Feb 22, 2026

Before you draw up another dose, stop. Look at the liquid in your vial, pen, or syringe. What color do you see? Clear? Pink? Red? Something yellowish that was not there last week? The color of your semaglutide tells you more than you might realize. It tells you whether your medication is safe to inject, whether it has been stored properly, whether it contains additional ingredients, and whether you are holding the real thing or a counterfeit product that could put your health at serious risk.
This matters more than most people think.
The FDA has seized multiple batches of counterfeit Ozempic in recent years, and the demand for compounded semaglutide has created a marketplace where color variations are common but not always understood. Some colors are perfectly normal. Others are warning signs that something has gone wrong with your medication. And a few specific color changes mean you should discard the product immediately, no questions asked. Knowing the difference between a safe variation and a dangerous one could protect you from injecting degraded, contaminated, or counterfeit semaglutide into your body. This guide covers every form of semaglutide available, from FDA-approved injectable pens to oral formulations to compounded versions from specialty pharmacies. You will learn exactly what each form should look like, what the pen colors mean, why some compounded versions are pink or red, and which color changes demand immediate action. SeekPeptides created this reference so you never have to wonder whether what you are holding is safe to use.
What color should semaglutide be
The answer depends on which form of semaglutide you have. But the baseline is simple. FDA-approved injectable semaglutide, whether branded as Ozempic or Wegovy, should be clear and colorless. Always. No exceptions.
Think of it like water. The liquid inside your pen should look essentially identical to water in terms of color and clarity. It may be very slightly more viscous than water because of the inactive ingredients in the formulation, but your eyes should not detect any tint, hue, cloudiness, or floating particles when you hold the pen up to the light. If you see anything other than a perfectly transparent, colorless solution inside an Ozempic or Wegovy pen, something is wrong.
The FDA prescribing information for Ozempic describes the medication as "a sterile, aqueous, clear, and colorless solution." Wegovy carries the same description. These are not subjective standards. They are precise pharmaceutical specifications that every legitimate pen must meet before leaving the manufacturing facility.
Here is where it gets more nuanced. Compounded semaglutide from specialty pharmacies can look different. If your compounded formulation includes vitamin B12 (cyanocobalamin), the liquid may appear pink, light red, or even deep magenta depending on the B12 concentration. This is normal for that specific formulation. The B12 causes the color, not a problem with the semaglutide itself.
But a compounded semaglutide that does NOT list B12 on the label should still be clear and colorless. If it is not, contact your compounding pharmacy immediately before using it.
Research-grade semaglutide typically arrives as a white lyophilized powder that, when properly reconstituted, produces a clear and colorless solution. And oral semaglutide tablets, sold under the brand name Rybelsus, come as solid tablets with colors that vary by dose strength.
The bottom line is straightforward. Clear and colorless is the standard for injectable semaglutide. Any deviation from that standard needs an explanation, and if you cannot find one, do not inject.
Ozempic pen colors by dose
Ozempic uses a color-coded pen system to help patients and healthcare providers quickly identify which dose strength they are working with. The pen itself, the outer housing that you hold in your hand, comes in three distinct colors. But here is the critical distinction that confuses many people: the pen color refers to the packaging and housing, NOT the liquid inside.
The medication inside every Ozempic pen, regardless of pen color, is always clear and colorless.
Red pen (0.25 mg and 0.5 mg)
The red Ozempic pen is the starting pen for most patients. It contains 1.5 mL of semaglutide solution at a concentration that delivers either 0.25 mg or 0.5 mg per injection. Your prescriber will tell you which dose to dial. During the first four weeks, most patients use the red pen at the 0.25 mg setting, allowing the body to adjust to the medication. After that initial period, the dose typically increases to 0.5 mg, still using the same red pen. Each red pen delivers four doses total, so one pen lasts approximately four weeks when used once per week as prescribed.
The dose counter on the red pen shows numbers from 0 to either 0.25 or 0.5, depending on which dose you select. If you receive a pen with a dose counter that goes up to 80 or shows unusual numbering, that is a sign of a counterfeit product, and you should not use it.
Blue pen (1 mg)
The blue pen delivers the 1 mg dose of semaglutide. This is the first maintenance dose for many patients, prescribed after they have completed at least four weeks on the 0.5 mg dose. The blue pen also contains 1.5 mL of solution and delivers four weekly doses per pen. For patients whose blood sugar levels or weight loss goals require more aggressive treatment, the blue pen represents the middle tier of Ozempic dosing.
Again, the liquid inside is clear and colorless. The blue color is only on the pen housing.
Yellow pen (2 mg)
The yellow pen is the highest-dose Ozempic pen, delivering 2 mg of semaglutide per injection. It contains 3 mL of solution, twice the volume of the red and blue pens, and delivers four weekly doses per pen. Prescribers typically reserve the yellow pen for patients who need the maximum available Ozempic dose for optimal glycemic control.
The liquid inside the yellow pen is clear and colorless. Same as every other Ozempic pen.
Why pen color matters
The color-coding system exists for safety. It prevents dosing errors. Imagine accidentally using a 2 mg yellow pen when you are supposed to be on the 0.25 mg starter dose. That is a significant overdose. The distinct colors, red for starting doses, blue for medium, yellow for maximum, create an immediate visual check that helps patients confirm they are using the correct pen. If your pharmacy hands you a blue pen but your prescription says 0.25 mg, you know instantly that something does not match.
When examining your Ozempic pen, check the color of the housing AND the liquid. The housing should match your prescribed dose. The liquid should always be clear and colorless.
Wegovy pen identification by label color
Wegovy uses a different identification system than Ozempic. While both medications contain semaglutide made by Novo Nordisk, Wegovy is approved specifically for weight management and uses a five-tier dosing escalation with five different label colors to distinguish each dose level. Unlike Ozempic multi-dose pens, each Wegovy pen is a single-dose prefilled injector.
That is an important distinction. You do not dial a dose on a Wegovy pen. Each pen contains exactly one dose.
Green label (0.25 mg)
The green-labeled Wegovy pen contains 0.25 mg of semaglutide in 0.5 mL of solution. This is the starting dose. Patients typically use one green-labeled pen per week for the first four weeks as their body adjusts to the medication. The escalation schedule for Wegovy is gradual and deliberate, designed to minimize gastrointestinal side effects like constipation and nausea.
Pink label (0.5 mg)
After four weeks on the green pen, patients move to the pink-labeled pen delivering 0.5 mg of semaglutide in 0.5 mL. Another four weeks at this level before the next increase. The pink label is easy to spot and prevents confusion with the green starter dose.
Brown label (1 mg)
The brown-labeled pen delivers 1 mg of semaglutide in 0.5 mL. Patients reach this dose during weeks 9 through 12 of their titration schedule. Some patients experience the most noticeable appetite suppression at this dose level, though individual responses vary significantly.
Blue label (1.7 mg)
The blue-labeled Wegovy pen delivers 1.7 mg in 0.75 mL of solution. Note the slightly larger volume compared to the lower doses. Patients use this dose during weeks 13 through 16. By this point, many patients have experienced measurable weight loss results and are approaching the maintenance dose.
Black label (2.4 mg)
The black-labeled pen is the full maintenance dose, delivering 2.4 mg of semaglutide in 0.75 mL. This is the target dose for ongoing weight management. Clinical trials demonstrated the most significant weight loss at this dose level, and most patients remain on the black-labeled pen for the duration of their treatment.
The liquid inside every Wegovy pen
Regardless of label color, the liquid inside every legitimate Wegovy pen is clear and colorless. The white pen body is the same across all doses. Only the label color changes. If you receive a Wegovy pen and the liquid inside appears cloudy, tinted, or contains any visible particles, do not use it. Contact your pharmacy immediately. Legitimate Wegovy pens arrive sealed with a gray cap and should show no signs of tampering, cracking, or prior use.
For patients tracking their progress on semaglutide, understanding which pen you are using helps you correlate your results with your current dose level. Many people notice the most dramatic changes during the titration from 1 mg to 1.7 mg, though your experience may differ.
Rybelsus tablet colors and identification
Rybelsus is the oral tablet form of semaglutide, and it looks completely different from the injectable versions. Instead of a clear liquid, you are working with solid tablets that come in three dose strengths, each with its own visual characteristics. Knowing what your Rybelsus tablets should look like helps you verify authenticity and avoid taking the wrong dose.
3 mg tablets
The 3 mg Rybelsus tablet is white to light yellow in color. It is oval-shaped and film-coated, with the number "3" embossed on one side and the word "novo" on the other side. This is the starting dose. Patients typically take the 3 mg tablet once daily for the first 30 days before increasing. The tablet should be taken on an empty stomach with no more than 4 ounces (120 mL) of plain water, at least 30 minutes before any food, other beverages, or other oral medications.
7 mg tablets
The 7 mg Rybelsus tablet is also white to light yellow in color, oval-shaped, and film-coated. It has the number "7" embossed on one side and "novo" on the other. This is the first maintenance dose for many patients. For those exploring oral semaglutide options, the 7 mg tablet represents the dose where many patients begin experiencing meaningful appetite suppression effects.
14 mg tablets
The 14 mg Rybelsus tablet is white in color, oval-shaped, and film-coated. It has "14" on one side and "novo" on the other. This is the highest available oral semaglutide dose. The embossed numbers make it simple to verify you have the correct strength at a glance.
Key identification features for all Rybelsus tablets
All three strengths share the same oval shape and the "novo" embossing on one side. The dose number on the opposite side is the primary visual differentiator. The tablets should be kept in their original bottles with desiccant, not transferred to pill organizers, because moisture exposure degrades the medication. If you notice tablets that are crumbling, discolored beyond the normal white-to-light-yellow range, or missing their embossed markings, do not take them. Contact your pharmacy for replacements.
Understanding the visual differences between injectable and oral semaglutide helps you make informed decisions about which delivery method works best for your situation.
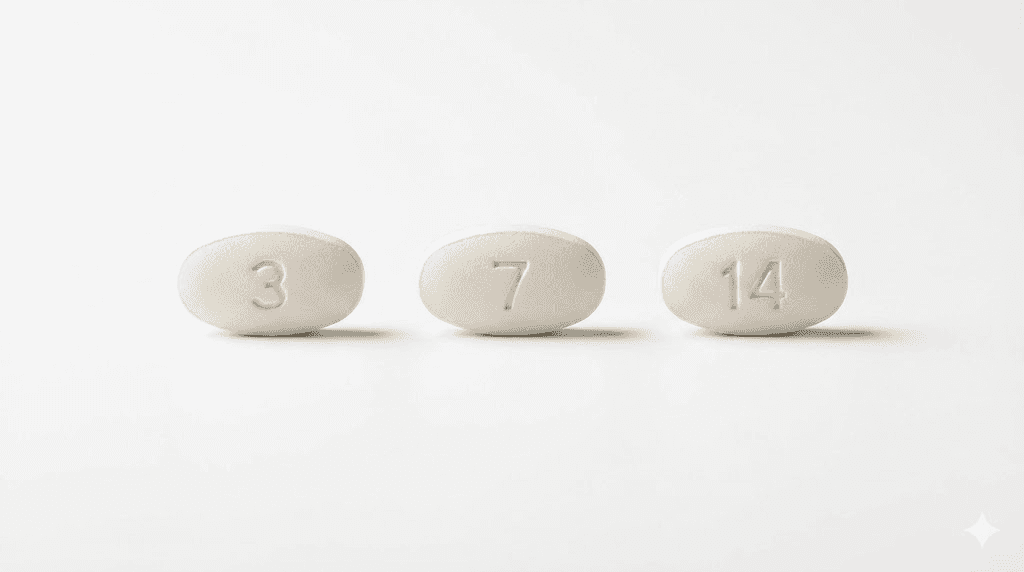
What color is compounded semaglutide
Compounded semaglutide is where the color question gets genuinely complicated. Unlike FDA-approved products that follow strict manufacturing standards and always produce a clear, colorless solution, compounded semaglutide can look quite different depending on what ingredients the pharmacy includes in the formulation.
The most important thing to understand: the semaglutide molecule itself is colorless when dissolved in solution. Any color you see comes from other ingredients in the compound, not from the semaglutide.
Standard compounded semaglutide (no additives)
A standard compounded semaglutide formulation without additional active ingredients should be clear and colorless, just like the brand-name versions. If your label lists only semaglutide (and standard inactive ingredients like bacteriostatic water, sodium chloride, or glycine), the solution should look like water. Any color in a formulation that should be colorless is a red flag.
Compounded semaglutide with B12 (cyanocobalamin)
This is the most common reason people see colored semaglutide. When a compounding pharmacy adds vitamin B12 (cyanocobalamin) to the semaglutide formulation, the liquid turns pink to red. The intensity of the color depends on how much B12 the pharmacy adds. Lower concentrations produce a light pink or salmon color. Higher concentrations create a deeper red or magenta.
Cyanocobalamin is naturally a deep red compound. Even small amounts added to a clear solution produce a visible tint. This is completely normal and does not affect the efficacy of the semaglutide. Many compounding pharmacies add B12 because it may help with energy levels and because some research suggests it supports overall metabolic health during weight loss.
If your label says "semaglutide with B12" or "semaglutide with cyanocobalamin" and the liquid is pink or red, everything is working as expected. The color is actually reassuring because it confirms the B12 was properly mixed into the formulation.
Compounded semaglutide with methylcobalamin
Methylcobalamin is a different form of vitamin B12. It also produces a pink or red color when added to semaglutide formulations. The shade is similar to cyanocobalamin-containing formulations, with the intensity varying based on concentration. Some patients and providers prefer methylcobalamin because it is the active, bioavailable form of B12 that the body can use directly without conversion.
The visual appearance of methylcobalamin-containing and cyanocobalamin-containing compounded semaglutide is essentially identical. You cannot reliably distinguish between them by color alone.
Compounded semaglutide with glycine
Glycine is an amino acid that some compounding pharmacies add as a stabilizer. It does not add any color to the solution. A semaglutide-glycine compound should be clear and colorless. If glycine AND B12 are both present, the solution will show the characteristic pink or red color from the B12.
Lyophilized (freeze-dried) compounded semaglutide
Some compounding pharmacies and research suppliers provide semaglutide as a white lyophilized powder that must be reconstituted before use. The powder itself should be white to off-white in appearance. Once properly reconstituted with bacteriostatic water or sterile water, the resulting solution should be clear and colorless with no visible particles.
If the powder appears yellow, brown, or any color other than white, it may have degraded. If the reconstituted solution is cloudy or contains particles that do not dissolve after gentle swirling, do not use it. Proper reconstitution technique matters. Let the water flow slowly down the inner wall of the vial rather than spraying it directly onto the powder, which can cause foaming and denaturation of the peptide.
Why is my semaglutide red or pink
You pulled your semaglutide out of the refrigerator and the liquid is pink. Or red. Or somewhere in between. Should you panic?
Probably not. But you should investigate.
The most common reason for pink or red semaglutide is the addition of vitamin B12 to the formulation. This is by far the most likely explanation, and it applies exclusively to compounded semaglutide from specialty pharmacies. FDA-approved Ozempic and Wegovy should NEVER be pink or red.
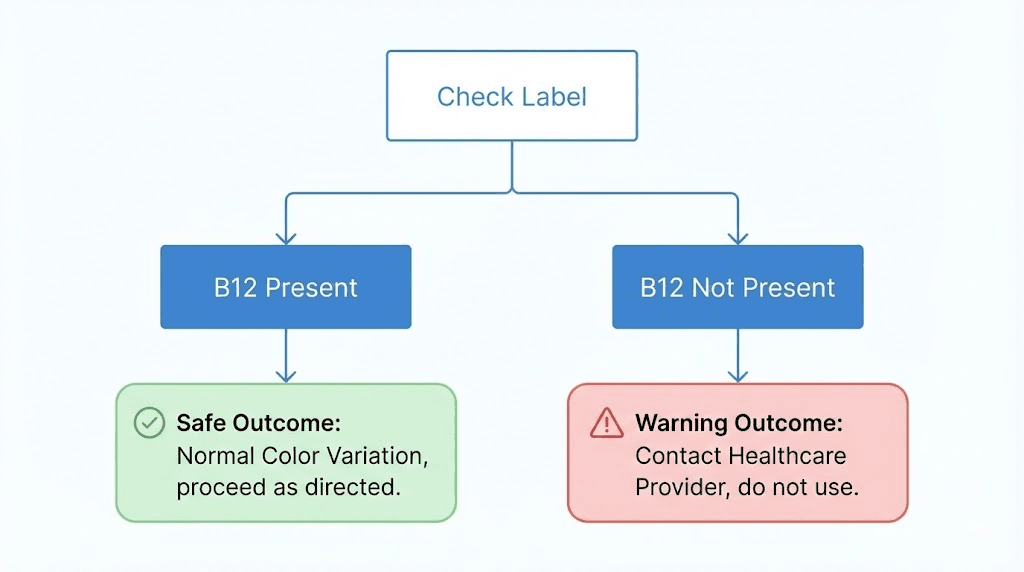
Step 1: Check your label
Look at the label on your vial or syringe. Does it mention any of the following?
Cyanocobalamin (a form of vitamin B12)
Methylcobalamin (another form of vitamin B12)
Vitamin B12 or B12
If any of these appear on your label, the pink or red color is expected and normal. The B12 is causing the color. Your semaglutide is likely fine.
Step 2: Assess the shade
B12-containing semaglutide typically ranges from light pink to deep red or magenta. The exact shade depends on the B12 concentration. A light pink with low-concentration B12. A deeper red with higher concentrations. The color should be uniform throughout the solution, not streaky or layered. And the solution should still be clear, meaning you can see through it even though it has color. Clarity and color are separate qualities. A solution can be colored (pink) and still clear (transparent, no cloudiness).
Step 3: If your label does NOT mention B12
This is when you should be concerned. If your semaglutide label lists only semaglutide (with or without glycine and other stabilizers) but the liquid is pink or red, do not inject it. Contact the compounding pharmacy that produced it. Ask them to verify the ingredients. There may be a labeling error, or there may be a contamination issue. Either way, do not use it until you have confirmation from the pharmacy about what is actually in the vial.
Step 4: If you have brand-name Ozempic or Wegovy
If your brand-name Ozempic or Wegovy pen contains pink or red liquid, do NOT use it. Period. These products should never contain B12 or any other color-producing additive. Pink or red liquid in an Ozempic or Wegovy pen may indicate contamination, counterfeiting, or product tampering. Contact your pharmacy and report it to the FDA MedWatch program.
For those using compounded formulations, SeekPeptides provides comprehensive guides on understanding different pharmacy formulations, ingredient lists, and what to expect from various compounded products.
Semaglutide color changes that signal danger
Some color changes are harmless. The pink of B12 is one example. But other color changes indicate that your semaglutide has degraded, been contaminated, or been improperly stored, and using it could be ineffective at best and dangerous at worst.
Here are the warning signs you should never ignore.
Yellow or brown discoloration
If your semaglutide solution has turned yellow, amber, or brown, it has almost certainly undergone chemical degradation. This typically happens when the medication is exposed to heat or light over an extended period. The peptide bonds in the semaglutide molecule break down, creating degradation products that absorb light at different wavelengths, producing the visible color change.
Yellow or brown semaglutide is no longer effective. The active ingredient has partially or fully broken down. But the bigger concern is safety. Degradation products are unpredictable. They may be inert, or they may cause adverse reactions. There is no way to know without laboratory testing, which is obviously not practical for individual patients.
If your semaglutide has turned yellow or brown, discard it. This can happen when medication is exposed to excessive heat, left in direct sunlight, or stored beyond its expiration date. Understanding how long semaglutide lasts helps prevent this scenario entirely.
Cloudy or turbid appearance
Cloudiness in semaglutide is a serious warning sign. A solution that was previously clear but has become hazy, milky, or opaque may be experiencing protein aggregation, bacterial contamination, or chemical precipitation. None of these are safe to inject.
Protein aggregation occurs when the semaglutide molecules clump together, forming particles too small to see individually but large enough to scatter light and create a cloudy appearance. This can happen due to agitation (shaking the pen roughly), temperature extremes, or contamination. Aggregated proteins can trigger immune responses when injected.
Bacterial contamination produces cloudiness as the microorganisms multiply in the solution. Injecting contaminated medication can cause infection at the injection site or, in severe cases, systemic infection. This is especially concerning with multi-use vials of compounded semaglutide that are accessed with a needle multiple times.
Do not inject cloudy semaglutide. Ever.
Visible particles or debris
Hold your vial or pen up to a light source and look carefully. Do you see any floating specks, fibers, crystals, or sediment? Any visible particulate matter means the product should not be used. Particles can form from crystallization of the medication, precipitation of inactive ingredients, contamination from the rubber stopper, or introduction of foreign material during use.
Before every injection, perform a quick visual inspection. This takes five seconds and could prevent a serious problem. If you spot particles, discard the product and contact your pharmacy for a replacement.
Frozen and thawed semaglutide
Semaglutide should never be frozen. If your medication accidentally freezes, whether in a refrigerator set too cold, during shipping, or during travel, the freeze-thaw cycle damages the peptide structure. Frozen semaglutide may appear cloudy or contain particles after thawing, but it can also look normal while being structurally compromised.
The conservative approach: if you suspect your semaglutide froze, even if it looks fine after thawing, discard it. The structural damage from freezing is not always visible, but it can reduce efficacy and potentially cause adverse reactions from aggregated proteins.
Color change summary table
Here is a quick reference for evaluating semaglutide color changes:
Clear and colorless = Normal for brand-name and standard compounded semaglutide
Pink to red = Normal ONLY if label lists B12, cyanocobalamin, or methylcobalamin
Yellow or brown = Degradation from heat or light exposure, do not use
Cloudy or milky = Contamination or protein aggregation, do not use
Contains particles = Crystallization or contamination, do not use
Any unexpected color = Contact pharmacy before using
When in doubt, do not inject. The cost of replacing a vial or pen is nothing compared to the risk of injecting degraded or contaminated medication. Patients tracking their results on semaglutide should know that color changes during a treatment cycle can explain unexpected plateaus or lack of weight loss, because degraded medication simply does not work as intended.
How storage affects semaglutide appearance
Proper storage is the single biggest factor in maintaining your semaglutide color, clarity, and potency. The medication is a biological molecule, a peptide, and peptides are sensitive to temperature, light, and physical agitation. Understanding storage requirements does not just preserve your medication. It protects your investment and your health.
Refrigeration requirements
Before first use, all injectable semaglutide products should be stored in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). This applies to Ozempic, Wegovy, and compounded semaglutide alike. The refrigerator provides a stable, cool, dark environment that slows chemical degradation and prevents microbial growth.
Store your semaglutide in the main body of the refrigerator, not in the door (temperature fluctuates too much), not in the back near the cooling element (risk of accidental freezing), and not in a produce drawer (variable humidity). The middle shelf is ideal. Keep it in the original packaging or box to protect from light.
Room temperature allowances
After you begin using a pen, both Ozempic and Wegovy can be stored at room temperature for a limited time. The specifics differ between products.
Ozempic: After first use, the pen can be stored at room temperature (59 to 86 degrees Fahrenheit, 15 to 30 degrees Celsius) or in the refrigerator for up to 56 days. After 56 days, discard the pen regardless of how much medication remains. This is one of the most generous room-temperature allowances among injectable peptide medications.
Wegovy: After first use, pens can be stored at room temperature (46 to 86 degrees Fahrenheit, 8 to 30 degrees Celsius) for up to 28 days. After 28 days, discard the pen. The shorter window compared to Ozempic means patients need to be more diligent about tracking when they opened their Wegovy pen. Knowing whether semaglutide expires after 28 days and understanding the specifics of using expired semaglutide is essential for anyone on this medication.
Compounded semaglutide shelf life varies by pharmacy and formulation. Some compounded products have a shelf life of 30 days, others up to 90 days. Always follow the beyond-use date (BUD) printed on the label from your compounding pharmacy.
Heat exposure and color changes
Heat is the enemy of semaglutide. When the medication is exposed to temperatures above the recommended range, the peptide structure begins to break down. This degradation is not instantaneous. Brief exposure to slightly elevated temperatures (a few minutes at 90 degrees Fahrenheit while transferring between locations) is unlikely to cause noticeable damage. But prolonged exposure to high heat, such as leaving a pen in a hot car for several hours, can cause irreversible degradation.
The visible signs of heat damage include yellowing of the solution, cloudiness, and particle formation. But here is the tricky part: early-stage degradation may not produce any visible changes. The medication can lose potency before it looks any different. This is why following storage guidelines matters, not just inspecting the solution visually.
For patients who travel with semaglutide, using an insulated medical travel case with a cool pack keeps the medication within the safe temperature range. Avoid checking semaglutide in airline luggage, where cargo holds can reach extreme temperatures.
Light exposure
Light, especially direct sunlight and fluorescent lighting, can accelerate the degradation of semaglutide. This is why the medication comes in opaque packaging and why pens are designed with light-blocking materials. Store your semaglutide away from windows, light sources, and areas where it might receive prolonged light exposure.
If you keep your pen on a bathroom counter or kitchen shelf where it sits in ambient light for hours each day, consider moving it to a drawer or cabinet. Even room-temperature storage works fine for semaglutide within the allowed timeframe, but minimizing light exposure adds an extra layer of protection.
Never freeze semaglutide
This point deserves special emphasis. Never freeze semaglutide. Freezing causes ice crystal formation within the solution, and those crystals physically damage the peptide molecules. When the solution thaws, the damaged molecules may aggregate, losing their biological activity. Frozen semaglutide cannot be "rescued" by thawing. The damage is permanent.
Check your refrigerator temperature regularly. Many home refrigerators have cold spots, particularly near the back wall or cooling element, where temperatures can dip below freezing. If your semaglutide has formed ice crystals, even partially, discard it.

How to verify your semaglutide is authentic
The surge in demand for semaglutide has created a parallel surge in counterfeit products. The FDA has issued multiple warnings about fake Ozempic and Wegovy pens entering the United States drug supply chain. Some of these counterfeits look remarkably convincing at first glance. But knowing what to look for can help you identify fakes before you inject them.
Packaging inspection
Start with the outer box. Genuine Ozempic and Wegovy packaging has specific features that counterfeiters often get wrong.
Look for dents, tears, or scratches on the outer carton. While minor shipping damage can happen to legitimate products, significant packaging damage can indicate the product was repackaged or tampered with. Check for misspellings on the label. Counterfeit products frequently have subtle spelling errors, incorrect capitalization, or grammatical mistakes in the prescribing information and label text.
Examine label alignment. Genuine Novo Nordisk labels are precisely aligned, with consistent spacing and straight edges. Misaligned or crooked labels suggest a counterfeit product. The font should match the official Novo Nordisk typography. If the lettering looks different from previous genuine pens you have used, be suspicious.
Lot number and expiration date verification
The FDA has identified specific counterfeit lot numbers in past seizures. For example, counterfeit Ozempic products were found bearing lot number NAR0074 and lot number PAR0362. While counterfeiters change lot numbers regularly, checking the FDA website for currently known counterfeit lot numbers is a simple precaution. The FDA MedWatch page maintains updated lists of recalled and counterfeit pharmaceutical products.
On genuine Ozempic pens, the format of the lot number and expiration date follows a consistent layout. In one identified counterfeit batch, the "EXP/LOT" text was positioned to the left of the date and lot number on the pen label instead of above it, which is the correct position on genuine products. These small formatting details can reveal counterfeits.
Pen mechanical verification
The dose counter on a genuine Ozempic pen operates smoothly and clicks distinctly with each dose increment. The red pen dose counter goes from 0 to 0.25 or 0.5. The blue pen goes to 1. The yellow pen goes to 2. If your pen dose counter displays numbers higher than these (some counterfeits have been found with dose counters going up to 80), the product is fake.
The pen cap should fit securely and click when attached. The injection button should depress smoothly and spring back. The needle attachment point should accept standard pen needles without wobbling. Any mechanical irregularity should raise concern.
The liquid itself
As emphasized throughout this guide, the liquid inside any genuine Ozempic or Wegovy pen must be clear and colorless. Hold the pen up to light. Tilt it gently. Look for any color, cloudiness, or particles. If you see anything other than a crystal-clear, water-like solution, do not use it.
Source verification
The single most effective way to avoid counterfeit semaglutide is to obtain your medication from a licensed, verified pharmacy. This means brick-and-mortar pharmacies, verified online pharmacies through your insurance network, or legitimate compounding pharmacies that are licensed by state boards of pharmacy.
Be extremely cautious about online sellers offering semaglutide at prices that seem too good to be true. The FDA has found that many online sellers market counterfeit or substandard semaglutide products. A study published in the Journal of Medical Internet Research analyzed semaglutide products sold by online sellers without prescriptions and found significant quality and safety concerns across multiple products tested.
For those researching their options, SeekPeptides provides detailed guides on verified semaglutide providers including Empower Pharmacy, Olympia Pharmacy, and BPI Labs to help you make informed decisions about sourcing.
What to do if you suspect a counterfeit
If you believe you have received counterfeit semaglutide, take these steps immediately. Do not inject the product. Photograph the product, packaging, and labels from multiple angles. Contact the pharmacy where you obtained it. Report the product to the FDA MedWatch program at www.fda.gov/medwatch or by calling 1-800-FDA-1088. If you already injected the product and experience any unusual symptoms, contact your healthcare provider immediately.
Research peptide semaglutide appearance
Research-grade semaglutide follows different manufacturing and packaging conventions than pharmaceutical products. If you are working with research-grade semaglutide, understanding its expected appearance helps you assess quality and proper handling.
Lyophilized powder form
Most research-grade semaglutide arrives as a lyophilized (freeze-dried) powder. The lyophilization process removes water from the peptide solution while preserving the molecular structure, creating a stable powder that can be stored at controlled temperatures for extended periods.
The powder should appear white to off-white in color. It typically looks like a small, fluffy cake or plug at the bottom of the vial. Some suppliers press the lyophilized material into a compact disc or pellet, while others leave it as a loose powder. Both presentations are normal. The key visual indicator of quality is the color. White to off-white is good. Yellow, brown, or darkened powder may indicate degradation during manufacturing, shipping, or storage.
Reconstitution process and expected appearance
When you reconstitute lyophilized semaglutide, the result should be a clear, colorless solution with no visible particles. The reconstitution technique matters significantly for achieving this result.
Add your chosen diluent, typically bacteriostatic water or sterile water, by directing the stream down the inner wall of the vial. Do not spray liquid directly onto the powder. Do not shake the vial vigorously. Instead, gently swirl or roll the vial between your palms until the powder is fully dissolved. Aggressive shaking can cause foaming and denature the peptide, creating aggregates that may appear as particles or cloudiness.
If you have properly reconstituted the semaglutide and see undissolved particles, let the vial sit in the refrigerator for 15 to 30 minutes and then gently swirl again. If particles persist, the powder may have degraded before reconstitution. A sterile syringe filter can remove visible particles, but this does not address underlying degradation. Using the peptide reconstitution calculator helps ensure you add the correct volume of diluent for your desired concentration.
For those working with different vial sizes, guides on reconstituting 10 mg semaglutide and 5 mg semaglutide provide specific volume recommendations for common concentration targets.
Storage after reconstitution
Once reconstituted, research-grade semaglutide should be stored at 2 to 8 degrees Celsius (standard refrigeration) and used within 30 days for most formulations reconstituted with bacteriostatic water. Sterile water reconstitutions have shorter stability windows, typically 7 to 14 days, because they lack the antimicrobial preservative (benzyl alcohol) found in bacteriostatic water.
For long-term storage of reconstituted semaglutide (if not used within the standard window), some researchers add a carrier protein such as bovine serum albumin (BSA) at 0.1% to prevent adsorption of the peptide to the glass vial walls. However, this is primarily relevant for very dilute solutions used in laboratory research rather than typical reconstitution scenarios.
Monitor the appearance of your reconstituted semaglutide at each use. If it was clear when you reconstituted it but has become cloudy or developed particles during storage, the peptide has degraded and should be discarded. Gradual changes in appearance over days or weeks are a normal signal that the reconstituted solution is reaching the end of its useful life.
Semaglutide vs tirzepatide appearance comparison
With both semaglutide and tirzepatide widely used for weight management and diabetes, patients sometimes need to visually distinguish between these medications. While they serve similar purposes, they have distinct visual characteristics that make identification straightforward once you know what to look for.
Injectable appearance comparison
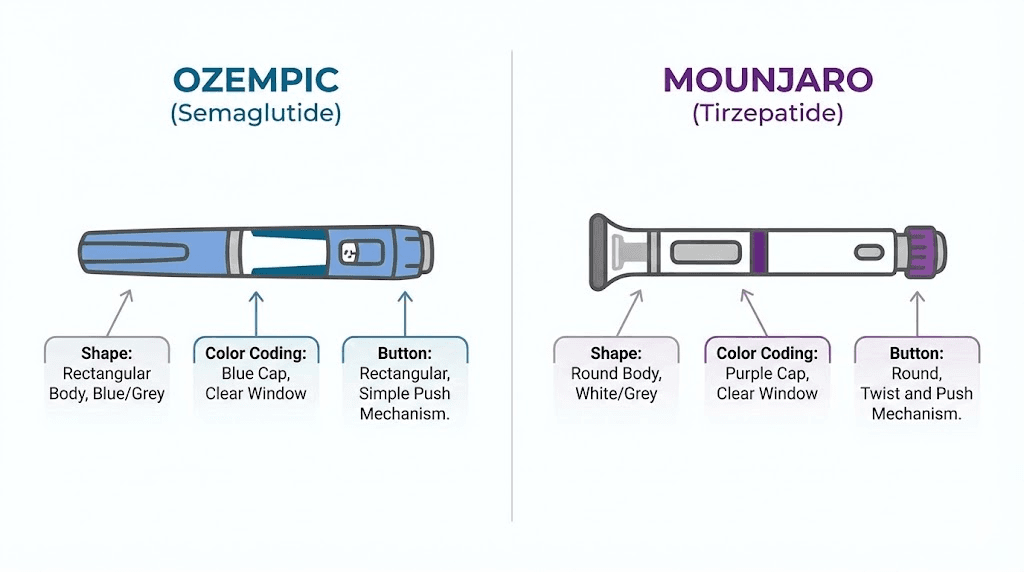
Both medications, in their FDA-approved injectable forms, appear as clear solutions when examined in the pen. However, there is a subtle but important difference. Semaglutide (Ozempic, Wegovy) is described as "clear and colorless." Tirzepatide (Mounjaro, Zepbound) is described as "clear, colorless to slightly yellow." That "slightly yellow" qualifier is significant. A very faint yellow tint in tirzepatide is within the normal specification. The same faint yellow in semaglutide would be a warning sign of degradation.
This distinction matters most for patients who switch between medications or use both in sequence. A semaglutide to tirzepatide conversion requires understanding both medications' visual profiles. Do not assume that because one medication looks slightly different from another, something is wrong. They are different compounds with different specifications.
Pen identification differences
Ozempic pens use the red, blue, yellow color system described earlier. Mounjaro (tirzepatide) uses a completely different color scheme with more dose levels and more colors. Mounjaro pens share a common design, a cylindrical body with a gray triangular safety cover on one end and a purple injection button on the other. The dose-specific coloring appears on the label and cap area.
Mounjaro dose colors include gray (2.5 mg), maroon/dark red (5 mg), green (7.5 mg), tan (10 mg), and purple (12.5 mg), with additional dose strengths available. Understanding the tirzepatide dosing system and the tirzepatide dose chart helps patients track their progress through the titration schedule.
Wegovy pens use the green, pink, brown, blue, black label system. Zepbound (tirzepatide for weight loss) follows the same color scheme as Mounjaro. The key takeaway: these are completely different color systems, and a Wegovy pen label color means something entirely different from a Mounjaro pen label color.
Compounded versions comparison
Compounded semaglutide and compounded tirzepatide can look similar or identical depending on the formulation. Both are clear and colorless without additives. Both turn pink or red when B12 is added. You cannot reliably distinguish between compounded semaglutide and compounded tirzepatide by appearance alone. Label verification is the only reliable method.
For patients comparing these medications, understanding their side effect profiles, the dosage comparison chart, and the overall comparison between semaglutide and tirzepatide provides much more actionable information than visual inspection. Those considering a change in medication can review guidance on switching between these medications.
Research peptide form comparison
In lyophilized powder form, both semaglutide and tirzepatide appear as white to off-white powders. Their reconstituted forms are both clear and colorless (when properly prepared). Without labeling, you cannot visually distinguish between reconstituted semaglutide and reconstituted tirzepatide. This underscores the importance of proper labeling, storage, and handling protocols for anyone working with research peptides.
The broader landscape of GLP-1 peptide medications is evolving rapidly, with newer compounds like retatrutide and mazdutide entering the picture. Each has its own visual characteristics and identification requirements.
Beyond color: what to know once you start semaglutide
Confirming your semaglutide looks right is the first step. But once you begin treatment, a whole world of practical questions opens up. What should you eat? When should you inject? What side effects are normal? The color of your medication matters for safety. Everything else matters for results.
Most patients starting semaglutide wonder how quickly it works. The timeline varies significantly. Some people notice appetite suppression within the first week. Others take several weeks. The medication builds up in your system gradually, which is why the dose escalation schedule exists. Patience during those early weeks is critical.
Food choices play a major role in how well you tolerate the medication. Knowing which foods to avoid on semaglutide and having a clear semaglutide diet plan can dramatically reduce side effects like nausea and bloating. A solid list of recommended foods makes meal planning simpler during the adjustment period.
Side effects are part of the process for most people. Fatigue hits some patients hard in the first few weeks. Constipation is common and manageable. Excessive burping catches people off guard. Dizziness can occur, especially at higher doses. Even menstrual cycle changes have been reported. Understanding these possibilities helps you prepare rather than panic.
Some patients wonder about combining semaglutide with other medications. Whether phentermine can be taken alongside semaglutide is a common question, as is the comparison between phentermine and semaglutide for weight loss effectiveness. The relationship between semaglutide and GLP-1 receptor agonists as a drug class is worth understanding too, especially as newer medications like GLP-1 patches become available.
Lifestyle factors influence outcomes significantly. The question of whether you can drink alcohol on semaglutide comes up constantly. So does whether it is possible to lose weight on semaglutide without exercise. The honest answer to both involves nuance that goes beyond a simple yes or no.
Injection technique matters more than most people realize. Choosing the best injection site and picking the optimal time of day to inject can reduce side effects and improve consistency. For those using compounded semaglutide with syringes rather than prefilled pens, mastering proper injection technique and understanding GLP-1 injection fundamentals prevents common dosing errors.
Results tracking keeps you motivated and helps identify problems early. Knowing what before and after semaglutide results typically look like sets realistic expectations. If you hit a plateau or experience no weight loss after four weeks, that is not necessarily a failure. It may signal the need for a dose adjustment, a dietary change, or an investigation into whether your medication has been stored properly, which brings us right back to color and appearance.
For those considering the longer view, understanding how long you stay on semaglutide and what withdrawal symptoms might occur when stopping helps with treatment planning. The timeline for appetite suppression and the question of whether semaglutide gives you energy or drains it factor into quality-of-life decisions during treatment.
Every one of these factors connects back to the fundamental question this guide answers: is my medication safe to use? A clear, properly colored semaglutide solution that has been stored correctly and comes from a verified source gives you the foundation for everything else. Without that foundation, none of the other optimizations matter.
Practical tips for visual inspection before every injection
Making a visual inspection part of your injection routine takes less than ten seconds and could prevent serious problems. Here is a simple process to follow every time you prepare your semaglutide injection.
The five-point visual check
First, check the color. Is the solution the color you expect based on your formulation? Clear and colorless for brand-name products and standard compounded semaglutide. Pink to red for B12-containing compounds. Any other color is a potential problem.
Second, check clarity. Hold the vial or pen up to a light source. Can you see through the liquid clearly, like looking through a window? Or is there any haziness, cloudiness, or milky appearance? Even slight cloudiness is a reason to stop and investigate.
Third, check for particles. With the light source behind the vial or pen, look carefully for any floating specks, fibers, crystals, or sediment. Gently tilt the vial or pen to see if anything settles or floats. Particles of any kind mean do not inject.
Fourth, check the container. Is the vial intact with no cracks? Is the rubber stopper properly seated? For pens, is the cap secure, and does the pen mechanism function properly? For syringe preparation, is the syringe package sealed and the needle sterile?
Fifth, check the date. Is the medication within its expiration date or beyond-use date? For opened products, is it within the allowed room-temperature window (56 days for Ozempic, 28 days for Wegovy)?
These five checks become second nature after a few injections. They protect you from using compromised medication and give you confidence that what you are injecting is safe and effective.
When to contact your pharmacy
Contact your pharmacy if you observe any of the following: an unexpected color change, cloudiness that was not present when you first opened the product, particles or debris in the solution, a damaged pen or vial, a product that appears to have been tampered with or previously opened, or any discrepancy between the product appearance and what you have received previously.
Most pharmacies will replace compromised medications, especially if you are within the dispensing period. Document any visual anomalies with photographs before contacting them, as this helps them investigate potential batch issues.
Finding the best injection approach
Beyond visual inspection, proper injection technique affects your results. The best injection site for semaglutide varies by individual anatomy and preference, with the abdomen, thigh, and upper arm being the standard options. Rotating injection sites helps prevent lipodystrophy, where repeated injections in the same area cause changes in the fat tissue. Understanding the best time of day to take semaglutide also plays a role in optimizing your treatment.
Patients who combine their semaglutide with other medications should be aware of potential interactions. Guides on combining phentermine and semaglutide and the comparison between phentermine and semaglutide provide important safety information for those considering combination approaches.
Understanding semaglutide formulation ingredients and their visual effects
Knowing why your semaglutide looks the way it does requires understanding the ingredients that go into each formulation. The active ingredient, semaglutide itself, is colorless in solution. Everything else you see comes from the inactive ingredients, excipients, and additional active compounds that pharmacies include for various purposes.
Ozempic inactive ingredients
Ozempic contains disodium phosphate dihydrate (buffer), propylene glycol (solvent), phenol (preservative), and water for injection. None of these ingredients have any visible color. They contribute to the solution stability, pH, and antimicrobial properties but do not alter the clear, colorless appearance. Hydrochloric acid or sodium hydroxide may be added during manufacturing to adjust pH, again without any color effect.
Wegovy inactive ingredients
Wegovy contains a similar excipient profile, optimized for the higher concentrations used in weight management dosing. The preservatives and buffers keep the solution stable during the 28-day room-temperature window while maintaining the clear, colorless appearance that patients should expect.
Why compounding pharmacies add B12
Compounding pharmacies frequently add vitamin B12 to semaglutide for several reasons. B12 deficiency is common in the general population, and weight loss medications can potentially affect nutrient absorption. B12 supports energy metabolism, which may help counteract the fatigue that some patients experience on semaglutide. B12 also supports neurological function and red blood cell production.
From a practical standpoint, combining B12 with semaglutide in a single injection reduces the number of injections a patient needs. Instead of getting a semaglutide injection plus a separate B12 injection, patients receive both in one shot. This convenience factor drives much of the popularity of semaglutide with B12 formulations.
The color change from B12 actually serves an unintentional benefit: it provides visible confirmation that the compound was properly mixed. If a pharmacy claims to have added B12 but the solution is colorless, that would be a legitimate reason to question whether the formulation was prepared correctly.
The role of glycine in compounded formulations
Glycine serves as a stabilizer in many compounded semaglutide formulations. This amino acid helps maintain the peptide structure during storage and can improve the overall stability profile of the compound. Glycine is colorless in solution and does not affect the visual appearance of the medication.
Some pharmacies include glycine alongside B12, creating a formulation that is pink or red (from the B12) but benefits from improved stability (from the glycine). The glycine and B12 compound approach has become increasingly popular among compounding pharmacies serving the weight management market.
Bacteriostatic water vs sterile water
The diluent used in compounded semaglutide also matters, though it does not affect color. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, allowing multi-use vial access over an extended period. Sterile water contains no preservatives and should be used for single-use preparations only.
Both diluents are clear and colorless. Neither will change the appearance of your semaglutide solution. However, the preservative in bacteriostatic water provides antimicrobial protection that helps prevent the bacterial growth that can cause cloudiness in multi-dose vials over time. This is why most compounding pharmacies use bacteriostatic water for semaglutide preparations intended for multiple draws from the same vial.
Special considerations for different semaglutide delivery methods
Semaglutide comes in several delivery formats beyond the standard prefilled pen and compounded vial. Each format has its own visual characteristics worth understanding.
Oral semaglutide drops
Oral semaglutide drops represent a newer delivery method. These liquid formulations are designed for oral or sublingual administration. The appearance can vary depending on the formulation, but most are clear liquids, sometimes with a slight color depending on additional ingredients or flavorings. Always compare the appearance of your oral drops to the description on the label and contact the pharmacy if there are discrepancies.
Semaglutide in GLP-1 combination products
The GLP-1 medication category continues to expand, with new formulations and combination products entering the market. Some of these products combine semaglutide or other GLP-1 agonists with additional active ingredients. Products like GLP-1 patches have entirely different visual presentations. Each product type has its own expected appearance, and patients should familiarize themselves with what their specific product should look like.
For injectable GLP-1 medications in general, the injection technique and injection site selection remain consistent regardless of the specific product. Visual inspection before injection is a universal best practice.
Managing side effects and what color does not tell you
While visual inspection tells you about the physical condition of your medication, it does not tell you everything about how the medication will affect you. Side effects like constipation, burping, dizziness, and fatigue can occur even when the medication looks perfectly normal. Similarly, effects on menstrual cycles and energy levels are related to the pharmacological action of the drug, not its appearance.
If you are experiencing side effects, the color and clarity of your semaglutide are unlikely to be the cause, assuming the medication passes your visual inspection. Discuss side effects with your healthcare provider to determine whether dose adjustments are needed. Understanding the withdrawal symptoms if you discontinue semaglutide is also worth reviewing with your provider.
Diet, lifestyle, and maximizing semaglutide results
Now that you know exactly what your semaglutide should look like, let us briefly touch on maximizing the results from your properly stored, visually verified medication. Color and clarity tell you the medication is physically intact. But getting the best outcomes requires more than just injecting a good product.
Following a structured diet plan while on semaglutide dramatically improves outcomes compared to medication alone. Understanding which foods to eat on semaglutide and which foods to avoid helps you work with the medication rather than against it. Many patients find that semaglutide changes their food preferences naturally, reducing cravings for highly processed foods while increasing tolerance for protein-rich meals.
For patients who feel their progress has stalled, understanding the semaglutide plateau phenomenon and investigating reasons for not losing weight on semaglutide can reveal adjustable factors. Some patients report four weeks on semaglutide with no weight loss during the initial dose-titration period, which is often normal and resolves as the dose increases.
Whether you can lose weight on semaglutide without exercise is a common question. The clinical data shows that weight loss occurs with semaglutide alone, but combining the medication with physical activity improves body composition, preserves muscle mass, and produces better long-term outcomes. Reviewing before and after semaglutide results from real patients provides realistic expectations.
Questions about alcohol and semaglutide are also frequent. Whether you can drink on semaglutide depends on several factors, including your individual health status, the amount consumed, and your tolerance level. Most providers recommend moderation and awareness that alcohol can affect blood sugar levels and gastrointestinal comfort.
SeekPeptides members access comprehensive protocol guides that go beyond medication appearance, covering optimized dosing schedules, dietary frameworks, exercise integration, and side effect management strategies. For researchers serious about maximizing outcomes, having a structured approach alongside visual medication verification creates the foundation for consistent, measurable results.
Unit conversions and dosage reference
Understanding what your semaglutide looks like is one piece of the puzzle. Knowing your exact dose is another. Many patients, especially those using compounded semaglutide with insulin syringes rather than prefilled pens, need to convert between milligrams and units. This is where dosing errors commonly occur, and the semaglutide dosage calculator becomes an invaluable tool.
Common conversion questions include how many milligrams are in 40 units of semaglutide, 50 units, or 100 units. The answer depends entirely on the concentration of your specific formulation. A 5 mg/mL solution yields very different milligram-per-unit values than a 2.5 mg/mL solution. Always verify your concentration before drawing a dose.
For patients using specific dose targets, understanding conversions like 10 units in milligrams, 20 units in milligrams, or 20 units in milliliters helps ensure accurate dosing. The question of whether 20 units is too much also depends on your concentration and prescribed dose.
Wegovy patients transitioning from prefilled pens to compounded semaglutide often need to know conversions like how many units equal 1.7 mg or how many units equal 2.4 mg. The peptide calculator and peptide cost calculator provide quick, accurate answers for these common conversion questions.
Frequently asked questions
What color should Ozempic liquid be?
Ozempic liquid should always be clear and colorless, regardless of which pen color (red, blue, or yellow) you are using. The pen color identifies the dose, not the medication color. If the liquid inside your Ozempic pen has any tint, cloudiness, or visible particles, do not use it and contact your pharmacy immediately. Learn more about Ozempic dosing to understand the relationship between pen color and dose strength.
Is it normal for compounded semaglutide to be pink?
Yes, if your label lists vitamin B12, cyanocobalamin, or methylcobalamin as an ingredient. The B12 causes the pink to red color, and it does not affect the semaglutide efficacy. If your label does NOT mention B12 but the solution is pink, contact your compounding pharmacy to verify the formulation before using it.
Can I use semaglutide that has turned yellow?
No. Yellow discoloration in semaglutide indicates chemical degradation, typically from heat exposure or improper storage. Degraded semaglutide may be ineffective and could potentially cause adverse reactions. Discard it and obtain a fresh supply. Understanding how long semaglutide remains viable helps prevent this situation.
What do Wegovy pens look like?
Wegovy pens are white single-dose prefilled injectors with color-coded labels: green (0.25 mg), pink (0.5 mg), brown (1 mg), blue (1.7 mg), and black (2.4 mg). Each pen has a gray cap. The liquid inside every Wegovy pen should be clear and colorless regardless of the label color.
How can I tell if my semaglutide has gone bad?
Look for these warning signs: any unexpected color change (especially yellow, brown, or amber), cloudiness or haziness, visible particles or debris floating in the solution, or a broken seal on the pen or vial. Also check the expiration date and, for opened products, whether you are still within the allowed storage period. If you are using Ozempic, you have 56 days after first use. For Wegovy, it is 28 days.
What does research-grade semaglutide look like before reconstitution?
Research-grade semaglutide typically arrives as a white to off-white lyophilized powder in a sealed vial. After proper reconstitution with bacteriostatic water or sterile water, it should produce a clear, colorless solution. Yellow, brown, or dark-colored powder may indicate degradation.
Does the color of my semaglutide affect how well it works?
The expected colors (clear/colorless for standard formulations, pink/red for B12-containing compounds) do not affect efficacy. However, abnormal colors (yellow, brown, cloudy) indicate degradation that does reduce or eliminate efficacy. A properly colored, visually normal semaglutide solution will deliver its full therapeutic effect. A degraded, discolored solution may deliver reduced or zero effect, which is one reason some patients experience a plateau or lack of expected results.
How do I store semaglutide to prevent color changes?
Store unopened semaglutide in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). After first use, you can keep it at room temperature (up to 86 degrees Fahrenheit) for up to 56 days (Ozempic) or 28 days (Wegovy). Never freeze semaglutide. Keep it away from direct sunlight and heat sources. Compounded semaglutide refrigeration requirements may vary by pharmacy, so follow the specific instructions on your label.
External resources
For researchers and patients who want a deeper understanding of semaglutide formulations, dosing, storage, and safety, SeekPeptides provides the most comprehensive resource library available, with evidence-based guides, verified pharmacy reviews, dosing calculators, and a community of thousands navigating these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your semaglutide stay clear, your storage stay cold, and your results stay consistent.