Feb 22, 2026

Before you inject another dose from that unmarked vial, stop. Read this first. Because what you do not know about grey market tirzepatide could put you in the emergency room.
That is not exaggeration. The FDA has received more than 545 adverse event reports linked to compounded tirzepatide alone, and grey market products bypass even the minimal oversight that compounding pharmacies provide. Some of those reports involved hospitalizations. Some involved severe hypoglycemia so dangerous that patients needed emergency treatment. And those numbers almost certainly underrepresent the real scope of the problem, because state-licensed pharmacies are not required to report adverse events to the FDA.
The grey market for tirzepatide has exploded. Costs for brand-name Zepbound run around $349 per month at the lowest dose through the manufacturer direct program, and many patients face costs of $1,000 or more when insurance will not cover it. That price gap has pushed thousands of people toward unregulated sources, buying raw active ingredients from overseas and preparing injections at home. The appeal is obvious. The risks are not.
This guide covers everything you need to know about grey market tirzepatide: what it actually is, why the FDA is sounding alarms, what contamination testing reveals about these products, how to spot unsafe sources, and what safer alternatives exist for people who cannot afford brand-name tirzepatide. Whether you are currently using grey market sources or considering them, the information here could save your health.
What grey market tirzepatide actually is
Understanding the grey market requires separating it from two other categories that people often confuse: compounded medications and counterfeit drugs. Each carries different risk levels, and the distinctions matter.
Grey market tirzepatide refers to the active pharmaceutical ingredient purchased through unauthorized channels, typically from overseas chemical suppliers, and prepared by the end user rather than a licensed pharmacy. These products are sold labeled as "for research purposes only," which is a legal workaround that allows suppliers to avoid pharmaceutical regulations. The buyer receives raw peptide powder, reconstitutes it with bacteriostatic water, and self-administers injections without medical supervision.
This is fundamentally different from compounded tirzepatide.
Compounded versions come from licensed 503A or 503B pharmacies that operate under FDA oversight. These pharmacies must follow Current Good Manufacturing Practice regulations, perform sterility and potency testing, and fill patient-specific prescriptions from licensed providers. Compounding is legal and regulated. Grey market purchasing is not.
Then there are outright counterfeits. The FDA has documented cases where products were labeled as coming from specific compounding pharmacies, but the pharmacies listed on the labels either did not exist or had never compounded those products. These fraudulent products represent the most dangerous end of the spectrum, because the buyer has zero information about what the vial actually contains.
How the grey market supply chain works
The typical grey market purchase follows a predictable pattern. A buyer finds a supplier, usually based in China, through online forums or dedicated websites. They order raw tirzepatide powder, often in quantities like 10 vials of 30mg each. The product ships internationally, sometimes disguised as cosmetics or skincare products to avoid customs scrutiny. One NPR report documented a buyer who received his tirzepatide in a box labeled "Multi-effect, young skin set."
Once the powder arrives, the buyer must reconstitute the tirzepatide themselves. This involves calculating the correct amount of bacteriostatic water to add, maintaining sterile technique during mixing, and determining the correct dosage in units for each injection. Every step introduces potential error. Every step lacks professional oversight. And every step assumes the raw powder is actually what the label claims.
The financial incentive is enormous. Brand-name Zepbound costs roughly $349 per month at the lowest dose. One grey market buyer reported spending approximately $350 for what he estimated was a ten-month supply. That price difference is what drives the entire market.
The scale of the problem
Grey market peptide imports from China reached an estimated $328 million in a single year. This is not a niche phenomenon. It is a massive, growing market fueled by the gap between what tirzepatide costs through legitimate channels and what people can afford. The Obesity Action Coalition, the American Association of Clinical Endocrinology, and multiple state attorneys general have all issued warnings about the safety implications of this trend.
Why people turn to grey market tirzepatide
Nobody buys unregulated peptides from overseas for fun. People turn to the grey market because the legitimate healthcare system has failed them in one or more ways, and understanding those failures matters for evaluating the full picture.
Insurance coverage remains the primary barrier. When insurance covers tirzepatide, copays can be as low as $25 per month. When it does not, patients face costs that can exceed $1,000 monthly. And coverage changes happen without warning. The NPR case study documented a patient named Phil who started on Ozempic with a $25 copay, lost weight, improved his bloodwork, and then watched his insurance change and his costs jump to over $1,000 per month. His results on the medication were excellent. His ability to afford it vanished overnight.
That is the pattern. People start tirzepatide or semaglutide through legitimate channels, experience genuine health improvements, and then lose access. For someone who has experienced real tirzepatide results, going back to their previous health situation feels unacceptable. The grey market becomes the alternative they can access.
Supply shortages compound the problem. During periods of high demand, even patients with valid prescriptions and willing insurance cannot always get their medication filled. Compounding pharmacies have partially addressed this gap, but they too face regulatory uncertainty as the FDA navigates the complex legal landscape around peptide regulation.
There is also the access barrier of the medical system itself. Getting a tirzepatide prescription requires a provider visit, which means insurance, time off work, and willingness to discuss weight with a medical professional. For some people, especially those without a primary care provider, the grey market feels simpler.
None of these reasons make grey market use safe. But they explain why rational people make what appears to be an irrational decision.
The real risks of grey market tirzepatide
The risks are not theoretical. They are documented, measured, and in some cases, life-threatening. Breaking them down into categories reveals just how many ways grey market tirzepatide can go wrong.
Contamination and purity failures
A study published in Drug Testing and Analysis examined internet-purchased research peptides and found alarming results. Thirty percent contained incorrect amino acid sequences. That means nearly one in three products was not even the right molecule. The buyer thought they were injecting tirzepatide. They were injecting something else entirely.
Even more concerning: 65% of tested products exceeded safety thresholds for endotoxin contamination. Endotoxins are lipopolysaccharides from bacterial cell walls. They are invisible to standard peptide purity testing methods like HPLC. A product can test at 99% chemical purity while being heavily contaminated with bacterial byproducts that trigger inflammatory responses, fever, and in severe cases, septic shock.
This distinction between purity and sterility is critical.
Chemical purity measures whether the expected peptide molecule is present. Sterility testing measures whether the product is free from living microorganisms. Endotoxin testing measures whether bacterial cell wall fragments are present below dangerous thresholds. Grey market products might pass the first test and fail the other two spectacularly. And most grey market suppliers only provide HPLC purity certificates, if they provide any testing documentation at all.
The FDA has received reports of compounded and grey market tirzepatide products arriving with visible contamination, improper coloring, and particulate matter in solution. If products from regulated compounding pharmacies can fail safety standards, imagine the failure rates for products with zero regulatory oversight.
Dosing errors and their consequences
Self-dosing tirzepatide without medical supervision is inherently risky. The tirzepatide dosing protocol involves carefully titrated escalation from 2.5mg to potentially 15mg over many weeks. Getting this wrong is not a minor inconvenience.
Counterfeit tirzepatide products have been found with wildly inconsistent concentrations. Some contained zero active ingredient. Others contained dangerously high doses that triggered severe hypoglycemia requiring emergency treatment. When you reconstitute a grey market vial, you are trusting that the powder inside is accurately dosed to the labeled amount. Without independent verification, that trust is blind.
The math matters here. If a vial is labeled as 30mg but actually contains 45mg, every injection delivers 50% more than intended. For someone following a tirzepatide dosage calculator based on the labeled amount, they would unknowingly be taking a significantly higher dose. At 15mg intended, they would actually receive 22.5mg, a dose that has never been studied in clinical trials.
The FDA specifically noted that "multiple reports of adverse events, some requiring hospitalization" were related to dosing errors with compounded and grey market injectable tirzepatide products. These are not abstract risks. They are sending people to emergency rooms.
No medical oversight
A prescription from a licensed provider is not just a permission slip. It is a safety checkpoint. Before prescribing tirzepatide, a provider evaluates whether the patient has contraindications, checks for drug interactions, establishes baseline bloodwork, and creates a monitoring plan for side effects.
Grey market users skip all of that.
They do not get screened for conditions that make GLP-1 receptor agonists dangerous, like a personal or family history of medullary thyroid carcinoma. They do not get monitored for gastrointestinal side effects that could signal more serious complications. They do not have a provider who can recognize early signs of pancreatitis, a potentially life-threatening condition that GLP-1 medications can trigger.
Dr. Scott Isaacs, president of the American Association of Clinical Endocrinology, summarized the risks plainly: "They could get an infection. They could have a reaction. They could have severe side effects from dosing errors." Dr. Tony Cirillo, president of the American College of Emergency Physicians, emphasized that these "are not benign medications without proper monitoring and supervision."
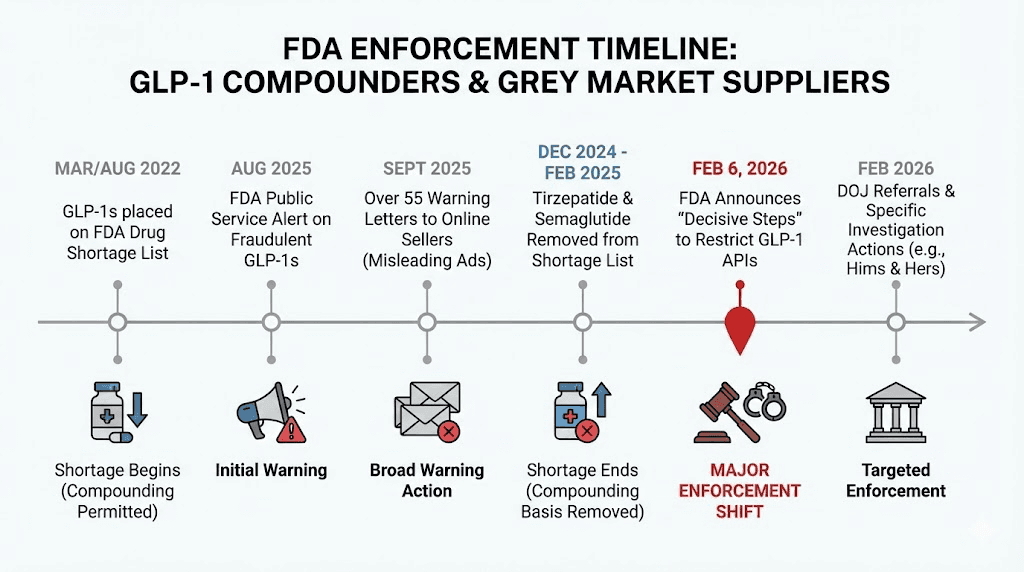
FDA warnings and adverse event data
The federal government has been increasingly vocal about the dangers of unregulated GLP-1 receptor agonists. The data behind their warnings deserves careful examination.
The numbers
As of July 31, 2025, the FDA had received a combined 1,150 adverse event reports related to compounded versions of semaglutide and tirzepatide. Breaking that down: 605 reports for compounded semaglutide and 545 reports for compounded tirzepatide. And these numbers come with an enormous caveat. Federal law does not require state-licensed pharmacies that are not outsourcing facilities to submit adverse events to the FDA. The actual number of adverse events is almost certainly much higher.
These reports include cases of severe pancreatitis requiring hospitalization, gallbladder disease, severe hypoglycemia with dangerously low blood sugar, and acute kidney injury from dehydration caused by gastrointestinal effects. The kidney injury pathway is particularly insidious: tirzepatide can cause nausea, vomiting, and diarrhea as normal side effects. Without medical monitoring, these symptoms can lead to dehydration severe enough to damage the kidneys.
Fraudulent product warnings
The FDA has identified specific instances of fraud in the tirzepatide supply chain. In some cases, the compounding pharmacies listed on product labels did not actually exist. In others, real licensed pharmacies were named on labels but had never compounded those specific products. Someone was slapping legitimate pharmacy names on fraudulent vials.
One documented adverse event from a fraudulent compounded tirzepatide product included redness, site swelling, pain, and a red lump at the injection site. While that particular case was not life-threatening, it illustrates that even seemingly minor reactions can signal serious problems with product quality, problems that would be invisible to the person self-administering an injection from an unverified source.
Warning letters and enforcement actions
The FDA has dramatically increased enforcement activity. Warning letters to GLP-1 compounders increased 50% in a single fiscal year. The agency sent warning letters to more than 50 compounding pharmacies and manufacturers, citing violations ranging from sterility failures to subpotent products to outright insanitary conditions.
One notable case involved a Pennsylvania compounding pharmacy inspected by the FDA. Between July of one year and May of the next, the facility distributed sterile drug products, including tirzepatide injectables, with multiple lots failing both sterility and potency testing. A patient received an injectable tirzepatide product that the pharmacy's own laboratory had determined was not sterile. The pharmacy did not notify the patient.
If a regulated, inspected compounding pharmacy can fail this badly, consider what happens with grey market suppliers who face zero inspection, zero testing requirements, and zero accountability.
Grey market versus compounded versus brand-name tirzepatide
Not all non-brand-name tirzepatide is created equal. Understanding the spectrum from safest to most dangerous helps contextualize where grey market products fall.
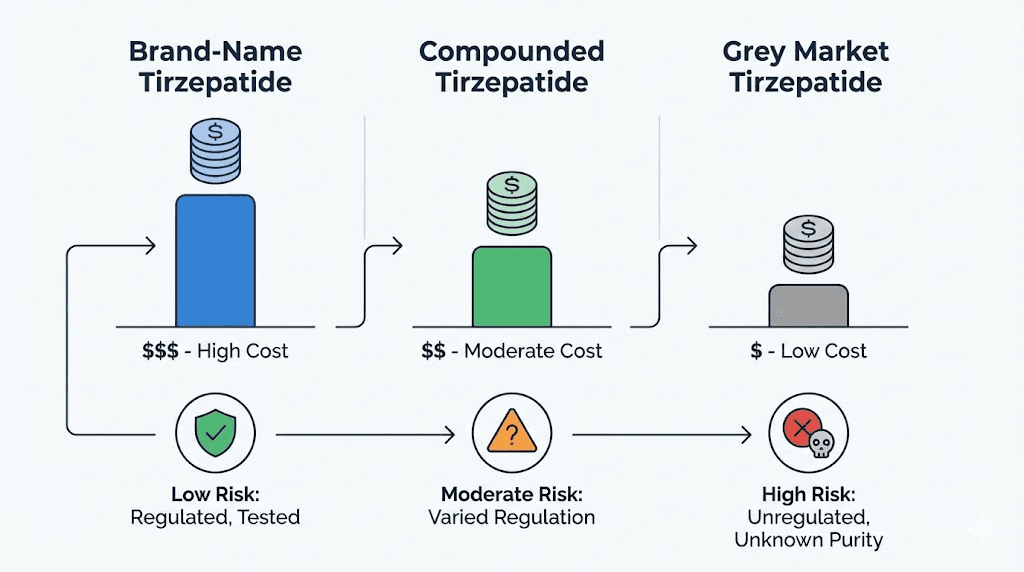
Brand-name tirzepatide (Mounjaro and Zepbound)
This is the gold standard. Manufactured by Eli Lilly under strict FDA oversight, every batch undergoes rigorous testing for potency, sterility, endotoxin levels, and stability. The dosing is precise and standardized. The delivery mechanism, a prefilled pen, eliminates reconstitution errors entirely. The cost is the barrier, not the quality.
Compounded tirzepatide from licensed pharmacies
Compounded versions are prepared by licensed 503A or 503B pharmacies based on valid prescriptions from licensed providers. These pharmacies must follow Good Manufacturing Practice standards, though the rigor of oversight varies. Quality compounding pharmacies perform batch testing and can provide Certificates of Analysis. The products are meaningfully safer than grey market sources, but they are not identical to FDA-approved brand-name medications.
The main risks with compounded tirzepatide involve potency variation between batches, potential sterility issues at lower-quality compounding facilities, and the fact that compounded versions may use different salt forms of the active ingredient. The FDA has noted that semaglutide sodium and semaglutide acetate are different active ingredients than those used in approved drugs, and similar concerns may apply to compounded tirzepatide formulations.
Grey market tirzepatide
This category represents the highest risk. No regulatory oversight. No required testing. No prescription. No medical supervision. The buyer becomes their own pharmacist, their own prescriber, and their own quality control department, roles they are almost certainly not qualified to fill.
Here is how the three options compare across key safety factors:
Factor | Brand-name | Compounded | Grey market |
|---|---|---|---|
FDA oversight | Full approval | Regulated under 503A/503B | None |
Sterility testing | Every batch | Required, frequency varies | Not performed |
Potency verification | Precise, standardized | Batch-tested | Unknown |
Endotoxin testing | Below USP limits | Usually performed | Not performed |
Medical oversight | Required prescription | Required prescription | None |
Adverse event reporting | Mandatory | Varies by pharmacy type | None |
Monthly cost | $349-$1,000+ | $200-$500 | $35-$50 |
Risk level | Lowest | Low to moderate | Highest |
The price column tells the story of why the grey market exists. But the risk column tells the story of why it should not.
How to identify grey market tirzepatide
Knowing the warning signs can protect you or someone you know from accidentally purchasing grey market products when they believe they are getting legitimate compounded tirzepatide.
Red flags in purchasing
The most obvious indicator is the "research purposes only" label. This phrase is the legal shield grey market suppliers use. If a product is explicitly labeled as not for human use, it is not regulated as a pharmaceutical product. Any tirzepatide sold with this designation is grey market by definition, regardless of how the seller markets it.
Other warning signs include no prescription requirement, direct sales from overseas suppliers, payment methods that avoid traditional banking like cryptocurrency, products shipped in nondescript packaging without proper pharmaceutical labeling, and prices that seem impossibly low compared to even the most affordable legitimate sources.
Websites dedicated to grey market sourcing guides exist openly. They provide supplier recommendations, dosing calculators, and community-verified purity results. The infrastructure looks professional. The quality control is nonexistent.
Red flags in the product itself
Physical product red flags include vials without proper pharmaceutical labeling, missing lot numbers or expiration dates, powder that appears discolored or clumped, reconstituted solutions that are cloudy or contain visible particles, and packaging that does not match pharmaceutical standards.
Documentation red flags include Certificates of Analysis from unknown or unverifiable testing laboratories, HPLC purity results without corresponding endotoxin or sterility testing, and testing performed by the seller rather than an independent third-party laboratory.
A product can have a 99% HPLC purity result and still be dangerous. Purity testing measures chemical composition. It does not measure sterility. It does not measure endotoxin levels. It does not verify that the product is safe for injection into a human body. Understanding peptide testing limitations is essential for anyone evaluating product quality claims.
Red flags in the seller
Legitimate compounding pharmacies have verifiable state licenses, physical addresses, and licensed pharmacists on staff. They require prescriptions from licensed providers. They can be looked up through state boards of pharmacy.
Grey market sellers operate through websites that may lack physical addresses, use customer service through messaging apps rather than phone lines, cannot provide verifiable pharmacy licenses, and change domain names frequently. Some sellers use social media platforms and encrypted messaging groups as their primary sales channels.

What happens when the dose is wrong
Dosing errors with tirzepatide are not minor inconveniences. They can be medically dangerous, and the grey market makes them far more likely.
The reconstitution problem
Brand-name tirzepatide comes in a prefilled pen. The dose is set by the manufacturer. There is no reconstitution, no calculation, no room for error.
Grey market tirzepatide arrives as a lyophilized powder. The user must add the correct volume of bacteriostatic water, mix without damaging the peptide, and then calculate how many units to draw in a syringe for each dose. Each step introduces error.
Consider a common scenario. A buyer receives a vial labeled as 30mg of tirzepatide. They add 3ml of bacteriostatic water, creating a concentration of 10mg per ml. They want a 2.5mg dose, so they draw 25 units in an insulin syringe (0.25ml). Simple math.
But what if the vial actually contains 20mg instead of 30mg? Now their 25-unit draw delivers only 1.67mg instead of 2.5mg. They wonder why the tirzepatide is not working. They increase the dose. They add more units. And they have no idea their baseline calculation was wrong from the start.
Flip the scenario. The vial contains 45mg instead of 30mg. Their 25-unit draw now delivers 3.75mg instead of 2.5mg. That is a 50% overdose from day one. By the time they escalate to what they think is 10mg, they are actually taking 15mg, the maximum dose studied in clinical trials, without the gradual titration that makes higher doses safer.
What overdosing looks like
Tirzepatide overdose can cause severe nausea and vomiting leading to dehydration, hypoglycemia (dangerously low blood sugar) especially in people who also take diabetes medications, acute pancreatitis requiring hospitalization, severe gastrointestinal symptoms including uncontrollable diarrhea, and cardiovascular effects from rapid fluid and electrolyte shifts.
The hypoglycemia risk deserves special attention. While tirzepatide alone rarely causes severe hypoglycemia in non-diabetic individuals, an overdose in someone who is also taking sulfonylureas, insulin, or other glucose-lowering medications can create a medical emergency. Blood sugar can drop low enough to cause seizures, loss of consciousness, and brain damage. Without medical monitoring, the person may not recognize the symptoms until they are in crisis.
What underdosing causes
Underdosing seems less dangerous, and physically it usually is. But the downstream effects create their own problems. Someone taking sub-therapeutic doses experiences minimal appetite suppression and minimal weight loss results. They conclude that tirzepatide does not work for them. They may increase the dose beyond what would be appropriate, creating the overdose scenario described above. Or they may abandon treatment entirely, losing the metabolic benefits that a properly dosed protocol would have provided.
The FDA has noted that lack of therapeutic effect is "probably the biggest thing" clinicians observe with grey market products. People are injecting substances that either contain too little active ingredient or the wrong molecule entirely, and they interpret the lack of results as a personal failure rather than a product quality failure.
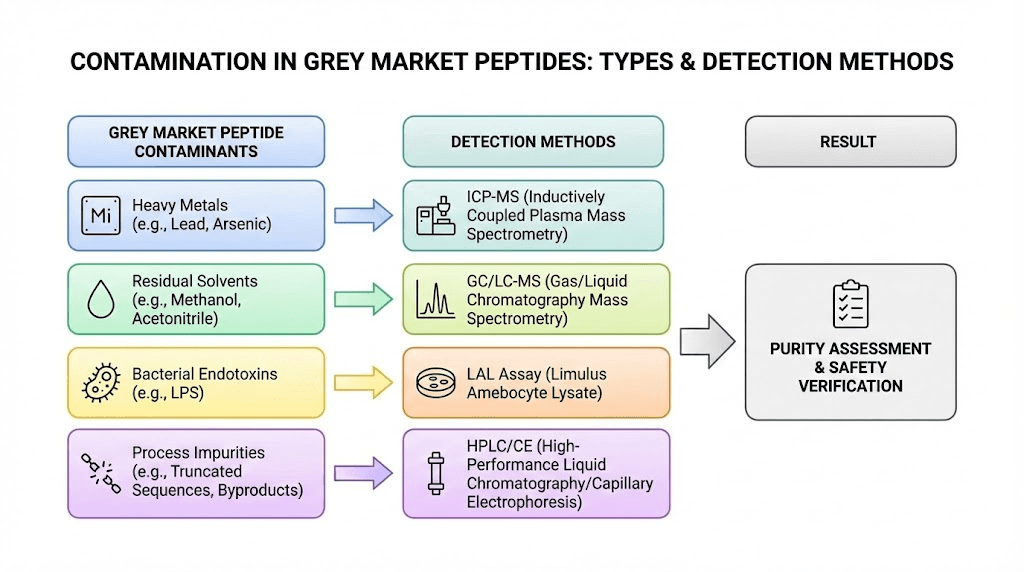
The contamination problem in detail
Contamination in grey market tirzepatide takes multiple forms, each with distinct health implications. Understanding these categories helps explain why a "99% pure" label means far less than most buyers assume.
Bacterial contamination
Sterile manufacturing requires cleanroom environments, validated sterilization procedures, and rigorous environmental monitoring. Grey market suppliers in countries without pharmaceutical oversight may produce peptides in facilities that would horrify a pharmacist. The equipment may not be properly sterilized between batches. The water used in processing may contain microorganisms. The packaging may introduce contamination during filling.
Injecting a bacterially contaminated solution can cause injection site infections ranging from mild redness to serious abscesses, systemic infections if bacteria enter the bloodstream, and immune reactions that mimic allergic responses. For someone injecting tirzepatide subcutaneously, even a localized infection can require medical treatment and antibiotics. Systemic infections from contaminated injectables can become life-threatening.
Endotoxin contamination
This is the hidden danger that even informed grey market users often miss. Endotoxins are fragments of bacterial cell walls (lipopolysaccharides) that persist even after bacteria are killed. A product can be technically sterile, containing no living bacteria, while still containing dangerous levels of endotoxins.
Endotoxin testing uses the Limulus Amebocyte Lysate (LAL) assay, which measures contamination in Endotoxin Units (EU). The research finding that 65% of internet-purchased peptides exceeded safety thresholds for endotoxin contamination is staggering. It means that even if the peptide itself is pure and the solution is technically sterile, the endotoxin load can cause fever, chills, inflammatory responses, and in extreme cases, endotoxic shock.
Standard HPLC purity testing does not detect endotoxins at all. A grey market supplier can truthfully claim their product is 99% pure by HPLC while that same product contains endotoxin levels that would disqualify it from any legitimate pharmaceutical application. This is why quality peptide handling and storage alone cannot make a contaminated product safe.
Wrong molecule entirely
Thirty percent of tested research peptides contained incorrect amino acid sequences. That statistic deserves to sink in. One in three products was not the molecule the buyer ordered.
Tirzepatide is a complex 39-amino acid peptide with a specific fatty acid moiety that enables its dual GIP/GLP-1 receptor agonist activity. Synthesizing it correctly requires sophisticated equipment and quality control. A mistake in the amino acid sequence could produce a molecule with reduced activity, no activity, or unexpected and potentially dangerous biological effects. The buyer would have no way of knowing.
Mass spectrometry can verify molecular identity, but it is expensive testing that most grey market suppliers do not perform or provide. Without it, you are trusting that an unregulated overseas lab correctly synthesized one of the most complex peptide drugs on the market. That is a significant amount of trust to place in a supplier you found on the internet.
Degradation products
Even if a grey market product starts pure, it may not stay that way. Tirzepatide stability depends on proper storage conditions, which grey market products may not receive during manufacturing, shipping, or storage. The FDA has received complaints about compounded GLP-1 drugs arriving warm or with inadequate ice packs, and grey market products shipped internationally face even more extreme temperature excursions.
Degraded tirzepatide may produce peptide fragments with unknown biological activity. It may lose potency unpredictably. And degradation products can trigger immune reactions, including the development of anti-drug antibodies that could affect a person's response to legitimate tirzepatide in the future.
Proper tirzepatide refrigeration is essential for maintaining potency, but the cold chain for grey market products is essentially nonexistent. A vial that sits in a hot warehouse for weeks before shipping, travels unrefrigerated across an ocean, and then sits in a mailbox on a summer afternoon has likely experienced temperature conditions that would render it ineffective or dangerous.
Legal considerations for grey market tirzepatide
The legal landscape surrounding grey market tirzepatide is more complex than most buyers realize, and the consequences extend beyond health risks.
Is buying grey market tirzepatide legal?
The short answer: it occupies a legal grey zone, which is how it got its name. Grey market tirzepatide is typically sold as a "research chemical" not intended for human use. This allows suppliers to operate outside pharmaceutical regulations. However, purchasing a prescription drug without a prescription and administering it to yourself is not legally sanctioned, regardless of how the product is labeled.
The FDA can and does take enforcement action against suppliers. State attorneys general have also gotten involved. Connecticut's attorney general sued a GLP-1 distributor for marketing grey market products. As regulatory attention increases, both sellers and buyers face growing legal exposure.
Understanding peptide legality requires recognizing that the rules are evolving rapidly. What was tolerated yesterday may be prosecuted tomorrow, and buyers who stockpiled grey market tirzepatide could find themselves holding products that are explicitly illegal to possess.
The liability vacuum
When something goes wrong with an FDA-approved drug, there are pathways for recourse. The manufacturer carries product liability insurance. The prescribing physician carries malpractice insurance. The pharmacy carries professional liability coverage. There are adverse event reporting systems that trigger investigations and recalls.
With grey market tirzepatide, none of those protections exist. If a contaminated vial causes an infection, there is no one to hold accountable. If an overdosed product causes hospitalization, the overseas supplier is beyond the reach of American courts. If a product turns out to be counterfeit, there is no recall system to notify other buyers.
The buyer assumes all risk. All of it. With no safety net, no insurance, and no legal recourse.
Safer alternatives to grey market tirzepatide
For people who cannot afford brand-name tirzepatide, safer options exist. None are as cheap as grey market sources, but all provide meaningfully better safety profiles.
Compounded tirzepatide from licensed pharmacies
The most direct alternative. Compounded tirzepatide from licensed 503A or 503B pharmacies requires a prescription but costs significantly less than brand-name. Prices typically range from $200 to $500 per month depending on the dose and pharmacy. These products undergo sterility and potency testing, are prepared under regulated conditions, and come with the oversight of a licensed pharmacist.
The regulatory landscape for compounded tirzepatide has shifted as the FDA addresses drug shortage designations, so availability may vary. Checking with multiple pharmacies and telehealth providers gives you the best chance of finding current options.
Manufacturer patient assistance programs
Eli Lilly offers savings programs for eligible patients. These can significantly reduce costs for brand-name Mounjaro and Zepbound. The specific programs and eligibility criteria change, so checking the manufacturer website directly provides the most current information.
Telehealth platforms with compounding pharmacy partnerships
Several telehealth platforms connect patients with licensed providers who can prescribe compounded tirzepatide and partner with vetted compounding pharmacies. These services typically bundle the provider consultation and medication into a single monthly cost. While not as cheap as grey market sources, they provide medical oversight, legitimate prescriptions, and regulated products.
Alternative GLP-1 medications
If tirzepatide specifically is inaccessible, other GLP-1 receptor agonists may be available at lower cost. Semaglutide is available in both brand-name and compounded versions. Orforglipron, an oral GLP-1 under development, may eventually provide another option. Discussing alternatives with a healthcare provider ensures you explore all legitimate pathways before considering unregulated sources.
Lifestyle and supplemental approaches while waiting
For people who previously used tirzepatide and lost access, maintaining results through dietary and exercise strategies can bridge the gap. Understanding which foods to avoid, following a structured tirzepatide-style diet plan, and incorporating appropriate supplements can support metabolic health while you work toward regaining access to legitimate medication.
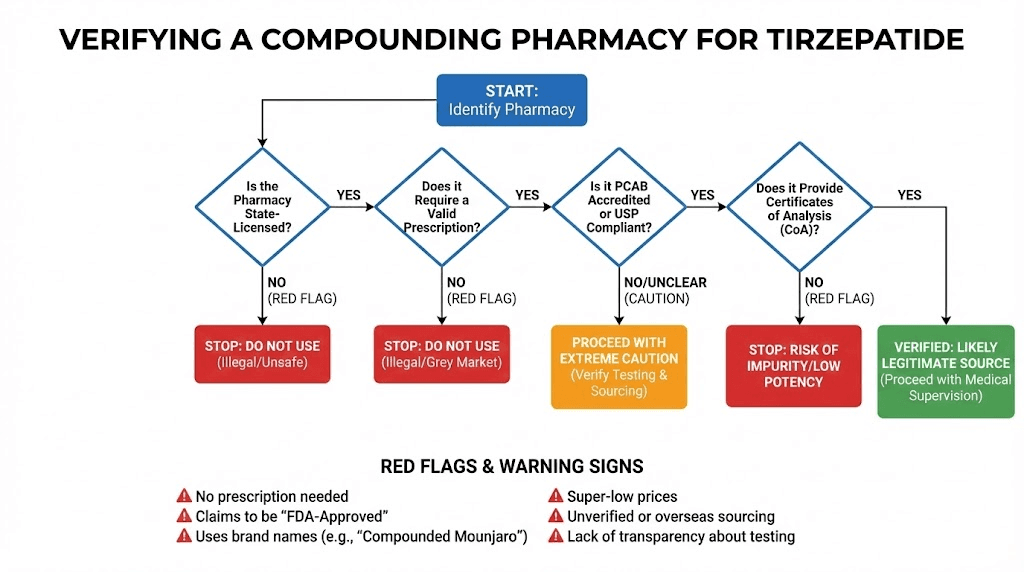
How to verify a legitimate compounding pharmacy
If you choose compounded tirzepatide, verifying your pharmacy is essential. Not all compounding pharmacies are equal, and the FDA warning letters demonstrate that even licensed facilities can have serious quality issues.
Verification steps
Start with the basics. Confirm the pharmacy holds a valid state license through your state board of pharmacy website. Check whether the pharmacy is a 503A (patient-specific compounding) or 503B (outsourcing facility) operation. 503B facilities face more stringent FDA oversight, including regular inspections.
Ask the pharmacy directly about their testing protocols. A quality compounding pharmacy should perform potency testing on each batch, sterility testing using validated methods, endotoxin testing (LAL assay) on injectable products, and beyond-use dating studies to verify how long their products remain effective. They should provide Certificates of Analysis upon request showing results from independent third-party laboratories, not just in-house testing.
Questions to ask
When evaluating a compounding pharmacy for compounded tirzepatide, ask these specific questions:
Where do you source your active pharmaceutical ingredient? Legitimate pharmacies use FDA-registered suppliers with drug master files. What testing do you perform on each batch? Look for potency, sterility, and endotoxin testing at minimum. Can you provide a Certificate of Analysis from a third-party laboratory? In-house testing alone is less reliable. What is your beyond-use dating, and what studies support it? This tells you how long the product remains effective after compounding. Have you received any FDA warning letters? This is public information, but asking directly shows you are an informed consumer. What is your protocol if a batch fails testing? Quality pharmacies have documented procedures for handling failures, including patient notification.
Red flags in compounding pharmacies
Be wary of pharmacies that cannot or will not provide testing documentation, offer prices significantly below market rates for compounded tirzepatide, do not require a valid prescription from a licensed provider, market directly to consumers through social media advertising, or cannot provide verifiable licensing information.
The FDA warning letter case study is instructive here. A licensed, inspected pharmacy released products that failed sterility testing without notifying patients. If that can happen at a regulated facility, imagine the standards at facilities you cannot verify.
Storage and handling risks with grey market products
Even if a grey market product starts in acceptable condition, maintaining it requires knowledge and equipment that many buyers lack.
Cold chain failures
Tirzepatide requires refrigeration. The shelf life in the fridge depends on proper storage from the moment of manufacture through delivery to your home. Grey market products may spend days or weeks in transit without adequate cold packaging. International shipping routes can expose packages to extreme heat. And even domestic delivery from an overseas warehouse may involve sitting in a hot delivery truck for hours.
The FDA specifically flagged that compounded GLP-1 drugs have arrived warm or with inadequate ice packs. Grey market products face the same risks multiplied by longer shipping times, less professional packaging, and zero quality monitoring during transit.
Once you receive a grey market vial, you have no way of knowing what temperatures it experienced. The powder may look normal. The reconstituted solution may appear clear. But if the peptide degraded during transit, potency is reduced and degradation products may be present. Understanding proper tirzepatide temperature stability is important, but it cannot fix damage that already occurred before you opened the package.
Reconstitution contamination
The process of reconstituting tirzepatide at home introduces contamination risks that professional compounding environments are designed to eliminate. Without a laminar flow hood, sterile gowning, and validated technique, bacteria from your environment can enter the vial during reconstitution. Your kitchen, bathroom, or desk is not a cleanroom. Every time the vial septum is pierced with a needle, there is a chance of introducing microorganisms.
Multi-dose vials that are accessed repeatedly over days or weeks accumulate contamination risk with each needle insertion. This is why bacteriostatic water contains a preservative (benzyl alcohol) that inhibits bacterial growth. But that preservative has limits. It slows bacterial growth rather than eliminating it entirely, and it does nothing to prevent the introduction of endotoxins or other non-living contaminants.
Dosing accuracy with consumer-grade equipment
Professional pharmacies use calibrated measuring equipment and validated procedures to ensure dose accuracy. A grey market user works with consumer-grade insulin syringes and mental math. The margin for error is significant, especially at lower doses where small volume differences translate to large percentage changes in the delivered dose.
Understanding how many units correspond to each mg requires knowing the exact concentration after reconstitution, which depends on the actual (not labeled) amount of peptide in the vial and the exact volume of water added. A syringe that measures to the nearest unit has an inherent imprecision that compounds with the uncertainty in the vial contents.
For context, the difference between 5mg and 7.5mg of tirzepatide is a clinically meaningful dose escalation. With consumer-grade equipment and uncertain vial contents, hitting those targets precisely is extremely difficult.
The broader context: why this problem exists
Grey market tirzepatide is a symptom of a larger healthcare access problem. Addressing it honestly means acknowledging the systemic failures that create demand for unsafe alternatives.
The cost crisis
GLP-1 receptor agonists represent some of the most effective treatments ever developed for obesity and type 2 diabetes. Clinical trials show tirzepatide producing significant weight loss results across diverse patient populations. For many people, these medications are transformative.
But transformative medications that cost $349 to $1,000+ per month, that insurers may or may not cover, that can be denied or discontinued at any time, create a painful gap between medical need and financial reality. The grey market fills that gap with cheaper, more accessible, and far more dangerous products.
The information asymmetry
Grey market communities often present themselves as informed, science-literate groups making rational decisions based on available data. Many participants genuinely try to verify product quality through third-party testing, community-shared lab results, and careful dosing protocols. This creates an illusion of safety that can be more dangerous than obvious quackery.
The problem is that even the best community testing cannot replicate the comprehensive quality assurance of regulated manufacturing. An HPLC test confirms chemical purity but not sterility. A mass spectrometry result confirms molecular identity but not endotoxin levels. Batch-to-batch consistency cannot be verified from a single test of a single sample. The testing that grey market communities perform is better than nothing, but dramatically short of what would be required for a product intended for injection into a human body.
The role of social media and forums
Online communities normalize grey market use. Success stories are amplified. Adverse events are underreported or attributed to user error. Suppliers cultivate reputations through community engagement, creating a trust dynamic that mimics but does not replicate the trust warranted by regulatory oversight.
Peptide forums and discussion communities serve valuable purposes for information sharing. But they can also create echo chambers where risk normalization occurs gradually. When everyone in a community is using grey market products without apparent problems, the perceived risk drops even as the actual risk remains constant.
What to do if you are currently using grey market tirzepatide
If you are reading this and currently using grey market tirzepatide, this section is for you. The goal is not to shame or judge. It is to help you transition to a safer approach.
Immediate steps
Schedule an appointment with a healthcare provider. Be honest about your grey market use. Most providers would rather help you transition to a safer option than have you continue using unregulated products. They can perform bloodwork to check your metabolic health, screen for complications, and write a prescription for a legitimate tirzepatide protocol.
If you experience any concerning symptoms, including unusual muscle pain, severe gastrointestinal symptoms, persistent headaches, signs of infection at injection sites, or unexplained body aches, seek medical attention immediately. Do not minimize symptoms because you are embarrassed about the source of your medication.
Transition planning
A cold-turkey stop from tirzepatide can cause rebound appetite and rapid weight regain. Work with a provider to either transition to a legitimate source of tirzepatide or taper your dose gradually while implementing lifestyle strategies. The dietary approaches that support tirzepatide use remain valuable during and after a transition period.
If cost is the barrier, explore compounded tirzepatide options, manufacturer assistance programs, and alternative GLP-1 medications with your provider. The tirzepatide market is evolving rapidly, and options that were unavailable months ago may now be accessible. Insurance coverage for GLP-1 medications continues to expand as more data demonstrates their health benefits beyond weight loss.
Reporting adverse events
If you experienced a negative reaction to a grey market or compounded tirzepatide product, consider reporting it to the FDA MedWatch program. These reports contribute to the data that drives regulatory action and helps protect others. Reports can be filed anonymously, and the FDA does not share reporter information with product manufacturers or law enforcement.
How SeekPeptides approaches peptide safety
Navigating the complex landscape of peptide sourcing, quality verification, and safe protocols requires reliable information. SeekPeptides exists specifically to fill this gap, providing evidence-based guidance that helps people make informed decisions about peptide research without relying on grey market communities or unverified sources.
Members access comprehensive dosing guides, detailed storage protocols, and safety frameworks built on published research rather than forum speculation. For anyone serious about understanding peptides safely, SeekPeptides offers the most comprehensive resource available, with protocols, calculators, and a community of thousands who prioritize doing things correctly.
Frequently asked questions
Is grey market tirzepatide the same molecule as Mounjaro or Zepbound?
It is supposed to be, but testing shows that 30% of internet-purchased research peptides contain incorrect amino acid sequences. Without independent verification through mass spectrometry, there is no way to confirm a grey market product contains the correct molecule. Even if the amino acid sequence is correct, differences in formulation, purity, and contaminant levels mean the product is not equivalent to properly manufactured tirzepatide.
Can I test grey market tirzepatide to make sure it is safe?
You can send samples to independent laboratories for HPLC purity testing and mass spectrometry identification. However, comprehensive testing including sterility and endotoxin assays is expensive, often costing more per test than the product itself. And testing a single vial does not guarantee that other vials from the same batch are identical. The only way to ensure product safety across all batches is the systematic quality control that regulated manufacturing provides.
What if I have been using grey market tirzepatide with no problems?
Absence of obvious adverse effects does not confirm product safety. Sub-therapeutic dosing may produce no noticeable effects, positive or negative. Low-level endotoxin contamination may cause chronic low-grade inflammation without acute symptoms. And the risk of receiving a contaminated batch exists with every new purchase, regardless of previous experiences. The NPR story documented a buyer who reported no problems over two years. That individual outcome does not change the population-level data showing significant contamination rates and adverse events.
How do grey market prices compare to compounded tirzepatide?
Grey market tirzepatide can cost as little as $35 to $50 per month. Legitimate compounded tirzepatide typically costs $200 to $500 per month. Brand-name Zepbound costs approximately $349 per month at the lowest dose through the manufacturer program, and potentially over $1,000 without insurance. The price gap is real, but it reflects the cost of manufacturing standards, testing, medical oversight, and regulatory compliance.
Can I switch from grey market to compounded tirzepatide without starting over?
Yes. A licensed provider can evaluate your current dosing level and prescribe a compounded product at an equivalent dose. You do not need to restart from the lowest dose if you have been stable on a higher dose, though your provider may adjust based on their clinical judgment. Honest communication about your grey market history helps them make the best decision.
Does the FDA distinguish between grey market and compounded products in their warnings?
The FDA uses the terms somewhat interchangeably in public communications, often grouping "compounded, counterfeit, and gray-market" products together. Their adverse event data captures reports from all non-brand-name sources. However, the agency clearly distinguishes between legally operating 503A/503B compounding pharmacies and unregulated grey market channels in their enforcement actions.
What are the most common adverse events reported with grey market or compounded tirzepatide?
The FDA reports include nausea, vomiting, diarrhea, abdominal pain, constipation, injection site reactions, and more severe outcomes including pancreatitis requiring hospitalization, gallbladder disease, severe hypoglycemia, and acute kidney injury. Many of the serious events are linked to dosing errors rather than contamination, highlighting the risks of self-dosing without medical supervision.
Is it safe to use grey market tirzepatide if I get it tested first?
Testing reduces risk but does not eliminate it. HPLC testing confirms purity but not sterility. Mass spectrometry confirms identity but not endotoxin levels. Sterility and endotoxin testing is expensive and only applies to the specific sample tested. Batch-to-batch variation means one good test result does not guarantee the next vial is safe. The only approach that systematically addresses all quality parameters is regulated manufacturing with comprehensive batch testing.
External resources
FDA: Concerns with unapproved GLP-1 drugs used for weight loss
Obesity Action Coalition: Statement on grey market GLP-1 products
National Association of Boards of Pharmacy: Verify your pharmacy
In case I do not see you, good afternoon, good evening, and good night. May your sources stay verified, your doses stay accurate, and your health stay protected.