Mar 1, 2026

Every vial of compounded semaglutide costs money. Real money. And every minute that vial sits on a countertop instead of a refrigerator shelf, you are gambling with your investment. The peptide does not care about your schedule, your forgetfulness, or the delivery driver who left the package in the sun. It degrades on its own timeline. Understanding that timeline is the difference between a protocol that works and one that quietly fails without warning.
This is the question that keeps researchers up at night. You pull the vial out for your weekly dose, get distracted by a phone call, and suddenly two hours have passed. Or maybe the power went out overnight. Or the ice packs in your delivery box arrived completely melted. In each scenario, the same thought hits: is my semaglutide still good? The answer depends on several factors, including temperature, formulation, duration of exposure, and whether the vial was previously opened. Most compounding pharmacies assign a beyond-use date based on stability testing specific to their formulation, and that date assumes proper refrigeration. Once you break that chain, different rules apply.
This guide covers everything you need to know about compounded semaglutide stability outside the refrigerator. We will walk through exact timeframes, temperature thresholds, degradation mechanisms, real-world scenarios, and the steps to take when storage goes wrong. Whether you are managing a weekly compounded semaglutide protocol or traveling with your medication, the information here will help you protect both your peptide and your results.
What makes compounded semaglutide different from branded versions
Before diving into storage timelines, you need to understand what you are actually storing. Compounded semaglutide is not the same product as Ozempic or Wegovy. Those branded medications contain proprietary stabilizers, preservatives, and buffer systems developed through years of pharmaceutical research. Novo Nordisk engineered those formulations to withstand specific temperature ranges for defined periods.
Compounded semaglutide is different. Compounding pharmacies create their own formulations using the same active ingredient but with different excipients, concentrations, and preservative systems. This means every compounding pharmacy produces a slightly different product. Some use vitamin B12 as an additive. Others include glycine for stability. Some combine multiple compounds in a single vial, like the popular semaglutide glycine B12 blend.
Why does this matter for storage? Because those different formulations have different stability profiles. A semaglutide methylcobalamin formulation may behave differently at room temperature than a plain semaglutide solution. The preservatives used, the pH of the solution, the concentration of the active ingredient, and the type of vial all influence how long the peptide remains viable outside refrigeration.
The critical takeaway is this: there is no single universal answer for how long compounded semaglutide can be unrefrigerated. The answer depends on your specific formulation. That said, we can establish general ranges based on available stability data, manufacturer guidelines, and the underlying science of peptide degradation.
How long compounded semaglutide can stay unrefrigerated
Here is the direct answer most people are looking for. Then we will break it down.
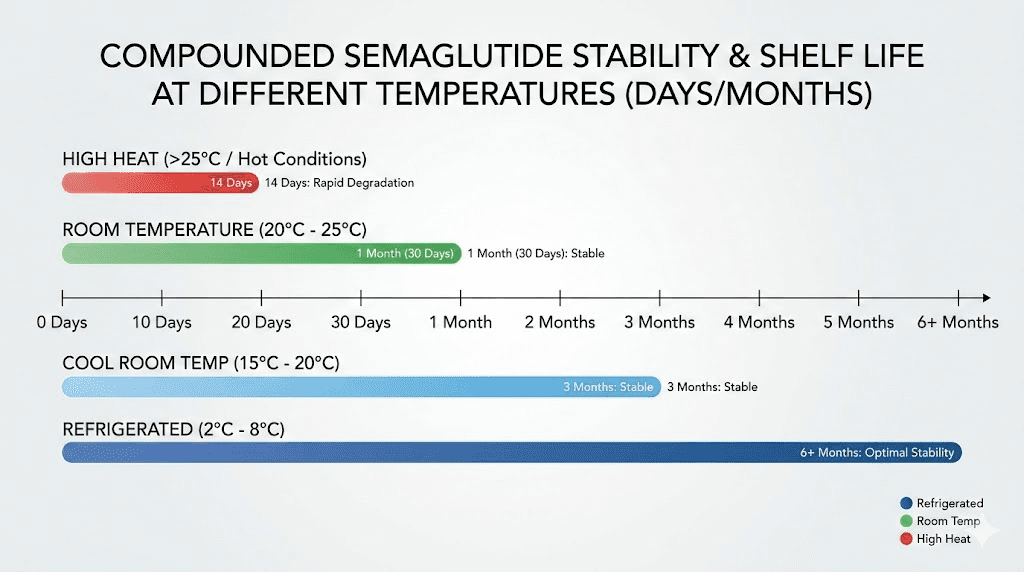
Most compounded semaglutide formulations remain stable at controlled room temperature, defined as 68 to 77 degrees Fahrenheit (20 to 25 degrees Celsius), for approximately 28 to 60 days. Some pharmacies report stability data supporting up to 60 days at controlled room temperature. Others assign more conservative windows of 14 to 28 days. The variation comes entirely from the specific formulation and the stability testing each pharmacy has conducted.
That is the range. But ranges are not very helpful when your vial has been sitting on the kitchen counter for six hours and you need to know whether to use it or throw it away. So let us get more specific.
Controlled room temperature: the 60-day window
When pharmacists refer to "controlled room temperature," they mean a narrow band between 68 and 77 degrees Fahrenheit. Not 80 degrees. Not 85 degrees. The specific range matters because peptide degradation rates increase exponentially with temperature, not linearly. At this controlled range, stability studies on compounded semaglutide formulations have shown the peptide maintains adequate potency for up to 60 days in some formulations.
This aligns with what we know about the branded products. Semaglutide storage data from the FDA prescribing information for Ozempic shows the medication can remain at room temperature (59 to 86 degrees Fahrenheit) for up to 56 days after first use. Semaglutide shelf life under these conditions has been well documented for branded versions, and compounded versions follow similar patterns when the formulation includes adequate stabilizers.
However, and this is important, your compounding pharmacy may assign a shorter window. If the label says 28 days unrefrigerated, follow the label. The pharmacy has either conducted stability testing specific to their formulation or is applying conservative guidelines to protect you. Either way, their number takes precedence over general estimates.
Temperature thresholds that change everything
The stability window shrinks dramatically as temperature rises. Think of it as a sliding scale where every degree above 77 Fahrenheit accelerates degradation.
At 77 degrees Fahrenheit (25 Celsius), the standard controlled room temperature, most formulations remain stable for 28 to 60 days. At 86 degrees Fahrenheit (30 Celsius), the upper limit of what most guidelines consider "room temperature," stability drops considerably. Most branded semaglutide products list 86 degrees as the absolute maximum, and compounded versions should not be expected to fare any better. Above 86 degrees, you are in uncharted territory for most compounded formulations. One telehealth provider reported their compounded semaglutide tolerates temperatures up to 95 degrees Fahrenheit for a maximum of 4 days, and up to 77 degrees for 15 days. But these numbers apply only to that specific formulation.
At temperatures above 95 degrees Fahrenheit, significant degradation can begin within hours. Leaving a vial in a hot car during summer, where interior temperatures can reach 130 to 170 degrees, could compromise the medication in minutes rather than hours. If you have ever wondered what happens if semaglutide gets warm, the answer is peptide denaturation and chemical breakdown that may not be reversible.
How your specific formulation affects the timeline
The concentration of semaglutide in your vial plays a role in stability. Higher concentrations may degrade differently than lower ones. If you are using a 5mg/mL formulation, its stability profile will differ from a 10mg formulation. The volume of solution remaining in the vial also matters. As you draw doses out, the headspace increases, exposing more of the solution surface to air and potential oxidation.
Preservatives are the other major variable. Bacteriostatic water containing benzyl alcohol inhibits microbial growth, which is a separate concern from peptide degradation. A vial reconstituted with bacteriostatic water will resist bacterial contamination longer than one reconstituted with sterile water, but the chemical stability of the semaglutide molecule itself depends on pH, buffer composition, and temperature rather than the preservative system.
Branded versus compounded: a storage comparison
Comparing storage requirements across different semaglutide products helps put the compounded version in context. Each product has been tested under specific conditions, and the differences are significant.
Ozempic storage at room temperature
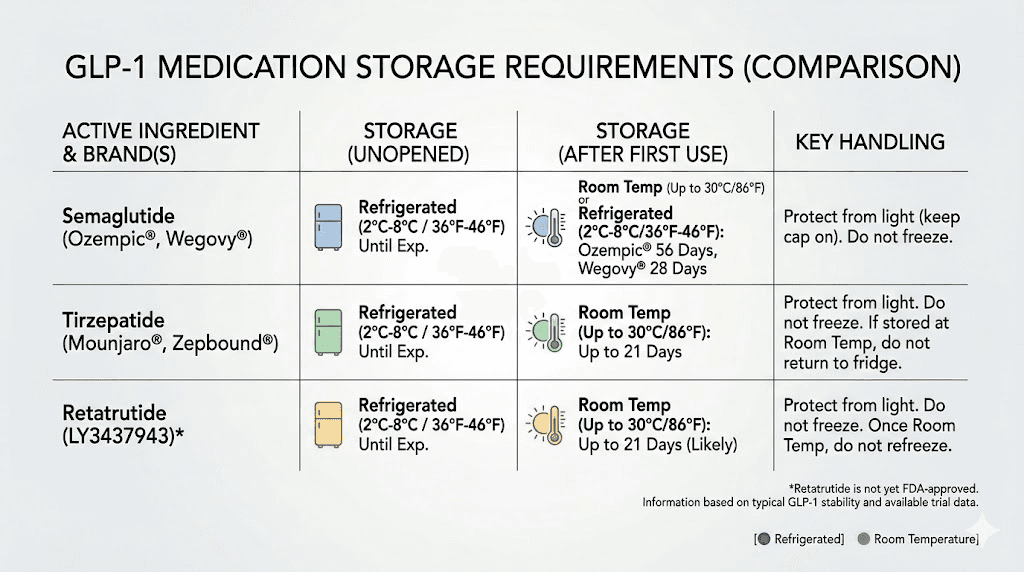
Ozempic, the most widely prescribed branded semaglutide injection, has the most generous room temperature allowance. According to the FDA prescribing information, an in-use Ozempic pen can be stored at 59 to 86 degrees Fahrenheit for up to 56 days. Before first use, unopened pens must be refrigerated at 36 to 46 degrees Fahrenheit. This 56-day window is based on extensive stability testing by Novo Nordisk and reflects the robust preservative and buffer system in the branded formulation.
For researchers comparing semaglutide versus tirzepatide, storage requirements are one practical factor worth considering in your protocol planning. Tirzepatide products have their own refrigeration requirements that differ from semaglutide.
Wegovy storage guidelines
Wegovy, the weight management version of semaglutide, has a shorter room temperature window. Unopened pens can be stored at 46 to 86 degrees Fahrenheit for up to 28 days. That is half the time Ozempic allows at room temperature. The difference likely reflects Wegovy formulation characteristics and its single-dose pen format rather than a fundamental difference in the semaglutide molecule itself.
Rybelsus oral tablets
Rybelsus, the oral semaglutide formulation, is the exception. These tablets are designed for room temperature storage from the start, with a shelf life of 24 months at 68 to 77 degrees Fahrenheit. The oral formulation uses different stabilization technology, including the SNAC absorption enhancer, which makes direct comparisons to injectable forms less meaningful.
Storage comparison table
Product | Refrigerated shelf life | Room temp (59-86F) | Max temp allowed | Format |
|---|---|---|---|---|
Ozempic | Until expiration date | 56 days (in-use) | 86F (30C) | Multi-dose pen |
Wegovy | Until expiration date | 28 days | 86F (30C) | Single-dose pen |
Rybelsus | Not required | 24 months | 86F (30C) | Oral tablet |
Compounded (typical) | 28-90 days (per BUD) | 14-60 days (varies) | 77-86F (varies) | Multi-dose vial |
The key lesson from this comparison is that even the most stable branded formulation has limits. Compounded versions, without the same level of proprietary stabilization, generally need more careful temperature management. If you are following a compounded semaglutide dose chart, protecting the integrity of your vial through proper storage directly affects whether you get the full therapeutic benefit from each dose.
What happens to semaglutide at room temperature
Understanding the science behind temperature-related degradation helps explain why storage matters so much. Semaglutide is a 31-amino acid peptide analog of human GLP-1. Like all peptides, its therapeutic activity depends on maintaining a precise three-dimensional structure. When that structure breaks down, so does the medication effectiveness.
Peptide denaturation explained
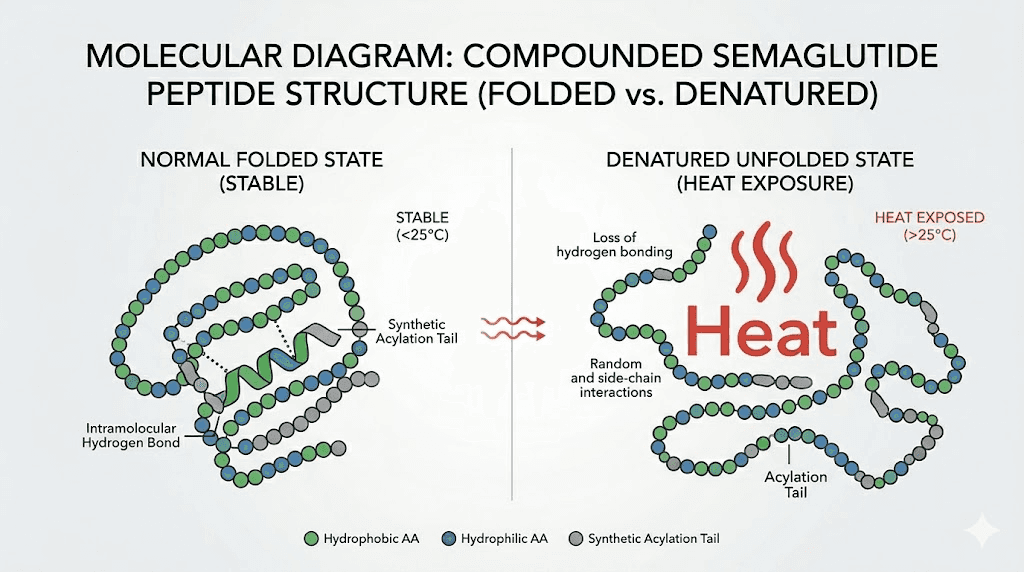
Denaturation is the unfolding of a peptide from its active conformation into a disordered state. For semaglutide, elevated temperatures disrupt two types of molecular bonds that hold the peptide together: hydrogen bonds and hydrophobic interactions. These forces are relatively weak individually, which is why they are sensitive to temperature changes. When enough of them break simultaneously, the peptide loses its ability to fit properly into the GLP-1 receptor.
The process resembles cooking an egg. The raw egg white is a clear, liquid protein solution. Apply heat, and the proteins denature into a solid white mass. You can not uncook the egg by cooling it down. Similarly, once semaglutide denatures beyond a certain point, returning it to the refrigerator does not restore its original structure.
A 2025 preformulation study published in the Journal of Peptide Science examined semaglutide stability across multiple temperature conditions. The researchers found that semaglutide remained stable for 3 hours even at 80 degrees Celsius (176 degrees Fahrenheit) in controlled laboratory conditions. This does not mean your compounded vial can handle those temperatures, as the study used purified semaglutide in specific buffer conditions, not a compounded formulation with multiple excipients. But it does demonstrate that the molecule itself has some inherent thermal resilience.
Chemical degradation pathways
Beyond denaturation, heat exposure promotes at least two other degradation pathways. Oxidation damages susceptible amino acid residues in the peptide chain, particularly methionine and tryptophan residues. Hydrolysis breaks the peptide bonds themselves, fragmenting the molecule into smaller, inactive pieces.
Research using liquid chromatography mass spectrometry (LC-HRMS) has identified at least 13 distinct degradation products that form when semaglutide breaks down. Each represents a different chemical modification to the original molecule. Some of these degradation products may be inactive. Others could potentially trigger immune responses if injected. This is why using degraded medication is not just a matter of reduced effectiveness. It introduces uncertainty about what you are actually injecting.
The pH of your compounded solution also influences degradation rates. Studies show semaglutide is relatively stable at lower pH values at room temperature but degrades faster at higher pH. This is one reason compounding pharmacies carefully formulate their solutions at specific pH levels and why proper reconstitution matters so much.
Why damage may be invisible
Here is the unsettling part. A vial of semaglutide that has undergone significant degradation may look completely normal. The solution can remain clear and colorless even after losing substantial potency. You would not know by looking at it, smelling it, or feeling the injection that the medication has been compromised.
Some visible changes do eventually appear with severe degradation. Semaglutide color changes like cloudiness, particulate matter, or discoloration indicate advanced breakdown. But by the time you can see these changes, the medication has likely been degraded far beyond the point of usefulness. Subtle potency loss from moderate temperature exposure produces no visible signs at all.
This invisible degradation is precisely why some people stop losing weight on semaglutide without understanding why. If storage has been compromised without their knowledge, they may blame the medication, their body, or their diet when the real problem is degraded peptide. Proper storage is not just about extending shelf life. It is about protecting your results.
Can refrigeration reverse the damage
Short answer: no. Once peptide denaturation or chemical degradation has occurred, returning the vial to the refrigerator does not undo the damage. Cold temperatures slow further degradation but do not repair what has already happened. Think of it as putting the brakes on a car that has already passed the exit. You stop moving further in the wrong direction, but you are still past where you needed to be.
This is why prevention matters more than recovery. The best strategy is never letting the vial reach damaging temperatures in the first place. If exposure has occurred, you need to assess the degree of damage based on temperature reached and duration, not simply refrigerate and hope for the best.
Common scenarios and what to do
Theory is helpful, but real life creates situations that do not fit neatly into storage guidelines. Here are the most common scenarios people face with compounded semaglutide and specific guidance for each one.
Left out overnight on the counter
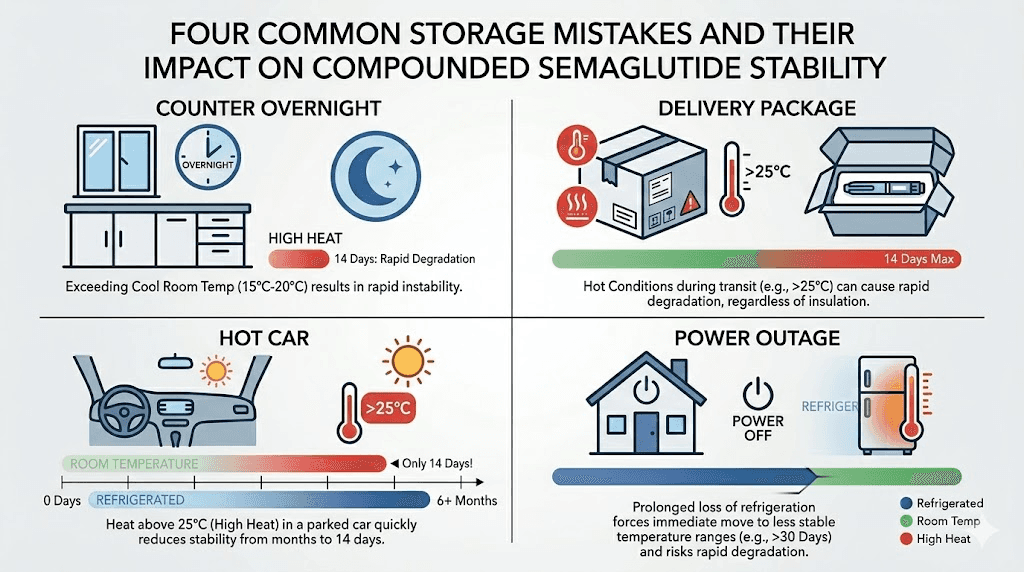
This is the most frequent storage mistake, and it happens to nearly everyone at some point. You pull the vial out for your dose, set it down, and forget about it until morning. By then, it has been at room temperature for 8 to 12 hours.
The good news: if your home temperature stayed between 60 and 77 degrees Fahrenheit, a single overnight exposure is almost certainly fine. Remember, most compounded formulations remain stable at controlled room temperature for weeks. One night is a small fraction of that window. Return the vial to the refrigerator immediately and note the exposure in case it becomes a pattern.
If you find yourself accidentally leaving semaglutide out overnight regularly, consider building a refrigeration step into your injection routine. Dose, cap, refrigerate, done. Make it automatic. Every time you leave the vial out, you are chipping away at the total stability window, even if each individual exposure seems minor. The effects are cumulative.
Delivery with melted ice packs
You come home to find your compounded semaglutide delivery sitting on the porch. The ice packs inside are completely liquid and warm to the touch. How long the package sat in the heat determines whether the medication is usable.
Most compounding pharmacies ship with enough cold packs to maintain safe temperatures for 24 to 48 hours in moderate weather. If the package arrived within that window and the ambient temperature was below 86 degrees, the medication is likely fine. Refrigerate it immediately and use it according to the normal schedule.
If the package sat in direct sunlight or in temperatures above 90 degrees for several hours, the vial may have reached damaging temperatures. Check with your compounding pharmacy before using it. Many pharmacies will replace shipments that were compromised during delivery at no additional cost, especially during summer months when heat damage is more common.
Forgot it in the car
Car interiors can reach extreme temperatures quickly. On a 90-degree day, the interior temperature can exceed 130 degrees within 30 minutes. On a 100-degree day, it can surpass 170 degrees. These temperatures will denature semaglutide rapidly.
If your vial was in a hot car for 15 minutes or less in moderate weather (below 85 degrees outside), it is probably fine. Refrigerate immediately. If it was in a hot car for an hour or more on a warm day, or any amount of time on a very hot day, discard the vial and contact your pharmacy for a replacement. The cost of a new vial is far less than the cost of weeks of injecting degraded medication that produces no results.
For context, this same principle applies to other peptide medications. If you are also managing a tirzepatide storage protocol or working with other temperature-sensitive compounds like peptides that require refrigeration, the same caution about vehicle storage applies across the board.
Power outage
A refrigerator maintains safe temperatures for approximately 4 hours during a power outage if the door remains closed. Opening the door repeatedly reduces this window. If power is restored within 4 hours and you did not open the refrigerator, your semaglutide almost certainly remained within the safe temperature range.
Extended outages of 12 to 24 hours are more concerning. The refrigerator interior will gradually warm to room temperature, which means your semaglutide transitions from cold storage to unrefrigerated storage. If your home is air-conditioned and remains below 77 degrees even without power, the medication enters the room-temperature stability window. That could still be 28 to 60 days depending on the formulation.
If your home temperature climbs above 80 degrees during an extended outage, which is common without air conditioning in summer, the degradation accelerates. Consider using a cooler with ice packs as a backup during known storm seasons. This is the same approach you would use when traveling with semaglutide, just applied to a home emergency situation.
Traveling without refrigeration
Travel is one of the most common reasons people need their semaglutide to survive without refrigeration. A weekend trip, a weeklong vacation, or even a long day of errands can take your vial out of cold storage for extended periods.
For short trips of 1 to 3 days, a quality insulated cooler bag with gel ice packs will keep the medication at safe temperatures. You do not need the vial to stay at refrigerator temperatures the entire time. Keeping it below 77 degrees is sufficient. Many travel cooler bags designed for medication can maintain this range for 12 to 24 hours with good ice packs.
For longer trips where refrigeration is genuinely unavailable, you are relying on the room-temperature stability window. If your hotel room stays at normal air-conditioned temperatures (68 to 72 degrees), your compounded semaglutide should remain stable for weeks. The risk increases in tropical climates, during summer outdoor activities, or when staying in accommodations without reliable climate control.
Plan your injection timing around your travel schedule. If you know you will be without refrigeration for a few days, time your dose so the vial spends the minimum time unrefrigerated before your next injection. And always keep the vial in the coolest, darkest spot available, like a bag in the interior of your luggage rather than a sunlit countertop.
Signs your semaglutide has degraded
Knowing how to inspect your medication after a potential storage breach can help you make informed decisions about whether to use it. Here is what to look for, starting with visual indicators and then moving to performance-based signs.
Visual indicators of degradation
Perform a visual inspection every time you draw a dose. Hold the vial up to a light source and look for any of these warning signs:
Cloudiness or haziness. Properly stored compounded semaglutide should be clear. Any cloudiness suggests protein aggregation, where degraded peptide molecules clump together. This is an immediate discard signal.
Particulate matter. Look for tiny floating particles, fibers, or specks in the solution. These can be degradation products, bacterial contamination, or foreign particles. Regardless of the cause, particulate matter means the vial should not be used.
Color changes. Fresh semaglutide solution is typically clear and colorless, though some formulations with B12 may have a slight pink or reddish tint. If you notice your semaglutide turning red or any unexpected color change from when you first received it, this warrants investigation. A significant shift in color that was not present initially suggests degradation or contamination.
Unusual texture. When drawing the solution into your syringe, it should flow smoothly with the same viscosity as water. If it feels thicker, stickier, or creates bubbles that do not dissipate normally, the formulation may have changed.
Performance indicators
Sometimes the first sign of degraded semaglutide is not something you see. It is something you feel, or rather, stop feeling. These performance changes may indicate compromised medication:
Reduced appetite suppression. One of the primary effects of semaglutide is appetite suppression. If you notice a sudden return of hunger or cravings at a dose that previously controlled your appetite, degraded medication could be the cause. This is particularly suspicious if the change happens mid-vial without any dose adjustment.
Weight loss plateau or reversal. If you were losing weight steadily and suddenly hit a semaglutide plateau that coincides with a known storage breach or a new vial from a different batch, medication quality should be on your list of possible explanations.
Loss of other effects. Semaglutide produces several effects beyond weight loss, including changes in energy levels, digestion speed, and food preferences. A sudden disappearance of these effects at a previously effective dose could signal compromised medication.
Keep in mind that performance changes can have many causes. Dose tolerance, lifestyle changes, stress, and other factors all influence results. But if you know or suspect a storage breach occurred, degradation should be high on your differential list. Do not waste weeks wondering if your protocol is failing when the answer might be sitting in a warm vial. If you are experiencing a plateau and have had storage issues, consider starting a fresh vial before troubleshooting other variables.
Traveling with compounded semaglutide
Travel is where storage theory meets real-world chaos. Airport security, hotel rooms, car trunks, beach bags. Every step of a trip introduces new temperature risks. Here is how to manage them without stress.
Short trips: 1 to 3 days
For a weekend getaway, temperature management is straightforward. Pack your vial in an insulated medication travel case with two to three gel ice packs. Do not use regular ice, which melts and creates a wet mess. Gel packs stay contained and maintain cooler temperatures longer. Place the ice packs so they surround the vial but do not touch it directly, as freezing is just as damaging as overheating. A small washcloth or paper towel between the vial and the ice packs provides a buffer.
Once you arrive, place the vial in the hotel room mini-fridge if one is available. If not, keep it in the insulated case in the coolest part of the room. Avoid the bathroom, which gets warm and humid during showers. A bedside drawer or closet shelf is a better option.
Extended travel: a week or more
Longer trips require more planning. If you are using a weekly injection schedule, a one-week trip means you need the vial to survive at least one full cycle outside normal refrigeration. Calculate how many doses you will need for the trip, consider bringing a fresh vial if your current one is partially used, and ensure you have all the supplies you need, from injection syringes to alcohol swabs.
For trips to warm climates, invest in a medical-grade travel cooler. These are different from standard insulated bags. They use phase-change materials that maintain a specific temperature range for 24 to 48 hours without ice and can be recharged by placing them in any freezer. Some models are specifically designed for injectable medication travel.
Air travel considerations
Always carry your medication in your carry-on bag, never in checked luggage. Cargo holds can reach temperatures below freezing, which damages semaglutide just as surely as heat does. TSA allows injectable medications through security. Bring your pharmacy label or prescription documentation to avoid delays, though in practice most TSA agents are familiar with medication vials and syringes.
Do not place the insulated case in the overhead bin next to the airplane cabin wall, which can get quite warm on the tarmac. Keep it under the seat in front of you where cabin temperature is more consistent. During layovers, keep the medication with you rather than storing it in airport lockers or leaving it in a hot terminal.
International travel
Research your destination medication regulations before traveling internationally. Most countries allow personal-use injectable medications with proper documentation. Carry a letter from your prescribing provider that includes the medication name, the reason for the prescription, and the expected duration of your trip. Having documentation in English and the local language of your destination adds an extra layer of preparation. For researchers also carrying other reconstituted peptides, the same documentation principles apply.
Seasonal storage considerations
Where you live and the time of year create unique storage challenges that generic guidelines do not address. Temperature management is not a one-size-fits-all situation.
Summer storage challenges
Summer introduces two major risks: ambient heat and interruptions to refrigeration. In regions where outdoor temperatures regularly exceed 90 degrees, any time your semaglutide spends outside the refrigerator carries more risk. Deliveries left on hot porches, car trips without insulation, and even carrying the vial from the pharmacy to your car can expose the medication to brief but significant heat.
During summer months, request signature-required delivery for your compounded semaglutide so it does not sit on a porch in the sun. If that is not possible, ask if the pharmacy can ship to your workplace where someone can bring it inside immediately. Track the delivery and try to be home when it arrives.
Power outages are also more common in summer due to storms and grid strain from air conditioning demand. Having a backup plan, such as a well-insulated cooler with frozen gel packs in the freezer, ready to go at all times, can protect your medication during unexpected outages. This backup approach is worth implementing for any temperature-sensitive peptide storage, not just semaglutide.
Winter freezing risks
Cold weather introduces the opposite problem. Freezing destroys semaglutide. Ice crystals form within the solution, physically shearing peptide molecules and disrupting the formulation structure. Even partial freezing, where ice crystals form and then thaw, can cause irreversible damage.
In winter, never leave semaglutide in a car, a mailbox, or any location where temperatures drop below 36 degrees Fahrenheit. The inside of a refrigerator door shelf is a better storage spot than the back of the fridge, where temperatures can drop closer to freezing. If your home refrigerator tends to freeze items, especially in the back or near the cooling element, store your vial on a middle shelf near the front.
Delivery packages during winter can also freeze. If your package arrives with frozen ice packs and the vial feels ice-cold or shows any evidence of ice crystals, contact your pharmacy before using it. Just as with overheating, freezing damage is not reversible by simply bringing the medication back to the correct temperature.

Storage best practices for maximum potency
Now that you understand the risks, here are the specific practices that maximize the potency and shelf life of your compounded semaglutide.
Ideal refrigerator placement
Not all spots in your refrigerator are equal. The door shelves experience the most temperature fluctuation from opening and closing, making them the worst spot for medication. The back of the main compartment stays coldest and most consistent, but can sometimes be cold enough to freeze liquids. The middle of a center shelf is the sweet spot, where temperatures stay most consistently in the 36 to 46 degree range.
Store the vial upright in a small container or ziplock bag to prevent it from rolling around and to protect it from spills. Keep it away from the back wall where the cooling element can create freezing zones. A properly refrigerated compounded semaglutide vial can last the full beyond-use date assigned by the pharmacy.
After reconstitution
If your compounded semaglutide arrives as a lyophilized (freeze-dried) powder that requires reconstitution, the storage clock starts ticking the moment you add the diluent. Unreconstituted powder is generally more stable at room temperature than the reconstituted solution, which is one reason some compounding pharmacies ship in lyophilized form.
After reconstitution with the appropriate amount of bacteriostatic water, refrigerate the vial immediately. The shelf life of reconstituted peptides in the fridge is significantly shorter than the powder form, typically 28 to 60 days depending on the formulation. Use the peptide reconstitution calculator to determine the correct diluent volume for your concentration, and refer to the semaglutide reconstitution chart for specific mixing ratios.
During use
When you take the vial out for your dose, minimize the time it spends at room temperature. Have your supplies ready before removing the vial from the fridge. Draw your dose, recap the vial, and return it to the refrigerator. The entire process should take less than 5 minutes.
Allow the injection solution to warm slightly before injecting, as cold injections can be more uncomfortable. One to two minutes at room temperature is sufficient. You do not need to wait for the vial to reach full room temperature. If you are new to the injection process, review the best injection sites for semaglutide and proper GLP-1 injection technique to make the process efficient.
Protecting from light
Temperature gets most of the attention, but light exposure also contributes to peptide degradation. Ultraviolet light from sunlight and some artificial sources can accelerate oxidation of the semaglutide molecule. Store the vial in its original box or a dark container inside the refrigerator. Never leave it on a windowsill, a sunlit counter, or anywhere with direct light exposure, even briefly.
This is especially important if your formulation comes in a clear glass vial rather than an amber one. Amber glass filters UV light, providing some built-in protection. Clear glass offers none. If your pharmacy ships in clear vials, keeping the vial in its box or wrapping it in aluminum foil provides additional light protection.
How proper storage protects your results
Storage is not just about keeping your medication technically "within specifications." It directly affects your results, your timeline, and your experience on semaglutide.
A fully potent dose of semaglutide suppresses appetite within a specific timeframe. Researchers typically notice appetite suppression beginning within hours to days of their first dose. As the protocol continues, effects build with each subsequent injection. How fast semaglutide works depends on consistent, potent dosing.
Now imagine what happens with a partially degraded vial. Instead of receiving your full prescribed dose, you might be getting 70 or 80 percent of the intended amount. The effects are weaker. Appetite suppression is partial rather than robust. Weight loss slows. You might think you need a higher dose when in reality, you need a properly stored one.
This creates a cascade of problems. You or your provider might increase the dose to compensate, which means you use the vial faster and spend more money. Or you might conclude that semaglutide is not working for you and switch medications entirely. If you have been wondering why you are not losing weight after 4 weeks on semaglutide, medication storage should be one of the first variables you investigate.
For those tracking their progress on a semaglutide diet plan or following a specific food protocol while on semaglutide, degraded medication undermines all the dietary effort you are putting in. The medication is supposed to be the foundation that makes the nutritional changes sustainable. When that foundation crumbles because of poor storage, everything built on top of it becomes harder.
SeekPeptides members frequently discuss storage protocols in the community, sharing practical tips for maintaining medication potency across different living situations. The consensus among experienced researchers is clear: investing a small amount of effort in proper storage pays enormous dividends in consistent results.
Reconstitution and storage timeline
Understanding the full lifecycle of your compounded semaglutide, from receipt through final dose, helps you plan storage at each stage.
Stage 1: Arrival and initial storage
When your compounded semaglutide arrives, refrigerate it immediately. Check the vial for any visible damage, leaking, or evidence of freezing. Note the beyond-use date on the label. If the vial arrives as a reconstituted solution, the BUD clock started at the pharmacy, not when you received it. This means some of your storage window has already been used during shipping.
If the vial arrives as lyophilized powder, you have more flexibility. The powder form is more stable and can tolerate brief temperature excursions during shipping better than the reconstituted solution. Refrigerate it and reconstitute when you are ready to begin your protocol.
Stage 2: Reconstitution
When you are ready to start, follow the reconstitution instructions for your specific concentration. Use the correct volume of bacteriostatic water as specified by your pharmacy. Swirl gently rather than shaking, as vigorous agitation can denature the peptide. Once reconstituted, label the vial with the date and refrigerate immediately.
The semaglutide mixing chart for your concentration will tell you how much diluent to add for each desired dose concentration. Getting this right at reconstitution sets you up for accurate dosing throughout the vial life. A mistake here compounds through every subsequent dose.
Stage 3: Active use
During active use, your vial will come out of the refrigerator briefly for each dose. These brief excursions are normal and expected. The storage guidelines account for them. What you want to avoid is cumulative room-temperature time that exceeds your formulation stability window.
Keep a running mental tally of unrefrigerated time. If your vial has been out for an hour here, two hours there, and then overnight once, that adds up. While each individual event might not exceed the stability limit, the total could. This is especially relevant toward the end of a vial when you have drawn most doses and the remaining solution has been through weeks of refrigerator-to-counter-to-refrigerator cycles. Use the semaglutide dosage calculator to plan how many doses remain in your vial so you can estimate the total unrefrigerated exposure time.
Stage 4: Approaching the beyond-use date
As you approach the BUD, the margin for storage errors shrinks. The peptide has been slowly degrading over its entire shelf life, even under perfect refrigeration. Degradation does not wait for the BUD to begin. It is a continuous process that refrigeration slows but does not stop. Near the end of the BUD, any additional unrefrigerated time carries proportionally more risk.
If you have doses remaining as the BUD approaches, use them according to schedule but be extra diligent about storage. Do not stretch the vial past its BUD, even by a few days. If you need guidance on what to do with expired semaglutide or what happens if you accidentally use expired semaglutide, err on the side of caution and start a fresh vial.
The cost of getting storage wrong
Let us talk money for a moment. Compounded semaglutide is not cheap. Vials can cost anywhere from $100 to $400 or more depending on the concentration, pharmacy, and formulation. Wasting even one vial to preventable storage damage is a direct financial hit.
But the indirect costs are worse. If you are using degraded semaglutide without knowing it, you are paying full price for partial results. You are investing time in a protocol that cannot deliver its full potential. You might spend weeks at a weight loss plateau before realizing the medication was the problem, not your body. Those are weeks you do not get back.
Researchers who supplement their semaglutide protocol with B12, glycine, or L-carnitine to enhance results are especially affected by storage issues. These complementary compounds work synergistically with semaglutide, but only if the semaglutide itself is delivering its full potency. Supporting compounds cannot compensate for a degraded primary medication.
The simple math: spending $15 on a good insulated travel case and $10 on quality gel ice packs can protect thousands of dollars in medication and weeks of progress. That is the best return on investment in your entire protocol.
Comparing semaglutide storage with other GLP-1 medications
If you are considering switching between GLP-1 medications or running parallel protocols, understanding the storage differences helps with logistics planning.
Compounded tirzepatide storage follows similar principles to compounded semaglutide but with its own specific stability profiles. Tirzepatide is a dual GIP/GLP-1 agonist with a different molecular structure, which means its degradation characteristics are not identical. If you are managing both medications, or considering a switch from tirzepatide to semaglutide or the reverse conversion, each medication needs its own storage protocol.
Retatrutide, the triple agonist GLP-1/GIP/glucagon receptor agonist, has its own reconstitution and storage requirements. Those interested in how retatrutide compares to semaglutide should note that newer peptides may have different stability profiles than established ones. The reconstitution protocols for retatrutide differ from semaglutide as well.
General peptide expiration principles apply across all these medications. All injectable peptides are temperature-sensitive. All degrade faster at higher temperatures. And all benefit from consistent, proper refrigeration. The specific timeframes differ, but the underlying biology remains the same.
Advanced storage considerations
Humidity and moisture
While temperature gets most of the attention, humidity can also affect compounded semaglutide, particularly the rubber stopper on the vial and the seal integrity. High humidity environments can promote microbial growth on the vial exterior, which increases contamination risk each time you insert a needle. Keep the vial in a sealed bag or container inside the refrigerator to protect against moisture accumulation.
Light exposure specifics
Semaglutide is sensitive to UV radiation, but normal indoor lighting poses minimal risk during brief exposure periods. The concern is prolonged exposure to direct sunlight or fluorescent lighting. If you are storing the vial on a counter for any reason, place it in a drawer or cabinet rather than leaving it exposed. Even 30 minutes of direct afternoon sunlight through a window can generate enough UV exposure to begin photo-oxidation of the peptide.
Vibration and physical stress
Physical agitation can promote peptide aggregation, particularly in reconstituted solutions. During travel, secure the vial so it does not bounce around in a bag or suitcase. Some researchers wrap their vials in a sock or small towel inside their travel cooler to minimize movement. Never shake a reconstituted peptide vial. If you need to mix it after it has been sitting undisturbed, swirl gently. This applies equally to reconstituted tirzepatide and other peptides mixed with bacteriostatic water.
Multiple vial management
Some researchers keep multiple vials at different stages. Perhaps one is in active use, another is reconstituted and waiting, and a third is still in lyophilized form. Each vial needs its own storage tracking. Label each with the reconstitution date and start of use date. Use the oldest reconstituted vial first to minimize waste. Store unreconstituted vials separately from reconstituted ones, and always use the reconstituted vial with the earliest date first.
When to discard and replace your vial
Knowing when to cut your losses and start fresh is just as important as knowing how to store properly. Here are the clear discard triggers:
Definite discard situations:
Any visible cloudiness, particles, or color change beyond what is normal for your formulation
Exposure to temperatures above 95 degrees Fahrenheit for more than 4 hours
Any evidence of freezing (ice crystals seen at any point)
Past the beyond-use date, even by one day
Dropped and cracked vial, even if the liquid looks fine
Known contamination (needle touch to non-sterile surface, etc.)
Probable discard situations (consult your pharmacy):
Extended exposure above 86 degrees for more than 24 hours
Multiple repeated room temperature excursions totaling more than the stability window
Delivery that sat in extreme heat or cold for an unknown duration
Sudden loss of effectiveness with no other explanation
Generally safe to continue:
Single overnight exposure at normal room temperature (68-77F)
Brief excursions of 1-2 hours during normal use
Exposure to temperatures up to 77F for less than 14 days total
Short car trips (under 30 minutes) in moderate weather
When in doubt, contact your compounding pharmacy. Many pharmacies have pharmacists available to assess specific exposure scenarios and advise whether the medication is still suitable for use. This professional guidance is far more reliable than guessing. For context, the same assessment principles apply if you are managing tirzepatide expiration concerns or wondering about whether semaglutide expires after 28 days.
Building a storage-proof protocol
The best storage strategy is one that eliminates opportunities for error. Here is a complete protocol for protecting your compounded semaglutide from the day it arrives to the last dose.
Arrival day: Receive the package personally if possible. Open immediately, inspect the vial, check the BUD, and refrigerate. If the package sat outside in extreme temperatures, photograph it and contact the pharmacy.
Storage setup: Designate a spot in your refrigerator, middle shelf, center area. Place the vial in a small container or bag to keep it upright and protected. If possible, keep a small thermometer in the fridge to verify the temperature stays between 36 and 46 degrees.
Injection routine: Prepare all supplies before opening the fridge. Remove the vial, draw your dose within 2 to 3 minutes, and return it immediately. Do not set it down and walk away. This discipline eliminates the most common source of accidental room temperature exposure.
Travel prep: Keep an insulated medication bag and gel packs ready at all times. When you know travel is coming, freeze the gel packs 24 hours in advance. Pack the bag the night before and transfer the vial from fridge to bag right before leaving.
Emergency backup: Keep a cooler and frozen gel packs in your freezer as a backup for power outages. If the power goes out, transfer the vial to the cooler immediately, and then do not open the cooler unless necessary.
For anyone serious about optimizing their semaglutide protocol, SeekPeptides provides comprehensive storage guides, stability databases, and handling protocols for members. The community includes experienced researchers who have navigated every storage scenario imaginable, from cross-country road trips to tropical vacations to extended power outages.
Frequently asked questions
Can I use compounded semaglutide that was left out for 24 hours?
If the room temperature stayed between 68 and 77 degrees Fahrenheit, a 24-hour exposure is within the stability window of most compounded formulations. Refrigerate immediately and continue use. If the temperature was higher, especially above 86 degrees, contact your pharmacy for guidance. One 24-hour exposure at controlled room temperature should not significantly impact a vial that was otherwise properly stored.
Does compounded semaglutide go bad faster than Ozempic at room temperature?
It depends on the formulation. Branded Ozempic contains proprietary stabilizers that Novo Nordisk developed specifically to extend room temperature stability to 56 days. Most compounded formulations do not have the same stabilizer profile, which is why their room temperature windows are sometimes shorter. However, some well-formulated compounded versions perform comparably. Check your pharmacy stability data for specifics.
What temperature is too hot for compounded semaglutide?
The general consensus is that temperatures above 86 degrees Fahrenheit (30 degrees Celsius) exceed the safe storage range. Brief exposures above this threshold, such as walking from a pharmacy to an air-conditioned car, are unlikely to cause meaningful degradation. Prolonged exposure above 86 degrees, especially above 95 degrees, can compromise the medication within hours. The effects of heat on semaglutide are cumulative and potentially irreversible.
Can I freeze compounded semaglutide to make it last longer?
Do not freeze reconstituted compounded semaglutide. Ice crystals damage the peptide structure through a physical mechanism different from heat damage but equally destructive. Lyophilized (freeze-dried) powder can be stored in the freezer before reconstitution if the pharmacy approves, but once reconstituted, refrigeration (36-46 degrees Fahrenheit) is the only appropriate storage temperature. Review our guide on compounded semaglutide refrigeration requirements for more details.
How do I know if my semaglutide lost potency from heat exposure?
Unfortunately, there is no reliable home test for potency. Visual inspection can catch severe degradation, such as cloudiness, particles, or color changes, but moderate potency loss produces no visible signs. The most reliable indicator is a sudden change in medication effectiveness. If your appetite suppression weakens, your weight loss stalls, or your expected monthly results fall short without any other explanation, degraded medication should be investigated.
Is it safe to inject semaglutide that has been at room temperature for a week?
At controlled room temperature (68-77 degrees Fahrenheit), one week of exposure is well within the stability window of most compounded semaglutide formulations. It should be safe to use. However, the cumulative unrefrigerated time matters. If the vial had previous room temperature exposures before this week, add them together. Follow your pharmacy beyond-use date guidance, and when in doubt, ask your pharmacist.
What should I do if my semaglutide was delivered in a hot package?
Document the delivery conditions, including approximate outdoor temperature and how long the package may have been sitting outside. Refrigerate the vial immediately. Contact your compounding pharmacy with the details. Most reputable pharmacies will replace heat-compromised shipments, especially during summer. Do not use the medication until you have received guidance from the pharmacy. Many delivery storage issues are discussed in detail in our complete compounded semaglutide guide.
Does the type of vial affect how long semaglutide can be unrefrigerated?
Yes. Amber glass vials provide UV protection that clear glass vials do not, which can slow photo-oxidation degradation. The type of rubber stopper and seal also affect stability. However, these factors are secondary to temperature. The vial type might make a marginal difference over weeks of storage, but it will not save a vial that was left in a 100-degree car for an afternoon.
External resources
For researchers serious about protecting their semaglutide investment and optimizing every aspect of their protocol, SeekPeptides offers the most comprehensive resource available. Members access detailed storage guides, evidence-based protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your results stay consistent.