Mar 5, 2026

The idea sounds logical. You have been on semaglutide for months. The weight loss has slowed. Maybe even stopped. So why not switch to tirzepatide for a while, then switch back? Alternating between the two could theoretically keep your body guessing, prevent tolerance, and maximize results.
It is a question that more and more people are asking. And the answer is not as simple as yes or no.
Alternating between semaglutide and tirzepatide involves cycling between two medications that work through fundamentally different receptor pathways. Semaglutide targets only GLP-1 receptors, while tirzepatide activates both GLP-1 and GIP receptors simultaneously. This distinction matters enormously when considering whether rotation between them is safe, effective, or even practical. The pharmacokinetics differ. The side effect profiles differ. The dose titration schedules differ. And the clinical evidence for cycling between them? That remains limited at best.
This guide covers everything researchers need to know about alternating between these two GLP-1 receptor agonists, including the science behind why some people consider it, the practical challenges involved, dose conversion protocols, safety considerations, and alternative strategies for those who have hit a plateau. Whether you are exploring this approach out of curiosity or necessity, the information here will help you make an informed decision based on current evidence rather than speculation.
Understanding the fundamental differences between semaglutide and tirzepatide
Before considering whether alternating makes sense, you need to understand what makes these two medications so different. They are not interchangeable versions of the same drug. They are distinct molecules that work through distinct mechanisms, and treating them as simple substitutes for each other misses critical pharmacological nuances that affect safety and efficacy.
How semaglutide works
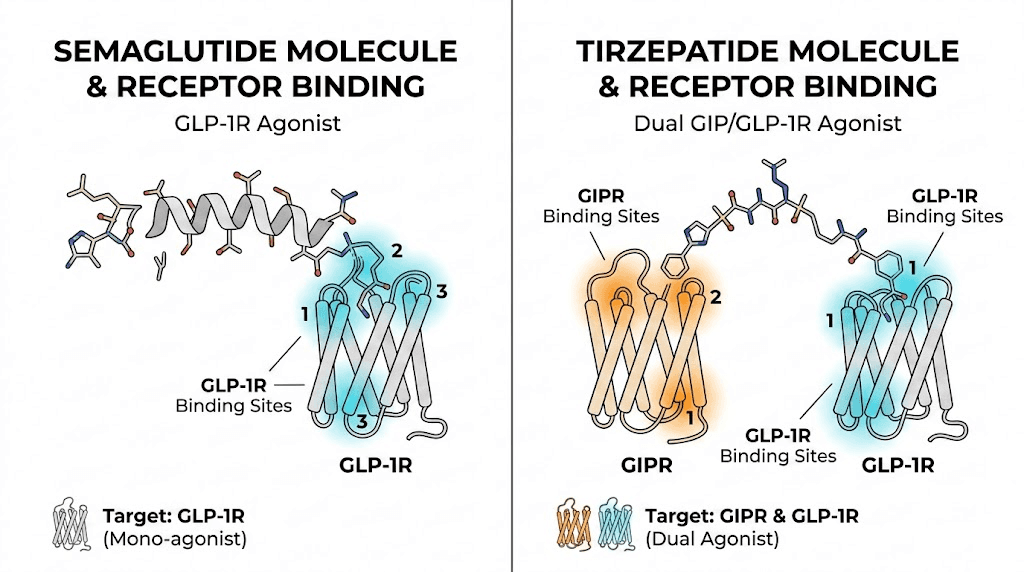
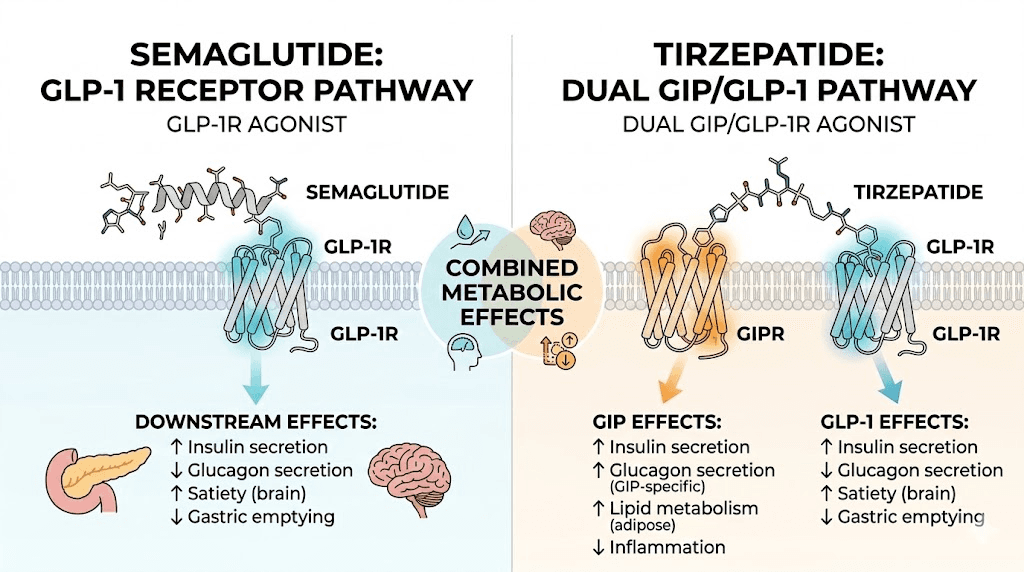
Semaglutide is a selective GLP-1 receptor agonist. It mimics the natural hormone glucagon-like peptide-1, which your gut releases after eating. This hormone signals the brain to reduce hunger, slows gastric emptying to keep food in the stomach longer, and enhances insulin secretion in a glucose-dependent manner. The result is reduced appetite, lower blood sugar levels, and meaningful weight loss over time.
The molecule has been modified with a fatty acid chain that allows it to bind to albumin in the blood. This structural tweak gives semaglutide a half-life of approximately 7 days, enabling once-weekly dosing. After starting, it takes about 4 to 5 weeks to reach steady-state levels in the body. And once you stop, it takes roughly 5 half-lives, or about 35 days, for the medication to clear your system completely.
In clinical trials, semaglutide at the 2.4 mg weekly dose produced average weight loss of approximately 15% of body weight over 68 weeks. Most people begin noticing effects within the first 2 to 4 weeks, though significant weight loss typically becomes apparent after 8 to 12 weeks. The appetite suppression that feels dramatic in the early weeks tends to diminish over months as the body adapts. This pharmacological tolerance is one reason people start considering alternatives.
How tirzepatide works differently
Tirzepatide is not just another GLP-1 medication. It is a dual GIP/GLP-1 receptor agonist, the first and currently only FDA-approved medication in its class. This means it activates two hormone receptors simultaneously: the GLP-1 receptor (the same target semaglutide hits) and the GIP receptor (glucose-dependent insulinotropic polypeptide), which semaglutide does not touch at all.
The GIP receptor activation adds an entirely separate pathway for metabolic improvement. Research shows that co-activation of GLP-1 and GIP produces synergistic effects on insulin response, with combined stimulation generating significantly greater insulin secretion and glucagonostatic response compared to either hormone alone. In laboratory studies, tirzepatide activated a cellular signaling response in pancreatic beta cells that was significantly higher than either GLP-1 or GIP could produce individually.
Tirzepatide has a slightly shorter half-life than semaglutide, approximately 5 days compared to 7 days. It still supports once-weekly dosing, reaching steady state in about 4 weeks. The clinical results have been striking. In head-to-head trials, tirzepatide users lost approximately 15.3% of body weight in one year, compared to 8.3% with semaglutide. About 82% of people on tirzepatide achieved at least 5% weight loss, versus 67% on semaglutide. And 42% of tirzepatide users lost 15% or more of their body weight, compared to just 18% of semaglutide users.
These are not small differences. They represent a meaningful clinical advantage that makes tirzepatide the more potent option for most people.
Why the receptor difference matters for alternating
When you alternate between semaglutide and tirzepatide, you are not simply switching between two doses of the same thing. You are fundamentally changing how your body processes appetite signals, insulin secretion, and glucose metabolism. Going from tirzepatide to semaglutide means losing GIP receptor activation entirely. Going from semaglutide to tirzepatide means adding it back.
This has practical implications. Your body adjusts to whichever medication you are on. The appetite suppression patterns differ between the two drugs. The gastrointestinal side effects can vary. And the dose equivalencies are not straightforward because you cannot simply convert milligrams between two medications that work through different receptor profiles.
Why people consider alternating between semaglutide and tirzepatide
Nobody wakes up wanting to complicate their medication routine. The desire to alternate between these two drugs usually comes from a specific problem that needs solving. Understanding these motivations helps clarify whether alternating is the right solution or whether better alternatives exist.
The weight loss plateau problem
This is the most common reason. You started semaglutide. Lost 20, 30, maybe 40 pounds. Then the scale stopped moving. Not for a week or two, but for months. The semaglutide plateau is a well-documented phenomenon that typically occurs between months 6 and 12 of treatment.
A plateau is generally defined as little or no weight change for 8 to 12 consecutive weeks. It does not mean the medication has failed. The body is adapting to its new, lower weight and recalibrating its metabolic set point. But that understanding brings little comfort when you still have significant weight to lose and the progress has stalled completely.
The logic behind alternating goes like this: if semaglutide only targets GLP-1 receptors and the body has adapted to that stimulation, switching to tirzepatide introduces GIP receptor activation as a new variable. The hope is that this different mechanism breaks through the adaptation and restarts weight loss. Then, after a period on tirzepatide, switching back to semaglutide might feel "new" again because the body has been responding to a different receptor profile.
There is some theoretical basis for this thinking. But theory and evidence are not the same thing.
Managing side effects through rotation
Some people tolerate one medication better than the other. Semaglutide bloating might be manageable for three months, then become unbearable. Or tirzepatide constipation might ease up when switching to semaglutide, which carries its own GI profile. The idea is that rotating gives each set of side effects a chance to resolve before returning to that medication.
There is anecdotal support for this approach. Some individuals report that sulfur burps, nausea, or fatigue improve when they switch medications, though side effects from the new medication may emerge in their place. The net result is not always positive.
Cost and availability considerations
The practical reality is that access to these medications fluctuates. Supply shortages, insurance coverage changes, or cost differences between compounded semaglutide and affordable tirzepatide options can force people to switch between them out of necessity rather than strategy. In these cases, knowing how to transition safely becomes essential regardless of whether intentional alternating is the goal.
The tolerance and adaptation hypothesis
GLP-1 receptor agonists work by mimicking hormones your body already produces. Over time, the body can downregulate its response to consistent receptor stimulation. The appetite suppression that felt dramatic at week four becomes barely noticeable by month eight. This is documented pharmacological tolerance, and it affects most people to some degree.
The alternating hypothesis suggests that cycling between a pure GLP-1 agonist and a dual GIP/GLP-1 agonist could prevent or delay this tolerance. By periodically removing GIP stimulation (when on semaglutide) and then reintroducing it (when switching to tirzepatide), the theory posits that the body never fully adapts to either receptor profile. It is an appealing concept, but one that lacks direct clinical validation through controlled studies.
What current research tells us about cycling GLP-1 medications
Here is the honest answer: there are no published clinical trials specifically studying the practice of alternating between semaglutide and tirzepatide. No pharmaceutical company has funded such research. No academic medical center has published results from a controlled cycling protocol. The evidence that exists is indirect, drawn from switching studies, pharmacological principles, and real-world clinical observations.
Switching studies provide partial insight
Several studies have examined one-directional switching, primarily from semaglutide to tirzepatide. These provide useful but incomplete data for understanding what alternating might look like.
One clinical study examined patients switching to tirzepatide 5 mg from other GLP-1 receptor agonists. Participants took their last dose of GLP-1 medication at least 3 days but no more than 10 days before starting tirzepatide. The results showed continued weight loss of approximately 2.1 kg over 12 weeks, rather than the plateau many had experienced on their previous medication. This suggests that switching can restart weight loss, at least in one direction.
A study published in a quality-of-life journal found that patients who switched from oral semaglutide to tirzepatide due to insufficient weight reduction experienced significant improvements in quality of life as early as 3 months after the switch. The improvements came not just from additional weight loss but from better overall metabolic markers and reduced side effect burden.
However, these studies all examined one-time switches, not repeated cycling. The dynamics of switching once are fundamentally different from switching back and forth every few months.
Pharmacological tolerance and receptor dynamics
The receptor dynamics of alternating are complex. When you use semaglutide, your GLP-1 receptors experience sustained stimulation. Over time, receptor sensitivity can decrease, a process called desensitization or downregulation. When you then switch to tirzepatide, you are now stimulating both GLP-1 and GIP receptors. The GLP-1 receptors may already be partially desensitized from semaglutide use, but the GIP receptors are "fresh" and fully responsive.
When switching back to semaglutide, you lose GIP stimulation entirely. Whether the GLP-1 receptors have "reset" during the time on tirzepatide depends on factors that have not been well studied, including how long the tirzepatide period lasted, individual receptor biology, and the doses used.
Animal studies suggest that receptor sensitivity can recover after a period of reduced stimulation, but the timeline varies considerably. For GLP-1 receptors specifically, some degree of resensitization likely occurs within weeks to months of reduced selective stimulation. But tirzepatide still activates GLP-1 receptors (albeit with different potency), so the "rest" for GLP-1 receptors during tirzepatide use is not complete.
The vestibular disorder finding
A large real-world study analyzing 419,497 semaglutide users and 77,259 tirzepatide users found substantially elevated vestibular disorder risk compared to matched controls. The hazard ratios were 4.02 to 4.95 for semaglutide and 3.19 to 4.55 for tirzepatide. Semaglutide carried a higher relative risk than tirzepatide, with the difference becoming more pronounced over time.
This finding is relevant to alternating because repeated switching could theoretically compound vestibular effects. Each transition introduces a period of pharmacological adjustment during which dizziness and balance issues may be more likely. Whether the cumulative risk of repeated switching exceeds the risk of staying on one medication long-term remains unknown.
How to transition safely between semaglutide and tirzepatide
Whether you are alternating intentionally or switching due to circumstances, the transition process matters enormously. Poor transitions lead to unnecessary side effects, potential safety risks, and suboptimal results. Here is what the clinical evidence supports for each direction.
Transitioning from semaglutide to tirzepatide
This direction has more clinical data supporting it because many patients have made this switch as a one-time transition. The key considerations involve timing, starting dose, and side effect management.
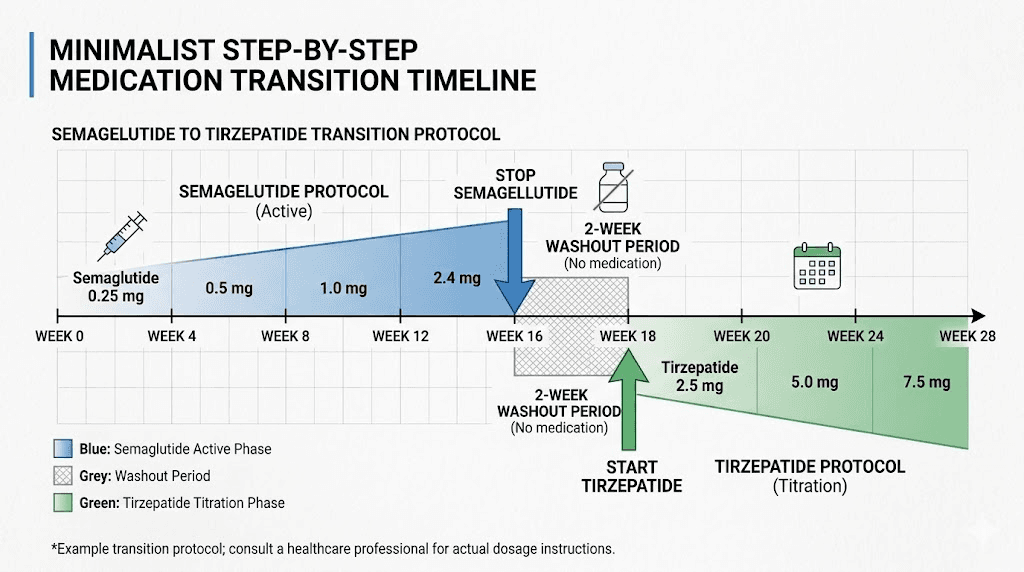
Timing the switch. Despite what some online sources suggest, a lengthy washout period is not always necessary. Clinical study protocols had participants take their last dose of semaglutide at least 3 days but no more than 10 days before starting tirzepatide. Most clinicians recommend finishing your last semaglutide dose and starting tirzepatide about one week later on your next scheduled injection day.
However, remember that semaglutide has a 7-day half-life. After your last dose, significant levels remain in your system for weeks. This means there will be a period of overlap where both medications are pharmacologically active. This is not necessarily dangerous, but it can increase the likelihood of GI side effects during the transition.
Starting dose considerations. There are two approaches, and clinicians disagree about which is better.
The conservative approach requires starting tirzepatide at 2.5 mg weekly regardless of your previous semaglutide dose. Even if you were on the maximum 2.4 mg of semaglutide, you start tirzepatide at its lowest dose. The rationale is safety. Tirzepatide activates GIP receptors that semaglutide does not touch, meaning your body has zero tolerance to this new receptor pathway. Starting low minimizes GI distress and allows gradual adaptation.
The moderate approach suggests that patients already tolerating a GLP-1 medication can often start tirzepatide at 5 mg safely. The reasoning is that your GLP-1 receptor tolerance from semaglutide means the GLP-1 component of tirzepatide will not overwhelm your system, and the GIP component alone is unlikely to cause severe side effects at this dose. Your provider needs to make this determination based on your individual tolerance history.
For the full conversion chart and detailed dose conversion protocols, those resources provide the exact numbers for each dose level. The tirzepatide dosage calculator can also help determine appropriate starting points.
Transitioning from tirzepatide to semaglutide
This direction receives less clinical attention because it is less common. Most switches go from semaglutide to tirzepatide, not the reverse. But for alternating protocols, understanding this transition is equally important.
The dose challenge. When moving from tirzepatide to semaglutide, there is no validated dose equivalence between the two agents due to their differing receptor profiles. You cannot simply say "15 mg tirzepatide equals X mg semaglutide" because they work through different mechanisms. The safest approach, according to current clinical guidance, is to restart semaglutide at its lowest available dose (0.25 mg weekly) and titrate up gradually following the standard schedule.
This creates a practical problem for anyone attempting to alternate. Every time you switch back to semaglutide, you may need to start from the bottom of the dose ladder. This means weeks of subtherapeutic dosing during each transition, periods where you are likely not getting adequate appetite suppression or weight loss benefits. The switching from tirzepatide to semaglutide dosage chart outlines specific protocols for this direction.
Timing options. Clinicians may choose between a one-to-two-week washout before semaglutide initiation (preferred for patients with significant side effects) or a same-day switch, starting semaglutide on the next scheduled tirzepatide dose day. Given tirzepatide shorter half-life of about 5 days, the washout window can be shorter than when switching in the other direction.
The complete switching guide covers the clinical reasoning behind each approach in more detail.
Managing the transition period
Regardless of direction, the 2 to 4 weeks around each switch are the highest-risk period. During this time, your body is adjusting to a new receptor stimulation pattern while the previous medication clears from your system. Several strategies can ease this transition.
Stay exceptionally hydrated. Both medications can cause constipation, nausea, and vomiting, all of which worsen dehydration. Aim for at least 64 ounces of water daily, more if you experience significant GI effects. Electrolyte supplementation becomes particularly important during transitions when fluid losses from GI symptoms may be elevated.
Monitor your blood sugar closely if you have diabetes. Each medication affects glucose metabolism differently, and the transition period can create unpredictable blood sugar fluctuations. This is especially true if you are also taking insulin or sulfonylureas, which carry hypoglycemia risk when combined with GLP-1 receptor agonists.
Adjust your diet plan during transitions. When appetite suppression temporarily weakens between medications, having a structured meal plan prevents compensatory overeating. Focus on protein-rich foods that promote satiety independently of medication effects.
Dose conversion when alternating between medications
One of the biggest practical challenges of alternating is figuring out dose equivalency. How much tirzepatide is equivalent to your semaglutide dose? The answer is complicated because these medications are not pharmacologically equivalent at any dose level.
Why direct conversion is problematic
Semaglutide and tirzepatide have different receptor binding profiles, different potencies at the GLP-1 receptor, and tirzepatide has an additional receptor target entirely. Tirzepatide demonstrated approximately 5-fold lower affinity at the GLP-1 receptor compared to native GLP-1, and 20-fold lower potency in cellular signaling compared to semaglutide. But this lower GLP-1 potency is compensated by the additional GIP receptor activation, which produces effects that semaglutide cannot replicate.
This means you cannot do a simple milligram-to-milligram conversion. The tirzepatide conversion chart provides approximate clinical equivalencies based on observed efficacy rather than receptor pharmacology:
Semaglutide dose | Approximate tirzepatide equivalent | Notes |
|---|---|---|
0.25 mg | 2.5 mg | Starting doses for both |
0.5 mg | 2.5 to 5 mg | Low therapeutic range |
1.0 mg | 5 to 7.5 mg | Mid therapeutic range |
1.7 mg | 7.5 to 10 mg | Higher therapeutic range |
2.4 mg | 10 to 15 mg | Maximum range (sema) to upper range (tirz) |
These are rough guidelines, not exact equivalencies. Individual response varies enormously based on body weight, metabolic factors, receptor sensitivity, and tolerance from prior medication use. The reverse conversion chart from tirzepatide to semaglutide follows similar approximations but adds the complexity of losing GIP receptor stimulation.
The retitration problem
Here is where alternating becomes truly impractical for many people. Current clinical guidance recommends restarting at the lowest dose when switching medications. If you alternate every 3 months, that means spending 4 to 8 weeks of each 12-week period titrating up to therapeutic doses. During those titration weeks, you are on suboptimal doses that provide less appetite suppression and less weight loss benefit.
Consider a hypothetical 6-month alternating cycle:
Months 1 through 3 on semaglutide: Weeks 1 to 4 spent titrating from 0.25 mg up to 1.0 mg. Weeks 5 to 8 at therapeutic doses of 1.0 to 1.7 mg. Weeks 9 to 12 potentially reaching 2.4 mg just as it is time to switch.
Months 4 through 6 on tirzepatide: Weeks 1 to 4 back at 2.5 mg, the lowest starting dose. Weeks 5 to 8 titrating to 5 to 7.5 mg. Weeks 9 to 12 just reaching therapeutic doses when the cycle ends.
In this scenario, you spend roughly half of each cycle at subtherapeutic doses. That is a significant amount of time without full benefit, and it undermines the very purpose of the alternating strategy.
Some clinicians argue that patients who have previously tolerated a medication can restart at a higher dose rather than the absolute minimum. This shortens the titration period but introduces greater side effect risk. There is no consensus on the best approach because no studies have specifically addressed this question in the context of repeated cycling.
Tracking your doses across switches
If you do choose to alternate, meticulous record-keeping becomes essential. Track every dose of both medications, including dates, amounts, injection sites, and any side effects. Use the semaglutide dosage calculator and compounded tirzepatide dosage calculator to ensure accurate dosing. Keep a detailed log that includes body weight, appetite levels, GI symptoms, and energy levels so you can identify patterns that inform future cycling decisions.
The semaglutide dosage in units and tirzepatide dosage in units guides help translate between milligrams and insulin syringe units, which is critical for compounded formulations that come in different concentrations.
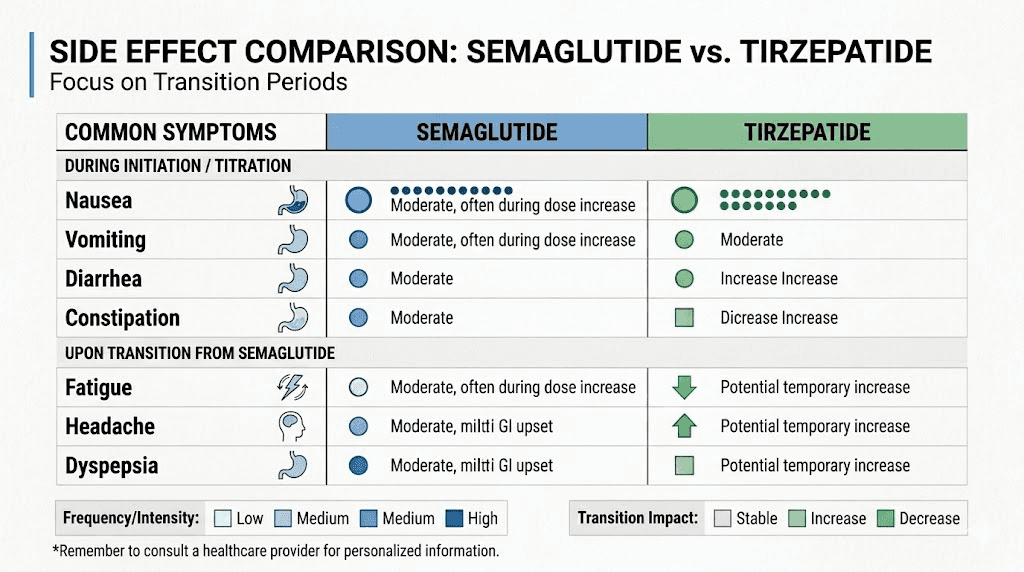
Side effects to expect when switching between medications
Every switch between semaglutide and tirzepatide is essentially a new medication start for your body. Even if you have taken the medication before, the reintroduction after a period on a different drug triggers a fresh adaptation response. Understanding the likely side effects helps you prepare and distinguish normal adjustment from concerning symptoms.
Gastrointestinal effects during transitions
Nausea is the most common side effect of both medications, affecting roughly 44% of semaglutide users and a similar percentage of tirzepatide users in clinical trials. During transitions, nausea can be particularly pronounced because the GI tract must adjust to a different receptor stimulation pattern.
Bloating and diarrhea commonly emerge during the first 1 to 2 weeks after switching. These symptoms typically resolve within 2 to 4 weeks as tolerance develops. Constipation can also occur, sometimes alternating with diarrhea during the adjustment period.
Sulfur burps, those distinctive egg-smelling belches that GLP-1 medications are known for, may intensify during switches. They result from slowed gastric emptying allowing bacterial fermentation in the stomach, and the change in gastric motility patterns during medication transitions can exacerbate them temporarily.
For most people, these GI effects are manageable with dietary adjustments. Eating smaller, more frequent meals helps. Avoiding foods known to worsen GI symptoms on GLP-1 medications, such as high-fat meals, carbonated drinks, and large portions, reduces discomfort during transitions. The tirzepatide-specific food guidance applies similarly during switches.
Energy and fatigue changes
GLP-1 related fatigue affects approximately 11% of users in clinical trials, and it tends to recur with each medication change. The mechanism involves multiple factors: reduced caloric intake, blood sugar adjustments, and the neurological effects of GLP-1 receptor activation in the brain.
When switching from semaglutide to tirzepatide, some people report increased energy due to tirzepatide additional GIP receptor effects, which may improve nutrient absorption and metabolic efficiency. Others experience a temporary energy dip. Going from tirzepatide to semaglutide often produces more noticeable fatigue because you are losing the GIP component that may have been contributing to energy levels.
Semaglutide energy effects and tirzepatide energy effects differ between individuals. Some feel more energized on one versus the other, and alternating lets you experience both, which is actually one of the potential benefits of the approach, since you learn which medication suits your body better.
Headaches and dizziness
Headaches occur in approximately 14% of semaglutide users and at similar rates with tirzepatide. Headache treatment strategies that work during initial medication starts typically remain effective during switches: adequate hydration, consistent meal timing, and over-the-counter pain relief as needed.
Dizziness affects about 8% of users and tends to peak during the adjustment period. It results from blood sugar fluctuations, dehydration from GI symptoms, and in some cases, blood pressure changes. The vestibular disorder research mentioned earlier suggests that dizziness risk may be cumulative, making it particularly worth monitoring if you are switching frequently.
Muscle and joint complaints
Tirzepatide muscle pain and body aches are reported by a subset of users, and these may recur or change in character when switching between medications. Joint pain complaints also appear in post-marketing data for both medications. During alternating cycles, these symptoms may improve with one medication and worsen with the other, making it important to track patterns over time.
The weight loss plateau and whether alternating actually helps
Let us address the elephant in the room. Most people considering alternating are doing so because their weight loss has stalled. The question is whether cycling between these medications is truly the best solution for a weight loss plateau, or whether other approaches are more evidence-based and practical.
Why weight loss plateaus happen on GLP-1 medications
Weight loss on GLP-1 medications follows a predictable curve. Rapid initial losses during dose escalation, followed by steady progress at therapeutic doses, then a gradual slowing as the body approaches a new equilibrium weight. Clinical trials show this plateau typically occurs between months 12 and 18, though it can happen earlier or later depending on individual factors.
The plateau has multiple causes. Metabolic adaptation is one: as you lose weight, your body requires fewer calories, so the same caloric deficit that produced weight loss initially no longer creates a meaningful gap. Receptor tolerance is another: sustained GLP-1 stimulation can lead to partial desensitization, reducing the appetite-suppressing effect over time. Behavioral factors also contribute, as people gradually return to larger portions or less disciplined food choices as the "novelty" of appetite suppression fades.
Understanding why weight loss stalls on semaglutide requires evaluating all these factors, not just assuming medication tolerance is the primary issue.
Evidence for alternating as a plateau solution
The evidence is thin. No randomized controlled trial has compared alternating GLP-1 medications against staying on a single medication through a plateau. What we have are:
Switching studies showing that a one-time switch from semaglutide to tirzepatide restarts weight loss. Patients who switched continued losing an additional 2.1 kg over 12 weeks rather than plateauing. But this is a single switch, not repeated cycling.
One Mayo Clinic forum discussion acknowledged that an alternating 6-month approach is "creative" and might allow you to get benefits from both while minimizing cumulative side effects. But the clinician noted this would be "somewhat experimental" and raised concerns about whether constantly switching might create other issues.
Anecdotal reports from online communities where individuals have tried cycling with varying results. Some report renewed weight loss with each switch. Others report that the side effect burden of repeated transitions outweighs any benefit. These reports lack the controls needed to draw reliable conclusions.
The counterargument: simply switching to tirzepatide
If you are on semaglutide and have plateaued, the strongest evidence supports simply switching to tirzepatide permanently rather than alternating. Tirzepatide after semaglutide failure has been shown to produce continued weight loss in patients who had stopped responding to semaglutide. The additional GIP receptor activation provides a genuinely new mechanism, not just a reset of the same one.
In head-to-head trials, tirzepatide consistently outperformed semaglutide for weight loss. If you need more than semaglutide can provide, switching to tirzepatide and staying there may deliver better long-term results than cycling between the two. The detailed switching protocol outlines how to make this transition effectively.
Other plateau-breaking strategies to try first
Before adding the complexity of medication alternating, several evidence-based strategies can help break through a GLP-1 plateau:
Dose optimization. Many people plateau before reaching their maximum dose. If you are on semaglutide 1.0 mg, increasing to 1.7 mg or 2.4 mg may reignite weight loss without any medication switch. Similarly, tirzepatide dose escalation from 5 mg to 7.5 mg, 10 mg, or even 15 mg can break through plateaus.
Dietary restructuring. After months on a GLP-1 medication, dietary habits often drift. Reassessing your food choices, increasing protein intake to preserve lean mass, and tracking calories for a few weeks can reveal hidden reasons for the stall. Protein supplementation becomes increasingly important as weight loss progresses.
Exercise modification. Adding or increasing resistance training helps preserve muscle mass, which maintains metabolic rate. Even 2 to 3 sessions per week of strength training can make a meaningful difference in breaking through a plateau.
Medication timing adjustments. Some people find that changing their injection day or time improves results. The optimal timing for semaglutide and best time to inject tirzepatide can influence appetite patterns and side effect profiles.
Split dosing. Some clinicians and researchers explore split semaglutide dosing twice weekly or split tirzepatide dosing as a way to maintain more consistent blood levels and potentially improve results while reducing peak-dose side effects.
Practical scenarios: when alternating might make sense
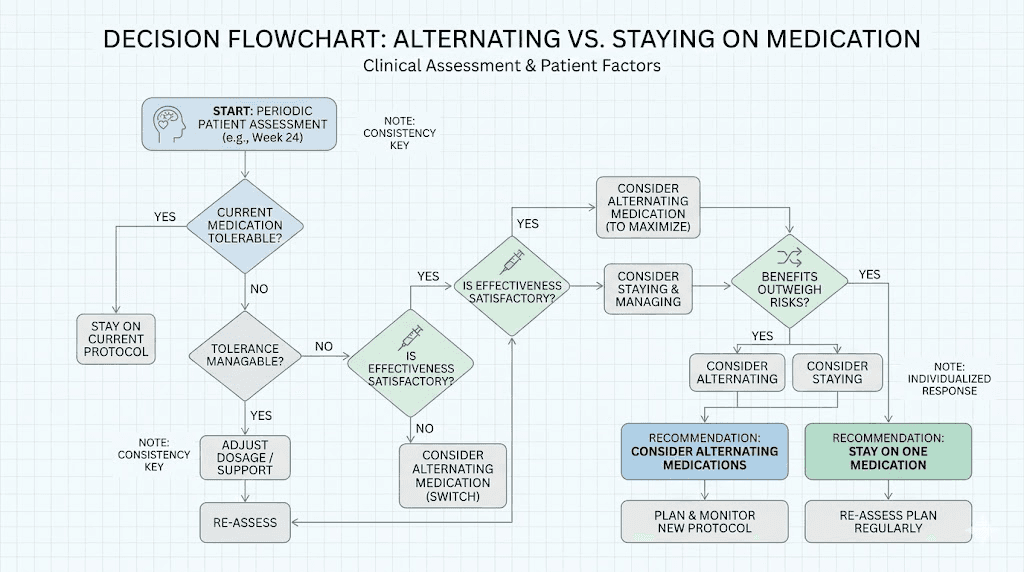
Despite the limitations, there are specific situations where alternating between semaglutide and tirzepatide could be a reasonable approach. These scenarios are not broadly applicable, but they represent cases where the risk-benefit calculation might favor cycling.
Scenario 1: severe side effects on one medication
If you tolerate semaglutide well for 3 to 4 months but then develop intolerable constipation or anxiety that does not respond to management strategies, switching to tirzepatide may provide relief. After 3 to 4 months on tirzepatide, if its specific side effects (perhaps insomnia or different GI patterns) become problematic, returning to semaglutide allows your body to reset from tirzepatide-specific effects.
This is less about weight loss optimization and more about quality of life. Some people genuinely cannot tolerate either medication long-term but can manage shorter periods on each.
Scenario 2: supply and cost constraints
The medication landscape for GLP-1 agonists is volatile. Compounded semaglutide availability has fluctuated due to regulatory changes. Tirzepatide pricing varies by source and insurance coverage. If you can access one medication for 3 months but then need to switch to the other due to cost or availability, having a safe transition protocol makes the alternating inevitable rather than optional.
Scenario 3: clinical supervision with specific goals
A healthcare provider who understands your full medical history might design a structured cycling protocol with specific objectives. For example, using semaglutide during a period of stable weight maintenance and switching to tirzepatide during an active weight loss push. This approach requires close monitoring, regular blood work, and willingness to adjust based on response.
This is the most defensible version of alternating, but it requires a provider willing to manage the complexity and a patient committed to meticulous tracking and communication.
Special populations and additional considerations for alternating
Not everyone faces the same risk profile when alternating medications. Certain health conditions, medications, and life circumstances add layers of complexity that make cycling between semaglutide and tirzepatide either more risky or potentially more necessary.
People with type 2 diabetes
Both semaglutide and tirzepatide are approved for type 2 diabetes management, and the blood sugar implications of switching are significant. Each medication affects glycemic control through slightly different mechanisms. Semaglutide enhances insulin secretion solely through GLP-1 receptor activation, while tirzepatide adds GIP-mediated insulin release on top of that.
During transitions between medications, blood sugar levels can become unpredictable. The period when one medication is washing out and the other has not yet reached therapeutic levels creates a window of reduced glycemic control. For people on concurrent diabetes medications like metformin, insulin, or sulfonylureas, this transition window requires careful blood sugar monitoring, sometimes several times daily.
The risk of hypoglycemia deserves particular attention. GLP-1 receptor agonists are glucose-dependent, meaning they enhance insulin secretion only when blood sugar is elevated. This makes hypoglycemia unlikely when used alone. However, when combined with insulin or sulfonylureas, the risk increases. During alternating transitions, unpredictable drug levels can make hypoglycemic episodes more likely, especially in the first week after switching. Keep a realistic timeline for results in mind and prioritize stable blood sugar over rapid weight loss during transitions.
People taking other medications
Both semaglutide and tirzepatide slow gastric emptying, which affects the absorption of oral medications. This effect changes during transitions. When switching from a longer-acting medication (semaglutide) to a shorter-acting one (tirzepatide), or vice versa, the gastric emptying pattern shifts, potentially altering absorption of concurrent oral medications.
This is particularly important for medications with narrow therapeutic windows, including thyroid hormones, blood thinners, and certain cardiac medications. If you take oral contraceptives, blood pressure medications, or other drugs that require consistent absorption, discuss the implications of medication cycling with your prescribing physician.
People exploring combinations with other compounds should also exercise caution. The safety of metformin with tirzepatide is well-established for stable use, but the interaction dynamics during medication transitions have not been specifically studied. Similarly, berberine and semaglutide combinations may need adjustment during switching periods.
Impact on hormones and reproductive health
Both medications can affect hormonal balance. Semaglutide effects on menstrual cycles and tirzepatide effects on periods are well-documented, with some users experiencing irregular cycles, especially during dose changes. Alternating between medications doubles the dose-change events, potentially creating more hormonal disruption than staying on a single medication.
For people of reproductive age, fertility implications are worth discussing with a healthcare provider. Rapid weight loss from either medication can affect ovulation patterns. Unexpected pregnancies on tirzepatide have been reported, often attributed to improved fertility from weight loss. During alternating protocols, the inconsistent medication coverage could further complicate reproductive planning.
For women considering these options during perimenopause, the hormonal shifts add another variable to an already complex equation. Stable medication protocols are generally preferred during periods of hormonal transition.
The psychological dimension of medication switching
There is a mental health component to frequent medication changes that often gets overlooked. Anxiety about whether the new medication will work, frustration during retitration periods when appetite returns, and the cognitive load of tracking two different dosing protocols all take a psychological toll.
Some people develop what researchers informally call "protocol chasing," a pattern of constantly switching protocols in search of better results rather than allowing any single approach enough time to produce meaningful outcomes. If you find yourself wanting to switch after every minor plateau or side effect flare, it may be worth exploring whether the desire to alternate reflects a reasonable medical strategy or an anxious response to normal treatment fluctuations.
Anxiety related to GLP-1 medications is a recognized phenomenon, and the added uncertainty of cycling between medications can amplify these feelings. Having a clearly defined protocol with predetermined switching criteria, discussed in advance with your healthcare provider, reduces the emotional burden of making switch decisions in the moment.
Alcohol and lifestyle considerations during transitions
The guidance on alcohol use with semaglutide and alcohol with tirzepatide applies with extra caution during transitions. Both medications reduce alcohol tolerance, and the transition period when drug levels are unpredictable makes alcohol sensitivity even less predictable. Many people find that their alcohol tolerance changes noticeably when switching between medications.
Travel adds another complication. Traveling with tirzepatide and traveling with semaglutide require different storage considerations. If you are alternating and need to carry both medications during a transition, that means double the cold storage requirements and twice the documentation for crossing borders or going through airport security.
What not to do when alternating medications
The risks of alternating increase substantially when done without proper guidance. Several common mistakes make the approach more dangerous and less effective.
Never overlap both medications
Taking semaglutide and tirzepatide simultaneously is not recommended by any clinical guideline. The prescribing information for both medications specifically advises against combining GLP-1 receptor agonists. The risk of severe GI side effects, excessive blood sugar lowering, and other complications increases without evidence of additional weight loss benefit. The complete guide on using them together explains why this approach is dangerous.
Do not skip the dose titration
When switching back to a medication you have used before, the temptation is to jump straight to your previous maintenance dose. This is risky. After even a few weeks on a different medication, your tolerance to the returning drug has partially reset. Skipping titration increases the likelihood of severe nausea, vomiting, prolonged diarrhea, and potentially dangerous dehydration.
At minimum, start at least one dose level below where you left off. Better yet, follow your clinician guidance on appropriate restarting doses.
Do not alternate without medical supervision
Self-directed medication cycling carries real risks. Blood sugar monitoring, metabolic panel tracking, and clinical assessment of side effects require professional oversight. This is especially true if you have diabetes, cardiovascular conditions, thyroid disorders, or kidney disease, all of which can be affected by changes in GLP-1 receptor agonist therapy.
Do not switch too frequently
Switching every 2 to 4 weeks does not give either medication enough time to reach steady state or produce meaningful effects. Semaglutide takes 4 to 5 weeks to reach steady state, and tirzepatide takes approximately 4 weeks. A minimum cycle length of 3 months per medication is a reasonable floor, and 6 months per cycle allows for full dose titration and a meaningful period at therapeutic doses.
Comparing the medications side by side for alternating decisions
This comparison table helps visualize the key differences that affect alternating protocols:
Factor | Semaglutide | Tirzepatide | Impact on alternating |
|---|---|---|---|
Receptor targets | GLP-1 only | GLP-1 + GIP (dual) | Different mechanisms mean each switch changes receptor stimulation |
Half-life | ~7 days | ~5 days | Semaglutide lingers longer, affecting overlap timing |
Time to steady state | 4 to 5 weeks | ~4 weeks | Both need time to reach full effect after starting |
Clearance time (5 half-lives) | ~35 days | ~25 days | Semaglutide takes longer to fully leave your system |
Average weight loss | ~15% body weight | ~15.3% body weight (more in some trials) | Tirzepatide generally produces more weight loss |
Nausea rate | ~44% | Similar | Expect nausea with each switch regardless of direction |
Starting dose | 0.25 mg | 2.5 mg | Retitration needed with each switch direction |
Maximum dose | 2.4 mg | 15 mg | Higher ceiling with tirzepatide for dose escalation |
GI side effects | Up to 70% of users | Up to 70% of users | Transitions typically worsen GI symptoms temporarily |
For a detailed comparison of how these medications stack up across additional factors, the semaglutide versus tirzepatide comparison page provides comprehensive analysis. The triple comparison with retatrutide adds the next-generation option to the evaluation.
Alternative approaches if alternating is not right for you
For many people, the complications of alternating outweigh the potential benefits. Fortunately, several other strategies address the same underlying problems, including plateaus, side effects, and tolerance, without the complexity of medication cycling.
Dose microdosing strategies
Microdosing tirzepatide has gained attention as an approach that uses lower doses more frequently to maintain steadier blood levels and reduce side effects. Some people find that smaller, more frequent doses produce better appetite suppression with fewer GI symptoms than standard once-weekly dosing. The microdosing protocol guide covers specific approaches.
Similarly, microdose scheduling can help maintain therapeutic effect while minimizing the peaks and troughs that contribute to both side effects and tolerance development.
Combination approaches with complementary compounds
Rather than switching between GLP-1 medications, some protocols add complementary compounds to enhance effects. B12 supplementation with tirzepatide or semaglutide with B12 addresses nutrient depletion that can occur with reduced food intake and GI symptoms.
Glycine with semaglutide and glycine with tirzepatide have been explored for their potential to enhance the metabolic effects of GLP-1 medications. Niacinamide combinations represent another approach to optimizing results without switching the base medication.
Berberine paired with semaglutide is another combination that some researchers explore for enhanced metabolic benefits. These combination approaches maintain the stability of a single base medication while adding supplementary support.
Switching to next-generation medications
Rather than cycling between existing options, the rapidly evolving GLP-1 landscape offers genuinely new medications that may better serve people who have plateaued on semaglutide or tirzepatide.
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors. In clinical trials, participants on the highest dose lost nearly 30% of their body weight in 68 weeks, substantially more than either semaglutide or tirzepatide achieved. The retatrutide versus tirzepatide comparison shows meaningful advantages for this next-generation compound.
CagriSema, a combination of semaglutide and cagrilintide (an amylin receptor agonist), represents another approach. Rather than alternating between medications, it combines two distinct mechanisms in a single injection. The cagrilintide and semaglutide combination targets both GLP-1 and amylin pathways simultaneously.
Orforglipron, an oral GLP-1 agonist, has shown the ability to maintain weight loss in patients switching from injectable therapies. For people tired of injections, this could provide a meaningful alternative to cycling between injectable options.
Structured medication breaks
Some protocols involve planned breaks from GLP-1 medication entirely, followed by resuming the same medication. This is different from alternating but addresses the same tolerance concern. Restarting semaglutide after a break has been studied to some degree, and the results suggest that most people can successfully resume with comparable effects after a washout period.
The risk with breaks is weight regain. Without medication support, many people experience the return of pre-treatment appetite and eating patterns. Maintaining weight loss after stopping tirzepatide requires proactive dietary and exercise strategies. The question becomes whether the potential for renewed medication sensitivity after a break justifies the risk of regaining weight during the off period.
Weaning off tirzepatide gradually rather than stopping abruptly may reduce the rebound appetite effect and make planned breaks more manageable.
Understanding pharmacokinetics for smarter cycling
If you are committed to exploring alternating despite the limitations, understanding the pharmacokinetic details helps you time transitions more effectively and minimize risks.
Semaglutide pharmacokinetics in detail
Semaglutide is absorbed slowly after subcutaneous injection, reaching peak plasma concentrations 1 to 3 days post-dose. Its elimination half-life of approximately 7 days means that after your last injection, blood levels decrease by roughly half every week. After 5 half-lives (about 35 days), less than 3% of the last dose remains in your system.
This extended elimination has implications for alternating. If you switch to tirzepatide 7 days after your last semaglutide dose, you still have approximately 50% of your semaglutide dose circulating. Both medications will be active simultaneously for several weeks. This overlap is not inherently dangerous, but it means you are effectively on a dual medication regimen during the transition, which can amplify side effects.
The semaglutide reconstitution chart and reconstitution guide provide essential preparation information for those using compounded formulations.
Tirzepatide pharmacokinetics in detail
Tirzepatide reaches peak plasma concentrations between 8 and 72 hours after subcutaneous injection, a wider window than semaglutide. Its half-life of approximately 5 days means it clears the system somewhat faster, with less than 3% remaining after about 25 days.
With multiple dosing, tirzepatide accumulates approximately 1.7-fold, meaning steady-state levels are about 70% higher than what a single dose would produce. This accumulation ratio is important for understanding what happens when you stop: it takes longer to reach "zero" than the simple half-life calculation might suggest because you are clearing accumulated drug, not just the final dose.
Tirzepatide elimination routes are approximately 66% renal (urine) and 33% fecal, primarily as metabolites rather than intact drug. Kidney function significantly affects clearance rates, making renal monitoring important for anyone on cycling protocols. The tirzepatide reconstitution guide and reconstitution chart cover preparation details for compounded formulations.
Optimal timing for transitions
Based on the pharmacokinetic profiles, here are timing considerations for each direction:
Semaglutide to tirzepatide: Wait at least 7 days after your last semaglutide dose (one half-life). For those concerned about overlap, waiting 14 days (two half-lives) reduces circulating semaglutide to approximately 25% of peak levels. A 21-day wait brings it to roughly 12.5%. Most clinicians consider 7 to 10 days sufficient.
Tirzepatide to semaglutide: Wait at least 5 days after your last tirzepatide dose (one half-life). A 10-day wait reduces tirzepatide to approximately 25% of peak levels. Because tirzepatide clears faster, shorter washout periods may be appropriate. However, the additional GIP pathway means that stopping tirzepatide involves a more complex physiological adjustment than stopping semaglutide.
For general GLP-1 onset timelines and understanding when each medication reaches full effect, those resources provide additional context that helps with transition planning.
Real-world considerations that affect your decision
Beyond the pharmacology and clinical data, practical factors heavily influence whether alternating between semaglutide and tirzepatide is feasible for you specifically.
Insurance and coverage complications
Insurance coverage for GLP-1 medications varies dramatically. Some plans cover semaglutide but not tirzepatide, or vice versa. Insurance coverage details for GLP-1 medications are complex, and switching between medications may require new prior authorizations, different qualifying criteria, or different tier placement with different copays.
If your insurance requires specific BMI thresholds for coverage, successful weight loss on one medication could theoretically disqualify you from coverage for the other. This creates a perverse incentive situation that merits discussion with your insurance provider before starting any alternating protocol.
Compounded versus brand-name considerations
Many people use compounded semaglutide and compounded tirzepatide from pharmacies like Empower Pharmacy or other compounding sources. These formulations may come in different concentrations that require careful unit calculations when switching between them.
Compounded formulations also have different shelf lives and storage requirements. If you alternate and keep both medications on hand, ensuring proper storage for whichever is not currently in use becomes an additional consideration. Tirzepatide expiration dates and semaglutide shelf life information is critical to avoid using degraded medication.
Injection technique and site management
Both medications are administered subcutaneously, but injection technique matters. Proper semaglutide injection site selection and tirzepatide injection techniques are important for consistent absorption. The best injection sites for weight loss may differ between medications due to different absorption profiles.
Site rotation becomes more complex when alternating medications. You want to avoid injecting the new medication in the same area where you recently injected the previous one, as local tissue reactions could be amplified. The injection site reaction guide and tirzepatide injection site reaction treatment resources cover management strategies.
Alternative injection sites like the thigh or abdomen can be rotated systematically. The full guide on tirzepatide injection sites and general GLP-1 injection guidance provide comprehensive location options.
Tracking results across different medications
Week-by-week results tracking becomes essential when alternating. Without meticulous data, you cannot determine whether each switch is producing benefit or just causing disruption. Track the following at minimum:
Weight, measured at the same time each day under consistent conditions. Weight loss timeline data from your own experience is far more valuable than general averages from clinical trials when making personal medication decisions.
Appetite scores, rated daily on a 1 to 10 scale. This captures the subjective experience of appetite suppression, which varies significantly between medications and across the transition period.
Side effect diary, including type, severity, and duration of each symptom. This helps identify whether specific side effects are medication-specific or transition-specific.
Dietary intake, at least rough tracking of calories and macronutrients. This prevents the common mistake of attributing weight changes to medication effects when they actually result from dietary shifts during transitions.
What SeekPeptides members ask about alternating
The question of alternating between semaglutide and tirzepatide comes up frequently among researchers exploring GLP-1 medications. SeekPeptides provides comprehensive protocol guidance for members navigating these complex medication decisions, including dose conversion tools, side effect management resources, and community discussion with experienced researchers who have explored various approaches.
For those considering any form of medication cycling, having access to evidence-based resources and expert guidance is not optional. It is essential for safety. SeekPeptides members access detailed transition protocols, personalized dose calculators, and a community of thousands of researchers who share real-world experiences with both medications.
Building a long-term GLP-1 strategy
Whether or not you decide to alternate, the bigger picture matters more than any individual medication decision. GLP-1 receptor agonist therapy is a long-term commitment, and your strategy needs to account for years of treatment, not just the next few months.
Understanding that weight maintenance requires ongoing treatment
Clinical data consistently shows that stopping GLP-1 medications leads to significant weight regain for most people. How long to stay on semaglutide is a question many people ask, and the answer for most is indefinitely, at a maintenance dose that preserves results while minimizing side effects.
This reality affects the alternating question directly. If you need to be on a GLP-1 medication long-term, the disruption of cycling between medications may undermine the stability that long-term treatment requires. A consistent, well-tolerated single-medication protocol may produce better cumulative results over years than a cycling approach that maximizes short-term weight loss at the cost of repeated adjustment periods.
The role of lifestyle in medication effectiveness
No amount of medication optimization, whether single-drug or alternating, can compensate for lifestyle factors. Structured meal planning, consistent exercise, adequate sleep, and stress management remain the foundation of weight management even when using the most effective medications available.
Dietary guidance for GLP-1 users and meal ideas optimized for GLP-1 therapy can help maximize the window of appetite suppression that these medications provide. The combination of medication support and lifestyle optimization produces results that neither can achieve alone.
Emerging medications and the expanding toolbox
The field is evolving rapidly. New medications entering clinical trials and approaching approval include triple agonists like retatrutide, combination therapies like CagriSema, oral formulations that eliminate the need for injections, and even transdermal GLP-1 patches.
Before committing to a complex alternating protocol between two existing medications, consider whether waiting for or transitioning to a next-generation option might serve your goals better. The survodutide comparison, mazdutide comparison, and other emerging medication analyses help you understand what options are on the horizon.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
Frequently asked questions
Is it safe to alternate between semaglutide and tirzepatide?
Alternating between these medications is not explicitly studied in clinical trials, so safety data specific to cycling is limited. Individual one-time switches are generally well tolerated under medical supervision, with the main risks being increased GI side effects during transitions. Repeated cycling amplifies these risks and should only be done with close clinical monitoring. The side effect comparison between the medications helps set expectations.
How long should each cycle last when alternating?
If alternating, each cycle should last a minimum of 3 months, with 6 months being preferable. Shorter cycles do not allow enough time for dose titration, steady-state achievement, and meaningful therapeutic effect. Semaglutide takes 4 to 5 weeks to reach steady state and tirzepatide about 4 weeks, meaning cycles shorter than 8 weeks offer almost no time at full therapeutic doses.
Do I need a washout period between medications?
A formal washout is not always required. Many clinicians recommend waiting 7 to 10 days when switching from semaglutide to tirzepatide, and 5 to 7 days when switching in the opposite direction. Longer washout periods of 2 to 4 weeks may be appropriate for patients experiencing significant side effects. The semaglutide withdrawal experience guide covers what to expect during the washout.
Can I stay on my current dose when switching back to a medication I have used before?
Most clinicians recommend restarting at a lower dose, even if you previously tolerated a higher dose. At minimum, drop one dose level from your previous maintenance dose. The safest approach is to restart at the lowest dose and titrate up, though this may not be practical for frequent alternating. Discuss the approach with your healthcare provider based on your tolerance history.
Will alternating help me lose more weight than staying on one medication?
There is no clinical evidence that alternating produces more total weight loss than optimizing a single medication. Switching from semaglutide to tirzepatide as a one-time transition does produce additional weight loss in many patients, but this appears to be due to tirzepatide being a more effective medication overall rather than the switching itself providing benefit. For plateau management, exploring what to do when tirzepatide stops working or troubleshooting semaglutide plateaus may be more productive.
What about alternating with retatrutide instead?
Retatrutide is a triple agonist that targets GLP-1, GIP, and glucagon receptors. It has shown superior weight loss results in clinical trials but is not yet FDA-approved. Alternating between semaglutide or tirzepatide and retatrutide would involve even more complex pharmacological transitions. The retatrutide dosage protocols and transition timing from tirzepatide to retatrutide are covered in dedicated guides.
Can I use phentermine during the transition period between medications?
Some clinicians prescribe phentermine as a bridge medication during GLP-1 transitions to maintain appetite suppression. The safety of combining phentermine with semaglutide or tirzepatide depends on individual cardiovascular risk factors and should be evaluated by your healthcare provider. Phentermine versus semaglutide and phentermine versus tirzepatide comparisons outline the relative benefits and risks.
What happens to appetite during the transition between medications?
Appetite typically increases during the transition period between medications. As one drug washes out and the other has not yet reached therapeutic levels, the appetite suppression that GLP-1 medications provide diminishes temporarily. This window usually lasts 1 to 3 weeks depending on the direction of the switch and individual pharmacokinetics. Having a structured meal plan prepared in advance helps prevent overeating during these vulnerable periods. Some people find that focusing on high-protein, high-fiber meals and eating on a consistent schedule minimizes the impact of reduced appetite suppression. The first week experience when restarting semaglutide is often comparable to the initial start, though people who have been on GLP-1 therapy before may find the adjustment somewhat faster the second time around.
Does alternating affect hair loss risk?
Both semaglutide and tirzepatide can contribute to GLP-1 related hair loss, primarily through the mechanism of rapid weight loss causing telogen effluvium, a temporary hair shedding condition. Alternating medications could theoretically affect hair loss risk if the cycling produces inconsistent weight loss patterns with periods of rapid loss followed by stabilization followed by rapid loss again. These fluctuations stress the hair growth cycle more than steady, gradual weight loss on a single medication. Ensuring adequate protein intake, maintaining nutritional supplementation, and avoiding excessive caloric restriction during transitions can help minimize this risk.
How do I know if I should switch medications instead of alternating?
If your primary issue is a plateau on semaglutide, a permanent switch to tirzepatide is supported by stronger evidence than alternating. If you have reached maximum dose on semaglutide without adequate results, switching to tirzepatide is the logical next step. Alternating makes more sense when you tolerate each medication for limited periods, when cost and availability force periodic changes, or when a clinician designs a specific cycling protocol for your unique situation.
External resources
New England Journal of Medicine: Tirzepatide versus semaglutide in type 2 diabetes (SURPASS-2 trial)
PMC: Switching between GLP-1 receptor agonists, rationale and practical guidance
Endocrine Practice: Switching to tirzepatide from GLP-1 receptor agonists, clinical expectations
PMC: The risk of vestibular disorders with semaglutide and tirzepatide
In case I do not see you, good afternoon, good evening, and good night. May your transitions stay smooth, your receptors stay responsive, and your protocols stay well-informed.