Mar 18, 2026

Experienced researchers know something beginners do not about combining metabolic supplements with GLP-1 receptor agonists. The question is never just "can I take this?" The real question is "what happens at the biochemical level when I do, and how do I manage those interactions intelligently?" When people ask whether they can take berberine and tirzepatide together, most online resources give one of two answers: a flat "no" based on vague safety concerns, or a flat "yes" with no nuance whatsoever. Neither answer is honest.
The truth lives in the mechanism. Berberine and tirzepatide both reach into glucose metabolism, but they use different doors. They share downstream effects, which creates both opportunity and risk. No official contraindication exists. No regulatory body has prohibited this combination.
But no clinical trial has studied it directly either, which means the research community is working from first principles, pharmacological reasoning, and clinical observation rather than clean randomized data.
That gap is exactly where this guide lives.
What follows is a comprehensive look at how these two compounds interact at the molecular level, where their effects overlap, what the practical risks actually are, how to time and dose them intelligently, and which populations should avoid the combination entirely. If you are already on tirzepatide dosing protocols and considering adding berberine, or vice versa, this is the resource that will help you make that decision properly. You will also find guidance on monitoring, GI management, and the CYP450 interactions that most articles skip entirely. SeekPeptides covers this topic because researchers deserve more than surface-level answers when stacking metabolic compounds.
What berberine does in the body
Berberine is an isoquinoline alkaloid found in several plants including barberry, goldenseal, and Oregon grape. It has been used in traditional medicine for centuries. Modern research has revealed mechanisms that explain those traditional uses in precise molecular terms, and what researchers have found is genuinely remarkable for a compound with less than 5% oral bioavailability.
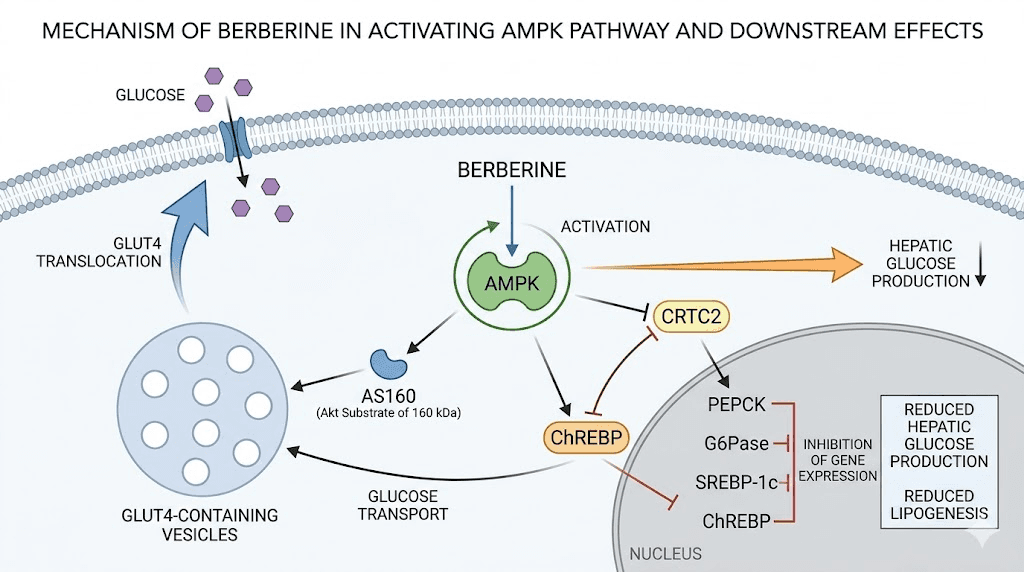
The primary mechanism is AMPK activation. Berberine phosphorylates AMP-activated protein kinase at the Thr172 residue, which triggers a cascade of metabolic effects throughout the body. AMPK acts as the master energy sensor in cells. When berberine activates it, cells behave as though they are in a low-energy state, even when they are not. This shifts metabolism toward fat oxidation, reduces hepatic glucose production, and improves insulin sensitivity in peripheral tissues. For anyone exploring peptides and compounds for fat loss, understanding AMPK activation is foundational. The best peptides for weight loss guide covers related compounds that work through overlapping metabolic pathways.
The GLUT4 translocation effect is equally important. Berberine increases the movement of GLUT4 glucose transporters to cell membranes, which allows cells to absorb glucose from the bloodstream more efficiently even in the absence of elevated insulin. This is one mechanism behind its blood sugar lowering effects, and it operates through a pathway largely independent of insulin receptor signaling. Research indicates berberine can reduce HbA1c by approximately 0.5 to 1.0% in people with type 2 diabetes, which is clinically meaningful and comparable to some pharmaceutical agents in mild-to-moderate cases.
Berberine also inhibits dipeptidyl peptidase-4 (DPP-4), which degrades endogenous GLP-1. Less DPP-4 activity means more active GLP-1 circulating in the system. On top of that, berberine stimulates endogenous GLP-1 secretion from L-cells in the intestinal lining through bitter taste receptor activation. This is a mechanism with enormous relevance to anyone already taking a GLP-1 receptor agonist, and it sets up the overlap discussion that matters most for this guide. Researchers interested in supplements to take with GLP-1 medications often encounter berberine near the top of those discussions precisely because of this shared mechanism.
Berberine and the gut microbiome
One underappreciated dimension of berberine is its effect on the gut microbiome. Research shows berberine shifts the gut bacterial community toward strains associated with metabolic health, including increased abundance of Akkermansia muciniphila. This particular species has been linked to improved gut barrier function, reduced metabolic endotoxemia, and better insulin sensitivity. Berberine also promotes short-chain fatty acid production by feeding beneficial bacterial populations, and SCFAs have their own positive effects on glucose metabolism and appetite regulation.
This microbiome effect may partly explain why berberine works better in some individuals than others. The baseline microbiome composition matters. People with more dysbiotic gut communities tend to see larger benefits. It also has implications for combination use with tirzepatide, since tirzepatide also appears to favorably influence gut microbiome composition through its effects on gastric emptying and intestinal transit time. The combination may produce additive microbiome benefits, though this has not been formally studied.
Berberine is also a modest inhibitor of several CYP450 enzymes, specifically CYP2D6, CYP2C9, and CYP3A4. This has implications for drug metabolism that deserve their own dedicated section later in this guide. The point here is that berberine is not a passive compound. It actively alters the enzymatic machinery that processes many medications, and researchers need to account for this when stacking it with other compounds.
How tirzepatide works (dual GIP/GLP-1 mechanism)
Tirzepatide is a dual agonist of two incretin hormone receptors: glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1). This dual mechanism is what separates it from earlier GLP-1 receptor agonists like semaglutide, and it is largely responsible for the compound superior efficacy data. The detailed comparison between semaglutide and tirzepatide illustrates exactly how this dual mechanism translates to better clinical outcomes. Understanding both arms of this mechanism matters when evaluating how berberine interacts with tirzepatide pharmacology.
The GLP-1 receptor arm is well understood at this point. GLP-1 receptors in the pancreas enhance glucose-dependent insulin secretion, meaning insulin release increases when blood glucose rises, but not when it is already normal or low. This glucose-dependency is central to the safety profile of GLP-1 agonists. GLP-1 receptor activation also suppresses glucagon secretion, slows gastric emptying, and acts on receptors in the hypothalamus to reduce appetite and food intake. Researchers familiar with the timeline for GLP-1 medications to start working and the GLP-1 injection technique guide will already understand the foundational pharmacology behind these receptor-mediated effects.
The GIP receptor arm is less intuitively obvious. GIP was historically considered the "incretin that failed" because GIP receptor agonism alone does not reduce blood sugar in people with type 2 diabetes. It actually appears to blunt GIP responsiveness in that population. But tirzepatide research revealed something surprising: when you activate both GIP and GLP-1 receptors simultaneously, synergistic effects emerge that exceed what either agonist produces alone. GIP receptor activation in adipose tissue appears to improve fat storage away from ectopic depots like the liver and muscle, which improves insulin sensitivity at those sites.
Tirzepatide clinical outcomes
The clinical data for tirzepatide is exceptional. In the SURPASS trial program, tirzepatide demonstrated HbA1c reductions of up to 2.5 percentage points and body weight reductions of 15 to 22% depending on dose and population studied. These are numbers that put tirzepatide ahead of every previously approved pharmaceutical for metabolic management. For researchers following tirzepatide weight loss timeline data, the trajectory is typically most pronounced between weeks 12 and 36 of continuous use. The tirzepatide speed of action guide and how long tirzepatide takes to work resource provide week-by-week expectations for researchers just starting the compound.
Tirzepatide is administered as a subcutaneous injection once weekly. The standard titration schedule begins at 2.5mg weekly, with dose escalation every four weeks up to a maximum of 15mg weekly. This slow titration is intentional and important. Gastrointestinal side effects are the primary reason people discontinue tirzepatide, and moving through doses too quickly dramatically increases that risk. Researchers using compounded tirzepatide starting doses follow the same general titration logic. Anyone exploring the full tirzepatide dose chart will find the gradual escalation structure clearly mapped. The first dose experience guide prepares researchers for what the initial weeks feel like before berberine is even in the picture.
Tirzepatide also demonstrates cardiovascular benefits including reductions in blood pressure, improvement in lipid profiles, and reductions in inflammatory markers. Its effects on metabolism go beyond simple weight loss. The compound improves hepatic steatosis, reduces visceral adiposity preferentially over subcutaneous fat, and improves markers of kidney function.
For researchers interested in the full range of tirzepatide weight loss results, understanding these systemic metabolic improvements contextualizes why the compound performs as well as it does. Tirzepatide also impacts testosterone levels and energy levels through its metabolic effects. These are systemic effects that berberine also touches through its own mechanisms, which creates both complementarity and the need for careful management.
Where berberine and tirzepatide overlap
The overlap is substantial. Both compounds lower blood glucose. Both improve insulin sensitivity. Both have favorable effects on lipid profiles. Both influence the GLP-1 axis, directly in tirzepatide case and indirectly through berberine stimulation of endogenous GLP-1 secretion. And both produce gastrointestinal side effects that worsen with rapid titration or inappropriate dosing. Researchers interested in the broader GLP-1 fat loss treatment landscape and the general question of what GLP-1 medications actually are will benefit from that foundational context. Understanding where these compounds meet is the prerequisite to understanding what happens when you combine them.
The GLP-1 axis overlap deserves the most attention. Tirzepatide provides exogenous GLP-1 receptor agonism at pharmacological concentrations. Berberine simultaneously increases endogenous GLP-1 secretion and reduces GLP-1 degradation by inhibiting DPP-4. The net effect when combining both is a state of prolonged and enhanced GLP-1 receptor activation. Whether this leads to receptor downregulation or saturation effects over time has not been studied, but the mechanistic logic suggests the combination produces greater GLP-1 axis stimulation than either compound alone. This is worth considering for researchers tracking tirzepatide not working anymore concerns, where receptor dynamics may play a role.
AMPK activation is another point of convergence. Tirzepatide influences AMPK indirectly through weight loss and improved metabolic status. Berberine directly phosphorylates AMPK at Thr172. The downstream effects of AMPK activation, including reduced hepatic glucose output and improved lipid metabolism, may be amplified when both compounds are present. This could be beneficial, representing additive metabolic improvement, or it could create situations where blood glucose drops further than intended, which circles back to the hypoglycemia risk that gets its own section below.
The shared GI vulnerability
Both compounds also share a vulnerability to gastrointestinal side effects. Tirzepatide slows gastric emptying significantly, particularly at higher doses. This is part of how it promotes satiety, but it also means food moves through the stomach and intestines more slowly than normal. Berberine irritates the intestinal lining at higher doses and produces diarrhea in 15 to 25% of users taking 1000 to 1500mg daily, according to clinical trial data. The diarrhea typically resolves by weeks three to four as the gut adapts, but the adaptation period is uncomfortable.
When you combine slowed gastric emptying from tirzepatide with berberine irritation of the intestinal lining, the resulting GI environment is more reactive than either compound creates alone. Gas, bloating, cramping, alternating constipation and diarrhea, and nausea can all worsen. This is the most common practical complaint from people combining these two compounds, and managing it requires specific strategies that this guide covers in a later section. Researchers already familiar with tirzepatide diarrhea duration will recognize that adding berberine can extend that adjustment period significantly. The GLP-1 headache guide and GLP-1 fatigue resource also cover symptoms that frequently accompany GI distress during the combination adjustment period.
Can you take berberine and tirzepatide at the same time
There is no official contraindication. No FDA drug interaction warning covers this combination. No major clinical guideline prohibits it. That is the accurate, honest starting point.
But "no contraindication" is not the same as "proven safe." It means the combination has not been formally studied in clinical trials and that regulators have not identified a prohibitory signal from available data. The absence of evidence is not evidence of absence. For researchers already taking supplements alongside tirzepatide, understanding this distinction matters enormously.
The practical answer is that many people do combine berberine and tirzepatide without serious adverse events. The combination is used in functional medicine settings where physicians supervise both compounds together. Diabetes management practices sometimes employ berberine as an adjunctive measure for patients on GLP-1 receptor agonists who need additional glycemic control. The overlap with the comparison compound is worth noting: researchers interested in berberine and semaglutide together will find very similar mechanistic logic applies to the tirzepatide combination, since semaglutide is a GLP-1 receptor agonist and tirzepatide adds GIP receptor agonism on top.
What the evidence actually says
No randomized controlled trial has examined berberine plus tirzepatide specifically. The closest analogs are studies combining berberine with metformin, berberine with other GLP-1 receptor agonists in animal models, and mechanistic studies in cell culture. From this adjacent literature, a picture emerges of additive glucose-lowering effects, additive GLP-1 axis stimulation, and additive GI burden.
A 2015 study in the journal Phytomedicine demonstrated that berberine combined with lifestyle intervention produced HbA1c reductions comparable to metformin monotherapy in newly diagnosed type 2 diabetes patients. This tells us berberine is pharmacologically meaningful, not a weak supplement. When you add something with genuine HbA1c-lowering power to tirzepatide, which is already the most potent approved agent for HbA1c reduction, the combined effect can push blood glucose lower than either party anticipated. Researchers considering this combination should also review the guide on metformin and tirzepatide together since the metabolic arithmetic is similar.
The bottom line: you can combine berberine and tirzepatide, but you need to do it with informed intentions, active blood sugar monitoring, appropriate dose sequencing, and a clear plan for managing GI side effects. This guide provides the framework for doing that intelligently.
Potential benefits of combining berberine with tirzepatide
When managed properly, the combination offers genuine potential advantages. Not every researcher will benefit from adding berberine to tirzepatide. But for certain profiles, the addition makes mechanistic sense and may produce outcomes that tirzepatide alone does not fully achieve.
The most obvious potential benefit is enhanced glycemic control. For researchers who are not reaching their target blood sugar or HbA1c goals on tirzepatide alone, berberine provides a second mechanistic pathway. AMPK activation is independent of GIP and GLP-1 receptor signaling. Adding a compound that works through a genuinely different mechanism creates genuine additive potential rather than redundant stimulation of the same pathway. Researchers tracking tirzepatide before and after results who plateau in glycemic improvement may find berberine helps move the numbers further. The general BMI requirements for GLP-1 medications resource also contextualizes where berberine addition may be most beneficial for researchers at different starting points.
Insulin sensitivity and metabolic flexibility
Both compounds improve insulin sensitivity, but through different mechanisms. Tirzepatide improves insulin sensitivity largely through weight loss, reduction of hepatic fat, and improvement of GIP receptor signaling in adipose tissue. Berberine improves insulin sensitivity directly through AMPK activation and GLUT4 upregulation at the cellular membrane level. These mechanisms are complementary rather than redundant. A person who is still insulin resistant despite tirzepatide use may respond well to the addition of berberine addressing cellular insulin signaling directly.
Berberine also reduces hepatic glucose production through AMPK-mediated inhibition of gluconeogenesis enzymes. Tirzepatide reduces hepatic glucose output through glucagon suppression. Both mechanisms reduce the liver contribution to fasting blood glucose, and combining them produces greater suppression of hepatic glucose output than either compound alone. For researchers whose fasting glucose remains elevated despite tirzepatide, this additive effect on hepatic glucose production is particularly relevant.
Microbiome synergy
The gut microbiome effects of both compounds may synergize in ways that support long-term metabolic health. Berberine directly increases Akkermansia muciniphila abundance and promotes short-chain fatty acid production. Tirzepatide alters gut microbiome composition indirectly through changes in gastric emptying, intestinal transit, and caloric intake. The combined modulation of gut bacteria toward a more metabolically favorable profile may support better gut barrier function, reduced systemic inflammation, and improved metabolic responses over time. Researchers interested in gut health alongside metabolic management should explore peptides for gut health as additional context.
Berberine also increases production of GLP-1 from intestinal L-cells. This mechanism may partially counteract any tachyphylaxis that develops to exogenous GLP-1 receptor agonism over time by maintaining endogenous secretion of the hormone. The clinical significance of this effect in humans on pharmacological doses of tirzepatide is theoretical, but the mechanistic logic is sound. It is also worth noting that researchers experiencing tirzepatide effectiveness decreasing over time sometimes find that supporting endogenous GLP-1 secretion through berberine and other means helps sustain the metabolic response.
Lipid and cardiovascular markers
Berberine has well-documented effects on lipid profiles, including reductions in total cholesterol, LDL cholesterol, and triglycerides, as well as modest increases in HDL. These effects are partially mediated through PCSK9 inhibition, since berberine reduces PCSK9 expression, which allows LDL receptors to be recycled more effectively rather than degraded. Tirzepatide also improves lipid profiles, particularly through weight loss and reduction of visceral adiposity. The combined lipid benefits may be additive, particularly for triglycerides, where both compounds work through complementary mechanisms. This cardiovascular benefit dimension adds to the benefits of tirzepatide beyond weight loss that researchers often underestimate.
Risks and side effects when taking both
The risks are real. Presenting the benefits without equal attention to the risks would be dishonest, and the risks require specific knowledge to manage properly.
Hypoglycemia is the primary concern. Both compounds lower blood glucose through multiple mechanisms. When combined, the glucose-lowering effects are additive. Researchers who are not using insulin and who have normal glucose regulation do not face severe hypoglycemia risk from this combination, because both compounds are largely glucose-dependent in their insulin-stimulating effects. But researchers who are also taking insulin, sulfonylureas, or other glucose-lowering agents face meaningful hypoglycemia risk. This warrants close attention and gets a dedicated section below.
Gastrointestinal side effects are additive and sometimes severe. This is the most commonly reported practical problem. Tirzepatide alone causes nausea in 20 to 40% of users during titration. Diarrhea affects 15 to 25% at the 2.5 to 5mg range. Berberine alone causes diarrhea in 15 to 25% of users at 1000 to 1500mg daily and gastrointestinal discomfort in up to 35% in clinical trials. When combined, nausea, diarrhea, abdominal cramping, and bloating are significantly more likely. The guide on tirzepatide bloating and the section on tirzepatide constipation treatment both address symptoms that can worsen when berberine is added to the protocol. Other GI symptoms covered in our sulfur burps on GLP-1 guide and the acid reflux resource may also appear during the adjustment period.
Drug interaction through CYP450 enzymes
Berberine inhibits CYP2D6, CYP2C9, and CYP3A4. These enzymes are responsible for metabolizing a large percentage of pharmaceuticals. Tirzepatide itself is not primarily metabolized by these enzymes, which means direct pharmacokinetic interaction between berberine and tirzepatide is not the main concern. The concern is for other medications a researcher might be taking that ARE metabolized by these enzymes. Blood pressure medications, antidepressants, anticoagulants, and cholesterol medications are commonly processed through CYP2D6 or CYP3A4. Adding berberine to any regimen containing such medications can raise their plasma concentrations and produce unexpected side effects.
This is covered in greater depth in the CYP450 enzyme section. The point here is that the interaction profile of berberine extends beyond its direct effects on blood sugar. It alters the enzymatic environment through which many other drugs pass, and researchers combining it with tirzepatide while also taking additional medications need to review their complete medication list for CYP450 interactions.
Blood pressure considerations
Both berberine and tirzepatide modestly lower blood pressure. Berberine appears to have antihypertensive effects through several mechanisms including activation of endothelial nitric oxide production and modest vasodilation. Tirzepatide lowers blood pressure through weight loss and reduced sympathetic nervous system activation. In researchers with normal or low-normal blood pressure, the additive blood pressure lowering effect of combining both compounds may produce orthostatic hypotension or lightheadedness, particularly when standing quickly. Researchers already monitoring tirzepatide fatigue should be aware that low blood pressure can contribute to that symptom profile, alongside other common side effects like dizziness, feeling cold, and body aches that may be exacerbated by combined hemodynamic changes.
The hypoglycemia question
This section deserves serious attention because hypoglycemia is the most consequential risk from this combination, and it is also the most misunderstood.
First, the reassuring part. Neither berberine alone nor tirzepatide alone has a strong propensity to cause hypoglycemia in people with normal pancreatic function who are not also taking insulin or secretagogues. The insulin-stimulating effects of tirzepatide are glucose-dependent, meaning they are robust when blood glucose is high but muted when blood glucose is normal or low. Berberine works primarily through insulin sensitization and reduced hepatic glucose output, not through forcing insulin secretion at inappropriate glucose levels. This glucose-dependent safety profile is a major advantage over older glucose-lowering agents like sulfonylureas.
The risk is real, but it is context-dependent.
When hypoglycemia risk is elevated
Risk becomes significant in several specific situations. If a researcher is also taking insulin, the additive glucose lowering from berberine plus tirzepatide may require insulin dose reduction to avoid hypoglycemia. If taking sulfonylureas, which force insulin secretion regardless of blood glucose level, the combination with berberine and tirzepatide creates genuine hypoglycemia risk. If the researcher is in a significant caloric deficit, fasting, or following a very low carbohydrate diet, the baseline glucose substrate available is lower, and the combined glucose-lowering effects of both compounds can push levels into symptomatic ranges. Researchers on aggressive weight loss protocols who are also combining these compounds should be particularly vigilant. The tirzepatide diet plan and foods to avoid on GLP-1 medications help ensure adequate caloric and carbohydrate intake that prevents unnecessary hypoglycemia risk during combined use.
The guide on phentermine and tirzepatide together covers a somewhat parallel discussion about combinations that affect appetite and metabolic rate simultaneously, and similar monitoring logic applies. Researchers using multiple metabolic compounds together need a systematic approach to blood sugar monitoring rather than assumption-based management.
Symptoms of hypoglycemia to know
Hypoglycemia symptoms typically begin when blood glucose drops below 70 mg/dL. Early symptoms include shakiness, sweating, heart palpitations, anxiety, hunger, and difficulty concentrating. These are often described as feeling "off" or having unusual hunger despite being on appetite-suppressing tirzepatide. As glucose drops further, toward 50 to 60 mg/dL, symptoms escalate to confusion, slurred speech, and visual disturbances. Severe hypoglycemia below 40 mg/dL can produce loss of consciousness. Researchers experiencing any of the early symptoms should check blood glucose immediately and consume fast-acting carbohydrates if levels confirm hypoglycemia.
The concern about hypoglycemia is more acute during the first several weeks of combining both compounds, before the body equilibrates to the combined glucose-lowering load. It is also higher after periods of extended fasting, intense exercise, or illness. The risk can be effectively managed through glucose monitoring and appropriate dose adjustment strategies outlined in the monitoring section below. Researchers experiencing anxiety symptoms, insomnia, or headaches should consider whether unrecognized mild hypoglycemia may be contributing to these symptoms. Anyone tracking maintaining weight loss after tirzepatide through lifestyle changes that involve fasting or low carbohydrate approaches should be especially attentive during transition periods.
Berberine timing and the glucose dip pattern
Many berberine users notice a predictable blood glucose dip pattern in the two to three hours following a berberine dose with meals. This post-berberine glucose nadir is typically modest in people with normal baseline glucose levels, dropping perhaps 10 to 20 mg/dL below post-meal peak, which is entirely normal and beneficial. In people on tirzepatide, where post-meal glucose responses are already blunted by slowed gastric emptying and enhanced insulin secretion, that same berberine-induced dip may bring glucose levels into the symptomatic hypoglycemia range more readily. This is why timing matters, and why the dosing strategies section below emphasizes specific timing windows for berberine relative to tirzepatide injections.
Timing and dosing strategies for taking both together
Approach this combination with a sequential introduction strategy rather than starting both compounds simultaneously. Beginning both at the same time makes it impossible to attribute side effects or unexpected glucose responses to the correct compound. Start tirzepatide first and allow several weeks at a stable dose before introducing berberine. This mirrors the broader principle that any metabolic compound stack should be built one layer at a time.
The standard approach for researchers on this combination is to establish a stable tirzepatide dose, at minimum reaching the 5mg weekly dose and tolerating it well, before adding berberine. Researchers following tirzepatide dosing protocols typically reach that 5mg level after eight weeks of standard titration. During those first eight weeks, using the compounded tirzepatide dosage calculator helps confirm accurate dosing before adding another variable to the equation.
Berberine introduction protocol
When introducing berberine to an established tirzepatide protocol, start low. Begin with 500mg once daily with the largest meal of the day. Hold at 500mg for two weeks while monitoring blood glucose responses. If tolerating well with no GI issues and no hypoglycemia signals, increase to 500mg twice daily with two meals. Hold for another two weeks. If continuing to tolerate well and glucose targets warrant additional support, move to the full standard dose of 500mg three times daily with meals.
The full berberine dosage range used in research protocols is 900 to 1500mg daily, divided into two or three doses taken with meals. The "with meals" instruction is important for two reasons. First, taking berberine with food reduces GI irritation significantly. Second, the glucose-lowering effect of berberine is most useful post-meal, when blood glucose is rising, which is also when the risk of hypoglycemia from berberine is lowest. Timing berberine with meals essentially aligns its mechanism with appropriate glucose physiology. Researchers using the tirzepatide meal plan approach can build berberine dosing directly into their meal structure.
Timing relative to tirzepatide injection
Tirzepatide is dosed once weekly, and its peak plasma concentration occurs approximately 24 to 72 hours after injection. The glucose-lowering effect is therefore distributed across the entire week rather than concentrated at injection time. This means there is no single "dangerous window" around the injection itself in terms of glucose interactions with berberine. However, researchers should be aware that gastric emptying is most significantly slowed in the 24 to 48 hours following tirzepatide injection, which is when berberine absorption may be most variable.
Space berberine doses from the actual tirzepatide injection day by a few hours if possible. This is not a hard rule but a precautionary measure during the initial combination period. After several weeks, most researchers find that the timing becomes less sensitive as the body adapts to both compounds. The guide on best time to take the tirzepatide shot covers injection timing optimization that can be coordinated with berberine dosing schedules. Related timing resources on best time of day to inject tirzepatide and best day to take tirzepatide help researchers optimize their weekly schedule for both compounds.
Dose considerations for tirzepatide when adding berberine
Some researchers find they achieve equivalent glycemic control on a lower tirzepatide dose once berberine is established in the protocol. This is a legitimate consideration, particularly for researchers experiencing dose-dependent side effects like nausea at higher tirzepatide levels. The guide on microdosing tirzepatide and the broader resource on how to microdose tirzepatide are relevant here because berberine addition can shift the optimal tirzepatide dose point downward. Researchers tracking tirzepatide dosage in units should update their glucose monitoring logs when adding berberine to see if dose adjustment becomes appropriate.
Blood sugar monitoring when combining berberine and tirzepatide
Monitoring is not optional when combining two blood-glucose-lowering compounds. It is the mechanism that keeps this combination safe and effective. Without monitoring, you are flying blind.
The minimum monitoring standard for the initial eight weeks of combining both compounds is fasting blood glucose each morning and a postprandial measurement two hours after the largest meal of the day. This gives you two data points per day that capture the most critical glucose windows: fasting baseline and the post-meal response. A standard glucometer with test strips is sufficient for this purpose. Continuous glucose monitors provide even richer data if accessible, showing the full glucose curve throughout the day rather than single-point snapshots.
Target ranges when combining both
Fasting glucose target while on this combination should generally be 80 to 110 mg/dL for most researchers. Below 70 mg/dL warrants attention and potential dose adjustment. Below 60 mg/dL is symptomatic hypoglycemia requiring immediate carbohydrate correction and medication review. Postprandial glucose at two hours should ideally stay below 140 mg/dL.
These targets are general guidelines. Individual metabolic status, other medications, diet composition, and activity level all affect what appropriate glucose targets look like for a specific researcher. For people managing type 2 diabetes with these compounds under medical supervision, individual targets may differ based on clinical history and complication risk. Researchers who also take insulin alongside tirzepatide and berberine, a situation explored in the guide on NAD and tirzepatide together in the context of stacking metabolic compounds, need individualized targets from their prescribing physician.
Bedtime monitoring and the overnight fast
Bedtime monitoring deserves specific attention. The overnight fasting period is the longest glucose-unstimulated period of the day, and it is when berberine doses from earlier meals are waning while the next morning dose has not yet been taken. For most researchers, this transition period is safe. But in the first few weeks of combined use, checking blood glucose at bedtime, around 10pm if waking at 6am, helps confirm that glucose is not dropping to problematic levels during sleep. A bedtime reading below 100 mg/dL in combination with berberine and tirzepatide is worth noting and potentially discussing with a physician. The guide on taking tirzepatide at night has relevant context on how injection timing affects overnight glucose patterns.
Keep a simple glucose log for at least the first eight weeks of combined use. Log fasting, postprandial, and bedtime readings alongside notes on meal composition, exercise, and any symptoms. This log will reveal patterns, such as consistent post-lunch dips or elevated morning glucose on certain days, that help you optimize both timing and dosing. If HbA1c monitoring is available through regular bloodwork, tracking HbA1c quarterly provides a long-term picture of combined glycemic control that fasting glucose snapshots cannot capture alone.
CYP450 enzyme interactions and medication safety
This is the section most online resources skip entirely, and it may be the most important for researchers on multiple medications.
Berberine is a known inhibitor of CYP2D6, CYP2C9, and CYP3A4. These three enzymes collectively handle the biotransformation of a substantial proportion of all pharmaceuticals. When berberine inhibits these enzymes, drugs that rely on them for clearance accumulate to higher plasma concentrations than intended. Higher plasma concentrations mean stronger effects, including stronger side effects, and in some cases toxicity.
Medications affected by CYP2D6 inhibition
CYP2D6 metabolizes many antidepressants, including fluoxetine, paroxetine, and many tricyclics.
It also processes several antipsychotics, opioid analgesics including codeine and tramadol, and beta-blockers like metoprolol and carvedilol. If a researcher on tirzepatide is also taking metoprolol for blood pressure and adds berberine, the metoprolol plasma concentration may rise substantially, potentially causing bradycardia or excessive blood pressure lowering. This is a real interaction that has been observed clinically.
Tramadol conversion to its active metabolite also requires CYP2D6. Berberine inhibition of this enzyme could reduce tramadol analgesic effectiveness. Codeine-to-morphine conversion is similarly CYP2D6-dependent. These are not theoretical concerns. They are documented pharmacokinetic interactions that anyone combining berberine with CYP2D6-substrate medications needs to know about. Researchers also taking compounds like tirzepatide with B12, tirzepatide with glycine, semaglutide with L-carnitine, or other metabolic cofactors should review their complete supplement and medication list for CYP450 considerations. Even combinations like semaglutide with glycine and semaglutide with B12 that seem straightforward deserve review in the context of berberine CYP450 inhibition.
Medications affected by CYP3A4 inhibition
CYP3A4 is perhaps the most prolific drug-metabolizing enzyme in the human body, handling roughly 30 to 40% of all pharmaceuticals. Berberine moderate inhibition of CYP3A4 is therefore significant. Relevant CYP3A4 substrates include statins like atorvastatin and simvastatin, calcium channel blockers, certain anticoagulants including apixaban and rivaroxaban, immunosuppressants like cyclosporine, and a wide range of other compounds. Someone on tirzepatide for metabolic management who is also on atorvastatin for lipids and adds berberine may experience elevated statin plasma levels, increasing the risk of myopathy or hepatotoxicity.
CYP2C9 inhibition by berberine affects warfarin metabolism particularly importantly. Warfarin is a narrow therapeutic index drug where small changes in metabolism can shift the anticoagulant effect into dangerous territory, either toward insufficient anticoagulation or toward bleeding risk. Anyone on warfarin should not add berberine without INR monitoring and physician guidance. For researchers exploring the semaglutide and liver enzyme data, the hepatic metabolic considerations of berberine are similarly relevant, since the liver is where most CYP450 activity occurs.
Tirzepatide and CYP450 interactions
Tirzepatide itself has minimal CYP450 interaction. It is primarily cleared through receptor-mediated endocytosis and proteolytic degradation, not through hepatic CYP450 metabolism. This means berberine CYP450 inhibition does not significantly alter tirzepatide pharmacokinetics directly. The concern is entirely about the broader medication context in which the combination sits. If a researcher is on tirzepatide plus berberine plus other medications, the berberine CYP450 effects on those other medications need assessment. This is a task best performed with a pharmacist or physician review of the complete medication list.
Managing GI side effects when taking both
Nausea. Diarrhea. Bloating. These are the three words that most people combining berberine and tirzepatide need to plan for, especially during the first four to six weeks.
The GI burden is real and additive. Berberine GI side effects in clinical trials affect 10 to 35% of users, with diarrhea reported specifically in 15 to 25% of those taking 1000 to 1500mg daily. These effects typically resolve by weeks three to four as the gut microbiome adapts and the intestinal epithelium adjusts to the compound. Tirzepatide GI side effects are most pronounced during titration, when gastric emptying slows significantly, and typically improve as the dose stabilizes. When both compounds are introduced in the same period, the combined GI burden can be substantial enough to cause people to abandon the protocol unnecessarily.
The sequential introduction advantage
Starting tirzepatide first and tolerating it well before introducing berberine staggers the GI adaptation periods. By the time berberine is introduced, tirzepatide GI side effects have largely resolved. The new GI burden from berberine is then easier to attribute and manage because it is the only new variable. This sequential approach is the single most important GI management strategy for this combination. Researchers who ignored this advice and started both simultaneously consistently report more difficult GI experiences than those who staggered introduction.
Researchers familiar with the full profile of semaglutide vs tirzepatide side effects comparison know that tirzepatide GI effects are manageable with proper titration. The same principle applies to berberine. Gradual dose escalation, as described in the timing section above, gives the gut time to adapt to berberine before reaching therapeutic doses.
Practical strategies for GI comfort
Take berberine with food and water, never on an empty stomach. The food buffer significantly reduces direct intestinal irritation. Divide the total daily berberine dose across three meals rather than two if GI symptoms are problematic, since smaller individual doses are better tolerated than larger ones. Berberine extended-release formulations, when available, spread absorption over a longer period and tend to produce fewer acute GI symptoms than standard-release capsules.
Probiotic supplementation during the GI adaptation period has supporting evidence from berberine research. A high-quality probiotic containing Lactobacillus and Bifidobacterium strains taken at a different time of day from berberine may help maintain gut microbiome balance during the transition. Researchers using probiotics alongside GLP-1 medications will find the same reasoning applies here, and the specialized formulations like Pendulum GLP-1 Probiotic Pro were specifically designed for this context. The gut response GLP-1 support guide covers additional strategies for managing GI adaptation on GLP-1 medications.
Dietary modification during the initial weeks helps substantially. Following a tirzepatide-appropriate diet that minimizes high-fat, greasy foods and excessive fiber loading gives the gut less total irritant load to manage. The resource on tirzepatide foods to avoid applies directly during combined use with berberine, since the same food choices that worsen tirzepatide GI symptoms tend to amplify berberine GI symptoms as well. The tirzepatide dietary guidance and the tirzepatide diet plan framework provide practical eating structures that complement berberine use.
Staying well hydrated matters more during berberine use because berberine can increase intestinal motility and transit speed, which reduces water reabsorption in the colon. Dehydration worsens both nausea and the fatigue that sometimes accompanies GI distress on tirzepatide. The symptom discussion in tirzepatide dry mouth is relevant here since both dry mouth and dehydration risk increase when GI side effects impair normal eating and drinking patterns.
Who should avoid combining berberine with tirzepatide
Not everyone should combine these compounds. There are specific populations where the risks outweigh potential benefits, and this section names them clearly.
Pregnant or breastfeeding researchers should not use berberine. Berberine has known potential for uterotonic effects and can cross the placenta, with evidence of fetal harm in some animal studies. This is not a gray area. The resource on tirzepatide while breastfeeding addresses the broader question of GLP-1 medications during this period, and berberine adds an additional layer of concern that makes the combination inappropriate entirely. The guide on GLP-1 use while breastfeeding covers the foundational question before berberine even enters the picture. Researchers who have experienced pregnancy on tirzepatide should discontinue both tirzepatide and berberine immediately upon discovering pregnancy and consult their physician. The effects of tirzepatide on menstrual cycles may also be relevant for those tracking reproductive health alongside metabolic management.
Researchers on warfarin or other narrow therapeutic index anticoagulants should avoid berberine unless under close medical supervision with regular INR monitoring. The CYP2C9 inhibition discussed above makes this combination genuinely risky. Researchers concerned about broader safety monitoring can review the blood work considerations for GLP-1 medications to understand what routine labs to request from their physician.
Other populations requiring caution or avoidance
Researchers already experiencing significant hypoglycemia episodes on tirzepatide alone should not add berberine without physician guidance and a clear monitoring plan. Adding a second glucose-lowering compound to a person already struggling with hypoglycemia is not a sensible approach.
People with significant liver disease should use caution. Berberine is hepatically metabolized, and impaired liver function alters berberine clearance and potentially its inhibition of CYP450 enzymes. Combined with tirzepatide, which also has hepatic metabolic effects, the interaction profile in the setting of liver disease is poorly characterized. The liver enzyme considerations for GLP-1 users provide important baseline knowledge for anyone managing hepatic health alongside these metabolic compounds. This is a situation requiring specialist input rather than self-directed combination.
Researchers on multiple CYP450-dependent medications, particularly those with narrow therapeutic windows, should review their complete medication list with a pharmacist or physician before adding berberine. This is not about whether berberine and tirzepatide interact directly with each other; it is about berberine interaction with the broader medication ecosystem. Someone on tirzepatide plus a statin plus an antidepressant, for example, faces a more complex interaction picture than someone on tirzepatide alone.
Researchers who have experienced severe berberine GI intolerance in the past should be cautious about adding it to tirzepatide, since the GI burden will be greater than berberine alone. The approximately 10% of people who develop persistent, significant GI symptoms on berberine even with appropriate dosing may find the combination genuinely intolerable. In such cases, exploring a tirzepatide alternative or reviewing alternatives to tirzepatide that pair more comfortably with berberine may be worth considering.
Berberine dosage adjustments while on tirzepatide
Standard berberine research dosing is 900 to 1500mg daily divided across two to three doses with meals. This is appropriate for most adults in most contexts. When adding berberine to an established tirzepatide protocol, the starting approach is more conservative: 500mg once daily with the largest meal, escalating as described in the timing section. Researchers using the peptide calculator for their broader protocols and tracking dosing with the tirzepatide dosage calculator should add berberine tracking to their monitoring logs.
The key principle for dose adjustment is to titrate berberine based on glucose response rather than following a rigid predetermined schedule. If blood glucose monitoring shows that fasting glucose is already in the low-normal range on tirzepatide, there is no need to push berberine to the maximum 1500mg daily. A lower dose that maintains glucose in target range while minimizing GI side effects is the optimal dose, not the maximum dose. SeekPeptides members working through personalized metabolic protocols consistently find that less-is-more applies when combining multiple glucose-modulating approaches.
Adjusting berberine based on tirzepatide dose escalation
As tirzepatide dose escalates, its glycemic impact increases. This means that a berberine dose that was appropriate at tirzepatide 2.5mg may produce excessive glucose lowering once tirzepatide reaches 10 or 15mg. Researchers should re-evaluate their berberine dose with each tirzepatide dose escalation, using glucose monitoring data to determine whether the berberine dose needs to be reduced as tirzepatide increases. This is particularly important during the jump from 5mg to 7.5mg tirzepatide, which many researchers find produces a step-change increase in glycemic effect. The detailed breakdown of tirzepatide dose effects in the tirzepatide dosage units guide helps researchers understand what metabolic shifts to anticipate at each dose level.
For researchers who reach their tirzepatide maintenance dose and want to maximize berberine's complementary effects, the standard 500mg three times daily with meals tends to be the optimal balance of efficacy and tolerability. Extended-release berberine at 1000mg twice daily is an alternative that provides similar total daily exposure with fewer dosing events and often better GI tolerability. Both approaches are used by researchers seeking sustained AMPK activation throughout the day rather than pulsatile peaks around individual doses. Some researchers using split tirzepatide dosing strategies find it easier to coordinate berberine timing with their adjusted injection schedule, since split dosing spreads the GLP-1 agonist effect more evenly across the week.
When to reduce or pause berberine
Reduce berberine dose when fasting glucose consistently falls below 75 mg/dL. Pause berberine entirely if experiencing confirmed hypoglycemia episodes, defined as blood glucose below 70 mg/dL with symptoms. Also pause berberine when starting a new CYP450-sensitive medication until drug interactions can be assessed. During periods of illness when food intake is reduced, the glucose-lowering effects of berberine become more unpredictable and the dose may need temporary reduction. Researchers following protocols for weaning off tirzepatide who are planning to maintain berberine through the transition should be aware that blood glucose may rise as tirzepatide is reduced, potentially requiring upward berberine dose adjustment to maintain metabolic targets.
What about berberine and semaglutide instead
Many researchers face a choice between tirzepatide and semaglutide as their GLP-1 foundation, with berberine as a potential addition to either. The mechanistic logic for combining berberine with semaglutide is very similar to the tirzepatide case, since semaglutide is a GLP-1 receptor agonist and the GLP-1 axis overlap with berberine applies to both. The comprehensive guide to berberine and semaglutide together covers that specific combination in equivalent depth.
The key difference between combining berberine with semaglutide versus tirzepatide comes from tirzepatide additional GIP receptor agonism. Semaglutide is purely a GLP-1 agonist. Tirzepatide activates both GIP and GLP-1 receptors, which produces greater weight loss, greater HbA1c reduction, and potentially greater effects on adipose tissue metabolism. For researchers who have compared outcomes on both compounds, the semaglutide vs tirzepatide guide and the comparison landing page provide the relevant head-to-head data.
Does tirzepatide or semaglutide combine better with berberine
From a pure mechanism perspective, berberine and tirzepatide is a more complementary combination than berberine and semaglutide, simply because tirzepatide dual mechanism means berberine AMPK-mediated glucose lowering adds to pathways that neither GIP nor GLP-1 receptor agonism directly covers. With semaglutide, the incretin side of glucose management is handled by GLP-1 receptor activation alone, and berberine augments endogenous GLP-1 on top of that. The additive effect is still present, but the mechanistic diversity is somewhat narrower.
That said, from a GI tolerability perspective, some researchers find the semaglutide plus berberine combination easier to manage than tirzepatide plus berberine, because semaglutide GI side effect profile tends to be slightly milder at equivalent glycemic efficacy doses. Researchers comparing semaglutide vs tirzepatide vs retatrutide in the context of metabolic compound stacking will find that tolerability considerations often drive compound selection as much as efficacy data does.
Regardless of whether berberine is paired with tirzepatide or semaglutide, the same core principles apply. Start the GLP-1 agonist first. Allow dose stabilization. Introduce berberine gradually. Monitor glucose actively. Manage GI side effects proactively. Review CYP450 interactions with the full medication list. These principles do not change based on which GLP-1 agonist is in the protocol. For researchers who have switched from one to the other, the guides on switching from tirzepatide to semaglutide and the semaglutide to tirzepatide conversion chart are useful references for the transition period.
For researchers who want the most comprehensive resource on all aspects of metabolic compound protocols, SeekPeptides membership provides access to detailed interaction guides, protocol builders, and a community of researchers actively managing these combinations. The depth of personalized guidance available through membership significantly exceeds what any single guide can provide, particularly for complex stacking decisions that depend on individual health context.
Tirzepatide formulation types and berberine interaction considerations
Tirzepatide is available in multiple formulations, and the formulation type matters when combining it with berberine. The standard injectable form, administered subcutaneously once weekly, has the most predictable absorption profile and the most consistent interaction dynamics with oral berberine. Researchers using the standard injection method or following the stomach injection technique should not worry about direct absorption competition between tirzepatide and berberine, since the routes are entirely separate (subcutaneous vs oral).
Oral tirzepatide changes this picture. When tirzepatide is taken orally, it passes through the same gastrointestinal tract as berberine. The potential for absorption interference increases. Berberine effects on intestinal motility, gut pH, and intestinal enzyme activity could theoretically influence oral tirzepatide bioavailability, though this specific interaction has not been studied. Researchers using tirzepatide drops or tirzepatide orally disintegrating tablets should space berberine doses at least two hours from oral tirzepatide to minimize any potential absorption competition.
Compounded tirzepatide blends add another layer to consider. Many compounded formulations include additional active ingredients like B12, glycine, niacinamide, or levocarnitine. These combination products have their own metabolic effects that stack on top of berberine when both are used. The guides on tirzepatide glycine B12 compounds, tirzepatide with B6, and tirzepatide with methylcobalamin cover these individual blend ingredients. When adding berberine on top of a multi-ingredient tirzepatide compound, the total metabolic load is higher, and conservative berberine dosing becomes even more important.
Long-term considerations for the berberine and tirzepatide combination
Short-term safety is one thing. What about taking both compounds for months or years? The long-term data simply does not exist for this specific combination. But we can extrapolate from what we know about each compound individually over extended use.
Berberine has been studied in trials lasting up to two years with acceptable safety profiles. The primary long-term considerations are GI tolerability (which tends to improve over time), liver function monitoring (berberine can occasionally elevate liver enzymes, particularly at higher doses), and the persistent CYP450 inhibition that requires ongoing awareness of medication interactions whenever the medication list changes. Researchers who add a new prescription medication after months of stable berberine use still need to evaluate that new medication for CYP450 interactions with berberine.
Tirzepatide long-term data extends through the multi-year SURPASS trial extensions and post-marketing surveillance. The compound carries a boxed warning for thyroid C-cell tumors based on animal data, which is a class-level concern shared with all GLP-1 receptor agonists. Long-term tirzepatide use requires monitoring for pancreatitis, gallbladder disease, and thyroid nodule changes. These monitoring requirements do not change when berberine is added, but researchers should be aware that both compounds place metabolic demands on the liver, and periodic liver function testing is prudent during long-term combined use.
Weight maintenance and the combination role
One of the most interesting long-term questions is whether berberine can support metabolic health during tirzepatide discontinuation or dose reduction. Many researchers eventually reduce or stop tirzepatide after achieving weight and metabolic targets. The challenge is maintaining weight loss after tirzepatide, which requires ongoing metabolic support. Berberine AMPK activation, insulin sensitization, and lipid-modulating effects may provide a degree of metabolic stability during the transition off tirzepatide that helps prevent full metabolic rebound.
This is speculative but mechanistically plausible. Researchers who are weaning off tirzepatide might find that established berberine use provides a metabolic "floor" that prevents the worst of post-tirzepatide metabolic regression. The insulin sensitization and hepatic glucose output reduction from berberine are independent of GLP-1 receptor agonism, which means they persist after tirzepatide is removed. Research in this area is needed, but the mechanistic rationale is sound.
When results plateau on the combination
Some researchers experience metabolic plateaus even on the combination. Blood sugar stabilizes at a higher point than desired, or weight loss stalls despite ongoing tirzepatide and berberine use. This is not uncommon and does not mean the combination has "stopped working." It often means that the metabolic variables have shifted and the protocol needs reassessment. The comprehensive guide to why weight loss stalls on tirzepatide covers the common causes, many of which apply regardless of whether berberine is in the protocol. Similarly, researchers experiencing persistent hunger on tirzepatide or unexpected weight gain should investigate those root causes before assuming berberine is insufficient.
Diet quality matters enormously for long-term results on this combination. Berberine works best when paired with a diet that does not overwhelm its glucose-modulating capacity. The food recommendations for GLP-1 medications, the GLP-1 breakfast ideas, and the GLP-1 dinner ideas provide practical eating frameworks that complement berberine use. Alcohol consumption also deserves attention. Both berberine and tirzepatide affect liver metabolism, and alcohol adds a third hepatic burden. The guide on drinking on tirzepatide and best alcohol choices on tirzepatide apply when berberine is also in the picture, with additional caution warranted since berberine CYP450 inhibition affects alcohol metabolism pathways as well.
Other supplement combinations researchers ask about
Berberine is rarely the only supplement in a researcher metabolic stack. Understanding how it fits alongside other commonly paired compounds with tirzepatide helps researchers build coherent protocols rather than haphazard combinations.
Metformin with tirzepatide is one of the most common pharmaceutical combinations. Adding berberine on top of metformin and tirzepatide creates triple AMPK activation (berberine and metformin both activate AMPK) plus dual incretin pathway stimulation. This requires closer glucose monitoring than berberine plus tirzepatide alone, since the additive AMPK effects may produce more pronounced glucose lowering than any two-compound combination.
Phentermine alongside tirzepatide is used by some researchers for additional appetite suppression and energy. Adding berberine to this combination does not introduce significant new interactions beyond what has been discussed, but the cumulative metabolic modulation becomes complex enough to warrant professional supervision. The comparison resources on phentermine versus tirzepatide and phentermine versus GLP-1 medications help researchers understand where each compound contributes to the overall protocol.
NAD+ precursors are another popular addition to tirzepatide protocols. The guide on NAD and tirzepatide together covers that combination directly. Berberine has minor interactions with NAD+ metabolism through its effects on mitochondrial function, but these are not clinically significant at standard doses. The combination of NAD+ precursors, berberine, and tirzepatide together has not been studied, and researchers running this triple stack should monitor liver function periodically given the combined hepatic metabolic demands.
Probiotics deserve specific mention because they interact positively with berberine gut microbiome effects. The best probiotics for GLP-1 users and the specialized Pendulum GLP-1 probiotic are commonly combined with both tirzepatide and berberine. Probiotics do not create safety concerns in this context and may improve GI tolerability during the initial berberine adaptation period.
For researchers managing specific health conditions alongside their metabolic protocols, conditions like Hashimoto thyroiditis, lipedema, or endometriosis bring their own metabolic dimensions that interact with both tirzepatide and berberine in condition-specific ways. These are conversations best had with physicians who understand both the condition and the metabolic compound stack.
Frequently asked questions
Is it safe to take berberine and tirzepatide together?
No official contraindication exists for combining berberine and tirzepatide. No regulatory body has prohibited the combination. However, no dedicated clinical trial has studied it either, and both compounds lower blood glucose through multiple mechanisms, creating additive hypoglycemia risk in certain populations. The combination is used in supervised functional medicine settings. With appropriate sequencing, glucose monitoring, and dose management, many researchers use both compounds without serious adverse events. The risk profile depends heavily on what other medications are present, the individual glucose baseline, and how carefully the introduction protocol is followed. Reading the supplements to take with tirzepatide guide provides broader context on managing multiple metabolic compounds together.
Can berberine replace metformin when on tirzepatide?
Berberine shares some mechanistic overlap with metformin, including AMPK activation and reduced hepatic glucose production. Several studies have shown comparable HbA1c reduction between berberine and metformin in mild-to-moderate type 2 diabetes. However, berberine is not approved as a pharmaceutical drug in most countries and lacks the decades of safety and efficacy data that metformin carries. The guide on metformin and tirzepatide together covers the metformin-tirzepatide combination specifically. Whether berberine can substitute for metformin in any individual case is a clinical decision requiring physician input, not a one-size-fits-all answer.
How long does it take for berberine to start lowering blood sugar on tirzepatide?
Berberine effects on blood glucose typically become apparent within one to two weeks of consistent use at therapeutic doses. The AMPK activation begins immediately upon dosing, but the downstream effects on insulin sensitivity, hepatic glucose production, and GLUT4 expression develop over days to weeks. In the context of tirzepatide, where glucose is already being managed, the incremental berberine effect may be more subtle and best detected through fasting glucose trends and HbA1c changes over eight to twelve weeks rather than day-to-day glucose readings. The tirzepatide response timeline guide contextualizes what timeframes to expect for metabolic changes from this class of compounds.
Should I adjust my tirzepatide dose when adding berberine?
Tirzepatide dose adjustment when adding berberine should be based on glucose monitoring data rather than a predetermined protocol. Many researchers do not need to adjust tirzepatide dose when adding berberine, particularly if the tirzepatide dose was already well-tolerated and glucose was well-controlled. However, if glucose monitoring shows that fasting glucose is consistently falling below target range after adding berberine, discussing tirzepatide dose reduction with a prescribing physician is appropriate. The tirzepatide dose chart and the dosage calculator help contextualize what dose adjustments look like in practice.
What should I do if I experience hypoglycemia symptoms while on both?
If experiencing symptoms of hypoglycemia (shakiness, sweating, confusion, heart palpitations, unusual hunger) while on both compounds, check blood glucose immediately if a glucometer is available. If blood glucose is below 70 mg/dL, consume 15 to 20 grams of fast-acting carbohydrates, such as glucose tablets, fruit juice, or regular soda, and recheck in 15 minutes. Document the episode including what was eaten that day, timing of medications, and activity level. Then reduce berberine dose as a first step and discuss the episode with your physician. If hypoglycemia episodes recur, consider pausing berberine entirely until medical review. The blood sugar monitoring section of this guide covers target ranges and monitoring protocols in detail.
Can I take berberine with other supplements alongside tirzepatide?
Many researchers take berberine as part of a broader supplement stack alongside tirzepatide. Common additions include magnesium, vitamin D, omega-3 fatty acids, and various B vitamins. Most standard supplements do not interact significantly with berberine or tirzepatide. The more complex question involves pharmaceutical medications and compounds with known metabolic effects. The CYP450 enzyme inhibition from berberine is the primary interaction concern with pharmaceutical agents. For common GLP-1 compound additions, the guides on tirzepatide with B12, tirzepatide with levocarnitine, and tirzepatide with niacinamide cover specific compound additions in detail.
Does berberine worsen tirzepatide side effects?
Berberine can worsen gastrointestinal side effects when added to tirzepatide, particularly during the initial weeks of berberine introduction. Nausea, diarrhea, abdominal cramping, and bloating are the most commonly reported additive effects. These symptoms typically improve as the gut adapts to berberine over three to four weeks, mirroring the adaptation pattern seen with berberine monotherapy. Managing this with gradual berberine dose escalation, consistent meal-time dosing, adequate hydration, and probiotic support reduces the severity and duration of GI discomfort substantially. The comprehensive section on managing GI side effects in this guide covers all practical strategies.
External resources
PubMed, National Library of Medicine, berberine and metabolic disease research database
FDA Drug Interactions Labeling and Clinical Pharmacology guidance
National Institute of Diabetes and Digestive and Kidney Diseases, diabetes management information
Endocrine Society, evidence-based weight management and metabolic health resources
For researchers navigating complex metabolic compound protocols, SeekPeptides provides the most comprehensive resources available, including evidence-based interaction guides, personalized protocol frameworks, and a community of thousands of researchers actively managing tirzepatide, semaglutide, and complementary metabolic compounds. Membership brings access to detailed stacking protocols, dosing guidance, and expert-reviewed content that goes well beyond what any single guide can cover, particularly when managing combinations that require ongoing monitoring and adjustment.
In case I do not see you, good afternoon, good evening, and good night. May your blood sugar stay stable, your GI adaptation stay brief, and your metabolic results stay consistent.