Feb 22, 2026

CagriSema and tirzepatide look nearly identical on paper. Both produce over 20% body weight reduction in clinical trials. Both require once-weekly injections. Both target multiple metabolic pathways simultaneously. But dig deeper into the clinical data, the receptor pharmacology, and the real-world side effect profiles, and these two treatments diverge in ways that matter enormously for researchers trying to decide between them.
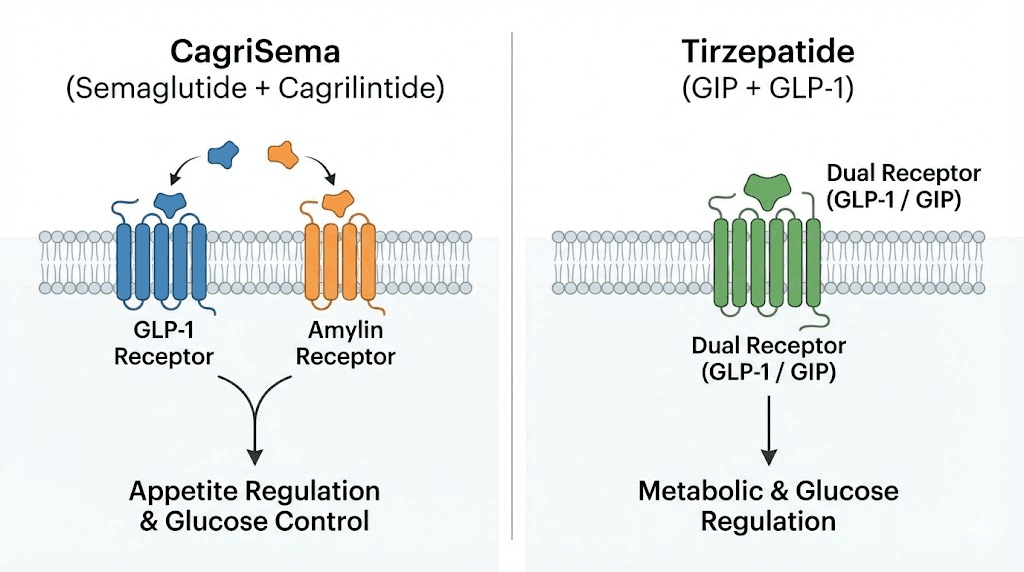
One is a single molecule activating two receptors. The other is a combination of two separate compounds working through entirely different signaling cascades. The distinction sounds academic until you consider what it means for appetite suppression, muscle preservation, metabolic adaptation, and long-term outcomes. These are not interchangeable treatments wearing different labels.
Tirzepatide arrived first, delivering unprecedented results in the SURMOUNT clinical trials and earning FDA approval as Zepbound. CagriSema followed closely behind, posting equally impressive numbers in the REDEFINE trials before filing for FDA review. The question everyone is asking is simple. Which one is better? The answer, as researchers exploring CagriSema dosing protocols and tirzepatide dosing for weight loss have discovered, depends entirely on what you prioritize. This guide breaks down everything you need to know. From receptor pharmacology and weight loss percentages to gastrointestinal side effects and body composition changes, every comparison here draws from published clinical trial data. Not speculation. Not marketing materials. The actual numbers from peer-reviewed research published in the New England Journal of Medicine and presented at major medical conferences.
SeekPeptides has compiled the most comprehensive analysis of these two treatments available anywhere, incorporating data from multiple phase 3 trials, mechanistic studies, and the latest regulatory filings.
What is CagriSema and how does it work?
CagriSema is a fixed-dose combination therapy containing two active compounds administered together in a single injection. The first component is cagrilintide, a long-acting amylin receptor agonist. The second is semaglutide, a GLP-1 receptor agonist that researchers already know from Wegovy and Ozempic. Together, these compounds activate two distinct satiety pathways simultaneously, something neither component achieves alone.
The concept behind CagriSema reflects a growing understanding that obesity involves multiple overlapping hormonal systems. Targeting just one pathway produces significant results. But adding a second pathway amplifies the overall effect. The whole becomes greater than the sum of its parts.
Novo Nordisk developed CagriSema as the successor to their blockbuster semaglutide therapy, recognizing that even 2.4 mg of standalone semaglutide left room for improvement. The combination approach was inspired by research showing that amylin and GLP-1 act on different brain regions to reduce appetite. By activating both systems simultaneously, CagriSema achieves deeper and more sustained appetite suppression than either compound alone.
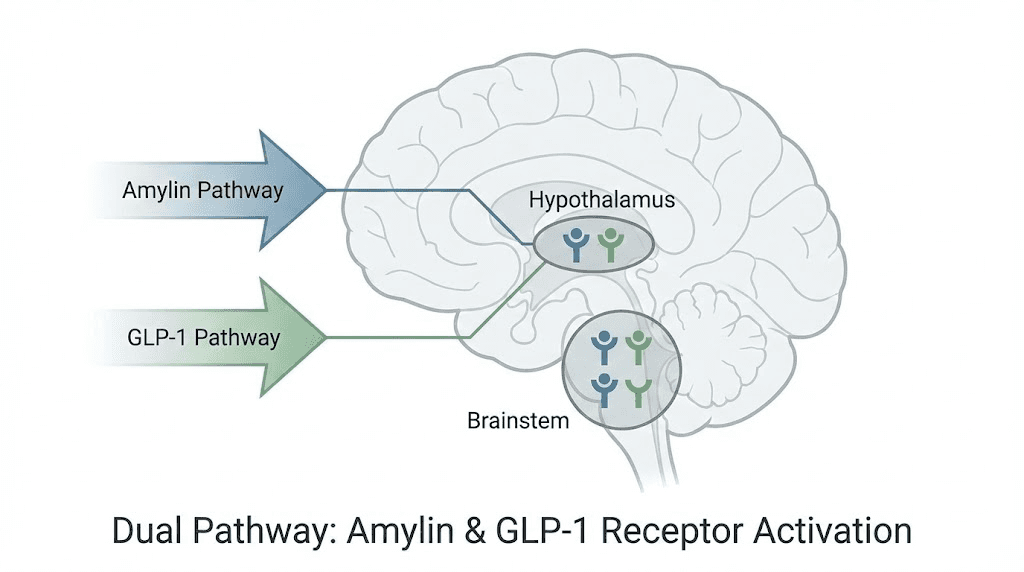
The amylin pathway explained
Amylin is a hormone co-secreted with insulin from pancreatic beta cells after meals. It serves as one of the primary satiety signals in the body, telling the brain that enough food has been consumed. Natural amylin slows gastric emptying, suppresses glucagon secretion, and activates receptors in the area postrema and nucleus of the solitary tract. These are two brainstem regions critical for controlling food intake and meal termination.
Cagrilintide is an engineered analog of amylin with a dramatically extended half-life. While natural amylin disappears from the bloodstream within minutes, cagrilintide maintains therapeutic levels for an entire week. This sustained activation of amylin receptors produces continuous appetite suppression between doses, and cagrilintide weight loss data confirms the compound works even as a standalone treatment.
The amylin pathway operates through distinct neural circuits compared to GLP-1. Where GLP-1 primarily activates hypothalamic neurons involved in energy balance, amylin targets hindbrain circuits that regulate meal-specific satiety and food reward. This is why combining the two pathways produces additive effects. They suppress appetite through different neurological mechanisms, covering more ground than either pathway can cover alone.
Research from the REDEFINE 1 trial shows that cagrilintide alone achieved 11.5% weight loss at 68 weeks. Respectable on its own. But when combined with semaglutide, that number nearly doubled. The synergy between cagrilintide and semaglutide is not merely additive. It appears to be genuinely synergistic, with the combination producing more weight loss than you would expect from simply adding the individual effects together.
The GLP-1 component
GLP-1, or glucagon-like peptide-1, is an incretin hormone released from intestinal L-cells after eating. It enhances insulin secretion, suppresses glucagon release, slows gastric emptying, and reduces appetite through central nervous system effects. The GLP-1 receptor appears throughout the brain, pancreas, and gastrointestinal tract, making it one of the most versatile metabolic targets available.
Semaglutide activates GLP-1 receptors with high potency and an extended duration of action. The molecule incorporates a C18 fatty acid side chain that promotes albumin binding, extending the half-life to approximately one week. This allows for once-weekly dosing while maintaining consistent receptor activation between injections.
Appetite suppression from GLP-1 activation occurs primarily through hypothalamic neurons, particularly in the arcuate nucleus. These neurons integrate energy balance signals and coordinate feeding behavior. When GLP-1 receptors on these neurons are activated, the result is reduced hunger, increased satiety, and decreased food preoccupation. Researchers studying how fast semaglutide works typically observe appetite changes within the first week, with clinically meaningful weight loss emerging by weeks 4 to 8.
For researchers familiar with semaglutide appetite suppression timelines, the GLP-1 component of CagriSema follows the same general pattern. The difference is that the amylin component adds a second layer of appetite control operating through completely separate neural circuits.
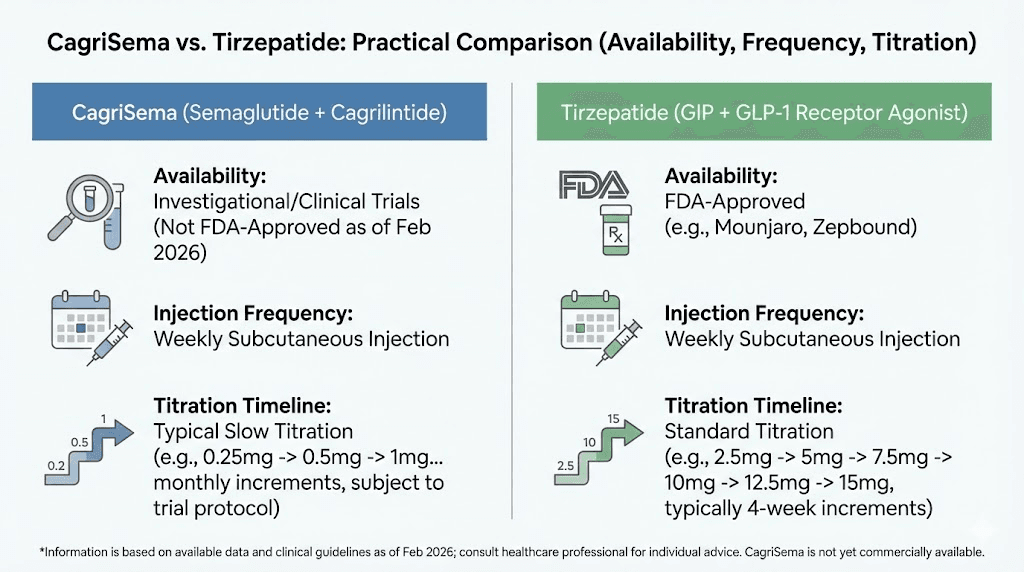
CagriSema dosing schedule
The CagriSema dosing protocol follows a gradual titration designed to minimize gastrointestinal side effects while reaching the full therapeutic dose. Treatment begins at 0.25 mg semaglutide combined with a proportional cagrilintide dose, with increases occurring every four weeks.
The typical escalation moves from 0.25 mg to 0.5 mg at week 5, then to 1 mg at week 9, followed by 1.7 mg at week 13, and finally reaching the full maintenance dose of 2.4 mg semaglutide with 2.4 mg cagrilintide at week 17. This 16-week titration period is essential. Jumping to the full dose immediately would produce severe nausea and vomiting in most people.
Researchers comparing this to standalone cagrilintide dosing will notice the escalation schedule mirrors what semaglutide requires on its own. The cagrilintide component does not add additional titration complexity because both compounds reach their maintenance doses on the same timeline. For those interested in combining cagrilintide with tirzepatide instead, the dosing considerations become more complex since that represents an off-label combination.
What is tirzepatide and how does it work?
Tirzepatide takes a fundamentally different approach. Rather than combining two separate compounds, tirzepatide is a single molecule engineered to activate two receptors simultaneously. It functions as a dual GIP/GLP-1 receptor agonist, meaning one peptide chain binds to and activates both the glucose-dependent insulinotropic polypeptide receptor and the GLP-1 receptor.
This is an important distinction. CagriSema is two drugs in one syringe. Tirzepatide is one drug that does two things. The practical implications of this difference extend to manufacturing, patent protection, dosing flexibility, and potentially even clinical outcomes.
Eli Lilly developed tirzepatide based on the observation that GIP and GLP-1 produce complementary metabolic effects. GIP was long considered a problematic hormone in obesity research because it appeared to promote fat storage. But newer research revealed that GIP receptor activation in the brain actually reduces appetite, and when combined with GLP-1 receptor activation, the metabolic benefits multiply.
The dual GIP/GLP-1 mechanism
The tirzepatide molecule was engineered with what researchers call imbalanced agonism. It activates the GIP receptor with approximately equal potency to native GIP, but activates the GLP-1 receptor with roughly five times lower potency than native GLP-1. This imbalance is intentional. The GIP component does much of the heavy lifting for insulin sensitization and fat metabolism, while the GLP-1 component handles appetite suppression and glucose regulation.
GIP receptor activation in the brain engages neural circuits involved in food reward and energy homeostasis. Unlike amylin, which primarily targets brainstem satiety centers, GIP acts on hypothalamic and mesolimbic pathways. This means tirzepatide suppresses appetite through a mechanistic profile that partially overlaps with GLP-1 but adds unique GIP-mediated effects on fat metabolism and energy expenditure.
In pancreatic beta cells, the combination of GIP and GLP-1 receptor activation produces synergistic insulin secretion. Studies show that tirzepatide elicits a cyclic AMP response in human beta cells significantly higher than what either GIP or GLP-1 produces alone. This enhanced insulin response contributes to superior glycemic control, which is why tirzepatide performs exceptionally well in populations with type 2 diabetes. Researchers tracking how fast tirzepatide works typically observe rapid improvements in blood sugar even before significant weight loss occurs.
The molecule incorporates a C20 diacid fatty acid side chain, slightly longer than the C18 side chain used in semaglutide. This modification promotes albumin binding and extends the half-life to approximately five days, enabling once-weekly dosing. The structural similarity to semaglutide is notable. Both are peptide chains with fatty acid modifications for extended duration. But the additional GIP receptor binding domain makes tirzepatide a fundamentally different pharmacological tool.
Tirzepatide dosing schedule
Tirzepatide follows a dose escalation from 2.5 mg to the maximum approved dose of 15 mg, with increases occurring every four weeks. The starting dose of 2.5 mg is maintained for four weeks, followed by 5 mg for four weeks, then 7.5 mg, 10 mg, 12.5 mg, and finally 15 mg. Not all researchers reach the maximum dose. Many find adequate results at 10 mg or 12.5 mg with fewer side effects.
The tirzepatide dosing in units can be confusing for those working with compounded formulations. The relationship between milligrams and units depends entirely on the concentration of the solution, and our tirzepatide dose chart breaks this down clearly. For compounded formulations, the tirzepatide compound dosing chart provides the specific calculations needed.
Some researchers explore microdosing tirzepatide as an alternative to full-dose protocols, particularly those who experience significant gastrointestinal side effects at standard doses. Oral tirzepatide formulations are also under development, and the oral versus injection comparison reveals meaningful differences in bioavailability and onset timing.
Weight loss results compared
This is where most researchers start. And the numbers are genuinely impressive on both sides.
CagriSema clinical trial results
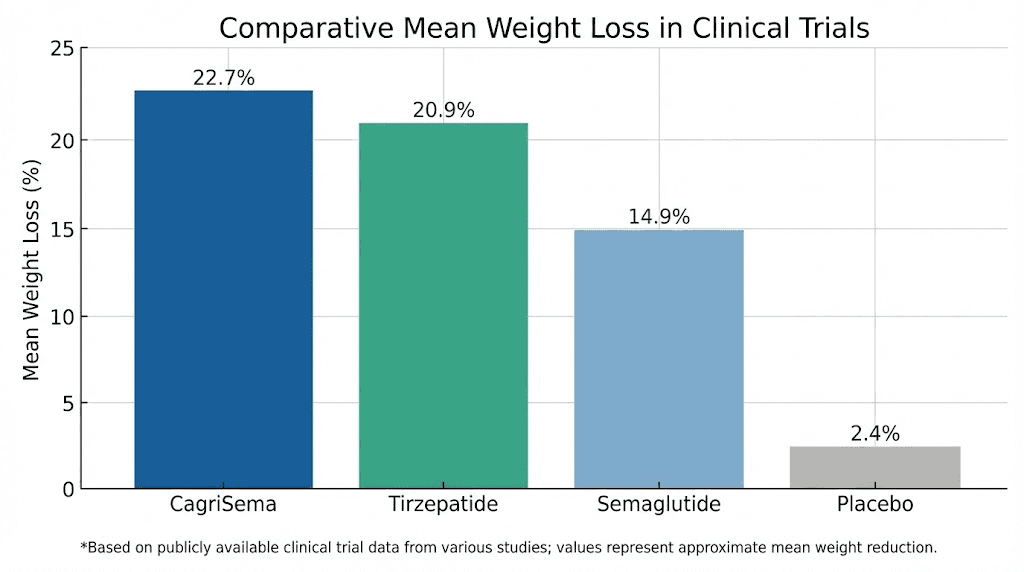
The REDEFINE 1 trial enrolled adults with obesity or overweight (BMI of 30 or greater, or 27 or greater with at least one weight-related comorbidity) without type 2 diabetes. At 68 weeks, CagriSema produced a mean body weight reduction of 22.7% from baseline. That number landed below the 25% target that Novo Nordisk had projected, causing brief concern among investors, but it still represents one of the largest weight loss outcomes ever achieved with a pharmacological intervention.
Looking deeper into the data, 60% of participants on CagriSema achieved at least 20% weight loss. Nearly one in four, 23% specifically, lost 30% or more of their body weight. These are surgical-level results from a once-weekly injection. For context, the placebo group lost just 3%, while standalone semaglutide 2.4 mg produced 16.1% weight loss, and standalone cagrilintide achieved 11.5%.
The REDEFINE 2 trial examined CagriSema in adults with type 2 diabetes, a population that typically shows less weight loss with GLP-1 therapies. Even here, CagriSema delivered 13.7% mean weight reduction at 68 weeks compared to 3.4% with placebo. Beyond weight loss, 73.5% of participants achieved hemoglobin A1c levels at or below 6.5%, compared to just 15.9% in the placebo group. The glycemic improvements were dramatic.
What sets these results apart is not just the magnitude of weight loss but the consistency. The response curves show that most participants were still losing weight at week 68, suggesting that longer treatment durations might produce even greater reductions. Researchers studying optimal treatment duration for GLP-1 therapies will find this particularly relevant.
Tirzepatide clinical trial results
The SURMOUNT-1 trial tested tirzepatide in adults with obesity or overweight without type 2 diabetes. The results established tirzepatide as the most effective anti-obesity medication available at the time of publication. At 72 weeks, the 15 mg dose produced a mean weight loss of 22.5%. The 10 mg dose achieved 21.4%, and even the 5 mg dose delivered 16.0%.
The response rates tell an equally compelling story. At the 15 mg dose, 63% of participants lost at least 20% of their body weight, and 36% lost 25% or more. These numbers were unprecedented when first published and set the benchmark that all subsequent obesity treatments have been measured against. For visual context, the tirzepatide before and after results documented across clinical trials and real-world reports show transformative changes, and before and after tirzepatide weight loss results continue to accumulate as more researchers complete full treatment courses.
In the SURMOUNT-2 trial for adults with type 2 diabetes, tirzepatide 15 mg produced 14.7% mean weight loss at 72 weeks. The glycemic improvements were also substantial, with significant reductions in A1c across all dose groups. These diabetes-specific results mirror the pattern seen with CagriSema in REDEFINE 2, though direct comparison requires caution because the trial populations and designs differed.
Researchers who have experienced the tirzepatide response timeline typically report noticeable appetite changes within the first two weeks, with measurable weight loss by weeks 4 to 6. The full effect takes several months to develop, particularly at lower doses during the titration phase. Those wondering about tirzepatide appetite suppression timing should expect gradual intensification as the dose increases.
Head-to-head comparison table
Measure | CagriSema (REDEFINE 1) | Tirzepatide 15 mg (SURMOUNT-1) |
|---|---|---|
Trial duration | 68 weeks | 72 weeks |
Mean weight loss | 22.7% | 22.5% |
Achieved 20%+ loss | 60% | 63% |
Achieved 25%+ loss | Not reported separately | 36% |
Placebo weight loss | 3.0% | 3.1% |
Population | BMI 30+ or 27+ with comorbidity | BMI 30+ or 27+ with comorbidity |
Mechanism | Amylin + GLP-1 (two compounds) | GIP + GLP-1 (single molecule) |
The numbers are remarkably close. CagriSema achieved 22.7% in 68 weeks. Tirzepatide achieved 22.5% in 72 weeks. On paper, CagriSema produced a marginally higher percentage in slightly less time. But clinical trial comparisons across different studies carry significant caveats. Different patient populations, different enrollment criteria, different sites, and different measurement protocols all introduce variability that makes direct numerical comparison imprecise.
This is exactly why the REDEFINE 4 trial exists. More on that later. For now, the most accurate statement is that both treatments produce approximately 20 to 23% weight loss in non-diabetic populations with obesity, and both are in a class far beyond anything that came before them. Researchers who have been tracking the semaglutide versus tirzepatide comparison will recognize that CagriSema fundamentally changes the competitive landscape by adding amylin to the equation.
Side effects and safety profiles
Weight loss numbers grab headlines. Side effects determine whether people actually complete treatment. Both CagriSema and tirzepatide carry gastrointestinal side effects as their primary safety concern, but the details diverge in meaningful ways.
CagriSema side effects
In the REDEFINE 1 trial, gastrointestinal adverse events affected 79.6% of participants receiving CagriSema, compared to 39.9% in the placebo group. The most common complaints were nausea (55% versus 12.6% with placebo), constipation (30.7% versus 11.6%), and vomiting (26.1% versus 4.1%). Diarrhea and abdominal pain also appeared frequently.
The severity profile matters more than the raw percentages. Most gastrointestinal events were classified as mild to moderate, and the majority were transient, resolving within the first several weeks of treatment or after dose stabilization. The discontinuation rate due to adverse events was 6% for CagriSema versus 3.7% for placebo. A 6% dropout rate for a treatment producing 22.7% weight loss is remarkably low.
Beyond gastrointestinal issues, CagriSema showed a pattern consistent with the known GLP-1 receptor agonist class. Injection site reactions occurred in a small percentage of participants. Increases in heart rate were observed, consistent with semaglutide therapy. And as with all effective weight loss treatments, there were cases of gallbladder-related events, which are associated with rapid weight reduction regardless of the method used.
Researchers concerned about specific side effects can explore detailed guides on cagrilintide side effects for information specific to the amylin component. Understanding which side effects come from which component helps in managing tolerance and making informed decisions.
Tirzepatide side effects
Tirzepatide side effects follow a similar gastrointestinal pattern but with some notable differences. In SURMOUNT-1, the most common adverse events at the 15 mg dose included nausea (31%), diarrhea (23%), constipation (11%), and vomiting (12%). These percentages are notably lower than those reported for CagriSema, though cross-trial comparisons are imperfect.
The semaglutide versus tirzepatide side effects comparison shows that tirzepatide generally produces less nausea than pure GLP-1 agonists at equivalent weight loss levels. The GIP component may actually buffer some of the gastrointestinal distress that GLP-1 activation alone produces. This is one of the theoretical advantages of the dual GIP/GLP-1 approach over the amylin/GLP-1 approach.
Fatigue with tirzepatide is a commonly reported concern, though clinical trial data does not list it as a top adverse event. Many researchers attribute tiredness to caloric restriction rather than a direct drug effect. Similarly, anxiety with tirzepatide and headaches appear in anecdotal reports more often than in controlled trial data. Muscle pain affects a subset of users and may relate to rapid body composition changes.
For managing gastrointestinal issues specifically, tirzepatide constipation treatment strategies and dietary modifications like those outlined in the tirzepatide foods to avoid guide can make a significant difference in tolerability. Proper nutrition during treatment, as described in our tirzepatide diet plan and what to eat on tirzepatide guides, helps minimize side effects while maximizing results.
Side effect comparison table
Side effect | CagriSema (REDEFINE 1) | Tirzepatide 15 mg (SURMOUNT-1) |
|---|---|---|
Any GI event | 79.6% | ~51% |
Nausea | 55% | 31% |
Vomiting | 26.1% | 12% |
Constipation | 30.7% | 11% |
Diarrhea | Reported frequently | 23% |
Discontinuation rate | 6% | 6.2% |
The nausea and vomiting rates for CagriSema are substantially higher than for tirzepatide. This likely reflects the additive gastrointestinal effects of combining two separate appetite-suppressing compounds. The amylin pathway slows gastric emptying through its own mechanism, and when layered on top of GLP-1 mediated gastric slowing, the combined effect on the gut is more pronounced.
However, the discontinuation rates are virtually identical. This suggests that while CagriSema causes more frequent gastrointestinal symptoms, those symptoms are generally manageable enough that participants continue treatment. The tolerability gap may be less significant in practice than the raw nausea percentages suggest. Many of the same management strategies that work for semaglutide constipation also apply to CagriSema side effects.
Body composition and muscle preservation
Weight loss alone tells an incomplete story. What kind of weight you lose matters enormously for long-term health and metabolic function. Losing primarily fat while preserving lean muscle mass produces far better outcomes than losing a proportional mix of both.
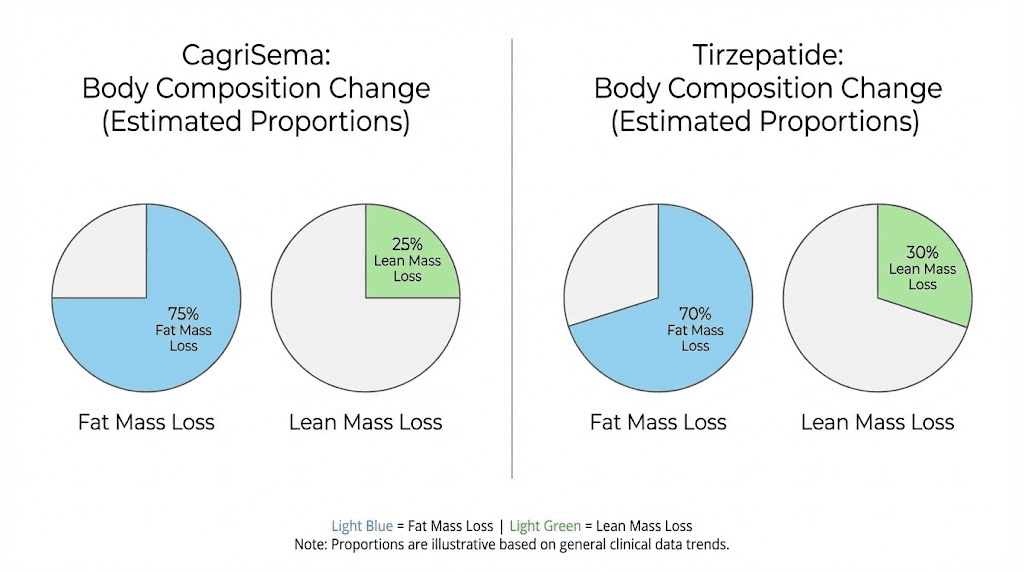
CagriSema data from body composition scans in REDEFINE 1 showed that approximately 67% of total weight loss came from fat mass and 33% from lean soft tissue mass. This ratio is consistent with what most pharmacological weight loss interventions produce, though it is slightly more favorable toward fat loss than what surgical interventions typically achieve.
Preclinical research on CagriSema revealed a particularly interesting finding. In animal models, CagriSema preserved energy expenditure during weight loss by maintaining mitochondrial leak respiration in skeletal muscle. This mechanism prevents the metabolic slowdown that typically accompanies significant weight loss. When the body loses weight, it normally reduces energy expenditure as a compensatory survival mechanism. CagriSema appears to partially prevent this adaptation, which could translate to more durable weight maintenance over time.
Tirzepatide body composition data from SURMOUNT-1 showed a similar pattern. Approximately two-thirds of weight loss was fat mass, with one-third coming from lean tissue. The GIP receptor activation in adipose tissue may provide additional benefits for fat mobilization and metabolism, though the clinical significance of these mechanistic differences requires further study.
For researchers concerned about muscle preservation during weight loss, several strategies can help regardless of which treatment is chosen. Adequate protein intake, resistance training, and proper supplementation during treatment all contribute to maintaining lean mass. Adding compounds like glycine with tirzepatide or glycine and B12 combinations may provide additional support for connective tissue and muscle metabolism. Some researchers also explore tirzepatide with B12 to support energy levels and prevent deficiency during rapid weight loss.
The bottom line on body composition is that neither treatment shows a clear advantage over the other based on available data. Both produce roughly two-thirds fat loss and one-third lean tissue loss. The real differentiator may be the metabolic preservation effects seen with CagriSema in preclinical models, but human data confirming this advantage is still limited.
Blood sugar control and metabolic benefits
Both CagriSema and tirzepatide produce substantial improvements in glycemic markers, but the mechanisms driving these improvements differ, and the magnitude varies between populations.
In populations with type 2 diabetes, tirzepatide has demonstrated exceptional glycemic control. The SURPASS trials showed A1c reductions of 1.24% to 2.58% depending on the dose and comparator. At the highest dose, more participants achieved normal A1c levels than with any other incretin-based therapy tested at the time. The dual GIP/GLP-1 mechanism provides synergistic insulin secretion from pancreatic beta cells. GIP enhances the first phase of insulin release, while GLP-1 sustains the second phase. Together, they produce insulin responses that neither receptor system generates alone.
CagriSema showed strong glycemic results in REDEFINE 2 as well, with 73.5% of participants achieving A1c levels at or below 6.5%. The amylin component contributes to blood sugar control through glucagon suppression and delayed gastric emptying, both of which reduce postprandial glucose spikes. While the mechanisms differ from tirzepatide, the clinical outcomes are comparable for most patients with type 2 diabetes.
Beyond glucose control, both treatments improve multiple cardiometabolic risk factors. Blood pressure reductions, improvements in lipid profiles (lower triglycerides, higher HDL), reductions in waist circumference, and decreases in inflammatory markers have been documented for both. CagriSema data additionally showed anti-inflammatory effects and significant blood pressure reductions in analyses presented at ObesityWeek, suggesting cardiovascular benefits that extend beyond weight loss alone.
An important finding for researchers monitoring metabolic health: 88% of CagriSema participants who had prediabetes at baseline returned to normoglycemia by the end of the REDEFINE 1 trial. This diabetes prevention effect could prove to be one of the most significant clinical benefits of effective obesity pharmacotherapy, regardless of which specific treatment is used. For those already researching GLP-1 based therapies, guides on the relationship between GLP-1 and Ozempic and GLP-1 companion resources provide additional metabolic context.
Availability, regulatory status, and access
Practical availability often matters as much as clinical efficacy. A treatment producing 25% weight loss is irrelevant if you cannot obtain it.
Tirzepatide has a significant head start. It received FDA approval as Mounjaro for type 2 diabetes and subsequently as Zepbound for weight management. It is commercially available now, prescribed by physicians, and covered by an increasing number of insurance plans. Compounded tirzepatide formulations are also widely accessible, and researchers can find detailed information in guides covering affordable tirzepatide options and compounded tirzepatide pricing.
For those working with compounded formulations, proper handling is essential. Understanding how to reconstitute tirzepatide, knowing refrigeration requirements, and following correct storage duration guidelines protect the integrity of the compound. The tirzepatide expiration considerations also matter for anyone managing inventory.
CagriSema is not yet available. Novo Nordisk filed a New Drug Application with the FDA in late December of the previous year for the weight management indication. The FDA review process typically takes 10 to 12 months from filing, meaning a potential approval decision could come in late this year or early next year. Even after approval, manufacturing ramp-up and commercial launch logistics mean CagriSema likely will not be widely available to patients until the following year.
This timeline gap matters. Researchers choosing between the two are not choosing between equivalent options today. They are choosing between a treatment available now (tirzepatide) and one that may become available in the next 12 to 18 months (CagriSema). For researchers who cannot wait, the decision is already made. Tirzepatide is the accessible option. Compounded sources for tirzepatide through pharmacies like Empower Pharmacy and other vendors provide current access.
Cost will also factor into the eventual comparison. Branded obesity medications carry significant price tags, and insurance coverage varies dramatically. The peptide cost calculator can help researchers estimate ongoing treatment expenses across different sources and formulations.
The REDEFINE 4 trial: the definitive showdown
Everything discussed above relies on cross-trial comparisons, which carry inherent limitations. Different patient populations, different trial designs, different enrollment criteria, and different measurement protocols all introduce noise that makes precise numerical comparisons between CagriSema and tirzepatide unreliable.
REDEFINE 4 is designed to eliminate that noise. This phase 3 trial directly compares CagriSema 2.4 mg/2.4 mg against tirzepatide 15 mg in approximately 800 adults with obesity. Both treatments are administered once weekly for 72 weeks. There is no placebo arm. The primary endpoint is percent change in body weight from baseline.
The trial is open-label, meaning both investigators and participants know which treatment they are receiving. This design choice introduces potential bias, particularly for subjective outcomes, but it reflects the practical reality that these treatments have different injection devices and titration schedules that would make blinding extremely difficult.
When REDEFINE 4 results are published, they will provide the first and only direct comparison between these two treatments using the same population, the same endpoints, and the same measurement protocols. Until then, every comparison, including this one, relies on imperfect cross-trial analysis.
What researchers should watch for in REDEFINE 4 beyond the headline weight loss number includes several secondary outcomes. Body composition differences, particularly the fat-to-lean mass ratio. Glycemic improvements in participants with prediabetes. Side effect profiles under identical monitoring conditions. Treatment satisfaction and quality-of-life measures. And perhaps most importantly, the rate of treatment discontinuation, which reveals how tolerable each treatment is in the real world rather than in the controlled environment of separate trials.
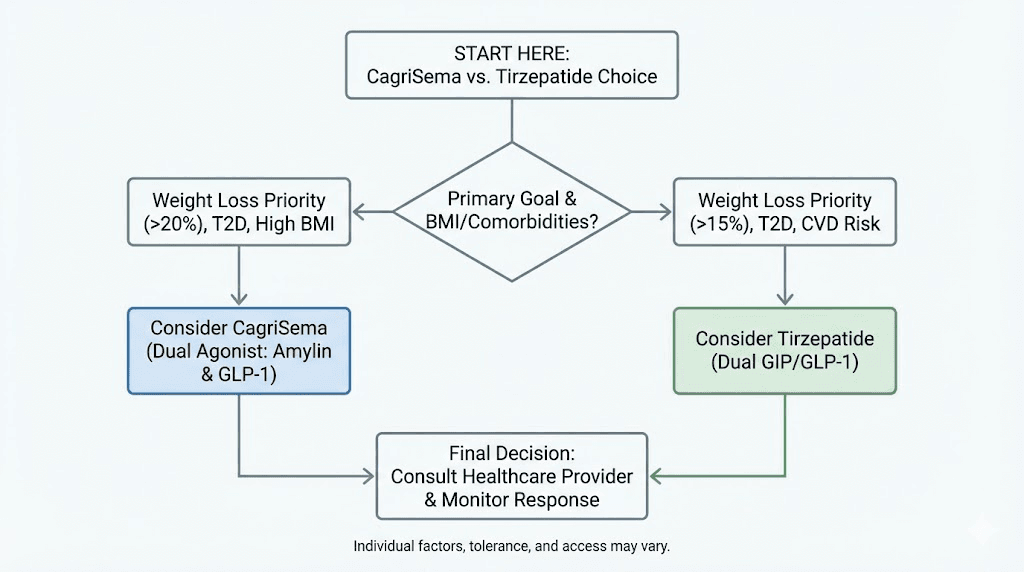
Which one should you choose?
The comparison between CagriSema and tirzepatide does not produce a single winner. Instead, it reveals two distinct approaches to the same problem, each with advantages in specific contexts.
CagriSema may be the better choice when
Researchers who have plateaued on standalone semaglutide therapy may find CagriSema particularly valuable. Since CagriSema adds the amylin pathway on top of GLP-1 activation, it offers a mechanistic upgrade for those who have already maximized the GLP-1 response alone. The semaglutide plateau troubleshooting guide covers common reasons for stalling, and CagriSema represents a pharmacological solution to that plateau by engaging an entirely new receptor system.
The metabolic preservation data is also noteworthy. If CagriSema truly prevents the metabolic slowdown associated with weight loss, as the preclinical data suggests, it could produce more durable outcomes with less weight regain after treatment completion. Researchers concerned about rebound effects after stopping GLP-1 therapies may find this mechanism particularly appealing.
CagriSema also makes sense for researchers interested in the amylin pathway specifically. The cagrilintide research for men and the broader cagrilintide weight loss data point to unique benefits of amylin receptor activation that extend beyond what GLP-1 or GIP agonism provides.
Tirzepatide may be the better choice when
Availability is the most obvious advantage. Tirzepatide is here now. CagriSema is not. For researchers ready to begin treatment, tirzepatide offers immediate access through both branded and compounded formulations.
Gastrointestinal tolerability is another significant factor. The lower nausea and vomiting rates with tirzepatide compared to CagriSema suggest it may be easier to tolerate, particularly during the titration phase. Researchers who are sensitive to gastrointestinal side effects might prefer the tirzepatide profile. The tirzepatide meal plan and dietary strategies can further improve tolerability.
For those with type 2 diabetes or significant insulin resistance, the dual GIP/GLP-1 mechanism may offer advantages for glycemic control. The synergistic insulin secretion produced by simultaneous GIP and GLP-1 receptor activation is mechanistically distinct from what amylin/GLP-1 combinations provide, and the SURPASS trial data in diabetic populations is among the strongest in the field.
Dose flexibility is another consideration. Tirzepatide is available in multiple dose strengths from 2.5 mg to 15 mg, allowing researchers to find their optimal balance between efficacy and side effects. CagriSema, at least in its current clinical development program, targets a single maintenance dose of 2.4/2.4 mg, offering less room for individualization.
Researchers who have struggled with tirzepatide losing effectiveness over time might consider whether switching to CagriSema when it becomes available could reignite weight loss through the different amylin-based mechanism. Conversely, those exploring switching between treatments should understand the conversion charts and dose equivalence guides to manage transitions safely.
How CagriSema and tirzepatide compare to other weight loss peptides
The obesity treatment landscape extends well beyond these two options. Understanding where CagriSema and tirzepatide sit relative to other emerging therapies provides essential context for informed decision-making.
Versus retatrutide (the triple agonist)
Retatrutide represents the next frontier. As a triple GLP-1/GIP/glucagon receptor agonist, it adds glucagon receptor activation on top of the dual mechanisms that tirzepatide already provides. Phase 2 data showed mean weight loss of up to 24.2% at 48 weeks, which, if confirmed in phase 3 trials, would surpass both CagriSema and tirzepatide.
The glucagon component increases energy expenditure and fat oxidation, potentially addressing the metabolic adaptation problem from yet another angle. Researchers tracking retatrutide versus semaglutide data and retatrutide dosing protocols will find that this triple agonist could eventually overtake both CagriSema and tirzepatide. The retatrutide dose schedule and dosage calculator provide practical guidance for researchers already exploring this compound.
For a more detailed tirzepatide comparison, the retatrutide versus tirzepatide dosage chart lays out the differences in dosing approaches. The retatrutide availability situation and cost considerations remain significant barriers to access, with options for purchasing retatrutide and finding retatrutide online still limited compared to tirzepatide.
Versus semaglutide alone
Semaglutide 2.4 mg (Wegovy) produces approximately 15 to 17% weight loss in clinical trials. Both CagriSema and tirzepatide substantially outperform this benchmark. For researchers currently on semaglutide who feel they have room for more weight loss, either CagriSema or tirzepatide represents a meaningful upgrade.
The transition from semaglutide to tirzepatide is well documented. The tirzepatide versus semaglutide dosage chart and semaglutide to tirzepatide dose conversion guide provide the practical framework for switching. Since CagriSema already contains semaglutide 2.4 mg, transitioning from standalone semaglutide to CagriSema would theoretically be simpler, though clinical guidance on this transition is limited.
Understanding semaglutide treatment timelines and appetite suppression onset helps researchers set realistic expectations when comparing single versus combination therapies. The semaglutide dosage calculator remains a useful tool for those still using the standalone formulation.
Versus survodutide and mazdutide
The weight loss peptide pipeline includes additional contenders. Survodutide, a dual GLP-1/glucagon receptor agonist, takes yet another approach by combining GLP-1 with glucagon rather than GIP or amylin. Early data shows promising weight loss and particularly strong effects on liver fat reduction, making it potentially valuable for patients with non-alcoholic fatty liver disease.
Mazdutide, developed by Innovent Biologics, is another GLP-1/glucagon dual agonist showing competitive weight loss results in Asian populations. The mazdutide versus retatrutide comparison highlights how different dual and triple agonist approaches are all converging on the same goal of maximizing weight loss while preserving metabolic health.
The broader picture is that the next generation of obesity treatments involves multi-target approaches, whether through single molecules activating multiple receptors (tirzepatide, retatrutide, survodutide, mazdutide) or through combination therapies pairing separate compounds (CagriSema). Both strategies are proving far more effective than single-target GLP-1 agonism.
Practical considerations researchers should know
Beyond the clinical data, several practical factors influence the CagriSema versus tirzepatide decision that many comparison articles overlook.
Injection preparation and storage
Tirzepatide in its commercial form (Mounjaro/Zepbound) comes in pre-filled auto-injector pens requiring no preparation. Compounded tirzepatide typically comes as lyophilized powder requiring reconstitution. The reconstitution process is straightforward but requires attention to detail. Proper storage, as covered in our guides on compounded tirzepatide storage duration and refrigeration requirements, protects potency and safety.
CagriSema in its anticipated commercial form will likely also come as a pre-filled pen. Compounded or research-grade combinations of cagrilintide and semaglutide require separate reconstitution and potentially separate injections, adding complexity. Information on purchasing cagrilintide covers the current options for researchers working with these compounds individually.
Dietary and lifestyle considerations
Both treatments work best when combined with proper nutrition. The foods to eat during GLP-1 therapy apply to both CagriSema (which contains semaglutide) and tirzepatide (which activates the same GLP-1 pathway). High-protein, nutrient-dense meals support muscle preservation while the appetite suppression from either treatment naturally reduces caloric intake.
Alcohol consumption is a common question. Guides on drinking on tirzepatide and drinking on semaglutide apply to CagriSema as well. The general recommendation is moderation, as both treatments can alter alcohol tolerance and increase the risk of hypoglycemia when combined with alcohol.
For researchers looking at broader peptide stacking strategies, the peptide stack calculator helps determine compatible combinations. And for those tracking overall treatment costs across different approaches, the peptide cost calculator provides useful estimates.
Injection technique and site selection
Proper injection technique matters for both treatments. The GLP-1 injection site guide and GLP-1 injection technique guide cover the fundamentals. Rotating injection sites reduces the risk of lipohypertrophy and local reactions. The abdomen, thigh, and upper arm are all acceptable sites for both CagriSema and tirzepatide.
The optimal injection timing guidance for semaglutide applies to the semaglutide component of CagriSema. Consistency matters more than the specific time of day, though some researchers prefer morning injections to manage initial nausea during waking hours when they can eat lightly and stay hydrated.
For researchers exploring delivery alternatives, tirzepatide drops, tirzepatide tablets versus injections, and sublingual tirzepatide dosing represent alternative administration routes worth investigating. Similarly, oral semaglutide drops and even GLP-1 patches are expanding the options for non-injectable delivery.
The bigger picture: why multi-target therapies represent the future
The CagriSema versus tirzepatide comparison is really a proxy for a larger question in obesity medicine. Is it better to combine separate compounds targeting different pathways, or to engineer single molecules that activate multiple receptors simultaneously?
The combination approach (CagriSema) offers theoretical advantages in flexibility. Each component can be dosed independently in research settings, and new combinations can be created without engineering entirely new molecules. If a third pathway proves beneficial, you simply add a third compound.
The single-molecule approach (tirzepatide, retatrutide) offers advantages in simplicity and potentially in pharmacokinetics. One injection, one molecule, coordinated receptor activation with a defined ratio of potencies. Manufacturing is simpler, and there are fewer drug-drug interaction concerns because there is only one drug.
Both approaches are producing results that seemed impossible just a decade ago. Pharmacological weight loss of 20% or more was the domain of bariatric surgery. Now, injectable therapies are matching and in some cases exceeding surgical outcomes without the invasiveness, recovery time, or irreversibility of surgery.
For researchers following the broader GLP-1 therapy landscape and monitoring developments in peptides for fat loss and fat-burning peptide protocols, the next several years will bring even more options. The GLP-1 plotter and tracking tools from SeekPeptides will continue to evolve alongside these treatments, helping researchers make sense of an increasingly complex landscape.
What about combining cagrilintide with tirzepatide?
Some researchers have asked whether combining cagrilintide (the amylin component of CagriSema) with tirzepatide (the GIP/GLP-1 dual agonist) could produce even greater results. After all, this would theoretically activate three distinct receptor systems: amylin, GIP, and GLP-1.
The cagrilintide dosage with tirzepatide guide explores this concept in detail. While the theoretical rationale is sound, no clinical trial has tested this specific combination, and the safety profile of triple-pathway activation through separate compounds is unknown. The gastrointestinal side effect burden could be substantial, given that CagriSema already produces higher nausea rates than either component alone.
Interestingly, retatrutide achieves something similar by activating GLP-1, GIP, and glucagon receptors through a single molecule. The fact that retatrutide has shown weight loss potentially exceeding both CagriSema and tirzepatide suggests that more receptor targets may indeed produce better outcomes. But the optimal combination of targets, and the safest way to activate them, remains an active area of research.
Understanding long-term outcomes and weight regain
One critical question that neither CagriSema nor tirzepatide has fully answered is what happens after treatment stops. The neurological effects of both treatments depend on continued drug exposure. When treatment is discontinued, appetite returns, metabolic rate adjustments reverse, and weight regain begins.
Data from semaglutide discontinuation studies show that approximately two-thirds of lost weight is regained within a year of stopping treatment. Researchers studying semaglutide withdrawal patterns will recognize this as a significant limitation of all incretin-based therapies. The same pattern likely applies to both CagriSema and tirzepatide, though long-term discontinuation data for both is limited.
The CagriSema metabolic preservation mechanism, specifically the maintenance of mitochondrial function in skeletal muscle during weight loss, could theoretically slow the rate of weight regain by preventing some of the metabolic adaptation that makes regain almost inevitable. If participants on CagriSema maintain higher energy expenditure after stopping treatment compared to participants on tirzepatide, the weight regain trajectory could differ meaningfully. This is one of the most important questions future research needs to answer.
For researchers managing the transition off treatment, the troubleshooting slow response and tirzepatide plateau management guides provide strategies for maximizing results during active treatment, which can improve long-term outcomes by establishing better metabolic baselines before discontinuation.
For researchers serious about optimizing their peptide protocols and staying current with the rapidly evolving treatment landscape, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members access detailed comparison tools, protocol databases, and expert recommendations that go far beyond what any single article can provide.
Frequently asked questions
Is CagriSema stronger than tirzepatide for weight loss?
Based on cross-trial data, CagriSema and tirzepatide produce nearly identical weight loss results, approximately 22 to 23% in non-diabetic populations with obesity. CagriSema achieved 22.7% at 68 weeks while tirzepatide achieved 22.5% at 72 weeks. The REDEFINE 4 head-to-head trial will provide definitive comparison data. Until then, neither treatment shows a clear weight loss advantage over the other based on available evidence.
Can you take CagriSema and tirzepatide together?
No clinical trial has tested this combination, and it is not recommended. Combining CagriSema (which contains semaglutide) with tirzepatide (which activates GLP-1 receptors) would result in excessive GLP-1 pathway stimulation, likely producing severe gastrointestinal side effects. The cagrilintide with tirzepatide combination (without semaglutide) is a different consideration that some researchers explore, but it remains untested in clinical trials.
When will CagriSema be available?
Novo Nordisk filed a New Drug Application with the FDA for CagriSema in late December. The FDA typically takes 10 to 12 months to review new drug applications, suggesting a potential approval decision by late this year. Commercial availability would follow several months after approval, with widespread access likely beginning the following year. Tirzepatide is available now for researchers who do not want to wait.
Does CagriSema cause more side effects than tirzepatide?
CagriSema produces higher rates of nausea (55% versus 31%) and vomiting (26% versus 12%) compared to tirzepatide in their respective clinical trials. However, discontinuation rates are similar for both treatments (approximately 6%), suggesting that while CagriSema causes more frequent gastrointestinal symptoms, they are generally manageable. Most side effects are transient and resolve within the first weeks of treatment. Detailed information on managing side effects is available in the cagrilintide side effects guide.
Is CagriSema just semaglutide with cagrilintide added?
Yes, CagriSema is a fixed-dose combination of semaglutide 2.4 mg and cagrilintide 2.4 mg administered as a single once-weekly injection. The cagrilintide and semaglutide combination leverages two distinct satiety pathways (amylin and GLP-1) for enhanced appetite suppression and weight loss beyond what either compound achieves alone.
Which has better muscle preservation, CagriSema or tirzepatide?
Both treatments show approximately 67% fat loss and 33% lean tissue loss in body composition analyses. Preclinical CagriSema data suggests it may preserve energy expenditure by maintaining mitochondrial function in skeletal muscle, which could theoretically support better muscle preservation over time. However, human head-to-head body composition data is not yet available. Regardless of treatment choice, adequate protein intake and resistance training remain the most important factors for preserving muscle during weight loss.
How does CagriSema compare to retatrutide?
Retatrutide is a triple GLP-1/GIP/glucagon receptor agonist that showed up to 24.2% weight loss in phase 2 trials at 48 weeks, potentially exceeding both CagriSema and tirzepatide. However, retatrutide is earlier in clinical development and not yet available. The retatrutide comparison data and availability information provide more context on this emerging treatment.
Can I switch from tirzepatide to CagriSema?
When CagriSema becomes available, switching from tirzepatide will be possible but will require medical guidance. Since CagriSema contains semaglutide rather than tirzepatide, the transition involves switching GLP-1 agonists while adding an amylin component. The tirzepatide to semaglutide switching guide provides relevant context for the GLP-1 component transition. Clinical protocols for switching specifically to CagriSema will likely be established after the treatment receives approval and enters clinical practice.
External resources
REDEFINE 1 trial results published in the New England Journal of Medicine
REDEFINE 4 head-to-head trial registration on ClinicalTrials.gov
American Diabetes Association CagriSema clinical data summary
CagriSema energy expenditure preservation study in Nature Metabolism
In case I do not see you, good afternoon, good evening, and good night. May your protocols stay evidence-based, your side effects stay manageable, and your results stay beyond expectations.