Feb 19, 2026

After analyzing dozens of independent testing reports, reviewing hundreds of customer experiences, and comparing retatrutide results across multiple vendors, one pattern stands out with uncomfortable clarity. Not every supplier delivering research peptides meets the same standard. Some fall far short. And when it comes to a compound as novel and consequential as retatrutide, the gap between a reliable source and a questionable one is not just an inconvenience. It is a fundamental research integrity problem.
Peptide Sciences is one of the most recognized names in the research peptide market. The company has built a reputation over years of operation, and many researchers default to it out of familiarity. But recognition does not equal reliability, especially when independent third-party testing data tells a very different story than a vendor marketing page. The question researchers should be asking is not whether Peptide Sciences is well-known. The question is whether their retatrutide actually contains what the label claims.
This guide breaks down everything a researcher needs to know about sourcing retatrutide from Peptide Sciences. We cover the company background, the science behind retatrutide as a triple receptor agonist, real third-party testing data including identity failures and quantity discrepancies, how to read certificates of analysis properly, what quality markers matter most, and how Peptide Sciences stacks up against competing vendors. Whether you are new to buying retatrutide or reconsidering your current source, this analysis provides the data you need to make a genuinely informed decision. Use the peptide calculator at SeekPeptides alongside this guide to verify your own preparation numbers as you evaluate vendors.
What is Peptide Sciences
Peptide Sciences operates as a United States-based supplier specializing in research-grade peptides. The company has positioned itself as a premium vendor, emphasizing purity claims and professional packaging. Their website lists an extensive catalog of peptides spanning metabolic research compounds, growth hormone secretagogues, healing peptides, and more. For researchers exploring the retatrutide availability landscape, Peptide Sciences appears frequently in search results and vendor discussions.
The company claims to use solid-phase peptide synthesis, commonly abbreviated as SPPS. This is the standard manufacturing method for research peptides. They also claim that each batch undergoes High-Performance Liquid Chromatography testing and Mass Spectrometry analysis, with Certificates of Analysis provided for every product. On paper, these are exactly the quality assurance steps a researcher would want to see.
But there is a critical difference between running internal tests and submitting products to independent laboratories.
Peptide Sciences pricing sits at the higher end of the market. Their retatrutide cost per milligram exceeds what many competing vendors charge, sometimes by a significant margin. Premium pricing often implies premium quality, and many researchers accept this logic without questioning it. The assumption is reasonable but not always accurate. Pricing reflects manufacturing costs, overhead, and profit margins. It does not inherently guarantee that the compound in your vial matches the compound on the label. Understanding the full retatrutide online marketplace requires looking beyond price tags alone.
Customer reviews for Peptide Sciences paint a mixed picture. Positive feedback tends to highlight professional packaging, sealed vials, smooth reconstitution, and prompt delivery. Researchers who had no issues report satisfaction with the ordering process and the physical appearance of the product. Negative feedback clusters around a few recurring themes. Customer support response times can be slow. The company does not offer refunds or returns, and replacement policies are extremely limited. Some customers have reported receiving vials that appeared cloudy after reconstitution, or products that did not produce expected research outcomes. These experiences raise questions, but they do not constitute definitive evidence of quality failure on their own.
What does constitute definitive evidence is laboratory data. And that is where the story becomes considerably more concerning for researchers considering Peptide Sciences as their retatrutide peptide buy source. Before we examine that testing data, it helps to understand exactly what retatrutide is, why it matters, and what makes it particularly challenging to source reliably on the grey market.
Retatrutide explained
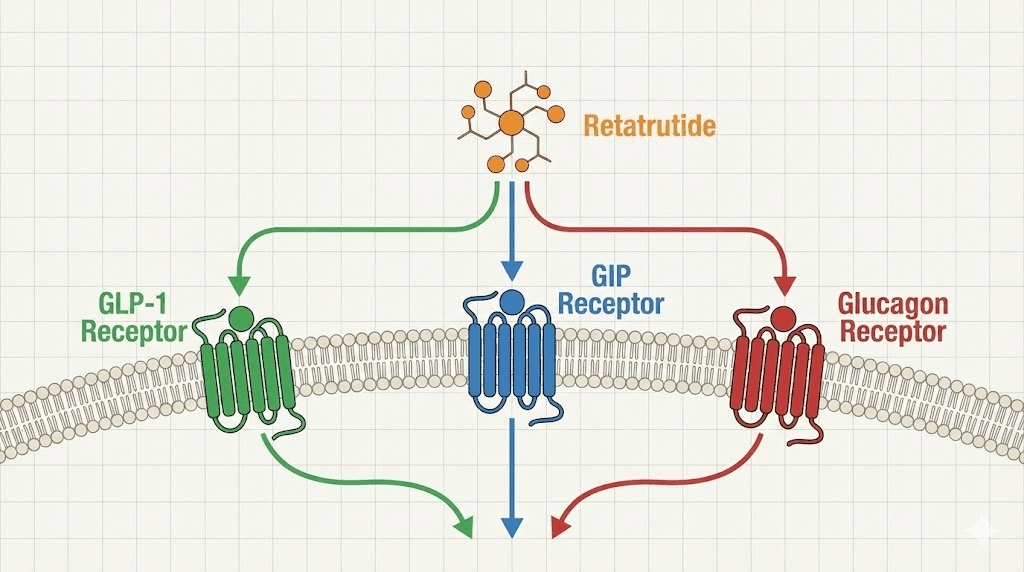
Retatrutide is not just another incretin mimetic. It represents a fundamentally different approach to metabolic modulation. While compounds like semaglutide target a single receptor and tirzepatide targets two, retatrutide activates three distinct hormone receptors simultaneously. This triple agonist mechanism is what separates it from everything that came before.
The triple receptor mechanism
Retatrutide, designated LY3437943 by its developer Eli Lilly, is a 39-amino acid peptide linked to a C20 fatty diacid moiety that extends its half-life to approximately six days. This structural feature allows once-weekly administration, similar to tirzepatide dosing schedules. But the pharmacological profile goes further than any dual agonist.
The three receptors it targets work in concert.
GLP-1 receptor activation enhances glucose-stimulated insulin secretion, slows gastric emptying, and promotes satiety. This is the same mechanism that makes semaglutide appetite suppression effective. It reduces caloric intake by signaling fullness to the brain and moderating postprandial blood sugar spikes. Most researchers are already familiar with GLP-1 agonism through compounds like semaglutide and liraglutide, and the comparison between retatrutide vs semaglutide starts here but extends much further.
GIP receptor activation facilitates insulin secretion in a glucose-dependent manner and plays an important role in lipid metabolism. This receptor contributes to reductions in fat deposition and improvements in overall energy balance. GIP agonism is what elevates tirzepatide above single-target GLP-1 drugs, and retatrutide incorporates this same advantage. Researchers studying the differences between semaglutide vs tirzepatide mechanisms will recognize the significance of adding GIP signaling.
Glucagon receptor activation is the third and most distinctive element. This is what makes retatrutide genuinely novel. Glucagon receptor agonism promotes energy expenditure through increased thermogenesis and lipid mobilization. It stimulates hepatic glucose production in a controlled manner while driving the body to burn stored fat more aggressively. The comparison between mazdutide vs retatrutide often centers on this glucagon component, as different triple agonist candidates weight these receptor interactions differently.
Together, these three mechanisms create a compound that simultaneously reduces appetite, improves insulin sensitivity, and actively increases energy expenditure. It is not simply suppressing hunger. It is reshaping metabolic function at multiple levels.
Clinical trial results
The clinical data for retatrutide is extraordinary by any standard in obesity pharmacotherapy.
In the Phase 2 trial published in the New England Journal of Medicine, the 12mg dose of retatrutide produced a mean weight reduction of 24.2% after 48 weeks. To put that in perspective, the 1mg group saw 8.7% reduction, the 4mg group achieved 17.1%, the 8mg group reached 22.8%, and the placebo group lost just 2.1%. These are numbers that reshape the entire conversation about pharmacological weight management. Researchers tracking the retatrutide dosage chart across these trials can see a clear dose-response relationship.
Phase 3 results from the TRIUMPH-4 trial pushed the data even further. At the 12mg dose over 68 weeks, participants achieved an average weight loss of 71.2 pounds, corresponding to a 28.7% reduction in body weight. The 9mg dose produced 26.4% weight loss. Placebo achieved just 2.1%. These results exceed anything previously documented for a single injectable compound and explain why interest in retatrutide dose protocols has surged across the research community.
Beyond weight reduction, the metabolic benefits extend to cardiovascular risk markers. Clinically meaningful improvements in non-HDL cholesterol, high-sensitivity C-reactive protein, and triglycerides were observed. The retatrutide dose schedule used in trials showed consistent improvements across these markers, suggesting systemic metabolic remodeling rather than simple caloric restriction.
Liver fat data is particularly striking. In patients with metabolic dysfunction-associated steatotic liver disease, retatrutide at 8mg and 12mg doses resolved steatosis in over 85% of subjects. Total liver fat content dropped below 5% in 89% of participants at the 8mg dose and 93% at 12mg. These numbers suggest a compound that addresses fatty liver disease with a potency that no existing treatment matches.
Safety profile from clinical trials
The most common adverse events in clinical trials were gastrointestinal in nature, consistent with the GLP-1 component of the mechanism. At the 12mg dose, nausea affected 43.2% of participants, diarrhea occurred in 33.1%, constipation in 25.0%, and vomiting in 20.9%. Placebo rates were substantially lower at 10.7%, 13.4%, 8.7%, and lower respectively. Researchers familiar with semaglutide vs tirzepatide side effects will recognize this profile as an amplified version of what single and dual agonists produce.
No cases of clinically significant hypoglycemia, medullary thyroid cancer, or C-cell hyperplasia were reported. This safety data comes from controlled clinical environments with pharmaceutical-grade retatrutide manufactured under GMP conditions. It does not apply to grey market research compounds of uncertain purity, identity, or concentration.
That distinction matters enormously. And it leads directly to the question of what Peptide Sciences is actually delivering when researchers order retatrutide from their catalog.
Peptide Sciences retatrutide product analysis
Peptide Sciences lists retatrutide in their catalog as a research peptide available in lyophilized form. The product comes in standard research vial sizes, typically 5mg and 10mg quantities. Their product page emphasizes purity testing via HPLC and identity confirmation through mass spectrometry, with a Certificate of Analysis available for each batch. The packaging is professional, with sealed vials, clear labeling, and tamper-evident closures. On the surface, this mirrors what researchers expect from a premium vendor.
Pricing for Peptide Sciences retatrutide sits notably above the market average. Researchers comparing retatrutide cost across vendors will find that Peptide Sciences charges more per milligram than many competing suppliers. The premium pricing creates an implicit suggestion of superior quality. Many researchers, particularly those new to the peptide sourcing landscape, accept higher prices as a proxy for higher reliability. This is a dangerous assumption to make without verifying it through independent data.
The company COA for retatrutide typically shows purity readings in the high 98-99% range, molecular weight confirmation via mass spectrometry, and standard appearance and solubility documentation. These internal COAs represent the company own testing, performed on their own equipment, with their own standards. They are not meaningless documents, but they lack the objectivity that independent third-party testing provides.
For researchers calculating their preparation parameters, the retatrutide dosage calculator on SeekPeptides can help determine exact reconstitution volumes. But those calculations are only meaningful if the vial actually contains the stated amount of the correct compound. This is the fundamental issue that separates theoretical preparation from practical research reliability, and it is where third-party testing data becomes essential reading for every researcher.
What the COA claims versus what the lab finds
A Certificate of Analysis from a vendor is a statement of what they believe, or claim, their product to be. An independent laboratory test is a statement of what the product actually is. When these two documents agree, the researcher can proceed with confidence. When they diverge, the vendor COA becomes a marketing document rather than a quality assurance document.
The divergence between Peptide Sciences internal COA claims and independent testing results is significant enough to warrant serious concern. Understanding how to evaluate these documents is a skill every researcher should develop, particularly when working with novel compounds where the stakes of misidentification are high. We cover this in detail in the third-party testing section below.
Third-party testing results
This is where the conversation moves from marketing claims to verified data. And the data is not favorable for Peptide Sciences retatrutide.
Finnrick Analytics overview
Finnrick Analytics operates as an independent product testing platform for research peptides. They use accredited laboratories to perform identity verification, purity analysis, and quantity measurement on peptide samples sourced from registered vendors. Their database provides a vendor-neutral assessment that researchers can use to evaluate suppliers on the basis of actual laboratory performance rather than marketing claims. The platform has tested over 1,700 retatrutide samples from more than 130 vendors, creating one of the most comprehensive quality databases available for this compound.
Each vendor receives a rating based on their aggregate test performance. Ratings range from A (Great) to E (Bad), with the rating reflecting consistency across multiple tests, not just a single batch result. This cumulative approach means that a vendor cannot game the system with one good test while delivering inconsistent quality across other batches.
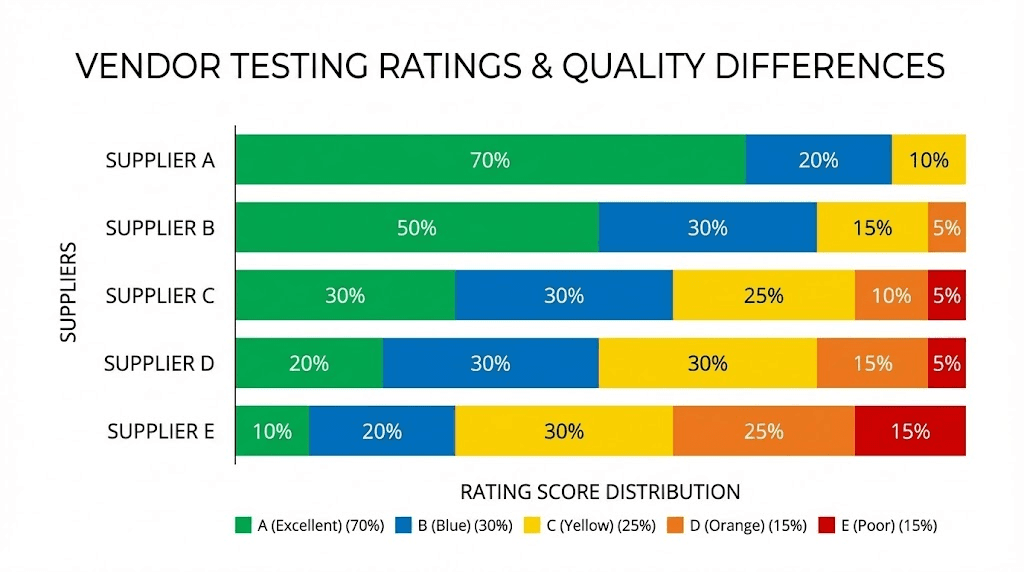
Peptide Sciences retatrutide rating: E (Bad)
Peptide Sciences received an E rating for retatrutide from Finnrick Analytics. This is the lowest possible rating.
That sentence deserves to stand alone. Let it sink in.
The E rating was assigned based on 36 tested samples collected between late December of a recent testing period through early the following year. The average score across these samples was 6.1 out of 10, with individual scores ranging from 2.0 to 10.0. This range is itself a problem. A score of 2.0 indicates a catastrophic quality failure. A range that spans from 2.0 to 10.0 shows extreme inconsistency from batch to batch. A researcher ordering retatrutide from Peptide Sciences has no way of knowing whether they will receive a 10.0 batch or a 2.0 batch until they test it themselves, or experience unexpected results in their research. Researchers comparing this with vendors like those reviewed in our Paradigm Peptides retatrutide analysis will see a stark contrast.
Identity failures
The most alarming finding from Finnrick testing is not purity variation. It is identity failure.
Multiple samples purchased from Peptide Sciences and labeled as retatrutide were found to actually contain tirzepatide or unidentified compounds entirely. This means researchers who ordered retatrutide, a triple GLP-1/GIP/glucagon agonist, received a dual GLP-1/GIP agonist instead, or something that could not even be identified. The implications are severe. Any research results generated with a misidentified compound are fundamentally compromised. The data cannot be trusted. The conclusions drawn from that data cannot be trusted. The entire research effort is contaminated at its foundation.
For a researcher studying the differences between retatrutide vs semaglutide or investigating the transition from tirzepatide to retatrutide, receiving tirzepatide in a vial labeled retatrutide would produce profoundly misleading results. The glucagon receptor component that makes retatrutide unique would be entirely absent, yet the researcher would attribute the observed effects to a triple agonist mechanism. This is not a minor quality variation. It is a complete misrepresentation of the product.
The identity verification process uses mass spectrometry to confirm the molecular weight and fragmentation pattern of the compound. When mass spectrometry identifies a different compound than what the label states, there is no ambiguity. The product is not retatrutide. It is something else. Researchers familiar with Peptide Sciences tirzepatide products should note the irony. Some retatrutide vials contained the very compound sold separately under a different product listing.
Quantity discrepancies
Beyond identity failures, Finnrick testing revealed significant quantity discrepancies in samples that did contain actual retatrutide. One 6mg vial was found to contain 8.75mg, representing a 45.8% overfill. While receiving more product than advertised might initially seem beneficial, it creates serious problems for research accuracy. A researcher using the retatrutide dosage calculator to determine precise concentrations would be working with fundamentally incorrect numbers. Every downstream calculation, from concentration per unit volume to administered research quantities, would be wrong.
In another direction, a 12mg vial contained only 9.36mg, a 22.0% shortfall. This means a researcher paid for 12mg, performed reconstitution calculations based on 12mg, and administered research quantities that were 22% lower than intended. At early stages of dose-response research, this level of inaccuracy can make the difference between observing an effect and missing it entirely. It can also lead researchers to incorrectly conclude that higher doses are needed when the actual problem is underfilled vials.
These quantity variations are not within acceptable manufacturing tolerances. Pharmaceutical manufacturing standards typically allow for variation of plus or minus 5% to 10%, depending on the product. A 45.8% overfill or 22.0% underfill is wildly outside any reasonable quality specification. Understanding how much retatrutide is actually in a vial becomes impossible without independent verification under these conditions.
Purity versus identity
One detail worth noting from the Finnrick data is that purity readings for Peptide Sciences samples ranged from 98.83% to 100%. On the surface, this sounds excellent. But purity without correct identity is meaningless. A vial that is 99.5% pure tirzepatide is not 99.5% pure retatrutide. It is 0% retatrutide. Purity measures how much of the sample is the identified compound versus impurities. If the identified compound is wrong, the purity number tells you that you have a very clean sample of the wrong peptide. This distinction is critical and often misunderstood by researchers who focus on purity percentages without verifying identity first.
How Peptide Sciences compares to other retatrutide vendors
Context matters. An E rating for Peptide Sciences carries more weight when you see what other vendors achieve on the same tests, from the same independent laboratories, using the same methodologies.
Top-rated vendors
Several vendors received A ratings from Finnrick Analytics for retatrutide. These include Paradigm Peptide, Orbitrex, and Peptide Partners. An A rating indicates consistent quality across multiple tests, with high identity confirmation rates, accurate quantity measurements, and strong purity scores. The gap between an A-rated vendor and an E-rated vendor is not a subtle difference. It represents fundamentally different levels of product reliability. Our detailed Paradigm Peptides retatrutide review covers one of these top-performing vendors in depth.
For researchers who have been defaulting to Peptide Sciences based on name recognition, the independent testing data suggests that switching to a higher-rated vendor could dramatically improve research reliability. The retatrutide buy guide on SeekPeptides covers how to evaluate vendors using multiple criteria beyond brand familiarity.
The pricing paradox
Peptide Sciences charges more than many vendors who receive significantly better independent testing ratings. This creates what researchers sometimes call the "premium pricing paradox." The expectation is that higher prices correlate with higher quality. In the case of Peptide Sciences retatrutide, the data shows the opposite relationship. Researchers are paying more money for less reliable product.
Some competing vendors with A ratings offer retatrutide at lower per-milligram costs. This does not mean the cheapest vendor is always the best, as cheap compounded options sometimes carry their own risks. But it does mean that price alone is an unreliable indicator of quality. Independent testing data is the only metric that actually answers the question of what is in the vial.
Vendor consistency versus single-batch results
One argument sometimes made in defense of vendors with mixed testing results is that a single bad batch does not represent the overall quality. This is true in the narrowest sense. But Peptide Sciences E rating is not based on one bad test. It reflects 36 samples tested over more than a year. The pattern is established. The inconsistency is the defining characteristic. A researcher cannot reliably predict whether their specific order will be among the well-tested batches or the failed ones. This uncertainty alone should give any serious researcher pause when comparing retatrutide online sources.
High-rated vendors demonstrate consistency across dozens of tests. Their quality is predictable, batch after batch. This consistency is what enables reliable research. Without it, every new vial introduces an uncontrolled variable into the experimental design, and that variable could be as extreme as receiving an entirely different compound.
Other vendor comparisons in the GLP-1 space
The pattern of vendor quality variation extends beyond retatrutide. Researchers working with other incretin mimetics should apply the same scrutiny. Those exploring tirzepatide sources should read about Lavender Sky tirzepatide, Empower tirzepatide, and Peptide Sciences tirzepatide reviews. For semaglutide, vendor analyses include Olympia semaglutide, Empower pharmacy semaglutide, and Direct Meds semaglutide. The GLP-1 agonist marketplace includes vendors across a wide quality spectrum, from Thrive GLP-1 and Willow GLP-1 to Evolv GLP-1 and LifeVantage GLP-1 offerings. Each requires its own due diligence.
Understanding certificates of analysis
A Certificate of Analysis is only as useful as the researcher ability to read it critically. Too many researchers glance at the purity percentage, see a number above 98%, and assume everything is fine. That approach misses the most important information on the document.
The three pillars of COA evaluation
Identity confirmation is the first and most critical element. This tells you whether the compound in the vial is actually what the label says it is. Identity is typically confirmed through mass spectrometry, where the observed molecular weight is compared against the theoretical molecular weight calculated from the amino acid sequence. For retatrutide, the theoretical molecular weight is specific to its 39-amino acid structure linked to the C20 fatty diacid moiety. If the observed mass does not match the theoretical mass within accepted tolerance, usually plus or minus 0.5 to 1.0 Daltons for singly charged ions, the identity is not confirmed. Period. No amount of high purity compensates for wrong identity.
Purity analysis is the second pillar. HPLC separates the target peptide from synthesis byproducts, deletion sequences, truncated fragments, and other impurities. The resulting chromatogram should show a single dominant peak comprising 95% or more of the total area. Secondary peaks indicate impurities. Purity above 98% is generally considered research-grade, while purity above 99% is excellent. Researchers working with reconstituted peptides should note that purity can degrade after reconstitution if storage conditions are improper.
Quantity verification is the third pillar, and it is the one most often overlooked. A COA should state the amount of peptide in the vial. But without independent verification, you are trusting the vendor claim that a 10mg vial actually contains 10mg. As Finnrick data showed for Peptide Sciences, actual quantities can deviate by 22% below to 46% above the labeled amount. Researchers relying on a retatrutide dosage calculator are only getting accurate results if the input quantity is itself accurate.
Red flags on a COA
Several indicators should raise immediate concern when reviewing a Certificate of Analysis.
Missing lot or batch numbers make it impossible to trace the COA to a specific production run. A COA without a lot number could be a generic template applied to multiple batches regardless of their actual test results. Always verify that the lot number on your vial matches the lot number on the COA.
Absence of chromatograms or spectra is another red flag. A complete COA should include the actual HPLC chromatogram and mass spectrum, not just the reported numbers. Without these raw data outputs, there is no way to verify that the testing was actually performed. Some vendors provide only a table of results without supporting analytical data.
Internal-only testing, meaning testing performed only by the vendor without independent laboratory verification, reduces the objectivity of the results. It is not that internal testing is worthless. But a vendor has an inherent financial interest in their products passing quality tests. Independent third-party testing removes this conflict of interest. When evaluating any vendor, including those reviewed in our ZLZ peptide retatrutide analysis, always ask whether the COA represents internal or independent testing.
Suspiciously perfect results across every batch should also raise questions. Real manufacturing processes produce variation. If every single COA from a vendor shows exactly 99.2% purity and exactly the labeled quantity with no variation whatsoever, the testing may be less rigorous than it appears. Legitimate testing reveals real variation within acceptable ranges.
How to request and verify a COA
Responsible researchers should request the COA before purchasing, not after. This allows evaluation of the vendor quality documentation before committing funds. When you receive a COA, cross-reference the lot number with your received product. Verify that the molecular weight matches published values for the compound. Check that the chromatogram shows a clean, dominant peak. And whenever possible, compare the vendor COA with independent testing data from platforms like Finnrick. The peptide cost calculator can help you understand whether you are paying a fair price relative to the documented quality level.

Retatrutide quality markers every researcher should check
Beyond the COA itself, several quality markers help distinguish reliable retatrutide from questionable product. These apply regardless of which vendor you are evaluating.
Physical appearance of lyophilized powder
High-quality lyophilized retatrutide should appear as a white to off-white powder or compact cake inside the vial. The powder should be dry and free of visible moisture. Discoloration, particularly yellowing or browning, can indicate degradation, contamination, or improper manufacturing. While appearance alone cannot confirm identity or purity, it serves as a basic initial screening. If the powder looks wrong before you even reconstitute it, something is wrong.
The seal on the vial should be intact and tamper-evident. Any sign that a vial has been opened, resealed, or compromised should lead to immediate rejection. Research peptides should arrive in sealed, labeled vials with clear batch identification.
Reconstitution behavior
When properly manufactured retatrutide is reconstituted with bacteriostatic water, it should dissolve completely within a few minutes of gentle swirling. The resulting solution should be clear and colorless to very slightly opalescent. Cloudiness, particulate matter, gel formation, or failure to dissolve completely are all warning signs. Researchers encountering these issues should not proceed with using the product.
Multiple customer reviews of Peptide Sciences products, across various peptides not limited to retatrutide, have reported issues with cloudy reconstitution and gel formation. While these reports do not definitively prove a quality problem with every vial, they are consistent with the pattern of inconsistency identified in Finnrick testing. The reconstitution calculator on SeekPeptides can help you determine the correct water volume, but if the product does not dissolve properly, no calculator can fix a manufacturing issue.
Storage and stability indicators
Retatrutide in lyophilized form should be stored at controlled temperatures, ideally between 2 and 8 degrees Celsius in a refrigerator. The vendor should ship with appropriate cold chain packaging, particularly during warm weather months. Peptides that arrive warm after extended transit may have already begun degrading. Researchers investigating peptide refrigeration requirements should note that retatrutide follows the same general cold storage principles as other peptide therapeutics.
Once reconstituted, the solution should remain stable for approximately 28 to 30 days when stored at 2 to 8 degrees Celsius. Similar principles apply to reconstituted tirzepatide fridge life and compounded semaglutide fridge storage. Freezing reconstituted peptide solutions can disrupt the three-dimensional structure, potentially reducing or eliminating biological activity. The same concerns apply to tirzepatide out of fridge scenarios and compounded semaglutide refrigeration requirements.
Batch-to-batch consistency
Perhaps the most important quality marker for any ongoing research program is consistency between batches. A researcher who orders retatrutide from the same vendor multiple times should receive product that performs identically each time. If research results vary dramatically between orders despite identical protocols, the most likely explanation is product inconsistency rather than biological variation.
Peptide Sciences 36-sample testing data showing scores ranging from 2.0 to 10.0 represents the definition of inconsistency. A researcher lucky enough to receive a 10.0 batch would have a very different experience than one who received a 2.0 batch. Neither researcher would know which batch they received without independent testing. This unpredictability is incompatible with reliable research design.
Reconstitution and storage after purchasing
Assuming a researcher has evaluated the vendor data and decided to proceed with a purchase, proper handling becomes the next critical factor. Even a perfect peptide product can be rendered useless by improper reconstitution or storage. This section covers the practical steps for handling retatrutide after it arrives.
Before you begin
Allow the vial to reach room temperature before opening. If the peptide was shipped cold or stored in a refrigerator, removing it and allowing 15 to 20 minutes of equilibration at room temperature prevents condensation from forming inside the vial and interfering with the reconstitution process. Researchers who have worked with reconstituting semaglutide or reconstituting tirzepatide will be familiar with this standard preparation step.
Gather all necessary supplies before beginning. You will need bacteriostatic water, sterile insulin syringes, alcohol wipes, and a clean work surface. The bacteriostatic water guide for retatrutide covers specific volume calculations. Similar principles apply to bac water for semaglutide and other peptide reconstitution preparations.
Step-by-step reconstitution
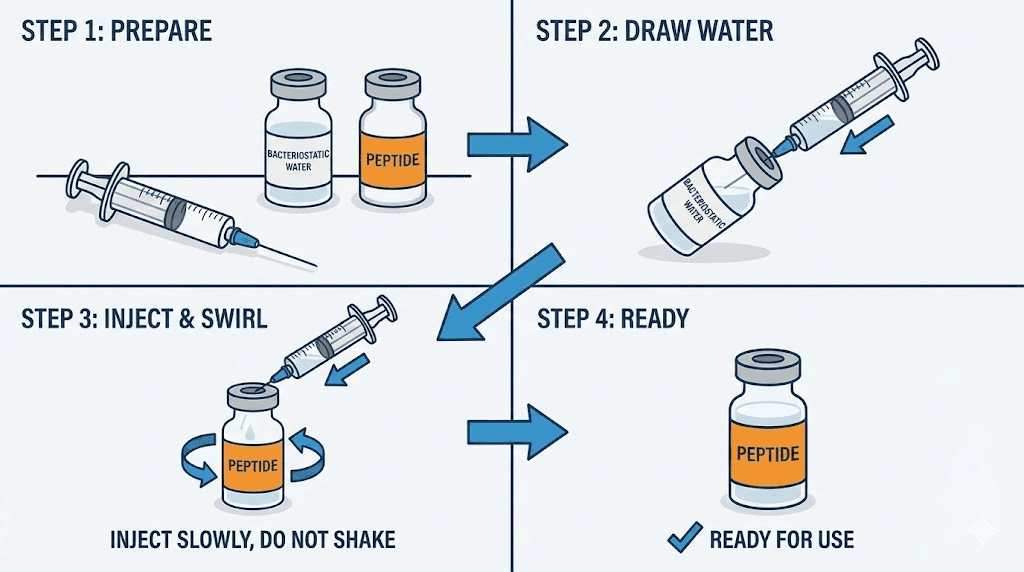
Clean the rubber stopper of both the peptide vial and the bacteriostatic water vial with an alcohol swab. Allow the alcohol to evaporate completely before proceeding. This takes approximately 30 seconds.
Draw the desired volume of bacteriostatic water into a sterile syringe. The volume depends on the concentration you want to achieve, which should be calculated in advance using a retatrutide dosage calculator. Common reconstitution volumes for a 10mg vial range from 1mL to 2mL, depending on the intended research concentration.
Insert the syringe needle through the rubber stopper of the peptide vial at an angle. Direct the stream of water down the inside wall of the vial, not directly onto the lyophilized powder. This prevents the peptide from foaming, which can damage the molecular structure. Release the water slowly and steadily.
Do not shake the vial. Shaking introduces air bubbles, creates foam, and can damage the peptide through mechanical stress. Instead, gently swirl the vial in a circular motion. Allow the water to gradually dissolve the powder. Most properly manufactured retatrutide will dissolve completely within 2 to 5 minutes of gentle swirling. If dissolution takes longer than 10 minutes, this may indicate a quality issue with the product.
Inspect the solution carefully. It should be clear and free of visible particles. Any cloudiness, gel formation, or undissolved material is a cause for concern.
Storage after reconstitution
Immediately refrigerate the reconstituted vial at 2 to 8 degrees Celsius. The bacteriostatic water preservative, benzyl alcohol, provides antimicrobial protection that allows the solution to remain usable for approximately 28 to 30 days when properly stored. Sterile water, which lacks this preservative, would only support stability for a few days.
Never freeze reconstituted retatrutide. Freezing can disrupt the peptide three-dimensional folding structure, potentially eliminating biological activity. The same principle applies to tirzepatide expiration and storage concerns, as well as expired semaglutide considerations. Temperature stability is essential for maintaining peptide integrity.
Minimize the number of times you puncture the rubber stopper. Each needle insertion creates a potential contamination pathway. Use the smallest gauge needle practical for your needs, and always swab the stopper with alcohol before each withdrawal. Researchers handling multiple peptides simultaneously should be careful not to cross-contaminate vials.
Injection site considerations for research
For researchers working with animal models or in vitro preparations, where to inject retatrutide depends on the specific research protocol. Subcutaneous administration is the route used in clinical trials. Proper injection technique, including where to inject GLP-1 agonists and how to inject GLP-1 compounds, follows standard subcutaneous protocols. Site rotation prevents tissue irritation or lipodystrophy at the injection location.
Safety considerations for research peptide sourcing
Research peptides exist in a regulatory grey area. They are sold labeled "for research use only" or "not for human consumption." This labeling allows vendors to operate without the oversight that pharmaceutical manufacturers face. The implications for researchers are significant and extend beyond simple product quality into questions of safety, legality, and risk management.
The grey market reality
The research peptide market operates without standardized dosing requirements, without prescription oversight, and without mandatory quality testing. Vendors set their own quality standards, perform their own testing, and report their own results. There is no regulatory body verifying that a vendor claims of 99% purity are accurate, or that the compound in the vial matches the label. This is the environment in which Peptide Sciences and all other research peptide vendors operate.
Some vendors invest heavily in quality control and third-party testing because they recognize that their reputation depends on delivering reliable product. Others cut corners because there is no enforcement mechanism to prevent it. The Finnrick testing data helps researchers distinguish between these two categories. But even the best testing platform can only test a sample of what a vendor produces. Individual vials that were not tested could still deviate from tested batches.
Contamination risks
Grey market peptide manufacturing processes can introduce several types of contamination. Heavy metals such as lead, arsenic, and mercury may be present as residual impurities from raw materials or synthesis equipment. Chemical solvents used in peptide synthesis, including trifluoroacetic acid and acetonitrile, can remain in the final product if purification is inadequate. Bacterial contamination, particularly endotoxins, can occur if manufacturing environments are not properly controlled. For compounds being studied in biological systems, these contaminants introduce uncontrolled variables that can confound results or cause harm.
The risk profile becomes especially concerning with novel compounds like retatrutide where manufacturing experience across the supplier network is limited. Established peptides like compounded semaglutide have been manufactured by hundreds of laboratories over many years, allowing processes to be refined. Retatrutide manufacturing expertise is still developing, and not all vendors have achieved reliable production at scale.
Identity misrepresentation as a safety concern
The Finnrick finding that Peptide Sciences retatrutide samples sometimes contained tirzepatide instead of retatrutide is not just a quality issue. It is a safety concern. Tirzepatide and retatrutide have different receptor binding profiles, different potency at shared targets, and different pharmacological effects. A researcher who believes they are administering retatrutide but is actually administering tirzepatide would be working with incorrect assumptions about mechanism, potency, and expected effects.
The glucagon receptor component absent from tirzepatide but present in retatrutide drives specific metabolic effects including increased thermogenesis and hepatic glucose modulation. These effects would be missing from a misidentified product. Any research conclusions drawn from such data would be fundamentally flawed. This is why identity verification through mass spectrometry is the most important quality test for any research peptide, and why researchers should not rely solely on vendor-provided COAs.
The importance of independent testing
Every researcher working with grey market peptides should consider independent testing as part of their quality assurance process. Organizations like Finnrick provide a service that partially addresses this need by testing vendor samples and publishing results. But researchers with access to analytical laboratory equipment can also verify their own purchases before using them.
At minimum, researchers should check that the molecular weight matches the expected value for the purchased compound. HPLC purity analysis can be performed at most research institutions with basic analytical chemistry facilities. These steps add time and cost to the research process, but they protect against the far greater cost of generating unreliable data from misidentified or degraded compounds. Researchers studying research vs pharmaceutical peptides should understand that this self-verification step is one of the key differences between the two categories.
Comparing grey market to pharmaceutical-grade compounds
The retatrutide used in Eli Lilly clinical trials was manufactured under Good Manufacturing Practice conditions with rigorous quality control at every step. Raw materials were verified, synthesis was controlled, purification met established specifications, and every batch was tested by qualified quality assurance personnel before release. The safety and efficacy data from those trials applies specifically to that pharmaceutical-grade product.
Grey market retatrutide, even from the best vendors, is manufactured under different conditions with different oversight. Researchers should keep this context in mind when interpreting clinical trial data alongside their own observations. The differences between lyophilized vs liquid peptides and between injectable vs oral peptides are additional variables that affect outcomes beyond the basic question of compound identity and purity.
Making an informed vendor decision
The data presented in this guide points to clear conclusions about Peptide Sciences retatrutide quality. But the broader lesson extends beyond a single vendor. Researchers need a systematic framework for evaluating any peptide supplier, and that framework should prioritize evidence over assumptions.
A decision framework for vendor selection
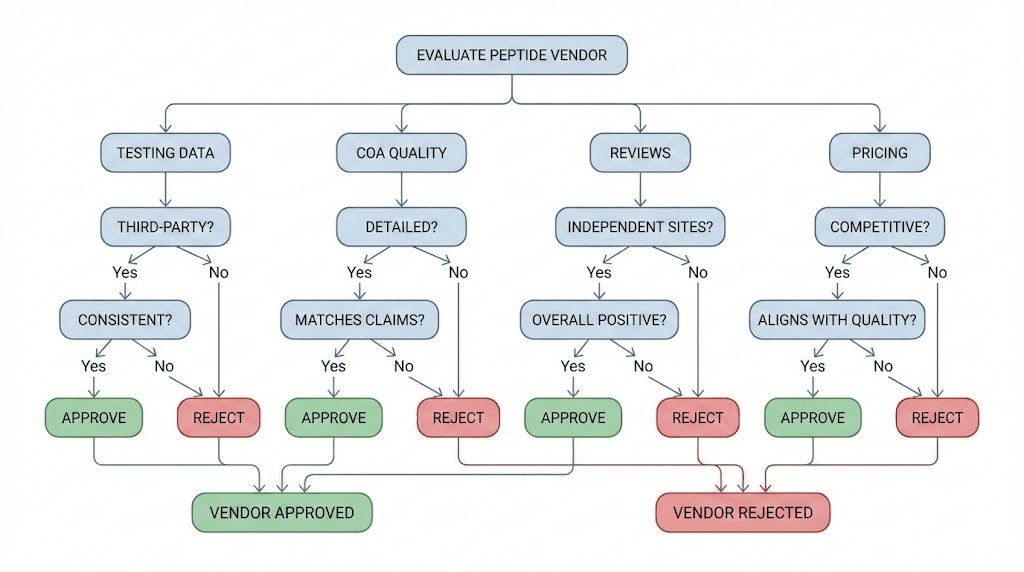
Start with independent testing data. Before considering any other factor, check whether the vendor has been tested by Finnrick or similar independent platforms. If testing data exists, examine the identity confirmation rate, the quantity accuracy, the purity consistency, and the overall rating. This is the most objective information available and should carry the most weight in your decision.
Second, evaluate the COA quality. Request a COA before purchasing. Look for lot-specific testing, chromatograms and spectra (not just numbers), molecular weight confirmation, and ideally evidence of third-party testing. A vendor that provides comprehensive, verifiable COAs is demonstrating a commitment to transparency. A vendor that provides minimal or generic-looking COAs is not.
Third, assess customer feedback patterns. Individual reviews can be biased in either direction. But patterns across many reviews are informative. If multiple independent reviewers report the same issue, such as cloudy reconstitution, inconsistent results, or unresponsive customer support, these patterns likely reflect genuine experiences. Reviews for Peptide Sciences show exactly this type of pattern clustering.
Fourth, consider the vendor refund and replacement policy. A vendor confident in their product quality should offer reasonable recourse for defective products. Peptide Sciences does not offer refunds and has very limited replacement policies. This shifts all risk to the researcher. Compare this with vendors who stand behind their products with more comprehensive quality guarantees.
Fifth, evaluate pricing in context. Price should be the last factor considered, not the first. A lower price from a vendor with A-rated independent testing results is objectively a better value than a higher price from a vendor with E-rated results. The peptide cost calculator can help you compare per-milligram costs across vendors, but remember that cost per milligram only matters if the milligrams actually contain the correct compound.
When to switch vendors
If your current vendor has poor independent testing ratings, it is time to switch. If your research results have been inconsistent despite controlling all other variables, your peptide source may be the uncontrolled factor. If your vendor COAs lack supporting analytical data, you do not have enough information to trust their quality claims. If your vendor does not offer reasonable recourse for quality issues, they are not investing in your success as a researcher.
Switching vendors does introduce a transition period where you should overlap testing to compare results. Order from both your current vendor and the new one. Test both products independently if possible. Compare reconstitution behavior, physical appearance, and research outcomes. This controlled transition protects against the possibility that even a well-rated vendor might have an off batch.
Thinking about the broader peptide research landscape
Retatrutide is one compound in a rapidly expanding category of metabolic research peptides. The same vendor evaluation principles apply to tirzepatide dosing research, semaglutide dosage studies, and emerging compounds like cagrilintide and orforglipron. The pipeline of next-generation metabolic peptides continues to grow, with mazdutide vs tirzepatide comparisons and other head-to-head analyses becoming increasingly relevant.
Researchers who establish good vendor evaluation habits now will be well-positioned as new compounds enter the research market. The skills of COA interpretation, independent testing data evaluation, and systematic vendor comparison do not become obsolete. They become more valuable as the peptide research space continues to expand and diversify.
SeekPeptides members access detailed vendor comparison tools, protocol databases, and expert-reviewed sourcing guides that help navigate these decisions with confidence. The platform provides continuously updated information as new testing data becomes available, ensuring researchers always have access to the latest vendor quality assessments.
Retatrutide in context: the metabolic peptide landscape
Understanding Peptide Sciences retatrutide offering requires placing it within the broader context of metabolic peptide research. Retatrutide does not exist in isolation. It represents one point on a spectrum of incretin-based compounds, each with distinct mechanisms, clinical data, and sourcing considerations.
How retatrutide compares to dual agonists
The comparison between retatrutide vs semaglutide highlights the fundamental difference between single and triple receptor targeting. Semaglutide activates only GLP-1 receptors. Clinical data shows approximately 15-17% weight reduction at optimal doses over similar timeframes. Retatrutide Phase 2 data showed 24.2% at 12mg over 48 weeks. The additional 7-9 percentage points of weight reduction likely stem from the GIP and glucagon receptor contributions.
Researchers studying how fast semaglutide works and comparing it to retatrutide onset will find different timelines for each compound. The triple mechanism of retatrutide creates a different pharmacodynamic profile, with appetite suppression, insulin sensitization, and energy expenditure acceleration occurring simultaneously. Single agonists like semaglutide primarily drive appetite suppression, with metabolic effects arising more indirectly through weight loss itself. For researchers considering the semaglutide to tirzepatide conversion pathway, understanding the differences in mechanism and potency is essential before considering a transition to a triple agonist.
The tirzepatide vs semaglutide dosage comparison shows that dual agonists already outperform single agonists. Tirzepatide Phase 3 data showed approximately 20-22% weight reduction at the highest dose, positioning it between semaglutide and retatrutide. The mazdutide vs retatrutide comparison represents the next frontier, as mazdutide also targets glucagon receptors but with a different receptor potency balance.
Side effect profiles across the spectrum
Gastrointestinal side effects tend to intensify as more receptors are targeted. Researchers monitoring semaglutide constipation or tirzepatide constipation should expect similar or more pronounced gastrointestinal effects with retatrutide. The semaglutide fatigue that some researchers observe may also manifest differently with triple agonist compounds, as the glucagon receptor component can affect energy levels through different pathways.
The relationship between dosing protocols and side effects is well-documented across the incretin class. Starting at lower doses and gradually titrating upward helps manage gastrointestinal tolerance. This approach is reflected in the retatrutide dose schedule used in clinical trials, as well as in microdosing tirzepatide strategies and semaglutide dosage in units protocols. The general principle of starting low and adjusting based on tolerance applies across the entire compound class.
Hair loss concerns have emerged across GLP-1 agonist research. The question of whether retatrutide causes hair loss mirrors similar investigations into GLP-1 hair loss more broadly. The mechanism appears related to rapid weight loss rather than direct peptide effects, but this remains an active area of investigation. Researchers tracking these side effects should ensure they are working with verified compounds, as attributing side effects to a compound you have not verified is scientifically meaningless.
Dietary and lifestyle interactions
Retatrutide research occurs alongside dietary and lifestyle variables that significantly affect outcomes. The literature on tirzepatide foods to avoid and what to eat on tirzepatide provides relevant context for retatrutide research as well, given the shared GLP-1 and GIP mechanisms. Dietary planning resources like tirzepatide diet plans, semaglutide diet plans, and tirzepatide meal plans offer frameworks that researchers studying retatrutide may find relevant for controlling nutritional variables.
The question of alcohol interaction, explored in guides on drinking on tirzepatide and drinking on semaglutide, extends to retatrutide as well. Alcohol affects gastric emptying, hepatic metabolism, and caloric intake, all of which interact with the pathways that retatrutide modulates. Controlling for alcohol consumption in research protocols improves data quality.
Supplementation strategies, including supplements with tirzepatide, tirzepatide niacinamide, tirzepatide glycine, and semaglutide with B12, reflect the broader research community effort to optimize outcomes when using incretin-based compounds. The role of semaglutide methylcobalamin combinations and semaglutide with glycine protocols continues to evolve as researchers gather more data.
Compound formulation variations
The peptide research landscape includes multiple formulation approaches. The comparison between tirzepatide oral vs injection and the development of oral tirzepatide reflects industry efforts to improve administration convenience. Similarly, tirzepatide drops and oral semaglutide drops represent alternative delivery methods. The Onmorlo GLP-1 patches offer yet another route of administration. For retatrutide, subcutaneous injection remains the standard research administration route, consistent with the clinical trial protocol.
Compounding approaches add another dimension. Resources on tirzepatide compound dosage charts, compounded tirzepatide calculators, and tirzepatide glycine B12 compound guides demonstrate the complexity of working with compounded formulations. Understanding the tirzepatide tablets vs injections comparison helps researchers evaluate different formulation approaches within the broader research context. The same analytical rigor that reveals problems with Peptide Sciences retatrutide should be applied to any compounded formulation.
What happens when researchers do not verify vendor quality
The consequences of using unverified peptide sources extend beyond wasted money. They cascade through the entire research process and can affect outcomes in ways that are not immediately obvious.
Wasted research time and resources
Consider a researcher who designs a dose-response study using retatrutide from Peptide Sciences. They carefully calculate doses using the retatrutide dosage calculator, follow proper reconstitution protocol using the bac water for retatrutide guide, and maintain rigorous experimental controls. If the vial they received contained tirzepatide instead of retatrutide, every data point from that study is compromised. The dose-response curve they generate reflects tirzepatide pharmacology, not retatrutide pharmacology. But the researcher attributes it to retatrutide because that is what the label said.
Weeks or months of research time are wasted. Resources consumed in the study are not recoverable. And if the researcher publishes or reports these findings, the contamination of the scientific record extends beyond their own laboratory. This is not a hypothetical scenario. The Finnrick identity failures documented for Peptide Sciences retatrutide mean this has actually happened to real researchers.
Incorrect dose-response conclusions
Quantity discrepancies create a subtler but equally damaging problem. If a 12mg vial actually contains 9.36mg (the 22% shortfall documented by Finnrick), every dosing calculation is wrong. The researcher administers less compound than they believe. If the study is comparing different dose levels, the entire dose-response relationship is shifted. Conclusions about minimum effective doses, maximum tolerated doses, or optimal dose ranges would all be incorrect.
Similarly, the 45.8% overfill in a 6mg vial means the researcher administers significantly more than intended. In a side effect tolerability study, this could lead to concluding that lower doses produce more side effects than expected, when in reality the actual dose was much higher than the nominal label. These errors propagate through research conclusions and can mislead future studies that rely on the published data.
The cost of switching later
Some researchers continue using a vendor they suspect may be unreliable because switching feels disruptive. They have historical data generated using that vendor product, and switching introduces a confounding variable into longitudinal studies. This is a real concern but it leads to a problematic conclusion. Continuing to use an unreliable source to maintain consistency with previous unreliable data does not make the data more reliable. It compounds the problem.
The better approach is to acknowledge the limitation, switch to a verified source, and clearly document the vendor change in all records. Future analysis can then control for the vendor variable. This is preferable to indefinitely generating questionable data from a source with documented quality failures. When making this transition, researchers may want to review tirzepatide not working discussions, not losing weight on semaglutide analyses, and 4 weeks semaglutide no weight loss case studies, as some of these outcome failures may actually reflect compound quality issues rather than biological non-response.
Advanced considerations for retatrutide research
For researchers who have addressed the sourcing question and are working with verified retatrutide, several advanced topics merit attention.
Dose titration strategies
Clinical trial protocols for retatrutide used gradual dose escalation to manage gastrointestinal tolerance. The retatrutide dose schedule from the Phase 2 trial started at lower doses and escalated over several weeks. This approach mirrors what is used in tirzepatide dose protocols and semaglutide dosage charts. The rationale is straightforward. GLP-1 receptor agonism causes nausea and other gastrointestinal effects that diminish as the body adapts. Starting at a lower dose allows this adaptation to occur before increasing to a therapeutic level.
For retatrutide specifically, the glucagon receptor component may introduce additional considerations for titration. Glucagon receptor activation affects hepatic glucose output and energy expenditure in ways that may require monitoring beyond what single or dual agonists demand. Understanding how much retatrutide to administer at each stage of titration requires balancing efficacy goals against tolerability constraints.
Transition protocols between compounds
Researchers transitioning from other incretin mimetics to retatrutide face unique challenges. The question of retatrutide after tirzepatide involves considerations of receptor cross-tolerance, wash-out periods, and dose equivalency. There is no standardized conversion chart for retatrutide the way there is for semaglutide to tirzepatide conversion. The addition of glucagon receptor agonism means that even matched GLP-1 and GIP activity would produce different metabolic effects.
For researchers who have been working with switching tirzepatide to semaglutide or vice versa, the transition to retatrutide adds another layer of complexity. The pharmacokinetics differ, the receptor engagement profile differs, and the downstream metabolic effects differ. Each transition should be treated as a new experimental condition, not simply a continuation of previous protocols.
Monitoring outcomes
Comprehensive outcome monitoring for retatrutide research should extend beyond weight measurement. Given the compound effects on hepatic metabolism, lipid profiles, inflammatory markers, and insulin sensitivity, researchers should consider monitoring a broad panel of metabolic indicators. The tirzepatide before and after documentation approach, expanded to include metabolic panels, provides a useful template. Similar documentation methods are used in tirzepatide weight loss results tracking and men before after tirzepatide case studies.
Specific parameters worth tracking include fasting glucose, fasting insulin, HOMA-IR for insulin resistance, triglycerides, LDL and HDL cholesterol, high-sensitivity C-reactive protein, liver enzymes including ALT and AST, and body composition measurements beyond simple body weight. These markers correspond to the endpoints measured in clinical trials and allow researchers to compare their observations against published Phase 2 and Phase 3 data.
Understanding non-response
Not every subject responds identically to retatrutide, even in clinical trials with pharmaceutical-grade product. But in the grey market context, apparent non-response may have a much simpler explanation: the product is not what it claims to be. Before concluding that retatrutide is ineffective for a particular application, researchers must first rule out product quality as a variable. This means verifying identity, purity, and quantity through independent testing before attributing outcomes, positive or negative, to the compound pharmacology.
The parallel to experiences documented in semaglutide appetite immediately discussions and semaglutide energy observations is instructive. Many reports of variable response in the research community may reflect variable product quality rather than variable biological response. Without product verification, the two cannot be distinguished.
The broader question of peptide research integrity
This review of Peptide Sciences retatrutide quality illuminates a broader challenge facing the research peptide community. The gap between vendor marketing claims and independently verified quality is, in many cases, substantial. This gap affects individual researchers, but it also affects the collective body of knowledge generated by the peptide research community.
When researchers publish findings based on unverified compounds, those findings enter the scientific literature with an unacknowledged uncertainty. Other researchers may build on those findings, creating a chain of work that rests on an unstable foundation. The compound quality variable is rarely discussed in research publications, yet it may explain more inconsistencies in the literature than any other single factor.
Efforts by organizations like Finnrick to create transparent quality databases represent an important step toward addressing this problem. SeekPeptides provides additional resources for researchers navigating the complex landscape of peptide sourcing, including vendor analyses, quality comparison tools, and protocol guides that account for compound variability. These resources help researchers make decisions based on data rather than marketing, and they contribute to a research environment where product quality is treated as the controlled variable it needs to be.
The future of metabolic peptide research depends on addressing the quality gap. As compounds like retatrutide move toward potential FDA approval and as the retatrutide availability landscape evolves, the distinction between pharmaceutical-grade and grey market products will become even more important. Researchers who establish rigorous vendor evaluation practices now are investing in the reliability of their future work. Those who do not are building on uncertain ground.
For researchers serious about optimizing their peptide protocols and making evidence-based sourcing decisions, SeekPeptides offers the most comprehensive resource available, with vendor quality databases, expert-reviewed protocols, and a community of experienced researchers who have navigated these exact challenges.
Frequently asked questions
Is Peptide Sciences retatrutide legitimate?
Peptide Sciences is a real company that sells research peptides, including retatrutide. However, independent testing by Finnrick Analytics gave their retatrutide an E rating (Bad) based on 36 tested samples. Multiple samples were found to contain tirzepatide or unidentified compounds instead of retatrutide. While the company provides internal COAs, the independent data raises serious quality concerns that researchers should consider before purchasing. Always compare vendor testing data through independent platforms before making a retatrutide peptide purchase decision.
What did Finnrick testing find in Peptide Sciences retatrutide?
Finnrick testing of 36 Peptide Sciences retatrutide samples revealed an average score of 6.1 out of 10, with scores ranging from 2.0 to 10.0. Critical findings included identity failures where samples labeled as retatrutide actually contained tirzepatide or unidentified compounds. Quantity discrepancies were also documented, including a 6mg vial containing 8.75mg (+45.8%) and a 12mg vial containing only 9.36mg (-22.0%). Purity ranged from 98.83% to 100%, but high purity of the wrong compound does not help a researcher studying the retatrutide dose response.
How does Peptide Sciences retatrutide compare to other vendors?
Peptide Sciences received the lowest possible Finnrick rating (E for Bad) for retatrutide. By comparison, vendors like Paradigm Peptide, Orbitrex, and Peptide Partners received A ratings (Great). The difference in independent testing performance is dramatic. Researchers comparing the Paradigm Peptides retatrutide profile against Peptide Sciences will see why vendor selection matters critically for research reliability.
Is retatrutide the same as semaglutide or tirzepatide?
No. Retatrutide is a triple receptor agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. Semaglutide targets only GLP-1 receptors. Tirzepatide targets GLP-1 and GIP receptors. The addition of glucagon receptor agonism in retatrutide drives increased energy expenditure and thermogenesis, contributing to the higher weight reduction percentages seen in clinical trials. Understanding the retatrutide vs semaglutide differences is essential for researchers selecting compounds for specific study designs. The semaglutide vs tirzepatide comparison page provides additional context on how these compounds differ at the receptor level.
How should I reconstitute retatrutide after purchasing?
Add bacteriostatic water to the lyophilized powder by directing the stream down the inside wall of the vial. Use the bac water for retatrutide guide to determine the correct volume. Gently swirl, never shake, the vial until the powder dissolves completely. The solution should be clear and particle-free. Store at 2 to 8 degrees Celsius and use within 28 to 30 days. The reconstitution calculator provides exact volume calculations based on your desired concentration.
What clinical trial results exist for retatrutide?
Phase 2 trials published in the New England Journal of Medicine showed 24.2% body weight reduction at the 12mg dose over 48 weeks. Phase 3 TRIUMPH-4 results showed an average weight loss of 71.2 pounds (28.7% body weight) at 12mg over 68 weeks. Additionally, retatrutide resolved liver steatosis in over 85% of subjects with MASLD at higher doses. These results represent pharmaceutical-grade retatrutide manufactured under GMP conditions, which is not equivalent to grey market research products. Review the complete retatrutide dosage chart for detailed dose-response data.
Should I switch from Peptide Sciences to another retatrutide vendor?
Based on independent testing data, switching to a higher-rated vendor would likely improve research reliability. Vendors with Finnrick A ratings demonstrate consistent identity confirmation, accurate quantities, and high purity across multiple tests. The retatrutide buy guide on SeekPeptides provides a comprehensive vendor evaluation framework. When switching, consider ordering from both vendors simultaneously to compare product quality during the transition.
Can I trust Peptide Sciences Certificate of Analysis?
Peptide Sciences provides internal COAs with their products. These documents reflect the company own testing, not independent third-party verification. Given that independent Finnrick testing found significant identity failures and quantity discrepancies that were not reflected in company COAs, researchers should treat these documents as vendor claims rather than objective quality verification. Always cross-reference vendor COAs with independent testing data when available. The research vs pharmaceutical peptides guide explains why this distinction matters.
External resources
Finnrick Analytics - Peptide Sciences Retatrutide Testing Data
Obesity Action Coalition - Grey Market GLP-1 Product Safety Statement
In case I do not see you, good afternoon, good evening, and good night. May your vendors stay verified, your peptides stay pure, and your research stay reliable.