Feb 5, 2026

Something strange is happening on TikTok and Amazon. Patches labeled "GLP-1" are selling by the thousands, promising weight loss results that sound suspiciously similar to what semaglutide and tirzepatide deliver. Onmorlo is one of the most searched brands in this category, and the questions flooding forums tell the real story: people want to know if these patches actually do anything.
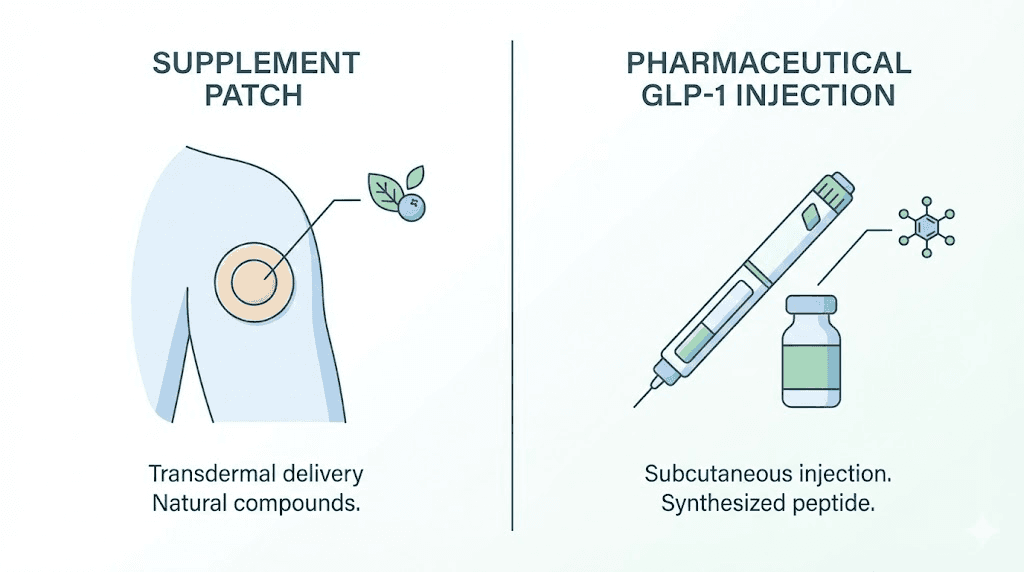
They do not contain GLP-1. Not a single milligram. Not a modified version. Not a "natural" form. The patches contain a blend of herbal ingredients, primarily berberine, pomegranate extract, cinnamon, and L-glutamine, delivered through adhesive patches worn on the skin for eight hours at a time. The marketing borrows heavily from the language of prescription GLP-1 receptor agonists like semaglutide and tirzepatide, but the product itself belongs to an entirely different category.
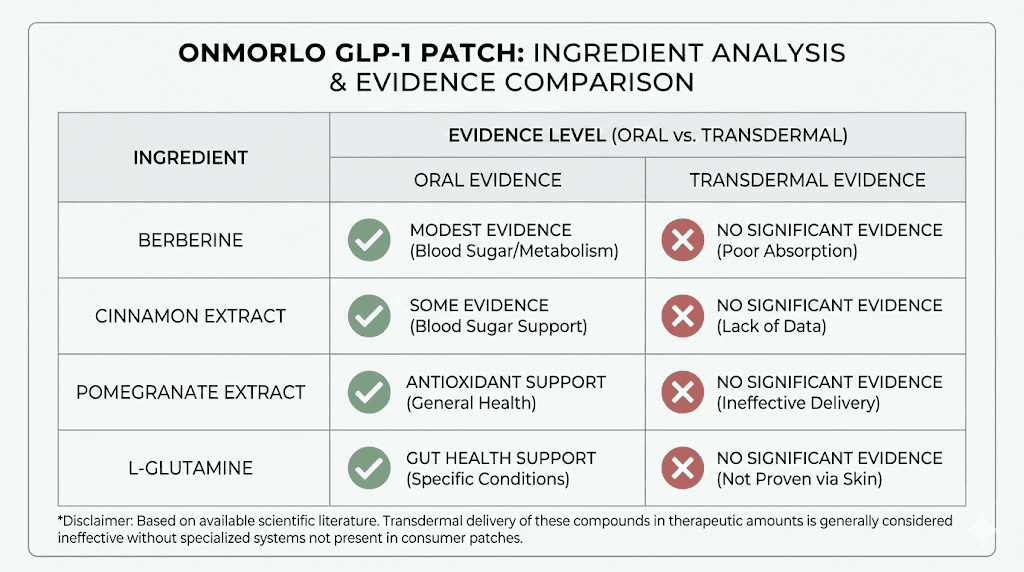
That does not mean the individual ingredients are worthless. Berberine has genuine clinical data behind it. Pomegranate shows interesting GLP-1 stimulating properties in research. Cinnamon has measurable metabolic effects.
But the question is not whether these compounds do something when swallowed. The question is whether they do anything when stuck to your skin. And the answer to that question matters enormously for the thousands of people spending money on these patches every month. This guide breaks down every ingredient, every claim, and every piece of evidence, so you can make a decision based on research rather than marketing.
What Onmorlo GLP-1 patches actually contain
Before evaluating whether these patches work, you need to understand exactly what is in them. The name "GLP-1 patch" creates a powerful association with prescription medications that have transformed weight loss treatment, but the ingredient list tells a completely different story.
The ingredient breakdown
Onmorlo patches contain four primary active ingredients. Berberine is the star of the formula, a plant alkaloid extracted from several herbs including goldenseal, barberry, and Oregon grape. It has been used in traditional Chinese medicine for centuries and has accumulated a respectable body of modern clinical research. Pomegranate extract provides polyphenols and ellagic acid. Cinnamon contributes cinnamaldehyde and other bioactive compounds. L-glutamine rounds out the formula as the most abundant amino acid in the human body.
None of these are peptides. None are GLP-1 receptor agonists. None function anything like semaglutide, tirzepatide, or retatrutide. The "GLP-1" in the product name refers to a marketing claim that these ingredients may support natural GLP-1 production in the body, not that the patches contain the actual incretin hormone or any synthetic analog of it.
This distinction matters more than anything else in this entire article.
How GLP-1 receptor agonists actually work
Real GLP-1 medications are synthetic peptides engineered to mimic and amplify the effects of a hormone your body naturally produces. Glucagon-like peptide-1 is secreted by L-cells in your intestine after you eat. It signals your pancreas to release insulin, slows gastric emptying, and tells your brain you are full. The natural hormone gets broken down by an enzyme called DPP-4 within about two minutes.
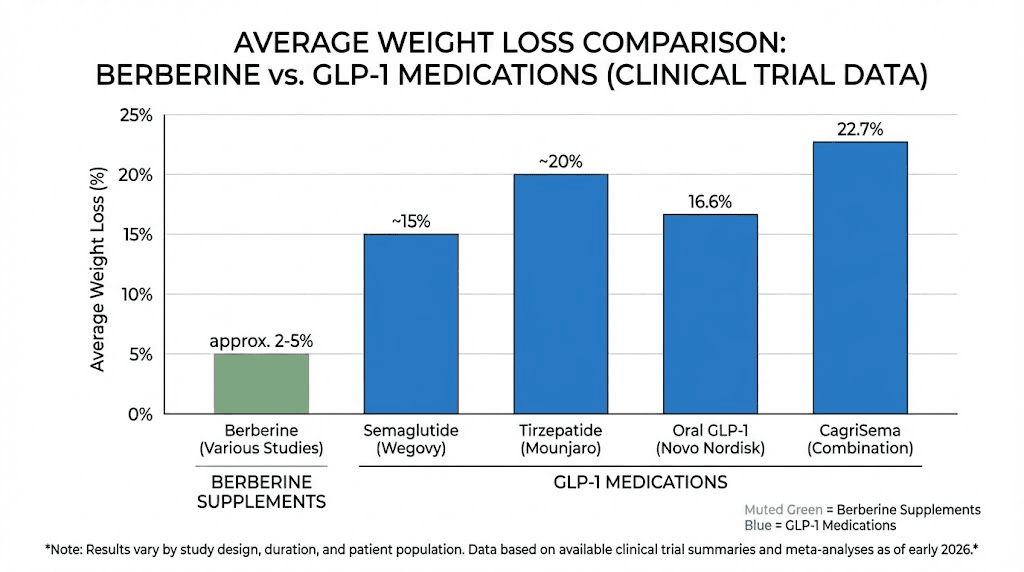
Pharmaceutical GLP-1 receptor agonists solve the rapid degradation problem. Semaglutide has been structurally modified to resist DPP-4 breakdown and bind to albumin in the blood, extending its half-life to approximately 168 hours. That is why a single weekly injection produces sustained appetite suppression, reduced food intake, and an average weight loss of 15-20% of body weight in clinical trials. Tirzepatide goes further by activating both GLP-1 and GIP receptors simultaneously, producing even greater weight loss in head-to-head studies.
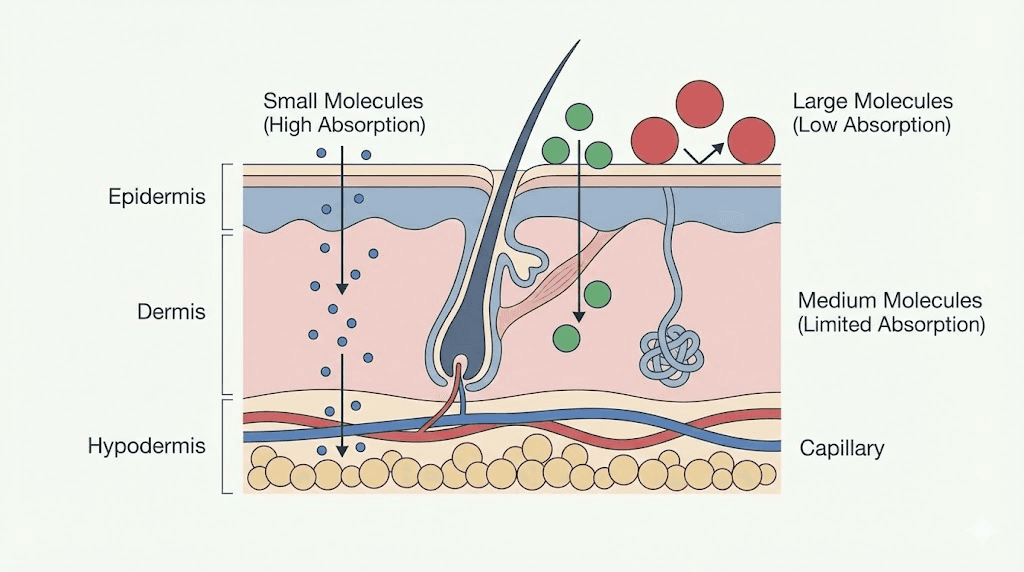
These are large, complex molecules. Semaglutide has a molecular weight of approximately 4,114 daltons. For context, most drugs that successfully penetrate the skin weigh less than 500 daltons. This is not a minor technical challenge. It is a fundamental barrier that no supplement patch has overcome.
What the patches do not contain
The patches contain zero milligrams of semaglutide, tirzepatide, liraglutide, dulaglutide, exenatide, or any other GLP-1 receptor agonist. They contain no synthetic peptides of any kind. They contain no prescription medications. They are classified as dietary supplements, though the FDA has raised questions about whether transdermal patches even qualify for that classification since dietary supplements are legally defined as products intended for ingestion.
A study published in the Annals of Pharmacotherapy specifically flagged transdermal "natural GLP-1" dietary supplements as potentially violating federal law and placing patients at risk. The researchers noted that these products cannot legally be marketed as dietary supplements because they are not intended for ingestion, and they cannot be marketed as drugs because they have not undergone FDA approval.
The transdermal delivery problem
Even if we set aside the GLP-1 naming issue entirely, a fundamental question remains. Can these ingredients pass through your skin and enter your bloodstream in meaningful quantities? The science here is not encouraging.
Your skin is a barrier, not a sponge
Human skin evolved specifically to keep things out. The outermost layer, the stratum corneum, consists of dead skin cells arranged like bricks in a wall, sealed together with a mortar of lipids. This barrier is remarkably effective at preventing molecular penetration. When something spills on your skin, you wipe it off. You do not absorb it into your bloodstream.
Transdermal drug delivery does work for certain medications. Nicotine patches, fentanyl patches, and hormone patches are all FDA-approved and clinically validated. But these medications share specific properties that make transdermal delivery feasible. They are small molecules (typically under 500 daltons). They are lipophilic (fat-soluble), which helps them pass through the lipid-rich stratum corneum. And they are effective at very low doses, meaning even limited skin absorption delivers therapeutic concentrations.
Berberine does not fit this profile well. Its molecular weight (336 daltons) is within the acceptable range for transdermal delivery in theory, but berberine is poorly absorbed even through the gastrointestinal tract, where conditions are far more favorable for absorption. Oral bioavailability of berberine is estimated at less than 1%. The idea that significant quantities would pass through the skin barrier, enter the bloodstream, and reach therapeutic concentrations is not supported by any published human study.
What the vitamin patch research shows
The most relevant clinical data comes from studies on vitamin and nutrient patches, which use similar delivery methods. A study on gastric bypass patients compared vitamin pills versus vitamin patches and found that the patch group had significantly lower levels of vitamins D, B1, and B12 compared to the pill group. A separate study on athletes with low iron showed that those using iron pills had significantly higher serum ferritin than those using iron patches.
No published clinical trial has demonstrated that berberine, pomegranate extract, cinnamon, or L-glutamine achieves meaningful blood concentrations when delivered through a skin patch. This is not a gap that can be dismissed with "more research needed." It is a fundamental question about whether the delivery method works at all, and the existing evidence from similar products suggests it does not.
Berberine: the most researched ingredient in the formula
If any ingredient in Onmorlo patches deserves a fair hearing, it is berberine. This compound has real clinical data. Real mechanisms. Real effects. But context matters enormously when evaluating whether those effects translate to a patch you stick on your arm.
What berberine actually does when taken orally
Berberine activates an enzyme called AMP-activated protein kinase (AMPK), which plays a central role in cellular energy metabolism. This mechanism has led to berberine being compared to metformin, a prescription diabetes medication, and the comparison is not entirely unfair. Both compounds activate AMPK. Both have demonstrated effects on blood sugar regulation. Both have shown modest weight-related benefits in clinical research.
A systematic review of 18 studies found that berberine supplementation produced significant decreases in both body weight and BMI. A meta-analysis of randomized controlled trials found that oral berberine reduced body weight by an average of 2.07 kg (about 4.5 pounds), BMI by 0.47 kg/m2, and waist circumference by 1.08 cm. Some individual studies reported weight loss between 5% and 7% of body weight over 8-12 weeks at doses of 1,000 mg to 1,500 mg daily.
Those numbers are real. They are also dramatically different from what prescription GLP-1 medications deliver. Semaglutide produces average weight loss of 15-17% of body weight. Tirzepatide averages 20-25%. The gap between berberine and actual GLP-1 receptor agonists is not small. It is enormous.
Berberine and blood sugar
Where berberine shows its strongest evidence is in glycemic control. Multiple studies demonstrate that oral berberine at 1,000-1,500 mg daily can reduce fasting blood glucose, HbA1c, and insulin resistance. A meta-analysis found significant improvements in fasting plasma glucose and HOMA-IR (a measure of insulin resistance) across multiple randomized controlled trials.
This matters for weight management because insulin resistance is a major driver of weight gain and difficulty losing weight. By improving insulin sensitivity, berberine may create metabolic conditions that make weight loss easier, even if it does not directly suppress appetite the way tirzepatide suppresses appetite through receptor binding.
But here is the critical problem. Every study showing these benefits used oral berberine. Swallowed. Digested. Processed through the gastrointestinal tract. Not one single published study has evaluated berberine delivered through a skin patch for metabolic effects.
The bioavailability problem
Berberine already has terrible bioavailability when you swallow it. Oral bioavailability sits below 1%, meaning that out of a 500 mg capsule, your body absorbs less than 5 mg into the bloodstream. The rest gets metabolized by gut bacteria or excreted. Researchers have spent years trying to improve oral berberine absorption using nanoparticles, liposomes, and other pharmaceutical technologies.
If berberine struggles to get absorbed through the gut, where your body is specifically designed to absorb nutrients, the idea that it would successfully penetrate the skin barrier in therapeutic quantities requires extraordinary evidence. That evidence does not exist.
The dose problem
Clinical studies showing benefits use 1,000-1,500 mg of oral berberine daily. Given oral bioavailability below 1%, roughly 10-15 mg actually reaches systemic circulation. A transdermal patch would need to deliver at least this amount through the skin. But patch dosing for Onmorlo is not disclosed in milligrams, making it impossible to evaluate whether the patch contains enough berberine to produce any measurable effect, even if transdermal absorption somehow worked.
Researchers who study peptide dosing and drug delivery understand that the route of administration fundamentally changes everything about how a compound behaves. A molecule that works orally will not necessarily work transdermally, and vice versa. Assuming otherwise is not just optimistic. It is pharmacologically illiterate.
Pomegranate extract and GLP-1 stimulation
The inclusion of pomegranate extract is likely where the "GLP-1" marketing claim gets its thinnest scientific justification. There is actual research connecting pomegranate to GLP-1 production. But the gap between what the studies show and what the patches promise is wide enough to drive a truck through.
What the pomegranate research actually shows
A randomized controlled trial studied women with type 2 diabetes who consumed 100 ml of pomegranate juice daily for eight weeks. The results showed enhanced GLP-1 levels and decreased DPP-4 and glucose levels. DPP-4 is the enzyme that breaks down natural GLP-1, so inhibiting it allows your body's own GLP-1 to last longer. This is actually the same mechanism used by prescription DPP-4 inhibitor medications like sitagliptin.
Pomegranate peel also acts as a prebiotic, feeding beneficial gut bacteria that produce short-chain fatty acids. These fatty acids stimulate L-cells in the intestine to secrete more GLP-1. The prebiotic mechanism is interesting and represents a real biological pathway.
But notice something critical about both mechanisms. They require interaction with the digestive system. GLP-1 is produced by L-cells in your intestine. DPP-4 operates primarily in the gut. Prebiotic effects require gut bacteria. A pomegranate extract sitting on top of your skin cannot interact with any of these systems because it never reaches your intestine.
Pomegranate through the skin: no evidence
Topical pomegranate extract has been studied for skin-specific benefits like UV protection, antioxidant activity, and anti-aging effects. Some pomegranate polyphenols may penetrate the outer skin layers and provide local antioxidant protection. But local skin effects are completely different from systemic metabolic effects. A compound sitting in your epidermis cannot stimulate GLP-1 production in your gut.
There are zero published studies showing that transdermal pomegranate extract produces measurable increases in circulating GLP-1 levels, reduces appetite, or contributes to weight loss. The research connecting pomegranate to GLP-1 stimulation only applies when the extract is consumed orally and reaches the gastrointestinal tract.
Cinnamon: metabolic effects and limitations
Cinnamon is one of the most studied spices in metabolic research. Its inclusion in Onmorlo patches is not random. There is real science here. But once again, the delivery method creates problems the marketing conveniently ignores.
Oral cinnamon and weight loss
An umbrella meta-analysis combining data from seven separate meta-analyses found that cinnamon supplementation significantly reduced body weight by an average of 0.67 kg, BMI, and waist-hip ratio. The greatest effects appeared at dosages of 2 grams or more per day taken for at least 12 weeks. Body weight reduction followed a non-linear pattern, with benefits plateauing after a certain dose threshold.
Cinnamon appears to work through multiple mechanisms. It improves insulin sensitivity. It may influence adipokine levels, including leptin and adiponectin, which regulate appetite and fat metabolism. A study found that 56 days of cinnamon supplementation enhanced adiponectin levels and decreased leptin levels in overweight women. Cinnamaldehyde, the compound responsible for cinnamon's distinctive flavor, has shown direct effects on fat cell metabolism in laboratory studies.
Average weight loss from cinnamon supplementation: less than 1.5 pounds. Compare that to the 30-50+ pounds that tirzepatide dosing protocols deliver over similar timeframes. The scale of effect is fundamentally different.
Transdermal cinnamon delivery
Cinnamaldehyde is a small, lipophilic molecule that theoretically could penetrate skin to some degree. Essential oils containing cinnamaldehyde are known skin penetration enhancers, meaning they can help other molecules cross the skin barrier. However, this property relates to local skin effects, not systemic metabolic effects.
No clinical trial has measured whether transdermal cinnamon extract produces meaningful changes in blood glucose, insulin sensitivity, body weight, or any metabolic parameter. The assumption that patch delivery would replicate the effects seen with oral supplementation is unsupported by evidence.
L-glutamine: the amino acid addition
L-glutamine is the most abundant amino acid in the human body. Your muscles, gut, and immune system use it constantly. Including it in a weight loss patch seems like an odd choice until you look at the limited research connecting glutamine to appetite regulation.
Glutamine and appetite
L-glutamine supports serotonin production, and serotonin plays a role in appetite regulation. A pilot study in non-dieting obese women found that glutamine supplementation produced modest weight loss compared to placebo. A six-week study in 66 people with type 2 diabetes found that glutamine reduced both belly fat and body fat. A two-week study observed decreased waist circumference in 39 people with overweight or obesity.
The evidence is preliminary. Sample sizes are small. Results are modest. And most importantly, all studies used oral supplementation.
Glutamine and gut health
Where L-glutamine shows its strongest evidence is in gut health. It serves as the primary fuel source for enterocytes, the cells lining your intestinal wall. Supplementation has shown benefits for intestinal barrier integrity, reduced gut inflammation, and improved microbiome diversity. Combined with a low-FODMAP diet, glutamine supplementation may improve IBS symptoms.
These gut-related benefits are interesting but require the glutamine to actually reach your gut. A patch on your arm does not deliver glutamine to your intestinal lining. The gut-specific mechanisms that make glutamine useful for digestive health cannot operate when the compound never contacts your digestive system.
Transdermal glutamine: the evidence gap
L-glutamine is a hydrophilic (water-soluble) amino acid. Water-soluble compounds face significant challenges in transdermal delivery because the skin's lipid-rich barrier preferentially allows fat-soluble molecules to pass. Transdermal delivery of water-soluble vitamins like B12 and C has already been shown to underperform compared to oral supplementation in clinical studies.
No published research has evaluated transdermal L-glutamine for weight loss, appetite control, or any metabolic effect. The assumption that a glutamine-containing patch would replicate the modest benefits seen with oral supplementation has no scientific basis.
The TikTok and Amazon marketing phenomenon
Understanding how these patches became so popular requires looking beyond the ingredients and into the marketing ecosystem that drives supplement sales. The GLP-1 patch trend did not emerge from scientific discovery. It emerged from social media algorithms and affiliate marketing structures.
How GLP-1 patches went viral
GLP-1 medications like Ozempic and Mounjaro became cultural phenomena. Celebrity endorsements, dramatic before-and-after photos, and widespread media coverage created enormous consumer demand. But prescription GLP-1 agonists require a doctor's prescription, cost hundreds of dollars per month, and involve self-injection. Millions of people wanted the results without the barriers.
Supplement companies recognized the opportunity. By creating products with "GLP-1" in the name and marketing them with language borrowed from pharmaceutical advertising, they could capture search traffic from people looking for cheaper, easier alternatives to prescription medications. TikTok's algorithm amplified the trend, with influencer-created content driving millions of views to GLP-1 patch products.
Media Matters for America reported that unregulated GLP-1 patches exploded on TikTok Shop despite the platform's stated ban on weight loss product advertising. Fast Company documented how TikTok was flooded with GLP-1 patch promotions. NBC's Today Show consulted doctors who uniformly expressed skepticism about the products' claims. Rolling Stone investigated the influencer economics behind GLP-1 patch promotion.
The marketing language problem
Onmorlo and similar brands walk a careful line with their marketing. They do not explicitly claim to contain semaglutide or tirzepatide. But the "GLP-1" branding, combined with weight loss imagery and language about "appetite support" and "metabolic enhancement," creates an implied association with prescription medications that the products do not share.
The Better Business Bureau reported a spike in scam complaints related to weight loss supplements in late 2025, many involving products marketed with GLP-1 terminology. AI-generated advertisements featuring deepfake celebrity endorsements added another layer of deception to the marketing ecosystem.
PBS News characterized the GLP-1 supplement space as a "gray world" where health experts urge caution. The fundamental problem is straightforward: when a product uses the same terminology as a prescription medication, consumers naturally expect similar results. Those expectations are not met.
Amazon reviews: what buyers report
Customer reviews for Onmorlo and similar GLP-1 patches show a predictable pattern. Some users report reduced appetite or mild weight loss, which could reflect genuine effects from the herbal ingredients, placebo response, or coincidental changes in diet and behavior during the trial period. Other users report no effects whatsoever. Some report skin irritation, rashes, and in rare cases, blistering from the adhesive.
The lack of controlled clinical trial data makes it impossible to distinguish genuine pharmacological effects from placebo responses. In weight loss research, placebo effects are substantial. Clinical trials of weight loss interventions consistently show 1-3% body weight reduction in placebo groups, which means some people will lose weight simply because they believe a treatment is working and unconsciously modify their behavior.
What real GLP-1 patches would look like
Legitimate pharmaceutical companies are working on transdermal delivery systems for actual GLP-1 medications. These products look nothing like what is currently sold on Amazon, and they are years away from commercial availability.
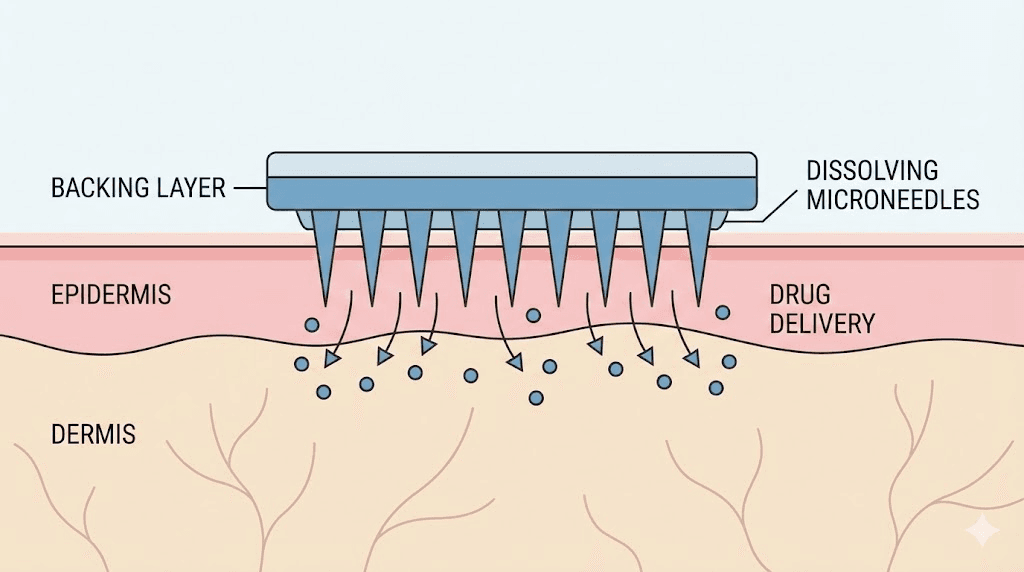
Microneedle technology
The most promising approach uses microneedle patches, which are fundamentally different from adhesive supplement patches. Microneedle patches contain hundreds of tiny dissolving needles, typically 600-800 micrometers long, that physically penetrate the skin's outer barrier and deliver medication directly into the dermis. This bypasses the stratum corneum entirely, solving the molecular size problem that prevents large peptides from crossing the skin.
A preclinical study demonstrated a semaglutide-loaded microneedle patch that achieved comparable plasma drug concentrations to subcutaneous injection in obese mice. The researchers developed a self-boosting system that enabled repeated release of semaglutide every seven days from a single application, sustaining drug efficacy for an entire month.
This technology is genuinely exciting. It could eventually replace weekly injections with a painless patch application. But it requires actual semaglutide loaded into dissolving microneedles, not herbal extracts sitting on an adhesive backing. The technology is in preclinical and early clinical development, meaning it has not yet completed the years of human trials required for FDA approval.
Anyone researching microneedle peptide patches will find that the technology is promising but not yet commercially available for GLP-1 delivery. The oral tirzepatide development pathway offers a closer analogy for non-injection delivery, and even that has required years of pharmaceutical engineering to overcome absorption challenges.
The fundamental difference
Real GLP-1 transdermal patches, when they eventually arrive, will be prescription medications requiring FDA approval, clinical trials with thousands of participants, and physician oversight. They will contain actual GLP-1 receptor agonists delivered through sophisticated microneedle technology. They will produce measurable changes in HbA1c, body weight, and metabolic parameters that can be verified through blood tests.
Supplement patches sold on Amazon bypass all of this. No FDA approval. No clinical trials. No physician oversight. No actual GLP-1 medication. The only thing they share with future pharmaceutical GLP-1 patches is that both are called "patches."
FDA regulation and legal considerations
The regulatory status of GLP-1 supplement patches exists in a genuinely problematic gray area that consumers need to understand before spending money on these products.
Dietary supplements cannot be patches
Under the Dietary Supplement Health and Education Act (DSHEA), a dietary supplement is legally defined as a product intended for ingestion. The FDA has explicitly stated that topical patch products and other products not intended for ingestion are not dietary supplements. Products cannot be injected, administered sublingually, or delivered transdermally and still qualify as dietary supplements.
This creates an immediate legal problem for GLP-1 supplement patches. If they are marketed as dietary supplements, they cannot legally be patches. If they are marketed as transdermal delivery systems, they are classified as drugs and require FDA approval before being sold. Most GLP-1 patch companies thread this needle by listing their products as dietary supplements on Amazon while selling them in patch form, essentially occupying a regulatory gap that has not yet been fully addressed by enforcement.
No pre-market approval
Unlike prescription medications, dietary supplements do not require FDA approval before being sold. Supplement manufacturers are responsible for ensuring their products are safe and accurately labeled, but they do not need to demonstrate efficacy. The FDA can take enforcement action after the fact if a product is found to be adulterated or misbranded, but this reactive approach means products can be sold for months or years before problems are identified.
The FDA has issued warning letters to companies marketing transdermal patches as dietary supplements. In one case, the FDA told a supplement company that their patch products were being marketed as unapproved new drugs because the company made specific disease-treatment claims. The regulatory framework exists. Enforcement simply has not caught up with the explosion of GLP-1 supplement patches on marketplace platforms.
What this means for consumers
When you buy a prescription GLP-1 medication like semaglutide, you are getting a product that has undergone Phase I, II, and III clinical trials involving thousands of participants. The manufacturing facility is inspected regularly. Batch testing ensures consistent potency. Adverse events are tracked through post-market surveillance.
When you buy Onmorlo patches on Amazon, none of these safeguards apply. You are trusting the manufacturer's claims about what is in the product, how much of each ingredient is included, and whether the patch delivers any of those ingredients into your body. There is no independent verification of any of these claims.
Comparing real options for natural GLP-1 support
If the goal is to support your body's natural GLP-1 production without prescription medications, there are approaches with substantially better evidence than transdermal patches. Every option discussed here has been studied in oral or dietary form, where absorption is at least possible.
Oral berberine supplementation
If berberine interests you, take it orally. Standard dosing is 500 mg two to three times daily with meals, totaling 1,000-1,500 mg per day. Studies using this protocol have demonstrated modest but measurable effects on body weight (average reduction of 2.07 kg), blood glucose, and insulin sensitivity. Effects become measurable after 8-12 weeks of consistent use.
Berberine does cause gastrointestinal side effects in some people, including nausea, abdominal pain, bloating, constipation, and diarrhea. Starting with a lower dose (500 mg once daily) and increasing gradually can minimize these effects. Berberine interacts with several medications, including metformin, blood thinners, and certain blood pressure medications. Consult a healthcare provider before starting.
Anyone who is not losing weight on semaglutide or other GLP-1 medications should address underlying issues with their protocol rather than supplementing with berberine patches.
Dietary approaches that stimulate GLP-1
Your body produces GLP-1 naturally, and certain dietary choices can enhance that production. Protein-rich meals stimulate significantly more GLP-1 release than carbohydrate-heavy meals. Fiber, particularly soluble fiber from sources like oats, legumes, and vegetables, feeds gut bacteria that produce short-chain fatty acids, which in turn stimulate L-cells to secrete GLP-1.
Fermented foods support a diverse gut microbiome that enhances GLP-1 production. Pomegranate juice (consumed orally, not through a patch) has demonstrated GLP-1 stimulating effects in clinical research. Green tea catechins, olive oil polyphenols, and even certain spices consumed in food have shown modest effects on incretin hormones.
These dietary approaches produce subtle effects compared to prescription medications. Nobody is going to replicate semaglutide results by eating more fiber. But they are free, backed by actual evidence, and do not require purchasing patches from Amazon.
Exercise and GLP-1
Both aerobic exercise and resistance training stimulate GLP-1 secretion. The combination of resistance training and pomegranate juice consumption produced enhanced GLP-1 levels in a randomized controlled trial of women with type 2 diabetes. Exercise improves insulin sensitivity independently of weight loss, creating metabolic conditions that support better appetite regulation.
For people interested in targeting belly fat, combining exercise with appropriate peptide stacking protocols represents a more evidence-based approach than relying on supplement patches.
Prescription GLP-1 medications
For significant weight loss, prescription GLP-1 receptor agonists remain the gold standard. Semaglutide and tirzepatide have been studied in trials involving tens of thousands of participants. Their efficacy, safety profiles, and optimal dosing are well established. Retatrutide represents the next generation, targeting three receptors simultaneously with even more impressive clinical trial results.
These medications require a prescription and medical monitoring for good reason. They are potent drugs with real side effects, including nausea, fatigue, gastrointestinal symptoms, and in rare cases, more serious complications. The medical oversight ensures appropriate dosing, side effect management, and monitoring for complications.
The cost barrier is real. Without insurance, semaglutide and tirzepatide can cost $800-1,300+ per month. Compounded versions are available at lower cost through telehealth platforms, though availability and legality vary by jurisdiction. These economic realities partly explain why affordable alternatives like GLP-1 patches attract such strong consumer interest, even when the evidence behind them is weak.
Side effects and safety concerns
GLP-1 supplement patches are generally considered low-risk because the ingredients are common supplements and the amounts likely to be absorbed through the skin are minimal. But "low-risk" does not mean "no-risk," and several safety considerations warrant attention.
Skin reactions
The most commonly reported side effects involve the skin at the application site. Redness, itching, and mild irritation are relatively common with adhesive patches of any kind. Some users of GLP-1 patches have reported more severe reactions including rashes, burns, and blistering. These reactions may result from the adhesive itself, the herbal ingredients, or penetration enhancers added to promote skin absorption.
People with sensitive skin, eczema, psoriasis, or other skin conditions should exercise particular caution. Applying herbal extracts to compromised skin barriers increases the risk of irritation and allergic reactions. If you experience any skin reaction beyond mild redness, remove the patch immediately and clean the area with mild soap and water.
Medication interactions
Even with limited transdermal absorption, the ingredients in GLP-1 patches could theoretically interact with medications. Berberine interacts with CYP450 enzymes, which process many prescription drugs. It can increase the effects of blood sugar-lowering medications, potentially causing hypoglycemia. It may enhance the effects of blood thinners. It can interact with immunosuppressants, certain antibiotics, and statins.
The uncertainty about transdermal absorption actually makes these interactions harder to predict, not easier. If you are taking any prescription medications, the safest approach is to either avoid these patches entirely or discuss them with your healthcare provider.
The delayed treatment concern
Perhaps the most significant safety concern is not what the patches do, but what they prevent. If someone with genuine metabolic disease, significant obesity, or uncontrolled diabetes uses GLP-1 patches instead of seeking proper medical treatment, the patches become actively harmful through inaction. The modest-at-best effects of herbal supplement patches cannot substitute for medications that produce 15-25% body weight reduction and significant improvements in glycemic control, cardiovascular risk, and metabolic parameters.
Anyone researching peptides for weight loss should understand the fundamental difference between evidence-based peptide therapies with published clinical trial data and supplement patches marketed with borrowed pharmaceutical terminology.

The cost-effectiveness calculation
Onmorlo patches typically sell for $20-40 for a 30-day supply (60 patches, worn for 8 hours each). This seems affordable compared to prescription GLP-1 medications. But cost-effectiveness is about what you get for your money, not just what you spend.
Cost per pound of weight loss
If Onmorlo patches produce the same weight loss as oral berberine in the most optimistic scenario (which is unlikely given the transdermal delivery issues), that would be roughly 2 kg (4.5 pounds) over 8-12 weeks. At $30 per month, that is $60-90 for 4.5 pounds of weight loss, or about $13-20 per pound.
Oral berberine supplements cost $10-20 per month for clinically studied doses (1,000-1,500 mg daily). Using the same optimistic weight loss estimate, oral berberine costs $4-9 per pound of weight loss, roughly half the cost of patches for the same ingredient with better-documented absorption.
Prescription semaglutide at $800-1,300 per month produces average weight loss of 2-4 pounds per week after the initial titration period. Over 12 weeks, total weight loss averages 15-25 pounds, resulting in a cost of $32-52 per pound. More expensive in absolute terms, but dramatically more effective per dollar in total weight management impact.
The real comparison
When you buy Onmorlo patches, you are paying $30 per month for a product with zero clinical trial evidence, questionable absorption, and ingredients you could obtain for less than $15 per month in oral form with better-documented effects. The premium you pay for the patch format buys you convenience and a placebo delivery mechanism that looks and feels like a real treatment.
If you are serious about using berberine, cinnamon, or pomegranate for metabolic support, oral supplementation is cheaper and backed by actual clinical data. If you are serious about significant weight loss, prescription GLP-1 medications, while more expensive, deliver results that are orders of magnitude greater.
What the medical community says
The medical establishment's position on GLP-1 supplement patches is remarkably unified. Physicians, pharmacists, endocrinologists, and obesity medicine specialists who have commented publicly on these products share the same assessment.
Doctor perspectives
When NBC's Today Show asked doctors about GLP-1 patches, the response was uniform skepticism. Physicians emphasized that these patches do not contain actual GLP-1 medications, that the transdermal delivery of the included ingredients has not been validated, and that consumers should not expect results comparable to prescription GLP-1 agonists.
The Cleveland Clinic's assessment of berberine as a weight loss supplement acknowledges the compound's modest evidence for oral use while noting that it is "not Ozempic" and should not be treated as an equivalent. UCLA Health characterized berberine as "so-called nature's Ozempic" while pointing out the enormous gap between berberine's effects and actual GLP-1 medications.
The National Center for Complementary and Integrative Health (NCCIH), part of the National Institutes of Health, specifically addressed berberine and weight loss, noting that there are not enough clinical studies to say with certainty how much weight people will lose, and that most existing studies had limitations including small sample sizes and inconsistent formulations.
Pharmacist and researcher warnings
A paper published in the Annals of Pharmacotherapy explicitly warned that transdermal "natural GLP-1" dietary supplements "violate law and place patients at risk." The researchers argued that these products occupy an illegal gray area: they cannot be dietary supplements because they are not intended for ingestion, and they cannot be drugs because they have not been approved.
This is not a fringe opinion. It represents the mainstream scientific assessment of a product category that has grown primarily through social media marketing rather than clinical validation.
Alternatives that actually work for appetite and weight management
If you landed on this article because you want help managing appetite or losing weight, here are options sorted from least to most aggressive, all backed by better evidence than GLP-1 supplement patches.
Dietary and lifestyle modifications
Increasing protein intake to 1.2-1.6 grams per kilogram of body weight per day consistently reduces appetite and supports lean mass preservation during weight loss. Soluble fiber from whole food sources stimulates natural GLP-1 production. Regular exercise, particularly resistance training, improves insulin sensitivity and metabolic rate. Adequate sleep (7-9 hours) and stress management support healthy cortisol levels, which directly influence appetite and fat storage.
These approaches produce modest but sustainable results. They are free, universally applicable, and supported by decades of clinical research.
Evidence-based oral supplements
If you want to supplement, choose compounds with actual absorption data. Oral berberine at 1,000-1,500 mg daily has the strongest evidence in this category. Psyllium husk fiber (5-10 grams daily) provides soluble fiber that feeds GLP-1 producing gut bacteria. Chromium picolinate (200-1,000 mcg daily) has modest evidence for blood sugar regulation. Green tea extract standardized for EGCG shows small but consistent effects on metabolic rate.
None of these will produce dramatic weight loss. All of them have more clinical support than transdermal delivery of the same compounds.
Prescription peptide therapies
For significant results, prescription peptides for weight loss remain the most effective pharmacological option. Semaglutide at therapeutic doses produces 15-17% body weight reduction. Tirzepatide produces 20-25%. Retatrutide has shown up to 24% in clinical trials targeting three receptors simultaneously. Cagrilintide combined with semaglutide represents another emerging approach with impressive early data.
These medications require medical supervision, produce real side effects, and cost significantly more than supplement patches. But they also produce real, measurable, clinically validated results. Understanding the difference between marketing claims and clinical evidence is essential for making informed decisions about your health.
SeekPeptides members access comprehensive protocol guides, dosage charts, and evidence-based information for every major peptide therapy. Whether you are researching GLP-1 agonists, BPC-157 for recovery, or optimal peptide stacks for weight loss, the platform provides the detailed protocols and community support that generic supplement marketing cannot replicate.
What happens when you wear the patch
For complete transparency, here is the most honest assessment possible of what likely happens when someone applies an Onmorlo GLP-1 patch.
Hour 0-1: Application
You apply the patch to clean, dry skin. The adhesive holds the patch in place. The herbal ingredients in the patch matrix sit against your skin surface. Some volatile compounds from cinnamon oil may produce a mild warming or tingling sensation. This sensation feels like "something is happening" and reinforces the belief that the product is working.
Hours 1-8: Wear period
During the wear period, some small molecules from the herbal ingredients may penetrate the outer skin layers. Whether any reach the deeper dermis or enter the bloodstream in measurable quantities is unknown because no clinical study has measured this. Your skin may become slightly irritated from the adhesive, especially in warm or humid conditions.
If you experience reduced appetite during this period, it could result from trace amounts of herbal compounds reaching your bloodstream, from the placebo effect of wearing a treatment, from your attention to food intake being heightened by the act of using a weight loss product, or from any number of other factors unrelated to the patch.
Hour 8: Removal
You remove the patch. If there is skin redness, this is from the adhesive, not from the ingredients being "absorbed." You apply a new patch. The cycle repeats.
Weeks 1-4: Evaluation period
Over the first month, some users will lose weight. Some will not. Those who lose weight may attribute it to the patch, but without a control group, it is impossible to determine whether the weight loss resulted from the patch ingredients, the placebo effect, increased attention to diet and exercise during the trial period, or normal weight fluctuations.
Onmorlo recommends using the patches for 1-3 months before evaluating results. This is a standard supplement marketing practice, as longer usage periods increase the chance that natural weight fluctuations, seasonal dietary changes, or lifestyle modifications will produce results that can be attributed to the product.
The bottom line on Onmorlo GLP-1 patches
Let me be direct.
The individual ingredients in Onmorlo patches have some clinical evidence behind them, but only when taken orally. Berberine can produce modest weight loss when swallowed at 1,000-1,500 mg daily. Pomegranate extract may support GLP-1 production when consumed as juice. Cinnamon shows small metabolic effects when ingested at adequate doses.
None of these ingredients have been demonstrated to produce metabolic effects when delivered through a skin patch. The transdermal delivery method used by Onmorlo and similar products has no clinical validation for these specific compounds. The "GLP-1" label is marketing, not pharmacology. These patches contain zero GLP-1 medication.
The expected weight loss, even in the most optimistic scenario, would be a fraction of what prescription GLP-1 agonists deliver. If the patches work at all (unproven), the magnitude of effect would be comparable to or less than taking the same ingredients orally, which costs less and has actual absorption data behind it.
For anyone serious about evidence-based weight management, the hierarchy is clear. Dietary and lifestyle modifications form the foundation. Oral supplements with clinical evidence can provide modest additional support. Prescription GLP-1 medications deliver dramatic results with medical oversight. GLP-1 supplement patches occupy none of these categories convincingly.
Spend your money on food that stimulates GLP-1 naturally. Spend it on oral berberine if you want the metabolic support. Spend it on a gym membership. Spend it on a consultation with a physician about prescription options. Any of these choices will produce better-documented outcomes than an herbal patch stuck on your arm for eight hours.
For researchers navigating the complex landscape of peptide-based weight loss, dosing protocols, and evidence-based protocols, SeekPeptides provides the most comprehensive resource available. Members access detailed tirzepatide dosing guides, retatrutide dosage charts, peptide calculators, and a community of thousands who have navigated these exact questions with real data rather than marketing claims.
Frequently asked questions
Do Onmorlo GLP-1 patches contain actual GLP-1?
No. Onmorlo patches contain berberine, pomegranate extract, cinnamon, and L-glutamine. They contain zero GLP-1 medication, no synthetic peptides, and no pharmaceutical compounds. The "GLP-1" in the name refers to a marketing claim that these ingredients may support natural GLP-1 production, not that the product contains the hormone or any receptor agonist.
Can berberine be absorbed through the skin?
Berberine has a molecular weight (336 daltons) that theoretically allows some skin penetration, but no published clinical study has demonstrated that transdermal berberine achieves therapeutic blood concentrations. Even oral berberine has bioavailability below 1%. Researchers who study peptide reconstitution and drug delivery understand that route of administration fundamentally changes pharmacokinetics.
Are GLP-1 patches FDA approved?
No GLP-1 supplement patches are FDA approved. The FDA has not approved any transdermal patch for weight loss that contains herbal ingredients. Additionally, the FDA has stated that transdermal patches do not qualify as dietary supplements, creating a regulatory gray area that these products currently occupy.
How much weight can you lose with GLP-1 patches?
No clinical trial has measured weight loss specifically from GLP-1 supplement patches. If the patches deliver similar effects to oral berberine (unproven), expected weight loss would be approximately 2 kg (4.5 pounds) over 8-12 weeks. By comparison, prescription semaglutide produces 15-17% body weight reduction and tirzepatide produces 20-25%.
Are Onmorlo patches safe?
The ingredients are generally recognized as safe when taken orally. The most common side effects from the patches involve skin irritation, redness, and in rare cases, blistering from the adhesive. People taking blood sugar medications, blood thinners, or immunosuppressants should avoid berberine-containing products or consult their physician due to potential interactions.
What is better than GLP-1 patches for weight loss?
Oral berberine supplementation (1,000-1,500 mg daily) provides better-documented effects at lower cost. Dietary modifications including increased protein and fiber stimulate natural GLP-1 production. Prescription GLP-1 medications deliver dramatically greater weight loss with clinical trial validation. All of these options have more evidence behind them than transdermal herbal patches.
Are microneedle GLP-1 patches real?
Pharmaceutical companies are developing microneedle patches that could deliver actual GLP-1 medications through the skin using tiny dissolving needles. Preclinical studies have shown promising results with semaglutide-loaded microneedle patches in animal models. However, these products are years away from potential FDA approval and are completely different from the herbal supplement patches currently sold online.
Can I use GLP-1 patches with semaglutide or tirzepatide?
Using herbal supplement patches alongside prescription GLP-1 medications is not recommended without physician approval. Berberine can enhance the blood sugar-lowering effects of GLP-1 agonists, potentially causing hypoglycemia. If you are already taking a prescription GLP-1 medication, the patches are unlikely to add any benefit and may complicate your treatment. Anyone taking peptides with Ozempic should consult their prescribing physician about potential interactions.
External resources
National Center for Complementary and Integrative Health: Berberine and weight loss
Cleveland Clinic: Berberine for weight loss
In case I do not see you, good afternoon, good evening, and good night. May your supplement research stay evidence-based, your weight loss stay sustainable, and your health decisions stay informed.