals more variability and, in some cases, more encouraging outcomes. A large-scale analysis using electronic health records found that weight regain after GLP-1 discontinuation was less rapid in clinical practice than in controlled trials.
The numbers paint a mixed picture. About 17.7% of patients regained all their weight or more within one year of stopping. That is concerning. But more than half maintained at least some degree of weight loss after one year. And a meaningful subset maintained most of their weight loss, particularly those who had established strong exercise habits, dietary modifications, and behavioral changes during treatment.
The rate of regain also varied. The average was approximately 0.8 kg per month, about 1.8 pounds, but individual trajectories ranged widely. Some people regained rapidly in the first few months and then stabilized. Others maintained their weight for several months before gradual regain began. A smaller group maintained their weight loss indefinitely, suggesting that for some individuals, the treatment period may be sufficient to reset behavioral patterns and metabolic function.
These individual differences highlight why a one-size-fits-all approach to semaglutide discontinuation does not work. Your outcome depends heavily on what you do during and after the transition. For people exploring the best peptide options for weight management, understanding these patterns helps set realistic expectations and inform strategy.
Why does the weight come back so aggressively
Three factors converge to drive weight regain after semaglutide discontinuation.
First, the hormonal rebound described earlier creates a powerful biological drive to eat more. Ghrelin rises, leptin falls, and the entire appetite regulation system shifts toward energy acquisition and storage. This is not about willpower. This is endocrinology.
Second, metabolic adaptation reduces energy expenditure. Weight loss decreases resting metabolic rate beyond what would be expected from the reduction in body mass alone. This phenomenon, sometimes called adaptive thermogenesis or metabolic adaptation, means your body burns fewer calories than a person of the same size who was never heavier. Semaglutide may partially counteract metabolic adaptation through its effects on energy expenditure, and when it is removed, the full extent of adaptation becomes apparent.
Third, behavioral patterns may revert. The medication made it easier to eat less, resist cravings, and maintain portion control. Without that pharmacological support, maintaining the same behaviors requires significantly more conscious effort. For many people, the cognitive load of constant food vigilance becomes unsustainable over time, and old patterns gradually return.
Understanding these mechanisms is not about resignation. It is about preparation. When you know what you are fighting against, you can develop targeted strategies for each challenge. And people who actively work on managing weight loss through multiple approaches tend to maintain more of their progress than those who relied solely on the medication.
Blood sugar and metabolic changes after discontinuation
Weight regain dominates the conversation about semaglutide discontinuation, but the metabolic changes are equally important and, for some individuals, more medically significant. Semaglutide was originally developed as a diabetes medication, and its effects on blood sugar, insulin sensitivity, and cardiovascular risk factors are substantial. When treatment stops, these benefits begin reversing.
Blood glucose and A1C changes
Semaglutide improves blood sugar control through multiple mechanisms. It enhances glucose-dependent insulin secretion from the pancreas. It suppresses inappropriate glucagon release. It slows gastric emptying, which reduces postprandial glucose spikes. And it promotes weight loss, which improves insulin sensitivity independently.
When you stop semaglutide, all of these mechanisms cease. The result is a predictable rise in blood glucose levels. Research suggests blood glucose can increase by 20 to 30 mg/dL within the first two weeks after the final dose. For someone with well-controlled type 2 diabetes, this could mean the difference between an A1C of 6.5% and one above 7.0%. For someone with prediabetes, it could mean crossing the threshold into the diabetic range.
The STEP 1 extension documented that improvements in A1C levels achieved during treatment reversed substantially after discontinuation. The reversal was not complete in all participants, likely because some maintained lifestyle modifications that independently improved insulin sensitivity, but the trend was clear and consistent across the study population.
If you have type 2 diabetes, discontinuing semaglutide requires careful medical management. Your healthcare provider may need to adjust other diabetes medications, increase monitoring frequency, or implement alternative treatment strategies. This is not a transition to manage alone. The safety considerations involved with peptide use extend to discontinuation planning as well.
Cardiovascular risk factor changes
The metabolic improvements from semaglutide extend well beyond blood sugar. Treatment is associated with reductions in blood pressure, improvements in lipid profiles including LDL cholesterol and triglycerides, decreases in inflammatory markers like C-reactive protein, and improvements in waist circumference, a marker of cardiovascular risk.
The STEP 1 extension showed that these improvements reversed in parallel with weight regain. Blood pressure increased. Lipid profiles worsened. Inflammatory markers rose. Waist circumference expanded. The cardiometabolic risk profile shifted back toward its pre-treatment state.
This is particularly important for individuals who were prescribed semaglutide specifically for cardiovascular risk reduction. The SELECT trial demonstrated that semaglutide reduces major adverse cardiovascular events in patients with obesity and established cardiovascular disease. Discontinuing treatment in this population requires a careful risk-benefit discussion with a cardiologist or endocrinologist.
Insulin resistance and metabolic rate
Insulin resistance tends to worsen after semaglutide discontinuation, driven by both weight regain and the loss of direct metabolic effects from GLP-1 receptor stimulation. The pancreatic beta cells, which received sustained support from semaglutide, must now manage blood sugar regulation independently. For people with compromised beta cell function, this can lead to more significant blood sugar elevation.
Metabolic rate, already depressed by weight loss-induced adaptive thermogenesis, may drop further as weight is regained. This creates a frustrating paradox where you need fewer calories to maintain a higher weight than you did before you started the medication. The body essentially becomes more metabolically efficient, which from an evolutionary standpoint is a feature, not a bug, but from a weight management standpoint is deeply counterproductive.
There is one piece of encouraging news regarding body composition. Research suggests that lean mass and skeletal muscle tend to recover following treatment discontinuation. While fat mass increases, the muscle mass that may have been lost during rapid weight loss on semaglutide can be rebuilt, particularly with resistance training. This argues strongly for incorporating strength training into your post-semaglutide lifestyle plan. The relationship between weight loss and muscle preservation through peptide approaches highlights why body composition matters more than scale weight alone.
Digestive system changes when stopping semaglutide
Your gastrointestinal system undergoes significant recalibration after semaglutide discontinuation. The medication profoundly alters gut motility, gastric emptying, and digestive hormone signaling. When these effects reverse, you will notice changes in how food moves through your system, how much you can eat comfortably, and how your digestion feels day to day.
Gastric emptying normalization
One of semaglutide primary mechanisms for appetite suppression is delayed gastric emptying. Food sits in your stomach longer, creating sustained feelings of fullness and reducing the desire to eat. When semaglutide clears your system, gastric emptying returns to its normal rate.
This change is noticeable. Meals that previously kept you satisfied for four or five hours may now leave you hungry within two to three hours. The sensation of fullness after eating diminishes. You may find that you can comfortably eat larger portions than you could on the medication, not because your stomach has physically expanded, but because food is moving through it at normal speed again.
The transition can cause temporary digestive discomfort. Some people experience bloating, mild nausea, or irregular bowel movements as motility patterns readjust. These symptoms are typically transient, resolving within one to three weeks as the gut establishes its new baseline.
Nausea resolution and other GI improvements
If you experienced gastrointestinal side effects while on semaglutide, stopping brings welcome relief. Nausea, the most common side effect during treatment, typically resolves completely within days of the medication clearing your system. Constipation, another frequent complaint, usually improves as well. Vomiting episodes cease. Acid reflux may decrease.
For some individuals, this GI relief is one of the primary motivations for discontinuing treatment. The trade-off between effective weight management and persistent nausea or constipation is a personal calculation that each individual must make in consultation with their healthcare provider. If GI side effects were driving your decision to stop, know that these will resolve. The challenge is managing the other consequences of discontinuation. Detailed guidance on supporting gut health through peptide research may provide additional insights for your transition.
Appetite and satiety signal changes
The gut produces numerous hormones that communicate with the brain about nutritional status. Semaglutide amplifies some of these signals and dampens others. When treatment stops, the entire gut-brain communication system recalibrates.
Cholecystokinin, released from the small intestine in response to fat and protein, returns to its pre-treatment signaling pattern. Peptide YY, which suppresses appetite in the postprandial period, decreases. The incretin effect, where gut hormones enhance insulin secretion in response to oral glucose, returns to baseline. Each of these changes subtly shifts the balance toward increased food intake.
You may notice that the types of foods you crave change as well. On semaglutide, many people report reduced interest in fatty, sweet, and highly processed foods. This effect is mediated partly through GLP-1 receptors in the brain reward circuitry. When semaglutide clears, the reward value of these foods increases again. A pizza that tasted mediocre on the medication suddenly tastes incredible. A dessert that held no appeal becomes irresistible. This is not psychological weakness. It is a neurochemical shift in reward processing.
Psychological and mood effects of semaglutide withdrawal
The psychological dimension of semaglutide discontinuation deserves far more attention than it typically receives. While the physical changes are well documented in clinical literature, the emotional and cognitive effects are just as real, just as impactful, and often more distressing for the people experiencing them.
The return of food noise
Food noise. Two words that did not exist in clinical vocabulary before GLP-1 medications entered mainstream use. The term describes the constant mental preoccupation with food, that running background commentary about what to eat next, when to eat, what sounds good, what is in the refrigerator. For many people with obesity, food noise is a defining feature of daily life. And for many semaglutide users, the quieting of this noise was the most transformative effect of the medication.
When semaglutide stops, food noise returns. Sometimes gradually. Sometimes all at once. The quiet mind you experienced on the medication gives way to a renewed preoccupation with food that can feel overwhelming, especially after experiencing what life felt like without it. Some people describe it as going back into a room with a persistent buzzing sound that you had forgotten was there until it stopped and then started again.
This return of food noise is rooted in neurobiology. GLP-1 receptors in the hypothalamus, nucleus accumbens, and other brain regions modulate appetite salience, essentially how important food appears in your mental hierarchy of concerns. When semaglutide stimulates these receptors, food drops lower on the priority list. When stimulation ceases, it rises again. Understanding how semaglutide affects appetite suppression helps clarify why this reversal occurs.
Mood changes and emotional effects
Research on the psychological effects of semaglutide discontinuation is still emerging, but several patterns have been documented. Some individuals report mood changes after stopping, including increased irritability, mild anxiety, and in some cases, depressive symptoms. These effects appear to be mediated through multiple pathways.
GLP-1 receptors are present in brain regions that regulate mood, not just appetite. Semaglutide may have direct mood-modulating effects through these receptors. Additionally, the emotional impact of seeing weight regain after working hard to lose it can trigger grief, frustration, and feelings of failure. The loss of the sense of control that the medication provided can be psychologically destabilizing.
There is an important distinction to make here. The FDA has conducted extensive evaluation and concluded that GLP-1 receptor agonists do not increase the risk of suicidality or serious mental health events. The mood changes associated with discontinuation are typically mild to moderate and transient. They are uncomfortable but not dangerous for most people.
However, if you have a history of depression, anxiety, or other mental health conditions, discuss your discontinuation plan with both your prescribing physician and your mental health provider. The transition period warrants closer monitoring for individuals with pre-existing psychological vulnerability. Resources on peptides and their relationship to mood regulation provide additional scientific context on how these pathways interact.
Identity and self-concept shifts
This is rarely discussed in clinical literature but frequently reported in patient communities. Many semaglutide users undergo significant identity shifts during treatment. They develop new relationships with food. They adopt new eating patterns. They may change their social circles, wardrobe, activity levels, and self-image. Some describe feeling like a different person.
Discontinuation can threaten this new identity. As appetite returns and weight begins to regain, the fear of "going back to who I was" can trigger significant psychological distress. This is compounded by cultural messaging that ties weight to moral worth and personal discipline.
Acknowledging these feelings without judgment is important. The changes you made while on semaglutide are real. The habits you developed are real. The knowledge you gained about nutrition, portion control, and your body are real. Those things do not disappear because the medication does. They may be harder to implement without pharmacological support, but they remain available to you.
Fatigue and energy changes
Many people report increased fatigue in the weeks following semaglutide discontinuation. This likely stems from multiple sources. Blood sugar fluctuations can cause energy dips. The metabolic shift toward energy conservation reduces overall vitality. Sleep disruption from mood changes or anxiety can compound the issue. And the psychological burden of managing increased appetite and food cravings creates a form of decision fatigue that is genuinely exhausting.
This fatigue typically improves as the body adjusts to its new metabolic baseline, usually within four to eight weeks of the last dose. In the meantime, prioritizing sleep quality, maintaining consistent meal timing, and avoiding dramatic caloric restriction can help stabilize energy levels. Understanding how long peptide effects persist after discontinuation provides useful context for setting timeline expectations.
How to safely taper off semaglutide
While semaglutide can be stopped abruptly without medical danger, growing evidence suggests that a gradual taper may produce better long-term outcomes. The rationale is straightforward. A slow reduction gives your body time to adjust to decreasing levels of GLP-1 receptor stimulation, allows you to practice appetite management with diminishing pharmaceutical support, and provides a more psychologically manageable transition.
Tapering evidence and protocols
Research presented at the European Congress on Obesity examined outcomes in patients who tapered semaglutide over an average of nine weeks before discontinuation. The results were striking. Participants who tapered lost approximately 15% of body weight during treatment and maintained a stable weight for at least 26 weeks, roughly six months, after stopping the medication. This stands in sharp contrast to the rapid regain typically seen with abrupt discontinuation.
A practical tapering protocol based on current clinical practice might look like this. If you are on the full 2.4 mg weekly dose, the first step would be reducing to 1.7 mg for four weeks. Then dropping to 1.0 mg for another four weeks. Then moving to 0.5 mg for a final four weeks before stopping entirely. The total taper period spans approximately nine to twelve weeks.
Some clinicians extend the taper even further, particularly for patients who have been on the medication for extended periods or who lost significant amounts of weight. A slower taper of 12 to 16 weeks may be appropriate for patients at higher risk of rapid weight regain. The key is that each dose reduction should be accompanied by a period of stability where you confirm that your appetite, blood sugar, and weight remain manageable at the new dose before reducing further.
Not every clinical situation allows for gradual tapering. Insurance changes, medication shortages, and financial constraints sometimes force abrupt discontinuation. If you must stop without tapering, the strategies outlined in the maintenance section become even more critical. Using resources like the semaglutide dosage calculator can help you understand the dosing levels during a structured taper.
Monitoring during the taper
Active monitoring during the tapering process helps identify problems before they become serious. At each dose reduction, track the following metrics.
Weight. Check weekly, at the same time, under the same conditions. A gain of more than 2 to 3 pounds at any step may warrant slowing the taper.
Appetite levels. Rate your hunger on a 1 to 10 scale three times daily, before each meal. Significant increases suggest the current dose reduction is being felt and you may need more time at this level before reducing further.
Blood glucose. If you have diabetes or prediabetes, check fasting glucose daily and post-meal glucose at least twice weekly. Rising levels indicate the need for potential medication adjustments.
Mood and energy. Note any changes in sleep quality, motivation, irritability, or emotional well-being. These soft metrics matter and can indicate that the transition is moving too quickly.
Digestive symptoms. Track any bloating, changes in bowel habits, or nausea patterns. These typically stabilize at each dose level within one to two weeks. Comprehensive peptide dosage reference charts can support tracking alongside your taper protocol.
Working with your healthcare provider
Tapering off semaglutide should not be a solo project. Your healthcare provider can adjust the taper based on your individual response, modify other medications if needed, order laboratory tests to monitor metabolic changes, and provide support and accountability throughout the process.
Before your tapering appointment, prepare by documenting your current dose and how long you have been on it, your total weight loss on the medication, your reasons for discontinuing, any other medications you take, your exercise and dietary habits, and your support system. This information helps your provider create a personalized tapering plan that accounts for your specific circumstances and risk factors. Learning about online peptide therapy options may also help you understand the full landscape of available support during your transition.
Strategies to maintain weight loss after stopping semaglutide
The data on weight regain is real, but it is not destiny. Research consistently shows that individuals who combine medication discontinuation with aggressive lifestyle modification maintain more of their weight loss than those who rely on willpower alone. The following strategies are evidence-based approaches that target the specific mechanisms driving post-semaglutide weight regain.
Exercise, the single most important factor
If you implement only one strategy from this entire guide, make it this one. Exercise.
Research indicates that maintaining weight loss after GLP-1 discontinuation requires approximately 250 or more minutes per week of moderate-intensity physical activity. That is significantly more than the 150 minutes recommended for general health. It translates to roughly 35 to 40 minutes of activity every day, or 50 minutes five days a week.
But not all exercise is equal for this purpose. The optimal approach combines three modalities.
Resistance training builds and preserves lean muscle mass, which is the primary driver of resting metabolic rate. More muscle means you burn more calories at rest. During semaglutide treatment, rapid weight loss can include muscle loss along with fat loss. Resistance training during and after treatment helps restore this lean mass. Aim for two to three sessions per week targeting all major muscle groups. Understanding how peptides support muscle growth provides useful context for why muscle preservation matters during weight transitions.
Aerobic exercise burns calories directly and improves cardiovascular fitness, insulin sensitivity, and mood. It also has a direct effect on GLP-1 production. Research shows that exercise combined with GLP-1 receptor agonist use produces a 25% increase in late-phase postprandial GLP-1 response. Even after stopping the medication, regular aerobic exercise helps maintain higher natural GLP-1 levels, partially offsetting the loss of pharmaceutical stimulation. For context on how activity supports fat loss specifically, research on optimizing fat loss provides complementary strategies.
High-intensity interval training offers metabolic benefits beyond steady-state cardio, including greater excess post-exercise oxygen consumption (EPOC), improved insulin sensitivity, and favorable hormonal responses. Two to three HIIT sessions per week, interspersed with lower-intensity activity, provides an excellent foundation.
The data on exercise and GLP-1 maintenance is compelling. Studies show that keeping up with exercise after stopping GLP-1 receptor agonists was associated with significantly less weight regain compared to those who became sedentary. Performance-focused peptide research continues to explore how different compounds can support exercise adaptations.
Nutrition strategies that mimic semaglutide effects
You cannot fully replicate semaglutide effects through diet alone, but you can partially mimic them. The goal is to create the same feeling of fullness and satisfaction that the medication provided, using food choices and timing rather than pharmacology.
Protein is your most powerful tool. It is the most satiating macronutrient, meaning it keeps you feeling full longer per calorie than carbohydrates or fat. Aim for 25 to 35 grams of protein at every meal. This typically means including a palm-sized portion of meat, fish, eggs, or legumes at each sitting. High-protein meals also have a higher thermic effect, meaning your body burns more calories digesting protein than other macronutrients.
Fiber comes second. High-fiber foods, including vegetables, fruits, whole grains, and legumes, add volume to meals without adding proportional calories. They slow gastric emptying, partially mimicking one of semaglutide primary mechanisms. They also feed beneficial gut bacteria that produce short-chain fatty acids, which have been shown to support satiety signaling. Aim for 30 or more grams of fiber daily.
Meal timing matters. Eating at consistent times helps regulate hunger hormones and reduces the likelihood of impulsive eating. Front-loading calories, eating a larger breakfast and lunch with a smaller dinner, aligns with circadian metabolic rhythms and may improve weight maintenance.
Volume eating, the practice of choosing foods with high water content and low caloric density, can help bridge the gap between reduced pharmaceutical satiety and the amount of food needed to feel physically full. Large salads with lean protein, vegetable-based soups, and roasted vegetable medleys are practical applications of this approach.
Tracking what you eat, at least temporarily, provides accountability and awareness. A food journal or tracking app can reveal unconscious caloric creep that often accompanies the return of appetite. Many common mistakes in peptide and weight management protocols stem from inadequate monitoring during transition periods.
Sleep and stress management
Sleep deprivation and chronic stress are independent drivers of weight gain, and they become particularly problematic during the post-semaglutide period when appetite regulation is already compromised.
Poor sleep increases ghrelin and decreases leptin, exactly the hormonal profile you are trying to counteract. It impairs glucose regulation, reduces executive function needed for food decisions, and increases cortisol, which promotes visceral fat storage. Aim for seven to eight hours of quality sleep per night. Consistent sleep and wake times, a cool dark bedroom, limited screen exposure before bed, and avoidance of caffeine after early afternoon all support sleep quality.
Chronic stress elevates cortisol, which directly promotes fat storage, particularly visceral fat, and drives stress-eating behaviors. Stress management is not a luxury during this transition period. It is a medical necessity. Regular exercise serves double duty here, reducing both stress and weight regain risk. Mindfulness practices, social support, time in nature, and limiting overcommitment all contribute to a lower stress baseline.
Behavioral and cognitive strategies
On semaglutide, healthy eating felt automatic. Without it, you need to make it manual. That means developing systems, habits, and environmental controls that support your goals when willpower fluctuates.
Meal prep removes decision fatigue. When healthy meals are already prepared, the barrier to eating well drops dramatically. Dedicate a few hours weekly to preparing protein, vegetables, and grains that can be quickly assembled into meals.
Environmental design matters. Remove or reduce access to highly palatable, calorie-dense foods in your home and workplace. This is not about restriction. It is about reducing the number of times per day you need to exercise willpower against a craving. Every food decision you do not have to make conserves cognitive resources for the decisions you cannot avoid.
Accountability structures help. Whether it is a dietitian, a support group, a workout partner, or a spouse who understands your goals, having someone to answer to improves adherence to lifestyle changes. The peptide community forums can provide peer support from others navigating similar transitions.
Mindful eating practices rebuild your relationship with hunger and fullness cues. On semaglutide, the medication handled satiety signaling. Off the medication, you need to re-learn how to recognize and respond to your body natural hunger and fullness signals. Eating slowly, without screens, paying attention to taste and texture, and checking in with your hunger level midway through a meal are all practical techniques.

When to consider restarting semaglutide
Stopping semaglutide does not have to be permanent. For many individuals, the evidence supports long-term or intermittent use as part of a chronic disease management strategy. Understanding when restarting makes clinical sense, and when other approaches may be more appropriate, helps you make informed decisions about your ongoing treatment.
Signs that restarting may be appropriate
Significant weight regain despite maximum lifestyle efforts is the clearest indication. If you have implemented all the strategies outlined above, maintained 250 or more minutes of weekly exercise, optimized nutrition, managed sleep and stress, and still find weight rapidly returning, the biology may simply be stronger than behavior alone can manage. This is not failure. It is the chronic nature of obesity asserting itself.
Worsening metabolic markers are another strong signal. If blood glucose, A1C, blood pressure, or lipid profiles deteriorate meaningfully after discontinuation, the health risks of remaining off medication may outweigh the reasons for stopping. Your healthcare provider can help you evaluate these trade-offs objectively.
Deteriorating quality of life matters too. If the return of food noise, constant cravings, and the psychological burden of weight management is significantly impacting your daily functioning, relationships, or mental health, restarting medication is a legitimate and compassionate choice.
The return of obesity-related complications, including joint pain from weight regain, worsened sleep apnea, reduced mobility, or return of GERD symptoms, all suggest that the medication was providing meaningful health benefits beyond weight loss.
The case for long-term treatment
The medical community increasingly recognizes that obesity, like hypertension or type 2 diabetes, may require indefinite treatment for optimal outcomes. Just as stopping blood pressure medication leads to blood pressure rising again, stopping obesity medication leads to weight regain. This parallel has shifted the conversation from "how long should I take this" to "what is the right long-term management strategy."
For some individuals, that strategy involves continuous semaglutide use at the lowest effective dose. For others, it may involve cycling, periods of treatment followed by planned discontinuation and lifestyle-intensive maintenance phases, then restarting when needed. For others still, it may involve switching to a different medication or combination approach. Exploring peptide cycling strategies provides a framework for thinking about intermittent treatment protocols.
Restarting after a break
If you decide to restart semaglutide after a period of discontinuation, you will likely need to re-titrate from the starting dose. Your body GLP-1 receptor sensitivity will have changed during the medication-free period, and jumping back to a high dose can cause the same GI side effects you experienced when you first started treatment.
A typical restart protocol involves beginning at 0.25 mg weekly for four weeks, then 0.5 mg for four weeks, then 1.0 mg for four weeks, then 1.7 mg for four weeks, before reaching the maintenance dose of 2.4 mg. Your provider may modify this schedule based on your tolerance and previous treatment history.
The good news is that semaglutide typically works just as well the second time around. Response rates are similar to initial treatment, and most individuals achieve significant weight loss again upon restarting. The medication does not lose effectiveness through prior exposure. Using the semaglutide dosing reference guides can help you track your re-titration progress accurately.
Semaglutide alternatives for continued weight management
If you are stopping semaglutide but want to continue pharmacological support for weight management, several alternatives exist. Each has different mechanisms, efficacy profiles, and side effect considerations. Understanding the landscape helps you have informed conversations with your healthcare provider about what comes next.
Tirzepatide, the dual agonist approach
Tirzepatide targets two receptors: GLP-1 and GIP (glucose-dependent insulinotropic polypeptide). This dual mechanism has shown superior weight loss compared to semaglutide in head-to-head trials. The SURMOUNT trials demonstrated average weight loss of approximately 22% at the highest dose, compared to roughly 17% with semaglutide.
For individuals switching from semaglutide to tirzepatide, the transition can provide continued or even enhanced weight management. The dual receptor mechanism may address aspects of appetite regulation and metabolic function that semaglutide alone does not fully capture. However, tirzepatide carries similar GI side effects and requires the same gradual dose titration. Detailed information on tirzepatide dosing protocols covers the specifics of initiating treatment, and a comprehensive semaglutide versus tirzepatide comparison helps clarify the differences between these two options.
Microdosing tirzepatide is gaining attention as a maintenance strategy for individuals who achieved their weight loss goals on semaglutide and want to maintain without full-dose treatment. A microdosing tirzepatide reference outlines how lower doses might support maintenance with fewer side effects.
Retatrutide, the triple agonist frontier
Retatrutide represents the next evolution in obesity pharmacology, targeting three receptors: GLP-1, GIP, and glucagon. Phase 2 clinical trials showed extraordinary weight loss of up to 28.7% at the highest dose over 48 weeks. This triple receptor approach addresses energy intake (GLP-1 and GIP) and energy expenditure (glucagon) simultaneously.
Retatrutide is not yet approved for clinical use, but its clinical development is advancing rapidly. For individuals who found semaglutide effective but insufficient, or who are looking ahead to potential future treatment options, retatrutide represents the most promising compound in the pipeline. The retatrutide versus semaglutide comparison provides a detailed breakdown of how these compounds differ. For dosing specifics, the retatrutide dosage chart and dosage calculator offer practical reference points.
Cagrilintide and combination approaches
Cagrilintide is a long-acting amylin analog that works through different mechanisms than GLP-1 agonists. Amylin, naturally produced by the pancreas alongside insulin, reduces appetite, slows gastric emptying, and suppresses glucagon secretion through pathways distinct from GLP-1. The combination of cagrilintide with semaglutide, known as CagriSema, has shown particularly promising results in clinical trials, with weight loss exceeding what either compound achieves alone.
For individuals stopping semaglutide, understanding the role of amylin in appetite regulation opens doors to alternative or complementary approaches. Resources on cagrilintide for weight management and cagrilintide dosing protocols provide detailed information. The combination approach outlined in the CagriSema dosing guide may represent the future of obesity pharmacotherapy. Information on how cagrilintide works alongside semaglutide explains the synergistic mechanisms.
Other GLP-1 alternatives
Beyond the newest compounds, several established options exist for continued weight management. The comprehensive guide to Ozempic alternatives covers the full landscape, including oral formulations that some individuals prefer over injections. The oral tirzepatide guide explores one such option. For an even broader perspective, the emerging research on GLP-3 peptides represents an entirely different approach to metabolic regulation that is being explored in early-stage studies.
Peptide stacking for maintenance
Some individuals explore combining different peptides to maintain weight loss results after stopping semaglutide. This approach requires careful planning and medical supervision, but can potentially address multiple aspects of weight management simultaneously. The peptide stacking guide covers the principles of combining compounds, while the peptide stack calculator helps with practical planning. For weight loss specifically, optimized peptide stacks for weight management outline evidence-based combinations.
Understanding the broader landscape of what peptides are and what they are used for provides the foundational knowledge needed to evaluate these options critically. The latest peptide research and clinical studies continue to expand our understanding of how these compounds can support long-term weight management.
Lifestyle-only maintenance, is it enough
For some individuals, lifestyle modification alone can maintain weight loss after semaglutide. The research identifies characteristics of those most likely to succeed without ongoing medication. They typically had moderate weight loss goals. They established strong exercise habits during treatment, consistently hitting 250 or more minutes per week. They made permanent dietary changes rather than temporary restrictions. They had strong social support networks. And they had metabolic health that was less severely compromised at baseline.
If this describes you, a medication-free approach is reasonable. Build your strategy around the pillars discussed earlier, high protein nutrition, regular exercise including resistance training, adequate sleep, and stress management, and monitor your weight and metabolic markers closely. If the trajectory starts trending in the wrong direction despite maximum effort, know that returning to medication remains an option. Tracking your results, as described in peptide before and after documentation guides, helps objectively evaluate your progress.
Regardless of which path you choose, working with qualified healthcare providers who understand peptide therapy ensures your decisions are medically informed. The cost considerations outlined in the peptide therapy cost guide and peptide cost calculator can help with practical financial planning for ongoing treatment.

Practical preparation checklist for stopping semaglutide
Preparation is the difference between a controlled transition and an overwhelming one. The following checklist, organized by timing, gives you a concrete action plan for managing semaglutide discontinuation.
Four weeks before your last dose
Schedule a comprehensive appointment with your healthcare provider. Discuss your tapering plan, establish monitoring protocols, and adjust any concurrent medications. Get baseline labs including fasting glucose, A1C, lipid panel, and any other relevant markers. These baselines allow you to track changes objectively during the transition.
Begin increasing protein intake to 1.0 to 1.2 grams per kilogram of body weight daily if you have not already. Start or intensify your resistance training program. If you are not currently exercising at 250 minutes per week, begin building toward that target now, while the medication is still supporting your appetite and energy.
Set up your tracking systems. Whether you use an app, a journal, or a spreadsheet, create a daily tracking method for weight, food intake, exercise, sleep, mood, and appetite levels. These data points become invaluable for identifying trends and making adjustments early.
During the taper or first two weeks off medication
Stock your kitchen strategically. Remove or reduce highly palatable, calorie-dense processed foods. Replace with high-protein options, pre-cut vegetables, soups, and meals that require minimal preparation. Make the healthy choice the easy choice.
Establish a meal prep routine if you do not have one. Prepare three to four days of meals at a time. Focus on protein-rich, high-fiber, volume-based meals that promote satiety without excessive calories. Pre-portioning meals removes the decision-making burden when cravings strike.
Identify your support network. Tell your partner, family, close friends, or support group about your transition. Ask for specific help, whether that means not bringing junk food into the house, joining you for walks, or simply being understanding if you are irritable. Connecting with communities like the peptide research community forums provides peer support from others navigating similar challenges.
Weeks 2 through 8, the critical transition window
Monitor weight weekly but do not react to daily fluctuations. A gain of one to two pounds per week warrants attention. More than that warrants a conversation with your healthcare provider about whether the transition is manageable or whether additional intervention is needed.
Check blood glucose regularly if you have diabetes or prediabetes. Report any fasting glucose consistently above 130 mg/dL or post-meal glucose above 200 mg/dL to your provider promptly.
Practice active appetite management. When cravings hit, implement a 15-minute delay. Drink water. Go for a short walk. Call a friend. Many cravings pass within 15 to 20 minutes if you can ride them out. If the craving persists, eat something protein-rich and portion-controlled rather than reaching for the most readily available calorie-dense option.
Maintain your exercise routine even when motivation dips. The fatigue and mood changes common in this period can make exercise feel harder. Do it anyway. Even a shortened workout provides metabolic, hormonal, and psychological benefits that compound over time.
Ongoing maintenance
Follow up with your healthcare provider at six weeks, three months, and six months post-discontinuation. These appointments allow for objective assessment of metabolic markers and weight trajectory, medication adjustments if needed, and early intervention if weight regain becomes significant.
Continue tracking key metrics. Weight, waist circumference, exercise frequency, and general well-being provide the data needed to evaluate your trajectory and make proactive adjustments. People who monitor consistently maintain more weight loss than those who stop tracking. Thorough guidance on managing your overall peptide and health optimization journey reinforces the importance of systematic, evidence-based approaches.
Specific populations and special considerations
Not everyone faces the same risks or challenges when stopping semaglutide. Several specific populations warrant additional discussion because their experiences and needs differ meaningfully from the general population.
Women and hormonal considerations
Women may experience semaglutide withdrawal differently due to the interplay between metabolic hormones and reproductive hormones. Estrogen, progesterone, and their fluctuations throughout the menstrual cycle influence appetite, weight distribution, and metabolic rate. Stopping semaglutide during certain phases of the menstrual cycle may produce more pronounced appetite rebound.
For perimenopausal and menopausal women, the challenges are compounded. Declining estrogen levels independently promote visceral fat accumulation, reduce metabolic rate, and increase appetite. Removing semaglutide support during this hormonal transition creates a double challenge. Resources on peptide approaches for menopause-related weight management and peptide options during perimenopause offer targeted guidance. Broader information on peptide approaches for women over 40 and safe peptide options for women provides additional context. The intersection of peptides and hormone balance is particularly relevant for women navigating this transition.
For women specifically considering weight management options, the peptides for weight loss in women guide addresses the unique physiological considerations that apply.
People with type 2 diabetes
Stopping semaglutide when you have type 2 diabetes requires the most careful medical management of any population. The medication may have been controlling your blood sugar as its primary function, with weight loss as a secondary benefit. Removing it without adequate replacement therapy can lead to significant hyperglycemia.
Your endocrinologist should develop a comprehensive transition plan that includes alternative glucose-lowering medications, increased blood sugar monitoring frequency, potential insulin adjustments, dietary modifications optimized for glycemic control, and clear parameters for when to seek urgent care. This is not a transition to navigate with internet advice alone. Professional medical guidance is essential.
Men, body composition, and muscle
Men stopping semaglutide often have specific concerns about muscle mass, body composition, and physical performance. The muscle loss associated with rapid weight loss on GLP-1 agonists can be particularly frustrating for men who value strength and physical capability.
The encouraging news is that research suggests lean mass and skeletal muscle recover following treatment discontinuation, particularly with resistance training. Prioritizing strength training, adequate protein intake of 1.2 to 1.6 grams per kilogram body weight, and potentially incorporating safe peptides for muscle growth support can accelerate the restoration of lean mass. The complete guide to peptides for men covers the full spectrum of options available, while fat-burning peptide approaches for men and the best peptides for muscle growth provide targeted information for specific goals.
People on concurrent medications
If you take medications alongside semaglutide, discontinuation may affect their absorption, metabolism, or required dosing. Semaglutide delayed gastric emptying can affect the absorption of oral medications. When gastric emptying normalizes after discontinuation, medications that were being absorbed slowly may now be absorbed faster, potentially changing their effective concentration.
Discuss ALL your medications with your prescribing provider before stopping semaglutide. Blood pressure medications, thyroid medications, diabetes medications, and psychiatric medications are among those most likely to require adjustment. Understanding peptide safety profiles and drug interactions provides valuable context for these discussions, and knowledge about the legal landscape of peptide therapies helps ensure your overall treatment plan remains compliant with current regulations.
The bigger picture, obesity as a chronic disease
Semaglutide withdrawal symptoms highlight a fundamental truth that the medical community has increasingly embraced. Obesity is a chronic, relapsing condition driven by complex interactions between genetics, environment, hormones, and neural circuitry. It is not a character flaw. It is not a failure of willpower. It is a disease, and like other chronic diseases, it often requires ongoing management.
When someone with hypertension stops taking blood pressure medication, their blood pressure rises. We do not blame them for lacking discipline. We recognize that the medication was managing a biological condition, and removing it allows that condition to reassert itself. The same logic applies to obesity medications. Weight regain after stopping semaglutide is not failure. It is the natural trajectory of a chronic condition that was being medically managed.
This perspective shift matters because it changes how we approach the problem. Instead of asking "how can I permanently fix my weight with a temporary medication," we can ask "what is the right long-term management strategy for my specific situation." For some, that strategy includes lifelong medication. For others, it involves intermittent medication use. For others, it means medication as a bridge to establishing sustainable lifestyle changes that can independently maintain results.
There is no single right answer. The right answer depends on your individual biology, your goals, your resources, and your preferences. What matters is making informed decisions based on data rather than hope, and having realistic expectations about what different approaches can and cannot achieve.
SeekPeptides exists to help you navigate these decisions with evidence rather than guesswork. The combination of comprehensive educational resources, detailed protocol guides, precise dosing calculators, and community support from thousands of researchers who have faced similar decisions makes the journey less isolating and more informed. Whether you are stopping semaglutide, switching to an alternative, or considering restarting, having access to the latest research and evidence-based guidance transforms uncertainty into confidence.
Understanding the current regulatory landscape around peptide therapies and knowing what healthcare providers can and cannot prescribe empowers you to advocate for yourself in clinical settings. The real transformation stories from people who have navigated these transitions successfully remind us that challenging does not mean impossible.
Frequently asked questions
How long do semaglutide withdrawal symptoms last?
Most physical symptoms of semaglutide discontinuation resolve within five to eight weeks as the medication fully clears your system. The half-life of semaglutide is approximately 7 days, and it takes about 35 days, or five half-lives, for the drug to be essentially eliminated. However, the metabolic and hormonal effects of withdrawal, including increased appetite and hormonal rebound, can persist for months as your body readjusts. Weight regain patterns continue for 12 to 18 months in many individuals. The timeline varies significantly based on individual factors, duration of treatment, and lifestyle modifications implemented during the transition. The general timeline for peptide effects provides useful context for understanding how long biological transitions take.
Can you stop semaglutide cold turkey?
Yes, semaglutide can be stopped abruptly without medical danger. It does not cause physical dependence or classical withdrawal syndrome. However, research suggests that a gradual taper over approximately nine weeks produces better weight maintenance outcomes, with some studies showing stable weight for at least six months after a structured taper compared to the rapid regain typically seen with abrupt discontinuation. If you must stop suddenly due to supply issues or insurance changes, focusing aggressively on lifestyle modifications, particularly exercise and protein-rich nutrition, becomes especially important.
Will I gain all the weight back after stopping semaglutide?
Not necessarily, but significant regain is common. The STEP 1 extension trial showed that participants regained approximately two-thirds of their lost weight within one year of stopping. Real-world data presents a more variable picture, with about 17.7% regaining all weight or more, but more than half maintaining at least some weight loss. Your outcome depends heavily on exercise habits, dietary modifications, metabolic health, and whether you continue any form of pharmacological support. Proactive lifestyle changes during and after treatment are the strongest predictors of long-term success. Exploring the best approaches for sustained fat loss can help optimize your maintenance strategy.
Does semaglutide withdrawal cause depression?
The FDA has concluded that GLP-1 receptor agonists do not increase the risk of suicidality or serious mental health events. However, some individuals do report mood changes after stopping semaglutide, including mild depressive symptoms, irritability, and increased anxiety. These effects typically stem from multiple factors including the loss of GLP-1 receptor stimulation in mood-related brain regions, the psychological impact of seeing weight regain, and the return of food-related stress. Case reports suggest that mood symptoms associated with semaglutide use generally resolve after discontinuation. If you have a pre-existing history of depression or anxiety, discuss monitoring plans with your mental health provider before stopping.
How much exercise do I need after stopping semaglutide?
Research suggests approximately 250 or more minutes per week of moderate-intensity physical activity for weight maintenance after GLP-1 discontinuation. That is roughly 35 to 40 minutes daily or 50 minutes five days per week. This exceeds the standard health recommendation of 150 minutes per week. The ideal program combines resistance training two to three times weekly for muscle preservation and metabolic rate support, plus aerobic exercise and ideally some high-intensity interval training. Exercise also helps maintain higher natural GLP-1 production, partially offsetting the lost pharmaceutical stimulation. Using the peptide dosing and activity planning resources can help structure your approach.
Is semaglutide withdrawal worse than stopping tirzepatide?
Both medications show similar patterns of weight regain after discontinuation. A meta-analysis found that pooled weight regain was approximately 9.69 kg for combined semaglutide and tirzepatide users. The experience of withdrawal symptoms, including increased appetite, cravings, and metabolic changes, is comparable between the two medications. The semaglutide versus tirzepatide comparison provides detailed analysis of how these medications differ across multiple dimensions. In general, the discontinuation experience is driven more by the degree of weight loss achieved and individual metabolic factors than by which specific medication was used.
Can I take other peptides to help with semaglutide withdrawal?
Several peptide-based approaches may support the transition off semaglutide, though any new treatment should be discussed with your healthcare provider. Peptides that complement GLP-1 treatment may help address specific concerns like muscle preservation, gut health, or metabolic support. The bioactive precision peptides for weight management represent emerging approaches to maintaining results. Resources on available injectable peptides and the comparison between injectable and oral delivery methods help you understand the options. The longevity peptide category also offers compounds that support overall metabolic health during transitions.
When should I see a doctor about semaglutide withdrawal symptoms?
Contact your healthcare provider if you experience fasting blood glucose consistently above 200 mg/dL, rapid weight regain of more than 5 pounds in a single week, severe mood changes including persistent depression or anxiety lasting more than two weeks, gastrointestinal symptoms that do not resolve within three to four weeks, or worsening of any obesity-related health condition. For individuals with type 2 diabetes, the threshold for medical contact should be lower, and any blood sugar above 300 mg/dL or symptoms of diabetic ketoacidosis require immediate medical attention. General peptide safety and risk management guidance provides a framework for knowing when professional medical intervention is warranted.
External resources
For researchers serious about optimizing their weight management protocols, SeekPeptides offers the most comprehensive resource available. Members gain access to evidence-based guides covering every major GLP-1 medication, detailed dosing calculation protocols, reconstitution calculators, and a community of thousands who have navigated these exact transitions. Whether you need help planning your taper, choosing an alternative medication, or building a lifestyle strategy for long-term maintenance, SeekPeptides provides the tools, protocols, and support to make every decision count.
In case I do not see you, good afternoon, good evening, and good night.