Feb 6, 2026

You cannot walk into a pharmacy and buy retatrutide. Not today. Not this year. Probably not until early 2027. The most promising weight loss peptide in development, showing 24.2% body weight reduction in clinical trials, remains locked behind FDA approval timelines while counterfeit versions flood online markets and fitness forums promote untested powders with unknown contents.
The disconnect creates a dangerous situation. Real demand meets fake supply.
People searching for legitimate retatrutide find research chemical vendors, compounding pharmacies making illegal claims, and underground sources selling substances that may contain anything from properly synthesized peptide to contaminated powder to nothing at all. The FDA has issued multiple warnings. Compounding pharmacies have received cease and desist letters. Yet the listings multiply.
This guide covers every legitimate pathway to access retatrutide, the research chemical market reality, quality testing that separates safe from dangerous, legal considerations that matter, and practical alternatives while you wait for FDA approval. You will learn which vendors actually test their products, how clinical trial enrollment works, what compounding pharmacy claims to ignore, and the specific timeline for pharmaceutical availability. No guesswork. No assumptions. Just documented facts about how to navigate the retatrutide landscape safely until proper prescriptions become possible.
SeekPeptides provides the most comprehensive resource for understanding peptide access, quality verification, and safe research practices while navigating the complex landscape between clinical trials and FDA approval.
Understanding retatrutide current legal status
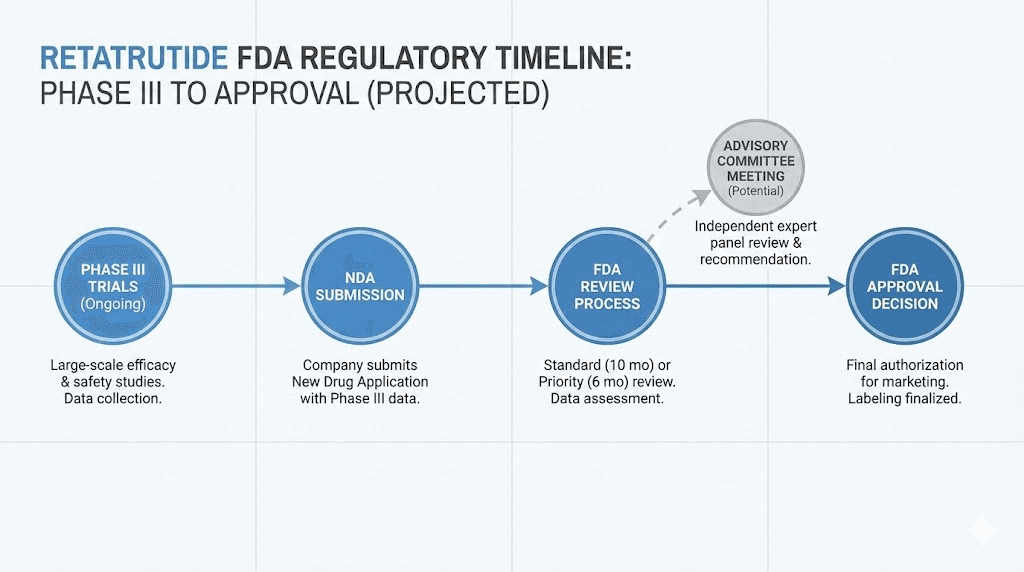
Retatrutide exists in regulatory limbo. Clinical trials continue. Phase III data impresses. Eli Lilly projects FDA submission in 2026. But today, right now, no legal prescription pathway exists outside research studies. The peptide remains investigational, which carries specific implications for anyone searching for legitimate access.
The FDA classifies retatrutide as an unapproved drug under active investigation. This means doctors cannot prescribe it. Pharmacies cannot dispense it. And compounding pharmacies, despite what some advertise, absolutely cannot legally compound it. Federal law prohibits compounding with bulk substances not on the approved list. Retatrutide is not on that list. Any compounding pharmacy offering retatrutide violates federal regulations and faces potential shutdown.
Phase III trials for weight loss peptides like retatrutide typically conclude in 2025-2026, with FDA review processes adding 6-12 months minimum. The realistic availability timeline for legitimate pharmaceutical retatrutide lands somewhere in early to mid 2027, assuming trials show no unexpected safety signals and Lilly submits applications on schedule.
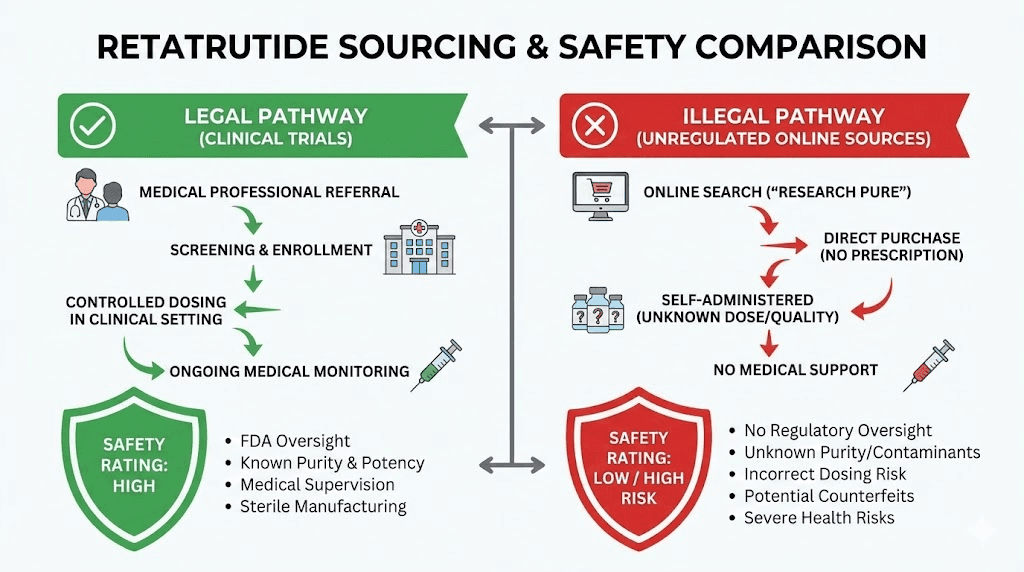
What this means practically is simple. If someone offers to prescribe you retatrutide today through telehealth, they operate outside legal boundaries. If a compounding pharmacy ships you retatrutide, they violate federal law. If an online vendor sells "pharmaceutical grade" retatrutide for human use, they lie. The only legal access pathway involves clinical trial enrollment through approved research sites.
Research chemical vendors operate in a different space. They sell retatrutide marked "not for human consumption" and "research use only." These products target laboratory research, cell studies, and animal models. Purchasing research chemicals for personal use exists in legal gray zones, with the substances themselves not illegal but their intended use potentially violating FDA regulations around unapproved drugs.
The FDA has issued warning letters to multiple entities selling or promoting retatrutide outside approved research contexts. These warnings target both online sellers making therapeutic claims and medical practices offering off-label compounded versions. The agency monitors the market actively. Enforcement actions continue. Anyone considering retatrutide purchase through unofficial channels should understand the regulatory landscape clearly.
Clinical trial access remains the only legitimate option
Eli Lilly operates multiple Phase III retatrutide trials across North America. These studies provide the only legal pathway to access pharmaceutical-grade retatrutide under medical supervision. Trial participants receive genuine medication, professional monitoring, and contribute to the data determining whether FDA approval proceeds.
The ClinicalTrials.gov registry lists active retatrutide studies with specific inclusion criteria. Most trials target obesity or overweight individuals with at least one weight-related comorbidity. BMI requirements typically start at 30 kg/m² or 27 kg/m² with complications. Age ranges vary but usually include adults 18-75 years old.
Enrollment involves extensive screening. Medical history review. Physical examination. Laboratory testing. Exclusion criteria eliminate candidates with GLP-1 contraindications including personal or family history of medullary thyroid carcinoma, multiple endocrine neoplasia syndrome type 2, or severe gastrointestinal disease. The screening process protects both participants and trial integrity.
Trial participation requires commitment. Regular clinic visits. Adherence to dosing schedules. Completion of questionnaires and assessments. Blood draws and vital sign monitoring. Most trials run 48-72 weeks with ongoing follow-up periods. Geographic proximity to trial sites matters as in-person visits cannot be skipped.
The upside balances the requirements. Participants receive genuine retatrutide at no cost. Medical supervision throughout the trial period. Professional management of any adverse effects. And crucially, they avoid the contamination risks, dosing uncertainties, and legal complications of underground sources.
What FDA approval will actually change
FDA approval transforms everything about retatrutide access. Legitimate prescriptions. Pharmacy dispensing. Insurance coverage negotiations. Patient assistance programs. The entire pharmaceutical infrastructure that makes drugs accessible to millions.
When retatrutide gains approval, doctors can prescribe it for labeled indications. Most likely obesity treatment initially, potentially expanding to diabetes management or cardiovascular risk reduction based on trial outcomes. Prescriptions flow through normal channels. Pharmacies order from licensed distributors. Patients pick up medication just like any other prescription.
The cost structure follows pharmaceutical norms. Eli Lilly will price retatrutide competitively with semaglutide and tirzepatide, likely landing in the $1000-1500 monthly range before insurance. Insurance coverage depends on formulary decisions, prior authorization requirements, and demonstrated medical necessity. The cost analysis for GLP-1 medications provides relevant comparison.
Compounding pharmacies gain no new rights. Even after FDA approval, retatrutide will not appear on the bulk substances list that permits compounding. The pathway requires Lilly patents to expire and FDA to specifically approve bulk substance use, a process taking years beyond initial approval. Any compounding pharmacy claiming they can provide retatrutide immediately after approval misleads customers.
Off-label prescribing becomes technically possible once approval exists, but practically unlikely. The peptide targets specific obesity indications. Insurance will not cover off-label use. And given the monthly costs, out-of-pocket payments for non-indicated use exceed what most patients consider reasonable. Expect strict adherence to labeled indications initially.
Research peptide vendor landscape and quality concerns
Research chemical suppliers sell retatrutide today. Lots of them. A quick search returns dozens of vendors offering 10mg vials, 20mg vials, bulk powder. Prices range from suspiciously cheap to somewhat reasonable. Quality varies from legitimate research grade to potentially dangerous contamination. The market lacks regulation. Buyers bear all risk.
These vendors target academic research, pharmaceutical development, and chemical biology studies. Their terms of service explicitly state "not for human consumption" and "research use only." This legal disclaimer protects them while creating a fig leaf of legitimacy. Everyone understands the actual customer base includes individuals using peptides personally, but vendors maintain plausible deniability.
The fundamental problem is verification. No regulatory body tests these products. No FDA oversight ensures quality. Vendors may provide certificates of analysis showing 99%+ purity, but these documents are easily fabricated and often represent one tested batch while shipping untested material. Independent testing reveals the scope of the problem.
Finnrick Analytics, an independent testing service, has evaluated 1,602 retatrutide samples from 128 different vendors between December 2024 and January 2026. Purity ranged from 62.38% to 100%. Let that sink in. Some samples contained less than two-thirds actual retatrutide. Others tested at pharmaceutical grade. Customers have no reliable way to distinguish between vendors without independent testing.
Quantity diverged by up to ±260% versus advertised amounts. A vial labeled 10mg might contain 3.6mg or 36mg. Dosing becomes impossible without verification. Underdosed vials waste money and produce no results. Overdosed vials risk serious adverse effects as users inadvertently inject multiple times their intended dose.
Endotoxin contamination presents the most immediate danger. Recent FDA investigations found 78% of research-grade retatrutide samples failed endotoxin screening with levels exceeding 100 EU/mg compared to the safe pharmaceutical limit of 0.1 EU/mg. Endotoxins cause fever, inflammation, and potentially severe immune reactions. Injecting contaminated peptides risks serious medical complications.
Peptide fragmentation under mass spectrometry affected 63% of tested samples. Fragmented peptides may retain some activity or none at all. Users cannot know if their injections work as intended. The variability makes dosing protocols unreliable and results unpredictable.
Vendor quality tiers and testing results
Finnrick testing categorizes vendors into quality tiers based on consistent performance across multiple batches. These ratings provide the only objective quality assessment available to researchers and consumers navigating the unregulated market.
A-tier vendors (rating: Great) show test scores averaging over 7 with no scores below 6 across at least 6 independent tests. These vendors demonstrate consistent quality, accurate labeling, and low contamination rates. Paradigm Peptides achieved A-tier status with retatrutide testing. Their COA-verified purities exceed 99.4% consistently.
E-tier vendors (rating: Bad) produce test scores averaging below 4 or show some scores below 3 across multiple tests. These vendors ship inconsistent quality, potentially dangerous contamination, and significantly mislabeled quantities. Peptide Sciences received E-tier rating for retatrutide despite marketing claims of pharmaceutical quality.
The gap between tiers is not subtle. A-tier vendors provide research-grade material suitable for cellular studies and animal models. E-tier vendors ship substances that may contain correct peptide mixed with unknown contaminants, incorrect quantities, and potentially harmful endotoxins. Price differences between tiers often amount to $50-100 per vial, trivial compared to contamination risks.
Customer reviews on platforms like Trustpilot show mixed feedback even for established vendors. Some Peptide Sciences customers report satisfactory experiences while others note clumpy reconstitution, cloudy solutions, and lack of expected results. These inconsistencies align with variable testing results suggesting batch-to-batch quality fluctuation.
Understanding proper reconstitution technique matters, but technique cannot fix fundamentally contaminated or mislabeled product. Users may attribute poor results to personal error when product quality actually caused the problem.
Certificate of analysis interpretation
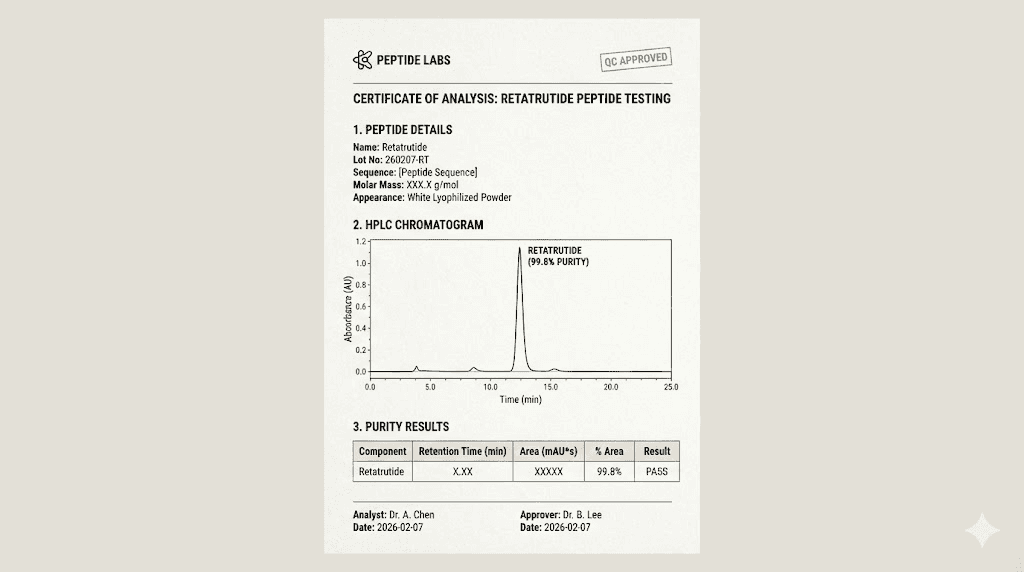
Every research peptide vendor provides certificates of analysis with their products. These documents supposedly verify purity, identity, and quality. In reality, COAs range from legitimate third-party testing to complete fabrications. Learning to evaluate COA credibility separates informed researchers from victims.
Legitimate COAs come from independent analytical laboratories using validated testing methods. High-Performance Liquid Chromatography identifies and quantifies peptide purity. Mass spectrometry confirms molecular identity. Endotoxin assays measure bacterial contamination. Each test should list the laboratory name, test date, methodology, and specific results.
Batch-specific COAs matter most. Generic or representative COAs from months or years past tell you nothing about your actual product. Trustworthy vendors provide COAs dated within weeks of your purchase, testing the specific batch you receive. They include batch numbers matching your vial labels. And they come from recognized analytical facilities, not in-house testing.
Red flags include missing laboratory information, suspiciously perfect results showing exactly 100% purity, identical COAs for multiple products, or documents lacking standard analytical details like retention times or mass/charge ratios. These suggest fabricated testing rather than legitimate analysis.
The HPLC chromatogram itself provides valuable information. Pharmaceutical-grade peptides show a single dominant peak representing the target compound with minimal additional peaks indicating impurities. Complex chromatograms with multiple significant peaks suggest contamination, degradation products, or synthesis byproducts remaining in the final product.
Mass spectrometry confirms you received the correct compound. The molecular weight should match retatrutide specifications exactly. Differences of even a few daltons indicate wrong peptide, fragmented sequences, or chemical modifications. This test answers the fundamental question of whether your vial contains retatrutide at all.
For researchers without analytical chemistry backgrounds, third-party verification services like Finnrick provide COA validation and independent testing. Sending samples for verification costs $100-200 but confirms whether expensive peptide purchases contain advertised contents. The investment makes sense for anyone planning extended research protocols with significant peptide expenditures.
Domestic versus international suppliers
Research peptide vendors split between domestic US operations and international sources, primarily Chinese manufacturers. The distinction matters for legal considerations, shipping reliability, quality assurance, and pricing. Each model carries specific advantages and risks.
Domestic US vendors typically resell peptides manufactured overseas but operate warehouses, customer service, and shipping from American locations. This structure provides faster shipping, easier communication, domestic legal jurisdiction for disputes, and theoretically better quality control. Prices run 20-40% higher than direct international purchase.
Established domestic vendors like Paradigm Peptides maintain US facilities, employ customer service staff in similar time zones, and process orders rapidly. Paradigm Peptides retatrutide testing shows A-tier quality ratings with consistent purity and accurate labeling. The premium pricing reflects verification costs, domestic overhead, and quality assurance processes.
International direct purchase from Chinese manufacturers offers significant cost savings. A 10mg retatrutide vial might cost $150-200 from US vendors but $80-120 shipped directly from Chinese suppliers. The savings scale with larger orders. Researchers planning extended protocols save hundreds or thousands of dollars through international sourcing.
The tradeoffs include longer shipping times of 2-4 weeks, customs clearance uncertainties, communication challenges across time zones and languages, and variable quality control. Some Chinese manufacturers produce pharmaceutical-grade material for research institutions worldwide. Others ship questionable substances with fabricated testing documents. Distinguishing between them requires investigation.
Customs considerations affect international shipments. US Customs and Border Protection may inspect packages containing research chemicals. Shipments require proper documentation declaring contents accurately. Mislabeled packages risk seizure. Even properly labeled research peptides may face customs holds or questions, delaying delivery.
Payment methods differ significantly. Domestic vendors accept credit cards through normal processors. International suppliers often require cryptocurrency, wire transfer, or payment services like Alibaba Trade Assurance. These payment methods offer less buyer protection than credit cards, increasing financial risk if vendors fail to deliver or ship incorrect products.
Customer support quality varies. US vendors typically respond to inquiries within hours during business days. International suppliers may take days to respond due to time zone differences and language barriers. Resolving shipping issues, quality concerns, or order errors takes longer and requires more persistent communication.
Cold chain maintenance during shipping affects peptide stability. Peptide storage requirements specify temperature ranges for both lyophilized powder and reconstituted solution. International shipments spend weeks in transit, potentially exposing peptides to temperature extremes. Domestic shipments arrive within days, reducing degradation risk.
Clinical trial enrollment process and requirements
Enrolling in retatrutide clinical trials provides the only legitimate method to access pharmaceutical-grade peptide under medical supervision while contributing to research determining FDA approval. The process involves specific steps, strict requirements, and significant time commitment. Understanding eligibility criteria and trial expectations helps potential participants determine if this pathway suits their situation.
ClinicalTrials.gov maintains the comprehensive registry of active retatrutide studies. Searching "retatrutide" returns current Phase III trials recruiting participants. Each listing provides study location, inclusion/exclusion criteria, trial duration, visit schedule, and contact information for enrollment inquiry.
Eligibility requirements typically include BMI thresholds of 30 kg/m² or higher for obese individuals, or 27 kg/m² with at least one weight-related comorbidity like hypertension, dyslipidemia, or prediabetes. Age restrictions usually permit adults 18-75 years old. Both men and women qualify, though women of childbearing potential must use effective contraception throughout the trial.
Exclusion criteria eliminate candidates with medical conditions contraindicated for GLP-1 receptor agonists. Personal or family history of medullary thyroid carcinoma disqualifies participants. Multiple endocrine neoplasia syndrome type 2 excludes enrollment. Severe gastrointestinal disease including inflammatory bowel disease or gastroparesis prevents participation. Previous bariatric surgery within the past year typically excludes candidates.
Psychiatric considerations matter. Active eating disorders, current suicidal ideation, or psychiatric hospitalizations within recent months may disqualify candidates. Trial protocols require stable mental health because weight loss medications can affect mood and researchers need baseline psychiatric stability to identify any medication-related effects.
Medication restrictions include prior use of GLP-1 medications. Many trials exclude candidates who used semaglutide, tirzepatide, or other GLP-1 agonists within the previous 3-6 months. This washout period ensures baseline measurements reflect natural state rather than residual effects from previous medications.
The screening process begins with initial phone contact. Research coordinators verify basic eligibility before scheduling in-person screening visits. These visits involve comprehensive medical history review, physical examination, vital sign measurement, ECG recording, and extensive laboratory testing including complete blood count, comprehensive metabolic panel, liver function tests, thyroid function, and lipid panel.
Laboratory results determine final eligibility. Significant kidney dysfunction, severe liver disease, or uncontrolled thyroid disorders may exclude candidates. Researchers need participants healthy enough to complete the long trial duration while having sufficient obesity-related health concerns to justify experimental treatment.
Geographic proximity to trial sites matters critically. Most retatrutide trials require in-person visits every 4 weeks throughout the 48-72 week trial duration, plus additional screening visits and follow-up appointments. Participants living more than an hour from trial sites struggle with attendance requirements. Remote participation is not possible for trials requiring vital signs, laboratory draws, and physical assessments.
Visit schedules include dose escalation appointments, safety monitoring, efficacy assessments, and blood draws. Each visit takes 2-4 hours including check-in, assessments, study drug administration or pickup, and exit procedures. Participants should expect to spend 3-4 hours monthly at the trial site for the entire study duration.
What trial participation actually involves
Understanding daily trial life helps potential participants make informed decisions about enrollment. The commitment extends beyond monthly clinic visits to include medication administration, symptom tracking, dietary monitoring, and adherence to protocol requirements.
Study medication administration follows strict protocols. Retatrutide trials use subcutaneous injections similar to other GLP-1 medications. Participants either self-inject at home following training or receive injections during clinic visits depending on specific trial design. Most trials employ home administration with provided prefilled pens or syringes.
Dose escalation schedules start at lower doses and gradually increase to target levels over 4-8 weeks. This titration minimizes gastrointestinal side effects common with GLP-1 medications. Participants cannot adjust doses independently. Protocol violations risk disqualification from the study.
Symptom tracking requires daily or weekly logging of side effects, gastrointestinal symptoms, injection site reactions, and overall wellbeing. Many trials use electronic diaries or smartphone apps for real-time symptom reporting. This data helps researchers understand medication tolerability and identify safety signals.
Dietary monitoring may include food diaries, 24-hour dietary recalls, or structured meal plans. Weight loss trials need accurate nutritional data to distinguish medication effects from dietary changes. Some trials provide nutritional counseling to all participants, creating standardized dietary interventions across treatment and placebo groups.
Physical activity tracking through accelerometers or activity logs documents exercise patterns. This data controls for activity differences between participants and helps researchers understand whether weight loss results primarily from appetite suppression, increased activity, or metabolic effects.
Safety monitoring extends beyond scheduled visits. Participants receive contact information for reporting severe adverse events, concerning symptoms, or protocol questions. Research staff provide 24/7 coverage for urgent issues. Any serious medical events require immediate reporting and may trigger unscheduled safety assessments.
Laboratory monitoring tracks metabolic parameters, liver function, kidney function, and other safety measures. Blood draws occur regularly throughout the trial. Some studies include specialized testing like oral glucose tolerance tests, body composition scans, or cardiovascular assessments requiring additional time commitments.
Placebo controls mean some participants receive inactive treatment throughout the trial. Most Phase III trials use randomized, double-blind, placebo-controlled designs. Participants have a 30-50% chance of receiving placebo rather than active retatrutide. Neither participants nor research staff know which treatment individuals receive until trial completion.
Compensation and costs for participants
Trial participation economics matter to potential enrollees. While study medication comes free, participants invest significant time. Understanding compensation structures and covered costs helps evaluate whether trial enrollment makes financial sense.
Study medication at no cost represents the primary financial benefit. Pharmaceutical-grade retatrutide worth potentially $1000-1500 monthly comes free to all participants for the trial duration. This benefit accrues even to placebo recipients who receive free monitoring and transition to active medication in extension phases.
All trial-related medical care comes at no cost. Screening tests, laboratory monitoring, ECGs, physical examinations, and any assessments required by the protocol receive full coverage. Participants pay nothing for these services that would cost thousands of dollars in normal clinical practice.
Compensation for time and travel varies by trial. Some studies provide $50-150 per completed visit to offset parking, gas, time off work, and inconvenience. Others offer no monetary compensation beyond free treatment and monitoring. ClinicalTrials.gov listings sometimes specify compensation details, or participants can ask research coordinators during initial screening contact.
Treatment for adverse events follows standard medical pathways. If participants develop side effects requiring medical attention beyond trial-provided care, their regular health insurance covers these costs. Trials provide free treatment for serious adverse events directly caused by study medication, but routine healthcare for unrelated conditions remains the participant's responsibility.
Lost wages for visit attendance become participant responsibility unless employers provide paid time off. Multiple-hour quarterly visits may require half-day absences from work. Self-employed individuals sacrifice billable hours.
The time investment totals 40-60 hours across the full trial duration.
Compounding pharmacy claims and legal reality
Some compounding pharmacies advertise retatrutide availability through telehealth prescriptions. These claims violate federal law. Understanding the legal framework governing pharmaceutical compounding explains why such offers represent either ignorance of regulations or willful law-breaking.
The FDA maintains a list of bulk drug substances that compounding pharmacies may use. This list determines which medications pharmacies can compound legally when meeting other regulatory requirements. Retatrutide does not appear on this list. Its absence makes all retatrutide compounding illegal under federal law regardless of prescription status.
The Alliance for Pharmacy Compounding, an industry organization representing compounding pharmacies, has explicitly warned members against compounding retatrutide. Their notices state clearly that retatrutide and related compounds like cagrilintide cannot be compounded legally. Any pharmacy doing so violates federal regulations and risks enforcement action.
FDA enforcement confirms the regulatory stance. The agency has issued warning letters to compounding pharmacies offering retatrutide. These letters cite violations of the Federal Food, Drug, and Cosmetic Act and demand immediate cessation of retatrutide compounding. Pharmacies that ignore warnings face potential civil penalties, product seizures, and license revocation.
The legal framework makes no exception for prescriptions. A valid prescription from a licensed physician does not make retatrutide compounding legal. The prescription may be legitimate, but compounding pharmacies still violate law by preparing the medication. Both the pharmacy and potentially the prescribing physician face regulatory risk.
Some compounding pharmacies claim they use legal workarounds or exemptions. These claims lack merit. No regulatory pathway permits retatrutide compounding today. Pharmacies making such claims either misunderstand regulations or deliberately mislead customers. Either situation should prompt serious caution.
Telehealth providers partnering with compounding pharmacies to offer retatrutide create particularly concerning scenarios. Patients receive online prescriptions after brief consultations, then compounding pharmacies ship medication directly. This model circumvents traditional medical oversight while providing illegal products. Patients bear health risks from potentially contaminated or mislabeled compounds while pharmacies generate revenue from legally prohibited services.
Quality concerns with compounded products
Even if compounding retatrutide were legal, quality concerns would remain. Compounding pharmacies lack the quality control infrastructure, testing capabilities, and regulatory oversight that ensure pharmaceutical-grade medication safety. The peptide synthesis and handling complexities make compounding risks particularly acute.
Pharmaceutical manufacturers produce GLP-1 peptides through multi-step synthesis with extensive purification and testing. Each production batch undergoes sterility testing, endotoxin analysis, potency assays, purity verification, and stability testing. These processes occur in FDA-inspected facilities following Current Good Manufacturing Practices with multiple quality control checkpoints.
Compounding pharmacies operate under less stringent requirements.
They prepare medications in smaller batches, perform limited testing, and lack the analytical equipment for comprehensive peptide characterization. Most compounding facilities cannot verify peptide identity through mass spectrometry, quantify impurities through HPLC, or measure endotoxin levels accurately.
The raw materials compounding pharmacies use raise additional concerns.
Where do they source retatrutide for compounding? The peptide is not available through pharmaceutical wholesalers because it lacks FDA approval.
This forces compounding pharmacies to purchase from research chemical suppliers, the same unregulated market supplying underground users. The quality variability affecting research chemicals therefore affects compounded products equally.
Sterility assurance presents challenges.
Peptide compounding requires aseptic technique, appropriate facilities, and rigorous environmental monitoring. Not all compounding pharmacies maintain cleanrooms or perform regular sterility testing. Injectable peptides contaminated with bacteria or endotoxins cause serious infections and inflammatory reactions.
Stability testing rarely occurs. Peptides degrade over time, particularly in solution. Pharmaceutical retatrutide includes excipients and preservatives optimized through stability studies to maintain potency throughout labeled shelf life. Compounded versions may lack proper stabilizers or use inadequate preservatives, leading to rapid degradation and unpredictable potency.
Dosing accuracy depends on proper compounding calculations and technique. Errors in reconstitution concentrations, vial filling volumes, or strength calculations lead to underdosing or overdosing. Without analytical verification, neither the pharmacy nor the patient knows if the prescribed dose matches actual vial contents.
Financial and legal risks for patients
Patients receiving retatrutide from compounding pharmacies face financial exploitation and legal complications beyond health risks. Understanding these dimensions helps evaluate whether such arrangements make sense even if tempting.
Pricing for compounded retatrutide typically ranges from $400-800 monthly, presented as significantly cheaper than eventual pharmaceutical pricing. However, patients have no assurance they receive genuine retatrutide at labeled amounts. They may pay $500 for a month of contaminated, underdosed, or completely incorrect medication with no recourse for refund or compensation.
Insurance coverage for compounded retatrutide is essentially nonexistent. Because the compounding violates federal law, insurance companies will not reimburse costs. Patients pay entirely out-of-pocket with no assistance from health plans. This creates perverse economics where illegal products cost more over time than waiting for legitimate FDA-approved medication with insurance coverage.
Medical liability becomes murky. If compounded retatrutide causes adverse effects, who bears responsibility? The prescribing physician may claim they ordered legitimate treatment. The compounding pharmacy operates outside legal bounds. The patient may struggle to identify culpable parties or recover damages for harm caused by illegal products.
Legal risks for patients remain unclear. Receiving prescription medication, even illegally compounded versions, differs from purchasing research chemicals without prescription. Most enforcement targets pharmacies and physicians rather than patients. However, no absolute guarantee protects patients from legal entanglement if authorities investigate prescribing physicians or compounding operations.
Record-keeping poses problems. Medical records documenting illegal medication use may create issues for future healthcare, life insurance applications, or other contexts where comprehensive health histories matter. Patients should consider whether accepting off-book retatrutide prescriptions creates documentation they would rather avoid.
Practical alternatives while waiting for approval
For individuals seeking weight loss benefits similar to retatrutide but requiring solutions available today through legitimate channels, several FDA-approved medications provide proven efficacy. These alternatives lack retatrutide's triple-agonist mechanism but offer substantial weight loss with legal prescriptions and insurance coverage.
Semaglutide represents the most established GLP-1 option. Marketed as Wegovy for weight loss and Ozempic for diabetes, semaglutide produces average weight loss of 15-17% in clinical trials. The medication has been available since 2021, with extensive real-world experience confirming both efficacy and safety profile. Semaglutide dosing follows clear protocols with established titration schedules.
Tirzepatide, the newest approved option before retatrutide, offers dual GIP/GLP-1 agonism. Sold as Zepbound for obesity and Mounjaro for diabetes, tirzepatide achieves 20-22% average weight loss, approaching retatrutide's results. The medication received FDA approval in 2023. Tirzepatide dosing protocols provide detailed guidance for optimal results.
Comparing semaglutide vs tirzepatide helps determine which medication best suits individual circumstances. Tirzepatide produces slightly greater weight loss on average but costs more. Semaglutide has longer track record with more extensive safety data. Both require weekly injections and cause similar side effect profiles.
Insurance coverage varies significantly between medications. Some plans cover Wegovy or Zepbound for obesity treatment while others only approve diabetes indications. Prior authorization requirements, step therapy mandates, and BMI thresholds differ by insurer. Working with prescribing physicians to navigate insurance processes maximizes coverage chances.
Patient assistance programs from Novo Nordisk and Eli Lilly provide cost support for qualifying individuals. These programs offer substantial discounts or free medication for patients meeting income thresholds or lacking insurance coverage. Applications require documentation but can reduce monthly costs from $1000+ to $25-100.
Telehealth prescribing services like Ro, Calibrate, and Found have emerged specifically for weight loss medication access. These platforms connect patients with physicians, handle insurance processes, and coordinate medication delivery. While adding service fees, they simplify obtaining prescriptions for individuals whose primary care physicians hesitate to prescribe weight loss medications.
Research peptide alternatives
For researchers interested in weight loss mechanisms beyond FDA-approved medications, several research peptides offer distinct approaches with varying evidence bases and safety profiles. These compounds remain investigational with research-use-only status, carrying the same quality and legal considerations as retatrutide.
Semaglutide and tirzepatide both exist in research chemical markets for laboratory use. Quality testing shows better consistency for these established peptides versus newer compounds like retatrutide. More vendors have standardized synthesis protocols. Testing services report higher average purity and fewer contamination issues. The research use considerations still apply fully.
AOD-9604, a modified fragment of human growth hormone, targets fat metabolism through different mechanisms than GLP-1 agonists. Research suggests lipolytic effects with minimal impact on insulin sensitivity. The peptide has undergone human trials with generally favorable safety profiles, though FDA approval for clinical use remains absent.
5-Amino-1MQ represents a newer research direction, targeting nicotinamide N-methyltransferase inhibition to increase NAD+ levels and metabolic rate. Early research shows promise but human data remains extremely limited. Quality testing for this compound reveals significant vendor variability and contamination concerns exceeding even retatrutide problems.
CJC-1295 and ipamorelin combine as growth hormone secretagogues promoting natural GH release. While not directly targeting weight loss, increased growth hormone levels may improve body composition. These peptides have longer research history than newer compounds but still lack FDA approval. Understanding CJC-1295 protocols requires comprehensive review.
Stacking multiple peptides amplifies both potential benefits and risks. Some researchers combine GLP-1 agonists with growth hormone peptides or metabolic modulators. These combinations lack clinical trial data. Interaction effects remain unknown. Side effect risks compound. The peptide stacking calculator helps plan combinations but cannot predict safety outcomes.
Lifestyle optimization strategies
While awaiting retatrutide availability or using current alternatives, optimizing fundamental health behaviors produces meaningful results and creates better foundation for future peptide use. These strategies cost nothing, carry no legal risks, and provide benefits regardless of eventual medication use.
Protein intake optimization supports both weight loss and muscle preservation. Research consistently shows high-protein diets of 1.6-2.2g per kilogram body weight improve satiety, thermogenesis, and body composition during caloric restriction. This macronutrient approach complements GLP-1 medications by addressing appetite through both pharmaceutical and nutritional mechanisms.
Resistance training preserves lean mass during weight loss. GLP-1 medications including retatrutide cause total body weight reduction with some unavoidable muscle loss. Progressive resistance training minimizes muscle loss and maintains metabolic rate. Studies show individuals combining GLP-1 treatment with resistance training maintain significantly more lean mass than those using medication alone.
Sleep optimization improves metabolic health and weight management outcomes. Poor sleep increases ghrelin, decreases leptin, impairs glucose metabolism, and drives appetite for calorie-dense foods. Prioritizing 7-9 hours quality sleep nightly creates metabolic conditions favoring weight loss with or without pharmaceutical support.
Stress management through meditation, breathing exercises, or other techniques reduces cortisol-driven abdominal fat accumulation. Chronic stress impairs weight loss through hormonal dysregulation regardless of caloric deficit. Addressing stress as a health fundamental improves outcomes across all interventions.
Tracking food intake through apps like MyFitnessPal or Cronometer provides objective data about caloric consumption.
Many individuals significantly underestimate intake without tracking. Awareness itself often produces behavioral changes leading to reduced consumption. This practice complements medication use by ensuring adequate protein intake and appropriate caloric deficits.
Cost analysis and financial planning
Understanding the complete financial picture for retatrutide access through various pathways helps make informed decisions about timing, sourcing, and alternatives. Costs vary enormously between legal and illegal options, with quality and safety differences justifying premium pricing for legitimate products.
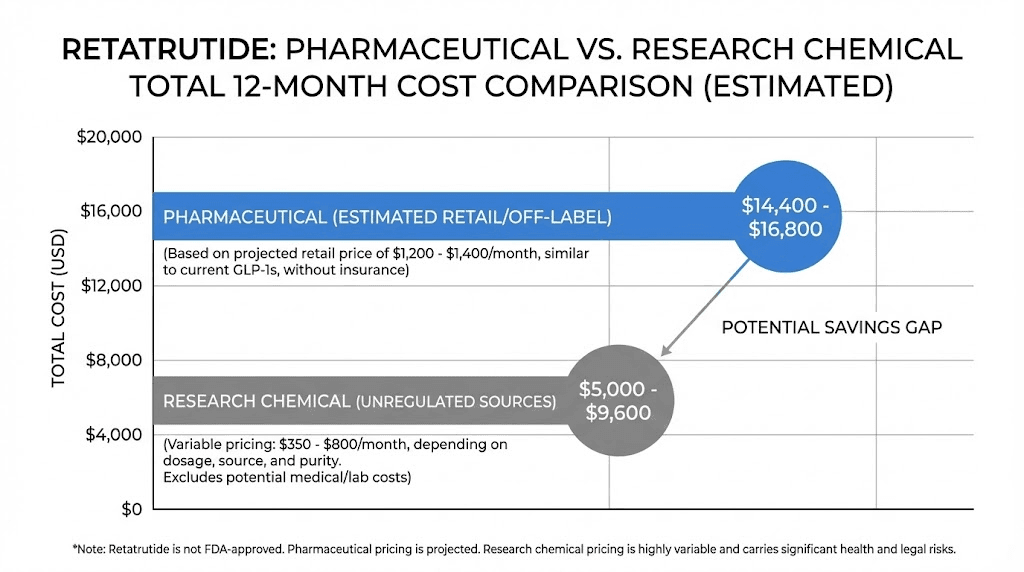
Projected pharmaceutical pricing for FDA-approved retatrutide will likely mirror current GLP-1 medications. Semaglutide (Wegovy) costs approximately $1,300-1,400 monthly list price. Tirzepatide (Zepbound) runs $1,000-1,100 monthly. Retatrutide will probably fall within this range, possibly at the higher end given superior efficacy data. Without insurance, expect $12,000-17,000 annual costs.
Insurance coverage dramatically changes economics. Plans covering obesity treatment typically require $25-100 monthly copays with prior authorization approval. Annual out-of-pocket costs drop to $300-1,200 versus $12,000+ without coverage. However, many insurance plans still exclude obesity medications entirely or impose strict criteria including documented diet/exercise failures and BMI thresholds above 35-40.
Patient assistance programs provide critical support for qualifying individuals. Novo Nordisk's program for Wegovy offers free medication to patients meeting income guidelines or lacking insurance coverage. Eli Lilly provides similar programs for Zepbound. These programs will likely extend to retatrutide post-approval, potentially reducing costs to $0 for eligible patients.
Research chemical pricing ranges from $150-400 per 10mg vial depending on vendor tier and purchase volume. A typical protocol using 8-12mg weekly requires 4-5mg daily, making 10mg vials last 2-2.5 weeks. Monthly costs run $300-800 before factoring reconstitution supplies, testing verification, and injection equipment. Annual research chemical expenses total $3,600-9,600.
Add independent testing costs if verifying product quality. Finnrick Analytics charges $100-200 per sample for comprehensive peptide analysis including purity, quantity, and contamination screening. Testing every shipment adds $1,200-2,400 annually. Skipping verification saves money but risks receiving contaminated, underdosed, or fake products.
Compounding pharmacy pricing advertised at $400-800 monthly appears intermediate between research chemicals and pharmaceutical pricing. However, the illegal nature means no insurance coverage, no patient assistance, and no recourse for poor quality. Annual costs total $4,800-9,600 with significant quality risks and zero legal protections.
Injection supplies add incremental costs across all sourcing methods. Insulin syringes cost $20-30 for 100-count boxes. Alcohol swabs run $5-10 for 100-count packages. Bacteriostatic water for reconstitution costs $10-20 per 30mL bottle. These supplies add roughly $10-20 monthly or $120-240 annually.
The peptide cost calculator helps estimate total expenses across different sourcing scenarios accounting for dosing schedules, vial sizes, and ancillary supplies. Running calculations for 6-12 month protocols reveals total investment requirements before committing to specific vendors or approaches.
Return on investment considerations
Evaluating peptide expenses requires considering health outcomes and long-term value rather than simply comparing prices. The cheapest option often provides worst value when accounting for effectiveness, safety, and sustainability.
Pharmaceutical retatrutide at $1,200 monthly with insurance provides the highest probability of genuine medication, accurate dosing, medical supervision, and sustained results. The annual cost of $14,400 produces documented 24% average weight loss in clinical trials. This translates to $600 per 1% body weight lost for a 200-pound individual, or $7.50 per pound lost.
Research chemicals at $400 monthly provide unknown efficacy due to purity and potency variations. If actual retatrutide content averages 80% of labeled amount and user receives average 19% weight loss instead of 24%, the cost per pound lost increases despite lower sticker price. Poor quality products may produce minimal results, making any cost wasteful.
Time value matters significantly. Using legitimate pharmaceutical retatrutide produces reliable results within 6-12 months. Experimenting with research chemicals of variable quality may require 12-24 months of trial and error, vendor changes, and protocol adjustments to achieve comparable outcomes.
The additional time represents opportunity cost and prolonged health risks from excess weight.
Medical supervision included with pharmaceutical prescriptions provides value beyond medication itself. Regular monitoring identifies side effects early. Professional guidance optimizes protocols. Support for sustainable behavior change improves long-term maintenance. These services contribute meaningfully to success rates even if difficult to quantify financially.
Long-term maintenance costs require planning. Weight regain after medication discontinuation affects many patients. Sustained treatment may require years rather than months of medication. Annual costs extending 3-5 years for weight maintenance change the economic calculation significantly. Insurance coverage becomes even more critical for long-term affordability.
Safety considerations and risk mitigation
Regardless of sourcing pathway, retatrutide carries inherent pharmacological risks beyond product quality concerns. Understanding the medication's safety profile, contraindications, and monitoring requirements enables informed risk assessment and appropriate mitigation strategies.
The most common adverse effects mirror other GLP-1 receptor agonists. Gastrointestinal symptoms including nausea, vomiting, diarrhea, and constipation affect 40-60% of users particularly during dose escalation phases. These effects typically diminish over 2-4 weeks as tolerance develops. Starting at low doses and titrating gradually minimizes severity.
Managing GLP-1 nausea requires strategic approaches including smaller frequent meals, avoiding high-fat foods, staying hydrated, and timing injections appropriately relative to meals. Ginger supplements, vitamin B6, and prescription anti-nausea medications help severe cases. Most users adapt within the first month of treatment.
Gallbladder complications occur at increased rates with rapid weight loss. GLP-1 medications slow gallbladder emptying while weight loss increases cholesterol saturation in bile. This combination elevates gallstone formation risk. Users experiencing right upper quadrant pain, especially after meals, require medical evaluation for possible cholecystitis.
Pancreatitis represents a rare but serious risk. Clinical trials show slightly elevated pancreatitis rates with GLP-1 agonists compared to placebo. Symptoms include severe abdominal pain radiating to the back, nausea, vomiting, and fever. Any suspected pancreatitis requires immediate medical attention and permanent medication discontinuation.
Thyroid concerns emerged from rodent studies showing medullary thyroid carcinoma with GLP-1 medications. Human relevance remains uncertain but package inserts include black box warnings. Personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 absolutely contraindicates retatrutide use.
Hypoglycemia risk increases for users taking insulin or sulfonylureas alongside retatrutide. The peptide's glucagon receptor agonism complicates glucose dynamics compared to pure GLP-1 agonists. Users on diabetes medications require dose adjustments and careful monitoring to prevent dangerous blood sugar drops. Understanding peptide insulin interactions becomes critical.
Cardiovascular effects require consideration.
Clinical trials show generally favorable cardiovascular outcomes with GLP-1 medications, but individual responses vary. Users with cardiovascular disease history require medical supervision to monitor heart rate, blood pressure, and any cardiac symptoms during treatment.
Monitoring protocols and safety labs
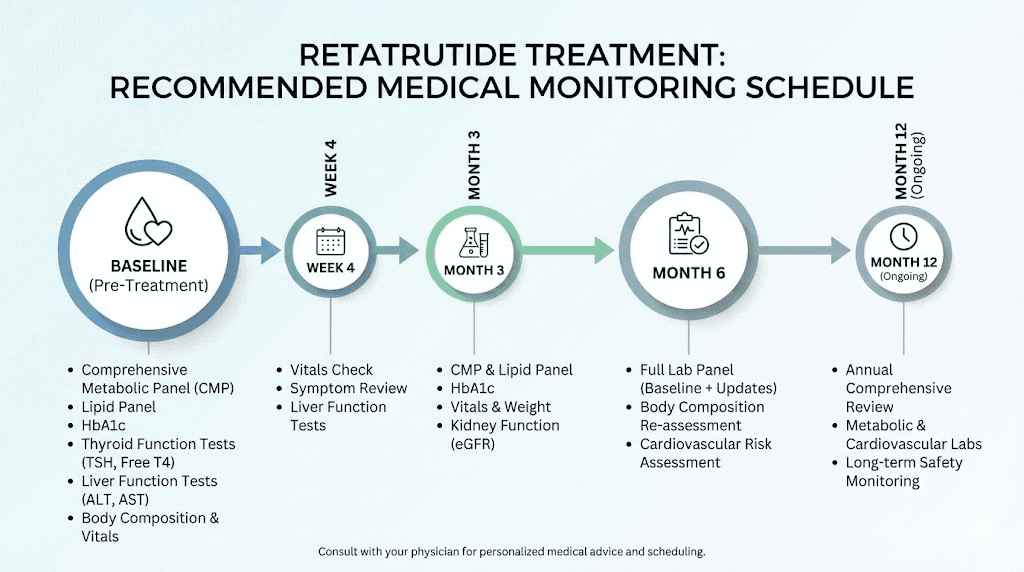
Appropriate medical monitoring during retatrutide use identifies adverse effects early and guides dosing adjustments. Users accessing peptides through legitimate pharmaceutical channels receive standardized monitoring. Those using research chemicals should implement equivalent safety measures independently.
Baseline laboratory testing establishes starting points for comparison. Essential tests include complete blood count, comprehensive metabolic panel, liver function tests, lipid panel, hemoglobin A1c, and thyroid function. These labs identify pre-existing conditions and provide reference values for tracking changes during treatment.
Monthly weight and body composition tracking documents efficacy and helps optimize dosing. Simple home scales suffice for weight monitoring. DEXA scans or bioelectrical impedance analysis every 8-12 weeks quantifies fat loss versus muscle preservation. Disproportionate lean mass loss warrants protocol adjustments including increased protein intake and resistance training.
Quarterly laboratory monitoring tracks metabolic effects and identifies potential complications. Repeating comprehensive metabolic panel detects kidney function changes. Liver function tests identify hepatic stress. Lipid panels document cardiovascular improvements. Hemoglobin A1c tracks glucose metabolism changes relevant for users with insulin resistance or diabetes.
Blood pressure and heart rate require regular home monitoring. Weekly measurements identify trends requiring attention. Significant heart rate increases or blood pressure elevations warrant dose reduction or medical consultation. Home blood pressure monitors cost $30-50 and provide valuable ongoing data.
Symptom tracking through journals or apps captures subjective experiences including energy levels, mood changes, gastrointestinal symptoms, and overall wellbeing. This data helps distinguish transient side effects from persistent problems requiring intervention. Many users find symptom patterns correlate with injection timing and dosing schedules.
The peptide cycle planning approach helps structure monitoring schedules, plan dose adjustments, and determine break periods for users implementing research chemical protocols without medical supervision.
Drug interactions and contraindications
Retatrutide interacts with various medications and medical conditions requiring careful assessment before starting treatment. Some interactions simply require monitoring while others absolutely contraindicate use.
Diabetes medications require dosing adjustments. Insulin doses typically decrease 20-50% when starting retatrutide due to improved glycemic control and reduced food intake. Sulfonylureas similarly require reduction to prevent hypoglycemia. SGLT2 inhibitors and metformin generally continue without adjustment but may require monitoring.
Warfarin levels fluctuate during GLP-1 treatment due to changing vitamin K intake from altered diet composition. Users on warfarin require more frequent INR monitoring with dose adjustments as needed to maintain therapeutic range. Direct oral anticoagulants like rivaroxaban or apixaban show less interaction but still warrant attention.
Oral medications with narrow therapeutic windows may show altered absorption due to delayed gastric emptying. Levothyroxine should be taken separately from retatrutide injection timing. Oral contraceptives maintain effectiveness but some users experience nausea affecting pill tolerance.
Psychiatric medications require consideration. Weight loss itself affects mood and anxiety in complex ways. SSRIs, SNRIs, and other psychiatric medications may require dose adjustments as body weight changes. Close monitoring with prescribing psychiatrists ensures mental health stability during significant weight loss.
Immunosuppressants including tacrolimus and cyclosporine used by transplant recipients may require monitoring due to weight loss affecting drug distribution and metabolism. These users require close coordination between transplant teams and any providers managing weight loss medications.
Alcohol consumption warrants moderation during retatrutide treatment. Many users report reduced alcohol tolerance and increased nausea when drinking. The combination may also increase risk of gastrointestinal symptoms and dehydration. Understanding GLP-1 alcohol interactions guides safer use.
Future availability timeline and market dynamics
Understanding when and how retatrutide reaches legitimate pharmaceutical markets helps plan timing for those waiting for FDA approval rather than pursuing underground sources. Multiple factors influence availability beyond simple trial completion dates.
Phase III trials for obesity treatment continue enrollment and data collection throughout late 2025 and into early 2026. Eli Lilly typically requires 48-72 week trial durations to capture long-term efficacy and safety data. Trials starting in 2024 will not complete until mid-to-late 2026 at the earliest.
FDA submission timing depends on trial results and Lilly's strategic planning.
Assuming positive trial outcomes, the company will likely submit New Drug Applications in late 2026 or early 2027. The submission process itself requires months to compile comprehensive safety data, efficacy analyses, manufacturing information, and proposed labeling.
FDA review processes typically take 10-12 months for standard reviews or 6-8 months for priority reviews. Priority review designation requires the drug to show significant improvement over existing treatments. Retatrutide's superior weight loss compared to tirzepatide may qualify for this expedited pathway, though FDA makes this determination during the review process.
Manufacturing scale-up introduces additional delays. Eli Lilly must build adequate production capacity to meet anticipated demand. GLP-1 medications face persistent supply constraints. Launching retatrutide without sufficient manufacturing capacity risks repeating the semaglutide shortage problems that frustrated patients and providers throughout 2022-2024.
The realistic availability timeline for FDA-approved retatrutide lands in mid-to-late 2027 for the most optimistic scenario. More conservative estimates push availability into 2028. This assumes no unexpected safety signals from ongoing trials and no manufacturing delays complicating launch preparations.
Market positioning relative to existing GLP-1 medications will shape insurance coverage and access. If retatrutide costs significantly more than tirzepatide while providing only marginally better results, insurers may restrict coverage heavily. Conversely, if pricing remains competitive while clearly superior efficacy emerges, retatrutide may become preferred formulary option rapidly.
Compounded medication future
The compounding pharmacy landscape for GLP-1 medications will shift dramatically as FDA addresses ongoing illegal practices. Understanding likely regulatory changes helps anticipate how unofficial access might evolve or disappear.
FDA enforcement actions against compounding pharmacies offering retatrutide and other non-approved peptides will likely intensify. The agency has issued warning letters and statements clarifying legal boundaries. Future enforcement may escalate to product seizures, pharmacy inspections, and civil penalties for continued violations.
State pharmacy boards follow FDA guidance and conduct their own oversight of compounding practices. Pharmacies losing state licenses cannot operate even if federal enforcement moves slowly. The multi-level regulatory structure creates redundant oversight making persistent illegal compounding increasingly risky for pharmacies.
Some compounding pharmacies will pivot to legal alternatives as enforcement pressures increase. Medications properly on the bulk substances list provide compliant revenue opportunities.
The risk-reward calculation for continuing illegal retatrutide compounding becomes less favorable as scrutiny increases and penalties mount.
Patient demand will persist regardless of legal status until FDA approval provides legitimate access. This guaranteed demand makes underground markets unlikely to disappear entirely. Instead, more cautious operators exit while less scrupulous providers continue serving willing customers despite legal risks. Quality and safety concerns intensify as legitimate pharmacies exit the market.
Congressional interest in compounding practices may drive legislative changes. Recent discussions about pharmacy compounding regulations, drug importation, and pharmaceutical pricing create political momentum for comprehensive reform. These changes could either expand legal compounding options or restrict them further depending on legislative outcomes.
International availability varies
FDA approval only affects United States access.
Other countries follow independent regulatory pathways with different timelines and approval criteria. International availability patterns may create medical tourism opportunities or prescription importation channels before US approval.
European Medicines Agency reviews typically run parallel to FDA processes. Major pharmaceutical companies including Eli Lilly typically pursue simultaneous approvals in US and European markets. Retatrutide will likely reach European Union countries within months of US approval rather than years.
Canada's drug approval system closely follows FDA with some independent evaluations. Health Canada typically approves major medications within 6-12 months of FDA approval. Canadian availability may create cross-border prescription filling opportunities for Americans, though importation legality remains complex.
Mexico operates less stringent approval processes for many medications. Some US-approved drugs reach Mexican pharmacies faster than Canadian ones. However, quality control concerns and counterfeit medication risks make Mexican sources problematic for sophisticated peptides requiring cold storage and proper handling.
Asian markets including Japan, South Korea, and Singapore operate independent approval processes. These countries may approve retatrutide on different timelines based on local trial data requirements. Japan typically requires domestic clinical trials before approval, potentially delaying availability years beyond US approval.
Medical tourism for retatrutide seems unlikely before widespread approval.
The medication requires ongoing use rather than one-time procedures. Monthly international travel for prescription refills makes no economic sense. Most medical tourism focuses on procedures, surgeries, or treatments delivered during single visits.
Frequently asked questions
Can I buy retatrutide legally today?
No legal pathway exists to purchase retatrutide outside clinical trial enrollment. The peptide lacks FDA approval and cannot be prescribed. Compounding pharmacies offering retatrutide violate federal law. Research chemical vendors sell for laboratory use only with no legal provision for personal consumption. Anyone offering retatrutide prescriptions through telehealth services operates illegally.
How do research chemical vendors differ from pharmaceutical sources?
Research vendors sell peptides marked "not for human use" targeting academic laboratories and research institutions. These products lack FDA oversight, quality consistency, and therapeutic guarantees. Testing shows purity ranging 62-100% across vendors with contamination risks significantly higher than pharmaceutical products. Pharmaceutical sources provide FDA-approved medications manufactured under strict quality controls with consistent potency and safety profiles. The quality verification process differs completely between categories.
What weight loss can I expect from retatrutide?
Phase II clinical trial data shows 24.2% average body weight reduction over 48 weeks at the 12mg dose. Individual responses vary from 15-35% weight loss depending on starting weight, adherence, diet quality, and physical activity levels. These results exceed semaglutide's 15-17% average and tirzepatide's 20-22% average. However, research chemical retatrutide of uncertain quality may produce substantially different results if purity and potency differ from pharmaceutical versions tested in trials.
How long until I can get a legitimate retatrutide prescription?
The realistic timeline extends to mid-2027 at the earliest, possibly into 2028. Phase III trials complete in late 2025 or early 2026. FDA submission follows, requiring 6-12 months review. Manufacturing scale-up adds further time. The gap between trial completion and pharmacy availability consistently runs 18-24 months for complex medications. Understanding FDA approval timelines explains the specific delays involved.
Are there any legal ways to use research peptides personally?
The legal framework remains murky. Research chemicals sold "not for human use" technically prohibit personal consumption. However, no specific law criminalizes individual possession or use of research peptides. Enforcement targets vendors making therapeutic claims rather than individual users. This creates a gray area where personal use occurs widely despite technical legal prohibitions. Users bear all health risks and have no legal recourse for contaminated or mislabeled products.
What happens if I experience side effects from research chemical peptides?
Medical treatment for adverse effects proceeds normally through emergency departments or primary care physicians. However, explaining you used research chemicals may affect provider responses and medical records. No pharmaceutical company stands behind the product. No formal adverse event reporting system captures problems. Users cannot sue for damages from contaminated products lacking FDA approval. The side effect management strategies for approved medications may not apply to research chemicals of uncertain identity and purity.
Should I wait for FDA approval or use research chemicals now?
This decision depends on individual risk tolerance, financial resources, and health urgency. Waiting for FDA approval provides pharmaceutical-grade medication with medical supervision and legal protections at the cost of 18-24+ months delay. Using research chemicals provides immediate access at lower financial cost but exposes users to contamination risks, legal uncertainties, and no medical oversight. People with serious obesity-related health conditions may reasonably judge that immediate intervention justifies research chemical risks. Others prefer waiting for legitimate options. The risk-benefit framework helps structure this decision systematically.
Can I import retatrutide from other countries?
Importing prescription medications for personal use exists in legal gray zones. FDA technically prohibits importing unapproved drugs but typically tolerates small quantities for personal use. However, retatrutide is not approved in any country currently, eliminating the "foreign prescription drug importation" justification. Customs may seize packages containing unapproved peptides. No legitimate international pharmacies sell retatrutide because no country has approved it yet. Any international source claiming to sell pharmaceutical retatrutide sells research chemicals or counterfeits. Understanding international pharmaceutical sourcing explains the specific legal constraints.
External resources
For researchers serious about navigating peptide access safely, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, quality verification frameworks, vendor testing databases, and a community of thousands who have navigated these exact questions. The platform provides detailed protocols for both legitimate pharmaceutical pathways and research chemical evaluation, safety monitoring frameworks, and realistic timelines for future availability.
In case I don't see you, good afternoon, good evening, and good night. May your peptide sources stay verified, your doses stay accurate, and your research stay safe.