Feb 25, 2026

Conflicting information about GLP-1 medications floods every corner of the internet. Search this question and you will find answers ranging from dangerously vague to misleadingly optimistic. Some sources suggest combining semaglutide and tirzepatide could accelerate results. Others say absolutely not. Very few explain the pharmacology behind the answer.
Can you take semaglutide and tirzepatide together? The question seems straightforward. The answer is not.
No. You should not take them together. But understanding why requires more than a one-word response, and the reasoning matters for anyone trying to get the most from their weight loss or blood sugar management protocol. These two medications share overlapping mechanisms of action, target many of the same receptors, and produce similar side effects. Combining them does not double the benefits. It amplifies the risks. The FDA has not approved this combination for any purpose. Clinical guidelines specifically advise against stacking incretin-based therapies. And the pharmacology behind receptor saturation explains exactly why more GLP-1 activation does not translate into more weight loss.
This guide covers the complete picture. You will learn the specific molecular reasons why combining these medications fails, what the landmark SURPASS-2 trial revealed when comparing them head-to-head, the exact protocols for safely switching between them, and evidence-based strategies for getting better results from a single medication than most people expect from two. Whether you have hit a semaglutide plateau and wonder if adding tirzepatide could help, or you are comparing tirzepatide vs semaglutide side effects to choose between them, the answer starts with understanding how each medication works at the cellular level. SeekPeptides built this guide to be the most thorough resource available on this exact question.
The short answer
You cannot safely take semaglutide and tirzepatide at the same time. No clinical trial has tested this combination in humans. The FDA has not approved it. And the pharmacological reasoning is clear: both medications activate the GLP-1 receptor, which means taking both would oversaturate a single pathway without meaningful additional benefit.
Think of it this way. If you are already receiving optimal GLP-1 receptor stimulation from semaglutide, adding tirzepatide does not unlock new receptors. Those receptors are already occupied. The additional GLP-1 activation has nowhere productive to go, and instead creates excessive signaling that translates directly into amplified side effects.
Tirzepatide does offer something semaglutide cannot: GIP receptor activation. But that advantage is already built into tirzepatide as a standalone medication. You do not need semaglutide to get it. The entire point of choosing between these medications is selecting the one that best matches your needs, not combining them and hoping the effects stack. The right question is not whether you can take both. The right question is which one works best for your situation, and what to do if it stops delivering results.
How semaglutide works inside the body
Understanding why these medications cannot be combined requires first understanding how each one works independently. Semaglutide belongs to a class of drugs called GLP-1 receptor agonists. These medications mimic the action of glucagon-like peptide-1, a hormone your gut naturally produces after eating. The synthetic version is engineered to last much longer in the body than the natural hormone, which degrades within minutes. Semaglutide has a half-life of approximately seven days. This is why it works as a once-weekly injection and why understanding GLP-1 medications matters for anyone considering combination therapy.
GLP-1 receptor activation
When semaglutide binds to GLP-1 receptors in the pancreas, it triggers a cascade of metabolic responses. The first and most immediate is glucose-dependent insulin secretion. This means the medication stimulates insulin release only when blood sugar levels are elevated, which is why GLP-1 agonists carry a lower risk of hypoglycemia compared to older diabetes medications like sulfonylureas.
The receptor activation also suppresses glucagon, the hormone responsible for telling the liver to release stored glucose into the bloodstream. Together, these effects create a powerful two-directional control over blood sugar levels.
But semaglutide does not just work in the pancreas. GLP-1 receptors exist throughout the body, including the brain, the stomach, the heart, and the kidneys. When semaglutide activates receptors in the hypothalamus, it directly influences appetite regulation and satiety signaling. This is the mechanism behind the dramatic appetite suppression that makes these medications so effective for weight loss. Understanding how quickly this appetite effect develops helps set realistic expectations for anyone starting therapy.
Appetite suppression and gastric emptying
The weight loss effects of semaglutide come from two complementary mechanisms. First, the direct appetite suppression through hypothalamic receptor activation reduces hunger signals and increases feelings of fullness after smaller meals. People taking semaglutide consistently report thinking about food less frequently and feeling satisfied with smaller portions.
Second, semaglutide significantly slows gastric emptying. Food stays in the stomach longer. This prolongs the feeling of fullness after eating. This is also why nausea is the most common side effect, particularly during dose escalation. The stomach is holding food longer than it normally would, and the body needs time to adjust. Managing this adjustment period with proper nutrition planning and understanding which foods to avoid makes a meaningful difference in tolerability.
Together, these mechanisms typically produce weight loss of 12-15% of total body weight in clinical trials. Real-world results vary, and some people experience a plateau after several months. Our guide on why you might not be losing weight on semaglutide covers the most common reasons for stalled progress and practical solutions. Tracking progress with realistic benchmarks, like those in our semaglutide one month results guide, helps keep expectations aligned with typical outcomes.
Blood sugar regulation beyond the pancreas
Semaglutide also influences blood sugar through mechanisms beyond simple insulin and glucagon modulation. Research shows it improves beta cell function over time, meaning the insulin-producing cells in the pancreas work more efficiently with continued use. It reduces hepatic glucose output, slowing liver production of new glucose molecules. And the cardiovascular benefits, which initially surprised researchers, appear connected to anti-inflammatory effects and improvements in endothelial function.
For researchers tracking their progress, understanding semaglutide dosage in units and how the medication scales across different concentrations is essential. Our semaglutide 5mg/ml dosage chart breaks down exact measurements for compounded formulations, while the semaglutide dosage calculator handles the math automatically. Understanding unit-to-milligram conversions becomes especially important when comparing doses between compounded semaglutide and brand-name formulations.
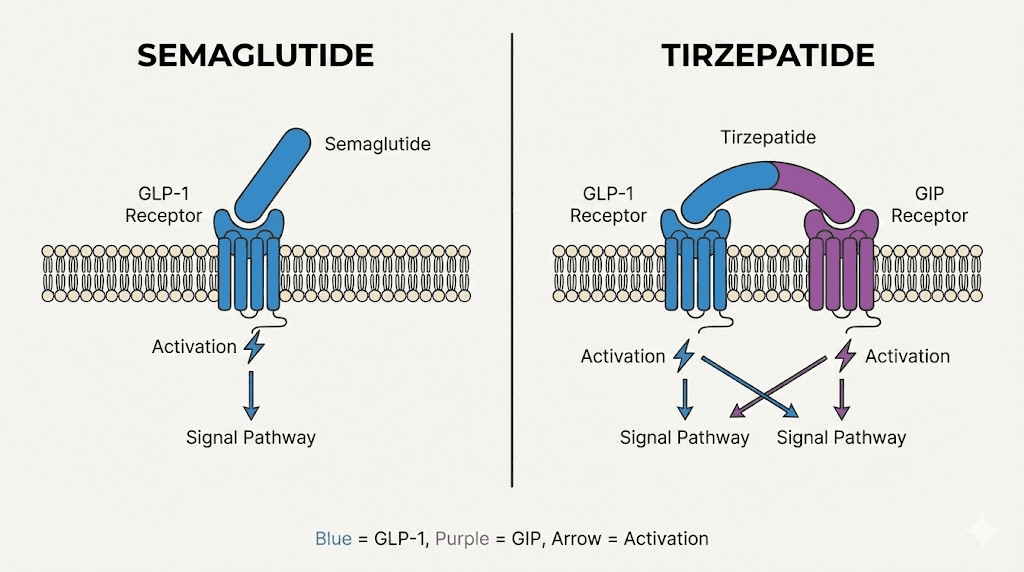
How tirzepatide works differently
Tirzepatide is not simply a stronger version of semaglutide. It is a fundamentally different type of molecule. While semaglutide activates only GLP-1 receptors, tirzepatide activates two receptor types simultaneously: GLP-1 receptors and GIP receptors. GIP stands for glucose-dependent insulinotropic polypeptide, another incretin hormone that plays a distinct role in metabolism. This dual mechanism is why tirzepatide is classified as a twincretin rather than a simple GLP-1 agonist, and understanding how tirzepatide works is essential before considering any combination approach.
Dual GIP and GLP-1 receptor agonism
The GIP receptor and the GLP-1 receptor serve different but complementary functions. GLP-1 receptor activation suppresses appetite, slows gastric emptying, and improves glucose-dependent insulin secretion. GIP receptor activation does something additional. It enhances fat metabolism, improves insulin sensitivity in adipose tissue, and may play a role in how the body distributes and utilizes stored energy.
Research published in JCI Insight showed that co-infusion of GLP-1 and GIP produces a synergistic effect, resulting in significantly increased insulin response and glucagonostatic response compared to separate administration of each hormone. This synergy is the entire reason tirzepatide was developed. Rather than simply adding more GLP-1 stimulation (which hits a ceiling), the addition of GIP receptor activation opens a second pathway that works alongside GLP-1 signaling.
This is also the critical reason why combining semaglutide with tirzepatide makes no pharmacological sense. Tirzepatide already provides GLP-1 receptor activation. Adding semaglutide on top of it would not open a new pathway. It would just pile more agonism onto a receptor that tirzepatide is already activating. For a complete breakdown of dosing differences, our tirzepatide dosage in units guide and tirzepatide dose chart cover the specifics.
Why dual action translates to greater weight loss
Clinical trial data consistently shows tirzepatide produces more weight loss than semaglutide. The landmark SURPASS-2 trial compared them directly, and we will cover those results in detail later in this guide. But the reason for the difference comes down to complementary receptor activation.
GLP-1 receptor activation primarily drives appetite reduction and gastric slowing. GIP receptor activation primarily drives metabolic improvements, fat utilization, and insulin sensitivity. When both pathways activate simultaneously, the body experiences a more comprehensive metabolic shift than either pathway could achieve alone.
This also explains tirzepatide weight loss timelines. Many researchers report noticeable results within the first four weeks, with progressive improvements over the standard escalation schedule. Real-world tirzepatide before and after results confirm these patterns, and our detailed weight loss results gallery showcases outcomes across various demographics. Men using tirzepatide report particularly strong results when combined with resistance training.
The imbalanced agonist advantage
Here is where the pharmacology gets particularly interesting. Tirzepatide is not a 50/50 agonist at both receptors. Research from JCI Insight describes it as an imbalanced and biased dual agonist. Specifically, tirzepatide shows comparable affinity with GIP and full agonist activity at the GIP receptor. But at the GLP-1 receptor, tirzepatide demonstrates a 5-fold lower affinity and 20-fold lower potency compared to dedicated GLP-1 agonists like semaglutide.
This sounds like a weakness. It is actually a strength.
The lower GLP-1 receptor potency means tirzepatide achieves its effects with less GLP-1 receptor occupation than semaglutide requires. Research shows that 5mg of tirzepatide (occupying roughly 3% of GLP-1 receptors) matches or exceeds the efficacy of 1mg semaglutide (which requires significantly higher receptor occupancy). The GIP receptor activation compensates for and surpasses what lower GLP-1 engagement alone would produce.
This imbalanced design also explains why tirzepatide tends to produce fewer GLP-1-related side effects relative to its overall efficacy. The gastrointestinal effects primarily driven by GLP-1 receptor activation can be somewhat mitigated by the lower GLP-1 receptor occupancy, while the GIP receptor activation contributes to efficacy through a gentler pathway. That said, GI side effects still occur with tirzepatide. Our guides on tirzepatide diarrhea duration, tirzepatide constipation treatment, and tirzepatide headaches cover management strategies in detail. Understanding potential body aches and fatigue patterns helps researchers prepare for the adjustment period.
Why combining semaglutide and tirzepatide is not recommended
The reasons go beyond the FDA not approving it. The pharmacological, clinical, and practical objections to combining these medications are substantial and well-documented. Anyone considering this combination should understand the full scope of evidence against it before making a decision that could compromise their health. Our complete semaglutide vs tirzepatide comparison provides a thorough framework for choosing between them rather than combining them.
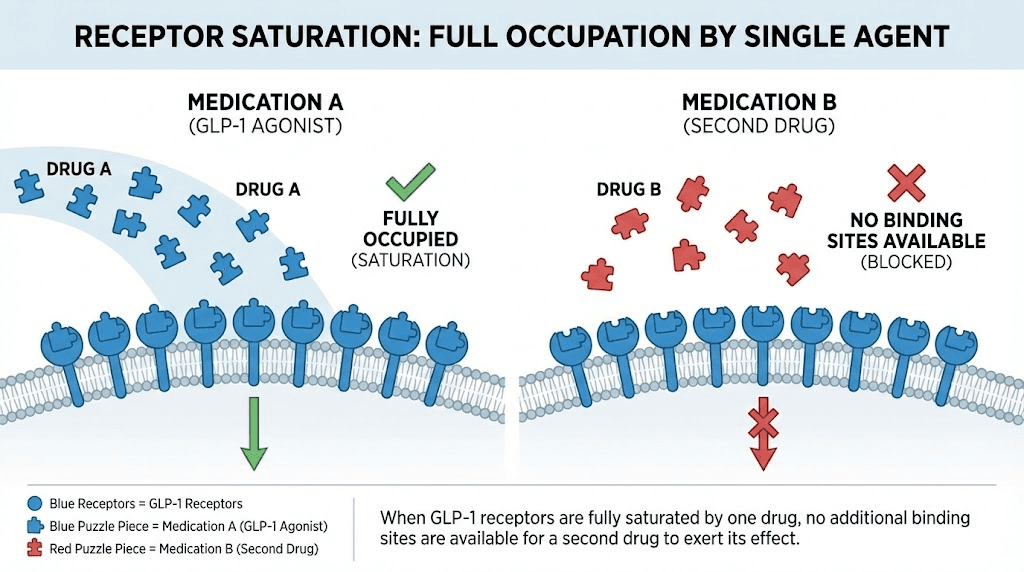
Receptor saturation makes additional GLP-1 activation pointless
Every receptor in the body has a saturation point. Once all available GLP-1 receptors are occupied by a medication, adding more of the same type of molecule has nowhere to bind. The therapeutic effect plateaus. But the circulating drug levels continue to rise, increasing the potential for off-target effects and side effect amplification.
At therapeutic doses, both semaglutide and tirzepatide achieve significant GLP-1 receptor occupancy. Semaglutide at 2.4mg (the maximum weight loss dose) produces near-maximal receptor engagement. Adding tirzepatide on top of this does not unlock additional receptor capacity. The GLP-1 receptors are already occupied.
The one unique receptor tirzepatide targets, the GIP receptor, is already built into tirzepatide as a single medication. You do not need semaglutide to get GLP-1 activation when tirzepatide already provides it. And you do not need to combine them to get GIP activation, because semaglutide does not activate GIP receptors at all. The math is simple. Semaglutide provides GLP-1 activation. Tirzepatide provides GLP-1 activation plus GIP activation. Combining them provides double GLP-1 activation (redundant and saturated) plus GIP activation. The only additional element is excess GLP-1 stimulation that has no productive destination.
Overlapping mechanisms with diminishing returns
Beyond receptor saturation, the downstream effects of GLP-1 activation follow a dose-response curve that flattens at higher levels. Appetite suppression does not scale linearly with increasing GLP-1 receptor activation. There is a ceiling effect. Past a certain point, more activation does not produce more appetite reduction. It produces more nausea.
The same applies to gastric emptying. Slowing digestion is beneficial up to a point. Past that point, the result is gastroparesis-like symptoms: severe nausea, vomiting, bloating, and potentially dangerous bowel obstructions. Combining two medications that both slow gastric emptying dramatically increases this risk. Understanding GLP-1 fatigue patterns and how long GLP-1 medications take to reach full effect reinforces why overloading these pathways is counterproductive.
Research on GLP-1 receptor pharmacology confirms that therapeutic doses of single agents achieve sufficient receptor engagement for maximum clinical benefit. The ceiling is already being reached with one medication. Two medications do not raise the ceiling. They just push you further past it into adverse effect territory.
No FDA approval and no clinical evidence
As of the latest available data, no clinical trial has studied the combination of semaglutide and tirzepatide in human subjects. This is not an oversight. There is no pharmacological rationale that would justify such a trial. The expected outcome based on receptor pharmacology is increased adverse effects without proportionally increased benefits.
The FDA labeling for tirzepatide (Mounjaro, Zepbound) specifically notes that the medication has not been studied in combination with other GLP-1 receptor agonists. This is a deliberate exclusion, not an invitation to experiment. Without safety data, combining these medications means accepting unknown risks with no evidence of additional benefit. Proper administration of either medication individually, including understanding how to inject GLP-1 medications, optimal injection sites, and best timing for injections, is far more likely to improve outcomes than adding a second medication.
Clinical guidelines explicitly advise against it
Medical guidelines published in recent years specifically recommend against combining medications within the same incretin class. The reasoning includes concerns about hypoglycemia in diabetic patients, increased gastrointestinal adverse events, and the absence of evidence showing clinical superiority of combination use over optimized monotherapy.
These guidelines reflect the consensus of endocrinologists, obesity medicine specialists, and pharmacologists who have reviewed the available evidence. The recommendation is clear: optimize single-agent therapy before considering a switch, and switch sequentially rather than stacking. For a broader perspective on the GLP-1 medication landscape, our semaglutide vs tirzepatide vs retatrutide comparison covers all three major options, while comparisons with emerging agents like CagriSema, survodutide, mazdutide, and orforglipron show where the field is heading.
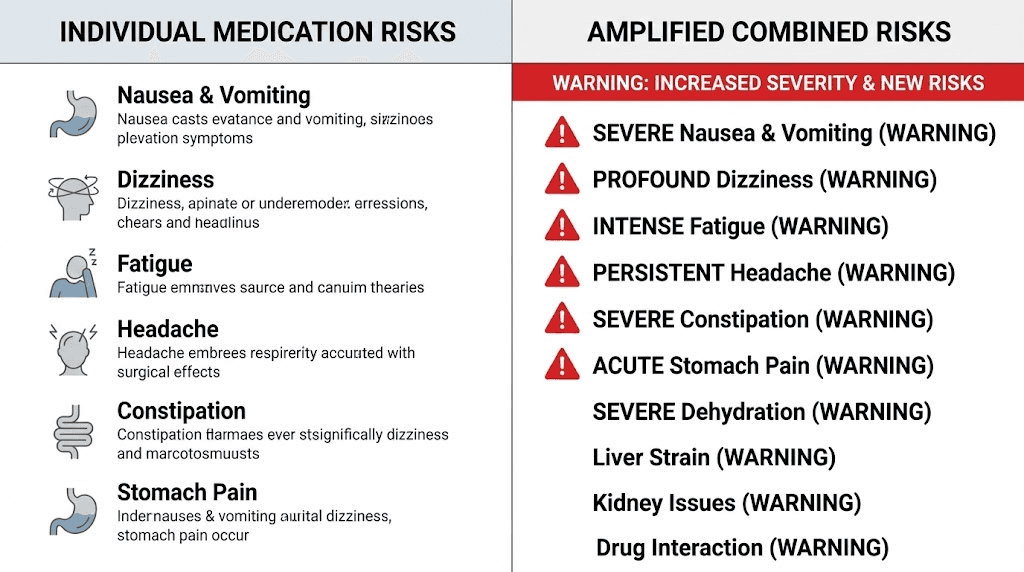
Side effect risks of taking both medications simultaneously
Even without formal clinical trial data on the combination, the expected adverse effect profile can be extrapolated from what we know about each medication individually and the pharmacology of excessive GLP-1 receptor activation. The risks are not theoretical. They follow directly from established pharmacological principles.
Intensified gastrointestinal problems
Gastrointestinal side effects are the most common complaints with both semaglutide and tirzepatide individually. A meta-analysis found that tirzepatide carries a relative risk of 2.94 for gastrointestinal adverse events compared to placebo, while semaglutide carries a relative risk of 1.68. These events include nausea, vomiting, diarrhea, constipation, and abdominal pain.
Combining both medications would predictably amplify these effects. Both drugs slow gastric emptying through GLP-1 receptor activation. Double the gastric slowing means dramatically increased nausea risk, worsened bloating, and a significantly elevated chance of more serious complications like bowel obstruction or ileus.
Most GI side effects with individual medications are mild to moderate and occur primarily during dose escalation periods. But combining two full-dose GLP-1 agonists eliminates the gradual escalation that makes these medications tolerable. The body would face the equivalent of maximum dosing on two medications simultaneously, with no time to adapt. For managing GI side effects on a single medication, our guides on semaglutide constipation, tirzepatide constipation, semaglutide burping, and tirzepatide diarrhea provide evidence-based solutions.
Hypoglycemia and dangerous blood sugar drops
GLP-1 receptor agonists individually carry a relatively low risk of hypoglycemia because their insulin-stimulating effect is glucose-dependent. The medication signals insulin release when blood sugar is elevated, not when it is already low. This built-in safety mechanism is one of the advantages of GLP-1 agonists over older diabetes medications.
However, doubling GLP-1 receptor stimulation could overwhelm this glucose-dependent mechanism. With excessive receptor activation, the threshold at which insulin secretion triggers could shift, potentially causing insulin release even at lower blood sugar levels. For people with type 2 diabetes who also take sulfonylureas or insulin alongside their GLP-1 medications, the combination of semaglutide and tirzepatide could create dangerous hypoglycemic episodes.
Even for non-diabetic individuals using these medications for weight loss, the additive effect on blood sugar regulation could produce symptoms of low blood sugar: shakiness, confusion, sweating, rapid heartbeat, and in extreme cases, loss of consciousness. Understanding whether your current medication affects energy levels or causes fatigue is important context. Doubling down on GLP-1 stimulation would only worsen GLP-1-related fatigue.
Dehydration and electrolyte imbalances
Nausea, vomiting, and diarrhea all cause fluid loss. When these effects are amplified by combining two medications, the risk of significant dehydration increases substantially. Dehydration from GLP-1 medications is already a recognized clinical concern with single-agent therapy.
Electrolyte imbalances from persistent vomiting or diarrhea can cause muscle cramps, cardiac arrhythmias, confusion, and kidney dysfunction. These risks compound when the source of fluid loss is doubled. Kidney function monitoring becomes especially critical, as GLP-1 receptor agonists have been associated with acute kidney injury in cases of severe dehydration. Our guide on supplements to take with tirzepatide covers electrolyte management and hydration strategies that apply to any GLP-1 medication user.
Excessive appetite suppression and malnutrition
One of the less obvious dangers of combining these medications is the potential for excessive appetite suppression leading to inadequate nutrition. Both medications powerfully reduce appetite. Both slow gastric emptying. The combined effect could reduce food intake to dangerously low levels.
Malnutrition during rapid weight loss is already a concern with single GLP-1 medications. Protein intake often drops below recommended levels, leading to muscle mass loss alongside fat loss. Hair loss, fatigue, and weakened immune function are documented effects of inadequate nutrition during GLP-1 therapy. Combining two powerful appetite suppressants could make eating enough food genuinely difficult.
The result would not be faster weight loss in a positive sense. It would be starvation-level caloric intake with all the metabolic consequences that follow: muscle wasting, bone density loss, hormonal disruption, and metabolic adaptation that makes future weight management even harder. Proper nutrition planning is critical even on a single medication. Our guides on foods to eat on semaglutide, what to eat on tirzepatide, and protein shakes for GLP-1 users provide practical meal planning strategies. Understanding which foods to avoid on tirzepatide and following a structured tirzepatide diet plan or semaglutide diet plan helps maintain adequate nutrition during therapy.
Head-to-head comparison: what the SURPASS-2 trial revealed
Rather than combining these medications, the relevant question is how they compare when used individually. The SURPASS-2 trial provides the most direct answer available from rigorous clinical research.
Study design and participants
SURPASS-2 was a 40-week randomized, open-label clinical trial published in the New England Journal of Medicine. The study enrolled 1,879 adults with type 2 diabetes and randomized them to receive one of three tirzepatide doses (5mg, 10mg, or 15mg weekly) or semaglutide 1mg weekly, all as add-on therapy to metformin.
Participants had a mean diabetes duration of 8.6 years, a baseline HbA1c of 8.28%, and a baseline weight of approximately 93.7 kg. These were not newly diagnosed patients with mild disease. They represented a population with established diabetes that had not been adequately controlled with metformin alone. The study design gave semaglutide every opportunity to compete. The 1mg dose was the maximum approved dose for type 2 diabetes at the time of the trial. Understanding tirzepatide dosing in units and semaglutide dosing in units provides context for how these trial doses translate to real-world use.
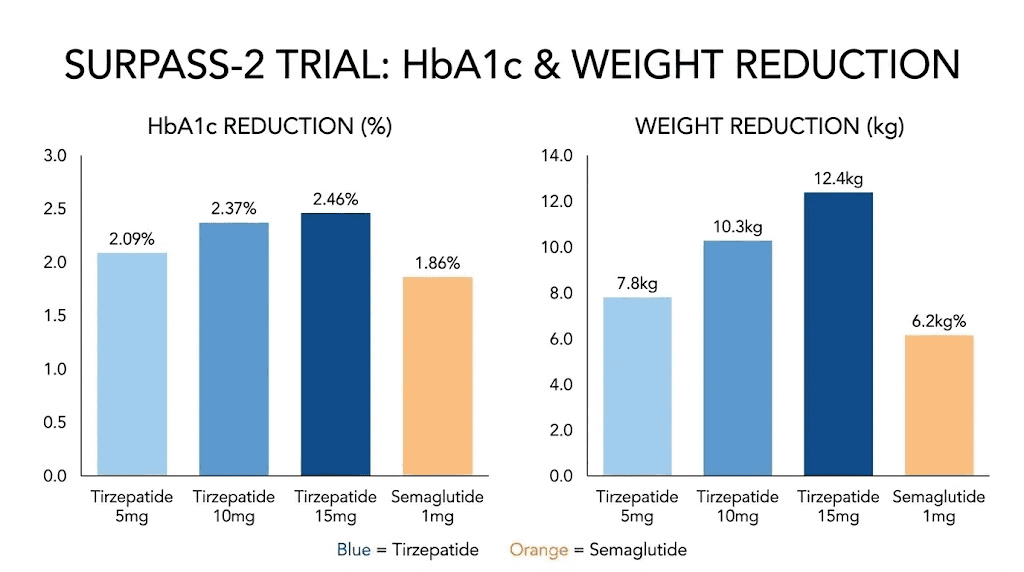
HbA1c reduction results
All three tirzepatide doses achieved superior blood sugar reduction compared to semaglutide 1mg. The estimated mean change from baseline in HbA1c was:
Tirzepatide 5mg: -2.01 percentage points
Tirzepatide 10mg: -2.24 percentage points
Tirzepatide 15mg: -2.30 percentage points
Semaglutide 1mg: -1.86 percentage points
Even the lowest tirzepatide dose outperformed semaglutide on blood sugar control. The highest tirzepatide dose reduced HbA1c by nearly half a percentage point more than semaglutide, a clinically significant difference that translates to meaningfully better glycemic management over time. For researchers starting tirzepatide, our compounded tirzepatide starting dose guide covers the initial dosing protocol.
Weight loss data
The weight loss differences were even more striking. All tirzepatide doses produced significantly greater weight reduction compared to semaglutide 1mg:
Tirzepatide 5mg: 1.9 kg more weight loss than semaglutide
Tirzepatide 10mg: 3.6 kg more weight loss than semaglutide
Tirzepatide 15mg: 5.5 kg more weight loss than semaglutide
All comparisons reached statistical significance (P less than 0.001). The highest tirzepatide dose produced an average of 5.5 kg (about 12 pounds) more weight loss than semaglutide over 40 weeks.
A composite endpoint of achieving HbA1c below 6.5% AND weight loss of 10% or more (without clinically significant hypoglycemia) showed even sharper differences. Only 22% of semaglutide patients achieved this composite, compared to 32% with tirzepatide 5mg, 51% with tirzepatide 10mg, and 60% with tirzepatide 15mg. These results demonstrate that switching to tirzepatide is far more logical than adding it on top of semaglutide. The dual-receptor mechanism provides advantages that additional GLP-1 activation alone cannot match.
Safety profiles in the trial
The most commonly reported adverse events across all treatment groups were gastrointestinal. Nausea, diarrhea, and vomiting occurred at similar rates in all groups, though they were somewhat more frequent with higher tirzepatide doses. Most GI events were mild to moderate in severity and occurred primarily during dose escalation phases.
Hypoglycemia rates were low across all groups, consistent with the glucose-dependent mechanism of both medications. Serious adverse events were comparable between tirzepatide and semaglutide groups, suggesting that the superior efficacy of tirzepatide does not come at the cost of significantly worse tolerability. Our comprehensive semaglutide vs tirzepatide side effects comparison covers every documented adverse effect, management strategies, and when to seek medical attention. Specific issues like tirzepatide injection site reactions and GLP-1 headaches are covered in dedicated guides.
How semaglutide and tirzepatide compare when used individually
Beyond the SURPASS-2 trial, real-world data and additional clinical evidence paint a complete picture of how these medications compare across multiple dimensions.
Weight loss effectiveness
Large-scale real-world studies consistently support the clinical trial findings. A study of over 18,000 patients found that those taking tirzepatide lost approximately 15.3% of body weight over one year, compared to 8.3% with semaglutide. That is nearly double the weight loss.
However, individual responses vary significantly. Some people respond exceptionally well to semaglutide and achieve weight loss comparable to average tirzepatide results. Others plateau early on either medication. The best predictor of success is not which medication you choose but how well you optimize the supporting factors: nutrition, protein intake, activity level, sleep, and consistent dosing. Our semaglutide before and after guide and tirzepatide before and after results showcase real outcomes across various starting points. Some researchers lose weight on semaglutide without exercise, though adding activity typically improves body composition.
Onset of action and timeline
Semaglutide typically takes 4-5 weeks to reach steady-state blood levels due to its 7-day half-life. Most people begin noticing appetite suppression within the first week, but significant weight loss usually becomes apparent around weeks 4-8. Our guide on how fast semaglutide works covers the detailed timeline.
Tirzepatide has a slightly shorter half-life of approximately 5 days but follows a similar escalation pattern. Meaningful appetite changes often appear within the first 1-2 weeks, with significant weight loss becoming noticeable by weeks 4-6. The full dose titration from 2.5mg to the target dose of 10mg or 15mg takes several months. Both medications require patience during the escalation phase. Our how fast tirzepatide works guide provides specific benchmarks for each dose level.
Side effect profiles
Both medications share similar GI side effect profiles, but the distribution and intensity differ somewhat. Semaglutide side effects tend to include nausea, constipation, diarrhea, and fatigue. Some users report specific issues like dizziness, insomnia, or excessive burping.
Tirzepatide side effects overlap significantly but may include injection site reactions more frequently and body aches that some users attribute to the GIP receptor activation. Tirzepatide headaches, muscle pain, and joint pain have been reported, though these are generally mild and temporary. Some users also report insomnia or anxiety during dose escalation phases.
How long each medication stays in the body
Understanding the pharmacokinetics of each medication is crucial for anyone considering switching between them.
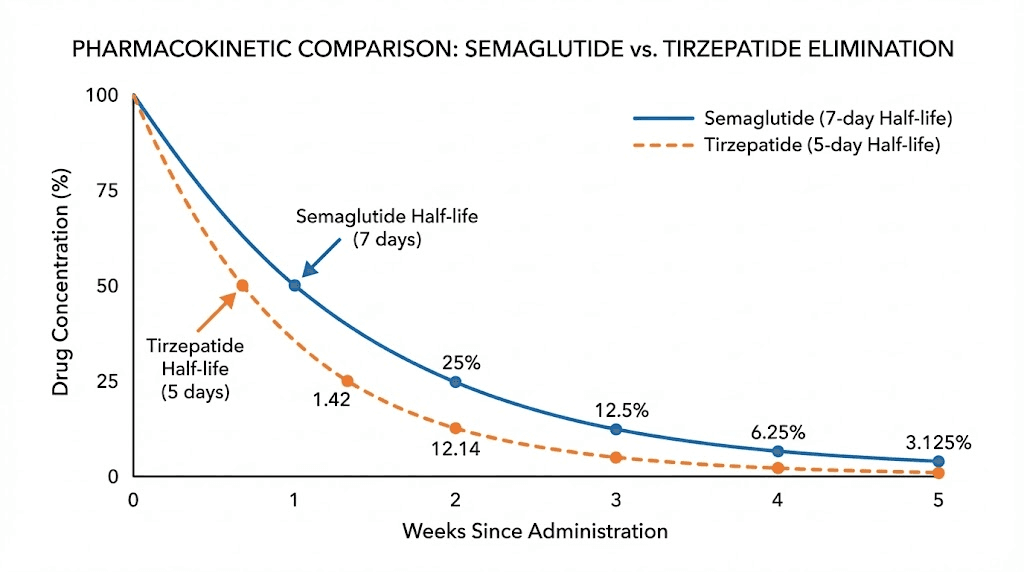
Semaglutide has a half-life of approximately 7 days. After the final injection, it takes roughly 5 weeks (35 days) for 97% of the medication to clear from the body. This extended presence is why weekly dosing works and why washout periods matter when switching. Our guide on semaglutide withdrawal symptoms covers what to expect as levels decline.
Tirzepatide has a half-life of approximately 5 days (120 hours). Complete elimination takes roughly 25-30 days after the final injection. The shorter half-life means tirzepatide clears the system somewhat faster than semaglutide. For proper storage during use, our guides on semaglutide shelf life and tirzepatide shelf life cover stability considerations. These numbers become critically important when transitioning from one medication to the other, which is the evidence-based alternative to combining them.
When switching between medications makes more sense than combining
If one medication is not delivering the results you expected, the evidence-based approach is switching, not stacking. Here are the most common scenarios where a transition is warranted.
You have hit a plateau
Weight loss stalls are common on both medications. After several months of steady progress, many people reach a point where the scale stops moving despite consistent dosing and reasonable nutrition habits. A semaglutide plateau does not necessarily mean the medication has stopped working. It often means the body has adapted to the current level of GLP-1 receptor stimulation and established a new metabolic equilibrium.
Switching to tirzepatide introduces GIP receptor activation as a new stimulus, which can break through a GLP-1-only plateau. Our detailed guide on 4 weeks on semaglutide with no weight loss covers all potential causes and solutions, including non-medication strategies that should be tried first. Similarly, if tirzepatide stops working, understanding why weight loss stalls on tirzepatide helps determine whether switching or dose optimization is the right next step.
Side effects are intolerable
Some people tolerate semaglutide well but struggle with tirzepatide, and vice versa. The side effect profiles overlap but are not identical. Someone experiencing severe semaglutide fatigue might find that tirzepatide produces less fatigue due to its different receptor binding profile. Conversely, someone with significant injection site reactions on tirzepatide or persistent redness and itching at injection sites might have a smoother experience with semaglutide. The key is that switching addresses the tolerability problem while maintaining the therapeutic benefit. Combining both medications would only add more side effects on top of the ones already causing problems.
Cost or availability changes
Practical factors sometimes drive medication switches. Compounded semaglutide and compounded tirzepatide have different pricing structures and availability that can change based on regulatory actions, manufacturing capacity, and pharmacy policies. Our peptide cost calculator helps compare pricing across formulations, and guides on affordable tirzepatide and cheap compounded tirzepatide cover the most cost-effective sourcing options.
How to safely switch from semaglutide to tirzepatide
This is the most common switching direction, given that tirzepatide typically produces greater weight loss. The process requires careful planning to minimize side effects and maximize the transition effectiveness.
The washout period
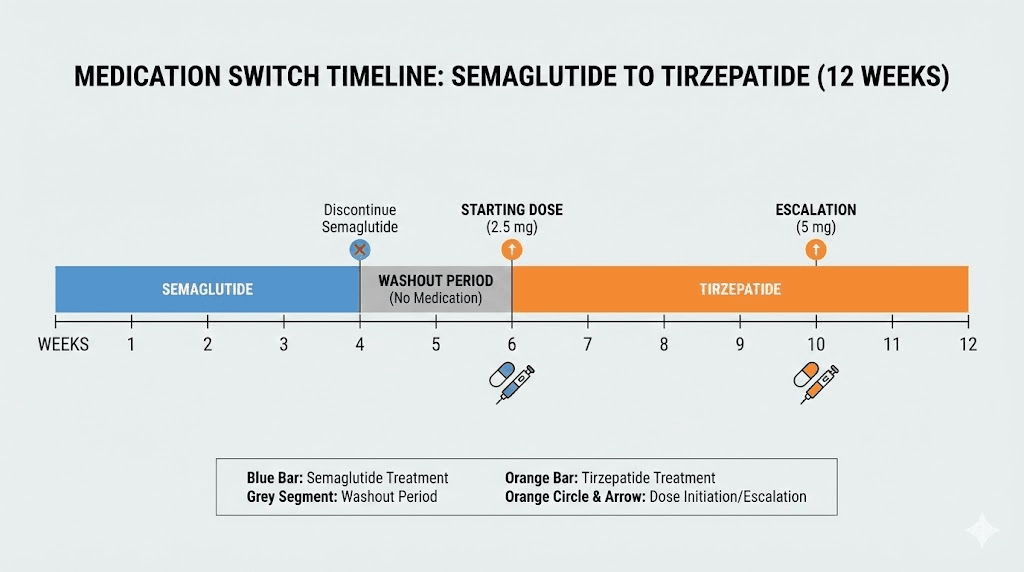
The recommended approach is to stop semaglutide and wait approximately 7 days before starting tirzepatide. This washout period allows semaglutide levels to begin declining before introducing a new GLP-1-active medication. Some healthcare providers recommend a shorter washout of 3-5 days, while others prefer a full week. The 7-day window aligns with the semaglutide half-life and the standard weekly injection schedule, making it the most practical approach.
Simply take your last semaglutide injection on the usual day. Skip the following injection day. Begin tirzepatide on what would have been the next injection day. Keep in mind that semaglutide takes approximately 5 weeks to fully clear the system. During the early weeks of tirzepatide use, some residual semaglutide will still be present. This is generally well-tolerated because tirzepatide starting doses are low and the overlapping period is brief. Our semaglutide to tirzepatide conversion chart provides a detailed visual guide for the transition timeline, while the switching dosage chart covers dose mapping between the two medications.
Starting dose protocol
Regardless of your semaglutide dose before switching, the standard recommendation is to start tirzepatide at 2.5mg weekly. This is the lowest available dose and serves as the initiation dose for all patients, whether new to GLP-1 therapy or transitioning from another medication.
Starting at 2.5mg may feel like a step backward if you were on high-dose semaglutide. Weight loss may slow temporarily. Appetite suppression may decrease. This is normal and expected. The low starting dose allows the body to adjust to tirzepatide GIP receptor activation, which produces effects your body has not experienced on semaglutide alone.
After 4 weeks at 2.5mg, the dose can be increased to 5mg. Subsequent increases of 2.5mg every 4 weeks can continue up to the maintenance dose, which is typically 10mg or 15mg depending on individual response and tolerability. For exact dosing calculations with compounded formulations, our tirzepatide dosage in units guide and the tirzepatide syringe dosage calculator provide measurement-level precision. Reconstituting tirzepatide correctly is also essential for compounded formulations, and our bacteriostatic water mixing guide covers exact ratios.
What to expect during the transition
The first 2-4 weeks of the transition are the most unpredictable period. Common experiences include temporary return of appetite as semaglutide clears and tirzepatide has not yet reached therapeutic levels. Some weight regain is possible during this window. New or different GI side effects may appear as the body adjusts to GIP receptor activation. Even if you tolerated semaglutide well, tirzepatide may produce different sensations.
Injection site differences can also occur. Tirzepatide may cause more localized reactions at the injection site compared to semaglutide. Our guide on tirzepatide injection site reaction treatment covers identification and management. Understanding proper injection technique can minimize these issues.
Most people stabilize within 4-6 weeks and begin experiencing the benefits of dual-receptor activation. By the 8-week mark, most researchers report comparable or improved appetite suppression compared to their previous semaglutide protocol.
Managing side effects during the switch
The transition period demands extra attention to self-care. Stay hydrated aggressively, as GI side effects may return during the dose escalation phase of tirzepatide even if they resolved on semaglutide. Prioritize protein intake, aiming for at least 1.0-1.2 grams per kilogram of body weight daily to preserve muscle mass during the transition.
Eating smaller, more frequent meals can help manage nausea during the early weeks. Our detailed tirzepatide diet plan and tirzepatide foods to avoid guides provide specific meal frameworks designed for the escalation phase. Supplementation strategies can also support the transition, and understanding whether alcohol is safe during tirzepatide use matters for those who drink socially. If side effects become severe, do not hesitate to slow the titration schedule. Some researchers stay at each dose level for 6-8 weeks before escalating, and this approach often produces better long-term tolerability.
How to safely switch from tirzepatide to semaglutide
While less common than the reverse direction, switching from tirzepatide to semaglutide does happen and requires its own set of considerations.
Reasons for switching to semaglutide
Several legitimate scenarios might prompt this switch. Availability disruptions can make tirzepatide temporarily inaccessible. Cost differences between compounded formulations may favor semaglutide. Some people experience side effects specifically related to GIP receptor activation that resolve on a GLP-1-only medication. Our guide on switching from tirzepatide to semaglutide covers the complete protocol, and the conversion chart provides dose mapping guidance.
Dose considerations
There is no standardized dose conversion between tirzepatide and semaglutide. The medications work through different mechanisms and have different potency profiles, so a direct milligram-to-milligram conversion does not apply. However, general guidelines suggest starting semaglutide at 0.5mg-1.0mg from tirzepatide 5mg, at 1.0mg-1.7mg from tirzepatide 10mg, and at 1.7mg-2.4mg from tirzepatide 15mg.
These are approximate starting points. Starting at a lower semaglutide dose than the conversion suggests is always safer than starting too high. Our semaglutide vs tirzepatide dosage chart provides a detailed comparison framework, and the semaglutide dosage calculator handles exact measurement amounts for compounded formulations. Understanding unit-to-milligram conversions and proper reconstitution ensures accurate dosing from the start.
Timeline expectations
Expect a transition period of 4-8 weeks before semaglutide reaches full therapeutic effect. During this window, appetite suppression may decrease compared to tirzepatide, as the GIP receptor component is no longer active. Some weight regain during the transition is possible but typically stabilizes once semaglutide reaches steady-state levels. The washout from tirzepatide takes approximately 25-30 days for complete elimination. Starting semaglutide 5-7 days after the last tirzepatide injection is the standard approach. Our best time of day to take semaglutide guide and best injection site guide help optimize administration from day one.
What doctors actually recommend instead of combining
If one GLP-1 medication is not producing the results you want, medical professionals have several evidence-based strategies that do not involve stacking two medications with overlapping mechanisms.
Optimizing single medication dosing
The most common reason for suboptimal results is inadequate dosing. Many people stall at a mid-range dose without fully titrating to the maximum. Semaglutide can be escalated to 2.4mg weekly for weight loss, and tirzepatide to 15mg weekly. If you have not reached these maximum doses, the first step is always to complete the titration.
Beyond standard dosing, some healthcare providers explore microdosing protocols for people sensitive to GI side effects at standard escalation rates. Our microdose tirzepatide schedule covers alternative approaches that allow for slower but more tolerable dose increases. Timing also matters. Adjusting when you inject or how you prepare your medication can affect tolerability. Our guides on best time to take semaglutide and best time for GLP-1 injections cover the evidence.
Adding complementary medications that use different mechanisms
Rather than combining two GLP-1 active medications, a more logical approach is adding a medication that works through an entirely different mechanism. This creates genuine synergy rather than receptor saturation.
Phentermine works through norepinephrine release, a completely different appetite suppression pathway. Our guides on combining phentermine and semaglutide and combining phentermine and tirzepatide discuss the evidence and practical considerations. For a direct comparison, our phentermine vs semaglutide and phentermine vs GLP-1 guides cover the full picture.
B12 and glycine additions are popular with compounded GLP-1 formulations. Semaglutide with B12, tirzepatide with B12, and tirzepatide with glycine and B12 compounds are widely available and may offer supportive benefits. Semaglutide with glycine and tirzepatide with glycine formulations focus on anti-nausea support. Methylcobalamin formulations of both semaglutide and tirzepatide are also used. Tirzepatide with niacinamide has gained attention as well.
AOD-9604, a peptide fragment with fat-loss properties, has been explored alongside tirzepatide for its complementary mechanism. Our AOD-9604 with tirzepatide guide discusses the research and practical protocols.
Lifestyle modifications that enhance medication results
No medication works optimally in a vacuum. The difference between average and exceptional GLP-1 results often comes down to supporting lifestyle factors that SeekPeptides members focus on alongside their protocols.
Protein intake is the single most important dietary factor during GLP-1 therapy. Adequate protein preserves muscle mass, supports metabolism, and improves body composition outcomes beyond what the medication alone achieves. Our protein shakes for GLP-1 users guide covers practical supplementation strategies.
Exercise during GLP-1 therapy, particularly resistance training, counteracts the muscle mass loss that commonly accompanies rapid weight loss. Even moderate strength training 2-3 times per week can significantly improve body composition results. Sleep quality directly affects hunger hormones, insulin sensitivity, and metabolic rate. Stress management follows a similar pattern, as elevated cortisol promotes visceral fat storage and can blunt the metabolic benefits of GLP-1 therapy. For comprehensive nutritional guidance, our foods to eat on semaglutide and what to eat on tirzepatide provide practical meal-level recommendations. Understanding how long to stay on semaglutide and whether you can drink alcohol on semaglutide helps build a sustainable long-term plan.
Next-generation alternatives worth watching
The GLP-1 medication landscape is evolving rapidly. Several next-generation options in development may offer advantages over both semaglutide and tirzepatide.
Retatrutide is a triple-receptor agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. Early clinical trial data suggests weight loss exceeding 24% of body weight, surpassing both semaglutide and tirzepatide. Our retatrutide vs semaglutide comparison covers the current evidence.
CagriSema combines cagrilintide (an amylin analog) with semaglutide in a single injection. This represents a genuinely different approach to combination therapy because cagrilintide works through amylin receptors, not GLP-1 or GIP receptors. Our CagriSema vs tirzepatide comparison and cagrilintide and semaglutide guide examine the data. Survodutide targets GLP-1 and glucagon receptors for obesity and metabolic liver disease. Orforglipron is an oral GLP-1 agonist that could eliminate weekly injections entirely, similar to oral tirzepatide formulations and GLP-1 patches being developed.
These next-generation medications represent the future of weight loss pharmacotherapy. Rather than combining current-generation medications inappropriately, waiting for purpose-designed combination products is the safer and likely more effective approach. Use the GLP-1 plotter tool to visualize how different medications compare over time.
For researchers committed to optimizing their GLP-1 protocols, SeekPeptides provides the most comprehensive resource available. Members access detailed dosing protocols, conversion calculators, evidence-based guides, and a community of experienced researchers who have navigated these exact medication decisions. From choosing between semaglutide and tirzepatide to managing side effects and optimizing results, SeekPeptides offers the depth and specificity that generic medical websites simply cannot match.
Frequently asked questions
Is it safe to take semaglutide and tirzepatide on the same day?
No. Taking both medications on the same day is not safe. Both activate GLP-1 receptors, and simultaneous administration would create excessive receptor stimulation, amplify gastrointestinal side effects, and provide no additional benefit beyond what either medication delivers individually. The correct approach is choosing one medication and optimizing the dose, or switching sequentially with an appropriate washout period. Our semaglutide to tirzepatide conversion chart covers the proper transition protocol.
Can I take a low dose of each to get both GLP-1 and GIP activation?
This idea has a logical appeal but does not work in practice. Tirzepatide already provides both GLP-1 and GIP receptor activation as a single medication. Taking a low dose of semaglutide plus a low dose of tirzepatide would give you less GIP activation than full-dose tirzepatide alone, while still creating redundant GLP-1 stimulation. If you want both receptor pathways active, tirzepatide alone at an appropriate dose is the correct choice. Our tirzepatide dosing guide covers the proper escalation schedule.
What if my doctor prescribed both at the same time?
If a healthcare provider has prescribed both medications simultaneously, it is worth seeking a second opinion or asking them to explain the pharmacological rationale. No published clinical guideline supports this approach. However, medical practice occasionally includes off-label combinations, and individual providers may have specific reasoning based on unique patient circumstances. Always follow healthcare provider guidance, but an informed conversation about the evidence is appropriate.
How long should I wait between stopping semaglutide and starting tirzepatide?
The recommended washout period is approximately 7 days. Stop semaglutide on your usual injection day, skip the next scheduled injection, and begin tirzepatide 2.5mg on what would have been the following injection day. Our detailed switching from semaglutide to tirzepatide dosage chart covers the complete timeline and dose escalation recommendations.
Will I gain weight during the switch between medications?
Some temporary weight gain during the transition is possible. As semaglutide clears your system and tirzepatide has not yet reached therapeutic levels, there can be a window of reduced appetite suppression. Most people regain 1-3 pounds during this period, which resolves quickly once tirzepatide reaches effective blood levels. Maintaining consistent nutrition habits and adequate protein intake during the transition minimizes this effect.
Is combining semaglutide and tirzepatide different from CagriSema?
Yes, fundamentally different. CagriSema is a purpose-designed combination of semaglutide and cagrilintide (an amylin analog). Cagrilintide works through amylin receptors, which are entirely different from GLP-1 and GIP receptors. This means CagriSema activates two distinct, non-overlapping pathways. Combining semaglutide and tirzepatide, by contrast, creates redundant GLP-1 stimulation because both medications activate the same receptor. Our CagriSema vs tirzepatide comparison explains the mechanistic differences in detail.
Which medication should I choose if I have never tried either?
The choice depends on individual factors including insurance coverage, cost, medical history, and provider recommendation. Tirzepatide generally produces greater weight loss in clinical trials, but semaglutide has a longer track record and more published safety data. Many researchers start with semaglutide due to wider availability and then switch to tirzepatide if results plateau. Our complete semaglutide vs tirzepatide comparison covers all the factors to consider, and understanding BMI requirements for GLP-1 medications is an important first step.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your protocols stay optimized, your transitions stay smooth, and your results stay consistent.