Feb 27, 2026

Tirzepatide and retatrutide look similar on paper. Both target GLP-1 receptors. Both produce significant weight loss in clinical trials. Both use once-weekly subcutaneous injections. But in practice, these are fundamentally different compounds with different receptor profiles, different side effect landscapes, and different outcomes for the people who use them.
The decision to switch is not simple.
Tirzepatide is a dual agonist. It hits GLP-1 and GIP receptors with precision, and it has delivered some of the most impressive weight loss data the pharmaceutical world has seen. But retatrutide goes further. It is a triple agonist, adding glucagon receptor activation to the mix, and that third receptor changes everything from thermogenesis to liver fat metabolism to the total percentage of body weight people lose in controlled studies. Phase 2 data showed up to 24.2% body weight reduction at the highest dose over 48 weeks. That number stopped the research community in its tracks.
So why does anyone hesitate to switch?
Because the transition itself carries real risks. Overlapping two GLP-1 class drugs can amplify gastrointestinal side effects to an unbearable degree. Starting retatrutide at the wrong dose after months on tirzepatide can trigger nausea, vomiting, and diarrhea severe enough to make people abandon the protocol entirely. And the washout period between stopping one and starting the other remains one of the most misunderstood aspects of the entire process. This guide covers every element of switching between GLP-1 compounds, with specific protocols, dose mapping, timeline expectations, and the science behind why the glucagon receptor makes retatrutide a compelling option for people who have hit their ceiling on tirzepatide. SeekPeptides has compiled the most current clinical data and community protocols into one resource so you can make an informed decision about whether this transition makes sense for your research goals.
Why people switch from tirzepatide to retatrutide
Nobody switches medications without a reason. The decision to move from tirzepatide to retatrutide usually comes from one of several frustrations, and understanding your specific motivation matters because it shapes how you approach the transition.
The tirzepatide plateau problem
Plateaus happen. They happen to almost everyone on GLP-1 receptor agonists, and they happen regardless of how disciplined the protocol is. After months of consistent weight loss on tirzepatide, the body adapts. Metabolic rate adjusts. Appetite suppression weakens. The scale stops moving even at the maximum tirzepatide dose.
This is not failure. This is biology.
When researchers explore why tirzepatide stops working, they find that the body reaches a new metabolic equilibrium. The GLP-1 and GIP receptor pathways have been maximally stimulated, and the system has recalibrated around that stimulation. Adding the glucagon receptor, which is what retatrutide does, introduces an entirely new metabolic lever that the body has not adapted to. It forces the system to recalibrate again, often producing renewed weight loss in people who had completely stalled on compounded tirzepatide.
The glucagon receptor advantage
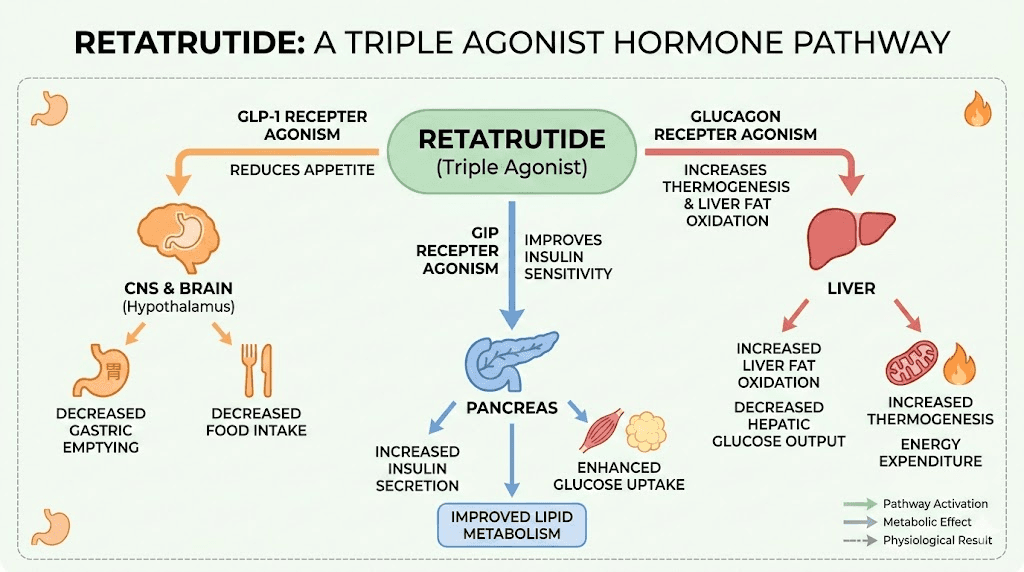
This is the real story behind the switch. Tirzepatide targets two receptors. Retatrutide targets three. That third receptor, the glucagon receptor, does something neither GLP-1 nor GIP can do on their own. It directly increases energy expenditure through thermogenesis. It promotes lipolysis, the breakdown of stored fat. And it reduces lipogenesis, meaning the body creates less new fat from the food you eat.
Think about what that means in practical terms.
With tirzepatide, your body reduces appetite and improves insulin sensitivity. Excellent mechanisms. But with retatrutide, you get those same appetite and insulin benefits plus your body actively burns more calories at rest while simultaneously breaking down stored fat more efficiently. The comparison between retatrutide and tirzepatide reveals this as more than a marginal improvement. It represents a fundamentally different approach to metabolic intervention, one that addresses energy expenditure in addition to energy intake. For researchers interested in the most effective peptides for weight loss, the glucagon component is what makes retatrutide stand apart from every other GLP-1 class compound currently available.
Clinical weight loss data that drove the conversation
Numbers matter. In the Phase 2 trial, participants on the 12mg dose of retatrutide lost up to 24.2% of their body weight over 48 weeks. The Phase 3 TRIUMPH-4 trial showed an average loss of 71.2 pounds, with additional benefits for osteoarthritis relief. A meta-analysis comparing the two compounds directly found retatrutide produced an average of 16.34 kg of weight loss compared to 11.82 kg for tirzepatide.
That is a 38% greater reduction.
For someone who has been tracking their tirzepatide weight loss timeline and watching progress slow, those numbers are hard to ignore. The three-way comparison between semaglutide, tirzepatide, and retatrutide consistently shows retatrutide at the top for total body weight percentage lost, and the difference becomes more pronounced at higher doses and longer treatment durations. Researchers looking at retatrutide dosage charts for weight loss can see the dose-response relationship clearly in the clinical data.
Liver fat reduction and metabolic health
Weight loss is not the only reason people consider the switch. Retatrutide Phase 2 data showed remarkable improvements in metabolic associated steatotic liver disease, previously called NAFLD. The glucagon receptor activation drives this effect by promoting hepatic fat oxidation, essentially telling the liver to burn its stored fat reserves. For researchers managing metabolic health beyond the number on the scale, this benefit has no equivalent in tirzepatide protocols. Understanding fat-burning peptide mechanisms helps explain why that third receptor makes such a profound difference in liver health outcomes.
Understanding the key differences between tirzepatide and retatrutide
Before you switch, you need to understand exactly what you are switching to. These compounds share some mechanisms, but they diverge in ways that directly affect dosing, side effects, and outcomes. The retatrutide versus tirzepatide dosage chart reflects these structural differences at every escalation point.
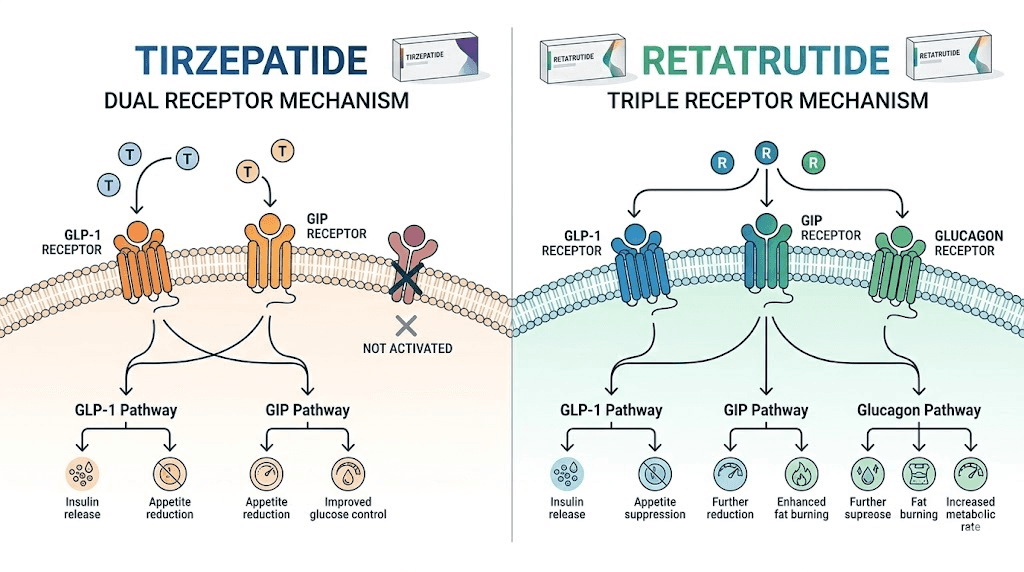
Dual versus triple agonist mechanism
Tirzepatide is built on a GIP backbone. It activates GLP-1 receptors with strong potency and GIP receptors with even stronger potency. These two pathways work together to reduce appetite, slow gastric emptying, and improve how the body handles glucose and insulin. For many people, this dual mechanism delivers transformative results, which is why tirzepatide works quickly for most users.
Retatrutide is also built on a GIP backbone, a 39-amino-acid peptide that activates all three receptors, but with different potency ratios. Its GLP-1 activation is moderate. Its GIP activation is strong. And its glucagon receptor activation is the defining feature, the piece that no other approved or near-approved weight loss compound offers at this potency level.
The glucagon receptor does several things simultaneously. It increases hepatic glucose output in the short term, which sounds counterintuitive for a metabolic drug, but the body compensates through increased energy expenditure. It activates brown adipose tissue. It promotes the breakdown of fatty acids. And in the liver specifically, it drives fat oxidation at rates that produce the MASLD improvements seen in trials. The researchers studying retatrutide dosing in detail have mapped how these three pathways interact at each dose level.
Pharmacokinetic differences that matter for switching
Half-life determines how long a compound stays active in your system. Tirzepatide has a half-life of approximately 5 days. Retatrutide runs slightly longer at approximately 6 days. Both support once-weekly dosing, which simplifies administration protocols. But when switching between them, these half-lives determine the critical washout period you must observe.
Five half-lives is the standard pharmacokinetic benchmark for clearing a drug from the system. For tirzepatide, that means 25 days minimum before less than 3% of the final dose remains active. For those who want to be conservative, 30 days provides additional clearance buffer. Understanding how long retatrutide lasts in the body is equally important because you need to account for the accumulation phase once you begin the new compound.
Storage and handling also differ slightly between the two. If you have been following tirzepatide refrigeration protocols, the same general principles apply to retatrutide, but you should review proper peptide storage after reconstitution for the new compound to ensure potency throughout your transition.
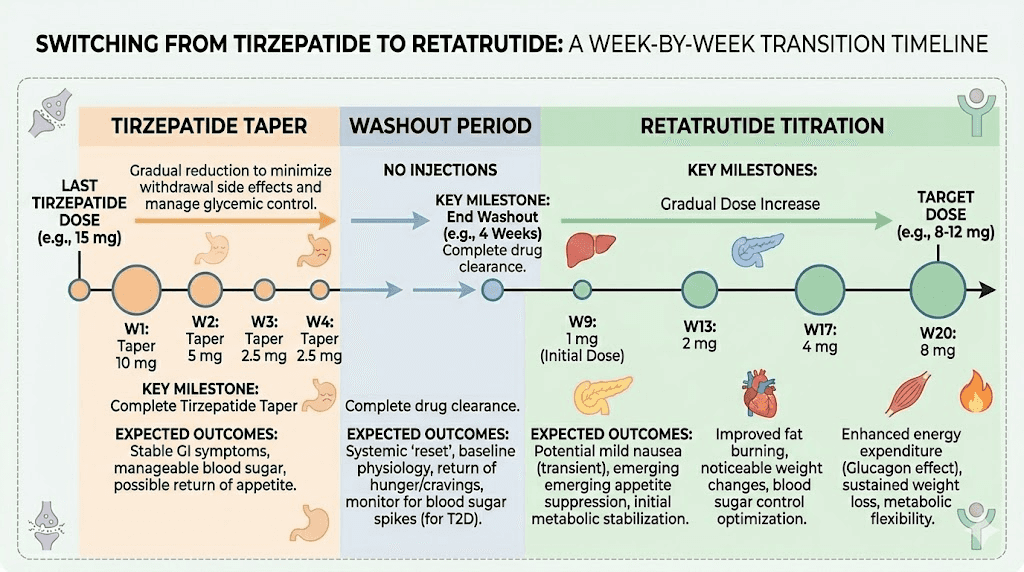
The washout period: what it is and why it matters
This section could save you weeks of unnecessary suffering. Skip it at your own risk.
The washout period is the time between your last tirzepatide injection and your first retatrutide injection. It exists for a critical reason. Both compounds act on GLP-1 and GIP receptors. If they overlap in your system, you get additive stimulation of those receptors, and the result is not better efficacy. It is worse side effects. Dramatically worse, in many cases.
Why overlapping two GLP-1 class drugs is dangerous
When two GLP-1 agonists are active simultaneously, the gastrointestinal effects stack. Nausea becomes severe nausea. Reduced gastric motility becomes gastroparesis-level stasis. The risk of vomiting, dehydration, and electrolyte imbalances climbs sharply. None of the clinical trials studied concurrent use because no responsible research protocol would design that overlap intentionally.
The side effect profiles of GLP-1 agonists are dose-dependent. Stacking two compounds effectively doubles the receptor stimulation at GLP-1, which means you are operating at a stimulation level that exceeds what any single-agent trial has tested. People who have experienced tirzepatide diarrhea or tirzepatide constipation already know these side effects can be difficult to manage. Doubling the receptor load makes them potentially unmanageable.
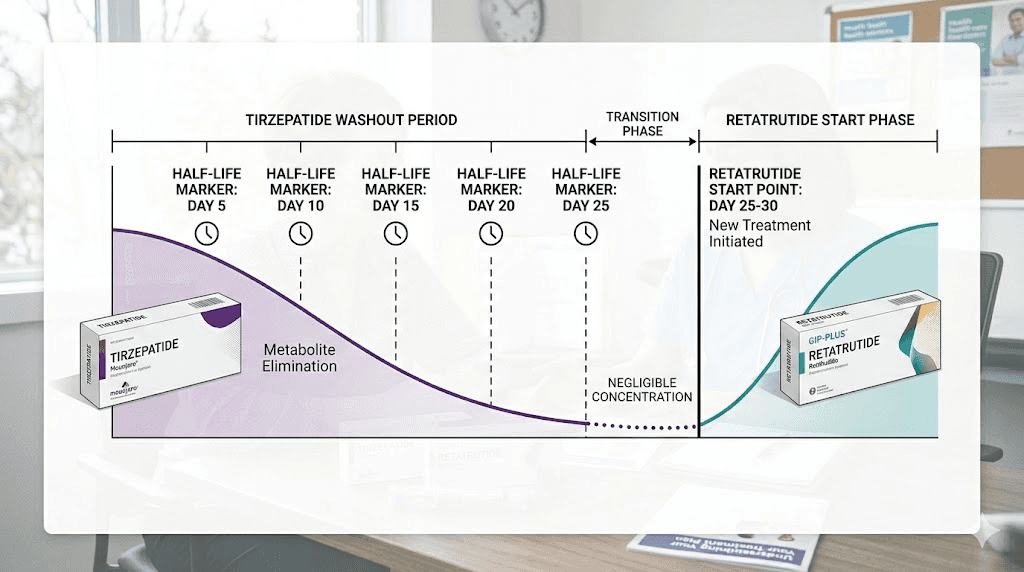
Half-life calculations for safe clearance
Tirzepatide half-life: approximately 5 days. After one half-life, 50% remains. After two half-lives (10 days), 25% remains. After three (15 days), 12.5%. After four (20 days), 6.25%. After five half-lives (25 days), approximately 3.1% of the last dose remains in circulation.
That 3% threshold is the standard clearance benchmark.
However, if you were on a high dose of tirzepatide, even 3% of a 15mg dose means roughly 0.45mg of active compound still circulating. That is not negligible when you are simultaneously introducing a new GLP-1 agonist. This is why conservative recommendations push the washout to 30 full days, particularly for those transitioning from higher tirzepatide doses.
Timeline recommendations by previous dose
For those coming off low-dose tirzepatide (2.5 to 5mg), a 20 to 25 day washout is generally sufficient. The absolute amount of residual compound is small, and most people tolerate the transition well at this range.
Mid-dose tirzepatide users (7.5 to 10mg) should plan for 25 to 30 days. The residual compound load is higher, and GI sensitivity tends to increase with longer tirzepatide exposure at these doses.
High-dose users (12.5 to 15mg) should commit to a full 30-day washout, possibly extending to 35 days if they experienced significant GI side effects on tirzepatide. For those who want to understand this transition in the context of other compounds, the guide on conversion between GLP-1 compounds provides useful reference points for dose equivalency thinking.
What to expect during the washout
Appetite will return. This is perhaps the most psychologically challenging aspect of the washout period. The appetite suppression that tirzepatide provided will fade as the compound clears your system. Research shows that approximately two-thirds of patients regain weight when they stop GLP-1 receptor agonists, and even a 25 to 30 day gap can trigger some weight regain if dietary habits are not maintained.
Prepare for this. Maintain your protein intake. Keep your meal schedule consistent. Track your calories manually if needed. The washout is temporary, and the weight regain during this window is typically modest, perhaps 2 to 5 pounds, if you stay disciplined with nutrition. For dietary guidance during this period, the recommendations in foods to avoid on GLP-1 therapy still apply, as your GI system will be readjusting to normal motility. A structured diet plan can help maintain progress during this gap.
Step-by-step transition protocol
This is the section you will bookmark. The actual protocol for getting from tirzepatide to retatrutide safely and effectively.
Step one: preparing for the switch
Before your last tirzepatide injection, gather everything you need for the retatrutide phase. This includes the compound itself, bacteriostatic water for reconstitution, appropriate syringes, and a clear understanding of your retatrutide reconstitution protocol. Do not wait until the washout period begins to source your supplies. The washout should be spent preparing your body, not scrambling for materials.
Document your current state. Record your weight, waist circumference, blood pressure if available, and a subjective rating of appetite suppression, energy levels, and GI comfort. These baseline measurements become critical reference points once you start retatrutide.
Review your reconstitution skills because the process for retatrutide follows the same fundamental principles but may use different concentrations. The peptide reconstitution calculator at SeekPeptides can help you determine exact volumes for your specific vial concentration.
Step two: the last tirzepatide dose
Take your final tirzepatide injection on your normal schedule day. Do not take a higher dose thinking it will "carry you through" the washout. Do not split the dose across multiple days. Just take your standard dose and mark the calendar. The washout clock starts from this injection.
Step three: the washout period (days 1 through 25-30)
During this period, focus entirely on maintaining your results without pharmaceutical support. Your nutritional framework should remain in place. High protein intake is especially important because it provides the greatest satiety per calorie, partially compensating for the loss of GLP-1 mediated appetite suppression.
Expect appetite to return gradually between days 7 and 14 as tirzepatide levels drop below therapeutic thresholds. This is normal. It does not mean the compound "stopped working." It means it is clearing your system as intended. The information about weaning off tirzepatide provides additional strategies for managing this transition period.
Step four: starting retatrutide
After the full washout period, you begin retatrutide. But here is where prior tirzepatide experience changes the standard protocol. The typical retatrutide titration for GLP-1 naive individuals starts at 1mg weekly. You are not GLP-1 naive. Your body has already adapted to GLP-1 and GIP receptor agonism, which means you can potentially start at a slightly higher dose.
The recommendations differ based on your previous tirzepatide dose.
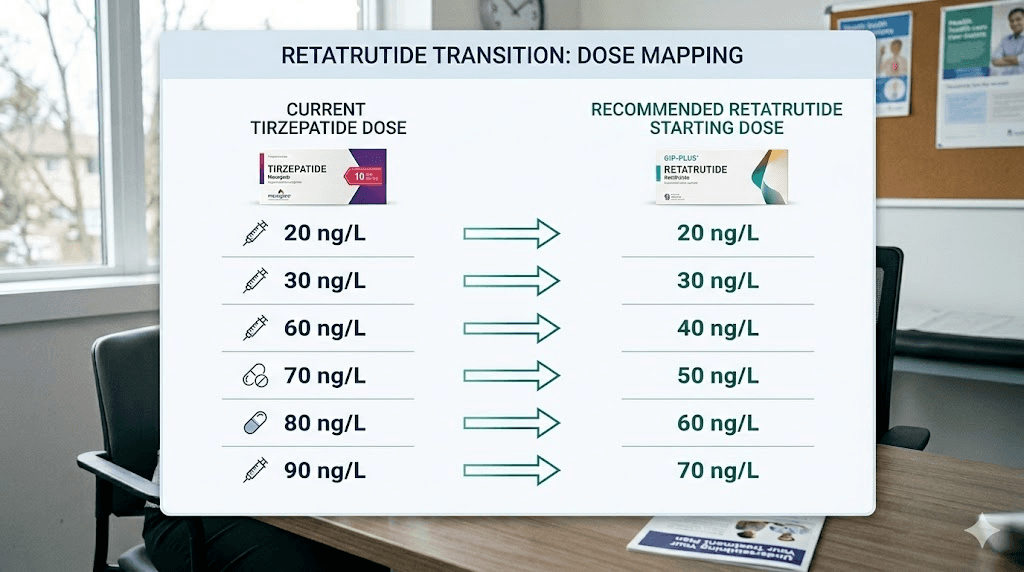
If you were on 2.5 to 5mg tirzepatide, start retatrutide at 1mg weekly. You had relatively modest GLP-1 exposure, and starting at the standard introductory dose is prudent. Follow the standard retatrutide starting dose protocol with escalation every 4 weeks.
If you were on 7.5 to 10mg tirzepatide, you can start retatrutide at 2mg weekly. Your body has demonstrated tolerance to significant GLP-1/GIP agonism, and the 2mg dose introduces the glucagon receptor component without overwhelming GI tolerance. The retatrutide dosage schedule after high-dose tirzepatide covers this scenario in detail.
If you were on 12.5 to 15mg tirzepatide, starting at 2mg retatrutide is still recommended. Despite your high GLP-1 tolerance from tirzepatide, retatrutide introduces the glucagon receptor, which is entirely new to your system. Starting at 2mg respects that new mechanism while acknowledging your existing receptor adaptation.
Step five: the escalation schedule
Once you have established your starting dose, follow a 4-week escalation pattern. The standard retatrutide dose schedule looks like this for someone starting at 2mg after tirzepatide experience:
Weeks 1 through 4: 2mg weekly. Monitor GI tolerance carefully. The glucagon receptor effects may produce sensations you did not experience on tirzepatide, including mild thermogenic warmth and occasional changes in skin sensitivity.
Weeks 5 through 8: 4mg weekly. This is where most people begin noticing the difference between tirzepatide and retatrutide. Appetite suppression returns to familiar levels, but you may also notice increased energy expenditure, subtle changes in body temperature regulation, and more pronounced fat loss even without additional caloric restriction.
Weeks 9 through 12: 8mg weekly. Therapeutic effects become pronounced. Weight loss accelerates. The combination of all three receptor pathways operating simultaneously produces metabolic effects that exceed what tirzepatide delivered at any dose. Reference the retatrutide dosing guide for additional adjustments based on tolerance.
Weeks 13 and beyond: 12mg weekly (maintenance). This is the dose that produced the 24.2% body weight reduction in Phase 2 trials. Not everyone needs to reach this dose. Some researchers find their optimal response at 8mg. The right maintenance dose is the one that produces consistent results with tolerable side effects. The complete retatrutide dosage chart provides visual reference for this entire trajectory.
Managing GI side effects during the transition
Gastrointestinal side effects are the number one reason people abandon GLP-1 receptor agonist therapy. They are also the number one reason people botch the tirzepatide to retatrutide transition. Understanding why they happen and how to manage them is not optional. It is essential.
Why GI symptoms may intensify during the switch
Even with a proper washout, your GI system has been operating under GLP-1 mediated slowed gastric emptying for months. When you introduce retatrutide, you are reintroducing that gastric slowing while simultaneously adding glucagon receptor effects. The glucagon receptor can cause nausea through a different pathway than GLP-1, meaning some people experience a qualitatively different type of nausea than what they felt on tirzepatide.
Clinical data supports this concern. Nausea rates in retatrutide trials reached approximately 45% at the maximum dose, compared to roughly 31% for tirzepatide. That is a meaningful difference, and it reflects the additive nausea pathways from three receptors versus two. For context on managing these effects, the approaches used for GLP-1 constipation and GLP-1 bloating apply here with some modifications.
Prevention strategies that actually work
Eat smaller, more frequent meals. This is standard advice for GLP-1 therapy, but it becomes even more critical during the transition. Smaller meals reduce the volume the slowed stomach must process, decreasing the trigger for nausea and reflux.
Stay hydrated aggressively. Dehydration worsens every GI side effect and is a common cause of the headaches associated with GLP-1 therapy. Aim for a minimum of 80 ounces of water daily, more if you experience any vomiting or diarrhea.
Avoid high-fat meals during the first 4 weeks of retatrutide. The glucagon receptor activation changes how your body processes dietary fat, and combining that with the gastric slowing from GLP-1 agonism creates a perfect storm for nausea. The meal planning strategies from tirzepatide protocols translate well here, with even greater emphasis on lean protein and complex carbohydrates during the initial weeks.
Consider the timing of your injection. Many researchers find that injection timing affects the severity of GI side effects. Taking the injection before bed allows you to sleep through the peak of initial nausea, which typically occurs 6 to 12 hours after administration.
When to slow down the escalation
Not everyone should follow the standard 4-week escalation. If you experience any of the following, remain at your current dose for an additional 2 to 4 weeks before escalating:
Nausea lasting more than 3 days after injection
Vomiting more than once per week
Inability to maintain adequate caloric intake (under 1,000 calories daily)
Signs of dehydration despite increased fluid intake
New onset of significant fatigue that interferes with daily function
Slowing escalation is not failure. It is intelligent protocol management. The clinical trials allowed for dose adjustment based on tolerability, and your protocol should too. Some people reach their optimal retatrutide dose in 13 weeks. Others take 20. Both timelines can produce excellent outcomes.
Monitoring your progress after switching
What you measure determines what you can improve. Tracking the right metrics during your transition from tirzepatide to retatrutide provides objective data about whether the switch is working, whether your dose needs adjustment, and whether you are experiencing the full benefit of triple receptor agonism.
What to track
Body weight is the obvious metric, but it is not the most informative during the first 4 to 6 weeks. Water weight fluctuations from changing GI motility, shifts in food volume, and the body adjusting to new receptor stimulation all create noise in the scale data. Weigh yourself weekly at the same time, under the same conditions, but do not overreact to short-term fluctuations.
Waist circumference provides a better early signal. The glucagon receptor activation in retatrutide preferentially drives visceral fat loss, and waist measurement captures this before the scale does. For those interested in targeting visceral fat specifically, this metric becomes your primary outcome measure.
Track your appetite on a simple 1 to 10 scale daily. This helps you understand when the retatrutide is reaching therapeutic levels and how the appetite suppression compares to what you experienced on tirzepatide. Most people find that retatrutide appetite suppression at 4mg equals what they experienced at roughly 7.5 to 10mg tirzepatide.
Energy levels matter too. The glucagon receptor increases energy expenditure, and many people report a subtle but noticeable increase in baseline energy once they reach the 4 to 8mg dose range. This is different from stimulant energy. It is more of a metabolic warmth, a sense that the body is running at a slightly higher RPM. The metabolic effects of these compounds become more apparent when you have experienced both.
Note any new side effects that you did not experience on tirzepatide. Dysesthesia, a skin tingling or sensitivity, was reported in approximately 20.9% of participants at the highest retatrutide dose. This is a glucagon receptor mediated effect that has no equivalent in tirzepatide therapy. It is typically mild and often resolves with continued use, but it should be documented.
Expected timelines for results
Understanding how long retatrutide takes to work sets appropriate expectations. If you are starting at 2mg after tirzepatide, the first 4 weeks are primarily about establishing tolerance and allowing the glucagon receptor pathway to activate. Weight loss during this period is often modest, perhaps 1 to 3 pounds, because the dose is below the threshold where all three receptors are fully engaged.
Weeks 5 through 8 at 4mg is where most people begin seeing meaningful results. Weight loss accelerates. Appetite suppression deepens. The thermogenic effect becomes noticeable. Expect 4 to 8 pounds during this phase if your nutrition is consistent.
Weeks 9 through 12 at 8mg typically produces the most dramatic changes. This is the therapeutic sweet spot for many researchers, where the balance of efficacy and tolerability optimizes. Weight loss of 8 to 12 pounds during this phase is consistent with clinical trial trajectories.
For a deeper analysis of when effects manifest, how long retatrutide takes to kick in after tirzepatide provides specific timelines based on prior tirzepatide dose levels.
Common mistakes when switching from tirzepatide to retatrutide
The transition between these two compounds has enough nuance that even experienced researchers make avoidable errors. Knowing the mistakes before you encounter them is the difference between a smooth transition and an abandoned one.
Mistake one: starting retatrutide at too high a dose
The logic seems sound. You were on 15mg tirzepatide. You tolerated it well. You can handle a high starting dose of retatrutide. Wrong. Tirzepatide tolerance does not transfer cleanly to retatrutide because of the glucagon receptor. Your body has never experienced pharmaceutical glucagon receptor agonism at these potencies. The GLP-1 and GIP tolerance you built on tirzepatide is relevant, but the glucagon component is a wild card.
Starting at 4mg or higher "to save time" is the fastest way to end up nauseated, dehydrated, and questioning why you switched in the first place. Respect the 2mg starting recommendation. It exists for a reason. The common mistakes in peptide research consistently include dosing too aggressively with new compounds.
Mistake two: skipping or shortening the washout
Impatience kills protocols. Some researchers take their last tirzepatide injection and start retatrutide 10 or 14 days later, reasoning that "most of the tirzepatide should be gone." Most is not all. At 14 days, roughly 14% of the last tirzepatide dose remains active. That is not a trace amount. That is enough to produce meaningful GLP-1 and GIP receptor stimulation that stacks with the retatrutide you are introducing.
The result is predictable. Severe nausea. Potential vomiting. Possible need to abandon the retatrutide dose and extend the washout anyway, setting you back further than if you had just waited the full 25 to 30 days initially.
Mistake three: ignoring new side effects unique to retatrutide
Dysesthesia catches people off guard. If your experience with GLP-1 compounds is limited to tirzepatide or semaglutide, you have never encountered this particular side effect. It manifests as tingling, numbness, or altered skin sensitivity, most commonly in the extremities. At the highest retatrutide dose, about 1 in 5 participants experienced it.
This is not dangerous. But it is alarming if you are not expecting it.
Similarly, the thermogenic effect of glucagon receptor activation can feel unusual. Some people describe it as a mild flushing or warmth, similar to the feeling after taking niacin but less intense. Others notice increased sweating during exercise. These are signs that the glucagon receptor is doing its job, not signs of a problem. Understanding musculoskeletal side effects from the tirzepatide experience can help you differentiate between expected transitional effects and genuine concerns.
Mistake four: not adjusting diet and supplementation
Your nutritional strategy on tirzepatide was calibrated for dual agonist therapy. Retatrutide changes the metabolic picture. The increased energy expenditure from glucagon receptor activation means your total daily energy expenditure is higher, and your caloric needs shift accordingly. Maintaining the same caloric deficit you had on tirzepatide without accounting for the additional expenditure can push you into an excessive deficit that promotes muscle loss, fatigue, and metabolic adaptation.
Protein intake should increase by 10 to 20% when transitioning to retatrutide to account for the higher metabolic rate and protect lean mass. Consider supplementation strategies including electrolytes, B vitamins, and magnesium, all of which support the increased metabolic demands. Protein shakes designed for GLP-1 users offer a convenient way to maintain intake when appetite is heavily suppressed.
The glucagon receptor difference: what it means for your results
The glucagon receptor is not just an add-on. It fundamentally changes what this class of drugs can achieve. Understanding the mechanism helps you appreciate why retatrutide produces different results than tirzepatide and why the transition is worth the effort for the right candidates.
Thermogenesis and energy expenditure
Glucagon receptor activation increases energy expenditure through multiple pathways. It stimulates brown adipose tissue, which burns calories to generate heat rather than storing them as white fat. It increases hepatic energy consumption as the liver ramps up fat oxidation and gluconeogenesis. And it has direct effects on resting metabolic rate that compound over time.
In practical terms, this means retatrutide users burn more calories at rest than tirzepatide users at equivalent appetite suppression levels. The weight loss is driven not just by eating less, but by burning more. This dual mechanism, reduced intake plus increased expenditure, explains why the Phase 2 data showed such dramatically higher weight loss percentages than any dual agonist trial. For researchers exploring the best peptide stacks for weight loss, understanding this thermogenic component reframes how you think about metabolic intervention entirely.
Liver fat reduction and MASLD improvement
The liver data from retatrutide trials may end up being more consequential than the weight loss data for public health. Glucagon receptor activation tells the liver to oxidize its fat stores. In the Phase 2 trial, participants on retatrutide showed significant reductions in hepatic fat content, with some achieving near-complete resolution of metabolic associated steatotic liver disease.
Tirzepatide also improves liver fat, primarily through weight loss and improved insulin sensitivity. But retatrutide adds a direct hepatic mechanism on top of those indirect benefits. The liver is being told to burn fat from two directions: indirectly through reduced caloric intake and improved metabolism, and directly through glucagon receptor mediated fat oxidation. For people transitioning specifically because of metabolic health concerns beyond weight, this distinction matters enormously.
Lipid metabolism changes
The glucagon receptor also reduces lipogenesis, the process by which the body converts excess calories into new fat stores. Combined with the increased lipolysis (fat breakdown) and thermogenesis (fat burning), the net effect on body composition is more favorable than what either GLP-1 or GIP agonism can produce alone. Researchers comparing different GLP-1 class compounds consistently find that the triple agonist approach produces the most comprehensive metabolic improvement when all three pathways are measured.
Retatrutide dosing protocols after tirzepatide by previous dose level
Your previous tirzepatide dose determines not just where you start retatrutide, but how quickly you should escalate and what your target maintenance dose should be. Here are three distinct protocols based on where you are coming from.
Protocol for those on low-dose tirzepatide (2.5 to 5mg)
You had relatively mild GLP-1/GIP receptor exposure. Your body adapted to these pathways at low intensity. This actually makes your transition slightly easier in some ways and slightly harder in others.
Easier because: your GI system was not heavily challenged by tirzepatide, so the transition to a new compound is less of a shock. The washout is shorter at the minimum recommended 20 to 25 days. And your baseline tolerance is more predictable.
Harder because: you lack the deep receptor adaptation that higher-dose tirzepatide users have, meaning the GLP-1 and GIP components of retatrutide may hit you harder than expected at relatively low doses.
Recommended protocol:
Washout: 20 to 25 days
Starting dose: 1mg weekly
Week 5: escalate to 2mg
Week 9: escalate to 4mg
Week 13: escalate to 8mg
Week 17: consider 12mg based on response and tolerance
This protocol follows the standard retatrutide peptide dosage chart exactly, treating you similarly to a GLP-1 naive individual. The starting dose considerations from your tirzepatide experience still inform your approach, but conservatism serves you well here.
Protocol for those on mid-dose tirzepatide (7.5 to 10mg)
You are the most common transition candidate. You achieved meaningful results on tirzepatide, potentially hit a plateau, and are looking for the next level of metabolic intervention.
Recommended protocol:
Washout: 25 to 30 days
Starting dose: 2mg weekly
Week 5: escalate to 4mg
Week 9: escalate to 8mg
Week 13: escalate to 12mg based on response
This accelerated schedule saves 4 weeks compared to the standard titration. You skip the 1mg introductory phase because your GLP-1/GIP receptor tolerance has already been established. The retatrutide 10mg dosing information helps you understand what to expect as you approach higher therapeutic doses. Use the retatrutide dosage calculator to verify your specific volumes based on reconstitution concentration.
Protocol for those on high-dose tirzepatide (12.5 to 15mg)
You have maximized tirzepatide. The GLP-1 and GIP pathways have been pushed to their pharmacological limits in your system. Your transition is motivated by either a weight loss plateau, a desire for the metabolic benefits of glucagon receptor activation, or both.
Recommended protocol:
Washout: 30 to 35 days (conservative, recommended)
Starting dose: 2mg weekly (resist the urge to start higher)
Week 5: escalate to 4mg
Week 9: escalate to 8mg
Week 13: escalate to 12mg
Yes, you start at 2mg despite coming from 15mg tirzepatide. The glucagon receptor is new territory regardless of your tirzepatide experience. The detailed guide on retatrutide dosing after high-dose tirzepatide explains the pharmacological reasoning behind this conservative approach. Your injection technique should follow standard subcutaneous protocols, and selecting appropriate injection sites helps minimize local reactions during the transition.
What to expect week by week after switching
Knowledge of what is coming reduces anxiety and prevents premature protocol abandonment. Here is a realistic week-by-week breakdown based on clinical data and community reports from researchers who have made this transition.
Weeks 1 through 4: the initial adjustment
The first month on retatrutide after tirzepatide feels like starting over. Your body is encountering a new compound with a new receptor profile, and even though two of the three pathways are familiar, the experience is different.
Week one is the most uncertain. Many people feel relatively little on 2mg retatrutide, especially if they are accustomed to high-dose tirzepatide. Appetite suppression may be mild. Weight loss may be negligible or even slightly positive due to the washout period weight regain stabilizing. This is normal. This is expected.
By week two, the GLP-1 and GIP effects begin registering at a noticeable level. Appetite starts decreasing again. Gastric motility slows. The familiar GLP-1 sensations return, but with a slightly different character. Some people describe the appetite suppression as "wider" on retatrutide, affecting not just hunger but food preoccupation and cravings in a way that feels broader than tirzepatide.
Weeks three and four solidify the adaptation. GI symptoms, if present, typically peak during week two and begin improving by week three as the body adjusts. The early experience on GLP-1 compounds provides useful parallels, though the retatrutide experience includes the unique glucagon elements.
Weeks 5 through 8: building therapeutic effects
At 4mg weekly, retatrutide begins showing its true character. The glucagon receptor, which was mildly active at 2mg, reaches a potency level where thermogenic effects become perceptible. Many researchers report feeling slightly warmer, especially during physical activity. Some notice increased sweating. Others describe a subtle increase in baseline energy that feels different from caffeine or stimulant energy.
Weight loss during this phase typically accelerates to 1 to 2 pounds per week. Fat loss specifically may be even higher, masked on the scale by simultaneous improvements in lean mass preservation from the glucagon-driven metabolic increase. This is where tracking waist circumference becomes more informative than the scale alone.
GI side effects usually stabilize during this phase. Most people have found their dietary rhythm and know which foods their system handles well on retatrutide.
Weeks 9 through 12: reaching therapeutic range
At 8mg weekly, you are in the dose range where Phase 2 data showed significant metabolic improvements across every measured parameter. Weight loss accelerates further. Liver fat reduction, if relevant to your situation, reaches clinically meaningful levels. Insulin sensitivity improvements compound with the changes already driven by weight loss.
This is also the phase where some people experience dysesthesia for the first time. At 8mg, the glucagon receptor stimulation is strong enough to produce the tingling and altered skin sensitivity reported in trials. It typically affects the hands and feet, is mild to moderate in intensity, and does not require dose reduction in most cases. Understanding the injection site reactions you experienced on tirzepatide helps distinguish between local injection effects and the systemic dysesthesia that is unique to retatrutide.
Weeks 13 and beyond: full therapeutic effect
At 12mg weekly maintenance, you are at the dose that produced the headlines. Up to 24.2% body weight reduction over 48 weeks. Average 71.2 pounds of weight loss in the TRIUMPH-4 trial. These results were achieved with consistent dosing over extended periods, and the trajectory shows continued improvement even beyond the 48-week mark in those who maintained therapy.
Not everyone needs 12mg. Some researchers find that 8mg provides an optimal balance of results and side effects for their specific physiology. The right maintenance dose is the one you can sustain long-term with acceptable tolerability and continued progress. Tracking your results with tools like the GLP-1 plotter helps visualize your trajectory and identify whether you are on track or need dose adjustment.
Comparing weight loss outcomes: tirzepatide versus retatrutide
The data tells a clear story. Both compounds produce clinically significant weight loss. But retatrutide produces more.
Phase 2 trial data
Tirzepatide Phase 3 trials (SURPASS and SURMOUNT programs) demonstrated weight loss of 15 to 22.5% body weight depending on dose and study population. These were landmark numbers that established tirzepatide as the most effective approved weight loss medication at the time of its launch.
Retatrutide Phase 2 showed up to 24.2% body weight reduction at the 12mg dose over 48 weeks. This exceeded tirzepatide benchmarks in a shorter study duration with a smaller sample size, generating enormous interest in the Phase 3 program that followed.
Phase 3 data: TRIUMPH-4
The TRIUMPH-4 trial focused on participants with osteoarthritis and demonstrated an average weight loss of 71.2 pounds along with significant improvements in joint pain and function. While this study population was specific, the magnitude of weight loss aligns with and extends the Phase 2 findings, suggesting that the 24% range is reproducible across populations at the highest dose.
Direct comparison meta-analysis
When researchers pooled data from comparable studies, retatrutide produced an average of 16.34 kg of weight loss compared to 11.82 kg for tirzepatide. That is a difference of approximately 10 pounds in the metric often used for regulatory assessment. The broader comparison across GLP-1 compounds consistently places retatrutide at the top of efficacy rankings, though with correspondingly higher side effect rates.
For researchers considering other alternatives alongside this transition, understanding how retatrutide compares to survodutide, CagriSema, orforglipron, and mazdutide provides the full landscape of next-generation options. The mazdutide versus retatrutide comparison is particularly relevant for those weighing triple agonist alternatives.
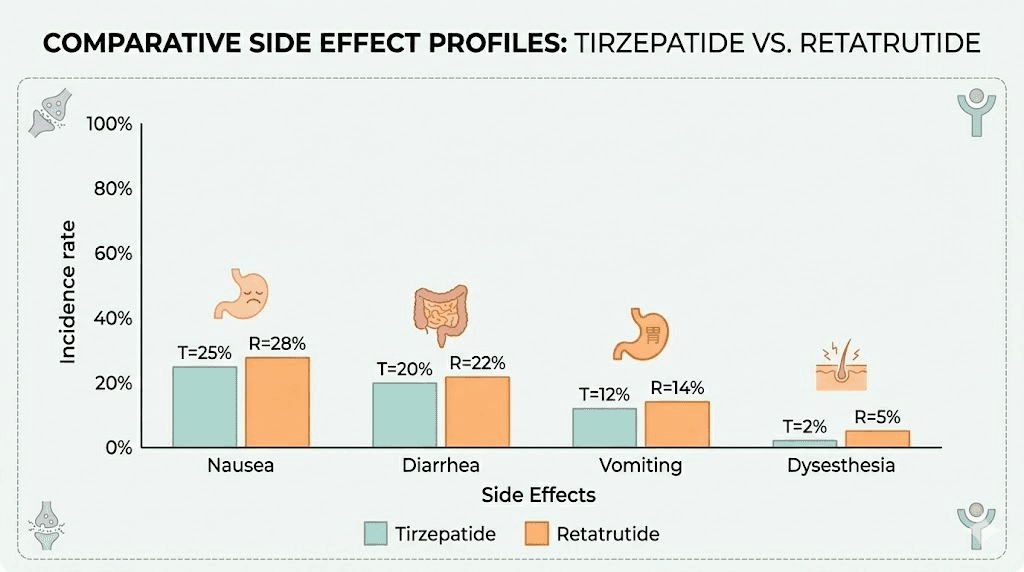
Side effect comparison: tirzepatide versus retatrutide
Understanding the side effect differences helps you prepare for what the transition may bring. Here is a comprehensive comparison based on clinical trial data.
Side effect | Tirzepatide (max dose) | Retatrutide (max dose) | Notes |
|---|---|---|---|
Nausea | ~31% | ~45% | Glucagon receptor adds a separate nausea pathway |
Diarrhea | ~17% | ~20-25% | Similar mechanism, slightly higher with triple agonism |
Vomiting | ~10% | ~12-15% | Often related to dose escalation speed |
Constipation | ~12% | ~10% | Slightly lower with retatrutide, possibly glucagon effect |
Dysesthesia | Rare | ~20.9% (at 12mg) | Unique to retatrutide, glucagon receptor mediated |
Fatigue | ~8% | ~10% | Usually transient during dose escalation |
Injection site reactions | ~5% | ~5-7% | Similar rates with proper technique |
Decreased appetite | ~15% | ~20% | Classified as side effect when extreme |
Discontinuation rate | ~4-7% | ~6-16% | Higher variability in retatrutide due to dose range |
Several patterns emerge from this data. GI side effects are uniformly higher with retatrutide, reflecting the additional receptor stimulation. The discontinuation rate range is wider, suggesting that tolerability varies more between individuals with the triple agonist. And dysesthesia is essentially a retatrutide-specific phenomenon that has no meaningful equivalent in tirzepatide therapy.
The body aches sometimes reported on tirzepatide and the anxiety some users experience do not appear to be more common on retatrutide, suggesting these effects are GLP-1/GIP mediated rather than glucagon-dependent. Similarly, menstrual cycle effects appear comparable between the two compounds based on available data. Sleep disruption, a concern for some tirzepatide users dealing with insomnia, does not appear to worsen with retatrutide.
When switching from tirzepatide to retatrutide is NOT a good idea
Not every plateau justifies a switch. Not every researcher benefits from triple agonism. Here are scenarios where staying on tirzepatide, adjusting the current protocol, or exploring other options makes more sense than switching to retatrutide.
You have not maximized tirzepatide
If you are on 5mg or 7.5mg tirzepatide and experiencing a plateau, the first step is not switching compounds. It is escalating your current dose. Tirzepatide goes up to 15mg, and many people experience renewed weight loss at higher doses. The tirzepatide dosage guide covers the full escalation pathway. Switching to retatrutide before exploring the full tirzepatide dose range means you may be abandoning a simpler solution.
Your plateau is not actually a plateau
Weight loss stalls are not always compound-related. Sometimes the issue is caloric creep, gradual increases in portion sizes or snack frequency that accumulate without being noticed. Sometimes it is a training change that shifted body composition toward muscle gain, masking fat loss on the scale. Sometimes it is another factor entirely that has nothing to do with the medication.
Before switching, spend 2 to 3 weeks tracking calories meticulously, maintaining consistent exercise, and measuring body composition, not just weight. If the plateau persists despite confirmed caloric deficit and consistent protocol, then it is likely genuine receptor adaptation and a reasonable candidate for transition.
You have severe GI sensitivity
If you struggled with GI side effects on tirzepatide, even at lower doses, retatrutide is likely to be more challenging. The side effect data is clear on this point. Retatrutide has higher nausea, vomiting, and diarrhea rates across all dose levels. If you needed split dosing or microdosing approaches to tolerate tirzepatide, moving to a compound with higher GI burden is a significant risk.
You have unmanaged cardiovascular conditions
The glucagon receptor affects heart rate and blood pressure differently than GLP-1 alone. While cardiovascular outcome data for retatrutide is still accumulating, the prudent approach for anyone with active cardiovascular disease is to remain on compounds with established cardiovascular safety profiles. Tirzepatide has more cardiovascular outcome data at this point.
Maintaining weight loss after stopping is your primary concern
If your plan involves eventually stopping peptide therapy, the question becomes: which compound produces more durable results after cessation? The data on this is limited for retatrutide since it is newer, but the general pattern with GLP-1 agonists suggests that maintaining weight loss after stopping requires lifestyle modifications regardless of which compound you used. Switching compounds does not solve the cessation problem.
Practical considerations: sourcing, storage, and handling
The logistics of switching involve more than just changing your injection schedule. Retatrutide availability, storage requirements, and handling protocols all factor into a successful transition.
Retatrutide is a research peptide that requires proper reconstitution before use. If you have been using pre-filled tirzepatide pens, the switch to reconstituting from lyophilized powder represents a practical skills change. The bacteriostatic water calculations for retatrutide depend on your desired concentration, and the reconstitution calculator simplifies this math considerably.
Sourcing matters. The quality of your retatrutide directly affects your outcomes and safety. Researching where to buy retatrutide thoroughly before purchasing is essential. Consider verified vendors with third-party testing, and review resources like the complete retatrutide buying guide and evaluations of specific suppliers such as Peptide Sciences, Paradigm Peptides, and ZLZ Peptide. For those considering bulk purchasing to reduce cost over the longer treatment course, the bulk retatrutide guide covers volume sourcing strategies and the cost guide breaks down pricing at various quantities.
Storage follows standard peptide protocols. Keep lyophilized (unreconstituted) retatrutide at controlled room temperature or refrigerated. Once reconstituted, store at 2 to 8 degrees Celsius (standard refrigerator temperature) and use within the recommended timeframe. The peptide refrigeration guide covers shelf life expectations in detail, and understanding peptide expiration helps you plan your supply appropriately.
SeekPeptides members access detailed protocols for reconstitution, storage optimization, and vendor evaluation that go beyond what any single guide can cover. The platform also provides tools like the peptide calculator and cost calculator that help you plan the financial and logistical aspects of your transition.
Stacking and combination considerations during the transition
Some researchers do not use GLP-1 agonists in isolation. If you were combining tirzepatide with other compounds, the switch to retatrutide requires reassessing those combinations.
The interaction between retatrutide and other peptides depends entirely on the mechanisms involved. Compounds like tesamorelin may complement the metabolic effects of retatrutide through growth hormone releasing hormone pathways that do not overlap with any of retatrutide three receptor targets. The cagrilintide and retatrutide combination has generated interest because amylin receptor agonism addresses appetite through yet another independent pathway.
However, stacking should be reintroduced gradually. During the first 8 weeks of retatrutide transition, many researchers prefer to run retatrutide as a standalone compound to isolate its effects and understand their individual response. Once stable at a therapeutic dose, additional compounds can be reintroduced one at a time. The peptide stack calculator helps plan multi-compound protocols, and the dosage calculation guide ensures accuracy across multiple compounds. For a broader perspective, the peptides for weight loss overview and the women-specific weight loss peptide guide cover the full range of options that can complement retatrutide therapy.
Understanding retatrutide availability and access
Retatrutide is not yet FDA approved. It remains in Phase 3 clinical trials, which means access is limited to research peptide suppliers. This is fundamentally different from tirzepatide, which is available by prescription as Mounjaro and Zepbound. The current retatrutide availability landscape is evolving, and understanding the distinction between pharmaceutical grade medications and research peptides is critical for anyone considering this transition.
The legal status of research peptides varies by jurisdiction, and researchers should understand their local regulations before purchasing. The online retatrutide purchasing guide covers the most current options available, and the vendor evaluation guide helps you identify suppliers with transparent testing practices.
For researchers new to handling research peptides who may have only used pharmaceutical tirzepatide products previously, the getting started with peptides guide covers the fundamentals of working with lyophilized compounds, including reconstitution, injection technique with proper GLP-1 injection methods, and optimal injection site selection.
Long-term considerations for the tirzepatide to retatrutide switch
Switching compounds is not just about the first few months. The long-term implications deserve consideration before you commit to the transition.
Weight regain risk during the transition
Research consistently shows that approximately two-thirds of patients regain weight when they stop GLP-1 receptor agonist therapy. The washout period represents a brief cessation, and while 25 to 30 days is not long enough for full weight regain, it is enough for 2 to 5 pounds of regain in most people. This is primarily water weight and glycogen restoration as gastric motility normalizes, and it reverses quickly once retatrutide reaches therapeutic levels.
The psychological impact is sometimes worse than the physical reality. Seeing the scale move upward after months of consistent loss can be demoralizing. Knowing this is expected and temporary helps maintain perspective. For weight loss that may have stalled on tirzepatide, the resources about plateaus on GLP-1 therapy provide useful framing that applies regardless of which specific compound is involved.
Can you switch back to tirzepatide later?
Yes. The transition is not a one-way door. If retatrutide does not work for your specific physiology, if the side effects are not manageable, or if access becomes difficult, you can switch back to tirzepatide using the same washout principles in reverse. The same 25 to 30 day clearance period applies when going from retatrutide to tirzepatide, and you would restart tirzepatide based on your previous tolerated dose.
Some researchers have even explored cycling between compounds as a strategy for overcoming persistent plateaus, though this approach lacks formal study data and should be considered experimental. The broader question of duration of GLP-1 therapy applies to this conversation as well.
What happens if tirzepatide gets warm during the transition
If you have leftover tirzepatide from your previous protocol, proper storage remains important even if you do not plan to use it immediately. Understanding temperature excursion effects on tirzepatide helps you decide whether kept supplies remain usable. Similarly, tirzepatide shelf life and compounded tirzepatide expiration information lets you make informed decisions about retaining backup supplies. Some researchers keep their remaining tirzepatide as a safety net during the early weeks of retatrutide transition.
The role of diet and lifestyle during the switch
Medication transitions do not happen in a vacuum. Your diet, exercise, sleep, and stress management all influence how smoothly the switch goes and how quickly you see results on retatrutide.
During the washout period, maintaining the dietary framework you used on tirzepatide is non-negotiable. High protein intake, minimum 1 gram per pound of lean body mass, provides satiety and preserves muscle tissue during the brief gap in pharmaceutical appetite suppression. The tirzepatide diet plan framework translates directly to the washout period.
Once on retatrutide, caloric needs shift. The increased energy expenditure from glucagon receptor activation means your total daily expenditure is higher than it was on tirzepatide at equivalent body weight. This has two implications. First, you may need slightly more calories to avoid excessive deficit. Second, your fat loss rate may exceed what the scale shows if you are also maintaining or building lean tissue. The concept of whether these compounds increase energy levels takes on new meaning with retatrutide, where the thermogenic effect provides a tangible baseline energy increase that many people notice.
Alcohol is worth mentioning specifically. If you were following alcohol guidelines for tirzepatide, the same caution applies to retatrutide with potentially greater intensity. The combination of three-receptor GI effects with alcohol represents a higher nausea and dehydration risk than dual agonist therapy alone.
Frequently asked questions
Can I take my retatrutide dose a day early if I am feeling good?
Consistency matters more than acceleration. Stick to your weekly schedule. The same reasoning behind not taking tirzepatide a day early applies to retatrutide. Both compounds have long half-lives that maintain steady-state levels when dosed on a consistent schedule. Moving doses earlier creates peaks and troughs that increase side effect variability.
How does the weight loss on retatrutide compare to what I saw on tirzepatide?
Meta-analysis data shows retatrutide producing approximately 38% greater weight loss than tirzepatide (16.34 kg versus 11.82 kg). However, individual results vary based on starting weight, dose, diet adherence, and metabolic factors. The tirzepatide before and after results and comparable retatrutide data suggest that the glucagon receptor component produces additional fat loss beyond what dual agonist therapy achieves, particularly for visceral fat.
Will I experience the same appetite suppression on retatrutide as I did on tirzepatide?
The appetite suppression profile is similar because both compounds activate GLP-1 receptors, but many users report that retatrutide appetite suppression feels qualitatively different. It tends to develop more gradually during titration and affects food preoccupation in addition to physical hunger. Most people find that retatrutide at 4 to 8mg produces appetite suppression equivalent to or greater than tirzepatide at 10 to 15mg. The speed at which appetite suppression develops depends on your dose escalation pace.
Is the washout period absolutely necessary?
Yes. There is no safe shortcut. Overlapping two GLP-1 class drugs produces additive GI side effects that can be severe enough to require medical attention. The 25 to 30 day washout is the minimum for safe transition, and extending it slightly for those on higher tirzepatide doses is prudent risk management.
Should I continue taking the same supplements on retatrutide?
Most supplements recommended alongside tirzepatide remain appropriate for retatrutide. Electrolytes, B vitamins, magnesium, and fiber support are still valuable given the GI effects. You may want to add additional magnesium and potassium if you experience increased sweating from the thermogenic effect, as mineral losses through perspiration increase with higher energy expenditure.
What if retatrutide does not work as well for me as tirzepatide did?
Individual response varies. Some people respond better to dual agonism than triple agonism, just as some respond better to semaglutide than tirzepatide. If you reach 8 to 12mg retatrutide and your results are inferior to what tirzepatide produced, switching back is a reasonable option. The transition is reversible, and your tirzepatide sensitivity should return after appropriate clearance time.
Does retatrutide cause hair loss like some people report on tirzepatide?
Hair thinning reported on GLP-1 agonists is generally related to rapid weight loss rather than the specific compound. The mechanism is telogen effluvium, where the stress of significant caloric deficit and weight loss pushes hair follicles into a resting phase. Since retatrutide may produce faster weight loss, the risk of temporary hair thinning could theoretically be equal or slightly higher. The details about retatrutide and hair loss provide specific management strategies.
How does the cost compare between tirzepatide and retatrutide?
The economics differ significantly because tirzepatide is available as a commercial pharmaceutical product while retatrutide is currently a research compound. Research peptide pricing for retatrutide varies by vendor, vial size, and purity. The retatrutide cost guide breaks down pricing across major suppliers, and the peptide cost calculator helps estimate monthly expenses based on your specific dose and concentration.
External resources
For researchers serious about optimizing their transition from tirzepatide to retatrutide, SeekPeptides offers the most comprehensive resource available, with evidence-based protocols, dosing calculators, vendor evaluations, and a community of thousands who have navigated these exact questions. The platform provides detailed retatrutide dosing guides, personalized protocol builders, and the support structure that makes complex transitions like this manageable and optimized.
In case I do not see you, good afternoon, good evening, and good night. May your washout stay clean, your titration stay smooth, and your results stay exceptional.