Feb 19, 2026

Beneath your tongue sits one of the most efficient drug absorption surfaces in the entire human body. A thin layer of highly vascularized tissue, rich with capillaries, capable of shuttling compounds directly into systemic circulation without the brutal gauntlet of the digestive tract. This is not a new discovery. Physicians have used sublingual delivery for nitroglycerin since the 1800s. But applying this route to a 4,114-dalton peptide like semaglutide? That is a much newer conversation, and it is changing how people think about GLP-1 receptor agonists for weight management.
Semaglutide sublingual refers to placing a liquid formulation of semaglutide under the tongue and holding it there while the medication absorbs through the oral mucosa. No needles. No swallowing a tablet with a specialized absorption enhancer. Just a measured dose held in place for a few minutes, absorbed directly into the bloodstream through tissue that sits millimeters from major blood vessels.
The appeal is obvious. Weekly injections work, but they come with needle anxiety, injection site reactions, and the kind of medical ritual that makes some people avoid treatment altogether. Oral semaglutide tablets addressed part of this problem, but they carry their own baggage: strict fasting requirements, a narrow absorption window, and bioavailability that hovers around 1% under ideal conditions. Sublingual delivery promises something different. It promises better absorption, fewer gastrointestinal side effects, and a daily routine that does not revolve around an empty stomach and a 30-minute waiting period. Whether it delivers on those promises depends on the science, the formulation, and the details most guides skip entirely. This is the guide that does not skip them.
What sublingual semaglutide actually is
Sublingual semaglutide is a liquid formulation of the GLP-1 receptor agonist semaglutide designed for absorption through the tissue beneath the tongue. The word sublingual comes from Latin: sub (under) and lingua (tongue). Unlike injectable semaglutide delivered subcutaneously, or oral tablets swallowed and absorbed through the gastrointestinal tract, sublingual formulations rely on the mucosal membrane to transfer the active compound directly into the bloodstream.
This is not the same as oral semaglutide.
That distinction matters. Oral semaglutide in tablet form (brand name Rybelsus) uses a compound called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) to protect the peptide from stomach acid and enhance absorption through the gastric lining. Even with this enhancer, bioavailability sits between 0.4% and 1%. That means for every 14mg tablet swallowed, the body actually uses roughly 0.06 to 0.14mg. The rest gets destroyed by digestive enzymes, degraded by stomach acid, or eliminated through first-pass hepatic metabolism.
Sublingual delivery sidesteps this entire process. The medication never enters the stomach. It never encounters digestive enzymes. It never passes through the liver before reaching systemic circulation. Instead, it crosses the thin sublingual epithelium, enters the capillary network beneath the tongue, and moves into the bloodstream directly. In preclinical studies, this route achieved an area under the curve (AUC) of 82.53 ng*h/ml compared to just 15.08 ng*h/ml for the oral route at equivalent doses. That is a roughly 5.5-fold improvement in drug exposure.
Currently, there is no FDA-approved sublingual semaglutide product on the market. The sublingual formulations available come from compounding pharmacies and pharmaceutical companies in various stages of development. This matters for understanding both the opportunity and the limitations of this delivery route.
How sublingual absorption works
Understanding why sublingual delivery offers advantages for semaglutide requires a basic grasp of what happens beneath the tongue at the tissue level. The mechanisms are straightforward, but the details separate effective sublingual use from wasted medication pooling in saliva.
The oral mucosa pathway
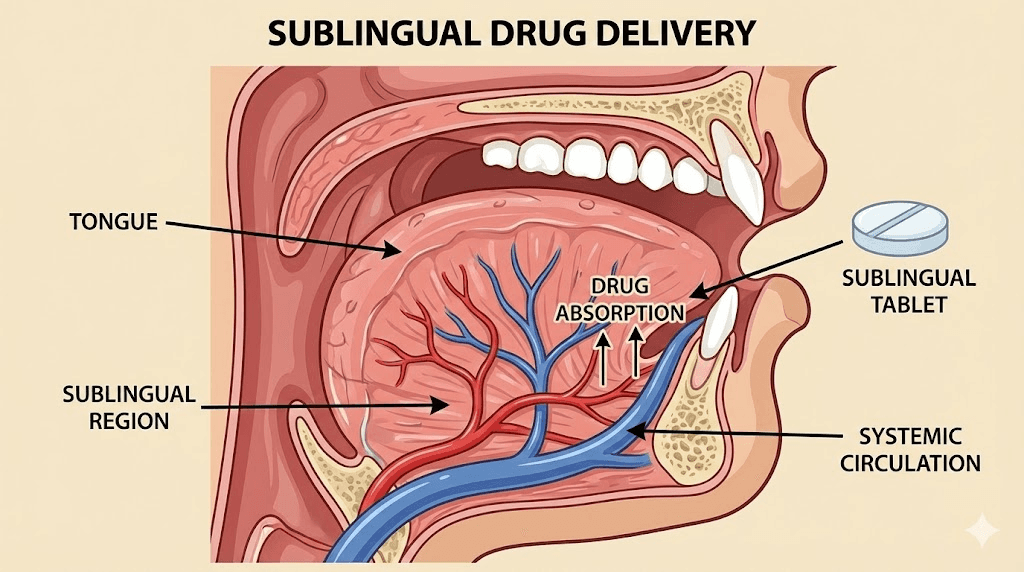
The floor of the mouth and the underside of the tongue are lined with non-keratinized stratified squamous epithelium. Unlike the tough, keratinized tissue on the roof of the mouth or the gums, this sublingual tissue is thin, highly permeable, and sits directly above a dense network of blood vessels. The lingual vein, the sublingual vein, and their tributaries run just beneath this membrane.
When a compound is placed under the tongue, it begins diffusing across this epithelium through the intercellular route, passing between cells rather than through them. Lipid-organized layers in the superficial epithelium represent the primary permeability barrier. Once a molecule crosses this barrier, it enters the capillary bed and drains into the systemic venous circulation, bypassing the portal vein entirely.
This is the critical difference.
Bypassing first-pass metabolism
When you swallow a medication, it travels through the stomach, into the small intestine, and absorbs through the intestinal wall into the portal vein. The portal vein carries everything to the liver first. The liver then metabolizes a significant portion of the drug before it ever reaches the rest of the body. For peptides, this first-pass effect is devastating. Liver enzymes, proteases, and clearance mechanisms can destroy the vast majority of an oral peptide dose before it does anything useful.
Sublingual absorption avoids this entirely. Blood from the sublingual capillaries drains into the internal jugular vein, which feeds directly into the superior vena cava and then the heart. No liver detour. No first-pass destruction. The full absorbed dose reaches systemic circulation intact. For a peptide like semaglutide, which is already challenging to deliver orally, this bypass mechanism is what makes sublingual delivery theoretically superior to standard oral peptide administration.
Why the sublingual route matters for peptides
Peptides are notoriously difficult to deliver orally. They are large molecules. Semaglutide weighs 4,114 daltons. For reference, most drugs that absorb well orally weigh under 500 daltons. The peptide bond structure is susceptible to enzymatic degradation throughout the gastrointestinal tract. Stomach acid, pepsin, trypsin, chymotrypsin, and dozens of other proteases all work to break peptides apart before they can absorb.
The sublingual mucosa has far fewer proteolytic enzymes. The contact time with destructive environments is minimal. And the absorption surface, while smaller than the intestine, offers direct vascular access that compensates for the reduced area. Research on sublingual peptide delivery has shown that smaller peptides (under 1,000 daltons) can cross the buccal mucosa without assistance. Larger peptides like semaglutide typically need permeation enhancers or specialized formulations, but the absorption advantage over oral delivery remains significant.
Sublingual semaglutide vs injectable semaglutide
The weekly subcutaneous injection remains the gold standard for semaglutide delivery. It is the most studied route, the most prescribed, and the one with the strongest clinical evidence behind it. But that does not mean it is the only route worth considering. Understanding how sublingual compares to injectable helps clarify where each option fits.
Bioavailability comparison
Subcutaneous injection delivers semaglutide with near-complete bioavailability. Estimates range from 89% to 100% of the injected dose reaching systemic circulation. This is the benchmark against which every other delivery route is measured.
Sublingual bioavailability is lower but meaningfully higher than oral tablets. Some compounding pharmacy sources cite bioavailability potentially reaching 40% for sublingual semaglutide, though this figure has not been confirmed in large human studies. The preclinical rat study comparing routes found the sublingual AUC was roughly 5.5 times higher than oral, suggesting substantially better absorption. However, rats and humans have different oral mucosa characteristics, so these numbers need clinical validation.
The practical implication: sublingual doses need to be higher than injectable doses to achieve equivalent blood levels, but lower than what oral tablets require. A 2.4mg weekly injection might translate to daily sublingual doses in the 1-3mg range, depending on the formulation and individual absorption characteristics. This is why working with a healthcare provider who understands semaglutide dosing across delivery routes is essential.
Onset of action
Sublingual delivery achieves measurable plasma concentrations faster than oral tablets. The medication does not need to survive stomach transit, wait for intestinal absorption, or pass through the liver. From under the tongue, semaglutide can begin entering the bloodstream within minutes.
That said, semaglutide is not a fast-acting medication regardless of route. Its mechanism of action involves sustained GLP-1 receptor activation over time. Appetite suppression and metabolic effects develop gradually over days and weeks, not hours. A faster initial absorption does not necessarily mean faster weight loss results. It means more consistent drug levels and potentially fewer missed absorption windows.
For injectable semaglutide, peak plasma concentration occurs 1-3 days after injection. For sublingual formulations taken daily, steady-state levels build over the first week of consistent dosing. Clinical effects typically become noticeable within 2-4 weeks regardless of delivery method.
Side effect profiles
One of the most frequently cited advantages of sublingual semaglutide is a potentially reduced gastrointestinal side effect burden. This makes physiological sense. When semaglutide is swallowed (either as a tablet or inadvertently with sublingual drops), it contacts the GI tract directly, which can trigger local nausea, vomiting, and digestive discomfort. When absorbed sublingually, the medication bypasses the gut entirely and exerts its effects through systemic GLP-1 receptor activation instead.
The systemic effects of semaglutide, including appetite suppression, delayed gastric emptying, and fatigue, will occur regardless of delivery route because they result from the drug reaching GLP-1 receptors throughout the body. But the intensity and frequency of direct GI irritation may be lower with sublingual use.
Clinical data supporting this claim remains limited to anecdotal reports and small clinical observations rather than large controlled trials. Users considering sublingual semaglutide for its gentler side effect profile should understand that nausea and GI effects can still occur through the systemic mechanism, even if local GI irritation is reduced.
Convenience and adherence
Injectable semaglutide requires a once-weekly subcutaneous injection. Some people find this convenient. Others dread it. Needle anxiety is a real barrier to treatment adherence, and it affects a larger population than most providers realize.
Sublingual semaglutide requires daily administration but eliminates needles entirely. No syringes, no injection technique to learn, no injection site rotation, no sharps disposal. For some people, a quick daily drop under the tongue is far easier to maintain than a weekly injection ritual.
The tradeoff is frequency. Daily dosing requires daily remembering. Weekly injections can be tied to a specific day and time. The better option depends entirely on individual preference, lifestyle, and which routine a person is more likely to maintain consistently. Consistency matters more than route when it comes to semaglutide results.
Sublingual semaglutide vs oral tablet semaglutide
If you are comparing sublingual semaglutide to the FDA-approved oral tablet (Rybelsus or the newer Wegovy oral formulation), the differences center on three main areas: absorption efficiency, dosing practicality, and the SNAC enhancer system.
Absorption differences
Oral semaglutide tablets rely on SNAC to create a localized pH change in the stomach lining that temporarily allows semaglutide to cross the gastric epithelium. This is an ingenious pharmaceutical solution, but it is inherently inefficient. Bioavailability hovers around 0.4-1%. That means a 14mg oral tablet delivers roughly the same systemic exposure as a 1mg injection. The other 13mg-plus gets destroyed.
Sublingual delivery offers meaningfully better absorption because the mechanism does not depend on surviving stomach conditions. The preclinical data showing a 5.5-fold AUC improvement over oral administration suggests that sublingual formulations could achieve therapeutic levels at much lower total doses. This has implications for both efficacy and cost, since less raw semaglutide is needed per dose to achieve the same blood levels.
For people who have tried oral semaglutide without satisfactory results, sublingual delivery may provide better absorption and more consistent drug exposure. Variable gastric absorption is one of the reasons some people respond poorly to oral tablets, and the sublingual route removes that variable entirely.
Dosing flexibility
Oral semaglutide tablets come in fixed doses: 3mg, 7mg, and 14mg. The titration schedule is set. You start at 3mg daily for 30 days, move to 7mg for 30 days, then reach the maintenance dose of 14mg. There is limited room for fine-tuning between those steps.
Sublingual semaglutide, particularly from compounding pharmacies, offers much more granular dosing control. Liquid formulations allow adjustment in 0.1mg or even 0.05mg increments. This means slower titration for people sensitive to side effects, faster escalation for those who tolerate it well, and precise dose optimization that fixed-dose tablets cannot match.
This flexibility is particularly valuable during the critical early weeks when appetite suppression is building and GI side effects are most common. Being able to increase by 0.25mg instead of jumping from 3mg to 7mg can make the difference between tolerating treatment and abandoning it.
The SNAC absorption enhancer consideration
SNAC is generally well tolerated, but it adds a layer of complexity to oral semaglutide dosing. The tablet must be taken on a completely empty stomach with no more than 4 ounces of plain water. No food, no other medications, no supplements for at least 30 minutes afterward. Some providers recommend waiting 60 minutes for optimal absorption.
These restrictions exist because food, other liquids, and medications all interfere with the SNAC-mediated absorption mechanism. Even a small snack before the waiting period ends can reduce absorption dramatically. This makes oral tablets impractical for people with certain morning routines, medication schedules, or lifestyle patterns.
Sublingual semaglutide has its own timing requirements (empty stomach, hold for several minutes, avoid eating/drinking afterward), but these are generally less restrictive than the oral tablet protocol. The hold time is shorter, the fasting window is less rigid, and there is no SNAC interaction to worry about.
How to take sublingual semaglutide correctly
Proper administration technique is the single biggest variable determining whether sublingual semaglutide actually works. Do it wrong and you are essentially swallowing expensive liquid that will get destroyed in your stomach. Do it right and you maximize absorption through the mucosal pathway.
Step-by-step administration
The process itself is simple. Executing it with the discipline required for consistent absorption takes attention to detail.
Step 1: Gently roll the bottle between your hands before each use. Some formulations contain suspensions that settle between doses. Do not shake vigorously, as this can create air bubbles that affect measurement accuracy.
Step 2: Draw the prescribed amount into the provided oral syringe. Check the measurement carefully. Dosing errors are the most common mistake with liquid formulations.
Step 3: Lift your tongue and place the liquid into the sublingual space, the pocket beneath the tongue where the tissue is thinnest and most vascularized. Aim for the area just behind the lower front teeth.
Step 4: Close your mouth and hold the liquid under your tongue. Do not swallow. Do not talk. Do not move the liquid around with your tongue. Keep it pooled in the sublingual space where absorption occurs.
Step 5: Hold for the prescribed duration. This is where most people fail, and the next section explains why hold time matters so much.
Step 6: After the hold period, you may swallow any remaining liquid. Some absorption continues through buccal tissue (the inner cheek) even after you resume normal mouth activity.
Timing and empty stomach requirements
Sublingual semaglutide should be taken on an empty stomach. Food in the GI tract does not directly affect sublingual absorption, but two related factors make fasting important.
First, eating stimulates saliva production. More saliva means faster dilution and swallowing of the sublingual dose, reducing contact time with the absorptive tissue. Second, some of the dose will inevitably be swallowed, and an empty stomach allows whatever small amount of GI absorption occurs to happen more efficiently.
The best time to take sublingual semaglutide is first thing in the morning, before eating or drinking anything. This ensures minimal saliva production, no food interference, and a consistent daily routine. After taking the dose, wait at least 15-30 minutes before eating, drinking, or brushing your teeth.
Hold time matters
This is the most critical variable. And it is the one most people underestimate.
The minimum recommended hold time is 30 seconds to 2 minutes depending on the formulation and provider guidance. But minimum is not optimal. Research on sublingual drug delivery consistently shows that longer contact time with the sublingual mucosa increases absorption. The Professional Compounding Centers of America (PCCA) recommends 5-15 minutes of sublingual contact for optimal peptide absorption.
Think about what that means practically. Five to fifteen minutes with liquid pooled under your tongue, mouth closed, no talking, no swallowing. That is a significant commitment. But it is the difference between absorbing a meaningful dose and swallowing most of it into digestive destruction.
Practical tip: time your sublingual dose with a quiet activity. Reading, watching something on your phone, doing light stretching. Build the hold time into a morning routine so it does not feel like wasted time. Some people set a timer and use the period for meal planning or reviewing their daily schedule.
What to avoid before and after dosing
Before dosing, avoid anything that increases saliva production or alters the pH of the oral environment. This includes:
Food or beverages within 30 minutes prior
Brushing teeth or using mouthwash (the detergents and pH changes can affect mucosal permeability)
Chewing gum or mints
Smoking or vaping (irritates and changes blood flow to oral tissue)
After dosing, avoid eating, drinking, or consuming alcohol for at least 15-30 minutes. This allows any residual medication still in contact with oral tissue to complete absorption. Drinking water too soon dilutes the remaining dose and washes it into the stomach.
Sublingual semaglutide dosing protocols
Dosing sublingual semaglutide is not as straightforward as following an injectable or oral tablet titration schedule. The bioavailability differences mean direct milligram-to-milligram comparisons do not work. Every dosing protocol needs to account for the absorption characteristics of the specific sublingual formulation being used.
Starting doses and titration
Most compounding pharmacy protocols start sublingual semaglutide at 0.5mg to 1mg daily. This is intentionally conservative. Even though sublingual bioavailability is lower than injectable, starting low allows the body to acclimate to GLP-1 receptor activation and minimizes the nausea that accompanies initial dosing.
A typical starting protocol looks like this:
Weeks 1-2: 0.5mg daily sublingual
Weeks 3-4: 1mg daily sublingual
Weeks 5-8: 1.5-2mg daily sublingual
Weeks 9-12: 2-3mg daily sublingual
Week 13 onward: 3mg daily sublingual (maintenance)
Individual response varies significantly. Some people achieve satisfactory appetite suppression and metabolic effects at 1-2mg daily. Others need the full 3mg maintenance dose. The advantage of liquid sublingual formulations is the ability to titrate in smaller increments than fixed-dose options allow.
Standard titration schedules
One commonly prescribed compounding protocol uses a 13-week escalation:
Fill 1 (Weeks 1-4): Semaglutide 1mg/ml suspension, 0.5ml under the tongue daily for 2 weeks, then increase to 1ml daily
Fill 2 (Weeks 5-8): Semaglutide 2mg/ml suspension, 1ml under the tongue daily
Fill 3 (Weeks 9-13): Semaglutide 3mg/ml suspension, 1ml under the tongue daily, then maintain
This schedule provides a steady escalation from 0.5mg to 3mg daily over approximately three months. Healthcare providers adjust based on tolerance, side effects, and therapeutic response. If nausea becomes significant at any step, staying at the current dose for an additional 1-2 weeks before escalating is a standard approach.
Use a semaglutide dosage calculator to cross-reference sublingual doses with equivalent injectable doses, keeping in mind that the conversion is approximate due to variable bioavailability.
Adjusting based on response
Sublingual semaglutide response monitoring follows the same principles as injectable semaglutide treatment. Key indicators include:
Appetite changes: Noticeable reduction in hunger and food fixation is the first sign of adequate dosing
Weight trajectory: Steady loss of 0.5-1% body weight per week suggests appropriate dosing
Side effect tolerance: Mild nausea is expected during titration. Severe or persistent nausea suggests the dose is too high or escalation was too rapid
Energy levels: If fatigue becomes significant, it may indicate the dose needs adjustment or nutritional support needs optimization
If you reach the maximum recommended sublingual dose without adequate response, the issue may not be the dose itself. Absorption technique (hold time, empty stomach compliance), formulation quality, and individual metabolic factors all play roles in sublingual semaglutide effectiveness.
Converting from injectable doses
Converting between injectable and sublingual semaglutide requires understanding that no exact conversion exists. The absorption profiles are fundamentally different: injectable provides near-complete bioavailability in a single weekly dose, while sublingual provides partial bioavailability across daily doses.
As a rough guide based on available compounding pharmacy protocols and estimated bioavailability ranges:
Injectable 0.25mg/week is roughly equivalent to sublingual 0.5-1mg daily
Injectable 0.5mg/week is roughly equivalent to sublingual 1-1.5mg daily
Injectable 1mg/week is roughly equivalent to sublingual 1.5-2mg daily
Injectable 2.4mg/week is roughly equivalent to sublingual 2.5-3mg daily
These are approximations. Individual absorption through the sublingual route varies more than injectable absorption, meaning some people may need higher or lower sublingual doses than these estimates suggest. Healthcare providers should monitor clinical response rather than rely solely on dose equivalency charts. For detailed unit-to-milligram conversions for injectable semaglutide, see our guides on semaglutide dosing in units and conversion charts.
Compounded sublingual semaglutide
Since no FDA-approved sublingual semaglutide product exists, the only current source is compounding pharmacies. Understanding how compounded sublingual semaglutide works, what the regulatory environment looks like, and how to evaluate quality is essential for anyone considering this route.
What compounding pharmacies offer
Compounding pharmacies create customized medication formulations based on individual prescriptions. For sublingual semaglutide, this typically means a liquid suspension or solution containing semaglutide at a specified concentration (commonly 1mg/ml, 2mg/ml, or 3mg/ml), along with excipients designed to enhance sublingual stability and absorption.
Some compounding formulations add complementary ingredients. Semaglutide with B12 is a common combination, as vitamin B12 deficiency can develop during GLP-1 therapy. Other formulations include glycine or methylcobalamin to support metabolic function during weight loss.
The quality of compounded sublingual semaglutide varies significantly between pharmacies. Not all compounding pharmacies have the equipment, expertise, or quality control systems to produce reliable peptide formulations. Major compounding pharmacies with established reputations and third-party testing tend to produce more consistent products than smaller operations.
Regulatory landscape
The regulatory environment for compounded semaglutide shifted dramatically in February 2025 when the FDA declared the semaglutide shortage resolved. During the shortage (2022-2025), compounding pharmacies operated under Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act, which allowed them to compound copies of commercially available drugs when those drugs were in shortage.
With the shortage declared over, the rules tightened. As of April 2025, compounding pharmacies can only produce semaglutide if a prescriber documents a clinical reason why the FDA-approved version does not meet a specific patient need. Legitimate reasons include:
Allergy to an inactive ingredient in the commercial product
Need for a different dosage form (such as sublingual instead of injectable)
Requirement for a dose not available in commercial products
Medical need for a formulation without a specific component
The sublingual delivery route itself may qualify as a legitimate clinical need since no FDA-approved sublingual semaglutide exists. Patients who cannot tolerate injections and do not respond adequately to oral tablets have a documented clinical reason for a compounded sublingual formulation. However, the regulatory interpretation varies, and this landscape continues to evolve.
Quality and safety considerations
The FDA has warned about quality concerns with some compounded GLP-1 products. Issues include inaccurate potency (the actual semaglutide content not matching the label), sterility problems (particularly for injectable compounds), and the use of salt forms (like semaglutide sodium) that have different pharmacological properties than the base compound used in FDA-approved products.
For sublingual formulations specifically, key quality factors include:
Potency verification: Third-party testing confirming the stated concentration matches the actual semaglutide content
Stability data: Evidence that the sublingual formulation maintains potency throughout its assigned shelf life
Proper excipients: Ingredients that support sublingual absorption without causing mucosal irritation
pH optimization: The formulation pH should be compatible with sublingual tissue (typically pH 5.5-7.0)
When evaluating a compounding pharmacy for sublingual semaglutide, look for PCAB accreditation (Pharmacy Compounding Accreditation Board), certificates of analysis for each batch, and transparent communication about their testing and quality control procedures. Check our guide on peptide safety considerations for additional quality evaluation criteria.
Side effects of sublingual semaglutide
Every route of semaglutide administration shares a core set of side effects related to the drug mechanism itself. GLP-1 receptor activation throughout the body produces predictable effects. The delivery route modifies some aspects of the side effect experience but does not eliminate the fundamental pharmacological response.
Common side effects
The most frequently reported side effects of semaglutide, regardless of delivery route, include:
Nausea: The most common side effect, occurring in 20-44% of users depending on dose and study. Usually most intense during the first 4-8 weeks and during dose escalation. Tends to diminish with continued use.
Decreased appetite: This is both a therapeutic effect and a side effect. Significant appetite reduction is the mechanism of weight loss, but it can lead to inadequate nutrition if not managed.
Diarrhea: Affects 15-30% of users. May be intermittent rather than constant.
Constipation: Affects 10-24% of users. Semaglutide slows gastric motility, which can reduce bowel movement frequency. See our guide on managing semaglutide constipation for specific strategies.
Headaches: Common during the initial weeks, usually resolving as the body adjusts.
Fatigue: Can occur due to reduced caloric intake, metabolic changes, or the drug itself. Our semaglutide fatigue guide covers management strategies in detail.
Oral-specific side effects
Sublingual administration introduces a few side effects not seen with injectable use:
Oral irritation: Some users report mild tingling, numbness, or irritation in the sublingual area during and immediately after dosing. This is usually mild and transient.
Taste disturbance: Sublingual formulations may have an unpleasant taste that lingers after dosing. The taste varies by formulation and excipients used.
Increased salivation: Paradoxically, placing something under the tongue can trigger increased saliva production, which can dilute the dose and reduce absorption if the liquid is swallowed prematurely.
These oral-specific effects are generally mild and improve with continued use as the oral tissue adapts to regular exposure. They are a reasonable tradeoff for people who prefer sublingual delivery over injection techniques.
Managing nausea during titration
Nausea management with sublingual semaglutide follows the same principles as other semaglutide formulations:
Slow titration: Increase doses gradually. If the standard 4-week titration intervals cause significant nausea, extend to 6-8 weeks between increases.
Eat smaller meals: Large meals overwhelm a stomach that is emptying more slowly. Following a semaglutide-optimized eating plan reduces nausea significantly.
Stay hydrated: Dehydration amplifies nausea. Aim for adequate water intake throughout the day.
Avoid trigger foods: High-fat, greasy, or very spicy foods commonly worsen GLP-1-related nausea.
Ginger and peppermint: Natural antiemetics that can reduce mild nausea without interfering with semaglutide activity.
If nausea becomes severe enough to affect daily function, reduce the sublingual dose by 0.25-0.5mg and hold at that level for 2-4 additional weeks before attempting to increase again. The liquid formulation makes this kind of precise dose adjustment straightforward.
When to contact a healthcare provider
Seek medical attention promptly for:
Severe or persistent abdominal pain (potential pancreatitis indicator)
Vision changes
Signs of allergic reaction (swelling, difficulty breathing, rash)
Persistent vomiting that prevents maintaining hydration
Signs of significant hair thinning (may indicate nutritional deficiency)
Rapid heart rate or palpitations
Severe mood changes
These symptoms, while uncommon, require professional evaluation regardless of the semaglutide delivery route being used.
Who should consider sublingual semaglutide
Sublingual semaglutide is not the right choice for everyone. It occupies a specific niche between injectable and oral tablet formulations, and understanding who benefits most helps set appropriate expectations.
Needle-averse individuals
This is the most obvious candidate population. Needle phobia (trypanophobia) affects an estimated 20-25% of adults to some degree. For some, it is mild discomfort that they push through. For others, it is a genuine barrier that prevents them from starting or continuing injectable GLP-1 therapy.
Sublingual semaglutide eliminates needles entirely. No learning injection technique. No anxiety about weekly shots. No traveling with syringes. For needle-averse individuals, the daily sublingual routine may be dramatically more sustainable than weekly injections, even though the injection itself is quick and minimally painful.
People who struggle with oral tablets
Some individuals do not respond well to oral semaglutide tablets. The strict fasting requirements, the 30-60 minute waiting period, the variable absorption, or the GI side effects from the SNAC enhancer make the oral tablet impractical or ineffective for them.
Sublingual delivery offers a middle path. No needles, no SNAC, no tablet-specific fasting protocol. The hold time requirement is the main compliance factor, and many people find it more manageable than the oral tablet restrictions.
People with gastroparesis or other conditions that affect gastric motility may also absorb sublingual semaglutide more reliably than oral tablets, since the sublingual route does not depend on stomach function for absorption.
Travel and lifestyle considerations
Injectable semaglutide requires cold chain storage, syringes, alcohol swabs, and sharps containers. Traveling with injectable medications involves TSA considerations, hotel refrigeration, and maintaining proper temperature control throughout the journey.
Sublingual semaglutide, while still requiring careful storage and temperature management, eliminates the need for syringes, sharps disposal, and injection supplies. A small bottle of sublingual drops and an oral syringe take up minimal space and do not raise the security questions that injectable supplies sometimes do.
For frequent travelers, outdoor enthusiasts, or people with active lifestyles where injection routines are difficult to maintain, the simplicity of sublingual dosing offers a practical advantage. Still, proper storage remains important. Check our guide on semaglutide shelf life for specific storage requirements.
Storage and handling
Compounded sublingual semaglutide requires careful storage to maintain potency and safety. The specific requirements vary by formulation, but general principles apply across most compounded sublingual products.
Temperature requirements
Most compounded sublingual semaglutide should be stored in the refrigerator at 36-46 degrees Fahrenheit (2-8 degrees Celsius). Some formulations allow short-term room temperature storage (up to 28 days), but refrigeration is always preferred for maintaining maximum potency.
Do not freeze sublingual semaglutide. Freezing can damage the formulation structure, alter the concentration, and affect absorption characteristics. If your medication freezes accidentally, do not use it. Contact your pharmacy for a replacement.
Protect the bottle from direct light exposure. UV light can degrade semaglutide over time. Most compounding pharmacies dispense sublingual formulations in amber or opaque bottles for this reason. If yours came in a clear container, store it inside its outer packaging or in a dark area of the refrigerator.
Shelf life
Compounded medications typically have shorter shelf lives than commercially manufactured products because they lack the extensive stability testing and preservative systems of FDA-approved drugs. Most compounded sublingual semaglutide carries a beyond-use date of 30-90 days from compounding, depending on the pharmacy and formulation.
Follow the beyond-use date on your specific bottle. Using expired compounded semaglutide is not recommended. The potency may have degraded, leading to underdosing, and the preservative system may no longer prevent microbial contamination. Read more about expired semaglutide considerations for additional context.
Travel considerations
When traveling with sublingual semaglutide:
Use an insulated pouch with ice packs for trips exceeding a few hours
Request a mini-fridge at your hotel or accommodation
Keep the medication in your carry-on bag when flying (temperature fluctuations in cargo holds can be extreme)
Bring a copy of your prescription in case questions arise
Plan your supply carefully to avoid running out during extended trips
Our detailed guide to traveling with semaglutide covers airline regulations, storage solutions, and destination-specific considerations.
The current research landscape
Sublingual semaglutide sits at an interesting point in pharmaceutical development. The science supports the concept. The preclinical data is encouraging. But large-scale human clinical trials proving efficacy and establishing optimal dosing are still in progress or planned. Here is where things stand.
Preclinical studies
The most significant preclinical study published in early 2026 in a pharmaceutical research journal provided the first direct comparison of sublingual, oral, and injectable semaglutide pharmacokinetics in rats. Key findings included:
At 1mg/kg, sublingual achieved an AUC of 82.53 ng*h/ml versus 15.08 ng*h/ml for oral, a 5.5-fold improvement
Sublingual administration showed lower variability in plasma concentrations compared to oral dosing
The study validated the proof-of-concept for sublingual peptide delivery as a viable alternative to injection and oral routes
These results are promising but carry important caveats. Rat oral mucosa differs from human oral mucosa in thickness, permeability, and enzyme content. The 5.5-fold improvement seen in rats may not translate directly to humans. Clinical studies in human subjects are needed to establish real-world bioavailability and dose equivalency.
Companies developing sublingual formulations
Several pharmaceutical companies are actively working on sublingual GLP-1 formulations:
Aspire Biopharma announced in late 2025 that their scientists are in the final phases of developing a sublingual semaglutide formulation. The company plans to conduct a Phase 1 pharmacokinetic study in Q2-Q3 2026 with 6-8 volunteers to validate the formulation properties. If successful, this would be the first human clinical data specifically for sublingual semaglutide from a pharmaceutical development program.
Galmed Pharmaceuticals signed a binding term sheet in April 2025 for development of a proprietary Self-Emulsifying Drug Delivery System (SEDDS) designed for sublingual peptide absorption. This formulation technology creates microemulsions when placed under the tongue, enhancing the ability of large peptides like semaglutide to cross the mucosal barrier. Galmed stated the approach could potentially enhance bioavailability compared to existing oral forms while reducing the required daily dose.
Additional research into sublingual GLP-1 delivery includes nanoparticle formulations, lipid-based carriers, and novel permeation enhancers designed specifically for the sublingual environment. The sublingual route is attracting serious pharmaceutical investment because it could combine the convenience of oral dosing with meaningfully better absorption.
What clinical trials are coming
The Aspire Biopharma Phase 1 trial planned for mid-2026 will be the first formal clinical evaluation of sublingual semaglutide. Phase 1 studies focus on pharmacokinetics (how the drug is absorbed, distributed, metabolized, and excreted) rather than efficacy. This means it will establish what blood levels the sublingual formulation achieves in humans, but will not directly measure weight loss outcomes.
Assuming positive Phase 1 results, Phase 2 trials evaluating efficacy and dose-response would follow, likely in 2027. An FDA-approved sublingual semaglutide product is probably several years away from market, given the typical drug development timeline.
In the meantime, compounded sublingual semaglutide continues to fill the gap for patients and providers who want this delivery route now. The compounding approach provides real-world clinical experience that informs the formal development programs, even though compounded products lack the rigorous clinical trial data of FDA-approved formulations.
For the latest on peptide regulation developments and emerging delivery technologies, SeekPeptides tracks the research landscape and provides members with updated analysis as new studies publish.
Common mistakes with sublingual semaglutide
Sublingual delivery is deceptively simple. Place liquid under tongue. Hold. Done. But the details matter enormously, and getting them wrong means wasting medication and underperforming on results.
Mistake 1: Insufficient hold time. This is the number one error. Holding sublingual semaglutide for 30 seconds and swallowing is not sublingual administration. It is oral administration with extra steps. The mucosa needs contact time to absorb the peptide. Aim for 5-15 minutes. Anything less than 2 minutes is likely inadequate for meaningful sublingual absorption.
Mistake 2: Taking it with food in the stomach. While sublingual absorption does not depend on the GI tract, food stimulates saliva production and increases the likelihood of premature swallowing. Take sublingual semaglutide on an empty stomach, preferably first thing in the morning.
Mistake 3: Swallowing prematurely. The instinct to swallow is strong. Saliva builds up. The liquid feels uncomfortable. People swallow without thinking. Every swallow sends medication to the stomach instead of through the mucosa. If you cannot hold the full dose without swallowing, start with smaller volumes and work up to the prescribed amount as your technique improves.
Mistake 4: Incorrect placement. The liquid needs to go under the tongue, in the sublingual space where the tissue is thinnest and most vascularized. Placing it on top of the tongue, against the cheek, or in the general mouth area significantly reduces absorption efficiency. The sublingual fossa, the small pocket just behind the lower front teeth and under the tongue base, is the target zone.
Mistake 5: Brushing teeth immediately before dosing. Toothpaste contains surfactants that alter the oral mucosa environment. The mechanical action of brushing can also temporarily increase mucosal permeability in unpredictable ways. Wait at least 15-30 minutes after brushing before sublingual dosing, or brush after dosing instead.
Mistake 6: Inconsistent timing. Daily sublingual semaglutide works best at the same time every day. Inconsistent dosing leads to inconsistent blood levels, which reduces therapeutic effectiveness. Set a daily alarm or tie the dose to a fixed morning routine.
Mistake 7: Not tracking response. Without monitoring weight, appetite, side effects, and other clinical markers, there is no way to know if the current dose is optimal. GLP-1 tracking tools and journals help identify trends and inform dose adjustments. SeekPeptides members access progress tracking systems designed specifically for this purpose.
Mistake 8: Expecting identical results to injectable. Sublingual bioavailability is lower than injectable. The onset and peak effects may differ. Some people who switched from injectable to sublingual need dose adjustments before finding their optimal level. If you are switching routes, work with your healthcare provider and understand that a transition period is normal. See our guide on switching between GLP-1 formulations for transition strategies.
Sublingual semaglutide and other delivery methods compared
The GLP-1 delivery landscape extends beyond sublingual, injectable, and oral tablet. Understanding where sublingual fits among all available options provides context for making an informed choice.
Sublingual vs nasal spray
Nasal spray peptide delivery offers another needle-free option. The nasal mucosa is also highly vascularized and provides direct systemic access. However, nasal delivery for large peptides like semaglutide faces challenges with dosing consistency (how deeply you inhale affects absorption), mucosal irritation from repeated daily use, and clearance by the mucociliary system that clears the nasal passages.
Sublingual delivery generally provides more controlled dosing and less variability than nasal administration. The sublingual space is a closed environment where the dose stays pooled, while nasal sprays can drain into the throat or be cleared by nasal mucus before full absorption occurs.
Sublingual vs transdermal patches
GLP-1 patches represent another emerging needle-free delivery technology. Transdermal patches deliver medication through the skin over extended periods, providing steady drug levels without daily dosing decisions. However, skin permeability for large peptides is extremely limited without specialized microneedle or iontophoretic technology.
Sublingual delivery offers better absorption for peptides than conventional transdermal systems, though microneedle patch technology may change this equation as it matures.
Sublingual vs oral drops (troches, lozenges)
Some compounding pharmacies offer semaglutide as troches (slow-dissolving lozenges) or buccal drops designed to be held against the inner cheek rather than under the tongue. The buccal mucosa is thicker and less permeable than the sublingual mucosa, meaning buccal delivery is generally less efficient.
Troches dissolve slowly and keep the medication in contact with oral tissue for longer periods, potentially improving total absorption despite lower tissue permeability. The sublingual route remains the preferred oral mucosal pathway due to the thinner epithelium and higher vascular density.
Sublingual vs capsules
Peptide capsules are swallowed and absorbed through the GI tract, similar to oral tablets but without the SNAC enhancer system used in Rybelsus. Standard capsule formulations have very low bioavailability for semaglutide, similar to or worse than the oral tablet. Sublingual delivery is meaningfully superior to capsule delivery for semaglutide absorption.
Optimizing sublingual semaglutide results
Taking sublingual semaglutide correctly is necessary but not sufficient. Optimizing results requires attention to the same lifestyle factors that determine outcomes with any semaglutide formulation.
Nutrition during treatment
Semaglutide reduces appetite. This is the mechanism. But reduced appetite without nutritional awareness leads to muscle loss, nutrient deficiency, and metabolic slowdown. Following a structured semaglutide diet plan that prioritizes protein intake, micronutrient density, and adequate calories is essential for preserving lean mass during weight loss.
Key nutritional priorities:
Protein: 0.7-1g per pound of body weight daily to preserve muscle mass
Fiber: 25-35g daily to support gut motility (counteracts constipation)
Hydration: Minimum 64oz of water daily, more if active
Micronutrients: Supplement B12, vitamin D, magnesium, and zinc, which can become depleted during caloric restriction
Our comprehensive list of foods to eat while on semaglutide provides specific meal ideas and food categories optimized for GLP-1 therapy.
Exercise and activity
Resistance training is non-negotiable during semaglutide-assisted weight loss. Without it, up to 40% of the weight lost can come from lean tissue rather than fat. This muscle loss reduces metabolic rate, compromises physical function, and worsens body composition even as the scale number drops.
Strength training 3-4 times per week, combined with daily walking or light cardio, creates the optimal exercise foundation for semaglutide users. The medication handles appetite management. Exercise handles body composition. Together, they produce dramatically better outcomes than either alone.
Supplements to consider
Several supplements support health during semaglutide treatment:
B12/Methylcobalamin: GLP-1 agonists can reduce B12 absorption. B12 supplementation prevents deficiency-related fatigue and neurological symptoms.
Glycine: Supports metabolic function and liver health during weight loss. Some compounded formulations include glycine directly.
Magnesium: Caloric restriction commonly leads to magnesium depletion. Supplementation supports sleep, muscle function, and metabolic health.
Digestive enzymes: Can help manage GI side effects for some users.
Fiber supplements: Psyllium husk or similar can help manage semaglutide-related constipation.
Monitoring and tracking
Track the following metrics weekly to assess sublingual semaglutide effectiveness:
Body weight (same time, same conditions each week)
Waist circumference
Appetite level (subjective 1-10 scale)
Energy levels
Side effects and their severity
Sleep quality
Exercise performance
Tools like GLP-1 plotters and the GLP-1 companion app can help visualize trends and identify when dose adjustments are warranted. Consistent tracking data is invaluable for healthcare provider consultations.
Sublingual semaglutide cost considerations
Cost is a major factor in the semaglutide delivery route decision. The pricing landscape for compounded sublingual semaglutide differs significantly from both branded injectable and oral products.
Compounded sublingual semaglutide generally costs less than brand-name Wegovy or Ozempic injections, which can exceed $1,000-1,300 per month without insurance. However, compounded products are typically not covered by insurance, meaning patients pay out of pocket.
Pricing varies significantly between compounding pharmacies, formulation concentrations, and monthly supply volumes. Some pharmacies offer sublingual semaglutide starting at $150-400 per month depending on the dose and formulation. This is generally less than branded products but more than some other peptide therapy options.
When evaluating cost, factor in the total monthly expense including provider visits, lab work, and supplementation. Also consider that better absorption efficiency means potentially needing less total semaglutide per dose compared to oral tablets, which can partially offset the raw cost per milligram difference. Use the peptide cost calculator to estimate and compare costs across different delivery routes and providers.
How sublingual semaglutide compares to sublingual tirzepatide
Semaglutide and tirzepatide are the two dominant GLP-1 receptor agonists in weight management. Both are available in compounded sublingual formulations. Understanding how they compare in the sublingual context helps inform the choice between them.
Tirzepatide is a dual GIP/GLP-1 receptor agonist, meaning it activates two incretin receptors rather than one. In clinical trials, tirzepatide has shown greater weight loss than semaglutide at equivalent study durations. This advantage appears to extend to sublingual formulations as well, though head-to-head sublingual comparison data does not yet exist.
Sublingual tirzepatide formulations follow similar administration principles: hold under the tongue, empty stomach, extended contact time for optimal absorption. Tirzepatide drop formulations are available from many of the same compounding pharmacies that produce sublingual semaglutide.
For people considering switching between these medications, our semaglutide to tirzepatide conversion chart provides dosing guidance for the transition. Note that converting between sublingual formulations of different drugs adds another layer of complexity, since the bioavailability profiles may differ between the two peptides when delivered sublingually.
Frequently asked questions
Is sublingual semaglutide FDA approved?
No. There is no FDA-approved sublingual semaglutide product as of early 2026. The sublingual formulations currently available come from compounding pharmacies. Aspire Biopharma plans the first Phase 1 clinical trial of a sublingual semaglutide formulation in mid-2026, and Galmed Pharmaceuticals is developing a SEDDS-based sublingual GLP-1 delivery system. An FDA-approved product is likely several years away.
How long do I hold sublingual semaglutide under my tongue?
The minimum recommended hold time is 1-2 minutes, but optimal absorption requires 5-15 minutes of contact with the sublingual mucosa. Longer hold times allow more medication to cross the mucosal barrier and enter the bloodstream. Swallowing the liquid prematurely sends it to the stomach where most of it will be destroyed.
Can I switch from injectable semaglutide to sublingual?
Yes, with healthcare provider guidance. The transition requires dose adjustment because sublingual bioavailability is lower than injectable. Most providers recommend overlapping the end of an injection cycle with the start of daily sublingual dosing, then monitoring clinical response to determine the optimal sublingual dose. See our guide on semaglutide transition considerations for details on managing the switch.
Does sublingual semaglutide work as well as injectable?
The clinical evidence for sublingual semaglutide is limited compared to the extensive trial data supporting injectable semaglutide. Preclinical data suggests sublingual absorption is significantly better than oral tablets but lower than injection. Whether this translates to equivalent clinical weight loss depends on the dose, formulation, and individual absorption characteristics. At appropriate doses, sublingual semaglutide should produce similar effects, though some people may need higher milligram doses to achieve the same blood levels as injection.
What does sublingual semaglutide taste like?
Taste varies by compounding pharmacy and formulation. Some formulations are relatively tasteless. Others have a mildly bitter or medicinal taste. Many compounding pharmacies add flavoring agents to improve palatability. The taste is generally tolerable and improves with continued use as most people habituate to it.
Can I eat or drink after taking sublingual semaglutide?
Wait at least 15-30 minutes after taking sublingual semaglutide before eating, drinking, or brushing your teeth. This allows any residual medication in the oral cavity to complete absorption. Drinking water too soon can wash remaining medication into the stomach. Check our timing guide for optimizing your daily schedule around dosing.
Is sublingual semaglutide covered by insurance?
Generally not. Compounded medications are typically excluded from insurance formularies. Patients pay out of pocket for compounded sublingual semaglutide. Some FSA or HSA accounts may cover compounded medications with a valid prescription. Check with your specific plan for coverage details.
Can I combine sublingual semaglutide with other medications?
Semaglutide can interact with several medication classes regardless of delivery route. It slows gastric emptying, which can affect absorption of other oral medications. Combining semaglutide with other weight loss medications requires careful medical supervision. Always disclose all medications and supplements to your healthcare provider.
How should I store sublingual semaglutide?
Refrigerate at 36-46 degrees Fahrenheit (2-8 degrees Celsius). Protect from light. Do not freeze. Follow the beyond-use date on your specific bottle, typically 30-90 days from compounding. See our comprehensive peptide storage guide for detailed information.
Is sublingual semaglutide safe during pregnancy?
No. Semaglutide is contraindicated during pregnancy and should be discontinued at least 2 months before planned conception due to the long half-life of the drug. This applies to all delivery routes, including sublingual. Breastfeeding considerations also apply. Discuss family planning with your healthcare provider before starting any semaglutide formulation.
External resources
Single-dose pharmacokinetics of sublingual semaglutide in rats (ScienceDirect, 2026)
PCCA: Sublingual semaglutide dosing considerations and regulatory insights
Efficacy of semaglutide in subcutaneous and oral formulations (PMC)
Sublingual delivery: the future for GLP-1 receptor agonists (Labiotech)
For researchers serious about optimizing their GLP-1 protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, personalized dosing protocols, and a community of thousands who have navigated these exact questions about delivery methods, titration, and treatment optimization.
In case I do not see you, good afternoon, good evening, and good night. May your absorption stay optimal, your dosing stay consistent, and your results stay lasting.