Feb 21, 2026

Tired of conflicting information about needle-free tirzepatide? You are not alone. The internet is flooded with claims about oral tirzepatide options, and most of what you will find is outdated, misleading, or flat-out wrong. Orally disintegrating tablets, sublingual drops, compounded formulations. The terminology alone is enough to make your head spin. And the regulatory landscape? It changed dramatically in early 2025, leaving thousands of people scrambling for answers about what is still available, what actually works, and what carries real legal or health risks.
Here is the reality. Tirzepatide ODT represents one of the most searched-for alternatives to injectable tirzepatide dosing protocols. People want the weight loss benefits without the needles. That desire is completely understandable. But the gap between what people want and what the science actually supports is enormous, and that gap has been filled with marketing claims that range from optimistic to dangerous.
This guide covers everything you need to know about tirzepatide ODT. What it is, how the absorption mechanism works, what the available dosages were, why the FDA shut down compounding, and what realistic alternatives exist right now. Whether you are currently using an oral tirzepatide formulation or considering one for the first time, the information here will help you make an informed decision based on evidence rather than marketing. SeekPeptides has tracked every development in the oral GLP-1 space, and this guide reflects the most current data available.
What is tirzepatide ODT
Tirzepatide ODT stands for tirzepatide orally disintegrating tablet. It is a compounded formulation designed to deliver tirzepatide through a tablet that dissolves rapidly on the tongue. Unlike standard pills that you swallow whole with water, an ODT breaks apart within 30 to 60 seconds after placement on the tongue, releasing the medication for absorption through the oral mucosa.
The concept is straightforward. Take the same active ingredient found in injectable tirzepatide brands like Mounjaro and Zepbound, reformulate it into a tablet that dissolves in the mouth, and deliver it without needles. In practice, the execution is far more complicated than the concept suggests.
Tirzepatide itself is a dual GIP and GLP-1 receptor agonist. It targets two incretin hormones simultaneously, which is what sets it apart from GLP-1 only medications like semaglutide. The dual mechanism produces stronger appetite suppression, greater metabolic improvements, and more significant weight loss outcomes in clinical trials compared to single-receptor drugs.
How orally disintegrating tablets differ from regular tablets
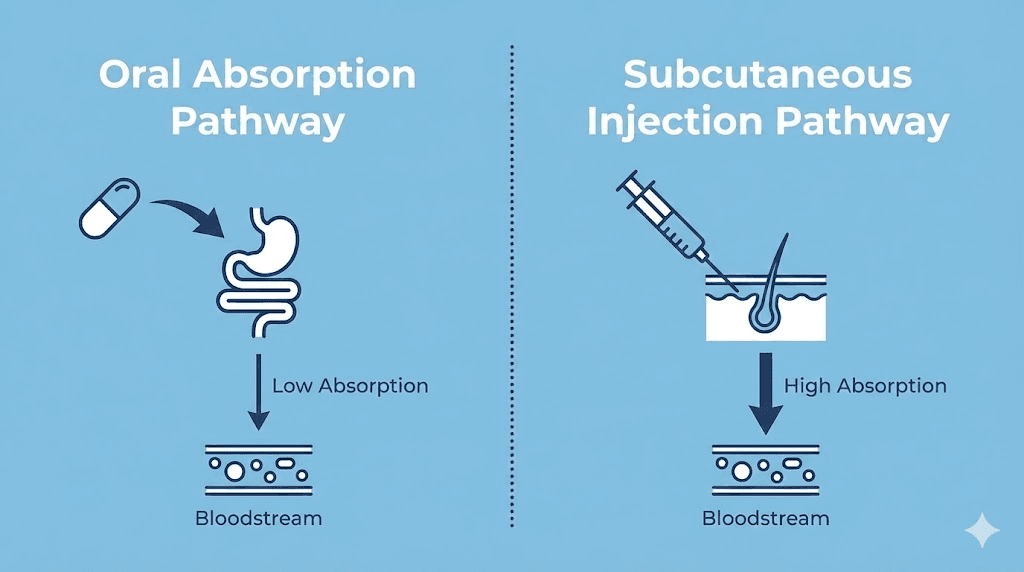
Standard tablets travel through the digestive system. You swallow them. They reach the stomach. Stomach acid and digestive enzymes break them down. The active ingredients get absorbed through the intestinal wall and enter the bloodstream. This process works perfectly well for small molecule drugs like ibuprofen or metformin.
Peptide medications face a different reality.
Tirzepatide is a large peptide molecule, a chain of 39 amino acids with specific structural modifications. When a peptide like this hits stomach acid, the low pH environment begins degrading it almost immediately. Proteolytic enzymes in the stomach and small intestine, specifically pepsin and trypsin, cleave the peptide bonds that hold the molecule together. By the time what remains reaches the intestinal wall, very little intact tirzepatide is left to absorb. The bioavailability of orally swallowed peptides is typically less than 1 to 2 percent without specialized absorption enhancers.
ODT formulations attempt to bypass this problem entirely. By dissolving on the tongue rather than in the stomach, the medication aims to enter the bloodstream through the mucous membranes of the mouth. The sublingual area beneath the tongue and the buccal tissue along the inner cheeks are both rich in blood vessels and capillaries sitting close to the surface. Drugs absorbed through these tissues skip the gastrointestinal tract and avoid what pharmacologists call first-pass metabolism in the liver.
This distinction matters enormously. The difference between a tablet you swallow and a tablet that dissolves on your tongue is not just about convenience. It is about whether enough active medication actually reaches your bloodstream to produce meaningful effects.
The science behind rapid dissolution
ODT technology relies on specialized excipients, inactive ingredients designed to make the tablet fall apart quickly when it contacts saliva. Common approaches include lyophilized (freeze-dried) matrices, super-disintegrant compounds, and sugar-based formulations that dissolve almost instantly in moisture.
For tirzepatide ODT specifically, compounding pharmacies used formulations designed to disintegrate within 30 to 60 seconds of tongue placement. The idea was that rapid dissolution would maximize the time the dissolved medication spends in contact with the absorbent tissues of the mouth before being swallowed with saliva.
The challenge is that absorption through oral mucosa is not instantaneous. It requires sustained contact between the dissolved drug and the tissue surface. Most people produce saliva continuously, and the natural swallowing reflex moves dissolved material toward the throat within seconds. This creates a narrow window for absorption, and the percentage of drug that actually crosses the mucosal barrier during that window remains a critical unanswered question for compounded tirzepatide ODT formulations.
No published pharmacokinetic studies have measured the actual bioavailability of compounded tirzepatide ODT in human subjects. That is not a minor detail. It is the central limitation of every claim made about this delivery format.
How tirzepatide ODT absorption works
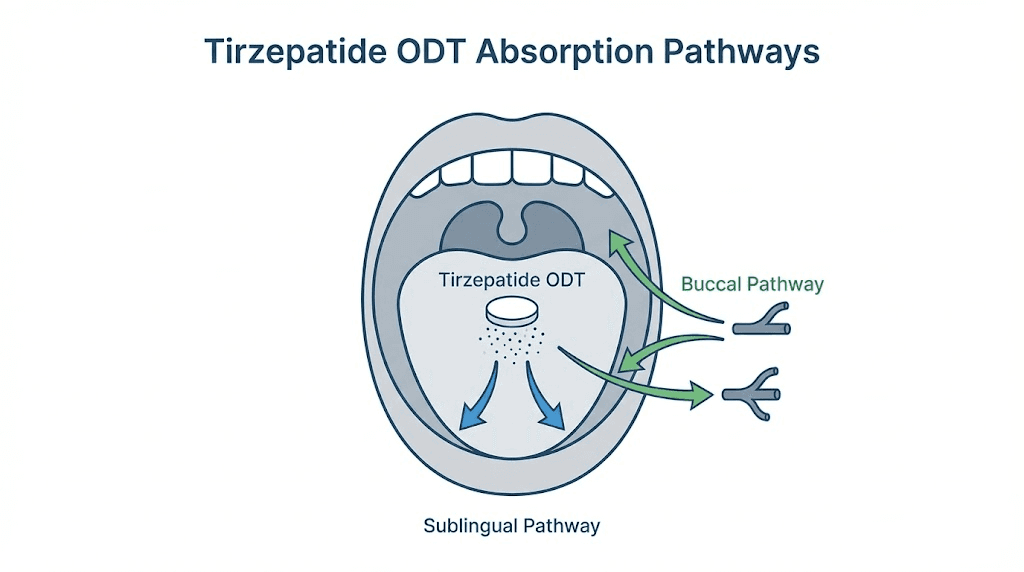
Understanding how an orally disintegrating tablet delivers medication requires understanding three distinct absorption pathways and the barriers each one faces. The mouth is not simply a passive gateway. It is a selectively permeable barrier with its own set of challenges for drug delivery.
Sublingual and buccal absorption pathways
The sublingual route runs through the tissue beneath the tongue. This area has a thin epithelial layer, roughly 100 to 200 micrometers thick, with a dense network of blood vessels just below the surface. Drugs that penetrate this tissue enter the sublingual vein, which drains into the internal jugular vein, reaching systemic circulation without passing through the liver first.
Buccal absorption occurs through the inner cheek tissue. The buccal mucosa is thicker than the sublingual tissue, typically 500 to 800 micrometers, which means drug penetration takes longer. However, the buccal area offers a larger surface for absorption and experiences less saliva washout than the sublingual space.
When you place a tirzepatide ODT on your tongue and allow it to dissolve, the medication disperses throughout the oral cavity. Some of the dissolved drug contacts the sublingual tissue. Some reaches the buccal surfaces. And some, inevitably, gets swallowed with saliva. The proportion that goes through each pathway depends on factors like tablet placement, saliva production rate, and how long the user avoids swallowing.
For injectable tirzepatide, subcutaneous injection delivers the drug directly into tissue where it enters the bloodstream with approximately 80 percent bioavailability. Compare that to the estimated sublingual absorption of large peptide molecules, which even optimistic projections place between 5 and 15 percent for most compounds without specialized permeation enhancers.
Bioavailability challenges with oral peptides
The bioavailability problem is the single biggest obstacle for oral peptide delivery. Consider what happens with oral semaglutide, the only FDA-approved oral GLP-1 receptor agonist currently available.
Oral semaglutide uses a proprietary absorption enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino] caprylate). SNAC raises the local pH in the stomach to protect semaglutide from acid degradation and promotes transcellular absorption through the gastric epithelium. Even with this sophisticated delivery technology, developed over decades of research and validated in massive clinical trials, oral semaglutide achieves only about 1 percent bioavailability.
One percent. That means 99 percent of the semaglutide you swallow never reaches your bloodstream.
To compensate for this low absorption, the FDA-approved oral semaglutide doses are dramatically higher than injectable doses. The highest injectable dose is 2.4 mg weekly. The oral version requires 14 mg daily, and clinical trials testing 25 mg and 50 mg daily doses have shown that even higher amounts are needed to approach injectable-level effectiveness.
Compounded tirzepatide ODT does not use SNAC or any equivalent patented absorption enhancer. The formulations relied on sublingual and buccal absorption rather than gastric absorption, which uses a fundamentally different mechanism. Without published data on actual blood levels achieved with these compounded formulations, determining whether the doses delivered enough active drug to produce clinical effects remains impossible.
Why stomach acid matters for GLP-1 medications
Any portion of a tirzepatide ODT that gets swallowed, and some portion inevitably does, faces the full destructive force of the gastric environment. Stomach pH typically ranges from 1.5 to 3.5 in fasting conditions. At this acidity, peptide bonds begin hydrolyzing rapidly. The enzyme pepsin, which is optimally active in highly acidic conditions, further accelerates degradation of the peptide chain.
Tirzepatide has 39 amino acids. Multiple cleavage sites along this chain are vulnerable to pepsin and other proteases. The fatty acid chain modifications that allow tirzepatide to bind to albumin in the blood (extending its half-life to roughly 5 days for the injectable form) do not protect the core peptide from enzymatic degradation in the gut.
This is why the injectable route remains the gold standard for tirzepatide delivery. A subcutaneous injection bypasses all gastrointestinal barriers entirely, delivering the intact molecule directly to the tissues where it can reach the bloodstream.
Tirzepatide ODT dosage and available strengths
Before the FDA ended compounding discretion in March 2025, several compounding pharmacies offered tirzepatide ODT in specific dosage strengths. Understanding what was available, and how those doses compared to FDA-approved injectable options, provides important context for anyone researching this topic.
Compounded ODT dosage options
The most commonly reported tirzepatide ODT strengths from compounding pharmacies were 3 mg, 4 mg, and 5 mg tablets. Some pharmacies offered additional strengths, but these three represented the core product line for most providers.
These daily doses were administered once per day, which represents a fundamental difference from injectable tirzepatide. The FDA-approved injectable versions, Mounjaro and Zepbound, are dosed once weekly. A daily oral dose and a weekly injection deliver medication through completely different pharmacokinetic profiles. The weekly injection creates a sustained blood level that rises after injection and slowly declines over seven days. A daily oral dose, assuming meaningful absorption occurs, would create daily peaks and troughs in blood concentration.
Whether the daily dosing pattern of compounded ODT produced equivalent appetite suppression and metabolic effects compared to weekly injections was never established through clinical research. The doses were determined by individual compounding pharmacies and prescribing providers, not through the kind of systematic dose-finding studies that preceded FDA approval of the injectable products.
How ODT dosing compares to injectable dosing
FDA-approved injectable tirzepatide comes in six dose strengths. The standard titration schedule starts at 2.5 mg weekly for four weeks, then increases to 5 mg weekly for at least four weeks, with potential escalation to 7.5 mg, 10 mg, 12.5 mg, and 15 mg based on tolerability and response.
At the highest dose of 15 mg weekly, clinical trials demonstrated average weight loss of approximately 20.9 percent of body weight over 72 weeks. When combined with intensive lifestyle modifications, that number approached 26 percent.
The compounded ODT doses of 3 to 5 mg daily might appear numerically similar to the lower injectable doses. But direct numerical comparison is misleading because the routes of administration have vastly different bioavailabilities. If sublingual absorption achieves 10 percent bioavailability (an optimistic estimate with no published data to support it), a 5 mg daily ODT would deliver approximately 0.5 mg of active tirzepatide to the bloodstream per day, or about 3.5 mg over a week. That would roughly correspond to the 2.5 mg weekly starting dose of the injectable, assuming the 10 percent bioavailability estimate is even accurate.
The math highlights a fundamental problem. Without knowing the actual bioavailability of the compounded formulation, calculating an equivalent effective dose is guesswork.
Starting doses and titration schedules
Providers who prescribed compounded tirzepatide ODT typically started patients at the lowest available dose, usually 3 mg daily, for two to four weeks before considering increases. This approach mirrored the gradual titration philosophy of the injectable protocol, where starting low reduces the severity of gastrointestinal side effects.
Some prescribers used a schedule like this:
Weeks 1 to 4: 3 mg daily
Weeks 5 to 8: 4 mg daily
Weeks 9 and beyond: 5 mg daily (if tolerated and additional effect desired)
Others kept patients at the starting dose longer if they were experiencing adequate appetite suppression. The variability in prescribing patterns reflected the absence of standardized dosing guidelines for compounded oral formulations, something that only comes from controlled clinical trials.
For anyone currently using or considering tirzepatide in any form, tools like the dosage calculator and compounded tirzepatide dosage calculator on SeekPeptides can help you understand dose conversions and unit measurements for the injectable formulations that remain available.
How to use tirzepatide ODT
For those who had access to tirzepatide ODT before the compounding ban, or who may still have remaining supply, understanding proper administration technique is important for maximizing whatever absorption the formulation can achieve.
Step by step administration
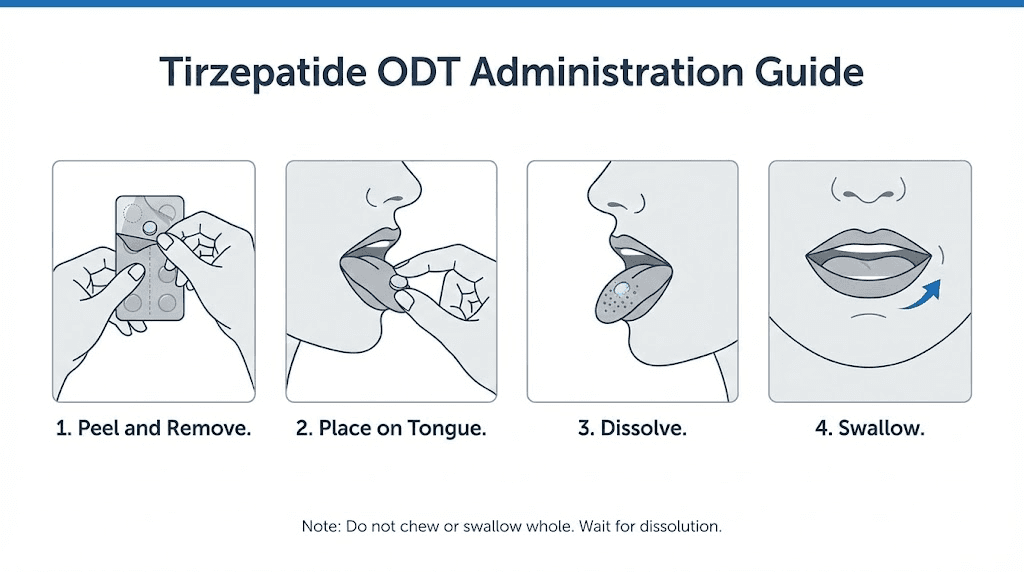
The administration process for tirzepatide ODT follows the standard protocol for any orally disintegrating medication:
Step 1: Preparation. Wash and thoroughly dry your hands. Moisture on fingers can begin dissolving the tablet prematurely, reducing the dose that makes it to your tongue intact.
Step 2: Removal from packaging. Peel back the foil packaging carefully. Do not push the tablet through the foil, as this can crumble or fragment the tablet. ODT formulations are deliberately fragile to enable rapid dissolution, which makes them susceptible to damage from rough handling.
Step 3: Placement. Place the tablet directly on the tongue. Most providers recommended placing it under the tongue (sublingual position) for maximum absorption through the highly vascular sublingual tissue. Allow it to dissolve completely, which typically takes 30 to 60 seconds.
Step 4: Avoid swallowing. During the dissolution period, try not to swallow. The longer the dissolved medication stays in contact with the oral mucosa, the more opportunity there is for absorption through the sublingual and buccal tissues. After the tablet has fully dissolved, you can swallow normally.
Step 5: Wait before eating or drinking. Most prescribers recommended waiting at least 30 minutes before consuming food or beverages. Eating too soon after administration could reduce absorption and increase the likelihood that the medication gets carried into the stomach with food rather than absorbed through the mouth.
Timing and food considerations
Tirzepatide ODT was typically prescribed for morning use, taken on an empty stomach at least 30 minutes before the first meal of the day. This timing recommendation served two purposes.
First, an empty stomach reduces the volume of saliva production triggered by food anticipation, which means less saliva to wash the dissolved medication toward the throat. Second, taking the medication well before eating ensures that the initial absorption window is not disrupted by the mechanical action of chewing and swallowing food.
The dietary considerations during tirzepatide use, regardless of formulation, remain the same. Understanding which foods to avoid while on tirzepatide and following a supportive eating plan can help maximize results. Many users found that combining their medication with a structured meal plan produced better outcomes than medication alone.
Common mistakes to avoid
Several administration errors were commonly reported among tirzepatide ODT users:
Handling tablets with wet hands. This is the most frequent mistake. Even slightly damp fingers can begin dissolving the tablet before placement, reducing the effective dose and creating an uneven dissolution on the tongue.
Swallowing too quickly. The natural impulse when something dissolves in your mouth is to swallow. But swallowing the dissolved medication sends it to the stomach, where the peptide faces degradation. Consciously delaying swallowing for at least 60 seconds after placement improves the sublingual absorption opportunity.
Drinking water immediately after. Some users would drink water right after the tablet dissolved, essentially flushing any remaining medication into the stomach. The 30-minute waiting period exists specifically to allow absorbed medication to enter the bloodstream through the oral tissues.
Storing tablets improperly. ODT formulations are extremely sensitive to moisture. Removing tablets from their individual packaging and storing them in pill organizers or open containers exposes them to ambient humidity, which can cause premature dissolution or degradation of the active ingredient.
Tirzepatide ODT vs injectable tirzepatide
This comparison is perhaps the most important section for anyone evaluating their options. The differences between ODT and injectable formulations are not just about needle avoidance. They span effectiveness, bioavailability, cost, and regulatory status.
Factor | Tirzepatide ODT (compounded) | Injectable tirzepatide (FDA-approved) |
|---|---|---|
FDA approved | No | Yes (Mounjaro, Zepbound) |
Clinical trial data | None | Extensive (SURPASS and SURMOUNT trials) |
Estimated bioavailability | Unknown (likely 5-15%) | Approximately 80% |
Dosing frequency | Daily | Weekly |
Available doses | 3 mg, 4 mg, 5 mg daily | 2.5 to 15 mg weekly |
Weight loss evidence | Anecdotal only | Up to 20.9% body weight in trials |
Current availability | Largely unavailable since March 2025 | Widely available |
Needles required | No | Yes (subcutaneous injection) |
Storage | Room temperature, protect from moisture | Refrigeration recommended |
Effectiveness and weight loss outcomes
Injectable tirzepatide has some of the most robust clinical evidence of any weight management medication in history. The SURMOUNT-1 trial enrolled 2,539 adults with obesity or overweight and demonstrated weight loss of 15 percent at the 5 mg dose, 19.5 percent at the 10 mg dose, and 20.9 percent at the 15 mg dose over 72 weeks. These results led to FDA approval of Zepbound for chronic weight management.
The SURPASS trials, which evaluated tirzepatide for type 2 diabetes under the brand name Mounjaro, showed substantial HbA1c reductions alongside significant weight loss. Across multiple trials, tirzepatide outperformed every comparator, including semaglutide at its highest dose.
Compounded tirzepatide ODT has zero clinical trial data. No randomized controlled trials. No dose-finding studies. No pharmacokinetic analyses published in peer-reviewed journals. The evidence for ODT effectiveness consists entirely of anecdotal reports from individual users and providers. While some of those reports describe meaningful before and after results, anecdotal evidence cannot control for placebo effects, concurrent lifestyle changes, or variations in formulation quality between different compounding pharmacies.
That matters. It matters a lot.
Bioavailability differences
The bioavailability gap between subcutaneous injection and oral mucosal absorption for a large peptide molecule like tirzepatide is substantial. Subcutaneous injection delivers approximately 80 percent of the dose to the bloodstream. Sublingual and buccal absorption of peptides without specialized enhancers typically ranges from 1 to 15 percent in published literature, depending on the specific molecule and formulation.
For context, if an injectable 5 mg dose delivers 4 mg of active tirzepatide to the bloodstream (80 percent of 5 mg), a daily 5 mg ODT with 10 percent bioavailability would deliver 0.5 mg per day, or 3.5 mg per week. But we do not actually know whether compounded tirzepatide ODT achieves 10 percent bioavailability. It could be higher. It could be much lower. Without pharmacokinetic data, this remains entirely speculative.
The oral versus injection comparison comes down to certainty. With an injection, you know how much drug is entering your system. With an ODT, you do not.
Convenience and compliance factors
The appeal of tirzepatide ODT is obvious. No needles. No injection sites to rotate. No need for reconstitution or bacteriostatic water mixing. No syringes to dispose of safely. For people with genuine needle phobia, which affects an estimated 20 to 25 percent of the adult population, these are not trivial benefits.
However, daily dosing versus weekly dosing creates its own compliance challenge. Missing one daily dose of a medication with uncertain absorption is very different from missing one weekly injection of a medication with proven bioavailability. Over the course of a month, a weekly injectable requires 4 doses. A daily ODT requires 28 to 30 doses. The more frequently a medication must be taken, the higher the probability of missed doses.
Users who value convenience should also consider that injectable tirzepatide comes in pre-filled pens that require no mixing, no measuring, and no specialized technique. The injection itself takes about 5 seconds and uses a very small needle that most users describe as virtually painless.
Cost considerations
Before the compounding ban, compounded tirzepatide ODT was generally less expensive than brand-name Mounjaro or Zepbound, which is one reason it attracted so many users. Compounded formulations from 503A pharmacies often cost a fraction of the branded product price.
However, comparing costs requires accounting for effectiveness. If a $200 per month compounded ODT delivers uncertain absorption while a $300 per month injectable delivers proven clinical results, the cheaper option may not actually be the better value. Cost per unit of actual drug absorbed is a more meaningful comparison than cost per tablet or cost per month.
For those evaluating price across different affordable tirzepatide options, the peptide cost calculator on SeekPeptides can help compare true costs across different formulations and providers.
Tirzepatide ODT vs drops and sublingual liquid
The oral tirzepatide market included more than just ODT tablets. Sublingual drops and liquid formulations also existed as compounded options. Understanding the differences between these formats helps clarify what each one was attempting to achieve and where each fell short.
Key differences between ODT and liquid drops
Tirzepatide drops were liquid formulations administered under the tongue using a dropper or syringe. The user would place a measured volume of liquid beneath the tongue, hold it there for 60 to 90 seconds, then swallow. This delivery method offered more precise dose control than ODT tablets, since volume could be adjusted in small increments.
ODT tablets, by contrast, came in fixed dose strengths. You could not easily split or adjust an orally disintegrating tablet. What came in the packaging was the dose you took. This made dose titration less flexible compared to liquid formulations.
The trade-off was convenience. ODT tablets required no measuring, no droppers, and no refrigeration in most cases. Liquid formulations typically needed refrigeration and had shorter shelf lives once opened. Tablets could be carried in a pocket or purse without worrying about leaking.
Absorption comparison between formats
Both ODT and sublingual drops rely on the same basic absorption mechanism, diffusion through the oral mucosa into the underlying capillary network. The theoretical difference is that a liquid held under the tongue maintains more uniform contact with the absorptive tissue than a dissolving tablet, which may distribute unevenly across the oral cavity.
In pharmacological terms, a pre-dissolved liquid should produce faster and potentially more consistent absorption than a tablet that must first dissolve before absorption can begin. The dissolution step in an ODT adds a delay and introduces variability based on individual saliva production rates, tongue positioning, and how completely the tablet dissolves before the user swallows.
That said, neither format has published bioavailability data for tirzepatide specifically. The comparison between ODT and drops is theoretical. Without head-to-head pharmacokinetic studies, no one can definitively state that one format delivers more active drug than the other.
Which format worked better
Anecdotal reports from providers and users were mixed. Some preferred the simplicity of ODT tablets. Others found that sublingual drops gave them more perceived appetite suppression, though this could reflect placebo effects, different compounding pharmacy formulations, or simply individual variation in absorption.
The honest answer is that without controlled studies, no one knows which format delivered more drug to the bloodstream. Both formats relied on the same absorption pathway with the same fundamental limitations. If you are comparing oral semaglutide drops and ODT formats for semaglutide, the same uncertainty applies.

Side effects of tirzepatide ODT
Side effects from tirzepatide ODT are expected to mirror those of injectable tirzepatide, since the active ingredient is the same molecule. However, the oral delivery route introduces some unique considerations that the injectable form does not share.
Gastrointestinal side effects
The most commonly reported side effects of tirzepatide, regardless of delivery method, involve the gastrointestinal system. This makes biological sense because GLP-1 and GIP receptors are heavily expressed throughout the gut, and activating them directly affects gastric motility, gastric emptying speed, and intestinal function.
Common GI side effects include:
Nausea reported in 30 to 40 percent of ODT users, compared to 18 to 24 percent for injectable tirzepatide. The higher incidence with ODT may reflect direct oral cavity and esophageal exposure to the medication before absorption.
Diarrhea affecting 12 to 20 percent of users, typically most prominent during the first two to four weeks of treatment or after dose increases.
Constipation affecting 6 to 10 percent, which may seem contradictory to diarrhea but reflects the complex and sometimes opposing effects of incretin hormones on different segments of the digestive tract.
Vomiting reported in 15 to 20 percent, again higher than injectable rates, possibly related to the taste or texture of the dissolving tablet.
Decreased appetite is technically a desired effect rather than a side effect, but the degree of appetite reduction can occasionally be excessive, leading to inadequate caloric intake in some users.
The gastrointestinal side effects of tirzepatide are generally dose-dependent. They tend to be worst during initiation and dose escalation, then improve as the body adjusts over two to four weeks at each dose level. This pattern holds for both injectable and oral formulations.
Non-gastrointestinal side effects
Beyond the GI tract, tirzepatide users may experience:
Headaches are reported by a meaningful percentage of users across all tirzepatide formulations. These typically occur early in treatment and resolve within the first few weeks. Dehydration related to reduced food and fluid intake may contribute.
Fatigue and tiredness can accompany the caloric restriction that tirzepatide promotes. When your body is running on significantly fewer calories than it is accustomed to, energy levels may dip, especially during the first month. Understanding GLP-1 related fatigue and how to manage it can help users adjust.
Body aches and muscle pain are reported by some users, particularly those losing weight rapidly. Rapid weight loss can stress the musculoskeletal system, and adequate protein intake becomes critical to preserving lean mass during treatment.
Sleep disturbances including insomnia have been reported, though the mechanism is not fully understood. Changes in blood sugar regulation, altered eating patterns, and the general metabolic shift that accompanies significant weight loss may all contribute.
Anxiety has been noted in some users, potentially related to hormonal shifts, changes in gut-brain signaling through the vagus nerve, or the psychological impact of rapid body changes.
Hair loss is a recognized side effect of rapid weight loss in general, not specific to tirzepatide. When the body loses weight quickly, it can redirect resources away from hair growth, leading to a temporary condition called telogen effluvium. This typically resolves once weight stabilizes.
Menstrual changes have been reported by female users. Fat tissue produces estrogen, and significant fat loss can alter hormonal balance enough to affect cycle regularity. Women who are or might become pregnant while on tirzepatide should consult their provider immediately, as the medication is contraindicated in pregnancy.
How ODT side effects compare to injectable
The anecdotal evidence from the compounded ODT era suggests slightly higher rates of nausea and vomiting compared to the injectable form. Several possible explanations exist for this observation.
Direct contact between the dissolving medication and the oral and esophageal mucosa may trigger local nausea responses that subcutaneous injection avoids. The taste of the dissolving tablet, even with flavoring agents, could contribute to emetic responses. And variable absorption from the oral route might create inconsistent blood levels that produce more gastrointestinal disturbance than the steady pharmacokinetic profile of weekly injections.
Conversely, ODT users avoided injection site reactions entirely. Redness, swelling, and itching at the injection site, which affect approximately 3 to 5 percent of injectable tirzepatide users, were not a concern with the oral format.
For a comprehensive comparison of tirzepatide side effects across formulations, including how they compare to semaglutide side effect profiles, the evidence strongly favors injectable delivery for both predictability and tolerability.
Storage and handling requirements
Proper storage is critical for maintaining the potency and safety of any medication, and tirzepatide ODT has specific requirements that differ from the injectable formulations.
Temperature and moisture considerations
Tirzepatide ODT should be stored at room temperature between 68 and 77 degrees Fahrenheit (20 to 25 degrees Celsius). Unlike injectable tirzepatide, which requires refrigeration, the ODT format was designed for room temperature storage.
However, the ODT formulation is extremely sensitive to moisture. The same property that makes the tablet dissolve rapidly on the tongue, its high affinity for moisture, means it will also begin dissolving if exposed to humidity in the air. This is why tirzepatide ODT tablets were individually packaged in foil-sealed blisters.
Never store ODT tablets in:
Pill organizers or weekly medication containers
Open containers or dishes
Bathrooms or kitchens where humidity levels fluctuate
Any environment where the tablets can contact moisture before use
If a tablet appears discolored, crumbled, or partially dissolved in its packaging, do not use it. These signs indicate moisture exposure that may have degraded the active ingredient.
Shelf life and expiration
Compounded medications typically have shorter beyond-use dates than commercially manufactured pharmaceuticals. Compounding pharmacies generally assigned tirzepatide ODT a shelf life of 60 to 90 days from the date of compounding, depending on their stability testing data.
Compare this to FDA-approved injectable tirzepatide pens, which have a shelf life of up to four years when stored properly under refrigeration. The expiration considerations for tirzepatide vary significantly between compounded and commercially manufactured products.
Using an expired or improperly stored compounded medication is particularly risky because degradation products from peptide breakdown are unpredictable. While an expired conventional tablet might simply lose potency, an expired peptide formulation could potentially contain degradation byproducts that cause unexpected effects.
FDA status and regulatory landscape
The regulatory story behind tirzepatide ODT is complex, and understanding it is essential for anyone evaluating their current options. The rules changed dramatically in 2025, and the implications are still unfolding.
Why tirzepatide ODT was never FDA approved
No oral tirzepatide product, including ODT, has ever received FDA approval. The FDA approved tirzepatide only as a subcutaneous injection, first as Mounjaro for type 2 diabetes in May 2022, then as Zepbound for chronic weight management in November 2023.
FDA approval requires extensive clinical trials demonstrating safety and efficacy for a specific formulation, dose, and route of administration. Eli Lilly, the manufacturer of tirzepatide, never submitted clinical trial data for an oral formulation because developing one that achieves adequate bioavailability remains a significant pharmaceutical challenge. The company may pursue oral delivery in the future, but as of early 2026, no oral tirzepatide clinical trials have been completed or published.
This means every compounded tirzepatide ODT that was ever dispensed in the United States was a non-FDA-approved product. Compounded medications, by definition, are not reviewed by the FDA for safety, efficacy, or quality before being dispensed to patients.
The compounding loophole explained
How did compounding pharmacies legally produce tirzepatide ODT if it was never FDA approved? The answer lies in a specific provision of federal pharmacy law.
Under the Federal Food, Drug, and Cosmetic Act, 503A compounding pharmacies can compound copies of commercially available drugs when those drugs appear on the FDA drug shortage list. When a medication is in shortage, meaning that the commercially available versions cannot meet patient demand, compounding pharmacies are granted temporary authority to prepare alternative versions to help fill the gap.
Tirzepatide appeared on the FDA drug shortage list beginning in late 2022. Demand for Mounjaro and later Zepbound dramatically exceeded supply as the medications gained attention for their weight loss effectiveness. During this shortage period, 503A compounding pharmacies legally compounded tirzepatide in various formulations, including compounded injectable preparations, sublingual drops, and ODT tablets.
The compounding pharmacies were not copying the exact commercial formulation. They were creating their own formulations of the same active ingredient, tirzepatide, using their own excipients and delivery formats. This meant wide variation in formulation quality, consistency, and bioavailability across different pharmacies.
The March 2025 compounding ban
On October 2, 2024, the FDA determined that tirzepatide was no longer in shortage. All doses of Mounjaro and Zepbound were deemed readily available to meet patient demand. This triggered a transition period during which compounding pharmacies were required to wind down their tirzepatide operations.
The FDA set March 19, 2025 as the final date for enforcement discretion. After that date, compounding pharmacies that continued producing any tirzepatide product, including ODT, were in violation of federal law. The legal basis was straightforward. The shortage that justified compounding had been resolved, which removed the legal authority for pharmacies to compound copies of the commercially available drug.
The US District Court for the Northern District of Texas upheld the FDA decision, concluding that the agency acted within its statutory authority in determining that tirzepatide was no longer in shortage. Legal challenges from compounding pharmacy associations were largely unsuccessful in reversing or delaying the enforcement deadline.
This regulatory change affected not only tirzepatide ODT but all compounded tirzepatide formulations, including injectables and sublingual drops. The entire compounded tirzepatide market was effectively shut down in a matter of months.
Current availability and legal considerations
The question everyone searching for tirzepatide ODT wants answered is simple. Can you still get it? The answer is more nuanced than a simple yes or no.
Can you still get compounded tirzepatide ODT
Officially, no. As of March 2025, compounding pharmacies in the United States are not legally authorized to compound tirzepatide in any form, including ODT. The FDA has been clear that continued compounding of tirzepatide constitutes a violation of the Federal Food, Drug, and Cosmetic Act now that the shortage has been resolved.
In practice, some websites and telehealth platforms continued offering products labeled as compounded tirzepatide after the March 2025 deadline. Reports indicate that certain online providers maintained availability for varying periods after the ban took effect. Whether those products actually contained tirzepatide, contained the labeled dose, or were produced in compliance with any pharmaceutical standards is impossible to verify independently.
Legal risks for pharmacies and patients
For pharmacies, the risks are substantial and clear. Any compounding pharmacy or outsourcing facility that continues preparing tirzepatide products faces potential FDA enforcement action, including warning letters, injunctions, and criminal prosecution in severe cases. Several pharmacies received FDA warning letters within weeks of the March 2025 deadline.
For patients, the legal risk is lower but the safety risk is significant. While patients are not typically targeted by FDA enforcement for purchasing compounded medications for personal use, buying from a source operating outside legal authority means there are no quality controls, no regulatory oversight, and no recourse if the product causes harm.
Products sold through unauthorized channels may contain incorrect doses, inactive ingredients that differ from the label, contaminants from unregulated manufacturing processes, or no tirzepatide at all. The FDA has specifically warned about counterfeit and adulterated GLP-1 products circulating after the compounding ban.
State by state considerations
Pharmacy regulation occurs at both the federal and state level. While the FDA ban on tirzepatide compounding applies nationally, individual state pharmacy boards may vary in their enforcement approaches and timelines. Some states moved quickly to enforce the federal directive, while others had slower implementation.
Additionally, some states have explored legislative measures related to compounding access for medications that were previously in shortage. The regulatory environment continues to evolve, and what applies in one state may not apply in another.
For the most current information on availability in your area, consulting with a licensed healthcare provider who stays current on pharmaceutical regulations is the safest approach. SeekPeptides maintains updated guides on the evolving legal landscape surrounding compounded GLP-1 medications.
FDA-approved alternatives to tirzepatide ODT
If tirzepatide ODT is no longer available, what options exist for people who want tirzepatide or similar medications without needles, or with needles that are as painless as possible?
Injectable tirzepatide with Mounjaro and Zepbound
The most direct alternative is the FDA-approved injectable tirzepatide itself. Mounjaro (for type 2 diabetes) and Zepbound (for weight management) deliver the exact same molecule with proven bioavailability and extensive clinical trial support.
Modern insulin-style pens use extremely thin needles, typically 31 or 32 gauge, that are 4 to 5 millimeters long. Most users report minimal to no pain during injection. The best injection sites include the abdomen, thigh, and upper arm, with the stomach being the most popular for its ease of access and consistent absorption.
For those concerned about needles, several practical strategies can help:
Applying a topical numbing cream (lidocaine) 15 to 20 minutes before injection
Using ice on the injection site for 30 seconds prior
Pinching the skin to reduce sensation during injection
Rotating injection sites to prevent tissue irritation
The weekly dosing schedule means only 52 injections per year. Many users report that their needle anxiety decreases significantly after the first few injections once they realize how minimal the sensation actually is.
Oral semaglutide as an alternative
Rybelsus (oral semaglutide) is currently the only FDA-approved oral GLP-1 receptor agonist. It uses SNAC technology to achieve gastric absorption and comes in 3 mg, 7 mg, and 14 mg daily tablets.
While Rybelsus is approved for type 2 diabetes rather than weight management, healthcare providers can prescribe it off-label for weight loss. Higher dose oral semaglutide (25 mg and 50 mg) has been studied in clinical trials and showed more substantial weight loss results, though these higher doses are not yet commercially available.
The key limitation of oral semaglutide compared to tirzepatide is the single-receptor mechanism. Semaglutide targets only GLP-1 receptors, while tirzepatide targets both GLP-1 and GIP receptors. Clinical trials consistently showed greater weight loss with injectable tirzepatide compared to injectable semaglutide at their respective maximum doses. The dosage comparison between tirzepatide and semaglutide reflects this difference in potency.
For those exploring the oral semaglutide landscape, understanding the absorption requirements is important. Oral semaglutide must be taken on a completely empty stomach with no more than 4 ounces of plain water, followed by at least 30 minutes of fasting before any food, drink, or other medications. These restrictions are necessary to maintain consistent absorption through the SNAC-mediated pathway.
Other GLP-1 options and future developments
The GLP-1 and dual-agonist medication space is evolving rapidly. Several developments are worth tracking:
Orforglipron is an oral non-peptide GLP-1 receptor agonist being developed by Eli Lilly, the same company that makes tirzepatide. Unlike tirzepatide, orforglipron is a small molecule rather than a peptide. This means it can potentially survive stomach acid and achieve meaningful oral bioavailability without specialized absorption enhancers. Phase 3 clinical trials have shown promising weight loss results, and if approved, orforglipron could become the first truly effective oral option in this drug class.
Survodutide is a dual glucagon and GLP-1 receptor agonist in development that targets a different receptor combination than tirzepatide. While currently only in injectable form, the diverse pipeline of incretin-based medications suggests that more options, including potentially oral ones, will become available in coming years.
GLP-1 patches represent another needle-free delivery approach in development. Transdermal patches that deliver GLP-1 receptor agonists through the skin could offer a future alternative for needle-averse patients, though no GLP-1 patches are currently FDA approved.
Mazdutide is another dual-agonist in development that has shown competitive weight loss results in Chinese clinical trials. While primarily injectable, the expanding competitive landscape should eventually drive development of more delivery options across the entire drug class.
Making the right choice for your situation
Choosing the right GLP-1 medication and delivery format depends on your individual circumstances, medical history, and treatment goals. There is no universal best option. There is only the best option for you, determined in partnership with your healthcare provider.
When injectable tirzepatide makes more sense
Injectable tirzepatide is the strongest choice when:
You want maximum proven effectiveness. No other currently available medication produces the degree of weight loss demonstrated by injectable tirzepatide at its highest doses. If your primary goal is maximum weight loss results, the injectable form has the strongest evidence base.
You want dosing certainty. With an injection, you know exactly how much drug enters your system. The precise dosing in units eliminates the absorption uncertainty that plagued oral formulations.
You prefer weekly convenience. One injection per week is less frequent than daily oral dosing. For busy people, the weekly schedule can actually be more convenient than remembering a daily tablet with strict timing and food restrictions.
You have tried oral GLP-1 options without adequate response. If oral semaglutide did not produce sufficient results, the superior bioavailability and dual-receptor mechanism of injectable tirzepatide may provide the additional efficacy needed.
When oral GLP-1 options fit better
Oral formulations may be preferable when:
You have severe needle phobia that cannot be managed. True needle phobia is a real medical condition. While many people overcome injection anxiety with practice and education, some individuals have phobic responses that make self-injection genuinely distressing. For these patients, oral semaglutide (Rybelsus) provides a legal, FDA-approved alternative.
You are starting GLP-1 treatment for the first time. Some providers prefer to start patients on oral semaglutide to establish tolerability before escalating to injectable tirzepatide. This step-wise approach can be particularly appropriate for patients who are hesitant about injections.
Your insurance covers oral but not injectable formulations. Coverage varies widely between insurance plans. In some cases, oral semaglutide may have lower out-of-pocket costs or fewer prior authorization requirements than injectable tirzepatide.
Questions to ask your healthcare provider
Before making a decision about any GLP-1 medication, discuss these questions with your provider:
Based on my health history and goals, which medication and delivery format do you recommend?
What weight loss can I realistically expect with each option?
What are the specific side effects I should watch for, given my medical history?
How will we monitor my progress and adjust dosing?
What dietary changes should I implement alongside medication? (See our guides on foods to avoid and supplements to consider)
What happens if I need to switch between medications?
Are there any drug interactions with my current medications?
A good provider will tailor their recommendation to your specific situation rather than applying a one-size-fits-all approach. They should be willing to discuss the evidence for each option honestly, including the limitations of any formulation you are considering.
Understanding compounded tirzepatide formulations
To place tirzepatide ODT in proper context, understanding the broader landscape of compounded tirzepatide formulations helps clarify what set ODT apart and where it fit within the compounding ecosystem.
Injectable compounded tirzepatide
Before the March 2025 ban, the most common compounded tirzepatide product was a subcutaneous injectable. These compounded tirzepatide injections used the same subcutaneous delivery route as the FDA-approved products and theoretically offered similar bioavailability, since the route of administration was identical.
Compounded injectables were available from pharmacies like Empower Pharmacy, which was one of the largest compounding providers during the shortage period. These injectables required reconstitution in some cases or came as ready-to-use solutions.
The key difference between compounded injectable and FDA-approved injectable was quality assurance. Brand-name Mounjaro and Zepbound are manufactured under rigorous FDA-inspected cGMP (current Good Manufacturing Practice) conditions. Compounded injectables were produced under state pharmacy board oversight, which varies significantly in stringency.
Enhanced formulations and compounds
Many compounding pharmacies offered tirzepatide in combination with other ingredients. Common additions included:
B12 (cyanocobalamin or methylcobalamin) was frequently added to tirzepatide formulations. The rationale was that GLP-1 medications can reduce nutrient absorption by slowing gastric emptying, and B12 supplementation might help prevent deficiency. The tirzepatide with B12 combination was one of the most popular compounded formulations.
Glycine was added to some formulations as a stabilizer and potential bioavailability enhancer. The tirzepatide glycine B12 compound became a particularly popular three-in-one formulation.
Niacinamide appeared in some compounded tirzepatide formulations based on claims that it could support metabolic function and reduce certain side effects.
Methylcobalamin, the active form of B12, was preferred by some providers over regular cyanocobalamin for its potentially better absorption and utilization.
Whether these additions provided meaningful clinical benefits beyond the tirzepatide itself was never established through controlled studies. They may have been beneficial, they may have been neutral, or in some cases they may have affected the stability or activity of the tirzepatide in the compounded formulation.
Microdosing and alternative protocols
Some users and providers experimented with non-standard dosing approaches during the compounding era. Microdosing tirzepatide involved using significantly lower doses than the standard protocol, sometimes as low as one-quarter of the typical starting dose, with the goal of reducing side effects while maintaining some appetite suppression.
The microdose schedule varied between providers but generally involved very gradual dose escalation over extended periods. This approach gained traction particularly among users who experienced significant gastrointestinal side effects at standard doses.
For users who have been on compounded tirzepatide and need to transition, understanding the conversion between different GLP-1 medications is important for maintaining treatment continuity.
Weight management beyond medication
Whether you use tirzepatide ODT, injectable tirzepatide, oral semaglutide, or any other GLP-1 medication, the medication works best as part of a comprehensive approach that includes dietary and lifestyle modifications.
Nutritional support during GLP-1 treatment
GLP-1 medications significantly reduce appetite, which creates both an opportunity and a challenge. The opportunity is obvious, reduced caloric intake drives weight loss. The challenge is ensuring that the calories you do consume provide adequate nutrition, particularly protein, to prevent excessive muscle loss during rapid weight reduction.
Following a structured tirzepatide diet plan that prioritizes protein intake, micronutrient density, and adequate hydration can make a significant difference in outcomes. Many users find that the foods recommended during GLP-1 treatment overlap substantially with evidence-based healthy eating patterns.
Protein shakes designed for GLP-1 users can be particularly helpful on days when appetite suppression makes eating full meals difficult. Aiming for 0.7 to 1.0 grams of protein per pound of body weight daily helps preserve muscle mass during weight loss.
Certain supplements may also support the body during the metabolic changes that accompany GLP-1 treatment. Electrolytes, fiber, and specific vitamins and minerals that may be under-consumed during periods of reduced food intake deserve particular attention.
Monitoring progress and adjusting course
Weight loss with GLP-1 medications does not follow a straight line. Plateaus are common and expected. Understanding that tirzepatide takes time to reach full effect helps manage expectations during the early weeks of treatment.
If you feel that your current medication is no longer producing results, several adjustments are worth discussing with your provider. Dose increases, dietary modifications, addition of physical activity, and in some cases, switching to a different medication can all help overcome stalled progress.
For women, being aware that tirzepatide can affect menstrual cycles helps distinguish between medication-related changes and other potential causes of cycle irregularity. Similarly, knowing that alcohol interacts with tirzepatide in ways that may worsen side effects and reduce treatment effectiveness is important for social situations.
SeekPeptides members access comprehensive tracking tools, detailed protocol guides, and a community of researchers who have navigated these exact challenges. The platform provides evidence-based guidance for optimizing GLP-1 treatment outcomes across all available formulations.
The future of needle-free tirzepatide
The compounding ban on tirzepatide ODT does not mean that needle-free tirzepatide delivery is permanently off the table. Several developments in pharmaceutical science and regulatory policy suggest that legitimate oral and non-injectable options may eventually become available.
Eli Lilly oral development programs
Eli Lilly, the manufacturer of tirzepatide, has invested heavily in oral peptide delivery research. Their most advanced oral program is orforglipron, a small-molecule GLP-1 receptor agonist that, unlike tirzepatide, is not a peptide and therefore does not face the same bioavailability challenges when taken orally.
Orforglipron has completed Phase 3 clinical trials showing weight loss in the range of 12 to 14 percent over 36 to 72 weeks. While this is less than the 20 percent achieved by injectable tirzepatide at the highest dose, it represents a meaningful result for an oral medication that does not require any special administration techniques or absorption enhancers.
Whether Eli Lilly will also develop a true oral formulation of tirzepatide itself remains speculative. The company has not publicly disclosed specific programs targeting oral tirzepatide delivery, but given the enormous commercial incentive and ongoing advances in oral peptide technology, it would be surprising if they were not exploring the possibility.
Advances in oral peptide delivery technology
The pharmaceutical industry has made significant progress in oral peptide delivery over the past decade. The success of SNAC-enhanced oral semaglutide demonstrated that meaningful oral bioavailability of a GLP-1 agonist is achievable, even if current absorption rates remain low compared to injection.
Next-generation approaches include:
Permeation enhancer technology that improves peptide transport across the intestinal wall without damaging the tissue.
Nanoparticle encapsulation that protects peptides from enzymatic degradation in the GI tract.
Microneedle patches that deliver peptides through the skin using microscopic needles that are painless and dissolve after insertion.
Mucoadhesive formulations that adhere to the intestinal wall and release peptide over extended periods for improved absorption.
These technologies are at various stages of development, and some may eventually enable oral or transdermal delivery of tirzepatide or similar dual-agonist compounds with bioavailability approaching that of subcutaneous injection.
Regulatory considerations for future products
Any future oral or non-injectable tirzepatide product would need to go through the full FDA approval process, including large-scale clinical trials demonstrating safety and efficacy. This process typically takes 5 to 10 years from initial development to market availability.
The FDA experience with compounded tirzepatide ODT, specifically the quality concerns and adverse event reports that accompanied unregulated formulations, may actually accelerate the regulatory pathway for legitimate oral tirzepatide products. Demonstrated patient demand combined with documented safety concerns from compounded versions creates a strong case for prioritizing the development and review of properly studied oral alternatives.
Practical guidance for current and former ODT users
If you previously used tirzepatide ODT and need to transition to another option, or if you are currently evaluating alternatives, this section provides actionable next steps.
Transitioning from compounded ODT to injectable
The transition from a compounded oral formulation to FDA-approved injectable tirzepatide requires coordination with a healthcare provider. Key considerations include:
Dose equivalence is uncertain. Because the bioavailability of compounded ODT was never measured, your provider cannot calculate a direct conversion from your ODT dose to an injectable dose. Most providers recommend starting injectable tirzepatide at the standard starting dose of 2.5 mg weekly and titrating up based on response, regardless of your previous ODT dose.
Side effects may differ. Even though you may have already adjusted to tirzepatide through the oral route, switching to injectable delivery changes the pharmacokinetic profile. You may experience a different side effect pattern, potentially less nausea than the oral route but with the addition of possible injection site reactions.
Timing matters. If you stop your compounded ODT, any tirzepatide in your system from the oral route will clear within a few days. Starting injectable tirzepatide can begin immediately after stopping the oral form, as there is no meaningful washout period needed.
Finding legitimate providers
With the compounding ban in effect, the safest route to tirzepatide is through legitimate prescribers who can prescribe the FDA-approved brands. Several types of providers can prescribe Mounjaro or Zepbound:
Primary care physicians
Endocrinologists
Obesity medicine specialists
Certain telehealth platforms that have been vetted for legitimacy
Be extremely cautious of any provider or website still offering "compounded tirzepatide" after March 2025. Legitimate providers will prescribe the FDA-approved products and can help with insurance authorization, manufacturer savings programs, and dose optimization.
For comprehensive information on different tirzepatide providers and pharmacy options, including reviews and comparisons, see our guides on ProRx Tirzepatide, Orderly Meds, Southend Pharmacy, Priority Meds, and BPI Labs offerings.
When to seek immediate medical attention
Regardless of which tirzepatide formulation you have used or are currently using, seek immediate medical care if you experience:
Severe abdominal pain that does not resolve (potential pancreatitis)
Signs of allergic reaction including swelling, difficulty breathing, or widespread rash
Persistent vomiting lasting more than 24 hours
Signs of dehydration including dark urine, dizziness, and rapid heartbeat
Vision changes (potential thyroid concerns)
Severe joint pain or muscle pain that interferes with daily function
These symptoms warrant professional evaluation whether you are on injectable, oral, or any other formulation of tirzepatide or GLP-1 medications.
Frequently asked questions
Is tirzepatide ODT still available?
Tirzepatide ODT is not legally available through US compounding pharmacies as of March 2025. The FDA resolved the tirzepatide shortage and ended enforcement discretion for compounding. Any source still offering compounded tirzepatide ODT is operating outside legal authorization. The FDA-approved injectable versions, Mounjaro and Zepbound, remain widely available through legitimate prescribers.
Is tirzepatide ODT as effective as the injection?
There is no clinical trial data comparing tirzepatide ODT to the injectable form. The bioavailability of oral peptides is generally much lower than subcutaneous injection. Injectable tirzepatide achieves approximately 80 percent bioavailability, while oral mucosal absorption of large peptides is estimated at 5 to 15 percent without specialized absorption enhancers. The injectable form has the strongest evidence for effectiveness.
What is the difference between tirzepatide ODT and tirzepatide drops?
Tirzepatide ODT is a solid tablet that dissolves on the tongue, while tirzepatide drops are a liquid formulation placed under the tongue. Both rely on sublingual and buccal absorption. Drops may allow more precise dose adjustments, while ODT tablets offer greater convenience and room-temperature stability. Neither format has published bioavailability data for tirzepatide.
Can I switch from tirzepatide ODT to the injection?
Yes. Most healthcare providers recommend starting injectable tirzepatide at the standard starting dose of 2.5 mg weekly regardless of your previous ODT dose, since the bioavailability difference means direct dose conversion is not possible. You can begin injectable tirzepatide immediately after stopping the oral formulation.
Why was compounded tirzepatide banned?
The FDA determined that tirzepatide was no longer in shortage as of October 2024, meaning all doses of Mounjaro and Zepbound were readily available. Under federal law, compounding pharmacies can only compound copies of commercially available drugs during documented shortages. When the shortage ended, the legal authority for compounding tirzepatide ended with it. The enforcement deadline was March 19, 2025.
What are the side effects of tirzepatide ODT?
Side effects are similar to injectable tirzepatide and primarily affect the gastrointestinal system. Nausea (30-40%), vomiting (15-20%), diarrhea (12-20%), and constipation (6-10%) are the most common. ODT users may experience slightly higher nausea rates than injectable users due to direct oral contact with the medication during dissolution.
Is there a pill version of tirzepatide from Eli Lilly?
No FDA-approved oral tirzepatide exists from Eli Lilly or any other manufacturer. Eli Lilly is developing orforglipron, a small-molecule oral GLP-1 agonist, but this is a different compound, not oral tirzepatide. Phase 3 trials for orforglipron have shown promising results, but it is not yet approved.
How should I store tirzepatide ODT?
Tirzepatide ODT should be stored at room temperature (68-77 degrees F) in its original foil packaging. Protect from moisture and humidity. Do not remove tablets from packaging until ready to use. Do not store in pill organizers or open containers. Check the expiration date and discard any tablets that appear discolored, crumbled, or partially dissolved.
External resources
For researchers serious about understanding all available GLP-1 options and optimizing their protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed formulation comparisons, and a community of thousands who have navigated these exact decisions.
In case I do not see you, good afternoon, good evening, and good night. May your formulations stay stable, your absorption stay consistent, and your decisions stay informed.