Feb 25, 2026

You are throwing money away. Right now. Every vial of compounded semaglutide sitting in your refrigerator is on a countdown timer, and most people have no idea how much time remains. The difference between a potent, effective dose and a degraded waste of money often comes down to a single number printed on the label, a number that most people either ignore or misunderstand entirely.
Compounded semaglutide does expire. But not in the way most medications do. Unlike the FDA-approved brand-name versions you might recognize from pharmacy shelves, compounded formulations operate under a completely different dating system called a beyond-use date. This distinction matters more than most people realize, because it affects potency, safety, and whether your compounded semaglutide is actually doing what you think it is doing.
This guide covers everything about compounded semaglutide expiration. You will learn how beyond-use dates differ from standard expiration dates, what factors accelerate degradation, how to recognize when your medication has gone bad, and the exact storage protocols that maximize shelf life.
Whether you are new to semaglutide or months into your protocol, understanding expiration is not optional. It is essential.
SeekPeptides has helped thousands of researchers navigate exactly these kinds of critical questions, and this guide reflects that same evidence-based approach.
Does compounded semaglutide actually expire?
Yes. Compounded semaglutide absolutely expires. There is no version of this medication, compounded or otherwise, that lasts forever. The active peptide degrades over time through natural chemical processes including oxidation, hydrolysis, and aggregation. These processes happen regardless of how carefully you store it.
But here is where it gets complicated.
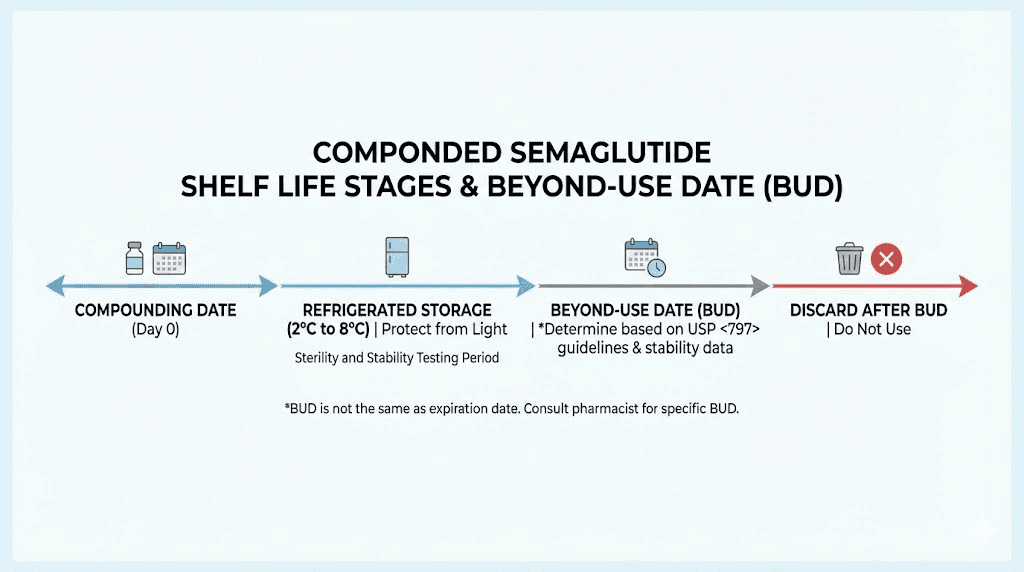
Compounded semaglutide does not carry a traditional expiration date. Instead, it carries something called a beyond-use date, or BUD. This is the date assigned by your compounding pharmacy that indicates the last day the medication should be used. After that date, the pharmacy can no longer guarantee the potency, sterility, or safety of the product.
The concept of a beyond-use date exists because compounded medications are prepared individually or in small batches. They do not undergo the same extensive stability testing that large pharmaceutical manufacturers conduct over years of development. A brand-name product like Ozempic has gone through rigorous multi-year stability studies. A compounded version relies on shorter-term stability data specific to that particular formulation and compounding process.
This does not mean compounded semaglutide is inferior. It means the dating system is different, and understanding that difference is critical for anyone using these formulations. If you have wondered whether your vial is still good, or if the medication sitting in your refrigerator has lost its effectiveness, the answer starts with understanding what your BUD actually means.
For a deeper look at how semaglutide compounding works overall, the complete guide to compounded semaglutide covers the fundamentals.
Beyond-use dates vs expiration dates explained
Most people use the terms interchangeably. They should not.
An expiration date appears on FDA-approved medications. It represents the date through which the manufacturer guarantees the product will maintain its labeled potency and safety when stored according to the label instructions. These dates are determined through International Conference on Harmonisation (ICH) stability testing protocols that can span years. The manufacturer submits this data to the FDA, and the FDA reviews and approves it.
A beyond-use date is fundamentally different. It appears on compounded medications and represents the date after which the preparation should not be used. The compounding pharmacy, not the FDA, assigns this date based on available stability data, USP guidelines, and the specific characteristics of their formulation. BUDs are typically much shorter than manufacturer expiration dates.
Here is why this matters for your semaglutide dosage protocol. A brand-name Ozempic pen carries an expiration date that might be two years from manufacture when unopened. Once opened, Novo Nordisk specifies it is good for 56 days. That 56-day window comes from extensive stability testing the company funded and submitted to the FDA.
Your compounded semaglutide vial, by contrast, might carry a BUD of 30, 60, or 90 days from the date of compounding. Some pharmacies that conduct rigorous in-house stability testing might assign BUDs up to 120 days. Others, relying on more conservative default USP guidelines, might assign BUDs as short as 14 days.
The variation is enormous. And it matters.
A 14-day BUD versus a 120-day BUD on the same active ingredient tells you something important about the pharmacy that made it. The pharmacy with the longer BUD has invested in stability testing to prove their formulation remains stable for that duration. The pharmacy with the shorter BUD is either using a less stable formulation or simply has not conducted the testing to prove otherwise.
Neither approach is necessarily wrong. But if you are trying to understand how long semaglutide is good for, you need to know which type of date you are looking at and what it actually represents. An expiration date on Ozempic means one thing. A BUD on your compounded vial means something different entirely.
The USP Chapter 795 (for non-sterile preparations) and USP Chapter 797 (for sterile preparations like injectable semaglutide) provide the framework pharmacies use to determine these dates. We will dig into USP 797 specifically later in this guide, because it directly affects how your compounded semaglutide is made, tested, and dated.
How long does compounded semaglutide last?
The answer depends on several variables. What formulation type did your pharmacy use? Has the vial been opened? How has it been stored? The timeline changes dramatically based on these factors.
Liquid formulations
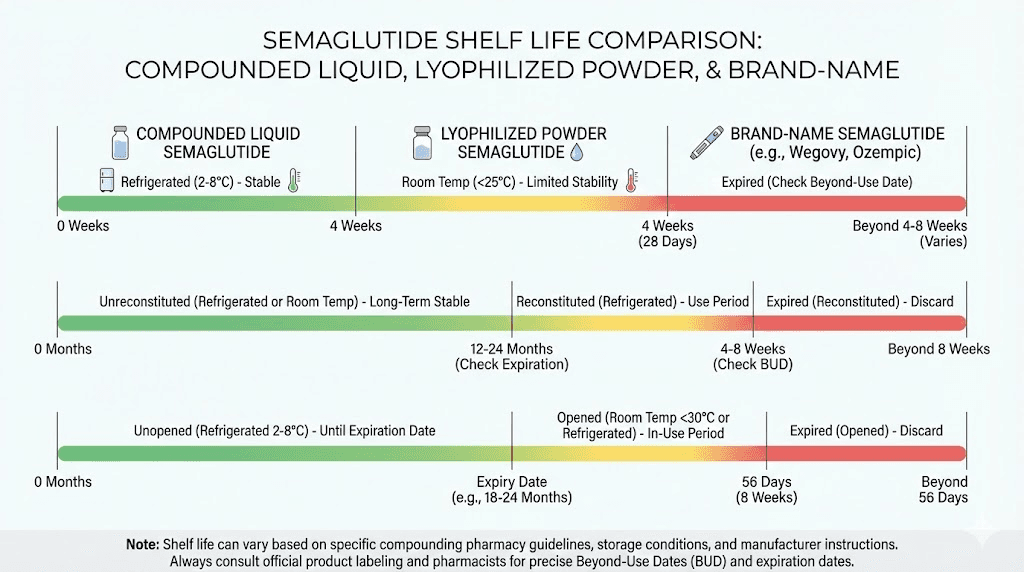
Most compounded semaglutide arrives as a ready-to-inject liquid solution. These formulations are the most common and also the most time-sensitive. When stored properly in a refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius), liquid compounded semaglutide typically remains stable for 4 to 6 weeks from the compounding date.
Some pharmacies achieve longer stability windows. Formulations from facilities with advanced stability testing programs may last up to 90 days when refrigerated and unopened. But 4 to 6 weeks is the general baseline for liquid preparations.
The reason liquid formulations have shorter shelf lives relates to the nature of peptides in solution. Once semaglutide is dissolved in a liquid carrier, it becomes more susceptible to chemical degradation. The peptide bonds can undergo hydrolysis, where water molecules break apart the amino acid chain. Oxidation reactions can alter specific amino acid residues. And the solution itself provides an environment where microbial contamination becomes possible if sterility is compromised.
If you are working with a liquid formulation, understanding proper refrigeration requirements becomes absolutely essential. Every hour outside the proper temperature range accelerates these degradation processes.
Lyophilized powder formulations
Lyophilized, or freeze-dried, semaglutide powder represents the more shelf-stable option. Before reconstitution, these preparations can remain stable for several months when stored properly. The removal of water during the lyophilization process dramatically slows the chemical degradation pathways that affect liquid formulations.
Think of it this way. Water is the enemy of peptide stability in long-term storage. Lyophilization removes that enemy. Without water, hydrolysis cannot occur. Oxidation slows considerably. Microbial growth becomes impossible in a dry, sealed vial.
However, once you reconstitute semaglutide by adding bacteriostatic water, the clock starts ticking at roughly the same rate as a pre-mixed liquid formulation. The advantage of lyophilized powder is in storage before reconstitution, not after. For guidance on mixing, check the 10mg semaglutide reconstitution guide or the 5mg semaglutide reconstitution guide.
After reconstitution
Once lyophilized semaglutide powder has been mixed with bacteriostatic water, the stability profile changes completely. The reconstituted solution typically needs to be used within 28 to 30 days when refrigerated. Some pharmacy labels may specify different windows, so always defer to what your specific pharmacy indicates.
The bacteriostatic water itself plays a role here. It contains benzyl alcohol as a preservative, which helps inhibit microbial growth. But it does not stop chemical degradation of the peptide. The semaglutide molecule will still undergo gradual breakdown through hydrolysis and oxidation, just as it would in any aqueous solution.
Use the peptide reconstitution calculator to determine your exact mixing ratios, and note the date you reconstitute on the vial. This becomes your personal starting point for tracking shelf life after mixing.
After first use
The first time you puncture the rubber stopper with a needle, you introduce another variable: potential contamination. Even with proper sterile technique, each needle insertion creates a microscopic pathway for bacteria. This is why many pharmacies specify a 28-day use window after first puncture, regardless of the original BUD.
Brand-name Ozempic pens allow 56 days after first use. Compounded vials are typically more conservative at 28 days. The difference comes down to the delivery mechanism. Pre-filled pens have closed systems that minimize contamination risk. Multi-dose vials with rubber stoppers have slightly higher exposure risk with each use.
If you are following a semaglutide dosage chart and your protocol spans several weeks, this 28-day window after first use becomes the practical limiting factor. Even if your BUD is 60 or 90 days out, once you start drawing from the vial, the 28-day clock takes priority.
Shelf life comparison table
Formulation type | Unopened shelf life | After first use | Storage temperature |
|---|---|---|---|
Liquid (pre-mixed) | 30 to 90 days (pharmacy dependent) | 28 days | 36-46F (2-8C) |
Lyophilized powder | Several months | N/A (not yet reconstituted) | 36-46F (2-8C) |
After reconstitution | 28 to 30 days | 28 days | 36-46F (2-8C) |
Brand-name Ozempic | Up to 2 years | 56 days | 36-46F (2-8C) |
The table above makes one thing clear. Compounded semaglutide has a significantly shorter usable life than brand-name alternatives. This is not a quality issue. It is a reality of the compounding process and the testing standards involved. Plan your dosing schedule accordingly to avoid waste.
Factors that affect compounded semaglutide expiration
Not all compounded semaglutide degrades at the same rate. Several factors either accelerate or slow the process. Understanding these factors gives you direct control over how long your medication remains effective.
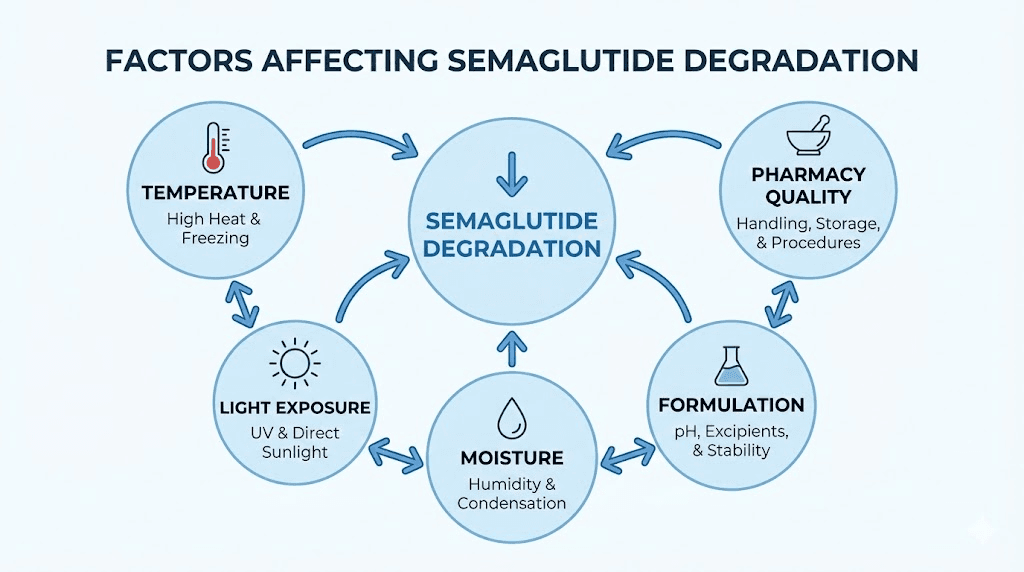
Temperature
Temperature is the single most important factor. Research published in the Journal of Peptide Science examined how temperature affects semaglutide stability at the molecular level. Studies conducted at 25, 40, and 55 degrees Celsius showed accelerating degradation at higher temperatures. Extended stability testing at 5, 25, 40, 60, and 80 degrees Celsius revealed that while semaglutide remained stable for 3 hours even at 80 degrees Celsius under stress conditions, real-world storage demands consistent cold temperatures for long-term stability.
The target range is 36 to 46 degrees Fahrenheit, or 2 to 8 degrees Celsius. This is standard refrigerator temperature. Fluctuations matter. Opening and closing the refrigerator door repeatedly, storing vials in the door compartment where temperature swings are greatest, or placing vials near the back where they might freeze, all of these common habits affect shelf life.
If your semaglutide has been exposed to warmth, the guide on warm-exposed semaglutide covers what to do. And if you accidentally left it out, this guide on overnight exposure explains the risks.
Room temperature exposure up to 86 degrees Fahrenheit for brief periods (a few hours) is generally tolerable. Beyond that, degradation accelerates rapidly. Never assume medication left at room temperature all day is fine. Time and temperature interact, and the damage accumulates.
Light exposure
UV and visible light trigger photochemical reactions in semaglutide. These reactions can damage sensitive amino acid residues within the peptide chain, reducing potency and potentially creating degradation products that were not in the original formulation.
This is why compounded semaglutide typically arrives in amber vials or opaque packaging. Store it in the original packaging whenever possible. If your pharmacy uses clear vials, keep them in a dark area of your refrigerator. Do not leave vials on countertops, near windows, or under bright lights.
The research on peptide stability consistently identifies light as a significant degradation factor, second only to temperature. If you want your medication to last as long as possible, darkness is your friend.
Moisture and contamination
For liquid formulations, contamination is the primary concern. Every time you insert a needle through the rubber stopper, you create a potential entry point for bacteria. Proper technique, using alcohol swabs on the stopper before each draw, using sterile needles, and minimizing the number of punctures, helps reduce this risk.
For lyophilized powder, moisture exposure before reconstitution is the enemy. If moisture enters the sealed vial through a compromised seal or improper handling, the powder can begin degrading through hydrolysis before you even mix it. Always inspect the seal integrity before reconstituting. If the powder appears clumped, discolored, or wet, do not use it. Understanding proper peptide storage after reconstitution helps you avoid these issues.
Formulation type and additives
The specific formulation your pharmacy uses affects stability. Semaglutide compounded with vitamin B12 may have different stability characteristics than plain semaglutide. Formulations containing glycine or methylcobalamin similarly have their own stability profiles.
The pH of the solution also matters enormously. Research has shown that pH is a key factor in the thermal degradation of semaglutide. Solutions at certain pH ranges maintain peptide stability better than others. The buffer type and concentration (molarity) further influence how quickly or slowly degradation occurs.
You typically cannot control these variables, as they are determined by the pharmacy during formulation. But you can ask your pharmacy what formulation they use and whether they have stability data specific to that formulation. A pharmacy that can answer these questions in detail is generally one that takes quality seriously.
Compounding pharmacy quality
Not all compounding pharmacies are equal. The quality of the compounding process directly impacts how long your semaglutide will last. Pharmacies operating in ISO Class 5 cleanroom environments with robust quality control programs produce more consistently stable products than those with less rigorous facilities.
Key quality indicators include: the pharmacy conducts in-house stability testing on their specific formulations, they perform sterility testing on each batch, they follow USP 797 guidelines for sterile compounding, and they can provide certificates of analysis upon request.
Pharmacies like Empower Pharmacy and Olympia Pharmacy are among the better-known compounding pharmacies for semaglutide. Understanding what differentiates quality compounding pharmacies helps you make informed decisions about where to source your medication.
Compounded vs brand-name semaglutide shelf life comparison
The differences between compounded and brand-name semaglutide shelf life are substantial. Understanding them helps you plan your protocol more effectively and avoid the frustration of wasted medication.
Brand-name semaglutide products, specifically Ozempic (for type 2 diabetes management) and Wegovy (for weight management), carry FDA-verified expiration dates. These dates are backed by extensive stability studies conducted by Novo Nordisk, the manufacturer. An unopened Ozempic pen stored in the refrigerator is typically good for up to two years from the date of manufacture. After first use, Novo Nordisk guarantees stability for 56 days whether refrigerated or stored at room temperature below 86 degrees Fahrenheit.
Compounded semaglutide operates in a fundamentally different regulatory framework. It is not FDA-approved. The FDA has not reviewed compounded semaglutide formulations for safety, effectiveness, or quality. Instead, compounding pharmacies operate under state pharmacy board oversight and must follow USP compounding standards.
This regulatory difference is why compounded formulations carry BUDs rather than expiration dates, and why those BUDs tend to be shorter. It is not because the semaglutide itself is different at the molecular level. It is because the testing and verification processes are different.
Factor | Brand-name (Ozempic/Wegovy) | Compounded semaglutide |
|---|---|---|
Regulatory oversight | FDA-approved | State pharmacy boards, USP standards |
Dating type | Expiration date | Beyond-use date (BUD) |
Unopened shelf life | Up to 2 years refrigerated | 30-120 days (varies by pharmacy) |
After first use | 56 days | Typically 28 days |
Stability testing | Multi-year ICH studies | Pharmacy-specific testing (varies) |
Delivery format | Pre-filled pen | Multi-dose vial (usually) |
For people comparing options between brand-name and compounded versions, the semaglutide vs tirzepatide comparison page covers broader differences between GLP-1 receptor agonists. And if you are evaluating whether compounded semaglutide is right for your situation, the GLP-1 and Ozempic relationship guide provides important context.
One practical implication of the shorter compounded shelf life: order smaller quantities more frequently rather than stocking up. A single vial that you can use within 28 days of opening is better than multiple vials that might sit beyond their BUD before you get to them. Use the compounded semaglutide dose chart to calculate how much you actually need per order.
USP 797 guidelines and what they mean for your medication
USP Chapter 797 is the United States Pharmacopeia standard governing the preparation of compounded sterile preparations. If you receive injectable compounded semaglutide, it should have been made in compliance with these standards. Understanding what USP 797 requires gives you insight into the quality controls behind your medication.
The key requirements include cleanroom environment standards, specifically ISO Class 5 or better air quality in the immediate compounding area. This means the air where your semaglutide is mixed contains fewer than 3,520 particles per cubic meter that are 0.5 micrometers or larger. For context, a typical room might have millions of such particles. The cleanroom standard dramatically reduces contamination risk.
Personnel training requirements under USP 797 mandate that pharmacy staff who compound sterile preparations undergo specific training, demonstrate competency through media-fill testing (essentially, proving they can compound without introducing contamination), and maintain ongoing competency through regular re-evaluation.
Environmental monitoring is another critical component. The pharmacy must regularly test the air, surfaces, and personnel in the compounding area for microbial contamination. These results must be documented and reviewed.
Beyond-use dating under USP 797 follows specific rules. If a pharmacy has not conducted sterility testing and stability studies on a particular formulation, USP 797 provides default BUD limits that are quite conservative, often just 14 days or less for certain preparation categories. Pharmacies that want to assign longer BUDs must conduct extended stability testing to justify those dates.
This is why BUDs vary so dramatically between pharmacies. A pharmacy assigning a 14-day BUD might be perfectly compliant with USP 797 but simply using the default limits. A pharmacy assigning a 90-day BUD has likely invested in testing that supports that longer window. Neither is necessarily better or worse, but the difference in usable time is significant for your protocol.
When choosing a compounding pharmacy for your semaglutide dosage protocol, asking about USP 797 compliance is one of the most important questions you can ask.
How to read your compounded semaglutide label
The label on your compounded semaglutide vial contains critical information. Here is what to look for and what each element means.
Beyond-use date (BUD). This is the most important date on the label. It tells you the last day the pharmacy guarantees the medication meets its quality standards. Do not use the medication after this date, even if it looks and smells normal. Degradation at the molecular level is not always visible.
Compounding date. Some labels include the date the medication was compounded. This helps you understand how fresh your supply is. If the compounding date is weeks before you received it, your actual usable window from the time it arrives may be shorter than you expect.
Concentration. Expressed in mg/mL, this tells you how much semaglutide is in each milliliter of solution. Common concentrations include 1 mg/mL, 2 mg/mL, 2.5 mg/mL, and 5 mg/mL. Knowing your concentration is essential for calculating accurate doses using a semaglutide syringe dosage chart.
Total volume. This tells you how much solution is in the vial. Combined with the concentration, this determines the total amount of semaglutide in the vial and how many doses you can draw from it.
Storage instructions. These typically read "refrigerate at 2-8 degrees Celsius" or "store at 36-46 degrees Fahrenheit." Follow these exactly. Deviations shorten shelf life.
Lot number. This identifies the specific batch your vial came from. If you ever need to report a quality concern or the pharmacy issues a recall, this number is essential.
Pharmacy information. The name, address, and contact information of the compounding pharmacy. If you have questions about your specific formulation, contact them directly.
Here is a practical tip. When your vial arrives, immediately write the date you received it and the date you first use it on the vial with a permanent marker or adhesive label. This gives you two reference points: the pharmacy BUD and your personal usage timeline. The earlier of these two dates determines when to stop using the vial.
If you are unsure about unit conversions for your specific concentration, guides like semaglutide units to mg conversion and how many units equal 1 mg help you dose accurately regardless of what concentration your pharmacy provides.
Signs your compounded semaglutide has expired or degraded
Not every degraded vial announces itself with obvious signs. But many do. Knowing what to look for can prevent you from injecting medication that has lost its effectiveness or, worse, that could cause adverse reactions.
Visual changes
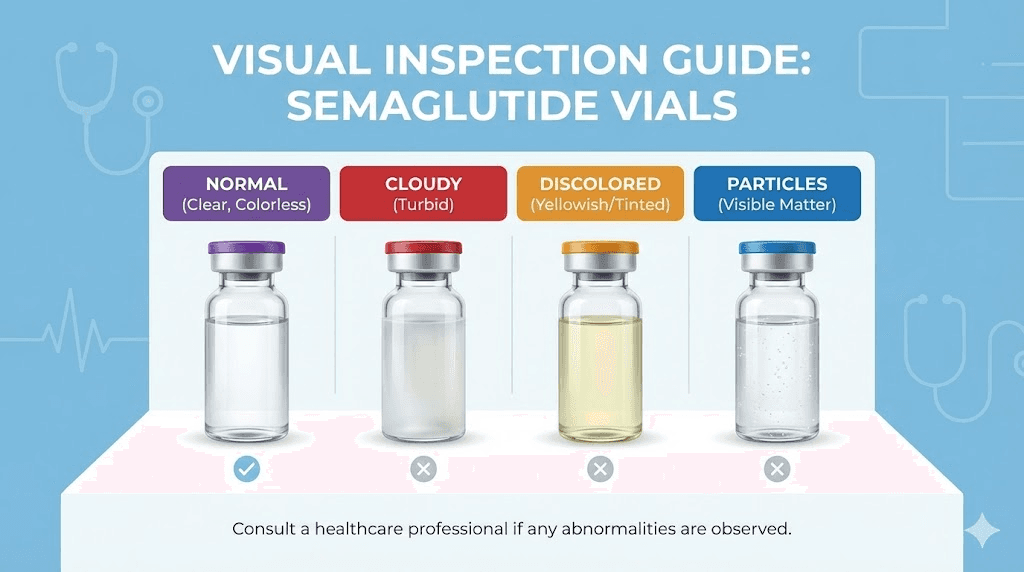
Normal compounded semaglutide solution is clear and colorless. Any deviation from this baseline should raise immediate concern.
Cloudiness or haziness indicates protein aggregation. When semaglutide molecules break down, they can clump together and form visible particles that scatter light. This is one of the most reliable visual indicators of degradation. If you hold the vial up to light and the solution looks anything other than perfectly clear, do not use it.
Discoloration is another warning sign. If your solution has turned yellow, pink, brown, or any other color, chemical degradation has occurred. For reference, our guide on what color semaglutide should be covers normal appearance in detail. And if you notice redness specifically, the guide on red semaglutide explains what that means.
Floating particles, sediment, or visible fibers in the solution indicate contamination or severe degradation. These should never be present in a sterile injectable product. Do not attempt to filter them out or inject around them. Discard the vial.
Odor changes
Normal compounded semaglutide has minimal to no odor. If your bacteriostatic water contains benzyl alcohol as a preservative, you might detect a very faint sweet smell, but nothing strong or unpleasant.
A chemical, foul, or unusual odor indicates breakdown products or microbial contamination. If you notice any strong smell when drawing from the vial, stop immediately. This is one of the clearest signs that the medication is no longer safe to use.
Performance changes
This is the subtle indicator that many people miss. If your semaglutide has been working consistently, with appetite suppression and other expected effects, and then suddenly those effects diminish or disappear, degraded medication could be the cause.
Reduced appetite suppression is often the first functional sign of potency loss. If you find yourself hungrier than usual on the same dose, or if your appetite suppression timeline seems to have shifted, check your vial dates before assuming you need a dose increase.
Weight loss stalling can also indicate degraded medication, though other factors like semaglutide plateau and dietary changes need to be ruled out first. The guide on why you are not losing weight on semaglutide covers all the possible reasons, including medication quality.
Less effective blood sugar management, for those using semaglutide for glycemic control, is another performance indicator. If your blood glucose readings start creeping up on the same dose and diet, expired or degraded medication should be on your list of potential causes.
What happens if you use expired compounded semaglutide
The consequences range from merely ineffective to potentially harmful. Understanding the risk spectrum helps you make informed decisions about vials that are at or near their BUD.
Reduced potency. This is the most common outcome. The active semaglutide peptide has partially degraded, meaning each dose delivers less medication than the label indicates. You might inject what you believe is 0.5 mg but actually receive only 0.3 mg of active semaglutide. The rest has broken down into inactive fragments. This means reduced effectiveness and slower results.
If you have been wondering why four weeks on semaglutide produced no weight loss, degraded medication is a real possibility.
Unpredictable effects. As semaglutide degrades, it can form breakdown products that were not in the original formulation. These degradation products have not been studied for safety. While most peptide degradation products are simply inactive fragments, the possibility of unexpected effects exists.
Contamination risk. Expired compounded medications, particularly liquid formulations that have been opened and used multiple times, carry elevated contamination risk. The preservatives in bacteriostatic water lose effectiveness over time. Bacteria that may have been introduced during needle insertion can multiply. Injecting a contaminated solution can cause injection site reactions, infections, or in severe cases, systemic illness.
Injection site reactions. Degraded medication may cause more irritation at the injection site than fresh medication. If you notice increased redness, swelling, or pain at your injection site that was not present earlier in your protocol, medication quality could be a factor.
For those who have accidentally used expired semaglutide, the accidental expired semaglutide guide covers immediate steps and what to watch for. The key takeaway: using expired medication once is unlikely to cause serious harm, but it should not become a habit. Replace expired vials promptly.
If you are also interested in how expiration affects other GLP-1 medications, the tirzepatide expiration guide and the expired tirzepatide guide cover similar territory for that medication.
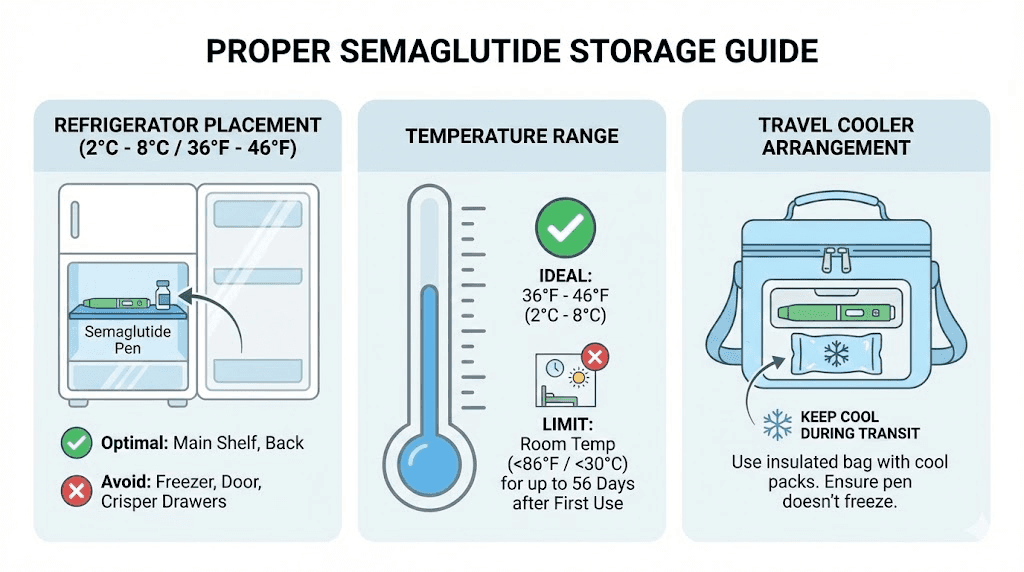
Storage best practices to maximize shelf life
Proper storage is the single most impactful thing you can do to maximize the life of your compounded semaglutide. Every storage decision either extends or shortens the time your medication remains effective.
Refrigeration protocol
Store your semaglutide in the main body of your refrigerator, not in the door. The door compartment experiences the most temperature fluctuation as you open and close the fridge throughout the day. Temperature swings, even brief ones, accelerate peptide degradation.
The ideal location is the middle shelf, toward the back. This area maintains the most consistent temperature. Avoid the very back wall if your refrigerator tends to freeze items there, because freezing semaglutide destroys its molecular structure. Ice crystals form within the solution, physically shearing peptide bonds and denaturing the protein. Frozen semaglutide should be discarded.
Use a refrigerator thermometer to verify your temperature stays within the 36 to 46 degree Fahrenheit range. Many household refrigerators run colder or warmer than their dial settings suggest. A $10 thermometer eliminates the guesswork.
Keep the vial in its original packaging or a small opaque container to protect from light. Every time you open the refrigerator, light enters. While brief exposures are not catastrophic, cumulative light exposure over weeks adds up.
For detailed refrigeration guidance specific to compounded semaglutide, the fridge storage duration guide goes deeper into best practices. And the broader semaglutide refrigerator storage guide covers both compounded and brand-name products.
Handling and injection hygiene
Every interaction with your vial is an opportunity to either maintain or compromise sterility. Follow these protocols consistently.
Always swab the rubber stopper with an alcohol pad before inserting a needle. Wait for the alcohol to dry completely before puncturing. Wet alcohol on the needle can introduce it into the solution, which is not ideal.
Use a new, sterile needle for every draw. Never reuse needles between draws. Besides the obvious infection risk, reused needles can introduce contaminants from the previous injection site back into the vial.
Draw your dose quickly and replace the vial in the refrigerator promptly. Do not leave the vial sitting on the counter while you prepare your injection site, find a mirror, or do anything else. Minimize the time outside refrigeration to the absolute minimum needed for drawing your dose.
Do not touch the rubber stopper with your fingers after swabbing. Do not blow on the needle. Do not set the needle down on any surface before inserting it into the vial. These habits sound minor, but each one represents a potential contamination vector that can shorten your vial life.
For proper injection technique that minimizes vial contamination, the semaglutide injection guide walks through the complete process step by step. And the GLP-1 injection guide covers technique fundamentals.
Travel and transport considerations
Traveling with compounded semaglutide introduces unique challenges. The medication needs to stay cold, protected from light, and handled carefully throughout transit.
A medical-grade travel cooler with gel ice packs is the minimum standard. Insulated lunch bags can work for short trips but lack the thermal stability for all-day travel or air travel. Never pack semaglutide in checked luggage, where temperature extremes in cargo holds can destroy it.
For flights, carry your medication in a small insulated pouch in your personal item. TSA allows injectable medications through security with proper documentation. The traveling with semaglutide guide covers TSA rules, international travel considerations, and packing strategies in detail.
If your semaglutide was exposed to heat during travel, the warm exposure guide helps you assess whether the medication is still usable.
Common mistakes that shorten shelf life
Most people make at least one of these mistakes. Some make several. Correcting them can add days or even weeks to your vial usable life.
Storing in the refrigerator door. Temperature fluctuation in the door compartment is 3 to 5 degrees higher on average than the main body. Over 28 days, those fluctuations accumulate and accelerate degradation. Move your vials to the middle shelf.
Not tracking dates. Without knowing when you received the vial, when the BUD is, and when you first used it, you are guessing at viability. Write dates directly on the vial. Use a permanent marker. Do it the moment the vial arrives and again when you first puncture it.
Leaving the vial out during injection preparation. Some people take the vial out, set it on the counter, prepare their injection site, draw the dose, inject, clean up, and then return the vial. That 10 to 15 minutes at room temperature, repeated every dose for weeks, adds up to hours of unnecessary heat exposure. Draw your dose first, return the vial, then inject.
Using the same needle to draw and inject. Some people draw from the vial and inject with the same needle to save supplies. This practice introduces skin bacteria and tissue particles back into the vial on the next draw. Always use a fresh needle for each step if possible, or at minimum use a new needle for each draw from the vial.
Shaking the vial. Vigorous shaking can cause semaglutide molecules to aggregate at the air-liquid interface, creating foam and accelerating degradation. If you need to mix the solution, roll the vial gently between your palms. Never shake it.
Ignoring visual changes. Many people notice slight cloudiness or a color change and convince themselves it is normal. It is not. Clear and colorless is the standard. Anything else means the medication has changed at a molecular level. Trust your eyes.
Buying more than you can use. Ordering multiple vials to save on shipping or take advantage of volume pricing seems smart. But if the BUD passes before you can use them, you have wasted money instead of saving it. Calculate your exact usage rate based on your vial dosage chart and order accordingly.
Not asking about formulation details. Your pharmacy should be able to tell you the exact formulation, concentration, preservatives used, and the stability data supporting their assigned BUD. If they cannot or will not answer these questions, consider that a red flag. Informed consumers get better outcomes, whether they are working with Direct Meds, BPI Labs, or any other provider.
What to do with expired compounded semaglutide
Do not use it. That is the short answer.
The longer answer involves proper disposal. Injectable medications should not be thrown in regular household trash or flushed down the toilet. The FDA recommends using drug take-back programs when available. Many pharmacies, hospitals, and law enforcement agencies host periodic drug take-back events.
If no take-back program is available, the FDA disposal guidance for household medications recommends mixing the medication with an unappealing substance like coffee grounds or cat litter in a sealed container before placing it in household trash. Remove or scratch out all personal information on the label before disposing.
Needles and syringes used with the expired medication should go in a sharps container. Never throw loose needles in regular trash. Most communities have sharps disposal programs, and many pharmacies accept full sharps containers.
After disposing of expired medication, contact your pharmacy to arrange a fresh supply. This is also a good time to re-evaluate your ordering schedule. If you are consistently finding expired vials, you might be ordering too much or too infrequently. Adjust your timing so that each vial arrives with maximum remaining shelf life and you can use it completely within the BUD window.
Keep a log of when you receive, open, and finish each vial. Over time, this data helps you optimize your ordering pattern and minimize waste. A simple spreadsheet or even a note on your phone works. The goal is never to face the question of whether an expired vial is safe to use, because you have already used it and moved on to a fresh one.
Choosing a compounding pharmacy with proper stability testing
Where you source your compounded semaglutide directly affects how long it lasts and how confident you can be in its quality. Not all compounding pharmacies are equal, and the differences can be significant.
Accreditation. Look for pharmacies accredited by the Pharmacy Compounding Accreditation Board (PCAB) or those that have voluntarily submitted to third-party quality audits. Accreditation is not required by law in most states, which means pharmacies that pursue it are going above the minimum requirements.
Stability testing. Ask whether the pharmacy conducts in-house stability testing on their semaglutide formulation. Pharmacies that do will assign longer, more accurate BUDs based on actual data rather than conservative USP defaults. This testing typically involves analyzing samples at various time points and temperatures to determine how long the formulation maintains its potency and sterility.
Sterility testing. Every batch of injectable medication should undergo sterility testing before release. Ask your pharmacy about their testing protocols. Do they test every batch? Do they use an independent lab or test in-house? What are their acceptance criteria?
Certificate of analysis. A reputable pharmacy should be able to provide a certificate of analysis (COA) for any batch of semaglutide they produce. This document confirms the identity, potency, sterility, and endotoxin levels of the medication. If a pharmacy cannot or will not provide this upon request, that is a concern.
Transparency. Good pharmacies are willing to discuss their processes. They can tell you about their cleanroom classification, their personnel training programs, their environmental monitoring results, and their quality control procedures. Evasive or vague answers suggest a pharmacy that might not be operating at the highest standards.
SeekPeptides members access detailed pharmacy comparison resources and community feedback on sourcing quality compounded medications. This kind of real-world experience data, from people who have actually used these pharmacies, can be more valuable than any marketing claim.
If you are evaluating specific pharmacies, guides on providers like Empower Pharmacy, Olympia Pharmacy, and Strive Pharmacy provide detailed information on what each offers.
Extending shelf life with proper protocol planning
Smart protocol planning minimizes waste and ensures you are always using fresh, potent medication. A few strategic decisions make a big difference.
Calculate your total usage per vial. Before ordering, determine exactly how many doses you will draw from each vial based on your current dosage and the vial concentration. If you are on 0.25 mg of semaglutide during your titration phase, a 5mg vial might last 20 doses, or roughly five months at weekly dosing. That is well beyond the BUD of any compounded formulation. You would need a smaller vial or a more concentrated solution.
Conversely, if you are at a higher dose like 2.4 mg, you will use a 5mg vial in about two weeks. At that rate, shelf life is rarely an issue because you finish the vial long before the BUD.
Match vial size to your dosing phase. During titration (the initial dose-escalation period), your dose changes every four weeks. This makes smaller vials more practical because you are not committing to a large supply at a dose you will soon increase. As you reach your maintenance dose and usage becomes predictable, you can order more efficiently.
Coordinate ordering with your pharmacy BUD. If your pharmacy compounds on specific days, try to time your orders so that you receive the freshest possible product. A vial compounded on Monday and shipped on Tuesday arrives with nearly the full BUD window intact. A vial that sat in the pharmacy for two weeks before shipping has already lost that time.
Consider lyophilized versus liquid formulations. If you have the knowledge and equipment to reconstitute semaglutide yourself, lyophilized powder offers the advantage of much longer pre-reconstitution storage. You can keep the powder for months and only reconstitute what you need for the next 28 days.
Use tools like the semaglutide dosage calculator and peptide cost calculator to determine the most cost-effective ordering strategy that also minimizes waste from expiration.
Semaglutide formulations and how they affect expiration
Not all compounded semaglutide is identical. Different pharmacies use different formulations, and these differences directly impact shelf life and stability.
Plain semaglutide. The simplest formulation contains semaglutide dissolved in a sterile solution, typically with a buffer to maintain optimal pH and bacteriostatic water as the solvent. This is the baseline formulation, and stability data for these preparations is the most widely available.
Semaglutide with B12. Many pharmacies add vitamin B12 (cyanocobalamin or methylcobalamin) to their semaglutide formulations. B12 can add its own stability considerations. The interaction between semaglutide and B12 in solution may affect the degradation profile of both compounds. If you are using a semaglutide with B12 formulation, the BUD assigned should specifically account for the combined stability, not just the semaglutide component.
Semaglutide with glycine. Glycine is sometimes added as a stabilizer or for its own therapeutic benefits. Glycine can actually improve peptide stability in solution by acting as a cryoprotectant and lyoprotectant. However, the specific interactions depend on concentration and formulation details.
Semaglutide with methylcobalamin. Methylcobalamin is a more bioactive form of B12. It is also more light-sensitive than cyanocobalamin, which means formulations containing methylcobalamin may have additional light protection requirements and potentially shorter shelf lives if not stored properly in opaque containers.
The key takeaway: ask your pharmacy specifically about the stability of their particular formulation, not just generic semaglutide stability data. A pharmacy that compounds semaglutide with three additional ingredients should have stability data for that specific combination, not just for semaglutide alone.
For those exploring alternative delivery methods, oral semaglutide drops and sublingual semaglutide have their own distinct stability profiles and storage requirements. These non-injectable formulations avoid some contamination risks but introduce different stability considerations related to their delivery mechanisms.
The science of semaglutide degradation
Understanding what happens to semaglutide at the molecular level as it degrades helps explain why storage conditions matter so much and why expiration dates exist in the first place.
Semaglutide is a 31-amino acid peptide analog of human glucagon-like peptide-1 (GLP-1). It has been modified from the natural GLP-1 sequence to resist degradation by the enzyme DPP-4, which is why it lasts much longer in the body than natural GLP-1. However, these modifications do not make it immune to chemical degradation in storage.
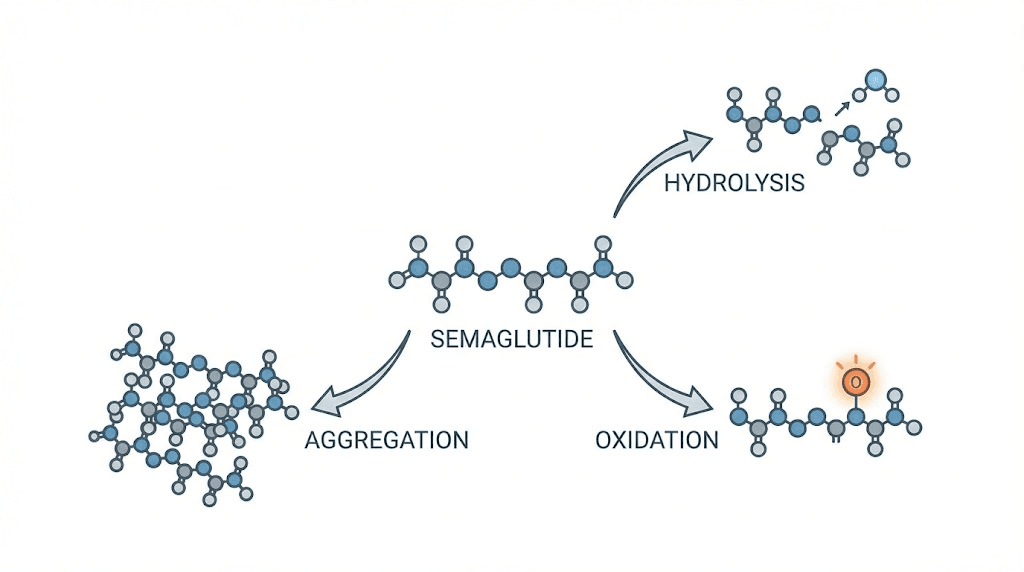
The primary degradation pathways include:
Hydrolysis. Water molecules attack peptide bonds in the amino acid chain, breaking semaglutide into smaller, inactive fragments. This reaction accelerates with higher temperatures and certain pH ranges. It is the dominant degradation pathway in liquid formulations, which is why lyophilized powder lasts longer, because removing water eliminates this pathway until reconstitution.
Oxidation. Oxygen reacts with specific amino acid residues, particularly methionine and tryptophan, altering the molecular structure. Oxidized semaglutide may have reduced receptor binding affinity, meaning it is less effective at activating the GLP-1 receptor. Light exposure accelerates oxidation through photochemical pathways.
Aggregation. Under stress conditions (temperature changes, agitation, air-liquid interface exposure), semaglutide molecules can stick together, forming dimers, trimers, and larger aggregates. These aggregates are less effective than individual molecules and can potentially trigger immune responses. Visible cloudiness in solution is often a sign of significant aggregation.
Deamidation. Asparagine and glutamine residues can undergo deamidation, where the amide group is lost. This changes the charge and structure of the peptide, affecting its biological activity. Deamidation is influenced by pH, temperature, and the surrounding amino acid sequence.
Recent preformulation research examined these pathways systematically. Studies used reverse-phase high-performance liquid chromatography (RP-HPLC) and liquid chromatography high-resolution mass spectrometry (LC-HRMS) to identify and quantify semaglutide degradation products under various stress conditions. This research demonstrated that pH was a key factor in thermal degradation, and that buffer type and strength significantly influenced stability.
For practical purposes, what this means is that your compounded semaglutide is in a constant, slow process of breaking down from the moment it is formulated. Proper storage slows this process dramatically. Improper storage accelerates it. And once enough degradation has occurred, the medication simply does not work as intended.
This is why the vial fridge life guide and the 28-day expiration guide exist. They translate this molecular science into practical timelines you can actually use.
Frequently asked questions
Can I use compounded semaglutide one or two days past the beyond-use date?
It is not recommended. The BUD represents the last day your pharmacy guarantees potency and safety. Going even a day or two past this date means you are in uncharted territory. The medication may still be fine, or it may have degraded past the point of effectiveness. The risk of reduced potency or contamination increases with every day past the BUD. When in doubt, start a fresh vial. For more on this topic, see the complete guide on using expired semaglutide.
Does compounded semaglutide expire faster than brand-name Ozempic?
In terms of assigned dating, yes. Brand-name Ozempic has a much longer shelf life (up to two years unopened, 56 days after first use) compared to compounded versions (30-120 days unopened, 28 days after first use typically). However, this difference largely reflects the different levels of stability testing, not necessarily a difference in the semaglutide molecule itself. The GLP-1 versus Ozempic comparison explores these differences further.
How can I tell how much time is left on my compounded semaglutide?
Check the BUD printed on the label. Then check when you first punctured the vial. The shorter of these two timelines determines your remaining time. If the BUD is 30 days away but you opened the vial 25 days ago, you effectively have about 3 days left (28-day post-opening limit). Track both dates carefully.
Is it safe to use compounded semaglutide that was left out of the fridge for a few hours?
Brief room-temperature exposure of a few hours is generally tolerable, provided the temperature stayed below 86 degrees Fahrenheit (30 degrees Celsius). However, each exposure shortens the remaining shelf life. If you accidentally left semaglutide out overnight, the risks increase significantly. Inspect the solution for visual changes and consider the total warm exposure time when deciding whether to continue using that vial.
Does the formulation type affect how quickly compounded semaglutide expires?
Absolutely. Lyophilized (freeze-dried) powder lasts significantly longer than pre-mixed liquid formulations before reconstitution. Once reconstituted, they have similar shelf lives. Formulations containing additional ingredients like B12 or glycine may have different stability profiles. Always check with your pharmacy for formulation-specific stability data. The lyophilized vs liquid peptide guide covers these differences in depth.
Should I switch pharmacies if my compounded semaglutide has a very short BUD?
A short BUD (14 days or less) might indicate the pharmacy is using conservative USP default limits rather than conducting their own stability testing. This does not necessarily mean the product is lower quality, but it does reduce your practical usage window. If the short BUD creates logistical problems for your protocol, asking the pharmacy about their stability testing or comparing with other providers is reasonable. The compounded semaglutide dosing chart can help you calculate whether a short BUD allows enough time for your specific dose schedule.
Can I freeze compounded semaglutide to extend its shelf life?
No. Never freeze liquid semaglutide. Freezing causes ice crystals to form within the solution, which physically damages the peptide through mechanical shearing of molecular bonds. The freeze-thaw cycle causes protein denaturation and aggregation that cannot be reversed. Frozen semaglutide should be discarded. This applies to both compounded and brand-name formulations.
What should I do if my semaglutide arrives warm from shipping?
Inspect the solution for visual changes (cloudiness, discoloration, particles). If it appears clear and colorless, refrigerate immediately and monitor for changes over the next 24 hours. Contact your pharmacy to ask about their shipping protocols and whether a cold chain was maintained. If you have concerns, request a replacement. The warm semaglutide guide provides a detailed assessment framework.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your storage stay cold, and your protocols stay effective.