Feb 5, 2026

Experienced researchers know something beginners do not about retatrutide dosing. The number on the vial matters far less than the schedule behind it. Grab any peptide forum thread, scroll past the first dozen posts, and you will find the same pattern repeating itself. Someone starts at 12mg because they read a headline about maximum weight loss. They get hit with nausea so severe they abandon the protocol entirely.
Meanwhile, the researcher who started at 1mg, titrated slowly over sixteen weeks, and respected every step of the escalation ladder is now twenty, sometimes twenty-four percent lighter, with side effects that barely registered. That gap between reckless dosing and strategic titration is where real results live.
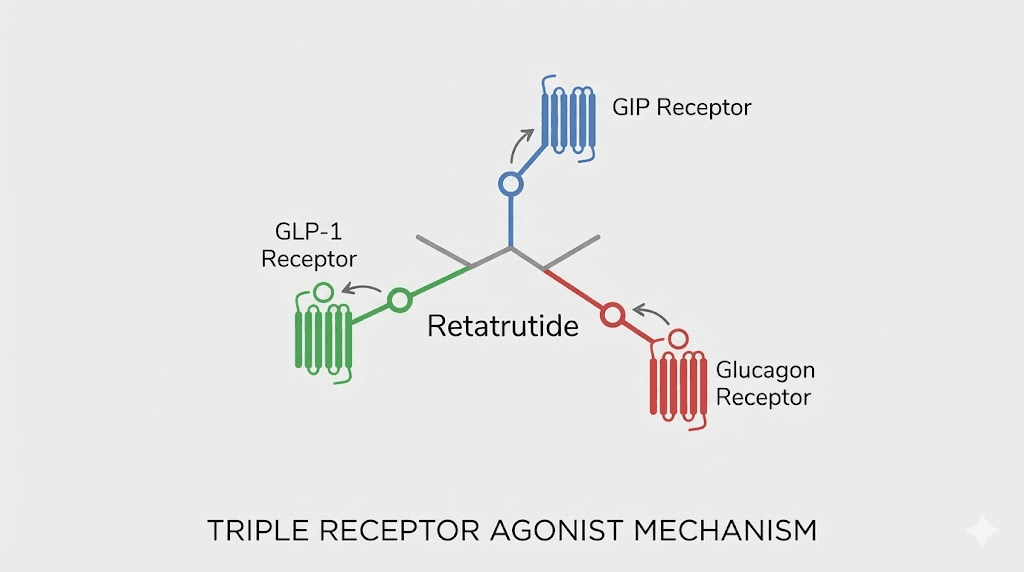
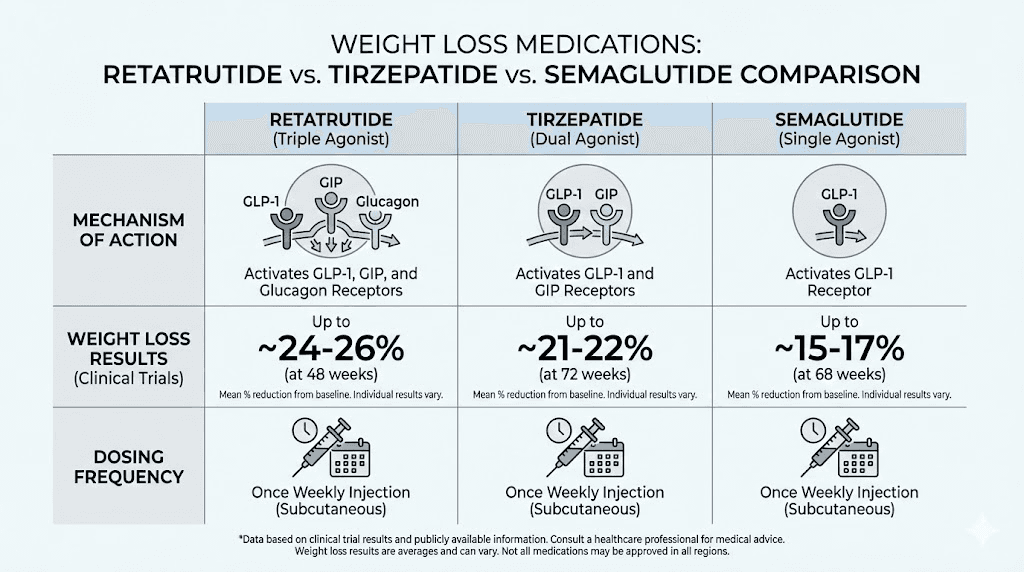
Retatrutide is not just another incretin mimetic. It is the first triple-agonist peptide to reach Phase 3 clinical trials, activating GLP-1, GIP, and glucagon receptors simultaneously. That triple mechanism changes everything about how the body processes fat, regulates appetite, and generates energy. But it also means the dosing protocol carries more complexity than a single-agonist compound like semaglutide or even the dual-agonist tirzepatide. Getting the retatrutide dose right requires understanding what each receptor does, how titration protects you from gastrointestinal distress, and why the clinical trials used the specific escalation schedules they did.
This guide covers every dose level tested in human trials, the exact titration timelines used by researchers, weight loss and liver fat data broken down by milligram, reconstitution and injection protocols, side effect management at each tier, and what the Phase 3 TRIUMPH program tells us about where dosing standards are heading.
Whether you are planning a first protocol or adjusting a current one, the information here comes directly from published clinical data, not forum speculation. SeekPeptides has compiled the most detailed dosing reference available so you can make informed decisions at every stage of the process.
How retatrutide works as a triple agonist
Every other weight loss peptide on the market targets one receptor. Maybe two. Retatrutide targets three. That distinction is not marketing language. It represents a fundamental shift in how pharmacological weight management works at the cellular level, and it explains why the retatrutide dosage chart produces outcomes that outpace everything else in the pipeline. For researchers already familiar with the broader peptide weight loss category, retatrutide represents a generational leap in mechanism sophistication.
GLP-1 receptor activation
The GLP-1 component does what researchers already know from semaglutide appetite suppression studies. It slows gastric emptying, meaning food sits in the stomach longer and satiety signals reach the brain sooner. It enhances glucose-dependent insulin secretion, keeping blood sugar stable without the hypoglycemic crashes that drive cravings. And it acts directly on hypothalamic appetite centers, reducing the neurological drive to eat.
But here is where retatrutide diverges. The GLP-1 activity does not work alone. It operates in concert with two additional receptor pathways, amplifying effects that a single agonist could never achieve on its own. Researchers who have tracked appetite suppression timelines with semaglutide onset data will notice that retatrutide produces similar GLP-1 driven satiety shifts, but with additional metabolic layers that pure GLP-1 agonists cannot replicate.
The GLP-1 pathway also connects to broader hormonal systems that researchers studying pancreatic peptide hormones will recognize. Insulin secretion becomes more glucose-dependent rather than basal, reducing the risk of hypoglycemia while simultaneously improving postprandial glucose control. This creates a metabolic environment where the body processes nutrients more efficiently at every dose level.
GIP receptor activation
Glucose-dependent insulinotropic polypeptide, or GIP, was long considered a problem hormone. Early research suggested it promoted fat storage. More recent data tells a different story. When GIP receptor activation combines with GLP-1 stimulation, as it does with tirzepatide dosing protocols, the result is enhanced insulin sensitivity, improved lipid metabolism, and a reduction in pathological fat deposition. Retatrutide leverages this same dual pathway while adding something tirzepatide cannot.
The GIP component also appears to improve beta-cell function over time, which has implications for researchers looking at metabolic health beyond simple weight reduction. For those comparing options, our retatrutide versus semaglutide comparison breaks down exactly where these mechanisms diverge.
Glucagon receptor activation
This is what makes retatrutide unprecedented. No approved obesity therapy directly activates the glucagon receptor. Glucagon increases hepatic glucose output, drives thermogenesis, mobilizes lipid stores, and accelerates energy expenditure. In simple terms, it tells the body to burn fuel rather than store it. Researchers who study the pancreatic peptide hormone system will recognize glucagon as the metabolic counterweight to insulin, and harnessing it pharmacologically opens therapeutic possibilities that the research community has pursued for decades.
The glucagon component is especially relevant for liver fat. Clinical data shows that retatrutide reduced hepatic fat content by over 80% at higher doses, a finding that stunned researchers studying metabolic-associated steatotic liver disease. The implications for visceral fat loss are significant, because glucagon preferentially targets the fat deposits surrounding organs rather than subcutaneous stores alone.
These three receptors working together create what pharmacologists call a synergistic metabolic cascade. Appetite drops. Energy expenditure rises. Fat mobilization accelerates. Liver fat melts. And insulin sensitivity improves across the board. Understanding this mechanism is critical for understanding why specific retatrutide doses produce the results they do.
The synergy also explains why dose titration matters so much more with retatrutide than with simpler compounds. When you increase a single-agonist like semaglutide, you are ramping one pathway. When you increase retatrutide, you are ramping three simultaneously. Each receptor system has its own adaptation timeline, its own threshold for side effects, and its own contribution to the overall metabolic response. Researchers familiar with GLP-3 peptide research and emerging gut hormone science will appreciate just how complex these receptor interactions become at higher dose levels.
For anyone evaluating whether the triple-agonist approach is worth the added complexity, the clinical data speaks clearly. No single or dual agonist has matched the combined weight loss, liver fat reduction, and body composition outcomes that retatrutide achieves. The peptides versus SARMs comparison highlights this distinction further for researchers weighing metabolic peptides against other compound categories entirely.
Retatrutide dose levels tested in clinical trials
The Phase 2 trial published in the New England Journal of Medicine tested four dose tiers against placebo over 48 weeks. Each tier tells a distinct story about efficacy, tolerability, and the tradeoffs researchers face when selecting a protocol. The retatrutide 20mg dosing guide covers extended protocols, but the foundational data starts here.
1mg weekly dose
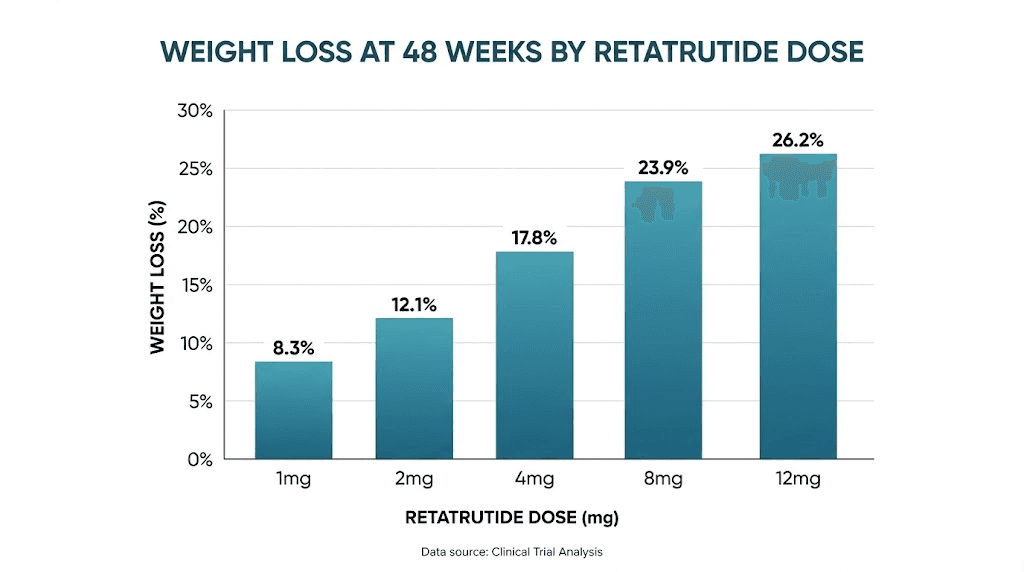
The lowest dose tested. Participants receiving 1mg weekly lost an average of 8.7% of their body weight at 48 weeks. That number might seem modest compared to the higher tiers, but context matters. An 8.7% reduction exceeds what most lifestyle interventions achieve. It exceeds what orlistat produces. And it comes with the mildest side effect profile of any group in the trial.
Nausea occurred in approximately 14% of participants at this dose. That is lower than many tirzepatide starting dose protocols. For researchers prioritizing tolerability over maximum effect, the 1mg tier provides a meaningful metabolic intervention with minimal disruption. Those curious about how much peptides cost will also find this dose tier the most budget-friendly option for sustained protocols.
The 1mg dose also serves a critical role as the universal starting point for all titration schedules. Even researchers targeting 12mg will spend their first four weeks at 1mg, making this the dose level every single protocol participant experiences. Understanding its effects establishes the baseline against which all subsequent escalations are measured.
4mg weekly dose
At 4mg, the weight loss curve steepens dramatically. Participants lost an average of 17.1% body weight at 48 weeks. That figure rivals the best results from semaglutide and tirzepatide head-to-head trials, and retatrutide achieved it with a shorter observation period.
Side effects increase proportionally. Nausea rates climbed to 18-36% depending on the titration speed, and gastrointestinal complaints became more common across the board. But for many researchers, a 17% reduction represents the threshold where metabolic health markers improve substantially, including fasting glucose, HbA1c, triglycerides, and blood pressure. This is the dose tier where the mainstream conversation about peptides typically focuses, because the results are dramatic enough to generate attention while remaining achievable for most participants.
The 4mg tier is also the lowest maintenance dose being tested in the Phase 3 TRIUMPH program, which signals that the sponsor considers it the minimum effective dose for a regulatory filing. Researchers building long-term protocols should note that 4mg may become the standard entry-level prescribed dose once approval occurs, making current data at this level especially relevant for understanding what the future standard of care might look like. Those who want to run the exact calculations for their own protocol can use the retatrutide dosage chart for reference.
8mg weekly dose
The 8mg tier produced 22.8% body weight loss at 48 weeks. Read that number again. Nearly a quarter of total body weight gone in under a year, from a once-weekly injection. This dose level also showed the greatest total fat mass reduction in the body composition substudy, with participants losing an average of 21.6kg of pure fat mass compared to placebo.
Nausea rates ranged from 17% to 60% depending on the specific protocol arm, highlighting why titration speed matters enormously at this level. Researchers who escalated too quickly bore the brunt of gastrointestinal side effects, while those who followed the standard four-week titration intervals reported significantly better tolerability. If you are exploring peptides for fat loss, the 8mg tier represents the intersection of near-maximum efficacy with manageable side effects.
The 8mg level is also where the glucagon receptor activation begins producing its most visible effects. Thermogenesis increases noticeably, with some researchers reporting feeling warmer than usual, increased sweating during physical activity, and elevated resting metabolic rate. These are signs that the glucagon component is driving energy expenditure at a level that meaningfully contributes to the overall weight loss beyond what GLP-1 and GIP provide alone. Researchers exploring complementary approaches may find the 5-amino-1MQ fat metabolism pathway an interesting parallel mechanism to understand how different compounds target energy expenditure through distinct biochemical routes.
12mg weekly dose
The highest dose tested produced 24.2% body weight loss at 48 weeks. Over 80% of participants on this dose lost more than 15% of their total body weight, a benchmark that correlates with resolution of obesity-related comorbidities in clinical literature.
But the gap between 8mg and 12mg is narrower than the gap between 4mg and 8mg. An additional 4mg yielded only 1.4 percentage points of additional weight loss, while side effects increased notably. Nausea hit 45% of participants.
This diminishing return is crucial information for anyone building a protocol, because it suggests that 8mg may represent the optimal balance point for many researchers. The retatrutide cost analysis becomes especially relevant here, since higher doses mean higher expenditure for incrementally smaller gains.
The complete retatrutide titration schedule
Titration is not optional. It is the single most important factor separating successful protocols from abandoned ones. The clinical trials used a structured escalation schedule designed to give the gastrointestinal system time to adapt to each new level of receptor activation. Skipping steps or accelerating the timeline almost always leads to intolerable nausea, vomiting, or diarrhea.
Standard four-week titration blocks
The Phase 2 trial protocol followed a consistent pattern. Researchers started every participant at the same base dose regardless of their target maintenance level, then increased the dose every four weeks until reaching the assigned tier.
Weeks 1 through 4: 1mg weekly. This introductory phase allows the body to adjust to GLP-1, GIP, and glucagon receptor activation simultaneously. Even participants destined for the 12mg arm started here. There are no shortcuts past this step.
Weeks 5 through 8: 2mg weekly for those heading to 4mg or above. The first escalation doubles the dose, which sounds aggressive but represents the smallest absolute increase in the protocol. Most participants tolerate this step with minimal new symptoms.
Weeks 9 through 12: 4mg weekly for those heading to 8mg or 12mg. This is where many researchers first notice significant appetite suppression. The GLP-1 component reaches a threshold level that produces noticeable changes in hunger patterns and food preferences.
Weeks 13 through 16: 8mg weekly for those heading to 12mg. The jump from 4mg to 8mg is the largest absolute increase in the protocol and typically produces the most intense gastrointestinal adjustment period. How long peptides take to work depends heavily on this transition phase, because the glucagon receptor activation reaches full effect here.
Weeks 17 onward: 12mg weekly maintenance for those in the highest arm. By this point, the body has had sixteen weeks to progressively adapt to triple-agonist activity. Side effects that seemed challenging at week 9 have typically faded by week 17.
Why four-week intervals matter
Four weeks is not arbitrary. Retatrutide has a half-life of approximately six days, meaning it takes roughly four to five half-lives, or about 24 to 30 days, to reach steady-state plasma concentrations at any given dose. Increasing before steady state means the body never fully adapts to one level before confronting the next. The result is stacking side effects that compound rather than resolve.
Researchers using the peptide calculator should factor this pharmacokinetic reality into their planning. A protocol that looks efficient on paper but compresses titration intervals will almost certainly produce worse outcomes than a patient, methodical escalation. SeekPeptides members access detailed titration planning tools that account for individual tolerance factors most generic charts ignore.
Modified titration for sensitive individuals
Not every researcher tolerates the standard schedule. Some experience persistent nausea even at the 1mg starting dose. For these individuals, a modified approach may be appropriate.
One strategy involves extending the initial 1mg phase to six or eight weeks instead of four. Another involves stepping from 1mg to 1.5mg before jumping to 2mg, creating an intermediate tier that the clinical trials did not formally test but that some researchers implement based on tolerability assessments. The key principle remains the same: never increase a dose while actively experiencing significant gastrointestinal symptoms from the current one.
This patient approach matters more than most people realize. A researcher who takes twenty weeks to reach 8mg but stays there for six months will achieve far more than one who reaches 12mg in eight weeks but quits after two months of intolerable nausea. The peptide community discussions are filled with stories of researchers who abandoned protocols prematurely because they rushed titration, and the pattern is remarkably consistent across compounds.
Titration for researchers switching from other compounds
A common question arises among researchers who have been using semaglutide or tirzepatide and want to transition to retatrutide. The temptation is to skip the low-dose titration phase, reasoning that GLP-1 receptor adaptation has already occurred. This reasoning is partially correct but dangerously incomplete.
While GLP-1 tolerance may carry over to some degree, the GIP and glucagon receptor components of retatrutide are entirely new stimuli for these researchers. The glucagon pathway in particular produces effects, including thermogenesis and hepatic lipid mobilization, that no prior GLP-1 or GLP-1/GIP compound prepared the body for. Starting at 1mg for at least two to four weeks remains the recommended approach even for experienced researchers transitioning from other incretin therapies.
The injectable versus oral peptide comparison is also relevant here, because some researchers transitioning from oral semaglutide may have different GLP-1 receptor adaptation profiles than those coming from injectable formulations. Oral bioavailability differs significantly from subcutaneous delivery, which means the degree of prior receptor adaptation is not directly comparable.
Weight loss results by retatrutide dose
Numbers tell stories. And the weight loss data from the retatrutide Phase 2 trial tells one of the most compelling stories in obesity pharmacotherapy. Every dose tier outperformed placebo, but the magnitude of difference between the lowest and highest tiers reveals exactly how dose-dependent the response curve is.
The dose-response curve explained
At the 1mg level, participants lost 8.7% of body weight over 48 weeks. At 4mg, that number nearly doubled to 17.1%. The jump from 4mg to 8mg added another 5.7 percentage points, reaching 22.8%. And the final step from 8mg to 12mg added just 1.4 points to reach 24.2%.
See the pattern. Each successive dose increase produces a smaller incremental gain. Pharmacologists call this a flattening dose-response curve, and it carries enormous practical implications. The difference between 1mg and 4mg is massive. The difference between 8mg and 12mg is minimal. This means researchers face a genuine cost-benefit analysis when choosing their target maintenance dose. Those exploring peptides for weight loss and muscle gain should weigh this curve carefully against side effect profiles and budget constraints.
Percentage of participants reaching weight loss thresholds
Raw averages obscure individual variation. The threshold data paints a clearer picture.
At 12mg, over 80% of participants lost 15% or more of their body weight. At 8mg, that figure was approximately 71%. At 4mg, roughly 50% of participants cleared the 15% threshold. And at 1mg, about 25% achieved that level of reduction.
The 15% threshold matters because clinical literature consistently identifies it as the point where obesity-related comorbidities begin resolving. Type 2 diabetes remission rates increase sharply. Sleep apnea improves or resolves. Joint pain decreases. Cardiovascular risk markers normalize. Reaching this threshold is often more clinically meaningful than any specific number on a scale. The tirzepatide before and after data shows similar threshold effects at the 15% mark, reinforcing that this is a physiologically meaningful breakpoint across compound classes.
For researchers comparing these outcomes against other compounds, our detailed retatrutide and semaglutide comparison contextualizes what these numbers mean relative to the current standard of care.
Rate of weight loss over time
Weight loss was not linear. The curve follows a characteristic pattern that researchers should anticipate.
Weeks 1 through 12 showed moderate but accelerating weight loss as titration progressed. Weeks 12 through 24 represented the steepest phase of the curve, with the highest rate of weekly loss occurring during this window. Weeks 24 through 36 showed continued loss but at a decelerating rate. And weeks 36 through 48 approached a plateau in most participants, with weight either stabilizing or declining very slowly.
This timeline matters for expectations. Researchers who evaluate their protocol at week 8 are seeing titration-phase results, not maintenance-phase results. The full picture does not emerge until week 24 at minimum. Anyone tracking results alongside fat burning peptide protocols should plan assessment windows accordingly.
Individual variation in response
Average weight loss numbers obscure tremendous individual variation. In the 12mg arm, some participants lost over 30% of their body weight while others lost under 15%. Genetic factors, baseline metabolic health, dietary compliance, physical activity levels, and even gut microbiome composition likely contribute to this variability.
Researchers should not panic if their results fall below the published averages at any given timepoint. The trajectory matters more than any single measurement. As long as the trend line moves in the right direction, the protocol is working. Plateau periods lasting two to four weeks are normal and do not necessarily indicate a need for dose adjustment.
Those tracking their results in detail will benefit from tools that account for normal fluctuations. The peptide calculator tools at SeekPeptides help researchers establish realistic expectations based on their starting parameters rather than relying solely on clinical trial averages that may not reflect individual circumstances.
Weight loss and metabolic health markers
Beyond the scale, retatrutide produced improvements in virtually every metabolic health marker measured. Fasting glucose decreased significantly at all dose levels above 1mg. HbA1c improved, with some participants in the higher dose arms achieving values consistent with prediabetes resolution. Triglycerides dropped. LDL cholesterol decreased. HDL cholesterol increased. Blood pressure improved.
These changes often precede visible weight loss on the scale, which is important context for researchers who feel discouraged during the early weeks. Internal metabolic improvement begins before external changes become obvious. The vilon peptide research on immune and metabolic modulation provides complementary context for understanding how peptide-driven metabolic shifts operate across multiple body systems simultaneously.
Retatrutide dose and liver fat reduction
If the weight loss data was impressive, the liver fat data was extraordinary. A substudy examining participants with metabolic-associated steatotic liver disease produced results that no pharmacological intervention had previously achieved.
Liver fat reduction by dose
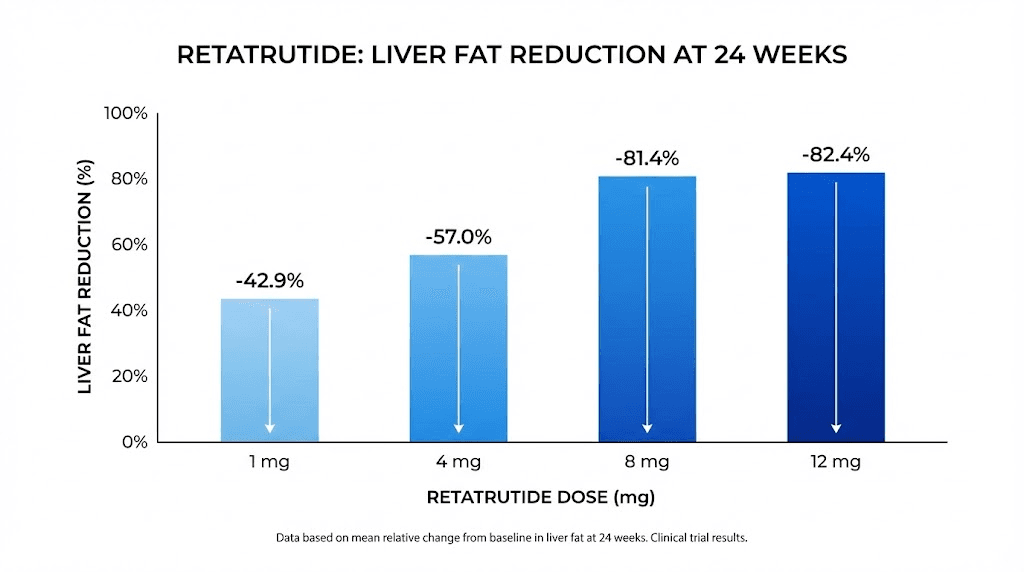
At the 1mg dose, liver fat decreased by 42.9% at 24 weeks. Already remarkable. At 4mg, the reduction reached 57.0%. At 8mg, it hit 81.4%. And at 12mg, liver fat dropped by 82.4%.
Those numbers deserve context. The pharmaceutical industry has spent decades trying to develop effective treatments for fatty liver disease. Most candidates produce reductions in the 15-30% range. Retatrutide more than doubled those figures at its lowest dose and nearly eliminated hepatic steatosis at its highest.
Resolution of fatty liver disease
Even more striking than relative reduction was the rate of normalization. Among participants receiving 12mg, 86% achieved normal liver fat levels by week 24. In the placebo group, that number was 0%. Not low. Zero.
The glucagon receptor component appears to drive much of this effect. Glucagon directly stimulates hepatic lipid oxidation, essentially telling the liver to burn its own fat stores for energy. Combined with the GLP-1-mediated improvements in insulin sensitivity and the GIP-mediated changes in lipid metabolism, the triple agonist creates conditions under which the liver clears accumulated fat at rates never before observed in clinical settings.
This has profound implications for researchers concerned about belly fat and visceral adiposity. Hepatic fat accumulation correlates closely with visceral fat deposits, and the mechanisms that clear one tend to clear the other. Researchers exploring 5-amino-1MQ and other metabolic peptides for similar goals will find the retatrutide liver data particularly relevant as a benchmark.
Clinical significance of liver fat clearance
Why do these liver numbers matter so much? Because fatty liver disease is not just an incidental finding. It is a progressive condition. Left untreated, hepatic steatosis advances to steatohepatitis, then to fibrosis, and eventually to cirrhosis. Each stage carries escalating risk and diminishing treatment options. A compound that can reverse steatosis in 86% of participants does not just improve a lab value. It potentially halts a disease trajectory.
The pharmaceutical industry currently has very few approved treatments for metabolic-associated steatotic liver disease. Resmetirom received FDA approval for the fibrotic stage, but the steatosis stage, where the vast majority of patients sit, has no approved pharmacological intervention. Retatrutide could become the first obesity therapy to carry a secondary indication for liver fat management, which would dramatically expand its clinical relevance beyond weight loss alone.
Researchers who are monitoring their own hepatic health should understand that liver fat reduction at the levels seen with retatrutide correlates with improvements in liver enzyme panels, specifically ALT and AST reductions that often normalize within the first 24 weeks of treatment. These markers are inexpensive to track through routine bloodwork and provide an objective measure of hepatic response to the protocol. The dosing chart data can help researchers correlate their specific dose level with expected liver fat improvements based on the clinical trial findings.
Body composition changes at each dose
Weight loss alone tells an incomplete story. What kind of weight a researcher loses, fat versus lean tissue, determines whether the metabolic outcome is beneficial or potentially harmful. The retatrutide body composition data addresses this question directly.
Fat mass reduction
The body composition substudy measured total fat mass changes using dual-energy X-ray absorptiometry, the gold standard for distinguishing fat from lean tissue. At the 8mg dose, participants lost an average of 21.6kg of fat mass compared to placebo. At 12mg, the fat mass reduction was 18.7kg.
Wait. The 12mg group lost less fat mass than the 8mg group? That finding surprised many researchers and requires careful interpretation. The 12mg group may have had different baseline compositions, or the additional weight loss at 12mg may have included a slightly higher proportion of lean tissue. This is exactly the kind of nuance that headline-level reporting misses but protocol-level planning demands.
Lean mass preservation
Lean mass was relatively preserved across all dose tiers, though not perfectly. Any intervention that produces 20%+ weight loss will involve some lean tissue reduction. The question is proportion.
In the retatrutide trials, the ratio of fat loss to lean loss was favorable compared to caloric restriction alone, where lean mass can represent 25-35% of total weight lost. The triple-agonist mechanism, particularly the glucagon component driving preferential fat oxidation, appears to shift the ratio toward greater fat selectivity.
Researchers interested in maintaining muscle mass during aggressive weight loss protocols may want to examine peptides for muscle growth and peptides versus TRT options as complementary strategies. The combination of a metabolic peptide for fat loss with an anabolic support agent is a protocol structure gaining traction in the research community.
Visceral versus subcutaneous fat
While the trial did not break down visceral and subcutaneous fat reduction separately using imaging for all participants, the liver fat data serves as a strong proxy. Visceral fat and hepatic fat accumulate and resolve through similar mechanisms. The dramatic liver fat clearance at higher doses strongly suggests proportional visceral fat reduction.
This matters because visceral adiposity drives metabolic disease far more potently than subcutaneous fat. A researcher who loses 15kg of visceral fat achieves more health benefit than one who loses 25kg of subcutaneous fat. The FTPP peptide adipotide research targets this same visceral compartment through entirely different mechanisms, providing additional context for researchers evaluating their options.
Implications for body composition optimization
The body composition data has practical implications for protocol design. Researchers who want to maximize fat loss while minimizing lean tissue reduction should consider several adjunct strategies during their retatrutide protocol.
Resistance training during treatment helps preserve muscle mass by maintaining the mechanical stimulus that signals the body to retain lean tissue even during caloric deficit. Protein intake becomes critical, with research suggesting that 1.2 to 1.6 grams per kilogram of body weight per day helps offset the lean mass losses associated with rapid weight reduction. Some researchers explore concurrent use of muscle-supporting peptides to further protect lean tissue during aggressive metabolic interventions.
The MOTS-c peptide research is particularly relevant here, as MOTS-c affects mitochondrial function and exercise capacity in ways that could complement the metabolic effects of retatrutide. Stacking strategies that pair metabolic peptides with performance-supporting compounds represent an emerging area of interest in the research community. Use the peptide stacking calculator to evaluate potential combination protocols.
For women navigating weight management alongside hormonal changes, the intersection of metabolic peptides and hormonal health adds another layer of complexity. Our guide on peptides for menopause weight loss addresses considerations unique to female researchers during perimenopause and postmenopause phases.
How to reconstitute and administer retatrutide
Theory means nothing without execution. The reconstitution and injection protocol for retatrutide follows the same general principles as other lyophilized peptides, but several details specific to this compound deserve attention. Getting these steps wrong can destroy potency before a single dose reaches the bloodstream.
Reconstitution protocol
Retatrutide arrives as a lyophilized powder, a freeze-dried cake or powder in a sealed vial. Converting it to an injectable solution requires bacteriostatic water and careful technique.
Step one: Gather supplies. You need the retatrutide vial, bacteriostatic water, an appropriately sized syringe for mixing, and alcohol swabs. Everything must be sterile.
Step two: Clean the vial tops. Wipe both the peptide vial and the bacteriostatic water vial with alcohol swabs. Let them air dry for ten seconds. Do not blow on them. Do not touch the cleaned surface. Contamination at this stage introduces bacteria that can multiply in the solution over the coming weeks, ruining the entire vial. The peptide vial handling guide covers sterile technique in extensive detail for researchers who want to minimize contamination risk throughout the process.
Step three: Draw bacteriostatic water. The volume you add determines the concentration of your reconstituted solution. Use the peptide reconstitution calculator to determine the exact volume based on your vial size and desired dose per unit. For most retatrutide vials, adding 1ml of bacteriostatic water to a 5mg vial produces a concentration of 5mg/ml, making dose measurement straightforward.
Step four: Add water to the peptide vial. Insert the needle into the vial at an angle and let the water run slowly down the inside wall of the vial. Do not inject the stream directly onto the powder cake. Direct force can damage the peptide structure.
Step five: Swirl gently. Do not shake. Shaking creates foam, introduces air bubbles, and can denature the peptide through mechanical stress. Hold the vial and roll it gently between your palms for 30 to 60 seconds. If powder remains undissolved, set the vial down and wait five minutes before swirling again. Clear solution indicates complete reconstitution.
Researchers new to this process should review our comprehensive guide on how to store peptides after reconstitution before proceeding. Proper handling from this point forward determines how long your reconstituted solution remains viable.
Storage after reconstitution
Once reconstituted, retatrutide must be refrigerated at 2 to 8 degrees Celsius. Room temperature storage degrades the peptide rapidly. Data on how long peptides last at room temperature consistently shows significant potency loss within hours for reconstituted solutions.
Reconstituted retatrutide remains stable for approximately four weeks under proper refrigeration. Mark the vial with the reconstitution date. Do not use solution that has been reconstituted for more than 28 days, even if it appears clear. Degradation products are not always visible. Researchers running high-dose protocols that deplete vials quickly face less stability concern, but those on lower doses like 1mg or 2mg may find that a single vial lasts close to the stability window, making careful date tracking essential.
Storage conditions between injections matter as well. Every time you remove the vial from refrigeration to draw a dose, you expose the solution to temperature fluctuation. Minimize the time the vial spends at room temperature. Draw your dose quickly, return the vial to the refrigerator immediately, and never leave a reconstituted vial on a countertop or in a travel bag for extended periods. The difference between four-week and two-week effective stability often comes down to these handling habits. Researchers who travel frequently should review the guidance on room temperature peptide stability to understand exactly how much potency they risk during transit.
For unreconstituted powder, storage guidelines differ. Check our guide on how long peptides last in powder form and the peptide vial research guide for detailed stability data across different storage conditions.
Injection protocol
Retatrutide is administered as a subcutaneous injection once weekly. The protocol is simple but requires attention to several details that affect absorption and comfort.
Syringe selection: Use an insulin syringe with a 29 to 31 gauge needle. Smaller gauge numbers mean larger needles, so 31 gauge is the finest option and typically the most comfortable. A 0.5ml or 1ml syringe provides adequate precision for most dose calculations.
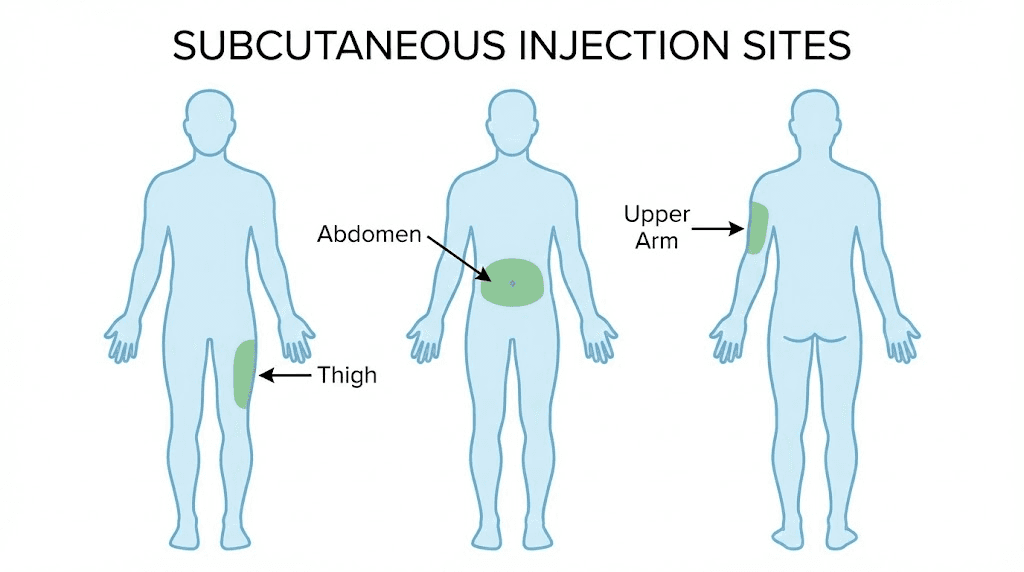
Injection sites: Three primary sites work for subcutaneous delivery. The abdomen, at least two inches from the navel, offers the most consistent absorption. The anterior thigh, in the outer middle third, provides ample subcutaneous tissue. The posterior upper arm works but is harder to self-administer.
Rotation: Rotate injection sites weekly. Using the same spot repeatedly causes lipodystrophy, a condition where subcutaneous fat thins or thickens at the injection site, altering absorption kinetics and potentially causing visible skin changes. Alternate between left abdomen, right abdomen, left thigh, right thigh in a consistent rotation.
Timing: Inject on the same day each week. Some researchers prefer morning administration, reasoning that any transient nausea will occur during active hours when it is easier to manage. Others prefer evening injection, hoping to sleep through the initial adjustment window. Neither timing has demonstrated superiority in clinical data. Consistency matters more than clock position.
Concentration calculations and unit conversions
Getting the math right prevents dosing errors. The relationship between vial size, reconstitution volume, and injection volume determines whether each dose delivers the intended milligrams.
Take a common example. A 5mg vial reconstituted with 1ml of bacteriostatic water yields a concentration of 5mg/ml. To draw a 1mg dose, you need 0.2ml, which equals 20 units on a standard U-100 insulin syringe. For a 4mg dose, you would draw 0.8ml, or 80 units. For an 8mg dose from this vial concentration, you would need 1.6ml, which exceeds the capacity of a single 1ml syringe, requiring either a larger reconstitution volume or two separate draws.
A more practical reconstitution for higher doses uses 2ml of bacteriostatic water in a 10mg vial, producing a 5mg/ml concentration where 8mg equals 1.6ml. Alternatively, using a 20mg vial with 2ml produces a 10mg/ml solution where even 12mg requires only 1.2ml, fitting comfortably in a standard insulin syringe. These calculations become second nature quickly, but errors during the first few reconstitutions are common. The reconstitution calculator eliminates guesswork by computing exact volumes for any vial size and target concentration.
Researchers working with multiple peptides simultaneously should be particularly careful about labeling. Mixing up concentrations between vials can lead to dramatically incorrect dosing. Our guide on lyophilized versus liquid peptides covers the practical differences in handling and storage that affect daily protocol management. The research versus pharmaceutical grade comparison also matters here, because pharmaceutical-grade formulations often come pre-reconstituted at standardized concentrations, eliminating the calculation step entirely.
Side effects at each dose level
Every effective intervention carries costs. Retatrutide is no exception. The side effect profile is dose-dependent, predictable, and in most cases manageable with proper preparation. But dismissing these effects or failing to plan for them is one of the fastest ways to derail a protocol.
Gastrointestinal effects
Nausea dominates the side effect landscape. At the 1mg dose, roughly 14% of participants reported nausea. At 4mg, rates ranged from 18% to 36%. At 8mg, the range widened to 17% to 60%, depending heavily on titration speed. At 12mg, nausea affected approximately 45% of participants.
Those ranges are not contradictions. They reflect different protocol arms within the same trial. Participants who titrated more slowly reported substantially less nausea than those on accelerated schedules. This is perhaps the most actionable finding in the entire side effect dataset. Slow titration does not just improve comfort. It improves adherence, which directly improves outcomes.
Beyond nausea, the gastrointestinal side effect cluster includes diarrhea, vomiting, constipation, and decreased appetite, which is technically a desired effect but can become problematic at extreme levels. Most of these symptoms peak during the first two to four weeks after each dose escalation and fade over the subsequent four to eight weeks as receptor adaptation occurs.
Cardiovascular effects
Heart rate increased by 5 to 10 beats per minute across the higher dose groups, peaking around week 24. This is consistent with the glucagon receptor activation driving increased sympathetic tone and thermogenesis. The increase was modest and did not produce clinical events in the trial population, but researchers with pre-existing cardiovascular conditions should factor this into their risk assessment.
Blood pressure actually improved in most participants, a benefit attributed to weight loss and improved insulin sensitivity outweighing any direct cardiovascular stimulation from the drug itself. Researchers with concerns about cardiovascular parameters should establish baseline readings before beginning any protocol and track them at regular intervals throughout treatment. The combination of improved blood pressure with a modest heart rate increase represents a net cardiovascular benefit for most individuals, but monitoring remains essential for those with pre-existing conditions.
Pancreatic considerations
Pancreatitis is a rare but serious risk associated with all incretin-based therapies. The retatrutide trials reported a small number of pancreatic events, consistent with the class effect seen in tirzepatide and semaglutide studies. Symptoms include severe, persistent abdominal pain radiating to the back, often accompanied by nausea and vomiting that differs qualitatively from the typical GI side effects.
Any researcher experiencing sudden, severe abdominal pain should discontinue administration immediately and seek medical evaluation. This is not a side effect to manage at home.
Managing side effects by dose tier
At 1mg, most researchers need no specific management. Mild nausea, if it occurs, typically resolves within the first two weeks without intervention.
At 4mg, meal timing becomes important. Eating smaller, more frequent meals rather than large portions reduces gastric distress. Avoiding high-fat meals on injection day helps. Staying hydrated addresses both nausea and constipation. Some researchers find ginger supplements or antiemetic medications helpful during the first few weeks at this tier.
At 8mg and 12mg, proactive management is essential. Researchers at these dose levels should have an antiemetic protocol ready before escalating. Meal composition shifts toward lean protein and complex carbohydrates, with fats reduced significantly during the adjustment phase. Fiber supplementation addresses constipation. And the extended titration approach, maintaining each step for six weeks instead of four, dramatically improves tolerability for sensitive individuals.
Resources on managing these effects overlap significantly with guidance for other GLP-1 based compounds. Our coverage of tirzepatide appetite suppression timelines addresses many of the same gastrointestinal management strategies applicable to retatrutide protocols.
Hair, skin, and other non-GI effects
Rapid weight loss from any mechanism can trigger telogen effluvium, a temporary increase in hair shedding that occurs when the body diverts resources away from hair follicle maintenance. This effect is not unique to retatrutide. It happens with bariatric surgery, extreme caloric restriction, and other pharmacological weight loss interventions. The shedding typically begins three to six months after the onset of rapid weight loss and resolves within six to twelve months as the body adapts to its new metabolic equilibrium.
Skin laxity is another concern with weight loss exceeding 15-20% of body weight. Subcutaneous fat loss occurs faster than skin elasticity can adapt, particularly in older researchers or those who carried excess weight for many years. Researchers interested in supporting skin health during rapid weight loss may want to explore peptides for anti-aging and collagen-supporting interventions that promote elastin and collagen synthesis.
Fatigue during the initial titration phases is commonly reported across all incretin therapies. The reduced caloric intake, combined with metabolic adaptation, can produce feelings of low energy during the first several weeks at each new dose level. This effect usually resolves as the body adjusts. Researchers experiencing persistent fatigue should ensure adequate hydration, electrolyte balance, and micronutrient intake before attributing the symptom to the compound itself. The discussion of tirzepatide and fatigue addresses this overlap in detail.
Retatrutide dose compared to tirzepatide and semaglutide
Comparison is inevitable. Researchers want to know where retatrutide fits in the landscape of metabolic peptides, and the data makes the positioning clear. But fair comparison requires acknowledging the differences in trial design, duration, and population that make direct head-to-head claims imprecise.
Weight loss head-to-head
Retatrutide at 12mg produced approximately 24% body weight loss at 48 weeks. Tirzepatide at its highest approved dose of 15mg produced approximately 22% weight loss, but over a longer 72-week trial period. Semaglutide at 2.4mg produced approximately 17% weight loss over 68 weeks.
The raw numbers favor retatrutide. It achieved more weight loss in less time. But these were different trials, different populations, and different primary endpoints. Only a head-to-head randomized controlled trial could definitively establish superiority, and that trial has not been conducted. Researchers who want to examine the granular differences between these compounds can start with the retatrutide versus semaglutide detailed breakdown and the semaglutide versus tirzepatide head-to-head comparison for the complete picture.
What can be said is that the triple-agonist mechanism appears to produce a steeper dose-response curve than either the dual-agonist (tirzepatide) or single-agonist (semaglutide) approach. The glucagon component adds a dimension of metabolic activation that the other compounds simply do not possess. Researchers weighing their options should read the complete retatrutide versus semaglutide analysis for detailed mechanistic comparisons.
Liver fat comparison
This is where retatrutide separates from the competition most decisively. Neither tirzepatide nor semaglutide has demonstrated liver fat reductions approaching the 80%+ figures seen with retatrutide. Tirzepatide shows promising hepatic effects in ongoing MASLD trials, and semaglutide has demonstrated modest liver fat reduction, but neither approaches the retatrutide benchmark.
The glucagon receptor, again, appears to be the differentiator. Direct hepatic lipid oxidation is a mechanism unique to the retatrutide triple-agonist profile. Researchers specifically targeting visceral and hepatic fat may find this advantage decisive in their compound selection.
Side effect comparison
All three compounds share a similar gastrointestinal side effect profile, which is expected given that all three activate the GLP-1 receptor. Nausea, diarrhea, vomiting, and constipation appear across all studies. The rates are broadly comparable when adjusted for dose equivalence.
Retatrutide adds the cardiovascular effect of modest heart rate elevation, which is less prominent with tirzepatide and semaglutide. This likely reflects the glucagon receptor activation increasing sympathetic tone and thermogenesis, the same mechanism that drives the additional metabolic benefits.
For a broader perspective on how these compounds fit within the peptide landscape, SeekPeptides provides comprehensive comparison tools and community discussions that cover real-world experiences beyond clinical trial data. The microdosing tirzepatide chart offers an alternative approach for researchers who find standard dosing too aggressive with any compound.
Cost per unit of weight loss
Beyond efficacy, cost efficiency matters for researchers planning sustained protocols. Retatrutide is not yet commercially available, meaning current pricing reflects the grey market peptide landscape rather than pharmaceutical pricing. Research-grade retatrutide typically costs more per milligram than semaglutide but less than some niche compounds.
When calculating cost per percentage point of weight loss, the 4mg dose often emerges as the most economically efficient tier. It achieves 17.1% weight loss at roughly one-third the compound cost of the 12mg dose, which achieves only 7.1 additional percentage points. The 8mg dose offers a middle ground, capturing 22.8% loss at two-thirds the compound cost. Researchers can run their specific numbers using the peptide cost calculator to determine exact per-week and per-percentage-point costs based on current sourcing prices.
Duration of treatment comparison
Trial duration differences complicate raw comparisons. Retatrutide achieved 24% loss in 48 weeks while semaglutide achieved 17% in 68 weeks and tirzepatide achieved 22% in 72 weeks. Normalizing for time, retatrutide produces approximately 0.5% body weight loss per week at its highest dose, compared to roughly 0.25% for semaglutide and 0.31% for tirzepatide.
That rate difference has practical significance. Faster weight loss means shorter treatment duration to reach a target, lower total compound consumption, and earlier access to the metabolic health benefits that accompany weight reduction. For researchers evaluating peptide options for weight loss, the speed factor may be as important as the total magnitude of effect. Faster protocols also mean less total time exposed to the gastrointestinal side effects that accompany all incretin therapies.
Adjusting your retatrutide dose
No protocol survives first contact with reality unchanged. Individual responses vary, life interferes with schedules, and side effects sometimes demand adjustments. Knowing how to modify a retatrutide dose intelligently separates experienced researchers from those following rigid scripts.
When to hold at a current dose
If you are still experiencing moderate to severe gastrointestinal side effects at your current dose level, do not escalate. This is the most common mistake. The titration schedule in the clinical trial was a minimum timeline, not a mandate. Extending any step by two, four, or even eight additional weeks is always preferable to escalating through symptoms.
Signs that you should hold include persistent nausea lasting more than seven days after a dose escalation, vomiting more than twice per week, diarrhea requiring dietary modification, or appetite suppression severe enough to prevent adequate protein intake. Any of these warrants staying at the current dose until symptoms resolve before considering the next increase.
When to reduce the dose
Dose reduction is not failure. It is informed adjustment. Reduce if gastrointestinal symptoms remain severe after six or more weeks at a given dose level, if significant dehydration results from persistent vomiting or diarrhea, if heart rate increases beyond 15 beats per minute above baseline, or if the side effect burden is causing protocol abandonment.
A reduction of one step, for example from 8mg back to 4mg, typically resolves acute symptoms within one to two weeks. After symptoms resolve, a slower re-escalation can be attempted, perhaps increasing by smaller increments than the standard protocol specifies. The semaglutide dosage calculator demonstrates how structured dose adjustments work for similar GLP-1 compounds, and the same principles of gradual escalation apply to retatrutide adjustments.
Remember that dose reduction does not erase the progress made at higher levels. A researcher who spent eight weeks at 8mg before reducing to 4mg retains the metabolic adaptations from that period. The body does not reset to zero. Weight loss continues at the lower dose, albeit at a slower rate, and the metabolic health improvements accumulated during the higher-dose phase persist as long as the protocol continues.
Handling missed doses
Missed doses happen. Travel, illness, supply disruptions, or simple forgetfulness can interrupt the weekly schedule. The approach depends on how many days have elapsed since the scheduled injection.
If less than four to five days have passed since your scheduled dose, administer it as soon as possible and resume your regular schedule the following week. If more than five days have passed, skip the missed dose entirely and resume on your next scheduled day. Never double a dose to make up for a missed one. The half-life of approximately six days means doubling produces plasma levels significantly higher than intended, increasing side effect risk without proportional benefit.
Multiple consecutive missed doses present a more complex situation. After two or more weeks without administration, restarting at the full maintenance dose risks severe gastrointestinal distress because receptor adaptation has partially reversed. The prudent approach is to restart at one step below your maintenance dose and hold for two weeks before returning to the target level. Three or more missed weeks may warrant restarting the titration from a lower base. Resources on peptide onset timelines provide context for understanding how breaks affect the pharmacokinetic steady state.
Optimizing the dose for individual goals
Not every researcher needs or wants maximum weight loss. The dose-response data supports targeting specific outcomes.
For moderate weight loss with minimal side effects, a maintenance dose of 4mg provides 17% body weight reduction with a tolerable side effect profile. This level suits researchers primarily interested in metabolic health improvements like insulin sensitivity and lipid panel normalization, where extreme weight loss is not the primary goal.
For aggressive fat loss with strong liver fat clearance, 8mg represents the sweet spot. The 22.8% weight loss and 81.4% liver fat reduction at this dose captures nearly all the benefit of the 12mg tier with meaningfully fewer side effects. The peptide cost calculator confirms that the cost-per-unit-of-benefit favors 8mg over 12mg for most researchers.
For maximum effect regardless of side effect tolerance, 12mg produces the highest absolute weight loss and liver fat clearance. But the marginal benefit over 8mg is small, and the marginal cost in side effects is real. This tier is best suited for researchers with specific clinical goals that require the last increment of pharmacological effect.
Combining retatrutide with other protocols
Some researchers explore adjunct compounds alongside their retatrutide protocol. The rationale varies. Some want to preserve lean mass during aggressive weight loss. Others target specific conditions like gut inflammation or joint recovery that retatrutide does not directly address.
Combination protocols carry added complexity and risk. Each additional compound introduces its own side effect profile, interaction potential, and pharmacokinetic considerations. The peptide stacking landscape has expanded rapidly, and the peptide stack calculator helps researchers model potential combinations before implementing them. Understanding peptides for gut health becomes especially relevant for researchers experiencing persistent GI symptoms who want to support gastrointestinal function during their metabolic protocol.
The decision to stack compounds should not be taken lightly. Begin with the retatrutide protocol alone, establish tolerability, confirm that the primary compound is producing the expected effects, and only then consider carefully selected adjuncts based on specific, measurable goals. Researchers who add too many variables simultaneously cannot identify which compound is producing which effect, making troubleshooting impossible. The peptide forum community is an excellent resource for finding researchers who have documented their combination protocol experiences, including both successes and failures.
For researchers specifically interested in the intersection of weight loss and anti-aging benefits, the overlap between metabolic peptides and longevity compounds represents a growing area of investigation. The anti-aging peptide landscape includes several compounds whose mechanisms complement rather than conflict with retatrutide action. Understanding how these different categories interact requires careful study of each compound individual profile, and the guides available at SeekPeptides provide the depth of information needed to evaluate these combinations responsibly.
What happens when you stop retatrutide
Every researcher eventually faces this question. Protocols do not run forever. Supply interruptions occur. Goals get met. Side effects sometimes necessitate discontinuation. Understanding what happens after cessation is as important as understanding the dosing protocol itself.
Weight regain patterns
The uncomfortable truth is that weight regain occurs in the majority of cases after discontinuing any GLP-1 based therapy. Data from semaglutide discontinuation studies shows that approximately two-thirds of lost weight returns within one year of cessation. Tirzepatide discontinuation data tells a similar story. There is no reason to expect retatrutide will be different.
The mechanism is straightforward. The pharmacological suppression of appetite and enhancement of energy expenditure cease when the compound clears the body. Metabolic adaptation, the body resetting its energy balance to a lower expenditure level during weight loss, means that the post-treatment metabolic rate is lower than before treatment began. Without the pharmacological support, the body gravitates back toward its previous setpoint.
This does not make the intervention pointless. Months of lower body weight produce lasting benefits including improved insulin sensitivity, reduced hepatic fat, decreased inflammatory markers, and potentially improved cardiovascular remodeling. Some of these benefits persist even after weight regain. But the weight itself tends to return.
Tapering versus abrupt cessation
The clinical trials did not specifically study tapering protocols, but physiological principles suggest a gradual dose reduction may produce a smoother transition than abrupt cessation.
A reasonable tapering approach would involve stepping down by one dose tier every four weeks. A researcher on 12mg would move to 8mg for four weeks, then 4mg for four weeks, then 1mg for four weeks before discontinuing entirely. This gives the appetite regulation system time to partially readapt at each level rather than experiencing the full rebound simultaneously.
Whether tapering actually reduces total weight regain remains unproven. But it almost certainly reduces the gastrointestinal rebound effects that some researchers report after abrupt cessation, including paradoxical increases in appetite, changes in bowel habits, and blood sugar fluctuations. The psychological component matters as well. A gradual step-down gives the researcher time to implement dietary and behavioral strategies at each lower dose level, building habits that can partially compensate for the loss of pharmacological support.
For researchers considering a taper schedule, the dosage calculator can help plan the exact reconstitution volumes needed at each reduced dose tier. Having the math sorted before starting the taper prevents logistical complications that might otherwise derail the gradual reduction plan. Each step down requires a new calculation if the reconstitution concentration does not divide evenly into the target dose, and the reconstitution calculator handles these conversions automatically.
Maintenance strategies after discontinuation
Researchers who have achieved significant weight loss with retatrutide should have a maintenance strategy in place before discontinuing. This typically includes a structured nutritional plan based on the lower caloric needs of a smaller body, a resistance training program to maintain or build lean mass that draws on the principles covered in our muscle growth peptide guide, and possibly a lower-dose maintenance protocol if long-term continuation is feasible.
Some researchers transition from retatrutide to a lower-cost compound for maintenance purposes. Moving from retatrutide to a moderate dose of semaglutide or tirzepatide can provide ongoing appetite suppression at a lower cost point. The retatrutide cost guide helps researchers evaluate these long-term economic considerations.
Others explore alternative peptide strategies for weight maintenance. Compounds like cagrilintide and amylin receptor agonists offer different mechanisms for appetite regulation that may complement the metabolic changes achieved during retatrutide treatment.
Long-term metabolic benefits that persist
Not all benefits disappear with the compound. Research on GLP-1 therapy discontinuation suggests that certain metabolic improvements show partial persistence even after weight regain. Hepatic fat reduction, in particular, may not fully reverse immediately, giving the liver additional time to recover from years of steatotic damage. Beta-cell function improvements, if they occurred during treatment, can persist for months beyond cessation.
Cardiovascular remodeling that occurs during significant weight loss may also provide lasting benefit. The heart adapts to the reduced body mass, and some of that adaptation persists even as weight returns. Inflammatory markers, while they do tend to rise with weight regain, may not return to their pre-treatment peaks if the regain is partial rather than complete.
The practical takeaway is that even a temporary course of retatrutide provides metabolic value beyond the weight loss itself. Researchers approaching discontinuation should focus not on whether weight will return, but on maximizing the duration and magnitude of the metabolic window that treatment provides. For those researching long-term peptide strategies, the broader landscape of weight management peptides offers numerous options for sustained metabolic support.
Phase 3 TRIUMPH trials and future dosing
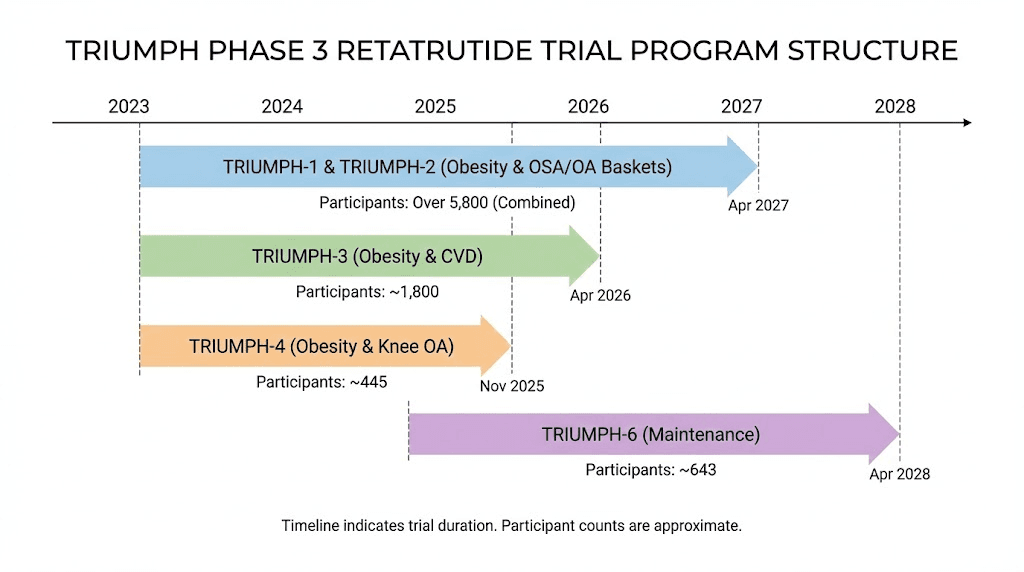
Retatrutide has not yet received regulatory approval. The Phase 2 data, while exceptional, is preliminary by regulatory standards. The Phase 3 TRIUMPH program will determine whether, and at what doses, retatrutide reaches the market.
TRIUMPH program overview
The TRIUMPH clinical development program includes multiple large-scale trials designed to confirm the Phase 2 findings in broader, more diverse populations.
TRIUMPH-1 enrolled approximately 2,300 patients and is expected to complete around mid-year. This trial tests retatrutide for obesity in adults without diabetes, using maintenance doses of 4mg, 9mg, and 12mg. The inclusion of a 9mg dose tier is noteworthy because it was not tested in Phase 2. Researchers can anticipate that the 9mg data will fill the gap between the 8mg and 12mg tiers, potentially identifying an optimal dose that balances efficacy and tolerability better than either existing data point.
TRIUMPH-2 enrolled approximately 1,800 patients with a completion timeline in early spring. This trial focuses on obesity in adults with type 2 diabetes, addressing the critical question of how retatrutide performs when glycemic control is a co-primary endpoint alongside weight loss.
TRIUMPH-4 is a more targeted study enrolling 445 patients with both obesity and knee osteoarthritis. The rationale is that weight loss beyond 15% often produces clinically meaningful improvements in joint symptoms, and retatrutide consistently crosses that threshold at moderate doses. This trial underscores the expanding scope of incretin therapy beyond simple weight management, a trend also visible in the growing body of research on peptides for athletic performance and recovery.
Additional trials in the TRIUMPH program may address retatrutide effects on cardiovascular outcomes, sleep apnea resolution, and kidney function, though these have not yet been formally announced. The breadth of the development program reflects confidence in the Phase 2 results and suggests that retatrutide, if approved, will be positioned as a comprehensive metabolic therapy rather than simply a weight loss drug. Researchers following the regulatory trajectory should monitor the peptide regulatory landscape for updates as trial results become available.
What the Phase 3 doses tell us
The Phase 3 maintenance doses of 4mg, 9mg, and 12mg represent a refinement of the Phase 2 protocol. Dropping the 1mg arm acknowledges that while it produced statistically significant weight loss, the magnitude was insufficient for a competitive regulatory filing. Adding the 9mg arm suggests the sponsor believes the 8mg to 12mg range contains the optimal therapeutic dose and wants more granular data to identify it precisely.
For researchers currently building protocols based on Phase 2 data, the Phase 3 dose selection validates the 4mg to 12mg range as the therapeutic window. The 9mg addition may eventually prove to be the ideal maintenance dose, capturing most of the 12mg efficacy with fewer side effects than either the 8mg or 12mg tiers. Future dosing recommendations will likely change based on these results, making it worth following the peptide regulation news for updates.
Expected timeline to market
If the TRIUMPH trials produce results consistent with Phase 2, regulatory submission could occur as early as late next year, with approval and market availability following twelve to eighteen months later. This places a realistic market availability estimate in the 2027 to 2028 timeframe.
Until then, retatrutide exists in the research compound space. Researchers sourcing it should understand the quality considerations involved. Our guides on grey market peptides, retatrutide sourcing, and vendor reviews for providers like QSC Peptides, Amo Peptides, and Asymchem Peptides provide critical due diligence information for navigating this landscape.
The ZLZ peptide retatrutide analysis and Paradigm Peptides retatrutide review offer additional sourcing perspectives. Researchers should also consider vendor comparisons from HK Peptides, Coast Peptides, Biomax Peptides, Klow Peptides, and Amopure Peptide reviews when evaluating sources for research-grade compounds.
Frequently asked questions
What is the best starting dose of retatrutide?
Every clinical trial protocol started all participants at 1mg weekly, regardless of their target maintenance dose. This applies universally. Starting at a higher dose skips the critical adaptation period for the GLP-1, GIP, and glucagon receptors and almost always produces severe gastrointestinal side effects. Use the retatrutide dosage calculator to plan your complete titration schedule from 1mg through your target maintenance dose.
How long does it take to see weight loss on retatrutide?
Most researchers notice initial changes within the first two to four weeks, primarily through reduced appetite and early water weight loss. Meaningful fat loss becomes measurable around weeks 8 through 12 as the dose reaches therapeutic levels. The steepest weight loss phase occurs between weeks 12 and 24. Results continue accumulating through week 48 in clinical trial data. For context on timelines with similar compounds, see our guide on semaglutide appetite suppression timelines.
Can I inject retatrutide daily instead of weekly?
No. Retatrutide has a half-life of approximately six days, which supports once-weekly dosing. Daily administration would produce accumulating plasma levels far beyond the studied range, dramatically increasing side effects without proportional benefit. The once-weekly injection schedule is both sufficient for maintaining therapeutic levels and the only regimen with clinical safety data supporting it.
Is retatrutide safe with other medications?
The clinical trials excluded participants on certain medications, so comprehensive interaction data is limited. The delayed gastric emptying caused by GLP-1 receptor activation can affect absorption of oral medications, particularly those with narrow therapeutic windows. Any researcher combining retatrutide with other compounds should consult relevant safety literature. The pancreatic peptide hormones guide provides context on how incretin-based compounds interact with endogenous hormone systems.
What happens if I miss two or more weeks of retatrutide?
After two or more missed weeks, receptor adaptation partially reverses. Restarting at your full maintenance dose risks severe nausea and gastrointestinal distress. The recommended approach is to restart one dose tier below your maintenance level, hold for two weeks, then resume your target dose. After three or more missed weeks, consider a more conservative re-titration starting two tiers below maintenance. Our guide on how long peptides take to work explains how pharmacokinetic steady states are affected by treatment interruptions.
How does retatrutide compare to tirzepatide for weight loss?
Retatrutide at 12mg produced approximately 24% weight loss at 48 weeks compared to tirzepatide at 15mg producing approximately 22% at 72 weeks. While these are different trials with different populations, the triple-agonist mechanism appears to produce faster and slightly greater weight loss. The liver fat advantage is more pronounced, with retatrutide achieving 82% reduction compared to more modest figures for tirzepatide. See our full semaglutide versus tirzepatide comparison for additional context on the incretin therapy landscape.
Do I need to diet while taking retatrutide?
The appetite suppression from retatrutide naturally reduces caloric intake without requiring a formal diet plan. However, nutritional quality matters. Protein intake should remain adequate, at minimum 0.8 grams per kilogram of body weight, to preserve lean mass during rapid weight loss. High-fat meals increase gastrointestinal side effects. And micronutrient supplementation may be warranted when total food volume decreases significantly. Researchers interested in complementary nutritional strategies can explore our guides on peptides for menopause weight loss and collagen peptides and weight loss for additional dietary considerations.
Where should I source retatrutide for research?
Retatrutide is not yet approved by any regulatory agency, meaning it is only available through research chemical suppliers. Quality varies enormously between vendors. Third-party testing certificates, published purity data, and community reputation are the primary indicators of reliability. Our comprehensive retatrutide buying guide covers vendor evaluation criteria in detail, and peptide forum discussions provide real-world sourcing experiences from the research community. The peptide pricing landscape changes frequently, so checking current rates before committing to a long-term protocol is essential.
Can I use retatrutide for gut health or other conditions beyond weight loss?
While retatrutide is being developed primarily as an obesity and diabetes treatment, the triple-agonist mechanism has implications beyond weight loss. The glucagon component enhances hepatic function, the GLP-1 component reduces systemic inflammation, and the GIP component improves metabolic signaling broadly. Some researchers hypothesize benefits for conditions involving metabolic dysfunction, though clinical data outside of obesity and diabetes remains limited. For targeted gut health support, dedicated compounds covered in our peptides for gut health guide may be more appropriate than relying on retatrutide alone.
External resources
New England Journal of Medicine: Retatrutide Phase 2 Trial Results
FDA Drug Safety and Availability Information
For researchers serious about optimizing their retatrutide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, personalized protocol tools, and a community of thousands who have navigated these exact dosing decisions. Members access detailed titration planners, peptide stacking calculators, vendor verification tools, and expert-reviewed protocols that account for individual factors most generic charts ignore. Whether you are starting your first protocol or fine-tuning an advanced regimen, the depth of information and community support at SeekPeptides turns complex dosing decisions into confident, data-driven choices.
In case I do not see you, good afternoon, good evening, and good night.