Feb 19, 2026
Two dual-agonist peptides. Both target GLP-1 receptors. Both produce dramatic weight loss in clinical trials. On paper, survodutide and tirzepatide look like the same drug wearing different labels. In practice? They activate completely different secondary pathways, produce different metabolic outcomes, and serve fundamentally different clinical purposes.
That distinction matters more than most comparison articles admit.
Tirzepatide pairs GLP-1 with GIP receptor activation. Survodutide pairs GLP-1 with glucagon receptor activation. Those second receptors determine everything, from how each drug burns fat to how it handles liver disease to how patients tolerate the side effects. One is FDA-approved and available at pharmacies right now. The other has breakthrough therapy designation for a liver condition that affects roughly 25% of the global population and is still working through Phase 3 trials.
This guide compares survodutide and tirzepatide on every metric that matters. Mechanism. Efficacy. Safety. Dosing. Availability. And the one question everyone asks but few can answer honestly: which one is actually better? The real answer, as SeekPeptides researchers know, depends entirely on what you are trying to achieve.
What is survodutide?
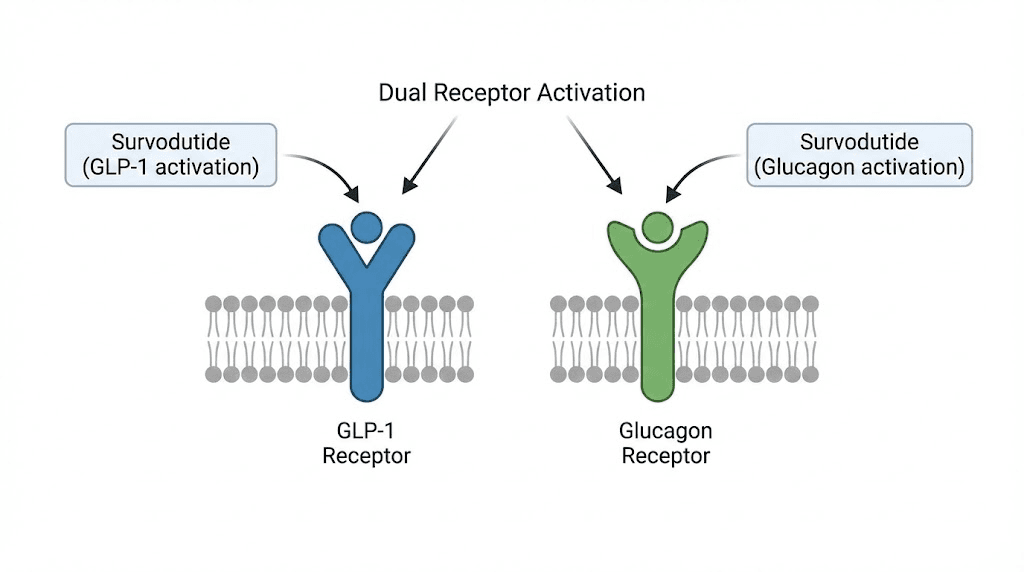
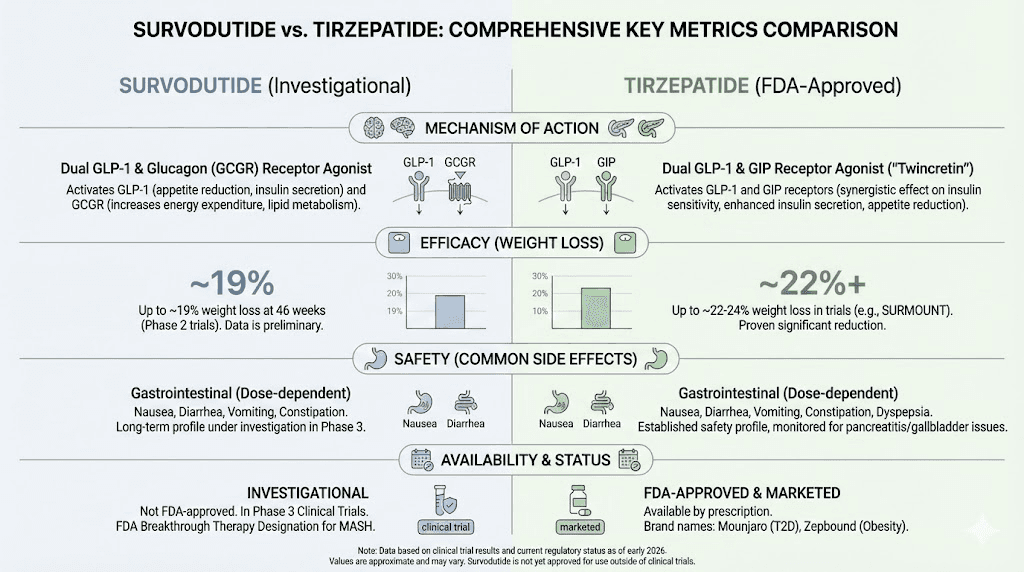
Survodutide (also designated BI 456906) is a synthetic peptide developed by Boehringer Ingelheim that activates two receptors simultaneously: the GLP-1 receptor and the glucagon receptor. This dual activation creates a metabolic profile that no single-agonist drug can replicate.
The GLP-1 component does what all GLP-1 receptor agonists do. It suppresses appetite by acting on hunger centers in the brain, slows gastric emptying to keep you feeling full longer, and improves insulin sensitivity. If you have used semaglutide or any other GLP-1 medication, you know these effects well.
The glucagon component is where survodutide gets interesting. Glucagon has historically been viewed as an anti-obesity paradox, a hormone that raises blood sugar but also increases energy expenditure and promotes liver fat oxidation. Survodutide is designed to harness the metabolic benefits of glucagon signaling while the GLP-1 component counterbalances the blood sugar elevation.
In laboratory studies, survodutide shows an EC50 of approximately 8 nM for the glucagon receptor and 1 nM for the GLP-1 receptor. That roughly 8:1 potency ratio means the GLP-1 activity dominates, which helps explain why the drug lowers blood sugar rather than raising it despite the glucagon activation.
The half-life of survodutide is approximately 109 to 115 hours, or roughly five days. This supports once-weekly dosing, matching the administration schedule of tirzepatide and semaglutide.
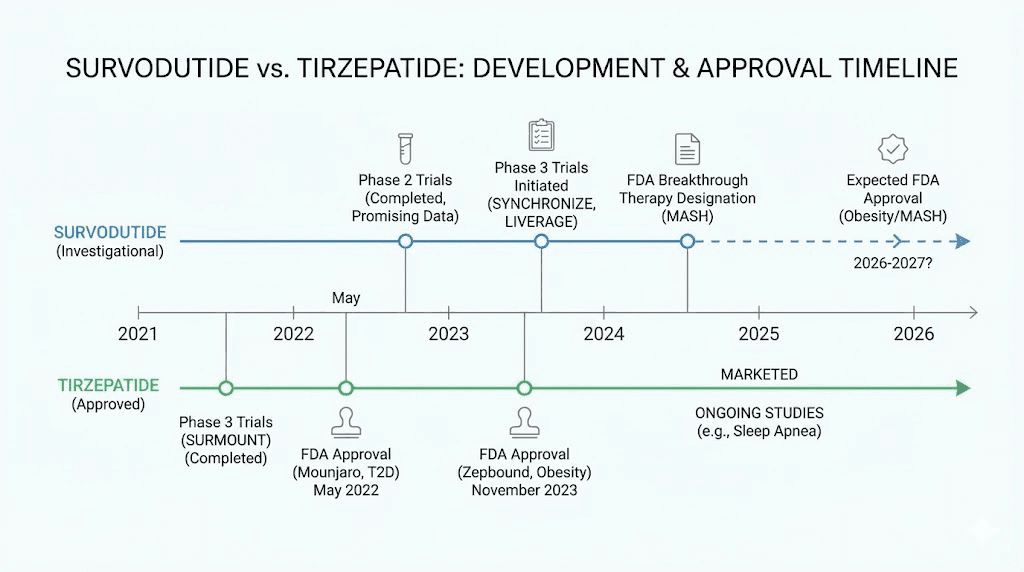
Survodutide is not yet FDA-approved for any indication. It has received FDA Breakthrough Therapy designation for MASH (metabolic dysfunction-associated steatohepatitis), the liver condition formerly known as NASH. Phase 3 trials for both obesity and MASH are ongoing, with results expected in the first half of this year.
What is tirzepatide?
Tirzepatide is a synthetic peptide developed by Eli Lilly that activates the GLP-1 receptor and the GIP (glucose-dependent insulinotropic polypeptide) receptor. It is marketed as Mounjaro for type 2 diabetes and as Zepbound for chronic weight management.
The GLP-1 component provides the same appetite suppression, gastric emptying delay, and insulin sensitization as other GLP-1 agonists. The GIP component adds a second layer of metabolic benefit. GIP enhances insulin secretion in a glucose-dependent manner, improves fat tissue metabolism, and may contribute to appetite regulation through mechanisms still being fully characterized.
Tirzepatide has been called a "twincretin" because it mimics two incretin hormones simultaneously. This dual incretin approach produces some of the most impressive weight loss results ever documented in clinical trials. The SURMOUNT-1 trial demonstrated up to 20.9% mean body weight reduction at the 15 mg dose over 72 weeks. No other approved weight loss medication had achieved numbers like that before tirzepatide.
The drug is available in pre-filled injection pens with doses ranging from 2.5 mg to 15 mg, administered once weekly. The standard tirzepatide dosing schedule starts at 2.5 mg for four weeks, then increases in increments every four weeks to the target maintenance dose.
Tirzepatide received FDA approval for type 2 diabetes (Mounjaro) in May 2022 and for chronic weight management (Zepbound) in November 2023. It is available by prescription and covered by many insurance plans for qualifying indications. For detailed dosing information, see our complete tirzepatide dosage guide in units.
The before and after results with tirzepatide have been extensively documented, with many users reporting substantial changes in body composition, appetite patterns, and metabolic markers within the first months of treatment.
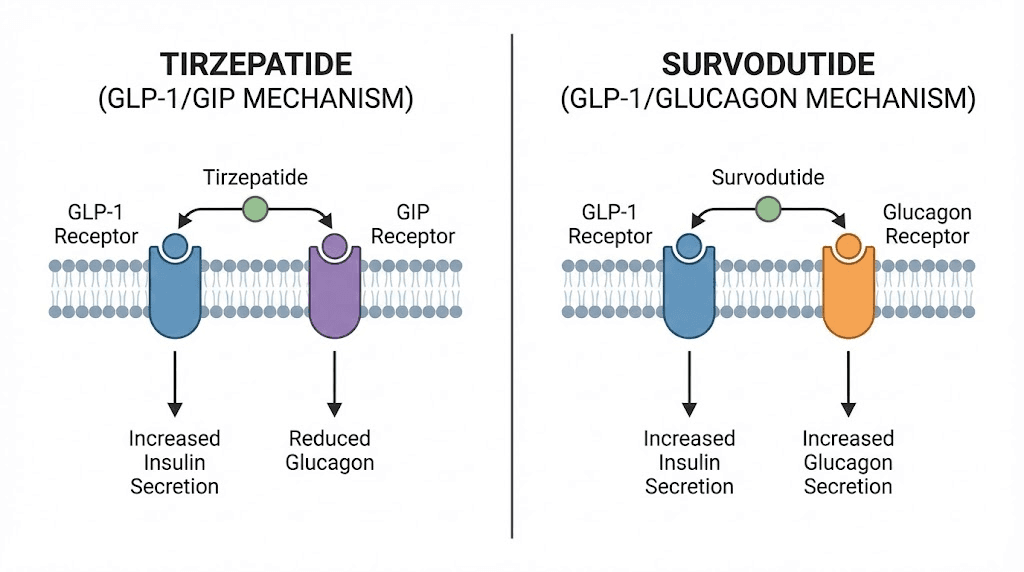
How survodutide and tirzepatide work differently
This is the core distinction that drives every difference between these two drugs. Both activate GLP-1 receptors. But their secondary targets create divergent metabolic profiles.
The GLP-1 foundation (shared)
Both survodutide and tirzepatide leverage GLP-1 receptor agonism for:
Central appetite suppression through hypothalamic signaling
Delayed gastric emptying, which prolongs satiety
Enhanced insulin secretion in response to meals
Improved glucose homeostasis
These shared effects explain why both drugs produce meaningful weight loss and glycemic improvement. The GLP-1 pathway is the proven backbone of modern weight loss peptide therapy. Understanding how peptides work at the receptor level helps explain why dual agonism amplifies single-target effects.
Tirzepatide second target: GIP receptors
GIP (glucose-dependent insulinotropic polypeptide) is an incretin hormone released from the gut after eating. When tirzepatide activates GIP receptors, several things happen.
First, insulin secretion increases in a glucose-dependent manner. This means the effect is strongest when blood sugar is elevated and diminishes when blood sugar is normal, reducing hypoglycemia risk. Second, GIP signaling appears to improve fat tissue biology by enhancing lipid storage in appropriate adipose depots rather than ectopic locations like the liver and muscle. Third, there is emerging evidence that GIP receptor activation in the brain contributes to appetite reduction through pathways distinct from GLP-1.
The combined GLP-1/GIP activation in tirzepatide creates a synergistic effect on appetite and glucose control that exceeds what either receptor alone could produce. This is why tirzepatide outperformed semaglutide in head-to-head trials. For a deeper comparison between these two, see our semaglutide vs tirzepatide analysis.
Survodutide second target: glucagon receptors
Glucagon is traditionally thought of as a counter-regulatory hormone that raises blood sugar. Activating glucagon receptors intentionally seems counterintuitive for a diabetes or obesity drug. But the story is more nuanced.
Glucagon receptor activation increases hepatic energy expenditure. Your liver burns more calories processing nutrients. This thermogenic effect is unique to glucagon signaling and not present with GIP activation. In preclinical models and early clinical trials, glucagon receptor activation increased total energy expenditure, meaning the body burns more calories even at rest.
Glucagon also promotes liver fat oxidation. It literally drives the liver to break down stored fat for energy. This is why survodutide shows such remarkable results in MASH trials, because the glucagon component directly targets the pathological fat accumulation that defines that disease.
Additionally, glucagon appears to reduce food intake through mechanisms distinct from GLP-1, providing a second independent appetite suppression pathway. Combined with GLP-1 appetite suppression, this dual approach attacks caloric intake from two different angles.
The trade-off is tolerability. Glucagon receptor activation causes more gastrointestinal distress than GIP activation, which partly explains why survodutide has higher discontinuation rates in clinical trials compared to tirzepatide.
Weight loss results: head-to-head data comparison
No direct head-to-head trial between survodutide and tirzepatide has been conducted. All comparisons rely on cross-trial analysis, which carries inherent limitations. Different patient populations, different trial durations, and different protocols make precise comparison difficult. That said, the available data tells a compelling story.
Tirzepatide weight loss data
The SURMOUNT-1 trial enrolled 2,539 adults with obesity or overweight (BMI 27 or higher with at least one weight-related comorbidity) without type 2 diabetes. After 72 weeks of treatment:
5 mg dose: 15.0% mean body weight reduction
10 mg dose: 19.5% mean body weight reduction
15 mg dose: 20.9% mean body weight reduction
Placebo: 3.1% mean body weight reduction
Among participants receiving 15 mg, approximately 57% achieved at least 20% weight loss. These results established tirzepatide as the most effective approved weight loss medication at the time.
The SURMOUNT-2 trial in patients with both obesity and type 2 diabetes showed somewhat lower but still impressive results: 12.8% to 14.7% weight loss over 72 weeks. The speed of tirzepatide results varies by individual, but most patients see meaningful changes within the first eight to twelve weeks. For people wondering how long tirzepatide takes to work, the data shows consistent progressive weight loss throughout the treatment period.
Survodutide weight loss data
The Phase 2 obesity trial enrolled 386 adults with BMI 27 or higher without type 2 diabetes. After 46 weeks of treatment:
0.6 mg dose: 6.2% mean body weight reduction
2.4 mg dose: 12.5% mean body weight reduction
3.6 mg dose: 13.2% mean body weight reduction
4.8 mg dose: 14.9% mean body weight reduction (all randomized)
4.8 mg dose: 18.7% mean body weight reduction (completers only)
Placebo: 2.8% mean body weight reduction
Among completers at the 4.8 mg dose, 68.8% achieved at least 10% weight loss, 67% achieved at least 15%, and 40% achieved at least 20% weight loss.
One critical observation: the weight loss curves had not plateaued at week 46. They were still trending downward. This suggests that a longer treatment duration, comparable to the 72 weeks used in tirzepatide trials, could yield even greater results.
Cross-trial comparison (with caveats)
Metric | Survodutide (4.8 mg) | Tirzepatide (15 mg) |
|---|---|---|
Trial duration | 46 weeks | 72 weeks |
Mean weight loss (all randomized) | 14.9% | 20.9% |
Mean weight loss (completers) | 18.7% | ~22.5% |
Achieved 10%+ loss | 68.8% | ~85% |
Achieved 15%+ loss | 67% | ~72% |
Achieved 20%+ loss | 40% | ~57% |
Weight loss still trending down | Yes (at week 46) | Plateauing (by week 72) |
Phase of trial | Phase 2 | Phase 3 |
Tirzepatide shows greater weight loss in the data we have. But the comparison is not entirely fair. Tirzepatide had 26 additional weeks of treatment. Survodutide weight loss was still accelerating. And Phase 2 trials typically use smaller, less optimized populations than Phase 3 trials. The SYNCHRONIZE Phase 3 results will provide a much clearer picture when they emerge.
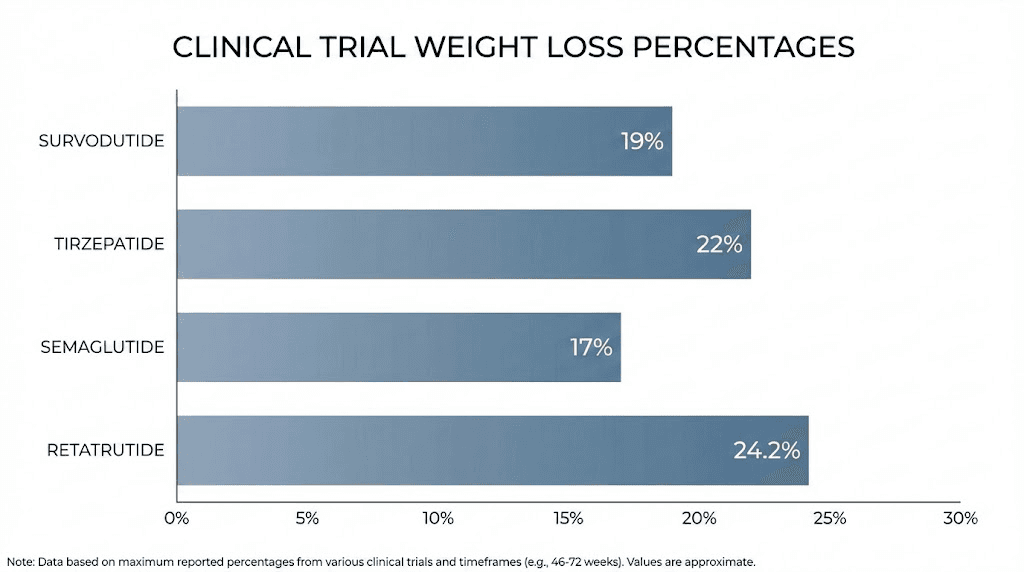
For context on how these results compare with single-agonist options, semaglutide (Wegovy) achieved 14.9% mean weight loss at 68 weeks in the STEP 1 trial, and retatrutide, a triple agonist targeting GLP-1, GIP, and glucagon receptors, showed up to 24.2% weight loss in Phase 2. Our guide on the best peptides for weight loss covers the full landscape of options.
Survodutide for MASH: the liver advantage
This is where survodutide separates itself from tirzepatide in the most clinically meaningful way. MASH (metabolic dysfunction-associated steatohepatitis) is a progressive liver disease characterized by fat accumulation, inflammation, and fibrosis. It affects approximately 25% of the global population in its earlier stages and can progress to cirrhosis and liver failure.
There are very few effective treatments for MASH. Survodutide may change that.
The Phase 2 MASH trial (published in NEJM)
This 48-week randomized, double-blind, placebo-controlled trial enrolled 293 adults with biopsy-confirmed MASH and fibrosis stages F1 through F3. Participants received weekly subcutaneous injections of survodutide at 2.4 mg, 4.8 mg, or 6.0 mg, or placebo.
The results, published in the New England Journal of Medicine, were remarkable.
MASH resolution without worsening of fibrosis:
2.4 mg dose: 47% of participants (vs 14% placebo)
4.8 mg dose: 62% of participants (vs 14% placebo)
6.0 mg dose: 43% of participants (vs 14% placebo)
Liver fat reduction of at least 30%:
2.4 mg dose: 63% of participants
4.8 mg dose: 67% of participants
6.0 mg dose: 57% of participants
Placebo: 14% of participants
Fibrosis improvement by at least one stage:
Survodutide groups: 34-36%
Placebo: 22%
The percentage change from baseline in liver fat content after 48 weeks was a staggering 62% reduction in the 6.0 mg group, compared to just 5.7% in the placebo group.
The 4.8 mg dose emerged as the optimal balance between efficacy and tolerability. The 6.0 mg dose showed slightly less MASH resolution than 4.8 mg, likely because higher dropout rates from side effects affected the intent-to-treat analysis.
Why glucagon activation drives liver fat reduction
This is not a coincidence. The glucagon receptor is predominantly expressed in the liver. When survodutide activates hepatic glucagon receptors, it directly stimulates liver fat oxidation, forcing the liver to break down its own fat stores for energy. This mechanism is fundamentally different from the indirect liver benefits seen with GLP-1 single agonists, which reduce liver fat primarily through weight loss and improved insulin sensitivity.
Survodutide attacks liver fat both directly (through glucagon-mediated hepatic fat oxidation) and indirectly (through GLP-1-mediated weight loss and metabolic improvement). This dual approach explains why the liver fat reduction numbers with survodutide exceed those seen with GLP-1 agonists alone.
How tirzepatide compares on liver endpoints
Tirzepatide has shown liver fat reduction in studies, primarily as a secondary benefit of overall metabolic improvement and weight loss. However, tirzepatide does not directly target liver fat oxidation the way survodutide does. The GIP receptor is not primarily expressed in the liver, so the glucagon-specific hepatic benefits are absent.
For patients whose primary concern is liver health, MASH, or metabolic liver disease, survodutide represents a fundamentally different therapeutic approach. This is reflected in the FDA granting survodutide Breakthrough Therapy designation specifically for MASH, an acknowledgment that the drug addresses an unmet medical need in a way existing treatments do not.
Side effects: a critical comparison
Both drugs produce gastrointestinal side effects consistent with the GLP-1 class. But the rates differ significantly, and the differences trace directly back to their secondary receptors.
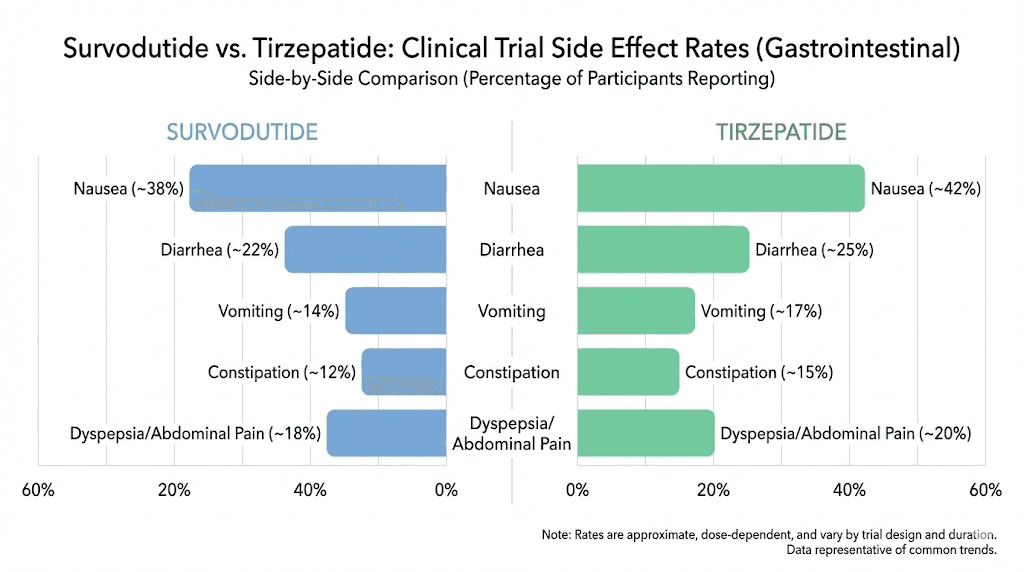
Survodutide side effects (Phase 2 data)
Nausea: 56% of participants
Vomiting: 27%
Diarrhea: 22%
Constipation: 21%
Discontinuation due to adverse events: 24.6%
Serious adverse events: 4.2%
In the MASH trial, rates were even higher: nausea 66%, diarrhea 49%, vomiting 41%. These elevated rates occurred primarily during the rapid dose-escalation phase used in Phase 2 trials. The Phase 3 SYNCHRONIZE trials use extended, flexible dose escalation, which is expected to substantially improve tolerability.
Tirzepatide side effects (Phase 3 data)
Nausea: 24-33% of participants (dose-dependent)
Diarrhea: 18-23%
Vomiting: 8-13%
Constipation: 11-17%
Discontinuation due to adverse events: 4.3-7.1%
Serious adverse events: similar to placebo
The difference in discontinuation rates is striking. About one in four survodutide participants dropped out due to side effects, compared to fewer than one in fourteen with tirzepatide. This is the single biggest practical difference between the two drugs for most patients.
Why survodutide causes more GI distress
Glucagon receptor activation directly stimulates gastric acid secretion and affects gut motility. Combined with GLP-1 effects on gastric emptying, this creates a more intense gastrointestinal experience than the GIP/GLP-1 combination in tirzepatide. GIP signaling does not have the same provocative effect on the gut.
However, context matters. The Phase 2 survodutide trials used rapid dose escalation over 20 weeks. The Phase 3 trials extend this escalation period and allow dose flexibility based on individual tolerance. If the Phase 3 protocol significantly reduces discontinuation rates, the tolerability gap between survodutide and tirzepatide could narrow considerably.
For anyone managing GI side effects with GLP-1 medications, strategies like timing meals appropriately, staying hydrated, and choosing the right foods can make a significant difference. Our guides on foods to avoid on tirzepatide, constipation management, and what to eat on tirzepatide apply broadly to GLP-1 class medications. The side effect comparison between semaglutide and tirzepatide provides additional context on GLP-1 tolerability patterns.
Side Effect | Survodutide | Tirzepatide |
|---|---|---|
Nausea | 56% | 24-33% |
Vomiting | 27% | 8-13% |
Diarrhea | 22% | 18-23% |
Constipation | 21% | 11-17% |
Discontinuation rate | 24.6% | 4.3-7.1% |
Dosing schedules compared
Both drugs use once-weekly subcutaneous injection with gradual dose titration. The specifics differ.
Survodutide dosing (based on clinical trials)
Survodutide is not yet commercially available, so dosing information comes from clinical trial protocols.
Phase 2 obesity trial doses: 0.6 mg, 2.4 mg, 3.6 mg, and 4.8 mg once weekly. The 4.8 mg dose showed optimal efficacy for weight loss.
Phase 2 MASH trial doses: 2.4 mg, 4.8 mg, and 6.0 mg once weekly with a 24-week rapid dose-escalation phase followed by a 24-week maintenance phase.
Phase 3 SYNCHRONIZE trials: Using extended, flexible dose escalation. Specific titration schedules are designed to minimize early GI side effects by increasing doses more gradually than in Phase 2.
The optimal dose appears to be 4.8 mg for both obesity and MASH indications. The 6.0 mg dose did not consistently outperform 4.8 mg, likely due to increased side effects and dropouts at the higher dose.
Tirzepatide dosing (FDA-approved schedule)
Period | Dose | Notes |
|---|---|---|
Weeks 1-4 | 2.5 mg | Starting dose (not therapeutic, just initiation) |
Weeks 5-8 | 5.0 mg | First therapeutic dose |
Weeks 9-12 | 7.5 mg | Increase if tolerated |
Weeks 13-16 | 10.0 mg | Common maintenance dose |
Weeks 17-20 | 12.5 mg | Further increase if needed |
Week 21+ | 15.0 mg | Maximum approved dose |
Tirzepatide comes in pre-filled pens, eliminating the need for vial-and-syringe dosing. This is a significant convenience advantage over survodutide, which would likely require manual injection from vials initially (assuming it follows the typical path of newer injectables). For those already working with injectable peptides, the process is familiar. Our guides on how to inject GLP-1 medications and tirzepatide injection technique cover proper administration.
The compounded tirzepatide dosage calculator helps users working with compounded formulations calculate accurate doses, and many people find microdosing tirzepatide useful for managing side effects during the titration phase.
Availability and regulatory status
This is the most practical difference between the two drugs right now.
Tirzepatide: available today
Tirzepatide is FDA-approved and commercially available under two brand names:
Mounjaro for type 2 diabetes (approved May 2022)
Zepbound for chronic weight management (approved November 2023)
It is available by prescription at pharmacies nationwide. Insurance coverage exists for qualifying indications, though coverage varies by plan and diagnosis. Affordable tirzepatide options exist through compounding pharmacies and manufacturer savings programs. Empower Pharmacy and other compounding pharmacies offer tirzepatide formulations, often combined with ingredients like B12, glycine, or niacinamide.
Alternative formulations are also emerging. Oral tirzepatide is in development, and tirzepatide sublingual drops offer a needle-free option through some compounding pharmacies. The oral vs injection comparison helps patients decide which route suits their preferences.
Survodutide: still in clinical trials
Survodutide has no FDA approval for any indication. It is available only through enrollment in clinical trials. The key regulatory milestones:
FDA Breakthrough Therapy designation for MASH (September 2024)
EMA PRIME designation for MASH (November 2023)
China NMPA Breakthrough designation (June 2024)
Phase 3 trials (SYNCHRONIZE-1 and SYNCHRONIZE-2) fully enrolled, results expected H1 of this year
Estimated FDA filing: Late this year at the earliest
Estimated FDA approval: Possibly next year, depending on Phase 3 results
If Phase 3 results confirm Phase 2 findings, survodutide could reach the market within the next 12-24 months. But until then, tirzepatide is the only dual-agonist option available outside of clinical trials.
For researchers tracking the evolving landscape of weight loss peptides, the pipeline includes several other promising compounds. Orforglipron is an oral GLP-1 agonist that could eliminate injections entirely. Retatrutide targets three receptors simultaneously. CagriSema combines amylin and GLP-1 agonism. Understanding these options helps with long-term planning.
Which one is better for weight loss?
Based on current data, tirzepatide produces more total weight loss. The 20.9% mean reduction at 72 weeks exceeds the 14.9% (or 18.7% in completers) seen with survodutide at 46 weeks.
But the answer is not that simple.
Survodutide weight loss was still accelerating at the end of its 46-week trial. If the treatment had continued to 72 weeks, the gap could have narrowed substantially. Phase 3 data from SYNCHRONIZE-1 and SYNCHRONIZE-2, which run for 76 weeks, will provide the first truly comparable duration endpoints.
Tirzepatide also has the advantage of established optimal dosing through extensive Phase 3 trials, while survodutide dosing is still being refined. Phase 3 may identify more effective titration strategies that improve both efficacy and adherence.
For pure weight loss today, tirzepatide wins by default because it is available and survodutide is not. For people not losing weight on tirzepatide, or those who have plateaued on GLP-1/GIP dual agonism, survodutide represents a mechanistically different approach that could work where tirzepatide has stopped working. The glucagon-mediated increase in energy expenditure is a pathway that tirzepatide simply does not activate.
Some research suggests that people who respond well to one dual-agonist mechanism may not respond as well to the other, and vice versa. Individual metabolic profiles, receptor sensitivity, and underlying conditions all influence response. This is exactly why having multiple dual-agonist options available is valuable for the field.
For those exploring alternatives while waiting for survodutide, our guides cover switching between GLP-1 medications, managing tirzepatide plateaus, and peptide stacking strategies for weight loss.
Which one is better for metabolic health?
This is where the comparison gets more nuanced. "Better" depends entirely on which metabolic condition you are trying to address.
For type 2 diabetes
Tirzepatide has proven, FDA-approved efficacy for HbA1c reduction. The SURPASS trials demonstrated reductions up to 2.4 percentage points in patients with type 2 diabetes. It improves insulin sensitivity through both GLP-1 and GIP receptor activation.
Survodutide has shown glycemic improvement in Phase 2 trials with T2D patients (up to 1.71 percentage point HbA1c reduction at 16 weeks with the 2.7 mg dose), but this data is from shorter, smaller studies. SYNCHRONIZE-2 will provide Phase 3 data in patients with both obesity and T2D.
Winner for diabetes: Tirzepatide (proven Phase 3 data, FDA approval, established track record).
For MASH and liver disease
Survodutide has Phase 2 data published in the NEJM showing 62% MASH resolution at the 4.8 mg dose, 62% liver fat reduction at the 6.0 mg dose, and meaningful fibrosis improvement. It has FDA Breakthrough Therapy designation specifically for MASH.
Tirzepatide has shown liver fat reduction in studies, but no specific MASH trial data with histological endpoints comparable to survodutide.
Winner for liver disease: Survodutide (direct mechanism, breakthrough therapy designation, NEJM-published data).
For cardiovascular health
Both drugs are expected to show cardiovascular benefit based on their metabolic effects. Tirzepatide cardiovascular outcome data is more mature. The SYNCHRONIZE cardiovascular outcomes trial (SYNCHRONIZE-CVOT) is ongoing for survodutide.
Winner for cardiovascular health: Too early to call. Both show promise but need more data.
For energy expenditure
Survodutide increases total energy expenditure through glucagon receptor activation, a unique advantage. Tirzepatide does not have this mechanism. For people concerned about metabolic adaptation during weight loss, survodutide theoretical advantage in maintaining energy expenditure could be significant.
Winner for energy expenditure: Survodutide (glucagon-mediated thermogenesis is unique to this mechanism).
The future of dual-agonist therapy
Survodutide and tirzepatide represent two branches of the same evolutionary tree in metabolic medicine. Both prove that targeting multiple receptors simultaneously produces better outcomes than hitting one receptor alone. But they prove it in different ways.
Tirzepatide proved that GLP-1/GIP dual agonism produces superior weight loss and glycemic control compared to GLP-1 alone. Survodutide is proving that GLP-1/glucagon dual agonism may offer unique benefits for liver disease and energy metabolism that GLP-1/GIP cannot match.
The next frontier is triple agonism. Retatrutide hits all three receptors, GLP-1, GIP, and glucagon, simultaneously. Phase 2 data showed up to 24.2% weight loss at 48 weeks, the highest ever recorded for any obesity medication. Understanding its dosage protocols, availability timeline, and expected costs helps researchers plan ahead.
For the broader comparison landscape, mazdutide offers another GLP-1/glucagon dual agonist developed in China with its own emerging dataset. The amylin receptor agonist class represents yet another complementary approach, and combinations like cagrilintide paired with semaglutide (CagriSema) are in late-stage development.
The future likely involves personalized dual-agonist selection based on individual metabolic profiles. A patient with MASH and moderate obesity might benefit most from survodutide. A patient with obesity and type 2 diabetes might respond best to tirzepatide. A patient who has plateaued on both could turn to triple agonism with retatrutide.
This is not a winner-take-all competition. It is an expanding toolkit. And for researchers navigating this landscape, SeekPeptides provides the most comprehensive, up-to-date resource for understanding these options, with evidence-based guides, detailed protocol databases, and a community of experienced researchers who track every development in real time.
Complete comparison summary
Feature | Survodutide | Tirzepatide |
|---|---|---|
Developer | Boehringer Ingelheim | Eli Lilly |
Mechanism | GLP-1/Glucagon dual agonist | GLP-1/GIP dual agonist |
Brand names | None (investigational) | Mounjaro, Zepbound |
FDA approval | Not approved | Approved (diabetes + obesity) |
Max weight loss (trials) | 18.7% at 46 weeks (completers) | 20.9% at 72 weeks |
MASH efficacy | 62% resolution (Phase 2) | Not specifically studied |
Liver fat reduction | Up to 62% from baseline | Indirect (via weight loss) |
Energy expenditure effect | Increases (glucagon-mediated) | No direct effect |
Common side effects | Nausea 56%, vomiting 27% | Nausea 24-33%, vomiting 8-13% |
Discontinuation rate | 24.6% | 4.3-7.1% |
Dosing frequency | Once weekly | Once weekly |
Dose range tested | 0.6-6.0 mg | 2.5-15 mg |
Availability | Clinical trials only | Prescription (pharmacies) |
Expected cost (post-approval) | Est. $1,000-1,400/month | ~$1,060/month (Zepbound) |
Frequently asked questions
Is survodutide more effective than tirzepatide for weight loss?
Current data shows tirzepatide produces slightly more total weight loss (20.9% vs 14.9-18.7%), but the trials used different durations. Survodutide weight loss was still trending downward at week 46, suggesting longer trials could narrow the gap. Phase 3 results expected this year will provide more comparable data.
Can I get survodutide now?
No. Survodutide is only available through clinical trial enrollment. It has not received FDA approval for any indication. The Phase 3 trials (SYNCHRONIZE-1 and SYNCHRONIZE-2) are fully enrolled. The earliest possible commercial availability would be next year, assuming positive Phase 3 results and FDA review.
Does survodutide work on the liver differently than tirzepatide?
Yes, fundamentally. Survodutide activates glucagon receptors in the liver, directly stimulating hepatic fat oxidation. This produces liver fat reductions of up to 62% from baseline, with 62% of patients achieving MASH resolution at the optimal dose. Tirzepatide does not have this direct hepatic mechanism. Its liver benefits come indirectly through weight loss and metabolic improvement.
Which drug has fewer side effects?
Tirzepatide has significantly better tolerability. Discontinuation rates due to adverse events are 4.3-7.1% for tirzepatide versus 24.6% for survodutide. Nausea rates are approximately half with tirzepatide compared to survodutide. However, survodutide Phase 3 trials use slower dose escalation, which may improve these numbers. See our GLP-1 side effects comparison for broader context.
Will survodutide replace tirzepatide?
Unlikely. These drugs serve overlapping but distinct clinical purposes. Tirzepatide excels at weight loss and glycemic control. Survodutide has unique advantages for liver disease and energy expenditure. The future likely involves both drugs coexisting as options for different patient profiles, similar to how semaglutide and tirzepatide both have their place in current practice.
What is the difference between GIP and glucagon receptor activation?
GIP (glucose-dependent insulinotropic polypeptide) receptor activation primarily enhances insulin secretion, improves fat tissue metabolism, and may contribute to central appetite suppression. Glucagon receptor activation primarily increases hepatic energy expenditure, promotes liver fat oxidation, and stimulates thermogenesis. The metabolic effects are complementary but distinct. Our guide on how peptides work covers receptor signaling in more detail.
Could I switch from tirzepatide to survodutide?
In theory, yes, once survodutide becomes commercially available. The drugs target different secondary receptors, so someone who has plateaued on tirzepatide could potentially benefit from survodutide approach to energy expenditure and liver fat metabolism. However, no clinical data exists on switching between these specific drugs. Work with your healthcare provider to evaluate options. For current switching guidance between available GLP-1 medications, see our GLP-1 switching guides.
Is retatrutide better than both survodutide and tirzepatide?
Retatrutide targets all three receptors (GLP-1, GIP, and glucagon) simultaneously, and Phase 2 data showed up to 24.2% weight loss at 48 weeks. It theoretically combines the advantages of both tirzepatide and survodutide. However, it is even earlier in development than survodutide, with Phase 3 trials ongoing. More receptors does not automatically mean better, as the side effect profile and optimal dosing still need to be established.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your liver stay healthy, and your metabolic toolkit stay ever-expanding.