Feb 13, 2026

That vial sitting in your refrigerator has a clock ticking. You might not hear it. You might not even think about it. But every day that passes, the tirzepatide inside is one step closer to losing the very thing that makes it work.
Most people never check expiration dates on their peptide medications until something feels off. The weight loss stalls. The appetite suppression weakens. The results that came so reliably in the first few weeks suddenly vanish. And they blame their body, their diet, their discipline, when the real culprit has been sitting on the second shelf of their refrigerator the entire time.
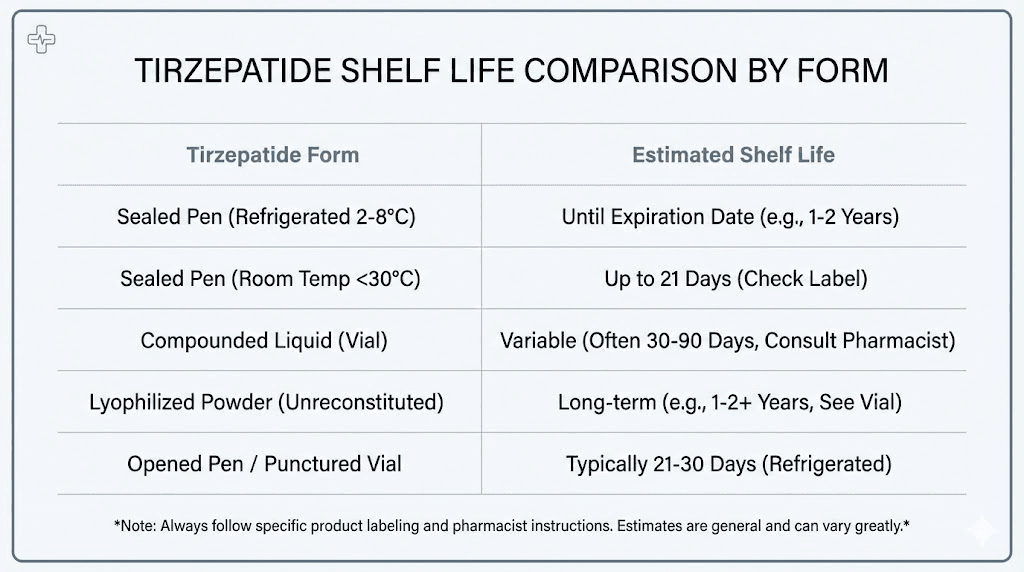
Tirzepatide absolutely expires. Every formulation, whether it is brand-name Mounjaro, Zepbound, or compounded versions, has a defined shelf life that depends on its form, how it has been stored, and whether it has been opened. The timelines range from as little as 21 days to as long as 24 months, and confusing one for the other can mean the difference between effective treatment and wasted medication.
This guide covers every scenario. Sealed vials. Opened vials. Reconstituted solutions. Lyophilized powder. Compounded formulations. Brand-name pens. You will know exactly how long each form lasts, how to tell when tirzepatide has gone bad, and what actually happens at the molecular level when it expires. Because guessing with peptide medications is not a strategy. It is a waste of money, time, and progress.

The short answer: yes, tirzepatide expires
Every tirzepatide product has an expiration date. This is not optional labeling. It is not a conservative estimate designed to make you buy more medication. The date printed on your vial, pen, or compounded preparation represents the last day the manufacturer or pharmacy can guarantee full potency and safety under proper storage conditions.
For brand-name products like Mounjaro and Zepbound, the manufacturer expiration date is typically 24 months from the date of manufacture when stored continuously at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). That is the maximum. The moment storage conditions deviate, that timeline shrinks dramatically.
Compounded tirzepatide operates under completely different rules. The beyond-use date assigned by a compounding pharmacy is typically 28 to 90 days from the date of preparation, depending on the formulation type and the pharmacy's own stability testing. This is not the same as a manufacturer expiration date. It is shorter, more conservative, and varies from pharmacy to pharmacy.
Understanding the distinction between these two timelines is critical. One represents pharmaceutical-grade stability testing backed by years of data. The other represents best estimates under USP guidelines. Neither should be ignored.
How long does tirzepatide last in different forms
The shelf life of tirzepatide depends entirely on what form it is in and how it has been handled. A sealed pen sitting in proper refrigeration is a completely different situation from a reconstituted vial that has been punctured three times and left on a bathroom counter.
Here is the breakdown.
Brand-name pens (Mounjaro, Zepbound): sealed and refrigerated
Unopened brand-name tirzepatide pens have the longest shelf life of any form. When stored at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit), they remain potent until the printed expiration date, which is usually about 24 months from manufacture.
Keep them in the original carton. Light degrades peptides. The box is not just packaging. It is protection.
These pens contain no antimicrobial preservatives, which is why proper storage matters so much. The sterile environment inside the pen is maintained by the sealed delivery system, not by chemical preservatives fighting off contamination.
Brand-name pens: room temperature
Life happens. Maybe your refrigerator broke. Maybe you are traveling. Eli Lilly, the manufacturer, has tested tirzepatide at room temperature and published clear guidelines.
Mounjaro and Zepbound can stay at room temperature for up to 21 days, as long as the temperature never exceeds 86 degrees Fahrenheit (30 degrees Celsius). After 21 days at room temperature, discard the pen even if the expiration date has not passed.
This is not a suggestion. This is the manufacturer telling you that beyond 21 days outside refrigeration, they cannot guarantee what you are injecting will work as intended.
One important detail that catches people off guard: you cannot re-refrigerate tirzepatide after it has been stored at room temperature. The temperature cycling from cold to warm to cold again creates stress on the peptide structure. Once it leaves the fridge and stays out, it is on a 21-day countdown with no reset button.
Compounded tirzepatide: liquid formulations
Compounded tirzepatide comes in several forms, and the most common is a ready-to-inject liquid solution. The shelf life here depends on the compounding pharmacy, the specific formulation, whether it contains preservatives, and the USP chapter under which it was prepared.
Most compounding pharmacies assign a beyond-use date of 28 to 90 days from preparation when stored at 2 to 8 degrees Celsius. Some pharmacies with proprietary stability data may extend this window, but 28 to 30 days is the most common assignment for preservative-free formulations.
The beyond-use date is not the same as a manufacturer expiration date. It is governed by USP Chapter 797 standards for sterile compounding, and it factors in both chemical stability and sterility risk. Even if the tirzepatide molecule itself might remain chemically active for longer, the sterility of the preparation becomes questionable after the assigned date.
Always follow the specific date printed on your compounded vial. Different pharmacies have different data, different formulations, and different testing. What is safe from one pharmacy is not automatically safe from another.
Lyophilized (freeze-dried) tirzepatide powder
Lyophilized tirzepatide is the most stable form. Without water present, the chemical reactions that degrade peptides, primarily hydrolysis, essentially stop. Properly stored lyophilized powder can last 12 months or longer at refrigerator temperatures, and some peptide manufacturers report stability beyond that.
The key word is "properly stored." That means:
Sealed, airtight vial
Refrigerated at 2 to 8 degrees Celsius
Protected from light
Never exposed to moisture
The moment you reconstitute lyophilized tirzepatide with bacteriostatic water or sterile water, the clock resets dramatically. Reconstituted solutions typically last 28 to 30 days refrigerated, or up to 90 days if the solution contains bacteriostatic water (which has a mild preservative effect from the benzyl alcohol).
This difference between powder and solution shelf life is massive. A vial of lyophilized tirzepatide sitting in your fridge might be good for a year. The moment you add water, you have roughly a month.
Opened and punctured vials
Once you puncture the rubber stopper of any tirzepatide vial, the sterility clock starts ticking regardless of the printed expiration date. Industry standard guidance gives 21 to 28 days for multi-dose vials after first puncture, even with preservatives present.
Each needle insertion introduces a microscopic risk of contamination. Good technique minimizes this, but does not eliminate it. The more times you puncture a vial, the higher the cumulative risk.
What actually happens when tirzepatide expires
Expiration is not a light switch. Tirzepatide does not go from 100 percent potent to zero overnight. It is a gradual process, and understanding what happens at the molecular level helps explain why expired medication behaves differently than fresh medication.
Peptide degradation pathways
Tirzepatide is a 39-amino-acid peptide with specific structural modifications that make it effective, including alpha-aminoisobutyric acid (Aib) substitutions at positions 2 and 13 (which protect against DPP-4 enzyme degradation in the body) and a C20 fatty diacid chain at position 20 (which enables albumin binding for a longer half-life).
When tirzepatide degrades outside the body, several chemical processes are at work.
Hydrolysis is the primary pathway. Water molecules attack peptide bonds, breaking the 39-amino-acid chain into shorter, inactive fragments. This happens faster at higher temperatures and at extreme pH levels. It is also why lyophilized powder (no water present) lasts so much longer than reconstituted solutions.
Deamidation occurs when asparagine and glutamine residues in the peptide chain lose their amide groups. This subtly changes the peptide structure in ways that reduce receptor binding affinity. Your body recognizes the deamidated peptide differently, and its ability to activate GIP and GLP-1 receptors diminishes.
Oxidation targets vulnerable amino acids, particularly methionine residues. Light exposure accelerates this pathway significantly. The resulting oxidized peptide has altered folding and reduced biological activity.
Aggregation happens when degraded peptide fragments stick together, forming larger clumps. This is what you see when a once-clear solution turns cloudy or develops visible particles. Aggregated peptides are not just ineffective. They can trigger immune responses.
Potency loss over time
The practical effect of all these degradation pathways is simple. Less active tirzepatide in every injection.
A vial that started at full potency might be at 90 percent after moderate degradation. Then 75 percent. Then 60 percent. At some point, the amount of active tirzepatide per injection drops below the therapeutic threshold, and results stop.
This is particularly problematic with tirzepatide dosing because the therapeutic window matters. The difference between 2.5mg and 5mg of tirzepatide produces meaningfully different outcomes. If degradation has reduced your effective dose by 30 or 40 percent, you are no longer on the protocol you think you are on.
The tricky part is that mild degradation produces no visible changes. The solution looks clear. It does not smell different. The only indication is that results have slowed or stopped, and by then you have been injecting subtherapeutic doses for weeks.
Safety concerns with expired tirzepatide
The good news, relatively speaking, is that tirzepatide degradation typically results in loss of activity rather than formation of dangerous toxic compounds. Degraded tirzepatide generally becomes less effective rather than harmful.
But that is not the whole picture.
Three legitimate safety concerns exist with expired tirzepatide.
First, reduced glycemic control. For people using tirzepatide for type 2 diabetes management, subtherapeutic doses mean inadequate blood sugar control. This is not a minor inconvenience. Poorly controlled blood glucose increases the risk of cardiovascular disease, nephropathy, retinopathy, and neuropathy. Using expired medication thinking it is still working can mask a developing problem.
Second, contamination risk. Expired medications, especially opened vials without preservatives, have had more time for potential microbial growth. The longer a vial sits, the higher the contamination risk, even in a refrigerator. Injecting contaminated solutions can cause infections ranging from mild injection site reactions to serious systemic infections.
Third, aggregated peptides and immunogenicity. When peptides aggregate, they can trigger immune responses. Aggregated tirzepatide looks different to your immune system than properly folded tirzepatide. This can potentially lead to the development of anti-drug antibodies, which could reduce the effectiveness of future tirzepatide treatments even with fresh medication.
The FDA does not recommend using any medication past its expiration date, and no regulatory body has published safety data on using expired tirzepatide.
Storage rules that determine how long your tirzepatide actually lasts
The printed expiration date only holds true under specific conditions. Deviate from those conditions, and the effective shelf life shrinks. Sometimes dramatically.
Temperature: the single biggest factor
Tirzepatide is a temperature-sensitive peptide. The recommended storage range is 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius) for long-term storage. This is standard refrigerator temperature, not the door shelf where temperature fluctuates every time you open the fridge.
Here is what temperature deviations do.
Room temperature (below 86 degrees Fahrenheit / 30 degrees Celsius): Tirzepatide remains stable for up to 21 days. After that, degradation accelerates to the point where potency cannot be guaranteed. This applies to brand-name products. Compounded formulations may have shorter room-temperature windows.
Above 86 degrees Fahrenheit (30 degrees Celsius): Tirzepatide essentially expires immediately when exposed to high heat, regardless of what the printed date says. The peptide structure degrades rapidly at elevated temperatures. A vial left in a hot car for an afternoon may look identical to a fresh vial, but the tirzepatide inside has suffered irreversible damage.
Freezing (below 32 degrees Fahrenheit / 0 degrees Celsius): Freezing permanently destroys tirzepatide. The ice crystal formation physically damages the peptide structure, and this damage does not reverse when the solution thaws. If your tirzepatide freezes, even briefly, discard it. Do not attempt to thaw and use it. The manufacturer is explicit about this: frozen tirzepatide must be discarded even after thawing.
Temperature cycling, going from cold to warm to cold again, is also damaging. Each temperature transition stresses the peptide structure. This is why the re-refrigeration restriction exists for brand-name products. Once removed from the fridge, the 21-day room-temperature clock starts and cannot be paused.
Light exposure: the silent degrader
Ultraviolet and visible light trigger photodegradation of peptides through oxidative pathways. This is why tirzepatide comes in opaque packaging or amber vials. It is why the manufacturer tells you to keep pens in the original carton.
A vial sitting on a windowsill or under fluorescent lights degrades faster than an identical vial stored in darkness. The degradation is invisible. No color change. No cloudiness. Just steadily declining potency.
Simple fix: keep tirzepatide in its original packaging until you need it, and store it in a dark area of your refrigerator.
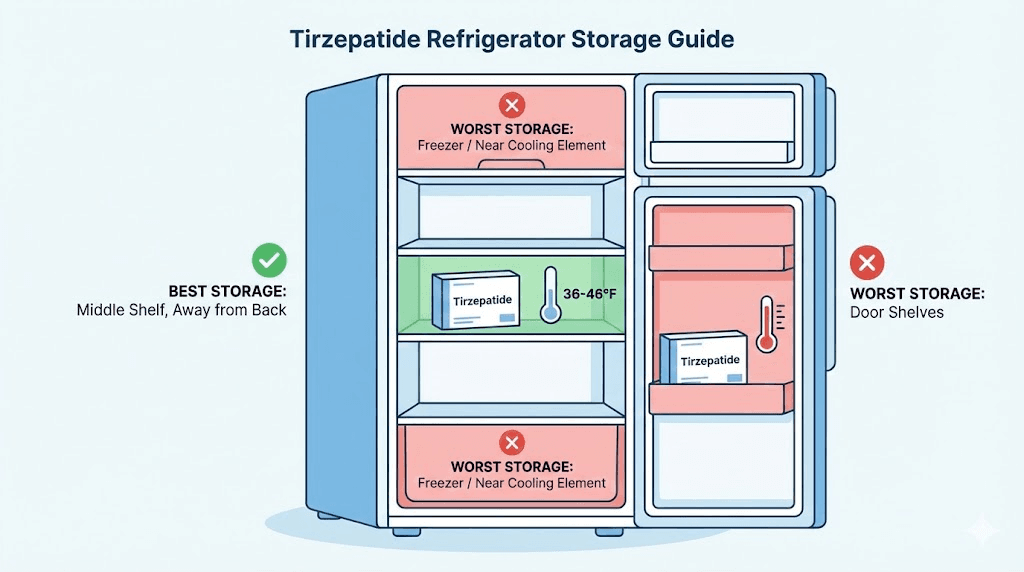
Proper refrigerator placement
Not all spots in your refrigerator are equal for peptide storage.
Best location: Middle shelf, toward the back. This area maintains the most consistent temperature with the least fluctuation from door opening and closing.
Worst locations: The door (temperature swings every time you open the fridge), the back wall of the fridge (risk of freezing if too close to the cooling element), and the crisper drawer (humidity concerns).
Some researchers invest in a small, dedicated medication refrigerator with a built-in thermometer. This eliminates the temperature fluctuations that come from a household refrigerator being opened multiple times daily. It also removes the risk of accidentally pushing a vial against the back wall where it might freeze.
If you use your household refrigerator, place a small thermometer near your medication to monitor actual temperatures. The thermostat setting and the actual internal temperature are not always the same.
How to tell if your tirzepatide has gone bad
Some signs of degradation are visible. Others are not. Knowing what to look for, and knowing the limitations of visual inspection, is essential for anyone using tirzepatide.
Visual indicators of degradation
Cloudiness. Fresh tirzepatide solution should be clear and colorless to slightly yellow. If the solution appears cloudy, hazy, or turbid, peptide aggregation has occurred. Do not use it.
Particles. Any visible particles, flakes, or sediment in the solution indicate significant degradation. These particles are aggregated peptide fragments and potentially other breakdown products. Discard immediately.
Color changes. A dramatic shift in color, particularly darkening or the appearance of a brownish tint, indicates oxidation. Compounded tirzepatide with B12 should remain light yellow to light pink. Shifts outside this range signal breakdown.
Container damage. Cracked vials, damaged pen cartridges, or compromised rubber stoppers eliminate any expiration date guarantee. The sterile barrier has been broken, and contamination is possible regardless of the medication age.
Leakage. Any evidence of leaking around the stopper or pen tip means the seal has failed. Discard the entire unit.
What visual inspection cannot tell you
Here is the uncomfortable truth. A vial of tirzepatide can look perfectly fine, clear, colorless, no particles, and still be significantly degraded. Early-stage hydrolysis, deamidation, and oxidation do not produce visible changes. The solution looks identical to a fresh preparation, but the active peptide content may be 20, 30, or even 50 percent reduced.
This is why expiration dates and beyond-use dates exist. Visual inspection is a necessary check, but it is not sufficient. You need both. If it looks bad, do not use it. If it looks fine but the date has passed, still do not use it.
Functional signs that your tirzepatide may have degraded
Sometimes the first indication is not what you see in the vial but what you feel, or stop feeling, in your body.
Appetite returning earlier than expected. If you have been on a stable tirzepatide dose and your appetite suppression suddenly weakens mid-cycle, degraded medication is a possible cause. This is especially suspect if nothing else in your routine has changed.
Weight loss plateau. A sudden stall in weight loss while on a previously effective dose can have many causes. But if the stall coincides with using medication from an older vial or one with questionable storage history, degradation should be on your list of suspects.
Blood glucose control slipping. For those using tirzepatide for diabetes management, unexplained increases in fasting glucose or postprandial spikes may indicate reduced medication potency rather than disease progression.
Increased nausea or unusual injection site reactions. While this seems counterintuitive (you might expect less side effects from weaker medication), degraded peptides with altered structures can sometimes trigger different inflammatory responses at the injection site.
Brand-name vs. compounded tirzepatide: shelf life differences that matter
The distinction between brand-name and compounded tirzepatide is not just about cost or availability. Their shelf life profiles are fundamentally different, and understanding why helps you manage each type appropriately.
Brand-name tirzepatide (Mounjaro, Zepbound)
Eli Lilly manufactures both Mounjaro and Zepbound under strict FDA oversight with extensive stability testing. The expiration dates on these products are backed by real-time stability data showing that the peptide maintains specified potency levels throughout the labeled shelf life when stored correctly.
Key characteristics:
24-month shelf life from manufacture when refrigerated
21 days at room temperature (below 86 degrees Fahrenheit)
No preservatives in the prefilled pens
Single-use pens designed for one injection
Extensive stability testing and quality control
Consistent potency from batch to batch
The single-use pen design eliminates one major variable: you never need to worry about how long an opened vial lasts because each pen is used once and discarded. This is a significant advantage from a shelf life perspective.
Compounded tirzepatide
Compounded tirzepatide operates in a different regulatory framework. Compounding pharmacies prepare these formulations under USP Chapter 797 guidelines for sterile compounding, and the beyond-use dates they assign are based on a combination of USP default dating, any proprietary stability testing they have conducted, and the specific formulation used.
Key characteristics:
28 to 90 day beyond-use date from preparation (varies by pharmacy)
May or may not contain preservatives
Multi-dose vials requiring multiple punctures
Potency can vary between batches
Different pharmacies produce different quality levels
Storage requirements may differ from brand-name
The variability between compounding pharmacies is significant. One pharmacy might assign a 28-day beyond-use date while another assigns 90 days for what appears to be a similar formulation. This difference reflects their individual stability testing, formulation specifics, and quality assurance practices.
Reputable compounding pharmacies invest in stability testing for their specific formulations and can provide data supporting their assigned beyond-use dates. If your pharmacy cannot explain how they determined the dating on your medication, that is a red flag.
Why the shelf life gap exists
The dramatic difference between 24 months (brand-name) and 28 to 90 days (compounded) has several explanations.
First, manufacturing environment. Eli Lilly produces tirzepatide in pharmaceutical-grade manufacturing facilities with precisely controlled conditions. Compounding pharmacies, even excellent ones, operate in different environments with different levels of environmental control.
Second, formulation specifics. Brand-name tirzepatide uses proprietary buffer systems, stabilizers, and packaging designed to maximize shelf life. Compounded formulations use different excipients and may not have the same stabilization advantages.
Third, testing depth. Eli Lilly has conducted years of stability studies on their specific formulations. Most compounding pharmacies rely on USP default dating guidelines rather than product-specific long-term stability data.
Fourth, regulatory requirements. FDA requires manufacturers to prove shelf life claims through rigorous testing. Compounding pharmacies operate under different regulatory standards that do not require the same level of stability documentation.
None of this means compounded tirzepatide is bad. It means its shelf life is shorter, and you need to manage it accordingly. Use it within the assigned beyond-use date. Do not assume it lasts as long as brand-name products.

The reconstitution factor: how mixing changes everything
If you are using lyophilized tirzepatide that requires reconstitution with bacteriostatic water, the act of mixing fundamentally changes the expiration timeline. Understanding this transition is essential for proper use.
Before reconstitution: maximum stability
Lyophilized tirzepatide powder in a sealed vial is the most shelf-stable form. With no water present, hydrolysis cannot occur. The powder can maintain potency for 12 months or longer when stored at 2 to 8 degrees Celsius in a sealed, light-protected container.
If you are stocking up on lyophilized tirzepatide, this is the form to store. Keep it sealed, keep it cold, and keep it dark. It will maintain its potency far longer than any liquid formulation.
After reconstitution: the clock resets
The moment you add water to lyophilized tirzepatide, every degradation pathway activates. Hydrolysis begins. The risk of microbial contamination becomes real. The shelf life drops from months to weeks.
With bacteriostatic water (which contains 0.9 percent benzyl alcohol as a preservative): The reconstituted solution typically lasts up to 28 to 30 days refrigerated. Some sources cite up to 90 days, but the more conservative 28 to 30 day window is generally recommended for peptides.
With sterile water (no preservative): The reconstituted solution should be used within 24 to 48 hours for single-use applications, or up to 7 to 10 days refrigerated if handling has been perfectly sterile. Without bacteriostatic preservative, there is nothing preventing microbial growth if any contamination occurred during reconstitution.
This is why bacteriostatic water is strongly preferred over sterile water for multi-dose reconstitution. The small amount of benzyl alcohol provides a meaningful buffer against microbial contamination that extends usable shelf life significantly.
Reconstitution best practices that maximize shelf life
How you reconstitute affects how long the solution remains viable.
Use proper technique. Swab the vial top with alcohol before puncturing. Use a sterile syringe. Do not touch the needle to anything non-sterile. Each contamination opportunity shortens the usable life of the reconstituted solution.
Do not shake the vial. Gently swirl or roll the vial to dissolve the powder. Vigorous shaking creates foam, introduces air, and can physically damage the peptide through agitation-induced aggregation. If your solution is foamy after mixing, you were too aggressive.
Record the reconstitution date. Write the date on the vial with a permanent marker. It is surprisingly easy to lose track of when you mixed a vial, especially if you have multiple vials in rotation. A simple date label prevents guesswork.
Use a proper reconstitution ratio. The amount of bacteriostatic water you add affects concentration and, indirectly, how many times you need to puncture the vial. Fewer punctures mean fewer contamination opportunities. Calculate your dosing so that you use the entire vial within the recommended window with minimal punctures.
What happens if you use expired tirzepatide
People ask this question for a practical reason. They have expensive medication that is just past the date, and they want to know if it is still usable. Here is the honest assessment.
Scenario 1: slightly past expiration, properly stored
If your tirzepatide is a few days past the printed expiration date and has been stored correctly at 2 to 8 degrees Celsius the entire time, it has likely not undergone dramatic degradation. Expiration dates include a safety margin. The medication did not go from effective to useless overnight.
That said, no regulatory body endorses using expired medication. The FDA explicitly advises against it. The potency is unknown, the safety is unverified, and you are making a decision without data.
Scenario 2: significantly past expiration
Medication that is weeks or months past expiration has had considerably more time for degradation. Even under ideal storage, chemical degradation accumulates. The further past the date, the more uncertain the potency.
Using significantly expired tirzepatide for weight loss likely means you are injecting less active peptide than you think. The dose you are counting on is not the dose you are getting. For diabetes management, this uncertainty is medically concerning because inadequate glucose control has real health consequences.
Scenario 3: past expiration with questionable storage
This is the worst combination. Expired medication that has also experienced temperature excursions, light exposure, or other storage violations has likely degraded significantly more than properly stored expired medication.
A vial that sat in a hot delivery truck for hours, then spent two months in a refrigerator, then passed its expiration date has experienced compounding damage from multiple sources. The effective potency could be a fraction of what the label states.
Do not use it. The cost of replacement medication is lower than the cost of weeks of ineffective treatment plus the potential health risks.
What the research says
There is no published clinical trial data on the efficacy or safety of expired tirzepatide in humans. The manufacturer's expiration date is the last point at which they can guarantee the product meets specifications. Beyond that date, you are in uncharted territory.
Some general pharmaceutical research, particularly the FDA's Shelf Life Extension Program (SLEP) which tests expired military stockpile medications, has found that many solid-dose medications retain potency well past their expiration dates. However, peptide medications are fundamentally different from pills. They are more fragile, more susceptible to degradation, and more dependent on proper storage. Findings from tablet stability studies do not transfer to injectable peptides.
Travel and tirzepatide: managing expiration on the go
Traveling with tirzepatide introduces storage challenges that can accelerate expiration. Whether you are flying across the country or driving across town in summer heat, the medication needs protection.
Flying with tirzepatide
Airlines are not kind to medications that need refrigeration. Checked luggage can experience temperatures below freezing in the cargo hold (which would destroy tirzepatide) or above 86 degrees Fahrenheit on a hot tarmac (also destructive).
Always carry tirzepatide in your carry-on luggage. Use an insulated medication travel case with gel packs that maintain 2 to 8 degrees Celsius without freezing the medication. Many companies make travel cases specifically designed for injectable medications.
A medical travel cooler is not a luxury item for peptide users. It is a necessity. The 21-day room-temperature window gives you some buffer, but why use up that buffer unnecessarily when a simple cooler keeps the medication at optimal temperatures?
Road trips and hot climates
A car interior in summer can exceed 140 degrees Fahrenheit. That is nearly three times the temperature at which tirzepatide expires immediately. Never leave medication in a vehicle, even "for a few minutes."
If you are driving, keep the medication in an insulated cooler in the passenger compartment (not the trunk, which gets hotter). If you stop for meals or errands, bring the cooler with you.
Extended travel without refrigeration
For situations where refrigeration is genuinely unavailable for extended periods, remember the 21-day room-temperature rule for brand-name products. Plan your travel medication supply accordingly. If your trip exceeds 21 days without reliable refrigeration access, you may need to arrange for a fresh supply at your destination.
For compounded tirzepatide, check with your pharmacy about room-temperature stability. It may be shorter than 21 days depending on the formulation. Some compounded preparations require continuous refrigeration with no room-temperature allowance.

Tirzepatide shelf life compared to other GLP-1 medications
Understanding how tirzepatide expiration compares to similar medications provides useful context, especially if you are switching between medications or deciding between options.
Tirzepatide vs. semaglutide shelf life
Semaglutide (brand names Ozempic and Wegovy) has similar storage requirements and shelf life to brand-name tirzepatide. Unopened semaglutide pens last until the manufacturer expiration date when refrigerated, and can survive at room temperature for limited periods.
The key difference is in the room-temperature window. Semaglutide pens (Ozempic) allow up to 56 days at room temperature below 86 degrees Fahrenheit. Tirzepatide allows only 21 days. This makes semaglutide considerably more travel-friendly and more forgiving of brief storage lapses.
If you are comparing the two medications and travel frequently or have unreliable refrigeration, semaglutide's longer room-temperature stability may be a practical advantage worth considering.
For compounded versions, both medications have similar beyond-use date ranges of 28 to 90 days depending on the pharmacy. The compounded semaglutide shelf life is governed by the same USP guidelines as compounded tirzepatide.
If you are curious about the dosage differences between the two, or wondering about conversion charts for switching, those are separate but related considerations.
Tirzepatide vs. retatrutide considerations
Retatrutide is a newer triple agonist (GIP, GLP-1, and glucagon) that is still in clinical trials. As a research peptide, it is primarily available in lyophilized form with shelf life characteristics similar to lyophilized tirzepatide: stable as powder for months, limited once reconstituted.
The same fundamental principles apply. Peptide structure determines degradation pathways. Temperature determines degradation speed. Storage determines actual shelf life regardless of printed dates.
Maximizing tirzepatide shelf life: a complete protocol
You have paid for this medication. You want every microgram to work as intended. Here is a comprehensive protocol for maximizing the useful life of your tirzepatide.
Storage protocol
Step 1: Immediate refrigeration. The moment you receive tirzepatide, put it in the refrigerator. Do not set it on the counter while you unpack other items. Do not leave it in the delivery box until later. Immediate refrigeration maximizes the total shelf life.
Step 2: Optimal placement. Place the medication on the middle shelf toward the back of the refrigerator, not in the door. Consider using a small plastic container to keep vials organized and prevent them from rolling against the back wall where they might freeze.
Step 3: Light protection. Keep medication in original packaging or store vials in an opaque container. Even the light from opening your refrigerator contributes to photodegradation over time.
Step 4: Temperature monitoring. Place a thermometer near your medication. Check periodically that temperatures stay between 36 and 46 degrees Fahrenheit. Adjust your refrigerator settings if needed.
Step 5: FIFO rotation. If you have multiple vials, use the oldest ones first. First in, first out. Label each vial with the date received to make this easy.
Handling protocol
Minimize room temperature exposure. Remove the vial or pen from the refrigerator only when you are ready to inject. Do not set it out to "warm up" for extended periods. A brief temperature equilibration of 5 to 10 minutes is sufficient if cold injections bother you.
Clean technique always. Alcohol swab on the vial top before every puncture. Clean hands. Sterile syringe. These practices do not just prevent infection. They extend the usable life of multi-dose vials by minimizing contamination introduction.
Record everything. Date opened. Date reconstituted (if applicable). Number of punctures. Expected discard date. A simple label on the vial prevents confusion and ensures you never accidentally use medication past its window.
When to discard
Discard tirzepatide immediately if:
The printed expiration date or beyond-use date has passed
The solution is cloudy, discolored, or contains particles
The vial has been at room temperature for more than 21 days (brand-name) or past your pharmacy's specified window (compounded)
The medication has been frozen at any point
The medication has been exposed to temperatures above 86 degrees Fahrenheit
An opened vial has been punctured for more than 28 days
You are unsure of the storage history
The container shows any damage or leakage
When in doubt, throw it out. The cost of a replacement vial is always less than the cost of using degraded medication that does not work.
Common mistakes that shorten tirzepatide shelf life
Even people who know the rules make these mistakes. Knowing the most common errors helps you avoid them.
Storing in the refrigerator door
This is the number one mistake. The door is the warmest part of the refrigerator and experiences the most temperature fluctuation. Every time you open the fridge, the door temperature swings. Over weeks and months, these repeated temperature cycles accelerate peptide degradation significantly.
Move your tirzepatide to the middle shelf. Today. Right now if you can.
Assuming compounded tirzepatide lasts as long as brand-name
People who switch from Mounjaro to compounded tirzepatide often carry over their assumption about shelf life. "My Mounjaro lasted months in the fridge, so this compounded vial should too." It does not. The beyond-use date on compounded tirzepatide is dramatically shorter, and exceeding it is not safe.
Re-refrigerating after room temperature storage
You took the pen out of the fridge for travel. You kept it at room temperature for a week. Now you are home and want to put it back in the fridge to "pause" the countdown. Unfortunately, the manufacturer says you cannot. The 21-day clock continues from the moment it left refrigeration, whether it goes back in the fridge or not.
Not tracking reconstitution dates
"I mixed this vial... sometime last month? Maybe?" If you cannot state with confidence when a vial was reconstituted, you cannot know whether it is within its usable window. The solution is simple: write the date on the vial every time.
Keeping "backup" vials too long
Stocking up on tirzepatide makes practical sense if supply is uncertain. But those backup vials have expiration dates too. If you buy three months of supply and your prescription changes or treatment pauses, those vials are still aging. Check dates regularly and use the oldest stock first.
Ignoring delivery conditions
Mail-order tirzepatide sits in delivery trucks, on porches, and in mailboxes. In summer, a package on your front porch for a few hours can experience temperatures well above 86 degrees Fahrenheit. If your medication arrives warm to the touch, contact the pharmacy immediately. They should be shipping with cold packs and insulated packaging. If they are not, find a pharmacy that does.

A complete shelf life reference table
For quick reference, here is a comprehensive summary of tirzepatide shelf life under various conditions.
Form | Storage condition | Shelf life | Notes |
|---|---|---|---|
Brand-name pen (sealed) | Refrigerated (2-8 C) | Until printed expiration (~24 months) | Keep in original carton |
Brand-name pen (sealed) | Room temperature (below 30 C) | 21 days maximum | Cannot re-refrigerate after |
Brand-name pen (sealed) | Above 30 C (86 F) | Expires immediately | Discard even if not used |
Brand-name pen (sealed) | Frozen | Destroyed immediately | Do not thaw and use |
Compounded liquid (sealed) | Refrigerated (2-8 C) | 28-90 days (per pharmacy BUD) | Varies by pharmacy |
Compounded liquid (opened) | Refrigerated (2-8 C) | 21-28 days after first puncture | Clean technique essential |
Lyophilized powder (sealed) | Refrigerated (2-8 C) | 12+ months | Most stable form |
Reconstituted with bac water | Refrigerated (2-8 C) | 28-30 days | Record reconstitution date |
Reconstituted with sterile water | Refrigerated (2-8 C) | 7-10 days (conservative) | No preservative protection |
This table covers the most common scenarios, but always defer to the specific guidance provided by your manufacturer or compounding pharmacy. Their data supersedes general guidelines.
The cost of ignoring expiration dates
Let us talk about money. Because expired tirzepatide is not just a health concern. It is a financial one.
Brand-name tirzepatide is expensive. Compounded tirzepatide is more affordable, but still represents a meaningful investment. Every degraded vial is money wasted.
But the real financial cost is not the medication itself. It is the lost progress.
If you have been using degraded tirzepatide for two months without knowing it, you have lost two months of potential weight loss or metabolic improvement. You have spent two months on a dose that was not actually what you thought it was. You cannot get those two months back. You will need additional time on effective medication to make up the lost ground.
Proper storage and attention to expiration dates is not obsessive. It is basic cost management. You are protecting your investment in both the medication and the results.
When to contact your pharmacy or provider
Certain situations warrant a conversation with the pharmacy or prescribing provider rather than making decisions on your own.
Medication arrived warm or damaged. Contact the pharmacy immediately. Most reputable pharmacies will replace medication that arrived outside specified temperature ranges. Document the condition with photos.
You are unsure about storage history. If there was a power outage, a refrigerator malfunction, or any other situation where you are uncertain whether the medication stayed within the proper temperature range, ask your pharmacy. They can advise based on the specific duration and temperature of the excursion.
Results have suddenly changed. If your results with tirzepatide have changed without any other explanation, discuss the possibility of medication degradation with your provider. They may recommend a new vial or pen to test whether the medication is the issue.
You have medication close to expiration that you will not use in time. Some pharmacies may offer exchanges or credits for unused medication approaching its expiration date. It never hurts to ask.
You are switching between brand-name and compounded. The different shelf life profiles mean your storage and usage habits may need to change. Your provider or pharmacist can help you adjust.
Frequently asked questions
Can I use tirzepatide one week after the expiration date?
The FDA advises against using any medication past its expiration date. While tirzepatide does not instantly become useless on the expiration date, its potency is no longer guaranteed. A week past expiration with perfect storage history presents lower risk than medication that is months expired or poorly stored, but no clinical data supports using it. The safest approach is replacement. If you are managing diabetes with tirzepatide, using potentially weakened medication is a medical risk you should not take without consulting your provider.
Does freezing tirzepatide make it last longer?
No. Freezing destroys tirzepatide permanently. Unlike some medications that can be frozen for long-term storage, tirzepatide's peptide structure is damaged by ice crystal formation. This damage does not reverse upon thawing. Never freeze tirzepatide, and if it freezes accidentally, discard it immediately. This applies to all forms: brand-name pens, compounded solutions, and reconstituted solutions.
How do I know if my compounded tirzepatide is still good?
Check three things. First, is it within the beyond-use date printed on the vial by your compounding pharmacy? Second, has it been stored at 2 to 8 degrees Celsius continuously? Third, does the solution look clear, colorless (or slightly yellow), and free of particles? If all three answers are yes, it should still be effective. If any answer is no, or if you are uncertain about storage history, contact your pharmacy.
Why does compounded tirzepatide expire so much faster than Mounjaro?
The difference comes down to manufacturing environment, formulation specifics, and testing depth. Eli Lilly manufactures Mounjaro in pharmaceutical-grade facilities with proprietary stabilizers and extensive stability testing. Compounding pharmacies work under different conditions with different formulations and are governed by USP guidelines that typically result in more conservative beyond-use dates. The shorter dating on compounded products reflects both genuine stability differences and regulatory caution.
Does the type of water used for reconstitution affect shelf life?
Yes, significantly. Bacteriostatic water contains 0.9 percent benzyl alcohol, which provides mild antimicrobial protection and extends the reconstituted solution's usable life to approximately 28 to 30 days. Sterile water without preservatives offers no microbial protection, limiting the usable window to 7 to 10 days at most. For multi-dose vials, bacteriostatic water is strongly preferred.
What should I do if my tirzepatide was left out of the fridge overnight?
One night at room temperature is well within the 21-day allowance for brand-name products, assuming your room temperature did not exceed 86 degrees Fahrenheit. The medication is fine. However, once removed from refrigeration, some manufacturers recommend not re-refrigerating, meaning the 21-day countdown has now started. For compounded tirzepatide, check with your pharmacy about room-temperature tolerance for your specific formulation.
Can I tell the potency of my tirzepatide without lab testing?
Not accurately. Visual inspection can identify obvious degradation (cloudiness, particles, color changes), but early potency loss produces no visible signs. The solution will look identical to fresh medication even with 20 to 30 percent potency reduction. This is why following expiration dates and proper storage is essential. You cannot see mild degradation, so you must prevent it through proper handling.
Is expired tirzepatide dangerous to inject?
Expired tirzepatide is more likely to be ineffective than dangerous. Peptide degradation typically produces inactive fragments rather than toxic compounds. However, three real risks exist: reduced therapeutic effect (dangerous for diabetes management), potential contamination in older opened vials, and possible immunogenic responses from aggregated peptides. The risk-benefit calculation strongly favors using fresh medication.
External resources
USP Chapter 797: Pharmaceutical Compounding, Sterile Preparations
Structural insights into tirzepatide pharmacological actions (NIH)
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based storage guides, proven protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members get access to detailed dosage calculators, safety databases, and expert guidance for every step of the process.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your refrigerator stay cold, and your expiration dates stay far in the future.