Mar 20, 2026
What if the medication you have been comparing to tirzepatide is not a different drug at all? What if Zepbound and tirzepatide are, at the molecular level, the exact same thing?
This confusion sends thousands of people searching every month. They read about Zepbound on one website and tirzepatide on another. They see different names, different packaging, different prices. Naturally, they assume these must be different medications with different mechanisms and different results. The assumption is wrong.
Zepbound is tirzepatide. Same molecule. Same mechanism. Same 39-amino-acid peptide chain that activates both GIP and GLP-1 receptors in your body. The difference is not in the chemistry. It is in the labeling, the FDA approval, the insurance coding, and the way you access it. And those practical differences matter enormously for your wallet, your coverage, and your treatment options. This guide breaks down every single distinction between brand-name Zepbound, its sister product Mounjaro, and compounded tirzepatide, so you can make a genuinely informed decision about which path makes sense for your situation. SeekPeptides members navigate these exact decisions with access to comprehensive protocol guidance and dosing tools that account for formulation differences most resources ignore.
The short answer: Zepbound is tirzepatide
There is no chemical difference between Zepbound and tirzepatide. None.
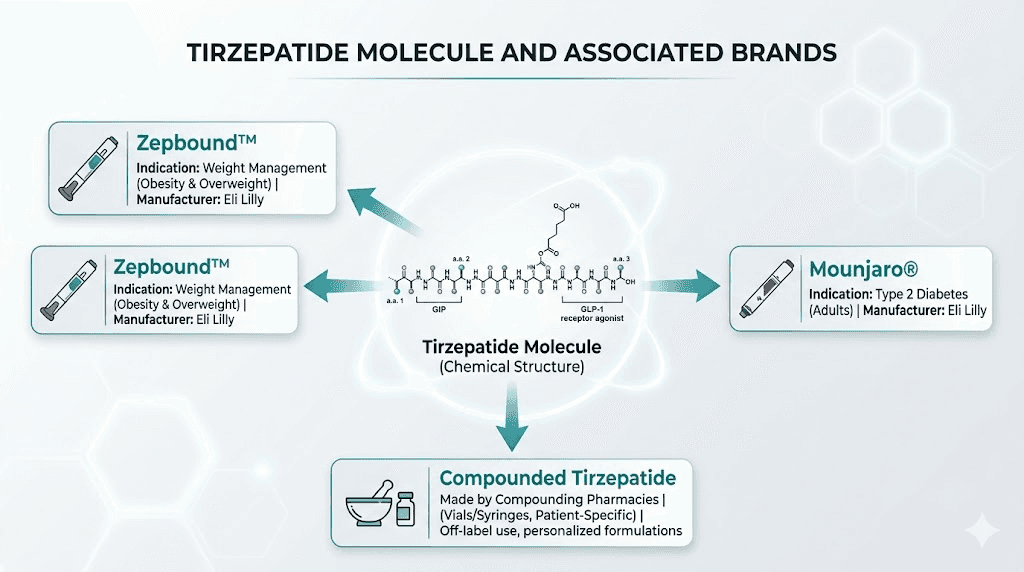
Tirzepatide is the active pharmaceutical ingredient. It is the generic name for the molecule itself. Zepbound is the brand name that Eli Lilly chose when marketing this molecule specifically for weight management. Think of it like acetaminophen and Tylenol. Same compound, different label. The distinction exists for regulatory and marketing purposes, not because there is any pharmacological difference between what you receive.
Eli Lilly manufactures both Zepbound and Mounjaro. Both contain tirzepatide. Both come in identical dose strengths ranging from 2.5 mg to 15 mg. Both are administered as once-weekly subcutaneous injections. The molecule sitting inside each pen or vial is structurally, functionally, and therapeutically identical. When you inject 5 mg of Zepbound, you are injecting the exact same thing as 5 mg of Mounjaro. Your body cannot tell the difference because there is no difference to detect.
So why do two brand names exist for the same drug?
The answer involves FDA regulations, insurance billing codes, and the way pharmaceutical companies navigate approval pathways. And understanding these distinctions could save you hundreds or even thousands of dollars per month on the same medication.
Understanding the molecule: what tirzepatide actually does
Before diving into the branding differences, it helps to understand what this molecule actually does inside your body. Tirzepatide is a synthetic peptide consisting of 39 amino acids. It was designed based on the native GIP (glucose-dependent insulinotropic polypeptide) sequence, modified with a 20-carbon fatty diacid chain that binds to albumin in the blood. This albumin binding is what gives tirzepatide its long half-life of approximately five days, enabling once-weekly dosing.
What makes tirzepatide unique among peptide therapies is its dual receptor activation. It binds to and activates both the GIP receptor and the GLP-1 receptor. Most other medications in this class, including semaglutide, only target the GLP-1 receptor. This dual action creates a synergistic effect that research has shown produces greater results than targeting either receptor alone.
The GLP-1 pathway
GLP-1 receptor activation does several things simultaneously. It stimulates insulin secretion from the pancreas in a glucose-dependent manner, meaning it only increases insulin when blood sugar is elevated. It suppresses glucagon release, reducing the liver output of glucose. It slows gastric emptying, which means food moves through your stomach more slowly. And it acts on appetite centers in the brain to reduce hunger signals.
This is why appetite suppression is one of the first effects people notice. The delayed gastric emptying also explains common side effects like bloating, constipation, and nausea during the early weeks.
The GIP pathway
GIP receptor activation adds another layer of metabolic benefit. It enhances insulin sensitivity in peripheral tissues, improves fat metabolism, and contributes to energy balance regulation. Research suggests that the combination of GIP and GLP-1 activation produces a significantly greater insulin response and glucagonostatic effect compared to either hormone working alone.
This dual mechanism is why tirzepatide consistently outperforms single-receptor GLP-1 agonists in clinical trials. The comparison between semaglutide and tirzepatide demonstrates this clearly, with tirzepatide producing 47% greater relative weight loss in head-to-head studies.
Why the molecule matters more than the brand
Whether you receive tirzepatide through Zepbound, Mounjaro, or a compounded formulation, the mechanism of action is identical. Your GIP receptors do not care what label was on the box. Your GLP-1 receptors do not check whether you paid brand-name prices. The molecule binds the same way, triggers the same cascades, and produces the same physiological effects regardless of how it was packaged or who manufactured it.
Understanding this removes a lot of confusion. The real questions are not about chemistry. They are about access, cost, regulation, and quality assurance.
Zepbound vs Mounjaro: same drug, different FDA labels
Here is where the practical differences begin.
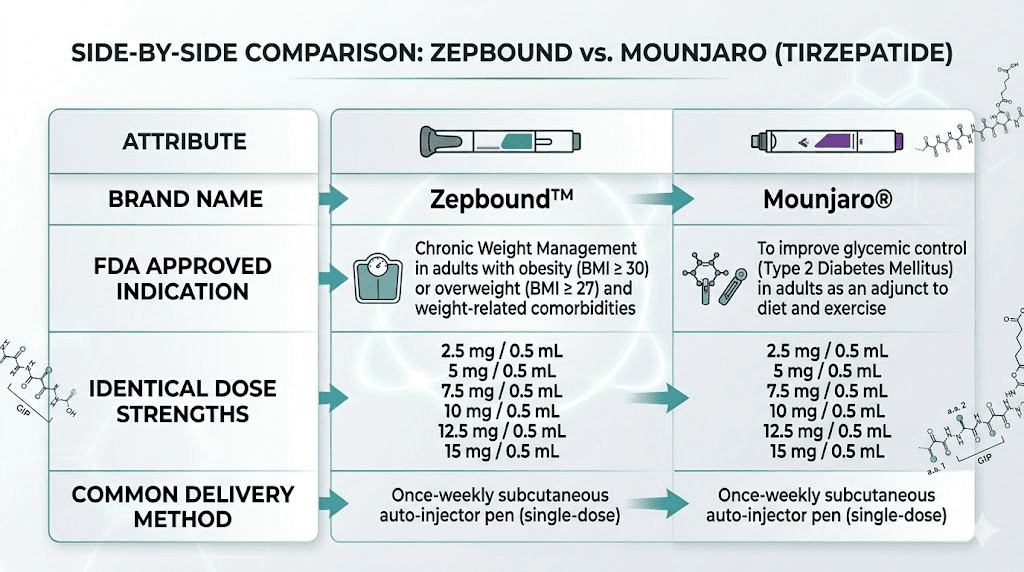
Mounjaro was approved by the FDA on May 13, 2022, for adults with type 2 diabetes. Its approval was based on the SURPASS clinical trial program, which demonstrated impressive glycemic control along with substantial weight loss as a secondary benefit. Doctors prescribe Mounjaro using ICD-10 codes related to diabetes management.
Zepbound received its FDA approval on November 8, 2023, roughly 18 months later. This approval was specifically for chronic weight management in adults with obesity (BMI of 30 or greater) or overweight (BMI of 27 or greater) with at least one weight-related comorbidity. The approval was based on the SURMOUNT trial program, which focused specifically on weight loss outcomes. Doctors prescribe Zepbound using ICD-10 codes related to obesity.
Same molecule. Different approved uses. Different insurance pathways.
Why this distinction matters for your prescription
Insurance companies care deeply about FDA-approved indications. If you have type 2 diabetes, your insurance may cover Mounjaro because it is approved for that condition. Zepbound, prescribed for weight loss, would likely be denied under the same plan because many insurers consider obesity treatment a "lifestyle" category and exclude it from coverage.
Conversely, if you do not have diabetes but meet the BMI criteria, Zepbound is the appropriate prescription for your situation. Your provider cannot prescribe Mounjaro off-label for weight loss and expect insurance to cover it, though some do try. The coding determines coverage, and the coding depends on which brand name is prescribed and for which indication. This is critical for understanding insurance coverage pathways and eligibility requirements.
Dose availability
Both Zepbound and Mounjaro are available in the same six dose strengths:
2.5 mg (starting dose for the first 4 weeks)
5 mg (first maintenance dose)
7.5 mg
10 mg
12.5 mg
15 mg (maximum dose)
All doses come as 0.5 mL injections in single-dose pens. Multi-dose vials are also available with FDA approval, containing enough medication for four weekly injections. The dosage in units is identical regardless of brand. If you need help converting between units and milligrams, tools like the 2.5 mg conversion guide or the 5 mg conversion guide can help with precise calculations.
The standard titration protocol starts at 2.5 mg weekly for four weeks, increases to 5 mg for another four weeks, and then adjusts upward in 2.5 mg increments every four weeks as needed. This is the same schedule for both brands. For a complete breakdown, see the full tirzepatide dose chart.
Compounded tirzepatide: the third option everyone asks about
Beyond the two brand-name options, there is a third path that has become enormously popular: compounded tirzepatide.
Compounding pharmacies create customized versions of medications using the active ingredient. In the case of tirzepatide, compounding pharmacies source the raw peptide and formulate it into injectable solutions, often at significantly lower cost than brand-name alternatives. Some pharmacies also offer oral drops, orally disintegrating tablets, and other delivery methods.
How compounded tirzepatide differs from Zepbound
The active molecule is the same. The differences lie in regulation, manufacturing standards, and additional ingredients.
Zepbound is manufactured by Eli Lilly under strict FDA oversight. Every batch undergoes rigorous quality control testing, potency verification, and sterility assurance. The FDA has reviewed and approved the specific manufacturing process, the excipients (inactive ingredients), the delivery device, and the stability data proving the product maintains potency through its expiration date.
Compounded tirzepatide does not go through this FDA approval process. Compounding pharmacies operate under different regulatory frameworks. 503A pharmacies compound medications on an individual patient basis with a prescription. 503B outsourcing facilities can produce larger batches but still operate outside the traditional FDA approval pathway. Both must follow current Good Manufacturing Practices, but the oversight is different from what a major pharmaceutical manufacturer faces.
Many compounding pharmacies add supplementary ingredients to their tirzepatide formulations. Common additions include vitamin B12, glycine, niacinamide, methylcobalamin, and levocarnitine. These additions may provide supplementary benefits, but they also mean the final product is not identical to brand-name Zepbound.
The FDA shortage designation factor
Compounding pharmacies were initially able to produce tirzepatide formulations because the FDA listed tirzepatide on its drug shortage list. Under federal law, compounding pharmacies can produce copies of FDA-approved drugs that are currently in shortage. This legal framework enabled the rapid growth of compounded tirzepatide availability.
The shortage designation has been a moving target, with periods where it was removed and then reinstated. This regulatory uncertainty affects the availability and legality of compounded tirzepatide at any given time. Staying informed about current availability status is essential for anyone relying on compounded formulations.
Quality considerations
Not all compounding pharmacies are equal. The quality of compounded tirzepatide can vary significantly between providers. Key factors include the source of the raw tirzepatide peptide, the accuracy of dosing, the sterility of the final product, and the stability of the formulation over time.
Choosing a reputable pharmacy matters enormously. Research reviews from established providers like Empower Pharmacy, Southend Pharmacy, ProRx, or Red Rock Pharmacy before committing to a provider. Third-party testing, Certificate of Analysis documentation, and transparency about sourcing are all indicators of a trustworthy pharmacy. For a broader overview of vendor options, the grey market tirzepatide guide covers what to watch for and what to avoid.
Cost comparison: what you will actually pay
Cost is the primary reason people research the difference between Zepbound and tirzepatide. The price gap is substantial.
Zepbound pricing
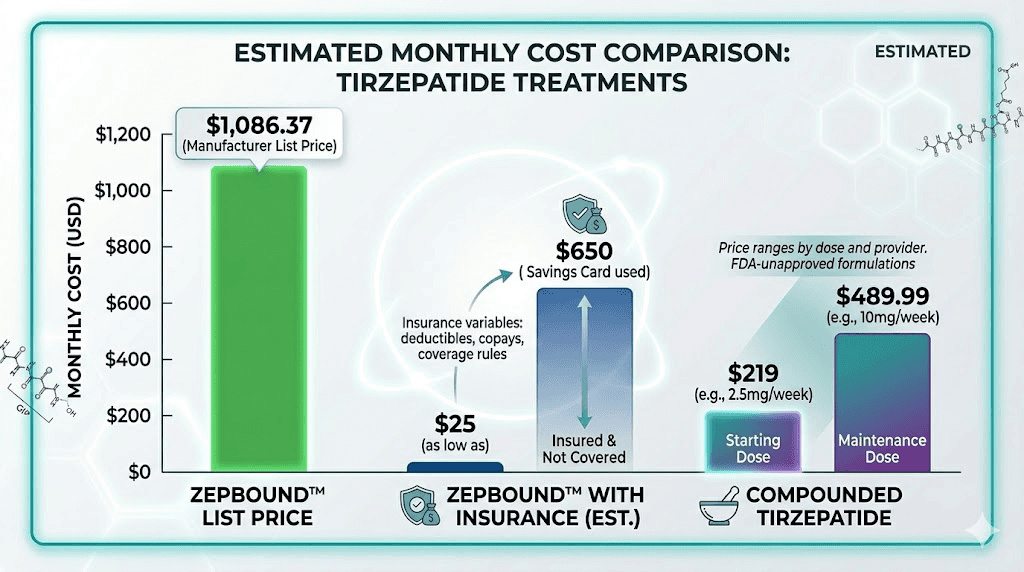
Zepbound has a list price of approximately $1,086 for a 28-day supply. That is the retail price without any insurance or discount programs. If your commercial insurance covers Zepbound, the manufacturer savings card can bring your copay as low as $25 for a one-month or three-month supply. Without insurance coverage, Eli Lilly offers a direct self-pay option around $499 per month.
Medicare does not cover weight loss medications. Most Medicaid programs exclude them as well. If you fall into one of these categories, brand-name Zepbound at full price is prohibitively expensive for most people.
Compounded tirzepatide pricing
Compounded tirzepatide typically costs between $150 and $400 per month, depending on the pharmacy, the dose, and whether additional ingredients are included. Some telehealth platforms offer bundled packages that include the medication, medical consultations, and shipping for a single monthly fee. For cost-conscious options, our guides on affordable tirzepatide and cheap compounded tirzepatide provide detailed comparisons across dozens of providers.
Insurance does not cover compounded tirzepatide. It is always a cash-pay medication. However, the base price is low enough that cash-pay compounded tirzepatide is often cheaper than the insurance copay for brand-name Zepbound, especially for people with high-deductible plans or limited pharmacy benefits.
Cost comparison table
Factor | Zepbound (brand) | Compounded tirzepatide |
|---|---|---|
List price | ~$1,086/month | $150-$400/month |
With commercial insurance | As low as $25/month | Not covered |
Self-pay (no insurance) | ~$499/month | $150-$400/month |
Medicare/Medicaid | Not covered | Not covered |
FDA approved | Yes | No |
Quality oversight | Full FDA manufacturing oversight | State pharmacy board regulation |
Additional ingredients | Standard excipients only | May include B12, glycine, etc. |
The right choice depends entirely on your insurance situation, your budget, and your comfort level with compounded medications. For many people without robust insurance coverage, compounded tirzepatide is the only financially viable path to accessing this medication. For those with good pharmacy benefits, brand-name Zepbound at $25 per month is hard to beat.
Individual pharmacy pricing varies widely. Platforms like Medvi, Zealthy, and Willow offer different pricing structures worth comparing before committing.
Insurance coverage and eligibility requirements
Insurance coverage for tirzepatide, under any name, is complicated. Understanding the landscape can save you months of denied claims and appeals.
Zepbound coverage
Zepbound is approved for chronic weight management in adults who meet specific BMI criteria. To be eligible, you need a BMI of 30 or greater, or a BMI of 27 or greater with at least one weight-related comorbidity such as hypertension, type 2 diabetes, dyslipidemia, or obstructive sleep apnea.
Even meeting these criteria does not guarantee coverage. Many commercial insurance plans explicitly exclude anti-obesity medications from their formularies. Others require prior authorization, step therapy (trying cheaper alternatives first), or documented failure of diet and exercise programs before approving Zepbound.
The coverage landscape varies dramatically by insurer and by plan. Blue Cross Blue Shield plans, for example, range from full coverage to complete exclusion depending on the specific plan and state. Anthem coverage varies similarly. Knowing what to say when requesting coverage from your insurance company can significantly improve your chances of approval.
Mounjaro coverage
If you have type 2 diabetes, Mounjaro coverage is generally easier to obtain. Diabetes medications are considered medically necessary by most insurance plans. Prior authorization may still be required, and your insurer may prefer that you try metformin or other first-line medications before approving a GLP-1 based therapy. But the overall approval rate for diabetes indications is substantially higher than for weight management.
Some people without diabetes have obtained Mounjaro through off-label prescriptions for weight loss. This approach is risky from an insurance perspective, as claims may be denied and you could be responsible for the full cost retroactively.
The BMI requirement debate
The BMI thresholds for Zepbound eligibility remain controversial. BMI is a flawed metric that does not account for muscle mass, body composition, or metabolic health. Someone with a BMI of 26 and significant metabolic dysfunction may benefit more from tirzepatide than someone with a BMI of 31 and good metabolic markers. Yet insurance draws hard lines at these numbers.
This rigidity pushes many people toward compounded tirzepatide, where there are no insurance gatekeepers and eligibility is determined by a prescribing provider assessment rather than an arbitrary BMI cutoff.
Clinical trial results: what the data actually shows
The clinical evidence for tirzepatide, whether branded as Zepbound or Mounjaro, is among the strongest in the weight management and diabetes treatment space. The SURMOUNT and SURPASS trial programs provide the foundation for understanding what to expect.
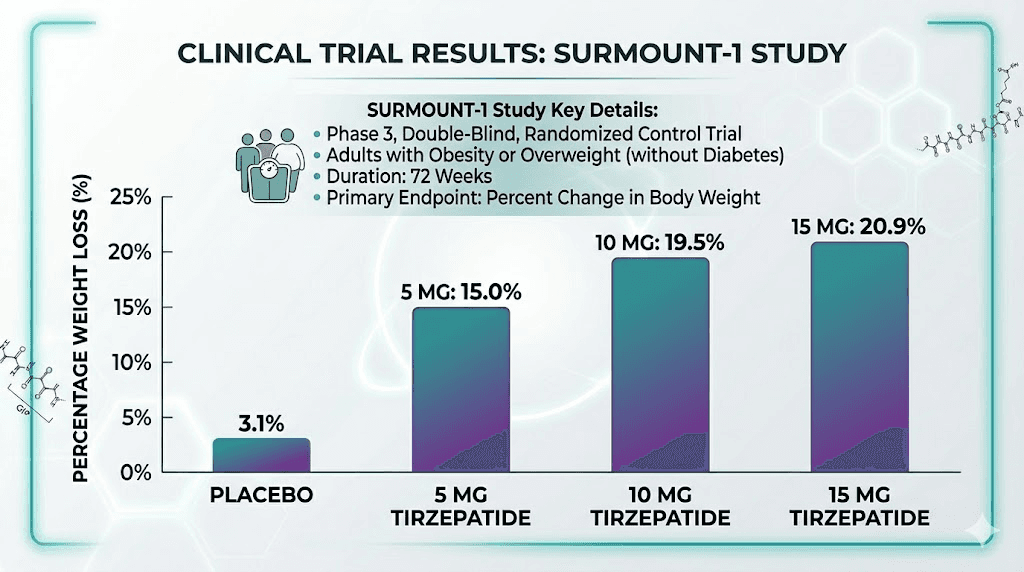
SURMOUNT-1: the weight loss evidence
The SURMOUNT-1 trial enrolled 2,539 adults with obesity or overweight with at least one comorbidity. None had diabetes. Participants received either tirzepatide (at 5 mg, 10 mg, or 15 mg) or placebo, administered weekly for 72 weeks.
Results were remarkable. The 15 mg group lost an average of 20.9% of body weight compared to 3.1% with placebo. But averages only tell part of the story. Among the 15 mg group, 56.7% of participants lost 20% or more of their body weight. Nearly 91% lost at least 5%. These numbers far exceeded anything previously demonstrated with pharmacotherapy for obesity.
For context on what this timeline looks like in practice, the tirzepatide weight loss timeline breaks down typical results week by week, and the first month expectations guide covers what you should realistically anticipate during the initial weeks.
SURMOUNT-5: head-to-head with semaglutide
The SURMOUNT-5 trial directly compared tirzepatide to semaglutide (Wegovy) in a head-to-head study. This was the trial everyone had been waiting for, pitting the two most popular weight loss medications against each other.
Tirzepatide won decisively. Participants on tirzepatide achieved an average weight loss of 20.2% at 72 weeks compared to 13.7% with semaglutide. In absolute terms, the tirzepatide group lost an average of 50.3 pounds versus 33.1 pounds in the semaglutide group. That is a 47% greater relative weight loss.
The side effect profiles were similar between both medications, with gastrointestinal symptoms being the most common in both groups. For people weighing their options between these two treatments, the full semaglutide vs tirzepatide comparison provides detailed analysis beyond just weight loss numbers.
SURPASS trials: the diabetes data
The SURPASS trials tested tirzepatide in people with type 2 diabetes. These studies demonstrated significant improvements in HbA1c levels, with many participants achieving blood sugar targets that had eluded them with previous treatments. The weight loss in the diabetes population was also substantial, though slightly less than in the non-diabetic SURMOUNT population.
These results apply equally to Zepbound and Mounjaro because, again, they contain the identical molecule. The clinical trial data generated for one brand applies directly to the other. Your body does not respond differently based on the label.
What about compounded tirzepatide data?
There are no large-scale clinical trials specifically testing compounded tirzepatide formulations. The assumption, supported by pharmacological principles, is that if the active ingredient is the same molecule at the same dose, the effects should be equivalent. However, this assumption relies on the compounding pharmacy accurately dosing and properly formulating the product.
Variations in potency, purity, and formulation quality between compounding pharmacies could theoretically affect outcomes. This is one reason why choosing a reputable, well-reviewed pharmacy matters so much.
Side effects and safety profile
Side effects are the same regardless of whether you take Zepbound, Mounjaro, or compounded tirzepatide. The molecule causes the same physiological responses no matter how it is packaged.
Common side effects
Gastrointestinal symptoms dominate the side effect profile. Nausea is the most frequently reported, occurring in approximately 20-30% of participants in clinical trials. It tends to be worst during the initial weeks and during dose escalation periods, then improves as the body adapts.
Diarrhea and constipation seem contradictory, but both are common. Different people respond differently to the slowed gastric emptying that tirzepatide causes. Some experience looser stools, others the opposite. Both tend to resolve within the first few weeks at each dose level.
Headaches affect roughly 5-10% of users. Fatigue is common in the early weeks, particularly as caloric intake decreases substantially. Body aches, insomnia, and anxiety have also been reported by some users.
Injection site considerations
Injection site reactions can include redness, itching, swelling, or minor bruising. Proper injection technique reduces these reactions significantly. Rotating between injection sites, including the abdomen, thigh, and upper arm, helps prevent localized reactions from developing.
For people using compounded tirzepatide from vials rather than pre-filled pens, proper syringe injection technique and accurate syringe dosage measurement become important skills. Brand-name Zepbound uses pre-filled auto-injector pens that simplify administration, while compounded versions typically require drawing from a vial with an insulin syringe.
Less common side effects
Some users report effects that receive less attention in clinical trial summaries. Menstrual changes have been noted by many women, including irregular cycles, heavier or lighter periods, and changes in timing. Testosterone effects in men have also been discussed in clinical contexts. Hair thinning is reported by a subset of users, likely related to rapid weight loss rather than the medication itself. Muscle pain and joint pain are reported occasionally.
These side effects occur at the same rates regardless of formulation. Brand-name Zepbound does not cause fewer side effects than compounded tirzepatide, and compounded versions do not cause more. The molecule is the same. The side effect profile is the same.
Managing side effects effectively
Dietary modifications play a major role in tolerability. Following a tirzepatide-optimized diet plan, knowing which foods to avoid, and planning appropriate meals can dramatically reduce gastrointestinal symptoms. Strategic supplementation also helps address common nutritional gaps that develop with reduced food intake.
The slow titration schedule exists specifically to minimize side effects. Jumping to higher doses too quickly is one of the most common mistakes. Starting at 2.5 mg and increasing only every four weeks gives the body time to adapt. Some people find that microdosing approaches work even better, using smaller incremental increases to find their optimal dose with minimal side effects.
Storage and handling differences
Storage requirements are one area where practical differences exist between brand-name and compounded tirzepatide.
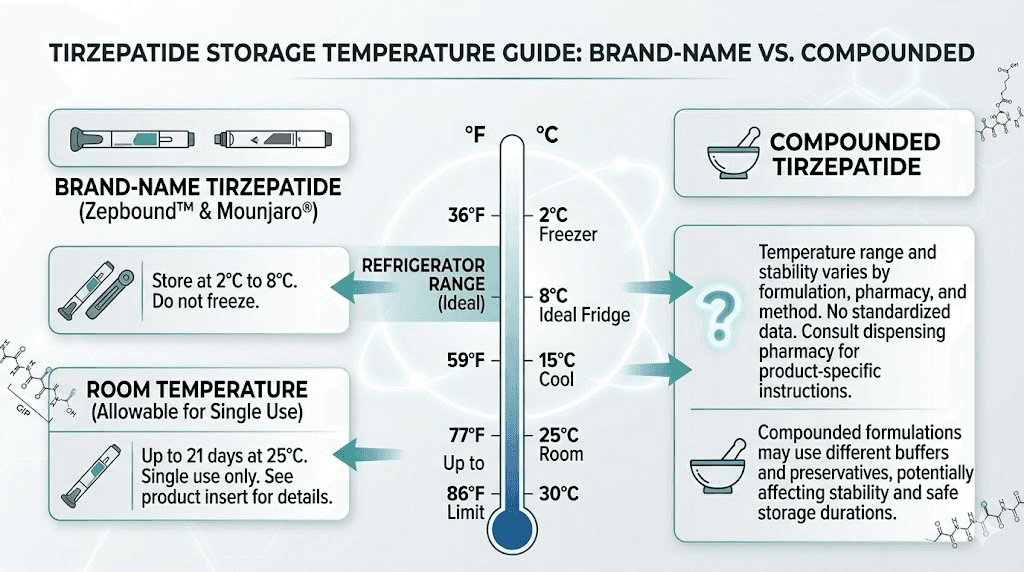
Zepbound storage
Zepbound pens should be stored in the refrigerator at 36-46 degrees Fahrenheit (2-8 degrees Celsius). An unopened pen can remain refrigerated until the expiration date printed on the packaging. Once you remove a pen from the refrigerator, it can be kept at room temperature (up to 86 degrees Fahrenheit or 30 degrees Celsius) for up to 21 days. Do not freeze Zepbound, and do not use it if it has been frozen.
Compounded tirzepatide storage
Compounded tirzepatide generally requires refrigeration as well, but the specific storage requirements depend on the formulation. Different pharmacies use different excipients and preservatives, which affect stability. Always follow the specific storage instructions provided by your compounding pharmacy.
Compounded tirzepatide may have a shorter shelf life than brand-name Zepbound. This is because compounding pharmacies may not conduct the same extensive stability testing that Eli Lilly performs. Understanding shelf life expectations, proper refrigeration requirements, and what happens if tirzepatide gets warm helps protect the potency of your medication.
For lyophilized (freeze-dried) tirzepatide, additional preparation steps are required. You need to reconstitute the powder with bacteriostatic water before use, following proper mixing ratios. Once reconstituted, the solution typically must be used within 28-30 days.
If you travel frequently, the tirzepatide travel guide covers how to maintain proper storage during trips.
Dosage and administration: what changes and what stays the same
The dosing protocol is identical regardless of formulation. But the administration experience differs between brand-name pens and compounded vials.
Brand-name administration
Zepbound and Mounjaro come in pre-filled, single-use auto-injector pens. You select your dose by choosing the appropriate pen strength. Press the pen against your injection site, push the button, and wait for the injection to complete. No measuring, no drawing from vials, no calculations. The pen delivers a precisely measured dose every time.
This simplicity is a genuine advantage. There is zero room for dosing error. The pen contains exactly the right amount of medication, and the injection mechanism ensures consistent delivery. For people who are new to self-injection or uncomfortable with syringes, the pen format significantly reduces the learning curve and the potential for mistakes.
Compounded tirzepatide administration
Most compounded tirzepatide comes in multi-dose vials. Administration requires drawing the correct volume with an insulin syringe, which means you need to understand unit-to-milligram conversions and dosing charts.
For example, if your vial contains 60 mg of tirzepatide in 6 mL of solution (a common concentration), each 0.1 mL equals 1 mg. To take a 2.5 mg dose, you would draw 0.25 mL. To take a 5 mg dose, you would draw 0.5 mL. The compounded tirzepatide dosage calculator handles these conversions automatically, but understanding the math provides an extra layer of safety.
Specific dose conversion guides are available for each common milligram amount: 7.5 mg, 10 mg, 12.5 mg, and 15 mg. Having the right reference for your specific dose eliminates guesswork.
Injection timing and frequency
All forms of tirzepatide are administered once weekly. The best time of day to inject and the best day of the week are personal preferences, but consistency matters. Pick a day, pick a time, and stick with it.
Some people ask about adjusting their schedule, whether they can take it a day early or two days early. Minor schedule adjustments are generally acceptable, but the medication works best with consistent seven-day intervals.
Others wonder about five-day dosing intervals or splitting doses across the week. While some practitioners experiment with modified schedules, the standard once-weekly protocol is what clinical trials validated.
How tirzepatide compares to other weight loss options
Understanding where tirzepatide fits in the broader landscape of weight management options helps put the Zepbound versus compounded decision in perspective.
Tirzepatide vs semaglutide
The semaglutide vs tirzepatide comparison is the most common question in this space. Semaglutide (Wegovy/Ozempic) targets only the GLP-1 receptor, while tirzepatide hits both GIP and GLP-1. The SURMOUNT-5 trial showed tirzepatide producing 47% greater weight loss. For people switching between the two medications, the dose conversion chart provides guidance on equivalent doses.
Some people start on semaglutide and wonder whether tirzepatide will work if semaglutide did not. The dual mechanism provides a different, often more effective approach. The reverse question also comes up when people consider alternating between the two.
Tirzepatide vs phentermine
Phentermine and tirzepatide work through completely different mechanisms. Phentermine is a stimulant that suppresses appetite through norepinephrine release. Tirzepatide works through incretin hormone pathways. Some people ask about combining both medications, though this approach requires careful medical supervision.
Tirzepatide vs newer agents
The GLP-1 space is evolving rapidly. Orforglipron is an oral non-peptide GLP-1 agonist in development. Survodutide targets glucagon and GLP-1 receptors. CagriSema combines cagrilintide and semaglutide. Mazdutide is another dual agonist. And retatrutide, a triple agonist hitting GIP, GLP-1, and glucagon receptors, has shown even greater weight loss in early trials. The difference between tirzepatide and retatrutide comes down to that third receptor target.
None of these newer agents change the fundamental Zepbound versus compounded tirzepatide decision. They are separate medications entirely. But they provide context for understanding where tirzepatide sits in the treatment hierarchy.
Diet and lifestyle optimization on tirzepatide
Regardless of whether you take Zepbound or compounded tirzepatide, dietary optimization makes a significant difference in both results and tolerability.
Nutrition fundamentals
Tirzepatide dramatically reduces appetite. This is the mechanism that drives weight loss. But reduced appetite can also lead to reduced nutrient intake if you are not deliberate about food choices. The foods to eat on tirzepatide guide emphasizes protein-rich, nutrient-dense options that maximize nutrition within a reduced caloric window.
Protein intake becomes especially important. Without adequate protein, a significant portion of weight loss comes from muscle rather than fat. This undermines long-term metabolic health and makes weight maintenance after discontinuation more difficult. The tirzepatide diet plan and printable meal plan are structured around protecting lean mass while maximizing fat loss.
Certain foods are best minimized or avoided while on tirzepatide. The foods to avoid guide identifies specific categories that commonly trigger or worsen gastrointestinal side effects.
Supplementation
Reduced food intake often means reduced vitamin and mineral intake. Key supplements to consider include a high-quality multivitamin, protein powder (particularly protein shakes formulated for GLP-1 users), and fiber supplements to address the constipation that many users experience. GLP-1 specific supplement stacks are becoming increasingly popular as more research identifies common nutrient deficiencies in this population.
Alcohol considerations
Drinking alcohol on tirzepatide requires caution. The medication changes how your body processes alcohol, often leading to faster intoxication, more severe hangovers, and increased gastrointestinal symptoms. If you choose to drink, understanding which alcoholic beverages are better tolerated can help minimize problems.
Exercise
You can absolutely lose weight on GLP-1 medications without exercise. But resistance training is strongly recommended to preserve muscle mass during rapid weight loss. Metabolic benefits compound when medication is paired with regular physical activity, and muscle preservation directly impacts long-term weight maintenance after eventual medication discontinuation.
What to expect when starting tirzepatide
Whether you start with Zepbound pens or compounded vials, the experience follows the same general pattern.
Week 1-4 (2.5 mg)
The first month is an adjustment period. The 2.5 mg starting dose is intentionally subtherapeutic for weight loss. Its purpose is to acclimate your body to the medication and minimize gastrointestinal side effects. Many people notice mild appetite reduction during this phase. Some notice nothing at all. Both responses are normal.
The first dose experience varies widely. Some people feel nauseous within hours. Others feel no effects for several days. A few report immediate and dramatic appetite suppression. There is no single "normal" response.
Week 5-8 (5 mg)
At 5 mg, most people begin noticing meaningful appetite suppression. This is the first true therapeutic dose for many users. Weight loss typically begins in earnest during this phase, with the first month of weight loss at therapeutic doses averaging 4-8 pounds, though individual variation is substantial.
Gastrointestinal side effects may reappear briefly with the dose increase, then settle again as the body adapts.
Months 3-6 and beyond
Progressive dose increases continue every four weeks as needed. Most people find their optimal dose somewhere between 5 mg and 15 mg. The weight loss timeline shows that results accelerate through the first six months and then gradually slow as you approach a new metabolic equilibrium.
Some people plateau during treatment. The troubleshooting guide for plateaus addresses common reasons why progress stalls and strategies for breaking through. Others wonder why they are not losing weight despite consistent dosing, a problem that usually traces back to dietary factors, dose optimization, or metabolic adaptation.
Discontinuation and long-term planning
Eventually, many people consider stopping tirzepatide. Whether due to cost, reaching goal weight, or personal preference, understanding the discontinuation process is important.
Stopping abruptly is not recommended. Gradual tapering off tirzepatide helps minimize rebound appetite and weight regain. The body needs time to readjust to functioning without the exogenous incretin support.
Weight regain after discontinuation is a documented reality. Clinical trial data shows that most people regain a significant portion of lost weight within 12-18 months of stopping medication. This is not a failure of willpower. It is biology. The same metabolic defense mechanisms that make initial weight loss difficult also push the body back toward its previous weight when pharmacological support is removed.
Maintaining weight loss after tirzepatide requires a deliberate strategy involving sustained dietary changes, regular exercise (particularly resistance training), and potentially a lower maintenance dose rather than complete discontinuation.
Special populations and considerations
Certain groups face additional considerations when choosing between Zepbound and compounded tirzepatide.
Women of reproductive age
Tirzepatide can affect fertility. Weight loss itself restores ovulatory function in many women with obesity-related anovulation, and tirzepatide accelerates this process. Some women have become pregnant unexpectedly while on tirzepatide. The medication should be discontinued before planned pregnancy, and women of reproductive age should use reliable contraception while taking it.
Breastfeeding while on tirzepatide is not recommended due to insufficient safety data. Menstrual cycle changes are commonly reported and usually normalize over time.
Post-surgical patients
People who have undergone surgery may need to time their tirzepatide use carefully. The medication slows gastric emptying and affects nutrient absorption, which can complicate surgical recovery. Knowing when to resume GLP-1 medication after surgery requires guidance from both your surgeon and prescribing provider.
People on other medications
Drug interactions exist with certain medications. Metformin and tirzepatide are commonly used together in diabetes management with a generally favorable interaction profile. Berberine is a supplement that some people combine with tirzepatide for additional metabolic benefits. Creatine and NAD+ are other common combinations. Always disclose all medications and supplements to your prescribing provider.
Making your decision: a practical framework
The choice between Zepbound and compounded tirzepatide is not about which one is "better." They contain the same molecule. The decision comes down to your specific circumstances.
Choose Zepbound if:
Your insurance covers it (check first, do not assume)
You qualify for the manufacturer savings card ($25/month)
You prefer the simplicity of pre-filled auto-injector pens
FDA approval and full manufacturing oversight matter to you
You do not want to measure doses or handle syringes
You can afford the $499/month self-pay price if insurance does not cover it
Choose compounded tirzepatide if:
Your insurance does not cover Zepbound
Cost is a primary concern ($150-400/month vs $499-1,086/month)
You are comfortable with syringe-based self-injection
You want a formulation with added ingredients like B12 or glycine
You prefer a specific provider or telehealth platform
You need flexible dosing options beyond the standard pen strengths
Regardless of your choice:
Follow the same titration schedule (start low, increase slowly)
Monitor for the same side effects
Optimize your diet and supplementation
Track your progress and adjust as needed
Work with a qualified prescribing provider
SeekPeptides provides the tools and resources to support either path. The peptide calculator handles dose conversions for compounded formulations. The dosage charts cover every common concentration. And the comprehensive protocol guides walk you through every step of the process, from first injection to long-term maintenance.
Frequently asked questions
Is Zepbound the same as tirzepatide?
Yes. Zepbound is a brand name for tirzepatide, the active pharmaceutical ingredient. They are the same molecule manufactured by Eli Lilly. Zepbound is FDA-approved specifically for chronic weight management, while Mounjaro (also tirzepatide) is approved for type 2 diabetes. For a full comparison of dosing protocols across formulations, see our complete guide.
Why is compounded tirzepatide cheaper than Zepbound?
Compounding pharmacies do not bear the same research and development costs, FDA approval costs, or marketing expenses as Eli Lilly. They source the raw peptide ingredient and formulate it at lower overhead costs. The tradeoff is that compounded products do not undergo the same regulatory review and manufacturing oversight. Our affordable tirzepatide guide compares pricing across dozens of providers.
Can I switch from Zepbound to compounded tirzepatide?
Yes. Since both contain the same active ingredient, switching is straightforward. Maintain the same milligram dose you were taking. The only adjustment is learning to draw from a vial with a syringe instead of using a pre-filled pen. The compounded tirzepatide starting dose guide covers the transition process in detail.
Is compounded tirzepatide safe?
When sourced from a reputable 503B outsourcing facility or licensed compounding pharmacy, compounded tirzepatide uses the same molecule as brand-name products. The safety risk comes from poorly run pharmacies that may have dosing inaccuracies, contamination, or stability issues. Research your pharmacy carefully, check for third-party testing, and review credentials before purchasing.
Does Zepbound work faster than compounded tirzepatide?
No. The rate of weight loss depends on the dose, your individual metabolism, diet, and activity level, not the brand. At the same milligram dose, brand-name and compounded tirzepatide produce the same effects. See the tirzepatide timeline for typical results by week.
Will my doctor prescribe compounded tirzepatide?
Some doctors prescribe compounded tirzepatide directly. Others refer patients to telehealth platforms that specialize in weight management prescriptions. The prescribing landscape varies by state and by provider. Many online platforms like IVIM, Elevate Health, and Belle Health handle both the prescription and the medication sourcing.
Can I use a Zepbound pen if I have a compounded tirzepatide prescription?
No. These are separate prescriptions for separate products. A prescription for compounded tirzepatide cannot be filled with Zepbound, and vice versa. Your provider needs to write a specific prescription for whichever product you intend to use.
What happens if compounded tirzepatide is no longer available?
Compounded tirzepatide availability depends on the FDA drug shortage designation. If the shortage is officially resolved and compounding is restricted, you would need to transition to brand-name Zepbound or Mounjaro, or explore alternative medications. Staying informed about regulatory changes helps you plan ahead.
External resources
For researchers serious about optimizing their tirzepatide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, dosing calculators, and detailed protocol databases that cover every formulation, every dose strength, and every practical question that arises during treatment.
In case I do not see you, good afternoon, good evening, and good night. May your doses stay accurate, your protocols stay consistent, and your results stay exactly what the data promised.