Feb 20, 2026

Before you add another pill to your tirzepatide protocol, stop. This is not a decision to rush. Combining phentermine with tirzepatide involves two powerful medications that affect the cardiovascular system, the nervous system, and metabolic pathways in ways that overlap, amplify, and sometimes conflict. The combination is not FDA-approved. No clinical trial has ever tested it. And yet thousands of people are doing it anyway, some under medical supervision and others entirely on their own.
That gap between clinical evidence and real-world practice is exactly why this guide exists.
If you are considering adding phentermine to your tirzepatide regimen, or if you are already taking both and wondering whether that was a good idea, you need specific answers. Not vague reassurances. Not scare tactics. Actual pharmacological data on what happens when these two mechanisms collide inside the same body. You need to understand the cardiovascular implications, the overlapping side effect profiles, and the realistic scenarios where this combination could make sense versus the scenarios where it could cause real harm.
This guide covers every angle. The pharmacology of both drugs. The overlapping side effects and cardiovascular concerns. The monitoring your provider should be doing if you are on both. When combination therapy might be justified, and when it absolutely should not happen. Whether you are working with a weight loss clinic prescribing compounded medications or managing your own weight loss research, understanding the interaction between phentermine and tirzepatide is not optional. It is essential.
The short answer
Yes, it is technically possible to take phentermine and tirzepatide at the same time. Some physicians prescribe them concurrently, particularly for patients who have plateaued on one medication alone. But "possible" and "recommended" are two very different things.
The FDA has not approved this combination. The phentermine label specifically warns against using it with other weight loss medications. No randomized controlled trial has ever evaluated phentermine plus tirzepatide for safety or efficacy. Every single prescription written for this combination is off-label.
That does not mean it never happens.
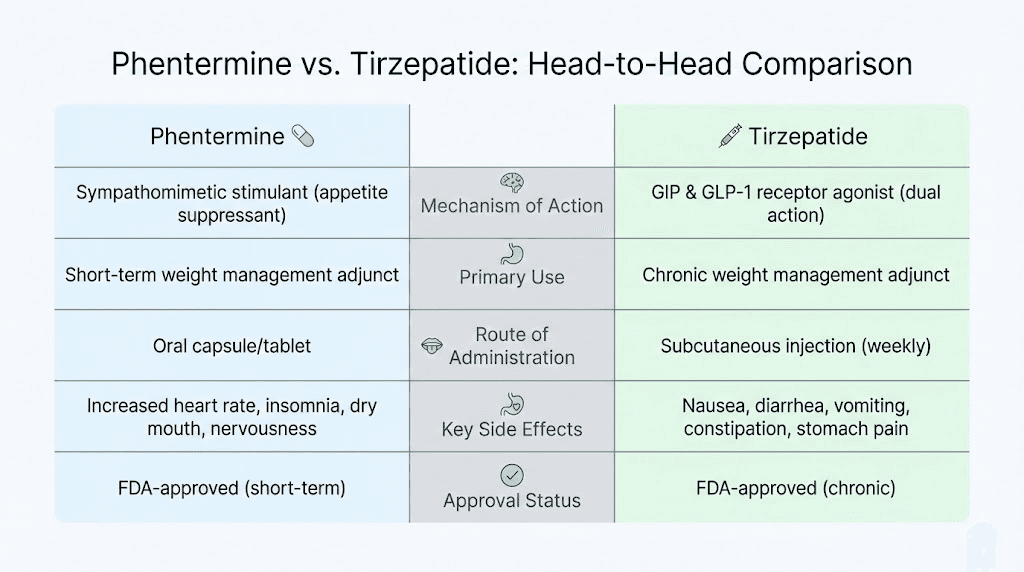
In clinical weight management practices across the country, some providers add phentermine to GLP-1 therapy when patients hit a weight loss plateau. The logic is straightforward: the two drugs work through completely different pathways. Phentermine targets norepinephrine in the brain to suppress hunger signals directly. Tirzepatide works through dual GIP and GLP-1 receptor activation to slow gastric emptying, enhance insulin response, and reduce appetite through gut-brain signaling. Different mechanisms. Different receptors. In theory, complementary effects that could produce additive weight loss.
But theory and practice do not always align. The same qualities that make these drugs effective individually, appetite suppression, metabolic shifts, cardiovascular stimulation, can compound in unpredictable ways when combined. The modest heart rate increase from tirzepatide (averaging 2-4 beats per minute in clinical trials) stacks on top of the sympathomimetic stimulation from phentermine. Gastrointestinal side effects that are manageable alone can become intolerable together. And the lack of clinical trial data means nobody truly knows the combined risk profile.
The bottom line: work with a qualified healthcare provider. Do not combine these medications without medical supervision. And do not assume that more medication automatically equals more weight loss. For many people, optimizing tirzepatide dosing alone produces results that make adding phentermine completely unnecessary.
How phentermine works in the body
Understanding why this combination carries risk requires knowing what each drug does on its own. Phentermine has been prescribed for weight loss since 1959. That makes it one of the oldest FDA-approved anti-obesity medications still in active use, and its mechanism has been thoroughly studied over more than six decades of clinical practice.
It is a sympathomimetic amine. That means it mimics the effects of the sympathetic nervous system, the same system responsible for the fight-or-flight response. Specifically, phentermine triggers the release of norepinephrine from presynaptic vesicles in the lateral hypothalamus, the brain region that regulates hunger and satiety signals.
The result is direct: reduced appetite.
When norepinephrine floods the hypothalamic synaptic cleft, it stimulates beta-2 adrenergic receptors. Research indicates that phentermine also inhibits neuropeptide Y, one of the primary signaling molecules that tells the brain to seek food. This creates a two-pronged suppression effect. The hunger signal gets blocked. The sensation of fullness gets amplified. And the person eating simply does not feel as driven to consume calories as they would without the medication.
Phentermine also has weaker effects on dopamine transporters, which may contribute to its mild stimulant properties and the increased alertness some users report. Some evidence suggests it increases resting energy expenditure, though appetite suppression remains the dominant mechanism behind weight loss results. If you have compared phentermine to newer GLP-1 medications, you already know the newer drugs work through entirely different systems.
Here is where the safety picture gets complicated.
Because phentermine activates the sympathetic nervous system, it raises heart rate and blood pressure. These are not side effects in the traditional sense. They are direct, predictable consequences of the drug mechanism. Every patient taking phentermine experiences some degree of cardiovascular stimulation. For most healthy individuals without underlying heart conditions, this is manageable and clinically insignificant. For people with uncontrolled hypertension, arrhythmias, or structural heart disease, it is potentially dangerous.
The FDA approves phentermine only for short-term use, typically 12 weeks or less. Standard dosing ranges from 15 mg to 37.5 mg taken orally once daily, usually in the morning to avoid the insomnia that comes with late-day sympathetic stimulation. The drug is classified as a Schedule IV controlled substance due to its stimulant properties and potential for psychological dependence over time.
Common side effects include dry mouth, insomnia, restlessness, elevated heart rate, irritability, and constipation. These effects connect directly to the norepinephrine mechanism and tend to be dose-dependent. Serious but rare complications include primary pulmonary hypertension and valvular heart disease, though those associations come primarily from older studies involving fenfluramine combinations rather than phentermine alone.
However, more recent research provides a somewhat reassuring picture for short-to-medium term use. A large electronic health record cohort study found that patients who used phentermine beyond three months did not show increased cardiovascular adverse events, including no elevated risk of heart attack, stroke, or death over three years of follow-up. This data suggests that for otherwise healthy patients, phentermine duration beyond the FDA label may be clinically reasonable under proper supervision.
For many patients exploring weight loss medication combinations, phentermine serves as a first-line option because of its low cost and decades of clinical history. But its limitations are significant. Tolerance develops. Effects diminish over weeks and months. And the cardiovascular profile creates legitimate concerns when you start layering additional medications on top of it.
How tirzepatide works in the body
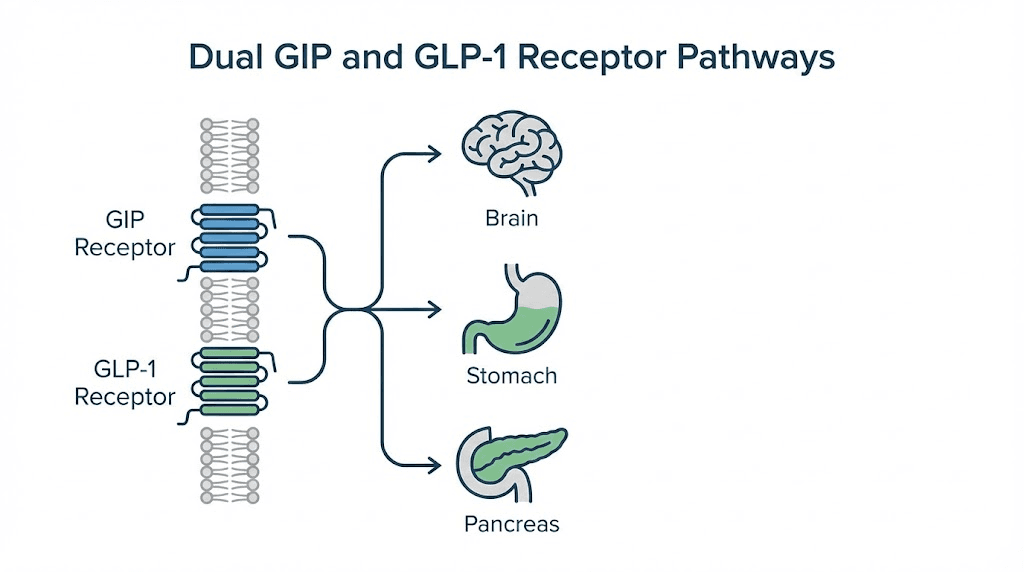
Tirzepatide operates through a fundamentally different system than phentermine. Where phentermine is essentially a central nervous system stimulant that suppresses hunger from the top down, tirzepatide works through the incretin system, a hormonal pathway that connects the gut, the pancreas, and the brain in a coordinated metabolic response to food.
The drug is a dual agonist. It activates two distinct receptors simultaneously: the GIP receptor (glucose-dependent insulinotropic polypeptide) and the GLP-1 receptor (glucagon-like peptide-1). This dual mechanism is what separates tirzepatide from older GLP-1 medications like semaglutide, which only activate the GLP-1 receptor. The addition of GIP receptor activation appears to amplify the metabolic and weight loss effects beyond what GLP-1 alone can achieve.
Here is what happens when tirzepatide enters the body.
First, it enhances glucose-dependent insulin secretion. When blood sugar rises after a meal, tirzepatide amplifies the natural insulin response from pancreatic beta cells. This is not a blanket insulin increase. It only occurs when glucose levels are elevated, which reduces the risk of hypoglycemia compared to older diabetes medications. Research comparing tirzepatide to semaglutide found that tirzepatide improved insulin sensitivity and insulin secretory responses to a greater extent, resulting in lower post-meal insulin and glucagon concentrations.
Second, it suppresses glucagon. GLP-1 receptor activation inhibits glucagon secretion during normal and high blood sugar states, though critically not during hypoglycemia. This glucagon suppression helps prevent excessive glucose production by the liver, contributing to both blood sugar control and metabolic efficiency.
Third, and most relevant to weight loss, tirzepatide dramatically slows gastric emptying. Food stays in the stomach longer. The signals that tell the brain "you are full" persist for extended periods. This is why many users report being unable to finish meals, losing interest in snacking, and experiencing profound changes in their relationship with food. Understanding how quickly tirzepatide suppresses appetite is critical context for anyone considering adding phentermine on top of an already powerful appetite suppression mechanism.
Fourth, it acts on the brain directly. Both GIP and GLP-1 receptors exist in brain regions controlling appetite and reward, which is why tirzepatide reduces food cravings and not just physical hunger. The satiety signaling goes beyond simply feeling full. It changes how the brain processes the desire for food at a neurological level.
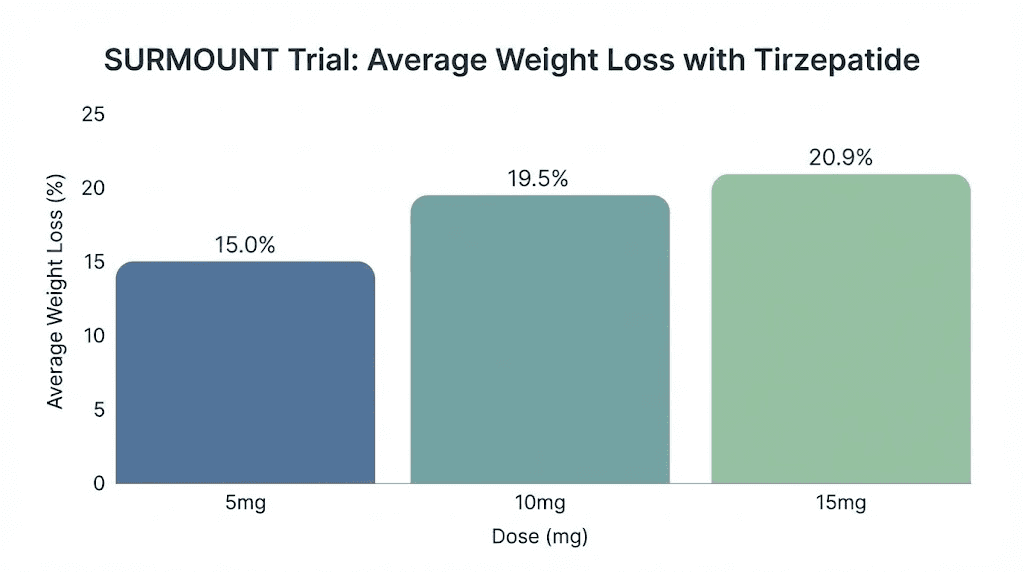
The clinical results reflect the power of this dual mechanism. In the SURMOUNT-1 trial, participants taking tirzepatide at the 15 mg dose lost an average of 20.9% of their body weight over 72 weeks. At the 10 mg dose, the average was 19.5%. Even the lowest dose of 5 mg produced 15% weight loss. For comparison, the placebo group lost 3.1%. The percentage of participants achieving at least 20% weight loss at the highest dose reached 56.1%, and 38.7% achieved 25% or greater weight loss. These are numbers that approach what bariatric surgery delivers, without any surgical intervention.
Tirzepatide is approved for long-term use, which stands in direct contrast to the short-term approval for phentermine. The standard tirzepatide dosing protocol starts at 2.5 mg weekly for the first four weeks, then increases in 2.5 mg increments every four weeks based on tolerability and response. The maximum approved dose is 15 mg per week. This gradual titration is specifically designed to minimize gastrointestinal side effects, which are the most common reason for discontinuation.
Common side effects of tirzepatide include nausea (affecting 24-29% of participants in trials), diarrhea (18-24%), vomiting (8-10%), and constipation (16-17%). There is also a modest increase in heart rate, averaging 2-4 beats per minute. Most gastrointestinal side effects are transient, peaking during dose escalation and improving as the body adapts. Proper dietary management and meal planning can significantly reduce GI discomfort during titration.
The weight loss efficacy of tirzepatide alone raises an important question that anyone considering adding phentermine should seriously contemplate: with 15-21% average weight loss as monotherapy, do you actually need a second medication?
Why some doctors combine phentermine and tirzepatide
Despite the lack of formal clinical trials, some weight management physicians do prescribe phentermine alongside tirzepatide. Understanding their reasoning, even if you ultimately decide against the combination, helps you have a more informed conversation with your own provider.
The primary rationale is complementary mechanisms. Phentermine and tirzepatide suppress appetite through completely different biological systems. Phentermine works fast, within hours, through direct norepinephrine stimulation in the brain. Tirzepatide works gradually, over days to weeks, through incretin-mediated gut-brain signaling and delayed gastric emptying. One is a central nervous system stimulant. The other is a hormonal modulator. In theory, targeting both pathways simultaneously could produce greater appetite suppression than either drug alone.
The second rationale is plateau management. Weight loss plateaus are common with every anti-obesity medication, and tirzepatide is no exception. If you have been on tirzepatide for months and your weight loss has stalled, some providers consider adding phentermine as a short-term boost rather than immediately increasing the tirzepatide dose. The thinking: if the incretin pathway has reached maximum effect, stimulating a completely different pathway might restart progress without the gastrointestinal burden of higher tirzepatide doses.
A third scenario involves transition patients. Some people start their weight loss journey on phentermine because it is inexpensive and readily available. When they later begin tirzepatide, there may be a period of overlap while tirzepatide titrates up to therapeutic levels. Tirzepatide takes weeks to reach full effect, especially since the starting dose of 2.5 mg is sub-therapeutic for weight loss. Rather than leaving the patient without appetite suppression during this ramp-up period, some providers maintain phentermine temporarily and then discontinue it once tirzepatide reaches effective doses.
From a clinical practice standpoint, some weight management specialists also point to the different timing profiles. Phentermine provides immediate, same-day appetite suppression but wears off by evening. Tirzepatide provides sustained, around-the-clock metabolic effects but takes weeks to reach full potency. The combination theoretically fills each medication gap.
But there is a critical counterargument that deserves equal weight in this discussion.
A randomized, placebo-controlled pilot study examined adding phentermine to liraglutide (a GLP-1 agonist similar in class to tirzepatide) in adults with obesity who had already been on liraglutide for one year and achieved approximately 12% weight loss. The result: adding phentermine did not provide meaningful additional weight loss compared to placebo. This suggests that once GLP-1 receptor agonism is maximized, adding a norepinephrine-based appetite suppressant may not offer the additional benefit that the complementary mechanism theory predicts.
Now, tirzepatide is not liraglutide. The dual GIP/GLP-1 mechanism is different and more potent. But this study introduces genuine doubt about whether the theoretical synergy between phentermine and incretin-based drugs translates into real-world clinical benefit. The combination might simply be adding cardiovascular risk without proportional weight loss improvement.
This is why the decision to combine these medications requires careful, individualized clinical judgment rather than a blanket recommendation in either direction. If your provider suggests this combination, ask them specifically what evidence they are relying on, what monitoring plan they have in place, and what their criteria are for discontinuing if the combination does not produce measurable additional benefit within a defined timeframe.
Safety concerns and drug interactions
The safety profile of combining phentermine and tirzepatide deserves a thorough, unflinching examination. These are not supplements or vitamins. Both are pharmacologically active compounds with documented effects on the cardiovascular system, the gastrointestinal tract, and metabolic regulation. When combined, these effects can amplify in ways that create genuine clinical risk.
Cardiovascular effects
This is the most significant safety concern with the combination. Phentermine, by design, stimulates the sympathetic nervous system. This raises heart rate, increases blood pressure, and puts additional demand on the heart. Tirzepatide independently causes a modest heart rate increase, averaging 2-4 beats per minute in the SURMOUNT clinical trials. Individually, neither increase is typically dangerous for healthy individuals. Together, the cumulative effect on heart rate and blood pressure becomes clinically significant, particularly for patients with pre-existing cardiovascular disease, uncontrolled hypertension, or arrhythmias.
The concern is not theoretical. Sustained elevation in heart rate, even by 5-10 beats per minute, increases long-term cardiovascular risk. When you combine the sympathomimetic stimulation of phentermine with the incretin-mediated heart rate increase from tirzepatide, monitoring becomes absolutely essential. Any patient on this combination should be checking blood pressure and resting heart rate at minimum weekly, with clear thresholds established for when to reduce or discontinue one or both medications.
Gastrointestinal side effects
Both phentermine and tirzepatide affect the gastrointestinal system, though through different mechanisms. Tirzepatide slows gastric emptying and causes nausea, diarrhea, vomiting, and constipation in a significant percentage of users. These effects are most pronounced during dose escalation and typically improve with time. Phentermine commonly causes constipation and dry mouth through its sympathomimetic effects.
When combined, gastrointestinal symptoms can intensify. The constipation from tirzepatide compounds with the constipation from phentermine. Nausea may worsen. And the combination can lead to dehydration and electrolyte imbalances if vomiting or diarrhea becomes persistent, which creates secondary risks for kidney function and overall metabolic stability. Understanding what to eat while on tirzepatide becomes even more critical when phentermine further reduces food intake and fluid consumption.
Blood sugar fluctuations
This interaction is particularly important for anyone using tirzepatide for type 2 diabetes management in addition to weight loss. Phentermine-induced weight loss can alter insulin sensitivity and change the dosage requirements for tirzepatide and other diabetes medications. As you lose weight on the combination, blood sugar may drop more than expected, creating a risk of hypoglycemia that requires more frequent monitoring and potential dose adjustments.
The drug interaction databases classify this as a moderate interaction, noting that "as you lose weight during treatment with phentermine, your dosage requirement of tirzepatide or any other diabetic medications may change." This is not a minor technicality. It requires proactive blood sugar monitoring and communication with your prescriber about dose adjustments as weight changes.
Medication absorption issues
Tirzepatide delays gastric emptying. That delay affects how other oral medications are absorbed, including phentermine. Slower stomach emptying can change the rate and extent of phentermine absorption, potentially altering its peak blood levels and duration of action. In some cases, this might make phentermine less effective. In others, it could lead to delayed but higher peak concentrations. The clinical significance of this interaction has not been formally studied for phentermine specifically, but it is a known consideration with all oral medications taken alongside GLP-1 receptor agonists.
If you are taking other medications beyond phentermine and tirzepatide, this absorption delay matters even more. Birth control pills, blood pressure medications, thyroid medications, and other drugs that depend on consistent absorption timing may all be affected by the delayed gastric emptying that tirzepatide produces. A thorough medication review with your pharmacist is essential before starting this combination.
Side effects when combining phentermine and tirzepatide
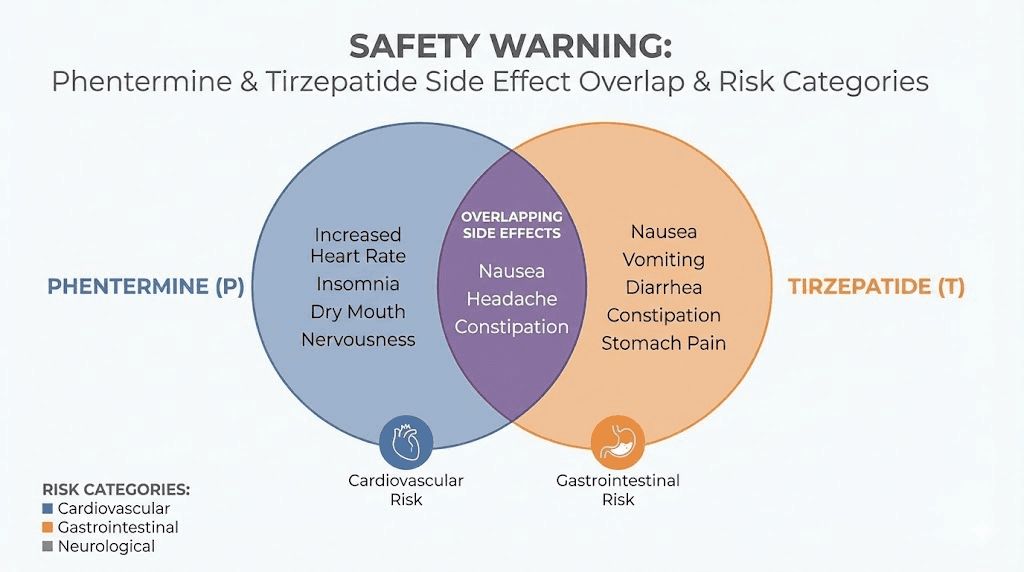
To make an informed decision, you need to understand exactly which side effects overlap, which amplify each other, and which are unique to each medication. Here is a comprehensive breakdown.
Side effect | Phentermine alone | Tirzepatide alone | Combined risk |
|---|---|---|---|
Elevated heart rate | Common | Modest (2-4 bpm) | Amplified |
Elevated blood pressure | Common | Rare | Increased |
Nausea | Occasional | Common (24-29%) | Amplified |
Constipation | Common | Common (16-17%) | Significantly amplified |
Diarrhea | Rare | Common (18-24%) | Tirzepatide-driven |
Insomnia | Common | Rare | Phentermine-driven |
Anxiety/restlessness | Common | Rare | Phentermine-driven |
Headache | Common | Common | Amplified |
Dry mouth | Very common | Occasional | Amplified |
Fatigue | Rare | Common | Variable |
Vomiting | Rare | Common (8-10%) | Tirzepatide-driven |
Dizziness | Common | Occasional | Amplified |
The most concerning overlaps are cardiovascular. Elevated heart rate from both medications stacking together is the primary risk clinicians worry about. But the gastrointestinal overlap deserves attention too. If tirzepatide is already causing significant nausea or headaches, adding phentermine can push side effects past the threshold of tolerability and lead to discontinuation of one or both medications.
There is also an interesting paradox with the combination. Phentermine commonly causes insomnia and restlessness due to its stimulant properties, while tirzepatide commonly causes fatigue and low energy, particularly during dose escalation. Some patients report that these opposing effects partially cancel each other out, with phentermine providing energy during the day while tirzepatide reduces caloric intake throughout the full 24-hour cycle. This is anecdotal, not evidence-based, but it is a pattern some clinicians observe.
The body aches that some tirzepatide users report may also be affected by phentermine, since sympathomimetic stimulation can increase muscle tension and restlessness. If you are already experiencing musculoskeletal side effects on tirzepatide, adding a CNS stimulant could worsen those symptoms.
For anyone currently on the combination who is experiencing side effects, keeping a detailed symptom log with dates, times, severity ratings, and food intake is invaluable. This data allows your provider to determine which medication is driving which symptoms and whether dose adjustments to one or both drugs could improve tolerability without sacrificing efficacy.
Who should never combine phentermine and tirzepatide
Certain medical conditions create absolute contraindications for this combination. If any of the following apply to you, combining phentermine and tirzepatide is not a matter of risk tolerance or medical judgment. It is simply unsafe.
Absolute contraindications
Cardiovascular disease. This includes coronary artery disease, heart failure, uncontrolled hypertension, arrhythmias, or history of stroke. The combined cardiovascular stimulation from both drugs creates unacceptable risk for patients with compromised cardiac function.
Hyperthyroidism. Phentermine is contraindicated in hyperthyroid patients because sympathomimetic stimulation compounds the already elevated metabolic state. Adding tirzepatide does not reduce this risk.
Glaucoma. Phentermine can increase intraocular pressure through its adrenergic effects. This is a hard contraindication that applies regardless of what other medications are being taken.
MAO inhibitor use. Patients taking monoamine oxidase inhibitors or those who have taken them within the past 14 days cannot take phentermine. The interaction can cause hypertensive crisis.
Personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2 (MEN2). This is a tirzepatide-specific contraindication based on preclinical findings in rodent models.
Pregnancy or active breastfeeding. Both medications are contraindicated during pregnancy. Tirzepatide and breastfeeding is a topic that requires careful discussion with a healthcare provider, and getting pregnant while on tirzepatide raises separate concerns about fetal exposure during the critical early weeks.
History of drug abuse. Phentermine is a controlled substance with stimulant properties. Patients with a history of substance abuse are at elevated risk for psychological dependence, and this risk does not disappear simply because a GLP-1 medication is being taken simultaneously.
Relative contraindications
These are situations where the combination might be possible under very careful supervision but carry elevated risk that must be weighed against potential benefit:
Controlled hypertension (even well-managed blood pressure requires closer monitoring on this combination)
History of anxiety disorders (phentermine can exacerbate anxiety through CNS stimulation)
History of pancreatitis (GLP-1 receptor agonists including tirzepatide carry a theoretical risk of pancreatitis)
Gallbladder disease (rapid weight loss from combination therapy increases gallstone risk)
Severe gastrointestinal conditions including gastroparesis (tirzepatide further slows already-compromised gastric motility)
Kidney disease (dehydration risk from combined GI side effects can worsen renal function)
If you fall into any of these categories, the conversation with your provider needs to be particularly thorough. The risks may or may not outweigh the benefits depending on severity, current management, and overall health status. But the discussion must happen before any prescription is written.
What research actually says about the combination
The most honest assessment of the clinical evidence is this: there is almost none specific to phentermine plus tirzepatide. The combination exists in clinical practice based on pharmacological reasoning and extrapolation from related studies, not direct evidence.
Direct evidence
Zero published randomized controlled trials have evaluated phentermine combined with tirzepatide. No prospective cohort studies. No large-scale observational analyses. The specific combination has simply never been formally studied. Every published source, from drug interaction databases to clinical reviews, acknowledges this evidence gap.
Related evidence from GLP-1 combinations
The closest available data comes from studies of other GLP-1 agonists combined with phentermine. In a randomized placebo-controlled pilot study, researchers examined whether adding phentermine to patients already on liraglutide (a GLP-1 agonist) for one year would produce additional weight loss. The patients on liraglutide had already achieved approximately 12% weight loss. The result was not encouraging: adding phentermine did not provide meaningful additional weight loss compared to adding placebo.
This is only one study, with a single GLP-1 agonist that is less potent than tirzepatide. It does not definitively prove that phentermine plus tirzepatide would fail. But it introduces legitimate doubt about the additive benefit hypothesis and suggests that once incretin-based appetite suppression is maximized, additional norepinephrine-based suppression may have diminishing returns.
Tirzepatide monotherapy evidence
What we do have is robust evidence for tirzepatide alone. The SURMOUNT trial program provides extensive data on what tirzepatide achieves without any additional weight loss medication:
5 mg dose: 15.0% average weight loss at 72 weeks
10 mg dose: 19.5% average weight loss at 72 weeks
15 mg dose: 20.9% average weight loss at 72 weeks
At 15 mg, 56.1% of participants lost at least 20% of body weight
At 15 mg, 38.7% of participants lost at least 25% of body weight
These numbers are remarkable. They approach bariatric surgery outcomes without any surgical intervention. And they were achieved with tirzepatide alone, without phentermine or any other complementary medication. This context is essential: before accepting the risk of combining two drugs, you need to consider whether the tirzepatide dose has actually been maximized. Many patients struggling with weight loss on tirzepatide have not yet reached the 15 mg dose, and increasing the dose within the approved range may produce better results than adding a second medication.
The SURMOUNT-4 continuation trial further demonstrated that maintaining tirzepatide long-term preserves weight loss results, while discontinuation leads to significant weight regain. This underscores the importance of understanding the long-term trajectory of tirzepatide therapy rather than looking for short-term boosts from additional medications.
Dosage considerations if your doctor approves the combination
If after thorough evaluation your healthcare provider determines that combining phentermine and tirzepatide is appropriate for your specific situation, the dosing strategy matters enormously. This is not a situation where standard doses of both drugs get prescribed without modification.
Phentermine dosing in combination
Standard phentermine monotherapy dosing ranges from 15 mg to 37.5 mg daily. When combined with tirzepatide, many providers start at the lower end, 15 mg daily, and only increase if needed and tolerated. Some prescribe the 8 mg formulation taken up to three times daily rather than a single larger dose, which allows for more flexible dose adjustment and lower peak blood concentrations.
Timing matters significantly. Phentermine should be taken in the morning, ideally before breakfast or one to two hours after breakfast. Late-day dosing causes insomnia, which is already a concern with the CNS stimulation this drug produces. When combined with tirzepatide, which is typically injected once weekly on a consistent day, there is no specific timing interaction between the two. However, taking phentermine in the morning and injecting tirzepatide at a separate time of day can help you track which medication is causing which effects.
Tirzepatide dosing in combination
The standard tirzepatide titration schedule should generally be followed even when phentermine is being taken concurrently. Starting at 2.5 mg weekly for four weeks, then increasing to 5 mg, then 7.5 mg, 10 mg, 12.5 mg, and ultimately 15 mg as needed and tolerated. The four-week minimum at each dose level allows the body to adapt to the gastrointestinal effects before adding more.
Some providers may titrate tirzepatide more slowly when phentermine is on board, reasoning that the combined appetite suppression might allow lower tirzepatide doses to achieve target weight loss. This approach has the advantage of reducing GI side effects and potentially lowering long-term tirzepatide costs for patients using compounded formulations.
Understanding the unit-to-milligram conversion is important for accurate dosing. If you are using compounded tirzepatide, familiarize yourself with how many units equal 2.5 mg, 5 mg, 7.5 mg, and higher doses to ensure accuracy. Using a tirzepatide dosage calculator can help prevent dosing errors that become more consequential when a second medication is in the mix.
Duration planning
Because phentermine is FDA-approved only for short-term use (12 weeks), the combination should have a built-in exit strategy. Before starting, your provider should establish clear criteria for success (typically 5% additional weight loss within 12 weeks of adding phentermine) and a timeline for discontinuing phentermine regardless of results. Tirzepatide can and should continue long-term, but phentermine should be viewed as a temporary addition rather than a permanent part of the protocol.
Monitoring requirements for combination therapy
If you are on both phentermine and tirzepatide, the monitoring requirements exceed what either medication demands individually. This is not negotiable. The combined cardiovascular, metabolic, and gastrointestinal effects require proactive tracking to catch problems early.
Cardiovascular monitoring
Check resting heart rate and blood pressure at minimum weekly for the first month, then biweekly after that. A home blood pressure monitor with heart rate display is essential. Establish your baseline before starting the combination, and set clear thresholds with your provider for when to contact them. A resting heart rate consistently above 100 bpm or blood pressure consistently above 140/90 should prompt immediate clinical reassessment.
Blood sugar monitoring
If you are using tirzepatide for diabetes management, increase blood sugar monitoring frequency when phentermine is added. The accelerated weight loss and changes in food intake can alter insulin sensitivity rapidly. Your tirzepatide dose may need adjustment downward as weight decreases, and this adjustment timeline compresses when phentermine is producing additional caloric restriction. The interaction databases specifically note that diabetic medication requirements may change during phentermine treatment.
Weight and body composition tracking
Weekly weigh-ins at the same time under the same conditions provide the data needed to assess whether the combination is actually producing additional benefit beyond what tirzepatide alone was achieving. If weight loss does not accelerate within 8-12 weeks of adding phentermine, the cardiovascular risk is not justified and phentermine should be discontinued. Tracking before and after results with photos and measurements adds context that the scale alone cannot provide.
Gastrointestinal symptom tracking
Keep a daily log of nausea severity, bowel patterns, vomiting episodes, and food intake for at least the first month on the combination. This data helps your provider distinguish between tirzepatide-related GI effects (which often improve with time) and phentermine-related effects (which may persist as long as the drug is taken). Persistent vomiting or diarrhea that leads to dehydration is a reason to reduce or stop one of the medications promptly.
Mental health monitoring
Phentermine is a CNS stimulant that can cause or worsen anxiety, agitation, and mood changes. If you have any history of anxiety or mood disorders, monitoring these symptoms is critical when adding phentermine to your regimen. The combination of reduced food intake (from tirzepatide), stimulant effects (from phentermine), and rapid body composition changes can create psychological stress that deserves attention alongside the physical monitoring.
Laboratory monitoring
Your provider should order baseline labs before starting the combination and repeat them at 4-8 week intervals. Key markers include comprehensive metabolic panel (to monitor kidney function and electrolytes), lipid panel, hemoglobin A1c (for diabetic patients), liver function tests, and thyroid function. The combination can affect all of these, and catching abnormalities early prevents complications.
Phentermine vs tirzepatide: do you actually need both?
Before committing to combination therapy, it is worth stepping back and asking a blunt question: does adding phentermine to tirzepatide actually provide enough additional benefit to justify the added risk, monitoring burden, and cost?
Here is how the two medications compare as standalone treatments:
Factor | Phentermine alone | Tirzepatide alone |
|---|---|---|
Average weight loss | 5-10% over 12 weeks | 15-21% over 72 weeks |
Mechanism | CNS norepinephrine release | Dual GIP/GLP-1 receptor agonist |
FDA-approved duration | 12 weeks (short-term) | Long-term use |
Route | Oral (daily pill) | Subcutaneous injection (weekly) |
Appetite suppression onset | Hours (same day) | Days to weeks |
Cardiovascular effects | Raises HR and BP | Modest HR increase (2-4 bpm) |
GI side effects | Mild (constipation, dry mouth) | Significant (nausea, diarrhea) |
Cost | Low ($15-75/month generic) | Higher (varies by source) |
Tolerance development | Common over weeks | Uncommon at stable doses |
Controlled substance | Yes (Schedule IV) | No |
The data tells a clear story. Tirzepatide monotherapy produces dramatically greater weight loss than phentermine monotherapy. For the vast majority of patients, optimizing tirzepatide alone, reaching the 10-15 mg dose range, following a proper meal plan, and giving the medication 6-12 months to reach full effect, will produce better results than starting phentermine at any point in the process.
If you are on tirzepatide at 5 mg or 7.5 mg and feeling like it is not working, the answer is almost certainly dose optimization rather than adding a second medication. The microdosing approaches some patients use may also be contributing to suboptimal results. Before adding phentermine, exhaust the tirzepatide dose range. Talk to your provider about moving to 10 mg, 12.5 mg, or 15 mg if side effects are manageable.
Even for patients already at the maximum 15 mg tirzepatide dose who have plateaued, the evidence for adding phentermine is thin. The liraglutide-plus-phentermine pilot study found no meaningful additional benefit. And the risks of combination therapy, cardiovascular strain, amplified side effects, drug interactions, additional monitoring requirements, are not trivial.
The scenario where adding phentermine makes the strongest case is this: a patient on maximum tirzepatide dosing for at least 6 months, with a documented weight loss plateau lasting 8 or more weeks, who has optimized diet, exercise, sleep, and stress management, and whose provider has ruled out medical causes for the plateau (thyroid dysfunction, medication interactions, fluid retention). Even then, the addition should be short-term with clear success criteria and a defined discontinuation timeline.
For a detailed comparison with a different GLP-1, see our guide on phentermine vs semaglutide or the broader analysis of combining phentermine with semaglutide.
Transitioning from phentermine to tirzepatide
A far more common and clinically supported scenario than combining both medications is transitioning from phentermine to tirzepatide. Many patients start with phentermine because it is inexpensive, readily available, and provides immediate appetite suppression. When they are ready for a more effective long-term solution, tirzepatide becomes the next step.
The transition requires planning because of the different onset profiles of these medications.
The overlap problem
Phentermine works the same day you take it. Within hours, appetite suppression kicks in. Tirzepatide takes weeks to reach therapeutic blood levels, and the starting dose of 2.5 mg is sub-therapeutic for weight loss in most patients. If you abruptly stop phentermine and start tirzepatide at 2.5 mg, you will likely experience a period of 4-8 weeks where appetite suppression is minimal, which can lead to weight regain and frustration.
This gap is why some providers maintain a brief overlap period, continuing phentermine during the initial tirzepatide titration and gradually tapering phentermine as tirzepatide doses increase. A typical transition timeline might look like this:
Weeks 1-4: Start tirzepatide 2.5 mg weekly while maintaining phentermine at current dose
Weeks 5-8: Increase tirzepatide to 5 mg weekly, reduce phentermine to 15 mg daily (if currently on a higher dose)
Weeks 9-12: Increase tirzepatide to 7.5 mg weekly, begin tapering phentermine to every other day
Week 13+: Discontinue phentermine, continue tirzepatide titration as needed
This is a general framework, not a prescription. The actual transition timeline should be individualized based on your response to both medications, your weight loss trajectory, and your side effect profile. Some patients can discontinue phentermine sooner because tirzepatide appetite suppression kicks in faster for them. Others may need a longer overlap.
What to expect during the transition
Expect some fluctuation in appetite and energy levels as you shift between the two mechanisms. Phentermine provides a stimulant-based appetite suppression that also boosts energy and alertness. Tirzepatide provides a more gradual, hormonally-mediated appetite reduction that some people experience as initial fatigue before the metabolic benefits become apparent. The first few weeks after dropping phentermine may feel like a step backward even though tirzepatide is building toward much greater long-term efficacy.
Some providers who manage both transitions between GLP-1 medications and transitions from traditional appetite suppressants to GLP-1 therapy report that patients who understand the transition timeline and set appropriate expectations have much better adherence and outcomes than those who expect immediate equivalence between the two medication types.
If you are currently on semaglutide rather than phentermine and considering tirzepatide, the process is different. See our guide on switching between tirzepatide and semaglutide for specific protocols.
Common mistakes when combining weight loss medications
Even when physician-supervised, combination weight loss therapy is prone to specific errors that undermine results and increase risk. Understanding these mistakes helps you advocate for yourself and ask the right questions.
Mistake 1: Not maximizing the single agent first
The most common mistake is adding phentermine before tirzepatide has been fully optimized. If you have not reached the 10-15 mg tirzepatide dose range, you have not given monotherapy a fair trial. Jumping to combination therapy at subtherapeutic tirzepatide doses means you are accepting the risks of two drugs without extracting the full benefit of the more effective one. Check your current dosage against the standard titration chart before considering additions.
Mistake 2: No exit strategy
Starting phentermine without a predefined discontinuation plan leads to indefinite use of a drug approved for only 12 weeks. Before the first pill, establish clear success criteria (e.g., additional 5% weight loss within 12 weeks) and a firm stop date. If the criteria are not met, phentermine gets discontinued, not continued indefinitely in hopes that it might eventually work.
Mistake 3: Ignoring diet optimization
Adding a second medication to fix a diet problem never works long-term. If your caloric intake is not aligned with your weight loss goals, or if you are eating foods that exacerbate tirzepatide side effects, adding phentermine will not solve the underlying issue. Address nutrition fundamentals and build a sustainable eating plan before layering on additional pharmacology.
Mistake 4: Inadequate monitoring
Combination therapy with no monitoring is reckless. If your provider prescribes both medications but does not establish a monitoring plan (blood pressure checks, labs, scheduled follow-ups), that is a serious red flag about the quality of care. You should be seen at minimum monthly while on combination therapy, with more frequent contact during the initial overlap period.
Mistake 5: Self-prescribing
Some individuals obtain phentermine through telehealth or other channels and add it to tirzepatide they are sourcing independently. This eliminates the medical oversight that makes combination therapy even remotely reasonable. Without a provider reviewing your cardiovascular health, checking for contraindications, and monitoring for complications, combining these drugs yourself is a gamble with genuinely dangerous stakes.
Mistake 6: Neglecting lifestyle factors
Weight loss medications work best as part of a comprehensive approach. Exercise, sleep quality, stress management, and proper supplementation all influence outcomes. If you are not sleeping 7-8 hours, not engaging in regular resistance training, and not managing stress, adding phentermine will not compensate for those deficits. Fix the foundation before adding more pharmacology.
What to do if you are currently taking both
If you are already on phentermine and tirzepatide together, this section is specifically for you. Whether your combination was prescribed by a physician or self-initiated, here is what you should be doing right now.
First, establish monitoring if you have not already. Get a home blood pressure monitor. Check your resting heart rate and blood pressure at the same time each morning before taking phentermine. Record the numbers. If resting heart rate is consistently above 100 bpm or blood pressure is above 140/90, contact your provider immediately. These thresholds are not arbitrary. They represent the point where cardiovascular risk becomes clinically significant.
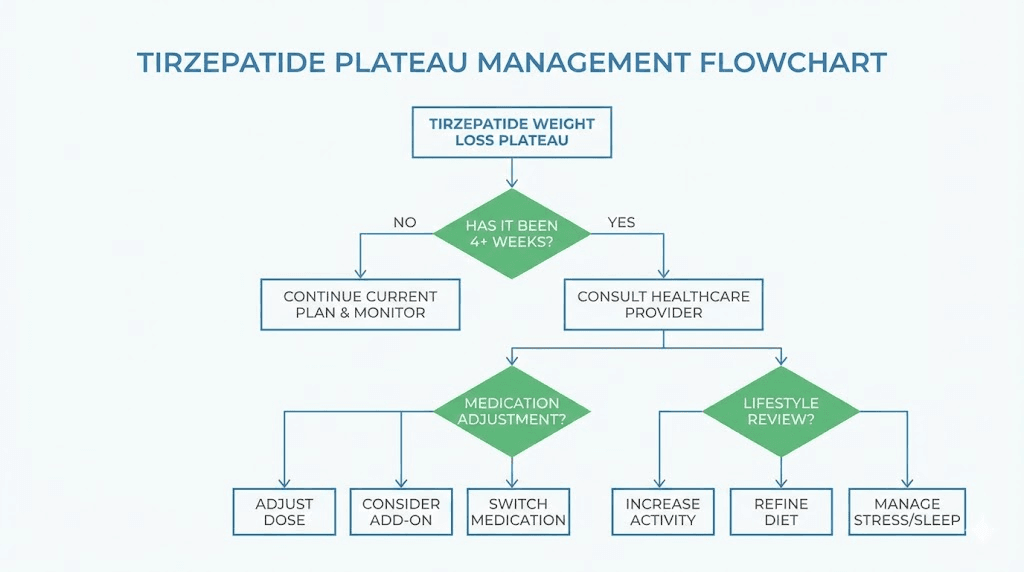
Second, track your results objectively. Weigh yourself weekly under consistent conditions (same time, same clothing, same scale). If you have been on the combination for 8 or more weeks without measurable weight loss improvement beyond what tirzepatide alone was producing, the phentermine is adding risk without benefit and should be discontinued.
Third, have an honest conversation with your prescriber. If different providers prescribed each medication, make sure both are aware of the combination. Fragmented care where one provider does not know what the other prescribed is a recipe for dangerous interactions going undetected. If you self-initiated the combination, talk to a provider about what you are doing. The information they need to keep you safe requires full transparency.
Fourth, have a discontinuation plan. Phentermine should not be taken indefinitely. Work with your provider to establish when you will stop phentermine, what the tapering schedule looks like, and how you will manage the transition to tirzepatide monotherapy. The goal is always to reach a sustainable, single-agent maintenance protocol for long-term weight management.
Fifth, optimize everything else. While on combination therapy, your body is under significant pharmacological pressure. Support it with proper hydration (dehydration risk increases with both drugs), adequate protein intake to preserve lean mass, nutrient-dense foods to prevent deficiencies from reduced caloric intake, and avoid alcohol which compounds both the cardiovascular and gastrointestinal risks.
Alternative approaches to consider before combining medications
Before taking the step of adding phentermine to tirzepatide, consider whether any of these evidence-based alternatives might address your plateau or treatment goals without the added risk of combination therapy.
Dose optimization
If you are not yet at the maximum tirzepatide dose of 15 mg weekly, increasing your dose is the most straightforward approach. Each dose increase from 5 mg to 10 mg to 15 mg produces substantially more weight loss in clinical trials. Review your current dosing with your provider and discuss whether titration upward makes sense for your situation.
Compound formulation changes
Some patients find that compounded tirzepatide with glycine and B12 or tirzepatide with niacinamide provides enhanced effects compared to standard formulations. While this is largely anecdotal, the addition of glycine in particular has gained attention for potentially supporting metabolic function during weight loss.
Dietary restructuring
A registered dietitian who understands GLP-1 therapy can help restructure your meal plan to maximize weight loss without additional medication. Caloric quality matters as much as quantity, and many patients plateau because of nutrient timing, macronutrient distribution, or hidden caloric sources rather than insufficient appetite suppression.
Exercise protocol adjustment
Adding or modifying resistance training can break weight loss plateaus by increasing basal metabolic rate through lean muscle preservation. Weight loss medications, including tirzepatide, cause some degree of lean mass loss alongside fat loss. Resistance training helps shift the ratio toward fat loss specifically, which may restart scale movement even when total caloric intake has not changed.
Switching GLP-1 medications
If tirzepatide has plateaued, some providers consider switching to or adding a different GLP-1 compound rather than adding phentermine. Newer agents like survodutide, orforglipron, or mazdutide are in various stages of development and clinical use, and retatrutide (a triple agonist targeting GIP, GLP-1, and glucagon receptors) shows even greater weight loss potential than tirzepatide in early trials.
Sleep and stress optimization
Chronic sleep deprivation and elevated cortisol from stress are proven contributors to weight loss resistance. Addressing these factors costs nothing and carries zero pharmaceutical risk. Before adding phentermine, get a sleep study if you suspect sleep apnea, establish consistent sleep and wake times, and implement evidence-based stress management practices.
For comprehensive support with all these approaches, SeekPeptides offers evidence-based guides, detailed protocols, and a community of experienced researchers who have navigated these exact optimization questions.
The role of proper storage and handling
When using any weight loss medication, and especially when using multiple medications concurrently, proper storage and handling directly affect efficacy. A degraded medication that has lost potency can mimic a plateau, leading you to add phentermine when the real problem is that your tirzepatide has been compromised.
Tirzepatide requires refrigeration before first use. Understanding tirzepatide storage requirements and how long it lasts in the fridge protects against potency loss. Compounded formulations may have different stability profiles, and knowing how long compounded tirzepatide can be unrefrigerated prevents using degraded medication. Check expiration dates and discard any medication that has been improperly stored.
For compounded tirzepatide that requires reconstitution, proper reconstitution technique is critical for accurate dosing. Incorrect reconstitution can result in either over-dosing (increased side effects) or under-dosing (perceived plateau that is actually a preparation error). If you are self-administering, review your injection technique periodically to ensure accuracy.
Phentermine, being an oral medication, is more straightforward to store but still requires protection from heat and moisture. Keep it in a cool, dry place in its original container. If you are traveling with both medications, travel storage guidelines for injectable peptides apply to tirzepatide.
Understanding your options beyond this combination
The weight loss medication landscape is evolving rapidly, and the phentermine-plus-tirzepatide combination represents just one possible approach among an expanding array of options. Understanding what else exists helps you make decisions with full context rather than tunnel vision focused on only these two drugs.
Oral tirzepatide formulations are in development and may eventually provide the convenience of a daily pill with the efficacy of current injectable tirzepatide. The comparison between oral and injectable tirzepatide is already a topic of active research and clinical interest. For patients who dislike injections, tirzepatide tablets or sublingual drops may eventually provide alternatives that achieve similar results without the need for combination therapy.
The addition of supportive compounds to tirzepatide is another area of active exploration. Tirzepatide with vitamin B12 and tirzepatide with glycine are formulations that some compounding pharmacies offer, aiming to support energy and metabolic function during weight loss. While these additions carry far less risk than combining with phentermine, evidence for their additional benefit is still limited.
Combination peptide approaches are also gaining attention. AOD-9604 with tirzepatide represents one such pairing that targets different fat loss mechanisms without the cardiovascular concerns associated with phentermine.
For those researching medications through SeekPeptides, the platform provides updated information on emerging weight loss compounds, including comparisons, protocols, and safety profiles that help researchers make informed decisions about their approach.
Frequently asked questions
Is combining phentermine and tirzepatide FDA-approved?
No. This combination has not been approved by the FDA, and no clinical trial has evaluated its safety or efficacy. All prescriptions for the combination are off-label. The phentermine prescribing information specifically warns against combining it with other weight loss medications.
Can phentermine help break a tirzepatide weight loss plateau?
The evidence is weak. A pilot study of phentermine added to liraglutide (a related GLP-1 agonist) showed no meaningful additional weight loss. While tirzepatide is a different and more potent drug, this study introduces doubt about the additive benefit theory. Before adding phentermine, consider optimizing your tirzepatide dose and addressing lifestyle factors.
What is the biggest risk of taking both together?
Cardiovascular strain. Phentermine raises heart rate and blood pressure through sympathetic nervous system stimulation. Tirzepatide causes a modest heart rate increase (2-4 bpm average). Combined, these effects can produce clinically significant cardiovascular stress, particularly in patients with pre-existing heart conditions or uncontrolled hypertension. Regular monitoring of side effects is essential.
How long can you take phentermine alongside tirzepatide?
Phentermine is FDA-approved for only 12 weeks (3 months) of continuous use. When combined with tirzepatide, most providers limit phentermine to the same timeframe or shorter. Clear success criteria should be established upfront, and phentermine should be discontinued if those criteria are not met within the defined period.
Does tirzepatide affect how phentermine is absorbed?
Yes, potentially. Tirzepatide slows gastric emptying, which can alter the absorption rate and peak blood levels of oral medications including phentermine. The clinical significance of this specific interaction has not been formally studied, but it is a known consideration with all oral medications taken alongside GLP-1 receptor agonists.
What should I eat while taking both medications?
Focus on nutrient-dense, easily digestible foods that minimize gastrointestinal distress. Lean proteins, cooked vegetables, and complex carbohydrates in small, frequent meals tend to be best tolerated. Review our guides on what to eat on tirzepatide and foods to avoid for detailed recommendations. Stay well-hydrated, as both medications can contribute to dehydration through different mechanisms.
Can I switch from phentermine to tirzepatide instead of combining them?
Yes, and this is the more commonly recommended approach. Transitioning from phentermine to tirzepatide involves a brief overlap period while tirzepatide titrates to therapeutic levels, followed by phentermine discontinuation. This approach gives you the superior long-term efficacy of tirzepatide, as demonstrated in the SURMOUNT trial results, without the ongoing risks of combination therapy.
Should I tell my doctor if I am taking both without a prescription?
Absolutely. Full transparency with your healthcare provider is essential for your safety. They cannot monitor for complications or adjust treatment appropriately if they do not know what you are taking. There is no judgment involved, only a genuine need to manage risk. Bring both medications to your next appointment and discuss your current protocol honestly.
External resources
For researchers committed to optimizing their weight loss protocols safely, SeekPeptides offers the most comprehensive resource available. Members access evidence-based medication guides, detailed dosing protocols, interaction databases, and a community of thousands who have navigated these exact decisions with real-world experience and clinical guidance.
In case I do not see you, good afternoon, good evening, and good night. May your medications stay properly dosed, your monitoring stay consistent, and your weight loss journey stay safe.