Feb 8, 2026

That vial has been sitting in your fridge for months. Maybe longer. You picked it up, checked the date, and felt that sinking feeling. Expired. But it looks fine. Clear solution, no particles, no weird color. So now you are wondering whether you can still use it.
You are not alone in asking this question. Thousands of people each month search for answers about expired semaglutide, and the confusion is understandable. Semaglutide is expensive. Getting a new prescription takes time. And throwing away medication that looks perfectly good feels wasteful, especially when you have been making real progress on your weight loss journey or finally getting your blood sugar under control.
But here is the problem. Semaglutide is not like the aspirin sitting in your medicine cabinet. It is not a simple chemical compound that stays stable for years past its printed date. Semaglutide is a peptide, a biologic medication made from complex protein structures that degrade in ways you cannot always see. And the consequences of using degraded semaglutide range from simply wasting your injection to genuinely risking your health.
This guide covers everything you need to know. We will walk through exactly what happens to semaglutide after it expires, why peptide medications behave differently from regular pills, how to tell if your medication has degraded, what to do if you accidentally used an expired dose, and the specific shelf life timelines for every form of semaglutide on the market. Whether you are using brand-name Ozempic, Wegovy, or compounded semaglutide, the information here applies to you.
Why semaglutide expiration matters more than regular medications
Most people grow up hearing that medication expiration dates are overly conservative. And for many drugs, that is actually true. The FDA Shelf Life Extension Program (SLEP), a joint initiative between the Department of Defense and FDA established in 1986, found that roughly 88% of tested medications remained stable an average of 66 months beyond their original expiration dates. Two-thirds of 122 medications tested stayed effective for at least four additional years.
So naturally, you might assume semaglutide follows the same pattern.
It does not.
Here is the critical distinction. The SLEP program explicitly excludes biologics. Peptide medications like semaglutide, BPC-157, TB-500, and other protein-based therapeutics are not eligible for shelf-life extension testing because they degrade through fundamentally different mechanisms than conventional small-molecule drugs.
A standard tablet like ibuprofen contains a relatively simple chemical structure. It might lose a small percentage of potency over years, but the molecule itself remains largely intact. Semaglutide, by contrast, is a 31-amino acid peptide chain with a complex three-dimensional structure held together by hydrogen bonds and hydrophobic interactions. When those bonds break down, the peptide does not just get weaker. It fundamentally changes.
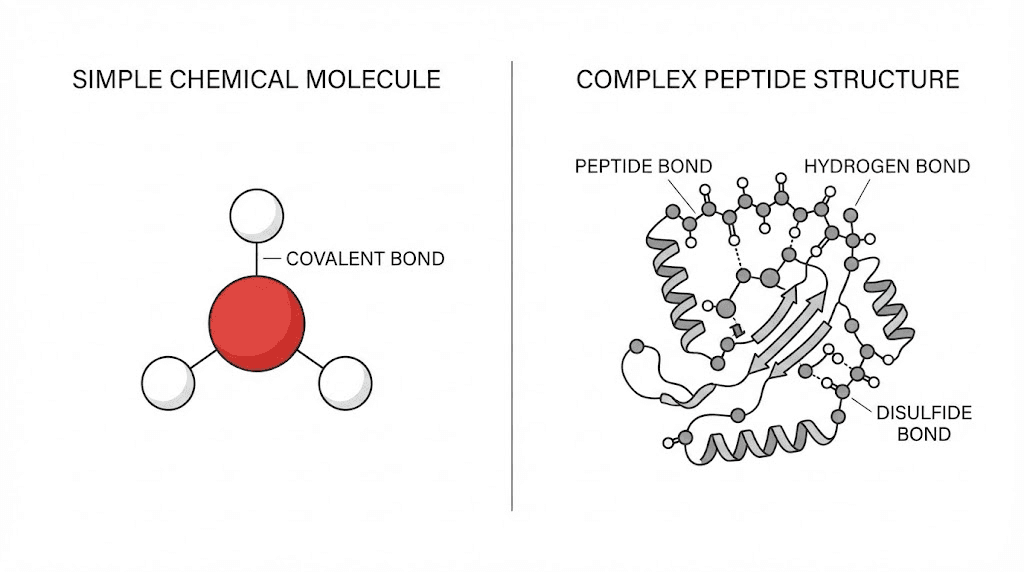
How peptide degradation works at the molecular level
When semaglutide degrades, several things happen simultaneously. The hydrogen bonds maintaining its active three-dimensional shape begin to break. This process, called denaturation, unfolds the peptide from its functional form into a shape that no longer fits the GLP-1 receptor properly. Think of it like a key that has been bent. It might still look like a key, but it will not open the lock.
Simultaneously, chemical degradation pathways activate. Oxidation attacks vulnerable amino acid residues. Hydrolysis breaks bonds in the peptide backbone itself. These reactions accelerate with heat, light exposure, and pH changes. Research published in the Journal of Peptide Science found that pH was a key factor in semaglutide thermal degradation, with higher degradation rates observed between pH 4.5 and 5.5, near the peptide isoelectric point of pH 5.4.
The degraded peptide fragments do not simply become inactive. They can aggregate into clumps or fibrils, tiny protein structures that your immune system may recognize as foreign. This aggregation is invisible to the naked eye in early stages, which is why expired semaglutide can look perfectly fine while being significantly compromised.
Understanding these mechanisms matters for anyone using semaglutide for dosing protocols. You could be injecting the right number of units but receiving a fraction of the actual active medication.
The real-world impact of reduced potency
Clinical research on semaglutide consistently shows that higher doses produce better results for both weight loss and blood sugar management. The dose-response relationship is well established. So what happens when your medication has lost 20%, 40%, or even 60% of its potency?
You get subtherapeutic dosing without knowing it.
For someone managing type 2 diabetes, this means blood glucose levels creep upward. You might attribute it to diet changes or stress. For someone using semaglutide for weight management, the appetite suppression weakens. Cravings return. Progress stalls. And you might blame yourself for lacking willpower when the real problem is sitting in your refrigerator.
This is arguably the most insidious risk of expired semaglutide. Not dramatic side effects, but quiet, invisible loss of the benefits you are counting on.
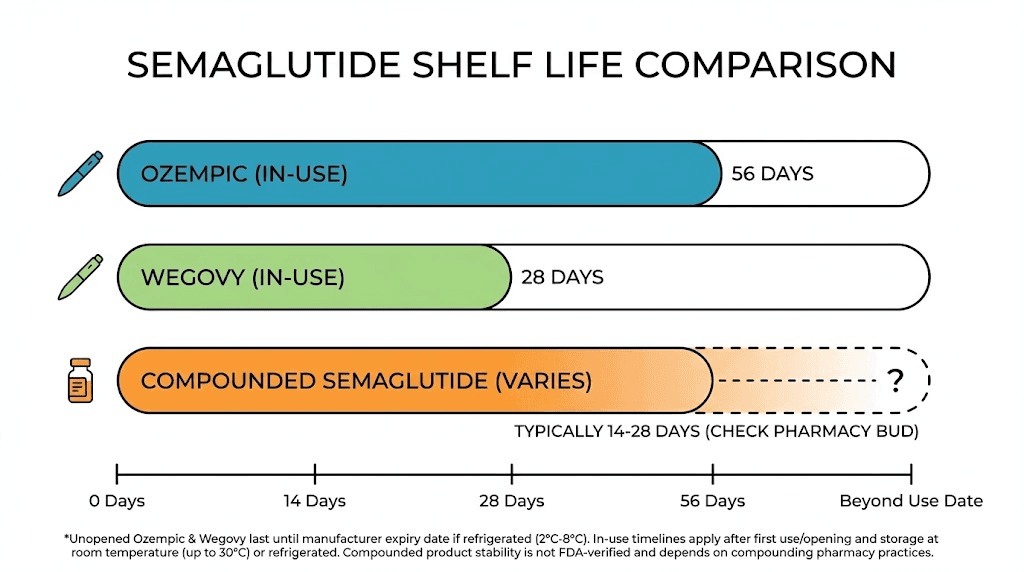
Exact shelf life timelines for every form of semaglutide
Different forms of semaglutide have different expiration windows. Knowing yours is the first step toward safe use. Here is every timeline you need, broken down by product type.
Brand-name Ozempic (injectable pen)
Unopened and refrigerated: 24 months from manufacture date. Some sources cite up to 36 months when stored at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Always check the specific expiration date printed on your pen.
After first use: 56 days. This applies whether you keep it refrigerated or store it at room temperature between 59 and 86 degrees Fahrenheit (15 to 30 degrees Celsius). After 56 days, discard the pen even if medication remains inside.
Temperature limits: Never expose to temperatures below 36 degrees Fahrenheit or above 86 degrees Fahrenheit. If either boundary is crossed, the medication may be compromised regardless of the expiration date.
These timelines matter for anyone following a semaglutide dosing schedule. Missing your window does not just waste medication, it potentially disrupts your entire protocol.
Brand-name Wegovy (injectable pen)
Unopened and refrigerated: Check manufacturer date, typically 24 months from production.
After first use: Wegovy pens are single-use. Each pen contains one dose. Use it and discard it. There is no multi-day storage window because the pen is not designed for multiple injections.
Temperature handling: Same parameters as Ozempic. Refrigerate until ready, avoid freezing, avoid temperatures above 86 degrees Fahrenheit.
Compounded semaglutide (vials)
This is where things get more complicated, and where most of the confusion around expired semaglutide actually originates.
Unopened vial: The beyond-use date (BUD) varies dramatically by pharmacy. Compounding pharmacies that conduct rigorous stability testing may assign BUDs of up to 120 days. Pharmacies relying on external stability data or less rigorous testing typically assign 14 to 28 days.
After first puncture: Most compounded semaglutide vials last 28 to 30 days once opened, assuming proper refrigeration. Some pharmacies specify shorter windows.
Critical difference: Compounded semaglutide does not undergo the same extensive stability testing as FDA-approved products. The formulation, preservatives, and concentration can vary between pharmacies. This means the shelf life printed on your compounded vial is often more conservative, but also less rigorously validated, than what you see on an Ozempic pen.
If you are using compounded semaglutide, always follow the specific instructions from your compounding pharmacy. When in doubt, call them directly. And if you are researching the differences between delivery methods, understanding injectable versus oral peptide options can help you make better decisions.
Oral semaglutide (Rybelsus)
Shelf life: Follow the manufacturer expiration date on the packaging. Store at room temperature, protected from moisture. Keep tablets in their original blister packaging until use.
Key difference: Oral semaglutide tablets are generally more stable than injectable forms because they do not rely on the same protein-in-solution structure. However, moisture exposure can degrade them, which is why the blister packaging matters.
What actually happens when you use expired semaglutide
Let us break down the specific risks, ranked from most likely to least likely. Understanding the probability helps you make informed decisions rather than panic.
Most likely: reduced effectiveness
This is the scenario that happens in the vast majority of cases. The semaglutide has lost some or most of its potency. You inject it. Nothing bad happens immediately. But you do not get the full therapeutic benefit.
For weight loss users, this means:
Appetite suppression weakens or disappears
Cravings return sooner than expected
Weekly weight loss slows or stalls
You might increase food intake without realizing the medication is the variable that changed
For diabetes management, this means:
Blood glucose readings drift upward
Post-meal spikes become more pronounced
A1C levels may rise at your next check
You might need medication adjustments that would not have been necessary with full-potency semaglutide
If you have been tracking your results and notice a sudden plateau, checking your medication expiration date should be one of the first things you investigate. The reasons for not losing weight on semaglutide are varied, but expired medication is one that people frequently overlook.
Moderately likely: gastrointestinal issues
Degraded semaglutide can sometimes trigger gastrointestinal side effects that differ from the normal nausea many users experience during dose escalation. This happens because the degraded peptide fragments interact with your GLP-1 receptors differently than the intact molecule.
Possible symptoms include:
Nausea that feels different from your usual semaglutide experience
Persistent stomach discomfort that does not follow the typical pattern
Diarrhea or vomiting beyond what you experienced when starting the medication
Bloating or cramping at unusual times relative to your injection
These symptoms overlap with normal semaglutide side effects, which makes them easy to dismiss. The key differentiator is the pattern. If you have been stable on your dose for weeks and suddenly experience GI symptoms without any dietary changes, degraded medication is worth investigating. For broader context on managing these effects, understanding semaglutide-related symptoms provides useful background.
Less likely but serious: infection risk
This is the risk that genuinely concerns medical professionals. After expiration, the preservative systems that maintain sterility in multi-dose vials and pens can become less effective. Research has found that approximately 5.6% of multi-dose medication vials develop bacterial growth over time.
Five point six percent might sound small. But consider what it means. If 100 people use expired multi-dose semaglutide vials, roughly 5 to 6 of them are injecting contaminated medication. The consequences of injecting bacteria directly into subcutaneous tissue include:
Injection site infections (redness, swelling, warmth, pain)
Abscess formation requiring medical drainage
Cellulitis (spreading skin infection)
In rare cases, bloodstream infections requiring hospitalization
This risk is higher with compounded semaglutide vials than with brand-name pens, because vials require repeated needle punctures through a rubber septum, creating more opportunities for contamination. Each puncture introduces a tiny pathway for bacteria. Over time, especially past the recommended use window, the cumulative risk increases.
Proper peptide storage after reconstitution practices reduce but do not eliminate this risk. And expired preservatives mean even careful handling may not be enough.
Rare: immune reactions from aggregated peptides
When semaglutide degrades, the peptide chains can clump together into aggregates and fibrils. These protein structures are larger and shaped differently than the original molecule. Your immune system, which is highly sensitive to unfamiliar protein shapes, may mount a response.
Potential immune reactions include:
Injection site reactions beyond normal (excessive redness, swelling, itching)
Systemic allergic responses (rash, hives, difficulty breathing in extreme cases)
Development of anti-drug antibodies that could reduce the effectiveness of future semaglutide doses
This last point is particularly concerning for long-term users. If your body develops antibodies against semaglutide fragments, it might start neutralizing even fresh, fully potent medication. One dose of expired semaglutide could theoretically compromise the effectiveness of your entire ongoing treatment.
This scenario is rare. But it is documented with other biologic medications, and the mechanism applies to semaglutide as well.

How to tell if your semaglutide has gone bad
Not all degradation is visible. But some is. Knowing what to look for can save you from injecting compromised medication.
Visual inspection checklist
Before every injection, examine your semaglutide against these criteria:
Clarity: Semaglutide should be completely clear and transparent. Hold the vial or pen up to light. The solution should look like water. Any haziness, cloudiness, or milky appearance indicates protein aggregation. Do not use it.
Color: Normal semaglutide is colorless. Any tint, whether pink, red, yellow, amber, or brown, signals degradation or contamination. Even a faint discoloration means the medication is compromised.
Particles: Look for floating specks, sediment at the bottom, crystal formations, or fibrous material. These indicate chemical precipitation or contamination. Even tiny particles disqualify the medication.
Consistency: The solution should flow freely when you tilt the vial. If it appears thickened, grainy, or syrupy, degradation has occurred.
Here is the catch, though. Early-stage degradation is invisible. The peptide can lose significant potency before any visual changes appear. A clear, colorless solution does not guarantee full potency. It only means the degradation has not progressed to the point of visible aggregation.
This is exactly why expiration dates exist. They mark the boundary beyond which the manufacturer cannot guarantee potency, even if the medication looks perfect.
Beyond visual inspection: other warning signs
Your body can tell you things your eyes cannot. Pay attention to these signals:
Changed injection experience. If the injection feels different, stings more than usual, or produces unusual redness at the site, the solution composition may have changed.
Altered effectiveness pattern. A sudden change in how the medication affects your appetite, energy, or blood sugar, without any other variable changing, could indicate potency loss.
Unusual side effects. New symptoms you have never experienced on semaglutide, especially at a dose you have been stable on, warrant investigation.
Tracking your progress systematically makes these changes easier to spot. SeekPeptides members use protocol tracking tools that help identify exactly when something shifts in their response pattern, making it easier to catch issues like expired medication before they derail progress.
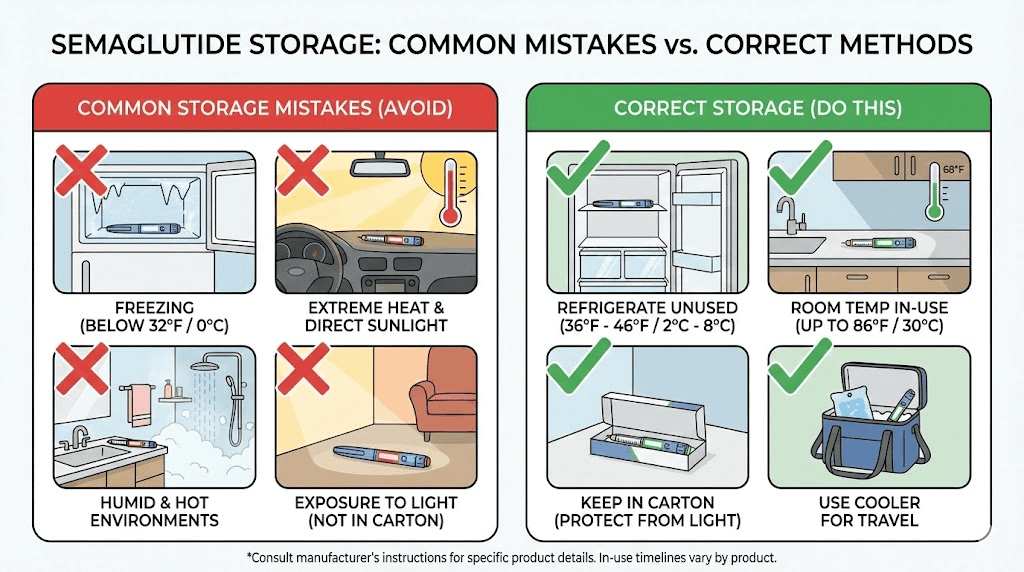
Storage mistakes that make semaglutide expire faster
Expiration dates assume proper storage. Improper handling can cause your semaglutide to degrade well before the printed date. Here are the mistakes that accelerate degradation.
Temperature excursions
This is the most common storage error, and the most damaging.
Leaving semaglutide out of the fridge too long. Every hour at room temperature counts against your medication stability. While Ozempic can tolerate room temperature for up to 56 days after first use, that clock starts ticking the moment you take it out. And compounded semaglutide may have a much shorter room-temperature tolerance.
Storing in the refrigerator door. The door is the warmest part of your fridge and experiences the most temperature fluctuation. Every time you open the door, the medication gets hit with warm air. Store semaglutide in the back of the main compartment, where temperature is most stable.
Accidental freezing. If semaglutide freezes, even briefly, the protein structure is permanently damaged. The ice crystals that form during freezing physically tear apart the peptide bonds. There is no recovery from this. If you suspect your semaglutide has frozen, discard it immediately, even if it thaws and looks clear. For deeper understanding of cold storage requirements, the peptide refrigeration guide covers the science behind proper temperature maintenance.
Heat exposure during shipping or travel. Semaglutide shipped in summer without cold packing, or left in a hot car, can degrade rapidly. Research shows semaglutide remains stable for only about 3 hours at 80 degrees Celsius (176 degrees Fahrenheit), demonstrating the peptide thermal limits. At more moderate but still elevated temperatures, degradation is slower but cumulative.
Light exposure
Ultraviolet light accelerates oxidation of vulnerable amino acid residues in the semaglutide peptide chain. This is why both brand-name pens and compounded vials use opaque or tinted packaging.
Common mistakes:
Removing pens from their outer carton before use
Storing vials on a countertop or windowsill
Using clear travel cases without UV protection
Keep semaglutide in its original packaging until you are ready to inject. If using a travel case, choose one that blocks light. These practices align with broader peptide storage best practices that apply to all protein-based medications.
Contamination through handling
Every time you puncture a multi-dose vial, you create a potential pathway for bacteria. The preservatives in the formulation, typically benzyl alcohol or phenol, are designed to handle some level of contamination. But they have limits, and those limits shrink as the medication ages.
Best practices:
Always clean the vial top with an alcohol swab before drawing medication
Never touch the rubber septum with your fingers
Use a fresh needle for every withdrawal
Do not store vials with needles attached
Keep the protective cap on when not in use
These steps apply to all peptide injection protocols and become even more critical as medication approaches or passes its expiration date.
Compounded versus brand-name semaglutide: expiration differences that matter
The distinction between compounded and brand-name semaglutide becomes especially important when discussing expiration. The two products follow different regulatory frameworks, different stability testing protocols, and different shelf-life designations.
Brand-name stability testing
Novo Nordisk, the manufacturer of Ozempic and Wegovy, conducts extensive stability studies as part of the FDA approval process. These studies test the medication at various temperatures and humidity levels over extended periods. The expiration date on your Ozempic pen represents the point beyond which Novo Nordisk can no longer guarantee the medication meets its quality specifications based on these rigorous studies.
The testing includes accelerated stability studies (exposure to elevated temperatures to predict long-term behavior) and real-time stability studies (actual storage over the full shelf-life period). This is why brand-name products can confidently assign 24-month or longer expiration dates.
Compounded stability testing
Compounding pharmacies operate under different rules. The beyond-use date (BUD) on your compounded semaglutide vial might be based on:
USP guidelines: For pharmacies following United States Pharmacopeia standards, the default BUD for aqueous preparations without stability data is 14 days under refrigeration. With proper stability testing, this can extend to 30, 60, 90, or even 120 days.
In-house testing: Some larger compounding pharmacies conduct their own stability studies. These results directly determine the BUD they assign. The quality and scope of these studies varies significantly between pharmacies.
Third-party data: Some pharmacies rely on stability data from ingredient suppliers or third-party laboratories. This data may or may not perfectly match the specific formulation, concentration, or packaging used by that pharmacy.
The practical impact? Two compounded semaglutide vials with the same concentration from different pharmacies might have very different BUDs, and both could be legitimate based on the available data. If you are comparing options, understanding the difference between research and pharmaceutical grade peptides provides important context.
What this means for you
If you are using brand-name Ozempic or Wegovy, the expiration date is well-validated. Trust it.
If you are using compounded semaglutide, the BUD is your hard stop. Do not assume it has the same margin of safety as a brand-name product. Contact your pharmacy if you have questions about their specific stability testing protocols. And never use a compounded product past its labeled BUD, even by a day or two.
I accidentally used expired semaglutide: what to do right now
First, do not panic. A single dose of recently expired semaglutide is unlikely to cause serious harm. But you should take specific steps to protect yourself.
Step 1: Stop using the expired medication
Do not take another dose from the same vial or pen. Switch to a fresh supply if available, or contact your prescriber for a replacement.
Step 2: Assess what you used
Consider these factors to gauge your risk level:
How far past expiration? A few days past is very different from several months past. Risk increases with time beyond the expiration date.
How was it stored? Properly refrigerated medication degrades slower than medication that experienced temperature excursions. If it was kept at the right temperature the entire time, the risk is lower.
What did it look like? If the solution was clear, colorless, and particle-free, the medication may have still been partially effective. If you noticed any visual changes, the risk is higher.
Brand-name or compounded? Brand-name products generally have more stability margin built into their expiration dates. Compounded products have less margin.
Step 3: Monitor for symptoms
Watch for these warning signs over the next 24 to 72 hours:
Injection site reactions. Redness, swelling, warmth, pain, or any discharge at the injection site. A small amount of redness immediately after injection is normal. Increasing redness, spreading warmth, or swelling that develops hours later is not.
Gastrointestinal symptoms. Severe or unusual nausea, vomiting, or diarrhea that differs from your normal semaglutide experience.
Signs of infection. Fever, chills, body aches, or general malaise. These could indicate bacterial contamination.
Blood sugar changes. If you use semaglutide for diabetes, monitor your blood glucose more frequently for the next few days. Readings above 300 mg/dL warrant immediate medical attention.
Step 4: Contact your healthcare provider
Call your prescriber or pharmacist and let them know what happened. They can:
Assess your individual risk based on your health profile
Recommend additional monitoring if needed
Prescribe a replacement dose so you do not miss your schedule
Advise whether the expired dose should count toward your treatment or be repeated
Step 5: Dispose of the expired medication properly
Do not flush semaglutide down the toilet or throw it in regular trash. Most pharmacies accept expired medications for safe disposal. Many communities also hold periodic drug take-back events. The FDA recommends mixing medications with coffee grounds or cat litter in a sealed container before disposal if no take-back options are available.
The FDA position on expired medications (and why biologics are different)
The FDA takes a clear position: do not use medications past their expiration dates. Period. But to understand why this matters more for semaglutide than for your leftover antibiotics, you need to understand what expiration dates actually mean in regulatory terms.
What an expiration date guarantees
When a pharmaceutical company sets an expiration date, they are making a legally binding guarantee. They are stating that the medication will maintain at least 90% of its labeled potency and meet all quality specifications (sterility, pH, particulate matter) through that date, assuming proper storage.
The FDA requires stability data to support every expiration date. For brand-name semaglutide, this includes real-time data from products stored under recommended conditions for the full shelf-life period. The testing is exhaustive, repeated, and documented.
What happens after the expiration date
After the expiration date, the guarantee ends. This does not automatically mean the medication is dangerous. It means nobody can tell you with certainty that it is safe and effective. The manufacturer liability ends. The FDA approval applies only to the product within its labeled shelf life. Your prescriber cannot guarantee outcomes.
For conventional small-molecule drugs like blood pressure pills or pain relievers, the gap between the expiration date and actual degradation is often large. Hence the SLEP findings about medications lasting years longer.
For biologics like semaglutide, the gap is much narrower, and the consequences of degradation are more unpredictable. Peptide degradation produces fragments and aggregates that may trigger immune responses. Small-molecule degradation typically just produces less active compound.
This is why every medical professional, every pharmacy, and every manufacturer gives the same advice about expired semaglutide. Do not use it. The risk-benefit calculation is fundamentally different from taking an expired ibuprofen.
Common myths about expired semaglutide
Misinformation about expired semaglutide circulates widely. Let us address the most common myths directly.
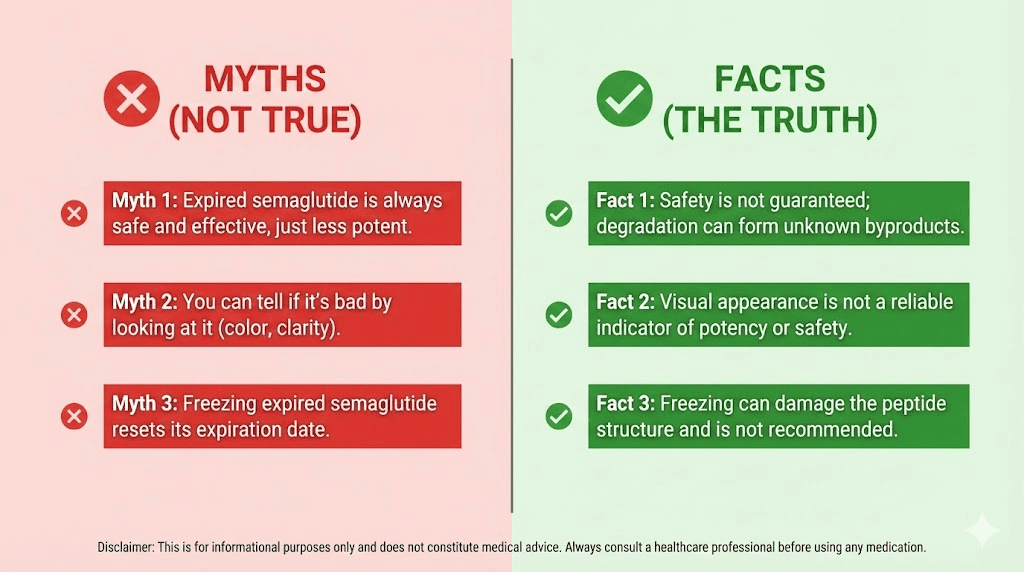
Myth 1: If it looks clear, it is still good
Wrong. Visual clarity tells you that gross degradation has not occurred. It tells you nothing about potency loss, chemical degradation at the molecular level, or early-stage aggregation. A vial of semaglutide that lost 50% of its potency looks exactly the same as a fresh one.
This myth is especially dangerous because it gives false confidence. People check the vial, see clear solution, and assume they are safe. Meanwhile, the therapeutic benefit they are counting on may be significantly reduced.
Myth 2: Expiration dates are just a marketing ploy to sell more medication
This conspiracy theory is popular but wrong, at least for biologics. Pharmaceutical companies would actually prefer longer shelf lives because it reduces waste, lowers return rates, and simplifies distribution. Novo Nordisk assigns the shelf life that their stability data supports. If they could justify a longer one, they would.
For conventional drugs, there is some truth to the idea that expiration dates are conservative. But for peptide medications, the expiration date often represents a genuine boundary beyond which the product cannot be trusted.
Myth 3: Keeping it refrigerated extends the expiration date
Proper refrigeration is necessary to reach the expiration date, not to extend past it. The printed expiration assumes proper refrigeration. If you have been storing semaglutide at the correct temperature, the expiration date is accurate. If you have not, the effective expiration date is earlier than what is printed.
Refrigeration slows degradation. It does not stop it. Chemical and physical degradation processes continue at refrigerator temperatures, just at a slower rate. This is why even perfectly stored semaglutide eventually expires.
Myth 4: A few days past expiration is fine
Maybe. But you are gambling. The expiration date is not a cliff edge where the medication is perfect on day zero and toxic on day one. Degradation is gradual and continuous. On the day after expiration, the medication is probably very similar to how it was the day before. A week later, less so. A month later, significantly less predictable.
The problem is that you cannot quantify the degradation without laboratory testing. You are making a blind guess about potency and safety. For an expensive medication with real health implications, that is a risky bet.
Myth 5: You can extend shelf life by freezing semaglutide
This is not just wrong, it is actively harmful. Freezing semaglutide destroys the medication. Ice crystals that form during freezing physically rupture the peptide protein structure. The damage is permanent and irreversible. Once frozen, semaglutide is useless, even if it thaws and appears clear.
This myth likely originates from the fact that some lyophilized (freeze-dried) peptides can be stored frozen in powder form. But reconstituted peptides in solution, which is what semaglutide pens and vials contain, cannot tolerate freezing. The difference between lyophilized and liquid peptide storage is critical here.
How to avoid wasting semaglutide (and never face the expiration dilemma)
The best approach to expired semaglutide is never having it in the first place. Here are practical strategies to prevent waste.
Timing your prescriptions
Work with your prescriber and pharmacy to align your refill schedule with your actual usage rate. If you inject weekly, a 4-pen supply should last about a month. Getting refills too early creates stockpile that might expire before you use it.
For compounded semaglutide with shorter BUDs, this becomes even more important. A 30-day BUD means you need to use the entire vial within 30 days of it being compounded, not 30 days from when you receive it. Clarify the BUD start date with your pharmacy.
First in, first out
If you have multiple vials or pens, always use the oldest one first. Write the date you received each one on the packaging. This prevents the scenario where a newer pen gets used while an older one sits untouched until it expires.
Track your doses
Keep a simple log of when you started each pen or vial and when you expect to finish it. This helps you identify potential timing problems before medication expires. Many people using semaglutide protocols already track their doses for efficacy monitoring. Adding an expiration check to that routine takes seconds.
Proper storage from day one
Start storing correctly the moment you receive the medication. Do not leave it on the counter while you unpack other items. Do not store it in the door of your fridge. Put it in the back of the main compartment immediately.
For travel, use an insulated medical cooler with gel packs. Not loose ice, which can cause freezing. Not a regular cooler without temperature monitoring. And never leave semaglutide in a car, even briefly. Car interior temperatures can exceed 140 degrees Fahrenheit in summer, far beyond the 86-degree limit.
Communicate with your pharmacy
If your dosing schedule changes, such as during dose titration when you are moving from lower to higher doses, let your pharmacy know. They can adjust your refill quantities and timing so you receive only what you will use within the shelf-life window.
Semaglutide degradation compared to other peptide medications
Understanding how semaglutide compares to other peptides helps put the expiration risk in context. Not all peptides degrade at the same rate or through the same mechanisms.
Why semaglutide is relatively stable (for a peptide)
Semaglutide was engineered for stability. Its design includes a fatty acid side chain (C-18 fatty di-acid) that allows it to bind to albumin in the blood, extending its half-life. This same structural modification also provides some protection against degradation in storage compared to unmodified peptides.
Research shows semaglutide remains stable for up to 3 hours at 80 degrees Celsius, which is remarkably robust for a peptide. By comparison, many research peptides like BPC-157 and TB-500 are significantly more fragile in solution.
The pH stability profile is also relevant. Semaglutide is most stable at pH values above 7.0, which is why commercial formulations are buffered to this range. Below pH 5.5, degradation accelerates significantly due to the peptide isoelectric point at pH 5.4.
Comparison with other GLP-1 medications
Different GLP-1 receptor agonists have different stability profiles:
Tirzepatide (Mounjaro/Zepbound): Similar storage requirements to semaglutide. Unopened pens last until the manufacturer expiration date when refrigerated. After first use, tirzepatide in the fridge follows its own specific timeline. The dual GIP/GLP-1 mechanism means degradation affects two receptor pathways rather than one.
Liraglutide (Saxenda/Victoza): Shorter half-life than semaglutide, requiring daily rather than weekly dosing. Storage profile is similar, but the daily dosing means vials and pens get used faster, reducing the chance of expiration.
Retatrutide: A newer triple-agonist compound still in clinical trials. Storage and stability data are less well-established. For those researching this compound, the retatrutide dosing guide covers what is currently known.
Regardless of which GLP-1 medication you use, the fundamental principle remains the same. Peptides degrade. Expiration dates exist for a reason. And the consequences of ignoring them are real, even if they are not always immediate or dramatic.
The cost factor: when money tempts you to use expired semaglutide
Let us address the elephant in the room. Semaglutide is expensive. Brand-name Ozempic can cost over $900 per month without insurance. Even compounded versions, while cheaper, represent a significant investment. When an entire month supply expires before you use it, the financial sting is real.
But consider the cost-benefit analysis honestly.
The cost of using expired semaglutide
Lost effectiveness: If the medication has degraded by even 30%, you are injecting a subtherapeutic dose. The medication effectively costs more per unit of benefit because you are paying full price for partial results.
Medical costs: An injection site infection from contaminated medication can easily cost thousands in medical treatment. An abscess requiring drainage means an emergency room visit, antibiotics, and follow-up care. These costs dwarf the price of a new semaglutide prescription.
Delayed progress: If expired semaglutide stalls your weight loss or destabilizes your blood sugar control, you lose weeks or months of progress. For diabetes patients, poor glucose control increases long-term complication risks that carry enormous costs.
Potential immune sensitization: If degraded peptide fragments trigger antibody production, your body might become less responsive to semaglutide itself. This could force you to switch to a different, potentially more expensive medication.
Better alternatives to using expired medication
If cost is the primary driver, explore these options instead:
Talk to your prescriber about compounded semaglutide, which is typically much less expensive than brand-name products
Check manufacturer patient assistance programs (Novo Nordisk offers several)
Explore insurance coverage options, as many plans now cover GLP-1 medications
Adjust your dosing schedule in consultation with your prescriber rather than stretching medication beyond its shelf life
Use peptide cost calculators to compare options and find the most cost-effective approach for your protocol
The bottom line? Using expired semaglutide is not saving money. It is gambling with your health and your progress. The math almost never works out in your favor.
Special situations: what about semaglutide that expired during dose transitions
Dose transitions create unique expiration challenges. Here are the most common scenarios and how to handle them.
You titrated up and have leftover lower-dose medication
When moving from 0.25mg to 0.5mg, or from 0.5mg to 1.0mg, you might have remaining medication at the lower dose. If it is still within the expiration window, some prescribers may advise continuing to use it if you need to step back down temporarily. But if it has expired, discard it and request a new prescription at the appropriate dose.
You paused treatment and your supply expired
Treatment breaks happen. Side effects, surgery, pregnancy planning, or simply running out of refills can pause your semaglutide use. If your supply expired during the break, you need fresh medication when restarting.
Important: when restarting semaglutide after a break, most prescribers recommend re-titrating from a lower dose rather than jumping back to your previous dose. This applies whether your medication expired or not. Using expired medication to restart at a lower dose is still not advisable. Get a fresh supply and titrate properly.
Your pharmacy sent medication close to expiration
This occasionally happens, especially with brand-name products that moved slowly through the supply chain. If you receive semaglutide with less than 6 months until expiration on a brand-name product, or less than the full BUD on compounded product, contact your pharmacy. You may be entitled to a replacement with a longer shelf life.
Document the expiration dates when you receive medication. Take a photo of the packaging and note it in your dosing log. This protects you in case of disputes and helps you track your supply timeline.
How temperature history affects expiration beyond the printed date
The expiration date on your semaglutide assumes something important: unbroken cold chain storage. If the cold chain was broken at any point, the effective expiration date is earlier than what is printed.
What is a cold chain break?
A cold chain break occurs whenever semaglutide leaves the recommended temperature range. Common scenarios include:
Sitting on a pharmacy counter during processing
Transport from pharmacy to home without insulation
Power outage affecting your refrigerator
Accidental placement in a freezer compartment
Leaving the pen out after an injection
Travel without proper cooling
Each temperature excursion accumulates. Semaglutide does not reset when returned to proper temperature. The degradation that occurred during the warm period is permanent. This cumulative effect means that even brief excursions, repeated over time, can significantly reduce actual shelf life.
How to track temperature history
For high-value medications like semaglutide, consider using a simple refrigerator thermometer to monitor your storage temperature. Digital thermometers with min/max recording show whether your fridge dipped or spiked during power fluctuations. Some models even have alarms.
If you travel frequently with semaglutide, temperature-indicating labels designed for pharmaceutical transport can show whether your medication exceeded safe temperatures during transit. These are inexpensive and provide peace of mind.
Maintaining proper room temperature storage limits for peptides is equally important for periods when the medication is outside the fridge.
Proper disposal of expired semaglutide
Once you determine your semaglutide has expired, proper disposal protects both you and the environment.
Preferred: pharmacy take-back programs
Most pharmacies accept expired medications for proper disposal. Call your pharmacy to confirm they participate. Many chain pharmacies have collection boxes in-store. This is the safest and most environmentally responsible option.
Alternative: DEA take-back events
The Drug Enforcement Administration holds periodic National Prescription Drug Take-Back events. While semaglutide is not a controlled substance, these events accept most medications.
Home disposal method
If no take-back option is available, the FDA recommends this process:
Remove the semaglutide from its original container
Mix it with an undesirable substance like used coffee grounds, dirt, or cat litter
Place the mixture in a sealed container such as a zip-lock bag
Remove or scratch out all personal information on the prescription label
Place the sealed container in household trash
For pens, remove the needle (if attached) and place the pen in a sharps container before disposal. Do not recap used needles.
Do not flush semaglutide. Do not pour it down the drain. The active compound can enter water systems and affect aquatic organisms.
The science behind semaglutide stability testing
For those who want to understand the deeper science, here is how pharmaceutical companies determine expiration dates for peptide medications.
ICH stability testing guidelines
The International Council for Harmonisation (ICH) sets global standards for pharmaceutical stability testing. These guidelines require testing under multiple conditions:
Long-term studies: Storage at recommended conditions (2-8 degrees Celsius for semaglutide) for the full proposed shelf life. Samples are tested at regular intervals, typically every 3 months for the first year, then every 6 months thereafter.
Accelerated studies: Storage at elevated temperatures (typically 25 degrees Celsius at 60% relative humidity, and 40 degrees Celsius at 75% relative humidity) for 6 months. These studies predict long-term stability by stressing the product.
Stress studies: Exposure to extreme conditions including high temperature, light, oxidation, and pH extremes. These identify degradation pathways and help develop stability-indicating analytical methods.
What they test for
At each time point, the medication is analyzed for:
Potency: Does it still contain at least 90% of the labeled active ingredient?
Purity: Have degradation products formed? Are they within acceptable limits?
Sterility: Is the product still free from microbial contamination?
pH: Has the solution acidity changed?
Particulates: Are visible or sub-visible particles present?
Appearance: Has the color or clarity changed?
Container closure: Is the packaging still maintaining its seal?
The expiration date is set at the point where any of these parameters first falls outside acceptable limits, with a safety margin. This means the medication might still be partially effective past its expiration date, but at least one quality attribute is no longer guaranteed.
Why you cannot replicate this at home
Some people argue that visual inspection is sufficient to determine if medication is still good. But most of the critical quality attributes, potency, purity, sub-visible particles, microbial load, require laboratory testing with specialized equipment like high-performance liquid chromatography (HPLC) and mass spectrometry. The peptide testing laboratory guide explains these analytical methods in detail.
You cannot test potency by looking at a vial. You cannot detect 5% bacterial contamination by examining the solution. You cannot measure pH changes by color alone. This is why the expiration date exists: it replaces laboratory testing with a simple date check that anyone can perform.
What healthcare professionals actually say about expired semaglutide
Medical consensus on this topic is unusually clear. Here is what different types of healthcare professionals advise.
Endocrinologists
Endocrinologists who prescribe semaglutide for diabetes management are unanimous: do not use expired medication. Their primary concern is blood sugar destabilization. A patient who relies on semaglutide for glucose control and unknowingly uses degraded medication could experience dangerous hyperglycemia. For patients already managing their semaglutide dosing calculations, expired medication introduces an uncontrolled variable that undermines the entire protocol.
Pharmacists
Pharmacists are trained to be the last line of defense for medication safety. They consistently advise against using expired semaglutide, citing both potency concerns and contamination risks. Many pharmacists specifically mention the preservative degradation issue in multi-dose vials as a key concern.
Weight loss physicians
Obesity medicine specialists who prescribe semaglutide for weight management echo the same advice. Their additional concern is the psychological impact. A patient who uses expired semaglutide, experiences reduced appetite suppression, and concludes the medication does not work for them might discontinue a therapy that would have been effective with proper medication.
The medical community alignment
It is rare for the entire medical community to agree so clearly on an issue. But the advice on expired semaglutide is unanimous across specialties, across professional organizations, and across national health authorities. Do not use it. Get a replacement. The risks outweigh any perceived savings or convenience.
Building a semaglutide management system that prevents waste
Rather than wondering about expired medication, build a system that prevents the problem entirely. Here is a practical framework.
The semaglutide tracking protocol
When you receive medication:
Photograph the expiration date or BUD
Write the received date on the packaging
Store immediately in the correct location
Add a calendar reminder for 2 weeks before expiration
When you open a pen or vial:
Write the date opened on the packaging with a permanent marker
Set a reminder for the end of the open-container use window (56 days for Ozempic, check your specific product)
Calculate how many doses remain and confirm you will use them before the window closes
Weekly checks:
Visual inspection before each dose (clarity, color, particles)
Confirm the medication is still within its use window
Check refrigerator temperature
Note any changes in effectiveness
This system takes minimal effort but prevents the expired medication dilemma entirely. SeekPeptides members access protocol tracking tools that make this kind of systematic medication management straightforward, with reminders and logging built in.
Working with your healthcare team
Communication prevents waste. Tell your prescriber and pharmacist:
Your exact injection schedule
Any planned breaks or dose changes
If you are accumulating unused medication
If you received medication close to expiration
They can adjust prescribing quantities, refill timing, and product selection to minimize the chance of medication expiring before you use it.
Frequently asked questions
How long past the expiration date can you use semaglutide?
The official medical recommendation is zero days past expiration. Using semaglutide beyond its printed expiration date or beyond-use date means the manufacturer and FDA no longer guarantee its potency, purity, or sterility. While degradation is gradual rather than instant, you have no way to know how much potency has been lost without laboratory testing. For a medication that affects blood sugar and appetite regulation, this uncertainty creates genuine risk.
Does expired semaglutide become toxic?
Expired semaglutide does not typically become acutely toxic like some medications. Instead, the peptide degrades into fragments that are less active. The primary risk is reduced potency rather than toxicity. However, bacterial contamination in expired multi-dose vials can cause serious infections, and peptide aggregates from degradation can potentially trigger immune responses. So while toxicity in the traditional sense is unlikely, the medication is not risk-free after expiration.
Can I tell if my semaglutide has expired just by looking at it?
Visual inspection can identify advanced degradation, including cloudiness, discoloration, and visible particles, but it cannot detect early or moderate degradation. Semaglutide can lose substantial potency while still appearing completely clear and colorless. Visual inspection is a necessary safety check before every injection, but it is not a substitute for respecting the expiration date. Always perform both checks.
Is compounded semaglutide more likely to expire quickly?
Yes. Compounded semaglutide typically has a shorter beyond-use date than brand-name products. Where Ozempic might have a 24-month unopened shelf life, compounded semaglutide BUDs range from 14 to 120 days depending on the compounding pharmacy stability testing. The formulation, preservatives, and packaging differ between compounders, making generalization difficult. Always follow the specific BUD assigned by your compounding pharmacy.
What should I do with semaglutide pens that still have medication inside but have expired?
Discard them properly. Do not use the remaining medication. Return them to a pharmacy for safe disposal, use a DEA take-back program, or mix the contents with coffee grounds or cat litter in a sealed container for household trash. Remove and safely dispose of any attached needles in a sharps container. The remaining medication, no matter how much is left, is not worth the risk of using after expiration.
Does storing semaglutide perfectly extend its shelf life beyond the expiration date?
No. The expiration date already assumes perfect storage at the recommended temperature of 36 to 46 degrees Fahrenheit. Storing it perfectly allows the medication to reach its full labeled shelf life. It does not extend beyond it. The degradation processes, while slowed by refrigeration, continue regardless. Consider the expiration date as the maximum life under ideal conditions, not a starting point for extension.
Can my doctor test whether my semaglutide is still good?
Your doctor does not have the equipment to test semaglutide potency. Testing requires analytical chemistry instruments like HPLC and mass spectrometry, found in pharmaceutical quality control laboratories. A specialized peptide testing lab could theoretically analyze a sample, but the cost would far exceed simply replacing the medication. If there is any question about your semaglutide quality, replacement is always more practical than testing.
I accidentally froze my semaglutide. Is it expired now?
Effectively yes, though for a different reason than time-based expiration. Freezing physically destroys the peptide structure through ice crystal formation. This damage is permanent and irreversible. Even if the solution looks clear after thawing, the active peptide has been damaged. Discard any semaglutide that has been frozen, regardless of what the expiration date says. This applies to all peptide medications stored in solution.
External resources
For researchers serious about managing their peptide protocols effectively, SeekPeptides provides comprehensive storage guides, stability databases, and protocol tracking tools designed to help you get the most from every dose while avoiding situations like expired medication.
In case I do not see you, good afternoon, good evening, and good night. May your semaglutide stay potent, your storage stay cold, and your progress stay on track.