Feb 12, 2026

Conflicting headlines are everywhere. One source says compounded semaglutide is perfectly safe and a fraction of the cost. Another says it is dangerous, unregulated, and possibly contaminated. A third tells you it has been banned entirely. You are trying to make an informed decision about your health, and the information landscape is a mess.
Here is the reality. Compounded semaglutide occupies one of the most complicated spaces in modern medicine. It involves pharmaceutical regulation, compounding pharmacy law, GLP-1 receptor science, and a supply chain that stretches across continents. Getting this wrong can cost you money. Getting it very wrong can cost you a trip to the emergency room.
This guide exists because you deserve clarity. Not marketing spin from telehealth companies trying to sell you something. Not fear-mongering from brand manufacturers protecting market share. Actual information, grounded in research, written for people who want to understand what they are putting into their bodies. SeekPeptides built this resource because navigating the compounded semaglutide landscape requires the kind of detailed, unbiased analysis that most sources simply do not provide.
We will cover what compounded semaglutide actually is at the molecular level, how it differs from brand-name products like Ozempic and Wegovy, what the FDA regulations actually say right now, how dosing protocols work, what side effects to watch for, how to store it properly, and what the future holds. Every claim backed by research. Every number verified.
If you have been confused, you will not be after reading this.
What compounded semaglutide actually is
Semaglutide is a glucagon-like peptide-1 receptor agonist. That is a mouthful, so here is the simple version. Your gut naturally produces a hormone called GLP-1 every time you eat. This hormone tells your pancreas to release insulin, tells your brain you are full, and slows down how fast food moves through your stomach. Semaglutide mimics this hormone, except it lasts much longer than the natural version.
The natural GLP-1 hormone survives in your bloodstream for about two minutes. Semaglutide? It lasts roughly a week. That is because the molecule has been engineered with a fatty acid chain that binds to albumin in your blood, protecting it from the enzymes that would normally break it down. One injection per week. Steady appetite suppression. Consistent weight loss results.
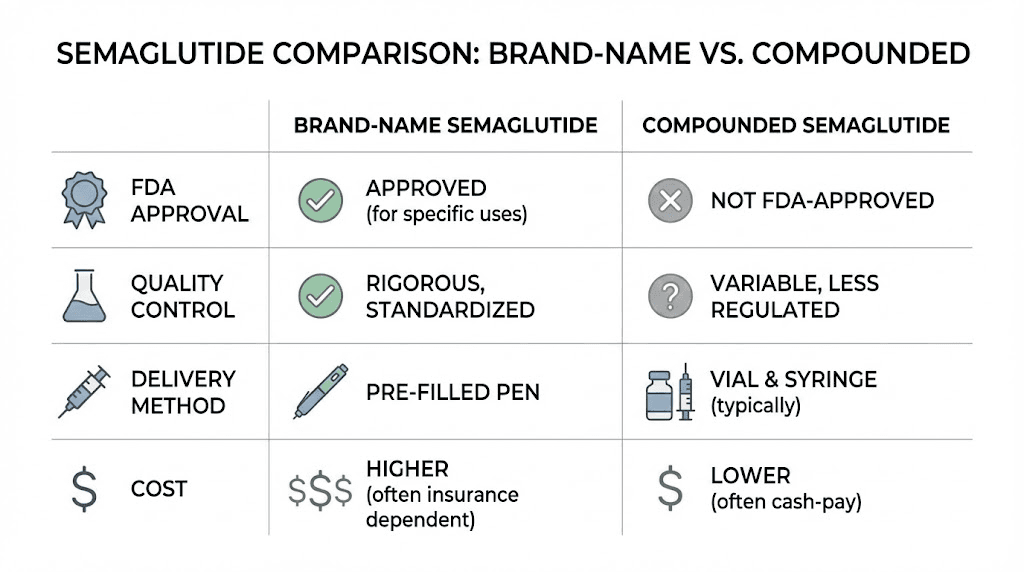
Brand-name semaglutide products like Ozempic and Wegovy are manufactured by Novo Nordisk, a pharmaceutical company based in Denmark. These products go through years of clinical trials, FDA review, and rigorous quality control before reaching patients. Ozempic is approved for type 2 diabetes management. Wegovy is approved specifically for weight management. Same molecule. Different approved uses. Different dosing protocols.
Compounded semaglutide is that same molecule, but prepared by a compounding pharmacy rather than Novo Nordisk. A compounding pharmacy takes the raw active ingredient, semaglutide, and combines it with other ingredients to create a finished injectable product. The concept is not new. Compounding pharmacies have existed for centuries, creating customized medications when commercial products do not meet a patient's specific needs.
But here is where things get complicated. And they get complicated fast.
The legal framework for compounding
In the United States, compounding pharmacies operate under two distinct regulatory categories defined by the Federal Food, Drug, and Cosmetic Act. Understanding these categories is essential for evaluating compounded medication safety.
503A pharmacies are traditional compounding pharmacies. They prepare medications based on individual prescriptions from licensed healthcare providers. Think of your local pharmacy mixing a specific cream or capsule that a doctor ordered for one patient. These pharmacies are primarily regulated by state boards of pharmacy.
503B outsourcing facilities are larger operations that can produce compounded medications in bulk without individual prescriptions. They register with the FDA and must comply with current good manufacturing practices, or cGMPs. This means more oversight, more testing, and theoretically more consistency.
Both types of facilities have been involved in compounding semaglutide. The regulatory rules governing when they can do this, however, changed dramatically in early 2025.
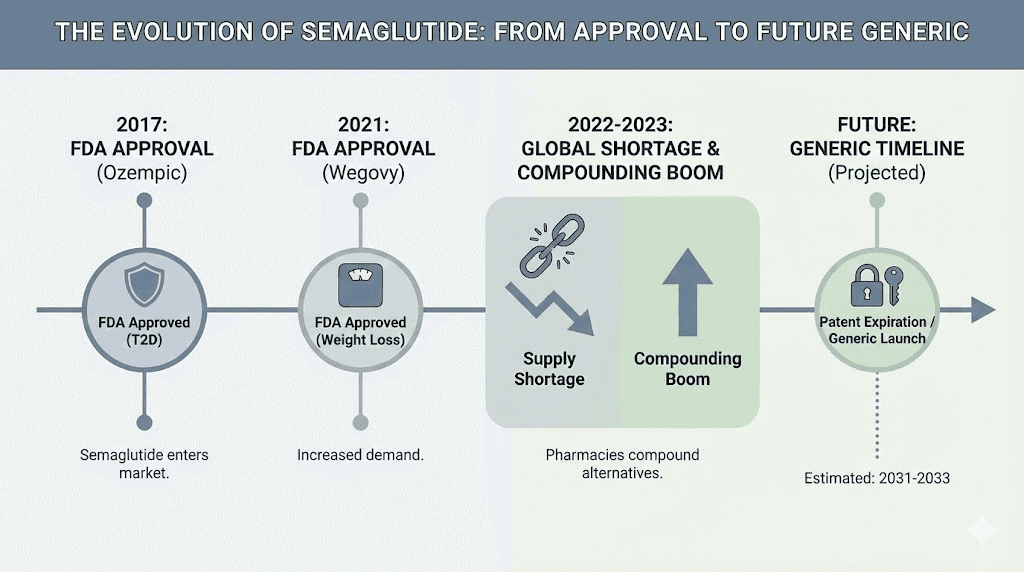
The shortage that started it all
The story of compounded semaglutide begins with a shortage. When demand for semaglutide and tirzepatide exploded in 2022 and 2023, Novo Nordisk could not keep up. Pharmacies across the country reported empty shelves. Patients who had been prescribed Ozempic or Wegovy found themselves unable to fill their prescriptions for weeks or months at a time.
Under federal law, when the FDA places a drug on its official shortage list, compounding pharmacies gain the right to produce copies of that drug. This is a safety valve built into the system. If patients cannot access their prescribed medication through normal channels, compounding pharmacies can step in to fill the gap.
And step in they did. Hundreds of compounding pharmacies began producing semaglutide. Telehealth companies built entire business models around prescribing and delivering compounded versions. The market grew from essentially zero to billions of dollars in revenue within two years.
Then, on February 21, 2025, the FDA announced that the semaglutide shortage was officially resolved.
How compounded semaglutide differs from Ozempic and Wegovy
This is not as simple as saying they are the same thing in different packaging. The differences matter, and understanding them could affect your health outcomes, your safety, and your wallet. Anyone considering peptide dosing needs to understand these distinctions clearly.
The active ingredient question
Ozempic and Wegovy contain semaglutide base. This is the exact molecular form that went through clinical trials, demonstrated safety and efficacy in thousands of patients, and received FDA approval. The base form has a specific molecular weight, specific binding properties, and specific pharmacokinetic behavior that researchers have studied extensively.
Compounded semaglutide should also contain semaglutide base. But many compounding pharmacies have used different chemical forms instead. Semaglutide sodium. Semaglutide acetate. These are salt forms of the molecule, created by combining semaglutide with sodium or acetic acid.
Why does this matter?
Salt forms can behave differently in the body. They may absorb at different rates. They may have different stability profiles. They may interact differently with other ingredients in the formulation. The FDA has explicitly stated that semaglutide salt forms are not the same as semaglutide base and should not be used for compounding purposes.
This is not a theoretical concern. When you receive a compounded semaglutide product, you may or may not know which form of the molecule you are getting. The label may say semaglutide, but the chemical identity of what is actually in the vial can vary depending on where the pharmacy sourced its raw materials.
Formulation differences
Brand-name Ozempic and Wegovy contain more than just semaglutide. They include a carefully designed formulation of inactive ingredients, buffers, preservatives, and stabilizers that work together to keep the molecule stable and functional throughout its shelf life. Novo Nordisk spent years optimizing these formulations.
Compounded versions may use entirely different inactive ingredients. Some pharmacies add vitamin B12 to their formulations. Others include glycine as a stabilizer and muscle-preserving agent. Some add L-carnitine for its purported fat-burning properties. A few include niacinamide or other supplements.
None of these combinations have been studied in clinical trials. There is no published research establishing that semaglutide plus B12 is safe or effective. There is no data showing that glycine enhances semaglutide absorption. These are additions that pharmacies make based on theoretical benefits, not proven outcomes. If you are interested in understanding how combining different compounds works, the research must be evaluated carefully.
Delivery system
Ozempic comes in a prefilled injection pen. You dial your dose. You press a button. The pen delivers a precise amount of medication every single time. The mechanism is engineered to prevent dosing errors. You cannot accidentally give yourself ten times the intended dose.
Compounded semaglutide comes in a vial. You draw up the medication yourself using an insulin syringe. This requires understanding how to read syringe markings, how to calculate the correct volume for your prescribed dose, and how to inject properly.
This difference is not trivial. The FDA has received reports of patients accidentally administering five to twenty times their intended dose because they misread syringe markings or confused units with milligrams. A study published in the Journal of Medical Toxicology documented multiple cases of compounded semaglutide overdoses reported to poison control centers, with symptoms ranging from severe nausea and vomiting to dehydration requiring hospitalization.
Quality control and testing
Every batch of Ozempic and Wegovy undergoes extensive quality testing before it reaches patients. Potency testing confirms the correct amount of active ingredient. Sterility testing confirms no bacterial contamination. Stability testing confirms the product will remain effective through its expiration date. Endotoxin testing checks for dangerous bacterial byproducts.
Compounding pharmacies have varying levels of quality control. 503B outsourcing facilities must follow cGMP standards, which include many of these same tests. 503A pharmacies have less stringent requirements. Some pharmacies conduct rigorous third-party testing. Others do the minimum required by their state board of pharmacy.
This variability means that the quality of compounded semaglutide can differ significantly from one pharmacy to another. The same prescription filled at two different compounding pharmacies could yield products with different potencies, different purity levels, and different shelf lives. Understanding proper peptide testing standards helps you evaluate what you are receiving.
FDA regulations and the current legal landscape
The regulatory environment surrounding compounded semaglutide has shifted dramatically, and it continues to evolve. If you are currently using compounded semaglutide or considering it, understanding these changes is not optional. It is essential.
The shortage resolution
On February 21, 2025, the FDA officially removed injectable semaglutide from its drug shortage list. This single action triggered a cascade of regulatory consequences that reshaped the entire compounded semaglutide market overnight.
When a drug is on the shortage list, compounding pharmacies have broad legal authority to produce copies. When it comes off the list, that authority evaporates. The FDA established transition periods to prevent abrupt disruption to patients already receiving compounded semaglutide. 503A pharmacies received until April 22, 2025, to wind down production. 503B outsourcing facilities received until May 22, 2025.
After those deadlines, compounding pharmacies could no longer produce products that are essentially copies of commercially available FDA-approved semaglutide products.
What compounding is still allowed
The end of the shortage did not completely eliminate compounded semaglutide. Federal law still allows compounding in specific, limited circumstances. A physician can prescribe compounded semaglutide if a patient has a documented medical need that the commercial product cannot meet.
The most common qualifying scenario involves allergies. If a patient has a documented allergy to one of the inactive ingredients in Ozempic or Wegovy, a compounding pharmacy can prepare an alternative formulation without that ingredient. The compounded product must be meaningfully different from the commercial product, not simply a cheaper copy.
Cost alone is not a valid reason for compounding. Convenience is not a valid reason. Preference is not a valid reason. The medical necessity must be real, documented, and specific.
Legal challenges and ongoing litigation
The compounded semaglutide market did not go quietly. On February 24, 2025, just three days after the FDA announced the shortage resolution, the Outsourcing Facilities Association filed a lawsuit challenging the decision. Their argument was that the FDA acted arbitrarily and recklessly in declaring the shortage resolved when access barriers still existed for many patients.
This lawsuit, and others like it, created a period of legal uncertainty. Some compounding pharmacies continued producing semaglutide while courts considered injunctions. Others stopped immediately. The legal landscape has been fluid, with different jurisdictions reaching different conclusions about enforcement timelines.
For patients, this means that the availability of compounded semaglutide may depend partly on where you live, which pharmacy you use, and how courts in your jurisdiction have ruled on pending litigation. Anyone using peptide products should stay informed about regulatory developments.
The salt form controversy
Separate from the shortage question, the FDA has taken a clear position on semaglutide salt forms. The agency has stated that only semaglutide base, the same form used in FDA-approved products, is appropriate for compounding. Semaglutide sodium, semaglutide acetate, and other salt forms are not acceptable.
This matters because many compounding pharmacies, particularly during the peak of the shortage, sourced their raw materials from suppliers that provided salt forms rather than the base form. The supply chain for pharmaceutical-grade semaglutide base is limited, and Novo Nordisk has aggressively protected its supply relationships.
If you have received compounded semaglutide, it is worth asking your pharmacy which form they used. If they cannot or will not answer, that itself is informative.
How semaglutide works in the body
Understanding the mechanism helps you understand why dosing, timing, and formulation quality all matter so much. Whether you are using brand-name or compounded products, the pharmacology is the same, but only if the active ingredient is truly equivalent.
The GLP-1 receptor system
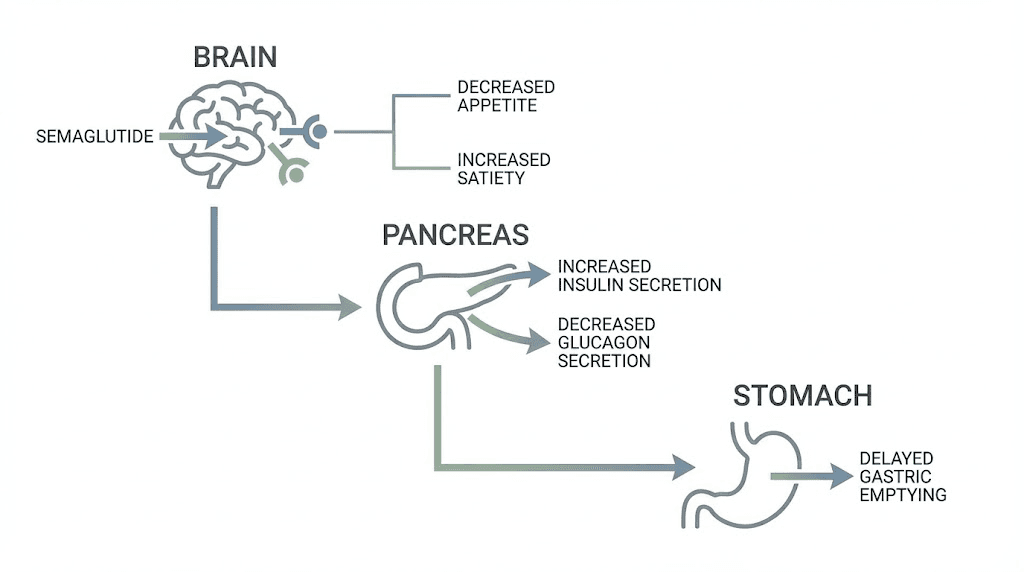
Your body has GLP-1 receptors in multiple locations. The pancreas. The brain. The stomach. The cardiovascular system. When semaglutide binds to these receptors, it triggers a coordinated response across all of these systems simultaneously. Understanding how peptides interact with receptors gives you a foundation for evaluating any GLP-1 therapy.
In the pancreas, semaglutide stimulates insulin secretion in a glucose-dependent manner. This means it only increases insulin when blood sugar is elevated, which reduces the risk of hypoglycemia compared to some other diabetes medications. It also suppresses glucagon, the hormone that raises blood sugar, when glucose levels are already adequate.
In the brain, semaglutide acts on appetite regulation centers, particularly in the hypothalamus. It reduces hunger signals, increases feelings of satiety, and may even reduce food cravings and the reward response to eating. This is why people on semaglutide often report not just eating less, but genuinely wanting less food.
In the stomach, semaglutide slows gastric emptying. Food stays in your stomach longer, which contributes to the feeling of fullness and reduces postprandial blood sugar spikes. This also explains the most common side effect, nausea, which occurs when the stomach processes food more slowly than the body expects.
Pharmacokinetics of semaglutide
After a subcutaneous injection, semaglutide reaches peak concentration in the blood within one to three days. Its half-life is approximately one week, which is why it only needs to be injected once per week. This extended half-life comes from the structural modifications Novo Nordisk engineered into the molecule.
The fatty acid chain attached to semaglutide binds to albumin, a protein that circulates abundantly in your blood. This albumin binding serves as a protective shield, preventing the enzymes dipeptidyl peptidase-4 and neutral endopeptidases from breaking down the molecule. It also creates a reservoir effect, with semaglutide slowly releasing from albumin over the course of the week.
Steady state, where the amount entering your system equals the amount being eliminated, occurs after four to five weeks of consistent dosing. This is why the full effects of a dose change take several weeks to manifest, and why the standard dosing escalation protocol increases doses in four-week intervals.
For compounded products, the pharmacokinetics should theoretically be identical if the active ingredient is truly the same. But if the formulation uses a different salt form, different stabilizers, or different concentrations, the absorption profile could change in unpredictable ways. This uncertainty is one of the core concerns with compounded products.
Dosing protocols for compounded semaglutide
Dosing is where the practical rubber meets the road. Get this wrong and you either waste your money with subtherapeutic doses or wind up in the emergency room with an overdose. The standard semaglutide dosing chart applies to both brand-name and compounded products, but the mechanics of measuring compounded doses require additional attention.
The standard escalation protocol
Whether brand-name or compounded, the dosing philosophy for semaglutide follows a gradual escalation designed to minimize gastrointestinal side effects while reaching therapeutic levels.
For weight management (Wegovy protocol):
Weeks 1 through 4: 0.25 mg once weekly
Weeks 5 through 8: 0.5 mg once weekly
Weeks 9 through 12: 1.0 mg once weekly
Weeks 13 through 16: 1.7 mg once weekly
Week 17 and beyond: 2.4 mg once weekly (maintenance dose)
For type 2 diabetes (Ozempic protocol):
Weeks 1 through 4: 0.25 mg once weekly
Weeks 5 through 8: 0.5 mg once weekly
If additional glycemic control needed: 1.0 mg once weekly
Maximum: 2.0 mg once weekly
These escalation timelines exist for a reason. Jumping to higher doses too quickly overwhelms the gastrointestinal system and dramatically increases nausea, vomiting, and diarrhea. Patience with the protocol typically produces better outcomes and better tolerability.
Compounded semaglutide dosing flexibility
One argument in favor of compounded semaglutide is dosing flexibility. Brand-name products come in fixed dose increments. Compounded products can be prepared at virtually any concentration, allowing for more granular dose adjustments.
Some providers start patients at doses as low as 0.125 mg or 0.15 mg per week, lower than any brand-name option allows. This ultra-low starting dose can benefit patients who are particularly sensitive to GLP-1 side effects or who want the most conservative possible introduction. If you are calculating doses, our semaglutide dosage calculator can help determine exact amounts.
Others use compounding to create micro-dosing protocols, maintaining lower doses for extended periods rather than escalating to the full therapeutic dose. The theory is that some patients achieve adequate appetite suppression at lower doses, reducing side effects while still losing weight. The evidence for this approach is largely anecdotal, drawn from provider experience rather than clinical trials. How fast semaglutide works depends significantly on which protocol you follow.
Concentration calculations
This is where compounded semaglutide becomes genuinely dangerous if you are not careful.
Compounded semaglutide vials come in various concentrations. Common options include 1 mg/mL, 2.5 mg/mL, 5 mg/mL, and even 10 mg/mL. The concentration determines how much liquid you need to draw up for your prescribed dose.
Example: If your dose is 0.5 mg and your vial concentration is 5 mg/mL, you need 0.1 mL, which equals 10 units on an insulin syringe. If you mistakenly draw up 50 units, thinking that is your dose, you have just injected 2.5 mg, five times more than intended.
These calculation errors happen frequently. A case series published by a poison control center documented multiple incidents where patients administered 5 to 20 times their intended dose due to confusion between milligrams, milliliters, and units. The resulting symptoms included severe vomiting, dangerous dehydration, and hypoglycemia requiring emergency treatment.
If you use compounded semaglutide, you must understand the relationship between your prescribed dose, your vial concentration, and the volume you need to inject. Write it down. Verify it with your pharmacist. Double-check every single time. Understanding how to calculate peptide dosages correctly is not optional when using compounded products.
Injection technique
Compounded semaglutide is administered as a subcutaneous injection, the same as brand-name products. The injection goes into the fatty tissue just beneath the skin, not into muscle. Common injection sites include the abdomen (at least two inches from the navel), the front of the thigh, and the upper arm.
Rotate injection sites each week. Using the same spot repeatedly can cause lipodystrophy, which creates lumps of hardened fatty tissue that affect absorption. If you are new to self-injection, our guide on where to inject GLP-1 medications covers proper technique in detail.
Proper technique matters for consistent absorption. Inject at a 45 to 90 degree angle depending on how much subcutaneous tissue you have at the injection site. Pinch the skin if using a shorter needle. Do not aspirate (pull back on the plunger). Hold the needle in place for 5 to 10 seconds after injecting to allow the full dose to deposit in the tissue.
Side effects and safety concerns
Side effects from semaglutide are well-documented from clinical trials of the brand-name products. The question with compounded semaglutide is whether the side effect profile changes when the formulation, source material, or quality control differs from the approved products.
Common side effects
The most frequently reported side effects of semaglutide, regardless of source, are gastrointestinal. These effects occur because semaglutide slows gastric emptying and changes signaling in the gut-brain axis.
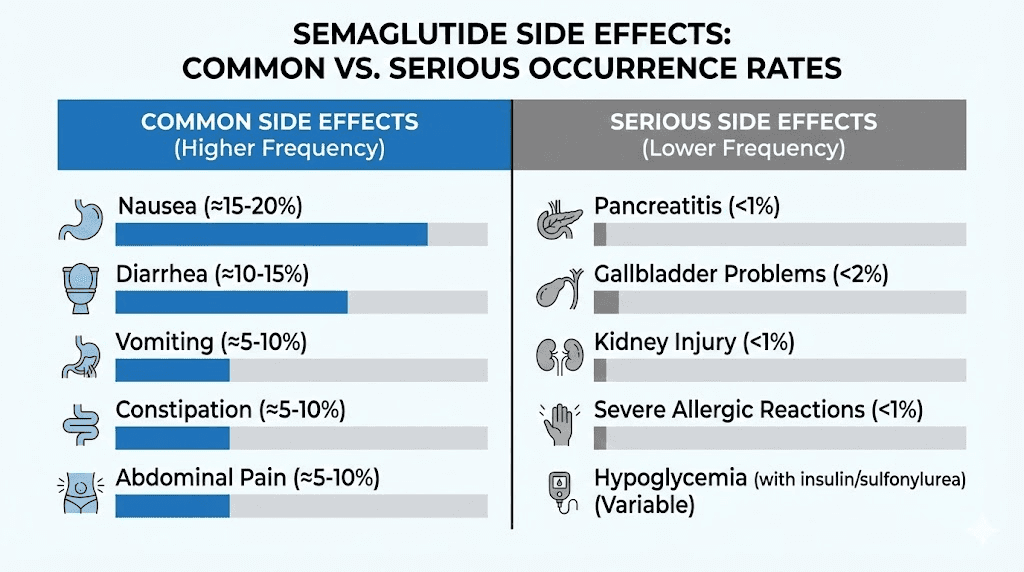
Nausea affects roughly 44% of patients at the 2.4 mg maintenance dose in clinical trials. It is most common during the first few weeks at each new dose level and typically improves with time. Eating smaller meals, avoiding fatty or greasy foods, and staying hydrated all help manage nausea. If you are struggling with side effects, understanding semaglutide fatigue and other common reactions can help you determine what is normal versus what requires medical attention.
Vomiting affects approximately 24% of patients at the maintenance dose. Like nausea, it tends to improve as the body adapts.
Diarrhea occurs in about 30% of patients. Staying hydrated is critical, as GLP-1 medications combined with reduced food intake can increase dehydration risk.
Constipation affects roughly 24% of patients. Adequate water intake and fiber supplementation can help.
Abdominal pain is reported by about 20% of patients. This is usually mild and related to the changes in gastric motility.
These side effects are typically dose-dependent. Higher doses produce more GI symptoms. This is exactly why the gradual escalation protocol exists. Rushing through dose increases dramatically worsens tolerability.
Serious side effects
Less common but more concerning side effects require awareness and monitoring.
Pancreatitis: Semaglutide carries a warning about the risk of acute pancreatitis. Symptoms include severe, persistent abdominal pain that may radiate to the back, often accompanied by vomiting. If you experience these symptoms, stop the medication and seek immediate medical attention.
Gallbladder problems: Clinical trials showed increased rates of cholelithiasis (gallstones) and cholecystitis (gallbladder inflammation) in semaglutide users. Rapid weight loss itself increases gallstone risk, so this may be partially related to the weight loss rather than the medication directly.
Thyroid concerns: In rodent studies, semaglutide caused thyroid C-cell tumors. While this has not been observed in humans, semaglutide carries a boxed warning about medullary thyroid carcinoma risk. It is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
Kidney issues: Dehydration from GI side effects can exacerbate existing kidney problems. Cases of acute kidney injury have been reported, particularly in patients who become severely dehydrated from persistent vomiting or diarrhea.
Hair loss has been reported by some users, likely related to the rapid weight loss and potential nutritional deficiencies that can accompany significant caloric restriction rather than a direct drug effect. If you experience this, evaluating your nutrition plan is the first step.
Compounded-specific safety concerns
Beyond the standard semaglutide side effects, compounded products carry additional risks that do not apply to brand-name medications.
Dosing errors are the most immediate danger. As discussed in the dosing section, the vial-and-syringe format creates opportunities for mistakes that the prefilled pen design specifically prevents. The FDA has linked compounded semaglutide dosing errors to approximately 10 deaths and 100 hospitalizations.
Contamination risk is elevated with compounded products. Sterility must be maintained throughout the compounding process. If a pharmacy cuts corners on sterile technique, the result can be bacterial or fungal contamination that causes infections ranging from mild injection site reactions to life-threatening sepsis.
Potency inconsistency means you may not be getting the dose you think you are getting. If a compounding pharmacy does not conduct rigorous potency testing on each batch, the actual amount of semaglutide in your vial could be higher or lower than labeled. Higher means unexpected side effects. Lower means you are paying for a product that does not work as expected.
Source material concerns: The raw semaglutide used by compounding pharmacies typically comes from overseas manufacturers. Not all of these suppliers maintain the same quality standards. Some have been found to provide product that does not meet pharmaceutical-grade specifications. When sourcing any research peptides, quality verification is paramount.
As of April 2025, the FDA had received 520 adverse event reports related to compounded semaglutide. The agency acknowledges that this number likely underrepresents the true scope, as many adverse events go unreported, particularly from state-licensed 503A pharmacies that are not required to submit reports to the FDA.
Cost comparison: compounded versus brand-name
Cost has been the primary driver of the compounded semaglutide market. The price difference between brand-name and compounded products is substantial, and for many patients, it has been the deciding factor.
Brand-name pricing
Without insurance, brand-name semaglutide is expensive. Ozempic costs approximately $900 to $1,000 per month at retail pharmacy prices. Wegovy costs approximately $1,300 to $1,400 per month. These prices are for the medication alone and do not include provider visits, lab work, or other associated costs.
Insurance coverage varies dramatically. Some insurers cover Ozempic for diabetes but not Wegovy for weight management. Some cover both. Some cover neither. Prior authorization requirements can delay access by weeks. Patients often discover their coverage status only after their physician has already prescribed the medication. If you are evaluating the cost of peptide therapy broadly, insurance coverage is a major variable.
Novo Nordisk offers savings programs for commercially insured patients, which can reduce out-of-pocket costs significantly. The NovoCare patient assistance program provides free medication to qualifying uninsured patients. But navigating these programs takes time and effort that many patients find frustrating.
Compounded pricing
Compounded semaglutide has historically cost a fraction of brand-name pricing. Typical monthly costs have ranged from $150 to $300 through telehealth platforms, with some pharmacies offering even lower prices for multi-month supplies.
This 70% to 85% cost savings explains the explosive growth of the compounded semaglutide market. For patients paying out of pocket, the math is straightforward. A year of Wegovy at retail pricing costs approximately $16,000. A year of compounded semaglutide costs approximately $2,000 to $3,600. The difference is life-changing for many people.
However, price should never be the only consideration. Cheap compounded semaglutide from a pharmacy that cuts corners on quality testing may cost you more in the long run, whether through ineffective treatment, adverse events, or emergency medical care. Understanding the true cost of peptide therapy requires factoring in these risks.
The post-shortage pricing landscape
With the FDA declaring the shortage resolved and limiting compounding, the market dynamics are shifting. Some telehealth platforms have pivoted to offering brand-name semaglutide at negotiated prices, with monthly costs around $300 to $500 through partnerships with pharmaceutical assistance programs. Others have shifted to offering compounded formulations that are meaningfully different from the commercial products, such as semaglutide with added B12 or glycine, arguing these qualify as non-copies.
The long-term pricing trajectory remains uncertain. As generic versions of semaglutide eventually enter the market, prices for both brand-name and compounded products will likely decrease. But that timeline is measured in years, not months.
Storage and handling
Proper storage is critical for any injectable medication, but it is especially important for compounded products that may have shorter shelf lives than brand-name alternatives. If you have questions about how long compounded semaglutide lasts in the fridge, the answer depends on several factors.
Refrigeration requirements
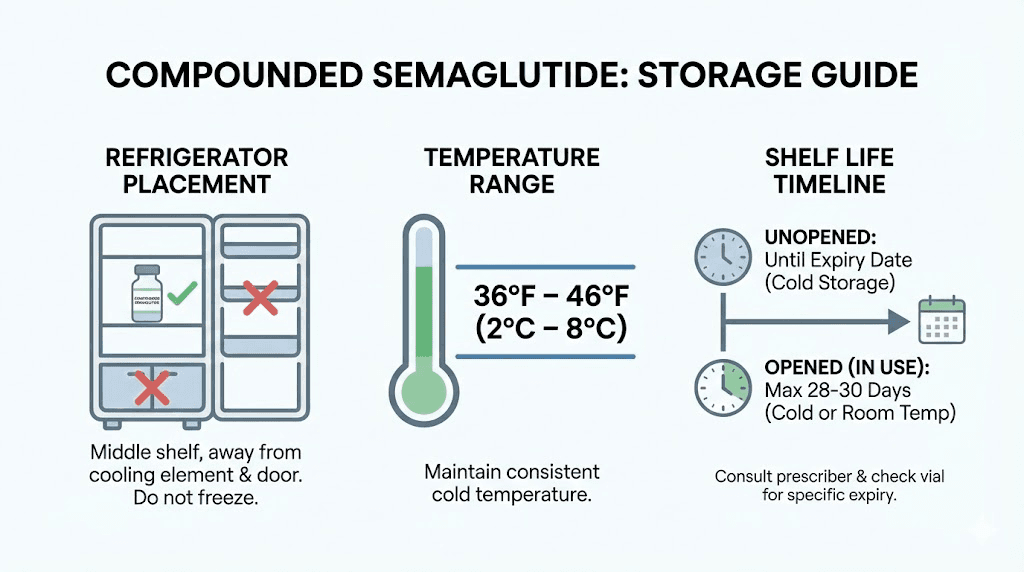
Compounded semaglutide must be stored in a refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). This is consistent with brand-name semaglutide storage requirements and with general peptide storage best practices.
Do not freeze compounded semaglutide. Freezing can damage the molecular structure and reduce potency. If your vial accidentally freezes, do not thaw and use it. Discard it and obtain a new vial.
Do not store it in the refrigerator door. The door is the warmest part of the refrigerator and experiences the most temperature fluctuation from repeated opening and closing. Place the vial on an interior shelf where the temperature remains most stable.
Beyond-use dates
Unlike brand-name products that have expiration dates established through extensive stability testing, compounded products have beyond-use dates (BUDs). The BUD represents the date by which the pharmacy guarantees the product meets its labeled potency and sterility specifications.
BUDs for compounded semaglutide vary widely depending on the pharmacy. Some assign 28-day BUDs. Others assign 60-day or 90-day BUDs based on their own stability testing. Pharmacies that conduct more rigorous testing may assign BUDs up to 120 days.
Once you puncture the vial with a needle for the first time, a separate clock starts. Most pharmacies recommend using the vial within 28 days of first puncture, regardless of the original BUD. Each time a needle enters the vial, there is a small risk of introducing contamination. The more punctures, the higher the cumulative risk. Following proper post-puncture storage guidelines helps minimize this risk.
Refrigeration is not optional for compounded semaglutide. Unlike some brand-name pen formulations that can tolerate limited room temperature exposure during use, compounded vial formulations are generally less stable and more susceptible to degradation at elevated temperatures.
Travel considerations
Traveling with compounded semaglutide requires planning. The medication must stay cold throughout transit. Insulated travel cases with gel packs work for short trips. For longer travel, consider asking your pharmacy about room-temperature stability data for their specific formulation.
Air travel adds another layer. You can carry injectable medications and syringes through airport security with a valid prescription. Keep the medication in its original pharmacy-labeled vial. Bring a letter from your prescribing physician if traveling internationally.
Do not check the medication in luggage, as cargo holds can freeze.
Compounded semaglutide with added ingredients
One of the distinguishing features of compounded semaglutide is the ability to add supplemental ingredients. These additions have become increasingly common and increasingly debated. Understanding what each additive claims to do, and what the evidence actually shows, helps you make informed decisions.
Semaglutide with B12 (cyanocobalamin or methylcobalamin)
B12 is the most common addition to compounded semaglutide formulations. The rationale includes energy support during caloric restriction, potential prevention of B12 deficiency from reduced food intake, and metabolic support for fat metabolism.
The reality is more nuanced. There is no published clinical evidence that adding B12 to semaglutide improves weight loss outcomes. There is no evidence that semaglutide causes B12 deficiency. Some patients do report improved energy levels with B12-fortified formulations, but this could be placebo effect, could be from B12 supplementation addressing a pre-existing deficiency, or could be genuine synergy. We simply do not have the data to know. Our detailed guide on semaglutide with B12 examines the evidence more thoroughly.
From a regulatory standpoint, adding B12 to semaglutide creates a product that is different from the FDA-approved formulation. Some compounding pharmacies have argued that this difference allows them to continue compounding even after the shortage resolution, since their product is not an essentially copy of the commercial product. This legal argument is being tested in various enforcement actions and lawsuits.
If you want B12 supplementation alongside your semaglutide therapy, you can also simply take an oral B12 supplement or receive periodic B12 injections separately. The convenience of a combined product is real, but it comes with the trade-off of using a formulation that has never been tested for safety in combination. Understanding how B12-fortified semaglutide dosing works is important if you choose this route.
Semaglutide with glycine
Glycine is an amino acid that serves multiple functions in compounded semaglutide formulations. As a stabilizer, it helps maintain the structural integrity of the semaglutide molecule during storage. As a supplemental ingredient, proponents claim it supports muscle preservation during weight loss, improves sleep quality, and provides anti-inflammatory benefits.
Some research supports glycine role in muscle preservation and metabolic health, but these studies examined glycine supplementation independently, not in combination with semaglutide. Whether glycine provides meaningful additional benefit when combined with semaglutide in a single injection is unknown.
Glycine is generally recognized as safe at supplemental doses. It is a naturally occurring amino acid that your body produces and uses daily. The risk of adverse effects from glycine in a semaglutide formulation is likely low, but the benefits are speculative.
Semaglutide with L-carnitine
L-carnitine is an amino acid derivative involved in fatty acid transport into mitochondria for energy production. The theoretical basis for adding it to semaglutide is that it could enhance fat burning beyond what semaglutide achieves alone.
The evidence for L-carnitine as a standalone weight loss supplement is mixed at best. Some studies show modest benefits. Others show none. Combining it with semaglutide has not been studied at all. The claim that this combination enhances results is marketing, not science.
Semaglutide with niacinamide
Some formulations include niacinamide (vitamin B3). The purported benefits include metabolic support, anti-inflammatory effects, and improved insulin sensitivity. As with other additions, there is no clinical evidence that combining niacinamide with semaglutide produces better outcomes than semaglutide alone. Our analysis of niacinamide additions to GLP-1 medications applies similar principles.
The bottom line on additives
None of the common compounded semaglutide additives have been studied in combination with semaglutide in controlled clinical trials. They may be safe. They may provide some benefit. But anyone telling you these combinations are proven or established is overstating the evidence.
If additives appeal to you, there is nothing stopping you from taking B12, glycine, or L-carnitine as separate supplements alongside brand-name semaglutide. This approach gives you the quality assurance of FDA-approved semaglutide plus the potential benefits of supplementation, without the risks of an untested combination product.
Who compounded semaglutide is and is not appropriate for
Not everyone who wants compounded semaglutide should use it. And not everyone who has been told they cannot use it should give up on it. The nuance matters.
Potentially appropriate candidates
Patients with documented allergies to inactive ingredients in Ozempic or Wegovy. This is the most clear-cut medical justification. If you have a verified allergy to an excipient in the brand-name product, a compounding pharmacy can formulate a version without that specific ingredient. This is the kind of patient-specific need that compounding was designed to serve.
Patients requiring doses not available in commercial products. If your physician determines that you need a dose that falls between the fixed increments available in Ozempic or Wegovy pens, a compounded formulation can provide that exact dose. This is particularly relevant for patients who are extremely sensitive to dose changes.
Patients in clinical situations where the commercial product is contraindicated but semaglutide itself is not. Rare but possible scenarios where an alternative formulation could allow treatment that would otherwise not be available.
Not appropriate candidates
Patients seeking compounded semaglutide solely because it is cheaper. Cost is understandable as a concern, but the FDA has explicitly stated that price is not a valid justification for compounding a copy of a commercially available drug. More importantly, the cost savings come with trade-offs in quality assurance and safety that you should weigh carefully.
Patients without a prescription from a licensed healthcare provider. Any semaglutide product, brand-name or compounded, requires a prescription. If someone is offering you compounded semaglutide without a physician evaluation, that is a red flag for your safety and legal standing.
Patients who cannot safely self-inject from a vial. The vial-and-syringe format requires understanding of dose calculation, sterile technique, and injection procedure. If you are not confident in your ability to perform these tasks correctly, the prefilled pen format of brand-name products is significantly safer.
Patients with a history of or family history of medullary thyroid carcinoma or MEN2. This contraindication applies to all semaglutide products, compounded or otherwise. No formulation change removes this risk.
Pregnant or breastfeeding individuals should not use any form of semaglutide. Animal studies have shown fetal harm. There is no human safety data for use during pregnancy or lactation.
How to evaluate a compounding pharmacy
If you determine that compounded semaglutide is appropriate for your situation, choosing the right pharmacy becomes the most important decision you make. The difference between a reputable compounding pharmacy and a substandard one can mean the difference between safe, effective treatment and a dangerous product.
Accreditation
Look for pharmacies accredited by the Pharmacy Compounding Accreditation Board (PCAB) or the Accreditation Commission for Health Care (ACHC). These voluntary accreditations indicate that the pharmacy has submitted to independent review of their facilities, processes, and quality standards. Not all good pharmacies are accredited, but accreditation provides an additional layer of assurance.
503B registration
If possible, choose a 503B outsourcing facility rather than a traditional 503A pharmacy. 503B facilities are registered with the FDA, required to follow current good manufacturing practices, and subject to FDA inspection. This means more consistent quality control, more rigorous testing, and more accountability.
Testing practices
Ask the pharmacy about their testing protocols. Specifically:
Do they test every batch for potency? They should. Do they test for sterility? Essential for injectables. Do they test for endotoxins? A marker of bacterial contamination. Do they conduct stability testing to establish their beyond-use dates? If they cannot or will not answer these questions, consider that a disqualifying factor.
Reputable pharmacies will provide Certificates of Analysis (COAs) for their products upon request. These documents detail the results of quality testing for specific batches. Ask for the COA for your specific lot number. Applying the same scrutiny you would to peptide testing and quality verification is appropriate here.
Source material verification
Ask where the pharmacy sources their raw semaglutide. Ideally, it should come from an FDA-registered facility that manufactures pharmaceutical-grade active ingredients. Ask specifically whether they use semaglutide base or a salt form. If they use a salt form, understand that this does not align with FDA guidance.
Red flags to watch for
Be cautious of pharmacies that offer semaglutide without a valid prescription, that guarantee specific weight loss results, that cannot answer questions about their quality testing, that offer prices dramatically below market rates (suggesting corners being cut), or that ship product without proper cold chain packaging. Understanding what separates reputable peptide vendors from questionable ones applies the same principles.
Transitioning between compounded and brand-name semaglutide
Whether you are moving from compounded to brand-name or vice versa, transitions require careful planning to maintain consistent therapeutic levels and avoid dose-related complications.
From compounded to brand-name
If you are transitioning to brand-name Ozempic or Wegovy from compounded semaglutide, the process is relatively straightforward if you know your current dose. Match the brand-name dose as closely as possible to your current compounded dose. If your compounded dose falls between available brand-name increments, your physician will determine whether to round up or down based on your tolerance and response.
Continue the same weekly injection schedule. Do not take both products in the same week. The transition from one to the other should be seamless from a pharmacokinetic standpoint, assuming the compounded product contained genuine semaglutide at the labeled concentration.
If you are considering switching from tirzepatide to semaglutide or between different GLP-1 medications entirely, the transition is more complex and requires physician guidance on dose equivalence.
From brand-name to compounded
Transitioning from brand-name to compounded semaglutide requires the same dose-matching approach, plus additional attention to the compounded product quality. Verify the concentration of the compounded vial carefully. Calculate your injection volume precisely. Consider having your pharmacy walk you through the first dose calculation if you are unfamiliar with vial-and-syringe administration.
What to watch for during transitions
Monitor for any changes in side effects, appetite suppression, or weight loss trajectory after switching. If the compounded product is truly equivalent, you should not notice any difference. If you do notice changes, either positive or negative, it may indicate that the compounded product differs from the brand-name in potency, absorption, or formulation in ways that affect its performance.
Keep your physician informed throughout the transition. Document your weekly weight, any side effects, and your subjective experience of appetite suppression. This data helps your physician evaluate whether the transition is working or whether adjustments are needed.
The future of compounded semaglutide
The compounded semaglutide landscape is evolving rapidly, driven by regulatory changes, legal challenges, market forces, and scientific developments. Here is what to watch for.
Ongoing litigation
Multiple lawsuits challenging the FDA shortage resolution remain active in federal courts. The outcomes of these cases will significantly influence whether and how compounding pharmacies can continue producing semaglutide. If courts side with the compounding industry, access may expand again. If they side with the FDA, the crackdown will intensify.
Generic semaglutide
Novo Nordisk patents on semaglutide will eventually expire. When they do, generic manufacturers will be able to produce FDA-approved semaglutide products that undergo the same rigorous review process as Ozempic and Wegovy but at lower prices. This could significantly reduce the cost gap that drives demand for compounded products.
The timeline for generic semaglutide depends on patent litigation, regulatory review periods, and manufacturing capacity build-out. Estimates range from the late 2020s to the early 2030s. Until then, the tension between brand-name pricing and compounded alternatives will persist.
New GLP-1 medications
The GLP-1 receptor agonist market is growing rapidly. New medications with different molecular structures, different dosing schedules, and potentially different side effect profiles are in development and entering the market. Retatrutide, CagriSema, survodutide, and orforglipron represent the next wave of weight loss peptide therapies.
As more options become available, the market pressure that created the compounded semaglutide boom may dissipate. If patients can access effective, FDA-approved alternatives at reasonable prices, the demand for compounded versions will naturally decrease.
Oral formulations
Rybelsus (oral semaglutide) is already FDA-approved for diabetes, and higher-dose oral formulations for weight loss are in development. Oral semaglutide drops and tablets could eliminate the injection barrier entirely, making the medication more accessible and removing the dosing error risk associated with compounded injectables.
Improved access programs
Novo Nordisk and other stakeholders are expanding patient access programs. Negotiated pricing through telehealth platforms, expanded insurance coverage mandates in some states, and new savings programs are all working to close the affordability gap. If brand-name pricing becomes more accessible, another major driver of compounded demand goes away.
Diet and lifestyle optimization while using semaglutide
Semaglutide is not a magic injection. It is a tool. And like any tool, its effectiveness depends on how you use it alongside other interventions. Whether your semaglutide is brand-name or compounded, these principles apply equally.
Nutrition during semaglutide therapy
Semaglutide reduces appetite significantly. This is the point. But reduced appetite can lead to reduced nutrient intake if you are not intentional about what you eat. When you eat less food overall, the quality of each bite matters more.
Protein becomes critical. Semaglutide-induced weight loss can include significant lean muscle loss if protein intake is inadequate. Research suggests consuming at least 1.0 to 1.2 grams of protein per kilogram of body weight daily to preserve muscle mass during GLP-1 therapy. Some experts recommend even higher intakes of 1.5 to 2.0 grams per kilogram for active individuals. Our semaglutide diet plan provides detailed nutritional frameworks.
Hydration is equally critical. The GI side effects of semaglutide, particularly nausea, vomiting, and diarrhea, can cause significant fluid loss. Reduced food intake also means reduced water intake from food sources. Aim for at least 64 ounces of water daily, more if you are experiencing GI symptoms.
Micronutrient supplementation deserves consideration. When eating less, you naturally consume fewer vitamins and minerals. A quality multivitamin, along with specific attention to vitamin D, iron, calcium, and B vitamins, helps prevent deficiencies that can develop during extended periods of reduced caloric intake. Knowing which foods to prioritize and which to avoid on GLP-1 therapy makes a real difference in outcomes.
Exercise and physical activity
Resistance training becomes more important, not less, when using semaglutide. The medication does not discriminate between fat loss and muscle loss. Your body will shed both unless you give it a reason to preserve muscle. That reason is resistance training.
Two to three sessions per week of progressive resistance training, targeting all major muscle groups, helps maintain lean mass during weight loss. This is not about bodybuilding. It is about preserving the metabolic tissue that keeps your metabolism healthy and your body functional as you lose weight.
Cardiovascular exercise complements the weight loss effects of semaglutide and provides independent cardiovascular health benefits. Walking is sufficient for most people starting out. As fitness improves, moderate-intensity activities like cycling, swimming, or jogging can be added.
Be aware that reduced caloric intake from semaglutide may temporarily reduce exercise capacity. You may feel less energetic during workouts, especially in the first few weeks at a new dose. This typically improves as your body adapts. Tracking your long-term timeline on semaglutide helps you understand what to expect at each phase.
Monitoring and tracking
Regular monitoring helps you and your healthcare provider evaluate treatment effectiveness and catch potential problems early.
Weekly weigh-ins: Same scale, same time of day, same conditions. Weight fluctuates daily, so weekly trends are more meaningful than daily numbers.
Body composition: If possible, track body fat percentage and lean mass in addition to weight. This helps distinguish between fat loss and muscle loss, which total body weight alone cannot do.
Lab work: Your physician should order baseline and periodic follow-up labs including fasting glucose, HbA1c, lipid panel, kidney function, liver function, and thyroid function. These help monitor both the therapeutic effects and potential adverse effects of semaglutide.
Side effect journaling: Keep a log of GI symptoms, energy levels, mood, and any other changes you notice. Patterns in this data can guide dose adjustments and identify problems early. SeekPeptides provides members with tracking tools and protocol guides to help organize this kind of data effectively.
Common mistakes people make with compounded semaglutide
After analyzing thousands of semaglutide protocols and user experiences, certain mistakes appear repeatedly. Avoiding these common pitfalls can dramatically improve your outcomes and safety.
Mistake 1: Escalating doses too quickly
Impatience is the most common enemy of successful semaglutide therapy. The escalation timeline feels slow when you want results now. But jumping from 0.25 mg to 1.0 mg in two weeks instead of eight is a recipe for miserable GI side effects that may cause you to abandon the medication entirely. The people who succeed with semaglutide are the ones who respect the titration schedule. Understanding why you might not lose weight initially and trusting the process is essential.
Mistake 2: Ignoring protein intake
This one is subtle and devastating. Semaglutide dramatically reduces appetite. You eat less. But if the food you do eat is low in protein, your body starts breaking down muscle for amino acids. Six months later, you have lost 30 pounds, but a significant portion of it was muscle. Your metabolism has slowed. You feel weak. You look "skinny fat" instead of healthy. Prioritize protein at every meal. Every single one.
Mistake 3: Not verifying vial concentration before drawing a dose
Compounded semaglutide comes in multiple concentrations. If your pharmacy switches you to a different concentration vial without clearly communicating the change, you could accidentally take double or half your intended dose. Always verify the concentration printed on your current vial before drawing up each dose. When in doubt, use our semaglutide dosage calculator to confirm your injection volume.
Mistake 4: Choosing a pharmacy based solely on price
The cheapest option is rarely the safest option. Pharmacies offering dramatically below-market prices are cutting costs somewhere. That somewhere might be quality testing, sterile processing, or source material verification. Saving $50 per month is not worth the risk of a contaminated or underpotent product.
Mistake 5: Stopping abruptly without a plan
Semaglutide suppresses appetite through active pharmacological intervention. When you stop, the appetite suppression goes away. Without a plan for maintaining your new habits, weight regain is common and can be significant. Research on semaglutide withdrawal shows that planning your exit strategy is just as important as planning your start.
Work with your physician to develop a maintenance plan before discontinuing. This might include a gradual dose reduction, increased focus on behavioral strategies, or a transition to a lower maintenance dose rather than complete cessation.
Mistake 6: Skipping medical supervision
Semaglutide is a prescription medication with real risks. Using it without regular physician oversight means nobody is monitoring your kidney function, checking for pancreatitis symptoms, adjusting your diabetes medications if applicable, or evaluating your overall response to treatment. The "set it and forget it" approach does not work safely with semaglutide, regardless of the source.
Frequently asked questions
Is compounded semaglutide the same as Ozempic?
Not exactly. Compounded semaglutide should contain the same active ingredient as Ozempic (semaglutide base), but it is prepared by a different facility with different inactive ingredients, different quality control standards, and without FDA approval. The delivery method is also different, with compounded products using vials and syringes instead of prefilled pens.
Can I still get compounded semaglutide after the shortage ended?
In limited circumstances, yes. If you have a documented medical need that the commercial product cannot meet, such as an allergy to an inactive ingredient, a physician can still prescribe compounded semaglutide. Cost alone is not a valid justification under current FDA regulations.
How do I know if my compounded semaglutide is safe?
Choose an accredited, FDA-registered 503B outsourcing facility whenever possible. Ask for Certificates of Analysis for your specific batch. Verify that the pharmacy uses semaglutide base (not salt forms). Ensure they conduct potency, sterility, and endotoxin testing on every batch. Following peptide safety protocols helps protect your health.
What is the difference between semaglutide base and semaglutide sodium?
Semaglutide base is the exact molecular form used in FDA-approved products. Semaglutide sodium is a salt form created by combining semaglutide with sodium. The FDA has stated that salt forms are not acceptable for compounding and may have different absorption, stability, and efficacy profiles compared to the base form.
How should I store compounded semaglutide?
Refrigerate at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Do not freeze. Store on an interior refrigerator shelf, not in the door. Once punctured, use within 28 days. Follow the beyond-use date on the label from your pharmacy. Our peptide storage guide covers these principles in comprehensive detail.
Can compounded semaglutide with B12 work better than plain semaglutide?
There is no clinical evidence that adding B12 to semaglutide improves weight loss outcomes. B12 may support energy levels independently, but the combination has not been studied in controlled trials. You can achieve similar potential benefits by taking B12 as a separate supplement alongside brand-name semaglutide.
What happens if I accidentally take too much compounded semaglutide?
Overdose symptoms typically include severe nausea, vomiting, diarrhea, and potentially dangerous dehydration. Contact your healthcare provider immediately or call Poison Control (1-800-222-1222). Severe cases may require emergency hydration and monitoring. Always double-check your dose calculations before injecting.
How long does it take for compounded semaglutide to start working?
Most people notice reduced appetite within the first one to two weeks. Weight loss typically becomes measurable after two to four weeks. Full therapeutic effects develop over several months as the dose is gradually escalated to maintenance levels. Our guide on how long semaglutide takes to work covers the complete timeline.
External resources
FDA: Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
PubMed: Administration Errors of Compounded Semaglutide Reported to Poison Control
Novo Nordisk: Difference Between FDA-Approved and Compounded Semaglutide
PubMed: Safety Analysis of Compounded GLP-1 Receptor Agonists (FAERS Study)
For researchers serious about understanding the nuances of GLP-1 therapy, SeekPeptides offers the most comprehensive resource available. Members get access to detailed protocol comparisons, evidence-based guides, dosing tools, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your formulations stay pure, your doses stay accurate, and your decisions stay informed.