Mar 5, 2026

The vial arrives in a small box. White powder sits at the bottom. A label claims 99% purity. A certificate of analysis comes folded inside, numbers and charts that look official enough. You paid somewhere between $40 and $200, depending on the vendor, the milligram count, and whatever sale was running that week.
But here is the question nobody answers honestly. What is actually in that vial?
Independent laboratory testing tells a story that most vendors would rather you did not hear. When researchers at ACS Peptide Testing Labs analyzed peptide samples submitted for verification, 73% showed measurable purity discrepancies compared to supplier certificates of analysis. For GLP-1 receptor agonists like semaglutide specifically, that number climbs to 81%. A separate study published in the Journal of Medical Internet Research tested semaglutide products purchased from online sellers and found measured purity ranging between 7.7% and 14.37%, numbers that are catastrophically far from the 99% printed on those labels.
This does not mean every vendor sells garbage. It means the market is unregulated, inconsistent, and stacked against buyers who do not know what to look for. And most buyers do not know what to look for. This guide changes that. Whether you are evaluating research peptides versus pharmaceutical compounds, comparing compounded semaglutide options, or trying to figure out if a vendor certificate means anything at all, the information here will help you make decisions based on evidence rather than marketing copy. SeekPeptides has compiled everything researchers need to navigate this landscape without getting burned.
Understanding semaglutide as a peptide compound
Semaglutide is a 31-amino-acid peptide with a C-18 fatty acid chain attached at position 26 via a linker. That fatty acid chain is what makes semaglutide different from natural GLP-1. It allows the molecule to bind to albumin in the blood, extending its half-life from roughly two minutes to approximately one week. This is elegant biochemistry. It is also what makes semaglutide exceptionally difficult to synthesize correctly.
Most peptides researchers work with are relatively straightforward chains of amino acids. Semaglutide is not. The fatty acid modification, the specific linker chemistry, and the length of the chain all create opportunities for synthesis errors. Truncated sequences, where the peptide chain terminates prematurely, are the most common impurity. These fragments often share similar chromatographic properties to the target peptide, which means they can hide in basic purity testing. A vendor running a quick HPLC analysis might report 98% purity while missing truncated variants that a more thorough peptide testing protocol would catch.
Understanding this complexity matters because it directly affects what you get when you buy semaglutide peptide from any source. The difficulty of synthesis explains why price varies so dramatically between vendors. It explains why purity claims are unreliable without independent verification. And it explains why pharmaceutical-grade semaglutide, manufactured under strict GMP conditions with extensive quality controls, costs what it does.
How semaglutide works at the molecular level
Semaglutide activates GLP-1 receptors throughout the body. In the pancreas, this stimulates insulin secretion in a glucose-dependent manner. In the brain, it acts on appetite centers in the hypothalamus, reducing hunger and increasing satiety. In the stomach, it slows gastric emptying, which means food stays in the stomach longer and you feel full after smaller meals.
The weight loss effects are significant. Clinical trials showed average weight loss of 14.9% of body weight over 68 weeks at the 2.4 mg weekly dose. But these results came from pharmaceutical-grade semaglutide with verified potency. When the active ingredient in your vial is actually 8% pure instead of 99% pure, the math changes dramatically. You might be injecting one-twelfth of the dose you calculated, which explains why some researchers report zero appetite suppression while others on the same nominal dose see dramatic results.
For researchers exploring how semaglutide affects the body, understanding the mechanism helps explain why consistent potency matters so much. Every downstream effect, from appetite suppression timing to how quickly results appear, depends on actually receiving the dose you intended.
Semaglutide salt forms and why they matter
Here is something most buyers never consider. Not all semaglutide is the same molecule.
FDA-approved semaglutide products use the base form of the peptide. But many compounding pharmacies and research peptide vendors use salt forms, specifically semaglutide sodium or semaglutide acetate. These are different active ingredients with different molecular weights. The FDA has explicitly stated that semaglutide sodium and semaglutide acetate are not the same as the semaglutide used in approved products like Ozempic and Wegovy.
Why does this matter? Because dosing calculations based on pharmaceutical semaglutide do not directly translate to salt forms. The molecular weight difference means milligram-for-milligram, you are getting a different amount of active peptide. Researchers using semaglutide dosage calculators need to know which form they have, or their calculations will be off from the start. If a vendor does not clearly state which salt form they sell, that alone is a red flag worth paying attention to.
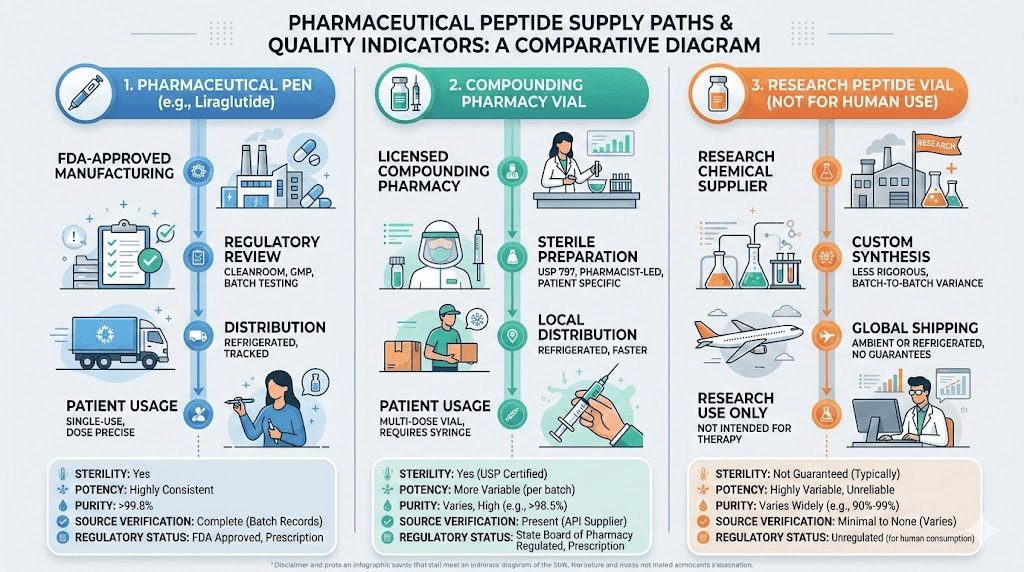
The three categories of semaglutide sources
When people search for where to buy semaglutide peptide, they encounter three fundamentally different categories. Each comes with its own risk profile, cost structure, and quality expectations. Confusing them is where most problems begin.
FDA-approved pharmaceutical products
Ozempic (injectable semaglutide for type 2 diabetes) and Wegovy (injectable semaglutide for weight management) are manufactured by Novo Nordisk under strict Good Manufacturing Practice conditions. Every batch undergoes extensive testing. The supply chain is regulated from raw material sourcing through final packaging. These products come in pre-filled pens with calibrated doses, eliminating the need for reconstitution or manual dose calculation.
The tradeoff is cost and access. Without insurance, branded semaglutide can exceed $1,000 per month. Obtaining it requires a prescription, which means a clinical evaluation, appropriate diagnosis, and ongoing medical oversight. For many people, this is the safest and most reliable option. It is also the most expensive and the hardest to access.
Compounded semaglutide from licensed pharmacies
Compounding pharmacies create customized medications based on individual prescriptions. They still require a prescription, but they can offer semaglutide at significantly lower prices than brand-name products. The quality control varies enormously depending on the type of pharmacy.
Section 503B outsourcing facilities operate under more stringent FDA oversight. They are inspected by the FDA, must follow current Good Manufacturing Practice requirements, and are required to report adverse events. These pharmacies represent the closest thing to pharmaceutical-grade quality outside of the brand-name manufacturers.
Section 503A pharmacies operate under state board of pharmacy regulations rather than direct FDA oversight. They are not required to report adverse events to the FDA, and their products are not tested by federal regulators. Quality varies from excellent to concerning, depending on the specific pharmacy, their sourcing practices, and their internal quality controls.
Many researchers explore specific compounding pharmacies when evaluating their options. Guides covering Empower Pharmacy semaglutide, Olympia semaglutide, Belmar Pharmacy semaglutide, and Direct Meds semaglutide provide detailed breakdowns of what each pharmacy offers, including their sourcing practices and testing protocols.
Research peptide vendors
This is where the landscape gets complicated. Research peptide vendors sell semaglutide labeled "for research purposes only" or "not for human consumption." These products exist in a regulatory gray area. They do not require prescriptions. They are not inspected by the FDA. Their quality control depends entirely on the vendor, and there is no external enforcement mechanism guaranteeing what is in the vial matches what is on the label.
Some research vendors maintain high standards. They use reputable synthesis facilities, conduct third-party testing through independent laboratories, and provide detailed certificates of analysis with batch-specific data. Others sell products with fabricated or recycled certificates, inadequate testing, or active ingredient levels that bear no resemblance to their labels.
The problem is telling them apart without specialized knowledge. That is exactly what the rest of this guide addresses.
How to evaluate a research peptide vendor before buying
Evaluating vendors is not about finding the cheapest price or the flashiest website. It is about identifying specific, verifiable indicators of quality and avoiding the red flags that signal potential problems. Here is a systematic approach that experienced researchers use.
Certificate of analysis verification
Every legitimate vendor provides a Certificate of Analysis for their products. But not all COAs are created equal.
A legitimate COA should include the peptide name and sequence, the batch or lot number that matches your specific product, purity percentage determined by HPLC, mass spectrometry confirmation of molecular identity, the name and credentials of the testing laboratory, the date of testing, storage conditions, and the laboratory contact information for verification.
Here is the critical step most buyers skip. Call or email the laboratory listed on the COA and verify that they actually tested that batch. Fabricated COAs are not uncommon in the research peptide market. A legitimate lab will confirm their testing records. A fabricated COA will lead to a dead end, a nonexistent lab, or a lab that has no record of the batch number in question.
Look specifically for third-party COAs issued by independent laboratories rather than in-house testing. When the vendor tests their own product, there is an inherent conflict of interest. When an independent lab like ACS Peptide Testing Labs or Janssen Labs conducts the analysis, the results carry significantly more weight. Understanding peptide testing laboratories and what their results actually mean is one of the most valuable skills a researcher can develop.
HPLC purity analysis explained
High-Performance Liquid Chromatography separates the components of a peptide sample and measures the proportion of each. The result is a chromatogram, a graph with peaks. The main peak represents the target peptide. Additional peaks indicate impurities, truncated sequences, or degradation products.
Purity is calculated as the percentage of the main peak area versus the total area of all peaks. A 98% HPLC purity means the target peptide accounts for 98% of the detected material. Sounds straightforward. It is not.
The problem with semaglutide specifically is that truncated sequences from incomplete synthesis can co-elute with the target peptide, meaning they appear as part of the same peak rather than as separate impurities. Standard HPLC methods might report 98% purity while missing significant contamination. This is why mass spectrometry confirmation is essential, it verifies the molecular weight matches the full semaglutide sequence rather than a truncated variant.
When reviewing a COA, look for both HPLC purity data AND mass spectrometry confirmation. If a vendor only provides HPLC results without mass spec, the purity number is less reliable, especially for a complex peptide like semaglutide.
Red flags that signal problems
Some warning signs are obvious. Others are subtle. Here are the patterns experienced researchers watch for.
Unrealistically low prices should make you pause. Synthesizing genuine, high-purity semaglutide is expensive due to its structural complexity. If a vendor sells it for 70% less than competitors, ask yourself what they cut to achieve that price point. The answer is usually purity, quality control, or both.
Generic or recycled COAs are a major red flag. If the COA does not include a batch-specific lot number, or if the same COA document appears across multiple product listings with different dates, the testing may be fabricated. Some vendors use a single COA for dozens of batches, which defeats the entire purpose of batch-specific quality testing.
Missing contact information or no verifiable laboratory affiliation suggests the vendor cannot or will not stand behind their testing claims. Legitimate operations provide clear channels for questions about their products, their sourcing, and their quality control processes.
No visible customer reviews or community presence can also be concerning. While new vendors exist, established ones have track records on peptide forums and research communities. Before purchasing from any vendor, search for independent reviews and user experiences beyond the vendor website itself.
Vendors who make therapeutic claims or provide dosing guidance for human use are operating outside the boundaries of legitimate research peptide sales. This does not necessarily mean their product quality is poor, but it does indicate a cavalier attitude toward regulatory compliance that may extend to their quality control practices.
What experienced researchers look for in a vendor
Consistent batch-to-batch quality, verified through independent testing over multiple orders, is the gold standard. Researchers who work with reputable peptide vendors often test early orders independently to establish a baseline of trust before committing to larger purchases.
Transparent communication matters enormously. A vendor who clearly states their synthesis source, provides batch-specific documentation, and responds promptly and knowledgeably to technical questions demonstrates the kind of operational integrity that correlates with product quality.
Proper packaging and shipping, including cold chain management where appropriate, temperature indicators, and protective packaging for lyophilized products, indicates attention to the details that preserve peptide integrity from manufacture to delivery.
Independent testing and why it matters more than you think
Let us talk numbers. The data from independent testing laboratories paints a picture that every semaglutide buyer needs to see.
ACS Peptide Testing Labs reports that 73% of all peptide samples submitted for independent verification show purity discrepancies compared to supplier COAs. For GLP-1 agonists like semaglutide, the discrepancy rate rises to 81%. Only 27% of supplier COAs exactly match independent testing results.
A study published in the Journal of Medical Internet Research went further. Researchers purchased semaglutide products from online sellers and submitted them for comprehensive analysis. The measured purity ranged between 7.7% and 14.37%. Every single sample contained detectable endotoxin levels, ranging from 2.1645 EU/mg to 8.9511 EU/mg. These are bacterial contamination markers that indicate inadequate manufacturing conditions.
Think about what that means practically. If you purchase a 5mg vial of semaglutide that is actually 10% pure, you have 0.5mg of active peptide. Your dosage calculations assume 5mg. Every dose you prepare will be roughly one-tenth of what you intended. This explains the wildly inconsistent results researchers report when using different vendors or even different batches from the same vendor.
How to get your peptides independently tested
Several laboratories offer peptide testing services for individual researchers. The process typically involves sending a small sample of your product to the lab, specifying the tests you want (HPLC purity, mass spectrometry, endotoxin screening), and receiving results within one to three weeks.
Costs for basic HPLC purity and mass spectrometry testing range from $100 to $300 per sample. That sounds expensive relative to a $60 peptide purchase, but consider the alternative. If you are basing weeks or months of research on a product that turns out to be 10% pure, the wasted time and unreliable data cost far more than a verification test.
Prioritize testing for your first order from any new vendor. If results match the supplied COA within acceptable margins (plus or minus 3% for purity is reasonable), you have established a baseline of trust. If results diverge significantly, you have saved yourself from building research on compromised material.
What endotoxin contamination means
Endotoxins are components of bacterial cell walls that trigger inflammatory responses. Their presence in a peptide product indicates contamination during manufacturing, likely from inadequate sterile technique, contaminated water sources, or poor environmental controls in the production facility.
The endotoxin levels found in the JMIR study (2.1 to 8.9 EU/mg) are significant. For context, the FDA limit for injectable products is 5 EU/kg/hour. While research peptides are not regulated to pharmaceutical standards, the presence of endotoxins indicates manufacturing conditions that likely compromise other aspects of product quality as well. If a facility cannot maintain basic sterile conditions, their synthesis accuracy and purity controls are likely equally inadequate.
Semaglutide peptide forms and what you should expect
Research-grade semaglutide typically arrives as a lyophilized (freeze-dried) white powder in a sealed vial. Understanding what to expect helps you identify potential problems before they affect your research.
Lyophilized powder characteristics
Properly lyophilized semaglutide should appear as a white to off-white powder or cake at the bottom of the vial. The powder should be dry with no visible moisture, clumping, or discoloration. A yellowish or brownish tint may indicate degradation, oxidation, or contamination.
The powder should dissolve completely when reconstituted properly. Particles, cloudiness, or visible matter after reconstitution suggest impurities, degradation, or contamination. These visual checks are not substitutes for analytical testing, but they provide a first-line quality assessment that costs nothing and takes seconds.
Storage matters enormously for maintaining potency. Lyophilized semaglutide is most stable at minus 20 degrees Celsius or colder. At refrigerator temperatures (2 to 8 degrees Celsius), it maintains stability for up to 36 months. At room temperature, degradation accelerates significantly. If your peptide arrived without cold packing in summer heat, potency may already be compromised before you open the vial. Researchers concerned about semaglutide shelf life should prioritize proper storage from the moment of delivery.
Reconstitution essentials
Reconstituting semaglutide requires specific technique to preserve the peptide structure. The bacteriostatic water should be added slowly, directed down the inside wall of the vial rather than sprayed directly onto the powder. Direct force can damage the peptide structure, specifically the fatty acid modification that gives semaglutide its extended half-life.
Never shake the vial. Gentle swirling allows the powder to dissolve without the mechanical stress that degrades peptide bonds. A fully reconstituted solution should be clear and colorless. Any cloudiness, particles, or color change indicates a problem.
The amount of bacteriostatic water you add determines the concentration of your solution, which directly affects dosing accuracy. Common reconstitution volumes for a 5mg vial include 1ml (creating a 5mg/ml solution) and 2ml (creating a 2.5mg/ml solution). Use our peptide reconstitution calculator to determine exact volumes based on your target concentration. For specific guidance on 5mg vials, see the complete 5mg semaglutide reconstitution guide. Researchers working with larger vials should consult the 10mg semaglutide reconstitution guide for appropriate water volumes.
Post-reconstitution storage
Once reconstituted, semaglutide has a much shorter usable life than its lyophilized form. With bacteriostatic water, the reconstituted solution remains stable for approximately 28 days when refrigerated at 2 to 8 degrees Celsius. Some researchers report usable stability for 30 to 45 days, but potency begins declining after the 28-day mark.
Never freeze reconstituted semaglutide. Ice crystal formation irreversibly damages the peptide structure. This is one of the most common mistakes researchers make, and it renders the product useless. Detailed guidance on how long semaglutide lasts in the fridge and compounded semaglutide fridge storage helps researchers maximize their product life without sacrificing quality.
Protect the solution from light. UV exposure accelerates peptide degradation. Store in the original amber vial if provided, or wrap clear vials in aluminum foil. Understanding what happens when semaglutide gets warm helps researchers handle temperature excursions appropriately.
Cost analysis across semaglutide sources
Price is often the primary driver of purchasing decisions. Understanding the true cost, including hidden costs of quality problems, helps researchers make informed choices.
Research peptide vendor pricing
Research-grade semaglutide typically costs between $30 and $200 for a 5mg vial, with prices varying based on claimed purity, vendor reputation, and purchase volume. A 10mg vial generally runs $50 to $350. Bulk pricing can reduce per-milligram costs by 30 to 50%.
But the sticker price is misleading if purity is significantly below claims. Consider a $60 vial labeled as 5mg at 99% purity. If independent testing reveals actual purity of 15%, you have approximately 0.75mg of active semaglutide. Your effective cost per milligram of actual active ingredient is $80/mg. Meanwhile, a $150 vial from a reputable vendor with verified 97% purity contains approximately 4.85mg of active compound, costing roughly $31/mg. The "expensive" option is actually 61% cheaper per unit of active ingredient.
This math applies to every aspect of semaglutide research. Peptide cost calculators can help you compare per-dose costs across different products and concentrations, but they only work when the purity inputs are accurate.
Compounded semaglutide pricing
Compounded semaglutide from licensed pharmacies typically costs between $150 and $500 per month, depending on the pharmacy, the dose, and whether additional compounds like B12 or glycine are included. This represents a significant savings over brand-name products but comes at a premium compared to research peptides.
The price difference buys you several things. A prescription means medical oversight and appropriate dose adjustments. Licensed pharmacies face regulatory accountability, even if the degree varies between 503A and 503B facilities. And the product is intended for human use, meaning quality standards, while imperfect, are higher than "for research purposes only" labeling requires.
Researchers exploring compounded options often compare specific pharmacy offerings. Semaglutide with B12 compounds are popular for their potential to address B12 depletion during GLP-1 use. Semaglutide with glycine formulations and semaglutide glycine B12 blends offer additional compound options. Some researchers also explore semaglutide with methylcobalamin combinations or semaglutide with niacinamide for enhanced protocols.
Hidden costs of low-quality products
Beyond the direct financial waste of paying for inactive material, low-quality semaglutide creates several hidden costs.
Wasted research time is perhaps the most significant. If your peptide is significantly underpotent, you may spend weeks or months evaluating protocols that appear ineffective, not because the protocol is wrong but because the product is inadequate. The data generated from compromised material is unreliable and potentially misleading.
Inconsistent dosing creates another problem. If purity varies between batches, your results will be inconsistent even with identical protocols. This introduces variables that confound interpretation and make it impossible to establish reliable baselines.
Contamination concerns add risk that is difficult to quantify. Endotoxins, residual solvents, heavy metals, and unidentified impurities found in tested samples represent contamination that no legitimate research protocol should tolerate. Researchers tracking their response to semaglutide who experience unexpected dizziness, fatigue, or bloating may be responding to contaminants rather than the peptide itself.
Legal considerations for buying semaglutide peptide
The legal landscape surrounding semaglutide purchasing is complex and evolving. Researchers need to understand the boundaries to make informed decisions.
Research peptide legality
Purchasing peptides labeled "for research purposes only" is generally legal in the United States. The key distinction is intent. Buying a research peptide for legitimate laboratory use is lawful. Buying a product labeled "for research" with the intent to use it as a therapeutic agent occupies legally ambiguous territory.
The FDA has taken enforcement action against vendors who market research peptides in ways that suggest human consumption. Warning letters, seizures, and injunctions have targeted companies that blur the line between research supply and therapeutic distribution. For buyers, the legal risk is minimal in most cases, but the regulatory environment is tightening as GLP-1 peptides gain mainstream attention.
Understanding peptide legality in your specific jurisdiction is essential before making any purchase. Regulations vary by state and country, and what is permissible in one location may be restricted in another.
Compounded semaglutide and the FDA
The FDA has been increasingly active in regulating compounded semaglutide. In particular, the agency has raised concerns about pharmacies using semaglutide salt forms (sodium, acetate) that differ from the approved active ingredient, pharmacies sourcing raw materials labeled "for research use only" that are not intended for human drug products, quality control failures that have resulted in hospitalizations, and dosing errors related to concentration variability between compounding pharmacies.
For researchers, this regulatory activity has practical implications. Some compounding pharmacies that previously offered semaglutide may have ceased production or changed their formulations in response to FDA pressure. The availability and legality of compounded semaglutide changes frequently, making it essential to verify current status before ordering.
Import considerations
Semaglutide purchased from international vendors, particularly those based in China, may face customs scrutiny. The FDA has authority to detain imported products that appear to be unapproved drugs. While many international shipments arrive without issue, some are detained or destroyed at the border.
Researchers who source internationally should be aware that customs detention rates increase during periods of heightened regulatory attention, that products without proper documentation are more likely to be detained, and that personal use exemptions that apply to some pharmaceutical products may not extend to research peptides. For those interested in sourcing considerations, the guide on Chinese peptide sourcing covers the specifics of international supply chains.
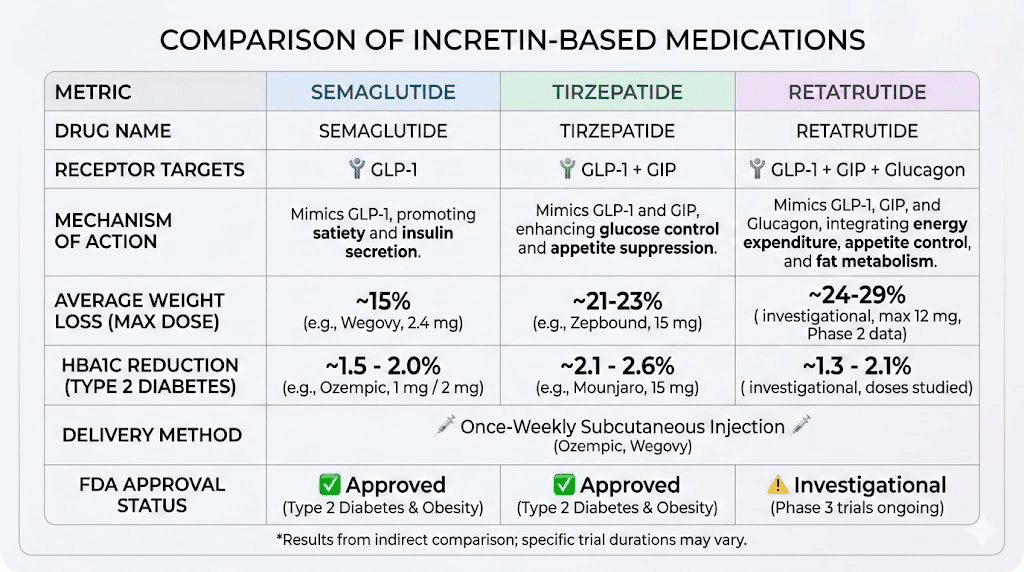
Comparing semaglutide to alternative GLP-1 peptides
Semaglutide is not the only GLP-1 receptor agonist available. Understanding the alternatives helps researchers make informed decisions about which compound best suits their needs and budget.
Semaglutide versus tirzepatide
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has shown even greater weight loss efficacy than semaglutide in head-to-head trials. The SURMOUNT trials demonstrated average weight loss of up to 22.5% of body weight at the highest dose, compared to approximately 15% for semaglutide.
The semaglutide versus tirzepatide comparison extends beyond efficacy numbers. Side effect profiles differ, with tirzepatide showing somewhat lower rates of nausea in some studies. Dosing schedules are similar (weekly injection for both), but the dose escalation protocols differ significantly. Researchers comparing these compounds should review the detailed side effect comparison and the dosage comparison chart to understand the practical differences.
From a purchasing perspective, tirzepatide faces similar quality and sourcing challenges as semaglutide. Research vendors sell both compounds, and the same quality verification principles apply. Peptide Sciences tirzepatide reviews and other vendor evaluations can help researchers compare sourcing options.
The triple agonist option
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors. Phase 2 trial data showed weight loss of up to 24.2% at 48 weeks, making it potentially the most effective weight loss peptide in development. Researchers interested in this compound can explore the retatrutide purchasing guide for sourcing information, or compare it directly against other options through the semaglutide versus tirzepatide versus retatrutide comparison.
The challenge with retatrutide is that it has not yet received FDA approval, making pharmaceutical-grade product unavailable. This means researchers are limited to compounded or research-grade sources, where quality verification becomes even more critical. For detailed dosing information, the retatrutide dose guide and retatrutide dosage calculator provide research-based protocols.
Oral versus injectable options
Some vendors now offer oral or sublingual semaglutide formulations. The oral semaglutide drops guide and sublingual semaglutide guide cover these delivery methods in detail. Oral bioavailability for peptides is typically very low (often below 1% without specialized absorption enhancers), so oral formulations require different evaluation criteria than injectable products.
The injectable versus oral peptides comparison explains the fundamental pharmacokinetic differences that affect efficacy. Researchers considering oral options should understand that dose requirements, onset timing, and bioavailability are fundamentally different from injectable formulations.
Dosing considerations when using research-grade semaglutide
If you have decided to purchase research-grade semaglutide, accurate dosing becomes both more important and more difficult than with pharmaceutical products. The variables multiply.
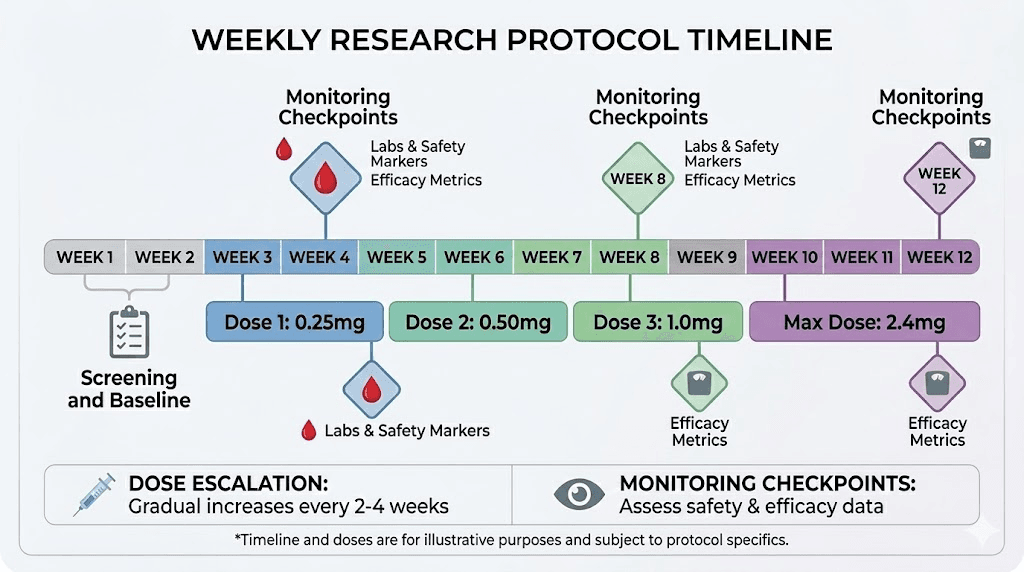
Starting dose protocols
Standard semaglutide dose escalation in clinical trials follows a specific pattern. Weeks 1 through 4 use 0.25mg weekly. Weeks 5 through 8 increase to 0.5mg. Weeks 9 through 12 move to 1.0mg. Weeks 13 through 16 reach 1.7mg. And the maintenance dose of 2.4mg begins at week 17.
This gradual escalation exists for good reason. Rapid dose increases dramatically worsen gastrointestinal side effects, particularly nausea, vomiting, and diarrhea. Even with verified pharmaceutical-grade semaglutide, skipping dose escalation steps increases adverse event rates significantly.
With research-grade semaglutide of unknown or uncertain purity, dose escalation becomes even more complicated. If purity is lower than claimed, your actual dose is lower than calculated. If you then switch batches or vendors and the new product has higher purity, your effective dose may jump dramatically. This is how serious adverse events happen with research peptides, not from the peptide itself, but from uncontrolled dose variation between batches.
Researchers working with semaglutide should thoroughly understand the semaglutide dosage in units conversion system, the 5mg/ml dosage chart, and how to use the semaglutide dosage calculator for precise calculations.
Unit conversion fundamentals
One of the most common sources of dosing errors is confusion between milligrams and units. Research-grade semaglutide is measured in milligrams, while insulin syringes used for injection are marked in units. The conversion depends entirely on the concentration of your reconstituted solution.
For example, if you reconstitute 5mg of semaglutide in 2ml of bacteriostatic water, you create a 2.5mg/ml solution. In a standard insulin syringe where 100 units equals 1ml, 10 units of this solution equals 0.1ml, which contains 0.25mg of semaglutide.
The math is straightforward, but mistakes are remarkably common. Resources covering how many mg is 40 units of semaglutide, 50 units conversion, 20 units to mg, and the complete units-to-mg conversion guide help researchers avoid calculation errors that can lead to significant over or under-dosing. The injection technique guide covers the physical administration process.
Timing and administration
Semaglutide is administered once weekly. The best time of day to take semaglutide is a common question among researchers. Clinical trials did not specify a particular time of day, but many researchers prefer morning administration to manage any nausea during waking hours rather than overnight.
Consistency matters more than the specific day or time. Choose a day and approximate time that works for your schedule and stick with it. Variation of a day or two is acceptable, but regular timing helps maintain stable blood levels. For researchers exploring splitting the dose to twice weekly, the pharmacokinetic considerations are different and require careful evaluation.
Injection site selection also affects absorption. The best injection sites for semaglutide include the abdomen, thigh, and upper arm. Subcutaneous injection depth matters. Too shallow hits the dermis, causing pain and poor absorption. Too deep hits muscle, potentially altering the absorption rate. The detailed injection guide covers proper technique for researchers unfamiliar with subcutaneous administration.
Managing side effects with research-grade semaglutide
Side effects from semaglutide are well documented in clinical literature. But research-grade products introduce additional variables that can modify the side effect profile in unpredictable ways.
Expected gastrointestinal effects
Nausea is the most common side effect, affecting approximately 44% of subjects in clinical trials at the 2.4mg dose. It typically peaks during dose escalation and improves over time as the body adapts. Other common GI effects include diarrhea, vomiting, constipation, and abdominal pain.
With research-grade semaglutide, the severity and pattern of GI effects can vary between batches if potency is inconsistent. A batch with lower-than-claimed purity might produce minimal nausea, leading the researcher to escalate faster than appropriate. When the next batch arrives with higher purity, the effective dose jumps, and severe nausea follows.
Detailed guidance on managing specific side effects is available for constipation treatment, burping and sulfur burps, bloating management, and foods to avoid during semaglutide use.
Non-GI effects to monitor
Beyond the digestive system, semaglutide can affect energy levels, mood, sleep, and metabolic markers. Many researchers report significant fatigue during the initial weeks, which typically improves as the body adjusts. Some experience sleep disruption or dizziness. Others notice effects on their menstrual cycle or libido.
Monitoring these effects helps researchers distinguish between expected pharmacological responses and potential contamination-related reactions. If side effects are unusual, inconsistent with the known semaglutide profile, or appear after switching batches, contamination or purity issues should be considered as possible explanations.
Researchers wondering whether semaglutide increases energy or shows up in blood work can find detailed analysis in our specialized guides. Understanding when side effects typically begin helps set appropriate expectations for the research timeline.
When to be concerned
Certain symptoms warrant immediate attention. Severe abdominal pain that does not resolve could indicate pancreatitis, a rare but serious potential side effect. Significant changes in vision or severe headache deserve medical evaluation. Allergic reactions, including hives, facial swelling, or difficulty breathing, require emergency attention regardless of whether the source is pharmaceutical or research-grade.
With research-grade products, additional concerns include injection site reactions beyond mild redness (potentially indicating contamination), symptoms inconsistent with semaglutide pharmacology (potentially indicating unknown contaminants), and dramatic changes in effect between batches (indicating potency inconsistency).
Building an effective semaglutide research protocol
For researchers committed to working with research-grade semaglutide, a systematic approach minimizes risk and maximizes data quality.
Protocol framework
Week 0: Preparation
Select and order from a vendor who meets the quality criteria outlined above. Request batch-specific COA with third-party verification. Prepare a testing sample for independent verification if this is your first order from this vendor. Gather all necessary supplies, including insulin syringes, bacteriostatic water, alcohol swabs, and appropriate storage containers.
Week 1 through 4: Low-dose initiation
Begin with the lowest practical dose. Document everything, including injection site, time, dose calculated versus drawn, any immediate effects, and any delayed effects over the following 48 to 72 hours. This establishes a baseline response profile for this specific batch.
Week 5 through 8: Gradual escalation
Increase dose according to standard escalation protocol, but only if the previous dose level was well tolerated. Never increase dose if experiencing significant adverse effects. If you receive a new batch during this period, consider restarting at the previous dose level until you confirm the new batch produces similar effects at similar doses.
Ongoing: Monitoring and documentation
Track weight changes, appetite levels, energy, sleep quality, and any adverse effects on a consistent schedule. This data is only meaningful if the peptide quality is consistent, which is why vendor selection and verification are so critical. Researchers can track their progress against benchmarks like week-by-week semaglutide results and one-month result patterns.
Dietary considerations during semaglutide research
Nutrition plays a significant role in semaglutide outcomes. Appetite suppression is one of the primary effects, which means caloric intake often decreases substantially. Without attention to nutritional quality, this caloric deficit can lead to muscle loss, nutrient deficiencies, and energy crashes.
Protein intake becomes especially important. Aim for at least 1.0 to 1.2 grams of protein per kilogram of body weight to preserve lean mass. The best foods to eat on semaglutide guide and the semaglutide diet plan provide structured approaches to maintaining adequate nutrition. The comprehensive food list makes meal planning straightforward.
Some researchers explore complementary compounds to enhance their protocols. Semaglutide with L-carnitine is popular for supporting fat metabolism. Berberine combined with semaglutide may offer complementary metabolic effects. And combining phentermine with semaglutide is a question many researchers investigate, though the phentermine versus semaglutide comparison suggests they work through different mechanisms.
Exercise and semaglutide
The question of whether exercise is necessary comes up frequently. While weight loss without exercise is possible on semaglutide, resistance training helps preserve muscle mass during the caloric deficit that semaglutide creates. The body will break down both fat and muscle for energy. Without resistance training stimulus, a larger proportion of weight loss comes from lean tissue, which negatively affects metabolic rate and long-term outcomes.
Shipping and handling considerations that affect product quality
The peptide in your vial might have been perfectly synthesized. It could have passed every quality test with flying colors. But if shipping and handling were careless, potency takes a hit before you ever open the package. This is one of the most overlooked factors in peptide purchasing.
Temperature during transit
Lyophilized semaglutide tolerates temperature excursions better than reconstituted solutions, but it is not invincible. Extended exposure to temperatures above 30 degrees Celsius accelerates degradation of the peptide backbone and the critical fatty acid modification. During summer months, packages can reach internal temperatures of 50 degrees Celsius or higher inside delivery trucks and on doorsteps.
Reputable vendors mitigate this with insulated packaging, gel ice packs, and expedited shipping options. Some include temperature monitoring strips that change color when the package exceeds a safe temperature threshold. If your vendor ships in a standard padded envelope without cold chain protection, the risk to product quality is real and measurable.
Researchers who have dealt with semaglutide left out overnight know that even brief temperature excursions can affect potency. The time limits for unrefrigerated semaglutide provide guidance on how long exposure remains within acceptable bounds.
Packaging integrity
The vial should arrive with its seal intact. No cracks. No signs of moisture intrusion. The cap should be secure and show no evidence of tampering. If the rubber stopper shows needle marks that you did not make, the product has been accessed before it reached you, which raises obvious concerns about contamination and accurate fill volume.
Lyophilized powder should sit as a solid cake or loose powder at the bottom of the vial. If it appears dissolved, partially dissolved, or wet, moisture has entered the vial during storage or shipping. This compromises both potency and sterility. A wet or dissolved lyophilized product should not be used, regardless of what the COA says about the batch.
Documentation and labeling
Proper labeling should include the compound name, the quantity (in milligrams), the batch or lot number, the synthesis date or expiration date, storage instructions, and the vendor identification. Products that arrive without clear labeling make it impossible to track batch quality, verify against COA documentation, or identify the product if questions arise later.
Some vendors include reconstitution instructions, which can be helpful for researchers new to peptide work. However, these instructions are no substitute for understanding the principles behind mixing peptides with bacteriostatic water and the correct water volumes for your target concentration.
Understanding semaglutide dosing charts and calculations
Accurate dosing is the foundation of reliable research. With pharmaceutical semaglutide in pre-filled pens, the manufacturer handles the math. With research-grade peptides, the math is your responsibility. And it is less straightforward than it appears.
Concentration-dependent dosing
When you reconstitute semaglutide, the volume of bacteriostatic water you add determines the concentration. That concentration determines how much liquid you draw for each dose. Get the concentration wrong and every subsequent dose is wrong too.
Here is a practical example. You have a 5mg vial. You add 2ml of bacteriostatic water. Concentration is 2.5mg per ml. To administer 0.25mg, you need 0.1ml, which equals 10 units on a standard U-100 insulin syringe. Simple enough. But what if the vial actually contains 3mg instead of 5mg because purity is 60%? Now your concentration is 1.5mg per ml, and 10 units gives you 0.15mg instead of 0.25mg. You think you are at a starting dose. You are actually at a sub-therapeutic level.
This is why verified purity matters for dosing accuracy, not just product quality in the abstract. The semaglutide dosage chart in units and the dosage chart in ml provide reference frameworks that assume accurate labeling. Researchers working with verified products can use these charts confidently. Those working with unverified products should treat them as approximations.
Syringe selection and reading accuracy
Insulin syringes come in multiple sizes. The 0.3ml (30 unit), 0.5ml (50 unit), and 1.0ml (100 unit) varieties are most common. Smaller syringes offer better precision for small doses. If your starting dose requires 10 units, a 30-unit syringe provides finer graduation marks and easier accuracy than a 100-unit syringe where 10 units occupies a tiny fraction of the barrel.
Reading the meniscus correctly matters at these volumes. The bottom of the curved liquid surface should align with your target marking. Parallax error from reading at an angle introduces dose variation of 5 to 10%, which compounds over multiple injections. The syringe injection technique guide covers these practical details that significantly affect dosing consistency.
Reconstitution math for common vial sizes
The 5mg reconstitution guide walks through the most common scenario. For those working with 10mg vials, the 10mg mixing guide covers the specific calculations. The semaglutide mixing chart and reconstitution chart provide quick reference for multiple concentration options.
Each concentration option has tradeoffs. Higher concentrations (5mg/ml) mean smaller injection volumes, which can be more comfortable but harder to measure precisely. Lower concentrations (1mg/ml) mean larger volumes that are easier to measure but require more injection time and potentially more discomfort. Most researchers settle on 2 to 2.5mg/ml as a practical middle ground that balances measurement precision with injection volume.
Research-grade semaglutide versus compounded formulations in practice
The distinction between research-grade and compounded semaglutide is not just regulatory. It has practical implications that affect every aspect of the research experience.
Formulation differences
Research-grade semaglutide typically arrives as a pure (or at least intended to be pure) lyophilized peptide. No excipients. No preservatives beyond what is in your bacteriostatic water. No additional compounds.
Compounded semaglutide, by contrast, often includes additional ingredients. B12 combinations are among the most popular, designed to address the vitamin B12 depletion that GLP-1 agonists can cause. Glycine formulations are another common option. Some pharmacies offer pyridoxine (vitamin B6) combinations as well.
These additions can be beneficial. But they also introduce complexity. Multi-compound formulations have more potential points of failure in manufacturing, more stability considerations in storage, and more variables in how the final product behaves. Research-grade pure semaglutide eliminates these variables, giving you a cleaner experimental condition, assuming the purity is verified.
Delivery method options
Compounding pharmacies offer semaglutide in various delivery forms beyond injectable solutions. Oral drops, sublingual formulations, and even troches are available from some compounders. The injectable versus oral peptide comparison details the pharmacokinetic differences that matter for research design.
Bioavailability varies dramatically between delivery methods. Injectable semaglutide has near-100% bioavailability. Oral formulations typically achieve 1 to 3% bioavailability without specialized absorption enhancers, meaning a much larger total dose is needed to achieve equivalent blood levels. Sublingual absorption falls somewhere between these extremes. Researchers must account for these differences when comparing results across delivery methods or transitioning between them.
Cost per effective milligram comparison
When comparing costs honestly, researchers need to calculate the cost per milligram of bioavailable active ingredient, not the sticker price per vial or per month.
Research-grade injectable semaglutide at verified 95% purity, priced at $120 for 10mg, delivers approximately 9.5mg of active compound at near-100% bioavailability. Effective cost: roughly $12.63 per bioavailable milligram.
Compounded injectable semaglutide at $300 per month for a four-week supply at 2.5mg weekly (10mg total), assuming pharmaceutical-grade purity from a 503B facility, delivers approximately 10mg at near-100% bioavailability. Effective cost: $30 per bioavailable milligram.
Oral compounded semaglutide at $200 per month might provide a nominal 100mg dose designed to deliver roughly 2 to 3mg of bioavailable peptide after accounting for the low oral absorption rate. Effective cost: $67 to $100 per bioavailable milligram.
Research-grade semaglutide from a low-quality vendor at $40 for 5mg but with actual purity of 12%, delivers only 0.6mg of active compound. Effective cost: $66.67 per bioavailable milligram, making the cheapest option per vial one of the most expensive per unit of actual active ingredient.
These calculations reinforce why purity verification is not academic. It is the single biggest factor in determining actual cost effectiveness. The peptide cost calculator helps researchers run these comparisons with their specific numbers.
Traveling with semaglutide peptides
Researchers who travel need to maintain cold chain integrity for reconstituted semaglutide and proper storage for lyophilized vials. This presents practical challenges, especially for air travel.
Lyophilized semaglutide is relatively travel-friendly. It tolerates room temperature for extended periods (up to 14 days at up to 30 degrees Celsius). Pack it in carry-on luggage rather than checked bags, where temperature extremes in cargo holds can exceed safe ranges.
Reconstituted semaglutide requires more careful handling. A small insulated cooler bag with a gel ice pack maintains appropriate temperature for 12 to 24 hours, sufficient for most domestic travel. For longer trips, plan access to refrigeration at your destination. The complete travel guide covers TSA regulations, international travel considerations, and practical packing strategies.
Carrying syringes and vials through airport security raises questions occasionally. Having a letter from a healthcare provider or research institution explaining the nature of the materials can streamline the process, though it is not legally required for domestic U.S. travel. Research peptides without clear pharmaceutical labeling may attract more scrutiny than products in original pharmacy packaging.
Specific peptide interactions to understand before buying
Semaglutide does not exist in isolation. Many researchers work with multiple compounds simultaneously, and understanding potential interactions is essential before adding semaglutide to an existing protocol.
GLP-1 combinations
Combining semaglutide with other GLP-1 receptor agonists is generally not recommended due to additive receptor activation that increases adverse event risk without proportional benefit. The question of whether semaglutide and tirzepatide can be taken together comes up frequently, and the short answer is that the overlap in mechanism makes this combination risky and unlikely to provide additive benefit.
However, researchers sometimes transition from one GLP-1 compound to another. The tirzepatide to semaglutide switch guide and the conversion chart help navigate these transitions safely, accounting for the different pharmacokinetic profiles of each compound.
Non-GLP-1 peptide stacking
Semaglutide can potentially be combined with peptides that work through entirely different mechanisms. Researchers interested in combining weight loss protocols with healing peptides, for example, might explore running semaglutide alongside BPC-157 or TB-500. Others combine GLP-1 protocols with growth hormone secretagogues like ipamorelin or CJC-1295.
The peptide stacking calculator helps researchers evaluate combination protocols. The peptide stacking guide covers the principles of combining multiple compounds, including timing, injection site rotation, and monitoring for interaction effects.
Pharmaceutical interactions
Semaglutide slows gastric emptying, which can affect the absorption of other oral medications. This is particularly relevant for medications with narrow therapeutic windows, including some blood pressure medications, thyroid hormones, and oral contraceptives. Researchers who take prescription medications should understand how semaglutide might alter their absorption profiles.
The question of combining phentermine with semaglutide is one of the most common pharmaceutical interaction queries. While they work through different mechanisms, combining appetite suppressants requires careful consideration of cardiovascular effects and should involve medical oversight.
Common mistakes when buying semaglutide peptide
Learning from other researchers' mistakes is cheaper than making your own. These are the errors that show up most frequently in research communities.
Prioritizing price over verification
The cheapest vial is almost never the best value. As demonstrated in the cost analysis section, a $40 vial at 10% actual purity costs more per milligram of active compound than a $150 vial at 97% purity. Yet price remains the primary selection criterion for most first-time buyers. The money saved on the initial purchase is lost many times over in unreliable results, wasted research time, and eventual reorders from better vendors.
Trusting vendor COAs without verification
A certificate of analysis is only as trustworthy as the laboratory that produced it and the integrity of the vendor presenting it. Without independent verification, at least for initial orders, you are taking the vendor at their word. Given that 81% of GLP-1 peptide COAs show discrepancies when independently tested, blind trust is statistically unwise.
Improper storage destroying potency
Semaglutide that arrives at room temperature in a heat wave has already begun degrading. Product stored in a non-temperature-controlled location after arrival continues degrading. Reconstituted solution left on a counter for hours between doses accumulates damage. Each of these scenarios is common and each reduces the active compound in your vial. The semaglutide shelf life guide covers proper handling from delivery through final use. And if you discover your semaglutide arrived hot, specific guidance helps you assess whether the product is still usable.
Skipping dose escalation
Impatience kills more protocols than bad peptides do. Researchers who jump to high doses because they feel nothing at 0.25mg are setting themselves up for severe gastrointestinal distress when they hit a properly potent batch. The escalation protocol exists for safety. It is not optional, and it is not a suggestion. Even researchers who have experienced four weeks with no weight loss should continue escalating gradually rather than jumping doses.
Failing to track and document
Without consistent documentation, you cannot distinguish between product quality variation, dose response, and normal day-to-day fluctuation. A simple daily log covering dose, time, injection site, appetite level, energy, and any adverse effects provides the data foundation needed for meaningful protocol assessment. Researchers who track meticulously can identify weight loss plateaus early and adjust accordingly, rather than guessing about what went wrong. Understanding why weight loss stalls requires the kind of data that only careful documentation provides.
What the research community says about semaglutide sourcing
Online research communities provide valuable, if unverified, data points about vendor quality and sourcing experiences. Patterns emerge from thousands of individual reports that are worth understanding.
Vendor reputation patterns
Community consensus tends to stabilize around a small number of vendors with consistent quality reports. These vendors typically charge moderate to premium prices, maintain responsive customer service, and show consistent results across multiple independent user reports. The comprehensive vendor guide compiles community feedback and quality indicators for major peptide suppliers.
Vendors with volatile reputations, periods of excellent product followed by batches of poor quality, represent a different risk profile. They may source from multiple synthesis facilities without consistent quality standards. The product arriving in your mailbox might be excellent or it might be substandard, and you have no way to know before testing.
Community testing initiatives
Some research communities organize group testing, where multiple members submit samples from the same vendor batch to independent laboratories. These initiatives generate the most reliable vendor quality data available because they aggregate multiple independent data points from the same production batch.
Participating in or following these community testing efforts provides information that no amount of individual research can match. The collective data reveals patterns, both positive and negative, that help researchers make better sourcing decisions.
Forum red flags
Be cautious of vendor recommendations that come from accounts with no posting history, suspiciously polished reviews that read like marketing copy, and claims of pharmaceutical-grade quality at research peptide prices. Legitimate community members share nuanced experiences that include both positives and negatives. Pure enthusiasm without any reservations often signals vendor promotion rather than genuine user experience.
Long-term considerations for semaglutide research
Researchers who plan to work with semaglutide over extended periods face additional considerations beyond initial vendor selection.
Maintaining consistent supply
Research peptide vendors go out of business, change suppliers, or experience quality fluctuations. Having a backup vendor identified and tested before your primary source becomes unavailable prevents research interruptions that can compromise ongoing protocols.
Buying larger quantities when you have verified a good batch can make sense, but only if you have appropriate storage. Lyophilized semaglutide stored at minus 20 degrees Celsius remains stable for extended periods, making bulk purchases of verified product a reasonable strategy. Understanding proper peptide storage is essential for anyone maintaining inventory.
Transitioning between sources
If you switch vendors or even receive a new batch from the same vendor, treat it as a potentially different product. The conservative approach is to reduce your dose back to a level where any potency difference would be manageable, then gradually return to your target dose while monitoring for changes in effect.
This is especially important for researchers who have been stable on a particular dose for weeks. A sudden shift in potency, whether higher or lower, disrupts the equilibrium your body has established. Researchers who have explored restarting semaglutide after a break understand that dose re-escalation is necessary even with the same product, and it is even more critical when the source changes.
Knowing when to consider alternatives
Some researchers find that semaglutide does not produce expected results despite using verified, high-purity product. Before assuming the peptide does not work, evaluate other factors. Inadequate protein intake, poor sleep, high stress, and certain medications can all blunt the response. The guide on how long to stay on semaglutide helps researchers set realistic timelines.
If genuine non-response is confirmed, alternatives exist. Switching from semaglutide to tirzepatide is a common next step, as the dual-agonist mechanism may succeed where a GLP-1-only approach did not. The semaglutide to tirzepatide conversion chart helps researchers navigate the transition.
For researchers ready to stop semaglutide entirely, understanding withdrawal symptoms and whether cold turkey cessation is advisable helps manage the transition. Weight regain prevention strategies matter, too, whether you are maintaining results after tirzepatide or any GLP-1 compound.
Frequently asked questions
Is it legal to buy semaglutide peptide online?
Purchasing peptides labeled for research purposes is generally legal in the United States. However, the regulatory environment is evolving, particularly for GLP-1 compounds. Check current regulations in your jurisdiction before purchasing. Understanding peptide legality helps you stay within legal boundaries.
How can I tell if my semaglutide is real?
Visual inspection (white powder, dissolves clearly) provides basic screening. Vendor COA verification with the listed laboratory adds another layer. Independent testing through a peptide testing lab provides the most reliable confirmation. No single method is foolproof, but combining all three gives reasonable confidence in product authenticity.
What purity should I expect from research-grade semaglutide?
Reputable vendors typically achieve and verify 95 to 98% purity for semaglutide, which is more realistic than the 99% many claim. Given the structural complexity of semaglutide, any vendor claiming 99%+ purity should provide exceptionally detailed testing data to support that claim. Independent testing data suggests most research-grade semaglutide falls between 90 and 97% when honestly assessed.
How much semaglutide peptide should I buy for a research cycle?
A standard dose escalation to 2.4mg weekly over 16 weeks requires approximately 20mg of active semaglutide. Accounting for reconstitution losses and potential purity below claims, purchasing 25 to 30mg provides a reasonable buffer. Use the peptide cost calculator to compare per-dose costs across different purchase sizes.
Can I combine semaglutide with other peptides?
Many researchers investigate peptide combinations. Popular questions include combining semaglutide with tirzepatide, pairing it with B12 compounds, or exploring stacks with non-GLP-1 peptides. The peptide stacking calculator helps researchers evaluate combination protocols.
What should semaglutide look like after reconstitution?
Properly reconstituted semaglutide should be a clear, colorless solution with no visible particles, cloudiness, or color change. Any deviation from this, including unusual color, reddish tint, or visible matter, indicates potential contamination or degradation and the product should not be used.
How should I store semaglutide peptide long term?
Lyophilized semaglutide stores best at minus 20 degrees Celsius in a dry, sealed container. At refrigerator temperature (2 to 8 degrees Celsius), stability extends up to 36 months. Reconstituted solutions should be refrigerated and used within 28 days. Never freeze reconstituted semaglutide. For detailed guidance, see the complete peptide storage guide.
What happens if my semaglutide was not kept cold during shipping?
Lyophilized semaglutide can tolerate temperatures up to 30 degrees Celsius for up to 14 days without significant degradation. If shipping took longer or temperatures exceeded this, potency may be reduced. The semaglutide arrived hot guide helps you assess whether your product remains viable.
External resources
For researchers serious about optimizing their peptide sourcing and protocols, SeekPeptides provides the most comprehensive resource available. Members access evidence-based vendor evaluations, detailed protocol guides, dosing calculators, and a community of thousands of experienced researchers who have navigated these exact purchasing decisions.
In case I do not see you, good afternoon, good evening, and good night. May your peptides stay pure, your vendors stay honest, and your protocols stay consistent.