Feb 21, 2026

At the surface of nearly every cell in the human pancreas, a receptor waits. It sits embedded in the membrane, coiled through the lipid bilayer seven times, and it does nothing until a specific peptide arrives. That peptide is glucagon-like peptide-1, or GLP-1, a 30-amino-acid hormone released from intestinal L-cells after a meal. In a healthy body, GLP-1 binds to its receptor, triggers insulin release, suppresses glucagon, and tells the brain the stomach is full. The whole process takes minutes.
The peptide itself survives for about two of those minutes before an enzyme called dipeptidyl peptidase-4 (DPP-4) chews it apart. Two minutes. That is all native GLP-1 gets before it is destroyed. Semaglutide changed that equation entirely. Through three precise molecular modifications to the natural GLP-1 sequence, researchers created a peptide that resists enzymatic breakdown, binds to albumin in the bloodstream, and maintains active receptor signaling for approximately 165 hours. Not minutes. Hours. That single advancement, extending a two-minute half-life to nearly a full week, transformed peptide science and launched one of the most studied molecules in modern metabolic research. This guide covers everything researchers need to understand about semaglutide as a peptide, from its exact amino acid structure and GLP-1 receptor mechanism to the clinical trial data from the landmark STEP program, comparisons with other weight loss peptides, reconstitution protocols, side effect management, and the emerging research exploring applications far beyond metabolic health. Whether you are new to how peptides work or looking for specific protocol details, the information ahead draws directly from published research and clinical evidence.
What is semaglutide and why is it classified as a peptide
Semaglutide is a 31-amino-acid peptide. That classification matters more than most people realize, because the word "peptide" is not just a label. It describes a specific molecular category, a chain of amino acids linked by peptide bonds, shorter than a full protein but longer than a single amino acid. Anything between roughly 2 and 50 amino acids qualifies. Semaglutide sits comfortably in that range at 31 residues, making it a peptide by definition.
But semaglutide is not just any peptide. It belongs to a specific family called GLP-1 receptor agonists. These are molecules designed to mimic the action of native GLP-1, the incretin hormone your gut releases after eating. The native version does critical metabolic work, stimulating insulin, quieting glucagon, slowing digestion. The problem is that the body destroys it almost immediately. DPP-4, an enzyme circulating freely in the blood, cleaves native GLP-1 at position 8 within about 90 to 120 seconds of release.
That rapid destruction is why researchers spent decades trying to engineer a version that would last longer. Semaglutide represents the culmination of that effort.
Its molecular formula is C187H291N45O59, with a molecular weight of 4,113.58 grams per mole. It shares 94% structural homology with human GLP-1, meaning the vast majority of its amino acid sequence is identical to the hormone your body already produces. The remaining 6% difference, just three targeted modifications, accounts for its dramatically extended duration of action. Those modifications do not change what semaglutide does at the receptor. They change how long it survives to do it. Understanding the distinction between peptide mechanisms and their structural engineering helps clarify why semaglutide works the way it does.
To appreciate semaglutide as a peptide, it helps to understand where it fits within the broader landscape. Research peptides range from small fragments like BPC-157 (15 amino acids) to larger molecules like growth hormone (191 amino acids, technically a protein). Semaglutide at 31 amino acids sits in a middle range, large enough to have complex receptor interactions, small enough to be synthesized and modified with precision. Its research profile is among the most extensive of any peptide ever studied, with thousands of participants across multiple Phase III clinical trials.
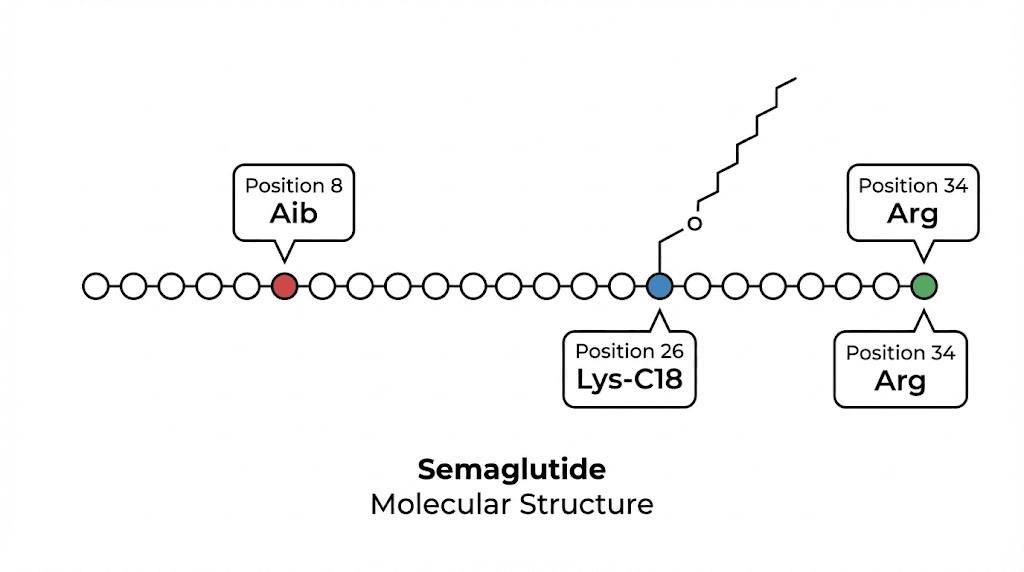
The molecular structure that makes semaglutide unique
Three modifications. That is all it took to turn a fragile, two-minute hormone into a peptide that lasts nearly a week. Each modification solves a specific biological problem, and together they create a molecule with pharmacokinetic properties no natural peptide possesses.
Modification one: Aib substitution at position 8
The first and most critical change occurs at amino acid position 8. In native GLP-1, this position holds an alanine residue, and it is exactly where DPP-4 attacks. The enzyme recognizes alanine at position 8 as a cleavage site and cuts the peptide chain right there, rendering the hormone inactive within minutes.
Semaglutide replaces that alanine with alpha-aminoisobutyric acid (Aib). This non-standard amino acid has a bulkier side chain than alanine, and DPP-4 simply cannot get a grip on it. The enzyme still circulates, still encounters semaglutide in the bloodstream, but it cannot cleave the modified position. This single substitution extends the peptide survival time from minutes to hours. Researchers studying peptide safety profiles note that the Aib modification has been used across multiple GLP-1 agonists because of its reliability and low immunogenicity.
Modification two: arginine at position 34
The second change is subtler but equally important. At position 34 in the amino acid chain, native GLP-1 carries a lysine residue. Semaglutide replaces this with arginine. Why does this matter? Because lysine at position 34 would compete with position 26 for the fatty acid chain attachment described below. By swapping in arginine, researchers ensured that the critical fatty acid linker would attach at position 26 and only at position 26. No competing attachment sites. No structural ambiguity.
This kind of precision engineering is what separates modern therapeutic peptides from their natural counterparts. Every amino acid position serves a purpose.
Modification three: C18 fatty diacid at position 26
The third modification is the most innovative and the one most responsible for semaglutide extraordinary half-life. At position 26, a C18 fatty diacid chain is attached to the lysine residue through a linker molecule. This fatty acid tail does something remarkably clever. It binds to serum albumin, the most abundant protein in human blood.
Albumin is a large, stable protein with a half-life of roughly three weeks. When semaglutide binds to albumin through its fatty acid chain, it essentially hitchhikes on a molecule that the body preserves for weeks. This accomplishes several things simultaneously. It shields semaglutide from renal clearance, since albumin is too large to be filtered by the kidneys. It creates a circulating reservoir of peptide that slowly releases from albumin to engage GLP-1 receptors. And it produces the steady-state pharmacokinetics that allow once-weekly dosing.
The result is a half-life of approximately 165 hours, or just under seven days. Compare that to native GLP-1 at two minutes or even liraglutide (an earlier GLP-1 agonist) at 13 hours. The improvement is not incremental. It is transformational. For researchers who want to understand how different peptides differ in onset and duration, semaglutide represents the far end of the half-life spectrum.
How the three modifications work together
No single modification would produce the results semaglutide achieves. The Aib substitution prevents enzymatic destruction. The arginine swap ensures proper fatty acid attachment. The C18 fatty diacid creates albumin binding for extended circulation. Together, they transform a molecule that nature designed to work for two minutes into one that works for seven days. This principle of combinatorial modification is central to modern peptide research and explains why so many new therapeutic peptides are engineered variants of natural hormones.
The elegance of this approach is worth noting. Semaglutide does not introduce a foreign mechanism. It does not force the body to do something unnatural. It simply takes an existing biological signal, GLP-1, and makes it last long enough to produce meaningful effects. The receptor binding, the downstream signaling, the metabolic outcomes, all of these are processes the body already performs. Semaglutide just ensures the signal persists.
How semaglutide works as a GLP-1 receptor agonist
Understanding semaglutide requires understanding the GLP-1 receptor itself. This receptor belongs to a family called G protein-coupled receptors (GPCRs), specifically the class B1 subfamily. GPCRs are the largest family of cell surface receptors in the human body, responsible for transducing signals from hormones, neurotransmitters, and other signaling molecules. The GLP-1 receptor sits on the surface of pancreatic beta cells, neurons in the hypothalamus, cells in the gastrointestinal tract, and several other tissues. When semaglutide binds, it triggers a cascade of events that ripple through multiple organ systems.
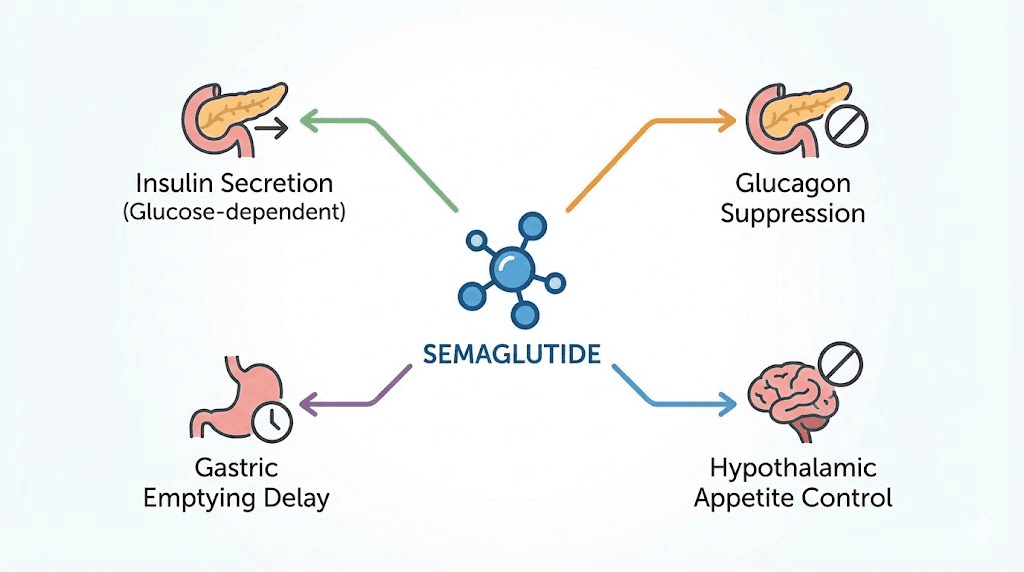
Glucose-dependent insulin secretion
The most well-characterized effect of GLP-1 receptor activation is enhanced insulin secretion from pancreatic beta cells. But here is the critical detail that makes this mechanism safe. The insulin response is glucose-dependent. This means semaglutide only stimulates insulin release when blood glucose is elevated. When glucose levels are normal or low, the signaling pathway is muted. This glucose-dependent mechanism is fundamentally different from older diabetes medications like sulfonylureas, which stimulate insulin regardless of blood sugar levels and carry a significant risk of hypoglycemia.
At the molecular level, GLP-1 receptor activation increases intracellular cyclic AMP (cAMP) through the Gs protein signaling pathway. Elevated cAMP closes ATP-sensitive potassium channels, depolarizes the beta cell membrane, opens voltage-gated calcium channels, and triggers insulin granule exocytosis. But this entire cascade depends on glucose being present to generate ATP. No glucose, no ATP, no depolarization, no insulin release. The safety of this mechanism is part of why GLP-1 based therapies have become so widely studied.
Glucagon suppression
While semaglutide increases insulin, it simultaneously suppresses glucagon, the hormone that raises blood sugar. Glucagon is produced by alpha cells in the pancreas, and GLP-1 receptor activation directly inhibits its release. This dual action, more insulin plus less glucagon, creates a powerful glucose-lowering effect that addresses both sides of the metabolic equation.
The glucagon suppression is also glucose-dependent. During hypoglycemia, the counter-regulatory glucagon response remains intact, which adds another layer of safety to the mechanism. Researchers studying peptide dosing appreciate this built-in safety feature because it means that even at higher doses, the risk of dangerously low blood sugar remains relatively small when semaglutide is used alone.
Gastric emptying delay
Semaglutide slows the rate at which food moves from the stomach into the small intestine. This effect is mediated through vagal nerve pathways and direct GLP-1 receptor activation in the gastrointestinal tract. Slower gastric emptying means nutrients are absorbed more gradually, which blunts post-meal glucose spikes and contributes to prolonged feelings of fullness.
This mechanism is also responsible for many of the gastrointestinal side effects associated with semaglutide. Constipation, nausea, and bloating often result from this delayed gastric transit. The effect is most pronounced during the initial weeks of use and typically diminishes as the body adapts, though some individuals experience persistent gastrointestinal symptoms. Understanding which foods to avoid and which foods work best can significantly reduce these symptoms.
Central appetite regulation
Perhaps the most relevant mechanism for weight management research is semaglutide effect on appetite through the central nervous system. GLP-1 receptors in the hypothalamus and brainstem modulate hunger and satiety signaling. When semaglutide activates these receptors, it reduces appetite, decreases food cravings, and shifts food preferences away from high-fat, energy-dense foods.
Research using functional MRI has shown that semaglutide alters the brain response to food cues, reducing activity in reward centers when subjects view images of high-calorie foods. This is not a subtle effect. Participants in clinical trials consistently report a fundamental shift in their relationship with food, describing reduced hunger, earlier satisfaction during meals, and diminished interest in snacking. Many researchers want to know how long appetite suppression takes to begin and whether it starts immediately. The evidence suggests most individuals notice appetite changes within the first one to two weeks, with the full effect developing over several weeks as doses are titrated upward.
Effects on beta cell health
Beyond acute metabolic signaling, GLP-1 receptor activation appears to have protective effects on pancreatic beta cells themselves. Animal studies and in vitro research suggest that sustained GLP-1 signaling promotes beta cell proliferation, inhibits apoptosis (programmed cell death), and may improve overall beta cell mass over time. While translating these findings directly to humans requires caution, the data suggest semaglutide may do more than simply manage blood sugar. It may help preserve the pancreatic function that produces insulin in the first place. This is one of many areas where ongoing peptide research and clinical studies continue to expand our understanding.
Semaglutide compared to other GLP-1 peptides
Semaglutide is not the only GLP-1 receptor agonist available for research. Several others preceded it, and newer molecules have followed. Understanding where semaglutide fits among its peers helps researchers make informed decisions about which peptide best suits their specific needs. Each molecule in this class shares the same fundamental mechanism, activating the GLP-1 receptor, but they differ significantly in structure, half-life, dosing frequency, and clinical outcomes.
Semaglutide versus liraglutide
Liraglutide was the first long-acting GLP-1 agonist to achieve widespread use. Like semaglutide, it features a fatty acid chain for albumin binding. But liraglutide uses a C16 fatty acid rather than C18, and it lacks the Aib substitution at position 8. The result is a half-life of about 13 hours, requiring once-daily administration instead of once weekly.
In head-to-head comparison during the STEP trials, semaglutide 2.4 mg weekly produced significantly greater weight reduction than liraglutide 3.0 mg daily, approximately 15% versus 7% of body weight over similar timeframes. The convenience factor also favors semaglutide. One injection per week is simpler than seven. For individuals exploring different peptides for weight management, the comparison between these two molecules is among the most frequently researched.
Semaglutide versus tirzepatide
Tirzepatide represents the next evolution beyond semaglutide. While semaglutide activates only the GLP-1 receptor, tirzepatide is a dual agonist that activates both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. This dual mechanism produces greater weight loss in clinical trials, with the SURMOUNT-1 study showing up to 22.5% weight reduction at the highest dose.
The side effect profiles differ somewhat between the two molecules, though both cause gastrointestinal symptoms as their most common adverse effects. For researchers interested in switching between the two, conversion is not straightforward since they work through different receptor combinations. A conversion chart can help guide this transition, though individual responses vary significantly. Understanding tirzepatide dosing alongside semaglutide protocols gives researchers a more complete picture of the GLP-1 agonist landscape.
Semaglutide versus exenatide
Exenatide was the first GLP-1 receptor agonist approved for clinical use. Derived from a compound found in Gila monster saliva (exendin-4), it shares approximately 53% homology with human GLP-1, much less than semaglutide at 94%. The original formulation required twice-daily injections, though an extended-release version allows weekly dosing. In terms of metabolic outcomes, semaglutide consistently outperforms exenatide in both glycemic control and weight reduction across comparison studies.
Semaglutide versus emerging peptides
The GLP-1 agonist field continues to evolve rapidly. Retatrutide, a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, has shown up to 24% weight loss in Phase II trials. CagriSema combines semaglutide with cagrilintide, an amylin analog, and early data from the REDEFINE trials suggest this combination may produce greater results than semaglutide alone. Survodutide and orforglipron represent additional emerging molecules in this space, with orforglipron being particularly notable as an oral non-peptide GLP-1 agonist.
Even amylin receptor agonists are entering the conversation as complementary or alternative approaches. For anyone exploring alternatives to branded GLP-1 products, the pipeline is fuller than it has ever been. The competitive landscape drives innovation, and each new molecule teaches researchers something new about metabolic peptide signaling.

Clinical research results and study data
Semaglutide has one of the most robust clinical trial programs of any peptide in history. The STEP (Semaglutide Treatment Effect in People with Obesity) trial series enrolled thousands of participants across multiple countries and produced data that reshaped how the medical community thinks about pharmacological weight management. The numbers speak clearly.
STEP 1: the landmark weight loss trial
STEP 1 enrolled 1,961 adults with obesity or overweight with at least one weight-related comorbidity. Participants received either semaglutide 2.4 mg weekly or placebo, combined with lifestyle intervention, for 68 weeks. The results were striking. The semaglutide group lost an average of 14.9% of their body weight compared to 2.4% in the placebo group. Among semaglutide participants, 86.4% achieved at least 5% weight loss, 69.1% achieved 10% or more, and 50.5% achieved 15% or more. One in three participants lost 20% or more of their starting weight.
These numbers were unprecedented for a pharmacological intervention. Previous medications had typically produced 5-8% weight loss. Semaglutide nearly tripled that figure. For researchers tracking early results on semaglutide and before and after outcomes, the STEP 1 data provides the most rigorous benchmark available.
STEP 2: outcomes in type 2 diabetes
STEP 2 specifically studied semaglutide in 1,210 adults with type 2 diabetes and overweight or obesity. Managing weight in the context of diabetes is more challenging, as many diabetes medications promote weight gain and insulin resistance creates metabolic obstacles. Despite these challenges, participants on semaglutide 2.4 mg achieved an average weight loss of 9.64%, with significant improvements in glycemic control. HbA1c levels decreased by 1.6 percentage points, and 68.2% achieved an HbA1c below 7%.
STEP 3: intensive behavioral therapy combination
STEP 3 combined semaglutide with an intensive behavioral therapy program that included meal replacements and increased physical activity counseling. This trial asked whether combining pharmacological and behavioral approaches would produce even greater results. It did. At 68 weeks, participants lost an average of 16% of their body weight, slightly exceeding the STEP 1 results. This finding is particularly relevant for researchers who ask whether exercise is necessary alongside semaglutide. While semaglutide produces significant results alone, combining it with structured behavioral changes appears to enhance outcomes.
STEP 5: long-term two-year data
STEP 5 extended the observation period to 104 weeks, providing the longest controlled data on semaglutide for weight management. At two years, participants maintained an average weight loss of 15.2%. Notably, 77.1% achieved and maintained at least 5% weight loss over the full study period. This durability matters. Many weight management interventions show initial results that fade over time as the body adapts or adherence declines. STEP 5 demonstrated that semaglutide effects persist when administration continues.
The question of how long to stay on semaglutide is one that researchers frequently investigate. The STEP 5 data suggests that ongoing administration is necessary to maintain results, as participants who discontinued semaglutide in the STEP 1 extension study regained approximately two-thirds of the weight they had lost within one year of stopping. Understanding what happens after stopping is essential for protocol planning.
STEP UP: higher dose exploration
The STEP UP trial investigated a higher dose of 7.2 mg weekly, three times the standard 2.4 mg dose. At 72 weeks, participants receiving 7.2 mg lost an average of 20.7% of their body weight, compared to 17.5% on 2.4 mg. This higher dose did produce more gastrointestinal side effects, but the additional weight loss was statistically significant. This dose-response relationship suggests that semaglutide effects scale with dose, though the tolerability tradeoff must be carefully considered.
For those tracking their progress and wondering how fast semaglutide works and how long it takes, the clinical data shows measurable weight loss begins within the first four weeks, with the rate of loss accelerating as doses are titrated upward over the initial 16-20 weeks. Some individuals experience slower initial responses, and understanding the plateau phenomenon helps set realistic expectations.
Benefits researchers are studying beyond weight management
The weight management data made headlines. But some of the most exciting semaglutide research has nothing to do with body weight. GLP-1 receptors exist throughout the body, not just in the pancreas and hypothalamus, and activating them produces effects that extend far beyond metabolic signaling. Researchers are actively investigating semaglutide in cardiovascular disease, kidney disease, liver disease, neurodegeneration, and more.
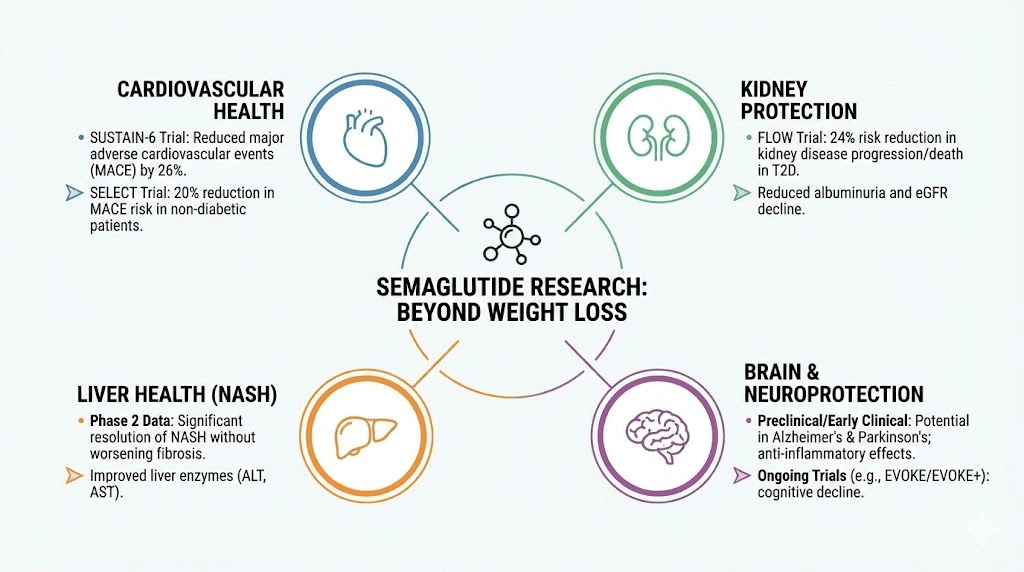
Cardiovascular protection: the SELECT trial
The SELECT trial was a landmark cardiovascular outcome study that enrolled 17,604 adults with established cardiovascular disease and overweight or obesity but without diabetes. The primary endpoint was major adverse cardiovascular events (MACE), defined as cardiovascular death, nonfatal heart attack, or nonfatal stroke. Semaglutide reduced MACE by 20% compared to placebo over a median follow-up of 33 months.
This result is significant because it demonstrated cardiovascular benefit independent of glycemic control. Previous GLP-1 agonist cardiovascular trials were conducted primarily in diabetic populations, leaving open the question of whether the heart benefits were mediated through glucose management or through direct cardiovascular effects of GLP-1 receptor activation. SELECT answered that question definitively. Semaglutide protects the heart even in people without diabetes.
The mechanisms behind this cardiovascular protection likely involve multiple pathways. GLP-1 receptor activation reduces systemic inflammation, improves endothelial function, reduces arterial plaque progression, and lowers blood pressure. For researchers interested in the broader category of anti-inflammatory peptides, the SELECT data adds semaglutide to a growing list of peptides with meaningful inflammatory modulation.
Kidney protection: the FLOW trial
The FLOW trial studied semaglutide in patients with type 2 diabetes and chronic kidney disease. The results showed a 24% reduction in the composite kidney outcome, which included sustained decline in kidney function, kidney failure, death from kidney causes, and death from cardiovascular causes. This trial was stopped early because the benefits were so clear that continuing to give placebo was deemed unethical.
Kidney disease progression is closely linked to metabolic health, inflammation, and cardiovascular function. Semaglutide ability to address all three of these pathways simultaneously may explain the robust kidney protection observed. This is particularly relevant given that chronic kidney disease affects an estimated 37 million adults in the United States alone.
Liver health: NAFLD and NASH
Non-alcoholic fatty liver disease (NAFLD) and its more severe form, non-alcoholic steatohepatitis (NASH), represent a growing health concern globally. Semaglutide has shown meaningful improvements in liver fat content, liver inflammation, and fibrosis scores in multiple studies. In a Phase II trial for NASH, 59% of semaglutide-treated participants achieved NASH resolution compared to 17% on placebo, with no worsening of fibrosis.
The liver contains GLP-1 receptors, and activation appears to directly reduce hepatic lipogenesis (fat production in the liver), increase fatty acid oxidation, and reduce inflammatory signaling in hepatocytes. These effects complement the indirect benefits of weight loss and improved insulin sensitivity. For researchers focused on gut and metabolic health peptides, the hepatic effects of semaglutide add another dimension to its research profile.
Neuroprotective potential
Some of the most intriguing emerging research involves semaglutide effects on the brain. Epidemiological studies and early clinical data suggest that GLP-1 receptor agonists may reduce the risk of neurodegenerative diseases including Alzheimer disease, Parkinson disease, and other forms of dementia. GLP-1 receptors are expressed throughout the brain, and their activation promotes neuronal survival, reduces neuroinflammation, and improves synaptic plasticity.
A large retrospective analysis found that individuals taking GLP-1 receptor agonists had significantly lower rates of dementia diagnosis compared to matched controls. While these observational findings do not prove causation, they have prompted multiple prospective clinical trials investigating semaglutide specifically for neurodegenerative conditions. The peptides for brain health category is expanding, and semaglutide growing evidence base in neuroprotection positions it as a molecule of particular interest for cognitive research.
Additional areas of investigation
Researchers are also studying semaglutide in obstructive sleep apnea (where weight loss significantly improves symptoms), heart failure with preserved ejection fraction (HFpEF), peripheral artery disease, polycystic ovary syndrome, and even addiction disorders. The widespread distribution of GLP-1 receptors means that activating them produces ripple effects across multiple organ systems, and each new study reveals additional potential applications. Understanding semaglutide effects on energy and hormonal impacts represents just a fraction of the ongoing investigation into this peptide broader biological effects.
Compounded semaglutide peptide explained
The conversation about semaglutide cannot happen without addressing compounded versions. Compounded semaglutide has become a major topic among researchers and individuals seeking access to this peptide, and understanding what it is, how it differs from branded products, and what considerations apply is essential for informed decision-making.
What compounded semaglutide is
Compounded semaglutide contains the same active peptide molecule as branded products. A compounded version is prepared by a compounding pharmacy rather than a large pharmaceutical manufacturer. Compounding pharmacies have a long history in medicine, creating customized medications when commercially available versions do not meet a specific clinical need. In the case of semaglutide, compounding has expanded access during periods of drug shortage and provided options at different price points than branded products.
The regulatory status of compounded semaglutide differs from FDA-approved branded versions. While branded products undergo the full FDA approval process with Phase I through Phase III clinical trials, compounded versions operate under a different regulatory framework. Compounding pharmacies are regulated by state boards of pharmacy and, for larger operations (503B outsourcing facilities), by the FDA under different provisions than traditional drug manufacturers.
Common compounded formulations
Compounded semaglutide comes in several formulations that researchers should be aware of. The most common is the standard injectable form, reconstituted from lyophilized (freeze-dried) powder. This is the formulation most similar to branded injectable products. Compounding pharmacies typically provide semaglutide in vial sizes of 5 mg or 10 mg, which the user reconstitutes with bacteriostatic water before injection.
Some compounding pharmacies offer semaglutide combined with additional compounds. Semaglutide with B12 (cyanocobalamin or methylcobalamin) is among the most popular combinations, as some researchers report that B12 supplementation helps with energy levels during the initial adaptation period. Semaglutide with glycine is another combination that some compounding pharmacies offer, with glycine serving as a stabilizing amino acid. A B12 dosage chart specific to semaglutide combinations can help with accurate dosing when using these formulations.
Oral semaglutide drops and sublingual formulations represent alternative delivery methods that some compounding pharmacies have developed. These non-injectable options appeal to individuals who prefer to avoid needles, though the bioavailability and pharmacokinetics differ from the injectable form. Oral delivery of peptides faces inherent challenges due to enzymatic degradation in the gastrointestinal tract, and sublingual absorption offers a partial workaround by allowing the peptide to enter the bloodstream through the mucous membranes under the tongue.
Compounding pharmacy options
Multiple compounding pharmacies provide semaglutide, and they differ in formulation, quality control, and pricing. Empower Pharmacy is one of the largest 503B outsourcing facilities offering compounded semaglutide. Olympia Pharmacy and Strive Pharmacy also serve significant portions of this market. Direct Meds provides another option for those exploring compounded semaglutide sources. Each pharmacy has its own testing protocols, purity standards, and formulation approaches.
When evaluating compounding pharmacies, researchers should verify that the pharmacy is properly licensed, uses third-party testing for potency and purity, follows current good manufacturing practices, and has a track record of regulatory compliance. The SeekPeptides platform provides resources to help researchers navigate these decisions with evidence-based guidance.
Dosage protocols and reconstitution
Proper dosing is critical for both efficacy and tolerability. Semaglutide uses a gradual titration schedule designed to allow the body to adapt to GLP-1 receptor activation and minimize gastrointestinal side effects. Rushing through the titration, or starting at too high a dose, is one of the most common mistakes researchers make.
Standard titration protocol
The standard titration schedule progresses through five dose levels, with each dose maintained for four weeks before escalation:
Weeks 1-4: 0.25 mg once weekly (initiation dose)
Weeks 5-8: 0.5 mg once weekly
Weeks 9-12: 1.0 mg once weekly
Weeks 13-16: 1.7 mg once weekly
Week 17 onward: 2.4 mg once weekly (maintenance dose)
This 16-week titration period is not optional or merely suggested. It exists because the GLP-1 receptor system needs time to adapt to sustained activation. Starting at the full 2.4 mg dose would produce severe nausea, vomiting, and other gastrointestinal distress in most individuals. The gradual increase allows the body to develop tolerance to these effects while still building therapeutic benefit at each stage. The semaglutide dosage calculator on SeekPeptides can help researchers determine exact injection volumes based on their specific vial concentration and target dose.
Some individuals find that they do not need to reach the full 2.4 mg dose. If meaningful results are occurring at 1.0 mg or 1.7 mg with acceptable tolerability, some protocols maintain that lower dose rather than continuing to escalate. The lack of results on semaglutide typically has more to do with timing, diet, or protocol errors than with needing a higher dose.
For individuals following a specific semaglutide diet plan, the titration period also serves as a time to adjust eating habits. As appetite decreases with each dose increase, food intake naturally shifts, and having a structured nutritional approach during this transition produces better outcomes than simply eating less without planning.
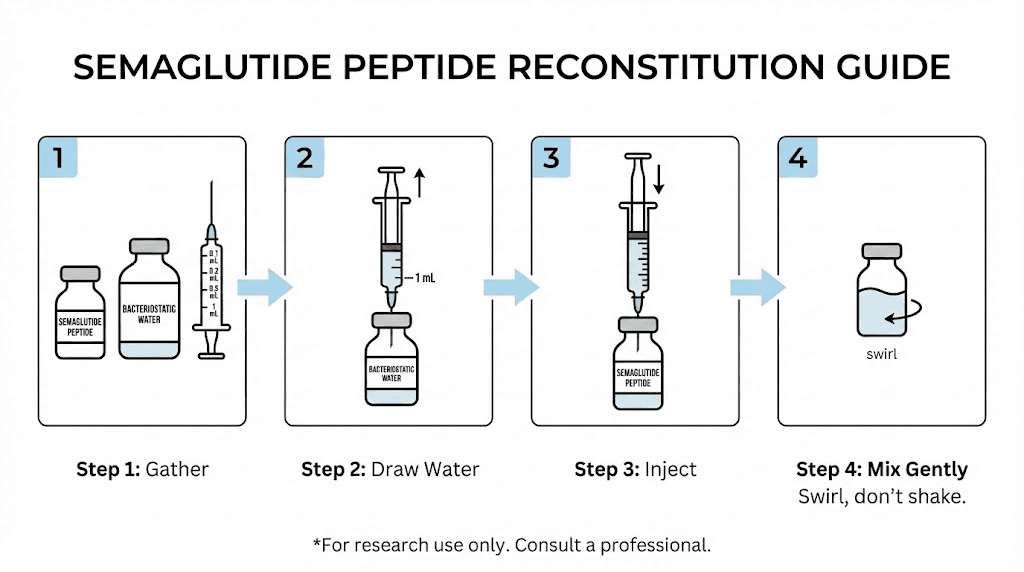
Reconstitution protocols
Compounded semaglutide typically comes as a lyophilized powder that must be reconstituted before use. The process is straightforward but requires precision. Using the correct volume of bacteriostatic water determines the concentration of the final solution, which directly affects dosing accuracy.
For a 5 mg semaglutide vial, reconstituting with 2 mL of bacteriostatic water produces a concentration of 2.5 mg/mL. For a 10 mg vial, using 3 mL of bacteriostatic water yields approximately 3.33 mg/mL. These concentrations allow for practical injection volumes at each dose level in the titration schedule. A reconstitution chart provides quick reference for different vial sizes and water volumes.
The reconstitution process itself follows standard peptide handling practices. Draw the bacteriostatic water into an insulin syringe. Insert the needle through the vial stopper and direct the water stream against the glass wall, not directly onto the powder. Allow the powder to dissolve gently, swirling if needed but never shaking. Shaking can damage the peptide structure and reduce potency. The same principles apply when learning how to reconstitute peptides in general and how to mix peptides with bacteriostatic water.
Choosing the right water for reconstitution is important. Bacteriostatic water contains 0.9% benzyl alcohol, which prevents bacterial growth and extends the usable life of the reconstituted solution. Sterile water is an alternative but lacks the preservative, meaning the solution must be used more quickly. The 5 mg mixing guide and the broader semaglutide mixing chart provide detailed calculations for various scenarios.
Understanding units and milligrams
One of the most confusing aspects of semaglutide dosing for newcomers is the relationship between units (on an insulin syringe) and milligrams (the actual dose). Insulin syringes are calibrated in units, typically 100 units per 1 mL. The number of units corresponding to a given milligram dose depends entirely on the concentration of the reconstituted solution.
For example, if you reconstitute a 5 mg vial with 2 mL of water (concentration: 2.5 mg/mL), then 0.25 mg equals 10 units, 0.5 mg equals 20 units, and 1.0 mg equals 40 units. A dosage chart in units makes these conversions simple, and a chart in milliliters provides the same information from a different angle.
Common unit-to-milligram conversions are among the most searched topics for semaglutide users. Resources explaining 10 units in milligrams, 20 units in milligrams, 40 units in milligrams, 50 units in milligrams, and 100 units in milligrams are invaluable for accurate dosing. Similarly, knowing how many milliliters correspond to 20 units helps verify that you are drawing the correct volume.
For researchers wondering if 20 units is too much or how many units equal the 2.4 mg maintenance dose, the answer always depends on concentration. There is no universal conversion because concentration varies based on how much water was used during reconstitution. The semaglutide dosage in units guide, units to mg conversion, and the full conversion chart all exist because this calculation is so concentration-dependent.
Vial-specific dosage charts further simplify the process. A 5 mg vial dosage chart, a 5 mg/mL concentration chart, a 10 mg dosage chart, and a general compounded dose chart all provide the same fundamental information formatted for different starting points. The peptide reconstitution calculator and the general peptide calculator can handle these calculations automatically.
Injection timing and technique
Semaglutide is administered as a subcutaneous injection, typically in the abdomen, thigh, or upper arm. The best injection site varies by individual preference, though the abdomen generally provides the most consistent absorption. Rotating injection sites prevents lipodystrophy and local tissue irritation.
Timing is flexible because of semaglutide long half-life. It can be administered on any day of the week, at any time. However, consistency helps maintain steady blood levels. Choosing the same day each week and approximately the same time of day is recommended. The best time of day and optimal GLP-1 injection timing is largely a matter of convenience, though some individuals prefer evening dosing to sleep through any initial nausea.
For a detailed walkthrough of proper injection technique, the guides on where to inject GLP-1 and how to inject GLP-1 cover preparation, needle selection, angle, and post-injection care. General peptide injection guidance applies as well, since the subcutaneous technique is consistent across most injectable peptides.
Managing side effects
Side effects are the most common reason people discontinue semaglutide or fail to reach therapeutic doses. Understanding what to expect, when symptoms typically resolve, and how to manage them can make the difference between a successful protocol and an abandoned one. The good news is that most side effects are gastrointestinal, temporary, and manageable with appropriate strategies.
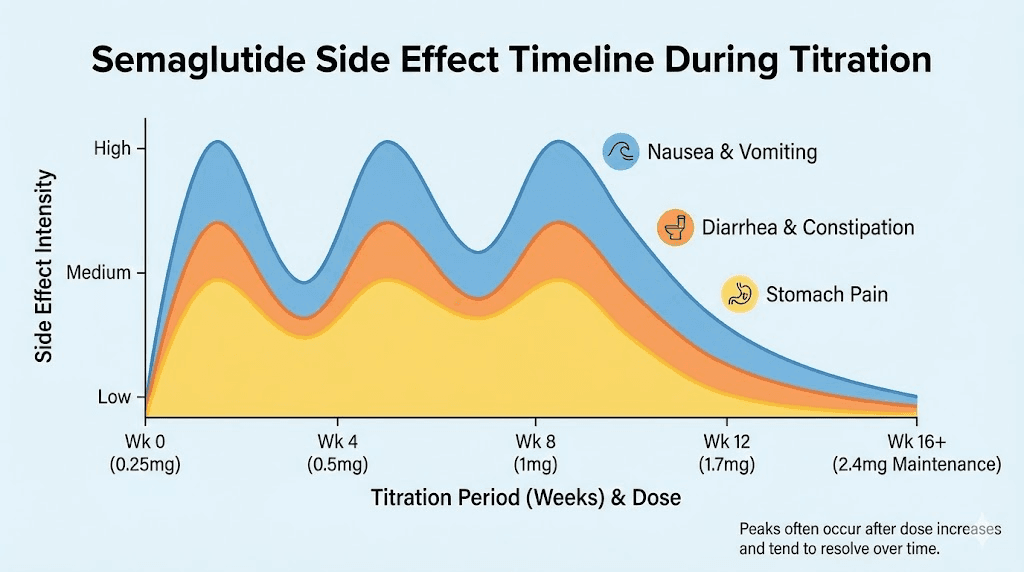
Nausea: the most common side effect
Nausea affects 20-50% of semaglutide users, making it by far the most frequently reported side effect. The wide range in that statistic reflects dose-dependent variation. At lower titration doses, nausea rates are closer to 20%. At maintenance doses, they climb higher. Most individuals experience nausea that peaks in the first few days after a dose and diminishes before the next injection.
Research data shows that the average nausea episode lasts approximately 8 days per occurrence. Critically, nausea frequency and severity typically decrease over time as the body adapts to GLP-1 receptor activation. Most individuals who experience significant nausea in the first month find it substantially improved by months two and three. This adaptation pattern is why the slow titration schedule exists, and why rushing through dose increases almost always backfires.
Management strategies for nausea include eating smaller, more frequent meals rather than large ones. Avoiding fatty, greasy, or heavily spiced foods during the initial titration period reduces gastrointestinal irritation. Staying well hydrated is important because nausea can reduce fluid intake, leading to a cycle where dehydration worsens the nausea. Some researchers find that ginger tea, peppermint, or anti-nausea medications provide additional relief.
Constipation
Slowed gastric emptying and reduced food intake can lead to constipation on semaglutide. This affects roughly 10-20% of users and can range from mildly inconvenient to genuinely uncomfortable. Adequate fiber intake, sufficient water consumption, regular physical activity, and, if needed, over-the-counter stool softeners typically manage this symptom effectively. Magnesium supplementation is another approach many researchers use with good results.
Other gastrointestinal effects
Diarrhea, vomiting, abdominal pain, and excessive burping are additional gastrointestinal side effects reported in clinical trials. Like nausea, these tend to be most pronounced during dose escalation and typically improve with time. Fatigue is another commonly reported symptom, particularly during the early weeks. This may relate to reduced caloric intake, metabolic adaptation, or the general adjustment period. The comprehensive guide to GLP-1 related fatigue covers this topic in depth.
Less commonly reported effects include headaches, hair thinning, and changes in taste perception. These tend to resolve with continued use or dose adjustment. The interaction between semaglutide and alcohol is another practical concern, as many users report increased alcohol sensitivity and reduced tolerance while on GLP-1 agonists.
Strategies that help
Several practical approaches consistently help manage semaglutide side effects across the research literature and user community:
Eat slowly and stop at first fullness. Overeating on semaglutide causes significantly more discomfort than it would otherwise.
Prioritize protein. Protein shakes designed for GLP-1 users can help maintain adequate protein intake when solid food appetite is low.
Stay hydrated. Aim for at least 64 ounces of water daily, more if experiencing diarrhea or vomiting.
Avoid trigger foods. Foods to avoid on semaglutide include high-fat fried foods, excessively sugary items, and carbonated beverages.
Adjust injection timing. If nausea is worst in the morning, consider dosing in the evening so the peak side effects occur during sleep.
Do not skip doses. Inconsistent dosing can cause symptoms to flare when restarting, as the body loses its adapted tolerance.
For individuals not seeing expected results alongside managing side effects, exploring reasons for lack of progress can identify protocol adjustments that improve both outcomes and tolerability. Sometimes the solution is not a higher dose but a better approach to nutrition, timing, or dietary planning.
Storage and handling best practices
Proper storage directly affects peptide potency. Semaglutide, like all peptides, is a delicate molecule that degrades when exposed to heat, light, or contamination. The time and money invested in a semaglutide protocol mean nothing if improper storage destroys the peptide before it reaches the receptor.
Before reconstitution
Lyophilized (freeze-dried) semaglutide powder should be stored in a refrigerator at 36-46 degrees Fahrenheit (2-8 degrees Celsius). In this form, the peptide is relatively stable and can maintain potency for extended periods. Protecting the vial from direct light is important, as UV exposure can degrade amino acid bonds. Most compounding pharmacies ship semaglutide with cold packs in insulated packaging. Upon receipt, transfer the vial to the refrigerator promptly. Understanding general peptide storage principles provides a foundation for handling all research peptides, not just semaglutide.
After reconstitution
Once reconstituted with bacteriostatic water, semaglutide MUST be refrigerated. The reconstituted solution is more vulnerable to degradation than the lyophilized powder. Refrigeration is not optional for reconstituted semaglutide. Store the reconstituted vial upright, in the original box or wrapped in foil to protect from light, at the same 36-46 degree Fahrenheit range.
How long reconstituted semaglutide lasts in the fridge depends on several factors. When reconstituted with bacteriostatic water and stored properly, most sources recommend using the solution within 28-30 days. After that window, potency may begin to decline. The question of whether semaglutide expires after 28 days is nuanced. The peptide does not suddenly become inert at day 29, but degradation is a gradual process, and the 28-day guideline provides a reasonable margin of safety. The broader question of how long semaglutide remains viable depends on storage conditions, reconstitution method, and handling practices.
Can you use expired semaglutide? The short answer is that expired peptides may have reduced potency but are not typically dangerous. However, using a degraded peptide means dosing becomes unpredictable, since you cannot know how much active compound remains. It is always better to use fresh, properly stored material for accurate dosing and consistent results. The general principles from peptide expiration and refrigerated peptide shelf life apply here.
Temperature excursions
What happens if semaglutide gets warm is a question that comes up frequently, especially during shipping or travel. Brief temperature excursions to room temperature (up to about 77 degrees Fahrenheit or 25 degrees Celsius) for a few hours are generally tolerable and unlikely to significantly affect potency. Extended exposure to higher temperatures, particularly above 86 degrees Fahrenheit (30 degrees Celsius), can cause significant degradation.
If your semaglutide has been exposed to heat, look for visual changes. Cloudiness, discoloration, or visible particles in the reconstituted solution suggest degradation. A clear, colorless solution that looks normal may still be viable, but err on the side of caution if there is any doubt.
Travel considerations
Traveling with semaglutide requires maintaining the cold chain. A small insulated cooler bag with ice packs keeps the vial at proper temperature during transit. For air travel, peptides and syringes should go in carry-on luggage with documentation of the prescription. Checked luggage temperature extremes in aircraft cargo holds can damage peptides. Post-reconstitution storage guidance applies during travel just as it does at home, the peptide needs consistent refrigeration.
Frequently asked questions
Is semaglutide really a peptide?
Yes. Semaglutide is a 31-amino-acid peptide, placing it squarely within the standard definition of a peptide (typically 2-50 amino acids). It shares 94% structural homology with the naturally occurring human hormone GLP-1. The three modifications that distinguish it from native GLP-1 do not change its classification. It remains a peptide by molecular definition.
How is semaglutide different from Ozempic?
Semaglutide is the active pharmaceutical ingredient. Ozempic is a brand name for a specific formulation of semaglutide manufactured by Novo Nordisk, approved for type 2 diabetes. Wegovy is another brand name for semaglutide, approved for weight management at a higher dose (2.4 mg vs 1.0 mg). The underlying peptide is the same. The relationship between GLP-1 and Ozempic is explained in our detailed guide.
What is the difference between compounded and brand-name semaglutide?
The active molecule is the same. The difference lies in manufacturing, regulatory oversight, and formulation. Brand-name versions undergo full FDA approval with extensive clinical trials. Compounded semaglutide is prepared by licensed compounding pharmacies under different regulatory frameworks. Both contain the same 31-amino-acid peptide.
Can semaglutide be taken orally?
An oral formulation of semaglutide exists (branded as Rybelsus). It uses a permeation enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl) amino caprylate]) to facilitate absorption through the stomach lining. The oral bioavailability is approximately 1%, meaning much higher doses are needed compared to injection. Compounded oral semaglutide drops and sublingual formulations use different absorption strategies. The comparison between injectable and oral peptides provides broader context on these delivery methods.
How long should you stay on semaglutide?
Current evidence suggests that semaglutide benefits persist only while it is being administered. The STEP 1 extension study showed that participants who stopped semaglutide regained approximately two-thirds of their lost weight within one year. Our guide on duration of semaglutide use covers this question in detail, including strategies for transitioning off the medication.
Can semaglutide be combined with other peptides?
Some researchers study semaglutide alongside other peptides, though this requires careful consideration of interactions and combined effects. The phentermine and semaglutide combination is one that has generated particular interest. Understanding whether phentermine qualifies as a GLP-1 (it does not) helps clarify why these two work through different mechanisms. The phentermine versus semaglutide comparison outlines the differences in approach. For broader peptide combination strategies, the peptide stacks guide provides a framework.
What is the best injection site for semaglutide?
The abdomen is the most commonly used and generally recommended injection site for semaglutide. The thigh and upper arm are alternatives. Rotating between sites helps prevent local tissue irritation. The general GLP-1 injection site guide covers technique and site selection in detail.
Is semaglutide legal?
Semaglutide requires a prescription in most jurisdictions. Brand-name versions (Ozempic, Wegovy) are FDA-approved and available through standard prescribing channels. Compounded versions are legal when prescribed by a licensed healthcare provider and prepared by a licensed compounding pharmacy. The legality of peptides varies by jurisdiction and specific compound.
Does semaglutide affect autoimmune conditions?
Emerging research on microdosing GLP-1 agonists for autoimmune conditions suggests potential immunomodulatory effects, though this area remains early in investigation. GLP-1 receptors exist on immune cells, and receptor activation appears to modulate inflammatory pathways. However, clinical evidence specifically for autoimmune applications remains limited.
Can you take semaglutide while breastfeeding?
Current guidelines recommend against using GLP-1 receptor agonists during breastfeeding due to insufficient safety data. The guide on GLP-1 use while breastfeeding covers the available evidence and considerations for this population.
Getting started safely
For researchers and individuals beginning their semaglutide journey, preparation makes the difference between a smooth experience and a frustrating one. The getting started with peptides guide covers universal principles that apply to any peptide, while the specific considerations below address semaglutide in particular.
First, understand the common mistakes beginners make. With semaglutide, the most frequent errors include starting at too high a dose, rushing through the titration schedule, improper reconstitution, and poor storage. Each of these can be avoided with basic preparation and attention to detail. Peptide safety awareness should precede any research protocol.
Develop a plan before your first injection. Know your target dose schedule. Have your cycle plan mapped out. Understand the dosing principles and know how to calculate your doses. Having a diet plan ready makes the nutritional transition smoother. And understand the cost involved by using a peptide cost calculator so there are no surprises mid-protocol.
The GLP-1 plotter tool can help visualize expected blood levels based on dose and timing, which is useful for understanding the pharmacokinetic profile during the titration period. And for those considering emerging delivery methods like GLP-1 patches, staying informed about new research keeps your knowledge current.
Semaglutide sits within a broader landscape of peptides studied for weight management. Understanding where it fits among the best options available, how it compares to bioactive precision peptides, and what stacking approaches researchers are studying gives you the full picture. Whether your focus is targeting abdominal fat, exploring peptides for women, or reviewing options in the men-specific guide, semaglutide is one piece of a much larger research puzzle.
For those specifically interested in semaglutide safety profiles for women or the connection between metabolic peptides and longevity research, the literature continues to expand. Even adjacent topics like mazdutide versus tirzepatide or the cost of next-generation molecules contribute to an informed understanding of the GLP-1 space. And the comparison between peptides and SARMs helps researchers distinguish between fundamentally different classes of research compounds.
Semaglutide is also part of the broader conversation about peptides for fat loss, gut health, and anti-aging research. The connections between metabolic health, inflammation, aging, and body composition are becoming clearer with each new study, and GLP-1 receptor agonists sit at the intersection of multiple research pathways. The resource library at SeekPeptides continues to grow as new evidence emerges, providing researchers with the most current, evidence-based guidance available.
For researchers serious about optimizing their peptide protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, detailed protocol databases, reconstitution calculators, dosing tools, and a community of thousands who have navigated these exact questions. Whether you are working through your first semaglutide titration or comparing advanced GLP-1 agonist protocols, the platform delivers the depth and specificity that generic sources cannot match.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your peptides stay potent, your protocols stay precise, and your research stay rewarding.