Mar 1, 2026

Why semaglutide stops working for some people
You did everything right. You followed the dosing schedule. You adjusted your diet. You showed up every week with the injection, watched the scale drop for months, and then it just stopped. Or maybe it never really worked in the first place. Maybe the appetite suppression that everyone raves about never kicked in for you. Maybe you lost five pounds in three months while your neighbor dropped thirty.
You are not alone.
Clinical trials funded by Novo Nordisk found that up to 23% of people taking semaglutide fell into the non-responder category, meaning they lost less than 5% of their body weight over the study period. That is roughly one in four people who take semaglutide and do not get the results they expected. And the frustration runs deep, because when everyone around you talks about how these medications changed their life, being the person it did not work for feels isolating.
Here is the good news. Semaglutide failing does not mean all GLP-1 based medications will fail. Tirzepatide works through a fundamentally different mechanism, targeting two receptors instead of one, and the clinical data shows it consistently outperforms semaglutide for both weight loss and metabolic improvement. But the answer to whether tirzepatide will work for you specifically depends on why semaglutide did not work in the first place.
This guide breaks down the science behind GLP-1 non-response, explains why tirzepatide often succeeds where semaglutide fails, walks you through the switching process step by step, and helps you set realistic expectations for what happens next. Whether you hit a plateau, never responded at all, or quit because of side effects, you will find specific, actionable guidance here.
Understanding why semaglutide fails: the five core reasons
Before jumping to tirzepatide as the answer, you need to understand why semaglutide did not work. The reason matters because it determines whether switching medications will actually help, or whether the real fix lies somewhere else entirely.
Genetic GLP-1 receptor variations
Your GLP-1 receptors are encoded by the GLP1R gene. And like most genes, this one varies from person to person. The single nucleotide polymorphism rs6923761, known as the Gly168Ser variant, is the most extensively studied GLP-1 receptor variation. It changes the structure of the receptor slightly, which means semaglutide may not bind as effectively or trigger the same downstream signaling cascade in your cells.
Think of it like a key and lock. Semaglutide is the key. Your GLP-1 receptor is the lock. If your lock is shaped slightly differently because of genetics, the key does not turn as smoothly. The medication still enters your body. It still circulates. But the cellular response is weaker, and the appetite suppression and metabolic effects you were counting on simply do not materialize the way they should.
This is one of the most compelling reasons to try tirzepatide. Because tirzepatide activates GIP receptors in addition to GLP-1 receptors, you get an entirely separate signaling pathway. Even if your GLP-1 receptors are genetically suboptimal, the GIP receptor pathway offers a different route to the same metabolic outcomes.
Metabolic adaptation and plateau
This one catches people off guard. Semaglutide was working. The weight was dropping. Then somewhere around month four or five, everything stopped. The scale froze. Maybe it even crept back up a pound or two.
This is metabolic adaptation. As you lose weight, your body requires fewer calories to function. Your total daily energy expenditure drops. The semaglutide plateau is not the medication failing. It is your body recalibrating to a new baseline. The medication still suppresses appetite and slows gastric emptying. But the calorie deficit that drove your initial weight loss has shrunk or disappeared entirely because your smaller body now burns less energy.
Switching to tirzepatide in this scenario often restarts weight loss because the dual receptor mechanism provides a stronger metabolic stimulus. The SURMOUNT-5 trial showed that tirzepatide produced 20.2% body weight reduction compared to 13.7% with semaglutide over 72 weeks, a significant gap that suggests tirzepatide may push past the metabolic ceiling where semaglutide stalls.
Inadequate dosing
Here is a simple but overlooked possibility. You may not have been on a high enough dose of semaglutide. The standard semaglutide dosing protocol starts at 0.25mg weekly and titrates up to 2.4mg over approximately 16 to 20 weeks. Many people experience side effects during titration that prevent them from reaching the therapeutic dose.
If you never made it past 1mg or 1.7mg because of nausea, constipation, or other gastrointestinal issues, you were never on a dose strong enough to produce maximum results. Interestingly, the SURMOUNT-5 trial found that gastrointestinal adverse events causing treatment discontinuation occurred in 5.6% of semaglutide users compared to only 2.7% of tirzepatide users. So tirzepatide may actually be easier to tolerate at therapeutic doses.
Lifestyle factors undermining the medication
No GLP-1 medication works in a vacuum. Semaglutide suppresses appetite and improves insulin sensitivity, but it cannot overcome certain lifestyle factors that actively work against weight loss.
Poor sleep quality disrupts leptin and ghrelin regulation. Chronic stress elevates cortisol, which promotes fat storage around the midsection. Alcohol consumption adds empty calories and impairs metabolic function. Inadequate protein intake leads to muscle loss, which further reduces metabolic rate. And some prescription medications, including certain antidepressants, beta-blockers, and corticosteroids, actively promote weight gain.
If lifestyle factors are the primary reason semaglutide did not work, switching to tirzepatide alone will not solve the problem. You need to address the underlying lifestyle issues first. Tirzepatide is more powerful, but it is not powerful enough to overcome chronic sleep deprivation or 2,000 excess calories from alcohol each week.
Underlying medical conditions
Certain conditions make GLP-1 medications less effective. Uncontrolled hypothyroidism slows metabolism significantly. Cushing syndrome floods the body with cortisol. Polycystic ovary syndrome creates insulin resistance that partially counteracts GLP-1 effects. And insulin resistance itself, when severe enough, can blunt the metabolic improvements these medications provide.
Getting these conditions diagnosed and managed is essential before switching medications. A provider who runs comprehensive bloodwork, including thyroid panel, cortisol, insulin, and HbA1c, can identify whether a treatable condition is sabotaging your results.
The semaglutide non-responder spectrum: where do you fall?
Non-response is not a binary. It is a spectrum. Understanding where you fall on that spectrum shapes your entire approach to tirzepatide and determines how likely it is to work.
Complete non-responders (less than 2% body weight loss)
You took semaglutide at 2.4mg for months. Nothing happened. Your appetite barely changed. The scale did not move. Your HbA1c stayed the same. This is true non-response, and it affects roughly 10 to 15% of people who try semaglutide.
Complete non-response often points to receptor-level issues, either genetic polymorphisms that alter GLP-1 receptor binding, or downstream signaling problems that prevent the receptor activation from translating into metabolic changes. For this group, tirzepatide's GIP pathway represents a genuine alternative route. If your GLP-1 receptors are the bottleneck, bypassing them through GIP activation can produce results that pure GLP-1 agonism never could. But success is not guaranteed, and expectations should be tempered.
Inadequate responders (2-5% body weight loss)
You lost some weight, maybe 10 to 15 pounds over six months. Enough to notice. Not enough to matter clinically. Your appetite decreased somewhat, but the "food noise" everyone talks about never fully disappeared. You still think about food. You still struggle with portions.
Inadequate response suggests partial GLP-1 pathway engagement. The mechanism is working, just not powerfully enough. This is actually the most promising group for tirzepatide switching because the additional GIP stimulation can amplify the partial response you already have. Think of it as adding a second engine to a car that was underpowered with one. The foundation works. It just needs more thrust.
Plateau responders (initial success followed by stall)
You had a great first three to five months. Maybe you lost 20, 30, even 40 pounds. Then it stopped. Completely. Despite being at maximum dose. Despite maintaining your diet and exercise habits. The semaglutide plateau hit hard and will not budge.
Plateau response is the most common reason people switch, and it has the highest success rate with tirzepatide. Your body adapted to GLP-1 agonism, but it has not adapted to GIP agonism because it has never been exposed to pharmacological GIP activation before. The novel stimulus restarts the metabolic momentum. Many plateau responders report their weight loss accelerating within weeks of reaching therapeutic tirzepatide doses, often surpassing the rate they experienced during their best months on semaglutide.
Side effect limited responders
The medication might have worked if you could have tolerated it. But the bloating, nausea, vomiting, or fatigue made it impossible to reach or maintain a therapeutic dose. You topped out at 0.5mg or 1mg because every increase brought unbearable gastrointestinal distress.
This group faces an interesting question. Will tirzepatide side effects be different? The data says possibly yes. The different receptor profile and biased agonism of tirzepatide produce a somewhat different side effect pattern. Some people who could not tolerate semaglutide at any dose find tirzepatide manageable. Others experience similar issues. The only way to know is a careful trial starting at the lowest dose with slow titration.
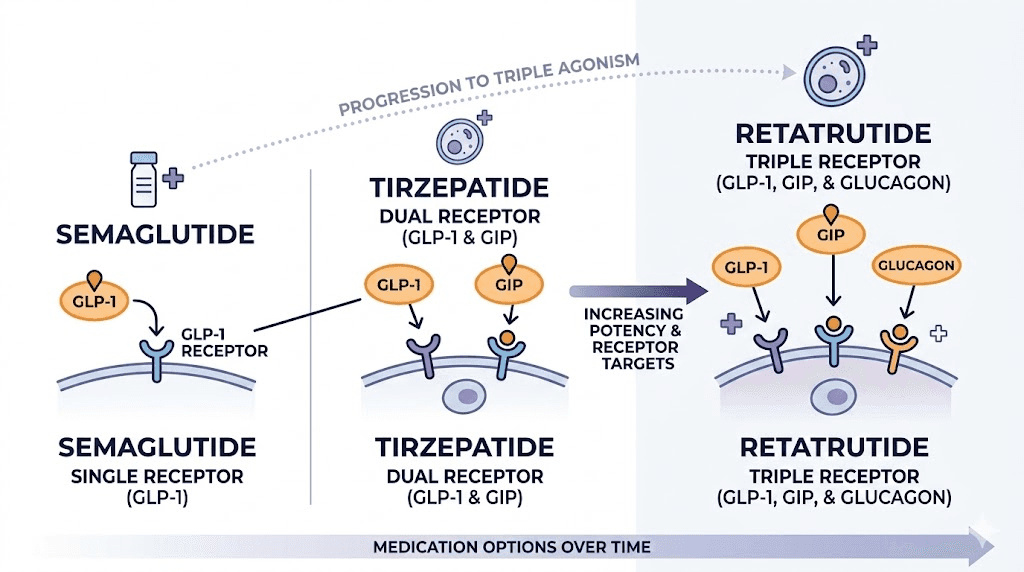
How tirzepatide works differently than semaglutide
This is where things get interesting. And this is the core reason why tirzepatide succeeds for many people who fail on semaglutide.
The dual receptor advantage
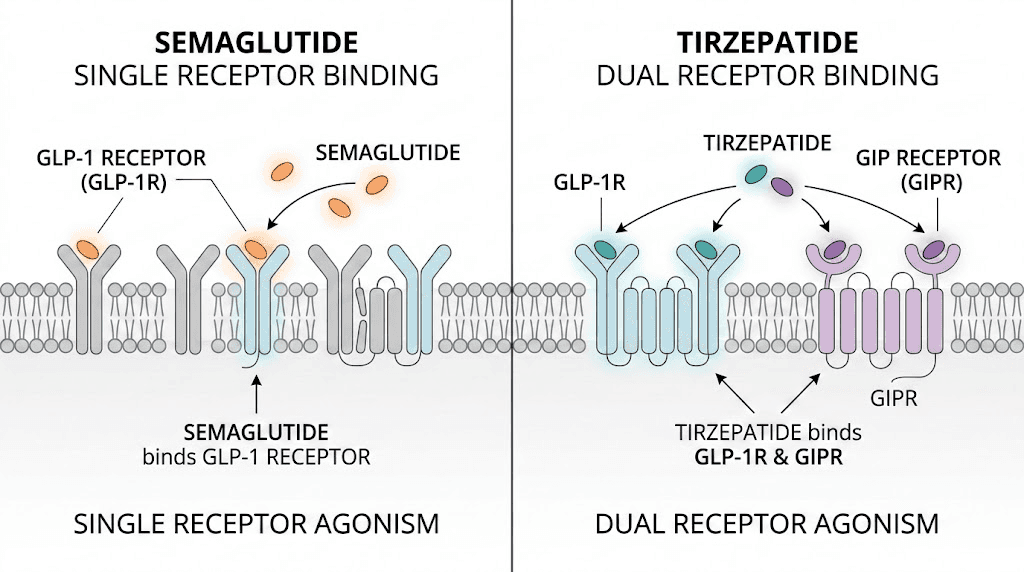
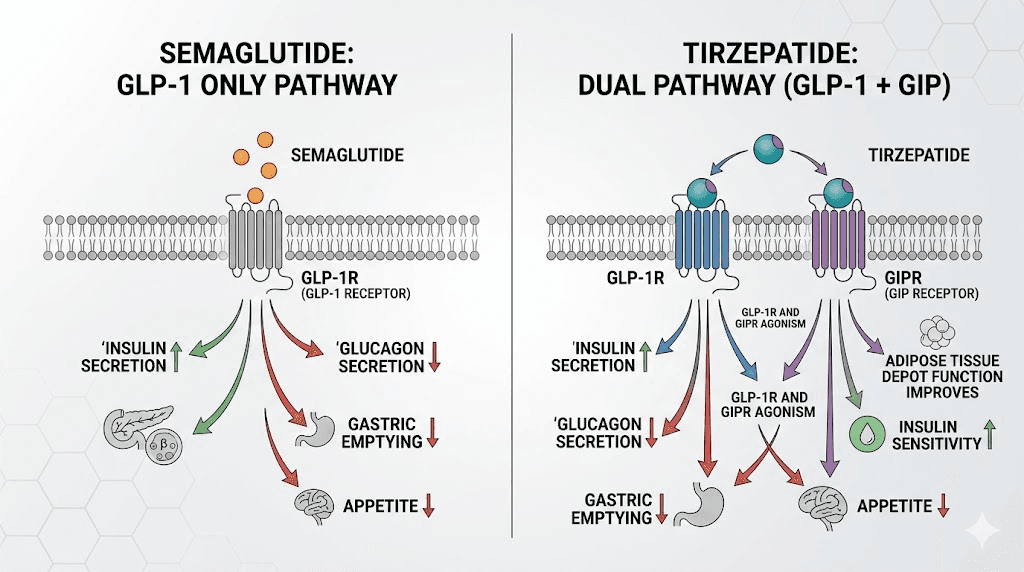
Semaglutide activates one receptor. The GLP-1 receptor. It binds, triggers a signaling cascade, slows gastric emptying, enhances insulin secretion, and suppresses appetite through central nervous system pathways. For many people, this is enough. But for the 15 to 25% who do not respond adequately, one receptor simply is not cutting it.
Tirzepatide activates both GLP-1 receptors and GIP receptors. GIP, or glucose-dependent insulinotropic polypeptide, is the other major incretin hormone. It is released from K-cells in the small intestine after eating and has its own distinct effects on metabolism, insulin secretion, and fat tissue.
The GIP receptor pathway does things the GLP-1 pathway cannot do alone. Research published in JCI Insight describes tirzepatide as an "imbalanced and biased dual agonist," meaning it actually engages the GIP receptor more potently than the GLP-1 receptor. This is not a weakness. It is a design feature. The GIP receptor activation provides additional metabolic benefits through pathways that pure GLP-1 agonists like semaglutide simply cannot access.
GIP receptor effects on fat tissue
GIP receptors exist on adipocytes, your fat cells. When tirzepatide activates these receptors, it influences how fat cells store and release energy. Studies show GIP receptor activation on fat tissue promotes improved lipid handling, enhanced insulin sensitivity in fat cells specifically, and changes in fat distribution patterns.
For people with significant visceral fat, the kind that wraps around internal organs and drives metabolic disease, this additional pathway matters enormously. The SURMOUNT-5 trial showed tirzepatide reduced waist circumference by 18.4cm compared to 13.0cm with semaglutide. That 5.4cm difference represents meaningful visceral fat reduction that pure GLP-1 agonism did not achieve.
Enhanced insulin signaling through biased agonism
Tirzepatide does something subtle but important at the molecular level. Research from Nature Metabolism shows that beta-arrestin1, a protein that normally limits the insulin response to GLP-1, does not limit the insulin response to GIP or tirzepatide. This biased agonism means tirzepatide can enhance insulin secretion more effectively than semaglutide, particularly in people whose GLP-1 signaling is partially blunted.
In plain terms, even if your body has developed some degree of resistance to GLP-1 signaling, the GIP pathway and the biased signaling properties of tirzepatide can still drive insulin response. It is like having a backup generator. When the main power line weakens, the backup kicks in.
Central nervous system appetite control
Both semaglutide and tirzepatide cross the blood-brain barrier and act on appetite centers in the hypothalamus. But they arrive through different receptor populations and trigger different downstream effects. GIP receptors in the brain influence reward pathways, food palatability perception, and satiety signaling through mechanisms distinct from GLP-1.
This explains a phenomenon many people report when switching. The appetite suppression with tirzepatide often feels qualitatively different from semaglutide. Not just stronger, but different in character. Food noise may diminish in ways it never did with semaglutide, particularly cravings for high-fat and high-sugar foods.
The clinical evidence: tirzepatide versus semaglutide head to head
Opinions are cheap. Data is not. Here is what the actual clinical trials show when these two medications go head to head.
SURMOUNT-5: the definitive comparison
SURMOUNT-5 is the first large-scale, randomized, controlled trial directly comparing tirzepatide and semaglutide for obesity treatment. Published in the New England Journal of Medicine, it enrolled 751 adults with obesity but without type 2 diabetes. Participants were randomly assigned to receive maximum tolerated doses of either tirzepatide (10mg or 15mg) or semaglutide (1.7mg or 2.4mg) weekly for 72 weeks.
The results were decisive.
Tirzepatide produced a mean body weight reduction of 20.2%. Semaglutide produced 13.7%. That is a 47% greater weight loss with tirzepatide. In absolute terms, tirzepatide users lost an average of 22.8kg (about 50 pounds) compared to 15.0kg (about 33 pounds) with semaglutide.
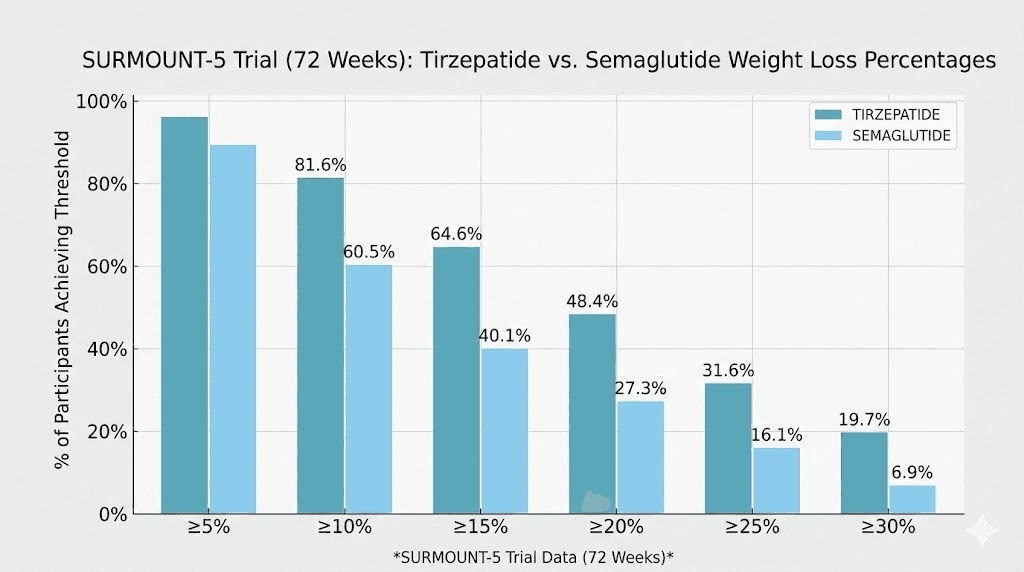
But the numbers get even more telling when you look at weight loss thresholds. Here is how many participants in each group hit specific milestones:
10% or more body weight loss: More participants achieved this with tirzepatide
15% or more body weight loss: Substantially more with tirzepatide
20% or more body weight loss: Significantly more with tirzepatide
25% or more body weight loss: 31.6% of tirzepatide users versus only 16.1% of semaglutide users
Nearly twice as many people on tirzepatide achieved 25% or greater body weight loss compared to semaglutide. That gap represents the difference between a good result and a transformative one for many people.
SURPASS trials: type 2 diabetes data
The SURPASS trial series examined tirzepatide versus semaglutide specifically in patients with type 2 diabetes. SURPASS-2 compared tirzepatide at 5mg, 10mg, and 15mg against semaglutide 1mg weekly over 40 weeks.
All three tirzepatide doses outperformed semaglutide 1mg for both HbA1c reduction and weight loss. The highest tirzepatide dose (15mg) produced an average weight loss of 11.2kg compared to 5.7kg with semaglutide 1mg. HbA1c reductions were similarly superior across all tirzepatide doses.
For people with type 2 diabetes who found semaglutide insufficient for blood sugar control or weight management, these numbers offer concrete evidence that tirzepatide provides a meaningful upgrade.
Real-world data confirms trial results
Clinical trials occur under controlled conditions. Real-world data tells you what happens when regular people use these medications in their actual lives, with all the missed doses, dietary inconsistencies, and imperfect compliance that reality involves.
A large retrospective study of over 18,000 patients found that those taking tirzepatide lost approximately 15.3% of body weight in one year compared to 8.3% with semaglutide. The real-world gap is slightly smaller than trial data, which is expected, but it remains clinically significant and consistently favors tirzepatide.
Another real-world study published in PMC examined patients with type 2 diabetes and confirmed that tirzepatide users had greater HbA1c and weight reductions at 12 months compared to injectable semaglutide users, regardless of whether they had previous GLP-1 receptor agonist experience. That last point matters enormously. Even patients who had already used another GLP-1 medication saw better results with tirzepatide.
Safety profile comparison
More effective does not always mean more side effects. In SURMOUNT-5, gastrointestinal adverse events were common in both groups, which is expected for this drug class. However, side effect profiles showed some interesting differences.
Treatment discontinuation due to gastrointestinal side effects occurred in 5.6% of semaglutide users versus 2.7% of tirzepatide users. So despite being more effective, tirzepatide was actually better tolerated in this trial. For people who quit semaglutide because of unbearable nausea, bloating, or constipation, this is encouraging data suggesting they may tolerate tirzepatide better.
Who is most likely to succeed on tirzepatide after semaglutide failure
Not everyone who fails on semaglutide will automatically succeed on tirzepatide. Understanding which categories of semaglutide failure respond best to switching helps you set realistic expectations.
Plateau responders: the strongest candidates
If semaglutide worked initially but stalled after 4 to 6 months, you are the ideal candidate for switching. Your body responded to GLP-1 agonism. The mechanism works for you. You simply need more metabolic stimulus to push past the adaptation your body has made.
Tirzepatide provides exactly that. The additional GIP receptor activation gives your metabolism a new signal it has not adapted to. Many people who plateau on semaglutide see renewed, significant weight loss within the first 8 to 12 weeks of tirzepatide. The timeline for tirzepatide to work tends to be faster for previous GLP-1 users because the body already has some metabolic priming from semaglutide exposure.
Partial responders: strong candidates
You lost some weight on semaglutide but never achieved a clinically meaningful amount, typically defined as 5% or more of body weight. Maybe you dropped 8 or 10 pounds over several months and then the loss stopped entirely.
Partial response suggests your GLP-1 pathway is active but insufficient on its own. The dual mechanism of tirzepatide often closes this gap. The SURMOUNT-5 data showing 47% greater weight loss with tirzepatide suggests that many partial semaglutide responders would cross the threshold into meaningful weight loss with the stronger medication.
Side effect quitters: promising candidates
You stopped semaglutide because of intolerable side effects, usually nausea, vomiting, or severe gastrointestinal distress. You never reached a therapeutic dose because your body could not handle the titration.
The SURMOUNT-5 data showing lower discontinuation rates with tirzepatide (2.7% versus 5.6%) is encouraging but not a guarantee. Some researchers theorize that tirzepatide's biased agonism, particularly its stronger GIP activity relative to GLP-1 activity, may produce a different side effect profile that certain people tolerate better.
Start at the lowest dose. Titrate slowly. Give each dose four full weeks before increasing. This is not the time to rush.
True non-responders: uncertain outcomes
If semaglutide produced absolutely no appetite suppression, no metabolic improvement, and no weight loss even at the maximum 2.4mg dose over 16 or more weeks, you may have fundamental resistance to GLP-1 receptor agonism.
Tirzepatide can still work for true GLP-1 non-responders because of the GIP pathway. But the outcomes are less predictable. Some people with GLP-1 receptor polymorphisms respond beautifully to tirzepatide because the GIP receptor carries the metabolic load. Others with broader metabolic issues may need different interventions entirely.
A provider who specializes in obesity medicine can help determine whether tirzepatide is likely to work or whether other approaches, such as combination therapy, adding phentermine, or investigating underlying conditions, make more sense.
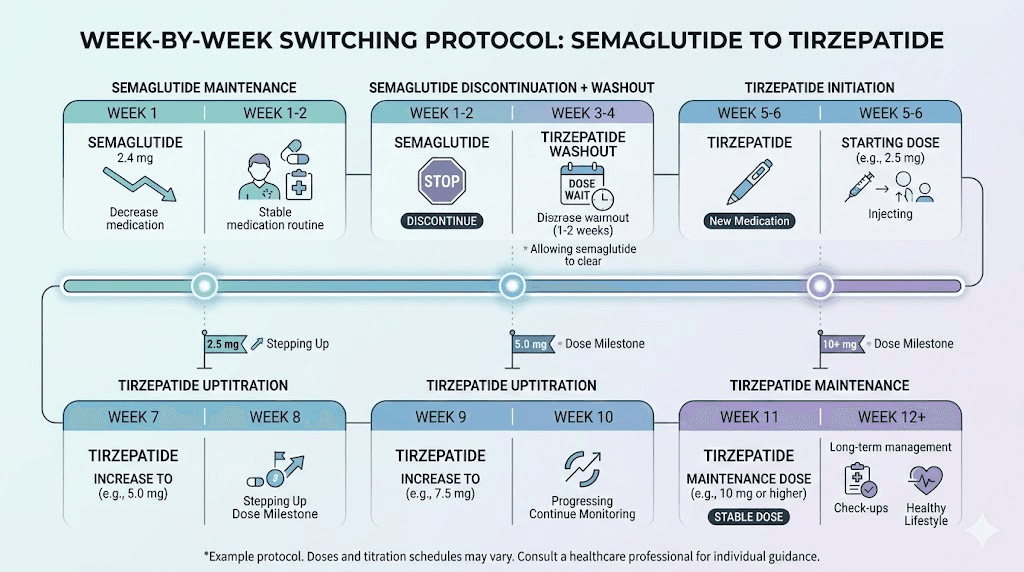
How to switch from semaglutide to tirzepatide: step by step protocol
If you have decided to make the switch, here is exactly how to do it safely and effectively. This section covers timing, dosing, what to expect, and how to manage the transition period.
Step 1: determine your last semaglutide dose
Take your final dose of semaglutide on your regular injection day. Note the date and the dose you were on. This information matters for managing your transition timeline and for communicating with your provider about your medication history.
Do not taper off semaglutide. There is no clinical evidence that tapering provides any benefit before switching. Simply take your last dose and prepare for the transition.
Step 2: wait one week before starting tirzepatide
Start tirzepatide approximately one week after your last semaglutide injection, which typically aligns with your next scheduled injection day. Both medications are administered weekly, so the timing usually works out naturally.
Semaglutide has a half-life of approximately 7 days, meaning it takes about a week for the drug levels to drop by half. Starting tirzepatide one week after your last semaglutide dose means some semaglutide will still be in your system. This overlap is generally well tolerated and prevents a gap in GLP-1 receptor coverage that could cause appetite rebound or blood sugar spikes.
Step 3: start tirzepatide at 2.5mg regardless of your semaglutide dose
This is critical. Even if you were on the maximum 2.4mg of semaglutide, you start tirzepatide at 2.5mg. There is no dose equivalence between these medications. They have different receptor profiles, different pharmacokinetics, and different potency curves.
Starting at the lowest dose allows your body to adjust to the additional GIP receptor activation, which is an entirely new stimulus your system has not experienced before. Skipping doses or starting higher increases the risk of severe nausea, vomiting, and other gastrointestinal side effects that could derail the entire switch.
Step 4: follow the standard tirzepatide titration schedule
The standard tirzepatide titration increases the dose every four weeks:
Weeks 1-4: 2.5mg weekly
Weeks 5-8: 5mg weekly
Weeks 9-12: 7.5mg weekly
Weeks 13-16: 10mg weekly
Weeks 17-20: 12.5mg weekly
Weeks 21+: 15mg weekly (maximum dose)
Not everyone needs to reach 15mg. Many people find their optimal response at 10mg or 12.5mg. Your provider will help you determine when you have reached the dose that balances efficacy with tolerability.
Step 5: monitor your response during the transition
During the first four to six weeks after switching, pay attention to these markers:
Appetite changes: Most people notice appetite suppression returning or intensifying within the first two weeks on tirzepatide, even at the starting 2.5mg dose. If you were a semaglutide non-responder, watch for any appetite changes at all. Even subtle shifts suggest the GIP pathway is engaging.
Gastrointestinal symptoms: Mild nausea is common during the first week at each new dose. It typically subsides within 3 to 5 days. If nausea persists beyond a week at any dose, discuss slowing your titration with your provider.
Energy levels: Some people experience a noticeable increase in energy on tirzepatide, potentially related to improved glucose metabolism and the effects of GIP receptor activation. Others feel fatigued initially, similar to what happens when the body adjusts to any new metabolic medication.
Weight changes: Do not expect dramatic weight loss in the first month at 2.5mg. The starting dose is designed for tolerability, not maximum efficacy. Real weight loss typically begins at the 5mg to 7.5mg range and accelerates as you titrate higher.
What the approximate dose conversion looks like
There is no officially validated dose conversion between semaglutide and tirzepatide. Their mechanisms are too different for a simple milligram-to-milligram comparison. However, clinical experience provides approximate equivalencies that help set expectations for where your therapeutic dose may land.
Approximate equivalency chart
These are rough guidelines based on clinical observation, not precise conversions:
Semaglutide 0.25-0.5mg corresponds roughly to tirzepatide 2.5-5mg
Semaglutide 1mg corresponds roughly to tirzepatide 5-7.5mg
Semaglutide 1.7mg corresponds roughly to tirzepatide 7.5-10mg
Semaglutide 2.4mg corresponds roughly to tirzepatide 10-15mg
These equivalencies suggest where you might eventually land, not where you should start. You still start at 2.5mg regardless. The conversion simply helps predict how quickly you might need to titrate to match or exceed your previous level of appetite suppression and weight loss. For a more detailed breakdown, see the semaglutide to tirzepatide conversion chart.
Why the conversion is not exact
Tirzepatide is not just "stronger semaglutide." It is a different type of medication with a different receptor profile. Comparing doses directly is like comparing aspirin milligrams to ibuprofen milligrams. Both reduce pain, but they work through different mechanisms at different potencies.
The GIP component of tirzepatide means that even at what might seem like a "low equivalent" dose, you could experience effects that semaglutide never produced. Conversely, some GLP-1 specific effects might feel different or delayed as your body adjusts to the new receptor balance.
Managing side effects during the switch
Side effects are the primary reason people abandon GLP-1 medications before reaching therapeutic doses. Since you are already experienced with GLP-1 side effects from semaglutide, you have some advantages. But tirzepatide introduces GIP-related effects that may feel new.
Gastrointestinal effects
Nausea, constipation, diarrhea, and reduced appetite are the most common side effects with both medications. During the switch, you may experience these symptoms even if you tolerated semaglutide well, because the GIP receptor activation is new to your system.
Practical management strategies include eating smaller, more frequent meals rather than two or three large ones, avoiding high-fat and fried foods especially during the first week at each new dose, staying hydrated with at least 64 ounces of water daily, and considering a tirzepatide-friendly meal plan that prioritizes lean protein and easily digestible foods.
Injection site reactions
Some people experience more injection site reactions with tirzepatide than they did with semaglutide. Redness, itching, or a small bump at the injection site can occur. These typically resolve within a few days and are more common at higher doses.
Rotating injection sites between abdomen, stomach, thigh, and upper arm minimizes this. Allow the medication to reach room temperature before injecting. And inject slowly rather than quickly.
Fatigue and energy shifts
During the first two to three weeks after switching, some people report temporary fatigue. This usually reflects the metabolic adjustment to dual receptor activation. Your body is literally rewiring its incretin signaling, and that takes energy.
Ensure adequate protein intake (at least 0.8 grams per pound of body weight), prioritize sleep, and avoid dramatic calorie restriction during the transition. Your body needs nutrients to adapt. Starving yourself during a medication switch is counterproductive.
Mood and cognitive effects
GIP and GLP-1 receptors exist in the brain. When you switch medications, the receptor activation profile in your central nervous system changes. Some people report temporary mood shifts, mild sleep disruption, or changes in food-related thoughts.
These effects are usually transient. If mood changes persist beyond three to four weeks or are severe, consult your provider. In rare cases, the change in central nervous system receptor activation can unmask or exacerbate anxiety or depression that the previous medication was inadvertently managing.
Realistic timelines: when to expect results after switching
Expectations kill more medication protocols than side effects do. Here is an honest, research-backed timeline for what to expect when switching from semaglutide to tirzepatide.
Weeks 1-4 (2.5mg): the adjustment phase
Weight loss during this period is typically minimal, 0 to 3 pounds at most. The 2.5mg starting dose is subtherapeutic for weight loss. Its purpose is tolerability and allowing your body to adjust to GIP receptor activation.
What you should notice: some appetite reduction, possibly different from what semaglutide produced. Some people describe it as a quieter appetite suppression, where food thoughts simply diminish rather than the strong "food aversion" some experienced with semaglutide.
Weeks 5-12 (5mg-7.5mg): early response phase
This is when meaningful weight loss typically begins. At 5mg, most people experience noticeable appetite suppression. By 7.5mg, the dual mechanism is fully engaging. Average weight loss during this phase ranges from 4 to 10 pounds, though individual variation is significant.
People who plateaued on semaglutide often see the most dramatic response during this phase. The new GIP receptor stimulus restarts metabolic pathways that had adapted to pure GLP-1 agonism. It is not uncommon for plateau patients to lose more weight in weeks 5 through 12 of tirzepatide than they lost in their final three to four months on semaglutide.
Weeks 13-24 (10mg-15mg): therapeutic phase
By this point you should be at or approaching your maintenance dose. Weight loss accelerates through this phase as the medication reaches full potency. Based on SURMOUNT-5 data, average monthly weight loss at therapeutic doses is approximately 1.5 to 2.5% of starting body weight.
For someone starting at 250 pounds, that translates to roughly 4 to 6 pounds per month during the therapeutic phase. Cumulative weight loss from the start of tirzepatide might reach 15 to 25 pounds by the six-month mark, with continued loss thereafter.
Months 6-18: sustained response
The SURMOUNT-5 trial ran for 72 weeks (about 18 months) and showed continued weight loss throughout the study period for most tirzepatide users. Unlike semaglutide, where plateaus are common around month 4 to 6, tirzepatide users in clinical trials showed more sustained weight loss curves, likely because the dual mechanism provides a broader metabolic stimulus that is harder for the body to fully adapt to.
Long-term, the 20.2% average weight loss seen in SURMOUNT-5 gives you a reasonable benchmark. Some people exceed this significantly. Others fall below it. But it represents the central tendency for what tirzepatide achieves at maximum tolerated doses over 18 months.
What to do if tirzepatide also does not work
Let us address the question nobody wants to ask. What if you switch to tirzepatide and it still does not work? What then?
First, define "not working"
Give tirzepatide a fair trial. That means reaching at least 10mg (ideally 15mg) and staying at that dose for a minimum of 12 weeks before concluding it has failed. Starting at 2.5mg and quitting at 5mg because you have not lost weight is not a fair trial. You would not judge semaglutide based on results at 0.25mg.
Define failure objectively. Less than 5% body weight loss after 16 or more weeks at a therapeutic dose (10mg or above) constitutes a poor response. Some appetite suppression with slow but steady weight loss at 1 to 2 pounds per month is not failure. It is a suboptimal response that may still be clinically meaningful.
Combination therapy approaches
If tirzepatide alone is insufficient, combination therapy may be the next step. Adding phentermine to tirzepatide provides sympathomimetic appetite suppression through a completely different pathway. This combination targets three separate appetite and metabolism pathways simultaneously: GLP-1, GIP, and norepinephrine.
Other combination options include metformin for insulin resistance, topiramate for appetite and food cravings, and naltrexone/bupropion for reward-driven eating patterns. These are prescription decisions that require provider evaluation and monitoring.
Investigating root causes
If two different incretin-based medications have failed, the problem may not be the medications. It may be something upstream that is preventing any weight loss medication from working effectively.
Comprehensive evaluation should include complete thyroid panel (not just TSH), fasting insulin and HOMA-IR for insulin resistance assessment, cortisol testing for adrenal dysfunction, sex hormone panel (testosterone, estrogen, progesterone), sleep study if sleep apnea is suspected, medication review for weight-promoting drugs, and psychological evaluation for eating disorders or food addiction.
Next-generation medications
The GLP-1 medication landscape is evolving rapidly. Retatrutide is a triple receptor agonist that targets GLP-1, GIP, and glucagon receptors simultaneously. Early clinical trials show weight loss exceeding 24% of body weight, surpassing even tirzepatide. For people who fail dual agonism, triple agonism may provide the additional stimulus needed.
CagriSema, a combination of cagrilintide (an amylin analog) and semaglutide, is another promising option that adds amylin receptor activation to GLP-1 agonism. Survodutide, a dual GLP-1/glucagon receptor agonist, takes yet another approach by leveraging glucagon's thermogenic properties.
The point is this: even if both semaglutide and tirzepatide fail, the medication pipeline contains multiple next-generation options targeting different receptor combinations. The field is moving fast, and options are expanding.
Optimizing your tirzepatide results: practical strategies
Switching medications alone is not enough. If you want to maximize your tirzepatide results, especially after semaglutide failure, these evidence-based strategies make a significant difference.
Protein prioritization
Inadequate protein intake is the single most common dietary mistake people make on GLP-1 medications. When your appetite is suppressed, you eat less of everything, including protein. This leads to muscle loss, which lowers your metabolic rate, which causes weight loss to stall.
Target a minimum of 0.8 grams of protein per pound of goal body weight. For someone targeting 160 pounds, that is at least 128 grams of protein daily. Spread it across at least three meals. Consider a protein shake if hitting your target through whole foods alone is difficult with a suppressed appetite.
Supplements that support tirzepatide include a high-quality multivitamin (to compensate for reduced food intake), magnesium citrate (helps with constipation and sleep), omega-3 fatty acids (anti-inflammatory support), and vitamin D (often depleted in people with obesity).
Resistance training
This cannot be overstated. Resistance training is not optional when you are on GLP-1 medications. Without it, up to 40% of the weight you lose may come from lean muscle mass rather than fat. This destroys your metabolic rate and sets you up for rebound weight gain.
You do not need to become a powerlifter. Two to three sessions per week of compound exercises, such as squats, deadlifts, rows, and presses, performed at moderate intensity, is sufficient to preserve muscle mass during weight loss. Even bodyweight exercises provide meaningful muscle preservation if you cannot access a gym.
Sleep optimization
Poor sleep disrupts leptin, ghrelin, and cortisol, the three hormones most directly involved in appetite regulation and fat storage. Seven to nine hours of quality sleep per night supports the metabolic environment that allows tirzepatide to work optimally.
If tirzepatide causes sleep disruption, try injecting earlier in the day rather than at night. Some people find that morning injections produce less sleep interference than evening injections.
Meal timing and composition
Tirzepatide slows gastric emptying, meaning food stays in your stomach longer. Eating large meals, especially high-fat meals, exacerbates nausea and bloating. Smaller, more frequent meals work better for most people on this medication.
A practical approach: three moderate meals plus one to two protein-rich snacks, totaling 1,200 to 1,800 calories daily depending on your size and activity level. Front-load protein at each meal. Include fiber-rich vegetables for satiety and gut health. Limit added sugars and highly processed foods, which can trigger cravings even on appetite-suppressing medications.
For specific meal ideas, explore our tirzepatide diet plan and foods to avoid on tirzepatide guides.
Hydration
Dehydration is sneaky on GLP-1 medications. Reduced food intake means reduced water intake from food sources. Add in potential diarrhea or vomiting during dose adjustments, and mild dehydration is common. This slows metabolism, increases constipation, and can cause dry mouth, dizziness, and fatigue.
Aim for a minimum of 64 ounces of water daily, more if you exercise or live in a hot climate. Electrolyte supplementation can help, especially during the first few weeks of a new dose.
Stress management and cortisol control
Chronic stress elevates cortisol. Cortisol promotes visceral fat storage, increases appetite for high-calorie foods, and disrupts insulin sensitivity. Even the most powerful GLP-1 medication cannot fully override chronically elevated cortisol levels.
If you are switching to tirzepatide after semaglutide failure, audit your stress levels honestly. Are you sleeping enough? Are you exercising too intensely (which raises cortisol)? Are work or relationship stressors consuming your mental energy? Addressing these factors does not just improve your tirzepatide results. It improves every aspect of your metabolic health.
Practical stress interventions that evidence supports include a 10-minute daily walking practice, progressive muscle relaxation before bed, limiting caffeine after noon, and reducing screen time in the hour before sleep. These are not luxury wellness add-ons. They are metabolic interventions that directly influence whether your medication works optimally.
Alcohol and tirzepatide
Drinking on tirzepatide requires more caution than most people realize. Alcohol provides empty calories (7 calories per gram) that bypass appetite regulation. It impairs liver function, which affects glucose metabolism. And it disrupts sleep architecture, even when it seems to help you fall asleep faster. The sleep you get after drinking is lower quality, which negatively affects next-day hunger hormones.
If you consumed alcohol regularly on semaglutide and did not lose weight, this could be a contributing factor regardless of which medication you take. During the tirzepatide transition, consider eliminating alcohol for at least 8 to 12 weeks to give the medication the best possible environment to work. Many people find that GLP-1 medications naturally reduce their desire to drink, and tirzepatide's additional GIP pathway may amplify this effect. For guidance on which drinks have the least impact, see best alcohol to drink on tirzepatide.
Cost considerations when switching from semaglutide to tirzepatide
The financial reality of switching medications matters. Insurance coverage, compounded alternatives, and long-term cost planning all factor into whether the switch is feasible.
Brand-name medication costs
Brand-name tirzepatide (Zepbound for weight loss, Mounjaro for diabetes) carries a higher list price than brand-name semaglutide (Wegovy for weight loss, Ozempic for diabetes). Without insurance, tirzepatide costs approximately $1,000 to $1,200 per month at retail pharmacies. Semaglutide runs approximately $800 to $1,100 per month.
However, manufacturer discount programs, pharmacy coupons, and insurance negotiated rates can significantly reduce these costs. Many patients pay far less than list price. If you have documented semaglutide failure, your provider can use this as leverage for insurance approval of tirzepatide coverage.
Compounded tirzepatide as an alternative
Affordable tirzepatide options exist through compounding pharmacies. Compounded tirzepatide typically costs $150 to $500 per month depending on the dose, pharmacy, and formulation. This represents significant savings compared to brand-name options.
When evaluating compounding pharmacies, look for 503B outsourcing facilities that operate under FDA oversight. These pharmacies have stricter quality controls than 503A pharmacies and must follow current good manufacturing practices. Reputable options include Empower Pharmacy, Strive Pharmacy, and others that have established track records with GLP-1 compounding.
Be cautious with grey market tirzepatide sources. Unregulated products may contain incorrect concentrations, contaminants, or entirely different compounds. The cost savings are not worth the risk of injecting something that could harm you or simply waste your time with an ineffective product.
The true cost of not switching
If semaglutide is not working, every month you continue paying for it is money wasted. Factor in the cost of the medication that is not producing results, the ongoing health costs of obesity (medications for blood pressure, diabetes, joint pain), and the opportunity cost of delayed weight loss.
Switching to tirzepatide, even at a higher monthly cost, may actually save money long-term if it produces the metabolic improvements that semaglutide could not. Reduced need for diabetes medications, blood pressure medications, and other obesity-related treatments can offset the medication cost difference within months.
Common questions people ask when considering the switch
Do I need a washout period between semaglutide and tirzepatide?
No formal washout period is required. Most providers recommend starting tirzepatide approximately one week after your last semaglutide dose. This typically aligns with your next scheduled injection day. Some overlap of the two medications in your system is expected and generally well tolerated.
Will my insurance cover tirzepatide if semaglutide did not work?
Coverage varies significantly by plan. Many insurance plans now recognize documented semaglutide failure as a valid reason to approve tirzepatide coverage, particularly for patients with type 2 diabetes or BMI above 30. Your provider can submit a prior authorization documenting your semaglutide trial, the inadequate response, and the clinical rationale for switching. Some plans require a step therapy failure (documented trial on semaglutide) before covering tirzepatide.
Can I switch back to semaglutide if tirzepatide does not work either?
Yes. Switching between GLP-1 medications is reversible. If tirzepatide does not produce better results, you can return to semaglutide or try a different approach entirely. There is no physiological reason that trying tirzepatide would make semaglutide less effective if you return to it.
Is compounded tirzepatide an option?
Compounded tirzepatide is available from compounding pharmacies and is typically less expensive than brand-name Zepbound or Mounjaro. Compounded formulations use the same active ingredient but may differ in concentration, requiring careful unit calculation. Use our compounded tirzepatide dosage calculator to ensure accurate dosing if you go this route.
How long should I try tirzepatide before deciding it does not work?
Give tirzepatide a minimum of 16 weeks at a therapeutic dose (10mg or higher) before evaluating efficacy. This means you need approximately 20 to 24 weeks total, accounting for the titration period. Judging the medication during the titration phase produces misleading conclusions because you are not yet at a dose designed to produce maximum weight loss.
What if I was already on the highest semaglutide dose?
If you were at 2.4mg semaglutide and it was not working, you still start tirzepatide at 2.5mg. However, you may need to titrate to the 15mg maximum dose of tirzepatide to see the full benefit. The dose conversion data suggests that maximum semaglutide roughly corresponds to tirzepatide 10-15mg, so maximum tirzepatide provides meaningfully more metabolic stimulus than maximum semaglutide.
Frequently asked questions
If semaglutide does not suppress my appetite will tirzepatide?
In many cases, yes. Tirzepatide targets both GLP-1 and GIP receptors, providing appetite suppression through two pathways instead of one. Even if your GLP-1 receptors are genetically less responsive, the GIP receptor pathway can still significantly reduce hunger and food noise. Clinical data shows tirzepatide produces greater appetite reduction than semaglutide for most people.
How much more weight will I lose on tirzepatide compared to semaglutide?
The SURMOUNT-5 trial showed tirzepatide produces about 47% more weight loss than semaglutide on average, which is 20.2% versus 13.7% body weight reduction over 72 weeks. For someone weighing 250 pounds, that difference translates to roughly 16 additional pounds lost over 18 months. Individual results vary, but the dual mechanism consistently outperforms single GLP-1 agonism in both clinical trials and real-world outcomes.
Are tirzepatide side effects worse than semaglutide?
Surprisingly, the SURMOUNT-5 trial found that fewer people discontinued tirzepatide due to side effects (2.7%) compared to semaglutide (5.6%). While gastrointestinal side effects are common with both, tirzepatide may actually be better tolerated for some people. The side effect profile is similar in type, with nausea, constipation, and diarrhea being most common, but the severity and tolerability may differ due to the different receptor activation pattern.
Can genetics explain why semaglutide failed but tirzepatide might work?
Yes. Polymorphisms in the GLP1R gene, particularly the rs6923761 variant, can reduce your response to pure GLP-1 agonists like semaglutide. Tirzepatide bypasses this issue by heavily engaging GIP receptors, which are encoded by a separate gene (GIPR). If your GLP-1 receptors are genetically suboptimal but your GIP receptors function normally, tirzepatide can deliver results through the alternative pathway.
Should I try a higher dose of semaglutide before switching to tirzepatide?
If you have not yet tried the maximum 2.4mg dose of semaglutide, it may be worth maximizing that dose first before switching. Some people see meaningful improvement at higher semaglutide doses after plateauing at lower ones. However, if you have been at maximum dose for 12 or more weeks without adequate response, or if side effects prevent further dose increases, switching to tirzepatide is a reasonable next step.
What happens to my blood sugar control when I switch?
For people with type 2 diabetes, blood sugar control typically improves or at least maintains during the switch. The SURPASS trials showed tirzepatide produced greater HbA1c reductions than semaglutide at all dose levels. However, there may be brief fluctuations during the transition period when tirzepatide is at its starting dose. Monitor blood sugar more frequently during the first four to six weeks after switching.
Can I combine semaglutide and tirzepatide?
Taking semaglutide and tirzepatide together is generally not recommended. Since tirzepatide already includes GLP-1 receptor agonism, adding semaglutide would double the GLP-1 stimulation without providing the additional GIP benefit. This increases side effect risk without proportional benefit. If tirzepatide alone is insufficient, combination with a different class of medication, like phentermine or metformin, is a more logical approach.
Is oral tirzepatide an option if I hate injections?
Oral formulations of tirzepatide are in development and early clinical trials show promising results. Oral tirzepatide and tirzepatide ODT (orally disintegrating tablets) may eventually offer needle-free alternatives. Tirzepatide drops and sublingual formulations are also being explored. For now, subcutaneous injection remains the standard delivery method.
Storage, handling, and practical logistics of switching
The practical side of switching medications gets overlooked in most guides. But logistics matter because poor storage destroys medication potency, and incorrect handling leads to dosing errors that undermine your results.
Storing tirzepatide properly
Tirzepatide requires refrigeration between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). If you are using compounded tirzepatide from a 503B compounding pharmacy, storage requirements may vary slightly depending on the formulation. Always check the specific storage instructions from your pharmacy.
How long does tirzepatide last in the fridge? Unopened vials typically remain stable for their labeled shelf life when refrigerated. Once reconstituted (for compounded versions), most formulations are good for 28 to 42 days in the refrigerator. Never freeze tirzepatide. And if your medication gets warm during shipping or a power outage, check the specific temperature and duration guidelines before using it.
Traveling during the transition
If you travel frequently, plan your switch timing carefully. The titration period requires consistent weekly injections. Missing doses during titration slows the entire process and may require restarting at a lower dose.
For travel logistics, use an insulated medication pouch with ice packs to maintain cold chain. Carry your prescription documentation for airport security. And consider timing your injection the day before you travel rather than mid-flight. For semaglutide travelers switching over, travel with GLP-1 medications follows similar principles for tirzepatide.
Reconstituting compounded tirzepatide
If you are using compounded tirzepatide in lyophilized (powder) form, you will need to reconstitute it with bacteriostatic water before injection. The amount of water you add determines the concentration, which directly affects your dosing accuracy.
For a 10mg vial reconstituted with 2ml of bacteriostatic water, the concentration is 5mg/ml. For a 30mg vial, refer to the 30mg tirzepatide reconstitution guide for exact calculations. Using the right amount of bacteriostatic water is critical. Too much water means underdosing. Too little means overdosing. Neither produces optimal results.
Use a peptide reconstitution calculator to determine exact volumes. Inject the bacteriostatic water slowly down the side of the vial. Swirl gently. Never shake. Allow the powder to dissolve completely before drawing your dose.
Injection technique for tirzepatide
Tirzepatide is injected subcutaneously, meaning into the fat layer just beneath the skin. The technique is essentially identical to semaglutide injection. If you were comfortable injecting semaglutide with a syringe, tirzepatide requires no new skills.
Preferred injection sites include the abdomen (at least two inches from the navel), the front of the thigh, and the back of the upper arm. Rotate sites with each injection to prevent injection site reactions and lipodystrophy. If you experienced injection site issues with semaglutide, try a different anatomical area with tirzepatide.
For the best time to take your tirzepatide shot, many people find morning injections work well because any nausea tends to subside by afternoon. Others prefer evening injection so they sleep through the initial GI effects. There is no pharmacological difference in efficacy based on injection timing. Choose what works best for your schedule and comfort.
Tracking your progress: what to measure and when
Switching medications without tracking your results is like driving without a map. You might get somewhere, but you will not know if you are on course.
Weight tracking protocol
Weigh yourself at the same time every day, preferably first thing in the morning after using the bathroom and before eating or drinking. Record the daily weight but focus on the weekly average. Daily weight fluctuates by 2 to 5 pounds based on water retention, sodium intake, bowel activity, and hormonal cycles. The weekly trend is what matters.
Create a baseline by calculating your average weight during the final week on semaglutide. Compare all tirzepatide progress against this baseline, not against your all-time highest weight. This gives you an honest picture of what the medication switch specifically contributed.
Body measurements
The scale does not tell the whole story. If you are doing resistance training (which you should be), you may gain muscle while losing fat, resulting in minimal scale movement but significant body composition improvement.
Measure waist circumference at the navel, hip circumference at the widest point, and chest circumference at the nipple line every two weeks. These measurements capture fat loss that the scale misses. The SURMOUNT-5 trial showed tirzepatide reduced waist circumference by 18.4cm, and tracking this number gives you clinical-trial-grade data on your own progress.
Subjective markers
Rate these on a 1 to 10 scale weekly: appetite level (how hungry you feel on average), food noise (how often you think about food), energy level, sleep quality, and mood. These subjective markers often change before the scale does and provide early signals about whether tirzepatide is engaging your metabolic pathways.
If appetite and food noise decrease within the first two weeks on tirzepatide, even at the starting 2.5mg dose, that is a strong positive signal. It means the GIP/GLP-1 dual pathway is activating your satiety centers. Weight loss typically follows within weeks.
Blood work timeline
Get baseline blood work before starting tirzepatide: complete metabolic panel, HbA1c, fasting insulin, lipid panel, and thyroid function. Repeat at 12 weeks and 24 weeks. This objective data tells you whether the medication is improving your metabolic health, which matters independently of weight loss. Some people see dramatic improvement in insulin sensitivity, triglycerides, and inflammatory markers even before significant weight loss occurs.
Special populations: specific considerations when switching
Women of reproductive age
Both semaglutide and tirzepatide can affect fertility and are not recommended during pregnancy or breastfeeding. If you are breastfeeding or planning to become pregnant, discuss timing with your provider before switching. Tirzepatide should be discontinued at least two months before planned conception due to its long half-life.
Weight loss itself can restore ovulation in women with obesity-related anovulation. Some women who were not ovulating on semaglutide may begin ovulating on tirzepatide as additional weight comes off. Use reliable contraception if pregnancy is not desired. Read more about how tirzepatide affects your period and getting pregnant on tirzepatide.
People with type 2 diabetes
Switching GLP-1 medications when you have diabetes requires careful blood sugar monitoring. Tirzepatide's greater glucose-lowering effect compared to semaglutide means hypoglycemia risk may increase, especially if you are also taking sulfonylureas or insulin.
Your provider may need to reduce concurrent diabetes medications during the transition. Monitor blood glucose at least four times daily for the first two to three weeks after switching. Watch for symptoms of hypoglycemia: shakiness, sweating, confusion, rapid heartbeat.
People taking other weight loss medications
Some people switch to tirzepatide while already taking other weight-related medications. Phentermine combined with semaglutide is a common combination. When switching the GLP-1 component to tirzepatide, your provider may adjust or temporarily discontinue the phentermine during titration to isolate the effects of the new medication and manage side effects individually.
Metformin is another common co-prescription. It generally continues unchanged during the switch because it works through an entirely different mechanism (reducing hepatic glucose production) and does not interact with incretin receptors. Metformin and tirzepatide together is a well-tolerated combination that many providers use for patients with type 2 diabetes.
If you were using berberine with semaglutide, the same principle applies when switching to tirzepatide. Berberine acts on AMP-activated protein kinase and can complement the incretin pathway without significant interaction risk. However, monitor blood sugar closely as the combined glucose-lowering effects may be stronger with tirzepatide than they were with semaglutide.
People on multiple medications
GLP-1 medications slow gastric emptying, which can affect absorption of oral medications. If you are taking medications that require consistent absorption, such as oral contraceptives, thyroid medications, or certain antibiotics, discuss timing adjustments with your provider.
This effect may be more pronounced with tirzepatide than semaglutide due to the dual receptor activation. Take critical medications at least one hour before your first meal, and consider whether any medication timing needs adjustment during the switch.
People with a history of pancreatitis or thyroid conditions
Both semaglutide and tirzepatide carry warnings about thyroid C-cell tumors (based on rodent studies) and pancreatitis risk. If you experienced any pancreatic symptoms on semaglutide, such as severe abdominal pain radiating to the back, discuss this thoroughly with your provider before starting tirzepatide. The risk profile is similar for both medications, but individual sensitivity varies.
People with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 should not use either medication. This contraindication applies equally to semaglutide and tirzepatide.
The role of SeekPeptides in your GLP-1 journey
Navigating GLP-1 medication decisions, from understanding why semaglutide failed to evaluating whether tirzepatide is the right next step, requires reliable, evidence-based information. SeekPeptides provides exactly that. With comprehensive dosing guides, dosage calculators, and detailed articles on every aspect of GLP-1 therapy, the platform helps you make informed decisions backed by clinical data rather than marketing hype.
SeekPeptides members get access to personalized protocol guidance, detailed conversion tools, and a community of researchers who have navigated these exact switching decisions. Whether you need help calculating your tirzepatide syringe dosage, understanding your reconstitution ratios, or planning your starting dose protocol, the resources are built for exactly this kind of decision-making.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
External resources
SURMOUNT-5 Trial: Tirzepatide vs Semaglutide for Obesity (New England Journal of Medicine)
SURPASS-2 Trial: Tirzepatide vs Semaglutide for Type 2 Diabetes (New England Journal of Medicine)
Tirzepatide Dual GIP/GLP-1 Receptor Agonist Mechanism (JCI Insight)
Why Weight Loss Drugs Do Not Work for Everyone: Genetics (Scientific American)
Factors Associated with Weight Loss Response to GLP-1 Analogues (PMC)
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your doses stay optimized, and your results stay consistent.