Mar 15, 2026

The outcomes could not be more different. Injectable semaglutide has years of clinical trials behind it, thousands of published studies, and FDA approval for both diabetes management and weight loss. The semaglutide troche, by contrast, sits in a different category entirely. It is a compounded formulation. It has no FDA approval. And it has almost no human clinical data supporting its specific delivery method.
Yet people are choosing it anyway. In growing numbers.
Why? Because needles are a barrier. A real one. The idea of weekly self-injection stops a significant percentage of candidates from ever starting a semaglutide protocol in the first place. Fear. Inconvenience. Travel logistics. The simple discomfort of pushing a needle into your own skin every seven days. These are not trivial objections, and dismissing them ignores the reality of human behavior.
The semaglutide troche promises a solution. A small medicated lozenge, placed under the tongue or between the cheek and gum, that dissolves slowly and delivers the same GLP-1 receptor agonist without a single needle. It sounds almost too good to be true. And in some ways, it is, because the science behind sublingual peptide delivery is more complicated than most marketing materials suggest.
This guide covers everything you need to know. What a semaglutide troche actually is. How sublingual absorption works at the molecular level. What the research says about bioavailability compared to injections. Proper dosing protocols. Side effects. Cost considerations. Regulatory status. And the critical questions you should ask before choosing this route over compounded injectable semaglutide or the FDA-approved alternatives. SeekPeptides has compiled the most current evidence available so you can make an informed decision rather than a hopeful one.
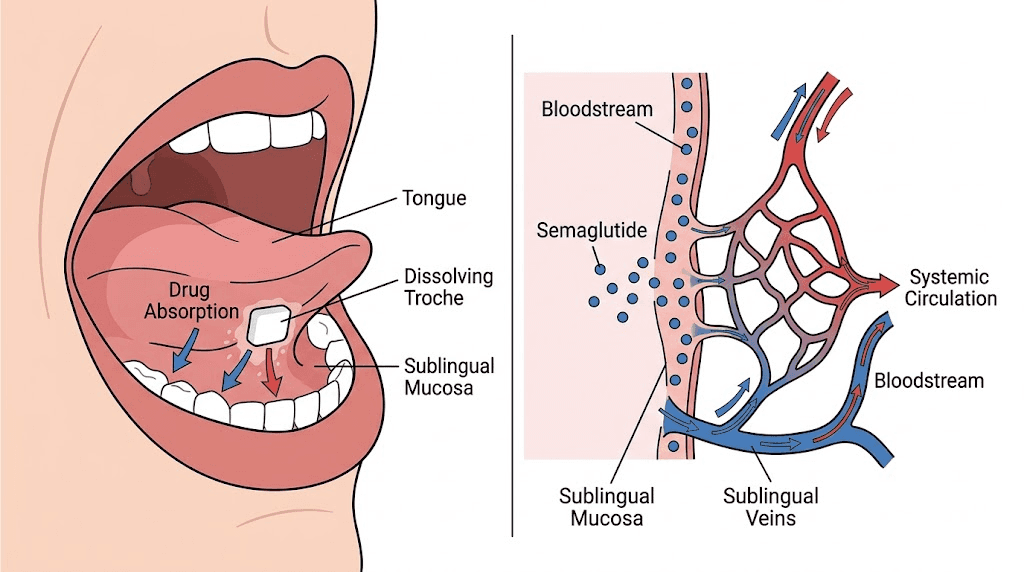
What is a semaglutide troche
A troche is a medicated lozenge. The word itself comes from the Greek "trokhiskos," meaning a small wheel or disk. In modern pharmacy, a troche refers to a compounded medication shaped into a small, flat tablet designed to dissolve slowly in the mouth. It is not swallowed whole like a pill. It is not chewed. It sits against the mucosal tissue inside your mouth and releases its active ingredient gradually.
A semaglutide troche contains the same active compound found in Ozempic, Wegovy, and Rybelsus. Semaglutide is a GLP-1 receptor agonist, a synthetic analog of the naturally occurring glucagon-like peptide-1 hormone. It binds to GLP-1 receptors throughout the body, primarily in the brain and gastrointestinal tract, to reduce appetite, increase feelings of fullness, and slow gastric emptying.
The difference is delivery.
Instead of subcutaneous injection, the troche delivers semaglutide through the oral mucosa. This is a fundamentally different absorption pathway from both injectable semaglutide and the oral tablet form (Rybelsus). When a troche dissolves under your tongue or against your cheek, the medication passes directly through the thin mucosal membrane and enters the bloodstream without first traveling through the digestive system.
This matters because peptides are fragile. They are chains of amino acids. The digestive tract is specifically designed to break amino acid chains apart, which is why oral bioavailability of most peptides hovers near zero.
Rybelsus solves this problem with a special absorption enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate) that temporarily makes the stomach lining more permeable. Even with SNAC, oral semaglutide achieves only about 1% bioavailability.
One percent. That means 99% of the semaglutide you swallow in a Rybelsus tablet never reaches your bloodstream.
The troche attempts a different workaround. By keeping the medication in contact with the sublingual or buccal mucosa, it bypasses the stomach entirely. No gastric acid degradation. No first-pass liver metabolism. The medication enters the bloodstream through the rich network of capillaries beneath the tongue and along the inner cheek. At least, that is the theory. The practical reality involves several important caveats that anyone considering this delivery method needs to understand.
How troches differ from sublingual drops
Semaglutide troches and sublingual semaglutide drops share the same basic concept: deliver semaglutide through the oral mucosa rather than through injection or the digestive tract. But the formulations differ in meaningful ways.
Drops are liquid. They are placed under the tongue and absorbed quickly, usually within 30-60 seconds. The contact time with mucosal tissue is relatively brief because saliva dilutes the liquid and triggers the swallowing reflex. Troches, by contrast, dissolve slowly over 5-15 minutes. This extended contact time theoretically allows more medication to cross the mucosal barrier before being swallowed.
Troches also typically contain additional ingredients that drops do not. Most semaglutide troches include vitamin B12 (cyanocobalamin or methylcobalamin), which serves dual purposes. B12 itself is well-absorbed sublingually, so it provides genuine nutritional supplementation. It also helps mask the taste of the semaglutide compound. Some troches contain flavoring agents, sweeteners, and binding agents that hold the lozenge together during the dissolution process.
The practical trade-off is convenience versus absorption consistency. Drops are faster to administer but harder to keep in contact with the mucosa. Troches take longer but maintain more consistent mucosal contact. Neither form has been subjected to rigorous clinical trials comparing them to each other or to injectable formulations.
The compounding pharmacy connection
Semaglutide troches are not manufactured by Novo Nordisk, the pharmaceutical company that produces Ozempic, Wegovy, and Rybelsus. They are created by compounding pharmacies, which are specialized facilities that mix medications in custom formulations based on individual prescriptions.
Compounding pharmacies have existed for centuries. They fill an important gap when commercially available medications do not meet a specific patient need, whether that involves a different dosage form, the removal of an allergen, or a combination of active ingredients. The compounding of semaglutide became particularly widespread during the FDA-declared shortage of injectable semaglutide products, which allowed compounders to legally produce semaglutide formulations under Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act.
This distinction matters. A compounded semaglutide troche is not a generic version of Ozempic or Rybelsus. It is a custom-prepared medication that uses the same active ingredient but may differ in purity, potency, absorption characteristics, and shelf stability. The FDA does not verify the safety or effectiveness of compounded medications the way it does for commercially manufactured drugs. Quality depends entirely on the compounding pharmacy, its facilities, its processes, and its quality control measures.
How semaglutide troches work at the molecular level
Understanding why a semaglutide troche might or might not work requires a brief look at the biology of mucosal absorption. The lining of your mouth is not just a passive barrier. It is a dynamic tissue with specific properties that determine what can and cannot pass through it.
Sublingual and buccal absorption pathways
The floor of the mouth, the area under your tongue, is covered by a thin, non-keratinized epithelium. Non-keratinized means it lacks the tough protective layer found on the roof of the mouth and the outer skin. This thinner tissue allows molecules to pass through more easily. Beneath this epithelium sits a dense network of blood vessels that feeds into the sublingual vein, which drains into the internal jugular vein and then directly to the heart. Medication absorbed here enters systemic circulation rapidly, bypassing both the intestinal wall and the liver.
Buccal absorption, through the cheek lining, works similarly but through slightly thicker tissue. The buccal mucosa is partially keratinized, which means absorption is somewhat slower and less efficient than the sublingual route. However, the cheek provides a larger surface area, and a troche placed between the cheek and gum can remain in contact with the tissue for longer without being dislodged by tongue movement.
For small molecules like nitroglycerin or certain hormones, sublingual absorption is well-established and highly effective. These molecules are small enough to pass through the mucosal cells via passive diffusion. But semaglutide is not a small molecule.
The size problem with peptide absorption
Semaglutide has a molecular weight of approximately 4,114 daltons. That makes it a large peptide, significantly bigger than the small molecules that absorb well sublingually. For reference, nitroglycerin has a molecular weight of 227 daltons. Testosterone is 288 daltons. Even most sublingual medications fall below 500 daltons.
Molecular weight matters because larger molecules have a harder time crossing biological membranes through passive diffusion. The mucosal epithelium acts as a selective barrier, and while it allows small lipophilic molecules through relatively easily, large hydrophilic peptides face significant resistance. Semaglutide has to navigate through or between epithelial cells, and its size makes both routes challenging without assistance.
This is the fundamental scientific question surrounding semaglutide troches: can a 4,114-dalton peptide achieve meaningful absorption through the oral mucosa?
A recent preclinical study published in 2025 offers some initial answers. Researchers administered sublingual semaglutide to rats and measured pharmacokinetic parameters. The results showed that sublingual delivery did produce measurable plasma semaglutide concentrations, and systemic exposure was higher than equivalent oral doses that had to pass through the GI tract.
However, animal pharmacokinetics do not always translate directly to humans, and the study used permeation enhancers that may not be present in all commercial troche formulations.
The first formal clinical evaluation of sublingual semaglutide in humans, a Phase 1 trial by Aspire Biopharma, is planned for mid-2026. Until those results are available, the actual bioavailability of semaglutide troches in humans remains uncertain. This uncertainty is perhaps the most important thing to understand about this delivery method. It might work very well. It might work poorly. The data to answer that question definitively does not yet exist in published human studies.
The GLP-1 mechanism once absorbed
Regardless of delivery method, once semaglutide reaches the bloodstream, it works the same way. It is a GLP-1 receptor agonist with a half-life of approximately 165 hours (about seven days), which is why the injectable form is dosed weekly.
Semaglutide binds to GLP-1 receptors in several key locations. In the hypothalamus, it activates satiety centers that reduce hunger and food cravings. In the gastrointestinal tract, it slows gastric emptying, which means food stays in the stomach longer and you feel full after eating less. In the pancreas, it stimulates glucose-dependent insulin secretion and suppresses glucagon release, which improves blood sugar control.
The appetite suppression effect is what most people seeking weight loss care about. When semaglutide suppresses appetite, it does so by acting on the brain directly, not just by making the stomach feel full. This central mechanism is why semaglutide produces more weight loss than older approaches that relied solely on gut-level satiety signals. Clinical trials of injectable semaglutide for weight loss showed average weight reductions of 12-15% of body weight over 68 weeks, with some participants losing significantly more.
The question for troche users is not whether semaglutide works. It does. The question is whether enough semaglutide gets absorbed through the troche to reach the therapeutic concentrations needed for these effects. And that brings us to dosing.
Semaglutide troche dosage and titration protocol
Dosing a semaglutide troche is fundamentally different from dosing an injection, and understanding why requires knowing how bioavailability affects effective dosing.
Why troche doses are higher than injection doses
With injectable semaglutide, nearly 100% of the administered dose reaches the bloodstream. The standard injectable semaglutide dosing schedule for weight loss starts at 0.25 mg per week and titrates up to 2.4 mg per week over 16-20 weeks. These are the doses that produced the weight loss results seen in clinical trials.
With a troche, bioavailability is lower. How much lower is uncertain, but it is definitely less than 100%. If sublingual absorption achieves, say, 10-20% bioavailability (which would be considered quite good for a peptide this size), then you would need a substantially higher nominal dose to achieve the same plasma concentrations as an injection.
This is why compounding pharmacies typically formulate semaglutide troches at daily doses of 0.5-2 mg rather than the 0.25-2.4 mg weekly doses used for injections. The math is straightforward: if you need 2.4 mg per week from an injection, and sublingual absorption is only 15% efficient, you would theoretically need roughly 16 mg per week sublingually, or about 2.3 mg per day, to achieve equivalent systemic exposure.
But here is the problem. Nobody knows the actual bioavailability with precision. Different compounding pharmacies use different formulations. Different patients produce different amounts of saliva, hold the troche for different durations, and have different mucosal tissue characteristics. The dose-response relationship for sublingual semaglutide has not been established in controlled human studies.
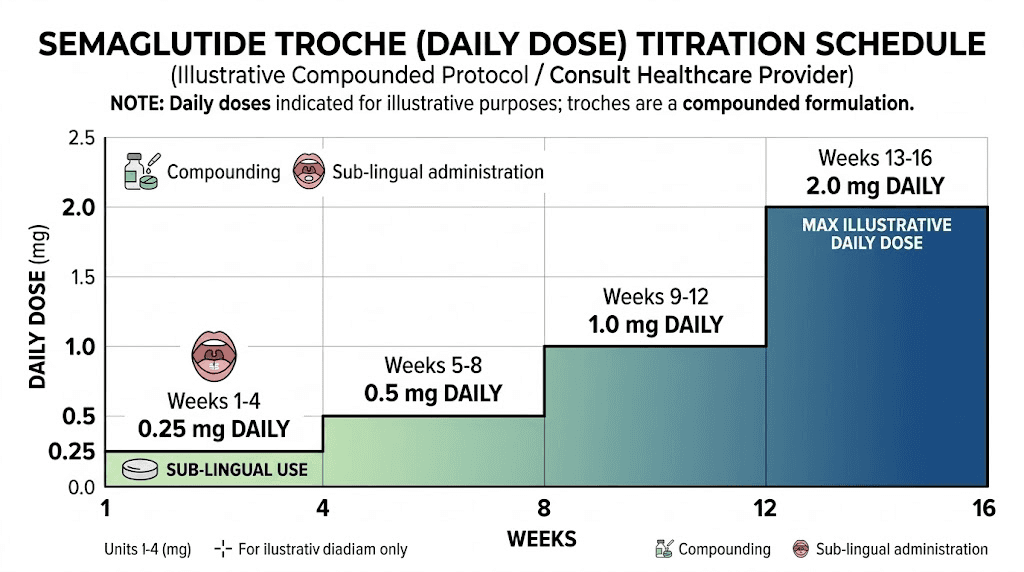
Typical titration schedules from compounding pharmacies
Most compounding pharmacies and prescribing clinicians follow a gradual titration approach for semaglutide troches, similar in principle to the injectable titration schedule but adapted for daily dosing:
Weeks 1-4: 0.25 mg to 0.5 mg daily. This is the assessment phase. The goal is not weight loss but tolerance evaluation. Starting low minimizes gastrointestinal side effects, which are the most common reason people stop semaglutide therapy early.
Weeks 5-8: 0.5 mg to 1.0 mg daily. If the initial dose was tolerated well, the prescriber increases to a moderate dose. Some patients begin experiencing appetite suppression at this level.
Weeks 9-12: 1.0 mg to 1.5 mg daily. This is where many patients reach a therapeutically effective dose range. Appetite suppression becomes more noticeable, and early weight changes may appear.
Weeks 13 and beyond: 1.5 mg to 2.0 mg daily, or higher based on response. The maximum dose varies by compounding pharmacy and prescriber, but most protocols cap at 2.0 mg to 3.0 mg daily.
Some clinicians prefer even more conservative starting doses, beginning at 0.25 mg daily and increasing by 0.25 mg every two to four weeks. This ultra-gradual approach reduces the risk of severe nausea but extends the time to reach a therapeutic dose.
If you are tracking your semaglutide dosage, understanding the difference between injectable units and troche milligrams is essential. The two are not interchangeable, and converting between them requires assumptions about bioavailability that have not been clinically validated. Do not attempt to self-convert your injectable dose to a troche dose or vice versa without guidance from your prescribing healthcare provider.
Dosing frequency: daily versus weekly
Injectable semaglutide is dosed weekly because its half-life of approximately seven days maintains stable plasma concentrations with once-weekly administration. A single injection on Monday produces therapeutic levels through the following Sunday.
Semaglutide troches are typically taken daily. The rationale is that sublingual absorption produces a smaller, shorter peak in plasma concentration compared to injection. To maintain more consistent levels, daily dosing smooths out the peaks and troughs. Some practitioners experiment with twice-daily dosing at lower individual doses, theorizing that this produces even more stable blood levels. However, this approach adds complexity and there is no published evidence comparing once-daily versus twice-daily sublingual semaglutide.
The daily dosing requirement is one of the practical trade-offs of the troche form. While you avoid needles entirely, you commit to daily medication administration instead of weekly. For some people, this is a worthwhile exchange. For others, the simplicity of a once-weekly injection is actually more convenient than remembering a daily lozenge with all its specific administration requirements. Whether daily dosing or split weekly dosing works better depends on your personal preferences and lifestyle.
How to take a semaglutide troche properly
The technique matters. Sublingual absorption is not as simple as popping a lozenge in your mouth and going about your day. How you take the troche directly affects how much medication actually reaches your bloodstream. Poor technique can reduce an already uncertain absorption rate to something genuinely ineffective.
Step-by-step administration
Step 1: Prepare your mouth. Do not eat or drink anything for at least 15 to 30 minutes before taking the troche. Food particles, beverages, and residual flavors stimulate saliva production. Excess saliva dilutes the medication and triggers swallowing, both of which reduce the amount of semaglutide that contacts and crosses the mucosal membrane. An empty, relatively dry mouth gives the troche the best chance of working.
Step 2: Place the troche correctly. Position the troche under your tongue (sublingual) or between your cheek and lower gum (buccal). The sublingual position provides thinner tissue and richer blood supply. The buccal position provides a stable resting place where the troche is less likely to be dislodged. Some practitioners recommend starting with the sublingual position and switching to buccal if the sublingual placement feels uncomfortable or if you produce too much saliva under the tongue.
Step 3: Hold without chewing or swallowing. Let the troche dissolve completely on its own. Do not chew it. Do not suck on it aggressively. Do not move it around with your tongue. The goal is sustained, steady contact between the dissolving medication and your mucosal tissue. This process takes anywhere from 5 to 15 minutes depending on the troche formulation and your mouth environment.
Step 4: Minimize swallowing. This is the hardest part. As the troche dissolves, saliva mixed with medication accumulates in your mouth. Your instinct is to swallow it. Resist as long as comfortable. Every swallow sends medication to your stomach, where it faces enzymatic destruction and achieves minimal absorption. The longer you can hold the dissolved medication against your mucosal tissue without swallowing, the more semaglutide gets absorbed through the intended route.
Ideally, hold for a minimum of 5 minutes. Ten to fifteen minutes is better.
Step 5: Wait after the troche dissolves. After the troche has fully dissolved and you have swallowed the remaining liquid, avoid eating or drinking for another 15 to 30 minutes. This allows any medication still in contact with the mucosal tissue to continue absorbing without being washed away by food or beverages. You may continue to taste the flavoring for an hour or more after administration, which indicates residual medication in the oral cavity.
Tips for maximizing absorption
Rotate the placement site. If you use the sublingual position today, try the left buccal position tomorrow and the right buccal position the day after. Rotating prevents localized tissue irritation and may expose the medication to fresh, undamaged mucosal tissue each time.
Take the troche at the same time each day. Consistency helps maintain stable blood levels and makes it easier to plan the fasting windows around administration. Many people find that taking the troche first thing in the morning, before breakfast, works best because the mouth is naturally drier after sleep and the 30-minute pre-dose fasting window is automatically satisfied.
Stay quiet during dissolution. Reading, watching a screen, or sitting calmly reduces jaw movement, tongue activity, and saliva production compared to talking, exercising, or eating. Some practitioners specifically recommend a quiet activity like reading while the troche dissolves.
Keep the troche stored properly. Most compounded semaglutide troches should be stored at room temperature away from moisture and heat. Some formulations require refrigeration. Follow your compounding pharmacy specific storage instructions, as improper storage can degrade the semaglutide and reduce potency. Storage considerations for compounded peptides are similar to those for refrigerated semaglutide formulations, though troches may have different stability profiles than liquid forms.
What to avoid while taking semaglutide troches
Do not eat or drink anything acidic within 30 minutes of administration. Acidic foods and beverages (citrus juice, coffee, soda) can alter the pH of the oral mucosa and potentially affect absorption. Neutral to slightly alkaline conditions in the mouth are generally more favorable for mucosal drug transport.
Do not smoke or use nicotine products immediately before or after taking the troche. Nicotine constricts blood vessels, including the sublingual and buccal vasculature. Reduced blood flow to the mucosal tissue means less medication gets carried away into systemic circulation, effectively reducing absorption.
Do not use mouthwash containing alcohol right before administration. Alcohol can irritate and temporarily alter the permeability of the mucosal membrane, and the effects on semaglutide absorption are unpredictable.
Semaglutide troche versus injectable semaglutide
This is the comparison most people want. Is a troche as good as an injection? The honest answer is: we do not know for certain. But we can lay out what is known and let the evidence guide the assessment.
Bioavailability comparison
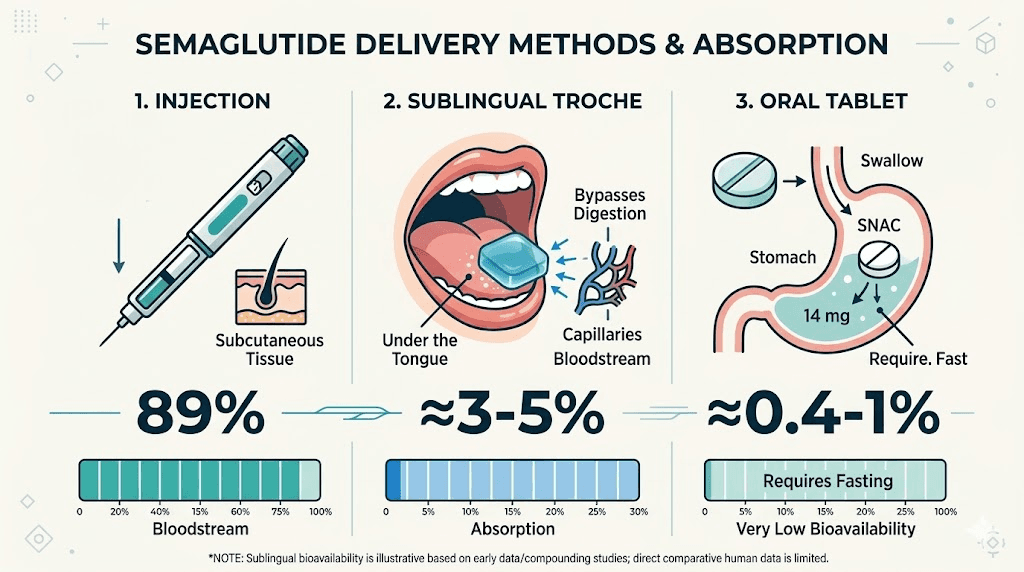
Injectable semaglutide (subcutaneous) achieves approximately 89% bioavailability. Nearly all of the injected dose reaches the bloodstream. This is well-established through extensive pharmacokinetic studies in the SUSTAIN and STEP clinical trial programs.
Oral semaglutide tablets (Rybelsus) achieve approximately 0.4% to 1% bioavailability, even with the SNAC absorption enhancer. This is why Rybelsus requires daily dosing at 7-14 mg to achieve effects roughly comparable to weekly injections of 0.5-1.0 mg. The comparison between injectable and oral peptides illustrates this bioavailability gap clearly.
Sublingual semaglutide troches likely fall somewhere between these two extremes. The sublingual route bypasses the GI tract, avoiding the massive degradation that oral tablets face. But the mucosal barrier still presents challenges for a peptide of semaglutide size. Estimated bioavailability for sublingual semaglutide ranges from 5% to 25% depending on the source, the formulation, and the assumptions used in the estimate. These are not firm numbers derived from controlled human studies. They are educated guesses based on the pharmacology of sublingual drug delivery and limited preclinical data.
Clinical evidence comparison
The evidence gap between the two delivery methods is vast.
Injectable semaglutide has been studied in over 20 major clinical trials, including the SUSTAIN program (diabetes), the STEP program (weight management), and the SELECT trial (cardiovascular outcomes). These trials enrolled tens of thousands of participants and produced some of the most impressive weight loss results ever documented for a pharmaceutical agent. In the STEP 1 trial, participants receiving 2.4 mg weekly semaglutide lost an average of 14.9% of their body weight over 68 weeks, compared to 2.4% with placebo. Week-by-week semaglutide results from these trials provide detailed timelines of expected progress.
Semaglutide troches have zero published clinical trials. Not one. There are no controlled studies comparing troche efficacy to injection efficacy in human subjects. The evidence supporting their use consists of: (1) the established efficacy of semaglutide as a molecule, (2) general pharmacological principles of sublingual drug delivery, (3) one preclinical study in rats showing measurable sublingual absorption, and (4) anecdotal reports from patients and clinicians.
This does not mean troches do not work. Many compounding pharmacy patients report appetite suppression, weight loss, and improved blood sugar control with semaglutide troches. But anecdotal reports are the weakest form of evidence. They are subject to placebo effects, reporting bias, and confounding factors like simultaneous diet and exercise changes.
Practical comparison table
Factor | Injectable semaglutide | Semaglutide troche |
|---|---|---|
FDA approved | Yes (Ozempic, Wegovy) | No |
Clinical trial data | Extensive (20+ trials) | None |
Bioavailability | ~89% | Estimated 5-25% |
Dosing frequency | Once weekly | Once daily |
Administration | Subcutaneous injection | Sublingual/buccal lozenge |
Needle required | Yes | No |
Onset of appetite suppression | 1-4 weeks typically | Variable, uncertain |
Storage | Refrigeration required | Room temperature or refrigerated |
Travel convenience | Requires cold chain, sharps disposal | Simple, room temperature |
Consistency of absorption | Highly consistent | Variable (technique dependent) |
The table tells a clear story. Injectable semaglutide wins on evidence, consistency, and proven efficacy. The troche wins on convenience, comfort, and accessibility. Which factors matter most depends on individual priorities. If you are considering the injectable route, our guide to GLP-1 injection sites covers proper technique for optimal results.
When a troche might be the better choice
Severe needle phobia is real. Clinical needle phobia (trypanophobia) affects an estimated 20-25% of adults to some degree, with 7-10% experiencing severe phobia that prevents them from receiving injections altogether. For these individuals, a troche that provides even partial semaglutide absorption may be preferable to an injection they will never take. A medication that achieves 15% bioavailability and gets used consistently is more effective than one with 89% bioavailability that stays in the refrigerator untouched.
Frequent travelers who cannot maintain cold chain storage may find troches more practical. Injectable semaglutide must be refrigerated until first use and can be kept at room temperature for limited periods thereafter. Troches are generally stable at room temperature, fit easily in a pocket or bag, and require no sharps disposal.
People who have experienced injection site reactions, such as redness, swelling, or lipodystrophy from repeated GLP-1 injections, may benefit from switching to a non-injection delivery method.
When injection is clearly superior
If you need proven, predictable, dose-dependent weight loss, injectable semaglutide is the evidence-based choice. The dose-response relationship is well-characterized. The outcomes are documented across thousands of patients. The timeline for semaglutide to work is well-established with injectable forms.
If you have diabetes and need reliable glycemic control, the uncertainty around troche bioavailability makes injections the safer option. Inconsistent drug delivery could lead to unpredictable blood sugar fluctuations.
If cost is a primary concern and you can access insurance coverage, brand-name injectable semaglutide may actually be more economical than out-of-pocket compounded troches, depending on your insurance plan.

Side effects of semaglutide troches
The side effect profile of semaglutide troches is expected to be similar to other forms of semaglutide, because the side effects are primarily driven by the pharmacological action of the drug rather than the delivery method. However, the troche form may introduce some unique considerations.
Common GLP-1 side effects
These occur regardless of delivery method and are the most frequently reported adverse effects of all semaglutide formulations:
Nausea is the most common side effect, reported by 20-44% of participants in clinical trials of injectable semaglutide. It is most pronounced during dose titration and typically improves over weeks as the body adjusts. Taking a semaglutide troche on an empty stomach, as recommended for absorption, may actually worsen nausea for some individuals because there is no food to buffer the GI effects. The relationship between semaglutide and fatigue is also well-documented across all delivery methods.
Vomiting occurs in approximately 5-15% of semaglutide users and is more common at higher doses or with rapid titration. Gradual dose escalation is the primary strategy for managing this.
Diarrhea affects roughly 8-15% of users, though interestingly some users experience the opposite problem. Constipation with semaglutide is also common, affecting 5-12% of patients in trials. The slowed gastric emptying that reduces appetite also slows intestinal transit, which can swing in either direction.
Abdominal pain and bloating are reported by 5-10% of users. These symptoms usually correspond to the delayed gastric emptying and altered gut motility caused by GLP-1 receptor activation.
Headache is reported by approximately 5-10% of users, particularly in the early weeks. This may be related to changes in eating patterns, blood sugar fluctuations, or direct central nervous system effects. Headache management strategies used for other GLP-1 agonists generally apply to semaglutide as well.
Troche-specific considerations
Beyond the standard GLP-1 side effects, semaglutide troches may produce some effects unique to the sublingual delivery method:
Oral irritation. Holding a dissolving troche against the mucosal tissue for 5-15 minutes daily can irritate the tissue, particularly if the same site is used repeatedly. Redness, mild soreness, or a tingling sensation at the placement site are possible. Rotating the placement location (left sublingual, right sublingual, left buccal, right buccal) helps minimize this.
Altered taste. Most troches contain flavoring agents, typically mint or citrus. Some users report a persistent metallic or chemical taste after the troche dissolves, which can last 30-60 minutes. This is generally mild and most people adapt to it within the first few weeks.
Excessive salivation. Having a foreign object dissolve in your mouth stimulates saliva production, which is exactly what you do not want for optimal absorption. Some people produce more saliva than others, and excessive salivation can reduce the effective dose by washing medication away from the mucosal surface and into the stomach.
Serious risks and safety warnings
The serious risks associated with semaglutide apply regardless of delivery method:
Thyroid C-cell tumors. Semaglutide carries a boxed warning (the most serious FDA warning) based on animal studies showing thyroid C-cell tumor development in rodents. It is unknown whether semaglutide causes these tumors in humans, but it is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
Pancreatitis. Cases of acute pancreatitis have been reported with semaglutide use. Symptoms include severe, persistent abdominal pain that may radiate to the back, often accompanied by nausea and vomiting. If you experience these symptoms, stop the medication and seek immediate medical attention.
Gallbladder problems. GLP-1 agonists have been associated with gallstone formation and cholecystitis. Rapid weight loss itself is a risk factor for gallstones, so it can be difficult to separate the drug effect from the weight loss effect. The connection between GLP-1 agonists and gallbladder issues applies across all formulations.
Kidney injury. Severe dehydration from GLP-1 related nausea, vomiting, or diarrhea can lead to acute kidney injury. Maintaining adequate hydration is essential, particularly during the titration phase when GI side effects are most pronounced.
Dosing errors with compounded products. This risk is unique to compounded formulations including troches. The FDA has reported instances where miscommunication between prescribers and compounding pharmacies led to patients receiving doses 5 to 20 times higher than intended. This is not a risk with FDA-approved products that come in standardized, pre-measured doses. Always verify your troche strength in milligrams and confirm it matches your prescribed dose.
Drug interactions to consider
Semaglutide slows gastric emptying, which can affect the absorption of oral medications taken around the same time. If you take other medications orally, discuss timing with your healthcare provider. Medications with narrow therapeutic windows, such as blood thinners, seizure medications, or thyroid hormones, require particular attention.
The interaction between semaglutide and phentermine is a common question, as some practitioners combine them. Similarly, questions about combining semaglutide with supplements like B12, glycine, or methylcobalamin are frequent. Many semaglutide troches already include B12 in the formulation, so additional supplementation may not be necessary.
Alcohol use while taking semaglutide troches follows the same general guidance as other semaglutide forms. The relationship between alcohol and semaglutide is an important consideration regardless of delivery method, as alcohol can worsen GI side effects and interfere with weight loss goals.
Compounding pharmacy considerations for semaglutide troches
Not all compounding pharmacies are equal. The quality of your semaglutide troche depends almost entirely on the pharmacy that makes it. Understanding what to look for, and what to avoid, can mean the difference between an effective medication and an expensive placebo.
503A versus 503B pharmacies
Compounding pharmacies in the United States operate under two primary regulatory frameworks:
503A pharmacies compound medications based on individual patient prescriptions. They are regulated primarily by state boards of pharmacy. They prepare medications in smaller quantities and are not required to follow Current Good Manufacturing Practices (CGMP). Quality oversight varies significantly by state.
503B outsourcing facilities operate under more stringent federal oversight by the FDA. They can compound medications in larger quantities without individual prescriptions, but they must follow CGMP, submit to regular FDA inspections, and report adverse events. They provide a higher baseline of quality assurance. Some of the more established compounding pharmacies for semaglutide, like Belmar Pharmacy and Empower Pharmacy, operate under these frameworks.
If you are choosing a pharmacy for semaglutide troches, a 503B facility generally offers more quality assurance than a 503A pharmacy. However, many excellent 503A pharmacies produce high-quality compounded medications. The key is due diligence.
What to ask your compounding pharmacy
Before ordering semaglutide troches from any compounding pharmacy, ask these questions:
What is the source of your semaglutide? Legitimate compounding pharmacies obtain semaglutide from FDA-registered suppliers that provide a Certificate of Analysis (COA) for each batch. The COA should confirm purity, potency, and identity testing. If a pharmacy cannot provide or will not discuss their sourcing, consider that a red flag.
Do you conduct third-party potency testing? Some pharmacies test their finished products through independent laboratories to verify that the actual semaglutide content matches the labeled amount. This is not legally required for 503A pharmacies but represents a commitment to quality.
What is your Beyond Use Date (BUD)? The BUD tells you how long the troche remains potent and stable after preparation. Shorter BUDs (30-90 days) may indicate a less stable formulation. Longer BUDs (6-12 months) suggest more rigorous stability testing. Compare this to the shelf life considerations for expired semaglutide products.
What excipients are in the troche? Excipients are the inactive ingredients that make up the troche base, including binders, sweeteners, flavorings, and potentially permeation enhancers. Some excipients can cause allergic reactions or GI discomfort. Knowing what is in your troche allows you to identify potential allergens and discuss alternatives if needed.
Do you provide a prescription verification process? Reputable pharmacies verify prescriptions with the prescribing provider, confirm dosing, and may offer pharmacist consultations. Pharmacies that ship compounded semaglutide without robust verification processes may be cutting corners elsewhere.
Red flags to watch for
Be cautious of any pharmacy or online seller that:
Sells semaglutide troches without requiring a prescription. Semaglutide is a prescription medication, period. Any source offering it without a prescription is operating illegally and cannot be trusted to provide a safe, accurately dosed product.
Claims their troches are "FDA-approved" or "clinically tested." No semaglutide troche has received FDA approval. Any such claim is false advertising and suggests the seller is willing to make misleading statements about their product.
Offers prices dramatically below market rates. Semaglutide is an expensive active pharmaceutical ingredient. If a troche is priced at a fraction of what legitimate compounders charge, the product may contain insufficient semaglutide, degraded semaglutide, or no semaglutide at all.
Cannot provide a Certificate of Analysis or refuses to answer questions about their sourcing and testing practices.

Cost comparison: semaglutide troche versus other formulations
Cost is a major factor driving interest in compounded semaglutide, including troches. The price difference between brand-name injectable semaglutide and compounded formulations is substantial.
Brand-name semaglutide pricing
Without insurance, the cost landscape for FDA-approved semaglutide products looks like this:
Ozempic (injectable, for diabetes): approximately $900-$1,100 per month at retail pharmacies. Insurance coverage varies widely, with some plans covering it fully and others requiring significant out-of-pocket costs.
Wegovy (injectable, for weight management): approximately $1,200-$1,500 per month at retail pharmacies. Insurance coverage for weight management medications is less consistent than for diabetes medications, and many patients pay a large portion out of pocket.
Rybelsus (oral tablets, for diabetes): approximately $900-$1,000 per month. This FDA-approved oral form requires the SNAC absorption enhancer and specific fasting protocols. It is covered by some insurance plans for diabetes management.
For many people, these prices are prohibitive. Even with manufacturer coupons and patient assistance programs, out-of-pocket costs for brand-name semaglutide often exceed $300-$500 per month. The peptide cost calculator can help you estimate and compare costs across different formulations.
Compounded semaglutide troche pricing
Compounded semaglutide troches typically cost between $150 and $400 per month, depending on the pharmacy, the dose, and the formulation. Some key pricing variables include:
Dose: Higher-dose troches cost more because they contain more active ingredient. A 0.5 mg daily troche is less expensive than a 2.0 mg daily troche.
Quantity: Some pharmacies offer price breaks for larger quantities (60-day or 90-day supplies).
Additional ingredients: Troches containing B12, glycine, or other additives may cost slightly more than plain semaglutide troches.
Pharmacy markup: Prices vary significantly between pharmacies. Shopping around is worthwhile, but remember that the cheapest option is not always the best quality option.
Compared to compounded injectable semaglutide, which typically costs $200-$400 per month, troches are generally in the same price range. The cost difference between compounded troches and compounded injectables is usually minimal, so the choice between them should be based on effectiveness and convenience rather than price.
The hidden cost consideration
Here is a cost factor most comparisons miss: effectiveness per dollar spent.
If injectable semaglutide achieves 89% bioavailability and produces 15% body weight reduction, and a troche achieves (hypothetically) 15% bioavailability, the troche delivers less actual medication per dollar. You might pay $300 per month for troches and receive the therapeutic equivalent of $50-$80 worth of injectable semaglutide. The remaining cost goes to semaglutide that does not get absorbed.
This calculation is speculative because the actual bioavailability of troches is unknown. But it is worth considering. If you are choosing troches primarily for cost reasons and you can tolerate injections, the compounded injectable form may actually provide better value, meaning more absorbed medication per dollar spent. Our guide to affordable compounded GLP-1 options explores these economic considerations in more detail.
Regulatory status and the FDA landscape
The regulatory environment for compounded semaglutide has shifted dramatically, and understanding the current rules is important for anyone considering a semaglutide troche.
The semaglutide shortage and compounding
From 2022 through early 2026, semaglutide was listed on the FDA Drug Shortage Database. During a declared shortage, Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act allows compounding pharmacies to produce copies of commercially available drugs. This legal provision enabled the widespread compounding of semaglutide in various forms, including injectable solutions, sublingual drops, and troches.
In February 2026, the FDA declared that the semaglutide shortage had ended. This decision had immediate implications for compounding pharmacies. When a drug is no longer in shortage, the legal basis for compounding copies of that drug becomes significantly narrower. Compounding pharmacies can still prepare semaglutide, but only when there is a documented medical need that the commercially available product cannot meet, such as:
A patient allergy to an inactive ingredient in the commercial product. A need for a dosage form not commercially available. A need for a specific dose strength not available commercially. A clinical necessity identified by the prescribing physician.
The "dosage form not commercially available" provision is the primary legal basis for continued semaglutide troche production. Since no FDA-approved semaglutide troche or sublingual formulation exists, there is an argument that compounding this specific dosage form meets the regulatory requirements even after the shortage ended. However, legal interpretations vary, and the regulatory landscape continues to evolve.
What this means for troche availability
The end of the semaglutide shortage has made compounded semaglutide troches somewhat harder to obtain and potentially more expensive. Some compounding pharmacies have reduced or stopped semaglutide production. Others continue to offer it with additional documentation requirements.
If you are currently using semaglutide troches or considering starting, be aware that your healthcare provider may need to provide clinical justification for the compounded formulation beyond simple preference for the dosage form. Your compounding pharmacy should be able to explain how they are complying with current regulations.
The upcoming clinical trial
Aspire Biopharma has announced plans for a Phase 1 clinical trial of sublingual semaglutide, expected to begin in mid-2026. This trial will be the first to formally evaluate the pharmacokinetics, safety, and efficacy of sublingually delivered semaglutide in human subjects. If successful, it could lead to FDA approval of a commercial sublingual semaglutide product, which would provide the validated formulation and clinical data that current compounded troches lack.
This is a potentially significant development. An FDA-approved sublingual semaglutide product would offer the convenience of needle-free delivery with the quality assurance, standardized dosing, and clinical evidence that compounded troches cannot currently provide. However, clinical trials take years, and FDA approval, if it comes, is likely several years away.
Who should consider semaglutide troches
The semaglutide troche is not for everyone. Understanding who benefits most, and who should look elsewhere, helps set realistic expectations.
Ideal candidates
People with genuine needle phobia. Not mild discomfort with injections, which most people experience, but clinical trypanophobia that prevents them from using injectable medications. For these individuals, a troche may be the only practical way to access semaglutide therapy. The alternative is not taking semaglutide at all, which means missing out on potential benefits entirely.
Frequent travelers. If your lifestyle involves constant travel, especially international travel or extended trips without reliable refrigeration, troches offer a practical advantage. No needles means no sharps disposal concerns. No required refrigeration (for most formulations) means no cold chain management. Troches fit in a carry-on bag alongside your regular medications.
People who have failed injectable therapy due to injection-related issues. Injection site reactions, lipodystrophy from repeated injections, or difficulty with self-injection technique are legitimate reasons to explore alternative delivery methods. If you have tried injections and experienced problems with administration, a troche eliminates those specific issues.
Those who prefer daily over weekly dosing. Some people actually prefer taking a daily medication over a weekly one. Daily dosing creates a routine that feels more natural and integrated into their health habits. If you are someone who thrives on daily health rituals, the troche fits that pattern.
Poor candidates
People who need clinically validated weight loss. If you need documented, evidence-based weight management, perhaps for a surgical requirement or medical necessity, injectable semaglutide provides the clinical evidence that troches cannot. Medical professionals evaluating your weight loss will be more confident in outcomes achieved with an FDA-approved formulation.
Diabetes patients requiring precise glycemic control. The uncertain bioavailability of troches makes them a risky choice for blood sugar management, where consistent drug delivery matters enormously. Inconsistent absorption could lead to unpredictable glucose fluctuations, which carry real health consequences.
People with dry mouth conditions. Ironic as it may seem, conditions that reduce saliva production (Sjogren syndrome, medication side effects, radiation therapy to the head and neck) can actually impair troche dissolution. Too little saliva means the troche does not dissolve properly, and the medication does not release from the matrix effectively.
People with oral mucosal diseases. Conditions affecting the mouth lining, such as oral lichen planus, aphthous ulcers, or oral candidiasis, can alter absorption characteristics and cause additional irritation when a troche is placed against affected tissue.
What to discuss with your healthcare provider
If you are considering a semaglutide troche, bring these specific questions to your healthcare provider:
What clinical evidence supports this delivery method for my specific health goals? How will we monitor whether the troche is achieving adequate blood levels? What metrics will we use to evaluate effectiveness, and at what point should we consider switching to injectable if the troche is not producing results? Is the compounding pharmacy you recommend a 503A or 503B facility, and what quality assurance measures do they follow?
SeekPeptides members access comprehensive protocol guides, dosing calculators, and expert-reviewed resources that can help you prepare for these conversations and make informed decisions about which semaglutide delivery method aligns with your goals and circumstances.
Common mistakes with semaglutide troches
Even people who choose troches for the right reasons can undermine their results with avoidable errors. These are the most common mistakes and how to fix them.
Mistake 1: Swallowing too quickly
The single biggest mistake is not holding the troche long enough. If you swallow the dissolved medication after two minutes instead of holding it for ten minutes, you send most of the semaglutide to your stomach, where it faces enzymatic destruction. The difference between a five-minute hold and a fifteen-minute hold could be the difference between meaningful absorption and almost none.
Fix: Set a timer. Do not rely on your sense of time, because your mouth will want you to swallow long before the optimal absorption period is over. Five minutes is the minimum. Ten to fifteen minutes is the target.
Mistake 2: Eating or drinking too soon
Consuming food or beverages within 30 minutes of taking the troche washes residual medication off the mucosal surfaces and introduces competing substances that can impair absorption. Many people take their troche and then immediately drink coffee or eat breakfast, nullifying much of the intended effect.
Fix: Plan your morning routine around the troche. Take it immediately upon waking, then shower, get dressed, or do other non-eating activities for 30 minutes before consuming anything. Align this with the optimal timing strategies that apply to all semaglutide formulations.
Mistake 3: Chewing the troche
Chewing breaks the troche into fragments that are immediately swallowed rather than held against the mucosa. Some people chew unconsciously, especially if the troche has a pleasant flavor. The entire point of the troche formulation is slow dissolution against tissue, and chewing defeats this mechanism.
Fix: Place the troche and then consciously relax your jaw. Do not move it with your tongue. Let it sit and dissolve on its own.
Mistake 4: Inconsistent timing
Unlike weekly injectable semaglutide, where a day or two of timing variation has minimal impact, daily troches produce smaller peaks in blood levels. Missing doses or taking them at wildly different times creates inconsistent semaglutide exposure, which can lead to fluctuating appetite and unpredictable results.
Fix: Take the troche at the same time every day. Morning administration, before breakfast, is the most practical for most people. If you are not seeing results after several weeks, inconsistent dosing is one of the first things to evaluate.
Mistake 5: Not tracking results objectively
Because troche bioavailability is uncertain, it is even more important to track objective markers of effectiveness. Many people assume the troche is working because they are taking it, without actually verifying that their appetite, weight, or metabolic markers are changing.
Fix: Track your weight weekly. Monitor your appetite on a simple 1-10 scale. Note food intake changes. If you are not seeing measurable changes after 8-12 weeks at a therapeutic dose, the troche may not be delivering adequate semaglutide to your system, and a conversation with your healthcare provider about alternative delivery methods is warranted. Tracking when weight loss begins on semaglutide helps set appropriate expectations for any delivery method.
Mistake 6: Comparing progress to injectable users
If a friend lost 30 pounds in three months on injectable semaglutide, that does not mean you should expect the same timeline with a troche. Lower bioavailability likely means slower, more modest results. Setting expectations based on injectable clinical trial data will lead to frustration.
Fix: Set your own baseline and track your own trajectory. Compare your results to your starting point, not to other people using a different delivery method with different pharmacokinetics.

Optimizing your semaglutide troche protocol
If you have decided that a semaglutide troche is the right choice for your situation, these strategies can help you get the most from it.
Nutrition while using semaglutide troches
The appetite suppression from semaglutide, when it occurs, creates a window of opportunity. Eating less does not automatically mean eating better. With reduced appetite, the quality of each meal matters more because you are consuming fewer total meals and smaller portions.
Protein intake becomes critical. Semaglutide-induced weight loss includes some muscle mass loss along with fat loss. Adequate protein intake (0.7-1.0 grams per pound of body weight daily) helps preserve lean mass. Prioritize protein at every meal, because with a suppressed appetite, you may only eat two meals per day instead of three. Following a structured semaglutide diet plan helps ensure nutritional adequacy even when eating less. The foods to prioritize while on semaglutide include lean proteins, fiber-rich vegetables, and nutrient-dense whole foods.
Hydration is equally important. Semaglutide can cause GI side effects that lead to fluid loss. Dehydration is a real risk, especially in the early weeks. Aim for at least 64-80 ounces of water daily, more if you are physically active or experiencing GI symptoms.
Exercise considerations
Resistance training is the most important exercise modality while taking semaglutide, regardless of delivery method. It directly counteracts the muscle loss that accompanies caloric deficit. Two to four resistance training sessions per week, focusing on compound movements, provides the stimulus needed to preserve lean mass.
Cardiovascular exercise remains beneficial for heart health, mood, and caloric expenditure, but it should complement, not replace, resistance training. The combination of semaglutide-reduced appetite, adequate protein, and resistance training produces the best body composition outcomes: preferential fat loss with muscle preservation. Peptides for fat loss work best when paired with appropriate exercise programming.
Supplementation while using semaglutide troches
Many semaglutide troches already contain B12, so additional B12 supplementation may be unnecessary. Check your troche formulation before adding standalone B12 supplements. Beyond B12, consider:
Vitamin D: Important for bone health, especially during weight loss when calcium metabolism can be disrupted. Many adults are deficient regardless of semaglutide use.
Magnesium: Supports hundreds of enzymatic reactions and can help with the constipation that semaglutide sometimes causes. Magnesium citrate or glycinate forms are generally well-tolerated.
Zinc: Important for immune function and metabolism. Caloric restriction can lead to zinc depletion over time.
Probiotics: The GI changes caused by semaglutide, including slowed motility, can affect the gut microbiome. A quality probiotic may help manage GI side effects. The best probiotics for semaglutide users focus on strains that support digestive comfort and regularity.
Adequate protein supplementation if dietary intake falls short. Whey, casein, or plant-based protein powders can help meet daily targets when appetite suppression makes it difficult to eat enough whole food protein.
Monitoring effectiveness
Because troche bioavailability is uncertain, monitoring is more important than it is for injectable users. Track these metrics:
Weight: Weekly, at the same time, under the same conditions. Expect slower changes than injectable users report.
Appetite: Rate daily on a 1-10 scale. A sustained decrease in appetite rating is the earliest sign that the troche is achieving therapeutic levels.
Waist circumference: Monthly measurement provides a more reliable indicator of fat loss than weight alone, especially if you are resistance training and building muscle simultaneously.
Blood glucose (if diabetic): If you are using semaglutide for glycemic control, monitor blood glucose closely, especially during dose changes. The variable absorption of troches means blood sugar responses may be less predictable than with injections.
GI symptoms: Track nausea, bowel habits, and appetite changes. GI side effects that worsen with dose increases actually serve as an indirect confirmation that semaglutide is being absorbed, because these effects are dose-dependent.
Semaglutide troche and other GLP-1 medications
Semaglutide is not the only GLP-1 receptor agonist available. Understanding how the semaglutide troche compares to other options in this class helps frame the broader decision landscape.
Semaglutide troche versus tirzepatide
Tirzepatide (brand names Mounjaro, Zepbound) is a dual GIP/GLP-1 receptor agonist that has shown even greater weight loss efficacy than semaglutide in head-to-head trials. In the SURMOUNT-1 trial, tirzepatide produced average weight loss of 20.9% at the highest dose, compared to 14.9% for semaglutide in STEP 1. Tirzepatide results have been impressive across multiple studies.
Tirzepatide is also available in compounded forms, including sublingual drops and oral formulations. The same bioavailability caveats that apply to semaglutide troches apply to compounded tirzepatide oral forms. Some patients explore switching between tirzepatide and semaglutide based on response and side effect profiles.
Semaglutide troche versus retatrutide
Retatrutide is a newer triple agonist (GIP/GLP-1/glucagon receptor agonist) that has shown remarkable weight loss results in Phase 2 trials, with participants losing up to 24% of body weight. It is not yet FDA-approved and is available only through research or compounding channels. The comparison between retatrutide and semaglutide is relevant for people exploring the full range of GLP-1-based therapies.
Semaglutide troche versus oral Rybelsus
Rybelsus is the FDA-approved oral semaglutide tablet. It uses the SNAC absorption enhancer and achieves about 1% bioavailability. Despite this low absorption, it has been shown effective for diabetes management in the PIONEER clinical trial program.
The key differences between a compounded troche and Rybelsus include: Rybelsus is FDA-approved with extensive clinical data. Rybelsus uses a proprietary absorption enhancement technology. Rybelsus is swallowed and absorbed through the GI tract, while a troche is designed for sublingual/buccal absorption. Rybelsus doses (3, 7, 14 mg) reflect its very low oral bioavailability. Troche doses are typically lower because sublingual absorption is theoretically more efficient than oral absorption.
If your primary motivation for a troche is avoiding injections while still getting an evidence-based product, Rybelsus deserves serious consideration. It has the clinical data that troches lack, and while it requires strict fasting protocols (no food or drink for at least 30 minutes after taking it), so do troches.
Combination approaches
Some clinicians prescribe semaglutide troches as part of combination protocols. Common combinations include semaglutide with vitamin B12 (often included in the troche itself), semaglutide with glycine for GI tolerability, or semaglutide alongside other weight management medications. Phentermine versus semaglutide is a common comparison, and some protocols use both together.
Any combination approach should be supervised by a healthcare provider who understands the pharmacology of all agents involved. Self-adjusting combination protocols based on internet research is not recommended, particularly with compounded medications where dosing precision is already less certain.
Long-term considerations for semaglutide troche use
Weight management with GLP-1 agonists is not a short-term intervention. Clinical data on injectable semaglutide shows that weight regain occurs in approximately two-thirds of patients who discontinue the medication. This has implications for long-term troche use.
Duration of therapy
If you are using a semaglutide troche for weight management, be prepared for extended use. The question of how long to stay on semaglutide does not have a simple answer. Current evidence suggests that continued use is necessary to maintain weight loss benefits. This means long-term daily troche administration, potentially for years.
Long-term safety data for compounded semaglutide troches does not exist. Long-term safety data for injectable semaglutide is reassuring through the major clinical trials (up to two years in some studies), and the SELECT cardiovascular outcomes trial showed cardiovascular benefits with long-term use. Whether these safety and benefit profiles apply equally to troches is unknown.
Discontinuation and weight regain
The withdrawal process from semaglutide is a critical consideration. Clinical studies show that after stopping semaglutide, most patients experience a return of appetite within weeks, followed by gradual weight regain over 12-18 months. Approximately two-thirds of lost weight is regained within one year of discontinuation.
This is not a failure of the medication. It reflects the biological reality that obesity is a chronic condition with strong physiological drivers. Semaglutide suppresses those drivers; it does not eliminate them permanently. Planning for discontinuation, including establishing sustainable dietary habits and exercise routines while the appetite suppression is active, can mitigate some of the rebound effect.
Cost sustainability
At $150-$400 per month, long-term troche use represents a significant ongoing expense. Over five years, that is $9,000-$24,000 in medication costs. Factor this into your decision, especially if insurance does not cover compounded medications (which is typical).
Compare this to the economic impact of the weight loss itself: potential reductions in diabetes medications, blood pressure medications, sleep apnea treatment costs, and obesity-related healthcare utilization. For many patients, the net economic impact of effective weight management is positive even after accounting for medication costs. But this calculus only works if the troche is actually producing meaningful weight loss, which circles back to the bioavailability question.
Frequently asked questions
How long does it take for a semaglutide troche to work?
If the troche is achieving adequate absorption, appetite suppression typically begins within 2-4 weeks of reaching a therapeutic dose. Weight loss follows as a secondary effect of reduced caloric intake. However, because troche bioavailability is uncertain, some users report slower onset compared to injectable semaglutide. If you see no appetite changes after 6-8 weeks at a therapeutic dose, discuss alternative delivery methods with your provider.
Can I switch from injectable semaglutide to a troche?
Yes, with provider guidance. The dose conversion is not straightforward because the bioavailability differs. Your healthcare provider will need to estimate an appropriate troche dose based on your current injectable dose, then monitor your response and adjust. Do not attempt this conversion on your own, as incorrect dosing could lead to loss of appetite suppression or, less commonly, overdosing.
Do semaglutide troches need to be refrigerated?
Most compounded semaglutide troches are stable at room temperature (below 77 degrees F / 25 degrees C) and do not require refrigeration. However, formulations vary by pharmacy, so follow your specific pharmacy storage instructions. Avoid exposing troches to excessive heat, moisture, or direct sunlight, as these conditions can degrade the active ingredient. Storage considerations differ from those for liquid compounded semaglutide, which typically does require refrigeration.
Are semaglutide troches safe?
Semaglutide as a molecule has an established safety profile from extensive clinical trials of injectable and oral forms. The troche-specific safety profile has not been independently evaluated. The primary safety concern unique to troches relates to compounding quality: is the product accurately dosed, pure, and stable? Choosing a reputable compounding pharmacy and working with a knowledgeable healthcare provider mitigates this risk but does not eliminate it entirely.
Can I take a semaglutide troche if I am pregnant or breastfeeding?
No. Semaglutide is contraindicated during pregnancy, and it should be discontinued at least two months before planned conception. The effects of semaglutide on fetal development have not been adequately studied in humans, but animal studies showed adverse developmental effects. The same guidance applies to breastfeeding while on GLP-1 medications.
How does a semaglutide troche compare to sublingual drops?
Both deliver semaglutide through the oral mucosa. Troches dissolve slowly (5-15 minutes), providing extended mucosal contact. Drops absorb more quickly but may be diluted and swallowed faster. Neither form has been subjected to comparative clinical trials. The choice between them is largely based on personal preference and what your compounding pharmacy offers. Our guide to oral semaglutide drops covers the liquid alternative in detail.
What happens if I accidentally swallow the troche whole?
You will absorb very little semaglutide. Without the SNAC absorption enhancer used in Rybelsus, orally swallowed semaglutide is almost entirely destroyed in the GI tract. You will not experience harmful effects from swallowing, but you will essentially miss that dose. Take your next dose at the normally scheduled time rather than doubling up.
Can I use semaglutide troches for diabetes management?
Some healthcare providers prescribe compounded semaglutide troches for diabetes management, but this is an off-label use of a compounded product. Injectable semaglutide (Ozempic) and oral semaglutide (Rybelsus) are both FDA-approved for type 2 diabetes. The uncertain bioavailability of troches makes precise glycemic control more challenging. If you choose troches for diabetes management, more frequent blood glucose monitoring is advisable.
External resources
For researchers serious about optimizing their GLP-1 protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your troches dissolve slowly, your absorption stay consistent, and your results speak for themselves.