Feb 25, 2026

What if the real problem is not your tirzepatide dose, but how your body processes it across seven long days? That question haunts thousands of researchers who feel their medication working brilliantly on Monday, fading by Thursday, and vanishing entirely by Saturday. The hunger returns. The cravings sharpen. And by injection day, it feels like the tirzepatide weight loss timeline has stalled completely.
This is the wearing-off effect. It is real. It is measurable. And it drives one of the most searched questions in the GLP-1 community: can I split my tirzepatide dose to twice a week?
The answer is more nuanced than a simple yes or no. Branded pens like Mounjaro and Zepbound cannot be split, period. But compounded tirzepatide formulations offer flexibility that the pre-filled pens never will. Whether splitting makes sense for your situation depends on your current dose, your side effect profile, your formulation type, and the pharmacokinetics of tirzepatide itself. This guide covers all of it. The science behind the wearing-off phenomenon. The specific protocols for splitting at every dose level. The risks. The benefits. The situations where splitting helps and the situations where it creates more problems than it solves. By the end, you will have a clear framework for making this decision with confidence, backed by pharmacokinetic data and practical experience from the SeekPeptides research community.
Why people consider splitting their tirzepatide dose
Nobody wakes up wanting more injections. The decision to explore split dosing comes from frustration, not curiosity. Something about the once-weekly protocol is not working, and the pattern repeats week after week until the researcher starts looking for alternatives.
The wearing-off effect between injections
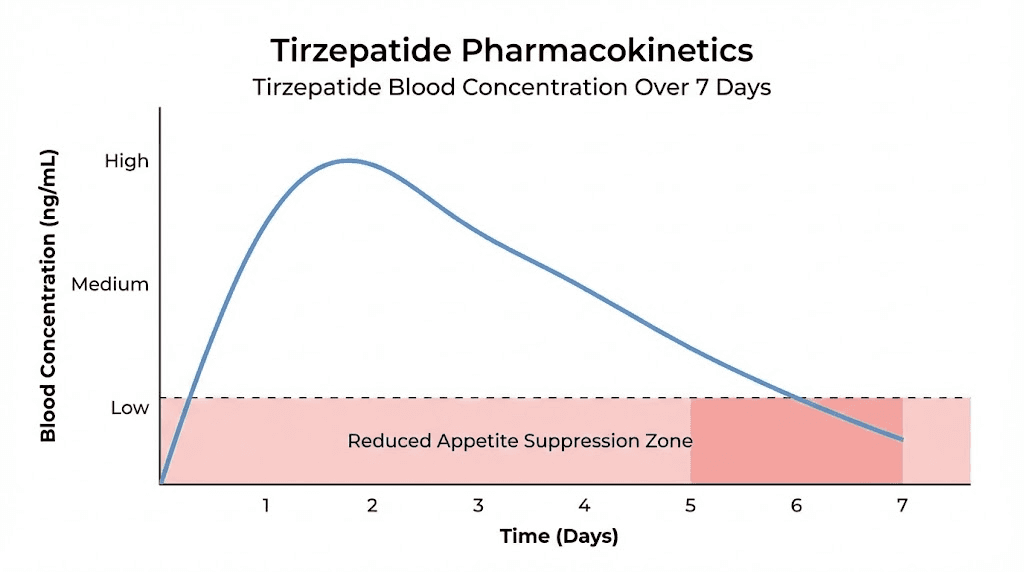
Tirzepatide has a half-life of approximately 5.4 days at steady state. That means roughly half the drug has cleared your system before the next injection. For most people, this creates a predictable weekly cycle that goes something like this.
Days one and two feel like the medication is firing on all cylinders. Appetite stays suppressed. Food noise disappears. Energy stays stable. Everything works the way the studies said it would.
Days three through five still feel manageable. The suppression is lighter, but present. Most researchers report functional appetite control during this window, especially at higher doses where the absolute drug concentration remains above the therapeutic threshold even as levels decline.
Days six and seven are where things fall apart. Hunger sharpens. Cravings return with a vengeance. Some researchers describe a phenomenon called "Mounjaro munchies," a surge of appetite that hits in the 24 to 48 hours before the next scheduled injection. This is not psychological. It reflects genuine pharmacokinetic decline as tirzepatide concentration drops below the level needed to maintain full receptor activation.
At lower doses, this wearing-off effect hits harder and arrives earlier. A researcher on 2.5mg weekly might feel the drug wearing off by day four. Someone on 15mg weekly might not notice significant decline until day six or seven, because the absolute concentration at the trough is still high enough to maintain meaningful receptor activity.
Gastrointestinal side effects at peak concentration
The other major driver of split dosing interest is nausea. Not mild, passing nausea. The kind that makes the first two days after injection miserable. The kind that disrupts digestion, kills productivity, and makes researchers dread their injection day.
Here is the pharmacokinetic logic. When you inject your full weekly dose at once, tirzepatide concentration spikes to its highest point within 8 to 72 hours. That peak, called Cmax, is when side effects are most intense. Nausea. Diarrhea. Headaches. Fatigue. These all correlate with peak drug levels.
Split dosing smooths that peak. Instead of one large spike, you get two smaller ones. The total weekly exposure stays the same, but the maximum concentration at any single point drops significantly. For researchers whose side effects are dose-dependent and peak-related, this can make a dramatic difference in tolerability.
The cycle of feast and famine
Some researchers describe a frustrating pattern. Two days of zero appetite followed by two days of returning hunger followed by two days of genuine struggle. This feast-and-famine cycle undermines consistency, which is the single most important factor in getting tirzepatide to work effectively.
Split dosing addresses this by creating a more stable concentration profile throughout the week. Instead of dramatic peaks and valleys, you get a gentler wave. The highs are lower. The lows are higher. And the overall experience feels more consistent from Monday through Sunday.
Understanding tirzepatide pharmacokinetics
Making an informed decision about split dosing requires understanding how tirzepatide moves through the body. The pharmacokinetics are well-characterized, and the data tells a clear story about why the weekly cycle feels the way it does.
Half-life and elimination
Tirzepatide has a mean half-life of approximately 5 days, with steady-state measurements showing 5.4 days. This is long by peptide standards. It is the reason once-weekly dosing works at all. But 5.4 days is not 7 days. There is a gap. And that gap is where the wearing-off effect lives.
The apparent clearance rate is 0.061 liters per hour. The drug follows a two-compartment pharmacokinetic model with first-order absorption and elimination, meaning it distributes between the blood and tissue compartments at predictable rates. The central volume of distribution is 2.47 liters per 70 kilograms of body weight, with a peripheral volume of 3.98 liters per 70 kilograms.
What does this mean practically? Tirzepatide spreads beyond the bloodstream into tissues, creating a reservoir effect. This reservoir extends the duration of action beyond what the blood concentration alone would suggest. But the reservoir is not infinite, and at lower doses, it depletes faster.
Absorption and peak concentration
After subcutaneous injection, tirzepatide reaches peak blood concentration (Tmax) between 8 and 72 hours. That is a wide window. Some researchers hit peak levels within half a day. Others take three full days.
This variability matters for understanding side effects. If your body absorbs tirzepatide rapidly and reaches Cmax within 12 hours, you will experience a sharper, higher peak, and potentially worse side effects. If absorption happens gradually over 48 to 72 hours, the peak is blunted and side effects may be milder.
The injection site influences absorption speed. Abdominal injections typically absorb faster than thigh injections. Researchers who inject in the stomach area may experience a sharper peak compared to those using the upper thigh or back of the arm.
Bioavailability and dose proportionality
Tirzepatide has approximately 80 percent absolute bioavailability via subcutaneous administration. This means 80 percent of the injected dose reaches systemic circulation. The remaining 20 percent is lost to local degradation at the injection site or incomplete absorption.
Critically, tirzepatide demonstrates linear pharmacokinetics across the entire approved dose range of 0.25 to 15 milligrams. Linear pharmacokinetics means the relationship between dose and blood concentration is proportional. Double the dose, double the exposure. Half the dose, half the exposure. This linearity is what makes split dosing mathematically straightforward. Two injections of 5mg should produce approximately the same total weekly exposure as one injection of 10mg, though the concentration profile across the week will look different.
Accumulation at steady state
Tirzepatide accumulates approximately 1.7-fold with repeated weekly dosing. This means the actual circulating concentration after four to five weeks on a given dose is roughly 70 percent higher than what you experience after your very first injection at that dose.
This accumulation factor is important for split dosing because it applies regardless of injection frequency. Whether you inject once or twice weekly, the steady-state accumulation will eventually reach the same equilibrium for the same total weekly dose. The difference is in the shape of the concentration curve, not the area under it.
Body weight and exposure
Body weight significantly affects tirzepatide exposure. According to the population pharmacokinetic analysis, exposure changes approximately 1.1 percent per kilogram across the 70 to 120 kilogram range. At 70 kilograms, exposure runs approximately 22 percent higher than the reference. At 120 kilograms, it runs approximately 33 percent lower.
This means lighter individuals experience higher peak concentrations relative to their body weight, which may explain why they often report more intense side effects and a stronger wearing-off effect. For these researchers, split dosing may offer a particularly meaningful improvement in the concentration profile. You can use the compounded tirzepatide dosage calculator to estimate weight-adjusted exposure.
Branded pens vs compounded tirzepatide: the split dosing divide
This distinction is fundamental. It determines whether split dosing is even possible for your situation. And getting it wrong carries real safety risks.
Why branded pens cannot be split
Mounjaro and Zepbound come in pre-filled, single-use auto-injector pens. Each pen contains one dose. There is no way to partially inject the contents and save the rest for later. The manufacturer, Eli Lilly, states explicitly that splitting doses is not possible with branded pens and should never be attempted.
Three critical reasons.
First, the pens contain no antimicrobial preservative. Once the seal is broken, there is nothing to prevent bacterial contamination of the remaining solution. A partially used pen sitting at room temperature becomes a bacterial growth medium within hours.
Second, the delivery mechanism is designed for single use. The auto-injector deploys once. You cannot partially activate it, stop midway, and come back later for the second half. The engineering does not allow it.
Third, dose accuracy becomes impossible. Even if you could somehow extract a partial dose, there is no reliable way to measure exactly how much remained in the pen. Dosing errors with a potent dual GIP/GLP-1 agonist carry real consequences.
Why compounded tirzepatide can be split
Compounded tirzepatide comes in multi-dose vials, not single-use pens. These vials contain lyophilized powder reconstituted with bacteriostatic water, which includes a benzyl alcohol preservative that prevents bacterial growth through multiple needle entries.
With a multi-dose vial, you draw the exact volume you need using an insulin syringe. Want to take 5mg on Monday and 5mg on Thursday instead of 10mg all at once? You simply draw half the weekly volume each time. The math is clean. The preservative protects the remaining solution. And the dosing accuracy depends on your syringe technique, which is controllable.
This is exactly why the reconstitution chart matters so much for split dosing. When you mix your tirzepatide with bacteriostatic water, the concentration you create determines how accurately you can measure split doses. Higher concentrations require smaller volumes per dose, which can be harder to measure precisely. Lower concentrations use larger volumes, making measurement easier but requiring more injection volume.
Storage considerations for split dosing
If you are using compounded tirzepatide for split dosing, the vial sits in the refrigerator between injections. Proper refrigeration between 36 and 46 degrees Fahrenheit is critical. The shelf life of reconstituted tirzepatide depends on storage conditions, and researchers should understand how long tirzepatide lasts in the fridge to avoid using degraded product.
Never leave the vial at room temperature for extended periods. If you need to travel with it, plan carefully. Understanding how long tirzepatide can be out of the fridge and what happens when it gets warm is essential for anyone splitting doses across multiple days.
How split dosing actually works
The concept is simple. The execution requires attention to detail.
The basic principle
Split dosing means dividing your total weekly tirzepatide dose into two (or occasionally three) smaller injections administered on different days. The total amount of tirzepatide per week stays exactly the same. You are not increasing your dose. You are not decreasing it. You are changing the delivery schedule.
Think of it like this. If your weekly dose is 10mg, you are currently getting all 10mg in one shot on one day. With split dosing, you would inject 5mg on Monday and 5mg on Thursday. Same total. Different distribution.
Timing the split injections
The most common split schedule divides the week into two roughly equal periods. For twice-weekly dosing, that means injections spaced approximately 3 to 4 days apart.
Popular schedules include Monday and Thursday. That gives you a 3-day gap followed by a 4-day gap. Some researchers prefer Monday and Friday for a cleaner 4-day and 3-day split. Others use Sunday and Wednesday. The exact days matter less than the spacing. You want roughly even intervals.
Avoid splitting into uneven gaps like Monday and Wednesday (2 days apart), because this creates one very short cycle and one very long cycle, defeating the purpose of smoothing out the concentration curve. Similarly, Monday and Saturday (5 days apart) leaves too long between the second and next first injection.
Even vs uneven splits
Most researchers divide the weekly dose evenly. A 10mg weekly dose becomes two 5mg injections. A 15mg weekly dose becomes two 7.5mg injections.
But even splits are not the only option. Some researchers use an uneven split where the larger dose comes at the beginning of the week. For example, a 10mg weekly dose might become 6mg on Monday and 4mg on Thursday. The rationale? You want slightly more suppression during weekdays when dietary discipline matters most, and you can tolerate a lighter effect on the weekend.
There is no published research comparing even versus uneven splits. This is entirely based on individual preference and practical experimentation. The microdosing tirzepatide chart provides additional context for those exploring non-standard dosing approaches.
The math of split dosing
Getting the volumes right requires understanding your vial concentration. Here is a practical example.
Say you have a 10mg vial of compounded tirzepatide reconstituted with 2 milliliters of bacteriostatic water. That gives you a concentration of 5mg per milliliter. For a total weekly dose of 10mg:
Once weekly: Draw 2 milliliters (the full vial), inject all at once
Split dosing: Draw 1 milliliter (5mg) on day one, draw 1 milliliter (5mg) on day four
For those working with different concentrations, the tirzepatide dosage chart in milliliters and the tirzepatide dosage chart in units provide the conversion references you need. Understanding the relationship between tirzepatide dosage in units and milligrams is essential for accurate split dosing.
Split dosing protocols by dose level
Not every dose splits equally well. Here are practical protocols for each standard tirzepatide dose level, along with realistic assessments of whether splitting makes sense at that dose.
2.5mg weekly (starting dose)
The starting dose of tirzepatide is 2.5mg per week. Splitting this into two 1.25mg injections is technically possible but rarely practical.
At 1.25mg per injection, you are working with very small volumes. With a concentration of 5mg per milliliter, each split dose would require drawing only 0.25 milliliters, which is 25 units on a standard insulin syringe. That is a tiny volume. Measurement errors become proportionally larger, and the actual drug concentration per injection may be insufficient to maintain meaningful receptor activation between doses.
The 2.5mg dose is an initiation dose, not a therapeutic dose. It exists to let your body adjust to the medication before escalating. Side effects at this level are typically mild. The wearing-off effect, while present, is expected because you have not yet reached steady state. Most researchers are better served by completing the standard 4-week initiation period on 2.5mg and then moving to 5mg rather than splitting the starting dose.
Recommendation: Do not split at 2.5mg. Proceed through standard titration.
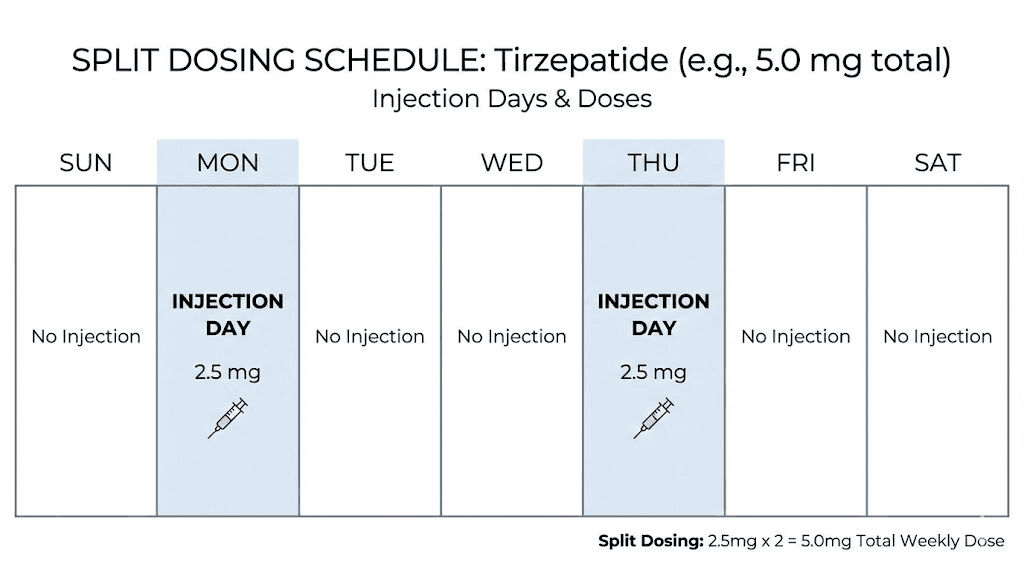
5mg weekly
Split protocol: 2.5mg on day one, 2.5mg on day four.
At 5mg, splitting becomes more practical. Each injection of 2.5mg still delivers a meaningful dose. This is the level where many researchers first notice the wearing-off effect strongly enough to consider alternatives.
The concentration math works cleanly. With a 10mg vial reconstituted in 2 milliliters, each 2.5mg dose requires 0.5 milliliters (50 units), which is easy to measure accurately.
Researchers at 5mg who experience significant end-of-week appetite return but tolerate the medication well otherwise are reasonable candidates for split dosing at this level. Check the compound tirzepatide dosing chart for volume references.
Recommendation: Reasonable to split if wearing-off is significant and you are using compounded formulation.
7.5mg weekly
Split protocol: 3.75mg on day one, 3.75mg on day four. Or asymmetric: 5mg on day one, 2.5mg on day four.
This dose level is where split dosing starts to show clear advantages for researchers with GI sensitivity. The 7.5mg dose represents the first true therapeutic escalation beyond the initiation doses, and the peak concentration after a single 7.5mg injection can trigger significant nausea in sensitive individuals.
Splitting to 3.75mg plus 3.75mg reduces the peak by roughly half while maintaining the same weekly exposure. For researchers who struggled with side effects during the standard titration schedule, this can mean the difference between tolerating the dose and needing to drop back down.
Recommendation: Good candidate for split dosing, especially during dose escalation.
10mg weekly
Split protocol: 5mg on day one, 5mg on day four.
Clean, even split. Each injection delivers the equivalent of a standard therapeutic dose. This is one of the most common split dosing protocols because the math is simple and the dose per injection is substantial enough to maintain strong receptor activation throughout the cycle.
At 10mg weekly, many researchers report excellent appetite suppression through day five with noticeable decline on days six and seven. Splitting to twice weekly often eliminates that late-week decline entirely, creating a more consistent experience. The 10mg dose in units is straightforward to calculate and measure.
Recommendation: Excellent candidate for split dosing. Clear benefits for most researchers.
12.5mg weekly
Split protocol: 6.25mg on day one, 6.25mg on day four. Or asymmetric: 7.5mg on day one, 5mg on day four.
The 12.5mg dose is not one of the standard FDA-approved dose levels (which jump from 10mg to 15mg), but it is commonly used in compounded protocols as an intermediate step. Splitting 12.5mg works well in practice. The tirzepatide dose chart can help map this to syringe volumes.
Recommendation: Strong candidate for split dosing, especially during the transition from 10mg to 15mg.
15mg weekly
Split protocol: 7.5mg on day one, 7.5mg on day four.
At the maximum approved dose, split dosing can be particularly valuable. A single 15mg injection represents a substantial drug load, and the peak concentration is the highest you will experience at any approved dose level. Researchers who tolerate 10mg well but struggle with the jump to 15mg often find that splitting resolves the issue.
The pharmacokinetic smoothing effect is most dramatic at this dose level because the absolute difference between peak and trough is largest. The 15mg dose in units requires careful measurement, and splitting it into two precise 7.5mg doses demands attention to reconstitution concentration.
Recommendation: Highly recommended for researchers experiencing side effects at 15mg or significant end-of-week wearing off.
When splitting your tirzepatide dose makes sense
Split dosing is not for everyone. It adds complexity and doubles the number of injections. But for specific situations, the benefits clearly outweigh the inconvenience.
Persistent GI side effects despite slow titration
You have followed the standard titration schedule. You have spent four weeks at each dose level. You have tried avoiding trigger foods. You have adjusted your meal timing and composition. And yet, every injection brings two days of misery. Nausea that makes eating impossible. Constipation that no amount of fiber fixes. Diarrhea that disrupts your routine.
If these side effects correlate specifically with the first two to three days post-injection and then improve, they are likely peak-related. Split dosing directly addresses peak-related side effects by lowering the Cmax.
Significant end-of-week appetite return
Your dose works beautifully for five days. Then the medication seems to vanish. Hunger returns to pre-treatment levels. Cravings become difficult to manage. And you spend the last day or two before your next injection fighting the same battles you started tirzepatide to escape.
This pattern is pharmacokinetic, not psychological. Your trough concentration is dropping below the therapeutic threshold. Split dosing lifts the trough by providing a second dose before levels fall too far. If you are experiencing this alongside stalled progress, check our guide on why weight loss stalls on tirzepatide for additional factors that may be contributing.
During dose escalation
The transition from one dose to the next is when side effects are most intense. Your body has adapted to the current dose, and the jump to a higher level creates a new, unfamiliar peak concentration. Split dosing during escalation allows you to increase total weekly exposure while keeping peak levels more manageable.
For example, instead of jumping from 5mg once weekly to 7.5mg once weekly, you could transition to 3.75mg twice weekly. Same total dose increase. But the peak after each 3.75mg injection is significantly lower than the peak after a single 7.5mg injection. This can make dose escalation smoother and more tolerable. The microdose tirzepatide schedule offers similar principles applied to even smaller increments.
Lower body weight researchers
If you weigh under 70 kilograms, your tirzepatide exposure per kilogram is significantly higher than someone weighing 100 kilograms on the same dose. The population pharmacokinetic data shows approximately 22 percent higher exposure at 70 kilograms versus the 90-kilogram reference. This means your peaks are sharper, your troughs are deeper (proportionally), and the wearing-off effect may be more pronounced.
Split dosing helps equalize the experience for lower body weight researchers by reducing that proportionally higher peak. If you are not sure what dose makes sense for your body weight, the peptide calculator can help estimate appropriate levels.
When you should NOT split your tirzepatide dose
Split dosing is a tool, not a universal improvement. Some situations make it inappropriate, unnecessary, or potentially harmful.
If you are using branded pens
This cannot be stated strongly enough. Mounjaro and Zepbound pens are single-use devices containing preservative-free solutions. Do not attempt to partially inject, save, and reuse the remainder. Do not try to extract solution from the pen into a syringe. Do not store a partially used pen. The contamination risk is real and the dosing accuracy is impossible to guarantee.
If you want to explore split dosing, you need to be using compounded tirzepatide from a multi-dose vial with bacteriostatic water. That is the only safe option for splitting.
If you are tolerating once-weekly dosing well
If your current schedule works, do not fix what is not broken. Split dosing adds complexity. Twice as many injections means twice as many injection sites to manage, twice as many opportunities for measurement errors, and twice as many disruptions to your weekly routine.
Some researchers explore split dosing out of curiosity rather than necessity. If you are losing weight steadily, tolerating your dose, and not experiencing significant wearing-off symptoms, the standard once-weekly protocol is working as designed. There is no evidence that split dosing produces better outcomes when the once-weekly protocol is already effective.
Without medical supervision
Split dosing is off-label. No clinical trial has evaluated tirzepatide given twice weekly. The pharmacokinetic reasoning is sound, and the logic is straightforward, but you are making changes to a potent medication protocol without direct clinical evidence supporting the modification.
This is a conversation to have with your healthcare provider. They can evaluate your specific situation, review your side effect history, and help determine whether the potential benefits justify the change. They can also monitor for unexpected effects that might arise from the altered concentration profile.
At starting doses during initiation
The first four to eight weeks on tirzepatide involve deliberate adaptation. Your body is adjusting to a new dual receptor agonist. Side effects during this period are expected and usually temporary. The timeline for tirzepatide to start working involves several weeks of adaptation that should not be short-circuited by prematurely modifying the dosing schedule.
Give the standard protocol a fair chance at each dose level before concluding that split dosing is necessary. Many side effects that feel unbearable in week one of a new dose resolve significantly by week three or four as your body adapts.
Managing side effects without splitting your dose
Before committing to split dosing, consider whether other adjustments might solve the problem with less complexity. Several strategies can reduce side effects or extend the effective duration of each dose without changing the injection schedule.
Slower titration
The standard protocol calls for four weeks at each dose level before escalating. But four weeks is a minimum, not a mandate. Spending six or eight weeks at a dose level gives your body more time to adapt, often reducing the side effects that make a higher dose feel intolerable.
The compound tirzepatide dosage chart includes intermediate dose levels (3.75mg, 6.25mg, 8.75mg, 11.25mg, 12.5mg) that are not available in branded pens but are standard in compounded protocols. Using these intermediate steps creates a gentler titration curve. Instead of jumping 2.5mg at a time, you increase by 1.25mg. The peaks are less dramatic. The adaptation is smoother.
Dietary adjustments
What you eat, and when, significantly impacts how tirzepatide side effects manifest. On injection day and the day after, eating smaller, more frequent meals can reduce nausea. Avoiding high-fat and greasy foods during the peak concentration window helps because tirzepatide slows gastric emptying, and heavy foods sit in the stomach longer.
Our guides on what to eat on tirzepatide and foods to avoid on tirzepatide provide specific recommendations. For a complete nutritional strategy, the tirzepatide meal plan maps out an entire week of eating optimized for the medication cycle.
For the end-of-week wearing-off effect specifically, protein-rich foods and high-fiber meals help maintain satiety even as drug levels decline. The right supplements alongside tirzepatide can also support appetite control and manage common nutritional gaps.
Hydration strategy
Dehydration worsens nausea, headaches, and fatigue. Tirzepatide reduces appetite, which means many researchers eat less, which means they also drink less, which compounds the side effects into a cycle that feels worse than the medication alone warrants.
Aim for at least 64 ounces of water daily. More on injection day. Electrolyte supplements can help if plain water is difficult to keep down during peak nausea periods.
Injection timing optimization
When you inject matters more than most researchers realize. Injecting in the evening means peak concentration hits while you are sleeping, which can blunt nausea symptoms because you sleep through the worst of them. Injecting in the morning means the peak hits during your active hours, when nausea is more disruptive.
The best time to take your GLP-1 shot depends on your personal schedule and side effect pattern. Experiment with different injection times before concluding that the only solution is split dosing.
Formulation changes
Different tirzepatide formulations can produce different side effect profiles. Tirzepatide with B12 may help with fatigue and energy. Tirzepatide with glycine can improve GI tolerability. Tirzepatide with niacinamide addresses metabolic support. The glycine and B12 compound combines multiple benefits.
Before splitting your dose, ask whether a different formulation might address your specific concerns more effectively.
The science behind peak and trough drug levels
Understanding what happens inside your body during a once-weekly versus twice-weekly dosing cycle helps explain why split dosing works for some researchers and why it is unnecessary for others.
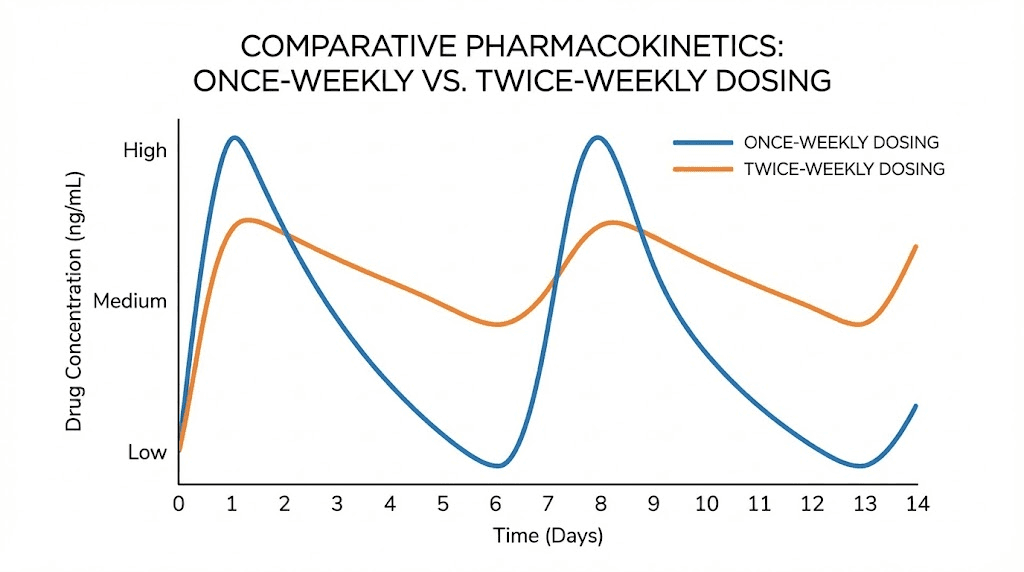
Once-weekly dosing: the roller coaster
With standard once-weekly dosing, the concentration profile looks like a series of peaks and valleys. Injection day creates a rapid rise in blood levels. Depending on your individual absorption rate, concentration peaks within 8 to 72 hours. Then a steady decline begins. By day five, roughly half the drug has been eliminated. By day seven, concentration has fallen further, though it remains above zero because the next dose adds to whatever remains.
At steady state (after four to five weeks on a given dose), the trough concentration, the lowest point just before your next injection, stabilizes. The 1.7-fold accumulation means your trough at steady state is meaningfully higher than your trough after the first injection. But the peak-to-trough ratio remains the same, and that ratio is what drives the roller coaster feeling.
The peak-to-trough ratio is most dramatic at lower doses. At 2.5mg, the absolute difference between peak and trough is small in milligrams but large relative to the total dose. At 15mg, the absolute difference is larger but proportionally similar. This is why some researchers tolerate once-weekly dosing fine at 15mg but struggled at 5mg, the relative fluctuation is the same but the absolute concentrations at the trough are much higher at 15mg, staying above the therapeutic threshold throughout the cycle.
Twice-weekly dosing: the smooth wave
Split dosing compresses the cycle. Instead of seven days between doses, you have three to four days. The drug does not have time to fall as far before the next injection tops it up.
The peak after each split dose is lower than the peak after the full weekly dose. How much lower depends on the split ratio, but for an even 50/50 split, the peak is roughly half what it would be with the full dose. The trough, conversely, is higher. The drug does not have seven days to decline; it only has three to four days before the next dose arrives.
The result is a narrower range between peak and trough. Less of a roller coaster. More of a gentle wave. The total area under the curve (total weekly drug exposure) remains the same, but the experience of that exposure changes fundamentally.
Why some people are more sensitive to fluctuations
Pharmacokinetic sensitivity varies between individuals. Some researchers barely notice the difference between peak and trough days. Others feel every percentage point of decline acutely. Several factors contribute to this variability.
Body composition affects distribution volume. Researchers with lower body fat may have different distribution kinetics than those with higher body fat, since tirzepatide distributes into adipose tissue.
Genetic variations in GLP-1 and GIP receptor density and sensitivity mean that some people need higher absolute concentrations to maintain the same level of receptor activation. For these individuals, even modest trough-level declines cross below their personal therapeutic threshold.
Metabolic rate affects clearance speed. Researchers with faster metabolism may eliminate tirzepatide more quickly, shortening the effective duration of each dose. If you notice that tirzepatide seems to stop working toward the end of each week, faster individual clearance may be a contributing factor.
Practical considerations for split dosing tirzepatide
The pharmacokinetics make sense on paper. But living with a split dosing schedule introduces real-world logistics that you should think through before making the switch.
Twice as many injections
This is the most obvious trade-off. Instead of one injection per week, you are doing two. For researchers who dislike injections, this doubles the unpleasant part of the protocol. For those who are indifferent to the injection itself, it is a minor inconvenience.
Each injection requires clean technique. Proper injection site selection, alcohol swab preparation, and correct injection technique apply to every injection, not just the once-weekly one. Cutting corners on hygiene because the doses are smaller is not acceptable.
Injection site management
With twice-weekly injections, you need to rotate injection sites more carefully. Injection site reactions including redness, swelling, and itchiness can develop more readily when the same area receives repeated injections without adequate rotation.
Use a systematic rotation pattern. Alternate between left and right sides of the abdomen, upper thighs, and backs of the upper arms. Never inject into the same spot within a two-week window. Keep a simple log of which site you used on which day.
Dosing accuracy with smaller volumes
Split dosing means drawing smaller volumes per injection. Smaller volumes require more precise syringe technique. A measurement error of 2 units on a 50-unit draw represents a 4 percent dose variance. The same 2-unit error on a 100-unit draw is only 2 percent.
Use insulin syringes with markings appropriate for your dose volume. For very small doses, consider syringes with half-unit markings for maximum precision. The tirzepatide syringe dosage guide explains how to select the right syringe for your specific protocol.
Reconstitution concentration also matters enormously for split dosing accuracy. If you reconstitute too concentrated, the volume per split dose becomes tiny and hard to measure. Too dilute, and each injection involves uncomfortably large volumes. Find the sweet spot that gives you measurable, manageable volumes for each split dose. The peptide reconstitution calculator helps determine the right water volume for your target concentration.
Record keeping
Split dosing demands more organized tracking than once-weekly dosing. You need to record the date, time, dose amount, injection site, and any side effects for each injection. You also need to track total weekly consumption to ensure you are not inadvertently under-dosing or over-dosing over time.
A simple spreadsheet or notebook works. The key is consistency. Skipping entries leads to confusion about which dose comes next, and confusion about dosing potent medications is something to avoid completely.
Travel and schedule disruptions
Once-weekly dosing is forgiving of schedule shifts. If your injection day is Monday but you travel on Monday, injecting Tuesday instead shifts everything by one day, and the impact is minimal.
Split dosing has less buffer. If your schedule is Monday and Thursday but Thursday becomes impossible, you need to decide whether to inject on Wednesday (two-day gap), Friday (one-day gap between Friday and the following Monday), or skip the Thursday dose entirely. Each option has different implications for your concentration profile.
Travelers who split dose should plan their injection schedule around their itinerary. Ensure proper storage, since tirzepatide temperature management during travel requires preparation.
Cost considerations
Split dosing does not change the amount of tirzepatide you use per week, so the per-milligram cost remains the same. However, you will use twice as many syringes, alcohol swabs, and possibly more bacteriostatic water (depending on your vial management). These are minor costs but worth acknowledging. For those budget-conscious about their protocol, our guides on affordable tirzepatide and cost-effective compounded tirzepatide provide relevant context, and the peptide cost calculator can help estimate total protocol expenses.
How to transition from once weekly to split dosing
If you have decided that split dosing is right for your situation, the transition should be methodical, not impulsive. Here is a step-by-step approach.
Step one: establish your baseline
Before changing anything, spend two to three weeks carefully tracking your current experience. Note when side effects peak, when they resolve, when appetite returns, and how you feel on each day of the weekly cycle. This baseline data tells you whether split dosing actually improves things once you switch.
Without a baseline, you have no way to objectively evaluate whether the new schedule is better. Memory is unreliable when it comes to gradual changes in how you feel.
Step two: calculate your split doses
Determine your total weekly dose. Divide it according to your chosen split ratio (even or uneven). Convert the split doses to syringe volumes based on your reconstitution concentration.
Double-check the math. Then check it again. The tirzepatide dosage calculator can verify your calculations. Getting the volumes wrong means getting the doses wrong, and dosing errors compound over weeks.
For the most common scenarios:
Step three: choose your schedule
Pick two days that work consistently with your lifestyle. The most important factor is that you can reliably inject on those days every week. A schedule you skip frequently is worse than the once-weekly protocol you are replacing.
Common schedules: Monday/Thursday, Tuesday/Friday, Sunday/Wednesday. Space injections 3 to 4 days apart. Mark them in your calendar. Set reminders.
Step four: make the switch
On your last once-weekly injection day, take your normal full dose. Then, seven days later, start the split schedule. Do not take a full dose and then start splitting three days later. That would mean excess medication in the first week.
Week one of split dosing may feel slightly different as the concentration profile adjusts. Give it at least three to four weeks before evaluating results, because you need to reach a new steady state with the altered injection schedule.
Step five: evaluate and adjust
After four weeks of split dosing, compare your experience to the baseline you established in step one. Are side effects reduced? Is end-of-week appetite better controlled? Is your overall experience more consistent?
If the answer to all three is yes, continue the split protocol. If some aspects improved but others did not, consider adjusting the split ratio or timing. If nothing improved or things got worse, return to once-weekly dosing. Not every modification works for every researcher.
Comparing split dosing with other dosing strategies
Split dosing is one of several non-standard approaches researchers use. Understanding how it compares to alternatives helps you choose the right strategy for your situation.
Split dosing vs microdosing
Microdosing tirzepatide involves taking doses smaller than the standard protocol, often daily or every other day. While split dosing maintains the same total weekly dose divided into fewer injections, microdosing reduces the total weekly dose itself.
The goals are different. Split dosing aims to smooth the concentration curve at full therapeutic levels. Microdosing aims to find the minimum effective dose for side effect minimization. Some researchers combine elements of both, using lower-than-standard doses administered more frequently, but this moves further from established protocols and requires even more careful monitoring. The microdose schedule provides detailed frameworks for this approach.
Split dosing vs dose reduction
If side effects are the primary concern, reducing the dose is the simplest solution. Instead of splitting 10mg into two 5mg injections, simply take 7.5mg once weekly. This reduces peak concentration without adding injection frequency.
The trade-off is lower total weekly exposure, which may mean less appetite suppression and slower weight loss. Split dosing maintains the higher exposure while reducing peaks, preserving the therapeutic benefit of the full dose. For researchers who need the full dose efficacy but cannot tolerate the full dose peak, splitting offers something that dose reduction cannot.
Split dosing vs switching medications
If tirzepatide side effects remain intolerable despite split dosing, switching to semaglutide is an option worth discussing with your provider. Semaglutide has different pharmacokinetics and activates only GLP-1 receptors (not dual GIP/GLP-1 like tirzepatide), which may produce a different side effect profile.
The semaglutide versus tirzepatide comparison explores the differences in detail. If you decide to switch, the conversion chart and switching dosage guide provide the transition frameworks.
For researchers interested in newer alternatives, articles covering survodutide versus tirzepatide, CagriSema versus tirzepatide, and orforglipron versus tirzepatide explore the next generation of GLP-1 medications. Retatrutide, a triple receptor agonist, represents another emerging option for researchers who have exhausted tirzepatide optimization strategies.
Safety considerations and risks of split dosing
Split dosing is generally considered low-risk when performed correctly with compounded formulations under medical guidance. But "low risk" is not "no risk," and understanding the potential issues helps you manage them proactively.
Drug accumulation with improper spacing
If injections are spaced too close together, tirzepatide concentration can build to levels higher than intended. The 5.4-day half-life means the drug is still substantially present three to four days after injection. Adding a second dose at that point does elevate the baseline compared to injecting into a deeper trough at seven days.
At standard split spacing (3 to 4 days), the accumulation effect is modest and generally well-tolerated because each individual dose is proportionally smaller. But if you accidentally inject two doses one day apart, or forget which day you are on and double up, the resulting concentration spike can cause significant side effects including severe nausea, gastrointestinal distress, and potentially hypoglycemia in those also taking diabetes medications.
Contamination risk with multi-dose vials
Every time you insert a needle into a multi-dose vial, you introduce a small contamination risk. With once-weekly dosing, the vial may only need four to eight entries (depending on vial size). With split dosing, that number doubles. Bacteriostatic water contains benzyl alcohol as a preservative, which provides protection, but the risk increases with more entries.
Mitigate this by swabbing the vial septum with alcohol before every draw, using a new needle each time, and monitoring the solution for any cloudiness, particles, or discoloration. If anything looks wrong, discard the vial. Understanding tirzepatide expiration and what happens with expired tirzepatide adds another layer of safety awareness.
Lack of clinical trial data
No randomized controlled trial has compared once-weekly tirzepatide to twice-weekly tirzepatide. The SURPASS and SURMOUNT studies that established efficacy and safety all used once-weekly dosing. While the pharmacokinetic reasoning for split dosing is sound, and while individual reports suggest it works well for many researchers, the formal evidence base does not yet exist.
This means unexpected interactions or effects are theoretically possible, though unlikely given the straightforward pharmacokinetic principles involved. It also means your healthcare provider may not be familiar with split dosing protocols and may be cautious about endorsing the approach.
Interaction considerations
If you are taking other medications alongside tirzepatide, the altered concentration profile from split dosing could theoretically affect drug interactions. Tirzepatide slows gastric emptying, which affects the absorption of oral medications. With split dosing, the gastric emptying effect is more constant throughout the week rather than strongest at the peak and weakest at the trough.
For most medications, this is unlikely to create clinically significant changes. But if you are on medications with narrow therapeutic windows (like certain heart medications or blood thinners), discuss the potential implications with your provider. If you are taking phentermine alongside tirzepatide, the interaction profile may shift slightly with split dosing.
What the research community reports
While formal clinical trials on split dosing do not exist, the GLP-1 research community has generated substantial anecdotal evidence. These reports are not clinical data, but they provide useful directional information.
Side effect improvement
The most consistently reported benefit is reduced nausea. Researchers who switched from once-weekly to twice-weekly dosing frequently describe a dramatic reduction in post-injection nausea. Instead of two days of significant nausea followed by five days of relative comfort, they experience mild or no nausea throughout the week. The side effect profile comparison between different GLP-1 medications provides additional context for what to expect.
More consistent appetite suppression
Many researchers report that split dosing eliminates or significantly reduces the end-of-week wearing-off effect. Instead of feeling the medication "turn off" on day six, they maintain relatively consistent appetite control throughout the entire week. This consistency often translates to better dietary adherence and more predictable weight loss patterns.
Improved energy levels
Some researchers report more stable energy throughout the week with split dosing. The once-weekly cycle can create energy fluctuations that mirror the drug concentration curve, with fatigue peaking alongside drug concentration and then returning in a different form as levels drop. Split dosing appears to moderate these fluctuations for some individuals.
No significant difference in weight loss
Importantly, researchers who switch to split dosing from once-weekly dosing at the same total weekly dose generally report similar weight loss outcomes. This aligns with the pharmacokinetic expectation: same total exposure should produce the same metabolic effects. The benefit is in tolerability and consistency, not in enhanced efficacy.
If your weight loss has stalled regardless of dosing schedule, the issue may lie elsewhere. Multiple factors beyond dosing schedule can cause tirzepatide to appear ineffective, and the expected weight loss timeline provides benchmarks for realistic expectations. Looking at tirzepatide before and after results and documented outcomes can help calibrate what to expect at each dose level.
Split dosing and other tirzepatide formulations
Tirzepatide is not limited to subcutaneous injection anymore. Understanding how split dosing relates to alternative formulations helps researchers evaluate all their options.
Oral tirzepatide
Oral tirzepatide is an emerging formulation that bypasses the injection question entirely. Daily oral dosing naturally creates a smoother concentration profile than weekly injections. For researchers whose primary motivation for split dosing is avoiding the peaks and troughs of weekly injections, oral formulations may eventually provide a better solution.
The comparison between oral versus injectable tirzepatide and the broader tablets versus injections analysis explore the trade-offs in detail.
Tirzepatide drops
Sublingual tirzepatide drops represent another alternative delivery method. Like oral tablets, drops can be administered daily, creating a naturally smoother pharmacokinetic profile without the need to manually split injectable doses.
Enhanced formulations
Compounded tirzepatide often includes additional ingredients that may influence the split dosing decision. B6-enhanced formulations, methylcobalamin compounds, and glycine-enhanced versions each address specific concerns. The color of different formulations can also help verify which compound you are working with.
For researchers serious about optimizing their tirzepatide protocols, SeekPeptides offers comprehensive guidance on formulation selection, dosing strategies, and protocol customization. Members access detailed comparison tools, expert-reviewed protocols, and a community of experienced researchers who have navigated these exact decisions.
Common split dosing mistakes to avoid
Researchers who have tried split dosing report a consistent set of avoidable errors. Learning from these mistakes saves time, frustration, and potentially wasted medication.
Mistake one: changing total weekly dose when switching to split
The most fundamental error is accidentally increasing or decreasing total weekly intake during the transition. Some researchers confuse splitting with dose escalation, thinking that two 5mg injections per week means they have moved from 5mg to 10mg weekly. Others reduce their total dose because the individual injections feel small. Neither is correct.
If your weekly dose is 10mg, split dosing means two 5mg injections. Not two 10mg injections. Not two 7.5mg injections. The total must remain exactly the same. Use the tirzepatide conversion chart to verify your numbers before starting any new protocol.
Mistake two: inconsistent timing between split doses
Split dosing works because it creates a predictable, smoother concentration curve. That predictability depends on consistent injection timing. Researchers who inject at random intervals, sometimes two days apart, sometimes five days apart, undermine the entire purpose of the split. The concentration profile becomes erratic rather than smooth.
Choose your two days and stick with them. Set calendar reminders. Treat each injection day with the same discipline you applied to your once-weekly schedule.
Mistake three: using the wrong reconstitution concentration
When you reconstitute your tirzepatide vial, the amount of bacteriostatic water you add determines the concentration. If you use too much water, each split dose requires a large injection volume. If you use too little, each split dose requires a tiny volume that is difficult to measure accurately.
For split dosing, aim for a concentration that puts each individual dose at 0.25 to 0.5 milliliters. This range is easy to measure with standard insulin syringes and comfortable to inject. The reconstitution chart provides the math for common vial sizes and target concentrations.
Mistake four: not tracking results objectively
Many researchers switch to split dosing and immediately declare it better or worse based on how they feel in the first few days. This is premature. The body needs three to four weeks to establish a new steady state on the split schedule. Changes in appetite, side effects, and energy during the first week may reflect the transition period, not the true experience of split dosing.
Track quantifiable metrics. Weekly weight. Waist measurement. Daily appetite rating on a 1-to-10 scale. Side effect severity. Compare week four of split dosing to your pre-switch baseline, not to the confusing first week of transition.
Mistake five: splitting branded pens by extracting solution
Some researchers attempt to draw solution from a branded pen using a syringe. This is dangerous for multiple reasons. The solution contains no preservative. The pen mechanism is not designed for this. The extracted volume cannot be measured precisely because pen mechanics vary. And the remaining solution in the pen degrades rapidly once the seal is compromised.
If you want to split dose, use compounded tirzepatide. There is no safe workaround for branded pens.
Mistake six: neglecting injection site rotation
With twice-weekly injections, the same injection site gets used twice as frequently if you are not careful about rotation. This accelerates lipohypertrophy (fatty tissue buildup at injection sites), which ironically impairs absorption of future injections, creating a self-defeating cycle.
Maintain at least six distinct injection sites and rotate systematically. The abdomen alone offers four quadrants. Add both thighs and both arms, and you have eight sites to rotate through, ensuring no single area is used more than once every four weeks.
Long-term considerations for split dosing
Split dosing is not just a short-term fix for acute side effects. Researchers who adopt it often continue for months or longer. Understanding the long-term implications helps set realistic expectations.
Adaptation over time
Just as the body adapts to once-weekly tirzepatide over the first few months, it adapts to the split dosing pattern as well. Some researchers find that the benefits of split dosing become less pronounced over time as their bodies adjust to the drug entirely. The nausea that motivated the switch may have resolved on its own with continued use.
Consider periodically retesting once-weekly dosing, perhaps every three to six months, to see whether you still need the split. Your body at month six is pharmacologically different from your body at month one, and the split dosing that was necessary early may become optional later.
Dose escalation with split dosing
If you are already split dosing at one level and need to escalate, increase the total weekly dose while maintaining the split. For example, moving from 10mg weekly (5mg + 5mg) to 12.5mg weekly (6.25mg + 6.25mg). Increase gradually, just as you would with once-weekly dosing.
Some researchers find that split dosing makes dose escalation smoother because each individual increase is smaller. Going from 5mg to 6.25mg per injection feels less dramatic than going from 10mg to 12.5mg in a single shot. This can allow faster titration to the target dose with fewer side-effect-related setbacks.
Maintaining compliance over months
The biggest long-term risk of split dosing is adherence fatigue. Two injections per week, every week, for months on end requires sustained discipline. Missing occasional doses is human. But with split dosing, a missed dose disrupts the concentration profile more quickly because there is less drug in reserve from each individual injection.
If you find yourself frequently missing split doses, consider whether the complexity is sustainable for your lifestyle. A consistently administered once-weekly dose may produce better results than an inconsistently administered split dose, even if the split is theoretically superior. The long-term treatment duration for GLP-1 medications is measured in months to years, making sustainable protocols essential.
Frequently asked questions
Is splitting my tirzepatide dose safe?
Splitting a compounded tirzepatide dose from a multi-dose vial is generally considered safe when done correctly. The total weekly dose remains the same, and the pharmacokinetic principles are straightforward. However, this is an off-label modification not evaluated in clinical trials, so it should be discussed with your healthcare provider. Never split branded pens like Mounjaro or Zepbound.
Can I split my Mounjaro or Zepbound pen?
No. Branded pens are single-use devices containing preservative-free solution. They cannot be partially injected and stored. Attempting to split a branded pen risks contamination, dose inaccuracy, and medication degradation. Split dosing is only possible with compounded tirzepatide in multi-dose vials containing bacteriostatic water.
How far apart should my split doses be?
Space split doses 3 to 4 days apart for optimal concentration balance. Common schedules include Monday/Thursday (3+4 day gaps) and Tuesday/Friday (3+4 day gaps). Avoid spacing less than 3 days apart, as this does not allow sufficient time between peaks and may increase side effects.
Will I lose more weight with split dosing?
Split dosing is not expected to increase weight loss compared to once-weekly dosing at the same total weekly dose. The total drug exposure over the week is the same, which means the metabolic effects should be equivalent. The benefit of split dosing is improved tolerability and more consistent appetite control, which may indirectly support better adherence and outcomes.
Does splitting reduce nausea?
For many researchers, yes. GI side effects are largely driven by peak drug concentration. Split dosing reduces the peak by distributing the dose across two smaller injections. If your nausea is worst in the first 24 to 48 hours after injection and then improves, it is likely peak-related and may respond well to split dosing.
Can I split semaglutide the same way?
Semaglutide has a longer half-life (approximately 7 days) compared to tirzepatide (approximately 5 days), which means the wearing-off effect is less pronounced with semaglutide. Split dosing semaglutide is less commonly discussed for this reason. However, the same general principles apply if using compounded semaglutide from multi-dose vials. The semaglutide dosage guide provides relevant information for researchers considering this approach.
What if I miss a split dose?
If you miss one of your split doses by a day or less, take it as soon as you remember and adjust the timing of your next injection accordingly. If you miss it by more than a day, skip the missed dose and take your next scheduled split dose at the regular time. Do not double up to compensate, as this defeats the purpose of split dosing and creates the same high peak you were trying to avoid.
Can I split my dose into three injections per week?
Technically yes, with compounded formulations. Some researchers use a Monday/Wednesday/Friday schedule with each injection containing one-third of the weekly dose. This creates an even smoother concentration profile but adds a third injection to the weekly routine. The benefit over twice-weekly dosing is marginal for most people, and the added complexity rarely justifies the slight improvement in concentration stability.
External resources
For researchers serious about optimizing every aspect of their peptide protocols, SeekPeptides provides the most comprehensive resource available. Members access evidence-based dosing guides, protocol optimization tools, and a community of thousands who have navigated these exact questions, from split dosing decisions to formulation comparisons to long-term protocol management.