Mar 1, 2026

You open the mailbox. The package feels warm. Maybe even hot. Your stomach drops because inside that box is semaglutide, a medication you have been waiting days to receive, and now you are standing in your driveway wondering if you just lost hundreds of dollars. The ice packs are melted. The insulation feels limp. And that vial or pen sitting inside? It is not cool to the touch.
This is not a rare scenario.
Every summer, thousands of people receiving compounded semaglutide or brand-name GLP-1 medications face this exact moment. The panic is real. The confusion is overwhelming. You do not know if you should inject it, throw it away, call the pharmacy, or start the whole ordering process over again. And the internet is full of conflicting advice, with some people saying a little warmth is fine and others insisting you should never use a medication that left the cold chain. The truth sits somewhere in the middle, and the details matter more than most guides admit. Temperature thresholds differ between formulations. Exposure duration changes everything. And the difference between a medication that lost 5% potency and one that lost 50% is not something you can see with your eyes.
This guide covers the exact temperature limits for every semaglutide formulation, how to assess whether your medication is still safe, the step-by-step process for getting a replacement, and the science behind what heat actually does to peptide molecules. Whether you received warm semaglutide from a pharmacy shipment or accidentally left it in a hot car, the protocols here will help you make an informed decision. SeekPeptides has compiled the most comprehensive temperature guidance available so you do not have to guess.
Why semaglutide temperature matters more than you think
Semaglutide is a peptide. That single fact determines everything about how it responds to heat. Unlike small-molecule drugs such as aspirin or ibuprofen, which can tolerate wide temperature swings without losing effectiveness, peptides are proteins with complex three-dimensional structures that depend on precise folding to function. When heat disrupts that folding, the medication stops working the way it should.
Think of it like an egg.
A raw egg has proteins in their natural folded state. Apply heat and those proteins unfold permanently. You cannot un-cook an egg. Peptide denaturation works on a similar principle, though the temperatures involved are much lower than what it takes to fry breakfast. The hydrogen bonds and hydrophobic interactions that hold semaglutide in its active configuration begin weakening at temperatures well below boiling, and once enough bonds break, the molecule cannot refold into its functional shape. This process, called denaturation, reduces the medication potency without necessarily changing how it looks.
The reason this matters for your shipment is straightforward. Semaglutide shelf life and potency depend on maintaining specific temperature ranges from the moment of manufacturing through the moment of injection. Every minute outside those ranges chips away at the active ingredient.
A short temperature excursion might cost you 2-3% potency. A long one could destroy the medication entirely. And the frustrating part is that degraded semaglutide often looks identical to fresh semaglutide, so you cannot rely on visual inspection alone to determine safety.
What makes GLP-1 receptor agonists particularly sensitive is their mechanism of action. Semaglutide works by mimicking the incretin hormone GLP-1, binding to receptors in the pancreas to stimulate insulin release and in the brain to reduce appetite. That binding depends on the molecule maintaining its exact three-dimensional shape. If even a small portion of the binding site denatures, the medication affinity for its receptor drops, and you need more of the drug to achieve the same effect. This is why someone injecting heat-damaged semaglutide might notice reduced weight loss results or a return of appetite that was previously well controlled.
The financial stakes are significant too. Brand-name Ozempic and Wegovy can cost over $1,000 per month without insurance. Even compounded semaglutide from telehealth providers runs $150-400 per month depending on the pharmacy and concentration. Injecting degraded medication means you are paying full price for partial results, and that lost potency compounds over weeks of a treatment protocol. Understanding temperature thresholds is not just about safety. It is about protecting your investment.
The exact temperature thresholds for every semaglutide formulation
Not all semaglutide is created equal when it comes to heat tolerance. The formulation, concentration, excipients, and packaging all influence how long the medication can survive outside refrigeration. Here are the exact numbers you need to know.
Unopened semaglutide (all formulations)
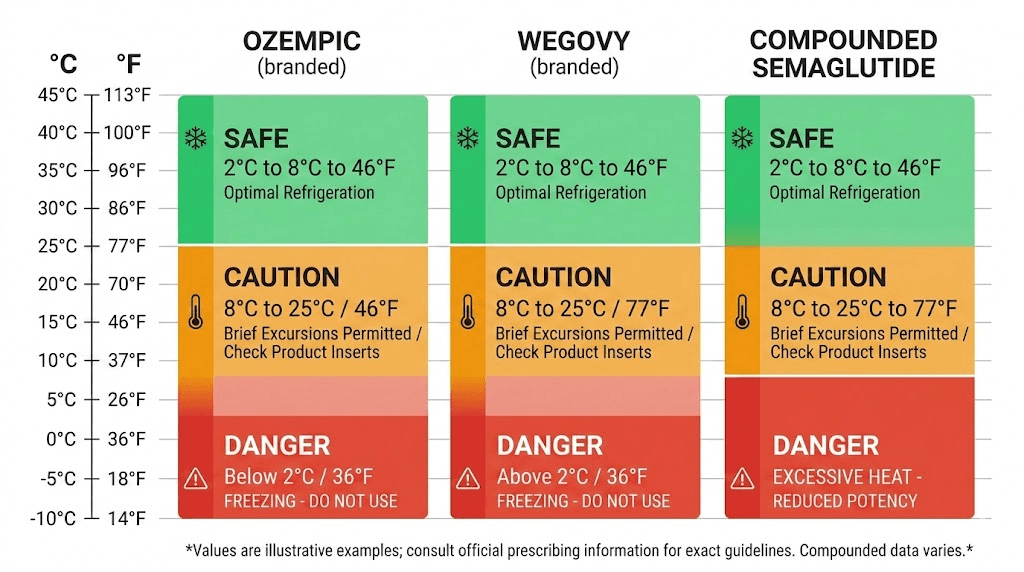
The universal rule for unopened semaglutide is simple. Store it at 36-46 degrees Fahrenheit, which is 2-8 degrees Celsius. This is standard refrigerator temperature. Every manufacturer, every pharmacy, and every clinical guideline agrees on this range. Unopened vials and pens should remain refrigerated from the moment they leave the manufacturing facility until you are ready to use them. If your compounded semaglutide needs refrigeration, this is the range to maintain.
Never freeze semaglutide. Freezing causes ice crystal formation that physically tears apart the peptide structure. While heat causes gradual denaturation, freezing causes immediate mechanical damage that is far worse. If your semaglutide arrived frozen, that is actually a bigger problem than if it arrived warm.
Ozempic (brand-name, Novo Nordisk)
Ozempic has the most generous room-temperature tolerance of the brand-name options. Once you start using a pen, Ozempic can be stored at room temperature up to 86 degrees Fahrenheit (30 degrees Celsius) for 56 days. That is eight full weeks outside the refrigerator, which gives you substantial flexibility for daily use and even traveling with semaglutide.
The 56-day window applies only after first use. Unopened Ozempic pens should stay refrigerated. And the 86-degree threshold is a hard ceiling, not a recommendation. Temperatures above 86 degrees Fahrenheit accelerate degradation significantly, and the 56-day timeline shortens dramatically with each degree above that mark.
Wegovy (brand-name, Novo Nordisk)
Wegovy is less forgiving than Ozempic despite containing the same active ingredient. Wegovy can be stored at room temperature up to 86 degrees Fahrenheit (30 degrees Celsius) for only 28 days. That is half the tolerance window of Ozempic, and the reason comes down to formulation differences, concentration variations, and the preservative system used in each product.
If your Wegovy arrived warm, you have a tighter margin for error. Combine shipping time with any additional room-temperature exposure during your use period, and you could exceed that 28-day window faster than you realize. Track the total time your Wegovy has spent outside refrigeration carefully, because those hours add up in ways that affect your first month results and beyond.
Compounded semaglutide
Compounded semaglutide from pharmacies like Empower Pharmacy, Olympia Pharmacy, or Direct Meds has different stability profiles than the brand-name products. Stability testing on compounded formulations shows that semaglutide can tolerate temperatures up to 95 degrees Fahrenheit (35 degrees Celsius) for 4 days and up to 77 degrees Fahrenheit (25 degrees Celsius) for 15 days while maintaining acceptable potency.
These numbers come from forced degradation studies that compounding pharmacies conduct as part of their quality assurance processes. The Beyond-Use Date (BUD) for most compounded semaglutide formulations is up to 60 days when stored under proper refrigeration, though individual pharmacies may assign shorter dating based on their specific testing. If you are wondering how long compounded semaglutide lasts in the fridge, the BUD on your vial label is your definitive answer.
One encouraging data point comes from stability studies on related formulations. A tirzepatide, glycine, and B12 compound tested at 104 degrees Fahrenheit (40 degrees Celsius) for 7 full days maintained 99% potency. While this was a different peptide, it suggests that well-formulated GLP-1 compounds with appropriate stabilizers can withstand significant heat exposure. Many compounded semaglutide formulations include glycine or B12 as stabilizing agents, which may improve heat tolerance beyond what the base peptide alone can handle.
Quick reference temperature table
Formulation | Storage temp | Room temp limit | Max duration at room temp | Upper danger threshold |
|---|---|---|---|---|
Ozempic | 36-46 F (2-8 C) | Up to 86 F (30 C) | 56 days | Above 86 F (30 C) |
Wegovy | 36-46 F (2-8 C) | Up to 86 F (30 C) | 28 days | Above 86 F (30 C) |
Compounded (liquid) | 36-46 F (2-8 C) | Up to 77 F (25 C) | 15 days | Above 95 F (35 C) |
Compounded (lyophilized) | 36-46 F (2-8 C) | Up to 77 F (25 C) | Varies by pharmacy | Above 95 F (35 C) |
Keep this table handy. If your semaglutide arrived warm, cross-reference the temperature your package likely reached with the formulation you are using. The combination of temperature and duration determines whether your medication is still viable, and those two variables interact in ways that are not always intuitive. A brief spike to 100 degrees Fahrenheit may cause less damage than a sustained 85-degree exposure over multiple days, depending on the formulation and stabilizers involved.
Brand-name versus compounded semaglutide heat tolerance
The differences between brand-name and compounded semaglutide go beyond price. They extend into stability, packaging, and heat resilience in ways that directly affect what happens when your medication arrives warm.
Why brand-name products have different stability profiles
Novo Nordisk, the manufacturer of Ozempic and Wegovy, invests millions in stability testing. Their formulations include proprietary excipient blends designed to protect the semaglutide molecule during temperature fluctuations. The pre-filled pen delivery system also reduces exposure to light, air, and contaminants that can accelerate degradation. This is one reason Ozempic gets that generous 56-day room temperature allowance while many compounded versions have shorter windows.
The concentration matters too. Ozempic pens contain semaglutide at relatively low concentrations in a carefully buffered solution. Compounded semaglutide often comes at higher concentrations, sometimes 5mg/mL or 10mg/mL, in multi-dose vials. Higher concentrations can actually improve stability in some cases because the peptide molecules essentially buffer each other, but they also create more opportunity for aggregation under heat stress.
Compounded formulations with added stabilizers
Many compounding pharmacies have gotten smart about heat stability. Formulations that include glycine and B12 tend to demonstrate better heat tolerance than plain semaglutide solutions. Glycine acts as a cryoprotectant and thermal stabilizer, helping maintain the peptide three-dimensional structure under stress. Methylcobalamin (B12) provides antioxidant protection that slows the oxidative degradation pathway heat accelerates.
Niacinamide-containing formulations and L-carnitine blends represent newer approaches to improving stability and adding complementary benefits. These additional compounds can influence the solution pH, ionic strength, and overall chemical environment in ways that either help or hinder the semaglutide molecule ability to survive temperature excursions.
The key takeaway is this. Not all compounded semaglutide is equally vulnerable to heat. A well-formulated product from a reputable pharmacy like Belmar Pharmacy with appropriate stabilizers may actually tolerate shipping temperatures better than a basic formulation from a less established source. Always ask your pharmacy about their stability testing data when placing an order, especially if you live in a warm climate.
Lyophilized versus liquid formulations
Lyophilized (freeze-dried) semaglutide has a significant advantage when it comes to shipping and heat tolerance. In its dried powder form, semaglutide is far more stable than when dissolved in solution. The lack of water removes the primary medium through which hydrolysis reactions occur, meaning the peptide can survive higher temperatures for longer periods before reconstitution.
If your compounded semaglutide arrives as a lyophilized powder that needs reconstitution, a warm arrival is less concerning than if it arrives as a pre-mixed liquid. The powder form gives you a built-in safety margin. Once you reconstitute the powder with bacteriostatic water, however, the clock starts ticking and proper refrigeration becomes critical. Use the peptide reconstitution calculator to determine exact mixing ratios for your specific vial.
How to assess if your semaglutide is still safe after heat exposure
Your semaglutide arrived warm. Now what? Before you make any decisions, you need to assess the situation systematically. Panic leads to waste, and assumptions lead to risk. Neither is acceptable when dealing with an expensive medication.
The visual inspection protocol
Start with what you can see. Hold the vial or pen up to a light source and examine the solution carefully. Healthy semaglutide should be clear, colorless to slightly yellow, and completely free of visible particles. The normal color of semaglutide ranges from water-clear to a very faint straw tint depending on the concentration and additives.
Look for these red flags.
Cloudiness or haziness. If the solution looks milky, foggy, or anything other than crystal clear, degradation has likely occurred. Cloudiness indicates protein aggregation, meaning semaglutide molecules have clumped together into larger particles that scatter light. This is a sign of significant structural damage, and cloudy semaglutide should not be injected.
Visible particles. Hold the vial still and look for tiny floating specks, fibers, or clumps. Then gently swirl (do not shake) and look again. Particles can indicate denatured protein precipitating out of solution, contamination, or crystallization. Any visible particulate matter disqualifies the medication from use.
Color changes. If the solution has turned red, pink, amber, dark yellow, or brown, chemical degradation has occurred. Oxidation of the peptide backbone or its amino acid side chains produces colored degradation products. Slight color change in compounded formulations with B12 is expected and normal since methylcobalamin has a natural pinkish hue. But dramatic color shifts in any formulation signal trouble.
Unusual consistency. The solution should flow freely like water when you tilt the vial. If it appears thick, gel-like, or viscous in ways that differ from when you first received it, the protein may have aggregated into a higher-order structure that changes the solution physical properties.
What the visual inspection cannot tell you
Here is the hard truth. Visual inspection catches severe degradation but misses moderate damage entirely. Semaglutide can lose 20-30% of its potency without any visible change to the solution. The molecule can partially denature, losing enough binding affinity to reduce effectiveness while still appearing perfectly clear and colorless. This is why temperature history matters so much. You cannot see the difference between full-potency and reduced-potency semaglutide, but your body will notice it through reduced appetite suppression, slower weight loss, or a plateau in progress.
Assessing temperature exposure duration
The next step is estimating how long your semaglutide was exposed to elevated temperatures. Check the tracking information for your shipment. When was it shipped? When was it delivered? How long did it sit in your mailbox or on your doorstep before you retrieved it? These details matter enormously.
A package shipped Monday evening with two-day delivery that arrives Wednesday afternoon has spent roughly 44 hours in transit. If the ice packs lasted 24-30 hours (which is typical), the medication may have been without active cooling for 14-20 hours. In moderate weather, that is unlikely to cause significant damage. In summer heat with ambient temperatures exceeding 95 degrees Fahrenheit, those same 14-20 hours could push the medication past safe thresholds.
Consider the conditions during transit too. Was the package sitting in a metal mailbox in direct sunlight? Mailbox temperatures can reach 150 degrees Fahrenheit on a hot day. Was it left on a concrete porch? Surface temperatures on sun-exposed concrete regularly exceed 130 degrees. Or was it in a shaded area or inside a building lobby? These scenarios produce dramatically different temperature exposures even on the same day. If you also think about how temperature issues relate to overall semaglutide shelf life, you can see why these variables matter so much.
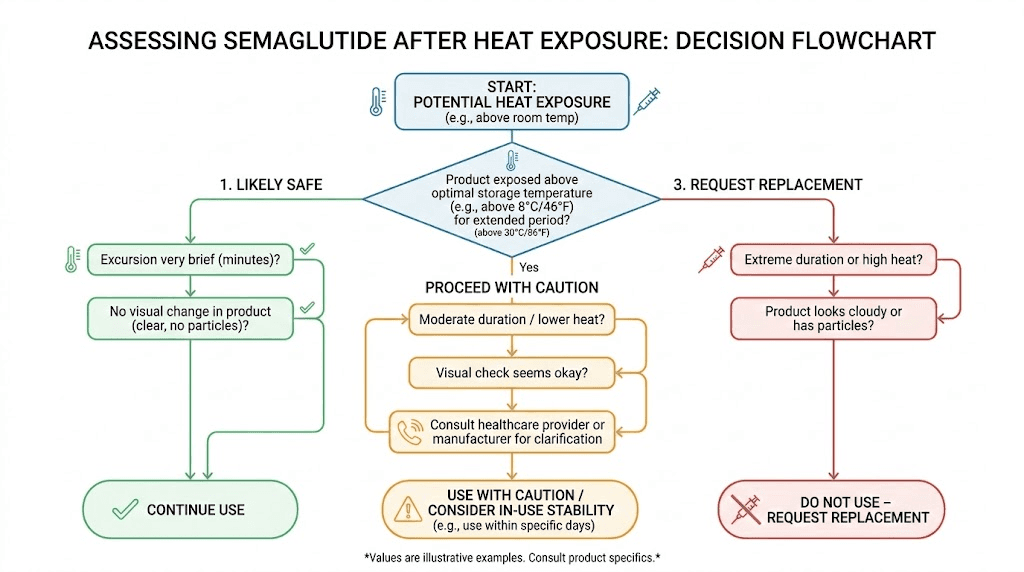
The decision framework
Use this systematic approach to decide whether to use or replace your semaglutide after heat exposure.
Likely safe to use: The ice packs are partially melted but still cool. The medication feels cool or room temperature (below 77 degrees Fahrenheit). Visual inspection shows a clear, colorless solution with no particles. Transit time was under 48 hours. Weather conditions during shipping were moderate. This scenario typically results in minimal potency loss, likely under 5%.
Proceed with caution: Ice packs are fully melted and warm. The medication feels warm to the touch but not hot. Visual inspection is normal. Transit time exceeded 48 hours or weather was warm (80-95 degrees Fahrenheit). In this case, the medication may have lost some potency but is likely still usable. You might notice slightly delayed appetite suppression or somewhat reduced effectiveness. Monitor your response carefully and contact your pharmacy.
Request replacement: The package is hot to the touch. Visual inspection shows cloudiness, particles, or color change. Transit exceeded 72 hours in warm weather. The medication was exposed to direct sunlight or extreme heat (above 100 degrees Fahrenheit) for any significant duration. Do not use this medication. Contact your pharmacy immediately for a replacement.
Step-by-step protocol when your semaglutide arrives warm
Do not panic. Do not throw it away immediately. And do not inject it without thinking. Follow this protocol in order.
Step 1: Document everything immediately
Before you do anything else, take photographs. Photograph the outer packaging, the insulation, the ice packs (noting their condition), the vial or pen, and the solution clarity. Take a photo of the shipping label with the tracking number visible. If you have an infrared thermometer or kitchen thermometer, measure and photograph the temperature of the medication, the ice packs, and the inside of the insulated container. This documentation is critical if you need to request a replacement or file a claim.
Write down the time you retrieved the package. Check your tracking information and note the delivery time. Calculate how long the package sat unattended. All of this creates a timeline your pharmacy will need.
Step 2: Refrigerate immediately
Get the semaglutide into your refrigerator right away. Even if you suspect it has been damaged, refrigeration stops any ongoing degradation. Place the vial or pen in the main body of your refrigerator (not the door, where temperatures fluctuate more) at 36-46 degrees Fahrenheit. Do not put it in the freezer. Proper refrigerator storage gives you time to assess the situation without further compromising the medication.
Step 3: Perform the visual inspection
After the medication has cooled in the refrigerator for 30-60 minutes, remove it and perform the visual inspection described above. Some cloudiness that appeared during heat exposure may actually resolve once the medication cools, as certain types of protein aggregation are reversible at lower temperatures. Check for clarity, color, and particulates. Compare what you see to the normal appearance of semaglutide.
Step 4: Contact your pharmacy within 24 hours
Whether the medication looks fine or not, contact your pharmacy within 24 hours of delivery. Report the warm arrival, share your photos, and describe the condition of the ice packs and packaging. Most pharmacies have specific protocols for handling temperature excursion reports. They may ask additional questions about the ambient temperature, how long the package was unattended, and whether you have used any of the medication yet.
Important: do not wait. Many pharmacies have strict timeframes for reporting temperature issues. Waiting three or four days to report a problem makes it much harder to get a replacement.
Step 5: Follow your pharmacy guidance
Your pharmacy will either advise you to use the medication, discard it, or send a replacement. If they offer a replacement, ask about expedited shipping with better insulation. If they advise using it, ask about expected potency and whether you should watch for signs of reduced effectiveness. If you are on a specific dosing schedule and need to delay injections while waiting for a replacement, ask your pharmacy how to handle the gap in treatment. A short break is manageable, but knowing the right approach matters for restarting semaglutide effectively.
Step 6: Monitor your response if you decide to use it
If you and your pharmacy decide the medication is likely fine, pay close attention to your body response after the next few injections. Track your appetite levels, weight changes, and any changes in how you feel. If you notice that your energy levels shift or your appetite returns earlier than expected between doses, the medication may have lost more potency than the visual inspection suggested. Document these observations and share them with your prescriber.
How to get a replacement from your pharmacy
Getting a replacement for heat-damaged semaglutide is not always straightforward, but knowing the process makes it much easier.
Compounding pharmacies
Most reputable compounding pharmacies have policies for replacing medications damaged during shipping. Pharmacies like Empower and Olympia often include temperature monitoring devices in their packaging or use shipping methods with built-in tracking. When you report a temperature excursion, they can cross-reference your claim with their shipping records.
Be direct and specific when you contact them. Tell them exactly when the package was delivered, what condition the ice packs were in, what the medication felt and looked like, and include your photographs. Do not exaggerate. Do not claim the medication was boiling hot if it was just warm. Honest reporting builds credibility and makes the replacement process smoother.
Most compounding pharmacies will reship at no additional cost for genuine temperature excursions, especially during summer months when the problem is well known. Some may ask you to return the original medication. Others will simply ask you to discard it. Follow their instructions exactly.
Brand-name through insurance or retail pharmacy
If you receive Ozempic or Wegovy through a retail pharmacy or mail-order service, the replacement process involves a few more steps. Contact the dispensing pharmacy first. They may need to coordinate with the shipping carrier and your insurance company. If the pharmacy uses a mail-order service like CVS Caremark, Express Scripts, or OptumRx, call their customer service line and request a temperature excursion investigation.
Insurance replacements can take longer because the payer needs to authorize a new fill outside your normal schedule. Be patient but persistent. Document everything in writing, including the names of representatives you speak with and any reference numbers they provide. You have a right to receive medication that was stored and shipped according to manufacturer specifications.
Telehealth providers
If you receive semaglutide through a telehealth weight loss service, contact their support team directly. These companies typically work with specific compounding pharmacies and have established processes for handling shipping issues. They may be faster than traditional pharmacies because maintaining customer satisfaction is central to their subscription-based business models. When your replacement arrives, double-check the expiration date and Beyond-Use Date on the new vial to make sure you are receiving fresh medication.
What if the pharmacy refuses to replace it?
This happens sometimes, and it is frustrating. If a pharmacy refuses your replacement request, escalate to a supervisor. Provide your temperature documentation and photos. Reference the manufacturer storage requirements (36-46 degrees Fahrenheit for storage, specific room-temperature limits for each formulation). If the pharmacy still refuses, file a complaint with your state Board of Pharmacy. Pharmacies have a legal obligation to ship medications in compliance with storage requirements.
If you are using a telehealth service and they refuse to help, consider switching providers. There are many options available, and reputable vendors stand behind their shipping practices.
What heat actually does to semaglutide at the molecular level
Understanding the science behind heat damage helps you make better decisions about your medication. This is not just academic knowledge. It directly informs how you assess risk.
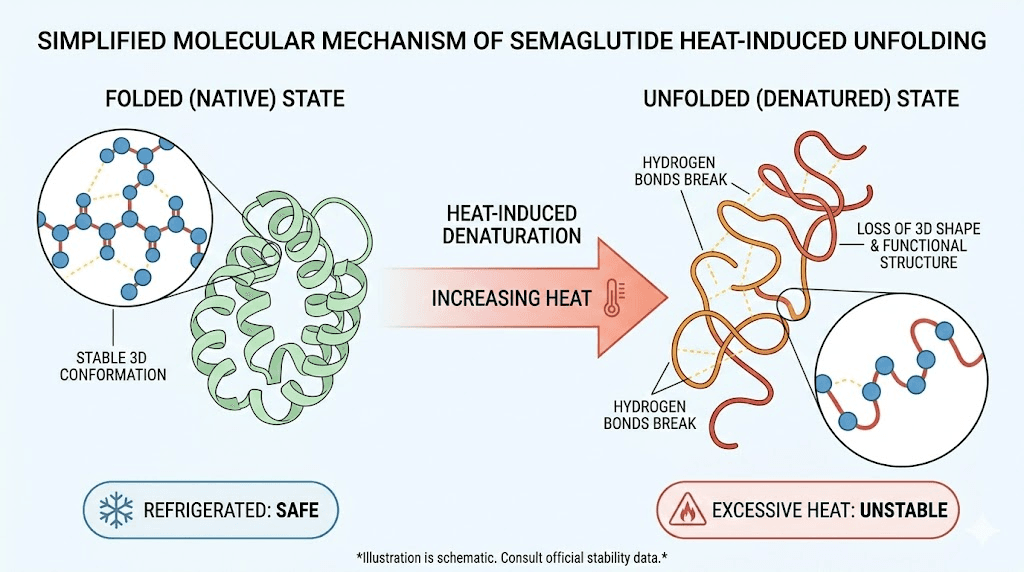
Peptide denaturation explained
Semaglutide is a 31-amino-acid peptide with a specific three-dimensional structure held together by several types of molecular forces. Hydrogen bonds connect different parts of the peptide backbone. Hydrophobic interactions cause oil-loving amino acid side chains to cluster together in the molecule interior. Van der Waals forces provide additional structural support. And a fatty acid chain (a C18 modification) anchors the molecule to albumin in the bloodstream, which is what gives semaglutide its long half-life.
Heat adds kinetic energy to the system. As molecules vibrate faster at higher temperatures, the weaker bonds begin to break. Hydrogen bonds go first, followed by hydrophobic interactions. The molecule begins to unfold, exposing its hydrophobic core to water. Once exposed, these hydrophobic regions seek each other out, causing multiple semaglutide molecules to stick together in a process called aggregation. This is the clumping that eventually becomes visible as cloudiness.
The process is generally irreversible. Unlike some proteins that can refold after mild heat exposure, peptide drugs typically lose their functional structure permanently once denaturation passes a critical threshold. This is why what happens when semaglutide gets warm matters so much for your treatment outcomes.
The three degradation pathways
Oxidation. Heat accelerates the oxidation of methionine and tryptophan residues in the semaglutide molecule. Oxidized semaglutide has reduced receptor binding affinity, meaning it does not activate GLP-1 receptors as effectively. This is one of the most common degradation pathways and one of the hardest to detect visually.
Hydrolysis. Water molecules attack the peptide bonds that link amino acids together, breaking the molecule into fragments. Heat dramatically increases the rate of hydrolysis. Fragmented semaglutide is biologically inactive because the receptor requires the full peptide sequence to bind properly. Hydrolysis products can sometimes trigger injection site reactions because the immune system may recognize fragments as foreign.
Aggregation. As described above, denatured molecules stick together. Aggregated semaglutide is not only inactive but can potentially cause injection site reactions or immune responses. Large aggregates are the particles you can sometimes see floating in degraded solutions. Smaller aggregates (called sub-visible particles) are too small to see but still problematic.
The safety question: is heat-damaged semaglutide dangerous?
Here is the most important point many articles miss. Denatured semaglutide does not become toxic. It becomes less effective. The degradation products of semaglutide are not known to cause dangerous adverse reactions beyond potential injection site reactions from aggregated particles. You will not poison yourself by injecting partially degraded semaglutide.
However, "not toxic" does not mean "worth using." Reduced potency means you are paying full price for partial results. It means your weight loss timeline stretches out. It means your semaglutide might take longer to work than expected. And it means you might mistakenly conclude that the medication does not work for you, when the real problem is degradation.
Lab stability data worth knowing
Published stability studies on semaglutide provide concrete numbers. The medication remains stable at 25 degrees Celsius (77 degrees Fahrenheit) for at least 28 days, with minimal potency loss in properly formulated products. At 40 degrees Celsius (104 degrees Fahrenheit), significant degradation occurs within 28 days, with potency drops accelerating after the first week. Interestingly, semaglutide remained stable for 3 hours at 80 degrees Celsius (176 degrees Fahrenheit) in controlled laboratory conditions, which suggests the molecule is fairly resilient to brief temperature spikes even at extreme levels.
The pH of the solution plays a critical role too. Semaglutide has an isoelectric point at pH 5.4, meaning the molecule carries no net charge at this pH and is most prone to aggregation. The worst degradation occurs at pH 4.5-5.5. Properly formulated products buffer the solution away from this dangerous range, which is another reason why quality compounding matters so much when you are considering how long your semaglutide vial stays good.
Understanding cold chain shipping for GLP-1 medications
Cold chain shipping is the backbone of safe medication delivery for temperature-sensitive drugs. Understanding how it works helps you evaluate whether your semaglutide was adequately protected during transit.
How cold chain packaging works
A properly designed cold chain shipping container has several layers. The outer box provides structural protection. Inside, an insulated liner (typically expanded polystyrene foam or insulated panels) creates a thermal barrier that slows heat transfer from the outside environment. Gel packs or phase-change materials sit inside this insulated space, absorbing heat energy as they melt and keeping the interior temperature low.
The design goal is to maintain 2-8 degrees Celsius inside the container for a specific duration, usually 24-48 hours for standard shipments or 48-72 hours for extended-range packaging. The system does not stay cold indefinitely. It is engineered to maintain temperature for long enough to cover the expected shipping time plus a safety margin.
Why ice packs melting does not automatically mean your medication is ruined
This is a critical misconception that causes unnecessary waste. Ice packs melting in transit is completely normal and expected after 24 hours. The phase-change materials in gel packs are designed to absorb heat as they transition from solid to liquid. A melted ice pack has done its job. The question is not whether the ice packs melted but whether the medication temperature exceeded safe limits after they melted.
Think of it this way. A refrigerator full of cold items will stay cool for hours after losing power because the cold mass inside absorbs ambient heat slowly. The insulated container in your shipping box works the same way. Even after the gel packs are fully melted and warm, the insulated space maintains a lower temperature than the outside environment for additional hours. The medication does not instantly jump to ambient temperature the moment the ice packs give out.
For most standard two-day shipments in moderate weather, the ice packs melt partway through transit but the medication arrives within safe temperature ranges. The problem occurs when shipping takes longer than expected, when weather is exceptionally hot, or when the package sits unattended in a hot location after delivery. These are the scenarios where your semaglutide that was left out is most at risk.
What good versus bad cold chain shipping looks like
Signs of good cold chain shipping: Thick insulated liner that fills the entire box. Multiple gel packs arranged around the medication on all sides. The medication is separated from direct contact with the gel packs (to prevent freezing). The outer box is clean, intact, and appropriately sized. Some pharmacies include a temperature indicator card that changes color if thresholds are exceeded.
Signs of poor cold chain shipping: Thin or absent insulation. Single gel pack tossed in loosely. Oversized box with lots of empty space (air gaps reduce insulation effectiveness). Medication in direct contact with a frozen gel pack (risk of freezing damage). No temperature monitoring. If you consistently receive poorly packaged shipments, switch pharmacies. The best peptide vendors invest in proper cold chain logistics.
Comparing tirzepatide shipping concerns
If you are also considering or using tirzepatide, the cold chain concerns are very similar. What happens when tirzepatide gets warm follows the same general degradation pathways as semaglutide, though the specific thresholds differ slightly. Tirzepatide refrigeration requirements are comparable, and tirzepatide fridge storage duration depends on the formulation and manufacturer. Some patients explore switching from semaglutide to tirzepatide and need to manage shipping logistics for both medications during the transition period.
Summer shipping survival guide for semaglutide
Summer is when most shipping problems occur. Temperatures inside delivery trucks can exceed 140 degrees Fahrenheit. Metal mailboxes become ovens. And even a few hours on a sun-exposed porch can push medication past safe limits. Here is how to protect your semaglutide during the hottest months.
Before you order
Request expedited shipping. Two-day or overnight shipping dramatically reduces exposure time. Yes, it costs more. But replacing a ruined vial of semaglutide costs even more. If your pharmacy or telehealth provider does not offer expedited options for temperature-sensitive medications during summer, that is a red flag about their quality standards.
Ask about packaging upgrades. Some pharmacies offer enhanced cold chain packaging for warm-weather shipments, including extra gel packs, thicker insulation, or phase-change materials rated for longer durations. Ask specifically what packaging changes they make during summer months. Pharmacies that ship the same way in January and July are not adequately protecting your medication.
Ship to a climate-controlled location. If possible, have your semaglutide shipped to your workplace, a staffed mailroom, or a location where someone can bring the package inside promptly upon delivery. Avoid shipping to a doorstep or mailbox that receives direct afternoon sun. This single change eliminates the most dangerous part of the delivery process, the hours between delivery and retrieval.
Time your orders strategically. Order early in the week so your package ships Monday or Tuesday and arrives Tuesday through Thursday. Avoid shipments that might spend a weekend sitting in a warehouse, delivery truck, or on your doorstep. Monday and Tuesday ship dates reduce the risk of weekend delays.
On delivery day
Track your package. Set up delivery notifications so you know the moment your semaglutide arrives. Most carriers offer text alerts, email notifications, and app-based tracking. The faster you retrieve the package after delivery, the less time it spends in uncontrolled temperatures.
Retrieve immediately. Do not let the package sit. If you cannot be home for delivery, arrange for someone else to bring it inside. Even 30 minutes in a hot mailbox can raise the temperature inside the shipping container significantly. If your mailbox hits 150 degrees Fahrenheit in direct sun, the insulation buys you time but not infinite time.
Open and refrigerate promptly. As soon as you have the package, open it and get the medication into your refrigerator. Check the gel packs and note their condition. Follow the assessment protocol outlined earlier in this guide to determine if any temperature excursion occurred. Being prompt and consistent about this protects both your semaglutide refrigerator shelf life and its effectiveness.
Long-term summer strategies
If you live in a hot climate and receive regular semaglutide shipments, consider these ongoing strategies. Ask your pharmacy about seasonal shipping upgrades that automatically apply during warm months. Some pharmacies add extra gel packs or use upgraded insulation between May and September without any additional request needed.
Consider ordering larger quantities less frequently during summer to reduce the number of shipments exposed to heat. A three-month supply shipped once is better than monthly shipments that each carry heat risk. Check with your pharmacy and insurance about multi-month dispensing options. This approach also aligns well with understanding how long you stay on semaglutide for your treatment plan.
If you notice a pattern of warm arrivals with a specific pharmacy, document each incident and escalate. Pattern documentation carries more weight than individual complaints. After three warm arrivals, most pharmacies will either upgrade your packaging permanently or switch you to a different shipping method.
How to properly store semaglutide after delivery
Getting semaglutide home safely is only half the battle. How you store it determines how long it stays potent and effective.
Refrigerator storage best practices
The ideal location inside your refrigerator matters more than most people realize. Place your semaglutide in the main body of the refrigerator, toward the back, where temperatures are most stable. The door shelves experience significant temperature fluctuations every time you open the refrigerator, sometimes swinging 10-15 degrees in a matter of seconds. The back of a middle shelf maintains the most consistent 36-46 degree Fahrenheit range.
Do not store semaglutide directly against the back wall where cooling elements might cause localized freezing. A thin layer of frost on the back wall of your refrigerator means temperatures in that immediate area periodically drop below freezing. Keep the vial or pen at least 2-3 inches from the back wall. And remember, never freeze. If you have ever accidentally left semaglutide out overnight, get it back into the refrigerator as soon as you notice and assess using the guidelines in this article.
After opening: room temperature guidelines
Once you start using your semaglutide, you have the option of keeping it at room temperature for convenience. The rules depend on your formulation. Ozempic can stay at room temperature (below 86 degrees Fahrenheit) for up to 56 days. Wegovy allows 28 days at room temperature. Compounded semaglutide varies by pharmacy, so check your specific label. Many compounded formulations recommend continued refrigeration even after opening to maximize the usable timeframe.
If you choose room temperature storage for convenience, keep the medication away from windows, heat sources, and direct sunlight. A bathroom medicine cabinet can be problematic because showers and baths create temperature and humidity fluctuations. A bedroom drawer or kitchen cabinet away from the stove is better. The complete peptide storage guide covers these considerations in more detail.
Protecting reconstituted semaglutide
If you are working with lyophilized semaglutide that you reconstitute yourself, storage after mixing requires extra care. Once you add bacteriostatic water to your peptide, the clock starts on a much shorter shelf life. Reconstituted semaglutide should be refrigerated immediately and used within the timeframe specified by your pharmacy, which typically ranges from 28-60 days depending on the formulation and concentration.
Use the semaglutide reconstitution chart to determine the correct amount of bacteriostatic water for your vial size. Incorrect dilution ratios can affect stability. An overly dilute solution may degrade faster, while an overly concentrated solution may have different aggregation characteristics. Getting the reconstitution right from the start is essential for maintaining potency throughout the vial lifespan. The reconstitution calculator takes the guesswork out of this step.
After reconstitution, store the vial upright in your refrigerator. Do not store it on its side, as this increases the surface area of solution exposed to air in the vial headspace, accelerating oxidative degradation. If your vial came with an aluminum crimp cap and rubber stopper, leave them in place. Each time you withdraw a dose, use a new sterile needle to minimize contamination risk. Proper post-reconstitution storage can mean the difference between a vial that stays potent for its full life and one that degrades prematurely.
Travel and temperature excursions with semaglutide
Shipping is not the only time semaglutide faces temperature challenges. Travel introduces a whole new set of risks, from hot cars to airport security to tropical destinations.
Car travel
Never leave semaglutide in a parked car. Vehicle interior temperatures can reach 170 degrees Fahrenheit within 30 minutes on a sunny day, even when the outside temperature is only 80 degrees. The dashboard and glove compartment can exceed 200 degrees. This is far beyond any safe threshold for any semaglutide formulation. Even the trunk, while cooler than the cabin, can reach 130-140 degrees.
When driving, keep your semaglutide in an insulated travel case with a cold pack. Place it on the floor of the passenger compartment, not in the trunk, because the passenger area benefits from air conditioning. If you stop for errands, take the medication with you in its insulated case. Never, under any circumstances, leave it in the vehicle. The complete travel guide for semaglutide covers every scenario in detail.
Air travel
Airplane cargo holds are not temperature controlled in the same way as the cabin. Temperatures in cargo areas can drop well below freezing at cruising altitude, which is actually more dangerous for semaglutide than moderate heat. Always carry your semaglutide in your carry-on luggage, never in checked bags.
TSA allows injectable medications in carry-on bags. You do not need a specific letter from your doctor, though having your prescription label visible on the vial or pen helps speed things along. Small gel packs are permitted for keeping medication cool. Declare your medication at the security checkpoint if asked, but most experienced TSA agents recognize injectable medications and process them quickly.
For long flights to warm destinations, bring an insulated medication travel wallet with a small gel pack. These wallets maintain cool temperatures for 8-12 hours, which covers even the longest commercial flights. Once you arrive, get the medication into a refrigerator as soon as possible. Hotel mini-fridges work, though they tend to run slightly warmer than home refrigerators. Set the mini-fridge to its coldest setting and check the temperature with a thermometer if possible.
Managing dose timing during travel
Travel disruptions can complicate your injection schedule. If you normally inject on a specific day and your travel throws that off, you have some flexibility. Semaglutide weekly injections can be given up to two days before or after the scheduled day without losing effectiveness. Adjust your timing to accommodate travel rather than skipping a dose entirely. You can even split your dose across two days if that works better for your travel schedule, though discuss this approach with your prescriber first.
Pack enough medication for your entire trip plus a buffer. Running out while traveling means finding a pharmacy in an unfamiliar area, and there is no guarantee you can get your prescription filled quickly. For extended trips, understanding how long your medication can stay out of the fridge helps you plan around situations where refrigeration is temporarily unavailable.
Beach, camping, and outdoor adventures
Outdoor activities present unique storage challenges. At the beach, never leave medication in a bag on the sand or in direct sunlight. Sand temperatures can exceed 150 degrees on hot days. Use a hard-sided cooler with ice packs and keep it in the shade. At campsites without electricity, a quality insulated cooler with frozen water bottles can maintain medication-safe temperatures for 24-48 hours depending on ambient conditions.
For backpacking or situations where carrying a cooler is impractical, consider the room-temperature windows described earlier. If your Ozempic is already in use, you have 56 days at temperatures up to 86 degrees. A weekend camping trip in moderate weather falls well within those parameters. Just keep the medication in the shade and insulated from direct heat sources.
Common mistakes that ruin semaglutide potency
Heat exposure during shipping is not the only way semaglutide loses potency. Many people unknowingly reduce their medication effectiveness through everyday handling mistakes.
Mistake 1: Storing in the refrigerator door
The refrigerator door is the worst location for temperature-sensitive medications. Every time you open the door, that shelf swings out into room-temperature air. Temperature monitoring studies show that door shelves can fluctuate between 35 and 55 degrees Fahrenheit throughout a typical day, with brief spikes even higher during extended door-open events like loading groceries. These repeated temperature cycles accelerate degradation far more than a single sustained temperature would.
Move your semaglutide to the middle of a main shelf. Today.
Mistake 2: Freezing by accident
Refrigerator temperature varies by location inside the unit. The area directly in front of the cooling vent or against the back wall can periodically drop below 32 degrees Fahrenheit, especially in older or poorly calibrated refrigerators. If your semaglutide freezes, the ice crystals physically rupture the peptide structure in a way that is irreversible and complete. Frozen semaglutide should not be used even if it thaws and looks normal. This applies equally to semaglutide stored long-term in the fridge.
Mistake 3: Shaking the vial
Vigorous shaking introduces air bubbles into the solution. Those bubbles create air-liquid interfaces where semaglutide molecules adsorb and denature. Every shake reduces potency slightly. Over weeks of daily agitation, the cumulative effect can be meaningful. When mixing or checking your semaglutide, swirl gently. Never shake. This is especially important when you reconstitute semaglutide from lyophilized powder. Use the semaglutide mixing chart for proper technique.
Mistake 4: Using expired medication
Expiration dates and Beyond-Use Dates exist for a reason. Stability testing shows that peptide potency declines over time even under perfect storage conditions. Using semaglutide past its labeled date means you are injecting medication with unknown potency reduction. Some people use expired semaglutide without realizing the risks. If you have accidentally taken expired semaglutide, the primary concern is reduced effectiveness rather than toxicity, but you should still discuss it with your prescriber.
Mistake 5: Inconsistent storage during use
Taking your semaglutide out of the refrigerator for injection, leaving it on the counter while you prepare, then forgetting to put it back for several hours. Doing this repeatedly, especially in a warm kitchen, adds up. Each room-temperature excursion chips away at the remaining potency. Create a habit: remove, prepare, inject, replace. Minimize the time your medication spends outside its storage temperature.
Mistake 6: Ignoring reconstitution ratios
For compounded lyophilized semaglutide, using the wrong amount of bacteriostatic water changes the solution concentration, pH, and stability profile. Too much water produces an overly dilute solution that may degrade faster. Too little water creates a highly concentrated solution that is prone to aggregation. Follow the 5mg reconstitution chart or the 10mg reconstitution chart precisely. The peptide reconstitution calculator removes all guesswork from this step and is one of the most popular free tools on SeekPeptides.
Mistake 7: Contaminating the vial
Using the same needle for multiple draws, touching the rubber stopper with bare fingers, or drawing from the vial in a non-sterile environment introduces bacteria that can degrade the peptide over time. Bacteriostatic water contains benzyl alcohol as a preservative, which helps, but it cannot compensate for gross contamination. Always use a new sterile needle for each draw and swab the stopper with an alcohol pad before inserting the needle. Understanding common peptide mistakes beginners make can save you from these errors.
How temperature issues affect your semaglutide results
You might wonder whether heat-damaged semaglutide is really that different in practice. The answer depends on how much potency was lost and what you are trying to achieve.
The potency-to-results relationship
Semaglutide dose-response curves are not linear. A 10% loss in potency does not simply produce 10% less weight loss. The relationship between dose and effect follows a sigmoid curve, meaning small potency losses at certain dose points might barely matter while the same percentage loss at other points could significantly reduce effectiveness. For someone on the lower maintenance doses, where they sit near the steep part of the dose-response curve, even modest potency reduction can noticeably affect how quickly semaglutide works.
If you are already experiencing a semaglutide plateau, using heat-damaged medication can make the problem dramatically worse. Reduced potency on top of metabolic adaptation creates a double barrier to continued progress.
Signs your semaglutide may have lost potency
Watch for these indicators after using medication that was exposed to heat. Your appetite returns earlier than usual between injections. You stop losing weight despite following your semaglutide diet plan consistently. Fatigue worsens or energy levels fluctuate more than expected. You experience less bloating or gastrointestinal effects than you did on previous doses, which paradoxically suggests the medication is weaker because those side effects correlate with drug activity.
None of these signs are definitive on their own. Many factors influence semaglutide effectiveness, including your diet, exercise, stress, sleep, and metabolic health. But if several of these indicators appear simultaneously after using heat-exposed medication, potency loss is a likely explanation.
What to do if you suspect reduced potency
Do not increase your dose to compensate for suspected potency loss. Self-adjusting doses without medical guidance is risky, and if the medication is not actually degraded, you could end up with a dose that causes excessive gastrointestinal side effects, dizziness, or excessive burping and nausea. Instead, contact your prescriber, explain the temperature excursion, and follow their guidance on whether to switch to a new vial or adjust your protocol.
Use the semaglutide dosage calculator to verify your current dosing and review the compounded semaglutide dose chart to make sure your protocol is appropriate for your goals. Sometimes what looks like a potency issue is actually a dosing issue, and fixing the root cause produces better results than chasing potency concerns.
Injection technique and heat-exposed semaglutide
How you inject semaglutide matters for results, and this is especially true if you are working with medication that may have partial potency reduction from heat exposure.
Maximizing absorption of every dose
Inject into subcutaneous fat, not muscle. Subcutaneous injection provides the slow, sustained absorption semaglutide was designed for. If you accidentally inject into muscle, the medication absorbs too quickly, producing a spike-and-crash pattern instead of steady levels. The best injection site for semaglutide is the abdomen, thigh, or upper arm, where subcutaneous fat is most accessible.
Proper injection technique with a syringe includes pinching a fold of skin, inserting the needle at a 45-90 degree angle (depending on the needle length and your body composition), injecting slowly, and holding the needle in place for 5-10 seconds after completing the injection to prevent medication from leaking out. These small details optimize the amount of semaglutide that reaches your system, which becomes critically important when working with medication that may already have reduced potency. Choosing the best injection site for weight loss can also influence absorption rates.
Dosing considerations with potentially compromised medication
If you and your prescriber decide to continue using heat-exposed semaglutide while awaiting a replacement, understand the dosing implications. Your semaglutide dosage in units remains the same on your syringe, but the effective dose you receive may be lower. This means you might temporarily experience what feels like being on a lower dose step, with less appetite suppression and slower weight loss. Refer to the syringe dosage chart to stay accurate with your measurements even in suboptimal conditions.
When your replacement medication arrives, do not start at a higher dose than what you were previously taking. Go back to your regular dose and give your body a full injection cycle (one week for weekly semaglutide) to recalibrate. You will likely notice a return of stronger appetite suppression and renewed progress within the first couple of doses on fresh medication. Understanding unit-to-milligram conversions and checking your dosing against the semaglutide units to mg reference helps keep your protocol precise.
Alternative formulations and their heat resilience
Not all ways of taking semaglutide carry the same shipping and storage risks. Understanding your options helps you choose the formulation best suited to your situation.
Oral and sublingual semaglutide
Oral semaglutide drops and sublingual formulations have different stability characteristics than injectable versions. Some oral formulations are designed to be stable at room temperature by default, which eliminates the cold chain shipping requirement entirely. The trade-off is that oral bioavailability is significantly lower than injectable, meaning you need a higher dose to achieve equivalent effects. The injectable versus oral comparison covers these differences in depth.
If you live in a hot climate and consistently struggle with temperature-damaged shipments, oral formulations may be worth discussing with your prescriber. The convenience of room-temperature storage and the elimination of cold chain risk can outweigh the bioavailability disadvantage for some patients.
Other GLP-1 options and their temperature profiles
Tirzepatide, the dual GIP/GLP-1 agonist sold as Mounjaro and Zepbound, has similar refrigeration requirements but may have slightly different heat tolerance based on its formulation. Tirzepatide shelf life and expiration guidelines are worth reviewing if you are considering alternatives. As mentioned earlier, a tirzepatide-glycine-B12 formulation maintained 99% potency at 104 degrees Fahrenheit for 7 days, which suggests excellent heat stability in well-formulated compounds.
The semaglutide versus tirzepatide comparison can help you evaluate which medication better fits your needs, including practical considerations like shipping and storage. Some patients find that the side effect profiles differ enough to make switching worthwhile for reasons beyond temperature concerns. SeekPeptides provides comprehensive comparison tools and protocols to help you make informed decisions about your GLP-1 medication.
When to be concerned versus when to relax
Not every warm package is a crisis. Knowing the difference between a minor temperature excursion and a genuine problem saves you stress, money, and unnecessary medication waste.
Situations where you can relax
Your package arrived with ice packs that are partially melted but still cool to the touch. The medication vial feels room temperature or slightly cool. The solution is clear with no particles. Standard two-day shipping in spring or fall weather. In this scenario, your medication almost certainly maintained safe temperatures for the vast majority of transit. Use it with confidence.
The ice packs are fully melted but the insulation is intact and the medication is not warm. Overnight shipping during moderate weather. This is normal. The ice packs did their job, and the insulation maintained an acceptable internal temperature even after the phase-change materials expired. The medication is fine.
You left your in-use semaglutide on the counter for a few hours in an air-conditioned home. Room temperature (68-76 degrees Fahrenheit) for a few hours is well within the safe window for all formulations. Put it back in the fridge and move on.
Situations where you should be concerned
The package feels distinctly hot to the touch. The medication vial is warm or hot. The insulation has collapsed or is minimal. Extended shipping during a heat wave. In this case, run through the full assessment protocol and contact your pharmacy.
The solution has changed color, shows particles, or appears cloudy. Regardless of shipping conditions, visual changes mean degradation has occurred. Do not use. Request a replacement.
The medication was left in a hot car, on a sun-exposed surface, or in a mailbox during extreme heat for more than 2-3 hours. The temperatures in these environments vastly exceed any safe threshold. Even brief exposure to 130-170 degrees can cause irreversible damage.
You notice reduced effectiveness after using medication from a warm shipment. Trust your body. If you are not losing weight on semaglutide and the timing correlates with a questionable shipment, the medication may be compromised. Discuss with your prescriber whether to continue or switch to a new supply. Looking at your before and after progress over time can help identify when effectiveness changed.
Interacting medications and heat sensitivity
If you are combining semaglutide with other medications, temperature damage to any one of them can affect your overall protocol.
Combination therapies to watch
Some patients combine semaglutide with other compounds. If you are taking phentermine and semaglutide together, the phentermine tablet has excellent heat stability (most tablets do), but the semaglutide component remains vulnerable. Those using berberine and semaglutide together should know that berberine supplements also tolerate heat well in their encapsulated form. The semaglutide is always the weak link in the chain.
For patients exploring whether semaglutide and tirzepatide can be used together, managing the cold chain for two temperature-sensitive injectables doubles the logistics challenge. Both medications require the same storage conditions, but receiving two separate shipments during summer doubles the risk of at least one arriving warm.
The alcohol question during warm-weather treatment
A side note related to warm weather and summer socializing. If you are wondering whether you can drink alcohol on semaglutide, the answer involves considerations beyond just medication interactions. Alcohol can worsen dehydration during hot weather, and some semaglutide side effects like dizziness and fatigue may intensify in the heat. Managing your medication storage and your overall health during summer months requires a holistic approach.
Long-term planning for medication stability
If you plan to be on semaglutide for months or longer, building good habits around medication handling pays dividends over time.
Creating your medication management routine
Set up a dedicated spot in your refrigerator for your medication. Use a small container or bag to keep it organized and prevent it from rolling around. Label it clearly so other household members know not to move it or confuse it with food items. Some people use a small insulated lunch bag inside the refrigerator as a dedicated medication zone, which adds an extra layer of thermal protection during normal door-opening events.
Track your injection dates, the condition of each new shipment when it arrives, and when you open each new vial or pen. This log helps you identify patterns with your pharmacy shipping quality and correlate any changes in your results with specific medication batches. It also helps if you ever need to reference your medication history for your prescriber, especially when discussing early experiences on semaglutide versus later responses.
Building a relationship with your pharmacy
A good pharmacy relationship is your best defense against temperature problems. Communicate proactively about your location, climate, and shipping preferences. Ask about their cold chain protocols and what upgrades they offer for summer shipping. Provide feedback on every shipment, positive or negative, so they can adjust their processes.
If you use a compounding pharmacy like Strive Pharmacy, IVIM Health, or others, understand that their shipping practices vary. Some invest heavily in packaging while others cut corners. The vendor comparison guide includes information about shipping quality among various providers.
Having a backup plan
If you depend on semaglutide for weight management, having a plan for when shipments are delayed or damaged reduces stress. Keep at least a one-week buffer supply if possible. Know whether your pharmacy offers emergency expedited shipping. Have your prescriber phone number readily available for urgent dosing questions. And understand the protocol for temporarily stopping semaglutide if you face a gap between a damaged shipment and its replacement. Knowing what to expect during even a brief pause in treatment helps you maintain your dietary plan and nutritional habits while waiting.
Special considerations for specific patient groups
Different people face different challenges when it comes to semaglutide temperature management.
People in rural or remote areas
If you live far from your pharmacy or in a rural area where delivery times are longer, your medication spends more time in transit and more time exposed to outdoor temperatures. Extended delivery windows of 3-5 days push beyond the design limits of standard cold chain packaging. Request enhanced packaging with extra gel packs. Consider overnight shipping even if it costs more. And have a neighbor or local contact who can retrieve your package if you are not home, since remote mailboxes often sit in full sun with no shade.
People in hot climates
Living in Arizona, Texas, Florida, or other hot states means dealing with extreme ambient temperatures for months at a time. Standard cold chain packaging may not be sufficient when outside temperatures exceed 110 degrees. Advocate forcefully with your pharmacy for enhanced packaging. Consider using a pharmacy with a distribution center closer to your location to reduce transit time. And seriously evaluate whether room-temperature-stable formulations like oral semaglutide might be more practical for your situation.
People who travel frequently
Frequent travelers face repeated temperature exposure events. Each trip introduces risk. Invest in a high-quality medical-grade insulated travel case. Research hotel refrigerator availability before booking. Carry a digital thermometer to check medication temperature after each transit period. Build a travel medication kit that is always ready to go so you never scramble to protect your semaglutide at the last minute. The semaglutide travel guide has packing lists and destination-specific advice.
People managing side effects
If you are already dealing with semaglutide side effects like insomnia, menstrual changes, or withdrawal symptoms between doses, using heat-compromised medication adds another variable that makes it harder to fine-tune your protocol. Knowing that your medication is fully potent removes one variable from the equation, making it easier to identify whether side effects are dose-related, timing-related, or caused by other factors. Peptide safety depends on both proper medication handling and proper dose management.
The economics of medication waste versus caution
There is a real tension between throwing away potentially fine medication and risking reduced results from degraded medication. Here is how to think about it rationally.
The cost of using degraded medication
Suppose your semaglutide lost 25% potency from heat exposure. You continue using it for a month before getting a replacement. During that month, your weight loss slows by roughly the same proportion, and you lose 3 pounds instead of the 4-5 you typically lose. The financial impact is not just the cost of the medication. It includes the extra month on your treatment plan, the emotional cost of frustration and doubt, and the potential for you to give up on a medication that was actually working, just not at full strength. People already questioning whether they can lose weight on semaglutide without exercise or whether the medication is right for them may incorrectly conclude that semaglutide is ineffective when the real problem was a warm shipment.
The cost of unnecessary waste
On the other hand, discarding medication that was perfectly fine wastes money and potentially delays your treatment. If your semaglutide experienced a brief, mild temperature excursion that caused negligible potency loss, throwing it away and waiting for a replacement creates a gap in your protocol. That gap can cause withdrawal effects, rebound appetite, and setbacks in your progress.
The rational approach is to use the assessment framework outlined earlier, consult with your pharmacy, and make a decision based on evidence rather than emotion. If the exposure was mild and the medication passes visual inspection, using it while requesting a replacement to have on hand is a reasonable middle ground. If the exposure was severe, discard it without hesitation.
How other peptides compare to semaglutide for heat sensitivity
If you work with multiple peptides, understanding the relative heat sensitivity of each helps you prioritize your cold chain efforts.
Most peptides share similar vulnerability to heat because they are all proteins with similar bond types holding their structures together. The fundamentals of how peptides work at the molecular level explain why temperature matters across the board. The general peptide storage guide covers principles that apply to all peptide-based medications. Understanding how peptides function helps contextualize why their three-dimensional structure is so critical and why heat disrupts their mechanism of action.
Some peptides are more resilient than others. Smaller peptides with fewer secondary structures tend to tolerate heat better because they have less complex folding to disrupt. Larger peptides and those with extensive post-translational modifications are generally more sensitive. Semaglutide falls in the middle range, more robust than some large proteins but more sensitive than small cyclic peptides. The peptide research section provides additional context on stability profiles across different peptide classes.
If you are exploring peptides beyond semaglutide, whether for weight loss, fat loss, or other goals, the same temperature management principles apply. Store properly, ship carefully, and assess any temperature excursions using the same systematic approach described in this guide.
Frequently asked questions
Is it safe to use semaglutide that was left out of the fridge overnight?
In most cases, yes. If your home is at normal room temperature (68-76 degrees Fahrenheit), an overnight excursion of 8-12 hours is well within the documented safe window for all semaglutide formulations. Ozempic tolerates up to 56 days at room temperature, and even the most conservative compounded formulations allow several days. Put it back in the refrigerator immediately and monitor the solution for any changes. Our complete guide to semaglutide left out overnight covers this scenario in detail.
Can I tell if my semaglutide is degraded just by looking at it?
You can detect severe degradation visually. Look for cloudiness, particles, or color changes, which indicate significant molecular breakdown. However, moderate degradation of 10-30% potency loss often produces no visible change. The solution can appear perfectly clear while containing partially denatured molecules. This is why understanding the temperature exposure history is so important. Review our guide to semaglutide appearance and color for a detailed visual reference.
Do I need to restart at a lower dose if my semaglutide was heat damaged?
Not necessarily. If you switch to a new, undamaged vial at the same dose, your body will respond to the full-potency medication normally. You do not need to re-titrate from the beginning. However, if you have been using degraded medication for several weeks and your body has partially adapted to the lower effective dose, the jump back to full potency might temporarily increase side effects. Monitor closely for the first one or two doses and consult your prescriber. Our guide on restarting semaglutide provides relevant protocol guidance.
Will my insurance cover a replacement if my medication arrived warm?
This varies by insurance plan and pharmacy. Most insurance companies will authorize an early refill for medications damaged during shipping, but you may need to provide documentation including photos, tracking information, and a statement from your pharmacy confirming the temperature excursion. Contact your insurance company directly and be prepared to escalate if the initial representative is unfamiliar with cold chain shipping requirements for injectable medications.
Should I switch from injectable to oral semaglutide to avoid shipping problems?
Oral semaglutide offers the advantage of room-temperature stability, but it comes with lower bioavailability, different dosing requirements, and potentially different side effect profiles. If repeated shipping problems are compromising your treatment, discuss the oral option with your prescriber. The injectable versus oral comparison can help you weigh the trade-offs.
How long can compounded semaglutide sit at room temperature before it is ruined?
Compounded semaglutide can tolerate room temperature (up to 77 degrees Fahrenheit) for approximately 15 days and temperatures up to 95 degrees Fahrenheit for about 4 days while maintaining acceptable potency. Beyond these thresholds, degradation accelerates significantly. For specific guidance based on your formulation and pharmacy, check the label or contact your pharmacy directly. Our guide on compounded semaglutide refrigeration covers this topic comprehensively.
Is frozen semaglutide worse than heated semaglutide?
Yes, generally. Freezing causes immediate, mechanical damage through ice crystal formation that physically ruptures peptide structures. Heat damage is typically gradual and depends on both temperature and duration. A semaglutide pen that was briefly frozen is more likely to be completely ruined than one that was warm for a few hours. Never freeze semaglutide, and if it arrived frozen, do not use it.
Can I use a home thermometer to check my semaglutide temperature?
Yes. A standard kitchen thermometer or infrared thermometer can help you assess the temperature of the medication, ice packs, and interior of the shipping container when your package arrives. An infrared thermometer is faster and does not require contact with the medication. Place a standard digital thermometer against the vial for about 30 seconds for an accurate reading. Any temperature at or below 77 degrees Fahrenheit (25 degrees Celsius) is reassuring for most formulations.
What if I used semaglutide that was definitely heat damaged, should I worry?
No need to worry about toxicity. Denatured semaglutide does not become poisonous. The primary consequence is reduced effectiveness, meaning the dose you injected simply did less than a full-potency dose would have. You may notice less appetite suppression or slower weight loss for that injection cycle. Resume your normal schedule with properly stored medication and your results will return to baseline. If you experience any unusual injection site reactions, such as increased redness, swelling, or itching, contact your prescriber.
Does semaglutide need to come to room temperature before injecting?
It is recommended but not required. Injecting cold semaglutide straight from the refrigerator can cause more discomfort at the injection site, including stinging and temporary redness. Letting the vial or pen sit at room temperature for 15-30 minutes before injection improves comfort without risking potency, since that brief exposure is well within safe limits for every formulation. This brief warm-up applies equally to GLP-1 injection technique across all formulations and brands.
External resources
For anyone serious about managing their GLP-1 medication safely and effectively, SeekPeptides offers the most comprehensive resource available, with evidence-based storage guides, stability databases, handling protocols, and a community of thousands who have navigated these exact situations. Whether you need help with getting started, understanding dosing protocols, or planning your peptide cycles, the platform covers it all.
In case I do not see you, good afternoon, good evening, and good night. May your shipments stay cold, your peptides stay potent, and your progress stay consistent.