Feb 27, 2026
You are throwing away potent semaglutide. Not on purpose. Not dramatically. But slowly, quietly, one degraded vial at a time. A vial left on the counter too long. A pen stored in the wrong spot. A reconstituted solution that sat three days past its window. Each mistake costs real money and real results. And the worst part? Most people do not even realize it is happening.
Semaglutide is not cheap. Whether you are using brand-name pens or compounded semaglutide from a pharmacy, every milligram matters. Waste a vial and you lose weeks of progress. Use degraded product and you wonder why the appetite suppression is not working, why the scale is not moving, why everything feels off. The answer often sits right there in your refrigerator, quietly losing potency while you assume it is fine.
Understanding semaglutide shelf life means understanding how this peptide degrades, what accelerates that process, and how to squeeze every drop of effectiveness from each vial or pen you purchase. It means knowing the difference between the manufacturer expiration date and the real-world window of maximum potency. It means recognizing that lyophilized powder, reconstituted solution, compounded liquid, and pre-filled pens all follow different timelines. Get this wrong and you are burning money. Get it right and you maximize every dose.
This guide covers every form of semaglutide, every storage scenario, every warning sign that something has gone bad. From the moment your semaglutide arrives to the moment you draw that final dose, you will know exactly how long you have and exactly how to protect what you have paid for. SeekPeptides has put together this comprehensive reference so you never have to guess about shelf life again.
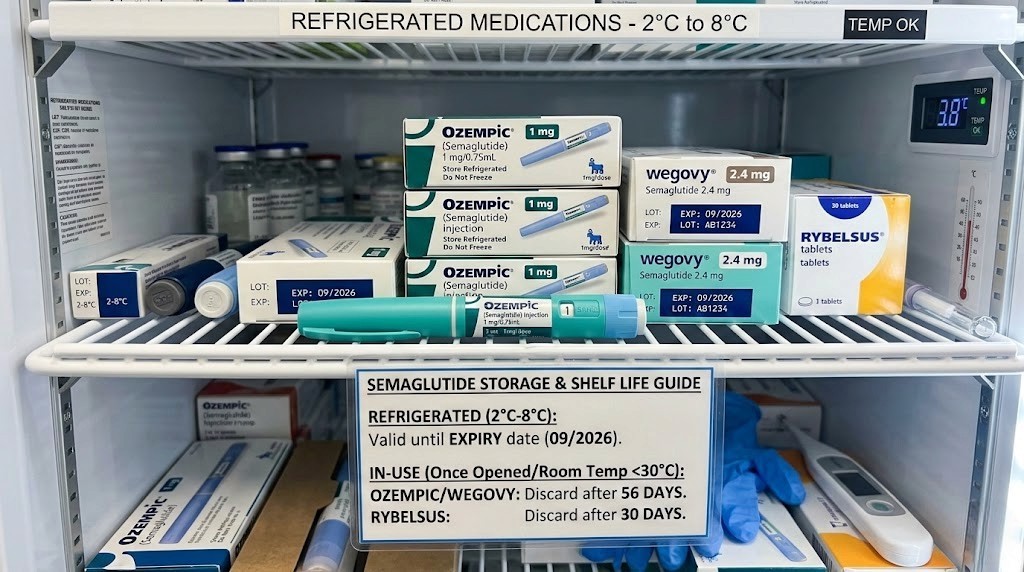
What determines semaglutide shelf life
Semaglutide is a peptide. That single fact governs everything about its shelf life. Peptides are chains of amino acids held together by chemical bonds that are sensitive to temperature, light, moisture, and bacterial contamination. Unlike a pill that sits stable in a bottle for years, semaglutide exists in a constant state of slow degradation from the moment it is manufactured. The question is never whether it will degrade. The question is how fast.
Several factors determine the rate of that degradation, and understanding them gives you control over how long your semaglutide actually lasts.
Temperature is the primary factor
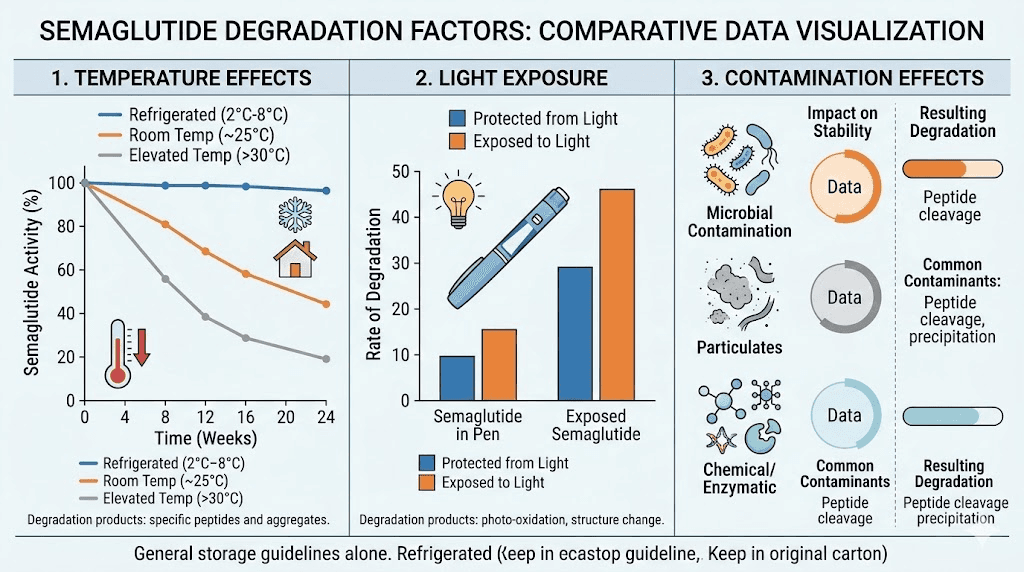
Heat accelerates every chemical reaction that breaks down peptide bonds. At refrigerator temperatures between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius), semaglutide degrades slowly. At room temperature, degradation speeds up significantly. At higher temperatures, like inside a hot car or near a window on a sunny day, the peptide can lose meaningful potency within hours. This is not theoretical. It is chemistry.
The relationship between temperature and degradation is not linear. A vial sitting at 86 degrees Fahrenheit does not degrade twice as fast as one at 43 degrees. It degrades far faster than that. Every 18 degree Fahrenheit increase roughly doubles the rate of chemical degradation for most peptides. So leaving your semaglutide in a warm environment for even a few hours can meaningfully reduce its effective shelf life.
Freezing creates a different problem entirely. When semaglutide solution freezes, ice crystals form within the liquid and physically damage the peptide molecules. They aggregate, clump together, and lose their biological activity. A frozen and thawed semaglutide pen or vial should be discarded. There is no recovering from freeze damage. The molecules have been structurally altered in ways that cannot be reversed by simply bringing the temperature back to normal.
Light exposure breaks peptide bonds
Ultraviolet light and even bright visible light can trigger photodegradation of semaglutide. This is why pens come in opaque packaging and why compounding pharmacies use amber vials. If you reconstitute semaglutide and store it in a clear vial on a brightly lit shelf, you are shortening its useful life with every passing hour. Keep it in the dark. The original packaging works. A closed refrigerator works even better.
Contamination starts a countdown
Every time a needle pierces a vial septum, bacteria have an opportunity to enter. This is why bacteriostatic water exists. The benzyl alcohol preservative in bacteriostatic water inhibits bacterial growth, extending the usable life of reconstituted peptides. Sterile water without preservatives offers no such protection. A vial reconstituted with plain sterile water should be used within 24 hours. A vial reconstituted with bacteriostatic water can last up to 28 days when properly refrigerated.
But bacteriostatic water is not magic. It slows bacterial growth. It does not stop it indefinitely. And it does nothing to prevent chemical degradation of the peptide itself. Even in bacteriostatic water, at perfect refrigerator temperature, in a dark vial, semaglutide is still slowly losing potency.
The form factor matters enormously
Lyophilized (freeze-dried) semaglutide powder is the most stable form. Remove the water and you remove the primary medium through which degradation reactions occur. Reconstituted semaglutide in solution is less stable. Compounded liquid semaglutide from a pharmacy follows its own rules based on the specific formulation. Pre-filled brand-name pens have been stability tested by the manufacturer and have clearly defined windows. Each form has a different shelf life, and confusing them is one of the most common and costly mistakes people make.
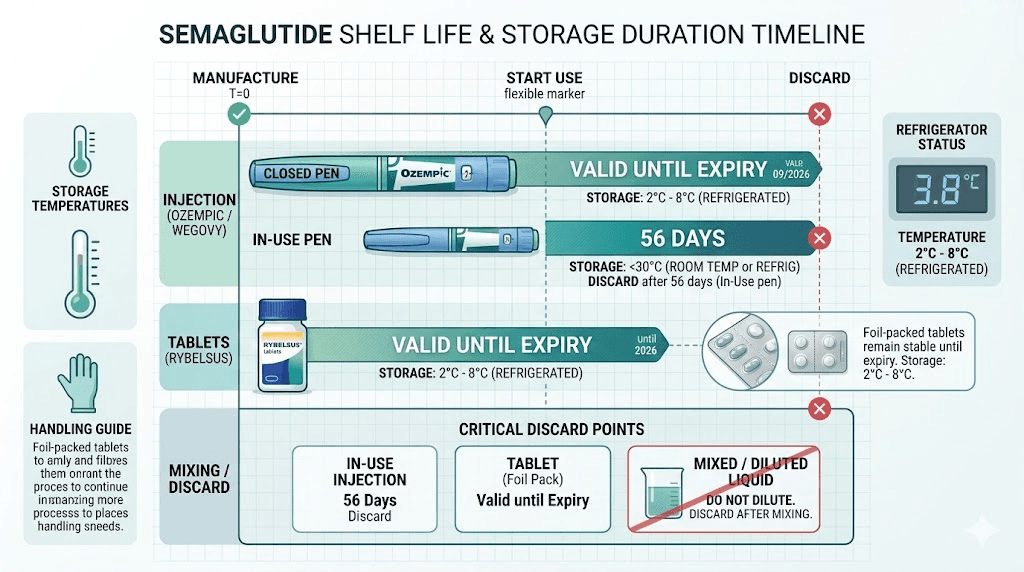
Unopened semaglutide shelf life by form
The shelf life of unopened semaglutide varies dramatically depending on what form you have. This distinction trips people up constantly. Someone reads that semaglutide lasts 36 months and assumes their compounded vial is good for three years. It is not. Someone else reads that reconstituted peptide only lasts 28 days and panics about their sealed pen. That is also wrong. Context matters.
Brand-name pens (Ozempic, Wegovy)
Ozempic and Wegovy pens, manufactured by Novo Nordisk, carry a shelf life of up to 36 months from the date of manufacture when stored in the refrigerator at 36 to 46 degrees Fahrenheit. This is the gold standard for semaglutide stability because these products undergo extensive stability testing, use pharmaceutical-grade preservatives, and are manufactured under strict Current Good Manufacturing Practice (cGMP) conditions.
The expiration date printed on the pen and its outer carton reflects this 36-month window. If your pen says it expires in March of next year, that date assumes proper refrigerated storage from the factory to your hands. If the pen was left on a loading dock in July heat for six hours, that expiration date no longer means what it says. You would never know.
One critical detail that catches people off guard: Wegovy pens are single-use. Each pen contains one dose. You inject it and throw it away, even if you can see liquid remaining. Ozempic pens are multi-dose and can be used multiple times over their in-use period. These are different products with different handling requirements, despite containing the same active ingredient. If you are comparing semaglutide versus tirzepatide, storage requirements are one factor worth examining for each.
Compounded liquid semaglutide
Compounded semaglutide is a different animal. These products come from compounding pharmacies, either 503A pharmacies that make patient-specific preparations or 503B outsourcing facilities that produce larger batches. The shelf life, called the Beyond-Use Date (BUD) in pharmacy terminology, varies significantly between providers.
A 503B facility with extensive stability testing data might assign a BUD of 90 to 120 days. A 503A pharmacy relying on default USP guidelines might assign a BUD of only 14 to 28 days. Same molecule. Wildly different shelf lives. The difference comes down to testing. Pharmacies that invest in stability studies can prove their formulation lasts longer and can label it accordingly.
When you receive compounded semaglutide, the BUD on the label is the number that matters. Not the 36-month figure from brand-name products. Not something you read online. The BUD on YOUR vial, from YOUR pharmacy, based on THEIR testing. This date typically ranges from 30 to 90 days for most compounded preparations, though some facilities with rigorous testing may extend it further.
Before using compounded semaglutide, understand the refrigeration requirements specific to your formulation. Some compounded formulations have different temperature tolerances than brand-name products.
Lyophilized semaglutide powder
Lyophilized semaglutide, the freeze-dried powder form used primarily in research settings, has the longest potential shelf life of any form. When stored desiccated (completely dry) at minus 20 degrees Celsius or colder, protected from light and moisture, lyophilized semaglutide can remain stable for months to years depending on the manufacturer and storage conditions.
At room temperature, sealed lyophilized powder remains stable for approximately three weeks. Refrigerated, it can last several months. Frozen at proper research temperatures, potentially much longer. The key factor is keeping it dry. The moment moisture reaches the powder, degradation begins. This is why these vials are often vacuum-sealed or packaged under inert gas.
Once you reconstitute lyophilized semaglutide, the clock starts ticking much faster. You have transitioned from the most stable form to one of the least stable. The shelf life drops from months or years down to days or weeks, depending on what you reconstitute with and how you store it.
Oral semaglutide (Rybelsus)
Oral semaglutide tablets have a shelf life determined by the manufacturer, typically printed on the blister pack or bottle. These tablets contain semaglutide co-formulated with SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate), an absorption enhancer. Store them at room temperature, below 86 degrees Fahrenheit, protected from moisture. Unlike injectable forms, these do not require refrigeration. The solid tablet form provides inherent stability that liquids lack.
After opening: the in-use shelf life
Opening a semaglutide pen or puncturing a vial septum starts a separate countdown. The unopened shelf life no longer applies. You are now working with the in-use shelf life, and this window is always shorter. Sometimes dramatically shorter.
Ozempic pen after first use
Once you use an Ozempic pen for the first time, it remains good for 56 days (8 weeks). During this 56-day window, you can store the pen either in the refrigerator or at room temperature up to 86 degrees Fahrenheit (30 degrees Celsius). After 56 days, discard the pen even if it still contains medication.
This 56-day window holds whether you used the pen once or twenty times. The clock starts on first use, not based on how many doses remain. If you only used half the pen in 56 days, you still throw it away. The stability data supports 56 days. Not 57. Not 60. Not however long you feel like pushing it. Trying to stretch this window is a false economy. You might save a few dollars on paper while injecting degraded medication that does not work properly, then wonder why you are not losing weight on semaglutide.
Wegovy pen after use
Wegovy pens are simpler because they are single-dose. You use the pen once and discard it. There is no multi-week in-use period to track. However, before that single use, the pen should have been stored in the refrigerator. Wegovy can also be kept at room temperature (up to 86 degrees Fahrenheit) for up to 28 days prior to use. After 28 days at room temperature, discard it even if the expiration date has not passed.
Compounded semaglutide vial after first puncture
This depends entirely on the formulation and the pharmacy. Generally, once you puncture the septum of a compounded semaglutide vial, the BUD on the label still applies, but contamination risk increases with each subsequent needle insertion. Some pharmacies specify a shorter in-use period after first puncture, separate from the overall BUD.
Best practice: note the date you first pierce the vial. If the pharmacy does not specify an in-use period, a conservative approach is to use the vial within 28 days of first puncture when stored refrigerated. Many people use their vials well within this window given typical semaglutide dosing schedules.
If you are drawing from the same vial over several weeks, use proper technique every time. Swab the septum with alcohol before each draw. Use a new needle. Do not touch the needle tip to anything before inserting it. Each clean draw extends the practical life of the vial. Each sloppy draw shortens it by introducing potential contaminants.
Reconstituted lyophilized semaglutide
Once reconstituted with bacteriostatic water, semaglutide solution should be refrigerated immediately and used within 28 days. With sterile water (no preservative), the window shrinks to 24 hours. Some researchers aliquot larger reconstitutions into smaller single-use volumes and freeze the extras at minus 20 degrees Celsius, thawing one at a time as needed. This approach extends total usable time while minimizing repeated freeze-thaw cycles.
The reconstitution process itself affects shelf life. Swirl the vial gently to dissolve the powder. Never shake it. Vigorous shaking can cause the peptide molecules to aggregate and lose biological activity. If you see foam or bubbles after mixing, you were too rough. Let it sit until the foam settles completely before using it or storing it. Proper reconstitution technique is not just about getting the right concentration. It is about preserving the peptide you just spent good money on.

Temperature storage guide: refrigerator, room temperature, and freezer
Temperature management is the single most impactful thing you can do to preserve semaglutide potency. Getting this right costs nothing. Getting it wrong costs everything.
Refrigerator storage (the gold standard)
Store semaglutide between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). This is standard refrigerator temperature. But where in the refrigerator matters more than you might expect.
The door is the worst spot. Every time you open the fridge, the door swings into room temperature air. Items in the door experience the most temperature fluctuation of anything in the refrigerator. Store semaglutide on a middle or upper shelf, toward the back, where temperature stays most consistent.
Avoid the back wall of the refrigerator too, especially in older models. The cooling element sits behind that wall, and items pressed against it can freeze. A frozen semaglutide vial is a ruined semaglutide vial. Leave a small gap between your medication and the back wall.
Consider purchasing a small refrigerator thermometer if you store expensive medications at home. Home refrigerators can vary more than you expect. The temperature setting on the dial is a suggestion, not a guarantee. Verify it with an actual thermometer. If you are spending hundreds on semaglutide each month, a five-dollar thermometer is the best investment you can make. For detailed guidance on how long semaglutide lasts in the fridge, the answer depends on the form but always starts with proper temperature control.
Room temperature storage
Sometimes refrigeration is not possible. You are traveling. You are at work. Your fridge broke at 2 AM. Knowing the room temperature limits prevents panic and prevents waste.
Ozempic pens can handle room temperature (up to 86 degrees Fahrenheit / 30 degrees Celsius) for up to 56 days after first use. Unused Wegovy pens can sit at room temperature for up to 28 days. Compounded semaglutide generally tolerates room temperature for much shorter periods, typically no more than 24 hours, though formulation-specific data may allow longer. If you accidentally left semaglutide out overnight, the brand-name pens are likely fine. Compounded preparations may not be.
If you are traveling with semaglutide, invest in a medication cooler bag with gel packs. These maintain refrigerator-range temperatures for 8 to 24 hours depending on the quality of the bag and ambient conditions. Never pack semaglutide in checked luggage where it could be exposed to extreme temperatures in the cargo hold. Carry it with you. The small inconvenience protects a significant investment.
Freezer storage: when it helps and when it destroys
This is where confusion thrives. For lyophilized (freeze-dried) powder, freezer storage is ideal. Store it at minus 20 degrees Celsius or colder, sealed and dry, and it remains stable for extended periods. This is the correct use of freezer storage for semaglutide.
For liquid semaglutide, whether in a pen, vial, or reconstituted solution, freezing is destructive. Period. Never freeze liquid semaglutide. The ice crystal formation damages peptide molecules irreversibly. A pen that was accidentally frozen and thawed might look exactly the same. The liquid might appear clear. But the molecules inside have been physically damaged in ways that reduce or eliminate biological activity. Throw it away.
The one exception involves intentional aliquoting of reconstituted solutions for research purposes. Some researchers reconstitute a large batch, divide it into single-use aliquots, and freeze them. Each aliquot gets thawed once and used immediately. This single freeze-thaw cycle causes less damage than multiple cycles. But it is still not ideal compared to using fresh reconstituted material, and each freeze-thaw event causes some degree of peptide degradation.
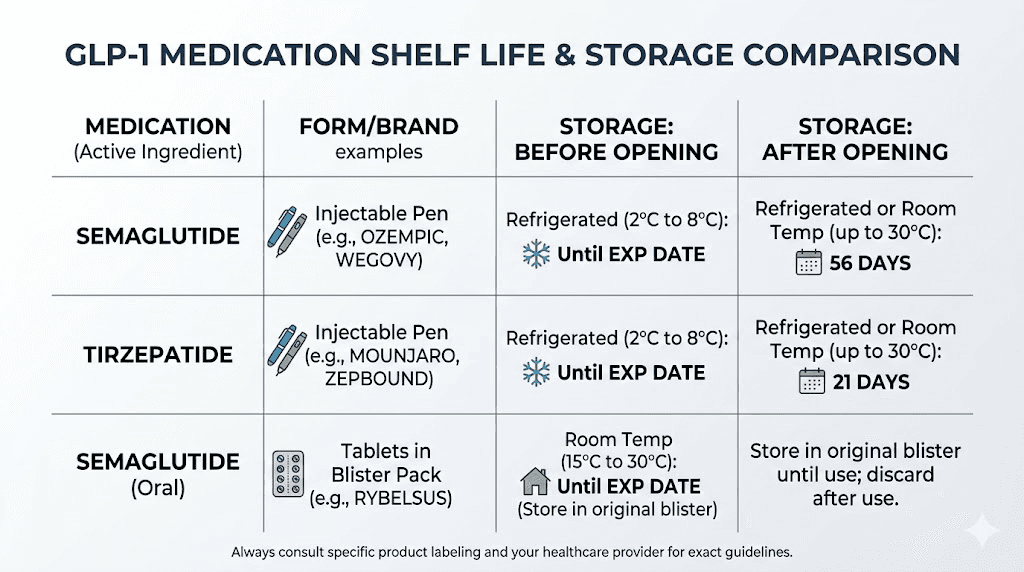
The semaglutide shelf life comparison table
Here is the reference table for every common semaglutide form:
Semaglutide Form | Unopened Shelf Life | Storage Condition | In-Use Period | Key Notes |
|---|---|---|---|---|
Ozempic pen | Up to 36 months | Refrigerated (36-46F) | 56 days after first use | Can store at room temp during in-use period |
Wegovy pen | Up to 36 months | Refrigerated (36-46F) | Single use, discard after | 28 days max at room temp before use |
Rybelsus tablets | Per manufacturer label | Room temp, below 86F | N/A (individual tablets) | Protect from moisture |
Compounded liquid (503B) | 90-120 days (BUD) | Refrigerated | Per pharmacy label | Stability-tested formulation |
Compounded liquid (503A) | 14-28 days (BUD) | Refrigerated | Per pharmacy label | Default USP guidelines apply |
Lyophilized powder | Months to years | Frozen (-20C) or refrigerated | 28 days reconstituted with bac water | 24 hours if using sterile water |
The numbers in this table represent ideal conditions. Deviation from proper storage reduces these windows. A pen that has been properly refrigerated its entire life may be fine at day 55 of its in-use period. A pen that spent a weekend in a hot car during shipping might be compromised on day one.
If you are calculating doses and want to ensure accuracy throughout the shelf life period, the semaglutide dosage calculator at SeekPeptides helps you plan exactly how much you need and how quickly you should use it.
How to tell if semaglutide has gone bad
Degraded semaglutide does not always announce itself. Sometimes the signs are obvious. Sometimes there are none. But knowing what to look for can save you from injecting compromised product and wondering why your results have stalled.
Visual inspection
Fresh semaglutide solution should be clear and colorless. Hold the vial or pen up to a light and look carefully. If you see any of the following, discard it immediately:
Cloudiness or haziness. Clear solution that has turned milky or cloudy indicates protein aggregation. The peptide molecules have clumped together. This is irreversible and the product is no longer effective. It is also a sign you should examine how you have been storing it, because something went wrong.
Particles or floaters. Any visible particles floating in the solution, settled at the bottom, or stuck to the glass indicate contamination or severe degradation. Do not use it. Do not try to filter it. Discard it.
Color changes. Semaglutide solution should be colorless. If it has turned yellow, pink, or any other color, degradation has occurred. For context on what normal looks like, check what color semaglutide should be. If yours is turning red or pink, that is a clear degradation signal.
Unusual odor. Open the cap and smell. Semaglutide solution should be essentially odorless. Any foul, sour, or unusual smell indicates bacterial contamination. Do not use it under any circumstances.
Performance signals
Sometimes degraded semaglutide looks perfectly fine but is not working anymore. If your semaglutide has been working well and you suddenly notice reduced appetite suppression, decreased weight loss momentum, or increased hunger despite no dose change, degradation is a possible cause. Before assuming your body has adapted, check your storage conditions and how long the current vial or pen has been open.
If you have been on a stable dose and experiencing good semaglutide results, a sudden drop in effectiveness paired with a vial nearing the end of its in-use period is a strong signal. Start a fresh vial and see if effectiveness returns.
This is especially relevant for anyone who has hit a semaglutide plateau. Before adjusting your protocol, before increasing your dose, make sure the product you are using is still potent. A degraded vial masquerades as a plateau, and increasing the dose of compromised product accomplishes nothing except burning through more degraded medication faster.
The injection site test
Fresh, potent semaglutide typically produces a slight burning or stinging sensation at the injection site, which resolves within minutes. If you have been experiencing this consistently and it suddenly disappears completely, the medication may have lost potency. This is not a definitive test, but it is one more data point. Pay attention to the injection site response as a subtle indicator of product quality.

Compounded semaglutide shelf life: the complete breakdown
Compounded semaglutide deserves its own detailed section because the rules are different, the variables are more complex, and the stakes are real. A growing number of people use compounded formulations, and understanding their shelf life requires understanding the compounding system itself.
503A versus 503B pharmacies
A 503A pharmacy compounds medications for individual patients based on a specific prescription. These are traditional compounding pharmacies. A 503B outsourcing facility produces larger batches under stricter manufacturing conditions similar to (but not identical to) pharmaceutical manufacturers. The distinction matters for shelf life because 503B facilities are required to conduct more rigorous stability testing.
When a 503B facility assigns a Beyond-Use Date of 90 or 120 days, that number is backed by actual laboratory testing of the specific formulation under defined storage conditions. When a 503A pharmacy assigns a BUD of 14 days, it may be using default USP (United States Pharmacopeia) guidelines rather than product-specific stability data. Both approaches are legal. Both produce safe products when followed. But the difference in assigned shelf life can be dramatic.
If shelf life matters to you (and it should, especially if you are buying in larger quantities to reduce cost), knowing whether your pharmacy is a 503A or 503B operation and understanding their stability testing methodology helps you make informed decisions. Longer BUDs mean less waste, which means more doses per dollar. For people using formulations like semaglutide with B12 or semaglutide with methylcobalamin, the additional ingredients can affect stability and BUD differently than plain semaglutide.
Factors that affect compounded semaglutide BUD
The concentration of semaglutide in the solution affects stability. Higher concentrations may degrade differently than lower concentrations. The pH of the solution matters. The specific buffer system used by the pharmacy matters. Whether preservatives are included and which ones. The type of vial and stopper material. The fill volume relative to the vial size (headspace affects oxygen exposure). Each of these variables influences how long the compounded product remains potent and safe.
This is why you cannot take the BUD from one pharmacy and apply it to another pharmacy's product. Even if both say "semaglutide 5mg/mL" on the label, the formulations may be different in ways that affect shelf life. Always follow the BUD on YOUR specific vial.
What the BUD label actually means
The Beyond-Use Date is the last date the pharmacy guarantees the product meets its labeled potency and sterility standards. After this date, the product has not necessarily gone bad. It simply has not been tested beyond that point, and the pharmacy cannot make guarantees about its quality.
Should you use compounded semaglutide past its BUD? No. Not because it instantly turns toxic at midnight on the BUD date. But because you are now in uncharted territory regarding potency and sterility. If your results become inconsistent, you would have no way to determine whether the problem is the medication, your body, or something else entirely. The small amount of money saved by stretching past the BUD is not worth the diagnostic confusion.
Read the compounded semaglutide expiration guide for more specific information about what happens as these products age and the specific signs to watch for with compounded formulations.
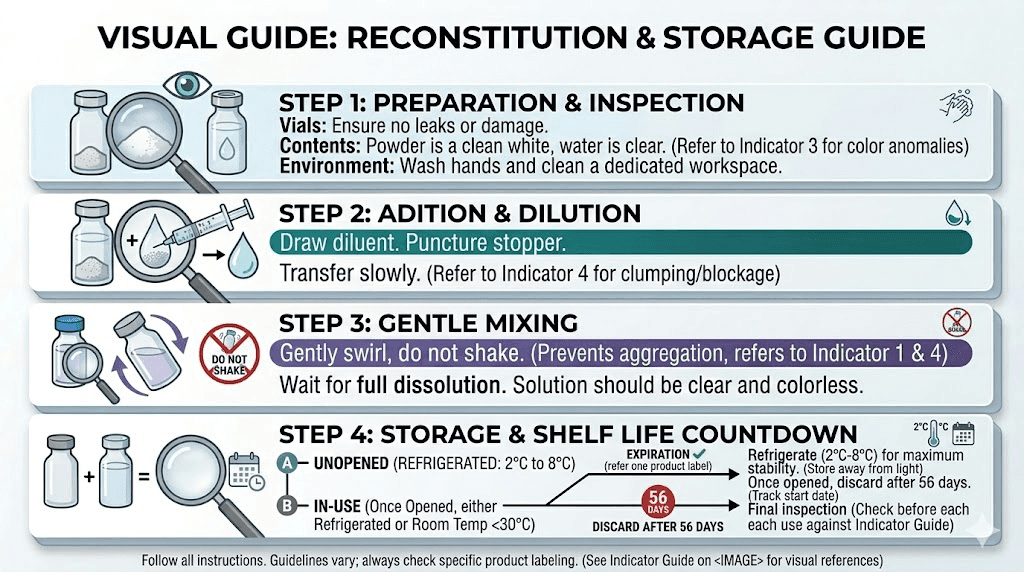
Reconstitution and shelf life: the critical connection
If you work with lyophilized semaglutide, reconstitution is the moment shelf life changes dramatically. Understanding this transition properly prevents the most common and costly storage mistakes.
Bacteriostatic water versus sterile water
This choice determines your post-reconstitution shelf life more than almost any other single decision.
Bacteriostatic water contains 0.9% benzyl alcohol, which inhibits bacterial growth. Reconstituted with bacteriostatic water and refrigerated, semaglutide typically remains usable for up to 28 days. This gives you a workable window for multi-dose use from a single reconstitution.
Sterile water contains no preservatives. Without anything to inhibit bacterial growth, a reconstituted vial becomes a race against contamination. Use it within 24 hours. If you reconstitute at 8 AM, plan to either use the entire vial by 8 AM the next day or discard whatever remains. There is no stretching this window safely.
For most people using reconstituted semaglutide over multiple doses, bacteriostatic water is the clear choice. The math supports it. A 5mg vial reconstituted with bacteriostatic water and used over four weekly doses wastes nothing. The same vial reconstituted with sterile water forces you to either use 5mg in a single day or throw away 75% of what you paid for.
The reconstitution process and shelf life
How you reconstitute affects how long the result lasts. Gentle swirling dissolves the powder while preserving peptide integrity. Vigorous shaking creates foam and can damage the molecules through a process called shear stress. This does not just affect the dose you are about to use. It affects every dose remaining in the vial.
The semaglutide mixing chart provides the proper ratios of water to powder for accurate concentration. Getting the ratio right means drawing accurate doses. But getting the technique right means those doses remain potent throughout the 28-day window. Both matter.
After reconstitution, immediately refrigerate the vial. Do not leave it on the counter while you prepare your injection or while you figure out where to store it. Every minute at room temperature in the initial hours after reconstitution matters more than you think, because the solution has not yet reached stable refrigerator temperature and degradation rates are at their highest.
Use the peptide reconstitution calculator to determine exactly how much bacteriostatic water to add for your desired concentration. Getting this step right from the beginning prevents dosing errors and wasted product throughout the entire shelf life window.
Semaglutide storage mistakes that shorten shelf life
Most shelf life problems are not caused by bad products. They are caused by good products stored badly. Here are the most common mistakes and what they actually cost you in terms of reduced potency and wasted medication.
Mistake 1: storing in the refrigerator door
The refrigerator door is the warmest, most temperature-variable spot in your entire refrigerator. Every time you open the door (and most people open their fridge 15 to 20 times per day), items stored in the door swing into room temperature air. Over the course of a day, a vial in the door experiences dozens of small temperature spikes. Over weeks, this cumulative exposure accelerates degradation significantly compared to a vial stored on a middle shelf toward the back.
Move your semaglutide off the door. Today. Put it on a shelf, behind the milk, where it stays cold and undisturbed. This single change could meaningfully extend the effective life of every vial you store.
Mistake 2: letting it warm up repeatedly
Some people take their semaglutide out of the fridge, draw their dose, and leave it on the counter while they inject. Then they put it back. Five minutes on the counter is fine. Thirty minutes starts to add up. An hour is problematic. And doing this weekly for a month means hours of cumulative room temperature exposure that the labeled shelf life did not account for.
The solution is simple. Take the vial out, draw your dose immediately, and put it back. Prepare your injection site and supplies before you touch the vial. Make the vial's time out of the fridge as short as physically possible. This is especially important for compounded semaglutide that may have shorter stability windows than brand-name products.
Mistake 3: not tracking dates
You opened the pen three weeks ago. Or was it four? Maybe five. You cannot remember, so you keep using it. This guesswork approach to shelf life management is incredibly common and incredibly wasteful in a different way. You might throw away a perfectly good pen because you are nervous, or you might keep using a compromised one because you cannot remember when you started.
Write the date on the pen or vial. Use a piece of tape and a marker. Write the date you opened it and the date it should be discarded. Remove all guesswork. This takes five seconds and eliminates one of the most common sources of semaglutide waste and uncertainty.
Mistake 4: buying more than you can use in time
Bulk purchasing compounded semaglutide to save money only saves money if you can use it before the BUD expires. If the BUD is 30 days and you use one dose per week, one multi-dose vial is probably fine. But buying three months worth means two of those vials might expire before you get to them.
Calculate your usage rate against the BUD before purchasing. Factor in your dosing schedule, the concentration per vial, and how many doses each vial provides. Then buy accordingly. The cheapest semaglutide is the semaglutide you actually use at full potency, not the semaglutide you bought at a discount and threw half away.
Mistake 5: using a vial after suspected freezing
Your refrigerator was turned up too high. You found ice crystals in the lettuce drawer. Your semaglutide vial feels suspiciously cold. The temptation is to thaw it and use it anyway because replacing it costs money and time.
Resist that temptation. If there is any chance your semaglutide froze, even partially, discard it. Frozen and thawed peptide solutions may look completely normal while being biologically inactive. You would inject it, wait a week, notice nothing happening, blame your body or your protocol, adjust your approach, waste another week, and eventually figure out the vial was bad all along. The total cost of that diagnostic odyssey far exceeds the cost of one replacement vial.
Shelf life and the semaglutide dosage connection
Your dose size and frequency directly affect how much shelf life matters to you. Someone on a high dose who goes through a vial quickly faces fewer storage challenges than someone on a low dose who needs the same vial to last a month.
Low-dose users: maximum shelf life exposure
If you are on a starting dose of 0.25mg per week, a single 5mg vial contains roughly 20 weeks of medication. But the vial only lasts 28 days after reconstitution. This is a fundamental math problem that many people do not discover until they have already reconstituted. You will waste the majority of a vial unless you plan ahead.
Solutions include: reconstituting with less bacteriostatic water to create a higher concentration (smaller volume per dose, same mg delivered), using the semaglutide 5mg dosage chart to plan exact draw volumes, or purchasing smaller vials (like 2mg or 3mg) that better match your monthly usage at lower doses. The goal is to use the entire vial within its shelf life window so nothing goes to waste.
Understanding the exact conversion between semaglutide units and milligrams helps you calculate precisely how many doses a vial provides at your specific dose. From there, you can determine whether the vial will be used up before its shelf life expires.
High-dose users: less waste, different concerns
If you are on 2.4mg per week, you will go through a 5mg vial in about two weeks. Shelf life is less of a concern because the vial is empty before degradation becomes significant. Your primary concerns shift to proper storage during those two weeks and ensuring you have your next vial ready when this one runs out.
High-dose users sometimes face supply concerns. If you are using Empower pharmacy semaglutide or Olympia pharmacy semaglutide, order timing matters. Running out and waiting for a new shipment means interrupted treatment. Maintaining a small buffer supply (one extra vial in the fridge) prevents gaps, as long as you track the BUD on that buffer vial and rotate stock properly. First in, first out. Always use the oldest vial first.
Dose escalation and shelf life planning
Most semaglutide protocols involve gradual dose increases. You start at 0.25mg, increase to 0.5mg after four weeks, then to 1mg, and potentially higher. This means your consumption rate changes over time, which means your shelf life calculations change too.
At 0.25mg per week, you might struggle to use a 5mg vial before it expires. At 1mg per week, that same vial lasts five weeks, still within the 28-day reconstitution window. At 2.4mg per week, you need a new vial every two weeks. Plan your purchases around your current dose, not your future dose. Buying ahead for a dose you have not reached yet creates shelf life risk if the dose increase gets delayed or adjusted.
For a complete dosing reference across different concentrations, the semaglutide dosing chart in units provides draw volumes at each dose level. Cross-reference that with your vial size and the 28-day post-reconstitution window to plan purchases that minimize waste.
Traveling with semaglutide: protecting shelf life on the go
Travel is where semaglutide shelf life gets put to the real test. Controlled refrigerator temperatures give way to hotel mini-fridges, airport security lines, and suitcases in overhead bins. A few hours of poor temperature control during travel can undo weeks of careful storage at home.
Short trips (1-3 days)
For short trips, a quality medication cooler bag with gel packs is sufficient. Pre-chill the gel packs thoroughly before packing. A good cooler bag maintains refrigerator-range temperatures for 8 to 12 hours, sometimes longer depending on ambient conditions and how often you open it.
Brand-name pens are the easiest for short travel because they tolerate room temperature for extended periods during their in-use phase. Compounded semaglutide requires more careful temperature management due to typically shorter room-temperature windows.
Longer trips (4+ days)
Longer trips require access to reliable refrigeration at your destination. Most hotels have mini-fridges, but their temperature control can be inconsistent. If possible, set the mini-fridge to its coldest setting and place the semaglutide in the back where temperature is most stable. Verify with a thermometer if you brought one.
Some travelers use portable medication coolers with active cooling (battery-powered or USB-charged) for trips where passive gel packs would not last. These are more expensive but provide guaranteed temperature control for days rather than hours. If you travel frequently and use temperature-sensitive medications, the investment pays for itself quickly in preserved medication.
Read the complete guide to traveling with semaglutide for detailed packing lists, TSA guidelines, and destination-specific storage solutions.
Air travel specifics
Keep semaglutide in your carry-on luggage, never in checked bags. Cargo holds can reach extremely cold temperatures at altitude (potentially freezing your medication) or extremely hot temperatures during ground operations. Neither is acceptable. TSA allows injectable medications in carry-on bags. Bring the medication in its original packaging with the prescription label visible. A letter from your prescriber can smooth things over if questions arise, though it is rarely needed.
Syringes and needles are allowed in carry-on when accompanied by injectable medication. If you carry injection supplies, pack them neatly in a clear bag alongside the medication to make security screening easy and quick.
Does semaglutide expire, and what happens if it does
Yes. All semaglutide products expire. The expiration date on brand-name products represents the manufacturer-tested window of guaranteed potency and safety. The BUD on compounded products serves the same purpose from the pharmacy. Passing either date does not mean the semaglutide instantly becomes dangerous. It means nobody can guarantee it still works properly.
What happens to expired semaglutide
After expiration, semaglutide gradually loses potency. The rate depends on how it was stored. A pen that expired yesterday after two years of proper refrigeration probably still contains highly active semaglutide. A pen that expired last month and spent weeks in a warm kitchen? Much less certainty.
The bigger risk with significantly expired semaglutide is not toxicity. It is contamination. Over time, the preservatives in multi-dose products become less effective. Bacterial contamination becomes more likely, especially if the product has been punctured multiple times. Injecting contaminated solution can cause infections ranging from mild injection site reactions to serious systemic infections requiring medical treatment.
If you are curious about using semaglutide slightly past its date, read the detailed analysis on whether you can use expired semaglutide. For situations where expired product was already used, the guide on accidentally taking expired semaglutide covers what to watch for and when to seek medical attention.
The 28-day rule for semaglutide
You may have heard that semaglutide expires after 28 days. This is a partial truth that causes significant confusion. The 28-day figure applies to specific situations, not universally.
Reconstituted semaglutide with bacteriostatic water: 28 days refrigerated. Wegovy pens stored at room temperature: 28 days maximum. Some compounded formulations: 28-day BUD depending on the pharmacy.
But Ozempic pens have a 56-day in-use period. Unopened pens last up to 36 months. Lyophilized powder lasts much longer than 28 days. The 28-day rule applies to specific forms in specific conditions, not to all semaglutide everywhere. Check what applies to YOUR product. The semaglutide 28-day expiration guide breaks down exactly when this timeline applies and when it does not.
Semaglutide shelf life versus other GLP-1 medications
If you are choosing between GLP-1 options and shelf life is a practical consideration (it should be), here is how semaglutide stacks up against alternatives.
Semaglutide versus tirzepatide shelf life
Tirzepatide shelf life follows similar patterns to semaglutide. Both are peptide-based GLP-1 receptor agonists that require refrigeration and have defined in-use periods. Brand-name tirzepatide (Mounjaro, Zepbound) pens have manufacturer-specified shelf lives and in-use periods comparable to Ozempic and Wegovy. Compounded tirzepatide follows BUD rules similar to compounded semaglutide.
The primary differences come down to specific formulations. Tirzepatide and semaglutide are different molecules with different stability profiles, which means the exact shelf life numbers can differ even under identical storage conditions. If you are switching between the two, do not assume the storage rules from one apply perfectly to the other. Check the specifics for each.
For a deeper comparison of how these medications differ beyond storage, the semaglutide versus tirzepatide comparison covers efficacy, side effects, dosing, and more. Both medications face similar practical challenges around fridge storage duration and refrigeration requirements.
Semaglutide versus retatrutide shelf life
Retatrutide, the triple-agonist GLP-1/GIP/glucagon receptor agonist currently in clinical trials, is primarily available in research settings as lyophilized powder. Its shelf life follows the general rules for lyophilized peptides: stable for extended periods when stored frozen and dry, limited shelf life once reconstituted. As retatrutide is not yet commercially available in pre-filled pen form, direct shelf life comparisons with brand-name semaglutide pens are not applicable.
Oral versus injectable shelf life considerations
Oral semaglutide (Rybelsus) has the simplest storage requirements of any semaglutide form. Tablets stored at room temperature, protected from moisture. No refrigeration needed. No reconstitution. No 28-day countdown. If storage simplicity and shelf life convenience matter to you, the injectable versus oral peptide comparison explores the broader trade-offs between these delivery methods.
Maximizing semaglutide shelf life: the complete protocol
Everything above distills into a practical protocol for getting the most out of every vial, pen, or tablet you purchase. Follow these steps consistently and you eliminate the most common sources of waste and potency loss.
Before purchase
Calculate your monthly usage based on your current dose. Use the semaglutide dosage calculator if needed. Compare your monthly usage against the product's shelf life or BUD. Buy only what you can use within the labeled timeframe. If using a compounding pharmacy, ask about their stability testing and BUD assignment methodology. A pharmacy with 90-day BUD testing saves you money compared to one with 28-day BUDs, assuming equivalent quality and pricing.
Upon receiving
Refrigerate immediately upon receipt. Check the expiration date or BUD. If the product was shipped, assess the cold chain. Was the cold pack still cold? Were the temperature indicators (if included) still within range? If the packaging arrived warm or the cold packs were completely melted, contact the supplier before using the product. Document the date received and the labeled expiration or BUD.
During storage
Store on a middle shelf toward the back of the refrigerator. Not in the door. Not against the back wall. Check refrigerator temperature periodically with a thermometer (aim for 36-46 degrees Fahrenheit). Keep the vial or pen in its original packaging or in an opaque container to protect from light. Do not move the product in and out of the refrigerator unnecessarily.
During use
Write the first-use date on the product with a marker. Minimize time out of the refrigerator when drawing doses. Swab vial septums with alcohol before each draw. Use a new needle each time. Swirl reconstituted solutions gently, never shake them. Return to refrigerator immediately after drawing your dose.
If you are using compounded semaglutide alongside supplements, timing and storage considerations may differ. Guides on combining semaglutide with glycine, semaglutide with B12, or other compounds each have their own storage nuances worth understanding.
Disposal
Discard on or before the expiration date, BUD, or in-use period end date, whichever comes first. Discard immediately if you observe any visual changes (cloudiness, particles, color change, odor). Discard if freezing is suspected. Do not donate, share, or save expired medication for future use. Dispose of sharps properly in an approved sharps container.
Semaglutide shelf life and cost optimization
Shelf life directly affects the cost-per-dose of semaglutide. Waste is a hidden cost that most people never calculate. Let us change that.
The real cost of waste
Assume a 5mg compounded semaglutide vial costs $150 and carries a 30-day BUD. If you are on 0.5mg per week, you use 2mg in 28 days, leaving 3mg unused and discarded. You paid $150 but used $60 worth of medication. Your effective cost per milligram just increased by 150%.
Now compare that to a pharmacy with a 90-day BUD. Same vial, same price. At 0.5mg per week, you use the entire 5mg within 10 weeks (70 days). But the BUD is 90 days, so you have a comfortable margin. Zero waste. You paid $150 for $150 worth of medication. Your cost per milligram stays at the labeled rate.
This is why shelf life should factor into your purchasing decisions alongside price per milligram, shipping costs, and product quality. The cheapest per-milligram price means nothing if half the vial ends up in the trash. Use the peptide cost calculator to factor waste into your true cost analysis.
Strategies for minimizing waste
Match vial size to dose. Lower doses need smaller vials. Higher doses can handle larger vials. Do the math before ordering.
Coordinate reconstitution timing. Do not reconstitute until you are ready to start using the vial. Every day between reconstitution and first use is a day of shelf life burned for nothing.
Consider concentration adjustments. Higher concentrations mean smaller draw volumes per dose, which can reduce the number of needle punctures per vial and slightly reduce contamination risk. Use the semaglutide 5mg mixing chart or the 10mg mixing chart to find the concentration that balances accurate dosing with practical shelf life considerations.
Rotate stock properly. If you keep a backup vial, always use the older one first. First in, first out. Label everything with receipt dates and BUDs.
Frequently asked questions
How long does semaglutide last unopened in the refrigerator?
Brand-name semaglutide pens (Ozempic, Wegovy) last up to 36 months unopened in the refrigerator at 36-46 degrees Fahrenheit. Compounded semaglutide lasts according to the Beyond-Use Date on the label, typically 30-120 days depending on the pharmacy. Semaglutide storage duration depends entirely on the product form and storage conditions.
Can I use semaglutide after the expiration date?
It is not recommended. After expiration, potency decreases and contamination risk increases. While slightly expired semaglutide is unlikely to be dangerous, you cannot guarantee it still works effectively. Read the full guide on using expired semaglutide for a detailed risk analysis.
How long does reconstituted semaglutide last?
With bacteriostatic water, up to 28 days refrigerated. With sterile water (no preservatives), use within 24 hours. Always refrigerate immediately after reconstitution and follow proper reconstitution technique to maximize the usable window.
What happens if semaglutide gets warm?
Brief exposure to room temperature (under 30 minutes) is generally fine for most forms. Extended warmth accelerates degradation. If semaglutide was exposed to temperatures above 86 degrees Fahrenheit for more than a few hours, potency loss is likely. Check the warm exposure guide for specific scenarios and recommendations.
Can I freeze semaglutide to make it last longer?
Only lyophilized (freeze-dried) powder benefits from freezer storage. Never freeze liquid semaglutide, whether in pens, vials, or reconstituted solution. Freezing destroys the peptide molecules through ice crystal formation. This damage is irreversible even after thawing.
How do I know if my semaglutide has gone bad?
Check for cloudiness, particles, color changes, or unusual odor. Also monitor for sudden loss of effectiveness at a previously working dose. Clear solution does not guarantee potency, but visible changes always indicate a problem. If the color of your semaglutide has changed from clear and colorless, discard it.
Does the 28-day rule apply to all semaglutide?
No. The 28-day figure applies to reconstituted semaglutide with bacteriostatic water and some compounded formulations. Ozempic pens have a 56-day in-use window. Unopened brand-name pens last up to 36 months. Check the 28-day expiration guide for specifics on which forms this applies to.
Is compounded semaglutide shelf life shorter than brand-name?
Generally yes. Brand-name products have extensive manufacturer stability testing supporting long shelf lives. Compounded products have pharmacy-determined BUDs that are typically shorter. However, well-tested 503B formulations can have BUDs of 90-120 days, which is adequate for most usage patterns.
External resources
For researchers serious about protecting their investment and optimizing their protocols, SeekPeptides provides comprehensive storage guides, stability databases, and handling protocols that help members get the most out of every vial. Whether you are navigating compounded formulations, planning dose escalations, or troubleshooting potency concerns, SeekPeptides members access the resources and community support to handle semaglutide with confidence.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your refrigerator stay calibrated, and your shelf life calculations stay accurate.