Mar 3, 2026

Some people notice changes within days. Others wait six weeks and wonder if anything is happening at all. The gap between fast responders and slow starters on semaglutide is not random, and it is not about genetics alone. It comes down to dose timing, body composition, dietary habits, and a handful of protocol details that most guides either skip entirely or bury under vague reassurances.
Here is the reality. Semaglutide does not work like a light switch. It works like a dimmer. The GLP-1 receptor activation builds gradually as you move through the dose titration schedule, and your body responds in phases that look nothing like the smooth curves in clinical trial graphs. Week one feels different from week four. Week four feels nothing like week twelve. And week twenty-four, for many researchers, is where the real transformation becomes undeniable.
This guide breaks down exactly what happens at every stage of semaglutide treatment, from the first injection through a full year and beyond. Not the sanitized version. The real version, backed by data from the STEP clinical trials, published research on GLP-1 receptor agonists, and documented experiences from thousands of researchers who have navigated this exact timeline. You will learn what to expect physically, what side effects peak and when they fade, how your appetite changes week over week, and the specific milestones that separate people who get exceptional results from those who plateau early. SeekPeptides has compiled this information from clinical data, community reports, and evidence-based protocols to give you the most complete week-by-week reference available.
How semaglutide produces results over time
Understanding the week-by-week timeline requires understanding the mechanism. Semaglutide is a GLP-1 receptor agonist, a synthetic version of a hormone your body already produces. When you eat, your gut releases natural GLP-1. That hormone signals your brain to stop eating, slows your stomach emptying, and tells your pancreas to release insulin. The problem is that natural GLP-1 breaks down within minutes.
Semaglutide lasts much longer. Its half-life sits at approximately 168 hours, roughly seven days, which is why the standard dosing protocol calls for once-weekly injections. But that long half-life also means the compound accumulates in your system over time. After your first injection, blood levels are relatively low. After four weeks of consistent dosing at the same level, steady-state concentration is approximately five times higher than after a single dose.
This accumulation effect is why results do not appear instantly.
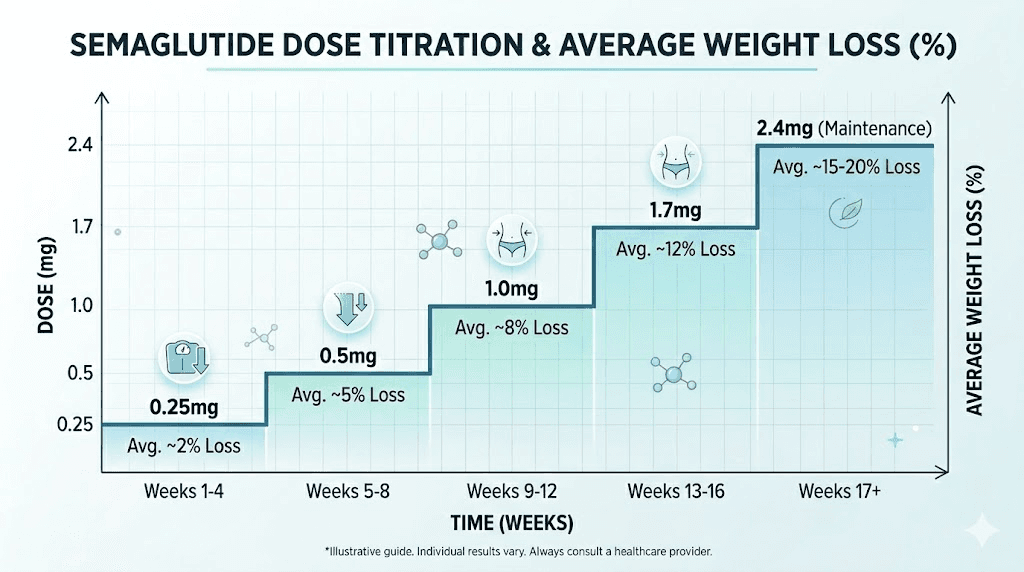
It is also why the standard titration protocol exists. Starting at 0.25 mg and gradually increasing to 0.5 mg, then 1.0 mg, then 1.7 mg, and finally 2.4 mg gives your GLP-1 receptors time to adjust. Each dose increase brings a new wave of receptor activation, and with it, a new phase of appetite suppression, metabolic changes, and potential side effects. The semaglutide dosage calculator can help you understand exactly where you are in this progression and what your target concentration looks like at each stage.
Three separate mechanisms drive the results you see on the scale. First, appetite suppression reduces caloric intake, often dramatically, within the first few weeks. Second, delayed gastric emptying makes you feel full longer after smaller meals. Third, and this one takes longer to manifest, changes in reward circuitry reduce what researchers call "food noise," those persistent thoughts about eating that have nothing to do with actual hunger. Each mechanism operates on a slightly different timeline, and together they create the layered response pattern that defines the semaglutide experience.
Weeks 1 and 2: the starting phase at 0.25 mg
The first two weeks catch most people off guard. Not because the effects are dramatic. Because they are subtle in ways you do not expect.
At 0.25 mg, you are on the lowest possible dose. This is intentional. The starting phase is about tolerance, not results. Your body is meeting a new compound, and the primary goal is letting your gastrointestinal system adjust before increasing the dose. That said, even at this low level, changes begin almost immediately for many researchers.
What happens in week 1
The first week on semaglutide typically brings a noticeable reduction in appetite within 24 to 72 hours of the initial injection. This is not the profound appetite suppression that comes later at higher doses. It is more like a gentle muting. Foods that normally trigger intense cravings become easier to resist. Portion sizes naturally decrease. You might find yourself forgetting about snacks you used to eat on autopilot.
Weight changes in week one usually range from 0 to 3 pounds. Some of this is water weight.
Side effects during the first week tend to be mild but present. Nausea affects roughly 15 to 20 percent of people at this dose, usually appearing within a few hours of injection and lasting 24 to 48 hours. Some researchers experience mild fatigue, a slight headache, or changes in bowel habits. The side effect onset timeline varies, but most people who will experience GI symptoms feel them within the first 48 hours post-injection.
Injection technique matters from day one. Whether you are using the abdomen, thigh, or upper arm, proper subcutaneous injection ensures consistent absorption. Researchers using compounded formulations should follow their specific syringe injection guide for accurate dosing.
What happens in week 2
By the second injection, your body has had seven days to process the first dose. Blood levels are building but have not reached steady state. Most researchers report that the appetite suppression from week one either maintains or slightly intensifies. You might notice that meals satisfy you faster, that you are reaching for seconds less often, or that evening snacking has dropped significantly.
The scale might show 1 to 4 pounds of total loss from baseline. Again, much of this is water and reduced food volume in the GI tract, not pure fat loss. Setting expectations correctly here prevents frustration later. Real fat loss at 0.25 mg is minimal, perhaps a quarter to half a pound of actual adipose tissue per week.
One thing that surprises many people during these first two weeks is the mental shift. Even though you are not losing significant weight yet, the relationship with food begins to change. That constant background hum of thinking about what to eat next, when to eat, what to order, starts to quiet. This is the "food noise" reduction that researchers describe as one of the most significant early effects, and it often happens before any meaningful number appears on the scale.
Weeks 3 and 4: completing the first month
The second half of month one is where patterns start to emerge. You have been on 0.25 mg for four weeks now, and your blood levels are approaching steady state for this dose. Your body has had time to adjust to the GLP-1 receptor activation, and the initial side effects, if you had any, have typically stabilized or resolved.
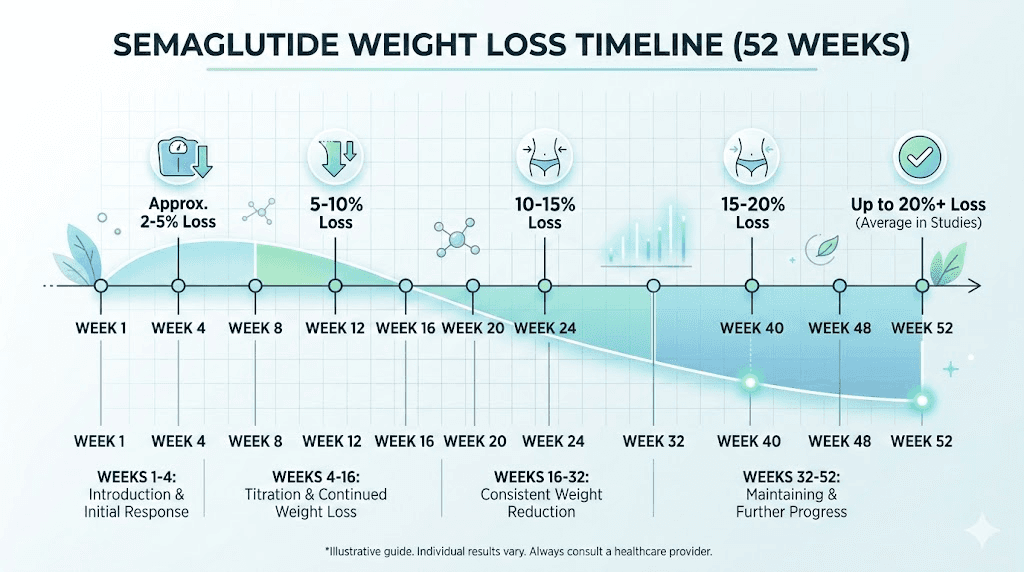
First month results in clinical trials showed an average weight loss of approximately 3.8 percent of baseline body weight at the 2.4 mg dose, but most researchers are still at 0.25 mg during this period. At the starting dose, realistic first-month losses typically fall between 2 and 6 pounds total, depending on starting weight, dietary changes, and individual metabolism.
Here is what gets interesting. For some people, weeks 3 and 4 bring a slight reduction in the appetite-suppressing effect compared to week 1. This is not the medication failing. It is your body achieving homeostasis at this dose level. The novelty of the GLP-1 signaling has worn off slightly, and your system has adapted. This is precisely why the titration protocol calls for a dose increase at week five.
Dietary patterns that emerge
By the end of month one, most researchers have naturally shifted their eating patterns. Food preferences often change without conscious effort. High-fat foods become less appealing. Sweets lose some of their pull. Protein-rich meals become more satisfying. These shifts are driven by the GLP-1 receptor activation in brain regions that process food reward, and they tend to intensify as the dose increases.
Smart researchers use this window to establish nutritional foundations. Building a food plan during month one, when appetite suppression is present but manageable, sets the stage for better results in months two through six. Focus on protein intake during this phase. Research shows that inadequate protein during GLP-1 therapy significantly increases lean mass loss, and the effects compound over time.
Also pay attention to what to avoid. Certain foods trigger worse GI symptoms on semaglutide, particularly high-fat meals, greasy foods, and large portions of anything. Eating smaller, more frequent meals during the first month helps your body adjust to the slowed gastric emptying.

Weeks 5 through 8: the 0.5 mg phase
Week five marks the first dose escalation. Moving from 0.25 mg to 0.5 mg doubles the amount of semaglutide in your system, and the effects reflect that increase. This is where many researchers first experience what they describe as "real" appetite suppression, the kind where you genuinely forget to eat, where a meal that used to seem normal now feels impossibly large, where the idea of a snack between meals simply does not occur to you.
The transition is not always smooth.
Dose increases commonly bring a temporary return of GI side effects. Bloating and nausea are the most reported symptoms during the 0.5 mg transition, affecting roughly 25 to 30 percent of researchers. These typically resolve within 7 to 14 days as steady state builds at the new dose. Constipation also becomes more common at this level, a direct consequence of the slowed gastric emptying that is actually helping you eat less.
Weight loss acceleration
Weeks 5 through 8 bring the first noticeable acceleration in weight loss. At 0.5 mg, the combination of stronger appetite suppression and your now-established dietary changes starts producing consistent results. Most researchers see 1 to 2 pounds per week of actual weight loss during this phase, though individual variation remains significant.
By the end of week 8, total weight loss from baseline typically ranges from 4 to 10 pounds. People with higher starting weights tend to lose more in absolute terms, while those closer to a healthy BMI may see smaller numbers but more significant body composition changes. The pace of weight loss at this stage depends heavily on whether you are complementing the medication with appropriate dietary and exercise habits.
Something critical happens during this phase that the numbers do not capture. Energy levels often improve. Not because semaglutide is a stimulant, it is not, but because reduced caloric intake paradoxically leads to better blood sugar stability. The crashes from large, carbohydrate-heavy meals disappear. Afternoon energy dips smooth out. Sleep often improves. Researchers report increased energy not from the drug itself but from the metabolic stabilization it produces.
The food noise continues to drop
At 0.5 mg, the reduction in food-related thoughts becomes even more pronounced. Researchers describe this as a quiet liberation. The constant mental negotiation around food, should I eat this, should I not, I really want that cookie, maybe just one, fades to near silence for many people. This is not willpower. It is pharmacological modulation of reward circuitry, and it is one of the most profound effects of GLP-1 therapy that clinical trials rarely capture in their data.
This mental shift has downstream effects that matter. Grocery shopping changes. Restaurant ordering changes. Social eating dynamics change. For people who have spent years or decades fighting food impulses, this quiet mind around eating feels transformative even before the scale shows dramatic numbers.
Weeks 9 through 12: the three-month milestone at 1.0 mg
The second dose escalation happens at week nine, bringing you to 1.0 mg weekly. This is a significant jump, quadrupling your original starting dose, and the effects typically match that increase. At the three-month mark, you are approaching therapeutic blood levels that mirror what clinical trials tested for weight management efficacy.
Clinical data from the STEP 1 trial shows average weight loss of approximately 5.9 percent of baseline body weight at 12 weeks. For a 200-pound person, that translates to roughly 12 pounds. For a 250-pound person, closer to 15. These are averages from the 2.4 mg dose group, so researchers still titrating through 1.0 mg might see slightly less, but the trajectory is clear.
What the three-month mark looks like
By week 12, the changes are visible. Not just on the scale. In the mirror. In how clothes fit. In face shape. Three months of consistent appetite reduction and improved dietary habits produces the kind of physical transformation that other people start to notice. Belt holes tighten. Rings loosen. Energy improves. Before and after comparisons at the three-month mark often show the first truly dramatic visual differences.
Blood work at this stage frequently reveals improvements beyond weight. Fasting glucose drops. HbA1c improves, sometimes significantly, even in people without diagnosed diabetes. Triglycerides decrease. LDL cholesterol often trends downward. These metabolic improvements are not just side benefits of weight loss. GLP-1 receptor agonists have direct effects on glucose metabolism and lipid profiles that operate independently of body weight changes.
This is also the phase where researchers need to be most vigilant about nutritional supplementation. Reduced food intake means reduced nutrient absorption. B12, iron, folate, vitamin D, and magnesium are the most common deficiencies reported during GLP-1 therapy. Some compounded formulations address this directly by including B12 in the semaglutide formulation or adding glycine for additional metabolic support.
Handling the second dose adjustment
The jump to 1.0 mg brings another round of potential side effects. Nausea returns for about 20 to 25 percent of researchers, typically lasting 5 to 10 days before subsiding. Dizziness can occur, especially if food intake drops too aggressively. Sulfur burping, one of the more unpleasant side effects, peaks for some researchers during this dose transition and typically resolves within two to three weeks.
Injection timing becomes more important at higher doses. Many researchers find that injecting in the evening, before bed, helps them sleep through the worst of any nausea. Others prefer morning injections so they can manage symptoms while awake. There is no single right answer, but consistency matters more than timing.
Weeks 13 through 20: mid-treatment acceleration
This is the phase that separates semaglutide from every diet you have ever tried.
Between months four and five, most researchers are at 1.7 mg (escalated at week 13) or approaching the full 2.4 mg dose. Blood levels of semaglutide are now in the range that clinical trials associate with maximum appetite suppression and metabolic modulation. The compound has been building in your system for over three months, steady-state concentration at these higher doses is fully established, and the cumulative effects are substantial.
Weight loss during this phase typically averages 1 to 2.5 pounds per week. Some weeks the scale barely moves. Other weeks it drops three pounds overnight. This variability is normal and reflects fluctuations in water retention, hormonal cycles, sodium intake, and dozens of other factors that temporarily mask fat loss. The trend line is what matters, not any individual week.
The metabolic shift
Something fundamental changes around weeks 13 to 16 that goes beyond appetite suppression. Researchers describe a metabolic "settling in" where their body seems to find a new equilibrium. Hunger signals become predictable. Energy levels stabilize. Sleep improves. The physical and mental chaos of the early titration phase gives way to a new normal that feels sustainable rather than forced.
This is the window where lifestyle habits either amplify results or limit them. Researchers who are eating according to a structured nutrition plan, maintaining adequate protein intake, and incorporating resistance training see significantly better outcomes than those relying on the medication alone. The data is clear on this point. In the STEP clinical trials, participants who combined semaglutide with lifestyle interventions lost up to 17.4 percent of baseline body weight, compared to 14.9 percent for those with less intensive behavioral support.
Do not underestimate the importance of exercise during this phase. Weight loss without exercise is possible on semaglutide, but the composition of that weight loss changes dramatically. Without resistance training, up to 40 percent of weight lost comes from lean mass, including muscle. With consistent strength training and adequate protein (1.0 to 1.2 grams per kilogram of body weight daily), lean mass preservation improves significantly.
By week 16 to 20
At the four to five month mark, total weight loss from baseline typically ranges from 8 to 15 percent of starting body weight. For a 220-pound starting weight, that puts you somewhere around 187 to 202 pounds. For a 180-pound starting weight, 153 to 166 pounds. These ranges are wide because individual response varies enormously based on starting BMI, metabolic health, adherence, and concurrent lifestyle modifications.
Researchers who started without seeing early results often find that weeks 13 to 20 bring the acceleration they were waiting for. Higher doses simply work better for some people. The GLP-1 receptor density varies between individuals, and some require more semaglutide to achieve the appetite suppression threshold that drives meaningful caloric reduction. If the first three months felt slow, this phase often delivers the results that validate patience and persistence.
Clothing sizes change during this period. Energy increases noticeably. Physical activity becomes easier as the load on joints decreases. Researchers who struggled with exercise at their starting weight find that 20 to 30 pounds of loss opens up new possibilities for movement, creating a positive feedback loop that accelerates further progress.
Weeks 21 through 32: the six-month mark and beyond
The six-month milestone is the benchmark that clinical trials use most frequently when reporting semaglutide outcomes. There is a reason for this. At 24 weeks, most participants have reached or are approaching the full 2.4 mg dose, blood levels are at steady state, and enough time has passed for meaningful fat loss to accumulate.
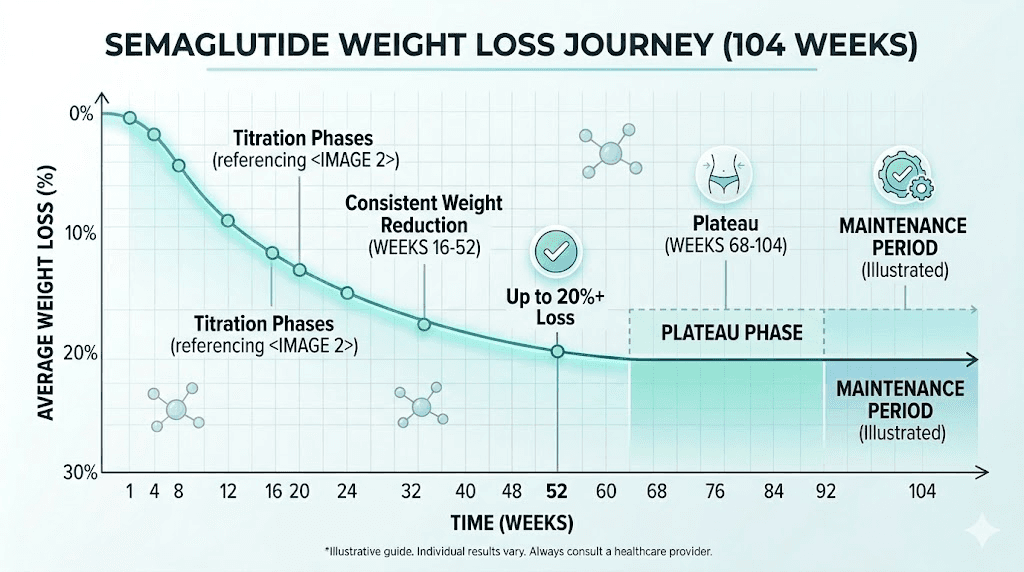
The numbers from STEP 1 are compelling. At 68 weeks, the semaglutide group averaged 14.9 percent weight loss from baseline, but the six-month data point shows approximately 13.8 percent, meaning most of the total weight loss has already occurred by this stage. The trajectory is steep through month six and then begins to gradual flatten, not because the medication stops working, but because the caloric deficit narrows as body weight decreases.
What 13 to 15 percent weight loss looks like
For a 230-pound person, 13.8 percent loss means roughly 32 pounds gone. That is not a cosmetic change. That is a medical transformation. Blood pressure drops an average of 4 to 6 mmHg systolic. HbA1c reduces by 0.5 to 1.5 percent in people with prediabetes or diabetes. Inflammatory markers like CRP often decrease by 20 to 40 percent. Joint pain diminishes. Sleep apnea symptoms improve or resolve. The downstream health effects of this magnitude of weight loss extend far beyond the number on the scale.
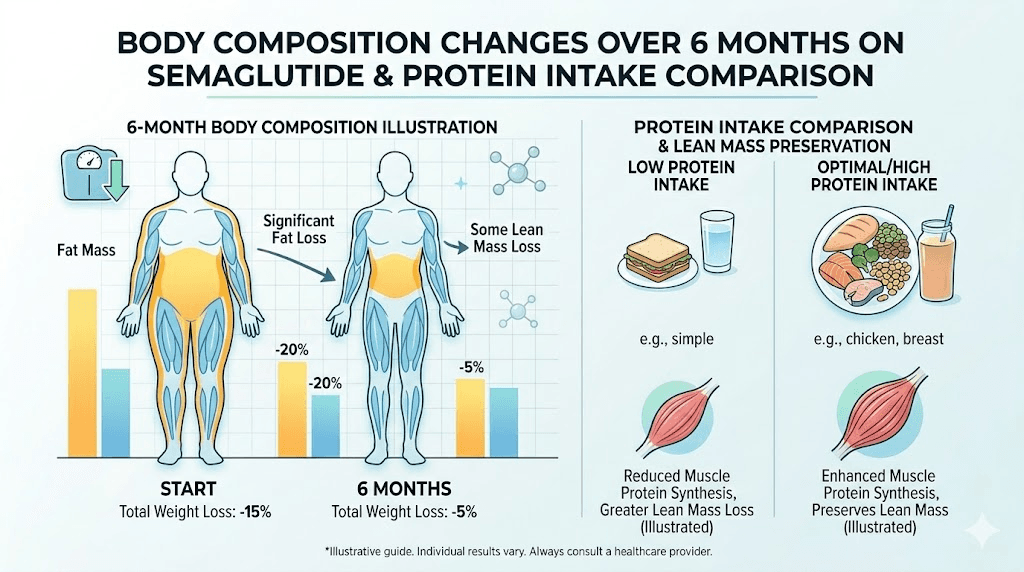
Body composition at six months shows a pattern worth understanding. Fat mass decreases substantially, with visceral fat (the dangerous fat surrounding internal organs) dropping proportionally more than subcutaneous fat. However, lean mass also decreases, and this is where smart protocol management makes the difference. Researchers who have maintained high protein intake and consistent resistance training throughout the process lose primarily fat, while those who relied on appetite suppression alone show concerning ratios of muscle loss.
SeekPeptides members frequently discuss strategies for optimizing body composition during this phase, including protein timing, resistance training protocols, and supplementation approaches that preserve lean mass while maximizing fat loss. Access to these detailed protocols helps researchers navigate the balance between aggressive weight loss and healthy body composition.
Side effects at six months
By the six-month mark, most GI side effects have resolved or become manageable. The body has fully adapted to the medication at the maintenance dose. Nausea, which peaked during the titration phase, affects fewer than 10 percent of researchers at steady state on 2.4 mg. Constipation is the most persistent side effect, affecting roughly 15 to 20 percent of long-term users, and often responds to increased fiber intake and hydration.
Two side effects deserve special attention at this stage. Hair thinning can appear around months 4 to 6, driven by the telogen effluvium response to rapid weight loss rather than the medication itself. This is usually temporary and resolves as weight stabilizes. Fatigue that appeared manageable early on can become more pronounced if nutritional deficiencies have developed due to prolonged reduced food intake. Regular blood work at the six-month mark is strongly recommended.
Weeks 33 through 52: approaching one year
The second half of year one on semaglutide is where the curve begins to flatten. This is not failure. It is physiology.
As you lose weight, your body requires fewer calories to function. A person who weighed 240 pounds at the start and now weighs 205 at week 33 has a basal metabolic rate roughly 250 to 350 calories lower than when they began. Meanwhile, the appetite suppression from semaglutide remains relatively constant at the maintenance dose. The result is a narrowing caloric deficit that progressively slows the rate of weight loss.
Between months 8 and 12, weight loss typically slows to 0.25 to 0.75 pounds per week. Some weeks the scale does not move at all. This is the phase where the semaglutide plateau becomes a common concern, and it is where many researchers either adjust their approach or lose motivation.
Understanding the plateau
A weight loss plateau on semaglutide does not mean the medication has stopped working. It means your body has reached a new equilibrium between caloric intake and expenditure at the current dose. The medication is still suppressing appetite. It is still slowing gastric emptying. It is still modulating reward circuitry. But the energy balance equation has shifted as your smaller body requires less fuel.
Researchers who hit a plateau between weeks 33 and 52 have several evidence-based options. Increasing physical activity, particularly high-intensity interval training and resistance work, can expand the caloric deficit without requiring further food restriction. Adjusting macronutrient ratios toward higher protein and moderate carbohydrate intake can improve satiety and support lean mass. Some providers recommend temporary caloric cycling, alternating between slightly higher and lower intake days, to prevent metabolic adaptation.
If you have been wondering why weight loss has stalled, this phase is the most common timeframe for plateaus. The good news is that maintaining a 10 to 15 percent weight loss is itself a significant health achievement, even if the scale stops moving. The metabolic improvements from that degree of weight reduction persist as long as the weight stays off.
The STEP 5 two-year data
Long-term data from the STEP 5 trial provides reassurance for researchers in this phase. Average weight loss at week 52 was 15.6 percent, and at week 104 (two years), it was 15.2 percent. That tiny difference, just 0.4 percentage points, tells an important story. Weight loss plateaus during the second half of year one, but it does not significantly reverse as long as treatment continues. The medication maintains the loss even after the rate of new loss slows to nearly zero.
This is fundamentally different from dieting without pharmacological support, where weight regain typically begins within 6 to 12 months and averages 30 to 50 percent of the total loss within two years. Semaglutide changes the weight maintenance equation by maintaining the appetite suppression and metabolic modulation that drove the initial loss.
Beyond week 52: long-term results and maintenance
How long should you stay on semaglutide? This question does not have a simple answer, and the week-by-week timeline beyond year one depends entirely on individual goals and medical context.
The clinical data is definitive on one point. Stopping semaglutide leads to weight regain for most people. The STEP 1 extension study followed participants who discontinued treatment after 68 weeks. Within one year of stopping, they regained approximately two-thirds of the weight they had lost. The appetite suppression fades. Food noise returns. The metabolic benefits gradually reverse. Withdrawal effects are not physically dangerous, but the rebound in hunger and cravings can be psychologically challenging.
For researchers who choose to continue treatment beyond one year, the trajectory is generally stable. Weight maintains within 1 to 3 percent of the nadir (lowest point), with seasonal fluctuations and life events causing temporary variations. The medication continues to suppress appetite, and the health benefits, lower blood pressure, improved glucose control, reduced inflammation, persist as long as treatment continues.
Some researchers explore dose reduction as a long-term strategy. Stepping down from 2.4 mg to 1.7 mg or even 1.0 mg after achieving goal weight can reduce side effects and medication cost while maintaining some appetite suppression. This approach works for some people but not others, and individual response to dose reduction is difficult to predict in advance.
Others transition to different GLP-1 agents. Switching between semaglutide and tirzepatide is increasingly common, especially for researchers who have plateaued on one compound and want to explore whether a dual agonist (GLP-1/GIP) provides additional benefits. The comparison between semaglutide, tirzepatide, and retatrutide shows distinct advantages and tradeoffs for each, and the optimal long-term strategy often involves individualized protocol adjustments over time.
What the STEP clinical trials actually show
Numbers matter. And the STEP (Semaglutide Treatment Effect in People with obesity) clinical trial program provides the most robust dataset available for understanding semaglutide results over time. Here is what each major trial found.
STEP 1: the foundational study
This trial enrolled 1,961 adults with BMI of 30 or higher (or 27 with comorbidities) and randomized them to semaglutide 2.4 mg weekly or placebo for 68 weeks. Results published in the New England Journal of Medicine showed that 86.4 percent of the semaglutide group lost at least 5 percent of body weight, compared to 31.5 percent of the placebo group. The average weight loss was 14.9 percent with semaglutide versus 2.4 percent with placebo.
Even more striking, 50.5 percent of semaglutide participants lost 15 percent or more, and 32.0 percent lost 20 percent or more. These numbers approach what was previously achievable only through bariatric surgery.
STEP 2: people with type 2 diabetes
Diabetes complicates weight loss, and the STEP 2 trial specifically enrolled participants with type 2 diabetes. The results were good but more modest, a mean weight reduction of 9.6 percent with semaglutide 2.4 mg versus 3.4 percent with placebo at 68 weeks. The reduced weight loss in diabetic participants likely reflects insulin resistance and other metabolic factors that attenuate the response.
STEP 5: the two-year study
Duration matters for understanding the full timeline. STEP 5 followed participants for 104 weeks (two years) on semaglutide 2.4 mg. Weight loss at week 52 averaged 15.6 percent and at week 104 averaged 15.2 percent. This minimal difference between year one and year two confirms that the medication maintains weight loss over extended treatment without significant regain.
The newer high-dose data
Recent trials have tested semaglutide at 7.2 mg weekly, triple the standard weight management dose. The results showed 20.7 percent weight loss with the higher dose versus 17.5 percent with 2.4 mg. This data suggests that dose-response continues above the current standard, though regulatory approval for the higher dose is still pending.
Understanding this clinical context matters because it sets realistic benchmarks for your own results. If you are at 12 weeks and have lost 6 percent of body weight, you are right on track with the trial averages. If you are at 24 weeks and below 10 percent, there may be protocol adjustments worth exploring. And if you are exceeding the averages, your combination of medication response and lifestyle modifications is producing above-average outcomes.
Side effects timeline: what to expect and when
Side effects follow their own week-by-week pattern that is just as important to understand as the weight loss timeline. Knowing when symptoms peak and when they resolve prevents unnecessary anxiety and helps you plan around the most challenging phases.
Weeks 1 through 4: initiation symptoms
Nausea affects 15 to 20 percent of researchers at 0.25 mg. It is typically mild, described as a queasy feeling rather than active vomiting, and peaks 12 to 24 hours after injection. Most find it resolves within 48 hours and decreases with each subsequent injection. The onset pattern is predictable enough that researchers can plan their injection day around it.
Mild fatigue and headache are common in the first two weeks and typically resolve by week three. Bowel changes, either constipation or loose stools, affect roughly 20 percent of users initially.
Weeks 5 through 12: dose escalation side effects
Each dose increase brings a temporary return of GI symptoms. The 0.5 mg jump is usually the easiest, with moderate nausea in 20 to 25 percent of researchers. The 1.0 mg jump is often the most challenging, with nausea rates climbing to 30 to 35 percent and lasting up to two weeks. Many researchers describe week 9 through 11 as the hardest stretch of the entire treatment.
This is when bloating can become pronounced. The slowed gastric emptying at 1.0 mg is significant enough that food sits in the stomach much longer than usual, creating a persistent fullness and distension that some find quite uncomfortable. Smaller meals, thorough chewing, and avoiding carbonated beverages help manage this symptom.
Weeks 13 through 24: adjustment and resolution
By month four, most GI side effects have resolved for the majority of researchers. The body has adapted to the higher doses, and the GI system has established a new normal with the slowed transit time. Roughly 10 to 15 percent continue to experience mild, manageable nausea at the 1.7 mg and 2.4 mg doses.
New side effects can emerge in this window. Some researchers report changes in taste perception, finding certain foods unpleasant that they previously enjoyed. Others notice increased sleep disturbances, though the direction varies, with some sleeping better and others worse. Menstrual cycle changes are reported by approximately 20 percent of female researchers, likely related to the hormonal shifts that accompany rapid weight loss rather than a direct effect of the medication.
Beyond week 24: long-term tolerability
The long-term side effect profile is reassuring for most researchers. By the six-month mark, the majority have either no side effects or mild, manageable symptoms that do not significantly impact quality of life. Constipation is the most persistent issue, affecting 15 to 20 percent of long-term users. Regular monitoring of blood work and nutritional status helps catch any developing deficiencies before they become symptomatic.
Body composition changes: what happens beneath the surface
The scale tells one story. Body composition tells a more nuanced one.
Research from the STEP 1 exploratory analysis using DEXA scans revealed that semaglutide-induced weight loss comprises approximately 60 percent fat mass and 40 percent lean mass. This ratio is consistent with what is seen in most weight loss interventions, but it raises important considerations for researchers tracking their results week by week.
Fat loss patterns
Visceral fat, the metabolically dangerous fat surrounding internal organs, decreases proportionally more than subcutaneous fat during semaglutide treatment. This is significant because visceral fat drives insulin resistance, inflammation, and cardiovascular risk to a degree that subcutaneous fat does not. Even modest weight loss of 5 to 10 percent, achievable within the first 8 to 12 weeks for most researchers, produces disproportionate reductions in visceral fat that improve metabolic health markers.
Subcutaneous fat loss follows a different pattern. It tends to reduce from the trunk first, then extremities. Many researchers notice their waistline shrinking before they see changes in arms or legs. This is partly because trunk fat is more metabolically active and responds more readily to the hormonal changes induced by GLP-1 therapy.
Lean mass considerations
The 40 percent lean mass loss figure from clinical trials represents participants who were not specifically optimizing for muscle preservation. Research published since the STEP trials shows that adequate protein intake (1.0 to 1.6 grams per kilogram of body weight daily) combined with resistance training two to three times per week can significantly reduce lean mass loss, potentially lowering it to 20 to 25 percent of total weight lost.
The SEMALEAN study provided even more nuance. Total fat mass decreased by 14 percent at seven months and 18 percent at twelve months, while lean mass initially declined by about 3 kilograms at seven months but then stabilized. Handgrip strength, a marker of functional muscle capacity, actually improved by 4.5 kilograms at twelve months. And the prevalence of sarcopenic obesity decreased from 49 percent at baseline to 33 percent at twelve months.
What does this mean for week-by-week tracking? It means the scale is an incomplete measure of progress. Protecting lean mass with proper nutrition is not optional if you want to maintain metabolic health and physical function throughout the weight loss process. Researchers who track body composition through DEXA scans, bioimpedance, or even simple waist-to-hip measurements get a much clearer picture of their actual progress than those who rely on scale weight alone.
How to optimize results at each phase
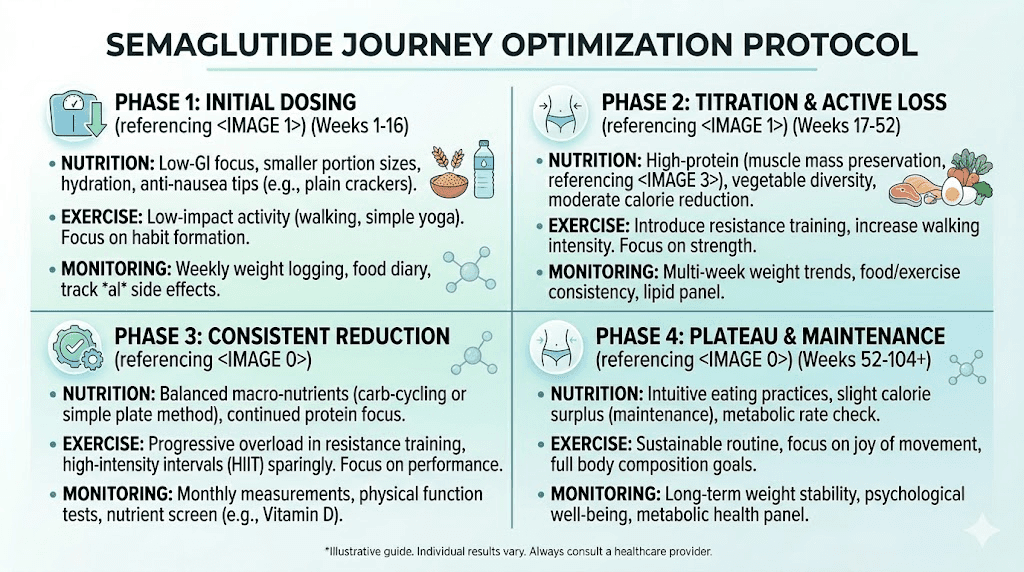
Knowing what to expect is valuable. Knowing how to maximize results at each phase is where the real advantage lies. Here is a phase-by-phase optimization framework built from clinical evidence and community experience.
Weeks 1 through 4: foundation building
The goal during the first month is not maximum weight loss. It is establishing the habits that will compound over the next six to twelve months.
Nutrition priorities: Start tracking protein intake immediately. Aim for at least 0.8 grams per kilogram of current body weight daily, with a target of reaching 1.0 to 1.2 grams per kilogram by month two. This means a 200-pound person should be consuming 73 to 109 grams of protein daily. Prioritize lean proteins, eggs, dairy, and legumes at every meal.
Hydration: Drink at least 64 ounces of water daily. GLP-1 therapy increases the importance of hydration because reduced food intake means less water from food sources. Dehydration worsens nausea, constipation, and fatigue, all common early side effects.
Exercise: Start with low to moderate activity, 150 minutes of walking, cycling, or swimming per week. Do not attempt aggressive exercise programs during the first month. Your body is adjusting to reduced caloric intake, and overtraining at this stage increases fatigue and muscle breakdown.
Weeks 5 through 12: acceleration phase
As the dose increases and appetite suppression intensifies, the risk of under-eating becomes real. Paradoxically, eating too little can slow results by triggering metabolic adaptation, where your body reduces energy expenditure to match dramatically reduced intake.
Nutrition priorities: Increase protein to 1.0 to 1.2 grams per kilogram daily. If nausea makes solid food difficult, protein shakes become essential. Do not let total caloric intake drop below 1,200 for women or 1,500 for men, even if appetite allows for less. Aim for structured meals rather than random eating.
Exercise: Introduce resistance training, two to three sessions per week focusing on compound movements like squats, deadlifts, presses, and rows. This is the most important exercise modification you can make for long-term body composition. Muscle preserved now means higher metabolic rate later, which directly affects how much total weight you lose and how easily you maintain it.
Monitoring: Take measurements, waist, hips, chest, thigh, and progress photos at week 5 and again at week 12. The scale during this phase can be misleading due to water retention fluctuations, especially around dose increases. Measurements and photos provide a more accurate picture of progress.
Weeks 13 through 24: maximization phase
This is the phase where researchers who optimized their foundations pull ahead dramatically from those who did not.
Nutrition priorities: Consider working with a dietitian to ensure adequate micronutrient intake. Supplement with a high-quality multivitamin, additional B12 (especially if not using a methylcobalamin-enhanced formulation), vitamin D, and magnesium. Track macronutrients rather than just calories, aiming for 30 percent protein, 35 percent fat, and 35 percent carbohydrates as a starting point.
Exercise: Increase resistance training intensity. Add a fourth weekly session if recovery allows. Consider adding high-intensity interval training (HIIT) once or twice per week to support cardiovascular fitness and increase post-exercise calorie burn. Monitor recovery closely, reduced caloric intake means reduced recovery capacity.
Mental health: The rapid body changes during this phase can trigger unexpected psychological responses. Body dysmorphia, anxiety about maintaining results, complicated feelings about changing social dynamics around food, these are all common and normal. SeekPeptides community discussions frequently address these topics, and having access to others who understand the experience can be genuinely therapeutic.
Weeks 25 through 52: maintenance and optimization
Nutrition priorities: As weight loss slows, caloric intake should stabilize rather than continuing to decrease. Calculate your new maintenance calories based on current weight and adjust intake to create a modest 250 to 500 calorie daily deficit. Attempting aggressive deficits during the maintenance phase increases muscle loss and metabolic adaptation without significantly accelerating fat loss.
Exercise: Maintain or increase resistance training. If you have not already, this is the time to invest in a structured progressive overload program. As body weight decreases, relative strength increases, meaning exercises become easier and you can handle heavier loads. Use this advantage to build muscle that will support long-term metabolic health.
Monitoring: Schedule comprehensive blood work at month 6 and month 12. Include thyroid function, metabolic panel, lipid panel, HbA1c, B12, ferritin, vitamin D, and a complete blood count. These results guide supplementation, identify developing deficiencies, and document the metabolic improvements that motivate continued treatment.
When to worry and when to be patient
Not every deviation from the expected timeline signals a problem. But some do. Here is how to tell the difference.
Normal variations that do not require concern
A week with no weight loss after several weeks of consistent loss is completely normal. Water retention from sodium intake, hormonal cycles, exercise-induced inflammation, or even stress can mask ongoing fat loss. Wait two to three weeks before concluding that progress has truly stalled.
A slight weight increase of 1 to 3 pounds between weeks is also normal, especially around dose increases, during the first few days of a new exercise routine, or during menstrual cycles. These fluctuations represent water, glycogen, and GI contents, not fat gain.
Side effects that return briefly with each dose increase are expected. The pattern of symptom, adaptation, resolution, then recurrence with the next increase, is the standard semaglutide experience for most researchers.
Signs that warrant medical attention
Severe or persistent vomiting (more than 24 hours or multiple times per day) requires medical evaluation. While nausea is common, actual vomiting that prevents keeping down fluids is not typical and can lead to dehydration and electrolyte imbalances. This is especially important during the higher dose phases.
Severe abdominal pain, particularly if it radiates to the back, could indicate pancreatitis, a rare but serious adverse event associated with GLP-1 therapy. This requires immediate medical attention.
No weight loss at all after 12 weeks at therapeutic doses (1.0 mg or higher) is unusual and warrants investigation. Thyroid dysfunction, undiagnosed medical conditions, medication interactions, or significant protocol deviations could be contributing factors. If you are in this situation, our guide on troubleshooting weight loss stalls covers the most common causes and solutions.
Unusual symptoms like severe dizziness, vision changes, kidney symptoms, or signs of blood clotting issues should be evaluated promptly, though these are rare.
The patience equation
Here is the uncomfortable truth about semaglutide timelines. The researchers who get the best long-term results are almost always the ones who can tolerate weeks of minimal scale movement without abandoning the protocol. Weight loss is not linear. It comes in waves. A frustrating two-week stall often precedes a whoosh of several pounds lost in a few days. This pattern repeats throughout treatment and is driven by fluid dynamics that scientists still do not fully understand.
If your results are slower than expected but moving in the right direction, patience is almost always the correct strategy. The clinical trials ran for 68 to 104 weeks for a reason, because the full effect of semaglutide takes time to manifest. Judging the medication after 4 or 8 weeks, when you are still on a fraction of the therapeutic dose, is like judging a marathon after the first mile.
The role of compounded formulations in results
Not all semaglutide formulations produce identical results on identical timelines, and understanding the differences helps set appropriate expectations for your specific protocol.
Compounded semaglutide products vary in concentration, reconstitution requirements, and additional ingredients that can influence the experience. Some formulations include vitamin B12 to address the nutritional deficiencies common during GLP-1 therapy. Others add glycine for its potential synergistic effects on metabolic health. Combination formulations like semaglutide glycine B12 aim to optimize the overall protocol by addressing multiple needs in a single injection.
Storage and handling directly impact potency, which impacts results. Refrigeration protocols must be followed carefully. Temperature excursions can degrade the peptide and reduce its effectiveness. If your results seem lower than expected, verifying proper storage is one of the first troubleshooting steps. Shelf life considerations also matter, especially for multi-use vials that are opened and used over several weeks.
For researchers using compounded vial formulations, accurate reconstitution is essential for proper dosing. Errors in bacteriostatic water volume directly affect the concentration per unit drawn. A 10 percent error in reconstitution means a 10 percent error in every dose throughout the vial. The peptide reconstitution calculator eliminates guesswork from this process and ensures your doses match your intended titration schedule.
Delivery method also influences the timeline. Sublingual semaglutide and oral drops have different bioavailability profiles compared to injectable forms, which can affect both the onset and magnitude of results. Injectable forms generally produce the most predictable and well-studied outcomes.
Lifestyle factors that accelerate or slow your timeline
The medication provides the foundation. What you build on that foundation determines whether you track with the trial averages, exceed them, or fall below.
Factors that accelerate results
Adequate protein intake: Researchers consuming 1.0 to 1.2 grams per kilogram daily consistently show better body composition outcomes than those eating less. Protein preserves muscle, maintains metabolic rate, and improves satiety, amplifying the appetite suppression already provided by semaglutide.
Consistent resistance training: Two to four sessions per week of progressive resistance training is the single most impactful lifestyle factor for long-term outcomes. It preserves lean mass, increases functional capacity, and improves metabolic health markers independent of weight loss.
Sleep quality: Seven to nine hours of quality sleep per night supports hormone regulation, recovery, and appetite control. Poor sleep increases ghrelin (the hunger hormone) and decreases leptin (the satiety hormone), partially counteracting the appetite suppression from semaglutide.
Stress management: Chronic stress elevates cortisol, which promotes fat storage, particularly visceral fat, and increases cravings for high-calorie foods. Managing stress through whatever methods work for you, exercise, meditation, social connection, therapy, removes a significant barrier to optimal results.
Factors that slow results
Alcohol consumption slows results through multiple mechanisms. It adds empty calories, impairs judgment around food choices, disrupts sleep, stresses the liver, and can worsen GI side effects. Researchers who eliminate or significantly reduce alcohol during treatment consistently see faster results.
Inconsistent dosing: Missing doses or varying injection timing disrupts the steady-state blood levels that drive consistent appetite suppression. The once-weekly schedule should be maintained within a 24-hour window of the same day each week. Splitting doses is sometimes considered for managing side effects, but this changes the pharmacokinetic profile and should only be done under guidance.
Ultra-low calorie intake: Eating fewer than 1,000 calories daily, which is easy to do accidentally when appetite is dramatically suppressed, triggers metabolic adaptation that actually slows weight loss over time. Your body downregulates thyroid function, reduces non-exercise activity thermogenesis (NEAT), and increases muscle catabolism when caloric restriction becomes too severe.
Sedentary behavior: While semaglutide works without exercise, a completely sedentary lifestyle limits results by allowing muscle loss, reducing daily energy expenditure, and missing the metabolic benefits of physical activity. Even moderate walking, 7,000 to 10,000 steps daily, meaningfully improves outcomes.
Comparing your results to averages: where do you stand
Here is a reference framework for understanding whether your results are on track, above average, or below expected. These benchmarks are derived from the STEP clinical trial data and adjusted for real-world conditions where adherence and lifestyle factors vary more than in controlled studies.
Week 4 benchmarks (still at 0.25 mg)
Below average: Less than 1 percent body weight loss. This is common and not cause for concern at the starting dose. The medication has not had time to build to therapeutic levels.
Average: 1 to 2 percent body weight loss (2 to 5 pounds for most starting weights). This is the expected range for the initial titration phase.
Above average: More than 2 percent body weight loss. Some researchers are highly responsive even at low doses and see early results that track ahead of the curve.
Week 12 benchmarks (at or approaching 1.0 mg)
Below average: Less than 3 percent body weight loss. If you are here, evaluate your dietary habits, protein intake, and dosing consistency. Also verify that your medication is still within its shelf life and has been stored properly.
Average: 5 to 7 percent body weight loss (11 to 18 pounds for a 220-pound starting weight). This aligns with the STEP trial trajectory at the three-month mark.
Above average: More than 8 percent body weight loss. You are tracking ahead of clinical trial averages, which suggests strong medication response combined with effective lifestyle modifications.
Week 24 benchmarks (at maintenance dose of 2.4 mg)
Below average: Less than 8 percent body weight loss. Consider discussing a protocol review with your provider. Dose verification and a comprehensive evaluation of contributing factors may be warranted.
Average: 10 to 14 percent body weight loss (22 to 31 pounds for a 220-pound starting weight). This is the sweet spot of expected results at six months.
Above average: More than 15 percent body weight loss. You are exceeding clinical trial averages, likely through a combination of strong drug response and excellent lifestyle habits.
Week 52 benchmarks (one year)
Below average: Less than 10 percent total body weight loss from baseline. At this point, switching to a dual or triple agonist like tirzepatide may provide additional benefit.
Average: 13 to 16 percent body weight loss. This aligns closely with the 14.9 percent average from STEP 1 at 68 weeks.
Above average: More than 17 percent body weight loss. Exceptional response territory that puts you in the top quartile of clinical trial outcomes.
Special populations and adjusted timelines
Not everyone follows the same week-by-week curve, and certain factors create predictable variations in how quickly results appear.
People with type 2 diabetes
Researchers with type 2 diabetes typically see slower weight loss on semaglutide compared to non-diabetic individuals. The STEP 2 trial showed 9.6 percent average weight loss at 68 weeks for diabetic participants versus 14.9 percent in STEP 1 (non-diabetic). This reduced response is driven by insulin resistance, diabetic medications (some of which promote weight gain), and metabolic dysfunction that blunts the full effect of GLP-1 therapy.
However, the metabolic benefits for diabetic researchers are often more significant than for non-diabetic users. HbA1c reductions, improved insulin sensitivity, and potentially reduced need for diabetes medications can be transformative health outcomes that the scale does not capture.
Women versus men
Men tend to lose weight faster on semaglutide during the first 12 weeks, partly due to higher baseline metabolic rates and greater starting weights. Women often see a more gradual initial response that catches up over time. Hormonal fluctuations throughout the menstrual cycle can also create more week-to-week variability in women, making the trend line harder to read but not fundamentally different in terms of total outcomes at six and twelve months.
Women should also be aware of potential menstrual cycle changes during treatment, as well as the increased risk of lean mass loss that research has identified in female participants.
Older adults
Researchers over 60 may see slightly slower weight loss and face a higher risk of sarcopenia (muscle loss) during treatment. The SEMALEAN study data suggests that older age is a significant predictor of lean mass loss on semaglutide. This makes protein optimization and resistance training even more critical for older researchers, who should aim for the higher end of protein recommendations (1.2 to 1.6 grams per kilogram daily) and prioritize strength training over cardiovascular exercise.
People with higher starting BMI
Individuals with BMI above 40 often see faster absolute weight loss in the early phases, simply because a higher body weight means a larger caloric expenditure even at rest, and the same percentage reduction in food intake creates a larger absolute caloric deficit. However, the percentage of body weight lost tends to be similar across BMI categories at 12 and 24 months, suggesting that the medication's effect on appetite is proportionally consistent regardless of starting weight.
Tracking your results effectively
How you measure progress significantly impacts your psychological experience of the semaglutide timeline. The scale is one data point. It should not be the only one.
Weekly weigh-in protocol
Weigh yourself at the same time each day, preferably first thing in the morning after using the bathroom and before eating or drinking. Record daily weights but evaluate only the weekly average. Daily fluctuations of 2 to 4 pounds are completely normal and meaningless in isolation. The weekly average removes this noise and reveals the actual trend.
Monthly measurements
Take circumference measurements monthly at consistent anatomical landmarks. Waist at the navel, hips at the widest point, chest at nipple level, and one thigh at the midpoint between knee and hip. These measurements capture body composition changes that the scale misses, particularly when you are simultaneously losing fat and gaining or maintaining muscle through resistance training.
Progress photos
Monthly front, side, and back photos in consistent lighting and clothing provide visual documentation that is invaluable for staying motivated during plateau phases. Our brains adapt to gradual change, making it difficult to appreciate transformation when you see yourself daily. Comparing month one to month four photos side by side often reveals dramatic changes that felt invisible while they were happening.
Non-scale victories
Track these alongside the numbers. Sleep quality. Energy levels. Medication reductions. Belt holes. Clothing sizes. Physical capacity, like the ability to climb stairs, walk distances, or complete workouts that were previously impossible. These markers of progress are often more meaningful than body weight and tend to improve before the scale catches up.
Comparing semaglutide results to other GLP-1 options
Context matters when evaluating your semaglutide results. How does the week-by-week timeline compare to other available GLP-1 therapies?
Tirzepatide, a dual GLP-1/GIP agonist, has shown superior weight loss results in head-to-head comparisons. The SURMOUNT clinical trials demonstrated average weight loss of 20 to 22 percent at 72 weeks, compared to semaglutide's 14.9 percent in STEP 1. The side effect profiles differ somewhat, with tirzepatide showing similar GI effects but potentially different patterns of onset and resolution.
Retatrutide, a triple agonist (GLP-1/GIP/glucagon), represents the next generation. Phase 2 trial data showed up to 24 percent weight loss at 48 weeks, suggesting even more potent effects than either semaglutide or tirzepatide alone. The week-by-week timeline for retatrutide appears compressed, with researchers reporting faster onset of appetite suppression and earlier weight loss milestones.
For researchers who have reached a plateau on semaglutide, transitioning to a dual or triple agonist is an increasingly common strategy. Switching from semaglutide to tirzepatide often restarts the weight loss trajectory, presumably by engaging additional receptor pathways that monotherapy does not activate.
What happens if you restart after a break
Life happens. Supply issues, cost concerns, side effects, surgery, or simply wanting a break can interrupt semaglutide treatment. Understanding what the timeline looks like when you restart after a gap is essential for managing expectations.
If the break is less than four weeks, many providers recommend resuming at the same dose you were previously taking. Blood levels have decreased but not fully cleared, and your GI system has recent memory of the medication. Side effects on resumption are typically mild.
If the break exceeds four weeks, most protocols recommend restarting the full titration from 0.25 mg. The rationale is twofold. First, GI tolerance resets after an extended break, and jumping back to a high dose risks severe nausea and vomiting. Second, the gradual build allows your body to re-establish steady-state levels safely.
Weight regain during breaks follows a predictable pattern. Most researchers regain 1 to 3 pounds in the first two weeks off (primarily water weight and increased GI contents), then 1 to 2 pounds per month thereafter as appetite normalizes and caloric intake creeps back up. The STEP 1 extension data showed approximately two-thirds of total weight loss regained within one year of discontinuation, emphasizing the chronic nature of obesity and the need for sustained treatment in most cases.
On restarting, the weight loss curve typically follows a compressed version of the original timeline, reaching previous weight within 60 to 75 percent of the original timeframe. This suggests that the body retains some metabolic "memory" of the previous treatment that accelerates the response on subsequent exposure.
The psychological timeline: how your relationship with food changes
The physical results get all the attention. The psychological transformation is often more profound.
Weeks 1 through 4: the quiet surprise
Most researchers describe the first month as a revelation. Not because of weight loss, but because of the sudden absence of food obsession. For people who have spent years or decades in a constant negotiation with hunger, cravings, and food choices, the silence is almost disorienting. You find yourself at 2 PM realizing you forgot about lunch. You walk past a bakery without the usual gravitational pull. The cookie jar sits unopened. Not through willpower. Through genuine indifference.
Weeks 5 through 12: the new normal
The novelty of reduced food noise fades, and a new baseline emerges. Eating becomes functional rather than emotional. This can trigger unexpected grief in researchers whose relationship with food was deeply tied to comfort, social connection, or identity. Some find this period emotionally challenging even as the physical results accelerate.
Months 3 through 6: identity shift
As your body changes, your self-concept begins to shift. Clothes shopping becomes exciting rather than dreaded. Physical activities that were off limits become possible. Social dynamics change. This period often brings a mixture of excitement and anxiety, the thrill of transformation combined with uncertainty about who you are becoming.
Beyond 6 months: integration
By the second half of the year, most researchers have integrated their new relationship with food into daily life. The changes feel normal rather than novel. Eating patterns have stabilized. Food choices are habitual rather than requiring constant decision-making. The psychological benefits of reduced food noise, combined with the physical confidence from weight loss, create a positive feedback loop that supports long-term adherence.
Frequently asked questions
How much weight can I expect to lose in the first month on semaglutide?
At the starting dose of 0.25 mg, most researchers lose 2 to 6 pounds during the first four weeks. Much of this initial loss is water weight and reduced GI contents rather than pure fat loss. The first month results are primarily about establishing tolerance and building toward therapeutic doses rather than maximizing weight loss.
When will I notice the most significant changes?
The most dramatic changes typically occur between weeks 12 and 24 (months 3 to 6), when you are at or approaching the full 2.4 mg dose and the cumulative caloric deficit has produced visible body changes. This is when before and after differences become most apparent to both you and others around you.
Is it normal to not lose weight in the first two weeks?
Yes. At 0.25 mg, the dose is intentionally sub-therapeutic for weight loss. Some researchers see no scale change at all during weeks one and two. This does not predict future response. The medication needs time to build in your system, and therapeutic doses are not reached until weeks 9 to 17 depending on the titration schedule. Weight loss typically begins in earnest at the 0.5 mg or 1.0 mg dose level.
What should I do if my weight loss plateaus?
First, confirm it is a true plateau, at least three weeks of no downward trend in the weekly average, rather than normal fluctuation. Then evaluate protein intake (increase to 1.0 to 1.2 g/kg), exercise habits (add or intensify resistance training), sleep quality, stress levels, and alcohol consumption. If the plateau persists beyond six weeks despite optimizing these factors, discuss a protocol review or potential switch to a dual agonist option with your provider.
Can I speed up the titration to get results faster?
Accelerating the titration schedule is not recommended. The four-week intervals between dose increases allow your GI system to adapt, reducing the severity of side effects and improving long-term tolerability. Researchers who skip ahead to higher doses too quickly often experience severe nausea and vomiting that forces them to pause treatment entirely, ultimately delaying results rather than accelerating them.
How do semaglutide results compare to tirzepatide?
In head-to-head comparisons, tirzepatide produces approximately 5 to 7 percent more total body weight loss than semaglutide at maximum doses. The week-by-week timeline for tirzepatide follows a similar pattern but reaches higher total weight loss by the end of the treatment period. Both medications show similar side effect profiles, though individual response to each can vary significantly.
What happens to my results if I stop taking semaglutide?
Research shows that most people regain approximately two-thirds of lost weight within one year of discontinuation. The appetite suppression fades, food noise returns, and caloric intake gradually increases. This is why semaglutide is increasingly viewed as a long-term or potentially lifelong medication for obesity management, similar to how blood pressure or cholesterol medications are used chronically.
Does the formulation type affect how quickly I see results?
Injectable semaglutide produces the most predictable and well-studied results. Oral and sublingual formulations have different bioavailability profiles that can affect both the timeline and magnitude of results. Compounded formulations with added ingredients like B12 or glycine may provide additional benefits but follow the same fundamental timeline for weight loss.
External resources
For researchers committed to maximizing their semaglutide results at every phase, SeekPeptides provides the most comprehensive protocol library available, with evidence-based dosing guides, body composition tracking tools, and a community of thousands who have navigated this exact timeline. Members access personalized protocol adjustments, expert-reviewed optimization strategies, and direct support for every stage of the journey.
In case I do not see you, good afternoon, good evening, and good night. May your titration stay smooth, your results stay consistent, and your protocols stay optimized.