Feb 26, 2026
Experienced researchers figured this out early. The powder form is not a limitation. It is not an inconvenience. It is the reason their results stay consistent month after month while others wonder why potency drops after two weeks in the fridge. Lyophilized tirzepatide, the freeze-dried powder that arrives in a sealed vial, represents the most stable and reliable way to work with this dual GIP/GLP-1 receptor agonist. And yet most people who encounter it for the first time feel intimidated. They see a dry white cake at the bottom of a vial and assume something is missing.
Nothing is missing.
What they are looking at is tirzepatide in its most potent, most shelf-stable, most chemically intact state. The moisture has been removed through a controlled freeze-drying process called lyophilization, halting the degradation reactions that destroy peptides in liquid form. Every major research supplier ships tirzepatide this way for a reason. The powder can survive temperature fluctuations during transit, sit in storage for months or even years without losing potency, and allow for precise custom dosing once reconstituted. This guide covers everything about lyophilized tirzepatide, from the science behind freeze-drying to step-by-step reconstitution, storage protocols, vial sizes, quality testing, and the common mistakes that waste product and compromise results. Whether you are handling your first vial or refining a protocol you have been running for months, SeekPeptides built this resource to be the only reference you need.
What lyophilized tirzepatide actually is
The word lyophilized comes from the Greek words for "solution loving," which is ironic because the entire point of the process is to remove the solution. Lyophilization is a pharmaceutical technique that freezes a substance and then reduces the surrounding pressure to allow frozen water to sublimate directly from solid ice to vapor. The result is a dry powder or cake that retains the original molecular structure of the peptide while eliminating the moisture that drives chemical degradation.
The lyophilization process explained
The manufacturing process involves three distinct phases. First, the tirzepatide solution is frozen to extremely low temperatures, typically -40 to -80 degrees Celsius. This converts all the water in the solution to ice while preserving the peptide structure within the ice matrix. Second, during primary drying, the chamber pressure is reduced to near-vacuum conditions while gentle heat is applied. Under these conditions, ice sublimates directly to water vapor without passing through a liquid phase, which would risk damaging the peptide. Third, secondary drying removes residual moisture bound to the peptide molecules, bringing the moisture content down to 1-3% or less.
This three-phase process takes 24-72 hours depending on the batch size and equipment. It is significantly more complex and expensive than simply preparing a liquid solution, which is why lyophilized products sometimes carry a premium price. But the stability advantage justifies the added manufacturing cost many times over.
What makes tirzepatide unique
Tirzepatide is a 39-amino-acid peptide that acts as a dual agonist, activating both GIP and GLP-1 receptors simultaneously. This dual mechanism is what separates it from single-agonist peptides like semaglutide. The GIP receptor activation enhances insulin secretion and promotes fat metabolism in ways that GLP-1 alone does not achieve. Meanwhile, the GLP-1 component suppresses appetite, slows gastric emptying, and supports blood sugar regulation. Together, these pathways create a synergistic effect that has shown superior efficacy in clinical trials compared to GLP-1-only agonists.
In its native state as a peptide chain in aqueous solution, tirzepatide is vulnerable to several degradation pathways. Hydrolysis breaks peptide bonds when water molecules attack the backbone. Deamidation alters asparagine and glutamine residues over time. Oxidation damages methionine residues and reduces biological activity. The 39-amino-acid chain includes a C20 fatty diacid moiety that enables albumin binding and extends the half-life in the body, but this fatty acid component also creates additional degradation vulnerability in solution.
Remove the water and you stop all three.
That is why pharmaceutical manufacturers and research suppliers default to the lyophilized form. The freeze-dried powder you receive in a sealed vial is tirzepatide at maximum potency, locked in a state where degradation reactions essentially cannot proceed. When stored correctly, lyophilized peptides can remain stable for years. Compare that to a reconstituted solution that begins losing potency within weeks, and the advantage becomes obvious.
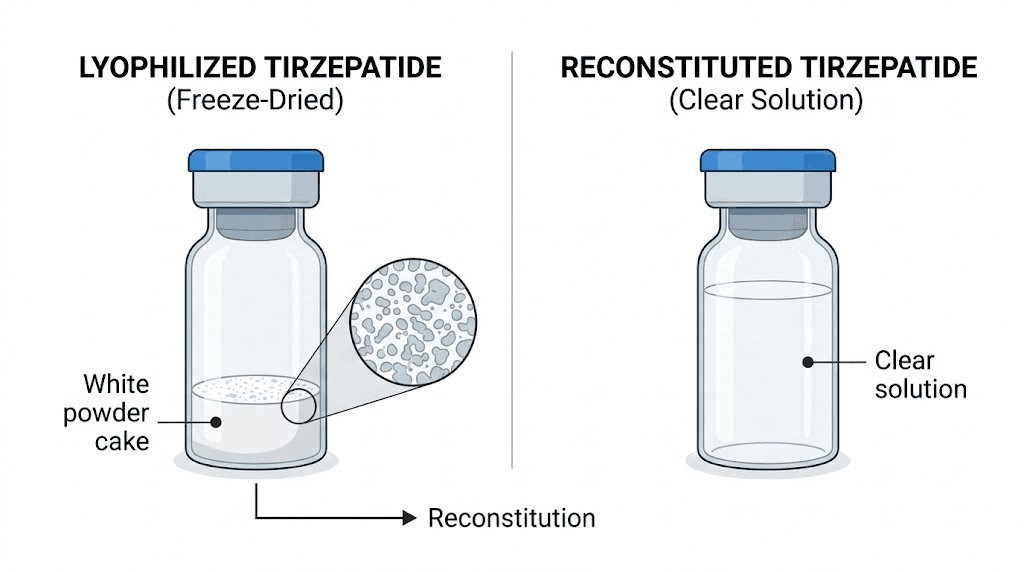
The visual appearance of lyophilized tirzepatide varies slightly between manufacturers. Some produce a compact white cake that fills the bottom portion of the vial. Others create a loose, fluffy powder. Both are normal. What matters is the color and consistency. The powder should be white to off-white. Any significant yellowing, brown spots, or gel-like texture suggests degradation or contamination. A properly lyophilized vial will dissolve readily when mixed with bacteriostatic water, producing a clear and colorless solution within minutes of gentle mixing.
Understanding the fundamental differences between lyophilized and liquid peptides is the first step to handling tirzepatide correctly. The form factor is not just a packaging choice. It is a deliberate pharmaceutical decision that directly impacts the potency, shelf life, and reliability of every dose you draw from that vial.
Why the lyophilized form matters for stability
Stability is not an abstract concept when it comes to peptides. It is the difference between a protocol that produces consistent results and one that quietly fails because the compound degraded in the vial before you ever drew a dose.
Three primary degradation pathways threaten tirzepatide in solution. Hydrolysis occurs when water molecules attack peptide bonds, literally breaking the amino acid chain apart. This reaction accelerates with temperature and proceeds even at refrigerator temperatures, just more slowly. Deamidation converts asparagine residues to aspartate through a reaction with water, altering the peptide structure and reducing receptor binding affinity. Oxidation targets methionine and cysteine residues, particularly when the solution is exposed to light or trace metals.
Lyophilization stops all of these simultaneously by removing the reaction medium: water.
In powder form, there is simply not enough molecular mobility for these degradation reactions to proceed at any meaningful rate. Research from peptide stability studies shows that lyophilized tirzepatide stored at -20 degrees Celsius retains greater than 98% potency for over 24 months. The same peptide in aqueous solution at refrigerator temperature (2-8 degrees Celsius) begins showing measurable degradation within 4-6 weeks. At room temperature, degradation accelerates dramatically, with significant potency loss possible within days.
This stability advantage extends beyond storage to shipping. Lyophilized vials can tolerate brief temperature excursions during transit without catastrophic potency loss. A liquid vial that sits in a hot delivery truck for several hours may lose significant activity. A lyophilized vial in the same conditions will be fine because the degradation reactions that heat accelerates require water to proceed.
The practical implication is straightforward. If you are serious about getting consistent, reliable results from your tirzepatide protocol, the lyophilized form is not just preferred. It is essential. Understanding tirzepatide expiration and degradation helps you plan purchases and storage, but starting with the most stable form gives you the widest margin for error.
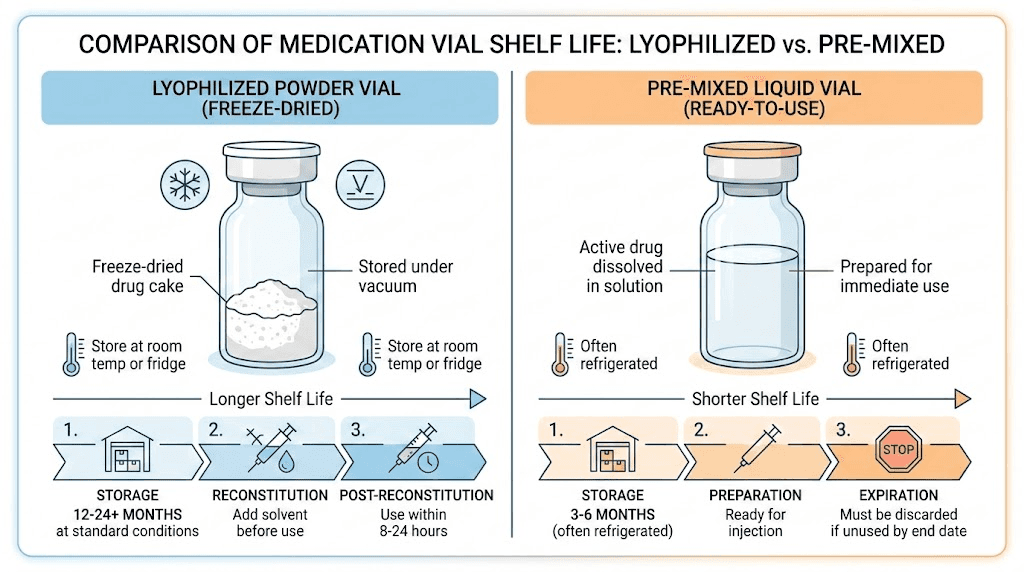
Lyophilized vs liquid tirzepatide
Not all tirzepatide products arrive as powder. Pre-mixed liquid formulations exist, typically from compounding sources that prepare ready-to-inject solutions. Understanding the tradeoffs between these forms helps you make informed decisions about which is right for your situation.
The key differences at a glance
Factor | Lyophilized (powder) | Liquid (pre-mixed) |
|---|---|---|
Shelf life (unopened) | 1-3+ years at -20C | 60-90 days refrigerated |
Shelf life (after opening) | 28-60 days refrigerated | 28-60 days refrigerated |
Shipping stability | Tolerates temperature excursions | Requires cold chain |
Dosing flexibility | Custom concentration based on reconstitution volume | Fixed concentration |
Convenience | Requires reconstitution step | Ready to inject |
Potency assurance | Higher (stable form) | Variable (degrades in solution) |
Cost per mg | Generally lower | Generally higher |
The convenience factor is the main appeal of liquid formulations. No mixing, no calculations, no bacteriostatic water. Draw your dose and inject. For people who feel intimidated by reconstitution or who simply want the simplest possible workflow, liquid makes sense.
But convenience comes at a cost.
Every day that tirzepatide sits in solution, degradation reactions chip away at its potency. The clock starts ticking the moment the powder dissolves. With liquid formulations, that clock started at the compounding pharmacy, meaning you have no idea how much shelf life remains by the time the vial reaches you. Factor in shipping time, warehouse storage, and delivery delays, and a liquid vial with a 90-day shelf life might arrive with only 50-60 usable days remaining.
Lyophilized vials, by contrast, arrive in their most stable state. You control when reconstitution happens. You start the clock yourself. For researchers running protocols that span months, this control is invaluable. You can purchase multiple vials, store them long-term as powder, and reconstitute each one only when you are ready to begin using it.
The choice between injectable forms also extends to oral versus injectable tirzepatide, with newer sublingual drops and orally disintegrating tablets entering the market. Each delivery method has its own stability profile, but for injectable protocols, lyophilized powder remains the gold standard for potency assurance.
Who should choose lyophilized
Lyophilized tirzepatide is the better choice for anyone running protocols longer than four weeks, anyone who wants to purchase in bulk, anyone who ships to locations with unreliable temperature control, and anyone who prioritizes maximum potency over maximum convenience. If you have five minutes to mix a vial and a basic understanding of the process, the lyophilized form is almost always the smarter investment.
Common vial sizes and what to expect
Lyophilized tirzepatide is available in several standard vial sizes. The most common include 5mg, 10mg, 15mg, 30mg, and 60mg. Some research suppliers also offer 20mg and 72mg options. The vial size you choose depends on your dosing protocol, how quickly you will use the reconstituted product, and whether you prefer to reconstitute smaller quantities more frequently or larger quantities less often.
Small vials: 5mg and 10mg
These are popular starting points. A 5mg vial provides enough tirzepatide for two weeks at the standard 2.5mg starting dose. A 10mg vial covers roughly four weeks at 2.5mg per week or two weeks at 5mg per week. The advantage of smaller vials is reduced waste. If you are new to tirzepatide and still assessing tolerance, a smaller vial means less product at risk if something goes wrong with storage or reconstitution.
The powder in a 5mg vial looks like a thin disc or a small amount of fluffy powder at the bottom. In a 10mg vial, the cake is slightly larger. Both should appear white to off-white with no discoloration.
Medium vials: 15mg and 30mg
Medium vials are the sweet spot for most ongoing protocols. A 30mg vial at a 5mg weekly dose lasts roughly six weeks, which falls comfortably within the recommended 28-60 day post-reconstitution window. These vials require slightly more bacteriostatic water during reconstitution to maintain a manageable concentration, and the powder cake is correspondingly larger.
For researchers working through the standard tirzepatide dose escalation schedule, a 30mg vial provides enough flexibility to move through 2.5mg, 5mg, and 7.5mg weekly doses within a single vial lifecycle.
Large vials: 60mg and 72mg
Large vials are designed for researchers at higher doses or those who want to minimize the frequency of reconstitution. A 60mg vial at 10mg per week lasts six weeks. At 15mg per week, roughly four weeks. These vials contain a substantial amount of powder and require more bacteriostatic water to achieve usable concentrations.
The main risk with large vials is shelf life after reconstitution. Once mixed, the solution should be used within 28-60 days depending on storage conditions. If your dose schedule means a 60mg vial will take longer than that to finish, a smaller vial reconstituted more frequently is the safer choice.
Choosing the right vial size for your protocol
The best vial size depends on three factors: your current weekly dose, the post-reconstitution shelf life window, and whether you want to minimize waste or minimize reconstitution frequency. Here is a practical framework.
If you are at the 2.5mg starting dose, a 5mg or 10mg vial makes the most sense. The 5mg vial gives you exactly two doses, meaning you will reconstitute every two weeks. Some researchers find this annoying. Others prefer the freshness guarantee that comes with a newly mixed vial every 14 days. A 10mg vial covers four weeks at 2.5mg, which fits neatly within the standard 28-day post-reconstitution window.
At maintenance doses of 7.5-15mg per week, larger vials become practical. A 30mg vial at 7.5mg weekly lasts exactly four weeks. At 10mg weekly, three weeks. A 60mg vial at 15mg weekly lasts four weeks. These timelines all fall within the safe use window, making larger vials both cost-effective and safe.
The calculation is straightforward. Divide the vial size in mg by your weekly dose in mg. That gives you the number of weeks the vial will last. If the answer is greater than four weeks, consider a smaller vial or accept that you may need to discard unused solution at the end.
Visual quality indicators
Regardless of vial size, here is what to check before reconstitution. The powder should be white to off-white. The vial seal should be intact with no signs of tampering. There should be no moisture droplets inside the vial, which would suggest compromised lyophilization. The powder should not appear clumped, gel-like, or yellowed. And the vial should have a clear label with batch number, manufacture date, and peptide quantity.
If you notice any of these warning signs, contact the supplier before reconstituting. Using a compromised vial means potentially injecting degraded or contaminated material, which is never worth the risk.
How to reconstitute lyophilized tirzepatide step by step
Reconstitution is the process of dissolving the lyophilized powder back into a liquid solution for injection. This is not complicated. But it does require attention to detail, clean technique, and patience. Rushing this step or cutting corners on sterility is the most common source of problems.
Supplies you need
Before you start, gather everything. You will need a vial of lyophilized tirzepatide, bacteriostatic water (BAC water containing 0.9% benzyl alcohol), alcohol swabs, a sterile syringe for drawing the water (typically a 3mL or 5mL syringe with a blunt-tip or standard needle), a clean flat work surface, and an insulin syringe for drawing doses after reconstitution.
A critical note on the solvent choice. Always use bacteriostatic water, not sterile water for injection. BAC water contains benzyl alcohol as a preservative that inhibits bacterial growth. Since you will be drawing multiple doses from the same vial over several weeks, bacterial contamination is a real risk with plain sterile water. The preservative in BAC water dramatically extends the safe use window of a multi-dose vial.
Workspace preparation
Clean your work surface with isopropyl alcohol or a disinfectant wipe. Wash your hands thoroughly. Some researchers use gloves, which adds an additional contamination barrier. Remove the lyophilized vial and BAC water from storage and allow them to reach room temperature before proceeding. Cold vials can cause condensation that introduces unwanted moisture, and temperature differences between solvent and powder can affect dissolution.
Step-by-step reconstitution
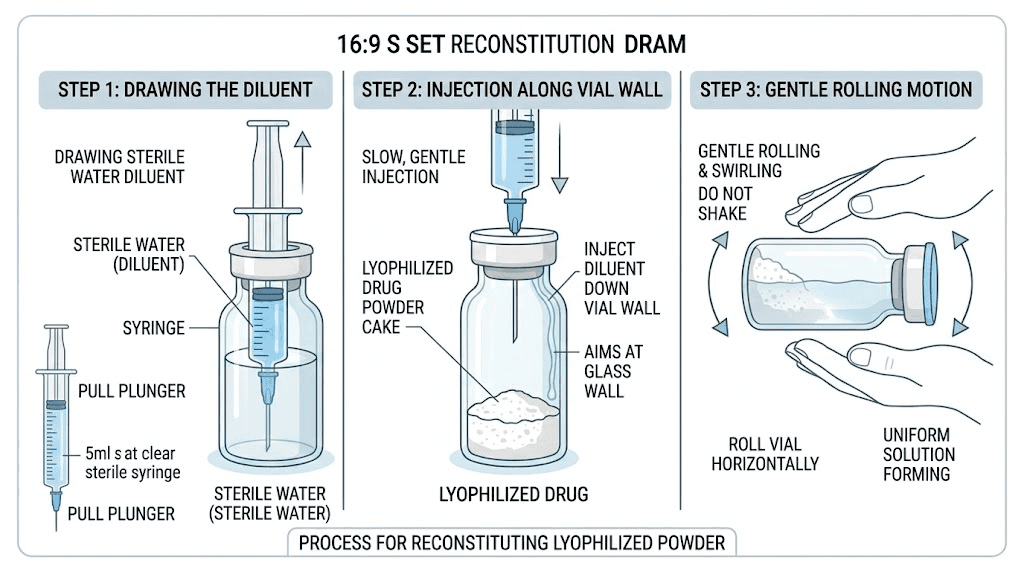
Step 1. Remove the plastic cap from the lyophilized tirzepatide vial and the BAC water vial. Wipe both rubber stoppers with a fresh alcohol swab. Allow the alcohol to dry completely before proceeding. This takes about 30 seconds.
Step 2. Draw the calculated amount of BAC water into your syringe. The exact volume depends on your vial size and desired concentration. Refer to the reconstitution ratio table in the next section for specific volumes.
Step 3. Insert the needle through the rubber stopper of the tirzepatide vial at a slight angle. Position the needle tip so it touches the inside wall of the vial, not the powder directly. This is important. Injecting water directly onto the lyophilized cake can cause foaming and protein damage from the impact force.
Step 4. Slowly depress the syringe plunger, allowing the BAC water to run gently down the inside wall of the vial. Do not rush this. A slow, controlled flow takes about 30-60 seconds for 2mL of water. The water will naturally flow down to the powder and begin dissolving it.
Step 5. Remove the syringe. Do not shake the vial. Shaking creates turbulence that can damage the peptide structure through a process called mechanical denaturation, where physical forces unfold and break the three-dimensional structure the peptide needs to function. Instead, gently tilt the vial on its side and slowly roll it between your palms. Some people prefer to let the vial sit undisturbed for 5-10 minutes, allowing the powder to dissolve passively.
Step 6. Check the solution. It should be clear and colorless with no visible particles, clumps, or cloudiness. A slight swirl when tilted is normal and will clear within seconds. If undissolved powder remains, continue gentle rolling until fully dissolved. Most lyophilized tirzepatide dissolves completely within 2-5 minutes. If particles persist after 10 minutes of gentle mixing, there may be an issue with the product.
Step 7. Label the vial with the reconstitution date and the concentration (mg/mL). Store immediately in the refrigerator at 2-8 degrees Celsius. The post-reconstitution clock has now started.
This process is nearly identical to reconstituting semaglutide or other lyophilized peptides. The peptide reconstitution calculator on SeekPeptides can help you determine exact water volumes for any vial size and target concentration, taking the guesswork out of the math.
Troubleshooting reconstitution problems
Sometimes reconstitution does not go perfectly. Here are the most common issues and how to handle them.
The powder is not dissolving. Most lyophilized tirzepatide dissolves within 2-5 minutes of gentle mixing. If chunks or clumps remain after 10 minutes, try placing the vial in the refrigerator for 15-20 minutes and then gently rolling again. Sometimes cold temperatures help stubborn formulations dissolve. If the powder still does not dissolve after 30 minutes, the product may be degraded or improperly lyophilized. Contact the supplier.
The solution looks cloudy. A properly reconstituted tirzepatide solution should be clear and colorless. Slight haziness immediately after mixing that clears within a few minutes is normal, caused by micro-bubbles from the dissolution process. Persistent cloudiness that does not clear suggests protein aggregation, which means the peptide has been damaged. Do not use a persistently cloudy solution.
There is foam on top. Foam indicates that the water was added too quickly or that the vial was agitated too vigorously. Let the vial sit undisturbed for 30-60 minutes. The foam will dissipate. Avoid drawing doses through the foam layer, as it can cause inaccurate measurements. Wait until the foam has completely resolved before drawing your first dose.
Small particles are floating in the solution. Visible particles after thorough mixing are a rejection criterion. They indicate either incomplete dissolution, aggregated peptide, or contamination. Do not filter and use the solution. Discard the vial and use a fresh one. This is one reason buying from suppliers with strong quality control matters.
Reconstitution ratios for every vial size
The amount of bacteriostatic water you add determines the concentration of your reconstituted solution. A higher concentration means less liquid per dose, which some researchers prefer for smaller injection volumes. A lower concentration means more liquid per dose but easier measurement of small doses. The right ratio depends on your weekly dose, syringe type, and personal preference.
Standard reconstitution ratios
Vial size | BAC water added | Concentration | Units per 2.5mg dose | Units per 5mg dose |
|---|---|---|---|---|
5mg | 1.0 mL | 5.0 mg/mL | 50 units (0.5 mL) | 100 units (1.0 mL) |
5mg | 2.0 mL | 2.5 mg/mL | 100 units (1.0 mL) | N/A (full vial) |
10mg | 2.0 mL | 5.0 mg/mL | 50 units (0.5 mL) | 100 units (1.0 mL) |
10mg | 1.0 mL | 10.0 mg/mL | 25 units (0.25 mL) | 50 units (0.5 mL) |
15mg | 2.0 mL | 7.5 mg/mL | 33 units (0.33 mL) | 67 units (0.67 mL) |
30mg | 3.0 mL | 10.0 mg/mL | 25 units (0.25 mL) | 50 units (0.5 mL) |
30mg | 2.0 mL | 15.0 mg/mL | 17 units (0.17 mL) | 33 units (0.33 mL) |
60mg | 3.0 mL | 20.0 mg/mL | 12.5 units (0.125 mL) | 25 units (0.25 mL) |
60mg | 6.0 mL | 10.0 mg/mL | 25 units (0.25 mL) | 50 units (0.5 mL) |
How to calculate your own ratio
The formula is simple. Concentration equals peptide amount divided by water volume. If you have a 10mg vial and add 2mL of BAC water, your concentration is 10 divided by 2, which equals 5mg per mL. To draw a 2.5mg dose from this solution, you need 0.5mL, which is 50 units on a standard U-100 insulin syringe.
To figure out units for any dose, use this formula: dose in mg divided by concentration in mg per mL, multiplied by 100. That gives you the number of units to draw on a U-100 syringe. The tirzepatide dosage calculator handles this math automatically and shows you exactly how many units to draw based on your vial size and reconstitution volume.
For specific unit conversions, these resources break down the math for common scenarios: 2.5mg in units, 5mg in units, 7.5mg in units, 10mg in units, 12.5mg in units, and 15mg in units.
Why concentration choice matters
Higher concentrations (like 10mg/mL or 20mg/mL) mean smaller injection volumes per dose. This is more comfortable, creates smaller injection site bumps, and reduces the amount of preservative injected per dose. However, measuring very small volumes (under 10 units) with a standard insulin syringe becomes imprecise, increasing the risk of dosing errors.
Lower concentrations (like 2.5mg/mL or 5mg/mL) mean larger injection volumes but more accurate measurement of each dose. This is especially important at lower doses like 2.5mg, where precision matters most for dose escalation protocols.
Most researchers find that a concentration between 5-10mg/mL offers the best balance of injection comfort and dosing accuracy. The tirzepatide dosage chart in units and reconstitution chart provide visual references for common configurations.
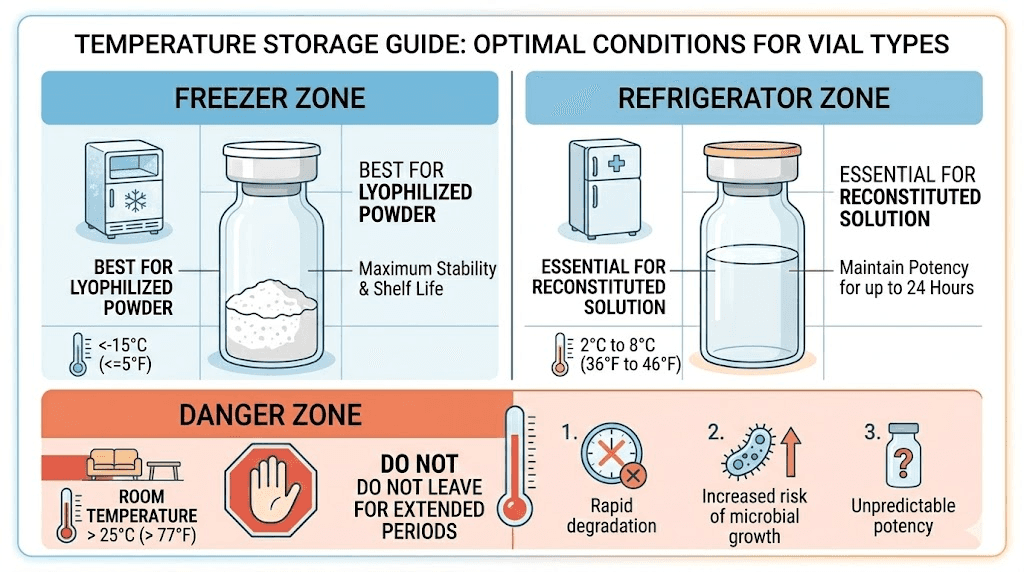
Storage requirements before and after reconstitution
Storage is where most people either get it right and enjoy full potency for the life of the vial, or get it wrong and watch their investment degrade in the refrigerator. The rules are different depending on whether the tirzepatide is still in powder form or has been reconstituted into solution.
Storing lyophilized powder (pre-reconstitution)
Unreconstituted lyophilized tirzepatide is remarkably stable. The ideal storage conditions are:
Long-term storage (months to years): Freezer at -20 degrees Celsius (-4 degrees Fahrenheit) in a sealed, dry environment away from light. This is the gold standard for maximum shelf life. Many researchers report maintaining potency for 2-3 years or longer under these conditions.
Medium-term storage (weeks to months): Refrigerator at 2-8 degrees Celsius (36-46 degrees Fahrenheit). Perfectly acceptable for vials you plan to use within the next few months.
Short-term storage (days to weeks): Room temperature at 15-25 degrees Celsius (59-77 degrees Fahrenheit) in a dry, dark location. The powder remains stable at room temperature for extended periods, though refrigeration or freezing is preferred when possible.
The key enemies of lyophilized peptides are moisture, light, and extreme heat. Keep vials sealed until ready to reconstitute. Store them in a dark location or wrap in aluminum foil if your storage area has light exposure. And never leave vials in a hot car, near a window, or in direct sunlight.
Storing reconstituted solution (post-reconstitution)
Once you add bacteriostatic water and the powder dissolves, the stability rules change completely. Reconstituted tirzepatide must be stored in the refrigerator at 2-8 degrees Celsius. Not in the freezer. Not at room temperature. The refrigerator, specifically.
Freezing reconstituted tirzepatide damages the peptide. Ice crystal formation during freezing physically disrupts protein structure, potentially rendering the peptide inactive. This is different from the controlled lyophilization process, which carefully manages ice crystal formation to preserve peptide integrity. Your home freezer does not offer this level of control.
Room temperature storage after reconstitution dramatically accelerates all the degradation pathways that lyophilization was designed to prevent. Studies show that peptides left at room temperature can lose significant potency within hours to days, depending on the specific peptide and temperature.
The standard shelf life for reconstituted tirzepatide with bacteriostatic water is 28 days, with some sources extending this to 60 days under ideal refrigeration conditions. The exact duration depends on several factors: the purity of the original peptide, sterility of the reconstitution process, consistent refrigeration temperature, and how often the vial is accessed for dose withdrawal.
Each time you insert a needle through the stopper to draw a dose, there is a small contamination risk. Using clean technique, wiping the stopper with alcohol before each entry, and using fresh sterile syringes minimizes this risk. But it is another reason why smaller vials used more quickly can be advantageous over larger vials used over longer periods.
Temperature excursions and what to do
Real life happens. Power outages, forgotten vials, delivery delays. If your reconstituted tirzepatide has been left out of the fridge, here is the general guidance: brief excursions of 1-2 hours at room temperature are unlikely to cause significant degradation. Longer periods, especially at higher temperatures, introduce increasing risk. If the solution has been at room temperature for more than a few hours, its remaining shelf life should be considered significantly shortened.
For lyophilized powder, temperature excursions are far less concerning. Even several days at room temperature will not significantly impact a properly sealed lyophilized vial. This is one of the biggest practical advantages of purchasing the powder form rather than pre-mixed solutions.
Shelf life of lyophilized tirzepatide
Shelf life varies dramatically depending on the form and storage conditions. Understanding these timelines helps you plan purchases, avoid waste, and ensure every dose delivers its intended potency.
Unreconstituted shelf life
Lyophilized tirzepatide stored at -20 degrees Celsius in a sealed vial retains potency for 1-3 years or longer, depending on purity and manufacturing quality. Most reputable suppliers assign a 12-month shelf life on the label, which represents the guaranteed stability period based on their accelerated stability testing. Actual stability often exceeds this by a significant margin.
At refrigerator temperatures (2-8 degrees Celsius), lyophilized tirzepatide remains stable for 6-12 months. At room temperature (15-25 degrees Celsius), expect 3-6 months of reliable stability, though this is the least recommended long-term storage option.
Reconstituted shelf life
After reconstitution with bacteriostatic water, the standard recommendation is to use the solution within 28 days. Some protocols and sources extend this to 60 days under strict refrigeration. The 28-day figure provides a conservative safety margin that accounts for gradual degradation and potential bacterial growth even with the BAC water preservative.
Understanding compounded tirzepatide expiration dates is especially important if you receive pre-reconstituted vials from a pharmacy. The clock on those vials started when the pharmacy mixed them, not when you received them.
Signs of degradation
Watch for these warning signs in reconstituted solution:
Cloudiness or turbidity that was not present immediately after reconstitution
Visible particles floating in the solution
Color change from clear to yellow, amber, or any other tint
Gel-like consistency or increased viscosity
Unusual odor that was not present before
Any of these signs suggest that the peptide has degraded, the solution has been contaminated, or both. Do not use a vial showing these characteristics. The risk of injecting degraded or contaminated material far outweighs the cost of a replacement vial.
If you use expired tirzepatide, the most likely outcome is simply reduced or absent efficacy rather than active harm, though contaminated solutions carry additional infection risks. The same shelf life principles apply to other lyophilized GLP-1 peptides like semaglutide.
Quality indicators and purity testing
Not all lyophilized tirzepatide is created equal. The source, manufacturing process, and quality testing determine whether that white powder in the vial is actually what it claims to be and whether it will perform as expected.
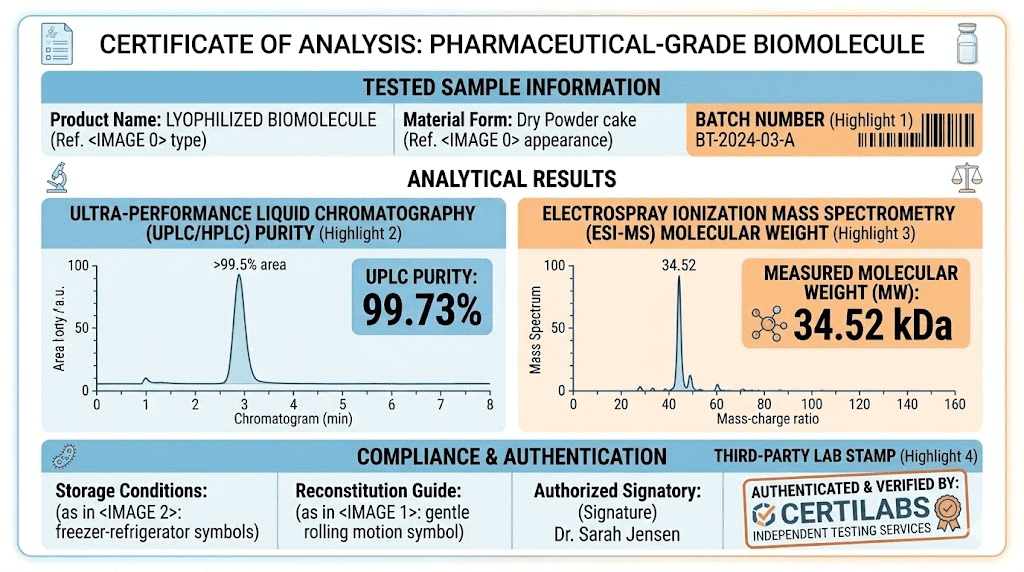
Certificate of Analysis (COA)
Every legitimate supplier should provide a Certificate of Analysis for each batch. This document contains the test results proving the product identity, purity, and safety. A proper COA includes the batch or lot number, the test date, the testing laboratory name, and results from multiple analytical methods.
The most critical distinction: third-party testing versus in-house testing. In-house COAs are produced by the supplier themselves, which creates an obvious conflict of interest. Third-party COAs come from independent laboratories with no financial stake in the results. Always prefer suppliers who provide third-party COAs.
HPLC testing
High-Performance Liquid Chromatography (HPLC) is the standard method for assessing peptide purity. This technique separates the components of a sample and quantifies the proportion that is actually tirzepatide versus impurities, degradation products, or other contaminants. For research-grade tirzepatide, a purity of 98% or higher is acceptable. Premium suppliers achieve 99% or above.
Purity below 95% is a red flag. It suggests either poor manufacturing quality, improper storage, or intentional under-filling of vials. Some grey market sources offer tirzepatide at suspiciously low prices, and low purity is one of the ways they cut costs.
Mass spectrometry
Mass spectrometry confirms the molecular identity of the peptide by measuring its exact molecular weight. Tirzepatide has a specific molecular weight (approximately 4,813.45 daltons), and mass spec verification ensures the product is actually tirzepatide and not a different peptide, a fragment, or a mixture. This is an identity test rather than a purity test, and it complements HPLC results.
Endotoxin and sterility testing
For injectable products, bacterial endotoxin testing (LAL test) and sterility testing are essential safety checks. Endotoxins are toxic components of bacterial cell walls that can cause fever, inflammation, and in severe cases, septic shock when injected. Sterility testing confirms the absence of viable microorganisms in the product.
Not all research-grade suppliers perform these tests. Pharmaceutical-grade products from compounding pharmacies are required to. Understanding the differences between research-grade and pharmaceutical-grade products helps you assess the quality assurance level of your specific source.
What to look for in a supplier
Reputable suppliers of lyophilized tirzepatide will offer third-party COAs with HPLC and mass spec data, clear batch numbering and expiration dates, proper packaging in sealed vials with crimped caps, visible lyophilized cake (not loose powder scattered around the vial, which suggests rough handling), and responsive customer service that can answer technical questions about their products.
Reviews and reputation matter too. Resources like the Peptide Sciences tirzepatide review and guides covering specific vendors like Empower, BPI Labs, and Strive can help you evaluate options before purchasing.
Dosing from a lyophilized vial
Once your tirzepatide is reconstituted, dosing follows the standard tirzepatide dose escalation protocol. The lyophilized form does not change the dosing, just the preparation required before the first dose.
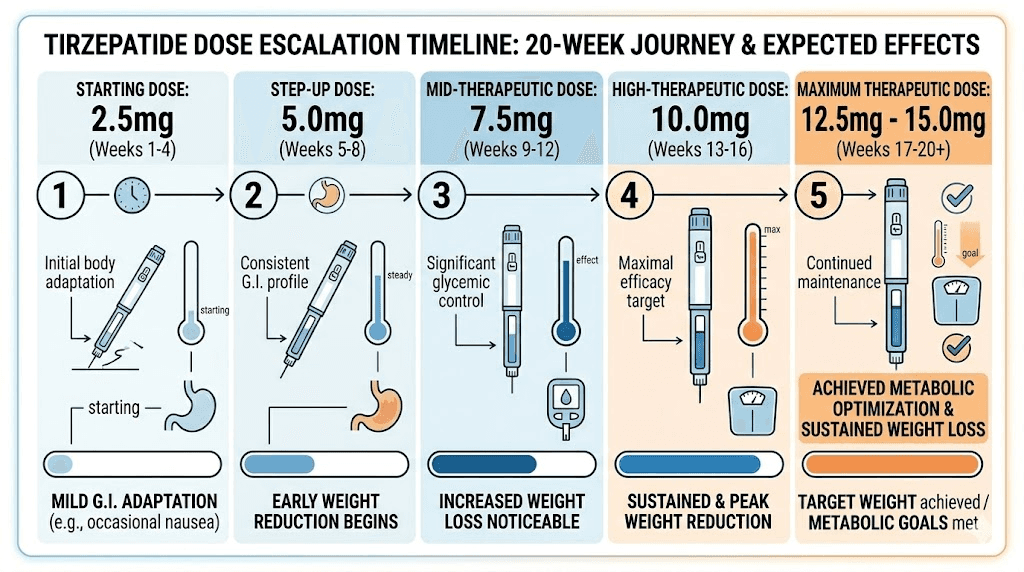
Standard dose escalation
The typical tirzepatide dose escalation schedule progresses as follows:
Weeks 1-4: 2.5mg once weekly
Weeks 5-8: 5.0mg once weekly
Weeks 9-12: 7.5mg once weekly
Weeks 13-16: 10.0mg once weekly
Weeks 17-20: 12.5mg once weekly
Weeks 21+: 15.0mg once weekly (maximum dose)
This gradual escalation allows the body to adapt to each dose level, minimizing gastrointestinal side effects that commonly occur when starting or increasing the dose. The starting dose of 2.5mg is deliberately low, designed primarily for tolerability rather than therapeutic effect. Most researchers begin to notice meaningful effects at the 5mg or 7.5mg level.
Drawing doses with an insulin syringe
Standard U-100 insulin syringes are the tool of choice for drawing and administering tirzepatide doses. Each unit marking on the syringe corresponds to 0.01mL of solution. Knowing your vial concentration, you can calculate exactly how many units to draw for any target dose.
For example, if your vial is reconstituted to 5mg/mL and you need a 2.5mg dose, you draw 50 units (0.5mL). If your vial is at 10mg/mL and you need 5mg, you also draw 50 units (0.5mL). The tirzepatide dosing in units guide provides a complete reference for common concentration and dose combinations.
For unit-to-mg conversions at specific doses: 10 units, 20 units, 30 units, 40 units, 50 units, and 75 units each contain detailed conversion tables.
Injection technique
Tirzepatide is administered subcutaneously, meaning into the fat layer just beneath the skin. Common injection sites include the abdomen (at least two inches from the navel), the front of the thighs, and the upper arm. Most researchers prefer the abdomen for its consistent absorption rate and larger injection area.
Rotate injection sites with each dose to prevent lipodystrophy (changes in fat tissue at the injection site) and reduce injection site reactions. If you notice redness, itching, or swelling at injection sites, the injection site reaction treatment guide covers management strategies.
Microdosing and split dosing
Some researchers use lower-than-standard doses for specific goals. Microdosing tirzepatide involves using sub-therapeutic doses, often for autoimmune or anti-inflammatory purposes rather than weight loss. The lyophilized form is particularly well-suited for microdosing because you can reconstitute to a low concentration that makes small doses easy to measure accurately.
Others prefer splitting their weekly dose into two injections, typically to reduce peak-related side effects. The microdose schedule and microdose chart provide specific protocols for these approaches.
Common mistakes with lyophilized tirzepatide
Mistakes happen. But the most costly ones are also the most preventable. Here are the errors that waste product, compromise potency, and undermine results.
Mistake 1: Shaking the vial during reconstitution
This is the single most common mistake. Vigorous shaking creates foam and turbulence that can denature the peptide, physically unfolding its three-dimensional structure and rendering it inactive. The instinct to shake is understandable since we shake so many other things to mix them. But peptides are not salad dressing. Gentle rolling, tilting, or passive dissolution is the only appropriate mixing technique.
If you accidentally shook the vial and see foam, let it sit undisturbed in the refrigerator for 30-60 minutes. The foam should dissipate. Some potency loss may have occurred, but a briefly shaken vial is not necessarily ruined. The key is not making it a habit.
Mistake 2: Using sterile water instead of bacteriostatic water
Sterile water for injection (SWFI) is fine for single-use vials that will be used immediately. But for multi-dose vials that you will draw from over several weeks, SWFI provides no protection against bacterial contamination. Every needle insertion through the stopper introduces a contamination risk. Without the benzyl alcohol preservative in BAC water, bacteria can colonize the vial within days.
Always use bacteriostatic water for multi-dose reconstitution. It is widely available and inexpensive. This is one of the cheapest and most effective investments in protocol safety you can make.
Mistake 3: Wrong storage temperature
Freezing reconstituted tirzepatide is a common mistake, especially among people who know that the lyophilized powder should be frozen. The rules change after reconstitution. Reconstituted solution goes in the refrigerator, never the freezer. Leaving reconstituted solution at room temperature for extended periods is the other common temperature error, particularly when people forget to put the vial back after drawing a dose.
Mistake 4: Injecting water directly onto the powder
Forcing water onto the lyophilized cake at high velocity can fragment the peptide cake and create localized high-concentration areas that dissolve unevenly. It can also generate foam. Always aim the needle at the inside wall of the vial and let the water run down gently to meet the powder at the bottom.
Mistake 5: Over-concentrating the solution
Using too little BAC water creates a very concentrated solution where tiny measurement errors in syringe volume translate to large dosing errors. If your solution is at 20mg/mL and your target dose is 2.5mg, you need to draw exactly 12.5 units, which is a very small and hard-to-read amount on most insulin syringes. A small misread of 2-3 units means a 16-24% dosing error. At 5mg/mL, the same dose requires 50 units, and a 2-3 unit misread is only a 4-6% error.
Mistake 6: Poor sterile technique
Failing to wipe vial stoppers with alcohol, reusing syringes, touching needle tips, or working on a dirty surface all introduce contamination risk. These shortcuts seem harmless in the moment but can lead to bacterial growth in the vial that causes injection site infections or worse. Every reconstitution and every dose withdrawal should follow clean technique as described in the reconstitution section above.
Mistake 7: Not tracking reconstitution dates
Without a date label on the vial, you have no reliable way to track how long the reconstituted solution has been in the fridge. Memory is unreliable, especially when you are managing multiple vials or running multi-month protocols. Label every vial with the reconstitution date and the concentration. Some researchers also add the discard date (reconstitution date plus 28 days) for easy reference.
Lyophilized tirzepatide and the compounding landscape
The regulatory environment around compounded tirzepatide has changed significantly. Understanding the current landscape helps you make informed sourcing decisions.
What compounding pharmacies do
Compounding pharmacies prepare custom medications tailored to individual patient needs. For tirzepatide, this historically meant preparing lyophilized vials or pre-mixed solutions at various concentrations, sometimes with added compounds like vitamin B12, glycine, or niacinamide. Compounded versions offered lower prices compared to brand-name Mounjaro, making tirzepatide accessible to more researchers.
Compounding pharmacies like Empower Pharmacy, ProRx, Southend Pharmacy, and others built significant businesses around compounded tirzepatide during the FDA drug shortage period. These pharmacies operated legally under Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act, which permits compounding of drugs on the FDA shortage list.
The regulatory shift
The FDA removed tirzepatide from the drug shortage list in October 2024, which impacts the legal basis for compounding. The full implications of this change are still unfolding, with legal challenges and regulatory clarifications ongoing. This does not affect research-use-only (RUO) lyophilized tirzepatide, which operates under different regulatory frameworks, but it has affected the availability and pricing of compounded prescription tirzepatide.
For the most current sourcing options, the affordable tirzepatide guide and compounded tirzepatide pricing overview provide updated information on available pathways.
Research-grade versus pharmaceutical-grade
Research-grade lyophilized tirzepatide is manufactured and sold for laboratory research purposes. It is typically 98-99%+ pure and comes with COAs, but it is not produced under the same Good Manufacturing Practice (GMP) standards required for pharmaceutical products intended for human use. The research vs pharmaceutical peptides comparison explains these differences in detail.
Pharmaceutical-grade tirzepatide, whether brand-name Mounjaro or products from licensed compounding pharmacies, is produced under GMP standards with extensive quality control, sterility testing, and regulatory oversight. The tradeoff is significantly higher cost.
The grey market peptide landscape sits between these two categories, and understanding where your specific source falls on this spectrum is important for making informed decisions about what you are putting in your body.
Comparing tirzepatide to other lyophilized peptides
The reconstitution and handling principles for lyophilized tirzepatide are broadly similar to other peptides, but there are important differences worth noting.
Tirzepatide versus semaglutide
Both are GLP-1 receptor agonists available in lyophilized form, though they differ in key ways. Semaglutide acts on GLP-1 receptors only, while tirzepatide acts on both GLP-1 and GIP receptors. In terms of handling, the reconstitution process is identical: BAC water, gentle mixing, refrigerated storage. The semaglutide reconstitution process and reconstitution chart mirror the tirzepatide approach.
The main practical difference is in dosing. Semaglutide doses are measured in milligrams at much lower absolute amounts (0.25mg to 2.4mg weekly) compared to tirzepatide (2.5mg to 15mg weekly). This means semaglutide solutions are typically reconstituted to lower concentrations to maintain measurable doses. The side effect profiles also differ, and some researchers switch between the two based on response and tolerance.
For those comparing all three major GLP-1 options, the semaglutide vs tirzepatide vs retatrutide comparison covers efficacy, dosing, and handling differences comprehensively.
Tirzepatide versus retatrutide
Retatrutide is a triple agonist (GIP, GLP-1, and glucagon receptors) that is also available in lyophilized form. The reconstitution process for retatrutide follows the same principles as tirzepatide. Storage requirements are identical. The primary difference is that retatrutide is a newer compound with less long-term stability data available, so following the conservative 28-day post-reconstitution window is especially important.
Tirzepatide versus non-GLP-1 peptides
Other common lyophilized peptides like BPC-157, TB-500, and CJC-1295 follow similar reconstitution procedures but have different stability profiles and sensitivity to handling errors. GLP-1 peptides like tirzepatide are generally more stable than smaller signaling peptides, which is good news for handling. The general peptide reconstitution and post-reconstitution storage principles apply across all lyophilized peptides, with tirzepatide being one of the more forgiving compounds in terms of stability.
Supplements and additives in compounded formulations
Lyophilized tirzepatide from compounding pharmacies sometimes includes additional compounds blended into the formulation. Understanding what these additives do and why they are included helps you evaluate whether a compounded blend is right for your protocol.
Vitamin B12 (cyanocobalamin and methylcobalamin)
Tirzepatide with B12 is one of the most common compounded blends. The rationale is that GLP-1 agonists can affect B12 absorption over time, and supplementing directly in the injection ensures adequate levels. Methylcobalamin is the bioactive form preferred by some researchers over cyanocobalamin. The addition of B12 can change the color of the reconstituted solution to pink or red, which is normal and does not indicate degradation.
Glycine
Glycine is an amino acid added as a stabilizer and to potentially reduce injection site discomfort. Some compounding pharmacies include glycine to improve the pH profile of the solution and enhance peptide stability after reconstitution. The tirzepatide/glycine/B12 triple compound is a popular formulation that combines all three.
Niacinamide (vitamin B3)
Niacinamide is sometimes added for its potential synergistic effects on metabolic health. B3 plays roles in cellular energy production and has been studied for its effects on insulin sensitivity, making it a logical addition to a GLP-1 agonist formulation.
Vitamin B6
Tirzepatide with B6 is less common but included by some compounders for its role in amino acid metabolism and neurotransmitter synthesis. B6 may also help with nausea management, which is a common side effect during the initial weeks of tirzepatide use.
When these additives are present in a lyophilized formulation, the reconstitution process remains the same. The powder may appear slightly different in color or texture compared to pure tirzepatide, and the reconstituted solution may have a faint color. These differences are expected and do not affect the reconstitution technique.
For researchers who prefer their tirzepatide without additives, plain lyophilized tirzepatide is widely available. The supplements to take with tirzepatide guide covers options for oral supplementation alongside a plain tirzepatide protocol, giving you the flexibility to choose your own supportive compounds.
What to expect when starting a lyophilized tirzepatide protocol
Understanding the timeline of effects helps set realistic expectations and prevents premature protocol abandonment. The lyophilized form does not change anything about how tirzepatide works in the body. Once reconstituted and injected, it behaves identically to any other form. The differences are purely in handling, storage, and preparation.
That said, there is a psychological dimension worth acknowledging. Researchers who reconstitute their own vials tend to be more engaged with their protocol. They know their exact concentration, they track their vials, and they develop habits around proper handling. This engagement correlates with better adherence and better outcomes, not because the powder form is somehow more effective, but because the ritual of preparation creates intentionality around the process.
First four weeks (2.5mg dose)
The starting dose is primarily about tolerability. Many researchers report subtle appetite reduction during the first week or two, but dramatic weight loss at 2.5mg is uncommon. The more common experience is mild gastrointestinal adjustment, including occasional nausea, mild constipation, or reduced appetite that comes and goes. Results typically become more noticeable as the dose increases over subsequent weeks.
Some people feel fatigue during the first week or two. Others report increased energy. Individual response varies significantly, and both are normal. If side effects like headaches, constipation, or diarrhea occur, they typically resolve within the first few weeks as the body adapts.
Weeks five through twelve (5mg-7.5mg)
This is where most researchers begin seeing meaningful results. Appetite suppression becomes more pronounced and consistent. Weight loss, for those using tirzepatide for that purpose, typically begins in earnest. The tirzepatide weight loss timeline shows that average results accelerate during this phase.
Side effects may temporarily increase with each dose escalation and then subside as adaptation occurs. Adjusting diet, eating the right foods, and following a structured meal plan can significantly reduce gastrointestinal discomfort during these transitions.
Beyond twelve weeks
Higher doses (10mg-15mg) produce the most significant metabolic effects. Before and after results from clinical studies and individual experiences show continued weight loss, improved metabolic markers, and stabilization of appetite at maintenance doses. Metabolic rate changes become measurable, and many researchers find a dose level where they achieve their goals without progressing to the maximum.
If results plateau or reverse, the tirzepatide not working anymore troubleshooting guide and weight loss stall analysis cover the most common causes and solutions.
Managing side effects during the protocol
The most commonly reported side effects of tirzepatide are gastrointestinal in nature. Nausea affects a significant percentage of users, particularly during the first few days after each dose increase. This is a direct consequence of the GLP-1 receptor activation slowing gastric motility and altering hunger signaling in the brain. For most people, nausea resolves within the first week at each dose level as the body adapts.
Body aches and muscle pain are reported less frequently but can be concerning when they occur. These symptoms are typically mild and transient, resolving within a few days. More persistent musculoskeletal discomfort may indicate rapid weight loss affecting muscle mass, which is why adequate protein intake and resistance training are commonly recommended alongside GLP-1 protocols.
Anxiety and sleep disturbances are less common but documented. Joint pain has also been reported by some users, particularly at higher doses. The side effect comparison between semaglutide and tirzepatide shows that while both share many gastrointestinal effects, the intensity and duration can differ due to the additional GIP receptor activation in tirzepatide.
Dietary modifications make a significant difference in managing gastrointestinal side effects. Eating smaller, more frequent meals, avoiding high-fat and greasy foods, staying well hydrated, and avoiding known trigger foods can substantially reduce nausea and digestive discomfort. The tirzepatide meal plan provides structured guidance for eating patterns that complement the protocol.
Supportive supplements can also help. Electrolytes combat dehydration from reduced food intake. Fiber supplements address constipation. Ginger and peppermint tea help with nausea. Protein supplementation protects muscle mass during weight loss. Building a supportive supplement regimen alongside your lyophilized tirzepatide protocol maximizes results while minimizing discomfort.
Traveling with lyophilized tirzepatide
One of the practical advantages of the lyophilized form is easier travel logistics compared to liquid formulations.
Unreconstituted lyophilized vials do not require cold chain storage, meaning they can travel at room temperature without concern. This eliminates the need for cooler bags, ice packs, and temperature monitoring during flights or road trips. Pack the sealed vials in their original packaging, keep them away from extreme heat, and they will be fine.
If you are traveling with already-reconstituted tirzepatide, the same cold chain requirements apply as with any liquid peptide. An insulated bag with a cold pack maintains appropriate temperatures for 12-24 hours. The traveling with GLP-1 peptides guide covers TSA guidelines, international travel considerations, and packing strategies in detail.
A practical tip for longer trips: bring lyophilized vials and a small vial of BAC water rather than pre-reconstituted solution. Reconstitute at your destination when you have access to refrigeration. This approach eliminates cold chain concerns during transit entirely and ensures maximum potency at the point of use.
Long-term considerations for lyophilized tirzepatide users
Running a lyophilized tirzepatide protocol for months or longer introduces considerations that short-term users may never encounter. Planning for these in advance prevents disruptions and maintains consistency.
Building a supply strategy
The lyophilized form enables a purchasing strategy that liquid formulations do not: buying ahead. Since unreconstituted powder stores for months to years, researchers can purchase multiple vials during favorable pricing periods and store them long-term. This protects against supply chain disruptions, price increases, and availability changes. Keep unreconstituted vials in the freezer at -20 degrees Celsius, clearly labeled with purchase dates, and rotate stock on a first-in-first-out basis.
A practical approach is to maintain a rolling supply of 8-12 weeks ahead of your current position in the protocol. This provides enough buffer to handle unexpected delays without creating excessive stockpiles that risk expiration. The peptide cost calculator helps you budget for bulk purchases based on your dose escalation schedule.
Transitioning between tirzepatide and other GLP-1 peptides
Some researchers transition between tirzepatide and other GLP-1 compounds during their protocol, either due to supply availability, side effect management, or protocol experimentation. The semaglutide to tirzepatide conversion chart and tirzepatide to semaglutide conversion chart provide dose equivalency guidance for smooth transitions.
When transitioning, keep in mind that the reconstitution process is identical regardless of which GLP-1 peptide you are using. The skills you develop handling lyophilized tirzepatide transfer directly to handling lyophilized semaglutide, retatrutide, or any other lyophilized peptide. The general bacteriostatic water mixing guide covers the universal principles that apply across all reconstitutable peptides.
Combining with other compounds
Some researchers stack tirzepatide with other peptides or compounds for synergistic effects. AOD-9604 with tirzepatide is a common combination targeting enhanced fat metabolism. Phentermine combined with tirzepatide has also been explored, though this combination requires careful consideration of cardiovascular effects. The peptide stacking calculator helps evaluate compatibility and dosing when running multiple compounds simultaneously.
When stacking multiple lyophilized peptides, each compound should be reconstituted and stored in its own separate vial. Never mix different peptides together in the same vial unless specifically directed by a compounding pharmacy formulation. Cross-contamination between peptides can affect stability, potency, and safety profiles.
Knowing when to maintain, reduce, or stop
Tirzepatide protocols are not meant to run indefinitely at maximum dose in most cases. Many researchers find an effective maintenance dose below the maximum 15mg and sustain results at that level long-term. Others cycle on and off, using lyophilized tirzepatide for a defined period and then transitioning to maintenance strategies that do not involve the peptide. The guide to weaning off tirzepatide covers tapering strategies that minimize rebound effects, and the maintaining weight loss after tirzepatide resource provides long-term strategies for sustaining results.
The beauty of the lyophilized form in this context is flexibility. If you decide to pause your protocol, any unreconstituted vials in your freezer will be waiting for you, fully potent and ready to mix whenever you decide to resume. This is not the case with pre-mixed liquid that expires within weeks regardless of whether you are using it.
Frequently asked questions
Is lyophilized tirzepatide the same as regular tirzepatide?
Yes. Lyophilized tirzepatide is the same molecule as any other form of tirzepatide. The term lyophilized simply describes the physical form, which is a freeze-dried powder. Once reconstituted with bacteriostatic water, it produces the same solution and works identically to pre-mixed liquid formulations. The only difference is that the powder form is more stable for storage and shipping.
How long does lyophilized tirzepatide last before reconstitution?
When stored at -20 degrees Celsius in a sealed vial, lyophilized tirzepatide can maintain potency for 1-3 years or longer. At refrigerator temperature, expect 6-12 months of reliable stability. At room temperature, 3-6 months. Most suppliers assign a 12-month expiration date, but actual stability under proper conditions typically exceeds this. Check the detailed shelf life guide for specific storage scenario timelines.
Can I freeze reconstituted tirzepatide?
No. Never freeze reconstituted tirzepatide. The uncontrolled ice crystal formation in a standard freezer damages the peptide structure and can significantly reduce or eliminate its biological activity. Only the lyophilized powder should be frozen. Once reconstituted, store exclusively in the refrigerator at 2-8 degrees Celsius. Understanding the refrigeration requirements prevents this common mistake.
What if my lyophilized tirzepatide looks yellow or discolored?
White to off-white is normal. Any significant yellowing, brown spots, or unusual coloration suggests degradation, improper lyophilization, or contamination. Do not reconstitute or use discolored product. Contact the supplier for a replacement. The exception is formulations containing B12, which may have a pink or reddish tint that is completely normal.
How much bacteriostatic water do I add to a 10mg vial?
The standard recommendation for a 10mg vial is 2mL of bacteriostatic water, producing a concentration of 5mg/mL. However, you can adjust this based on your preferred concentration. Adding 1mL creates a 10mg/mL concentration (more concentrated, smaller injection volume). Adding 2mL creates 5mg/mL (less concentrated, easier to measure small doses). The complete mixing guide for 10mg vials covers all common ratios.
Can I use the same reconstitution process for semaglutide?
Yes. The reconstitution technique is identical for all lyophilized peptides: add BAC water slowly along the vial wall, roll gently (never shake), and refrigerate immediately. The only differences are in the amount of water added and the resulting concentration, since semaglutide doses are much smaller in milligrams. See the semaglutide reconstitution guide for specific ratios.
Do I need a prescription for lyophilized tirzepatide?
Research-use-only (RUO) lyophilized tirzepatide is available without a prescription from research peptide suppliers. Pharmaceutical-grade tirzepatide intended for human use requires a prescription. The qualification requirements for GLP-1 medications vary by provider, and some telemedicine platforms offer consultations that streamline the process. The BMI requirements for GLP-1 prescriptions provide a starting point for understanding eligibility.
What happens if I accidentally leave my reconstituted vial out overnight?
A single overnight excursion at room temperature (8-12 hours) will reduce the remaining shelf life of the solution but is unlikely to render it completely useless. Return it to the refrigerator immediately and plan to use the remaining contents sooner than originally planned. If the solution appears cloudy or discolored after the excursion, discard it. For extended exposures, the out-of-fridge duration guide provides more specific guidance.
External resources
For researchers serious about optimizing their tirzepatide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, dosing calculators, and a community of thousands who have navigated these exact questions. Whether you are reconstituting your first vial or refining a protocol you have been running for months, SeekPeptides members get access to the tools and support that turn good protocols into great ones.
In case I do not see you, good afternoon, good evening, and good night. May your lyophilized powder stay potent, your reconstitution stay sterile, and your protocols stay consistent.