Feb 20, 2026

Before you attribute that new ache in your knees or hips to tirzepatide, consider this. Clinical trial data from the SURPASS and SURMOUNT programs, involving tens of thousands of participants, did not flag joint pain as a common adverse reaction. And a large population study of over 62,000 tirzepatide users actually found lower rates of joint pain compared to other weight loss treatments. Yet the question persists across forums, clinic waiting rooms, and late-night search sessions. Something does not add up.
The reality is more nuanced than a simple yes or no. Some people do experience joint discomfort after starting tirzepatide. But the mechanism behind that discomfort tells a different story than most assume.
Rapid weight loss changes how your body moves. Muscle loss weakens the scaffolding around your joints. Nutrient gaps from eating less quietly erode the building blocks your connective tissue needs. These factors, not the medication itself, appear to drive most of the joint complaints researchers have documented.
This guide breaks down every piece of evidence connecting tirzepatide to joint pain. You will find the actual clinical trial numbers, the biomechanical explanations, the timeline of when pain typically appears and resolves, and the specific strategies that prevent or manage it. Whether you are considering starting tirzepatide or already experiencing unexpected soreness, the answers are here.
What the clinical evidence actually says about tirzepatide and joint pain
Understanding whether tirzepatide causes joint pain requires separating clinical data from anecdotal reports. The distinction matters. One is measured under controlled conditions with thousands of participants. The other reflects individual experiences shaped by dozens of variables. Both have value, but they tell different stories.
SURPASS and SURMOUNT trial data
The pivotal clinical trials for tirzepatide span two major programs. SURPASS trials evaluated the drug for type 2 diabetes management. SURMOUNT trials tested it specifically for weight loss in people with obesity or overweight. Together, these programs enrolled over 20,000 participants across multiple countries and dosing protocols.
Joint pain was not identified as a common adverse event in either program.
The most frequently reported side effects were gastrointestinal. Nausea, diarrhea, decreased appetite, and vomiting dominated the adverse event profiles across all dose levels. Musculoskeletal complaints, including arthralgia (the clinical term for joint pain), occurred at rates comparable to placebo groups. This is a critical detail. When a side effect occurs at similar rates in people taking the drug and people taking a sugar pill, the drug itself is unlikely to be the cause.
The FDA prescribing information for both Mounjaro (tirzepatide for diabetes) and Zepbound (tirzepatide for weight management) does not list arthralgia as a common adverse reaction. Joint inflammation is not identified as a known pharmacologic effect of GIP or GLP-1 receptor activation. The side effect profile of tirzepatide centers firmly on the digestive system, not the musculoskeletal system.
Real-world population studies paint a surprising picture
Clinical trials have limitations. They run for defined periods, exclude certain populations, and cannot capture every real-world variable. That is why observational studies matter. And the largest one to date tells an unexpected story about tirzepatide and joint pain.
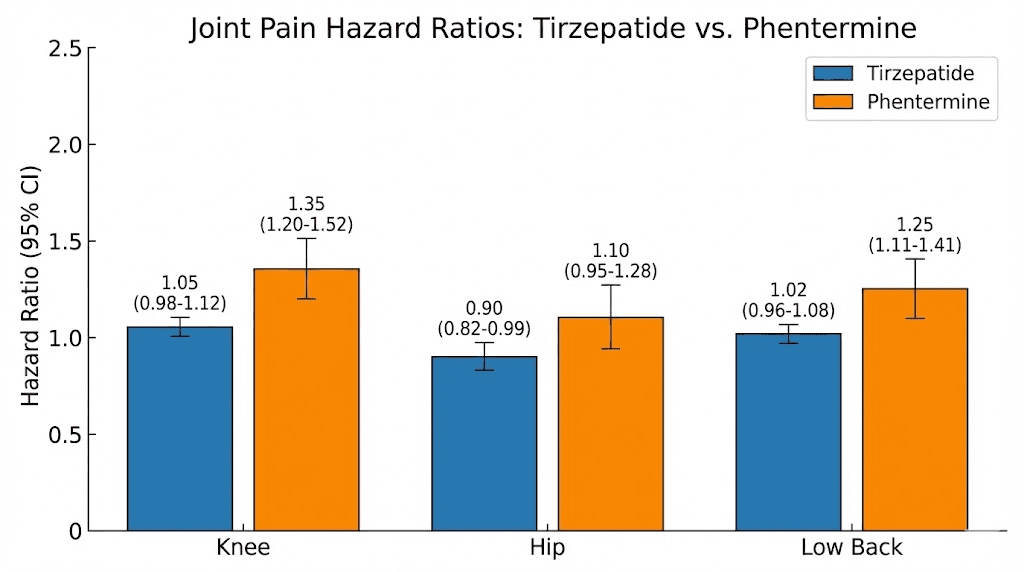
A study presented at the American College of Rheumatology examined musculoskeletal outcomes in over 62,000 tirzepatide users compared to matched patients using phentermine for weight loss. The results challenged the assumption that tirzepatide worsens joint pain.
Tirzepatide initiation was associated with an overall lower risk of joint pain, with a hazard ratio of 0.91. Breaking that down by location:
Knee pain: hazard ratio 0.84 (16% lower risk)
Hip pain: hazard ratio 0.88 (12% lower risk)
Low back pain: hazard ratio 0.89 (11% lower risk)
These numbers mean that, on a population level, people starting tirzepatide were actually less likely to develop joint pain than people using a different weight loss medication. The effect was most pronounced for knee pain, which makes biological sense given the relationship between body weight and knee joint stress.
The study also found that tirzepatide users had lower rates of NSAID and opioid prescriptions, suggesting they needed less pain management overall. For anyone concerned about joint health on tirzepatide, this data offers meaningful reassurance.
Postmarketing reports and what they mean
Once a drug reaches millions of patients in real-world settings, rare events surface that trials could not detect. Postmarketing surveillance has documented cases of musculoskeletal discomfort in tirzepatide users. One review of 500 patients found that approximately 15% reported some form of body aches after starting treatment. Another dataset suggested around 10% experienced muscle-specific pain.
These numbers sound concerning in isolation. Context changes the picture. Most reports describe generalized aches rather than specific inflammatory joint disease. The discomfort typically appears in the first four weeks, often coinciding with dose escalation, and tends to resolve by weeks four through eight. This temporal pattern suggests an adaptation process rather than cumulative joint damage.
Case reports documenting more serious musculoskeletal events exist but remain rare. A few published cases describe significant muscle pain or unusual reactions following tirzepatide administration. These cases highlight the importance of postmarketing monitoring but do not change the overall safety profile established in controlled trials. For the vast majority of users, joint-specific complaints either do not occur or resolve with time and simple interventions.
Why some people experience joint pain on tirzepatide
If tirzepatide does not directly damage joints, why do some users report pain? The answer lies not in the drug molecule but in the cascade of physical changes the drug triggers. Losing 15 to 25% of your body weight in months, not years, fundamentally alters how your body moves, supports itself, and maintains tissue health. That speed creates a window where things can hurt.
Rapid weight loss and biomechanical shifts
Your body adapted over years or decades to moving at a certain weight. Joints, tendons, ligaments, and the muscles surrounding them all calibrated to support that mass. When tirzepatide drives rapid weight loss, those structures must recalibrate quickly.
Every pound of body weight translates to roughly four pounds of force on the knee during walking. Lose 30 pounds and you remove 120 pounds of force from each step. That sounds purely beneficial, and long term it is. But the transition period introduces new mechanical stresses. Your gait pattern changes. Your center of gravity shifts. Muscles that once fired in a specific sequence to stabilize heavier movements must find new activation patterns.
Think of it like this. A building designed to support 100 tons does not simply become more comfortable if you remove 25 tons overnight. The structural balance shifts. Load-bearing elements that worked perfectly under the original weight may experience unusual stress angles during the transition. The same principle applies to your skeleton. Knees, hips, ankles, and the lumbar spine all undergo biomechanical reorganization during significant weight loss.
This explains why some people develop joint pain they never had before starting treatment. It is not the medication attacking the joints. It is the body learning a new way to move.
Muscle mass loss weakens joint support
This is arguably the most important factor. And it receives far too little attention.
Research shows that approximately 25% of weight lost on tirzepatide comes from lean mass rather than fat. For someone losing 40 pounds, that means roughly 10 pounds of muscle tissue gone. Muscle is not just about strength and appearance. It is the primary stabilizer of every joint in your body. The quadriceps protect your knees. The gluteus muscles protect your hips. The core musculature protects your spine.
Lose muscle and you lose the protective scaffolding around vulnerable joint structures. Cartilage, which has no nerve endings until it degenerates significantly, relies on surrounding muscle to absorb shock and distribute force evenly. Without adequate muscle support, joint surfaces bear loads they were never designed to handle alone.
The problem compounds when reduced appetite leads to insufficient protein intake. Without 1.2 to 1.6 grams of protein per kilogram of body weight daily, the body cannot maintain existing muscle during caloric restriction, let alone build new tissue. Many tirzepatide users eat significantly less and, without conscious effort, their protein consumption drops below maintenance thresholds.
The result? A double vulnerability. Less cushioning from reduced body fat. Less stabilization from reduced muscle mass. Joints caught in the middle.
Nutrient deficiencies from reduced food intake
Tirzepatide dramatically reduces appetite. That is its primary mechanism for weight loss. But eating less means absorbing less of everything, including nutrients critical for joint and connective tissue health.
Magnesium is essential for muscle relaxation and neuromuscular signaling. Deficiency causes muscle cramps, stiffness, and generalized aches that can mimic joint pain. Calcium supports bone density and the structural integrity of joint-adjacent tissues. Vitamin D regulates calcium absorption and plays a direct role in cartilage maintenance. Omega-3 fatty acids modulate inflammatory pathways throughout the body, including in joint synovial fluid.
When caloric intake drops from 2,500 to 1,200 calories daily, getting adequate amounts of these nutrients without supplementation becomes nearly impossible. The body begins prioritizing vital organ function over musculoskeletal maintenance. Joints feel the deficit.
Strategic supplementation addresses this gap directly. It is not about megadosing vitamins. It is about replacing what reduced food intake removes from your daily nutritional equation.
Dehydration and electrolyte imbalances
Water makes up roughly 80% of joint cartilage. Synovial fluid, the lubricant that allows smooth joint movement, depends on adequate hydration to maintain its viscosity and cushioning properties. When you are dehydrated, cartilage compresses more easily and synovial fluid becomes less effective.
Tirzepatide slows gastric emptying and reduces appetite. Many users drink less without realizing it because they feel full more often. Combine reduced fluid intake with the gastrointestinal side effects that can cause additional fluid loss (nausea, diarrhea), and the hydration equation tips negative quickly.
Electrolytes compound the issue. Sodium, potassium, and magnesium all influence muscle contraction, nerve signaling, and fluid distribution around joints. Imbalances create cramping, stiffness, and deep aches that localize around joints even when the joint structures themselves are healthy.
The fix is straightforward. Track water intake deliberately rather than relying on thirst signals, which tirzepatide may blunt along with appetite. Aim for a minimum of half your body weight in ounces daily, and consider electrolyte supplementation during the active weight loss phase.
Pre-existing conditions unmasked by weight changes
Many people starting tirzepatide carry pre-existing joint conditions they may not know about. Obesity itself is a risk factor for osteoarthritis, with excess weight accelerating cartilage degradation over years. Type 2 diabetes creates systemic inflammation that affects joint tissues. Metabolic syndrome damages blood vessels that supply nutrients to cartilage.
Weight loss can unmask these underlying issues in unexpected ways. Reduced inflammation from fat loss may reveal pain signals that chronic inflammation was paradoxically masking. Changed movement patterns may load damaged joint surfaces differently. Increased physical activity, common as people feel better and more mobile during weight loss, may stress joints that deteriorated silently under sedentary conditions.
This does not mean tirzepatide caused the joint problem. It means the weight loss process revealed something that was already there. Understanding this distinction matters for treatment decisions. The solution is not stopping tirzepatide. It is addressing the underlying joint condition while continuing to benefit from weight reduction.
The paradox: how tirzepatide can actually improve joint health
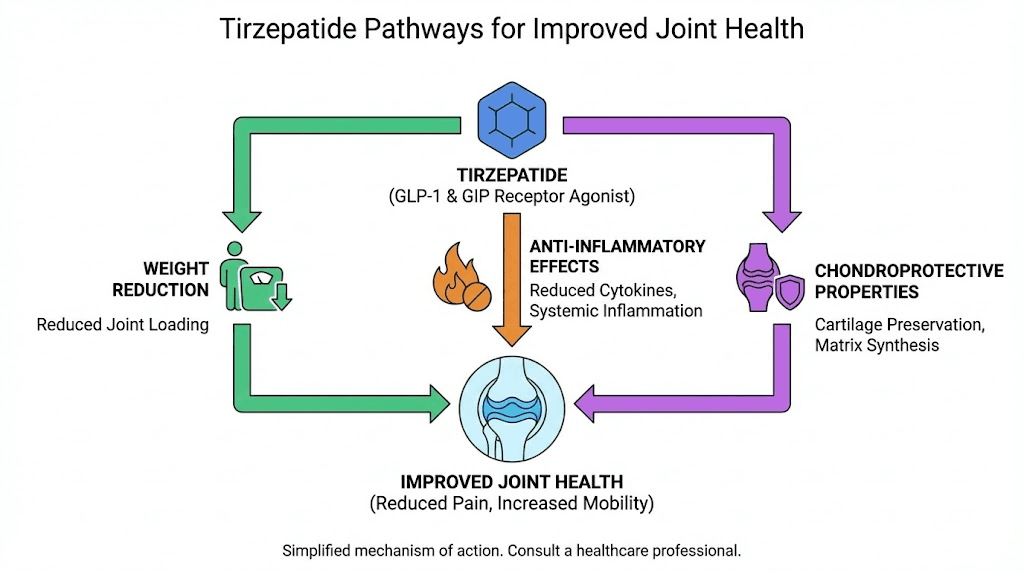
Here is where the story gets interesting. Despite individual reports of joint discomfort, the overall trajectory for joint health on tirzepatide points strongly positive. The mechanisms behind this improvement are both mechanical and molecular.
Weight reduction and mechanical stress relief
The math is simple and powerful. Every pound lost removes approximately four pounds of force from the knees during walking and up to six pounds during stair climbing. A person who loses 30 pounds on tirzepatide removes 120 to 180 pounds of cumulative force from their knee joints with every step.
Over the course of a day, an average person takes 6,000 to 10,000 steps. Multiply the force reduction per step by thousands of repetitions and the annual load reduction becomes staggering. This is why the ACR study found 16% lower knee pain risk in tirzepatide users. The weight reduction effect overwhelms any temporary discomfort from the transition period.
Hip joints benefit similarly. Low back pain improves as reduced abdominal mass decreases anterior pelvic tilt and the resulting lumbar compression. Even foot and ankle pain decreases as the structures supporting your entire body weight work under lighter loads.
For people with existing joint pain, this effect is transformative. Studies on GLP-1 receptor agonists and knee osteoarthritis show that the combination of weight loss and anti-inflammatory effects produces pain relief comparable to, and sometimes exceeding, conventional interventions. Research from Brigham and Women Hospital found tirzepatide and semaglutide more cost-effective than standard weight loss interventions for patients with knee osteoarthritis and obesity.
Anti-inflammatory properties of GLP-1 receptor agonists
Tirzepatide activates both GLP-1 and GIP receptors. Both receptor systems connect to anti-inflammatory pathways that extend far beyond glucose regulation and appetite suppression.
GLP-1 receptor activation inhibits the NF-kB signaling pathway, one of the master switches for inflammation throughout the body. NF-kB drives the production of pro-inflammatory cytokines like TNF-alpha, IL-1beta, and IL-6, all of which play documented roles in joint inflammation and cartilage degradation. By dampening this pathway, tirzepatide may provide direct anti-inflammatory benefits to joint tissues regardless of weight changes.
Animal studies support this mechanism. Liraglutide, another GLP-1 receptor agonist, activated PKA/CREB signaling pathways in rat models of knee osteoarthritis, reducing expression of inflammation-related proteins and demonstrating clear anti-inflammatory activity in joint tissues. While tirzepatide has not been studied as extensively for this specific application, it activates the same receptor system with even higher potency.
A growing body of rheumatology research suggests GLP-1 receptor agonists can help lower systemic inflammation with potential applications across multiple inflammatory joint conditions. This represents an emerging area of investigation that may eventually expand the therapeutic uses of medications like tirzepatide beyond metabolic diseases. Autoimmune and inflammatory conditions affecting joints may eventually become targets for GLP-1 based therapies.
Chondroprotective effects in research
Perhaps the most surprising finding comes from laboratory studies examining GLP-1 receptor agonist effects on cartilage cells directly. In vitro experiments have demonstrated chondroprotective effects, meaning these drugs may actually protect cartilage from degradation.
The proposed mechanisms include reduced matrix metalloproteinase activity (enzymes that break down cartilage), improved chondrocyte survival (the cells that maintain cartilage), and enhanced production of cartilage matrix components. While these findings have not fully translated to human clinical outcomes yet, they suggest the GLP-1 receptor system plays a role in joint tissue maintenance that researchers are only beginning to understand.
Population-level data supports this optimism. Patients treated with GLP-1 receptor agonists experienced a lower incidence of knee surgery compared to matched patients not receiving these medications. Whether this reflects direct cartilage protection, weight-related mechanical benefits, or anti-inflammatory effects (or some combination) remains under investigation.
The bottom line: tirzepatide appears to be a net positive for joint health over the medium and long term, even if a subset of users experiences temporary discomfort during the early weight loss phase.
Timeline of joint pain on tirzepatide: what to expect week by week
If you do experience joint discomfort on tirzepatide, understanding the typical timeline reduces anxiety and helps you distinguish normal adaptation from something requiring medical attention.
Weeks 1 through 4: the adjustment period
This is when most musculoskeletal complaints appear. The body is adjusting to reduced caloric intake, shifting fluid balance, and the initial metabolic effects of GLP-1 and GIP receptor activation. Body aches during this phase are common and generally mild.
What you might notice:
Generalized stiffness, particularly in the morning
Dull aches in weight-bearing joints (knees, hips, ankles)
Muscle soreness that feels disproportionate to activity level
Occasional sharp twinges during position changes
These symptoms typically coincide with dose titration. As your provider increases the tirzepatide dosage from the starting 2.5mg toward therapeutic levels, the body recalibrates repeatedly. Each dose increase can trigger a brief recurrence of adjustment symptoms.
Severity varies widely. Some users report mild stiffness they barely notice. Others describe aches significant enough to affect daily activities temporarily. Neither extreme predicts long-term outcomes.
Weeks 4 through 8: the adaptation phase
Most people who experienced initial joint discomfort see meaningful improvement during this window. The body stabilizes at the new caloric intake. Fluid balance normalizes. The musculoskeletal system begins adapting to changed biomechanics as weight loss progresses.
By week six, many users report that early aches have resolved completely. Those with ongoing discomfort typically describe it as less intense and less frequent than the initial presentation. The pattern suggests genuine physiological adaptation rather than masking or tolerance.
This is also the window where the benefits of weight reduction begin counteracting any initial biomechanical stress. Enough weight has been lost to meaningfully reduce joint loading, but not so much that dramatic gait changes continue destabilizing movement patterns.
Months 3 through 6: long-term outcomes
The population data becomes relevant here. By three to six months, the weight loss benefits for joint health dominate the picture. Tirzepatide before and after outcomes consistently show improved mobility, reduced pain medication use, and enhanced physical function.
Users who maintained adequate protein intake and some form of resistance exercise during the first months report the best joint outcomes. They preserved more muscle mass, provided better structural support to joints during the transition, and entered the maintenance phase with both reduced weight and maintained strength.
Persistent joint pain beyond three months warrants medical evaluation. While most tirzepatide-related musculoskeletal complaints resolve, ongoing pain may indicate an underlying condition that requires targeted treatment independent of weight loss medication.
How to prevent joint pain while taking tirzepatide
Prevention is dramatically more effective than treatment. These strategies address the root causes of joint discomfort during tirzepatide therapy, not just the symptoms.
Protein intake and muscle preservation
This is the single most impactful intervention. Without adequate protein, muscle loss accelerates, joint support deteriorates, and the risk of musculoskeletal pain increases substantially.
Target: 1.2 to 1.6 grams of protein per kilogram of body weight daily. For a 200-pound (91kg) person, that translates to 109 to 146 grams of protein per day.
Hitting this target on a reduced appetite requires deliberate planning. Protein-dense foods must take priority at every meal. Lean meats, fish, eggs, Greek yogurt, and whey protein supplements provide the highest protein-to-calorie ratios. When your appetite limits you to 1,200 or 1,500 calories daily, every calorie needs to work harder.
A structured diet plan helps ensure protein targets are met consistently. Planning meals in advance prevents the common pattern of eating whatever is convenient (usually carbohydrate-heavy) when appetite briefly returns. Consider our tirzepatide meal plan for specific daily meal templates designed around adequate protein intake.
Protein timing matters too. Distributing intake across three to four meals rather than concentrating it in one sitting maximizes muscle protein synthesis. Research shows the body can only utilize approximately 30 to 40 grams of protein per meal for muscle building, so spreading intake optimizes absorption and utilization.
Resistance training protocols
Exercise during tirzepatide treatment is not optional if you want to protect your joints. Specifically, resistance training preserves and builds the muscle tissue that stabilizes every joint in your body.
You do not need to become a powerlifter. Two to three sessions per week focusing on compound movements provides enormous joint protection. Squats strengthen the muscles around the knee and hip. Deadlifts build posterior chain strength that supports the spine. Rows and presses maintain upper body muscle that protects shoulder joints.
Start conservatively if you are new to resistance training. Bodyweight exercises, resistance bands, or light dumbbells provide sufficient stimulus for someone whose muscles are adapting to a new body composition. Progressive overload, gradually increasing resistance over weeks and months, builds strength without joint stress.
The combination of adequate protein and regular resistance training can shift the lean mass loss ratio significantly. Instead of losing 25% lean mass, properly supported individuals may lose only 10 to 15%, preserving the muscle critical for joint health throughout the weight loss process.
Hydration strategies
Deliberate hydration becomes essential when appetite suppression reduces natural drinking cues. Set specific targets rather than relying on thirst.
Minimum daily target: Half your body weight in ounces. A 200-pound person needs at least 100 ounces (roughly 3 liters) daily. During active exercise or in warm climates, increase by 20 to 30%.
Practical strategies that work:
Keep a measured water bottle and track refills
Set hourly reminders during the first month until the habit forms
Front-load water intake in the morning when nausea from tirzepatide may be lower
Include electrolyte-enhanced water or low-sugar electrolyte supplements
Count herbal tea, broth, and water-rich foods toward your total
Adequate hydration maintains synovial fluid quality, supports cartilage hydration, facilitates nutrient delivery to joint tissues, and helps flush inflammatory metabolites from the musculoskeletal system. It is the simplest and cheapest joint protection strategy available.
Key supplements for joint support
When caloric intake drops significantly, targeted supplementation fills nutritional gaps that diet alone cannot cover. These supplements have the strongest evidence base for joint health during weight loss.
Magnesium (400 to 600mg daily): Supports muscle relaxation, reduces cramping and stiffness, and plays a role in over 300 enzymatic reactions including those involved in cartilage maintenance. Magnesium glycinate offers the best absorption with the least gastrointestinal side effects, an important consideration when tirzepatide already affects digestive function.
Vitamin D3 (2,000 to 5,000 IU daily): Regulates calcium absorption, supports bone density, and directly influences cartilage health. Many people with obesity have insufficient vitamin D levels due to sequestration in fat tissue. As fat cells shrink during weight loss, stored vitamin D may be released, but supplementation ensures consistent levels throughout the process.
Omega-3 fatty acids (2 to 3 grams EPA/DHA daily): Modulate inflammatory pathways in joint synovial tissue. Multiple meta-analyses demonstrate reduced joint pain and stiffness with consistent omega-3 supplementation, particularly in people with osteoarthritis. These fatty acids complement the anti-inflammatory properties of tirzepatide itself.
Collagen peptides (10 to 15 grams daily): Provide the specific amino acids (glycine, proline, hydroxyproline) that cartilage requires for maintenance and repair. Studies show collagen peptide supplementation reduces joint pain in physically active individuals and may support cartilage integrity during periods of stress. For comprehensive information on collagen supplementation, see our guide to collagen hydrolysate versus collagen peptides.
Calcium (1,000 to 1,200mg daily): Essential for bone density maintenance during weight loss. Reduced caloric intake almost universally reduces calcium consumption, and the body will leach calcium from bone to maintain blood levels if dietary intake falls short.
The glycine compound formulations of tirzepatide may provide additional joint support through glycine, an amino acid critical for collagen synthesis. Some compounding pharmacies now offer tirzepatide with glycine and B12 specifically to address muscle and connective tissue support during treatment. For those interested in the specific role of glycine in tirzepatide formulations, our dedicated guide covers the evidence and protocols in detail.
Dose titration and timing considerations
How you escalate your tirzepatide dose affects the degree of musculoskeletal adjustment your body must handle. Faster escalation means more rapid weight loss, greater biomechanical shifts per unit of time, and potentially more joint stress during the transition.
Standard titration starts at 2.5mg weekly for four weeks before increasing to 5mg. Some providers move to higher doses more aggressively based on tolerance and response. If joint pain is a concern, discuss a more conservative titration schedule with your provider. Slower dose increases give the musculoskeletal system more time to adapt to each phase of weight loss.
Microdosing approaches represent another strategy. Rather than standard weekly dosing, some protocols use smaller, more frequent doses to smooth out the peaks and troughs that can trigger side effects. While not standard practice, this approach has gained attention for reducing GI side effects and may theoretically reduce the biomechanical stress of rapid weight fluctuations.
Use our compounded tirzepatide dosage calculator to determine precise dosing based on your specific formulation and target dose.
Managing joint pain if it occurs
Despite best prevention efforts, some people will experience joint discomfort during tirzepatide treatment. Having a management plan prevents overreacting to normal adaptation symptoms while ensuring genuine problems receive appropriate attention.
Immediate relief strategies
For mild to moderate joint discomfort during the adaptation phase:
Movement, not rest. This seems counterintuitive when joints hurt. But gentle movement maintains synovial fluid circulation, prevents stiffness from prolonged immobility, and gradually trains the musculoskeletal system to function at the new body weight. Walking, swimming, cycling, and yoga all provide joint-friendly movement that supports adaptation without excessive stress.
Temperature therapy. Ice reduces acute inflammation and provides pain relief for 15 to 20 minutes per application. Heat relaxes surrounding muscles and increases blood flow to joint tissues. Alternating between the two, starting with ice for acute pain and transitioning to heat for chronic stiffness, addresses both inflammatory and muscular components.
Anti-inflammatory support. Short-term NSAID use (ibuprofen, naproxen) can bridge severe discomfort while the body adapts. However, long-term NSAID use carries its own risks, particularly for people with reduced food intake and potentially compromised stomach lining from GI effects. Natural anti-inflammatories like turmeric (with piperine for absorption), ginger, and the omega-3 supplements discussed above provide gentler ongoing support.
Topical treatments. Topical diclofenac, menthol rubs, and arnica gels provide localized relief without systemic effects. These work well for specific joint pain (one knee, one hip) rather than generalized aches.
When to talk to your provider
Not all joint pain on tirzepatide is benign adaptation. Seek medical evaluation if you experience:
Joint swelling, redness, or warmth suggesting active inflammation or infection
Pain that worsens progressively rather than fluctuating or improving over weeks
Pain localized to a single joint with sudden onset, which could indicate gout, septic arthritis, or other specific joint pathology
Pain accompanied by fever suggesting systemic inflammatory or infectious process
Numbness, tingling, or weakness accompanying the joint pain, which may indicate nerve involvement
Pain lasting beyond eight weeks despite implementing prevention strategies
Pain severe enough to limit daily function including sleep, work, or basic mobility
Your provider can order imaging (X-ray, MRI) to evaluate joint structures, blood work (inflammatory markers, uric acid, autoimmune panels) to identify systemic causes, and may adjust your tirzepatide protocol if the musculoskeletal effects outweigh the metabolic benefits in your specific situation.
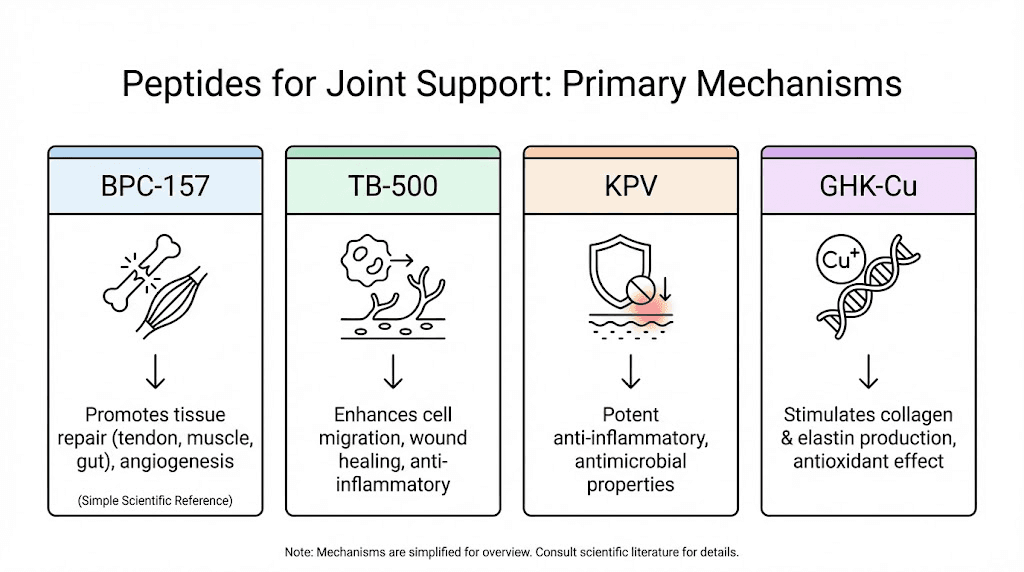
Supporting peptides for joint health
For researchers interested in peptide-based joint support, several compounds demonstrate relevant properties in the literature.
BPC-157 is the most extensively studied peptide for musculoskeletal repair. This 15-amino-acid sequence derived from human gastric juice demonstrates wound healing acceleration, tendon repair support, and anti-inflammatory effects in animal models. Research protocols typically use 200 to 300mcg administered subcutaneously, with studies showing improvements in tendon-to-bone healing and joint tissue recovery. For detailed dosing information, see our BPC-157 dosing guide.
TB-500 supports tissue repair through upregulation of actin, a protein critical for cell migration and wound healing. Studies show benefits for soft tissue recovery, reduced inflammation, and improved flexibility. The combination of BPC-157 and TB-500 represents one of the most popular stacking protocols for musculoskeletal health in the research community.
KPV peptide demonstrates potent anti-inflammatory properties through inhibition of pro-inflammatory signaling. While primarily studied for gut inflammation, its systemic anti-inflammatory effects may benefit joint tissue as well. The complete KPV benefits profile includes modulation of multiple inflammatory pathways relevant to joint health.
GHK-Cu supports collagen synthesis, tissue remodeling, and anti-inflammatory processes. As a copper peptide, it plays roles in wound healing and tissue regeneration that extend to connective tissue surrounding joints. Consult the GHK-Cu dosage guide for research protocols.
For a comprehensive overview of peptide options for musculoskeletal support, our guide to the best peptides for joint pain covers mechanisms, protocols, and evidence for each compound. Additionally, tissue repair peptides provide broader context on peptide-based recovery strategies.
Tirzepatide joint pain compared to other side effects
Putting joint pain in context with the broader side effect profile helps set realistic expectations. Most tirzepatide users experience some side effects, but the type, severity, and duration vary considerably.
Side effect | Frequency | Typical onset | Duration | Severity |
|---|---|---|---|---|
Nausea | 20-30% | Week 1 | 2-8 weeks | Mild to moderate |
Diarrhea | 12-18% | Week 1-2 | 1-4 weeks | Mild to moderate |
Decreased appetite | 15-25% | Week 1 | Ongoing (intended) | Mild |
6-12% | Week 2-4 | Variable | Mild to moderate | |
5-15% | Week 1-4 | 4-8 weeks | Mild to moderate | |
5-10% | Week 1-2 | 1-3 weeks | Mild | |
5-12% | Week 1-4 | 2-6 weeks | Mild to moderate | |
3-8% | Week 1-4 | Variable | Mild | |
3-5% | Variable | Variable | Mild | |
3-8% | Month 2-4 | Temporary | Mild |
Several patterns emerge from this comparison. Joint pain ranks in the middle tier of tirzepatide side effects, less common than GI symptoms but more common than rare reactions. Its onset overlaps with the initial adjustment period shared by most side effects. And like the majority of tirzepatide side effects, it tends to be self-limiting.
The key difference between joint pain and GI side effects is the underlying mechanism. GI symptoms result directly from GLP-1 and GIP receptor activation in the digestive system. Joint pain results indirectly from the metabolic and physical changes the medication produces. This distinction matters because GI symptoms may require dose adjustment or anti-nausea medication, while joint pain responds better to the nutritional and exercise strategies outlined above.
For people considering whether to start tirzepatide, this comparison offers perspective. Joint pain is a manageable and typically temporary possibility, not a reason to avoid a medication that provides substantial metabolic and cardiovascular benefits. The overall safety profile of peptide-based therapies supports this assessment.
Specific joint areas affected and what each means
Where you experience discomfort provides diagnostic clues about the underlying cause and the most effective response.
Knee pain on tirzepatide
Knees are the most commonly reported joint pain location during weight loss therapy, and for good reason. As the largest hinge joint in the body, knees bear the highest cumulative force during daily activities. They are also the most sensitive to changes in body weight and lower extremity biomechanics.
Knee pain during tirzepatide treatment typically falls into two categories. The first is mechanical adaptation pain, a dull ache around or behind the kneecap that worsens with stairs or prolonged sitting. This reflects the patellar tracking changes that occur as quadriceps strength decreases relative to body weight changes. Strengthening the vastus medialis oblique (the inner quad muscle) often resolves this within weeks.
The second is pre-existing osteoarthritis becoming more noticeable. As mentioned earlier, weight loss can paradoxically reveal joint damage that obesity-related inflammation was partially masking. If knee pain persists or includes grinding sensations, swelling, or locking, imaging evaluation is warranted.
The good news is unambiguous. Long-term weight loss produces the best outcomes for knee health. The 16% reduction in knee pain risk seen in the ACR tirzepatide study reflects the powerful mechanical benefit of reduced body weight on these vulnerable joints. For those with knee concerns specifically, peptides for bone and cartilage repair may provide additional support.
Hip pain and lower back pain
Hip and lower back pain during tirzepatide treatment share a common biomechanical origin. Abdominal fat creates anterior pelvic tilt, pulling the lower spine into excessive lordosis and shifting hip joint loading posteriorly. As abdominal fat reduces, pelvic alignment changes, lumbar curvature decreases, and hip joint mechanics shift.
This transition is ultimately beneficial. Reduced anterior pelvic tilt decreases lumbar disc compression and normalizes hip joint loading. But the adaptation period can produce unfamiliar sensations, particularly in people who have carried abdominal weight for years.
Core strengthening addresses both locations simultaneously. The transverse abdominis, internal obliques, and multifidus muscles form a natural corset that stabilizes the spine and pelvis. When these muscles strengthen as body weight decreases, both hip and low back pain typically resolve.
Our guide to the best peptides for back pain explores additional approaches for those dealing with persistent lumbar discomfort. The shoulder pain guide covers upper body joint concerns that may also arise during the weight loss transition.
Wrist, finger, and small joint discomfort
Small joint pain during tirzepatide treatment is less about biomechanical changes and more about systemic factors. These joints bear minimal body weight, so the mechanical weight loss explanation does not apply.
Electrolyte imbalances hit small joints disproportionately. Fluid shifts during rapid weight loss can cause transient swelling in fingers and wrists. Dehydration reduces synovial fluid volume, and small joints with less fluid reserve notice the deficit sooner than larger joints.
Inflammatory conditions like gout can be triggered by rapid weight loss. When fat cells break down quickly, they release purines that convert to uric acid. Elevated uric acid crystallizes in joints, most commonly the big toe, but potentially any small joint. This is a distinct condition from general weight loss adaptation and requires specific treatment.
If small joint pain is accompanied by redness, warmth, or extreme tenderness, uric acid testing can rule out gout. Simple hydration optimization and electrolyte supplementation resolves most non-gout small joint complaints within two to three weeks.
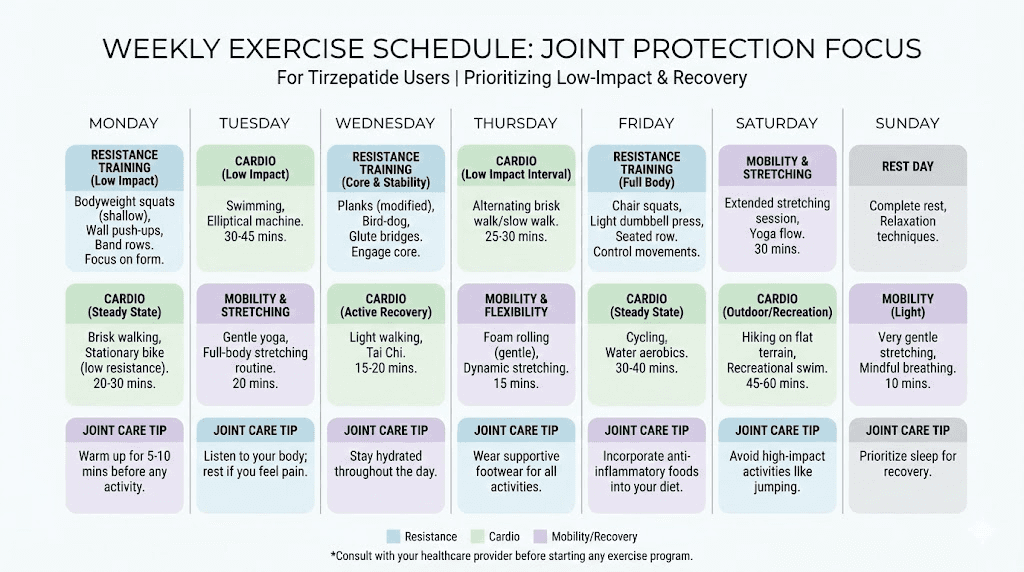
The role of exercise during tirzepatide treatment
Exercise deserves its own section because it simultaneously prevents joint pain, treats existing joint pain, preserves muscle mass, enhances weight loss quality, and improves nearly every outcome associated with tirzepatide use.
Yet many tirzepatide users reduce physical activity. Fatigue during the adjustment period, reduced energy from caloric restriction, and the misconception that the medication does the work alone all contribute to decreased exercise. This is a mistake with compounding consequences for joint health.
Resistance training preserves the muscle mass that stabilizes joints. Two to three sessions per week targeting all major muscle groups provides the minimum effective dose. Compound movements (squats, deadlifts, rows, presses) deliver the most joint-protective benefit per session because they strengthen entire kinetic chains rather than isolated muscles.
Cardiovascular exercise improves blood flow to joint tissues, supports cartilage nutrition (cartilage receives nutrients through compression and release during movement), and maintains cardiovascular fitness during caloric restriction. Walking, swimming, and cycling offer joint-friendly options that most people can perform even with existing joint discomfort.
Flexibility and mobility work addresses the range of motion changes that accompany body composition shifts. As fat pads reduce and muscle tension patterns change, joints may feel stiff in new positions. Regular stretching and mobility exercises maintain functional range of motion throughout the weight loss process.
The combination matters most. Research shows that pairing GLP-1 medications with structured exercise programs produces markedly better outcomes than medication alone, not just for weight loss but for body composition, joint health, metabolic markers, and quality of life. SeekPeptides members access personalized exercise protocols designed specifically for people using peptide-based weight loss therapies, integrating resistance training with appropriate recovery strategies.
Comparing joint pain risk: tirzepatide versus semaglutide
Many people choosing between tirzepatide and semaglutide wonder whether one medication carries higher joint pain risk. The available evidence does not show a meaningful difference in musculoskeletal side effects between the two.
Both medications produce similar rates of joint-related complaints in clinical trials and postmarketing surveillance. Both drive significant weight loss that creates the same biomechanical adaptation challenges. And both activate GLP-1 receptors, providing similar anti-inflammatory benefits to joint tissues.
The primary difference is that tirzepatide also activates GIP receptors, and early research suggests GIP signaling may have additional anti-inflammatory properties beyond those of GLP-1 alone. Whether this translates to meaningfully better joint outcomes remains unclear from current data.
Tirzepatide typically produces greater weight loss than semaglutide at comparable dose levels. Greater weight loss means larger biomechanical shifts in a shorter timeframe, which could theoretically increase transient joint discomfort during the adaptation period. However, it also means greater long-term mechanical benefit from reduced joint loading.
For a detailed comparison of side effect profiles between these medications, our semaglutide versus tirzepatide comparison covers differences in efficacy, tolerability, and clinical outcomes. The dosage comparison chart helps visualize dose equivalencies between the two medications.
If joint pain is a primary concern, neither medication offers a clear advantage over the other. The prevention strategies outlined in this guide apply equally to both. Focus on the medication that provides the best overall metabolic benefit for your specific situation, and implement joint protection measures regardless of which you choose.
Long-term joint health outlook on tirzepatide
The one-year and beyond perspective is overwhelmingly positive for joint health. This matters because joint concerns during the first few months can create anxiety that leads to premature discontinuation, sacrificing the long-term benefits that make the temporary discomfort worthwhile.
Weight maintenance after reaching target provides the most sustained joint benefit. Reduced mechanical loading becomes the new normal. Joints operate under lighter conditions permanently. Cartilage that was under excessive stress gets a reprieve that may slow or halt degenerative processes.
The anti-inflammatory effects of maintained weight loss compound over time. Adipose tissue (body fat) produces inflammatory cytokines. Less fat means less chronic systemic inflammation. Joints benefit from this reduced inflammatory load continuously, not just during the active weight loss phase.
For those who experienced significant weight loss, the improvement in physical function often enables activities that further strengthen the musculoskeletal system. A virtuous cycle develops: weight loss improves joint function, improved function enables more exercise, more exercise strengthens joint support structures, stronger joints enable even more activity.
Emerging research on GLP-1 receptor agonists and orthopedic outcomes suggests patients on these medications may require fewer joint procedures over time. Lower rates of knee replacement, reduced need for pain management interventions, and improved post-surgical outcomes in patients who achieved significant weight loss before orthopedic procedures all point toward long-term joint health benefits that justify working through the temporary adaptation period.
For researchers serious about long-term musculoskeletal health alongside their peptide protocols, SeekPeptides provides comprehensive guides covering joint support strategies, exercise protocols, and supplement stacking approaches specifically designed for GLP-1 receptor agonist users. The community includes thousands of members navigating these exact questions with evidence-based guidance.
Frequently asked questions
Does tirzepatide directly cause joint inflammation?
No. Clinical trial data and the FDA prescribing information do not identify joint inflammation as a pharmacologic effect of tirzepatide. The GIP and GLP-1 receptor systems targeted by tirzepatide are not known to trigger inflammatory responses in joint tissue. When joint discomfort occurs, it typically results from the secondary effects of rapid weight loss, not from direct drug action on joints.
How common is joint pain on tirzepatide?
Postmarketing data suggests 5 to 15% of users report some form of musculoskeletal discomfort, but clinical trials found rates comparable to placebo. Most reports describe mild, generalized aches rather than specific joint pathology. The rates are significantly lower than GI side effects like nausea (20-30%) and diarrhea (12-18%).
When does tirzepatide joint pain typically start?
Most musculoskeletal complaints appear during weeks one through four, coinciding with dose initiation and early titration. Pain often peaks around dose increases and resolves within four to eight weeks as the body adapts to the new weight and metabolic state.
Should I stop tirzepatide if I get joint pain?
Generally, no. Mild to moderate joint discomfort during the adaptation phase is manageable with the strategies outlined in this guide and typically resolves spontaneously. Stopping the medication sacrifices substantial metabolic and long-term joint health benefits. Consult your prescriber if pain is severe, progressive, accompanied by swelling or redness, or persists beyond eight weeks.
Can tirzepatide improve existing arthritis?
Evidence suggests it can. Weight loss reduces mechanical stress on joints, and GLP-1 receptor activation provides anti-inflammatory effects. Population studies show tirzepatide users have lower rates of joint pain and lower need for pain medications compared to other weight loss approaches. Research on GLP-1 agonists and knee osteoarthritis specifically shows promising results.
What supplements help prevent joint pain on tirzepatide?
The strongest evidence supports magnesium (400-600mg daily), vitamin D3 (2,000-5,000 IU daily), omega-3 fatty acids (2-3g EPA/DHA daily), collagen peptides (10-15g daily), and calcium (1,000-1,200mg daily). These address the nutritional gaps created by reduced food intake during treatment. Check our full supplements guide for detailed recommendations.
Does the B12 formulation of tirzepatide help with joint pain?
Tirzepatide with B12 primarily addresses energy levels and neurological function during weight loss. While B12 does not directly target joint pain, adequate B12 status supports nerve health (reducing referred pain from nerve compression) and energy levels (enabling the exercise critical for joint protection). The compounded formulations offer convenience for users supplementing multiple nutrients.
Is joint pain worse on higher doses of tirzepatide?
Higher doses produce faster weight loss, which can increase the biomechanical adaptation stress on joints. However, they also provide stronger anti-inflammatory effects and greater long-term weight reduction benefit. The relationship is not linear, as individual factors like baseline fitness, diet quality, and exercise habits matter more than dose level for predicting joint outcomes.
Can exercise make tirzepatide joint pain worse?
High-impact exercise during the adaptation phase may temporarily aggravate joint discomfort. However, appropriate exercise, particularly resistance training and low-impact cardio, prevents and treats joint pain more effectively than rest. The key is matching exercise intensity to current tolerance while progressively building capacity. Avoid jumping, running on hard surfaces, and heavy plyometrics during weeks one through four if joints are sensitive.
Do other GLP-1 medications cause less joint pain than tirzepatide?
Available evidence does not show meaningful differences in joint pain rates between tirzepatide, semaglutide, or other GLP-1 receptor agonists. All produce weight loss that creates similar biomechanical adaptation challenges. The joint protection strategies in this guide apply regardless of which medication you use. Our comparison of side effects across these medications provides additional context.
External resources
American College of Rheumatology: Impact of Tirzepatide on Musculoskeletal Pain
PubMed Central: Targeting GLP-1/GLP-1R Axis to Treat Osteoarthritis
Effects of GLP-1 Agonists on Musculoskeletal Health and Orthopedic Care
Journal of Inflammation: GLP-1 Agonist Ameliorates Inflammation in Knee Osteoarthritis
For researchers serious about optimizing their peptide protocols alongside joint health, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, personalized dosing protocols, calculators for precise measurements, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your joints stay pain-free, your weight loss stay consistent, and your protocols stay optimized.