Feb 5, 2026

The gap between those two experiences is not random. It is not luck. And it has almost nothing to do with willpower. The speed at which semaglutide works depends on a specific set of biological, pharmacological, and behavioral factors that most guides completely ignore. They hand you a timeline, tell you to be patient, and leave it at that. That is not good enough. Not when you are injecting yourself weekly and watching the scale with increasing frustration. Not when your friend lost 15 pounds in two months and you have barely lost four.
Here is what actually determines your results. Your starting weight matters. Your metabolic health matters. Your dose escalation schedule matters enormously. But the factor that separates fast responders from slow ones is something far less obvious, and clinical trial data from the STEP program reveals exactly what it is. This guide breaks down every dimension of how semaglutide works, not just appetite suppression timelines, but the full picture. Weight loss velocity by month. Body composition shifts. Metabolic marker improvements. Cardiovascular protection. Side effect patterns. And the specific week-by-week trajectory backed by data from trials involving tens of thousands of participants.
Whether you are two weeks in and wondering if anything is happening, or six months in and hitting a plateau, this is the reference you need. SeekPeptides built this guide from clinical evidence and real-world outcomes so you can stop guessing and start understanding exactly what semaglutide is doing inside your body right now.
What determines how fast semaglutide actually works
Speed is a misleading word when it comes to GLP-1 receptor agonists. Semaglutide does not flip a switch. It builds. The drug reaches pharmacological steady state after four to five weekly injections at any given dose, which means the concentration in your bloodstream needs roughly a month to stabilize. Before that point, every injection is building toward the therapeutic level your body needs to respond meaningfully.
The half-life of semaglutide is approximately 168 hours. One full week. That is why it works as a once-weekly injection, unlike older GLP-1 agonists that required daily dosing. But that long half-life also means accumulation takes time, and dose escalation extends the timeline further.
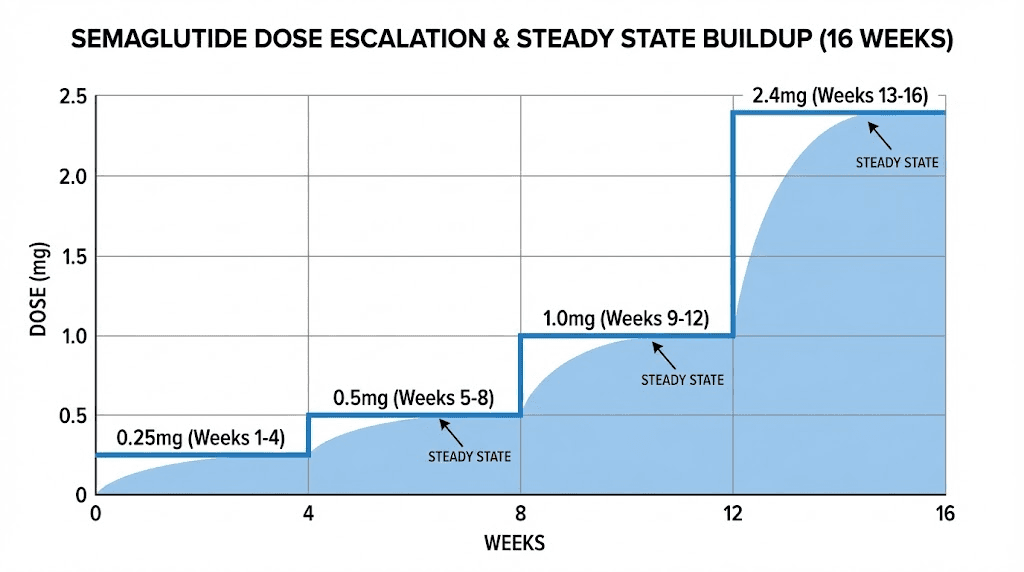
Most protocols start at 0.25 mg for the first four weeks. This is not a therapeutic dose. It exists solely to let your gastrointestinal system adjust. Then you move to 0.5 mg for another four weeks. Some people begin noticing effects here. The clinical dose for weight management is 2.4 mg, which most people do not reach until week 16 or 17 of their protocol. So when someone asks how fast semaglutide works, the honest answer depends entirely on which effect you are measuring and where you are in the dose escalation process. Understanding how long peptides take to work requires patience and context, and semaglutide is no exception.
Pharmacokinetics in plain language
After a subcutaneous injection, semaglutide absorbs slowly from the injection site. Peak plasma concentration occurs between 24 and 72 hours post-injection. The molecule binds to albumin in your blood, which protects it from degradation and gives it that unusually long half-life. This albumin binding is what makes weekly dosing possible.
But here is the part most people miss. Because the half-life is seven days, each new injection adds to what remains from the previous one. After your first injection of 0.25 mg, roughly half remains when you inject the second dose. After the second, you have the new dose plus the remnant. By injection four or five, the amount in your blood has reached a stable plateau, what pharmacologists call steady state. Only at steady state are you getting the full effect of that dose level.
This means the first real therapeutic window opens around weeks four to five at your current dose. When you escalate, the clock resets. New steady state. New adjustment period. New side effects, potentially. Understanding this timeline helps explain why results feel nonlinear, why you might feel nothing for three weeks and then suddenly everything shifts. Your body is not being unpredictable. The pharmacokinetics are simply playing out exactly as the chemistry dictates. For those who want to understand the precise measurements involved, our semaglutide dosage calculator can help you track where you are in the process, and our broader peptide calculator covers other compounds you may be considering alongside your protocol.
The dose escalation timeline and what happens at each stage
The standard semaglutide dose escalation for weight management follows a strict schedule. Each step lasts four weeks. Rushing it does not produce faster results. It produces worse side effects and higher discontinuation rates.
Weeks 1 through 4: the 0.25 mg introduction
This is the onboarding phase. The dose is sub-therapeutic for weight loss. Its sole purpose is gastrointestinal adaptation. Your GLP-1 receptors in the gut, pancreas, and brain are beginning to respond to the exogenous agonist, but the concentration is too low to produce dramatic effects.
What you might notice: mild appetite reduction, occasional nausea after meals, subtle changes in food preferences. Some people feel nothing at all. That is normal and expected. The peptide dosing guide principles apply here, starting low is always smarter than starting aggressively. If you are experiencing significant nausea or vomiting at this dose, it may signal that your tolerability profile needs monitoring.
Weight loss at this stage averages 1 to 2 percent of body weight. For a 200-pound individual, that is 2 to 4 pounds. Do not panic if the number is zero. The machinery is warming up.
Weeks 5 through 8: the 0.5 mg transition
Doubling the dose brings the first meaningful changes for many users. Appetite suppression becomes more noticeable. Portions decrease without conscious effort. The concept of "food noise," that constant background chatter about what to eat next, begins to quiet for some people. Others will not notice this shift until higher doses.
Nausea tends to return briefly after this escalation. It typically peaks during the first week at the new dose and settles by week two or three. This pattern repeats at every dose increase, and understanding it prevents unnecessary worry. The common mistakes beginners make often include panicking at side effects that are actually normal dose-adjustment responses.
By week 8, cumulative weight loss averages 3 to 5 percent of starting body weight. Metabolic improvements are beginning beneath the surface. Weight loss peptide research consistently shows that the visible results lag behind the invisible biochemical changes by several weeks.
Weeks 9 through 12: the 1.0 mg escalation
Now the effects compound noticeably. At 1.0 mg, most users report a clear reduction in hunger between meals, less interest in calorie-dense foods, and earlier satiety during meals. The GLP-1 receptor activation in the hypothalamus is reaching concentrations that meaningfully alter appetite signaling. Gastric emptying slows further, keeping food in the stomach longer and extending the feeling of fullness after eating.
Weight loss accelerates. Clinical data shows approximately 5 to 7 percent body weight reduction by this point. For our 200-pound example, that is 10 to 14 pounds. The trajectory is steepening, not plateauing. Blood sugar improvements become measurable if you are tracking them. Fasting glucose drops. Postprandial spikes flatten. Researchers monitoring peptides for fat loss often note that this dose level marks the transition from "something might be happening" to "something is definitely happening."
Weeks 13 through 16: the 1.7 mg push
Side effects may resurface briefly. The nausea pattern is familiar by now: a few days of discomfort after the dose increase, followed by adaptation. But the appetite suppression at 1.7 mg is substantially stronger than at 1.0 mg. Many users describe this as the dose where food genuinely stops occupying mental space. Meal planning becomes simpler because cravings have diminished to near silence.
Weight loss by week 16 typically reaches 7 to 10 percent of starting body weight. The rate of loss, roughly 1 to 2 percent per month, has been consistent since the beginning. What changes at this dose is not necessarily the speed but the sustainability. The appetite suppression is strong enough that caloric reduction feels effortless rather than forced. Understanding the best peptide stack for weight loss context helps you appreciate why semaglutide mono-therapy produces these results without requiring additional compounds for most people.
Week 17 onward: the 2.4 mg maintenance dose
This is the full therapeutic dose for weight management. Maximum GLP-1 receptor occupancy. Maximum appetite suppression. Maximum metabolic effect.
From here, weight loss continues but the rate gradually slows. Not because the drug stops working, but because your body reaches a new equilibrium between energy intake, expenditure, and metabolic adaptation.
The STEP 1 trial showed weight loss continuing from this point until approximately week 60, when average loss reached 14.9 percent of body weight. That is nearly 30 pounds for a 200-pound starting weight, achieved over roughly 15 months.
The trajectory from week 17 to week 60 is a gradual curve, not a straight line. The steepest losses occur in the first six months at full dose. The remaining months produce smaller incremental reductions as the body approaches its new set point. For broader context on the best peptides for weight loss, semaglutide at 2.4 mg remains one of the most effective single agents available, with results rivaling some combination approaches.
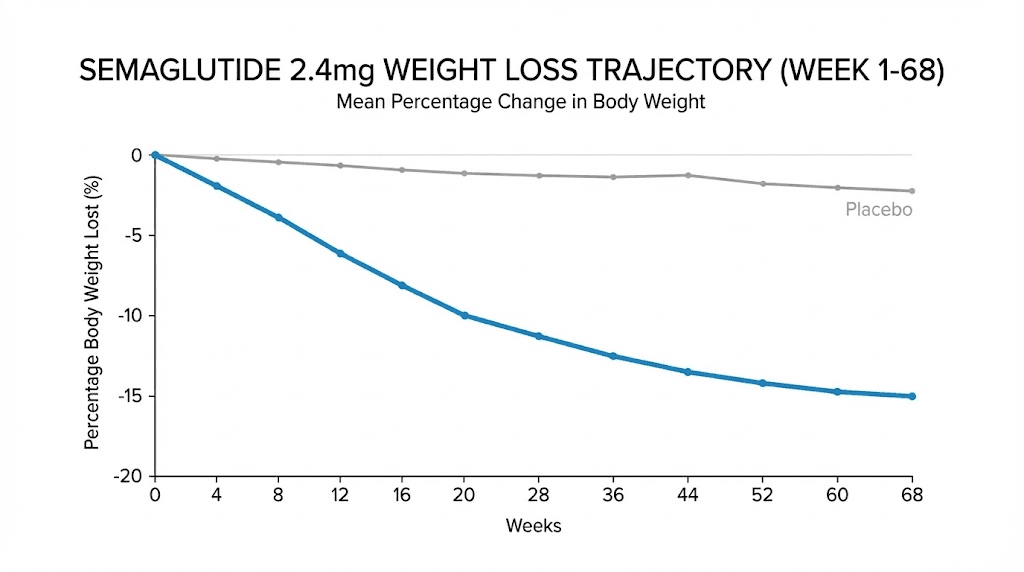
Weight loss results by month with clinical trial data
Abstract percentages are hard to feel. Let us translate the clinical data into concrete monthly expectations, drawn primarily from the STEP 1 trial, which remains the landmark study for semaglutide 2.4 mg in weight management.
Month 1: the slow start that means everything
Average weight loss: approximately 2 percent of body weight, with some trial participants losing up to 3.8 percent by week 4. For a 220-pound person, that translates to roughly 4.4 to 8.4 pounds. Most of this comes from reduced caloric intake rather than dramatic metabolic shifts, though visceral fat mobilization has already begun at the cellular level.
This month tests patience. The scale moves slowly. Friends and family do not notice. You might wonder if the injection is doing anything. It is. The GLP-1 receptor activation is reshaping your appetite neurocircuitry, slowing gastric emptying, and beginning to improve insulin sensitivity. These changes precede visible weight loss by weeks.
Month 2: momentum builds
Average cumulative loss: 4 to 6 percent. The rate is accelerating because you have transitioned to a higher dose (0.5 mg by now, possibly 1.0 mg depending on your protocol). Food noise reduction becomes apparent for most people. Portion sizes shrink naturally. The effort required to maintain a caloric deficit decreases noticeably.
For researchers tracking before and after results, month 2 is where the first visual changes often appear. Facial bloating reduces. Clothes fit differently. The mirror starts confirming what the scale has been suggesting.
Month 3: the confirmation point
Average cumulative loss: approximately 9.6 percent of starting body weight. This is the milestone where clinical benefit becomes unambiguous. For a 220-pound individual, 9.6 percent represents 21 pounds. Blood pressure often shows improvement by now. Fasting glucose has dropped measurably. Energy levels tend to increase as the metabolic burden of excess adiposity decreases.
The three-month mark also brings important body composition changes. Studies using dual-energy X-ray absorptiometry (DXA) show that fat mass loss is the dominant component of weight reduction at this stage. The body is preferentially burning stored fat, particularly visceral fat surrounding the organs. This is the most metabolically dangerous fat depot, and its reduction drives many of the cardiovascular and metabolic improvements that appear in bloodwork.
Month 6: the halfway mark with impressive numbers
Average cumulative loss: approximately 13.8 percent of body weight. The rate has slowed somewhat from the first three months, now running closer to 1 to 1.5 percent per month rather than the initial 2 percent pace. This deceleration is normal. It reflects metabolic adaptation, reduced body mass requiring fewer calories, and the body defending against further weight loss through hormonal adjustments.
At six months, the STEP 1 data shows fat mass reduced by 2.1 kg more than lean mass changes would predict. Visceral fat decreases by roughly 2.0 percent relative to total body mass. The ratio of fat to lean tissue shifts favorably, with lean body mass proportion increasing by approximately 3.0 percent. This matters enormously. You are not just lighter. You are compositionally different. Maintaining muscle while losing fat is one of the critical challenges in any weight loss protocol, and semaglutide performs better than caloric restriction alone on this measure.
Months 9 through 12: approaching the plateau
Weight loss continues but at a progressively slower rate. The cumulative average approaches 14 to 15 percent. Monthly losses may dip below 1 percent. This is not failure. This is physiology. Your body has found a new equilibrium at a significantly lower weight, and the metabolic, hormonal, and neurological adaptations that defend body weight are fully engaged.
The practical implication: if you started at 220 pounds, you are now around 187 to 189 pounds. That is 31 to 33 pounds lost. Your cardiovascular risk profile has likely improved substantially. Your metabolic markers may have normalized. The functional benefits, easier movement, better sleep, improved joint comfort, are tangible daily improvements. For those researching the broader landscape of weight loss peptides for women or fat burning peptides for men, semaglutide month-over-month data provides a realistic benchmark for what consistent GLP-1 agonist use can deliver.
The full clinical picture in one table
Timepoint | Average weight loss (%) | Example (220 lb start) | Monthly rate | Key milestone |
|---|---|---|---|---|
Month 1 | ~2% | ~4.4 lb | ~2% | Appetite changes begin |
Month 3 | ~9.6% | ~21 lb | ~2%/mo | Visible body changes |
Month 6 | ~13.8% | ~30 lb | ~1.5%/mo | Fat mass shift confirmed |
Month 12 | ~14.9% | ~33 lb | ~0.5%/mo | Approaching plateau |
Month 15 (wk 60) | ~14.9% | ~33 lb | Plateau | Maximum effect (nadir) |
These are averages. Individual variation is substantial. The STEP 1 trial showed that 86.4 percent of participants achieved at least 5 percent weight loss, 69.1 percent reached 10 percent, 50.5 percent hit 15 percent, and 32 percent achieved 20 percent or greater weight loss. Your position on this spectrum depends on factors we will explore shortly.
Appetite suppression and food noise reduction timeline
Weight loss is the outcome most people measure. But appetite suppression is the mechanism that drives it. Understanding when and how semaglutide changes your relationship with food is essential for setting realistic expectations.
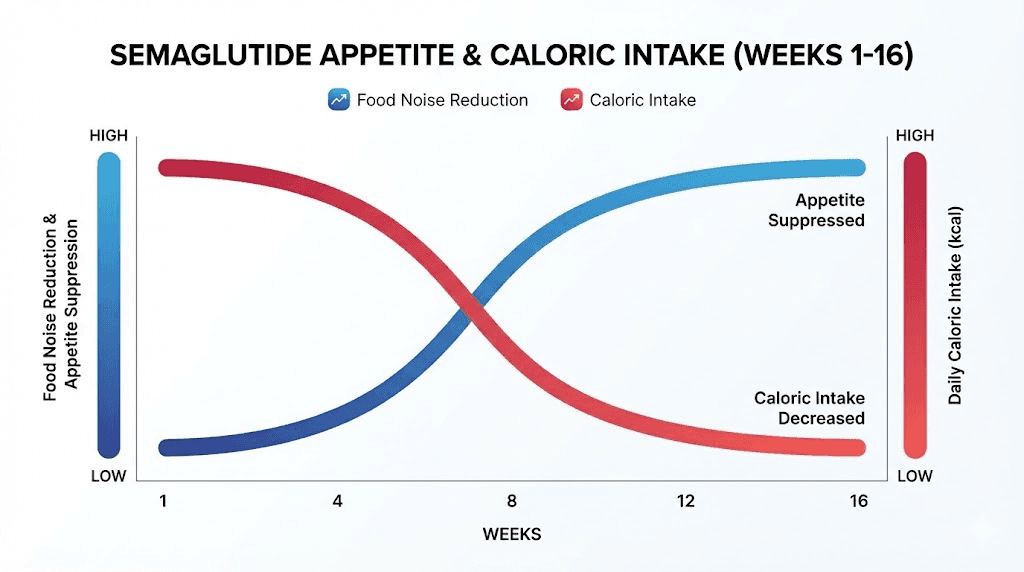
Weeks 1 through 3: subtle shifts
Most users describe the initial appetite changes as subtle. You finish a meal and realize you left food on the plate without thinking about it. A snack you normally reach for mid-afternoon does not cross your mind. These are not dramatic moments. They are quiet absences, things that used to happen that simply stop happening.
The science behind this involves semaglutide activating GLP-1 receptors in the nucleus tractus solitarius and the hypothalamic arcuate nucleus. These brain regions regulate hunger signaling and satiety. At 0.25 mg, the activation is partial. Enough to nudge the system. Not enough to overhaul it. For a deeper exploration of how long it takes for semaglutide to suppress appetite specifically, our dedicated article covers the appetite pathway in granular detail.
Weeks 4 through 8: food noise begins to quiet
"Food noise" has become the colloquial term for the persistent mental preoccupation with eating. What to eat next. When the next meal is. Whether there are snacks available. For people who have struggled with weight, this noise can be relentless, exhausting, and all-consuming.
Semaglutide turns down the volume. Between weeks 4 and 8, as the dose increases to 0.5 mg and the drug reaches steady state at that level, the mental chatter around food diminishes significantly. The Blundell study quantified this effect: a 24 percent reduction in total daily energy intake and a 35 percent reduction in ad libitum lunch intake. These numbers did not come from willpower. They came from neurochemistry. The participants were not trying harder. They were simply less driven to eat.
This is the period when many people report a profound psychological shift. Food stops being an adversary. Meals become functional rather than emotional events. The relief is difficult to overstate for those who have spent years or decades fighting constant hunger.
Weeks 8 through 16: appetite suppression at therapeutic levels
As the dose climbs to 1.0 mg, 1.7 mg, and eventually 2.4 mg, the appetite suppression deepens. Most users at the full dose describe their appetite as fundamentally different from pre-treatment. Not absent, which would be concerning, but manageable. Controllable. Proportionate to actual physiological need rather than driven by emotional or hedonic triggers.
Gastric emptying slows progressively with increasing doses, which means food stays in the stomach longer, prolonging physical fullness. Combined with the central appetite suppression in the brain, the result is a dual mechanism that reduces both the desire to eat and the physical capacity to overeat. Understanding what peptides are used for in the context of appetite and metabolism helps frame semaglutide within the broader category of signaling molecules that influence fundamental body processes. For researchers comparing options, the semaglutide versus tirzepatide comparison reveals how dual-agonist approaches may amplify these effects further.
Body composition changes: fat versus lean mass
Stepping on a scale tells you one number. It does not tell you what you lost. And with any weight loss intervention, the composition of that loss matters as much as the magnitude. Lose too much muscle and your metabolism drops, your functional capacity declines, and long-term weight maintenance becomes nearly impossible.
Semaglutide data on body composition is encouraging, though not without nuance.
What the DXA scans reveal
In the STEP 1 trial, researchers used DXA imaging to assess body composition changes. The results showed fat mass reduced by 3.5 percent relative to total body mass over the study period. Visceral fat, the metabolically active fat surrounding abdominal organs, decreased by 2.0 percent relative to total body mass. Meanwhile, lean body mass proportion actually increased by 3.0 percent. Read that carefully. The proportion of lean mass went up. The absolute amount of lean tissue did decrease somewhat, as it does in virtually any weight loss scenario, but the ratio shifted favorably toward muscle and away from fat.
At six months, fat mass reduction averaged 2.1 kg beyond what proportional weight loss alone would predict. This suggests semaglutide preferentially targets fat stores rather than causing indiscriminate tissue loss. The mechanism likely involves improved insulin sensitivity directing fuel utilization toward fat oxidation, combined with reduced caloric intake that still provides adequate protein for muscle maintenance in most cases.
The muscle preservation question
This is the concern that dominates online discussion. Does semaglutide cause muscle loss? The short answer: some lean mass loss occurs, as it does with any caloric deficit, but the proportion is better than diet-alone approaches. The longer answer involves understanding that maintaining muscle during weight loss requires two things that semaglutide does not directly provide: adequate protein intake and resistance training.
The drug reduces appetite. If appetite reduction leads to inadequate protein consumption, muscle loss will be more pronounced. This is a behavioral factor, not a pharmacological one. Researchers who prioritize protein intake (1.2 to 1.6 grams per kilogram of body weight daily) and engage in regular resistance exercise tend to preserve significantly more lean mass during semaglutide treatment. Understanding peptides for weight loss and muscle gain in tandem helps contextualize why some researchers pair semaglutide with compounds that support lean tissue preservation.
For those exploring the intersection of body composition optimization and peptide research, the peptides for muscle growth landscape offers complementary approaches. Growth hormone secretagogues like ipamorelin and CJC-1295 work through entirely different pathways that support lean tissue. While semaglutide handles the fat loss side of the equation, other safe peptides for muscle growth can address the anabolic side.
Timeline of body composition shifts
Weeks 1 through 4: minimal measurable composition change. Weight loss is predominantly water and early fat mobilization.
Weeks 4 through 12: fat mass begins declining measurably. Visceral fat reduction outpaces subcutaneous fat reduction. Lean mass remains relatively stable if protein intake is adequate.
Months 3 through 6: the most significant fat mass reduction occurs. DXA scans show clear reductions in trunk fat, visceral adipose tissue, and subcutaneous abdominal fat. Lean mass may decrease slightly in absolute terms but increases as a proportion of total body mass.
Months 6 through 12: composition changes continue at a slower pace. The body approaches a new equilibrium. Fat distribution patterns shift, with preferential loss from central deposits. This is precisely the pattern that correlates with reduced cardiovascular risk, reduced diabetes risk, and improved metabolic function. The peptide calculator for weight loss can help researchers plan dosing protocols that align with these body composition timelines.
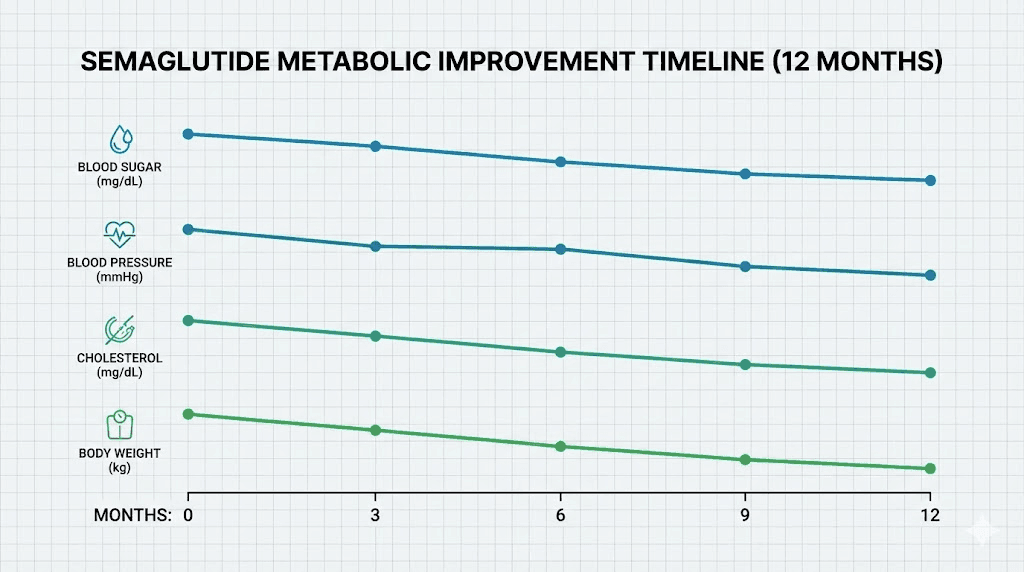
Metabolic improvements: blood sugar, A1C, cholesterol, and blood pressure
Weight loss is visible. Metabolic improvement is invisible. But the invisible changes may be more important for long-term health than the number on the scale. Semaglutide was originally developed as a diabetes medication, and its metabolic effects are profound and well-documented.
Blood sugar and A1C timeline
If you have elevated blood sugar or type 2 diabetes, semaglutide begins improving glycemic control almost immediately through direct pancreatic effects. The drug stimulates glucose-dependent insulin secretion and suppresses inappropriate glucagon release. These effects begin with the first injection, though they take weeks to produce measurable changes in HbA1c.
Significant blood sugar reductions typically become apparent after eight or more weeks of treatment. The magnitude depends on the dose. At 0.5 mg weekly, studies show HbA1c reductions of 1.2 to 1.5 percentage points. At 1.0 mg weekly, reductions reach 1.4 to 1.8 percentage points. For context, a 1.5-point reduction in HbA1c is clinically transformative, often representing the difference between uncontrolled and well-managed diabetes.
Even for individuals without diabetes, semaglutide improves insulin sensitivity and reduces fasting glucose. These changes correlate with reduced risk of developing type 2 diabetes and improved metabolic flexibility, the ability to efficiently switch between burning carbohydrates and fats as fuel sources. The peptide dosage chart resources can help researchers understand how different dose levels correlate with different magnitudes of metabolic response.
Cholesterol and lipid changes
Lipid panels begin improving within the first three months, though the full effect takes six months or longer to manifest. Semaglutide consistently reduces triglycerides, a direct consequence of reduced caloric intake and improved insulin sensitivity. LDL cholesterol shows modest reductions in most studies. HDL cholesterol may increase slightly, though this effect is less consistent.
The lipid improvements are partly weight-dependent and partly direct pharmacological effects. GLP-1 receptor activation in the liver influences lipoprotein metabolism independently of weight loss. This means some lipid improvement occurs even in participants who lose minimal weight, though greater weight loss produces greater lipid improvement. For a comprehensive understanding of how metabolic health intersects with peptide research, the peptides for hormone balance guide explores how multiple signaling pathways influence metabolic markers.
Blood pressure reduction
Blood pressure decreases gradually over the first six months. The mechanism is multifactorial: weight loss reduces the mechanical load on the cardiovascular system, improved insulin sensitivity reduces sodium retention, and direct vascular effects of GLP-1 receptor activation promote vasodilation.
In the SELECT trial, semaglutide-treated participants showed clinically meaningful reductions in both systolic and diastolic blood pressure. The improvement was evident by month 3 and continued through month 12. For individuals with borderline or stage 1 hypertension, semaglutide-induced weight loss may be sufficient to normalize blood pressure without additional antihypertensive medication.
The timeline for metabolic improvements follows a consistent pattern: early changes in glucose metabolism (weeks 4 to 8), followed by blood pressure improvements (months 2 to 6), followed by lipid optimization (months 3 to 12). This layered progression means that comprehensive metabolic panel improvement takes six months to a year to fully materialize, even though individual markers begin shifting much earlier.
Cardiovascular protection: what the SELECT trial revealed
The SELECT trial changed how physicians think about semaglutide. It was not a weight loss trial. It was a cardiovascular outcomes trial, designed to determine whether semaglutide could prevent heart attacks, strokes, and cardiovascular death in overweight or obese individuals with established cardiovascular disease.
The results were decisive.
The headline numbers
Semaglutide produced a 20 percent reduction in major adverse cardiovascular events (MACE), the composite of cardiovascular death, nonfatal heart attack, and nonfatal stroke. The primary composite outcome occurred in 6.5 percent of the semaglutide group compared to 8.0 percent in the placebo group over the trial period. A 1.5 percentage point absolute reduction and a 20 percent relative reduction.
Beyond the primary endpoint, the trial revealed a 22 percent reduction in a composite kidney endpoint, suggesting renal protective effects. Perhaps most striking was a 42 percent reduction in atrial fibrillation risk. Atrial fibrillation is strongly associated with obesity and metabolic dysfunction, and its reduction with semaglutide treatment suggests deep physiological remodeling beyond simple weight loss.
Improvements extended to inflammatory markers. C-reactive protein, a marker of systemic inflammation, decreased significantly. Waist circumference reduced substantially, reflecting the visceral fat loss that drives cardiovascular risk. These are not minor improvements. They represent fundamental shifts in cardiovascular risk architecture.
How fast do cardiovascular benefits appear
This is the nuanced question. The SELECT trial measured outcomes over years, not weeks. Cardiovascular event reduction is not something you feel on a daily basis. It manifests statistically in large populations over extended timeframes.
However, the mechanistic improvements that underlie cardiovascular protection begin much earlier. Blood pressure drops within months. Inflammatory markers decrease within the first three to six months. Lipid profiles improve over similar timeframes. Visceral fat, the primary driver of obesity-related cardiovascular risk, begins declining within weeks of starting treatment.
The practical takeaway: you will not "feel" cardiovascular protection. But the biological changes that produce it start accumulating from the first month of treatment. By six months, the metabolic environment surrounding your cardiovascular system is fundamentally different from baseline. The SELECT data simply quantifies the long-term clinical benefit of those early changes sustained over time. Researchers interested in complementary cardiovascular support may find the BNP level guide informative for understanding cardiac biomarkers, while longevity peptides cover the broader landscape of compounds that influence long-term health outcomes.
The side effects timeline: when they appear and when they fade
Side effects are the price of admission. Knowing when to expect them and when they pass is the difference between tolerating the protocol and abandoning it prematurely.
Week 1 through 2: initial onset
Nausea is the most common side effect and typically appears within the first few days of the initial injection. For some users, it manifests as a persistent mild queasiness. For others, it occurs specifically after meals, particularly large or fatty meals. The severity varies enormously between individuals. Some feel nothing. Others spend several days genuinely uncomfortable.
Constipation may also begin in the first two weeks, a consequence of slowed gastric motility. Reduced appetite can contribute by decreasing overall food and fiber intake. Gut health peptide research highlights the importance of gastrointestinal function during any protocol that affects digestive motility.
Weeks 2 through 4: peak and adaptation
Side effects tend to peak during weeks 2 through 4 of any given dose level. This is when the drug has reached or is approaching steady state at that dose, meaning maximum plasma concentration for that amount. Your body is adjusting to a new level of GLP-1 receptor activation across multiple organ systems.
By week 3 or 4 at the starting dose, most users report significant improvement in nausea. The gut adapts. Receptor sensitivity adjusts. The initial shock to the system fades. This adaptation is predictable and consistent across clinical trials. Understanding semaglutide withdrawal symptoms and side effect management helps researchers navigate both the adjustment period and any future protocol changes.
The dose escalation pattern: side effects recur
Here is what catches people off guard. Every time you increase the dose, the side effect cycle restarts. You feel fine at 0.5 mg, escalate to 1.0 mg, and the nausea returns. This is normal. Expected. Temporary.
The pattern: nausea returns within days of the dose increase, peaks during the first one to two weeks at the new dose, and improves over the following two to three weeks as your body adapts to the new steady state. Each cycle tends to be shorter and less intense than the previous one, because your GLP-1 receptors have partially adapted from the lower dose. But it still happens, and knowing this prevents the common mistake of thinking the medication has "stopped agreeing" with you.
For those experiencing persistent side effects, the troubleshooting guide for semaglutide covers strategies for managing common complaints, and our peptide injection guide addresses technique factors that can influence tolerability.
Common side effects and their typical resolution timeline
Side effect | Typical onset | Peak severity | Resolution | Management |
|---|---|---|---|---|
Nausea | Days 1-3 | Weeks 2-4 | Weeks 4-6 | Smaller meals, avoid fatty foods |
Constipation | Week 1-2 | Weeks 3-4 | Ongoing for some | Fiber, hydration, mild laxatives |
Diarrhea | Week 1-2 | Weeks 2-3 | Weeks 3-4 | Hydration, bland diet |
Fatigue | Week 1 | Weeks 2-3 | Weeks 4-6 | Adequate calories and rest |
Injection site reaction | Immediately | Hours | Days | Rotate sites, proper technique |
Headache | Days 1-3 | Week 1 | Weeks 2-3 | Hydration, electrolytes |
The overarching theme: most gastrointestinal side effects are transient and self-limiting. They improve as the body adapts to each dose level. Severe or persistent symptoms warrant medical evaluation, but the typical experience is discomfort that fades rather than worsens over time.
Why some people see faster results than others
The STEP trial data represents averages. But averages conceal an enormous range. Some participants lost over 20 percent of their body weight. Others lost less than 5 percent. Understanding the factors that separate fast responders from slow responders can help you optimize your own trajectory.
Starting body weight and BMI
Higher starting weight generally predicts greater absolute weight loss but not necessarily greater percentage loss. A 300-pound individual may lose 45 pounds (15 percent) while a 180-pound individual loses 22 pounds (12 percent). The absolute number is larger, but the percentage is similar. The dosage calculation for semaglutide does not adjust for body weight (unlike many peptides), which means heavier individuals receive a proportionally lower dose per kilogram. This partly explains why percentage-based losses converge across weight categories.
Insulin resistance and metabolic status
Individuals with significant insulin resistance, including those with type 2 diabetes or prediabetes, often experience slower initial weight loss. The metabolic dysfunction that accompanies insulin resistance creates hormonal barriers to fat mobilization. As semaglutide improves insulin sensitivity over weeks and months, these barriers diminish and weight loss accelerates. Paradoxically, the metabolically sickest individuals may see the most dramatic improvements in health markers even if their scale-based weight loss is slightly slower.
Dietary composition and protein intake
Semaglutide suppresses appetite but does not dictate food choices. Individuals who maintain adequate protein intake (1.2 to 1.6 grams per kilogram daily) tend to preserve more lean mass, which supports ongoing metabolic rate and continued fat loss. Those who allow protein intake to drop dramatically may experience faster initial scale weight loss (because they are losing muscle in addition to fat) but hit plateaus sooner as metabolic rate declines. The peptide cycle planning guide discusses how to structure protocols to optimize outcomes, and the same principles of nutritional support apply during semaglutide use.
Physical activity levels
Exercise does not make semaglutide work faster, per se. But it amplifies and extends the results. Resistance training preserves lean mass. Cardiovascular exercise improves the metabolic adaptations that semaglutide initiates. The combination of pharmacological appetite suppression and regular physical activity produces outcomes that exceed either intervention alone. Peptides for athletic performance and energy-boosting peptides can complement a fitness regimen during the adaptation period when energy may feel lower.
Adherence to the dose escalation schedule
This is the most controllable factor. Missing injections disrupts steady state. Skipping doses delays the accumulation needed for therapeutic effect. Irregular dosing creates valleys in plasma concentration where appetite suppression weakens and side effects may worsen when dosing resumes. Consistency is not glamorous, but it is the single most important behavioral predictor of results.
Genetics and individual variation
GLP-1 receptor density, distribution, and sensitivity vary between individuals. Some people are naturally more responsive to GLP-1 agonism. Polymorphisms in the GLP-1 receptor gene and related pathways influence how strongly the body responds to semaglutide. This is not something you can control or test for clinically, but it explains why two people on identical protocols can have different outcomes. The mechanism of peptide action differs across individuals based on receptor expression and downstream signaling efficiency.
What to do if semaglutide seems slow to work
Patience is necessary. Premature troubleshooting is not. But if you have been on semaglutide for eight or more weeks and are seeing minimal response, there are evidence-based adjustments worth considering before concluding the medication is not working.
Verify your dose is actually reaching therapeutic levels
If you are still at 0.25 or 0.5 mg, you are not at a therapeutic weight loss dose. Full appetite suppression and metabolic effects require higher doses for most people. Give the escalation schedule time. Judging semaglutide at sub-therapeutic doses is like judging an antibiotic after one day, you have not given it enough time or enough drug to work.
For researchers using compounded or research-grade semaglutide, verify the concentration and dosing accuracy. Our guide to semaglutide units and milligrams and the 40 units semaglutide conversion guide can help ensure accurate dosing. Using the peptide reconstitution calculator eliminates math errors that could result in under-dosing.
Assess caloric intake honestly
Semaglutide reduces appetite but does not prevent overeating. If you are consuming calorie-dense foods (liquid calories, high-fat snacks, alcohol) that bypass the satiety signals, your caloric intake may remain higher than you realize. Track your intake for one week using a food diary or app. The data often reveals patterns that appetite suppression alone cannot overcome.
Check protein intake
Low protein intake during weight loss causes disproportionate lean mass loss, which lowers resting metabolic rate, which slows further weight loss. It is a vicious cycle that semaglutide alone cannot break. Aim for 1.2 to 1.6 grams of protein per kilogram of body weight daily, prioritizing protein at every meal.
Rule out medical factors
Hypothyroidism, PCOS, certain medications (steroids, some antidepressants, insulin), and other medical conditions can impede weight loss even on semaglutide. If your response seems disproportionately low, a medical evaluation can identify treatable contributing factors. The peptide therapy near me guide can help you find qualified practitioners who understand how to optimize GLP-1 agonist therapy in the context of complex medical histories.
Consider whether expectations are calibrated
Two percent per month for the first six months is the average rate. If you are losing at that rate but expected five percent per month, the problem is not semaglutide. It is expectations. Recalibrate to the clinical evidence. Even the STEP 1 trial, which produced remarkable results, showed a gradual trajectory rather than rapid transformation. Sustainable weight loss measured in months, not weeks, is the hallmark of an approach that will actually last.
Comparing semaglutide speed to alternatives
Semaglutide is not the only GLP-1 based weight loss compound in research. How does its speed compare to the alternatives? Understanding these comparisons helps contextualize whether semaglutide is the right fit for a particular research protocol, or whether faster-acting alternatives exist.
Semaglutide versus tirzepatide
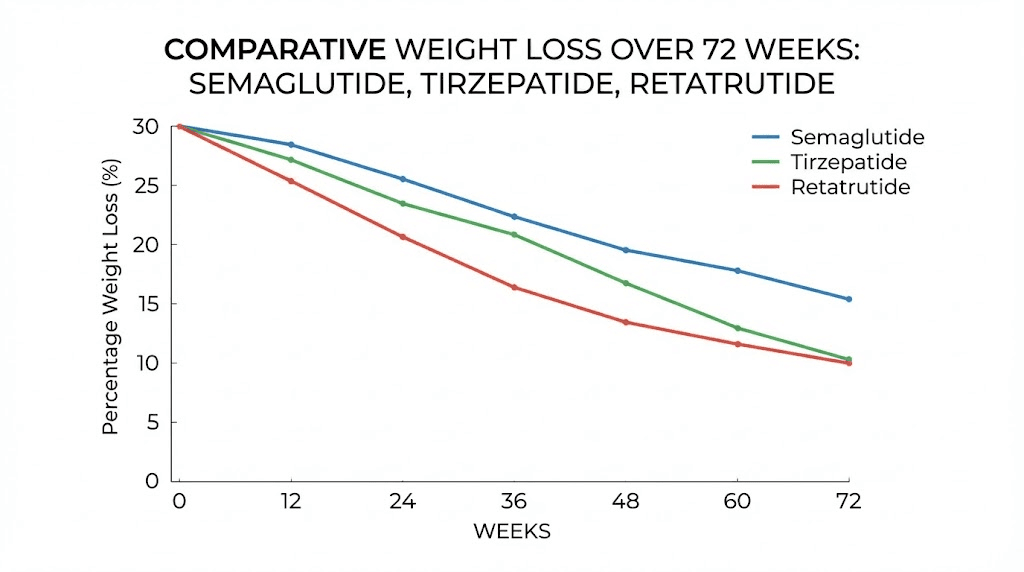
Tirzepatide versus semaglutide is the comparison that dominates current research discussions. Tirzepatide is a dual GIP/GLP-1 receptor agonist, meaning it activates two incretin pathways rather than one. The SURMOUNT trials showed tirzepatide producing average weight loss of 20 to 22.5 percent at the highest dose over 72 weeks, compared to semaglutide 14.9 percent in STEP 1.
Does tirzepatide work faster? The data suggests yes, marginally. Weight loss velocity with tirzepatide appears to be approximately 15 to 20 percent greater than semaglutide at comparable timepoints. The dual mechanism drives more pronounced appetite suppression and potentially greater metabolic improvement. For those exploring tirzepatide, our tirzepatide before and after analysis, microdosing tirzepatide chart, and tirzepatide dosing guide provide practical context. The tirzepatide unit conversion guide helps with accurate dosing, and our coverage of oral tirzepatide explores emerging delivery methods. If fatigue is a concern during treatment, our tirzepatide fatigue guide addresses this common side effect.
Semaglutide versus retatrutide
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors. Early phase 2 data showed average weight loss of approximately 24 percent at 48 weeks, with the STEP UP trial demonstrating 20.7 percent average weight reduction with the higher dose formulation at 72 weeks. If these results hold in phase 3 trials, retatrutide may be substantially faster than semaglutide in producing comparable or greater weight loss.
The triple agonism introduces glucagon receptor activation, which directly increases energy expenditure, a mechanism neither semaglutide nor tirzepatide possess. This thermogenic component could accelerate fat loss beyond what appetite suppression alone achieves. Our retatrutide versus semaglutide comparison breaks down the differences in detail, while the retatrutide dosage chart, dose guide, dose schedule, dosage calculator, and cost guide provide comprehensive coverage for researchers considering this compound.
Semaglutide versus cagrilintide combinations
Cagrilintide combined with semaglutide (CagriSema) represents a different approach: adding an amylin analog to GLP-1 agonism. Amylin, a hormone co-secreted with insulin, provides additional satiety signaling through distinct neural pathways. The combination has shown approximately 15 to 20 percent weight loss in early trials, though direct head-to-head data with semaglutide alone at equivalent doses is still emerging. Our coverage of cagrilintide for weight loss, cagrilintide for men, cagrilintide dosing, CagriSema dosing, and the amylin receptor agonist mechanism provides the full picture.
Speed comparison table
Compound | Mechanism | Avg weight loss at ~68-72 wk | Time to 5% loss | Time to 10% loss | Relative speed vs semaglutide |
|---|---|---|---|---|---|
Semaglutide 2.4 mg | GLP-1 | ~14.9% | ~8-12 wk | ~12-16 wk | Baseline |
Tirzepatide 15 mg | GLP-1 + GIP | ~20-22.5% | ~6-10 wk | ~10-14 wk | ~15-20% faster |
Retatrutide 12 mg | GLP-1 + GIP + Glucagon | ~20-24% | ~6-8 wk | ~8-12 wk | ~20-30% faster |
CagriSema | GLP-1 + Amylin | ~15-20% | ~8-10 wk | ~12-14 wk | ~5-15% faster |
Context matters more than raw speed. Semaglutide has the longest safety record, the most extensive clinical trial program, and the broadest approval basis among these options. Speed is one factor. Safety, availability, tolerability, and cost are others. The peptide therapy cost guide and best peptide vendors guide can help researchers evaluate the practical dimensions beyond pure efficacy data. For weight loss specifically, our peptides for fat loss overview and the peptides for menopause weight loss guide help match compounds to specific individual contexts.
The long-term trajectory: months 6, 12, and 24
Short-term results get the headlines. Long-term data determines whether those results matter.
Month 6: peak velocity has passed
At the six-month mark, you have likely lost 12 to 15 percent of your starting body weight if you reached the full 2.4 mg dose on schedule and responded at or above the average rate. The velocity of weight loss has begun to decrease, transitioning from roughly 2 percent per month to closer to 1 percent. Metabolic adaptation is the primary driver of this deceleration. Your body now weighs significantly less, burns fewer calories at rest, and has adjusted hormonal output (leptin, ghrelin, thyroid hormones) to defend against further loss.
This is not a plateau in the clinical sense. A true plateau means zero weight loss over an extended period. Most people at month 6 are still losing, just more slowly. The distinction matters psychologically. Knowing that deceleration is expected prevents the discouragement that leads to abandonment. Your protocol planning should account for this shift and set appropriate expectations for the second half of treatment.
Month 12: approaching maximum effect
The STEP 1 trial showed that average weight loss approached its nadir around week 60, roughly month 15. But most of the weight loss, over 90 percent of the total, occurs in the first 12 months. Month 12 weight loss averages 14 to 15 percent. The final 3 months to the nadir contribute less than 1 additional percentage point.
At 12 months, the body has reached or is very close to a new set point. Appetite suppression continues at the same level, but metabolic rate has adjusted to the new body weight, and caloric expenditure matches the reduced intake. Cardiovascular and metabolic improvements have fully manifested. The clinical benefit is at or near its maximum. The before and after results at the 12-month mark are typically the most dramatic, representing the full transformation trajectory.
Month 24: the STEP 5 extension data
The STEP 5 trial extended observation to 104 weeks, just over two years. At week 104, 77.1 percent of semaglutide-treated participants had achieved at least 5 percent weight loss, compared to only 34.4 percent of the placebo group. More importantly, the weight loss achieved by month 15 was maintained through month 24 with continued treatment.
This is the critical finding. Semaglutide is not a crash diet that works fast and then fails. It is a sustained pharmacological intervention that produces durable results as long as treatment continues. The two-year data demonstrates that the body does not develop tolerance, the weight does not inevitably rebound while on treatment, and the metabolic improvements persist over the long term.
For researchers thinking about long-term peptide use, the guide to concurrent peptide use and the peptide stacking guide provide frameworks for maintaining comprehensive protocols over extended periods. Understanding proper peptide storage and post-reconstitution storage becomes especially important during extended protocols where supply management is a practical concern.
What happens when you stop semaglutide
This is the question nobody wants to ask. But the data is clear, and avoiding it does more harm than addressing it directly.
The STEP 1 extension data
When semaglutide treatment was discontinued in the STEP 1 trial extension, participants regained approximately two-thirds of the weight they had lost within one year of stopping. This is not unique to semaglutide. It reflects the fundamental biology of weight regulation. The neurohormonal systems that semaglutide suppresses (hunger hormones, appetite signaling, metabolic adaptation) reactivate when the drug is withdrawn.
The regain was not instantaneous. It occurred gradually over 6 to 12 months, mirroring the gradual loss in reverse. Appetite returned to pre-treatment levels. Caloric intake increased. The metabolic defense mechanisms that had been suppressed by GLP-1 agonism reengaged fully.
What this means for planning
Semaglutide is not a cure. It is a treatment. Like blood pressure medication that controls hypertension while taken but does not eliminate the underlying condition, semaglutide manages weight while active but does not reprogram the body to maintain a lower weight without pharmacological support.
This does not diminish its value. The cardiovascular, metabolic, and quality-of-life benefits during treatment are substantial and well-documented. But realistic planning means understanding that discontinuation carries a high probability of significant weight regain. Strategies to mitigate this include gradual dose tapering rather than abrupt cessation, concurrent development of sustainable dietary and exercise habits, and potentially transitioning to lower maintenance doses rather than complete withdrawal.
Some researchers explore complementary approaches to support weight maintenance during and after GLP-1 agonist use. The 5-amino-1MQ guide covers a compound that influences fat metabolism through a different mechanism, while AOD-9604 dosage protocols and AOD-9604 side effects data cover a growth hormone fragment with fat-loss specific activity. The tesofensine guide explores yet another pathway for appetite and weight management. Our perimenopause peptide guide and peptides for women over 40 address the specific challenges that hormonal transitions add to weight management protocols.
Optimizing your semaglutide timeline for the fastest safe results
Speed matters, but not at the expense of safety or sustainability. Here are evidence-based strategies to ensure your semaglutide protocol delivers results as efficiently as possible.
Do not skip the dose escalation
Jumping to 2.4 mg on day one will not produce faster weight loss. It will produce severe nausea, vomiting, and a high probability of abandoning the medication entirely. The escalation schedule exists because it works. It maximizes the probability that you will tolerate the full therapeutic dose and maintain it long enough to achieve meaningful results.
Prioritize protein at every meal
Protein preserves muscle. Muscle preserves metabolic rate. Metabolic rate determines long-term fat loss. When semaglutide reduces your appetite, use that smaller appetite to eat higher quality food rather than simply eating less of the same food. Aim for 1.2 to 1.6 grams of protein per kilogram of body weight daily, spread across all meals.
Incorporate resistance training
Strength training is the most effective intervention for preventing lean mass loss during weight reduction. Two to three sessions per week focused on compound movements (squats, deadlifts, presses, rows) provide sufficient stimulus to signal your body that muscle tissue is needed and should be preserved. The combination of semaglutide for fat loss and resistance training for muscle preservation produces superior body composition outcomes compared to either alone. The athletic performance peptide research context supports this combined approach.
Stay hydrated and manage electrolytes
Reduced food intake means reduced dietary electrolyte intake. Combined with potential fluid losses from gastrointestinal side effects, dehydration and electrolyte imbalances can occur. Adequate hydration (minimum 2 liters daily) and attention to sodium, potassium, and magnesium intake prevents headaches, fatigue, and muscle cramps that can mimic or worsen semaglutide side effects.
Inject consistently on the same day each week
The 168-hour half-life means weekly injections maintain stable plasma levels. Varying the injection day disrupts this stability, creating peaks and valleys that worsen side effects and reduce consistent appetite suppression. Pick a day. Stick with it. If you need to change your injection day, do so gradually by shifting one day at a time over several weeks. The injection pen guide covers proper technique for consistent administration.
Track everything, but do not obsess over the scale
Weekly weigh-ins at the same time of day, same conditions, provide the most useful trend data. Daily weights fluctuate based on hydration, sodium intake, and bowel movements, creating noise that obscures the signal. Track waist circumference monthly. Take progress photos monthly. These measures reveal body composition changes that scale weight alone misses. Use our peptide cost calculator to budget your protocol realistically, and understand peptide costs for the long-term planning that sustained treatment requires.
Understanding the mechanisms behind semaglutide speed
For researchers who want to understand not just the timeline but the biology driving it, this section covers the key mechanisms that determine how quickly semaglutide produces its effects.
Central appetite regulation
Semaglutide crosses the blood-brain barrier and activates GLP-1 receptors in multiple brain regions involved in appetite regulation. The arcuate nucleus of the hypothalamus contains both appetite-stimulating (AgRP/NPY) and appetite-suppressing (POMC/CART) neurons. GLP-1 receptor activation shifts the balance toward suppression. It also acts on the nucleus tractus solitarius in the brainstem, which integrates peripheral satiety signals from the gut.
The central effects explain why semaglutide reduces not just physical hunger but psychological food preoccupation. The mesolimbic reward pathway, which drives hedonic eating, also contains GLP-1 receptors. Activation in this region reduces the rewarding properties of food, particularly high-calorie, high-palatable foods. This is the neurobiological basis of the "food noise" reduction that users describe. For a broader understanding of what peptides are and how they interact with neural pathways, the fundamentals provide essential context.
Peripheral metabolic effects
In the pancreas, semaglutide stimulates glucose-dependent insulin secretion and suppresses glucagon release. These effects improve glycemic control rapidly, often before significant weight loss has occurred. In the liver, GLP-1 receptor activation reduces hepatic glucose production and influences lipid metabolism. In adipose tissue, improved insulin sensitivity promotes appropriate fuel partitioning, directing excess energy toward oxidation rather than storage.
Gastric motility and the gut-brain axis
Slowed gastric emptying is a direct peripheral effect of GLP-1 receptor activation on the vagus nerve and enteric nervous system. Food remains in the stomach longer, prolonging mechanical distension signals that contribute to satiety. This effect is dose-dependent, meaning higher doses produce more pronounced slowing. The gut-brain axis, a bidirectional communication network between the gastrointestinal tract and the central nervous system, integrates these peripheral signals with the central appetite effects to produce comprehensive appetite suppression.
Understanding these layered mechanisms explains why semaglutide effects unfold over weeks rather than days. Each system has its own activation threshold, adaptation timeline, and dose-response relationship. The full therapeutic effect requires sufficient drug concentration across all relevant tissues simultaneously, which is precisely what the dose escalation schedule is designed to achieve. Resources on getting started with peptides and the peptide safety and risks overview help researchers build a solid foundation before engaging with any compound that operates across multiple physiological systems.
Special populations and adjusted timelines
Not everyone follows the standard trajectory. Certain populations have unique considerations that affect how fast semaglutide works.
People with type 2 diabetes
Glycemic improvements appear faster than weight loss in diabetic populations. Blood sugar reductions are often measurable within the first two weeks of treatment, driven by the direct pancreatic effects of GLP-1 receptor agonism. Weight loss, however, may be slightly slower compared to non-diabetic populations, likely due to the metabolic resistance created by chronic insulin dysregulation and the weight gain effects of concurrent diabetes medications like sulfonylureas or insulin.
People on other medications
Certain medications can accelerate or impede semaglutide effects. Metformin, when combined with semaglutide, may enhance glycemic improvements synergistically. Medications that cause weight gain (some antipsychotics, corticosteroids, certain antidepressants) can partially counteract semaglutide weight loss effects. Discussing potential interactions with a healthcare provider is essential before starting any protocol. The peptide therapy online guide discusses how telemedicine providers can help coordinate multi-medication management, and the testing labs guide covers how to monitor your markers during treatment.
Women in perimenopause and menopause
Hormonal fluctuations during perimenopause and menopause create additional metabolic challenges. Declining estrogen levels promote central fat accumulation, insulin resistance, and reduced lean mass. Semaglutide can still produce significant weight loss in this population, but the timeline may be extended, and the magnitude may be somewhat reduced compared to premenopausal women. The peptides for menopause guide and hormone balance peptide research address these specific considerations in detail.
Older adults
Age-related sarcopenia (muscle loss) makes the body composition question even more critical for older adults using semaglutide. Weight loss in this population must be carefully monitored to ensure that lean mass preservation is prioritized. The timeline for results may be similar, but the emphasis on protein intake and resistance training is amplified. Falls, fractures, and functional decline are real risks if muscle loss is not actively prevented during pharmacological weight reduction.
Real-world versus clinical trial timelines
Clinical trials represent controlled conditions. Real-world use introduces variables that trials minimize or eliminate. Understanding the gap helps calibrate expectations.
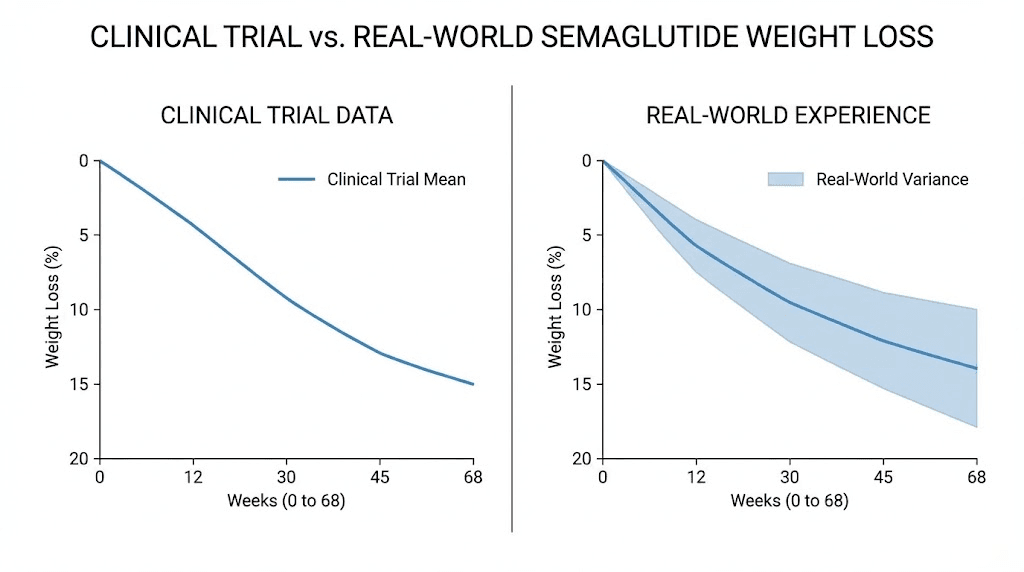
Why real-world results sometimes lag behind trial data
Trial participants receive regular monitoring, dietary counseling, and structured follow-up that promotes adherence and optimal behavior. They are often more motivated than average users because of the commitment involved in trial participation. Supply consistency is guaranteed in trials, whereas real-world users may face shortages, insurance issues, or cost barriers that create treatment gaps.
Additionally, trial participants who discontinue due to side effects or lack of efficacy are often excluded from certain analyses, which can inflate the apparent average effect. Real-world observational data, while more heterogeneous, provides a more realistic picture of outcomes across the full spectrum of users.
Why some real-world users exceed trial averages
Conversely, some individuals outperform trial averages. Higher starting BMI, younger age, absence of metabolic comorbidities, and aggressive lifestyle optimization (high protein, strength training, consistent dosing) can all push results beyond the mean. The 32 percent of STEP 1 participants who achieved 20 percent or greater weight loss were not outliers in the statistical sense. They represent the favorable end of the response distribution that real-world users can access with optimal conditions.
The peptide forum guide connects researchers with communities where real-world experiences and outcomes are shared, providing anecdotal but valuable context beyond clinical trial data. And for those considering the practical aspects of starting a protocol, our peptide reconstitution guide, mixing peptides with bacteriostatic water guide, and bacteriostatic water guide cover the fundamental preparation steps. Understanding peptide expiration, fridge shelf life, and powder form stability prevents wasted product and inconsistent dosing that could slow results.
The higher dose frontier: semaglutide 7.2 mg
Research continues to explore whether higher semaglutide doses produce faster and greater results. The STEP UP trial evaluated semaglutide doses up to 7.2 mg weekly, three times the currently approved weight management dose.
The results were striking. Average weight reduction reached 20.7 percent after 72 weeks at the 7.2 mg dose, compared to the 14.9 percent seen with 2.4 mg in STEP 1. The higher dose produced results comparable to tirzepatide, suggesting that dose intensity is a significant variable in GLP-1 agonist efficacy.
However, higher doses also produced higher rates of gastrointestinal side effects. The tolerability question is not trivial. A dose that works faster but causes 30 percent of users to discontinue may not be practically superior to a lower dose that more people complete. The peptide legality guide and research versus pharmaceutical peptides comparison address the regulatory landscape around different formulations and dose levels, while the drug testing guide covers detection considerations for those in tested environments. The choice between injectable versus oral peptides and understanding lyophilized versus liquid peptides adds further practical dimensions to protocol planning.
Complementary strategies that may accelerate semaglutide results
Semaglutide does not operate in isolation. The environment in which it operates, your body, your habits, your concurrent interventions, influences the speed and magnitude of results.
Nutrition optimization beyond caloric restriction
Meal timing, macronutrient ratios, and food quality all influence outcomes. Front-loading protein intake (larger protein portions at breakfast and lunch) can leverage semaglutide appetite suppression during the hours when cravings are naturally strongest. Reducing ultra-processed food intake improves insulin sensitivity independently, potentially amplifying semaglutide metabolic effects. Fiber intake supports gut health and prolongs satiety, complementing the gastric emptying effects of the drug.
Sleep optimization
Poor sleep increases ghrelin (hunger hormone) production, reduces insulin sensitivity, and promotes central fat accumulation. These effects directly counteract semaglutide mechanisms. Improving sleep quality and duration (7 to 9 hours nightly) removes a significant barrier to optimal drug response. This is one of the highest-impact lifestyle modifications for accelerating results, yet it receives far less attention than diet or exercise.
Stress management
Chronic stress elevates cortisol, which promotes visceral fat deposition, insulin resistance, and emotional eating. While semaglutide suppresses appetite pharmacologically, cortisol-driven eating operates through partially independent pathways. Stress reduction strategies (regular exercise, meditation, adequate rest, social connection) create a hormonal environment more conducive to rapid response.
Complementary peptides and compounds
Some researchers explore combinations that address aspects of weight loss and body composition that semaglutide does not directly target. Growth hormone secretagogues like sermorelin and ipamorelin may support lean mass preservation. The MOTS-c peptide influences mitochondrial function and metabolic efficiency. BPC-157 and TB-500 stacking addresses the tissue repair needs that can arise from increased physical activity during a weight loss protocol.
For researchers considering multi-compound approaches, the peptide stack calculator helps plan combined protocols, and the comprehensive stacking guide covers the principles of combining peptides safely. Understanding peptides versus steroids and peptides versus SARMs helps contextualize the safety profile of peptide-based approaches relative to alternatives. The BPC-157 overview, TB-500 benefits, and IGF-1 LR3 guide provide starting points for exploring supportive compounds. SeekPeptides members access detailed protocols for combining these compounds with GLP-1 agonists, along with community insights from researchers who have navigated these complex multi-compound approaches.
Reading beyond the scale: non-weight markers of semaglutide working
The scale is one data point. It is not the only one. And in the early weeks of treatment, non-weight markers may provide better evidence that semaglutide is active and effective than body weight changes alone.
Energy levels and sleep quality
Many users report improved energy levels within the first month, even before significant weight loss occurs. Reduced postprandial blood sugar spikes, improved insulin sensitivity, and lower systemic inflammation all contribute to a subjective increase in daily energy. Sleep quality may also improve as reduced food intake decreases nighttime GERD and sleep apnea symptoms associated with excess weight.
Reduced joint pain and improved mobility
Every pound lost removes approximately four pounds of stress from the knee joints. At just five pounds of weight loss, that translates to 20 pounds less force on each knee with every step. Users with joint pain often notice functional improvements before the scale changes enough to explain them, because the anti-inflammatory effects of GLP-1 agonism provide benefit independent of weight reduction. For researchers dealing with joint and tissue concerns, the peptide guide for joint pain and tendon repair peptide guide cover compounds that specifically target musculoskeletal healing.
Mental clarity and mood improvement
Reduced food noise frees mental bandwidth. Users consistently describe a clarity of thought that emerges when the constant preoccupation with food dissipates. This is not a placebo effect. The neurological changes driven by GLP-1 receptor activation in reward and executive function brain regions genuinely alter cognitive patterns related to food. Some researchers also note general mood improvement, potentially related to reduced inflammation and improved metabolic function. The peptides for depression and anxiety research, nootropic peptides, brain repair peptides, memory-enhancing peptides, and energy and focus peptides explore the cognitive dimension of peptide research in depth.
Skin, hair, and appearance changes
Weight loss and improved metabolic health often produce visible improvements in skin clarity, reduced facial puffiness, and an overall healthier appearance. These changes emerge gradually over months and are often noticed by others before the individual themselves. Reduced inflammation, improved circulation, and better nutrient partitioning all contribute. For those interested in the dermatological and aesthetic aspects of peptide research, the anti-aging peptide guide, wrinkle peptide guide, skin tightening peptides, and the GHK-Cu dosage guide cover compounds that specifically target skin health. Copper peptides for hair growth and the comprehensive hair growth peptide guide (along with the hair loss guide) address another visible dimension of health improvement.
Inflammation, immunity, and systemic effects timeline
Semaglutide does more than suppress appetite and reduce weight. Emerging evidence reveals significant anti-inflammatory and immunomodulatory effects that develop on their own timeline.
C-reactive protein and inflammatory markers
CRP, the most commonly measured marker of systemic inflammation, begins declining within the first three months of semaglutide treatment. The SELECT trial documented significant CRP reductions that correlated with but were not entirely explained by weight loss. This suggests direct anti-inflammatory effects of GLP-1 receptor activation in immune cells and vascular tissue.
For researchers tracking inflammatory markers, the reduction typically follows weight loss by a few weeks. Peak anti-inflammatory benefit appears to coincide with the period of most active weight loss (months 3 through 9), though improvements persist as long as treatment continues. The broader landscape of inflammation-targeting peptides, including KPV peptide and KPV for inflammation specifically, offers additional tools for researchers addressing chronic inflammatory conditions. The immune system peptide guide and autoimmune disease peptide research provide context for how immunomodulatory peptides complement metabolic interventions. Neuropathy, which can be driven by both metabolic dysfunction and inflammation, is covered in our neuropathy peptide guide, and bone health and bone healing peptide guides cover skeletal considerations during weight loss. Men may find our prostate health peptide guide relevant as weight loss intersects with hormonal health. For the full story on how popular figures discuss these compounds, our Joe Rogan peptides feature covers mainstream peptide conversations.
Frequently asked questions
How fast does semaglutide start working for appetite suppression?
Most people notice subtle appetite changes within the first one to two weeks of starting semaglutide at 0.25 mg. However, meaningful appetite suppression, the kind that significantly reduces daily caloric intake and quiets food noise, typically develops between weeks 4 and 8 as the dose increases to 0.5 mg and reaches steady state. The detailed appetite suppression timeline covers this process in granular detail.
How much weight can I expect to lose in the first month on semaglutide?
Clinical trial data shows an average of approximately 2 percent body weight loss in the first month, which translates to roughly 4 to 6 pounds for most starting weights. Some individuals lose more, particularly those with higher starting weights or those who are especially responsive to the appetite-suppressing effects. The first month is an introduction phase at a sub-therapeutic dose, so modest results are expected and normal.
Why did semaglutide stop working after a few months?
Semaglutide does not stop working. What changes is the rate of weight loss. After the initial rapid phase (months 1 through 6), weight loss decelerates as your body approaches a new metabolic equilibrium. This is not a plateau in most cases but rather a normal slowing of the trajectory. If you have truly stalled for eight or more weeks, review the troubleshooting strategies in this guide and consult the why you are not losing weight on semaglutide resource for specific solutions.
Does semaglutide work faster than tirzepatide?
No. Clinical evidence suggests tirzepatide produces weight loss approximately 15 to 20 percent faster than semaglutide at comparable timepoints, likely due to its dual GIP/GLP-1 mechanism. However, semaglutide has a longer track record and more extensive safety data. Our semaglutide versus tirzepatide comparison provides a comprehensive side-by-side analysis.
When do the cardiovascular benefits of semaglutide begin?
The biological changes that underlie cardiovascular protection, including reduced blood pressure, lower inflammatory markers, improved lipids, and decreased visceral fat, begin accumulating within the first three to six months. The clinical event reduction documented in the SELECT trial (20 percent reduction in MACE) reflects these early changes sustained over years. You will not feel cardiovascular protection on a daily basis, but the risk reduction begins early in treatment.
What happens if I miss a semaglutide dose?
If you miss a dose by fewer than five days, take it as soon as you remember and resume your regular schedule. If more than five days have passed, skip the missed dose and take the next one on your regular day. A single missed dose will cause a temporary dip in plasma levels but should not significantly disrupt results. Repeated missed doses, however, will prevent steady state maintenance and reduce efficacy.
Does semaglutide work for people without much weight to lose?
Semaglutide is most studied in populations with a BMI of 27 or higher. Individuals closer to a healthy weight range may experience less dramatic weight loss in absolute terms but can still benefit from metabolic improvements, appetite regulation, and body composition optimization. The percentage-based results may be lower, and the risk-benefit calculation is different for individuals without significant excess weight.
How long should I stay on semaglutide to see full results?
Clinical trial data shows maximum weight loss occurring around week 60 (approximately 15 months). The vast majority of weight loss, over 90 percent of the total, occurs within the first 12 months. For cardiovascular and metabolic benefits, longer treatment durations produce more sustained protection. Discontinuation typically leads to significant weight regain within 12 months, which is an important factor in long-term planning.
External resources
STEP 1 Trial - Once-Weekly Semaglutide in Adults with Overweight or Obesity (NEJM)
FDA Semaglutide Drug Safety Information
For researchers committed to optimizing their semaglutide protocols and understanding the full spectrum of peptide-based interventions, SeekPeptides provides the most comprehensive resource available. Members access evidence-based dosing protocols, detailed dosage charts, dosing calculators, expert-reviewed guides, and a community of thousands of researchers who have navigated these exact questions. Whether you are in week one wondering if anything is happening or month six looking for ways to push past a plateau, the tools and knowledge you need are there.