Feb 8, 2026
You are wasting money. Right now. Every single week. And the worst part is you probably do not even realize it.
Compounded semaglutide is not cheap. A single vial can represent a significant investment in your weight management research, and improper storage degrades that investment one molecule at a time. The compound breaks down silently, losing potency without any visible warning until you notice something alarming: your results have stalled, your appetite suppression has weakened, and you are injecting what amounts to expensive water.
Here is the number that should concern you. Researchers who store compounded semaglutide incorrectly lose up to 40% of their peptide potency before the vial is even halfway used. That means nearly half of what you paid for is sitting in your refrigerator, degrading, because of a temperature fluctuation you never noticed or a handling mistake you make every morning without thinking about it. The question of how long peptides last in the fridge is not a simple one, and the answer depends on factors most people never consider.
This guide breaks down exactly how long compounded semaglutide lasts under every storage condition, what causes it to degrade, how to tell if yours has gone bad, and the precise protocols that maximize every drop of your investment. Whether you are new to semaglutide and wondering how it works or an experienced researcher managing multiple vials, the storage science covered here will save you both money and frustration. SeekPeptides has compiled the most comprehensive storage reference available, drawing from pharmaceutical stability data, compounding pharmacy guidelines, and real-world handling experience to give you everything you need in one place.
What determines how long compounded semaglutide lasts in the fridge
The shelf life of compounded semaglutide is not a single number printed on a label. It is a calculated estimate called the beyond-use date, or BUD, and it varies dramatically depending on where your vial was compounded, how it was tested, and what conditions it has been exposed to since leaving the pharmacy.
Understanding BUD matters.
The beyond-use date represents the last date a compounded preparation can be expected to maintain its labeled potency and sterility under the specified storage conditions. For compounded semaglutide, this date is determined by the compounding pharmacy based on stability testing they have conducted on their specific formulation. Unlike manufactured drugs with extensive stability data spanning years of testing, compounded preparations operate under different regulatory frameworks that produce shorter, more conservative dating, and the range of BUDs you might encounter can vary wildly from one pharmacy to another.
USP 797 is the governing standard here. This chapter of the United States Pharmacopeia establishes requirements for sterile compounded preparations, including how pharmacies must determine BUDs. Under these guidelines, the default BUD for a sterile compounded preparation stored under refrigeration is relatively conservative unless the pharmacy has conducted extended stability testing on their specific formulation. Pharmacies that invest in rigorous stability testing, including potency assays, sterility verification, and endotoxin screening at multiple time points, can justify longer BUDs. Those that rely on default dating without extended testing must use shorter windows.
This creates enormous variation in the market. One pharmacy might assign a 90-day refrigerated BUD to their compounded semaglutide based on comprehensive stability data, while another assigns just 14 days because they have not invested in the same level of testing. Both are operating within regulatory guidelines. Both are providing a legitimate product. But the practical difference for the end user is massive.
The formulation itself plays a role too. Compounded semaglutide is not identical across pharmacies. Different compounding facilities may use different buffers, different pH levels, different preservative systems, and different concentrations. Each of these variables affects stability. A formulation buffered at pH 7.4 with appropriate stabilizing excipients will last longer than one compounded at a lower pH without the same level of formulation science behind it. When you are evaluating whether expired semaglutide is still usable, the formulation quality matters as much as the date on the vial.
The concentration matters too. Higher concentration formulations sometimes demonstrate different stability profiles than more dilute preparations, though this relationship is not always linear or predictable. Some pharmacies compound at concentrations that optimize both stability and dosing convenience, while others prioritize one over the other. Understanding your specific formulation, its concentration, and the pharmacy assigned BUD gives you the foundation for proper storage decisions. If you are working with unit-to-milligram conversions for semaglutide, knowing the concentration of your specific compounded product is essential for both accurate dosing and understanding stability. Similar concentration considerations apply to those using the tirzepatide unit conversion charts or calculating doses from the tirzepatide dosing tables for weight loss.
Compounded semaglutide shelf life by storage condition
Storage conditions create dramatic differences in how long your compounded semaglutide remains viable. The difference between proper refrigeration and leaving a vial on a bathroom counter for a few hours can mean weeks of lost shelf life. Here is what the data shows for each storage scenario, and why understanding these numbers could save you hundreds of dollars in wasted product.
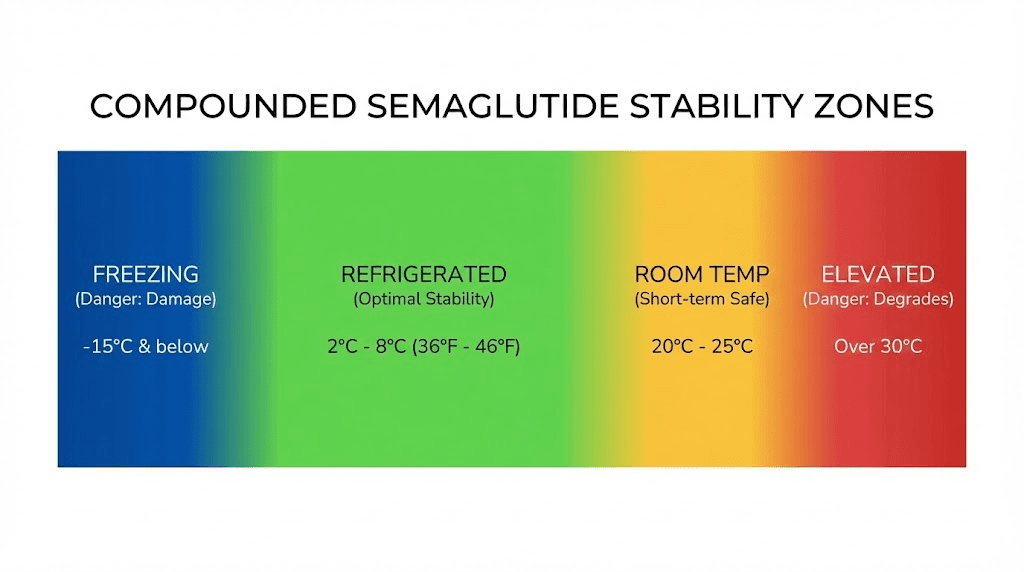
Refrigerated storage (2-8 degrees Celsius)
Refrigeration is the gold standard. Period.
Compounded semaglutide stored at 2-8 degrees Celsius (36-46 degrees Fahrenheit) maintains its potency for the longest possible duration. The typical beyond-use date for refrigerated compounded semaglutide ranges from 30 to 90 days, though this window deserves closer examination because the variation within it is significant and the reasons behind that variation affect your specific situation directly.
Pharmacies with the most rigorous stability testing programs, those conducting multiple potency assays at regular intervals throughout their testing period, can sometimes justify BUDs of up to 120 days under continuous refrigeration. These are typically larger 503B outsourcing facilities with dedicated analytical laboratories and the resources to conduct accelerated stability studies that demonstrate long-term potency retention. If your compounded semaglutide comes from one of these facilities, you have the luxury of a longer usage window, but you must still respect the specific conditions under which that dating was established.
On the shorter end, some pharmacies assign BUDs of just 14 to 28 days. This does not necessarily mean their product is inferior. It often means they have not invested in the extended stability testing required to justify longer dating. A 14-day BUD from a quality pharmacy using appropriate formulation science may actually produce a product that remains stable for months, but the pharmacy cannot legally claim that without the data to support it. For practical purposes, you should always follow the BUD assigned by your specific pharmacy.
Once you puncture the vial with a needle, a new clock starts. Most compounding pharmacies recommend using a multi-dose vial within 28 days of first puncture, even if the original BUD extends beyond that point.
The act of introducing a needle breaks the sterility barrier, and while the preservative system (typically benzyl alcohol) helps maintain sterility after puncture, it cannot do so indefinitely. Every puncture introduces a small risk of contamination, and 28 days represents a conservative but scientifically supported window for multi-dose use. This is similar to the considerations around how long reconstituted peptides last in the fridge after preparation.
Temperature consistency within the refrigerator matters more than most people realize. The 2-8 degree range exists for a reason. Below 2 degrees, you risk freezing, which causes irreversible damage. Above 8 degrees, degradation accelerates. Most household refrigerators fluctuate between 1 and 7 degrees Celsius depending on where in the unit the item is placed, how often the door is opened, and how well the thermostat is calibrated. The door shelves experience the most temperature variation. The back of the middle shelf maintains the most consistent temperature. Where you place your vial is not trivial, and understanding proper peptide storage protocols can make the difference between a vial that lasts its full BUD and one that degrades prematurely.
Room temperature tolerance
Sometimes refrigeration is not available. Travel happens. Power outages occur. You might forget to put your vial back after drawing a dose. Understanding the room temperature tolerance of compounded semaglutide prevents panic in these situations, but it also prevents complacency that could cost you potency.
Compounded semaglutide can tolerate temperatures up to 25 degrees Celsius (77 degrees Fahrenheit) for approximately 15 days without significant potency loss. This is not an invitation to store it at room temperature permanently. It is a tolerance window, a safety margin that accounts for real-world handling. If you accidentally leave your vial on the counter for an afternoon, it is almost certainly fine. If you leave it out for two weeks, the cumulative thermal exposure begins to matter.
Higher temperatures accelerate degradation exponentially. At 35 degrees Celsius (95 degrees Fahrenheit), the tolerance window shrinks to approximately 4 days. At 40 degrees Celsius (104 degrees Fahrenheit), you have roughly 3 days before significant degradation occurs. These are not hard cutoffs where the peptide suddenly becomes useless, but rather points at which potency loss becomes measurable and meaningful. The relationship between temperature and degradation follows Arrhenius kinetics, meaning that every 10-degree increase in temperature roughly doubles or triples the rate of chemical degradation. Understanding how peptides behave at room temperature is essential for making informed decisions during these situations.
The cumulative nature of thermal exposure is critical to understand. If your vial spent 6 hours at room temperature yesterday and 4 hours today, that thermal exposure is additive. You do not get a fresh 15-day window each time you return the vial to the refrigerator. The degradation that occurred during each room temperature excursion is permanent, and while the rate slows dramatically once refrigeration resumes, the damage already done cannot be reversed. Think of it like sunburn. You can get back in the shade, but the damage from each exposure accumulates.
Why freezing destroys compounded semaglutide
Do not freeze it. Ever.
This seems counterintuitive. If cold is good, colder should be better, right? Wrong. Freezing compounded semaglutide causes irreversible molecular damage through several mechanisms that cannot be undone by thawing, and the result is a product that may look perfectly normal while being significantly or entirely degraded.
When aqueous solutions freeze, ice crystals form. These crystals create mechanical forces at the molecular level that can physically break apart large peptide molecules like semaglutide. The peptide chains unfold, aggregate, and form structures that no longer match the active conformation required for biological activity. Even if the solution looks clear after thawing, the molecular damage has already occurred. The peptide molecules that were mechanically disrupted by ice crystal formation do not reassemble into their proper three-dimensional structure. They remain damaged, inactive, and potentially immunogenic.
Freeze-thaw cycles are particularly destructive. If your refrigerator has cold spots near the freezer compartment, or if you accidentally placed a vial in the freezer and then thawed it, the damage from a single freeze-thaw event can reduce potency by 30-50% or more. Multiple freeze-thaw cycles, which can happen inadvertently in a poorly calibrated refrigerator, will progressively destroy the remaining active peptide until the preparation is essentially inert. This is fundamentally different from lyophilized peptide powder, which is specifically designed to be stable in its dry, frozen state. The distinction between lyophilized and liquid peptide formats becomes critically important when storage conditions are less than ideal.
Cryoconcentration is another freezing hazard. As water in the solution freezes, the remaining liquid becomes increasingly concentrated with solutes, including the peptide, buffers, and preservatives.
This hyper-concentrated state can cause the peptide to aggregate, the pH to shift dramatically, and chemical reactions to accelerate in the unfrozen portion. Even partial freezing, where only some of the solution forms ice, can create these damaging conditions in the still-liquid portions.
Compounded vs brand-name semaglutide storage comparison
Understanding how compounded semaglutide storage compares to its brand-name counterparts helps put the shelf life question in proper context. Ozempic and Wegovy have undergone extensive stability testing by their manufacturer, Novo Nordisk, producing well-defined storage parameters. Compounded semaglutide operates under different constraints, but knowing the full picture helps you make better storage decisions regardless of which form you are using.
The comparison matters for practical reasons. If you have experience with brand-name semaglutide and are transitioning to a compounded version, your storage habits may need to change. If you are comparing options between semaglutide and tirzepatide, understanding the storage requirements of each helps with practical planning. And if you are evaluating the alternatives to brand-name Ozempic, storage shelf life is a legitimate factor in that decision.
Factor | Compounded semaglutide | Ozempic (brand) | Wegovy (brand) |
|---|---|---|---|
Refrigerated shelf life (unopened) | 30-90 days (varies by pharmacy) | Until expiration date (typically 2+ years) | Until expiration date (typically 2+ years) |
After first use | 28 days refrigerated (typical) | 56 days (room temp or refrigerated) | 28 days refrigerated |
Room temperature tolerance | Up to 77F for ~15 days | Up to 86F for 56 days | Up to 77F for 28 days |
Maximum temperature | 104F for ~3 days | 86F for 56 days | 77F for 28 days |
Freezing | Irreversible damage, discard | Do not freeze, discard if frozen | Do not freeze, discard if frozen |
Light sensitivity | Protect from light | Store in original carton | Store in original carton |
Stability testing basis | Pharmacy-specific (USP 797) | ICH guidelines (extensive) | ICH guidelines (extensive) |
Several things stand out in this comparison. Brand-name products benefit from years of stability testing conducted under ICH (International Council for Harmonisation) guidelines, which include accelerated aging studies, long-term storage studies, and stress testing that compounding pharmacies typically cannot replicate. This is why Ozempic can claim room temperature stability for 56 days at up to 86 degrees Fahrenheit, while compounded versions have more conservative room temperature tolerances.
The after-first-use window is particularly noteworthy.
Ozempic pens are designed for 56 days of use after first injection, which is significantly longer than the typical 28-day window for compounded vials. This difference reflects both the pen delivery system (which minimizes contamination risk compared to multi-puncture vials) and the extensive preservative efficacy testing conducted by the manufacturer. Compounded preparations in multi-dose vials face greater contamination risk with each needle puncture, which is why the post-opening window is shorter.
None of this means compounded semaglutide is inferior as a product. It means the storage requirements are more demanding and less forgiving. If you follow proper storage protocols, compounded semaglutide maintains its potency throughout its assigned BUD. The challenge is that proper storage requires more attention and discipline than the relatively forgiving storage parameters of brand-name products. Those looking into tirzepatide fridge storage will find similar considerations apply to that compound as well, since tirzepatide refrigeration requirements follow comparable principles. Researchers using the tirzepatide dosing guide for their protocols should apply these same storage standards to every vial in their research program.
The science behind semaglutide degradation
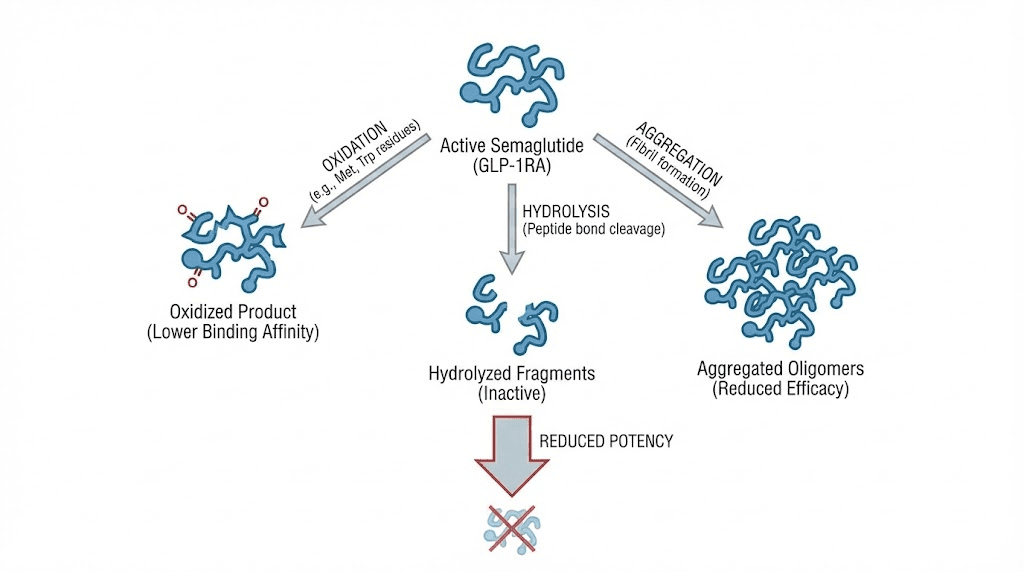
Knowing the storage timelines is important. But understanding why semaglutide degrades gives you the knowledge to make better decisions when situations are not black and white. The degradation of semaglutide follows predictable chemical and physical pathways, and each pathway responds to specific environmental conditions that you can control, or at least mitigate, once you understand the mechanisms.
Semaglutide is a modified GLP-1 receptor agonist with a molecular weight of approximately 4,114 daltons. It consists of a 31-amino acid peptide backbone with several structural modifications designed to extend its biological half-life, including a C-18 fatty acid chain attached via a linker to a lysine residue. These modifications, while brilliant from a pharmacological perspective, create additional points of vulnerability for chemical degradation. The molecule that works so effectively in the body, the one responsible for how quickly semaglutide works and how effectively it suppresses appetite, is precisely the molecule that requires careful storage to remain intact.
Temperature-dependent degradation
Temperature is the single most important factor controlling semaglutide stability. At the molecular level, heat provides energy that drives chemical reactions. Every chemical bond in the semaglutide molecule exists in an energy equilibrium, and as temperature increases, more bonds acquire enough energy to break. This follows Arrhenius kinetics, a fundamental principle of chemistry where reaction rates increase exponentially with temperature.
For peptides like semaglutide, the primary temperature-dependent degradation pathways include deamidation, where asparagine and glutamine residues lose their amide groups, and peptide bond hydrolysis, where the backbone of the molecule literally breaks apart. Both reactions require water (which is abundant in any liquid formulation) and energy (provided by heat). At refrigeration temperatures, these reactions proceed so slowly that the peptide maintains its integrity for weeks to months. At room temperature, the rate increases measurably. At elevated temperatures, it accelerates dramatically.
The practical implication is simple but important. Every minute your vial spends at a temperature above 8 degrees Celsius, degradation is happening faster than it needs to. Not catastrophically in most cases, but cumulatively. The vial you leave on the counter for 30 minutes while you prepare your dose is experiencing degradation at a rate roughly 2-3 times faster than it would in the refrigerator. Over weeks of daily use, these brief exposures add up.
pH and buffer effects
The pH of the formulation profoundly influences semaglutide stability, and this is where differences between compounding pharmacies can create real-world differences in shelf life. Semaglutide has an isoelectric point of approximately 5.4, which means it is least soluble and least stable at or near this pH. The molecule tends to aggregate and precipitate at its isoelectric point, forming particles that represent irreversible potency loss.
Research demonstrates that semaglutide is most stable at pH values above 7.0. At these slightly alkaline conditions, the molecule maintains its proper conformation, remains fully dissolved, and resists the aggregation that destroys potency. Formulations compounded at pH 7.4 (physiological pH) generally demonstrate superior stability compared to those at lower pH values, all else being equal.
Conversely, formulations at pH 4.5-5.5 (near the isoelectric point) degrade significantly faster. At these pH values, the peptide molecules carry minimal net charge, which reduces the electrostatic repulsion that normally keeps individual molecules separated. Without this repulsion, molecules collide, stick together, and form aggregates that can range from soluble oligomers (still somewhat active but less potent) to insoluble particles (completely inactive). The buffer system used, whether phosphate, citrate, histidine, or another buffer, also influences stability through its interaction with the peptide and its ability to maintain pH over time.
This is why asking your compounding pharmacy about their formulation is not being overly cautious, it is being appropriately informed. Understanding the fundamentals of peptide chemistry and how peptides work at the molecular level helps you appreciate why these seemingly technical details have practical consequences for your results.
Light and oxidation
Light is the silent destroyer of peptides. Ultraviolet and visible light provide energy that drives photochemical reactions in semaglutide, particularly oxidation of methionine residues and photo-degradation of tryptophan and tyrosine residues. These reactions produce degradation products that reduce potency and, in some cases, may increase immunogenicity.
Oxidation does not require light, but light accelerates it dramatically. Dissolved oxygen in the formulation reacts with susceptible amino acid residues, particularly methionine, cysteine, histidine, and tryptophan. This reaction produces modified residues that alter the peptide structure and reduce biological activity. Antioxidants in the formulation can slow this process, but they cannot eliminate it entirely, especially if the vial is repeatedly exposed to light during storage or use.
The practical takeaway is straightforward. Keep your vial in its original packaging or wrapped in aluminum foil when not actively in use. Do not store it on a windowsill, under bright kitchen lights, or in any location where it receives regular light exposure. The few seconds of light exposure during dose preparation are negligible. Extended exposure during storage is not. Many of the same principles apply to storing peptides after reconstitution, where light protection is equally important for maintaining potency.
How to tell if your compounded semaglutide has gone bad
Here is the uncomfortable truth. Significant potency loss can occur before any visible signs appear. A vial that looks perfectly clear and normal may have lost 20-30% of its active peptide through deamidation and hydrolysis without any visual indication. This is why following BUD dating and proper storage protocols is more important than relying on visual inspection alone.
That said, visual and physical signs of degradation do occur, and when they appear, they indicate advanced degradation that demands immediate action. Knowing what to look for helps you catch problems that have progressed beyond the invisible chemistry stage into obvious physical changes.
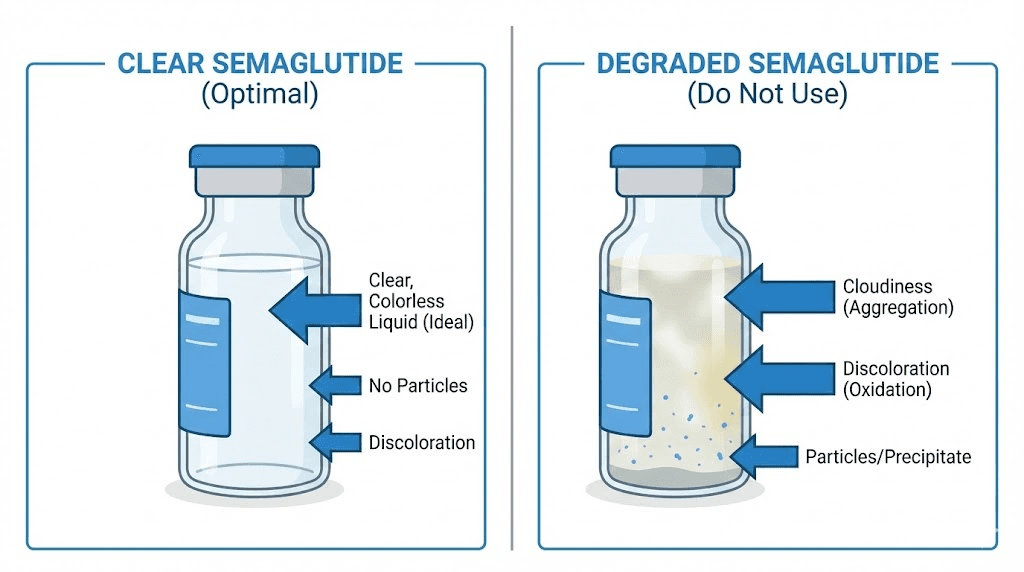
Cloudiness is the most common visible sign. A properly compounded semaglutide solution should be clear and colorless (or very slightly yellow for some formulations). Any haziness, turbidity, or cloudiness indicates protein aggregation, where peptide molecules have clumped together into particles large enough to scatter light. This is irreversible. No amount of gentle swirling, warming, or refrigeration will dissolve these aggregates. If your solution is cloudy, the potency has been significantly compromised.
Discoloration tells a story too. Watch for pink, yellow, brown, or amber tints that were not present when you first received the vial.
These color changes indicate oxidative degradation or other chemical reactions that have produced chromophoric (light-absorbing) degradation products. A slight yellowish tint may be acceptable in some formulations, but any color change from the original appearance warrants caution. Brown or amber discoloration indicates advanced degradation and the vial should be discarded. These same visual principles apply when evaluating whether any peptide has expired, making visual inspection a universal skill for anyone working with peptide compounds.
Particles are unambiguous. Visible particles floating in the solution, settled at the bottom of the vial, or appearing as specks when you hold the vial up to light indicate either contamination, aggregation, or both. Do not use a preparation with visible particles. Period. This represents either a sterility breach or advanced degradation, and neither scenario is acceptable for injection.
Unusual odor is rare but telling. Compounded semaglutide should be essentially odorless, with perhaps a faint smell from the preservative (benzyl alcohol has a mild, slightly sweet odor). Any strong, unusual, or unpleasant odor indicates bacterial contamination and the vial should be discarded immediately, regardless of the BUD date.
Changes in injection behavior sometimes signal degradation too. If the solution feels thicker or thinner than usual during injection, if it burns or stings more than it did initially, or if you notice increased injection site reactions, these could indicate formulation changes from degradation. While these signs are less specific than visual indicators, they are worth noting and investigating.
The bottom line on visual inspection is this: it is necessary but not sufficient. A vial that passes visual inspection may still have lost potency. A vial that fails visual inspection has definitely lost potency and possibly sterility. Use visual inspection as one layer of your quality control system, not as the only layer. Tracking your weight loss progress on semaglutide can also serve as an indirect indicator, since unexplained plateaus might sometimes trace back to potency issues from degraded product.
Proper storage protocol for maximum shelf life
Following a systematic storage protocol is not obsessive. It is protective. Every step in this protocol addresses a specific degradation pathway discussed above, and together they create a system that maximizes the useful life of every vial you purchase. The difference between casual storage and deliberate storage can be 2-4 additional weeks of usable product from every single vial.
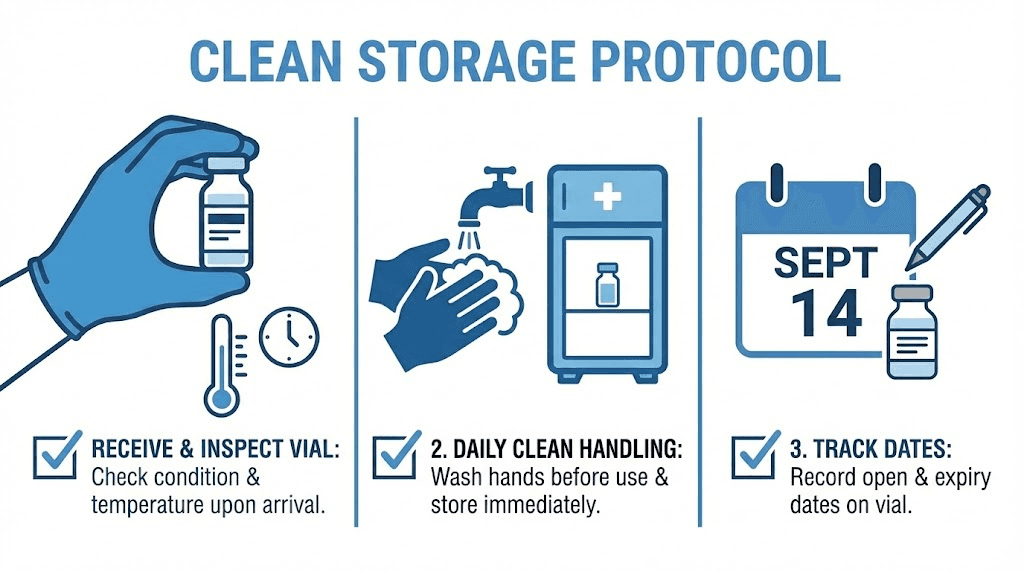
When you first receive your vial
The clock starts at delivery. How you handle your vial in the first hour sets the tone for its entire useful life. If your compounded semaglutide arrives via mail or courier, the temperature it experienced during shipping matters, and what you do immediately after receiving it matters even more.
Inspect the shipping package first. Quality compounding pharmacies ship semaglutide with cold packs in insulated packaging. Check that the cold packs are still cold (or at least cool) when you receive the package. If the cold packs are completely warm and the shipping took more than 48 hours, the vial may have experienced extended room temperature exposure during transit. Note this on your tracking log but do not panic, moderate room temperature exposure during shipping is usually within the tolerance window.
Inspect the vial immediately. Before refrigerating, hold it up to a light source and examine the solution. It should be clear, colorless (or very slightly yellow in some formulations), and free of particles. Check the crimp seal for integrity. Verify that the rubber stopper is intact and properly seated. Note the BUD assigned by the pharmacy and calculate your usage timeline.
Refrigerate immediately upon inspection. Do not leave the vial on your counter while you eat lunch, check your email, or go about your day. Every minute at room temperature is unnecessary thermal exposure. Place the vial in its original packaging (or wrap it in aluminum foil if it did not come in opaque packaging) and put it in the center of your refrigerator, not on the door shelf, not against the back wall where freezing is possible, and not near produce drawers that may be set to different temperatures.
Record the receipt date, the BUD, and the date you first puncture the vial. This creates a paper trail that eliminates guesswork later. SeekPeptides members use a systematic approach to vial management that eliminates guesswork and ensures you never accidentally use a vial beyond its safe window.
Daily handling best practices
Your daily routine around dose preparation has more impact on shelf life than most people realize. Each time you handle the vial, you are introducing variables: temperature changes from your hands, light exposure, potential contamination from the needle, and mechanical agitation from movement. Minimizing these variables through consistent handling practices extends your vial life.
Remove the vial from the refrigerator. Prepare your dose. Return the vial. This entire process should take 2-3 minutes, not 20. Do not set the vial on the counter while you shower. Do not carry it around the house while you decide where to inject. The shorter the time outside refrigeration, the better. If you are working with reconstituted peptide preparations, the same time-conscious handling applies.
Never shake the vial. Semaglutide, like all peptide solutions, is vulnerable to mechanical stress. Shaking creates air-liquid interfaces and shear forces that cause the peptide to unfold and aggregate at the surface of bubbles. If you need to mix the contents (some settling is normal with certain formulations), gently roll the vial between your palms or invert it slowly 2-3 times. The difference between shaking and rolling is the difference between creating foam (destructive) and gently redistributing the contents (safe).
Clean the stopper with an alcohol swab before every puncture. This is basic sterility practice, but it also matters for shelf life because microbial contamination accelerates chemical degradation. Bacteria produce enzymes that break down peptides, and even a small contamination event can dramatically shorten the useful life of a vial. The role of bacteriostatic water and preservatives in maintaining sterility is well documented, but proper aseptic technique is your first line of defense.
Use an appropriately sized needle. Larger gauge needles create larger puncture holes in the rubber stopper, which can compromise the seal over time with repeated punctures. Smaller gauge needles minimize this issue. If you are using a peptide injection pen, the needle gauge is typically standardized, but with manual syringes you have more control over this variable.
Recording and tracking dates
Memory is unreliable. Write it down.
On the vial itself (or on a label attached to it), record three dates: the date you received the vial, the BUD assigned by the pharmacy, and the date you first punctured it. With these three dates, you can always calculate whether you are within your safe usage window. The effective expiration for your vial is whichever comes first: the pharmacy BUD or 28 days after first puncture.
A simple spreadsheet or note in your phone works well for tracking multiple vials or for recording any temperature excursions that may have occurred. If the power went out for 6 hours last Tuesday, note it. If you accidentally left the vial out for 2 hours on Saturday, note it. These records help you make informed decisions about whether to continue using a vial or discard it early. Accurate tracking is just as important when managing peptide dosage calculations, where knowing exactly what you have and when you prepared it ensures accurate, safe dosing every time.
Traveling with compounded semaglutide
Travel introduces every risk factor simultaneously. Temperature changes. Mechanical agitation. Light exposure. Extended time away from your refrigerator. But with proper planning, you can travel with compounded semaglutide without significant potency loss, whether the trip is a weekend road trip or an international flight.
The foundation of travel storage is an insulated bag with cold packs. Medical-grade travel coolers designed for temperature-sensitive medications work best, but even a simple insulated lunch bag with a few small ice packs will maintain refrigeration-like temperatures for 8-12 hours, which covers most travel scenarios. The key is pre-chilling the insulated bag, using cold packs that have been frozen solid (not just cool), and minimizing how often you open the bag during travel.
For air travel, always carry your compounded semaglutide in your carry-on luggage. Never check it. Checked luggage compartments are not climate-controlled and can experience temperatures ranging from below freezing at altitude to above 40 degrees Celsius (104 degrees Fahrenheit) on tarmac during loading and unloading. Either extreme will damage your product. The TSA permits medically necessary liquids and injectable medications in carry-on bags, though you may be asked to declare them at security screening. Having your pharmacy label clearly visible on the vial simplifies this process.
Avoid direct sunlight during travel. A vial sitting in direct sunlight on a car dashboard, even for 20 minutes, can experience temperatures well above 40 degrees Celsius. Use opaque packaging or keep the vial wrapped in a small towel inside your insulated bag. UV exposure through glass is reduced compared to direct outdoor exposure, but the thermal effect of direct sunlight on a small vial can be dramatic and rapid.
Hotel refrigerators are acceptable but not ideal. Most hotel mini-fridges operate at temperatures between 4 and 10 degrees Celsius, which is slightly warmer than optimal but within an acceptable range for short stays. If your hotel fridge has a freezer compartment, keep the vial as far from it as possible to avoid inadvertent freezing. Many hotel refrigerators also have uneven cooling, so placing the vial in the center of the unit rather than against any wall provides the most consistent temperature.
For extended travel, consider your dosing schedule. If your trip is longer than 2-3 weeks, you may need to coordinate with your pharmacy for a fresh vial at your destination, or ensure your travel cooling solution can maintain appropriate temperatures for the full duration. Some researchers traveling for extended periods invest in portable medication coolers that use rechargeable cooling technology to maintain 2-8 degrees Celsius for days at a time. Planning your peptide cycle around your travel schedule can help avoid the need to transport sensitive compounds for extended periods.
Understanding the overall peptide dosing framework helps you determine whether adjusting your injection schedule around travel makes more practical sense than bringing your vial along. Some researchers prefer to time their weekly doses so that travel falls on days between injections, eliminating the transportation challenge entirely. For those exploring the emerging relationship between GLP-1 agonists and hair health or managing fatigue-related side effects of similar compounds, consistent dosing schedules also depend on consistent storage, so getting the logistics right matters for more than just convenience.
What happens if you use degraded semaglutide
The question everyone asks but few sources answer directly. What actually happens if you inject compounded semaglutide that has degraded? The answer depends on the degree and type of degradation, and the consequences range from merely frustrating to potentially concerning.
Reduced efficacy is the most common and most likely outcome. When semaglutide degrades through deamidation, hydrolysis, or oxidation, the resulting degradation products typically lack the biological activity of the intact molecule.
They do not activate the GLP-1 receptor with the same potency, which means your expected dose effectively becomes a smaller dose. If you were injecting what you believed to be 0.5 mg of semaglutide but the vial had degraded 30%, you were actually receiving the equivalent of roughly 0.35 mg. This manifests as reduced appetite suppression, less significant weight effects, and an overall sense that the medication is not working as well as it did initially.
This scenario is frequently misinterpreted. Researchers who experience diminished results often attribute it to tolerance, receptor downregulation, or metabolic adaptation, when the actual cause is a degraded vial that is delivering less active peptide than expected. Understanding semaglutide withdrawal symptoms versus degradation-related efficacy loss helps you distinguish between these very different scenarios and take the appropriate corrective action.
Aggregated semaglutide raises different concerns. Protein aggregates can potentially trigger immune responses, since the immune system may recognize aggregated forms of a peptide as foreign even when it tolerates the properly folded monomer. While clinically significant immunogenic reactions to degraded compounded semaglutide appear to be rare based on available data, the theoretical risk exists and represents a legitimate reason to discard visibly degraded product rather than attempting to use it.
Contaminated semaglutide, where sterility has been compromised, carries the most serious risks. Injection of bacterially contaminated solutions can cause local injection site infections, systemic infections, or sepsis in severe cases.
This is why the 28-day post-puncture window exists and why proper aseptic technique during every dose preparation is non-negotiable. The broader safety considerations for peptide use always include sterility as a foundational requirement, and the legal framework around peptides also governs the standards compounding pharmacies must meet. Storage conditions that compromise sterility should never be ignored.
Using slightly degraded product (one or two days past BUD, stored properly) is different from using severely degraded product (weeks past BUD, temperature abused, visually changed). The former is a calculated risk that many researchers accept with the understanding that BUDs include a safety margin. The latter is a genuinely poor decision with potential health consequences. Knowing where your specific situation falls on this spectrum requires honest assessment of your storage practices and careful attention to the visual and physical signs discussed earlier.
Common storage mistakes that shorten shelf life
Most potency loss from compounded semaglutide is preventable. The mistakes that cause premature degradation are nearly universal, they happen because they seem harmless, because old habits persist, or because nobody explained why these specific actions matter at the molecular level. Here are the most damaging mistakes and exactly how to avoid each one.
Storing on the refrigerator door. The door is the warmest part of your refrigerator and experiences the most temperature fluctuation. Every time the door opens, the items on the door shelves are exposed to room temperature air directly.
Internal shelves are buffered by the thermal mass of other items in the fridge. Temperature loggers placed on refrigerator doors consistently show fluctuations of 5-10 degrees with each opening, while the center of the middle shelf stays within 1-2 degrees. Move your semaglutide off the door.
Leaving the vial out during the entire injection routine. Some people remove the vial, place it on the bathroom counter, shower, shave, get dressed, eat breakfast, then come back to prepare their injection 30-45 minutes later. That entire time, the vial is at room temperature, warming up, and degrading faster than necessary. Prepare your dose first. Return the vial. Then proceed with your routine. This is one of the most common peptide mistakes beginners make, and it is the easiest to fix.
Exposing the vial to direct light. A vial sitting on a well-lit kitchen counter or near a window receives enough light to drive photodegradation reactions. Keep it in its box or wrap it in foil. The two seconds of light exposure during dose preparation is not the issue. The hours of light exposure during improper storage is.
Not cleaning the stopper. Each unclean puncture introduces a small but cumulative contamination risk. Over 10-15 injections from a single vial, these risks compound (no pun intended). Always swab with alcohol. Always let it dry before puncturing. This takes 10 seconds and protects the entire remaining contents of the vial.
Shaking the vial. Vigorous shaking is remarkably destructive to peptide solutions. The air-liquid interface created by bubbles acts as a denaturation surface where peptide molecules unfold and aggregate. A single vigorous shake can create billions of tiny bubbles, each one a site of potential peptide damage. If mixing is needed, roll gently. Never shake. The same principle applies to mixing peptides with bacteriostatic water during initial preparation.
Using the wrong needle gauge repeatedly. Large bore needles (18-20 gauge) used for drawing up doses create proportionally larger holes in the rubber stopper. Over multiple punctures, these holes can fail to seal completely, allowing air exchange and potential contamination. Use the smallest gauge needle that allows comfortable drawing and injection, typically 27-30 gauge for subcutaneous injection of semaglutide. Proper injection technique includes selecting appropriate needle sizes for each application.
Ignoring power outages. A 2-hour power outage in summer may mean your refrigerator internal temperature climbed to 15 degrees Celsius or higher. If you do not know about it (because it happened while you were at work), you cannot account for that thermal exposure. A simple maximum-minimum thermometer in your refrigerator or a smart temperature monitor that sends alerts to your phone eliminates this blind spot.
Storing multiple vials improperly. If you have backup vials, keep unopened ones in the coldest, most stable part of the refrigerator. Only open a new vial when you need it. Every vial that sits punctured but partially used is on a 28-day countdown regardless of the original BUD, so managing your inventory to minimize waste is an important part of cost-effective peptide research.
How to maximize every drop of compounded semaglutide
Compounded semaglutide represents a significant financial investment for most researchers. Wasting product through poor storage, improper handling, or inefficient dosing practices is throwing money away. Here is how to extract maximum value from every vial you purchase, and the strategies go beyond just storage into overall protocol efficiency.
Start by matching your vial size to your usage timeline. If you inject once weekly and your vial contains 8 doses, that vial needs to last 8 weeks. But if the BUD is only 30 days after first puncture, you will discard the last 4 doses. In this scenario, requesting smaller vials (4 doses per vial) or adjusting your protocol to align with the BUD eliminates waste. Understanding the relationship between units and milligrams in your specific formulation helps you calculate exactly how many doses each vial contains and whether the vial size matches your usage timeline.
Dead volume matters. Every syringe and needle combination traps a small amount of solution in the dead space of the hub. For insulin syringes with integrated needles, this dead volume is typically 0.5-2 units. For syringes with separate needles, it can be 5-10 units or more. Over the life of a vial, this dead volume waste can add up to 10-15% of the total vial contents. Using low dead volume syringes or syringes with integrated needles minimizes this loss. The peptide reconstitution calculator can help you plan concentrations that account for dead volume, ensuring your actual delivered dose matches your intended dose.
Dose accuracy prevents both underdosing (wasting time) and overdosing (wasting product). Ensure you know the exact concentration of your compounded semaglutide and that your syringe markings allow you to draw the precise volume needed for your target dose. A semaglutide dosage calculator takes the guesswork out of this process, translating your target dose in milligrams to the exact volume in units or milliliters for your specific formulation concentration. The peptide calculator provides additional functionality for researchers working with multiple compounds.
Consider your titration schedule carefully. Most semaglutide protocols involve gradual dose escalation over several weeks. If you start a new vial at a low dose and escalate over time, the vial will last more doses at the lower doses but fewer at the higher ones. Planning your vial purchases to align with your escalation schedule prevents the scenario where you have half a vial at a concentration you no longer need. Reviewing the peptide dosage chart alongside your titration plan helps you forecast your supply needs accurately.
Do not discard a vial just because it is a day or two past the BUD. We discussed this earlier, BUDs include a safety margin, and a vial stored perfectly at 2-4 degrees Celsius for its entire life may retain excellent potency for days beyond the assigned BUD. This is not permission to ignore dating, it is a recognition that strict BUD adherence sometimes leads to unnecessary waste. Use your judgment: if the vial was stored perfectly, passes visual inspection, and is only slightly past BUD, many researchers make the informed decision to continue using it. If storage was imperfect or the vial shows any visual changes, err on the side of caution.
Coordinate refills so you are not forced to rush through a vial or discard excess. If your pharmacy requires lead time for compounding, factor that into your ordering schedule so a new vial arrives before the current one expires. Understanding the cost structure of peptides and peptide therapy costs overall helps you make financially informed decisions about ordering quantities and frequencies.
Understanding your pharmacy beyond-use date assignment
Not all beyond-use dates are created equal, and understanding how your specific pharmacy arrived at their BUD helps you contextualize what that date actually means for your product. This is not about second-guessing your pharmacy. It is about being an informed consumer of a product where dating variability is enormous and the financial stakes are real.
503A pharmacies compound medications for specific patients based on individual prescriptions. These pharmacies typically assign more conservative BUDs because they may not have the analytical resources to conduct extended stability testing on every formulation they compound. A 503A pharmacy might assign a 14-day or 30-day BUD to their compounded semaglutide, not because the product fails after that period, but because their stability data only supports dating to that point. The product itself may be perfectly stable for longer.
503B outsourcing facilities operate under different regulatory frameworks and compound larger batches without patient-specific prescriptions. These facilities typically have more resources for stability testing, including HPLC potency assays, sterility testing, and endotoxin screening at multiple time points throughout the testing period. As a result, 503B facilities often assign longer BUDs, sometimes 60-90 days or more, because they have the data to support those extended dates.
When evaluating your pharmacy BUD, consider asking these questions. What stability testing was performed on this specific formulation? How frequently were potency assays conducted during the stability study? What storage conditions were maintained during testing? Were the results from the specific batch I am receiving, or from a representative batch of the same formulation? These questions are reasonable, and reputable pharmacies will answer them willingly.
The variation between pharmacies can be surprising. Two pharmacies might compound semaglutide at the same concentration using similar formulation approaches and assign wildly different BUDs, one at 28 days and another at 90 days, simply because one invested in more comprehensive stability testing. Neither pharmacy is wrong. But as a consumer, knowing the basis for the BUD helps you understand whether you are working with a conservatively dated product that likely has a longer practical shelf life, or a product with a BUD that represents the actual stability limit of that specific formulation.
This knowledge becomes particularly relevant when you are comparing the different sources and quality levels available in the compounded peptide market. The difference between research-grade and pharmaceutical-grade peptides extends to the rigor of their stability testing and the reliability of their assigned dating.
How compounding formulation affects storage life
The formulation science behind your specific compounded semaglutide has a direct and significant impact on how long it remains stable in your refrigerator. Two vials of "compounded semaglutide" from different pharmacies may look identical but behave very differently under the same storage conditions because of differences in their underlying formulation.
Buffer selection influences stability profoundly. Phosphate buffers are common in pharmaceutical formulations and maintain pH effectively, but they can interact with certain peptides in ways that promote degradation. Histidine buffers offer advantages for some peptide formulations, including better stabilization of certain amino acid residues. The choice of buffer, its concentration, and its pH all affect how the peptide behaves over time.
Preservative systems vary between pharmacies. Benzyl alcohol is the most common preservative in multi-dose compounded preparations. Its concentration matters, too little and sterility may not be maintained over the full use period, too much and it can interact with the peptide or cause injection site irritation. The optimal concentration balances antimicrobial effectiveness with peptide compatibility, and not all pharmacies optimize this balance identically.
Surfactants and stabilizers may or may not be included in your formulation. Some compounding pharmacies add polysorbate 80 or other surfactants to prevent peptide adsorption to vial surfaces and aggregation at air-liquid interfaces. These additives can significantly extend stability by preventing the surface-mediated degradation that occurs during normal handling. Their presence or absence in your specific formulation is another variable that affects how long the product remains stable.
Concentration effects on stability are sometimes counterintuitive. Higher concentrations can either improve or reduce stability depending on the specific peptide and formulation conditions. At higher concentrations, intermolecular interactions become more frequent, which can promote aggregation in some cases. In other cases, higher concentrations may actually be more stable because the peptide-to-surface ratio favors the peptide remaining in solution rather than adsorbing to vial surfaces. The ratio of solvent to peptide during preparation and in the final formulation both influence long-term stability.
Understanding these formulation variables helps explain why your specific product might last longer or shorter than generic timelines suggest. It also underscores why following your specific pharmacy instructions, rather than generic internet advice, is always the safest approach to storage and handling.
Semaglutide storage in context: comparing GLP-1 receptor agonist stability
Semaglutide is not the only GLP-1 receptor agonist in widespread use, and comparing storage requirements across this drug class provides useful perspective. Each compound has its own stability profile based on its molecular structure, formulation, and the data available from stability testing. Understanding these differences matters if you are transitioning between compounds or considering alternatives.
Tirzepatide, a dual GIP/GLP-1 receptor agonist, has storage requirements that closely parallel compounded semaglutide. The refrigerated shelf life of tirzepatide follows similar patterns, and the degradation mechanisms are comparable though not identical. Both compounds require refrigeration for optimal stability, both are damaged by freezing, and both tolerate brief room temperature excursions. For researchers comparing how tirzepatide performs versus semaglutide, the storage requirements are similar enough that transitioning between them does not require a fundamentally different storage approach.
The tirzepatide dosing schedule and dosage in units affect storage considerations in the same way semaglutide dosing does. How quickly you use a vial determines whether storage duration or post-puncture timing is the limiting factor. A vial used at higher doses will be depleted faster, potentially well within the BUD, while a vial used at lower titration doses may approach the BUD before all doses are drawn.
Retatrutide, a triple receptor agonist targeting GIP, GLP-1, and glucagon receptors, represents a newer addition to this class. While storage data for compounded retatrutide is less extensive, the general principles of peptide stability apply. Researchers exploring retatrutide dosing and how retatrutide compares to semaglutide should apply the same rigorous storage practices described throughout this guide.
Combination products like cagrilintide and semaglutide introduce additional stability considerations. When two peptides are combined in a single formulation, the stability of the least stable component determines the overall BUD. Combination formulations may also have different pH optima for each component, forcing a compromise pH that may not be ideal for either peptide individually. Understanding cagrisema dosing and the storage requirements of combination products requires attention to these additional complexities.
The broader context of peptides used for weight loss reveals that storage discipline is a universal requirement across this entire class of compounds. Whether you are working with semaglutide, fat-burning peptides, peptides targeting visceral fat, or exploring comprehensive fat loss peptide options, the fundamentals of cold chain maintenance, light protection, and aseptic handling apply universally. The AOD-9604 storage requirements and those for tesofensine follow similar principles. Even emerging GLP-1 platforms like GLP-3 and innovative delivery systems such as GLP-1 patches require the same foundational storage discipline, making the habits you develop with semaglutide transferable to virtually any peptide compound. Researchers exploring emerging products like oral tirzepatide or comparing mazdutide versus tirzepatide will find that storage remains the often-overlooked variable that separates successful protocols from disappointing ones.
Special situations and edge cases
Real life is not as neat as storage guidelines suggest. Situations arise that no FAQ on a pharmacy website fully addresses, and knowing how to navigate these edge cases can save both product and money.
What about semaglutide compounded with B12? Some compounding pharmacies offer semaglutide combined with vitamin B12. The addition of cyanocobalamin (B12) does not significantly alter the storage requirements for the semaglutide component, but B12 is light-sensitive and can degrade into inactive forms when exposed to light. This makes light protection even more important for combination formulations. Store these preparations in opaque packaging or foil wrapping and minimize light exposure during handling.
What if your fridge temperature fluctuates? All refrigerators fluctuate to some degree during normal operation (compressor cycles on and off). This normal cycling, typically within 1-2 degrees, is expected and accounted for in stability testing. What matters is the average temperature and the extremes. If your refrigerator regularly cycles between 1 and 7 degrees, that is fine. If it regularly dips below 0 or climbs above 10, you have a problem that needs addressing. A cheap digital thermometer with max/min recording tells you exactly what is happening in your fridge.
What about drawing multiple doses into syringes in advance? Some researchers pre-fill several syringes to save time during the week. This practice is generally discouraged for several reasons. The syringe barrel presents a different surface than the vial (usually polypropylene versus borosilicate glass), and peptide adsorption to syringe surfaces can reduce potency. The dead space and air-liquid interface in a pre-filled syringe are also less favorable for stability than a sealed vial. Pre-filled syringes also lack preservative protection if the dose is small enough that the preservative concentration is significantly diluted. If you must pre-fill, use the syringes within 24-48 hours and keep them refrigerated.
What about peptides shipped from overseas? Researchers sourcing peptides from international suppliers often face extended shipping times that challenge cold chain maintenance. If your compounded semaglutide spent 5+ days in transit without proper cold chain, the thermal exposure may have compromised stability. While the product might still be usable, it should be inspected carefully and potentially given a shorter personal use-by date. Having access to peptide testing laboratories that can verify potency can provide reassurance in these situations.
Can you switch between refrigerated and room temperature storage? Repeated temperature cycling (refrigerator to room temperature and back) is more damaging than consistent storage at either condition. Each cycle creates thermal stress and potentially condensation on the vial surface and stopper. If you must remove a vial from refrigeration for travel, try to keep it at a consistent temperature rather than cycling it in and out of cold storage multiple times.
The role of proper reconstitution in storage life
For researchers who receive compounded semaglutide in lyophilized (freeze-dried) form rather than as a pre-mixed solution, the reconstitution process directly affects how long the resulting preparation will last. Every decision made during reconstitution, from the diluent used to the technique employed, influences the stability of the final product.
The choice of reconstitution water matters more than most people realize. Bacteriostatic water (containing 0.9% benzyl alcohol as a preservative) provides antimicrobial protection that sterile water does not. This preservative extends the usable life of the reconstituted product by preventing microbial growth in the multi-dose vial. Sterile water without preservative creates a preparation that must be used much faster, typically within 24-48 hours when stored properly, because it lacks ongoing antimicrobial protection. For semaglutide preparations intended for multi-dose use, bacteriostatic water is the appropriate choice.
Reconstitution technique affects stability from the first moment of preparation. The standard reconstitution protocol calls for directing the diluent gently down the side of the vial, allowing it to contact the lyophilized cake gradually rather than blasting it with a direct stream. This gentle approach minimizes foaming, which creates the air-liquid interfaces that promote peptide denaturation. After adding the diluent, gentle swirling (never shaking) dissolves the peptide while maintaining its structural integrity.
The volume of diluent used determines the final concentration, which in turn affects stability and dosing convenience. Using the peptide reconstitution calculator ensures you achieve the exact concentration you need for your dosing protocol. A more concentrated solution means fewer doses per vial but potentially different stability characteristics, while a more dilute solution provides more doses but may have a shorter effective shelf life due to lower peptide-to-degradation ratios.
After reconstitution, the same storage rules apply that govern pre-mixed compounded preparations. Refrigerate immediately. Protect from light. Do not freeze. Use within 28 days of reconstitution (or within the timeframe specified for your specific product). Record the reconstitution date and the calculated expiration date on the vial.
Monitoring potency over time
For researchers who want to go beyond visual inspection and date tracking, there are options for monitoring the actual potency of their compounded semaglutide over time. This is not necessary for most users, but for those managing high-value research programs or working with products from unfamiliar sources, potency verification provides an additional layer of quality assurance.
High-performance liquid chromatography (HPLC) is the gold standard for peptide potency analysis. Several independent testing laboratories offer peptide purity and potency analysis for individual vials. The cost is typically modest relative to the value of the peptide being tested, and the results provide definitive evidence of whether your storage practices are maintaining potency at acceptable levels.
Indirect potency monitoring through outcome tracking is more practical for most researchers. If you are tracking your weight, appetite suppression, and semaglutide response timeline, an unexpected decline in these outcomes, particularly one that coincides with using an older vial or a vial that experienced a storage excursion, may indicate potency loss. This is not definitive evidence, as many factors affect treatment outcomes, but it can serve as a useful signal when interpreted alongside other information about your storage practices.
Establishing a baseline for comparison requires consistency. Use the same brand, concentration, and source for several vials while maintaining perfect storage. This establishes what "normal" response looks like. Any deviation from this baseline in subsequent vials, particularly those with different storage histories, becomes meaningful. SeekPeptides provides tracking tools and community insights that help researchers establish these baselines, aligning with the broader principle of treating peptide research with scientific rigor from the beginning.
Long-term storage planning for multi-month protocols
Semaglutide protocols typically span months. Some extend beyond a year. Planning your storage and supply chain over these extended timeframes prevents the common problem of running short, wasting excess, or compromising quality through poor inventory management.
Calculate your monthly consumption first. If you are injecting once weekly at a stable dose, your monthly consumption is straightforward. If you are still titrating, calculate the expected consumption at each dose level and plan accordingly. The semaglutide dosage calculator simplifies these calculations by translating your dose in milligrams to volume per injection, allowing you to determine how many injections a single vial provides.
Stagger your orders rather than stockpiling. It might seem efficient to order three months of supply at once, but compounded semaglutide has limited shelf life. Three vials sitting in your refrigerator for two months before you open them are two months closer to their BUD, potentially leaving you with compressed usage windows when you finally open them. Order one to two vials ahead at most, timed so each new vial arrives within a week or two of when you need it.
Build relationships with your pharmacy. Consistent ordering from the same pharmacy ensures formulation consistency, streamlines the ordering process, and often allows better communication about BUDs, formulation changes, and availability issues. Some pharmacies offer auto-ship programs that solve the timing problem automatically. Evaluating pharmacy quality using resources like the peptide solutions guide and purity and quality standards helps you identify pharmacies that invest in the rigorous stability testing that produces longer, more reliable BUDs.
Have a contingency plan. Supply disruptions happen in the compounding pharmacy world, whether from ingredient shortages, regulatory changes, or quality control holds. Having a backup pharmacy identified (and ideally having filled at least one prescription with them) ensures you are not scrambling to find a new source when your primary pharmacy cannot fill an order. Knowing your options across the peptide therapy landscape gives you flexibility when unexpected supply issues arise. Researchers can also evaluate where to source emerging compounds like retatrutide and monitor retatrutide availability as backup options within their weight management research programs.
For researchers considering whether to incorporate additional peptides into their protocol, understanding the storage requirements of each compound allows you to plan a unified storage strategy. Combining multiple peptides in a research program means managing multiple storage timelines, and the discipline you develop with semaglutide storage translates directly to managing compounds like BPC-157, TB-500, and others. The peptide stacking guide provides additional context on managing multiple compounds simultaneously, while the peptide stack calculator helps you plan combinations that account for storage logistics. Both men exploring peptide protocols and women researching peptide options benefit from establishing these storage systems early in their research journey.
Delivery format considerations for storage
Not all compounded semaglutide arrives in the same format, and the delivery format affects both storage requirements and practical handling. Understanding the differences between vials, pre-filled syringes, and alternative delivery methods helps you optimize your storage approach for your specific product.
Multi-dose vials are the most common format for compounded semaglutide. Glass vials with rubber stoppers and aluminum crimp seals provide good chemical inertness and barrier properties. Glass does not interact with the peptide, and the rubber stopper, when properly formulated, minimizes extractables and leachables that could affect stability. The primary storage concern with vials is maintaining sterility after first puncture and minimizing the number of punctures (and therefore potential contamination events) over the vial life.
Some compounding pharmacies now offer semaglutide in pre-filled syringes or cartridges. These formats reduce contamination risk (single use or limited puncture) and eliminate the dose-drawing step that creates some of the handling challenges discussed earlier. However, they present different storage considerations. The syringe materials (typically polypropylene or cyclic olefin polymer) may interact differently with the peptide than glass, and the air-liquid interface in a pre-filled syringe is less favorable than in a properly filled vial. Pre-filled formats should be stored tip-down to keep the air bubble away from the needle end, reducing surface-mediated degradation at the air-liquid interface.
Alternative delivery methods are emerging in the compounded semaglutide market. Sublingual formulations, oral capsules, and nasal sprays each have unique stability profiles that differ significantly from injectable solutions. These formats may use different stabilization strategies, different preservative systems, and different storage conditions. Always follow the specific storage instructions provided with these alternative formats, as the guidelines for injectable solutions do not necessarily apply.
The choice between injectable and oral peptide delivery carries storage implications beyond just the stability of the compound itself. Injectable preparations generally require stricter temperature control, while some oral and sublingual formats may be more temperature-tolerant due to the stabilizing excipients used in their formulation. The fundamentals of peptide injection include proper storage as a foundational requirement for safe and effective use.
Cost implications of proper vs improper storage
Numbers tell the story better than generalities. Let us calculate the actual financial impact of storage practices over the course of a typical semaglutide protocol.
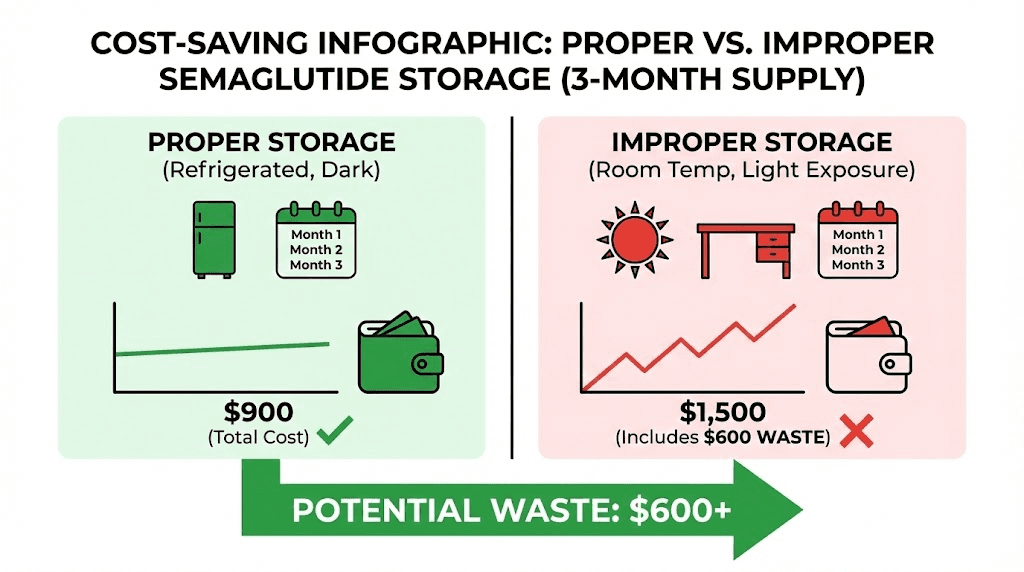
Consider a researcher using compounded semaglutide at a weekly dose of 1 mg, with vials containing 5 mg at a cost of roughly $150 per vial. Each vial provides 5 weeks of supply. Over a 6-month protocol (26 weeks), this researcher will use approximately 5.2 vials, spending roughly $780 on semaglutide alone.
Now consider the cost of improper storage. If poor handling causes 30% potency loss, that $150 vial is only delivering $105 worth of active peptide. Over 6 months, the researcher loses $234 to degradation, enough to buy more than 1.5 additional vials. But the cost does not stop at the peptide itself. Reduced potency means reduced effectiveness, which can mean a longer protocol to achieve the same results, additional medical consultations, and the psychological cost of plateau frustration.
From a different angle, proper storage essentially gives you free product. If your storage practices extend usable vial life from 20 days (poor handling) to the full 28 days (proper handling), you gain an extra week of doses from every vial. Over a year of use, that is an extra 3-4 weeks of supply, roughly the value of one additional vial, at no additional cost. Whether you are following documented weight loss journeys or building your own protocol using microdosing approaches, the financial math of proper storage is universally compelling.
The investment required for proper storage is minimal. A dedicated section of your refrigerator (free), a digital thermometer with min/max recording ($15-20), aluminum foil for light protection ($3), alcohol swabs for sterility ($5 for a box of 100), and a small notebook or phone app for tracking dates (free). Total investment: approximately $25 for equipment that protects hundreds of dollars in peptide product over its useful life.
This cost analysis extends to related compounds. Researchers also using tirzepatide for their research or exploring retatrutide pricing face similar financial incentives to maintain rigorous storage practices. The same logic applies to those researching cagrilintide for weight loss or reviewing cagrilintide dosing protocols, where each vial represents a financial commitment worth protecting. The peptide cost calculator helps researchers understand the true cost of their protocols when storage waste is factored into the equation. For women specifically, protocols involving peptides for weight loss or peptides for menopause weight management carry the same cost-protection incentives.
Frequently asked questions
Can I use compounded semaglutide that was left out overnight?
In most cases, yes. A single overnight room temperature excursion (approximately 8-10 hours at 20-25 degrees Celsius) falls well within the tolerance window for compounded semaglutide. The product can tolerate up to 15 days at temperatures up to 25 degrees Celsius (77 degrees Fahrenheit). However, note this exposure in your tracking log and subtract it from your remaining room temperature tolerance budget. Return the vial to the refrigerator as soon as possible, and if you have concerns about cumulative thermal exposure, inspect the solution carefully for any visual changes before your next use.
How can I tell if my refrigerator is maintaining the correct temperature?
Use a digital thermometer with minimum/maximum recording capability. Place it near where you store your semaglutide and check the readings weekly. The target range is 2-8 degrees Celsius (36-46 degrees Fahrenheit). If the maximum recorded temperature regularly exceeds 8 degrees, adjust your refrigerator thermostat. If the minimum drops below 2 degrees, move the vial away from the back wall and freezer compartment. Smart thermometers that send alerts to your phone when temperatures go out of range provide an extra layer of protection, especially for catching safety-critical temperature excursions during power outages or appliance malfunctions.
Is compounded semaglutide still good after the beyond-use date?
Beyond-use dates include a safety margin, and a vial stored under ideal conditions may retain potency for some time beyond the assigned BUD. However, using a product past its BUD means accepting a degree of uncertainty about potency and sterility that the pharmacy dating was designed to eliminate. If your vial is a day or two past BUD, was stored perfectly, and passes visual inspection, many researchers make the informed choice to continue using it. If it is significantly past BUD, was subject to temperature excursions, or shows any visual changes, discard it. For more detailed guidance on expiration considerations, review the complete guide to using expired semaglutide.
Does the concentration of compounded semaglutide affect how long it lasts?
Concentration can influence stability, though the relationship is complex and formulation-specific. Higher concentrations may offer some stability advantages in certain formulations because the peptide-to-surface ratio is more favorable, but they can also increase the risk of aggregation if the formulation is not optimized for that concentration. In practical terms, the BUD assigned by your pharmacy already accounts for the specific concentration of your product, so the assigned dating is your most reliable guide regardless of concentration. Understanding concentration-to-dose conversions helps you use your product efficiently within its assigned shelf life.
Should I let compounded semaglutide warm up before injecting?
Injecting cold semaglutide directly from the refrigerator is safe and does not reduce efficacy. Some researchers prefer to let the vial sit at room temperature for 5-10 minutes before injecting because cold injections can be slightly more uncomfortable. If you choose to warm the vial briefly, 5-10 minutes is sufficient and the thermal exposure is negligible. Do not heat the vial using warm water, a microwave, or any external heat source, as these can create localized hot spots that damage the peptide even if the overall solution temperature seems reasonable. Review the complete peptide injection guide for additional injection comfort tips.
Can I store compounded semaglutide in a wine fridge or beverage cooler?
Wine fridges and beverage coolers are not ideal for peptide storage. Most wine refrigerators operate at 10-18 degrees Celsius (50-64 degrees Fahrenheit), which is significantly warmer than the 2-8 degree range required for optimal semaglutide storage. Beverage coolers vary more widely, with some maintaining temperatures close to the appropriate range. If you must use an alternative cooling device, verify its temperature range with a thermometer before trusting it with temperature-sensitive medications. A standard household refrigerator set to 3-5 degrees Celsius is the most reliable option for most researchers.
How does semaglutide storage compare to other peptides I might be using?
Most reconstituted peptides require similar storage conditions: refrigeration at 2-8 degrees Celsius, protection from light, and use within a defined window after reconstitution. The specific timeframes vary by peptide and formulation. BPC-157 and TB-500 typically have similar post-reconstitution shelf lives when stored properly. Growth hormone releasing peptides like ipamorelin, sermorelin, and CJC-1295 follow comparable storage guidelines. The general peptide storage guide provides a comprehensive reference for managing multiple compounds with different stability profiles. For researchers exploring longevity peptides alongside semaglutide, a unified storage system keeps everything organized and properly maintained.
What is the absolute maximum time compounded semaglutide can last in the fridge?
The maximum documented refrigerated shelf life for compounded semaglutide from pharmacies with the most rigorous stability testing programs is approximately 120 days (about 4 months) when stored continuously at 2-8 degrees Celsius in the original sealed vial. This is the upper end. Most pharmacies assign BUDs between 30 and 90 days, and once the vial is punctured, the practical limit is typically 28 days regardless of the original BUD. These timelines assume continuous, perfect refrigeration with no temperature excursions, no light exposure, and proper handling at every dose withdrawal. In real-world conditions, the effective shelf life may be shorter. Understanding the peptide formulation fundamentals helps explain why some pharmacies achieve longer stability windows than others.
External resources
USP General Chapter 797: Pharmaceutical Compounding, Sterile Preparations
CDC Vaccine Storage and Handling Toolkit (Cold Chain Principles)
For researchers serious about proper peptide handling, SeekPeptides provides comprehensive storage guides, stability databases, and handling protocols for members. Whether you are managing a single semaglutide vial or a complex multi-peptide research program, the resources available through SeekPeptides help ensure every compound in your protocol maintains maximum potency from the first dose to the last.
The storage question is really about respect. Respect for the science behind the compound. Respect for the money you invested. Respect for the results you are working toward. Every degree of temperature control, every second of reduced light exposure, every careful handling decision compounds (again, no pun intended) over the life of each vial into measurable differences in potency, efficacy, and value. The researchers who get the best outcomes from peptide-based weight loss protocols are not necessarily using better products. They are taking better care of the same products. And now, so can you.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your refrigerator stay calibrated, and your results stay consistent. Join here.