Feb 6, 2026
After 48 weeks, one patient lost 20.1% of their body weight. Another lost 17.5%. Same duration, similar results. Different peptides. Mazdutide targets GLP-1 and glucagon receptors. Tirzepatide targets GLP-1 and GIP receptors. Both are dual agonists designed to produce superior weight loss compared to single-receptor medications like semaglutide, but their different receptor combinations create distinct metabolic effects, side effect profiles, and practical considerations that matter when choosing between them.
The question is not which peptide works. Both work. Clinical trials demonstrate substantial weight loss with both compounds. The question is which works better for specific goals, which is more tolerable, which has better long-term data, and most critically, which you can actually access. Tirzepatide has been available since 2022, with extensive global clinical experience and FDA approval. Mazdutide received approval in China in 2025 but remains unavailable in most markets, with international approval potentially years away.
This creates a practical paradox. Mazdutide shows promising results in head-to-head trials against semaglutide, demonstrating superior glycemic control and comparable or better weight loss. But tirzepatide has already proven itself across multiple large-scale trials involving tens of thousands of participants, with established dosing protocols, known side effect patterns, and widespread clinical use. Choosing between these peptides requires understanding not just what the data shows, but what the data means for real-world use.
This guide examines mazdutide and tirzepatide from every angle that matters.
We will explore their distinct mechanisms of action, compare clinical trial results, analyze side effect profiles, evaluate cost and availability, identify who benefits most from each option, and determine when one clearly outperforms the other. For those navigating the expanding landscape of dual-agonist peptides, SeekPeptides provides comprehensive comparisons, evidence-based protocols, and practical guidance for choosing and using these compounds effectively.
Mechanism comparison: GLP-1/glucagon versus GLP-1/GIP
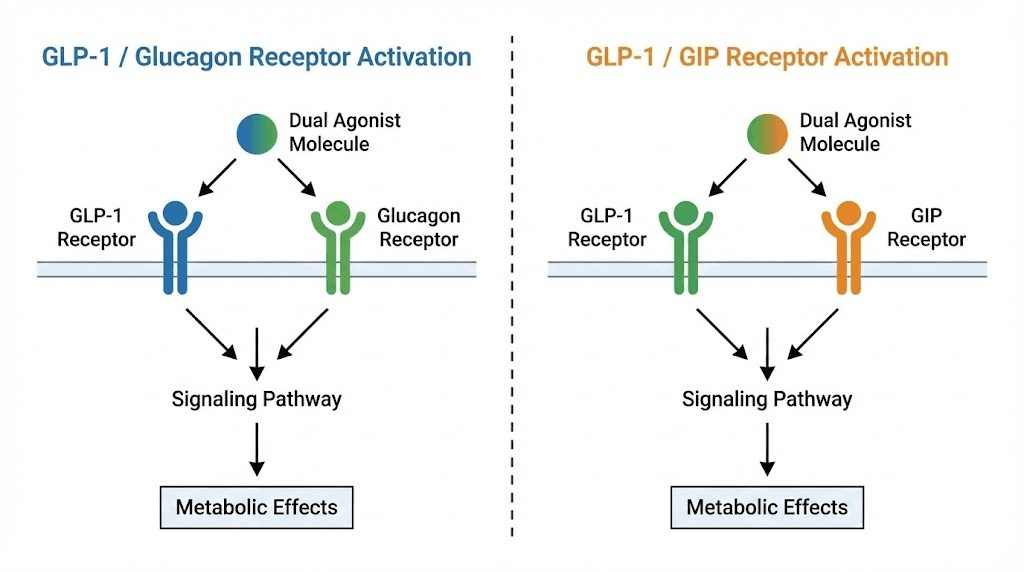
Both mazdutide and tirzepatide activate GLP-1 receptors, producing appetite suppression, delayed gastric emptying, and enhanced insulin secretion. This shared mechanism accounts for much of their weight loss efficacy. The critical difference lies in their second receptor target. Mazdutide activates glucagon receptors. Tirzepatide activates glucose-dependent insulinotropic polypeptide receptors.
These secondary targets are not interchangeable. They produce distinct metabolic effects that influence weight loss magnitude, body composition changes, glycemic control, and side effect patterns. Understanding these differences requires examining what glucagon and GIP actually do in the body, how their activation complements GLP-1 signaling, and why dual agonism produces superior results compared to single GLP-1 agonists.
Mazdutide: GLP-1 and glucagon receptor activation
Glucagon is typically known for raising blood glucose during fasting states. It stimulates hepatic glucose production and glycogenolysis, counteracting hypoglycemia. This seems paradoxical for a weight loss medication designed for people with obesity or type 2 diabetes. Why activate a hormone that raises blood sugar?
The answer lies in dose-dependent effects and receptor distribution. At therapeutic doses, mazdutide activates glucagon receptors in ways that enhance energy expenditure and promote fat oxidation without causing problematic hyperglycemia. Glucagon receptor activation increases metabolic rate by stimulating thermogenesis in brown adipose tissue and enhancing mitochondrial activity in hepatocytes and skeletal muscle.
Studies show that chronic glucagon receptor activation shifts substrate utilization toward fat oxidation. This is metabolically advantageous during weight loss because it preserves lean mass while preferentially mobilizing adipose tissue. The concern about hyperglycemia is mitigated by simultaneous GLP-1 receptor activation, which enhances insulin secretion and suppresses hepatic glucose production through different pathways. The net effect is improved glycemic control despite glucagon receptor stimulation. Understanding peptide receptor pharmacology clarifies how these seemingly contradictory effects balance in practice.
Mazdutide is structurally based on oxyntomodulin, a naturally occurring peptide produced by intestinal L-cells that activates both GLP-1 and glucagon receptors. Oxyntomodulin has been studied for decades as a potential obesity treatment but was limited by poor pharmacokinetics and the need for multiple daily injections. Mazdutide engineers improvements in receptor selectivity, half-life, and dosing convenience, allowing once-weekly administration.
Tirzepatide: GLP-1 and GIP receptor activation
Glucose-dependent insulinotropic polypeptide was historically called gastric inhibitory polypeptide. It is secreted by enteroendocrine K-cells in the proximal small intestine in response to nutrient intake, particularly fats and carbohydrates. Like GLP-1, GIP enhances insulin secretion in a glucose-dependent manner, reducing hypoglycemia risk compared to sulfonylureas or exogenous insulin.
GIP also influences fat metabolism, though its effects are complex and context-dependent. In adipose tissue, GIP promotes lipogenesis and fat storage under normal conditions. This initially seemed problematic for a weight loss medication. However, when GIP receptor activation is combined with GLP-1 agonism in the setting of calorie restriction, the outcome changes. Instead of promoting fat storage, dual agonism appears to enhance lipolysis and improve insulin sensitivity in adipocytes. The mechanisms behind this are not fully understood but likely involve changes in adipocyte function, inflammatory signaling, and nutrient partitioning.
Tirzepatide achieves superior weight loss compared to semaglutide despite both being GLP-1 agonists. Multiple head-to-head trials demonstrate 15-20% body weight reduction with tirzepatide at therapeutic doses versus 10-15% with semaglutide.
This additional efficacy is attributed to GIP receptor activation, though the precise mechanisms remain under investigation. Exploring tirzepatide mechanisms in detail provides additional context.
One hypothesis is that GIP enhances energy expenditure through effects on brown adipose tissue and skeletal muscle metabolism. Another possibility is that GIP reduces the metabolic adaptation that typically occurs during weight loss, where energy expenditure decreases more than predicted by changes in body mass. By maintaining higher metabolic rate during calorie restriction, GIP agonism may prevent the plateaus that limit weight loss with other interventions. Comparing tirzepatide versus semaglutide outcomes illustrates these differences clinically.
Why dual agonism outperforms single-receptor activation
Single GLP-1 agonists like semaglutide produce substantial weight loss, typically 10-15% body weight reduction over 6-12 months. This is clinically meaningful and superior to most other weight loss interventions. However, dual agonists consistently outperform them. The question is why.
One factor is additive effects. Activating two receptors produces overlapping but non-identical metabolic changes that sum to greater total impact. GLP-1 activation primarily reduces appetite and food intake. Adding glucagon or GIP receptor activation adds mechanisms that enhance energy expenditure, improve substrate utilization, and modify fat cell function. The combination produces weight loss through multiple complementary pathways rather than relying solely on reduced calorie intake.
Another factor is metabolic compensation. The body defends against weight loss through mechanisms that increase hunger, reduce energy expenditure, and enhance nutrient absorption efficiency. Single-receptor activation triggers these compensatory responses. Dual agonism may blunt some compensatory mechanisms by targeting multiple points in energy balance regulation. For example, if GLP-1 activation reduces appetite but also triggers compensatory reductions in metabolic rate, adding glucagon receptor activation that increases energy expenditure counteracts this adaptation. Understanding metabolic adaptation during weight loss explains why this matters.
A third factor is improved adherence. If dual agonists produce faster or more noticeable results, patients may maintain adherence better compared to slower-acting single agonists. Psychological factors influence real-world outcomes even when pharmacological mechanisms are superior. Seeing rapid initial results reinforces medication-taking behavior, while slow or modest results can lead to discontinuation before maximum benefit is achieved.
Clinical trial results: head-to-head data
Comparing mazdutide and tirzepatide requires examining their clinical trial results. Direct head-to-head trials between these two peptides do not yet exist. However, both have been compared to semaglutide and placebo in separate trials, allowing indirect comparisons. Additionally, mazdutide has been tested in head-to-head trials against semaglutide, providing insight into how it performs against the most widely used GLP-1 agonist.
Mazdutide phase 3 results in Chinese adults
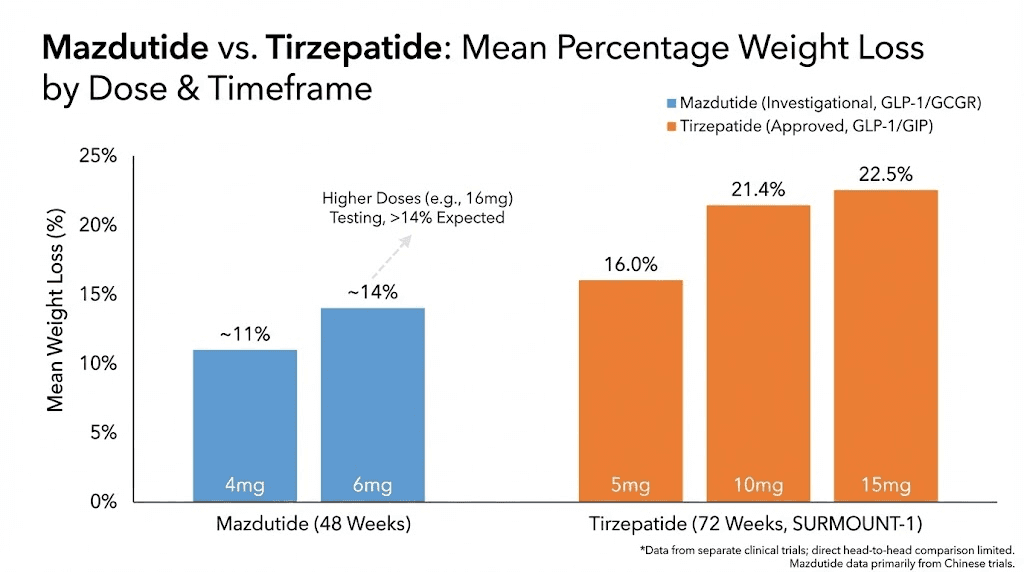
The GLORY-2 trial examined mazdutide in Chinese adults with obesity or overweight. This phase 3 randomized controlled trial enrolled participants with BMI over 24 kg/m² (the obesity threshold in Asian populations) or BMI over 28 kg/m² with comorbidities. Participants received either 4 mg mazdutide, 6 mg mazdutide, or placebo once weekly for 48 weeks. Results demonstrated dose-dependent weight loss. At week 48, participants receiving 4 mg mazdutide lost an average of 11.00% of body weight. Those receiving 6 mg lost 14.01%. Placebo participants gained 0.30%.
The proportion achieving clinically significant weight loss also increased with dose. In the 4 mg group, 35.7% achieved at least 15% weight reduction. In the 6 mg group, 49.5% reached this threshold. Only 2.0% of placebo participants achieved 15% weight loss. These results establish mazdutide as an effective weight loss medication, though the 14% average weight loss at 6 mg is lower than what tirzepatide achieves at comparable timeframes. Detailed clinical trial interpretation helps contextualize these numbers.
Higher doses are under investigation.
A phase 1 high-dose trial tested mazdutide at 9 mg and 10 mg weekly. At week 20, participants receiving 9 mg lost an average of 20.0% body weight. Those receiving 10 mg lost 21.0%. These results approach or match tirzepatide efficacy, suggesting that higher mazdutide doses may close the efficacy gap. Weight loss of 15% or greater was achieved by 66.7% of participants at 9 mg and 75.0% at 10 mg, indicating robust responses across most participants rather than outlier-driven averages.
Glycemic control also improved substantially. HbA1C reductions ranged from 1.2% to 1.8% depending on dose and baseline glycemia. Fasting glucose decreased by 20-30 mg/dL on average. Lipid profiles improved, with reductions in triglycerides and LDL cholesterol and increases in HDL cholesterol. Blood pressure decreased by 5-10 mmHg systolic and 3-5 mmHg diastolic. These cardiometabolic improvements are consistent with significant weight loss and suggest benefits beyond cosmetic weight reduction. Strategies for optimizing metabolic markers enhance these effects.
Mazdutide versus semaglutide head-to-head trial
The DREAMS-3 trial directly compared mazdutide to semaglutide in patients with type 2 diabetes. This is the most informative comparison available because it eliminates confounding variables present in indirect comparisons across different trials. Participants were randomized to receive either mazdutide 6 mg weekly or semaglutide 1 mg weekly for 24 weeks.
Results showed superior glycemic control with mazdutide. HbA1C reduction was significantly greater with mazdutide compared to semaglutide. Weight loss also favored mazdutide, with mean percentage weight reduction of 10.29% compared to 6.00% with semaglutide. This 4.3 percentage point difference is clinically meaningful and statistically significant. For a 100 kg individual, this represents an additional 4.3 kg of weight loss with mazdutide compared to semaglutide over 24 weeks.
These results position mazdutide as superior to semaglutide for both glycemic control and weight loss. However, the trial duration was only 24 weeks, and longer-term data may show different patterns. Additionally, the trial was conducted in patients with type 2 diabetes rather than individuals with obesity alone, which may influence generalizability. Nonetheless, demonstrating superiority to semaglutide, the current gold standard GLP-1 agonist, is a significant achievement. Comparing semaglutide dosing protocols with mazdutide provides additional perspective.
Adverse events were comparable between groups, with gastrointestinal side effects being the most common in both. This suggests that mazdutide does not sacrifice tolerability to achieve greater efficacy, though individual responses vary.
Tirzepatide SURMOUNT and SURPASS trial results
Tirzepatide has been studied extensively in the SURMOUNT program for weight management and SURPASS program for type 2 diabetes. These trials are among the largest peptide weight loss studies ever conducted, with thousands of participants across multiple continents and diverse populations.
SURMOUNT-1 enrolled adults with obesity or overweight without diabetes. Participants received tirzepatide 5 mg, 10 mg, or 15 mg once weekly or placebo for 72 weeks. Results at 72 weeks showed average weight loss of 15.0% with 5 mg, 19.5% with 10 mg, and 20.9% with 15 mg. Placebo participants lost 3.1%. The 15 mg dose produced average weight loss exceeding 20%, approaching surgical weight loss results without surgery. Proportion achieving 15% or greater weight loss was 85% at 10 mg and 91% at 15 mg, indicating that the vast majority of participants experienced substantial benefit. Exploring tirzepatide dosing strategies helps optimize outcomes.
SURMOUNT-2 focused on participants with obesity and type 2 diabetes. Average weight loss at 72 weeks was 12.8% with 10 mg and 14.7% with 15 mg tirzepatide, compared to 3.2% with placebo. Weight loss was slightly lower in diabetic participants compared to non-diabetic participants in SURMOUNT-1, consistent with metabolic differences and potentially greater insulin resistance in diabetic populations. However, glycemic control improved markedly, with HbA1C reductions of 2.0-2.5% from baselines typically around 8.0-8.5%.
SURPASS trials examined tirzepatide in type 2 diabetes across various comparators, including placebo, semaglutide, insulin degludec, and insulin glargine. SURPASS-2 directly compared tirzepatide to semaglutide 1 mg weekly. At 40 weeks, tirzepatide 5 mg, 10 mg, and 15 mg produced greater HbA1C reductions and greater weight loss than semaglutide 1 mg. Weight loss with tirzepatide 15 mg was 11.2 kg compared to 5.7 kg with semaglutide, nearly double. This establishes tirzepatide as superior to semaglutide for both weight loss and glycemic control in diabetic populations. Detailed tirzepatide versus semaglutide comparisons explore this further.
Cardiovascular outcomes are under investigation in the SURPASS-CVOT trial, which will determine whether tirzepatide reduces major adverse cardiovascular events in high-risk patients. Semaglutide has already demonstrated cardiovascular benefits in the SUSTAIN and SELECT trials, giving it an established safety profile for cardiovascular risk reduction. Tirzepatide is expected to show similar or better results, but until SURPASS-CVOT is completed, semaglutide maintains an advantage in this domain.
Indirect comparison: mazdutide versus tirzepatide
No direct head-to-head trial comparing mazdutide and tirzepatide exists. Indirect comparison across trials suggests tirzepatide produces greater weight loss at currently approved doses. Tirzepatide 15 mg produces approximately 20-21% weight loss at 48-72 weeks. Mazdutide 6 mg produces approximately 14% weight loss at 48 weeks. However, higher mazdutide doses (9-10 mg) show 20-21% weight loss at 20 weeks, suggesting that dose-escalation may equalize efficacy.
The critical question is whether mazdutide at higher doses maintains this efficacy over longer durations. Weight loss typically plateaus after 6-12 months, and early rapid loss does not always predict sustained long-term results. Tirzepatide has demonstrated sustained weight loss through 72 weeks with minimal regain, establishing its durability. Mazdutide needs longer-term data at higher doses to demonstrate comparable sustainability. Understanding long-term weight loss sustainability clarifies what these patterns mean.
Glycemic control appears similar between the two peptides. Both produce HbA1C reductions of 1.5-2.5% in diabetic populations, which is clinically significant and superior to most oral antidiabetic agents. Mazdutide showed superior glycemic control compared to semaglutide in DREAMS-3, and tirzepatide showed similar superiority in SURPASS-2. This suggests both dual agonists outperform semaglutide for diabetes management.
Side effect profiles appear comparable based on available data. Both peptides cause primarily gastrointestinal side effects, with nausea, vomiting, diarrhea, and constipation being most common. Incidence and severity are dose-dependent for both compounds. Strategies for managing GLP-1-related nausea apply to both peptides.
Side effect profiles and tolerability
Both mazdutide and tirzepatide share a common side effect profile dominated by gastrointestinal symptoms. This is expected given their shared GLP-1 receptor agonism, which slows gastric emptying and alters gut motility. However, subtle differences exist based on their secondary receptor targets and clinical trial safety data.
Gastrointestinal side effects: nausea, vomiting, diarrhea
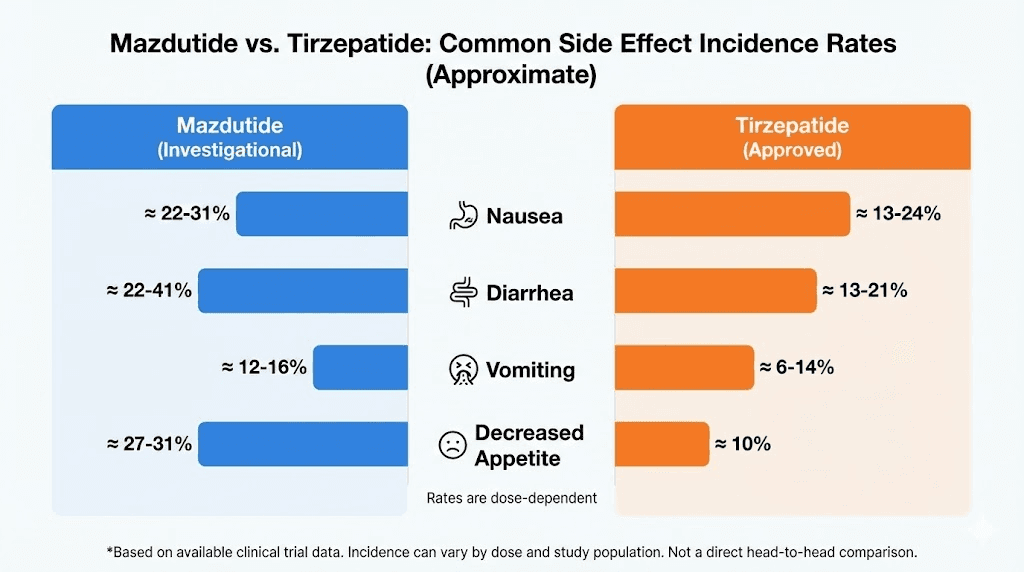
Nausea is the most frequently reported side effect for both peptides. Incidence ranges from 20-40% depending on dose, with higher doses producing higher incidence. Most nausea is mild to moderate and occurs during dose escalation. It typically diminishes after 2-4 weeks as the body adapts to slowed gastric emptying.
Vomiting occurs less frequently than nausea but is more distressing when present. Incidence is typically 5-15% across doses. Severe or persistent vomiting can lead to dehydration, electrolyte imbalances, and treatment discontinuation. Managing vomiting requires dose reduction, slower titration, dietary modifications, and occasionally antiemetic medications. Practical side effect management strategies reduce discontinuation rates.
Diarrhea affects 15-25% of users. Mechanisms include altered gut motility, changes in intestinal fluid secretion, and microbiome shifts. Diarrhea is usually self-limiting but can be problematic if severe or persistent. Increasing fiber intake gradually, staying hydrated, and using probiotics may help. Constipation is paradoxically common as well, affecting 10-20% of users. This reflects slowed gut motility taken to an extreme. Both diarrhea and constipation can occur in the same individual at different times during treatment.
Mazdutide trial data suggests gastrointestinal side effects are mostly mild to moderate in severity. Discontinuation rates due to GI intolerance were 3-7% across doses, comparable to tirzepatide. Tirzepatide trials report similar tolerability, with 4-8% discontinuation due to adverse events, predominantly gastrointestinal. This suggests both peptides have acceptable tolerability profiles for most users, though individual responses vary widely. Comparing tirzepatide side effect patterns with mazdutide reveals similarities.
Metabolic and endocrine side effects
Hypoglycemia risk is low with both peptides when used as monotherapy because their effects on insulin secretion are glucose-dependent. Insulin is only released when blood glucose is elevated, reducing hypoglycemia risk compared to sulfonylureas or exogenous insulin. However, when combined with insulin or sulfonylureas, hypoglycemia risk increases. Dose reduction of concomitant medications is typically required when initiating GLP-1 or dual-agonist therapy.
Thyroid concerns exist for GLP-1 agonists due to findings in rodent studies showing C-cell hyperplasia and medullary thyroid carcinoma. These findings have not been replicated in humans, and epidemiological data from millions of users suggests no increased thyroid cancer risk. Nonetheless, mazdutide and tirzepatide carry warnings about medullary thyroid carcinoma and are contraindicated in patients with personal or family history of this rare cancer or multiple endocrine neoplasia syndrome type 2. Monitoring thyroid safety with GLP-1 agonists provides reassurance.
Gallbladder disease incidence increases with rapid weight loss regardless of method. GLP-1 agonists, including dual agonists, are associated with increased risk of cholelithiasis and cholecystitis. Mechanisms include reduced gallbladder contractility and supersaturation of bile with cholesterol during rapid fat mobilization. Risk is dose-dependent and correlates with weight loss magnitude. Symptoms include right upper quadrant pain, nausea after fatty meals, and jaundice if biliary obstruction occurs. Ultrasound confirms diagnosis, and treatment ranges from watchful waiting to cholecystectomy depending on severity.
Pancreatitis is a rare but serious adverse event reported with GLP-1 agonists. Incidence is approximately 0.1-0.3%, only slightly higher than background population rates. Causality remains debated because obesity itself increases pancreatitis risk. Symptoms include severe epigastric pain radiating to the back, nausea, vomiting, and elevated pancreatic enzymes. Patients with history of pancreatitis should use these peptides cautiously if at all. Understanding peptide contraindications and warnings prevents serious complications.
Cardiovascular and renal effects
Heart rate increases of 2-5 beats per minute are commonly observed with GLP-1 agonists, including tirzepatide. This is thought to result from increased sympathetic tone or direct effects on cardiac pacemaker cells. The clinical significance is uncertain. Most patients tolerate this without symptoms, but those with underlying arrhythmias or cardiovascular disease should be monitored. Mazdutide data on heart rate effects is limited but likely similar given shared GLP-1 agonism.
Blood pressure typically decreases with both peptides, likely due to weight loss and improved insulin sensitivity rather than direct vascular effects. Reductions of 5-10 mmHg systolic and 3-5 mmHg diastolic are common. This is beneficial for most patients but may require adjustment of antihypertensive medications to prevent hypotension. Monitoring blood pressure changes during weight loss guides medication adjustments.
Renal function generally improves with GLP-1 agonists in diabetic patients, likely due to improved glycemic control and reduced intraglomerular pressure. Semaglutide has demonstrated reduction in progression of diabetic kidney disease. Tirzepatide is expected to show similar benefits, and mazdutide may as well, though specific renal outcome data is pending.
Injection site reactions and administration issues
Both peptides are administered via subcutaneous injection once weekly. Injection site reactions including redness, swelling, itching, or bruising occur in 2-5% of users. These are typically mild and resolve without intervention. Rotating injection sites between abdomen, thigh, and upper arm reduces reaction incidence. Proper injection technique minimizes discomfort and reactions.
Needle phobia is a barrier for some patients. Oral formulations are under development for various GLP-1 agonists, including oral semaglutide which is already available. Whether oral mazdutide or tirzepatide formulations will emerge remains to be seen. Comparing injectable versus oral peptides helps patients weigh convenience versus efficacy.
Availability, cost, and access considerations
Clinical efficacy matters little if a medication is unavailable or unaffordable. Mazdutide and tirzepatide differ dramatically in accessibility, creating a practical decision point that overrides theoretical efficacy comparisons for most patients.
Tirzepatide: widely available with established access
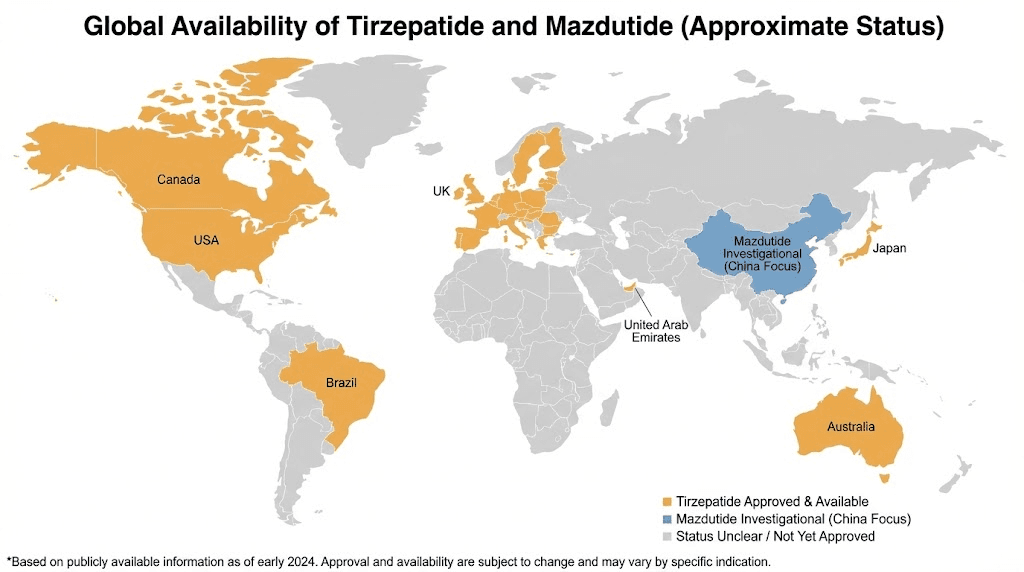
Tirzepatide is marketed as Mounjaro for type 2 diabetes and Zepbound for weight management. It received FDA approval in 2022 for diabetes and 2023 for obesity. It is available in the United States, European Union, United Kingdom, Canada, Australia, and many other countries. Availability continues expanding as regulatory approvals are granted in additional markets.
Dosing is standardized with prefilled autoinjector pens containing 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, or 15 mg doses. Patients start at 2.5 mg weekly for 4 weeks, then escalate by 2.5 mg increments every 4 weeks as tolerated until reaching maintenance dose, typically 10 mg or 15 mg. This titration schedule balances efficacy with tolerability by allowing gradual adaptation to GI side effects. Detailed tirzepatide dosage protocols guide optimization.
Insurance coverage varies. Many plans cover Mounjaro for diabetes but not Zepbound for weight management, citing obesity treatment as cosmetic rather than medical. Prior authorization requirements are common, requiring documentation of BMI over 30 kg/m² or over 27 kg/m² with comorbidities, failed attempts at diet and exercise, and sometimes trial of other weight loss medications. Denials are frequent, and appeals can take months.
Cost without insurance is approximately $1,000-1,200 per month for brand-name tirzepatide. Manufacturer savings cards can reduce copays to $25-50 per month for commercially insured patients, though restrictions apply. Medicare and Medicaid patients typically cannot use manufacturer coupons. Compounded tirzepatide from licensed pharmacies costs $300-600 per month depending on dose and source, offering a more affordable option with some tradeoffs in quality assurance and regulatory oversight. Evaluating peptide cost comparisons informs financial planning.
Mazdutide: limited availability, uncertain timeline
Mazdutide received approval in China in 2025 for treatment of obesity and type 2 diabetes. It is marketed there under the brand name Lirasen. However, it has not been approved in the United States, European Union, or most other markets. Regulatory submissions are in progress, but approval timelines are uncertain, likely 2026-2028 at earliest for major markets.
This creates a significant access barrier for patients outside China. Even once approved, insurance coverage will depend on cost-effectiveness analyses, formulary negotiations, and clinical positioning relative to tirzepatide and semaglutide. If mazdutide is not demonstrably superior to tirzepatide but costs similarly, insurers may not cover it as a preferred option.
Pricing is not yet established for markets outside China.
If priced competitively with tirzepatide or lower, it could gain market share. If priced higher without clear superiority, uptake will be limited. Cost in China is approximately $120-150 per month, substantially lower than tirzepatide in Western markets. Whether this pricing will translate internationally depends on manufacturing costs, regulatory expenses, and market competition. Understanding peptide pricing factors provides context for future cost predictions.
Compounded mazdutide is not currently available from research chemical suppliers or compounding pharmacies in the way that semaglutide and tirzepatide are. This may change if demand increases and synthesis becomes feasible, but for now, access is limited to approved markets.
Practical access strategies
For patients in markets where both peptides are available, tirzepatide is the practical choice due to established availability, known dosing protocols, extensive clinical experience, and insurance coverage pathways. Mazdutide may become an alternative if tirzepatide is not tolerated, not effective, or not covered by insurance, but only once it receives regulatory approval.
For patients in China, mazdutide is accessible and may be preferred if cost is a primary concern or if head-to-head data suggests equivalent or superior efficacy for individual circumstances. Tirzepatide is also available in China, so direct comparison and individualized choice are possible.
For patients unable to access either medication through prescription channels, compounded tirzepatide is more readily available than mazdutide.
Research chemical suppliers may offer tirzepatide, though quality, purity, and legality vary widely. Using non-prescription sources carries risks including contamination, incorrect dosing, lack of medical supervision, and legal consequences. Patients considering this route should understand the tradeoffs. Guidance on research versus pharmaceutical peptides clarifies risks and considerations.
Who benefits most from each peptide
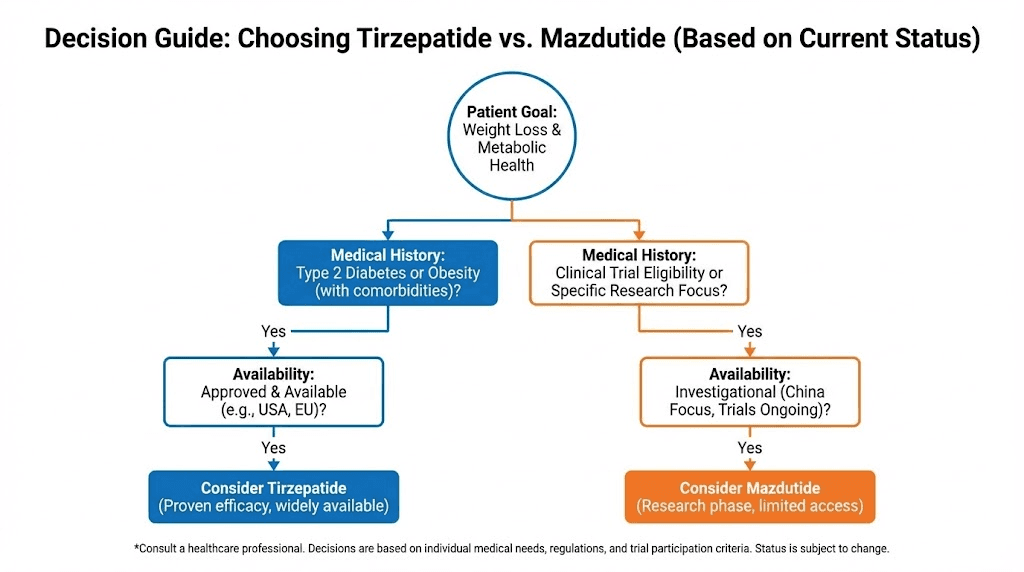
Choosing between mazdutide and tirzepatide depends on individual goals, medical history, access, cost, and preferences. Neither peptide is universally superior. Context determines the better choice.
When tirzepatide is the better choice
Tirzepatide is preferred for patients seeking maximum weight loss with the most robust clinical evidence. Its 20-21% average weight loss at 15 mg over 72 weeks is the highest among any approved weight loss medication. For individuals with severe obesity (BMI over 40 kg/m²) or those who have failed other interventions, tirzepatide offers the greatest likelihood of achieving substantial weight reduction.
Patients with type 2 diabetes benefit from tirzepatide extensive diabetes trial data. SURPASS trials demonstrate HbA1C reductions of 2.0-2.5% on average, superior to most oral antidiabetic agents and comparable to or better than insulin. For patients struggling with glycemic control despite multiple medications, tirzepatide provides a powerful tool. Understanding tirzepatide for diabetes management guides integration into treatment regimens.
Individuals in markets where mazdutide is unavailable have no choice but tirzepatide if they want a dual-agonist peptide. Given mazdutide limited availability outside China, tirzepatide is the practical default for most global patients.
Patients prioritizing long-term safety data benefit from tirzepatide more extensive clinical experience. Millions of patients have used tirzepatide since 2022, providing real-world safety data beyond what controlled trials capture. Rare adverse events, drug interactions, and long-term outcomes are better characterized for tirzepatide than mazdutide. For risk-averse individuals or those with complex medical histories, this established safety profile is reassuring.
When mazdutide is the better choice
Mazdutide is preferred for patients in China seeking cost-effective weight loss with dual-agonist benefits. At approximately $120-150 per month, it is significantly less expensive than tirzepatide while offering comparable efficacy at higher doses. For patients paying out-of-pocket, cost savings are substantial over time.
Individuals who have tried tirzepatide and experienced intolerable side effects may respond differently to mazdutide despite overlapping mechanisms. While side effect profiles are similar, individual responses vary, and switching between similar medications sometimes improves tolerability. This is not guaranteed, but for patients unable to tolerate tirzepatide, mazdutide represents an alternative worth trying once available. Strategies for switching between GLP-1 peptides reduce transition complications.
Patients interested in glucagon receptor agonism mechanisms may prefer mazdutide for theoretical reasons. If you are particularly interested in enhanced energy expenditure and fat oxidation via glucagon pathways, mazdutide offers this over tirzepatide GIP-based approach. However, practical outcomes matter more than mechanistic preferences, and tirzepatide superior clinical results suggest GIP agonism may be more effective overall.
Researchers and clinicians in China conducting studies on dual-agonist peptides have easier access to mazdutide and may choose it for practical reasons related to local availability and regulatory approval.
When neither is ideal and alternatives are better
Some patients are better served by single GLP-1 agonists like semaglutide. If you need moderate weight loss (10-15% body weight) and cannot tolerate dual-agonist side effects, semaglutide may provide sufficient benefit with better tolerability. Cost is also lower for semaglutide in some markets, especially with manufacturer savings programs. Exploring semaglutide as an alternative is worthwhile.
Patients with contraindications to GLP-1 agonists, including personal or family history of medullary thyroid carcinoma, history of pancreatitis, severe gastroparesis, or pregnancy, should not use either mazdutide or tirzepatide. Alternative weight loss interventions include phentermine/topiramate, naltrexone/bupropion, orlistat, bariatric surgery, or intensive lifestyle modification programs. Understanding weight loss alternatives provides options.
Individuals with minimal weight to lose (BMI under 27 kg/m² without comorbidities) do not meet clinical criteria for pharmacological weight loss treatment. Using potent medications for cosmetic purposes carries risks without proportional benefits. Diet, exercise, and behavioral interventions are more appropriate for modest weight management goals.
Combining mazdutide or tirzepatide with other interventions
Neither mazdutide nor tirzepatide should be used in isolation. Maximum benefit comes from integrating these peptides into comprehensive approaches that include dietary modifications, exercise, sleep optimization, stress management, and other interventions that support metabolic health and sustainable weight loss.
Dietary strategies to enhance dual-agonist effects
Protein intake is critical during weight loss to preserve lean mass. Aim for 1.6-2.2 grams of protein per kilogram of body weight daily, distributed across meals. Dual agonists reduce appetite, making it tempting to skip meals or eat minimally, but inadequate protein accelerates muscle loss. Prioritize lean protein sources like chicken, fish, eggs, Greek yogurt, cottage cheese, legumes, and protein powders. Detailed protein strategies with GLP-1 agonists prevent muscle loss.
High-fiber foods support satiety and gut health. Include vegetables, fruits, legumes, and whole grains in most meals. Fiber slows digestion, prolongs fullness, and feeds beneficial gut bacteria that influence metabolism and inflammation. Target 25-35 grams of fiber daily, increasing gradually to avoid GI discomfort.
Minimize ultra-processed foods high in refined carbohydrates, added sugars, and unhealthy fats. These foods are engineered to override satiety signals and drive overconsumption. Even with dual-agonist appetite suppression, hyperpalatable foods can undermine weight loss. Focus on whole, minimally processed foods that provide nutrients without excessive calories.
Hydration is essential. Dual agonists can blunt thirst perception, and dehydration worsens side effects like constipation, fatigue, and headaches. Drink at least 2-3 liters of water daily, more if exercising or in hot climates. Monitor urine color as a hydration indicator. Pale yellow indicates adequate hydration, while dark yellow suggests need for more fluid. Optimizing hydration during weight loss improves outcomes.
Exercise and physical activity optimization
Resistance training is non-negotiable during rapid weight loss. Dual agonists produce substantial weight loss, but 20-30% of lost weight can be lean mass if resistance training is not included. Muscle loss worsens metabolic rate, functional capacity, and long-term weight maintenance. Train 3-4 times per week, focusing on compound movements like squats, deadlifts, presses, rows, and lunges. Progressive overload, where weight or reps increase over time, stimulates muscle retention and growth. Practical resistance training programs prevent muscle loss.
Cardiovascular exercise supports calorie expenditure and cardiovascular health. Include 150-300 minutes of moderate-intensity activity per week, such as brisk walking, cycling, or swimming. High-intensity interval training can be effective but may be harder to sustain if appetite suppression reduces energy availability. Match exercise intensity to energy levels and recovery capacity.
Non-exercise activity thermogenesis contributes significantly to daily calorie expenditure. Walking more, taking stairs, standing while working, and staying active throughout the day adds several hundred calories of expenditure without formal exercise. This is especially important as formal exercise represents a small fraction of total daily activity for most people. Increasing NEAT for weight loss accelerates progress.
Sleep and stress management
Sleep deprivation undermines weight loss by increasing hunger hormones, reducing satiety hormones, impairing insulin sensitivity, and lowering willpower. Aim for 7-9 hours of quality sleep nightly. Prioritize sleep hygiene with consistent bed and wake times, dark and cool bedroom, limited screen time before bed, and avoidance of caffeine after early afternoon. Sleep optimization is detailed in sleep and metabolic health guides.
Chronic stress elevates cortisol, promotes fat storage, increases appetite, and impairs glucose metabolism. Stress management practices like meditation, deep breathing, yoga, nature exposure, and social connection mitigate these effects. Even 10-15 minutes daily of stress reduction practice improves hormonal balance and supports weight loss efforts.
Stacking with other peptides or medications
Combining dual agonists with other weight loss medications is generally not recommended. Stacking GLP-1 agonists with other appetite suppressants like phentermine increases side effect risk without clear additive benefit. Combining two dual agonists would be redundant and dangerous. Exploring peptide stacking safety prevents harmful combinations.
Some patients combine dual agonists with metformin for additional metabolic benefits. Metformin improves insulin sensitivity, reduces hepatic glucose production, and may have longevity benefits. Combining metformin with tirzepatide or mazdutide is safe and may enhance glycemic control in diabetic patients. Typical dosing is 500-1000 mg twice daily with meals.
Peptides like BPC-157 for gut healing or CJC-1295 with ipamorelin for lean mass preservation are sometimes stacked with dual agonists. BPC-157 may help manage GI side effects, though evidence is limited. Growth hormone secretagogues like CJC-1295 and ipamorelin may counteract muscle loss during calorie restriction, though adding complexity increases cost and risk. Practical peptide stacking strategies guide safe combinations.
Practical protocols for using each peptide
Effective use requires understanding proper dosing, titration schedules, injection technique, monitoring, and adjustment strategies. While both peptides share similarities in administration, subtle differences exist.
Tirzepatide dosing and titration protocol
Start tirzepatide at 2.5 mg once weekly for 4 weeks. This initial dose establishes tolerance and reduces side effect incidence. Many patients experience appetite suppression and mild weight loss even at this starter dose. Take the injection on the same day each week, at any time of day, with or without food. Rotate injection sites between abdomen, thigh, and upper arm to minimize reactions. Detailed injection technique instructions reduce discomfort.
After 4 weeks at 2.5 mg, increase to 5 mg weekly for 4 weeks. This is the first therapeutic dose for weight loss, and many patients see accelerated weight loss at this point. Monitor appetite, weight, and side effects closely during this transition.
After 4 weeks at 5 mg, increase to 7.5 mg weekly for 4 weeks. Continue this stepwise escalation by 2.5 mg every 4 weeks as tolerated, with goal of reaching 10 mg or 15 mg depending on response and tolerability. Some patients achieve adequate weight loss at 7.5 mg or 10 mg and do not require higher doses. Others need 15 mg to maximize effect.
If intolerable side effects occur during escalation, remain at current dose for an additional 4 weeks before attempting to increase, or drop back to the previous dose. Slower titration improves tolerability at the cost of delayed maximum efficacy. Individualize based on patient preference and side effect severity. Managing tirzepatide dose escalation optimizes outcomes.
Mazdutide dosing and titration protocol
Mazdutide dosing protocols are less standardized given its recent approval and limited global availability. Based on clinical trials, a reasonable approach starts at 3 mg once weekly for 4 weeks. This is lower than the therapeutic doses tested in trials but allows initial tolerance assessment.
After 4 weeks at 3 mg, increase to 4 mg weekly for 4 weeks. This corresponds to the lower therapeutic dose from GLORY-2. Monitor response and tolerability.
After 4 weeks at 4 mg, increase to 6 mg weekly, the higher dose from GLORY-2 that produced 14% average weight loss. Maintain 6 mg for at least 8-12 weeks to assess response before considering further escalation.
If weight loss is inadequate after 12 weeks at 6 mg, consider increasing to 9 mg or 10 mg based on phase 1 high-dose trial data showing 20-21% weight loss. These higher doses are not yet approved in most markets, so access may be limited. Safety and tolerability at higher doses appear acceptable based on preliminary data, but long-term experience is limited.
Monitoring and adjustment strategies
Weigh yourself weekly on the same day, at the same time, under similar conditions. Weekly averages provide clearer trends than daily measurements, which fluctuate with hydration, bowel movements, and sodium intake. Expect initial rapid weight loss of 2-4 pounds per week, slowing to 1-2 pounds per week after the first month, then 0.5-1 pound per week as you approach maintenance. Tracking weight loss progress helps identify plateaus early.
Measure waist circumference monthly. This tracks visceral fat reduction, which correlates more strongly with metabolic improvements than total weight. A shrinking waist despite stable weight suggests favorable body composition changes.
Monitor blood glucose if diabetic. Check fasting glucose weekly and HbA1C every 3 months. Hypoglycemia is rare with monotherapy but can occur if combined with insulin or sulfonylureas. Adjust other diabetes medications in consultation with a physician as glycemic control improves.
Track side effects daily for the first 2-3 months. Note severity, duration, and triggers for nausea, vomiting, diarrhea, constipation, or other symptoms. Patterns help identify strategies to minimize discomfort, such as avoiding specific foods, adjusting meal timing, or modifying dose escalation schedule.
Check liver function, kidney function, and lipid panels every 6 months. Weight loss and glycemic improvement typically enhance these markers, but monitoring confirms benefits and detects rare adverse effects. Annual thyroid function testing may be prudent given theoretical thyroid concerns, though risk appears minimal in humans. Comprehensive metabolic health monitoring ensures safety.
Troubleshooting inadequate response
If weight loss is minimal after 12-16 weeks at therapeutic doses, evaluate several factors. First, are you truly adherent? Missing doses, inconsistent timing, or failure to reach therapeutic dose levels reduces efficacy. Second, is calorie intake still excessive? Dual agonists reduce appetite but do not make weight loss automatic. If you are consuming high-calorie processed foods despite reduced appetite, weight loss will be limited. Third, is your metabolism unusually resistant? Some individuals have genetic or metabolic factors that blunt response to GLP-1 agonists. Exploring GLP-1 non-responder phenotypes provides insight.
If adherence and diet are optimized but response remains inadequate, consider switching peptides. If tirzepatide is not effective, mazdutide may work better due to its different secondary mechanism, and vice versa. Alternatively, consider non-peptide weight loss medications or bariatric surgery for individuals with severe obesity unresponsive to pharmacotherapy. Strategies for switching weight loss medications prevent gaps in treatment.
Frequently asked questions
Can I switch from tirzepatide to mazdutide or vice versa?
Yes, switching between dual-agonist peptides is possible. If you are discontinuing one peptide to start another, allow 1-2 weeks washout to minimize overlapping effects and side effects. Start the new peptide at its initial dose rather than attempting to match your previous dose, as potency and receptor affinity differ between compounds. Switching is most relevant for patients who cannot tolerate one peptide or who find one more effective than the other. Practical switching protocols reduce transition complications.
Which peptide has better long-term safety data?
Tirzepatide has substantially more long-term safety data given its earlier approval and wider use. Millions of patients have used tirzepatide since 2022, providing extensive real-world safety experience beyond controlled trials. Mazdutide safety data is limited to clinical trial populations, with real-world experience only beginning in China. For risk-averse patients, tirzepatide more established safety profile is reassuring. Understanding long-term peptide safety informs decision-making.
Do I need to stay on these peptides forever?
Most patients regain weight after discontinuing GLP-1 or dual-agonist peptides if they do not maintain the dietary and lifestyle changes that supported weight loss. Long-term or indefinite use is common for sustained weight maintenance. Some patients use peptides to lose weight, then transition to lower doses or intermittent use for maintenance. This requires careful monitoring and willingness to resume full-dose therapy if regain begins. Strategies for maintaining weight loss after stopping peptides improve long-term success.
Can I use these peptides if I do not have diabetes?
Yes, both mazdutide and tirzepatide are approved for weight management in individuals with obesity or overweight with comorbidities, regardless of diabetes status. Tirzepatide is marketed as Zepbound for non-diabetic weight loss and Mounjaro for diabetes. Mazdutide is approved for both indications in China. You do not need diabetes to benefit from these peptides. Non-diabetic use is covered in tirzepatide for non-diabetic weight loss guides.
Are these peptides safe for women who are pregnant or breastfeeding?
No, GLP-1 and dual-agonist peptides are contraindicated during pregnancy and breastfeeding. Weight loss is inappropriate during pregnancy, and effects on fetal development are unknown. Discontinue these medications at least 2 months before attempting conception to allow clearance from the body. Do not use while breastfeeding due to unknown effects on infant nutrition and development. Safety considerations are detailed in GLP-1 safety and contraindications guides.
What happens if I miss a dose?
If you miss a dose and remember within 4 days, take it as soon as possible and resume your regular weekly schedule. If more than 4 days have passed, skip the missed dose and take the next dose on the regularly scheduled day. Do not double dose to make up for a missed injection. Missing occasional doses reduces efficacy but is not dangerous. Consistent weekly dosing maximizes results. Managing missed doses prevents complications.
Can these peptides be combined with phentermine or other weight loss medications?
Combining dual-agonist peptides with other appetite suppressants like phentermine is generally not recommended due to increased side effect risk without clear additive benefit. Combining with metformin for diabetes management is safe and common. Combining with naltrexone/bupropion, orlistat, or topiramate has not been well-studied and should be approached cautiously under medical supervision. Exploring weight loss medication combinations clarifies risks and benefits.
For those committed to optimizing metabolic health through dual-agonist peptides, SeekPeptides members access comprehensive protocol libraries comparing mazdutide, tirzepatide, semaglutide, and emerging alternatives with detailed efficacy data, side effect management strategies refined through thousands of user experiences, personalized dosing calculators that account for individual factors most generic protocols ignore, and real-time updates as new clinical data emerges. The platform provides the depth of research and practical guidance needed to navigate the complex landscape of weight loss peptides with confidence.
In case I do not see you, good afternoon, good evening, and good night. May your appetite stay controlled, your metabolism stay optimized, and your weight loss stay sustainable.