Feb 12, 2026

Somewhere between the third contradictory Reddit thread and the fourth telehealth ad promising needle-free weight loss, you started wondering whether tirzepatide drops actually work. You are not alone. Thousands of people search for this exact thing every month, looking for an alternative to weekly injections that does not involve needles, alcohol swabs, or that anxious moment before the first subcutaneous stick. And the answer they find is complicated, because tirzepatide drops exist in a gray area between genuine pharmaceutical innovation and unproven compounded formulations that have never been tested in a single large-scale clinical trial.
That matters more than most sources will tell you.
The injectable versions of tirzepatide, sold as Mounjaro and Zepbound, went through years of rigorous clinical trials involving thousands of participants. Those trials produced some of the most impressive weight loss data in pharmaceutical history, with participants losing an average of 20.9% of their body weight at maximum doses and up to 26% when combined with lifestyle modifications. The sublingual drops being marketed online? Zero large-scale human trials. Zero FDA approval. Zero published bioavailability data specific to tirzepatide in drop form.
This guide covers everything researchers need to know about tirzepatide drops. How they work, what the dosing protocols look like, how they compare to injections on every measurable factor, what side effects to expect, how to store them properly, and most importantly, what the actual science says versus what the marketing claims. SeekPeptides provides evidence-based peptide education, and that means being honest about what we know and what we do not.
By the time you finish reading, you will understand exactly where tirzepatide drops stand in the weight management landscape, who might benefit from them, who should avoid them, and how to make an informed decision based on data rather than marketing.
What are tirzepatide drops and how do they work
Tirzepatide drops are compounded sublingual formulations designed to deliver tirzepatide, a dual GIP/GLP-1 receptor agonist, through the tissue under the tongue rather than through a subcutaneous injection. The concept is straightforward. Place the liquid under your tongue, hold it there for one to two minutes, and the medication absorbs through the dense network of blood vessels in the sublingual mucosa directly into the bloodstream.
That is the theory, at least.
The reality involves some significant biological challenges that anyone considering tirzepatide drops needs to understand. Tirzepatide is a peptide, a chain of 39 amino acids with a molecular weight of approximately 4,810 daltons. Peptides are notoriously difficult to deliver orally because digestive enzymes and stomach acid break them down before they can reach the bloodstream. The sublingual route attempts to bypass the digestive system entirely, and while the area under the tongue does have excellent blood supply, the absorption of large peptide molecules through this route remains unpredictable and inconsistent.
To understand why this matters, consider the comparison with oral semaglutide. Semaglutide is the only GLP-1 receptor agonist that has achieved FDA approval in an oral form, and even with a specialized absorption enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino] caprylate), its oral bioavailability sits at a dismal 0.4% to 1%. That means for every 100mg of semaglutide you swallow, less than 1mg actually reaches your bloodstream. The sublingual route may perform somewhat better than swallowing because it avoids stomach acid, but no published data confirms what percentage of tirzepatide actually makes it into circulation through sublingual delivery.
The dual receptor mechanism
What makes tirzepatide unique among weight loss peptides is its dual action on both the GIP (glucose-dependent insulinotropic polypeptide) receptor and the GLP-1 (glucagon-like peptide-1) receptor. Most other medications in this class, including semaglutide, only target GLP-1. The dual receptor activation produces several simultaneous effects that contribute to weight management.
GLP-1 receptor activation slows gastric emptying, meaning food stays in your stomach longer and you feel full faster. It also acts on the brain to reduce appetite signals and improve glucose-dependent insulin secretion. GIP receptor activation adds another layer entirely. It enhances insulin sensitivity, promotes fat metabolism, and appears to improve the body's ability to use stored energy rather than just reducing intake. The combination produces more substantial weight loss than GLP-1 agonism alone, which is why tirzepatide outperformed semaglutide in head-to-head comparisons.
Whether those same mechanisms function equally well when the peptide is absorbed sublingually rather than injected subcutaneously remains an open question. The mechanism of action does not change based on delivery route, but the amount of active peptide reaching the relevant receptors might. And that amount, the actual bioavailability, determines whether the drops produce the same results as injections or fall short.
How sublingual absorption works
The sublingual mucosa, the tissue under your tongue, is one of the most permeable areas in the oral cavity. It contains a rich capillary network that connects to the sublingual vein, which feeds into the external jugular vein and ultimately the systemic circulation. Medications absorbed here bypass first-pass metabolism in the liver, which is why sublingual delivery works well for small molecules like nitroglycerin.
For peptides, the picture gets more complicated. The sublingual epithelium is relatively thin compared to other oral tissues, which helps absorption. But peptide molecules are large. They do not cross biological membranes as easily as small molecules do. Some compounding pharmacies use absorption enhancers, permeation modifiers, or specialized carriers to improve sublingual peptide uptake, but the effectiveness of these approaches varies widely between formulations and between individual patients.
The practical result is inconsistency. Two people taking the same tirzepatide drop formulation from the same pharmacy might achieve very different blood levels of the medication. One might experience significant appetite suppression and weight loss. The other might notice almost nothing. This variability is fundamentally different from injectable tirzepatide, where subcutaneous delivery provides approximately 80% bioavailability with predictable, consistent absorption across patients.
Tirzepatide drops versus injections: the complete comparison
This is the question everyone asks first, and it deserves a thorough, honest answer. The comparison between tirzepatide drops and injections spans multiple dimensions, including efficacy, convenience, cost, safety, and regulatory status. On some of these factors, drops have a genuine advantage. On most, injections come out ahead. And on the most important factor, proven effectiveness, it is not even close.
Let us break it down.
Efficacy and clinical evidence
Injectable tirzepatide has some of the strongest clinical evidence of any weight management medication ever developed. The SURMOUNT trials enrolled thousands of participants and demonstrated weight loss results that fundamentally changed expectations in the field.
At the 2.5mg starting dose, participants lost an average of 5% to 7% of body weight. At 5mg, that increased to approximately 10% to 13%. The 10mg dose produced 15% to 18% losses. And at the maximum 15mg weekly dose, participants achieved an average of 20.9% body weight reduction, with some individuals losing more than 25% when combining medication with diet and exercise modifications.
For tirzepatide drops, the clinical evidence is essentially nonexistent. No randomized controlled trials. No published pharmacokinetic studies specific to sublingual tirzepatide. No head-to-head comparisons with injectable formulations. The only evidence comes from anecdotal reports, individual patient experiences shared by telehealth providers, and extrapolation from oral semaglutide data, which is a different molecule with different properties.
Some providers report that their patients see results with sublingual drops. Others report inconsistent or minimal effects. Without controlled studies, it is impossible to separate actual drug effects from placebo responses, lifestyle changes that coincided with starting the medication, or variations in formulation quality between compounding pharmacies.
Comparison table
Factor | Injectable tirzepatide | Tirzepatide drops |
|---|---|---|
FDA approval | Yes (Mounjaro, Zepbound) | No |
Clinical trials | SURMOUNT I-IV, thousands of participants | None published |
Average weight loss | 15-26% of body weight | Unknown/variable |
Bioavailability | Approximately 80% | Unknown, likely much lower |
Dosing frequency | Once weekly | Once daily |
Needle required | Yes | No |
Consistency | Highly consistent between doses | Variable absorption |
Insurance coverage | Possible with prior authorization | Generally not covered |
Typical monthly cost | $300-1,000+ (varies by source) | $150-500 (compounded) |
Regulatory oversight | FDA-regulated manufacturing | State-regulated compounding |
Convenience and lifestyle factors
This is where drops genuinely shine. For people with severe needle phobia, the sublingual route eliminates the biggest barrier to starting treatment. No syringes. No alcohol swabs. No injection site rotation. No subcutaneous technique to learn. You simply measure out your dose using the provided oral syringe, place it under your tongue, hold for one to two minutes, and go about your day.
The daily dosing schedule is a mixed bag, though. Injectable tirzepatide requires a single weekly injection. Drops require daily administration, typically first thing in the morning on an empty stomach, with restrictions on eating or drinking for 10 to 15 minutes afterward. Seven doses per week versus one dose per week means seven opportunities to forget, skip, or improperly administer the medication. For people who travel frequently, have irregular morning routines, or struggle with daily medication compliance, the weekly injection might actually be more convenient despite involving a needle.
Storage considerations also differ. Injectable tirzepatide pens from the manufacturer come ready to use and maintain potency for extended periods in the refrigerator. Compounded drop formulations may have shorter stability windows and more specific storage requirements depending on the pharmacy and formulation.
Dosing protocols for tirzepatide drops
Understanding tirzepatide dosing for sublingual drops requires recognizing that protocols vary significantly between providers and compounding pharmacies. Unlike FDA-approved injectable tirzepatide, which follows a standardized titration schedule established through clinical trials, drop dosing lacks a universally accepted protocol. That said, most providers follow a general framework that mirrors the injectable titration logic while accounting for the different delivery route.
Standard drop titration schedule
The most commonly reported titration for compounded tirzepatide drops follows this general pattern. The specifics may vary based on your provider and pharmacy.
Initiation phase (weeks 1 through 4): Most protocols start at 2.5mg daily, which is approximately 50 units when measured with the oral syringe provided by the pharmacy. This initial dose serves two purposes. It gives your body time to adjust to GLP-1 and GIP receptor activation, and it allows your provider to assess tolerance before increasing.
First escalation (weeks 5 through 8): If the starting dose is well tolerated, most providers increase to 5mg daily (approximately 100 units). This is often considered the first therapeutic dose, where most patients begin noticing meaningful appetite suppression and early weight changes.
Second escalation (weeks 9 through 12): Depending on response and tolerance, the dose may increase to 10mg daily (approximately 200 units). This represents a significant increase and is where GI side effects most commonly intensify if they are going to appear.
Maximum dose (if needed): Some protocols go up to 15mg daily (approximately 300 units). Not everyone needs this dose. Many patients achieve satisfactory results at 5mg or 10mg and remain there for the duration of treatment.
One critical difference between drops and injections that often gets overlooked: injectable tirzepatide maintains relatively stable blood levels throughout the week because it has a half-life of approximately five days. Daily sublingual dosing creates a different pharmacokinetic profile with daily peaks and troughs. Whether this matters clinically remains unknown because, again, no studies have characterized the pharmacokinetics of sublingual tirzepatide in humans.
How to administer tirzepatide drops correctly
Proper administration technique matters enormously with sublingual medications. Poor technique can drastically reduce absorption and effectiveness. Here is the step-by-step process most pharmacies recommend.
Step 1: Take the drops on an empty stomach, ideally first thing in the morning before eating or drinking anything.
Step 2: Draw the prescribed amount into the oral syringe provided by your pharmacy. Compounding pharmacies typically label their drops by concentration (mg/mL) and volume rather than total milligrams, so follow your specific prescription instructions carefully.
Step 3: Lift your tongue and dispense the liquid into the sublingual space, the pocket of tissue directly beneath the tongue.
Step 4: Hold the liquid under your tongue for at least one to two minutes without swallowing. Some providers recommend up to three minutes for maximum absorption. During this time, try not to talk, swallow, or move the liquid around your mouth.
Step 5: After the holding period, you can swallow any remaining liquid. However, the goal is for as much as possible to absorb through the sublingual tissue before swallowing.
Step 6: Do not eat or drink anything for at least 10 to 15 minutes after dosing. Some providers recommend 30 minutes for optimal absorption.
The difference between holding for 30 seconds versus two full minutes can meaningfully affect how much medication enters your bloodstream. Consistency in technique matters as much as consistency in timing. If you rush through the process some mornings and take your time on others, your effective dose will vary from day to day, contributing to the inconsistency that already challenges sublingual peptide delivery.
Dose measurement and concentration considerations
Compounding pharmacies formulate tirzepatide drops at various concentrations, and this is where confusion frequently arises. A pharmacy might dispense a solution at 10mg/mL concentration, meaning each milliliter contains 10mg of tirzepatide. If your prescribed dose is 5mg, you would draw 0.5mL. Another pharmacy might use a 5mg/mL concentration, requiring 1mL for the same dose.
Always verify three things with your pharmacy. The concentration of your specific formulation. The exact volume to draw for each dose. And how many doses the bottle contains. If you are using a tirzepatide dosage chart designed for injectable formulations, the numbers will not translate directly to drops because the delivery method, bioavailability, and dosing frequency are all different.
Understanding reconstitution and measurement basics helps avoid dosing errors regardless of which tirzepatide formulation you use. If your pharmacy provides tirzepatide that requires mixing or reconstitution before use, follow their specific instructions precisely. Using a peptide calculator can help verify measurements, though always defer to your prescriber and pharmacy for final dosing guidance.
Side effects of tirzepatide drops
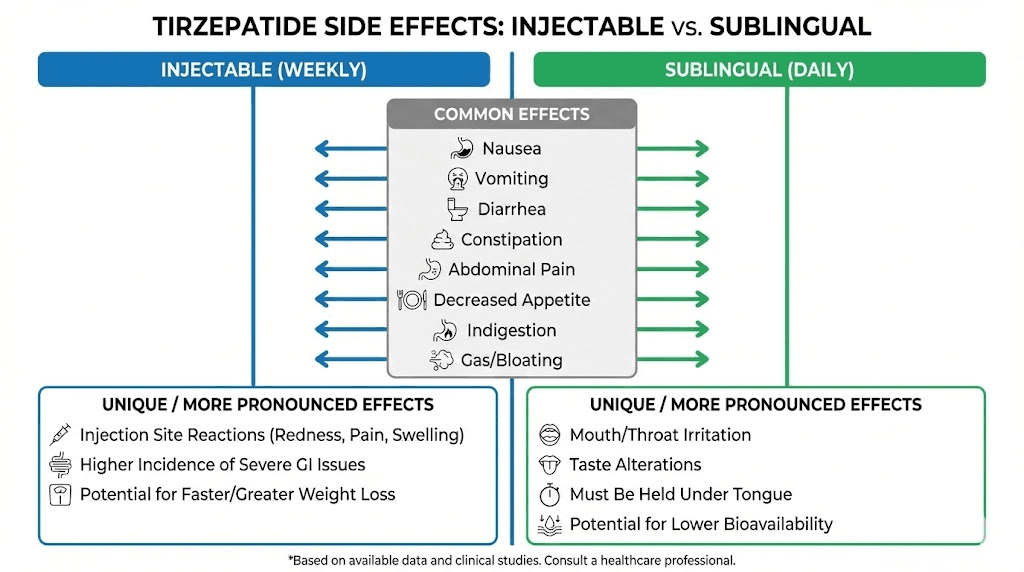
The side effect profile of tirzepatide drops shares significant overlap with injectable tirzepatide, but with some notable additions and potential differences in severity and timing. Understanding what to expect helps you distinguish between normal adjustment reactions and signals that require medical attention.
Gastrointestinal effects
The most common side effects with any form of tirzepatide involve the GI system. This makes sense given the mechanism of action. GLP-1 and GIP receptor activation directly affects gastric motility, appetite signaling, and digestive function. These effects are therapeutic features, not bugs, but they can be uncomfortable, especially during dose escalation.
Nausea affects 20% to 33% of injectable tirzepatide users in clinical trials. With drops, anecdotal reports suggest similar or potentially higher rates, possibly because daily dosing means daily exposure to the medication rather than a single weekly dose that gradually clears. The nausea is typically worst in the first one to two weeks at each new dose level and tends to diminish as the body adjusts.
Diarrhea occurs in 18% to 23% of injectable users. Drop formulations may produce slightly different GI patterns because of the daily dosing rhythm and potentially different absorption kinetics. Some patients report that the GI effects feel more constant with daily drops compared to the wave pattern common with weekly injections, where side effects peak in the first two to three days after injection and then fade.
Constipation, bloating, and abdominal discomfort round out the common GI complaints. Slower gastric emptying means food sits in your stomach and intestines longer, which can cause feelings of fullness, gas, and irregular bowel patterns. Maintaining adequate hydration and fiber intake helps manage these symptoms.
Reduced appetite is technically a therapeutic effect, not a side effect, but it can feel alarming if you suddenly have no interest in food whatsoever. This is more common at higher doses and typically moderates over time. If appetite suppression is so severe that you struggle to consume adequate nutrition, discuss dose adjustment with your provider.
Sublingual-specific effects
Drops can produce some side effects that injections do not, simply because of the delivery route.
Dry mouth is commonly reported with sublingual tirzepatide. The medication itself plus the instruction to avoid drinking for 10 to 15 minutes afterward can leave the mouth feeling parched. This is typically mild and resolves once you can drink normally.
Tongue or mouth irritation may occur from the formulation ingredients. Compounded sublingual solutions contain more than just tirzepatide. They include carriers, stabilizers, flavoring agents, and sometimes absorption enhancers. Any of these components could cause local irritation, especially with daily exposure to the same tissue.
Taste disturbance is another sublingual-specific complaint. Some patients describe a metallic or bitter taste that lingers after dosing. This varies by formulation and pharmacy.
Serious side effects to monitor
Whether delivered by injection or sublingually, tirzepatide carries the same serious risk warnings. These are rare but important to recognize.
Pancreatitis symptoms include severe, persistent abdominal pain that may radiate to the back, often accompanied by nausea and vomiting. Stop the medication and seek immediate medical attention if you experience this.
Gallbladder problems including gallstones and cholecystitis have been reported with GLP-1 receptor agonists. Symptoms include sudden, intense pain in the upper right abdomen, fever, and yellowing of the skin or eyes.
Kidney injury can occur, primarily through dehydration caused by persistent nausea, vomiting, or diarrhea. Maintaining hydration is essential, particularly during dose titration when GI side effects are most common.
Thyroid concerns represent a theoretical risk based on animal studies showing increased thyroid C-cell tumors with GLP-1 agonists. Tirzepatide carries a boxed warning about thyroid C-cell tumors, and it should not be used by anyone with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
One additional concern specific to compounded formulations is quality variability. FDA-approved injectable tirzepatide undergoes rigorous manufacturing quality controls. Compounded drops, while made by licensed pharmacies, do not undergo the same level of standardized testing. Potency, sterility, and stability may vary between batches, and in the worst cases, underdosing or overdosing could occur without the patient knowing. If you experience sudden changes in side effects or effectiveness after receiving a new batch from your pharmacy, this variability might be the reason.
The FDA and regulatory reality of tirzepatide drops
This section matters more than many people realize, and it is one that marketing materials for compounded drops tend to minimize or skip entirely. Understanding the regulatory landscape helps you assess the actual risk profile of what you are putting into your body.
What is FDA-approved
The FDA has approved tirzepatide in two branded injectable formulations. Mounjaro received approval for type 2 diabetes management, and Zepbound received approval for chronic weight management in adults with obesity or overweight with at least one weight-related comorbidity. Both are administered as once-weekly subcutaneous injections using pre-filled pens manufactured by Eli Lilly under strict FDA-regulated production standards.
That is it. No oral tablets. No sublingual drops. No nasal sprays. No patches. The only FDA-approved forms of tirzepatide require a needle.
How compounded drops enter the market
Compounded tirzepatide drops exist in a regulatory gray area. Under federal law, compounding pharmacies can prepare patient-specific medications when a commercially available form is medically unsuitable for a particular patient. This might apply when a patient has a documented allergy to an inactive ingredient in the commercial product, when a different concentration or route of administration is medically necessary, or historically when the commercial product was on the FDA drug shortage list.
The shortage exemption is particularly relevant here. Tirzepatide was on the FDA drug shortage list for an extended period due to overwhelming demand. During that time, compounding pharmacies could legally produce tirzepatide in various formulations, including sublingual drops. However, tirzepatide was removed from the FDA drug shortage list in October 2024, which significantly changed the legal landscape for compounding.
The current regulatory status of compounded tirzepatide is complex and evolving. Several lawsuits and FDA enforcement actions have created uncertainty about which compounded formulations remain legally available. The key point for consumers is that compounded tirzepatide drops are not the same as FDA-approved tirzepatide from a regulatory, quality assurance, or evidence standpoint.
Why this distinction matters for you
FDA approval means a medication has undergone extensive testing for safety, efficacy, and manufacturing quality. Every batch of Mounjaro or Zepbound contains exactly the amount of tirzepatide stated on the label, is free from contaminants, and maintains stability through the stated expiration date. These guarantees come from the multi-billion-dollar regulatory infrastructure that pharmaceutical manufacturers must maintain.
Compounded formulations undergo quality oversight from state boards of pharmacy rather than the FDA. Standards vary by state. Testing requirements vary by pharmacy. Some compounding pharmacies maintain excellent quality standards. Others have been cited for contamination, incorrect potency, or inadequate sterility procedures. The variability is the problem. You cannot easily verify which category your specific pharmacy falls into.
For people researching compounded tirzepatide options, understanding this regulatory distinction is not about fear-mongering. It is about informed decision-making. If you choose compounded drops, selecting a reputable 503B outsourcing facility (which undergoes more oversight than a standard 503A compounding pharmacy) and verifying that your pharmacy has current state licensure and a clean inspection record can mitigate some of the quality risks.
Who actually benefits from tirzepatide drops
Despite the limitations and unknowns, there are legitimate scenarios where tirzepatide drops might be a reasonable option. The key is matching the right person with the right formulation rather than treating drops as a universal alternative to injections.
People with severe needle phobia
Needle phobia is real. It is not a preference or a minor inconvenience. True trypanophobia can trigger vasovagal responses, panic attacks, and complete avoidance of medical care. For someone who simply cannot tolerate injections despite wanting treatment, sublingual drops may be the only viable route to access tirzepatide therapy.
If this describes you, the honest conversation with your provider should acknowledge that drops are less proven than injections, that results may be less predictable, and that you should monitor your response carefully. But accessing treatment in a less-proven form is arguably better than not accessing treatment at all, assuming the underlying medical indication is appropriate.
People who cannot use injectable formulations
Some individuals have medical reasons that make subcutaneous injections problematic. Certain connective tissue disorders, skin conditions at potential injection sites, or bleeding disorders might make injections inadvisable. In these cases, a sublingual formulation prescribed by a knowledgeable provider represents a legitimate medical adaptation.
People transitioning between formulations
Some providers use sublingual drops as a bridge. Perhaps a patient is switching between medications, titrating doses in smaller increments than the injectable pens allow, or supplementing injectable therapy with additional daily microdoses. These are nuanced clinical decisions that belong in the hands of experienced prescribers.
Who should avoid tirzepatide drops
If your primary motivation for choosing drops over injections is cost savings, convenience, or simply preferring not to inject, it is worth reconsidering. The cost savings may be offset by reduced effectiveness. The convenience of avoiding needles is balanced by the inconvenience of daily dosing with strict timing requirements. And if the drops do not produce adequate blood levels of tirzepatide, you may spend months and significant money achieving nothing.
People who should be especially cautious include those with a history of eating disorders (the variable appetite effects could be destabilizing), those taking medications with narrow therapeutic windows (unpredictable tirzepatide blood levels could affect interactions), and those with GI conditions that might be worsened by daily GLP-1 agonist exposure.
If you are currently not seeing results on tirzepatide injections, switching to drops is unlikely to improve things. The issue is almost certainly something other than the delivery route, and drops would provide less consistent dosing than injections, not more.
Storage and handling of tirzepatide drops
Proper storage is critical for maintaining the potency and safety of any peptide formulation, and compounded drops have their own specific requirements that differ from commercial injectable products.
Refrigeration requirements
Most compounded tirzepatide drops should be stored in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). This temperature range slows peptide degradation and helps maintain potency throughout the shelf life of the formulation. Your pharmacy should provide specific storage instructions with each dispensing, and those instructions take priority over general guidance.
Unlike FDA-approved injectable pens, which have been extensively stability-tested and come with precise expiration dates, compounded formulations may have shorter stability windows. Some pharmacies assign beyond-use dates of 30 to 90 days depending on the formulation. Check your specific product for this information and do not use it beyond the stated date.
Room temperature stability
For FDA-approved injectable tirzepatide, Eli Lilly states that pens can be stored at room temperature (below 86 degrees Fahrenheit or 30 degrees Celsius) for up to 21 days. After that, they should be discarded. The manufacturer also advises against returning the medication to refrigeration after room temperature storage.
Compounded drops may have different room temperature tolerances. Some formulations are more sensitive to temperature excursions than others depending on the carrier solution, preservatives used, and specific compounding methods. When in doubt, keep your drops refrigerated at all times except during the brief period needed for daily administration.
If you have questions about how long compounded tirzepatide can be out of the fridge, or whether tirzepatide needs to be refrigerated at all times, your dispensing pharmacy should be your primary resource. The answer depends entirely on their specific formulation.
Light protection and container requirements
Peptides are generally sensitive to light exposure, which can accelerate degradation. Keep your drops in the original container provided by the pharmacy, which is typically an amber or opaque bottle designed to block light. Do not transfer the solution to a different container unless specifically instructed to do so.
Store the bottle upright to minimize leakage and ensure accurate dosing. If the solution changes color, becomes cloudy, or develops particles, do not use it. These are signs of degradation or contamination. Understanding proper peptide storage principles helps protect your investment and safety regardless of the specific formulation.
Travel considerations
Traveling with compounded drops requires planning. You will need to maintain cold-chain storage, which means an insulated travel case with ice packs or a portable medication cooler. Unlike injectable tirzepatide pens that have a documented 21-day room temperature window, your compounded drops may not have the same tolerance.
If you travel frequently, discuss this with your provider. The once-weekly injectable format might actually be more practical for travel than daily drops that require strict storage and timing protocols.

Cost comparison and insurance considerations
Money matters in this discussion. Tirzepatide, regardless of form, is not cheap. But the cost equation differs significantly between FDA-approved injectables and compounded drops, and the cheapest option is not always the best value.
Injectable tirzepatide costs
Without insurance, brand-name Mounjaro or Zepbound can run $800 to $1,200+ per month depending on the pharmacy and any manufacturer discount programs. With insurance coverage and prior authorization, copays can drop to $25 to $150 per month, though coverage varies enormously by plan. Many commercial insurance plans now cover Zepbound for weight management, and Mounjaro for diabetes, though prior authorization requirements can be frustrating and time-consuming.
Compounded injectable tirzepatide, when legally available, typically costs $200 to $500 per month depending on the pharmacy and dose.
Tirzepatide drop costs
Compounded sublingual drops generally fall in the $150 to $500 per month range. This can appear cheaper than brand-name injectables, which drives much of the interest in drop formulations. But the cost calculation needs to account for effectiveness.
If injectable tirzepatide at $400 per month produces 20% body weight loss over a year, and drops at $200 per month produce 8% loss (hypothetically, since we do not have data), the injections delivered more than twice the result for twice the price. Cost per percentage point of weight loss actually favors the more expensive option. If drops produce no meaningful result, the $200 per month becomes pure waste.
This is not an argument against drops. It is an argument for honest cost-benefit analysis rather than making decisions based on sticker price alone.
Insurance realities
FDA-approved injectable tirzepatide has a path to insurance coverage. It is an uphill path involving prior authorizations, documented medical necessity, and sometimes appeals. But it exists. Compounded drops are almost universally out-of-pocket expenses. Insurance companies do not cover non-FDA-approved formulations, and HSA/FSA eligibility for compounded medications varies by plan administrator.
If you are exploring affordable tirzepatide options, consider all formulations and their relative effectiveness before defaulting to the lowest price point.
The science behind oral peptide delivery
To truly understand where tirzepatide drops stand, it helps to understand the broader scientific challenge of delivering peptides orally. This is not a new problem. Pharmaceutical researchers have been working on it for decades, and the progress has been slower than most people expect.
Why peptides are hard to take by mouth
Peptides are chains of amino acids, and the human digestive system is specifically designed to break amino acid chains into individual components for absorption. This is exactly what you want when you eat a steak. It is exactly what you do not want when you are trying to get an intact, functional peptide into your bloodstream.
Three main barriers stand between an orally administered peptide and therapeutic blood levels. Enzymatic degradation begins in the mouth with salivary enzymes and continues aggressively in the stomach with pepsin and hydrochloric acid. The small intestine adds pancreatic proteases and peptidases. By the time an unprotected peptide reaches the absorptive surface of the intestinal wall, most of it has been chopped into inactive fragments.
Mucosal permeability presents the second barrier. Even if a peptide survives enzymatic attack, it must cross the intestinal or sublingual epithelium to reach the bloodstream. Large molecules struggle to pass through tight junctions between epithelial cells, and transcellular transport (passing through the cells themselves) requires mechanisms that most therapeutic peptides lack.
First-pass metabolism hits oral medications after absorption. Blood from the GI tract passes through the liver before reaching systemic circulation, and liver enzymes can further degrade peptide molecules. The sublingual route avoids this barrier by feeding into veins that bypass the liver, which is one genuine advantage of drops over swallowed formulations.
The oral semaglutide precedent
The most relevant comparison for understanding tirzepatide drop potential is oral semaglutide (brand name Rybelsus). This is the only oral GLP-1 receptor agonist to achieve FDA approval, and its development story illustrates both the potential and the challenges of oral peptide delivery.
Oral semaglutide uses a proprietary absorption enhancer called SNAC that protects the peptide from stomach acid and promotes absorption through the gastric lining. Despite this sophisticated technology, oral semaglutide achieves a bioavailability of only 0.4% to 1%. To compensate, the oral dose (14mg daily for diabetes, with research into 25mg and 50mg for weight loss) is vastly higher than the injectable dose (1mg to 2.4mg weekly).
Even with these high doses, oral semaglutide drops produce more modest weight loss results than injectable semaglutide. The OASIS 1 trial showed that 50mg oral semaglutide achieved about 15% weight loss, approaching but not matching injectable results. The 14mg dose approved for diabetes produces significantly less weight loss than injectable formulations.
What does this mean for tirzepatide drops? It suggests that even with optimized delivery technology, oral peptide formulations struggle to match injectable efficacy. Compounded tirzepatide drops lack the sophisticated delivery technology that oral semaglutide uses, which raises legitimate questions about how much active peptide actually reaches the bloodstream.
Emerging delivery technologies
Researchers continue developing novel approaches to oral peptide delivery. Recent studies have explored using milk-derived small extracellular vesicles as delivery vehicles for both semaglutide and tirzepatide, with early results showing enhanced oral absorption. Other approaches include nanoparticle encapsulation, mucoadhesive formulations, and ionic liquid-based carriers.
These are exciting developments for the future of sublingual peptide delivery, but they remain firmly in the research phase. None are available in commercial or compounded formulations today. The tirzepatide drops currently on the market use comparatively simple formulation approaches that have not been validated for peptide bioavailability in published studies.
Making sense of tirzepatide drop claims and marketing
If you have spent any time researching tirzepatide drops online, you have encountered a wide spectrum of claims. Some are reasonable. Many are misleading. Knowing how to evaluate these claims protects you from making decisions based on hype rather than evidence.
Claims that are accurate
"Tirzepatide drops do not require needles." True. This is a straightforward factual statement about the delivery method.
"Sublingual delivery bypasses the digestive system." Partially true. It bypasses stomach acid and first-pass liver metabolism. It does not bypass all barriers to absorption.
"Tirzepatide acts on both GIP and GLP-1 receptors." True. The molecular mechanism of tirzepatide is the same regardless of delivery route. The question is whether enough active peptide reaches those receptors through sublingual delivery.
Claims that are misleading
"Same medication, no needles." While technically containing the same active ingredient, the bioavailability difference means you may not be getting the "same" effective dose. A 5mg sublingual dose does not produce the same blood levels as a 5mg injected dose.
"FDA-approved ingredient." The ingredient (tirzepatide) has FDA approval, but the sublingual formulation does not. This distinction matters enormously. Aspirin is an FDA-approved ingredient, but that does not make a homemade aspirin formulation FDA-approved.
"Clinically proven for weight loss." Injectable tirzepatide is clinically proven. Sublingual drops are not. Using injectable trial data to market sublingual formulations is a bait-and-switch that conflates two very different products.
"Results similar to injections." There is no published data supporting this claim. Individual testimonials do not constitute evidence, and selection bias (providers sharing success stories while omitting failures) makes anecdotal reports unreliable.
Red flags in provider marketing
Be cautious of any provider or pharmacy that refuses to acknowledge the limitations of sublingual tirzepatide. Legitimate prescribers will be upfront about the lack of clinical trial data, the potential for variable absorption, and the regulatory status of compounded formulations. If a provider makes it sound like drops are exactly equivalent to injections with zero downsides, that provider is prioritizing sales over informed consent.
Trustworthy providers discuss the evidence gap openly. They monitor your progress closely. They adjust your treatment plan based on actual results rather than assumed equivalence. And they present injections as the first-line recommendation while positioning drops as an alternative for specific circumstances.
Combining tirzepatide drops with lifestyle modifications
Regardless of which form of tirzepatide you use, the medication works best as part of a comprehensive approach that includes dietary modifications, physical activity, sleep optimization, and stress management. This is not filler advice. The SURMOUNT trials that produced those impressive weight loss numbers included structured lifestyle intervention alongside medication.
Dietary strategies while on tirzepatide drops
The appetite-suppressing effects of tirzepatide mean you will naturally eat less. The challenge becomes making sure the food you do eat provides adequate nutrition. When your intake drops from 2,500 calories to 1,200 calories, every bite matters more.
Prioritize protein intake. A minimum of 0.7 to 1.0 grams per pound of body weight helps preserve lean muscle mass during rapid weight loss. This is particularly important because GLP-1 agonists can cause loss of both fat and muscle, and inadequate protein accelerates muscle wasting. Quality tirzepatide diet plans emphasize protein-forward meals with each eating occasion.
Hydration becomes even more critical when taking tirzepatide in any form. GI side effects like nausea, vomiting, and diarrhea increase fluid losses. Slowed gastric emptying can reduce your natural thirst signals. Aim for at least 64 ounces of water daily, more if you experience GI symptoms. This also helps protect kidney function, which is important given the rare but real risk of kidney injury associated with GLP-1 agonists.
With drops specifically, remember the timing restrictions around dosing. Take your drops first thing in the morning before eating or drinking, then wait at least 10 to 15 minutes before consuming anything. Plan your morning routine around this window to ensure consistent dosing.
Exercise considerations
Resistance training is non-negotiable during any GLP-1 agonist therapy. The studies are clear. People who lose weight with tirzepatide without resistance training lose significantly more muscle mass than those who incorporate strength work. Two to three resistance training sessions per week using progressive overload principles helps maintain the muscle that keeps your metabolism functioning and your body composition improving rather than just shrinking.
Cardiovascular exercise supports heart health, mood, energy levels, and overall metabolic function. Start conservatively if you are reducing caloric intake significantly, as your energy availability will be lower than usual. Moderate-intensity activity like brisk walking, cycling, or swimming for 150 minutes per week aligns with general health guidelines and supports weight management without overtaxing a body under caloric restriction.
If you notice significant fatigue, lightheadedness, or exercise intolerance while on tirzepatide drops, discuss this with your provider. The variable bioavailability of drops means your effective dose could fluctuate, and intense exercise on days when drug levels happen to be higher could increase side effects like nausea or dizziness.
Sleep and stress management
Poor sleep and chronic stress both oppose weight loss through hormonal mechanisms. Cortisol elevation from stress promotes visceral fat storage. Sleep deprivation disrupts leptin and ghrelin, the primary hunger and satiety hormones, which can partially counteract the appetite-suppressing effects of tirzepatide.
Aim for seven to nine hours of quality sleep per night. If tirzepatide-related nausea is disrupting your sleep, taking your drops earlier in the morning and discussing anti-nausea strategies with your provider can help. Some patients find that adjusting the timing of their last meal relative to bedtime also improves sleep quality.

Monitoring your progress on tirzepatide drops
Because tirzepatide drops lack the clinical validation of injectables, monitoring your response becomes even more important. You cannot assume the drops are working just because you are taking them. Active tracking helps you and your provider determine whether sublingual delivery is producing adequate results or whether a switch to injectable formulation would serve you better.
What to track
Body weight: Weigh yourself at the same time daily (morning, after using the bathroom, before eating). Daily weights fluctuate, so look at weekly averages rather than day-to-day numbers. Most people on effective tirzepatide therapy lose 1 to 2 pounds per week after the initial water-weight phase.
Appetite and hunger levels: Rate your hunger on a 1 to 10 scale at consistent times each day. Effective tirzepatide therapy typically reduces baseline hunger by 3 to 5 points on this scale. If you notice no appetite change after four or more weeks at a given dose, the medication may not be reaching therapeutic levels in your system.
Measurements: Waist circumference, hip circumference, and other relevant measurements provide data independent of scale weight. Sometimes body composition is changing even when the scale is not moving.
Side effects: Track the type, severity, and duration of any side effects. This helps your provider optimize dosing and identify patterns. With daily drops, side effects may follow different patterns than with weekly injections.
Food intake: Even rough tracking of daily caloric intake helps contextualize weight loss results. If you are losing weight primarily because you are eating 800 calories a day due to severe nausea, that is different from losing weight at 1,500 calories with comfortable appetite management.
Timeline expectations
With injectable tirzepatide, most patients notice appetite changes within the first one to two weeks, early weight loss within two to four weeks, and significant results by the three to six month mark. How fast tirzepatide works depends on dose, individual metabolism, and lifestyle factors.
With drops, the timeline may be different because of variable absorption. Some patients report noticing effects within the first week. Others take several weeks to detect any change. If you have been on drops for eight or more weeks at a therapeutic dose (5mg or higher daily) and notice no appetite changes, minimal weight loss, and no GI side effects, there is a reasonable chance that sublingual absorption is inadequate for you specifically.
This is not a personal failure. It is a pharmacokinetic reality. Sublingual absorption varies between individuals based on factors including oral tissue thickness, blood flow, saliva production, and administration technique. If drops are not working, injectable tirzepatide with its 80% bioavailability and proven track record remains the gold standard option.
When to consider switching to injections
Have an honest conversation with your provider about switching from drops to injections if you experience any of the following after an adequate trial period.
No appetite suppression after six to eight weeks at therapeutic doses. No meaningful weight loss (less than 3% body weight) after three months. Persistent GI side effects without corresponding weight loss benefits. Highly variable day-to-day effects suggesting inconsistent absorption. Frustration with daily dosing requirements and timing restrictions.
Switching is not admitting defeat. It is optimizing your treatment based on your individual response, which is exactly what good medical care looks like.
Tirzepatide drops and other medications
Drug interactions with tirzepatide are important to understand regardless of the delivery route, but sublingual administration introduces some additional considerations.
Known tirzepatide interactions
Tirzepatide slows gastric emptying, which can affect the absorption of other oral medications. Drugs that require rapid or consistent absorption may have altered effectiveness. This includes oral contraceptives, antibiotics, and some cardiovascular medications. Your prescriber should review your full medication list before starting tirzepatide in any form.
Insulin and sulfonylureas carry an increased risk of hypoglycemia when combined with tirzepatide. Dose adjustments of these medications are typically necessary. If you are managing diabetes alongside weight loss goals, close glucose monitoring is essential, especially during dose escalation periods.
Sublingual-specific interaction considerations
If you take other sublingual medications, discuss timing with your provider. Taking multiple sublingual medications within the same window could affect absorption of one or both. Generally, space sublingual medications at least 30 minutes apart.
Mouthwashes, particularly alcohol-based ones, can alter the sublingual mucosa and potentially affect drug absorption. If you use mouthwash regularly, time it well away from your tirzepatide drop dosing.
Smoking and nicotine use can reduce sublingual blood flow and potentially impair absorption. If you smoke, this is another factor that might make injectable delivery more reliable than sublingual.
Certain supplements, particularly those containing B12, niacinamide, or glycine, are sometimes combined with tirzepatide in compounded formulations or taken alongside it. Discuss any supplements with your prescriber to ensure there are no interaction concerns.
The future of oral tirzepatide
While current compounded drops represent an imperfect solution, genuine pharmaceutical development of oral tirzepatide formulations is ongoing. Understanding where the science is heading provides context for current options.
Eli Lilly oral tirzepatide development
Eli Lilly, the manufacturer of Mounjaro and Zepbound, has acknowledged interest in developing oral formulations of tirzepatide. However, this is a fundamentally different undertaking than compounding pharmacies simply putting tirzepatide in a sublingual solution. Pharmaceutical-grade oral formulation development involves years of research into absorption enhancers, protective coatings, delivery vehicles, and extensive pharmacokinetic and clinical testing.
Any FDA-approved oral tirzepatide product is likely years away from market availability. If and when it arrives, it will have clinical trial data supporting its efficacy, a defined bioavailability profile, standardized dosing, and the quality assurances that come with FDA-regulated manufacturing. It will also likely cost significantly more than current compounded formulations.
Broader oral peptide research
The pharmaceutical industry is investing heavily in oral peptide delivery platforms. The success of oral semaglutide proved the concept is viable, and multiple companies are developing improved absorption technologies. Some approaches showing promise include enteric-coated nanoparticles that protect peptides through the stomach and release them in the intestine, intestinal patches that adhere to the mucosal wall and create a microenvironment for enhanced absorption, and novel absorption enhancers that are more effective than SNAC while maintaining safety.
For now, these remain research-stage technologies. The practical options for tirzepatide today are FDA-approved injectable formulations and compounded alternatives, including sublingual drops, that carry the limitations discussed throughout this guide.
Frequently asked questions
Do tirzepatide drops work as well as injections?
No published clinical data directly compares sublingual tirzepatide drops to injections. Injectable tirzepatide has approximately 80% bioavailability and extensive clinical trial support showing 15 to 26% body weight loss. Sublingual drops have unknown bioavailability and variable absorption that likely results in lower and less consistent blood levels of the medication. Individual results with drops range from significant weight loss to no detectable effect.
Are tirzepatide drops FDA-approved?
No. Only injectable tirzepatide (Mounjaro for diabetes, Zepbound for weight management) has FDA approval. Sublingual drops are compounded formulations prepared by compounding pharmacies. They have not undergone the clinical trials required for FDA approval and are not evaluated by the FDA for safety or efficacy. Learn more about oral tirzepatide options in our dedicated guide.
How long does it take for tirzepatide drops to start working?
Individual responses vary significantly. Some patients report appetite changes within the first week, while others notice nothing for several weeks. Most providers recommend evaluating response after at least four to eight weeks at a therapeutic dose. If you experience no appetite suppression, no GI effects, and no weight changes after this period, sublingual absorption may be inadequate. Compare this to injectable tirzepatide timelines where most patients notice effects within the first two weeks.
Can I switch from tirzepatide injections to drops?
Technically yes, with provider guidance, but this switch means moving from a proven delivery method to an unproven one. Most providers recommend the opposite direction, switching from drops to injections if drops are not producing adequate results. If your reason for switching is needle phobia that developed after starting treatment, discuss desensitization strategies with your provider before abandoning the more effective formulation.
How should I store tirzepatide drops?
Refrigerate between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). Follow your specific pharmacy instructions regarding room temperature stability, as this varies by formulation. Keep the bottle in its original container to protect from light, and do not use drops that have changed color, become cloudy, or developed particles. See our complete peptide storage guide for broader storage principles.
What is the difference between tirzepatide drops and oral tirzepatide tablets?
Both are non-FDA-approved compounded formulations, but they use different delivery approaches. Drops are liquid formulations administered sublingually (under the tongue) for direct absorption through the oral mucosa. Tablets, often called rapid-dissolve or sublingual tablets, dissolve under the tongue or on the tongue surface. The absorption mechanism is similar, but the formulation, carrier ingredients, and potentially the absorption characteristics differ. Neither has published clinical trial data supporting efficacy.
Can I take tirzepatide drops with other medications?
Tirzepatide can interact with certain medications regardless of delivery route. The most important interactions involve insulin, sulfonylureas, and medications that require consistent absorption timing. If you take other sublingual medications, space them at least 30 minutes apart from your tirzepatide drops. Always provide your complete medication list to your prescriber before starting any form of tirzepatide.
Why are tirzepatide drops daily while injections are weekly?
Injectable tirzepatide achieves high bioavailability (approximately 80%) and has a half-life of about five days, meaning a single weekly dose maintains therapeutic blood levels throughout the week. Sublingual drops have lower and more variable absorption, so daily dosing attempts to maintain more consistent drug levels. The different pharmacokinetic profile of sublingual delivery necessitates more frequent administration, though the optimal dosing frequency for sublingual tirzepatide has not been established through clinical research.
External resources
For researchers serious about making informed decisions about peptide protocols, SeekPeptides provides evidence-based guidance, comprehensive protocol databases, and a community of experienced researchers who can help you navigate the difference between marketing claims and actual science. Whether you are evaluating tirzepatide drops, optimizing injectable dosing, or exploring entirely different peptide approaches, SeekPeptides members access the detailed, research-backed information needed to make confident decisions.
In case I do not see you, good afternoon, good evening, and good night. May your peptide choices stay informed, your absorption stay consistent, and your results stay measurable.