Feb 8, 2026

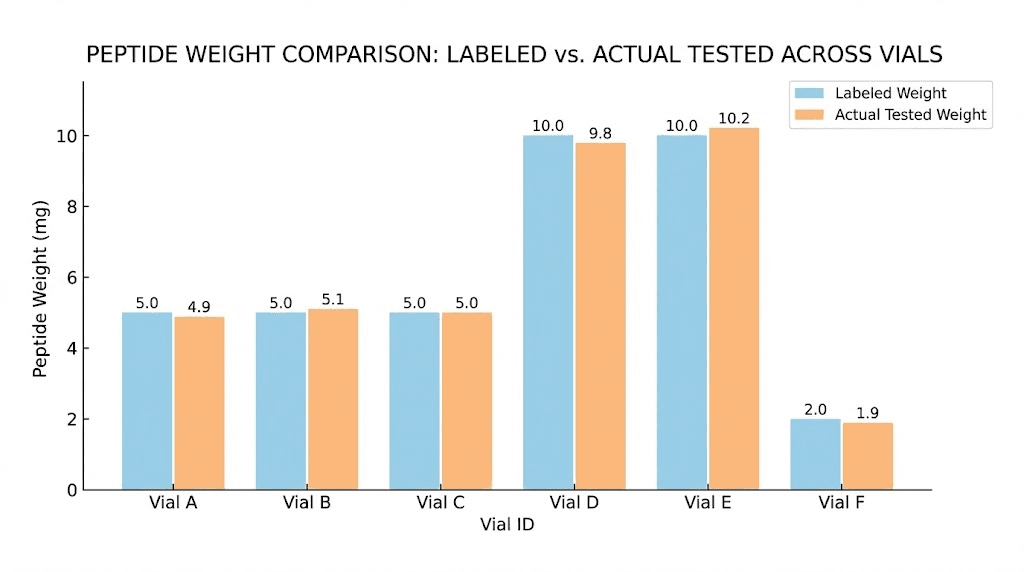
Independent testing scored Peptide Sciences a C rating for their tirzepatide. Purity looked good on paper, ranging from 98.73% to 99.95%. But the actual weight inside those vials told a different story. Samples labeled 5mg contained as little as 3.89mg. That is a 22.2% shortfall, and for a peptide where precise dosing matters enormously, that gap between what you pay for and what you receive changes everything.
This is the core tension surrounding Peptide Sciences and their tirzepatide offering. The company has built genuine credibility in the research peptide market over more than a decade. They ship fast. Their catalog runs deep. Their purity numbers consistently test high. But when tirzepatide entered the picture, a peptide commanding premium prices and attracting unprecedented demand, the cracks in their quality control became impossible to ignore.
And the landscape has shifted dramatically since those tests were conducted. Eli Lilly, the patent holder for tirzepatide, has launched an aggressive legal campaign against vendors and compounders. The FDA removed tirzepatide from its drug shortage list in December 2024. Compounding pharmacies faced mandatory shutdown deadlines. Research peptide vendors have been quietly pulling tirzepatide from their catalogs.
So what does all of this mean for researchers who have relied on Peptide Sciences for their weight loss peptide research? This guide breaks down the testing data, the availability timeline, the quality concerns, and the alternatives. Whether you are evaluating your current vendor or searching for a new source entirely, the information here will help you make a decision grounded in data rather than marketing claims. SeekPeptides has tracked these vendor developments closely, and what follows reflects the most current intelligence available.
Who is Peptide Sciences and why do researchers use them
Peptide Sciences operates out of Henderson, Nevada. They have been in the research peptide business since at least 2012, making them one of the longer-running domestic vendors in a market where companies appear and disappear with alarming regularity. That longevity alone sets them apart. Many researchers chose Peptide Sciences specifically because the company has not vanished overnight, has not rebranded under suspicious circumstances, and has maintained a consistent web presence for over a decade.
Their catalog covers an impressive range. BPC-157, TB-500, GHK-Cu, various growth hormone secretagogues, selank, semax, and dozens more. The company claims to synthesize peptides in-house using both automated and manual peptide synthesizers, with every product undergoing HPLC and mass spectrometry analysis. They provide Certificates of Analysis for their products and state that purity exceeds 99% across their lineup.
The customer experience reflects this mid-to-premium positioning. Orders placed before the daily cutoff typically ship same day. Domestic shipping within the United States is fast. Credit card payments are accepted, which removes the cryptocurrency barrier that frustrates researchers dealing with less established vendors. Their website is clean, professional, and well-organized.
But here is where the story gets complicated.
That 4.4 out of 5 average rating across 146+ reviews masks a pattern that becomes visible only when you read the negative reviews carefully. Customer service responses are inconsistent. Return and refund policies are restrictive, with replacements issued only for incorrect shipments. And the testing that supports those impressive purity claims? It is performed internally, not by independent third-party laboratories. For many experienced researchers who have learned the hard way that vendor claims and actual product quality do not always align, this distinction matters enormously.
The independent testing data on Peptide Sciences tirzepatide
Finnrick Analytics, an independent peptide testing organization, evaluated 12 samples of Peptide Sciences tirzepatide between March and July 2025. The results paint a nuanced picture that every researcher should understand before placing an order.
Purity tested well. Across those 12 samples, HPLC purity ranged from 98.73% to 99.95%. These are solid numbers. For context, most researchers consider anything above 98% acceptable for general research purposes, and anything above 99% to be high quality. On purity alone, Peptide Sciences delivered.
Weight accuracy told a different story entirely.
Samples showed significant shortfalls compared to their labeled amounts. Vials labeled as containing 5mg actually contained between 3.89mg and 4.29mg of tirzepatide. That is a shortfall ranging from 14.2% to 22.2% below the advertised amount. The 10mg vials performed somewhat better but still came up short, containing between 7.79mg and 9.65mg. Even at the best end of those ranges, researchers received less product than they paid for.
The overall test score averaged 6.7 out of 10, with individual scores ranging from 4.7 to 8.0. This earned Peptide Sciences a C rating, described as simply "Okay." For a vendor that positions itself as premium and charges accordingly, "okay" is not a ringing endorsement.
To put this in perspective, other vendors testing tirzepatide through the same independent lab achieved A ratings. Polaris Peptides was among those top-rated alternatives. The gap between a C and an A is not just academic. It represents real differences in what researchers receive for their money.
Why weight accuracy matters more than purity
Most researchers fixate on purity percentages. It makes sense. A 99% pure peptide sounds impressive. But purity tells you about what is in the vial, not how much is in the vial. If your 5mg vial is 99.95% pure but only contains 3.89mg of product, you have a very pure but significantly undersized sample.
For tirzepatide dosing calculations, this creates serious problems. Researchers who reconstitute based on the labeled amount will end up with a solution that is weaker than expected. Every subsequent dose drawn from that vial will deliver less active compound than intended. For a peptide where the difference between 2.5mg and 5mg represents a meaningful dose escalation, a 22% shortfall is not a rounding error. It fundamentally changes the research parameters.
This is precisely why SeekPeptides emphasizes third-party testing verification in all vendor evaluations. Internal testing can confirm purity. But only independent weighing and analysis can confirm that what is on the label matches what is in the vial.
Tirzepatide availability changes and why vendors are pulling it
The tirzepatide market for research peptides has undergone seismic shifts. Understanding these changes is critical for any researcher evaluating Peptide Sciences or any other vendor.
The timeline is straightforward but its implications are anything but simple.
In October 2024, the FDA determined that the tirzepatide injection shortage had been resolved. On December 19, 2024, tirzepatide was officially removed from the FDA drug shortage list. This single regulatory action triggered a cascade of consequences that reshaped the entire market for research peptides and compounded medications alike.
The compounding pharmacy shutdown
While tirzepatide remained on the shortage list, compounding pharmacies operated under a legal exemption. They could produce their own versions of the medication to address the supply gap. Once the shortage ended, that exemption evaporated. The FDA established two deadlines. Smaller state-licensed 503A pharmacies had until February 18, 2025. Larger 503B outsourcing facilities had until March 19, 2025. After those dates, compounding tirzepatide became a direct violation of the Food, Drug, and Cosmetic Act.
Some compounders pushed back. Legal challenges were filed. But on May 7, 2025, a US District Court in the Northern District of Texas upheld the FDA decision. The legal path for compounded tirzepatide narrowed to a sliver, available only for patients with verifiable clinical needs that the commercially available formulation could not meet.
Eli Lilly legal offensive
Eli Lilly did not wait for the courts to sort things out. In April 2025, the company filed the first wave of what industry experts predicted would be dozens of lawsuits. Initial targets included compounding pharmacies like Strive and Empower, which Lilly accused of continuing to produce mass-manufactured tirzepatide while falsely marketing it as "personalized" medication. By late April, Lilly had expanded its legal campaign to telehealth companies facilitating access to compounded tirzepatide.
The message was unmistakable. Eli Lilly intended to protect its tirzepatide patent aggressively, and any entity selling, compounding, or distributing unauthorized versions faced serious legal exposure.
Impact on research peptide vendors
Research peptide vendors occupy a different legal space than compounding pharmacies. They sell products labeled "for research purposes only" and are not directly subject to the same compounding regulations. However, the intensified enforcement environment created enormous pressure. Multiple vendors quietly removed tirzepatide and semaglutide from their catalogs throughout 2025.
Peptide Sciences appears to have followed this trend. The company has indicated a strategic pivot toward "novel, non-commercial sequences for preclinical investigation" rather than commercially available GLP-1 and GIP compounds. This shift is pragmatic. When a pharmaceutical giant with essentially unlimited legal resources is actively suing everyone in the supply chain, even vendors with strong legal positions have good reason to step aside.
For researchers, this means the window for obtaining tirzepatide through traditional research peptide channels has largely closed. Those still listing it may be operating on borrowed time, and the quality and authenticity of remaining inventory becomes even more questionable as established vendors exit the market.
How tirzepatide works and why researchers study it
Understanding the science behind tirzepatide explains why demand from the research community has been so intense, and why its removal from vendor catalogs represents such a significant loss for preclinical investigation.
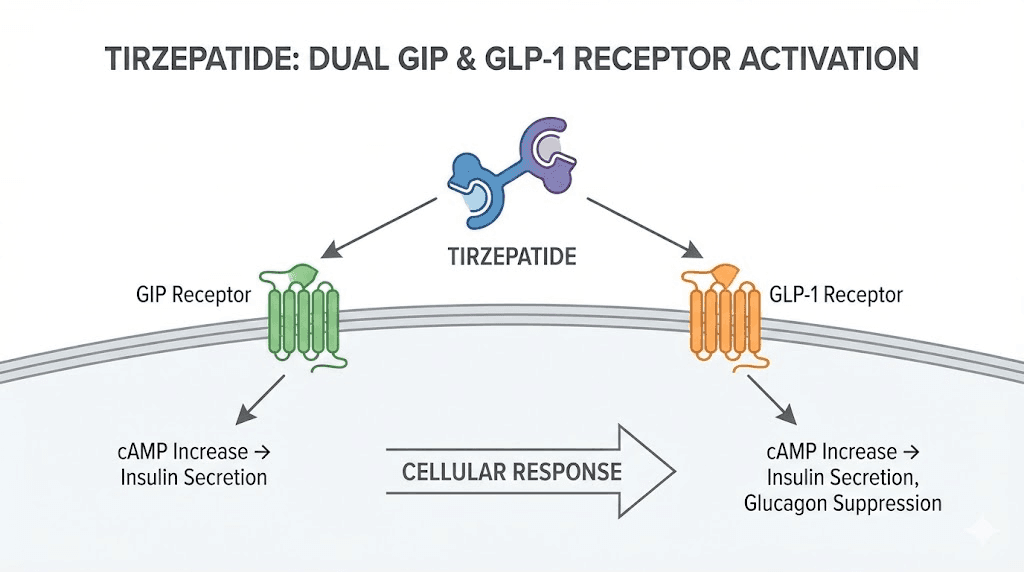
Tirzepatide is not just another weight loss peptide. It is a first-in-class dual agonist that simultaneously activates two distinct receptor pathways: the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. This dual mechanism sets it apart from single-target GLP-1 receptor agonists like semaglutide.
The peptide was engineered from the native GIP sequence. Its affinity for GIP receptors matches that of the naturally occurring GIP hormone. Its affinity for GLP-1 receptors is approximately five times weaker than native GLP-1, creating an intentional imbalance that researchers believe contributes to its unique efficacy profile.
The SURMOUNT clinical trial program
The clinical evidence supporting tirzepatide is extraordinary by any standard. The SURMOUNT trial program generated weight loss data that rewrote expectations for what pharmacological intervention could achieve.
SURMOUNT-1 enrolled adults with overweight or obesity. The results were striking. Participants receiving the 5mg dose achieved an average 16.0% body weight reduction. The 10mg dose produced 21.4% reduction. And the 15mg dose delivered 22.5% reduction in body weight. These numbers are not percentages of excess weight. They represent total body weight, meaning a 200-pound person on the highest dose lost approximately 45 pounds on average.
Perhaps more impressive than the averages was the consistency. Between 89% and 91% of participants on the 10mg and 15mg doses achieved at least 5% weight reduction. Across the full SURMOUNT program, mean weight reduction reached up to 26% in some analyses.
A direct comparison with semaglutide published in the New England Journal of Medicine confirmed what many researchers suspected. Among participants with obesity but without diabetes, tirzepatide was superior to semaglutide for both body weight reduction and waist circumference reduction at 72 weeks. The dual agonist approach was not just theoretically elegant. It produced measurably better outcomes.
Why the dual mechanism matters for research
The co-activation of GIP and GLP-1 pathways produces effects that neither pathway achieves alone. Research has demonstrated that simultaneous GIP and GLP-1 receptor stimulation creates a synergistic insulin response and glucagonostatic effect significantly greater than what separate administration of each hormone produces.
For preclinical researchers studying metabolic pathways, energy homeostasis, appetite regulation, and fat metabolism, tirzepatide represents a uniquely valuable tool. It allows investigation of dual-pathway activation in ways that mixing two separate compounds cannot replicate, because the pharmacokinetics of a single molecule activating both receptors differ fundamentally from two separate molecules each hitting one receptor.
This scientific value is precisely what makes the current supply disruption so frustrating for the research community.
Peptide Sciences pricing and value analysis
Peptide Sciences positions itself in the mid-to-premium price tier of the research peptide market. Their pricing is clearly listed on their website, which is a positive in an industry where some vendors hide costs behind account creation requirements or inquiry forms. Transparency in pricing at least allows researchers to make informed comparisons.
For tirzepatide specifically, Peptide Sciences charged prices consistent with their premium positioning. However, when you factor in the weight accuracy data from independent testing, the effective price per milligram of actual product received tells a very different story.
Consider the math. If a 5mg vial costs a certain amount but actually contains only 3.89mg, you are paying for 5mg but receiving 3.89mg. That is a 22.2% price premium beyond the listed cost, paid not for quality but for missing product. A researcher running a protocol that requires precisely calculated doses of tirzepatide would need to order approximately 22% more product to achieve the same total amount of active compound.
This changes the competitive equation dramatically. A vendor charging lower list prices but delivering accurate weights might actually provide better value than a premium-priced vendor with weight shortfalls. Without independent testing data, researchers have no way to make this calculation.
The hidden costs of internal testing only
Peptide Sciences provides Certificates of Analysis with their products. These COAs document purity via HPLC and mass spectrometry. The testing is conducted internally at their analytical laboratory. For many peptides, this level of quality documentation is better than what competing vendors provide, as some offer no testing documentation whatsoever.
But internal testing has inherent limitations. The entity producing the product is also the entity certifying its quality. This is not necessarily evidence of wrongdoing. Many legitimate manufacturers perform internal QC testing. However, it lacks the credibility that comes with independent verification, where the testing lab has no financial relationship with the manufacturer and no incentive to produce favorable results.
For researchers operating in institutional settings where third-party verification is expected or required, Peptide Sciences internal COAs may not meet compliance standards. This limits the utility of their products for certain research applications regardless of actual quality.
Shipping, customer service, and the ordering experience
Peptide Sciences handles logistics well. That is the consensus across hundreds of customer reviews and it deserves recognition. When the supply chain works, it works fast.
Orders placed before the daily cutoff ship same day from their Nevada facility. Domestic delivery within the United States is typically fast, with most researchers receiving their orders within a few business days. The company ships from within the US, which eliminates the customs delays and seizure risks that plague orders from overseas vendors. For researchers accustomed to waiting weeks for international shipments that may or may not clear customs, this domestic advantage is significant.
Payment processing is straightforward. Peptide Sciences accepts credit cards, removing the cryptocurrency-only barrier that many researchers find inconvenient with other vendors. The website interface is professional, products are well-organized, and the ordering process is intuitive.
Customer service, however, draws more mixed reviews.
Positive experiences exist. Some researchers report responsive communication and helpful interactions. But a consistent thread in negative reviews describes slow response times, difficulty reaching support when issues arise, and limited options for resolution when problems occur. The company does not offer refunds or returns except when the wrong product is shipped or an item arrives incorrect or incomplete. For a premium vendor, this inflexibility frustrates researchers who have legitimate concerns about product quality.
The no-refund policy becomes particularly problematic in the context of weight accuracy shortfalls. If a researcher receives a vial that independent testing would reveal contains 22% less product than labeled, there is no recourse through the company return policy. The product arrived. It is the correct product. That it contains less than advertised would need to be proven through independent testing that the researcher must fund separately.
How Peptide Sciences compares to other tirzepatide vendors
Context matters. A C rating from independent testing sounds mediocre in isolation, but how does Peptide Sciences stack up against the broader market?
The tirzepatide vendor landscape is divided into distinct tiers based on independent testing data. At the top, vendors achieving A ratings demonstrated both high purity and accurate weights. Polaris Peptides earned this top rating. These vendors deliver what their labels promise, both in terms of purity and quantity.
In the middle, vendors like Peptide Sciences occupy the C range. Good purity but inconsistent weight accuracy. The product works, but you may not get the full amount you paid for. The research is still possible, but protocol precision is compromised.
At the bottom, some vendors tested by independent labs showed purity as low as 75% and weight divergences of up to 640% from advertised amounts. These are not vendors cutting corners. These are vendors selling products that barely qualify as the advertised compound.
Compared to the worst actors in the market, Peptide Sciences is genuinely good. Their purity is consistently high. Their product is authentic tirzepatide. The shortfalls are in quantity, not identity or purity. For researchers whose primary concern is "is this actually tirzepatide," Peptide Sciences delivers.
But compared to the best vendors, Peptide Sciences leaves money on the table for researchers. A 22% weight shortfall at premium prices is hard to justify when alternatives exist that deliver accurate weights at competitive prices. The question each researcher must answer is whether the convenience of Peptide Sciences, including their established track record, fast shipping, and credit card processing, justifies the premium plus shortfall.
Vendor evaluation framework
When evaluating any research peptide vendor, researchers should assess five dimensions. Here is how Peptide Sciences performs on each.
Evaluation criteria | Peptide Sciences rating | Notes |
|---|---|---|
Purity | 9/10 | 98.73-99.95% HPLC purity, consistently excellent |
Weight accuracy | 5/10 | Shortfalls of 3.5-22.2% below labeled amounts |
Third-party testing | 4/10 | Internal testing only, COAs available but not independent |
Shipping and logistics | 9/10 | Same-day shipping, domestic US, credit cards accepted |
Customer service | 6/10 | Mixed reviews, no refund policy, inconsistent response times |
The pattern is clear. Peptide Sciences excels operationally but falls short on the two metrics that matter most for research integrity: weight accuracy and independent verification. Researchers must decide which factors carry more weight for their specific applications.
Reconstitution and handling considerations for tirzepatide
Assuming a researcher does obtain tirzepatide from Peptide Sciences or any other vendor, proper handling is essential to preserve whatever product is actually in the vial. Peptide reconstitution is not complicated, but it demands precision and attention to detail.
Tirzepatide ships in lyophilized form. This freeze-dried powder format dramatically extends shelf life and prevents degradation during transit. The reconstitution process converts this powder back into an injectable solution at a precise concentration.
Standard reconstitution protocol
A typical research reconstitution uses 2mL of sterile diluent per 10mg vial, creating a 5mg/mL solution. For 5mg vials, 1mL of diluent produces the same concentration. The choice of diluent matters significantly for multi-use research.
Bacteriostatic water is the standard for multi-use applications because it contains a preservative (0.9% benzyl alcohol) that inhibits bacterial growth. Plain sterile water works only if the entire vial will be used in a single session, as it contains no preservatives and contamination risk escalates rapidly after opening.
The technique matters as much as the materials. Insert the needle through the vial stopper and direct the stream of water slowly toward the inner wall of the vial. Never spray directly onto the powder. Gentle. Slow. Deliberate. This protects the peptide structure from mechanical damage.
Never shake the vial after adding diluent. Shaking introduces mechanical energy that can denature the peptide, breaking the bonds that maintain its biological activity. Instead, gently swirl the vial until the powder dissolves completely. If it does not dissolve within a few minutes of gentle swirling, set the vial in the refrigerator for 15-20 minutes and try again. Do not force dissolution through vigorous mixing.
Storage requirements
Proper storage directly impacts how long your reconstituted tirzepatide remains viable. The guidelines are specific and non-negotiable.
Lyophilized (unreconstituted) storage: Store at -20 degrees Celsius (-4 degrees Fahrenheit) in dry, dark conditions. Minimize moisture exposure. At this temperature, lyophilized tirzepatide maintains stability for extended periods. Some researchers store unreconstituted peptides at standard freezer temperatures for months without significant degradation.
Reconstituted storage: Refrigerate at 2-8 degrees Celsius (35.6-46.4 degrees Fahrenheit). Do not freeze reconstituted solution, as ice crystal formation can damage the peptide structure. Use reconstituted solution within 28 days. After 28 days, degradation may compromise research accuracy even if the solution appears unchanged.
For researchers working with tirzepatide from any source, proper refrigeration protocols are essential. The SeekPeptides reconstitution calculator can help determine exact dilution volumes for target concentrations.
The weight accuracy problem amplified
This is where the Peptide Sciences weight accuracy issue becomes particularly problematic. Standard reconstitution calculations assume the labeled amount is accurate. If you add 2mL of bacteriostatic water to a vial labeled 10mg, you expect a 5mg/mL solution. But if that vial actually contains 7.79mg (the low end of tested 10mg vials), your actual concentration is 3.895mg/mL, not 5mg/mL.
Every dose drawn from that vial will be 22% weaker than intended. A researcher targeting a 2.5mg dose would actually administer approximately 1.95mg. A 5mg target dose would deliver approximately 3.9mg. Over a multi-week protocol, this systematic underdosing compounds, potentially invalidating research outcomes.
Without sending samples for independent testing, researchers have no way to correct for this discrepancy. They would simply observe weaker-than-expected results and potentially conclude that tirzepatide was less effective at the intended dose, when the actual issue was receiving less product than paid for.
Is Peptide Sciences a compounding pharmacy
This question comes up frequently, and the distinction matters both legally and practically. Peptide Sciences is not a compounding pharmacy. They are a research chemical supplier. The difference is fundamental.
Compounding pharmacies are licensed to prepare customized medications for individual patients based on prescriptions. They operate under FDA oversight through either 503A (state-licensed) or 503B (outsourcing facility) designations. Their products are intended for human use. They must comply with good manufacturing practices and are subject to FDA inspections.
Research peptide vendors like Peptide Sciences sell products labeled "for research purposes only" and "not for human consumption." They are not pharmacies. They do not fill prescriptions. Their products are sold for in vitro research, preclinical studies, and scientific investigation. This distinction places them in a different regulatory category than compounding pharmacies.
This matters in the current tirzepatide enforcement environment because the FDA compounding shutdown and Eli Lilly lawsuits primarily target entities producing tirzepatide for human use. Research chemical vendors face different legal considerations, though the aggressive enforcement climate has still motivated many to remove tirzepatide from their catalogs voluntarily.
For researchers evaluating Peptide Sciences, understanding this distinction is important. Products from research chemical vendors have not undergone the same manufacturing oversight as compounded medications or FDA-approved pharmaceuticals. The quality assurance burden falls more heavily on the researcher to verify what they receive.
Alternatives for tirzepatide research
Given the availability challenges and quality concerns, researchers studying tirzepatide mechanisms have several paths forward. Each comes with tradeoffs.
Other research peptide vendors
Some vendors continue to offer tirzepatide. Those with independent testing data and higher ratings may provide better weight accuracy than Peptide Sciences. Polaris Peptides earned an A rating in independent testing. However, the enforcement environment means any vendor listing could change rapidly. Researchers should verify availability and obtain their own independent testing when possible.
Studying related GLP-1 compounds
For researchers whose work focuses on GLP-1 pathway mechanisms rather than tirzepatide specifically, related compounds remain more widely available. Retatrutide, a triple agonist targeting GIP, GLP-1, and glucagon receptors, is still available through some research vendors, though Eli Lilly (also its developer) may eventually extend enforcement to this compound as well.
Other incretin pathway compounds offer avenues for related research. Cagrilintide, an amylin analog, works through complementary mechanisms and is being studied in combination with semaglutide. These alternatives do not replicate tirzepatide dual agonism but allow investigation of overlapping metabolic pathways.
Institutional and pharmaceutical sources
Researchers in academic or institutional settings may be able to obtain tirzepatide through pharmaceutical partnerships, clinical trial material sharing agreements, or reference standard suppliers. These channels typically provide higher quality assurance than research peptide vendors, though access is more restricted and costs may be higher.
The case for comprehensive peptide education
Regardless of which vendor or compound a researcher chooses, the foundation of good research is understanding the molecules being studied. SeekPeptides provides comprehensive peptide education resources, dosage calculators, and protocol guidance that help researchers make informed decisions regardless of their peptide source.
Red flags to watch for when evaluating any tirzepatide vendor
The shrinking availability of tirzepatide through legitimate channels creates an environment ripe for exploitation. As established vendors exit the market, less scrupulous operators may fill the vacuum. Researchers should watch for specific warning signs.
Too-good-to-be-true pricing
Tirzepatide synthesis is expensive. The compound is a complex 39-amino-acid peptide with a C20 fatty diacid moiety. Legitimate synthesis at high purity requires sophisticated equipment and expertise. If a vendor offers tirzepatide at dramatically lower prices than established competitors, question how they achieve those savings. Cutting corners on synthesis, using lower-grade reagents, or simply mislabeling other compounds are all possibilities.
No testing documentation
Any vendor selling tirzepatide should provide a Certificate of Analysis at minimum. While internal COAs have limitations (as discussed regarding Peptide Sciences), they are still preferable to no documentation at all. Vendors who cannot or will not provide testing data for their tirzepatide products should be avoided entirely. The independent peptide testing landscape has matured enough that researchers can verify vendor claims if they choose to invest in third-party analysis.
New or unknown vendors suddenly stocking tirzepatide
When established vendors remove a product due to legal pressure, new entities sometimes appear offering that exact product. These pop-up vendors may lack the synthesis capabilities, quality control infrastructure, and track record that more established companies have built. A vendor with no history, no reviews, and no verifiable testing data should be approached with extreme caution regardless of their tirzepatide pricing or availability claims.
Claims of exemption from regulations
Some vendors may claim their tirzepatide products are exempt from current enforcement actions for various reasons. They might cite their research-use-only labeling, their location outside US jurisdiction, or creative legal interpretations. While the legal landscape for research peptides is genuinely complex, researchers should be wary of vendors who seem more focused on explaining why they can still sell tirzepatide than on demonstrating the quality of what they sell.
Pressure tactics and limited availability claims
"Order now before we sell out" messaging exploits the genuine scarcity of tirzepatide to pressure researchers into hasty purchasing decisions. Legitimate vendors with consistent supply do not need to create urgency. If a vendor's primary marketing message is about limited availability rather than product quality and testing data, their priorities may not align with your research needs.
Understanding tirzepatide dosing for research contexts
Researchers working with tirzepatide need to understand the established dosing frameworks from clinical trials, both to design appropriate protocols and to recognize when vendor weight shortfalls meaningfully impact their work.
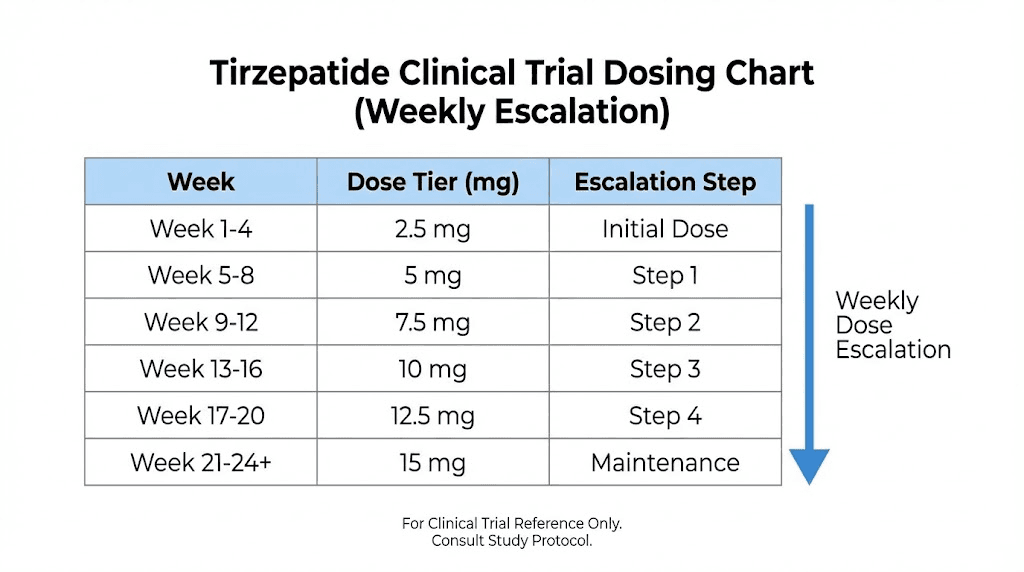
The clinical development program established five dose tiers: 2.5mg, 5mg, 7.5mg, 10mg, and 15mg, administered once weekly via subcutaneous injection. The standard clinical titration starts at 2.5mg for four weeks, then increases to 5mg. Subsequent dose escalations occur in 2.5mg increments at four-week intervals as tolerated, up to a maximum of 15mg weekly.
This escalation schedule exists for good reason. Side effects, primarily gastrointestinal, are dose-dependent and often diminish with continued use at each dose level. Rushing the escalation increases the likelihood and severity of nausea, which is the most commonly reported adverse effect.
Translating clinical doses to research concentrations
For researchers using reconstituted tirzepatide from research vendors, converting between mg doses and injection volumes requires attention to concentration calculations. Using the standard 10mg vial reconstituted with 2mL bacteriostatic water (5mg/mL) as an example.
Target dose | Volume at 5mg/mL | Volume if vial actually contains 7.79mg (3.895mg/mL) | Actual dose at intended volume |
|---|---|---|---|
2.5mg | 0.50mL (50 units) | 0.64mL would be needed | 1.95mg received |
5mg | 1.00mL (100 units) | 1.28mL would be needed | 3.90mg received |
7.5mg | 1.50mL (150 units) | 1.92mL would be needed | 5.84mg received |
10mg | 2.00mL (200 units) | 2.57mL would be needed | 7.79mg received |
The practical impact is clear. At every dose level, researchers using Peptide Sciences tirzepatide with low-end weight accuracy would systematically underdose by approximately 22%. This is not a trivial variance. It represents the difference between a therapeutic dose and a sub-therapeutic dose at multiple points in the escalation schedule. Our peptide dosage calculator helps researchers compute these concentrations accurately, but even the best calculator cannot correct for inaccurate starting material.
The importance of microdosing precision
Some researchers explore tirzepatide at doses below the standard clinical range. Microdosing protocols using 1mg or lower require even greater precision in reconstitution and vendor weight accuracy. When the target dose is already small, a 22% shortfall creates proportionally larger deviations from intended research parameters.
For any researcher pursuing precision dosing work, vendor weight accuracy is arguably the single most important factor in vendor selection, more important than purity, price, or shipping speed.
What the regulatory future looks like for research tirzepatide
The trajectory is unmistakable. Access to tirzepatide through research peptide vendors will continue to narrow. Several forces are converging to accelerate this trend.
First, Eli Lilly shows no signs of relaxing its enforcement posture. If anything, the company has expanded its legal campaign from compounding pharmacies to telehealth platforms to individual vendors. Each successful lawsuit strengthens the legal precedent and emboldens further action. The financial incentive is enormous. Tirzepatide (marketed as Mounjaro for diabetes and Zepbound for weight management) represents billions in annual revenue for Lilly. Protecting that revenue stream justifies significant legal spending.
Second, the SAFE Drugs Act and similar legislation has created new tools for enforcement against unauthorized drug sales. The regulatory framework is tightening, not loosening. Research peptide vendors that could previously operate in regulatory gray areas face a more clearly defined legal landscape with more explicit prohibitions.
Third, supply chain dynamics are shifting. As major vendors exit the tirzepatide market, the remaining supply becomes concentrated among smaller, potentially less reliable sources. This increases quality risk while simultaneously reducing researcher options. The vendors most likely to continue selling tirzepatide are those with the highest risk tolerance, which does not always correlate with the highest quality standards.
For the research community, the practical implication is adaptation. Researchers who have built protocols around research-grade tirzepatide need to either transition to alternative compounds, seek institutional pharmaceutical sources, or find the diminishing number of high-quality vendors who still carry it. SeekPeptides continues to monitor vendor availability and quality data to help researchers navigate these transitions.
The broader context of peptide vendor quality
The Peptide Sciences tirzepatide situation is not unique. It reflects systemic challenges in the research peptide industry that affect every compound, not just tirzepatide.
Independent testing across multiple vendors and compounds has revealed that weight accuracy issues are widespread. The finding that purity across tirzepatide vendors ranged from 75% to 99.6% and quantity diverged by up to 640% from advertised amounts reveals an industry where the gap between marketing and reality can be enormous.
This gap exists partly because the research peptide market operates with minimal regulatory oversight. Vendors are not required to submit products for independent testing. They are not subject to the manufacturing standards that govern pharmaceutical production. Customer protections are limited. And the technical knowledge required to independently verify product quality, including HPLC access and mass spectrometry capabilities, is beyond the reach of most individual researchers.
The result is an information asymmetry that favors vendors over researchers. Vendors know what is in their products. Researchers must trust vendor claims or invest in expensive independent testing. Organizations like Finnrick Analytics have begun to address this imbalance, but independent testing coverage remains limited to a fraction of available products and vendors.
What researchers can do to protect themselves
Several practical strategies can help researchers navigate this imperfect market.
Prioritize vendors with independent testing data. When third-party results are available, use them. Even imperfect independent data is more reliable than vendor self-testing alone.
Pool resources for independent testing. Individual testing is expensive, but researchers can share costs by organizing group testing of common products. This distributes the financial burden while generating valuable quality data.
Start with small orders. Before committing to a large purchase from any vendor, order small quantities and evaluate them. Look for consistency in appearance, dissolution behavior, and if possible, send samples for independent analysis.
Document everything. Keep records of lot numbers, appearance, reconstitution behavior, and research outcomes for every vendor and product. Over time, this data helps identify patterns and compare vendors even without formal laboratory testing.
Use established testing resources. Multiple services now offer peptide purity and mass testing at reasonable prices. The investment in a single test can save significantly more in wasted research time and invalid results from substandard products.
Stay informed. The vendor landscape changes rapidly. Companies that were reliable six months ago may have changed sourcing, synthesis methods, or management. Research communities and forums provide real-time intelligence that formal reviews cannot match.
Tirzepatide safety considerations for research protocols
Regardless of vendor source, researchers working with tirzepatide should understand its established safety profile from clinical trials. This knowledge informs both protocol design and the interpretation of research outcomes.
The SURMOUNT trials documented several common adverse effects. Gastrointestinal symptoms dominated, with nausea, diarrhea, and constipation being the most frequently reported. These effects were generally mild to moderate, dose-dependent, and tended to diminish with continued use at each dose level. The four-week titration between dose increases was specifically designed to minimize these effects by allowing GI adaptation before increasing the dose further.
Fatigue has been reported by some users. The mechanism likely involves the metabolic shifts associated with significant caloric intake reduction and rapid weight loss rather than a direct pharmacological effect of tirzepatide on energy pathways. Researchers should account for this when designing protocols that measure activity levels or energy expenditure.
More serious but less common adverse effects in clinical trials included pancreatitis (rare), gallbladder events (slightly elevated in treatment groups), and injection site reactions. These mirror the safety profile seen with GLP-1 receptor agonists broadly and are expected given the mechanism of action.
For researchers using tirzepatide sourced from vendors with known weight accuracy issues, an additional safety consideration arises. If a researcher adjusts their protocol upward to compensate for suspected underdosing and subsequently switches to a more accurately dosed source, the actual dose received could be significantly higher than intended. This kind of uncontrolled variable introduces risk that proper vendor verification would prevent.
How to verify tirzepatide authenticity independently
For researchers committed to quality verification, several testing options exist.
HPLC purity testing
High-Performance Liquid Chromatography remains the gold standard for peptide purity assessment. It separates the target compound from impurities and provides a purity percentage. Multiple commercial laboratories now offer HPLC testing for individual peptide samples at prices accessible to individual researchers. While HPLC confirms purity and identity, it does not directly measure total quantity in the vial.
Mass spectrometry confirmation
Mass spectrometry confirms molecular identity by measuring the mass-to-charge ratio of the compound. For tirzepatide, which has a well-characterized molecular weight (approximately 4813.45 Da), mass spec analysis can definitively confirm whether the sample is genuine tirzepatide or a different compound entirely. This test is particularly valuable when sourcing from new or unverified vendors.
Gravimetric analysis for weight verification
Weighing the actual peptide content requires careful technique. Lyophilized peptides are hygroscopic and low-density, making accurate weighing challenging without analytical balances capable of sub-milligram precision. For researchers with access to appropriate equipment, gravimetric verification provides the most direct measure of whether labeled amounts are accurate.
Third-party testing services
Several services specifically cater to peptide testing for the research community. These services accept individual samples, perform HPLC, mass spec, and sometimes gravimetric analysis, and provide independent certificates of analysis. The cost varies but typically ranges from $50-200 per sample depending on the scope of testing requested.
For high-value compounds like tirzepatide, the cost of a single independent test is a small fraction of the total purchase price and can prevent far more expensive consequences from using inaccurate or contaminated material.
Frequently asked questions
Does Peptide Sciences still sell tirzepatide?
Availability has been fluctuating due to the enforcement environment. Peptide Sciences has indicated a strategic shift toward novel, non-commercial sequences. Researchers should check their current catalog directly, but should be prepared for the possibility that tirzepatide may no longer be available or may be removed without notice.
Is Peptide Sciences tirzepatide the same as pharmaceutical tirzepatide?
Research-grade tirzepatide from any vendor is synthesized using peptide synthesis techniques, not manufactured under the same conditions as pharmaceutical-grade products. While the molecular structure is intended to be identical, the manufacturing standards, quality oversight, and regulatory compliance differ significantly. Independent testing showed Peptide Sciences tirzepatide had high purity but inconsistent weight accuracy.
How much tirzepatide is actually in a Peptide Sciences vial?
Independent testing of 12 samples showed 5mg vials contained 3.89-4.29mg and 10mg vials contained 7.79-9.65mg. Purity was high (98.73-99.95%), meaning what was in the vial was genuinely tirzepatide, but there was less of it than labeled. Use our peptide calculator to adjust reconstitution volumes if you suspect weight shortfalls.
Why did Peptide Sciences get a C rating for tirzepatide?
The C rating from Finnrick Analytics reflected the combination of high purity but significant weight shortfalls. While purity scores were excellent (98.73-99.95%), the quantity delivered fell 3.5-22.2% below labeled amounts across 12 tested samples. The average test score was 6.7 out of 10.
Can I still get compounded tirzepatide from a pharmacy?
Compounded tirzepatide became severely restricted after the FDA removed it from the drug shortage list in December 2024. Compounding pharmacies faced deadlines of February and March 2025 to cease production. A May 2025 court ruling upheld the FDA decision. Limited compounding may still occur for patients with verifiable clinical needs unmet by commercial formulations, but widespread availability has ended. Learn more in our tirzepatide availability guide.
What is the best alternative vendor for tirzepatide?
Polaris Peptides earned an A rating in independent tirzepatide testing. However, availability across all vendors is declining due to enforcement pressure. Researchers should verify current stock and obtain independent testing when possible. Our vendor comparison guide provides regularly updated assessments.
How should I store tirzepatide from Peptide Sciences?
Lyophilized tirzepatide should be stored at -20 degrees Celsius in dry, dark conditions. Once reconstituted with bacteriostatic water, refrigerate at 2-8 degrees Celsius and use within 28 days. Never freeze reconstituted solution. See our tirzepatide storage guide for detailed protocols.
Is tirzepatide legal to purchase for research?
The legality depends on jurisdiction and intended use. Research peptides sold for laboratory and educational use occupy a different legal category than medications sold for human use. However, the enforcement environment has intensified significantly, and researchers should understand the legal landscape for peptides in their jurisdiction before purchasing.
External resources
New England Journal of Medicine: Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1)
PubMed Central: Tirzepatide, a New Era of Dual-Targeted Treatment for Diabetes and Obesity
For researchers serious about optimizing their peptide protocols and navigating the rapidly shifting vendor landscape, SeekPeptides offers the most comprehensive resource available. Members access evidence-based guides, detailed dosing charts, precision calculators, and a community of thousands who have navigated these exact vendor questions.
In case I do not see you, good afternoon, good evening, and good night. May your vendors stay transparent, your vials stay accurate, and your research stay uncompromised.