Mar 21, 2026

Experienced researchers know something that beginners almost never figure out on their own. The peptide itself is rarely the problem. The source is. You can follow the perfect protocol, nail every dosing detail, and store your vials exactly right, but if the product you purchased was degraded, underdosed, or contaminated before it ever reached your door, none of that matters. You wasted time. You wasted money. And worse, you drew conclusions from bad data.
The GLP-1 peptide market has exploded. Dozens of vendors now compete for your attention, each promising 99% purity and fast shipping. Some deliver exactly what they claim. Others do not. The difference between a legitimate supplier and a questionable one often comes down to details that most buyers never think to check, things like lot-matched certificates of analysis, third-party HPLC verification, and proper cold chain shipping protocols.
This guide covers everything you need to know about buying GLP-1 peptides safely. From understanding the different types available and evaluating vendor quality, to reading lab reports, spotting red flags, and handling your purchase correctly after it arrives. Whether you are looking at semaglutide, tirzepatide, retatrutide, or the native GLP-1 peptide itself, the principles are the same. SeekPeptides has compiled the research, vendor data, and quality benchmarks so you do not have to learn these lessons the hard way.
What GLP-1 peptides are and why researchers study them
GLP-1 stands for glucagon-like peptide-1. It is a 30-amino-acid incretin hormone produced naturally in the intestinal L-cells after eating. The peptide binds to GLP-1 receptors throughout the body, triggering a cascade of metabolic effects that researchers have studied extensively for decades. Those effects include enhanced appetite suppression, improved insulin sensitivity, slowed gastric emptying, and potential neuroprotective properties that are still being characterized in clinical trials.
The native GLP-1 peptide has a half-life of roughly two minutes. It gets broken down almost immediately by the enzyme DPP-4. That limitation drove pharmaceutical development toward modified analogs that resist enzymatic degradation and last much longer in circulation.
Here is where things get interesting for researchers.
The GLP-1 receptor agonist class now includes several distinct compounds, each with unique pharmacological profiles. Semaglutide was among the first to achieve widespread clinical use, with modifications that extend its half-life to approximately seven days. Tirzepatide represents a dual-agonist approach, hitting both GLP-1 and GIP receptors simultaneously, which appears to produce stronger metabolic effects in clinical trials. And retatrutide takes things further still, acting as a triple agonist that targets GLP-1, GIP, and glucagon receptors all at once.
Each compound has different research applications. Semaglutide research often focuses on weight management timelines and cardiovascular outcomes. Tirzepatide studies frequently examine the comparative efficacy of dual versus single receptor activation. Retatrutide research explores whether triple agonism produces meaningfully different outcomes than dual or single approaches, with early data showing impressive dose-dependent responses in phase 2 trials.
Understanding what you are buying matters because these compounds are not interchangeable. The differences between semaglutide and tirzepatide extend beyond potency to mechanism of action, dosing schedules, side effect profiles, and storage requirements. Buying the wrong compound for your research question wastes resources. Buying a degraded or mislabeled product wastes everything.
The GLP-1 buying landscape explained
Not all GLP-1 peptides come from the same place, and the source determines almost everything about quality, legality, and reliability. The market breaks down into three distinct categories, and understanding the differences between them is not optional if you want consistent results.
Pharmaceutical-grade products
These are FDA-approved medications manufactured under strict GMP (Good Manufacturing Practice) conditions. Brand names like Ozempic (semaglutide), Wegovy (semaglutide), and Mounjaro (tirzepatide) fall into this category. They undergo rigorous quality control, batch testing, and regulatory oversight. The tradeoff is cost and accessibility. Understanding the relationship between GLP-1 and branded products helps researchers navigate which formulations serve different purposes.
For research applications, pharmaceutical-grade products serve as reference standards. They provide a benchmark against which other sources can be compared.
Compounded GLP-1 products
Compounding pharmacies create customized formulations of GLP-1 peptides, typically offering them at lower price points than brand-name equivalents. The quality varies enormously between pharmacies. Some operate under 503B registration with federal oversight and maintain excellent quality standards. Others cut corners on sterility testing, potency verification, and environmental controls.
The FDA has documented multiple recalls of compounded GLP-1 products. In one notable case, more than 15,000 vials of compounded semaglutide and tirzepatide were recalled due to sterility assurance failures. Testing revealed that some batches contained as little as 79.9% of the labeled potency for semaglutide and 86.8% for tirzepatide. These are not small discrepancies. A researcher working with a product at 80% of its stated concentration would draw fundamentally flawed conclusions about dose-response relationships.
When evaluating compounded sources, look for 503B-registered compounding pharmacies that provide batch-specific testing data. Pharmacies like Empower Pharmacy, Belmar Pharmacy, and others with established track records offer more reliability than unknown operations. The price of compounded semaglutide should raise questions if it falls significantly below market norms.
Research-grade peptide suppliers
This is the category most individual researchers encounter. Research peptide vendors sell lyophilized (freeze-dried) GLP-1 peptides intended for in vitro laboratory use. Quality in this space ranges from excellent to fraudulent, with little regulatory oversight to enforce standards. Understanding how to evaluate these suppliers is the single most important skill for any researcher purchasing GLP-1 peptides.
Research-grade peptides require reconstitution before use, come in lyophilized powder form, and must be stored correctly to maintain stability. The advantage is cost and accessibility. The disadvantage is that quality verification falls entirely on the buyer.

Vendor landscape for each major GLP-1 compound
The vendor landscape differs significantly depending on which GLP-1 compound you are purchasing. Some suppliers specialize in a single compound while others carry the full range. Understanding who sells what, and their reputation within the research community, helps narrow your options before you begin the detailed quality evaluation process.
Semaglutide vendor options
Semaglutide has the broadest vendor availability of any GLP-1 peptide. On the compounding pharmacy side, established names include Empower Pharmacy, which has been compounding semaglutide since the early shortage period and maintains 503B registration. Olympia Pharmacy is another large-scale compounder with established distribution networks. Smaller operations like Amino USA and BelleHealth serve specific market segments.
Research peptide suppliers including Peptide Sciences and others offer lyophilized semaglutide with batch-specific COAs. The research market is more fragmented, with quality varying significantly between suppliers. Community reviews from platforms like SeekPeptides provide the most reliable real-world quality data for these vendors.
One important distinction in the semaglutide market involves salt forms. Some compounding pharmacies use semaglutide sodium or semaglutide acetate rather than semaglutide base. These are chemically different compounds with different molecular weights. The FDA has raised concerns that salt forms have not been independently demonstrated to be safe or effective, as the clinical trials were conducted using semaglutide base. When purchasing compounded semaglutide, ask specifically which form the pharmacy uses and factor this into your evaluation.
Tirzepatide vendor options
The tirzepatide vendor landscape is slightly more consolidated than semaglutide. Major compounding pharmacies like Empower offer tirzepatide in multiple formulations, including standalone and blended versions with B12 or glycine. Specialty compounders like Red Rock Pharmacy, Boothwyn Pharmacy, and Southend Pharmacy have built reputations specifically in the tirzepatide space.
Newer entrants in the tirzepatide market include telehealth-integrated services like Brello Health, Trava, and Elevate Health. These platforms combine prescription services with compounding pharmacy partnerships, streamlining the acquisition process but adding another layer between the researcher and the actual manufacturing facility.
The grey market for tirzepatide also exists and warrants understanding. These are vendors operating in legal gray areas, often importing from overseas manufacturers or reselling without proper licensing. The savings can be substantial, but the quality risks multiply significantly.
Retatrutide vendor options
Retatrutide has the most limited vendor landscape because it remains in clinical development. No compounding pharmacies offer retatrutide because there is no FDA-approved reference drug to compound against. This means all retatrutide purchasing happens through research peptide suppliers.
The best sources for retatrutide tend to be established research peptide companies that have invested in synthesizing this more complex compound. Purchasing retatrutide online requires extra diligence because the limited supplier pool means fewer comparison points for quality benchmarking. Some researchers source lower-cost retatrutide from newer suppliers, but the quality verification burden increases proportionally with the price discount.
How to evaluate peptide vendor quality before you buy
This is where most buyers fail. They compare prices, read a few reviews, and place an order. That approach works for buying headphones. It does not work for buying research peptides where the difference between a legitimate and illegitimate product is invisible to the naked eye.
A systematic evaluation process protects your research investment and ensures reproducible results. Here is exactly what to check.
Certificate of analysis verification
The COA is the single most important document a peptide vendor provides. It should contain HPLC (High-Performance Liquid Chromatography) purity data showing the percentage of the target compound versus impurities, mass spectrometry data confirming the molecular identity matches the expected molecular weight, a batch or lot number that matches the label on your specific vial, and the date of testing.
A legitimate COA shows a clean HPLC chromatogram with one dominant peak representing 95-99% or more of the total area. Minor secondary peaks indicate trace impurities, which is normal at small percentages. The mass spectrometry data should confirm the molecular weight within plus or minus 1 Dalton of the theoretical value. For semaglutide, that theoretical molecular weight is approximately 4113.58 Da. For tirzepatide, it is approximately 4813.45 Da.
Here is the critical distinction most buyers miss. HPLC purity and net peptide content are different measurements. A peptide can test at 99% HPLC purity, meaning 99% of the peptide-related material in the sample is the target compound, while the net peptide content is only 75-85%. The remainder consists of water, counterions like TFA or acetate, and salts. This does not indicate a quality problem, but it does affect dosing calculations that researchers need to account for.
Third-party testing requirements
In-house testing means the vendor tested their own product. Third-party testing means an independent laboratory verified the results. The difference matters enormously. A vendor who only provides in-house COAs is essentially grading their own homework. Independent verification from an accredited lab, ideally one with ISO/IEC 17025 certification, carries significantly more weight.
Ask specifically whether testing was performed by an independent laboratory. Request the name of the testing facility. Look for vendors who proactively share this information rather than requiring you to dig for it. The most reputable suppliers make third-party COAs easily accessible on their product pages.
Business legitimacy checks
Before entering payment information on any peptide website, run a basic legitimacy check. Does the domain look professional, or is it misspelled, oddly hyphenated, or recently registered? Is there a real business address that resolves to an actual location? Is there a phone number? Does the checkout use HTTPS with a valid security certificate?
Look for state business registration. Check if the company has an established social media presence. Search for their name on Reddit, Trustpilot, and peptide research forums. SeekPeptides members often share vendor experiences and quality reports within the community, which provides real-world validation beyond what marketing claims can offer.
A vendor that has been operating for less than six months with no verifiable reviews should be approached with extreme caution regardless of their pricing or marketing claims.
Communication and support quality
Legitimate suppliers can answer basic technical questions because they have actual processes behind their products. Try asking a prospective vendor about their synthesis method, storage recommendations, or how to interpret their COA. The quality and speed of their response tells you a lot about the operation behind the website.
Vendors who cannot or will not answer technical questions, who respond only with marketing copy, or who take days to reply to straightforward inquiries are often not worth the risk, regardless of their pricing.
Reading a certificate of analysis like a researcher
Most buyers glance at the purity number on a COA and move on. That approach misses most of the information the document contains. A proper COA review takes five minutes and can save thousands of dollars in wasted research.
The HPLC chromatogram
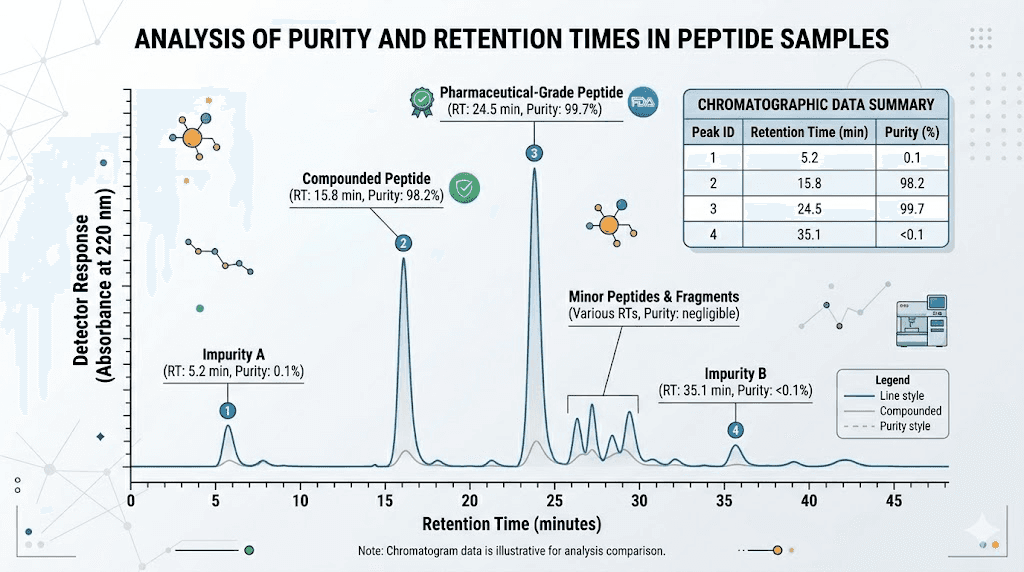
The chromatogram is a graph showing peaks at different retention times. Each peak represents a different compound detected in the sample. The main peak should be tall and dominant, with a clean baseline on either side. The area under this peak, expressed as a percentage of total peak area, gives you the HPLC purity value.
What to look for. A single dominant peak at greater than 95% area is acceptable. Greater than 98% is excellent. Multiple significant peaks suggest impurities or degradation products. An unusually broad main peak may indicate poor separation or a mixture of closely related compounds. If the vendor provides only a purity number without the actual chromatogram image, that is a red flag. Numbers can be fabricated easily. Chromatograms are harder to fake convincingly.
The retention time of the main peak should be consistent with the expected compound. While exact retention times vary by method and column, they should fall within a reasonable range for peptides of similar molecular weight. Researchers familiar with peptide dosing specifics understand that accurate purity data directly affects protocol reliability.
Mass spectrometry identity confirmation
While HPLC tells you how pure the sample is, mass spectrometry tells you what it actually is. The MS report should show a molecular ion peak matching the theoretical molecular weight of the target peptide. For larger peptides, you may also see multiply charged ions. A doubly charged ion appears at roughly half the molecular weight, and a triply charged ion at roughly one-third. These are normal and expected.
The key check is whether the observed molecular weight matches the expected value. A difference of more than 1-2 Daltons raises questions about whether the sample contains the correct compound. If the COA does not include mass spectrometry data at all, the identity of the peptide cannot be confirmed, only its purity relative to whatever is in the vial.
Batch number matching
The lot or batch number on the COA must match the number on your vial label. If they do not match, the COA does not apply to your product. This sounds obvious, but some vendors use a single COA for multiple batches, which defeats the purpose of batch-specific testing. Every batch of peptide should have its own COA because quality can vary between synthesis runs.
Also check the testing date. A COA from 18 months ago applied to a product you purchased today should prompt questions about storage conditions and potential degradation during that interval.
Red flags that signal a bad peptide vendor
Recognizing warning signs before you place an order is far cheaper than discovering quality problems after your research is compromised. These red flags have been documented across hundreds of vendor evaluations.
Pricing that defies manufacturing costs
Peptide synthesis has real costs. Raw materials, synthesis equipment, purification, lyophilization, quality testing, and proper storage all add up. When a vendor offers GLP-1 peptides at prices significantly below established market rates, something is being cut. It might be purity. It might be potency. It might be that the vial contains something other than what the label claims.
This does not mean the most expensive option is automatically the best. But if everyone else charges $80-150 for a product and one vendor charges $25, ask yourself what they are skipping to make those numbers work. Research into retatrutide pricing and compounded tirzepatide costs shows that legitimate suppliers operate within predictable price ranges. Outliers below those ranges warrant scrutiny.
Missing or generic documentation
Vendors who use phrases like "research grade" and "99% purity" without providing supporting chromatograms, identity data, or lot-matched documentation are making claims they cannot or will not substantiate. A COA that looks generic, lacks specific batch numbers, or uses identical formatting and data across multiple products is likely not from genuine batch testing.
Some vendors provide what appear to be COAs but are actually templates with numbers filled in. The tell is often in the details. Real COAs from real laboratories include method parameters, instrument identification, analyst names or initials, and specific testing conditions. Generic documents lack these elements.
Aggressive urgency marketing
Countdown timers. "Last chance" messaging. Flash sale claims of 50% off for the next hour. The peptide market does not operate on these timelines. Manufacturing and testing cycles make impulse-sale pricing logistically impossible for legitimate operations. These tactics target impulsive buyers and are strongly associated with low-quality or fraudulent vendors.
Limited payment protections
Payment methods that cannot be reversed or disputed, such as wire transfers, gift cards, or cryptocurrency-only options, reduce your recourse if the product never arrives or fails to meet specifications. Legitimate vendors typically accept credit cards and sometimes PayPal, which provide buyer protection mechanisms.
This does not mean every vendor accepting cryptocurrency is fraudulent. But if cryptocurrency is the only payment option, combined with other red flags on this list, proceed with extreme caution.
No verifiable business presence
No physical address. No phone number. No state business registration. Only anonymous forum posts or affiliate marketing as their public presence. These are characteristics of operations that can disappear overnight without accountability.
Cross-reference the business name with state business registries, the BBB, and domain registration records. A company that has been registered for years at a verifiable address carries more credibility than one registered last month at a P.O. box. Researchers using platforms like SeekPeptides benefit from community-verified vendor information that individual buyers would need to compile on their own.
Specific GLP-1 peptides available for purchase
The GLP-1 category encompasses several distinct compounds, each with unique characteristics that affect purchasing decisions. Knowing what you need before you start shopping prevents costly mistakes.
Semaglutide
The most widely available GLP-1 analog in the research market. Semaglutide features a C-18 fatty acid chain that enables binding to albumin, extending its half-life to approximately 7 days. It comes in both injectable and oral formulations, though the oral form requires specific absorption-enhancing excipients.
When buying semaglutide, verify the molecular weight (approximately 4113.58 Da) on the COA. Common vial sizes for research include 3mg, 5mg, and 10mg of lyophilized powder. Reconstitution with bacteriostatic water is straightforward but requires attention to concentration calculations. The semaglutide dosage calculator helps researchers determine precise measurements after reconstitution.
Semaglutide is also available in compounded forms with additives like vitamin B12, glycine, or niacinamide. These blends serve specific research purposes but add complexity to quality verification because each additional compound needs its own identity and purity confirmation.
Storage is critical. Reconstituted semaglutide should be refrigerated at 2-8 degrees Celsius. The shelf life of compounded semaglutide in the fridge varies by formulation but typically ranges from 28-56 days after reconstitution. Temperature excursions during shipping can compromise potency before you even open the package.
Tirzepatide
A dual GIP/GLP-1 receptor agonist that has shown remarkable efficacy in clinical trials. Tirzepatide is a 39-amino-acid synthetic peptide with a molecular weight of approximately 4813.45 Da. Its dual mechanism of action makes it particularly interesting for researchers studying metabolic pathways beyond what single-receptor agonists can illuminate.
Tirzepatide is available from research suppliers in lyophilized form, typically in 5mg, 10mg, and 30mg vials. Reconstitution requires careful bacteriostatic water calculations to achieve the desired concentration. The tirzepatide dosage calculator simplifies this process for researchers working with different vial sizes.
Compounded tirzepatide is also widely available, often blended with glycine and B12, niacinamide, or levocarnitine. Vendor reviews for compounded tirzepatide from suppliers like Empower, Simple Peptide, and IVIM Health provide real-world quality data that supplements COA verification.
Tirzepatide requires refrigeration after reconstitution. Unreconstituted lyophilized powder is more stable but should still be stored in cool, dry conditions away from direct light. Knowing how long tirzepatide lasts in the fridge prevents waste from expired product.
Retatrutide
The newest entrant in the GLP-1 agonist space, retatrutide is a triple receptor agonist targeting GLP-1, GIP, and glucagon receptors. Phase 2 trial data showed dose-dependent weight reduction of up to 24% at 48 weeks, which exceeded results from both semaglutide and tirzepatide monotherapy at comparable timepoints.
Retatrutide is primarily available through research peptide suppliers. It is not yet FDA-approved, which means pharmaceutical-grade options do not exist. This makes vendor quality evaluation even more critical for this compound. The current availability of retatrutide is more limited than semaglutide or tirzepatide, and pricing reflects its relative novelty and manufacturing complexity.
Common vial sizes include 5mg, 10mg, and 20mg. Reconstitution follows standard protocols using bacteriostatic water at calculated ratios. The dosing schedule typically involves weekly administration, similar to other long-acting GLP-1 agonists.
Some researchers source retatrutide in bulk quantities for extended research projects. Bulk purchasing introduces additional quality considerations around storage stability over longer periods and the need for representative sampling of larger batches.
Native GLP-1 and other analogs
The native GLP-1(7-36) amide peptide is available from biochemical suppliers and serves primarily as a reference standard or for in vitro receptor binding studies. Its extremely short half-life makes it impractical for most in vivo research, but it has applications in cell-based assays and receptor characterization studies.
Other GLP-1-related compounds entering the market include survodutide, CagriSema, and mazdutide. Availability of these newer compounds through research suppliers is still limited, but expanding as clinical trial data generates researcher interest. Understanding how these compounds differ from established GLP-1 agonists helps researchers select the right molecule for their specific question.

Cost comparison and getting real value for your money
Price matters. But price without quality context is meaningless. A $30 vial that contains 60% of its labeled potency is more expensive than an $80 vial at full potency because you need more product to achieve the same research outcomes.
What drives peptide pricing
Several factors determine the cost of GLP-1 peptides. Synthesis complexity is the largest factor. Longer peptides with complex modifications cost more to synthesize. Purification to high purity levels (greater than 98%) adds cost because it reduces yield. Third-party testing adds cost. Proper lyophilization, packaging, and cold-chain shipping add cost. Each of these steps contributes to product quality, and skipping any of them enables the low prices that should make buyers suspicious.
Semaglutide is generally the most affordable GLP-1 peptide from research suppliers because synthesis methods are well-established and multiple manufacturers compete on price. Compounded semaglutide pricing varies widely by pharmacy but typically ranges from $100-400 per month depending on dosage and formulation.
Tirzepatide pricing sits higher due to its longer amino acid sequence and dual-receptor modification. Tirzepatide costs from compounding pharmacies typically range from $200-600 per month, while research-grade vials vary by supplier and quantity.
Retatrutide commands the highest prices due to limited manufacturing competition and the compound novelty. The full retatrutide pricing landscape shows significant variation between suppliers. Use the peptide cost calculator to compare effective per-milligram costs across different vendors and vial sizes.
Bulk purchasing considerations
Buying in larger quantities typically reduces per-unit cost. However, bulk purchasing only makes sense if you can store the product correctly for the duration of your research timeline. Lyophilized peptides stored at minus 20 degrees Celsius remain stable for months to years. Reconstituted peptides degrade much faster, even under refrigeration.
Some researchers purchase bulk tirzepatide powder or bulk retatrutide for extended studies. The economics work if storage conditions are maintained and the product is used within its stability window. They do not work if improper storage causes degradation before the product is used.
The true cost of bad quality
Factor in these hidden costs when evaluating the cheapest option. Failed experiments that need to be repeated. Wasted consumables like syringes, bacteriostatic water, and disposables. Time lost to troubleshooting what turned out to be a product quality issue rather than a protocol problem. And the opportunity cost of not advancing your research during that wasted time.
One researcher on a peptide forum documented spending $450 on cheap peptides over three months before discovering the products were consistently underdosed. The replacement cost from a verified vendor was $600 for the same period, but produced reliable, reproducible results from the first vial. The "savings" from the cheap vendor actually cost an additional $450 and three months of unusable data.
There is also a compounding effect that many buyers overlook. When you start with a bad product, every observation you make is tainted. You might conclude that a particular compound does not work for your research application, when in reality the compound never reached effective concentration due to degradation or underdosing. You might adjust your protocol based on flawed results, moving further from optimal conditions rather than closer. And if you share your findings with other researchers, you propagate incorrect conclusions through the community. The ripple effects of one bad purchasing decision extend far beyond the initial wasted dollars.
Strategies for reducing costs without compromising quality
Saving money on peptide purchases is entirely possible without sacrificing quality. The key is knowing where legitimate savings exist. Volume discounts from established vendors provide real savings because manufacturing efficiencies genuinely reduce per-unit costs at scale. Loyalty programs and returning customer discounts are common among quality vendors. Seasonal promotions from legitimate vendors occasionally offer genuine discounts tied to inventory management rather than desperation pricing.
Comparing the per-milligram cost across vendors using the same quality benchmarks is the most direct way to find the best value. A vendor charging $100 for a 5mg vial at verified 98% purity is cheaper per milligram of active compound than a vendor charging $80 for a 5mg vial at 85% purity. The math is straightforward once you have reliable purity data from COAs.
Another legitimate savings strategy is purchasing lyophilized powder and reconstituting yourself rather than buying pre-constituted solutions. Pre-constituted products from compounding pharmacies include the pharmacy markup and the cost of the solution preparation. Lyophilized powder from research suppliers, reconstituted using the reconstitution calculator for precise measurements, typically costs significantly less per milligram while giving you more control over concentration and freshness.
Storage and handling after your purchase arrives
You found a quality vendor. You verified the COA. The package arrived with proper cold-chain shipping. Now what? How you handle the product from this point forward determines whether it maintains the quality you paid for.
Receiving and inspecting your order
When the package arrives, check the external temperature indicators if included. Many reputable vendors ship with cold packs or temperature monitoring strips. If the product arrives warm, that is a concern. Semaglutide that arrives hot may have experienced temperature excursions that compromised stability.
Inspect vials for cracks, broken seals, or discoloration. Lyophilized peptides should appear as a white to off-white powder or cake. Yellow, brown, or liquid contents in vials that should contain lyophilized powder indicate degradation or incorrect handling.
Verify the lot number on each vial against the COA provided. Document the condition of the shipment with photographs in case you need to file a quality complaint later.
Proper storage of lyophilized peptides
Unreconstituted, lyophilized GLP-1 peptides should be stored at minus 20 degrees Celsius for long-term stability. A dedicated laboratory freezer is ideal. If that is not available, the freezer compartment of a standard refrigerator works for shorter storage periods of a few months, though temperature cycling from the defrost mechanism is not ideal.
Keep vials sealed until ready for reconstitution. Moisture is the enemy of lyophilized peptides. Each exposure to air introduces humidity that can initiate degradation. Store vials in a desiccated container if possible. Protect from light, as UV exposure can degrade peptide bonds.
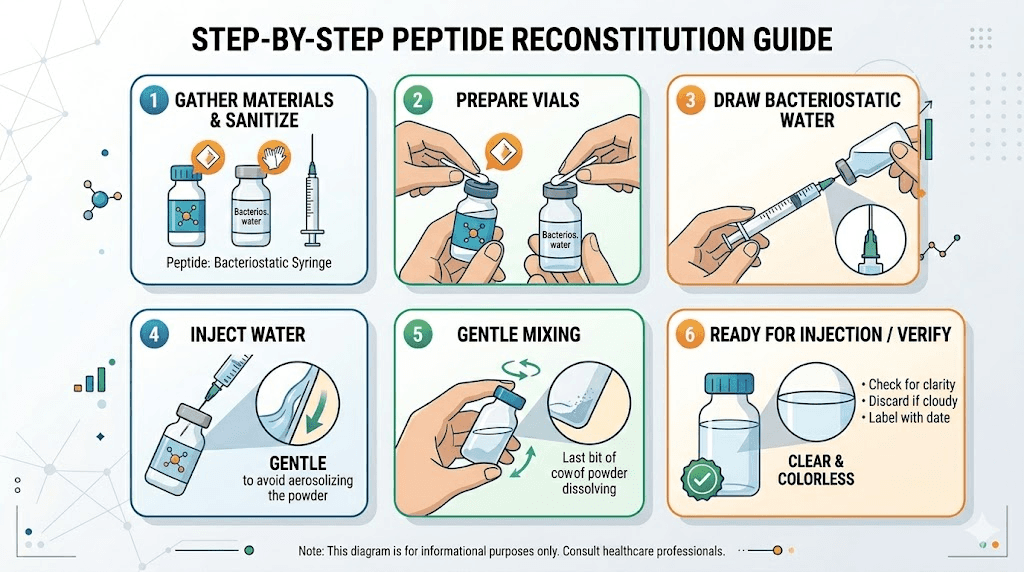
Reconstitution best practices
When ready to use, reconstitute with bacteriostatic water or sterile water. The choice depends on whether you need preservative protection for multi-use vials (bacteriostatic water contains 0.9% benzyl alcohol as a preservative) or single-use preparations (sterile water).
Add the water slowly, directed against the vial wall rather than directly onto the lyophilized cake. Swirl gently. Never shake. Aggressive agitation can damage peptide structure. Wait until the powder is fully dissolved before use. Most GLP-1 peptides dissolve within 1-5 minutes of gentle swirling.
The volume of water determines the concentration of the reconstituted solution. Detailed guides for reconstituting 5mg semaglutide, reconstituting tirzepatide, and calculating water volumes for retatrutide provide step-by-step instructions. The peptide reconstitution calculator automates the math.
Post-reconstitution storage
Once reconstituted, refrigerate at 2-8 degrees Celsius. Do not freeze reconstituted peptide solutions, as ice crystal formation can damage peptide structure. Freezing tirzepatide after reconstitution is a common mistake that can reduce potency.
The clock starts ticking once water is added. Semaglutide shelf life after reconstitution is typically 28-56 days with bacteriostatic water. Tirzepatide expiration follows similar timelines. With sterile water (no preservative), use within 24-48 hours or discard. Understanding expiration prevents researchers from using degraded product that yields unreliable results.
If the solution becomes cloudy, changes color, or develops visible particles after reconstitution, discard it. These changes indicate degradation or contamination.
Shipping considerations that affect quality
How a peptide gets from the vendor to your location matters almost as much as how it was manufactured. Temperature-sensitive compounds like GLP-1 agonists can degrade during transit if shipping protocols are inadequate.
Cold chain shipping requirements
Lyophilized peptides are more stable during shipping than reconstituted solutions, but they are not immune to heat damage. Extended exposure to temperatures above 40 degrees Celsius can accelerate degradation even in powder form. The best vendors ship with insulated packaging and cold gel packs, particularly during summer months when ambient temperatures during transit can exceed safe thresholds.
Reconstituted or pre-constituted solutions from compounding pharmacies are much more temperature-sensitive. The FDA has received complaints about compounded GLP-1 products arriving warm, with inadequate ice packs or insulation. If your semaglutide arrives hot, the potency may already be compromised. The safe practice is to not use any injectable peptide that arrives at a temperature obviously above refrigeration range.
Some vendors include temperature indicator strips in their packaging. These irreversible color-change indicators show whether the package exceeded a threshold temperature during transit. If your vendor does not include these, it is worth asking why. The cost is minimal, typically less than a dollar per shipment, and the information they provide is invaluable.
Shipping speed and timing
Faster shipping means less time for temperature exposure. Many quality vendors offer expedited shipping options specifically for temperature-sensitive products. If you are ordering during summer or to a warm climate, upgrading to two-day or overnight shipping is often worth the additional cost. A $20 shipping upgrade is cheap insurance compared to a $150 vial of degraded peptide.
Consider the timing of your order as well. Placing an order late on a Friday means the package may sit in a warehouse or delivery truck over the weekend. Ordering early in the week gives the package less idle time during transit.
International shipping complexities
International orders introduce additional variables. Customs holds can add days to transit time, during which temperature control is nonexistent. Customs documentation requirements vary by country, and packages flagged for inspection may be opened in environments that are not temperature-controlled.
Some countries restrict or prohibit the importation of certain peptides. Before placing an international order, verify that the specific compound you are purchasing can be legally imported to your jurisdiction. Failed shipments due to customs seizure represent a total loss in most cases.
Traveling with purchased peptides
Researchers who need to transport GLP-1 peptides between locations face additional handling challenges. Traveling with semaglutide and traveling with tirzepatide requires portable cooling solutions like insulated travel cases with cold packs. For air travel, carry peptides in your carry-on luggage rather than checked bags, where temperature extremes in the cargo hold can be severe. Keep documentation including prescriptions or COAs readily accessible in case of questions from security or customs personnel.
Common mistakes buyers make and how to avoid them
After analyzing hundreds of buyer experiences across research forums, certain mistakes appear repeatedly. Learning from others prevents repeating their expensive lessons.
Buying based solely on price
The most common mistake. Period. A researcher sees three vendors offering semaglutide at $120, $95, and $40, and orders from the $40 vendor without further investigation. Three weeks later, minimal results prompt questions. Was the protocol wrong? Was the dose insufficient? Usually, the answer is simpler. The product was underdosed or degraded.
Price should be one factor in your decision, not the only factor. A legitimate vendor charging $120 with third-party COAs, proper cold-chain shipping, and an established track record provides more value per dollar than a mystery vendor at $40 with no verifiable quality data.
Not checking the COA before using the product
Many buyers never look at the COA. They assume the vendor is honest and start using the product immediately. By the time they suspect a quality issue, they have already consumed most of the vial and cannot return it or have it independently tested. Check the COA before your first use. Verify the lot number, review the chromatogram, confirm the mass spec identity. Five minutes of verification can save weeks of compromised research.
Improper storage after receiving the order
The package arrives. It sits on the kitchen counter for a few hours. Maybe overnight. Then it goes into the fridge. That delay may not destroy the product, but it starts a degradation process that shortens the usable shelf life. Leaving semaglutide out overnight is a concern that many buyers only think about after the fact. Develop a habit of refrigerating or freezing your peptides immediately upon receipt.
Reconstituting too much at once
Some researchers reconstitute their entire supply at once for convenience. This is a mistake because reconstituted peptides degrade faster than lyophilized powder. Only reconstitute what you will use within the stability window, typically 28-56 days for bacteriostatic water preparations. If you have multiple vials, keep the extras in lyophilized form at minus 20 degrees Celsius until needed.
Ignoring expiration dates
Both lyophilized and reconstituted peptides have finite shelf lives. Using product past its expiration date introduces uncertainty about potency. While slightly expired lyophilized peptide stored at minus 20 degrees Celsius may retain most of its activity, the further past expiration you go, the less reliable your data becomes. Using expired semaglutide is a calculated risk that most serious researchers avoid.
Failing to document shipment condition
Take photographs of your package when it arrives. Note the condition of the cold packs, whether they are still cold or room temperature. Document any damage to vials or packaging. This documentation is essential if you need to file a quality complaint and is useful for your own records when evaluating vendor consistency over time.

Legal considerations and regulatory framework
The legal landscape around purchasing GLP-1 peptides is complex and varies by jurisdiction. Understanding it protects researchers from inadvertent legal exposure.
Research use classification
Research peptide vendors sell products labeled "for research purposes only" or "not for human consumption." This classification places them outside FDA drug regulation in most cases, but it also means these products have not undergone FDA review for safety, efficacy, or quality. The FDA has taken enforcement action against companies that market research peptides in ways that suggest human use, particularly in the GLP-1 space where consumer demand is extremely high.
The distinction between research and pharmaceutical-grade peptides is not just regulatory. It reflects fundamentally different manufacturing standards, quality oversight, and intended applications.
Compounding pharmacy regulations
Compounding pharmacies operate under different rules than research peptide suppliers. Section 503A pharmacies compound individual prescriptions, while Section 503B outsourcing facilities can produce larger batches under stricter oversight. The FDA has increasingly focused enforcement on compounding pharmacies producing GLP-1 agonists, particularly following the resolution of drug shortages that previously provided regulatory flexibility.
In December 2025, the WHO issued global guidelines on GLP-1 medicines for obesity treatment, further highlighting the regulatory attention these compounds receive. Researchers should stay current on regulatory developments that may affect availability and purchasing options.
Import considerations
Purchasing peptides from international suppliers introduces additional regulatory complexity. Customs regulations vary by country, and importation of research chemicals may require specific documentation or permits depending on jurisdiction. International purchasing options exist but carry additional logistical and legal considerations.
Injectable vs oral GLP-1 forms and what to buy
The GLP-1 market now includes both injectable and oral delivery forms. Understanding the differences affects purchasing decisions.
Injectable forms
The majority of GLP-1 research peptides are sold in injectable form, either as lyophilized powder for reconstitution or as pre-constituted solutions from compounding pharmacies. Injectable administration bypasses the gastrointestinal tract, providing more predictable bioavailability and avoiding the degradation that peptides experience in the acidic stomach environment.
For researchers new to injectable peptides, understanding how to inject GLP-1, where to inject, and optimal injection sites provides essential context. Subcutaneous injection using insulin syringes is the standard administration method.
The injectable vs oral peptides comparison highlights the practical differences between delivery methods. Injectable forms generally offer superior bioavailability, more precise dosing, and lower per-dose costs compared to oral alternatives.
Oral forms
Oral GLP-1 formulations have gained significant interest. Oral tirzepatide and oral semaglutide drops represent newer delivery approaches. These include sublingual formulations, orally dissolving tablets (tirzepatide ODT), troches (semaglutide troches), and sublingual semaglutide.
Oral forms appeal to researchers studying non-invasive delivery methods or comparing bioavailability across administration routes. The oral versus injection comparison for tirzepatide and tablets versus injections analysis provide data-driven comparisons.
Liquid oral formulations like GLP-1 six-in-one oral liquid and GLP-1 drops represent newer approaches that are still being characterized in research settings. Their convenience is appealing, but bioavailability data is less established than for injectable forms.
New delivery technologies like GLP-1 patches are also entering development, offering potential alternatives to both injection and oral routes.
Building a reliable vendor relationship
Once you identify a quality vendor, maintaining that relationship provides ongoing benefits that go beyond the first transaction.
Start with a test order
Before committing to a large purchase, place a small test order. Evaluate shipping speed, packaging quality, temperature control, COA accuracy, and customer service responsiveness. If possible, have the product independently tested to verify the vendor claims against an unbiased analysis. This small investment can save significant resources on future larger orders.
Document everything
Keep records of every order, including order confirmations, tracking information, photos of received packages, COAs, and any communications with the vendor. This documentation serves as both a quality log for your research and evidence if quality disputes arise.
Ask the right questions
Before placing your first order with any vendor, send them a short list of questions. How they respond tells you almost as much as their COA. Ask where their peptides are synthesized. Ask whether they use third-party testing and which laboratory performs it. Ask about their storage conditions between synthesis and shipment. Ask about their return or replacement policy for products that arrive damaged or fail to meet specifications.
Vendors who answer these questions quickly, specifically, and transparently are demonstrating operational confidence. Vendors who deflect, provide vague responses, or take days to respond to straightforward technical questions are signaling that their operation may not withstand scrutiny. This is free intelligence that costs nothing but a few minutes of your time.
Monitor consistency over time
A vendor who delivers excellent quality on the first order may not maintain that standard indefinitely. Manufacturing changes, supplier switches, or business pressures can affect product quality over time. Periodically verify quality through COA review and, when practical, independent testing. Compare results across batches to detect any drift in potency or purity.
Community resources help with ongoing monitoring. SeekPeptides provides updated vendor reviews, quality reports, and community feedback that helps researchers stay informed about changes in vendor quality over time. Members share batch-specific experiences that supplement individual COA reviews with real-world performance data.
Have a backup vendor
Supply disruptions happen. Vendors go out of business, experience manufacturing problems, or face regulatory actions. Having a pre-vetted backup vendor ensures your research continuity is not dependent on a single source. Apply the same quality evaluation criteria to your backup that you used for your primary vendor.

Understanding peptide degradation and why it matters for purchasing decisions
Peptide degradation is not a binary event. It is a gradual process that reduces potency over time. Understanding the mechanisms of degradation helps you make better purchasing decisions and recognize quality problems before they compromise your research.
Chemical degradation pathways
The primary chemical degradation pathways for GLP-1 peptides include deamidation, oxidation, and hydrolysis. Deamidation occurs when asparagine or glutamine residues lose their amide group, altering the peptide structure and potentially reducing receptor binding affinity. Oxidation affects methionine and tryptophan residues, and can be accelerated by light exposure, temperature, and dissolved oxygen in reconstituted solutions. Hydrolysis involves the cleavage of peptide bonds, breaking the molecule into smaller, inactive fragments.
Each of these pathways proceeds faster at higher temperatures, which is why cold storage is essential. The Arrhenius equation, a fundamental principle of chemical kinetics, tells us that reaction rates roughly double for every 10 degree Celsius increase in temperature. A peptide stored at 25 degrees Celsius degrades approximately 4-8 times faster than one stored at 4 degrees Celsius. This is not theoretical. It has direct implications for shipping conditions, storage practices, and the decision about when to reconstitute your supply.
Physical degradation and aggregation
Beyond chemical degradation, peptides can undergo physical changes that affect their activity. Aggregation, where peptide molecules clump together into larger structures, reduces the concentration of active monomeric peptide in solution. Aggregation can be triggered by temperature fluctuations, agitation (which is why you should never shake reconstituted peptides), and certain buffer conditions.
Visual signs of aggregation include cloudiness, visible particles, or gel formation in solutions that should be clear. If your reconstituted semaglutide or tirzepatide develops any visible change from its initial clear appearance, discard it. Using aggregated peptide can produce unpredictable results and, in some contexts, safety concerns.
What this means for buying decisions
Degradation awareness should influence your purchasing decisions in several specific ways. First, buy from vendors who demonstrate proper storage and shipping protocols, not just proper manufacturing. A peptide that was synthesized perfectly but stored at room temperature for months has degraded significantly before it reaches you. Second, prefer lyophilized (freeze-dried) formulations over pre-constituted solutions when possible, because lyophilized peptides are dramatically more stable during storage and transit. Third, consider the vendor location relative to yours. A local vendor in the same climate zone requires less aggressive cold-chain shipping than one halfway across the country.
When evaluating a new vendor, the condition of the first shipment tells you a great deal about their operational standards. Professional packaging with appropriate insulation, cold packs, and temperature monitoring suggests a vendor who understands peptide stability. A vial tossed in a padded envelope with no temperature protection suggests one who does not.
What to do after buying your GLP-1 peptide
Purchasing is only the first step. What you do with the product after it arrives determines whether your research produces meaningful results.
Establish your protocol
Before reconstituting, have your entire research protocol documented. This includes dosing calculations, administration schedule, outcome measurements, and timeline. The semaglutide dosage chart, tirzepatide dosage chart, and retatrutide dose schedule provide reference frameworks for protocol design.
For researchers comparing GLP-1 compounds, the tirzepatide versus semaglutide dosage chart and conversion chart help calibrate equivalent doses across different compounds.
Understand the timeline
GLP-1 agonists do not produce instant results. How long GLP-1 takes to work depends on the specific compound, dose, and individual factors. Semaglutide typically reaches steady-state concentrations after 4-5 weeks of weekly dosing. Tirzepatide follows a similar timeline. Setting appropriate expectations for when observable effects emerge prevents premature protocol changes based on incomplete data.
Early responses and first-week semaglutide experiences provide useful reference points for what to expect during the initial phase of research protocols. Researchers tracking week-by-week results and weight loss timelines can compare their observations against documented patterns.
Monitor for quality issues during use
Even with verified product, monitor for signs of degradation during use. Changes in solution clarity, color, or consistency after reconstitution suggest quality problems. Unexpected results, whether absence of expected effects or unusual side effects, should prompt consideration of product quality as a variable rather than immediately attributing the outcome to the protocol itself.
Common side effects of GLP-1 peptides are well-documented. Side effect comparisons between compounds help researchers distinguish expected pharmacological effects from unexpected responses that might indicate product quality issues. Known effects include fatigue, gastrointestinal effects, and headaches, all of which typically resolve with continued administration or dose adjustment.
Plan for the long term
Sustained research protocols require planning beyond the first purchase. Consider how long to continue GLP-1 protocols, maintenance dosing strategies, and what happens when research protocols conclude. The withdrawal and discontinuation considerations for GLP-1 agonists are an active area of research that informed investigators should understand.
Some researchers explore alternative compounds or switching between GLP-1 agonists as their research questions evolve. Understanding the transition dynamics between compounds helps design effective crossover protocols.
Supplementary compounds and combination purchases
Researchers often purchase GLP-1 peptides alongside complementary compounds. Understanding these combinations helps optimize your purchasing strategy and ensures you source everything you need from quality suppliers rather than scrambling for add-ons from unknown sources.
Supportive supplements
The most common supplementary purchases alongside GLP-1 peptides include supplements that support GLP-1 protocols. Probiotics rank high on this list, with research suggesting that gut microbiome composition influences GLP-1 receptor agonist effectiveness. GLP-1 specific probiotic formulations and general-purpose probiotics for semaglutide users address this connection. Fiber supplements help manage the gastrointestinal effects that are common with GLP-1 agonists, and creatine supports lean mass preservation during body composition changes.
Other frequently co-purchased items include electrolyte supplements, digestive enzymes, and tirzepatide-specific supplement protocols designed to mitigate common side effects like sulfur burps and bloating.
Combination research compounds
Some researchers investigate GLP-1 peptides alongside other research compounds, and this affects purchasing planning in important ways. Common combination protocols include phentermine with semaglutide, phentermine with tirzepatide, berberine pairings, berberine with tirzepatide, metformin co-administration, and even NAD+ with tirzepatide. Understanding potential interactions is essential before combining compounds. The peptide stacking calculator helps researchers plan multi-compound protocols.
When purchasing combination compounds, resist the temptation to source everything from the cheapest available vendor for each individual product. Using a single verified vendor for multiple compounds simplifies quality assurance, reduces shipping costs, and provides a single point of accountability if quality issues arise. However, if your primary vendor does not carry a specific compound you need, it is better to use a second verified vendor than to compromise on quality for convenience.
Nutritional support during GLP-1 research is another common consideration. Resources on optimal nutrition during semaglutide use, foods to avoid on tirzepatide, and meal planning for GLP-1 users help researchers account for nutritional variables in their protocols.
Using tools to optimize your purchase and protocol
Calculators and tracking tools eliminate guesswork from the buying and dosing process. SeekPeptides offers several free tools that researchers consistently find valuable.
The peptide calculator handles basic concentration and dosing calculations for any peptide. The compound-specific calculators for semaglutide and the reconstitution calculator account for the specific properties of each compound. The cost calculator enables direct vendor price comparison on a per-dose or per-milligram basis.
For researchers tracking their protocols, the GLP-1 plotter provides visualization tools, and monitoring tools help document outcomes over time. These resources turn raw data into actionable insights that improve protocol optimization across research cycles.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact purchasing and quality questions.
Frequently asked questions
Is it legal to buy GLP-1 peptides online?
Research peptides labeled "for research use only" or "not for human consumption" can generally be purchased legally for legitimate research purposes. However, regulations vary by jurisdiction, and the FDA has taken enforcement action against vendors marketing research peptides in ways that imply human use. Compounded GLP-1 medications require a valid prescription. Always verify the legal framework in your specific location before purchasing. The research vs pharmaceutical peptides comparison explains these distinctions in detail.
How can I tell if a peptide vendor is legitimate?
Check for batch-specific COAs with HPLC chromatograms and mass spectrometry data, third-party testing from independent laboratories, a verifiable business address and phone number, established online presence with independent reviews, and reasonable pricing consistent with manufacturing costs. Avoid vendors with aggressive urgency marketing, generic documentation, or payment methods that lack buyer protection.
What is the difference between HPLC purity and net peptide content?
HPLC purity measures the target peptide as a percentage of all peptide-related material in the sample. Net peptide content measures the target peptide as a percentage of everything in the vial, including water, salts, and counterions. A peptide can be 99% pure by HPLC while having only 75-85% net peptide content. Both measurements are important for accurate dosing calculations.
How should I store GLP-1 peptides after purchase?
Lyophilized (unreconstituted) peptides should be stored at minus 20 degrees Celsius for long-term storage. After reconstitution, refrigerate at 2-8 degrees Celsius and use within the stability window, typically 28-56 days with bacteriostatic water. Never freeze reconstituted peptides. Protect from light and moisture.
Why do GLP-1 peptide prices vary so much between vendors?
Price variation reflects differences in synthesis quality, purification methods, testing standards, and business overhead. The cheapest options often skip third-party testing, use lower-purity raw materials, or cut corners on storage and shipping. The most expensive options are not always the best either. Focus on value: quality verification per dollar spent rather than absolute price. The peptide cost calculator helps compare effective per-milligram costs.
Can I buy semaglutide, tirzepatide, and retatrutide from the same vendor?
Many research peptide vendors offer multiple GLP-1 compounds. Buying from a single verified vendor simplifies quality assurance and logistics. However, some vendors specialize in specific compounds and may offer better quality or pricing for their focus product. Compare retatrutide vs semaglutide and semaglutide vs tirzepatide options across vendors rather than defaulting to a single source.
What should I do if my peptide arrives warm or damaged?
Contact the vendor immediately and document the condition with photographs. Most reputable vendors have replacement policies for shipping damage. Do not use peptides that arrived under questionable temperature conditions, as temperature excursions degrade peptide potency. If the vendor is unresponsive, this tells you everything you need to know about their reliability.
How do I know which GLP-1 peptide to buy for my research?
Match the compound to your research question. Semaglutide is the most studied and widely available. Tirzepatide offers dual-receptor activation for comparative studies. Retatrutide provides triple-receptor activity for researchers exploring the frontier of incretin biology. The differences between compounds should drive your selection.
What supplies do I need besides the peptide itself?
At minimum, you need bacteriostatic water for reconstitution, insulin syringes for measurement and administration, alcohol swabs for sterile technique, and a refrigerator for storage. For lyophilized peptides stored long-term, a freezer capable of minus 20 degrees Celsius is ideal. The GLP-1 syringe guide covers needle gauge, syringe volume, and selection criteria in detail. Many researchers also keep a peptide calculator bookmarked for quick reference during reconstitution and dosing calculations.
Should I buy GLP-1 peptides from international or domestic vendors?
Domestic vendors generally provide faster shipping with fewer temperature control risks, easier returns or replacements, and clearer legal standing. International vendors sometimes offer lower prices due to differences in manufacturing costs, but introduce customs delays, potential temperature exposure, and regulatory uncertainty. For researchers in the United States, domestic vendors with third-party testing from US-based laboratories provide the most straightforward quality verification pathway. If considering international sources, factor in the additional risks and potential for total loss if a shipment is seized at customs.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vendors stay verified, your peptides stay potent, and your research stay reproducible.