Feb 24, 2026

After analyzing hundreds of retatrutide vendor listings, testing protocols, and third-party purity reports, one pattern keeps emerging. The difference between reliable research outcomes and wasted capital almost never comes down to which website looks the most professional. It comes down to verification. Specifically, whether the source you choose can prove what is actually inside the vial matches what the label claims.
That distinction matters more now than it ever has, because the retatrutide market has expanded rapidly in the wake of extraordinary clinical trial results. A 39-amino-acid synthetic peptide with three non-coded residues does not come cheap to manufacture correctly, and that manufacturing complexity has created a landscape where quality varies wildly from one vendor to the next. Independent testing of 1,866 peptide samples from 134 different vendors found purity ranging from 62% to 100%, with quantity accuracy diverging by as much as 10,000%. Even more concerning, 23% of tested retatrutide samples contained exendin-4 analogs rather than actual retatrutide. Those numbers should give any serious researcher pause. This guide exists to help you navigate that minefield with confidence, covering everything from retatrutide pricing structures to certificate of analysis interpretation, storage requirements to vendor red flags. SeekPeptides has assembled this resource because informed purchasing decisions protect both your research integrity and your budget. The goal is simple. By the time you finish reading, you will know exactly what to look for, what to avoid, and how to build a systematic process for evaluating any retatrutide source you encounter.
Why retatrutide sourcing quality matters more than price
Price is the first thing most researchers look at. It should be the last.
Retatrutide, designated LY3437943, is a triple agonist targeting GIP, GLP-1, and glucagon receptors simultaneously. That triple mechanism produced some of the most impressive weight reduction data in modern clinical research, with Phase 2 trials showing 24.2% mean body weight reduction at the 12mg dose over 48 weeks. The Phase 3 TRIUMPH-4 trial confirmed these results with 23.7% body weight reduction at 12mg and 20.0% at 9mg. Numbers like these have driven enormous demand, and enormous demand always attracts vendors who cut corners.
The problem is molecular complexity. Retatrutide contains three non-coded amino acid residues: two alpha-amino isobutyric acids at positions 2 and 20, plus an alpha-methyl-L-leucine at position 13. Synthesizing these correctly requires specialized equipment and expertise that not every peptide manufacturer possesses. When synthesis goes wrong, you do not just get a weaker product. You get something that may not be retatrutide at all.
Consider what poor sourcing actually costs. A vial that contains 70% purity retatrutide at a bargain price is not cheaper than a 98% purity vial at a premium price when you account for the dosing adjustments required, the inconsistent results, and the potential introduction of unknown impurities into your research. Factor in the 23% substitution rate mentioned earlier, and you begin to understand why the cheapest option often becomes the most expensive one over time. Researchers who have explored buying retatrutide online know that vetting vendors thoroughly is not optional. It is the single most important step in the entire research process.
Quality sourcing also protects reproducibility. If your dosage protocols are based on a specific purity level and your actual product deviates significantly from that level, every data point becomes unreliable. Your dose schedule falls apart. Your timeline expectations become meaningless. Months of careful planning get compromised by a purchasing decision that took fifteen minutes.
The parallel with other GLP-1 compounds is instructive. Researchers who have worked with compounded semaglutide understand that sourcing quality varies even for FDA-approved molecules. The situation with an unapproved investigational compound like retatrutide is exponentially more variable. There is no FDA manufacturing oversight. No standardized production requirements. No pharmacy board inspections. Quality exists only to the extent that individual manufacturers choose to maintain it, and not all of them choose well.
The financial argument for quality sourcing extends beyond the immediate purchase. A single vial of substandard retatrutide does not just waste the cost of that vial. It wastes every hour spent planning the protocol, every consumable used in the research process, and every day of timeline that produced unreliable data. The real cost of a bad purchase is measured in weeks of lost progress, not dollars per milligram. Researchers studying peptides for fat loss and metabolic research understand this principle deeply, because their protocols often span months and depend on consistent compound quality throughout.
The retatrutide quality spectrum explained
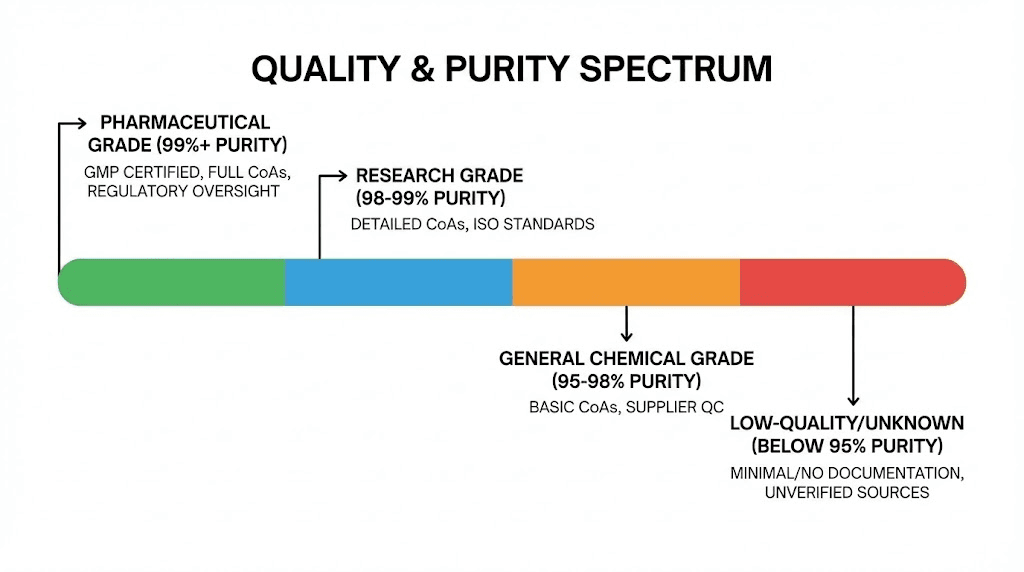
Not all retatrutide is created equal. The quality spectrum spans from pharmaceutical-grade synthesis to crude, barely functional knockoffs, and understanding where a product falls on that spectrum determines whether your research produces meaningful results.
At the top sits pharmaceutical-grade retatrutide. This is what Eli Lilly uses in clinical trials. Purity exceeds 99%, every batch undergoes extensive quality control, and the molecular weight of approximately 4,471 Daltons is confirmed through multiple analytical methods. Researchers outside of clinical trial settings rarely access this grade, but it represents the benchmark against which everything else should be measured.
Research-grade retatrutide occupies the next tier. Purity typically ranges from 95% to 99%, and reputable manufacturers provide detailed certificates of analysis with each batch. This grade serves the majority of legitimate research applications well. The key distinction between good research-grade and questionable research-grade lies in documentation. A vendor selling 98% purity retatrutide who provides an HPLC chromatogram, mass spectrometry data, and batch-specific testing results is operating at a fundamentally different level than one who simply prints "98% purity" on a label. When evaluating options from sources like Peptide Sciences or Paradigm Peptides, this documentation difference becomes immediately apparent.
Below research-grade, quality becomes unpredictable.
Some vendors sell retatrutide at 85% to 94% purity. For certain applications, this might be acceptable if the purity is accurately reported and the impurity profile is characterized. The danger zone begins when reported purity does not match actual purity, or when impurities include potentially harmful synthesis byproducts rather than benign truncated sequences. Vendors operating in this range who do not provide third-party testing data should be treated with extreme caution, particularly given the complexity of retatrutide purchasing decisions.
At the bottom of the spectrum sit products that claim to be retatrutide but contain something else entirely. The 23% substitution rate with exendin-4 analogs represents the most documented version of this problem, but other substitutions and adulterations exist as well. These products are not just ineffective. They introduce unknown variables into research and can produce misleading results that corrupt entire data sets.
This quality gap matters more than most researchers initially realize. Understanding where a product falls on this spectrum changes how you shop. Instead of asking "which vendor has the lowest price," the question becomes "which vendor can document where their product falls on the quality spectrum?" Documentation is the differentiator. A vendor who shows you exactly where their retatrutide sits, with evidence, is operating at a fundamentally different level than one who simply claims quality without proof. The difference between research and pharmaceutical grade peptides further illustrates why documentation standards matter so much in this market. The same principles apply whether you are sourcing retatrutide, evaluating cagrilintide vendors, or assessing any other next-generation research peptide.
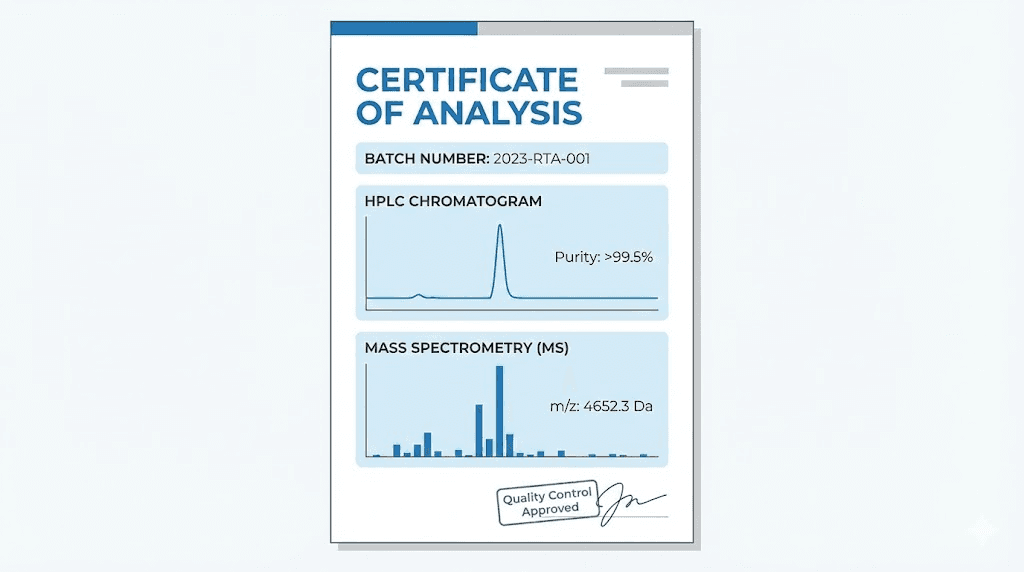
How to read a retatrutide certificate of analysis
A certificate of analysis is either your best defense against poor quality or a meaningless piece of paper. The difference depends entirely on what it contains, who produced it, and whether you know how to interpret it. Learning to read a COA properly is one of the highest-value skills any peptide researcher can develop, and it applies equally whether you are evaluating retatrutide availability from a new vendor or verifying a familiar source.
Essential elements of a legitimate COA
Every credible retatrutide COA should include a batch number that uniquely identifies the production run. This is not negotiable. Without a batch number, there is no way to trace the product back to a specific manufacturing event, and no way to verify that the COA you received corresponds to the vial you purchased.
HPLC chromatography data should be present. High-performance liquid chromatography separates the components of a sample and shows what percentage of the total material is the target peptide versus impurities. For retatrutide, look for a dominant peak representing the intact peptide at the correct retention time, with minimal secondary peaks. The purity percentage derived from this analysis should match or exceed the vendor stated purity.
Mass spectrometry data confirms molecular identity. The observed molecular weight should align with the expected value of approximately 4,471 Daltons, allowing for normal instrument variation. This test answers a fundamental question: is this actually retatrutide? Without mass spec data, you are relying entirely on the vendor assertion that the product is what they say it is. Given the substitution rates documented in independent testing, that reliance is not advisable. Researchers working with retatrutide dosing protocols need molecular identity confirmation before trusting any weight-based calculation.
The COA should also include appearance description, solubility data, endotoxin testing results, and amino acid analysis. Not every vendor provides all of these, but the more data points present, the more confidence you can place in the product.
Red flags in COA documents
Generic COAs that lack batch-specific information are the most common red flag. If every product from a vendor shares the same COA regardless of when it was manufactured, the document is essentially decorative. Real quality control generates unique data for each batch because each batch is a unique chemical event.
Watch for round numbers. A purity of exactly 99.00% should raise questions. Analytical instruments produce decimal results, and legitimate testing almost always yields values like 98.73% or 99.14%. Perfectly round numbers suggest the result was decided before the test was run.
Missing chromatogram images represent another warning sign. An HPLC result without the actual chromatogram is like a medical diagnosis without the imaging. The number alone tells you something, but the chromatogram tells you much more, including whether impurities are closely related peptide fragments or entirely different compounds. Understanding these details matters whether you are purchasing from ZLZ Peptide or any other source.
Be suspicious of COAs dated significantly before your purchase. Peptide quality can change during storage, particularly if storage conditions are suboptimal. A COA from six months ago may not accurately represent the product you receive today.
Third-party versus in-house testing
In-house testing means the vendor tested their own product. Third-party testing means an independent laboratory performed the analysis. The difference in credibility is substantial.
In-house testing is not inherently unreliable, but it creates an obvious conflict of interest. The entity with financial motivation to report high purity is also the entity determining purity. Reputable vendors address this by also providing third-party verification, usually from established analytical laboratories that specialize in peptide characterization.
Third-party testing eliminates this conflict. When an independent lab with no financial stake in the result confirms purity, identity, and quantity, you have a much stronger basis for confidence. Some vendors proactively submit samples for third-party testing. Others will do so upon request. Those who refuse third-party testing entirely should be avoided, especially when purchasing a complex molecule like retatrutide where the reconstitution process depends on accurate purity and quantity information.
Five categories of retatrutide sources
Understanding the vendor landscape requires categorization. Not every source operates the same way, carries the same risks, or offers the same level of accountability. Knowing which category a vendor falls into helps you calibrate your expectations and due diligence efforts appropriately.
Category 1: Established research peptide companies
These are companies that have operated in the research peptide space for years, sometimes over a decade. They maintain dedicated quality control laboratories, employ trained chemists, and have built reputations through consistent delivery of well-characterized products. Their pricing tends to be higher because their overhead, quality control, and customer service infrastructure is more extensive.
Established companies typically provide batch-specific COAs, offer third-party testing data, and maintain proper cold chain shipping protocols. They also tend to have responsive customer service and transparent return policies. The tradeoff is price. You will pay more per milligram from these sources, but you are also far more likely to receive exactly what you ordered at the stated purity. Detailed reviews of vendors like those covered in the Peptide Sciences retatrutide guide can help you evaluate specific companies in this category.
What distinguishes the best established vendors from merely adequate ones is consistency across batches. A vendor who delivered excellent product six months ago but has since changed manufacturers or quality control procedures may no longer meet the same standard. The longest-operating companies tend to have the most stable supply chains, which translates to the most predictable quality. Ask established vendors about their manufacturing relationships. How long have they worked with their current synthesis partner? Have they changed suppliers recently? These questions reveal operational stability that affects your research reliability. The Paradigm Peptides evaluation explores how established domestic vendors maintain quality over time.
Category 2: Newer domestic suppliers with third-party verification
A growing number of newer companies have entered the research peptide market with business models built around transparency and third-party testing. These vendors may not have the decade-long track record of Category 1 companies, but they compensate by making third-party test results readily available, often posting them directly on product pages.
The advantage here is often pricing. Newer companies operating with lower overhead can offer competitive rates while still maintaining quality standards. The risk is limited track record. A company that has been excellent for six months may or may not sustain that quality for six years. Evaluating these sources requires more ongoing vigilance and periodic verification of product quality.
Several newer suppliers have differentiated themselves by publishing comprehensive third-party testing results on their websites, sometimes including raw chromatogram data that more established vendors keep behind customer service requests. This transparency-first approach appeals to researchers who want to evaluate quality before placing an order rather than after. However, published test results from one batch do not guarantee the same quality in the next batch. Researchers working with newer vendors should plan on independent verification of at least their first order, treating the published data as a positive signal rather than a final guarantee. This careful verification approach is especially important for complex compounds like retatrutide, though the same principles apply to sourcing semaglutide from newer vendors or evaluating emerging tirzepatide sources. The research community benefits when newer vendors succeed at maintaining high standards, because increased competition drives quality improvements across the entire market.
Category 3: Direct manufacturer or bulk suppliers
Some researchers purchase directly from peptide synthesis facilities, often based in China. This approach can offer significant cost savings, particularly for bulk retatrutide purchases, but it comes with unique challenges. Communication barriers, longer shipping times, limited recourse for quality issues, and variable quality control standards all factor into the equation. The guide on bulk retatrutide from China covers these considerations in depth.
Direct manufacturer purchases absolutely require independent third-party testing before use. The quality control standards at synthesis facilities vary enormously, and the distance between you and the manufacturer makes resolving disputes difficult. Some researchers find excellent products through direct sourcing. Others discover that the savings evaporate when accounting for testing costs, shipping delays, and occasional batches that fail to meet specifications.
The language barrier deserves specific attention. Technical communication about peptide quality, purity specifications, and analytical methods requires precise vocabulary. Misunderstandings about what "98% purity" means, what analytical methods were used, or what impurity profiles look like can lead to receiving product that does not match expectations despite the vendor acting in good faith. Researchers who succeed with direct manufacturer purchasing typically have enough technical knowledge to communicate specifications clearly and evaluate responses critically. The lyophilized versus liquid peptide comparison illustrates the kind of technical nuance that matters when communicating specifications across language barriers.
Customs clearance adds unpredictable delays to international orders. Research peptides shipped from overseas may be held for days or weeks at customs facilities, often without temperature control. Summer orders are particularly vulnerable to heat degradation during extended customs holds. Some experienced researchers schedule their international orders during cooler months to minimize temperature-related risk. Others factor in the cost of occasional lost shipments as part of their total cost-of-ownership calculation.
Category 4: Compounding-adjacent sources
With the explosion of interest in GLP-1 receptor agonists, some sources have emerged that blur the line between research peptide suppliers and compounded pharmaceutical providers. These sources may market retatrutide alongside FDA-approved compounds like semaglutide and tirzepatide, sometimes implying a level of pharmaceutical oversight that does not actually exist for retatrutide as an investigational compound.
Approach this category with extra scrutiny. The regulatory framework for compounded pharmaceuticals does not extend to investigational research peptides, and a vendor association with compounding does not guarantee research peptide quality. Evaluate these sources using the same COA and testing criteria you would apply to any research peptide vendor. Some of the most well-known compounding pharmacies, like those detailed in the Empower Pharmacy semaglutide guide and Empower tirzepatide analysis, operate within FDA-regulated frameworks for approved compounds but may not apply those same oversight standards to investigational materials like retatrutide.
Category 5: Unverified online marketplaces and social media sellers
This category represents the highest risk tier. Vendors selling through social media platforms, general e-commerce marketplaces, or anonymous websites with no verifiable business identity present the greatest likelihood of quality problems. The 23% substitution rate and extreme quantity accuracy variations documented in independent testing are disproportionately concentrated in this category.
These sources often feature the lowest prices, which is precisely what makes them attractive and precisely what makes them dangerous. Without verifiable business identity, there is no accountability. Without batch-specific testing, there is no quality assurance. Without a reputation to protect, there is no incentive to deliver what was promised. Researchers who are exploring where to find retatrutide online should understand that the lowest price often correlates with the highest risk.

Red flags that signal a low-quality vendor
Spotting unreliable vendors before purchasing saves both money and research integrity. Certain warning signs appear consistently across problematic sources, and learning to recognize them quickly becomes second nature with practice.
Pricing dramatically below market average is the most obvious red flag. Manufacturing retatrutide properly costs real money. Three non-coded amino acid residues, a 39-residue chain, and rigorous purification processes all have minimum costs that cannot be eliminated through efficiency alone. When a vendor offers retatrutide at 50% or more below the average price from established suppliers, ask yourself what was cut to achieve that price. Usually, the answer involves purity, quantity, or both. Our retatrutide cost guide provides detailed pricing benchmarks for comparison.
No COA available is an immediate disqualifier. Any vendor who cannot provide a certificate of analysis for their product is telling you they either did not test it or do not want you to see the results. Neither option is acceptable.
Generic or template COAs shared across all batches indicate that testing is performative rather than genuine. Real quality control produces unique results for each production run.
Exaggerated claims should trigger skepticism. Vendors who promise "100% pure pharmaceutical-grade retatrutide" at research chemical prices are either lying about the grade or lying about the purity. Pharmaceutical grade implies GMP manufacturing and extensive regulatory oversight, which dramatically increases production costs.
Poor communication is a reliable predictor of problems. Vendors who take days to respond to pre-purchase questions, who cannot answer technical questions about their products, or who become evasive when asked about testing methodology are unlikely to handle post-purchase issues any better. A vendor who cannot explain how their retatrutide was synthesized, purified, and tested probably does not know, which means quality control is not a priority.
Website quality alone means nothing. Some of the worst vendors have beautiful websites. Some of the best have basic ones. Evaluate the substance behind the presentation, not the presentation itself. The same principle applies when comparing sources for grey market peptides of any kind.
Inconsistent product availability is another warning sign worth tracking. Vendors who frequently show products as "out of stock" and then suddenly have large quantities available may be sourcing opportunistically from whatever manufacturer offers the lowest price at any given moment. This inconsistency means the retatrutide you receive in one order may come from a completely different manufacturer than the next, with no quality continuity between batches. Consistent supply typically indicates stable manufacturing relationships, which correlates with consistent quality.
Unrealistic shipping promises deserve scrutiny as well. Vendors who claim same-day shipping from domestic warehouses but operate from overseas addresses, or who promise overnight delivery at standard shipping prices, may be misrepresenting their operations. Shipping logistics are verifiable. Track your first order carefully and compare the actual shipping timeline against what was promised.
Lack of any return or refund policy signals a vendor who does not stand behind their product. Legitimate vendors know that a small percentage of orders will have issues, and they plan for that with clear policies. Vendors who offer no recourse for quality problems are either confident you will not test the product or confident they will not be around when you try to complain. Neither scenario favors the researcher.
Green flags of a trustworthy retatrutide source
If red flags tell you who to avoid, green flags tell you who deserves a closer look. Trustworthy vendors share several characteristics that distinguish them from the rest of the market.
Batch-specific COAs with chromatogram images represent the gold standard. When a vendor provides HPLC data complete with the actual chromatogram for the specific batch you are purchasing, they are demonstrating genuine quality control. This level of transparency requires investment in analytical equipment and trained personnel, which itself signals commitment to quality.
Third-party testing availability matters enormously. Vendors who proactively share third-party test results, or who readily facilitate independent testing of their products, are confident in what they sell. That confidence is usually justified.
Consistent community reputation counts. Long-term positive feedback from multiple independent researchers carries more weight than any marketing claim. Look for patterns across forums, review aggregators, and research communities. A vendor who has consistently delivered quality products for years has demonstrated something that no amount of advertising can replicate. This is particularly true for complex molecules where resources like the retatrutide purchasing guide can help contextualize community feedback.
Proper storage and shipping protocols demonstrate understanding of peptide chemistry. Retatrutide is a protein-based molecule that degrades under improper conditions. Vendors who ship with appropriate cold packs, use insulated packaging, and clearly communicate storage requirements understand their product at a fundamental level.
Responsive, knowledgeable customer service rounds out the profile. When you can ask a technical question and receive a competent, specific answer, you are dealing with an organization that employs people who understand peptide science, not just sales representatives reading scripts.
Educational content creation is a subtle but powerful quality signal. Vendors who publish detailed guides about peptide handling, storage, reconstitution, and research methodology demonstrate expertise that typically extends to their manufacturing and sourcing practices. It takes real knowledge to create genuinely useful educational material, and vendors who invest in that knowledge tend to apply it across all aspects of their operations. This mirrors the approach SeekPeptides takes with comprehensive research guides, peptide stacking calculators, and detailed dosing charts that serve the research community.
Multiple vial size options often indicate a mature supplier. Vendors who offer 10mg, 20mg, and larger vials have invested in the equipment and processes to handle different fill volumes accurately. A vendor who only sells one vial size may be working with limited manufacturing capabilities. This is not always the case, but the availability of options suggests a more established operation.
Clear labeling with complete product information, including synthesis date, storage recommendations, and molecular weight, shows attention to detail. Vials that arrive with minimal or no labeling beyond a product name raise questions about how carefully the vendor manages other aspects of quality that are not visible from the outside.
How retatrutide pricing reflects quality
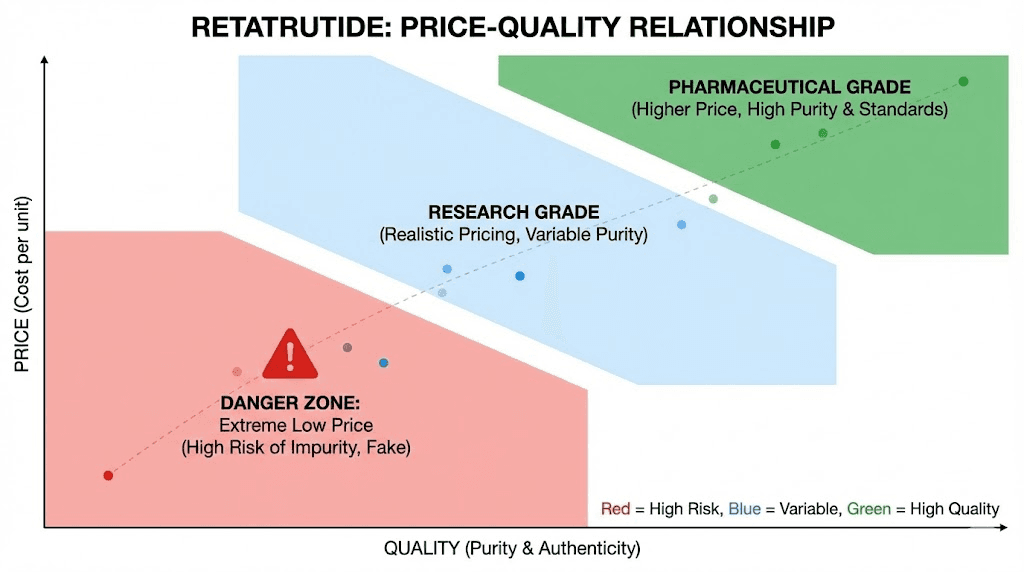
Pricing in the retatrutide market follows patterns that become predictable once you understand the underlying economics. The manufacturing cost floor for legitimate retatrutide is set by the complexity of the synthesis, and no amount of volume purchasing eliminates that floor entirely.
Three factors drive retatrutide manufacturing costs above those of simpler peptides. First, the three non-coded amino acid residues (two alpha-amino isobutyric acids and one alpha-methyl-L-leucine) require specialized building blocks that cost more than standard amino acids. Second, the 39-residue chain length means lower overall synthesis yields compared to shorter peptides, because each coupling step has a less-than-perfect success rate that compounds across the chain. Third, purification to research-grade purity requires multiple chromatography runs, each consuming time, solvents, and column capacity.
Together, these costs create a manufacturing floor that honest vendors cannot undercut without sacrificing quality. When vendors price dramatically below this realistic range, something has been compromised. Sometimes it is purity, with lower-grade product being sold at a claimed higher purity. Sometimes it is quantity, with vials containing less material than labeled. Sometimes it is identity, with a cheaper compound being substituted entirely. The complete retatrutide pricing guide breaks down these economics in detail.
That said, the most expensive option is not automatically the best. Some vendors charge premium prices without providing premium quality, relying on the assumption that buyers equate high price with high quality. The optimal purchasing strategy ignores both extremes and focuses on vendors whose pricing falls within the realistic range and whose quality documentation supports their claims.
Consider calculating the true cost per verified milligram rather than the sticker price per vial. If Vendor A charges $80 for a 10mg vial that independently tests at 99% purity with accurate fill weight, the cost per active milligram is approximately $8.08. If Vendor B charges $40 for a 10mg vial that tests at 85% purity with only 7mg actual content, the cost per active milligram is approximately $6.72, but you also need to account for the 15% impurity load and the unreliable dosing calculations. When factoring in the risk of research disruption, the "cheaper" option often ends up costing more in practical terms. The peptide cost calculator helps standardize these comparisons across vendors.
Seasonal pricing patterns deserve attention as well. Some vendors offer discounts during lower-demand periods that represent genuine value rather than quality compromises. Others raise prices during peak demand periods without any change in product quality. Understanding these patterns helps you time purchases strategically without sacrificing quality. Comparison shopping for research peptides follows principles similar to those outlined in guides about finding affordable tirzepatide and affordable GLP-1 options, where value and quality must be balanced carefully.
Volume purchasing can legitimately reduce per-milligram costs. Bulk retatrutide purchasing from established suppliers typically comes with quantity discounts that reflect actual savings in packaging and handling rather than quality compromises. The critical distinction is that legitimate bulk discounts are modest, perhaps 10% to 25%, while suspicious discounts are dramatic, 50% or more below market rates.
Storage and shipping standards that protect your purchase
Even perfectly synthesized retatrutide becomes worthless if storage and shipping degrade it before use. Understanding the stability profile of this peptide helps you evaluate whether a vendor handling practices are adequate and whether the product you receive is likely to be intact.
Lyophilized retatrutide, the freeze-dried powder form in which it is typically sold, demonstrates excellent stability when stored properly. At -20 degrees Celsius, lyophilized retatrutide remains stable for up to 48 months. This extended shelf life makes proper vendor storage practices critical, because a well-manufactured product that has been stored at room temperature for months may have lost significant potency before it ever reaches your laboratory.
Shipping introduces the most acute risk. Transit times, ambient temperatures, and packaging quality all affect whether the product arrives intact. Reputable vendors address this with insulated packaging, cold packs or dry ice for warm-weather shipments, and expedited shipping options. Some vendors include temperature indicators that reveal whether the package exceeded safe temperature ranges during transit. These details might seem minor, but they represent the difference between receiving what you paid for and receiving a degraded product.
Once you receive lyophilized retatrutide, storage at -20 degrees Celsius is ideal for long-term preservation. A standard freezer works well for this purpose. Reconstituted retatrutide requires different handling, with refrigeration at 2 to 8 degrees Celsius and use within one to two weeks for optimal potency. Room temperature tolerance for lyophilized powder extends to approximately 21 days below 30 degrees Celsius before significant degradation occurs, which provides some buffer for shipping delays but should not be relied upon for routine storage.
Evaluate your vendor shipping standards before purchasing. Ask what packaging they use. Ask about cold chain maintenance. Ask whether they offer expedited shipping during warm months. The answers will tell you a great deal about how seriously they take product integrity. The same storage principles apply to related compounds, as detailed in guides about tirzepatide temperature sensitivity and semaglutide temperature requirements. Proper handling of these peptides follows similar principles, and guides on tirzepatide refrigeration and semaglutide refrigeration provide additional context.
Your own storage infrastructure matters just as much as the vendor handling. Before your first retatrutide order arrives, verify that your freezer maintains a consistent -20 degrees Celsius. Temperature fluctuations caused by frequent door opening, power outages, or auto-defrost cycles can compromise peptide stability over time. A dedicated laboratory freezer with temperature logging provides the most reliable storage environment. For researchers who lack access to a -20 degree freezer, short-term refrigerator storage at 2 to 8 degrees Celsius works for lyophilized product that will be used within a few months, though the stability window is shorter than deep-freeze storage.
Long-term storage planning should account for the total quantity of retatrutide you expect to hold at any given time. Organizing vials by batch number and purchase date helps you rotate stock effectively, using older inventory before newer purchases. This "first in, first out" approach maximizes the usable life of every vial and prevents the accumulation of aging stock that gradually loses potency. The same organizational principles apply to researchers maintaining inventories of semaglutide, tirzepatide, or any other temperature-sensitive research compounds.
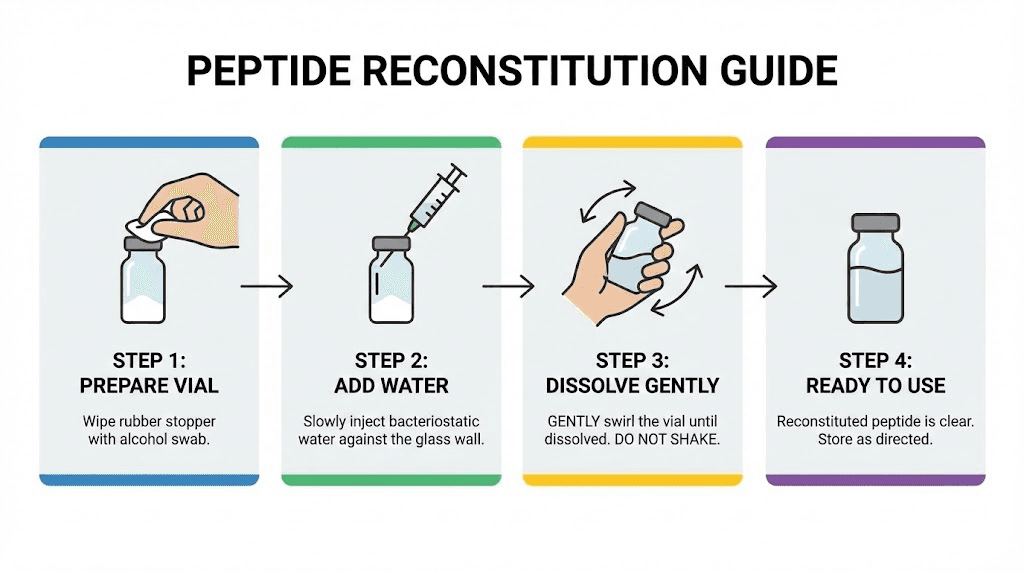
Reconstitution essentials for retatrutide researchers
Purchasing quality retatrutide is only half the equation. Proper reconstitution transforms the lyophilized powder into a usable solution, and errors at this stage can undermine even the highest-quality starting material.
Bacteriostatic water is the standard reconstitution solvent for retatrutide. The bacteriostatic agent, typically 0.9% benzyl alcohol, inhibits microbial growth and extends the usable life of the reconstituted solution. The volume of bacteriostatic water you add determines the concentration of the final solution, which directly affects dosing accuracy. For a 10mg vial, the bacteriostatic water volume guide provides specific calculations. Similar reconstitution principles apply across peptides, as covered in our guides for semaglutide reconstitution and tirzepatide reconstitution.
The reconstitution process itself requires care. Direct the bacteriostatic water stream against the glass wall of the vial, allowing it to flow gently down onto the lyophilized cake. Never inject water directly onto the powder with force, as this can damage the peptide through shear stress. Once the water is added, gently swirl the vial. Do not shake it. Vigorous agitation creates foam that indicates protein denaturation, the exact thing you are trying to avoid.
The retatrutide reconstitution chart provides volume-to-concentration tables that eliminate guesswork. Using the peptide reconstitution calculator on SeekPeptides adds another layer of precision by accounting for the specific vial size, desired concentration, and syringe markings you are working with.
After reconstitution, store the vial upright in a refrigerator at 2 to 8 degrees Celsius. Avoid repeated freeze-thaw cycles, which degrade peptide quality faster than almost any other handling error. If you reconstituted more than you will use within one to two weeks, consider dividing the solution into single-use aliquots before freezing, as covered in depth in peptide refrigeration duration guides. Related resources on semaglutide fridge storage and tirzepatide room temperature stability cover the same principles for related compounds.
Retatrutide dosage context for informed purchasing
Understanding dosage protocols before purchasing helps you buy the right quantity and avoid both shortages and waste. The amount of retatrutide you need depends on the protocol you intend to follow, the duration of your research, and whether you are titrating up from a starting dose.
Clinical trial protocols provide the most referenced dosing framework. The Phase 2 trial used doses ranging from 0.5mg to 12mg administered once weekly, with the highest dose producing the most dramatic results. The Phase 3 TRIUMPH-4 trial focused on 8mg and 12mg doses. For researchers planning protocols, understanding these dose ranges is essential for calculating total material requirements. The retatrutide dosage chart for weight loss research provides a structured overview of these clinical benchmarks.
Titration matters. Most protocols begin with lower doses and increase gradually. This approach allows for tolerance assessment and reduces the likelihood of adverse effects. A typical titration schedule might span several weeks before reaching the target dose, which means your initial purchase needs to account for both the lower titration doses and the eventual maintenance dose. The starting dose guide covers initial dosing considerations in detail.
With a half-life of approximately six days, retatrutide supports once-weekly dosing. This pharmacokinetic profile simplifies protocol planning and means that a single vial can last several weeks depending on the dose and vial size. The retatrutide dosage calculator helps translate weekly dose requirements into total milligram needs over a defined research period. Additional dosing resources include the detailed dosage calculator guide, the peptide dosage chart, and the comprehensive dosing overview.
The 20mg retatrutide dosing guide addresses higher-quantity vials specifically, which are relevant for researchers running extended protocols or working at higher dose ranges. Purchasing the appropriate vial size minimizes reconstitution events and reduces the risk of contamination or degradation from repeated access.
For researchers transitioning from other compounds, specific dosing considerations apply. The guides on transitioning from tirzepatide to retatrutide and retatrutide onset after tirzepatide address these scenarios with specific timeline and dosing recommendations.
Administration considerations also influence purchasing decisions. Retatrutide is administered subcutaneously, typically in the abdomen, thigh, or upper arm. The injection site selection guide covers the practical aspects of administration, while the injection technique guide addresses proper technique. Researchers need appropriate insulin syringes for accurate measurement. The syringe gauge and volume should match the reconstituted concentration and target dose. Using a 1ml insulin syringe with 100-unit markings provides the measurement precision needed for milligram-accurate dosing at most standard reconstitution concentrations.
Side effect awareness should inform both purchasing quantity and protocol planning. The most common adverse effects reported in clinical trials include nausea (38-43% at higher doses), diarrhea (33-35%), constipation (22-25%), and vomiting (20-21%). These gastrointestinal effects are most pronounced during dose escalation and tend to diminish over time. Understanding this profile helps researchers plan realistic timelines and avoid over-purchasing material for protocols that may need adjustment due to tolerability considerations. The broader side effect landscape for GLP-1 compounds is explored in resources about semaglutide constipation management, tirzepatide digestive side effects, and tirzepatide and anxiety.
How retatrutide compares to other GLP-1 options
Retatrutide does not exist in isolation. It belongs to a broader category of GLP-1 receptor agonists, and understanding how it compares to alternatives helps researchers make informed decisions about which compound best serves their specific research objectives. This comparison also provides context for evaluating whether the premium pricing of retatrutide over simpler compounds is justified for your particular application.
The fundamental distinction is receptor targeting. Semaglutide is a single GLP-1 receptor agonist. Tirzepatide is a dual GIP and GLP-1 receptor agonist. Retatrutide is a triple GIP, GLP-1, and glucagon receptor agonist. Each additional receptor target adds both complexity and potential efficacy, which is reflected in both the clinical data and the manufacturing costs. The three-way comparison guide provides the most detailed analysis of these differences.
In clinical trials, retatrutide produced the highest weight reduction percentages of any compound in its class. The 24.2% mean reduction at 12mg over 48 weeks exceeded the results seen with both semaglutide and tirzepatide in their respective trials. The retatrutide versus semaglutide comparison explores these efficacy differences in detail, while the retatrutide versus tirzepatide dosage comparison addresses how dosing schedules differ between the compounds.
Availability and sourcing complexity also differ significantly. Semaglutide is FDA-approved and available through pharmacies and compounding pharmacies. Tirzepatide is also FDA-approved with established supply chains, including options from specialized compounding sources and affordable alternatives. Retatrutide remains an investigational compound without FDA approval, which limits it to the research chemical market and introduces all the quality variability discussed throughout this guide.
Newer entrants in the GLP-1 space are also worth monitoring. The mazdutide versus retatrutide comparison and the Ozempic versus retatrutide analysis provide additional comparative context. Understanding the broader landscape, including what GLP-1 actually means relative to specific brand names, helps researchers position their work within the evolving field.
Side effect profiles differ across compounds as well. While all GLP-1 agonists share some common gastrointestinal effects, the triple agonism of retatrutide introduces glucagon receptor-mediated effects that the others do not produce. Researchers considering side effect management should review resources on semaglutide versus tirzepatide side effects, retatrutide and hair loss, and GLP-1 hair loss to understand the full picture. Fatigue is another consideration, with dedicated guides for GLP-1 fatigue, semaglutide fatigue, and tirzepatide fatigue.
Legal framework for research peptide purchasing
The legal status of retatrutide purchasing depends on jurisdiction, intended use, and how the compound is classified in your area. Understanding this framework protects you from inadvertent legal exposure and helps you identify vendors who operate within appropriate boundaries.
In the United States, retatrutide is not scheduled as a controlled substance. It is not FDA-approved, which means it cannot be legally sold for human consumption. However, it can be legally sold and purchased as a research chemical for legitimate research purposes. This "research use only" framework is the legal basis under which most domestic vendors operate. The distinction between research use and personal use matters significantly from a regulatory perspective.
Vendors who explicitly market retatrutide for human use, weight loss, or personal health benefits are operating outside the research chemical framework and exposing both themselves and their customers to regulatory risk. Legitimate vendors clearly label their products as "for research use only" and do not make therapeutic claims. This is not just a legal formality. Vendors who respect these boundaries tend to also respect quality standards, because both reflect an understanding of and commitment to operating within established rules.
International purchasing adds complexity. Customs regulations vary by country, and some jurisdictions restrict the importation of research peptides. If you are considering international vendors, understand the import regulations for your specific location before placing an order. Seized shipments represent both financial loss and potential legal complications. The research versus pharmaceutical peptide comparison provides additional context on how regulatory status affects purchasing options.
Payment method can also provide quality signals. Vendors who accept standard payment methods, credit cards, bank transfers, and established payment processors, operate within financial systems that provide some recourse for fraudulent transactions. Vendors who exclusively accept cryptocurrency or wire transfers to personal accounts remove that protection entirely.
State-level regulations add another layer of complexity for US-based researchers. Some states have enacted specific legislation regarding research chemicals that may affect purchasing, storage, or use of investigational peptides. While retatrutide itself is not controlled at the federal level, individual state laws can create additional requirements or restrictions. Checking your state specific regulations before purchasing is a prudent step that protects against unexpected legal complications.
For researchers affiliated with institutions, additional compliance requirements often apply. Institutional review boards, biosafety committees, and chemical safety offices may have specific protocols for procuring and storing investigational compounds. These institutional requirements exist separately from federal and state law and can impose additional documentation, storage, or handling obligations. Compliance with institutional protocols is not optional, and sourcing decisions should account for any documentation requirements your institution mandates. The peptides versus SARMs regulatory comparison provides useful context for understanding how different research compound categories are regulated.
International researchers face a patchwork of regulations that vary significantly by country. Some nations classify research peptides under pharmaceutical regulations regardless of intended use. Others have specific frameworks for research chemicals that impose requirements around documentation, end-use certification, or import licensing. Understanding your jurisdiction specific rules before ordering from an international vendor prevents shipment seizures, legal complications, and financial losses.
Common mistakes when sourcing retatrutide
Even experienced researchers make sourcing mistakes. Recognizing the most common errors helps you avoid them proactively rather than learning through expensive trial and error.
Prioritizing price over verification is the single most common mistake. The appeal of a lower price is understandable, especially when retatrutide costs significantly more than simpler peptides. But the cheapest vial that contains 70% purity or a substituted compound costs infinitely more than a slightly pricier vial that contains what the label says. Every dollar saved on a bad purchase is a dollar wasted.
Failing to verify COAs independently ranks second. Accepting a COA at face value without checking its internal consistency, comparing it against known standards, or requesting third-party verification leaves you reliant on vendor honesty alone. Given the documented quality issues in the market, that reliance is misplaced.
Ordering without a storage plan creates waste. Researchers who purchase retatrutide before their freezer space, reconstitution supplies, and protocol timeline are organized often end up storing product under suboptimal conditions. Plan your storage infrastructure before your purchase arrives, not after.
Ignoring shipping conditions during warm months is a seasonal version of the same problem. A product that ships perfectly in January may arrive degraded in July if the vendor does not adjust their packaging accordingly. Ask about warm-weather shipping protocols before ordering during summer months. The same principle applies to traveling with peptides, where temperature control becomes even more challenging.
Buying from a single source without comparison testing prevents you from establishing your own quality baseline. Experienced researchers often purchase small quantities from multiple vendors initially, submit samples for independent testing, and then scale up with the vendor that demonstrates the best quality. This upfront investment in comparison pays dividends in long-term research reliability.
Not checking expiration and manufacturing dates is surprisingly common. Peptides degrade over time even under proper storage, and purchasing old stock near its expiration means less usable life for your investment. The principles of peptide expiration and expired peptide viability apply equally to retatrutide.
Switching vendors mid-protocol without comparison testing introduces uncontrolled variables. Even if two vendors report identical purity levels, subtle differences in impurity profiles, actual peptide content, or formulation can affect research outcomes. If you must change vendors during a protocol, treat the new source as unverified. Test it independently before incorporating it into ongoing research. The compound switching guide illustrates similar transition challenges when researchers change between different peptides entirely.
Stockpiling without a consumption plan wastes capital and risks degradation. It is tempting to buy large quantities when a trusted vendor has a good price, but lyophilized peptides, while stable for years at -20 degrees Celsius, are not infinitely shelf-stable. Calculate your projected consumption rate over the next 6 to 12 months and purchase accordingly. Excess inventory ties up money that could be invested in other research needs, and even properly stored peptides gradually degrade. The peptide shelf life considerations and tirzepatide shelf life analysis explore degradation timelines that inform smart purchasing quantities.
Neglecting to document your vendor evaluation process is the final common mistake. Researchers who do not record which vendors they evaluated, what criteria they used, and what results they observed lose valuable institutional knowledge. When it comes time to reorder or when a colleague asks for a vendor recommendation, documented evaluations save time and improve decision quality. Keep a simple spreadsheet tracking vendor name, COA quality, independent test results, shipping quality, and overall satisfaction.

Building a systematic vendor evaluation process
Random vendor selection produces random results. A systematic evaluation process, applied consistently to every potential source, dramatically improves your odds of finding reliable quality.
Start with documentation review. Before purchasing anything, request a COA for the specific batch currently in stock. Review it against the criteria outlined earlier in this guide. Check for batch-specific data, HPLC chromatograms, mass spectrometry results, and reasonable purity claims. This step alone eliminates the majority of problematic vendors, because vendors who cannot or will not provide adequate documentation are telling you everything you need to know.
Research the vendor reputation independently. Search for reviews, feedback, and discussion in research communities. Look for patterns rather than individual opinions. A single negative review might reflect an isolated incident. A pattern of complaints about purity, quantity, or customer service reflects a systemic problem. When researching options, resources like the complete retatrutide buying guide provide structured evaluation frameworks.
Place a small test order first. Purchasing a single vial allows you to evaluate shipping speed, packaging quality, product appearance, and customer service responsiveness before committing to a larger purchase. If the test order meets your standards, scale up. If it does not, you have limited your financial exposure to a single vial rather than a bulk purchase.
Submit the test order for independent analysis if possible. Third-party testing of a sample from your first order provides objective quality data that no amount of vendor marketing can replicate. The cost of a single analytical test is trivial compared to the cost of running an entire research protocol with compromised material.
Document everything. Keep records of vendor COAs, your own test results, shipping conditions, and product performance. This documentation creates a vendor quality database that improves your purchasing decisions over time and provides evidence if you need to dispute a quality claim. Use the peptide calculator and cost calculator tools from SeekPeptides to standardize your cost-per-milligram comparisons across vendors and make your documentation even more useful.
Establish a reorder threshold based on your consumption rate and vendor lead times. Running out of retatrutide mid-protocol because you waited too long to reorder disrupts research timelines. Conversely, stockpiling excessive quantities ties up capital and risks degradation if storage conditions are not ideal. The balance point depends on your specific protocol requirements.
Periodically re-evaluate even trusted vendors. Quality can change when companies change ownership, manufacturing partners, or quality control procedures. Annual or semi-annual verification testing of products from your regular vendor ensures that the quality you originally confirmed still holds. This verification applies whether you are sourcing retatrutide, tirzepatide, or any other research peptide.
Build a vendor backup plan as part of your evaluation process. Relying on a single source creates vulnerability. If your primary vendor experiences stock shortages, quality problems, or operational disruptions, having a pre-vetted alternative prevents research interruptions. Maintain at least one backup vendor who has passed your evaluation criteria and from whom you have independently verified product quality. This redundancy may seem excessive for small-scale research, but the cost of a mid-protocol supply disruption far exceeds the modest investment of testing a backup source. The peptide research community benefits collectively when researchers share their evaluation findings, helping others identify quality sources more efficiently. Platforms like SeekPeptides facilitate this knowledge sharing through community forums, detailed vendor guides, and researcher-contributed quality reports.
Frequently asked questions
What purity level should I look for when buying retatrutide?
For most research applications, target a minimum of 95% purity with batch-specific HPLC verification. Higher purity, 98% or above, provides greater dosing accuracy and fewer impurity-related variables. Always verify claimed purity against a batch-specific COA that includes the actual chromatogram, not just a stated number. The retatrutide dose guide explains how purity levels affect protocol calculations.
How much does quality retatrutide typically cost per milligram?
Pricing varies by vendor category and quantity purchased, but legitimate research-grade retatrutide from established suppliers falls within a recognizable range. Prices significantly below the market average, particularly 50% or more below, almost always indicate quality compromises. The retatrutide pricing guide provides current market benchmarks and explains what drives cost differences between vendors.
Is it legal to buy retatrutide for research purposes?
In the United States, retatrutide is not a scheduled controlled substance and can be legally purchased as a research chemical for legitimate research use. It is not FDA-approved for human consumption, and vendors who market it for personal health use operate outside the established legal framework. International regulations vary by jurisdiction, so verify local laws before purchasing from overseas sources.
How can I verify that a vendor product is actually retatrutide and not a substitute?
Mass spectrometry data on the COA should confirm a molecular weight of approximately 4,471 Daltons, consistent with the retatrutide sequence. For definitive verification, submit a sample to an independent analytical laboratory for identity testing. Given that 23% of tested samples contained exendin-4 analogs rather than retatrutide, independent verification is strongly recommended for any new vendor.
What is the best way to store retatrutide after purchasing it?
Store lyophilized retatrutide at -20 degrees Celsius for maximum stability, where it remains viable for up to 48 months. After reconstitution, refrigerate at 2 to 8 degrees Celsius and use within one to two weeks. Avoid repeated freeze-thaw cycles. The peptide shelf life guide and semaglutide storage duration guide cover stability principles that apply broadly to research peptides.
Should I buy from domestic or international retatrutide vendors?
Both options can work well with proper verification. Domestic vendors offer faster shipping, easier returns, and no customs risk. International sources, particularly direct manufacturers, can offer lower pricing but require more due diligence and carry customs seizure risk. Regardless of origin, always verify quality through COA review and ideally independent testing.
How do I know if my retatrutide has degraded during shipping?
Lyophilized retatrutide should appear as a white to off-white powder or cake. Discoloration, visible moisture, or a collapsed, sticky appearance may indicate degradation. Some vendors include temperature monitoring indicators in shipments. If you suspect degradation, reconstitute a small amount and check for unusual cloudiness or particulate matter, which can signal protein aggregation. Understanding lyophilized versus liquid peptide stability profiles helps contextualize what to expect.
What injection supplies do I need alongside my retatrutide purchase?
A complete setup requires bacteriostatic water for reconstitution, insulin syringes for measurement and administration, alcohol swabs for sterile technique, and appropriate sharps disposal. The retatrutide injection site guide and injection technique guide cover the complete preparation and administration process. For general technique principles, the GLP-1 injection guide provides foundational instruction.
How does retatrutide compare to semaglutide and tirzepatide for research applications?
Retatrutide targets three receptors (GIP, GLP-1, and glucagon) compared to two for tirzepatide (GIP and GLP-1) and one for semaglutide (GLP-1). This triple mechanism produced 24.2% mean weight reduction in Phase 2 trials, exceeding both comparators. However, retatrutide is not FDA-approved, which means sourcing involves significantly more quality variability than the approved compounds. The three-way comparison guide provides detailed efficacy and mechanism analysis.
Can I combine retatrutide with other research peptides?
Combining research peptides requires careful consideration of mechanisms, potential interactions, and protocol complexity. Researchers exploring combinations should understand each compound independently before introducing them together. The peptide stack calculator helps plan combination protocols, and individual compound guides provide the foundational knowledge needed for informed stacking decisions. Always source each compound from verified vendors independently rather than purchasing pre-mixed combinations from unverified sources.
What happens if my retatrutide was exposed to heat during shipping?
Brief exposure to temperatures below 30 degrees Celsius, lasting less than 21 days, typically does not cause significant degradation of lyophilized retatrutide. Extended heat exposure or temperatures above 30 degrees may reduce potency measurably. If you suspect temperature compromise, inspect the lyophilized cake for discoloration or moisture. When in doubt, submit a sample for independent purity testing before using the material in research. The impact of temperature on semaglutide and temperature on tirzepatide follows similar degradation patterns.
External resources
For researchers serious about optimizing their retatrutide protocols and sourcing decisions, SeekPeptides offers the most comprehensive resource available. Members access detailed vendor evaluation frameworks, dosing calculators, reconstitution guides, and a community of experienced researchers who have navigated these exact purchasing decisions. Whether you are sourcing your first vial or refining an established protocol, the tools and knowledge within SeekPeptides help you make every milligram count.