Feb 26, 2026

On paper, retatrutide and tirzepatide look like close relatives. Both target GLP-1 receptors. Both produce dramatic weight loss. Both come from the same pharmaceutical company. But the outcomes tell a different story entirely.
Tirzepatide activates two receptors. Retatrutide activates three. That single extra receptor, the glucagon receptor, changes everything about how the body burns fat, processes liver lipids, and preserves metabolic function during aggressive weight loss. In Phase 3 clinical trials, participants on retatrutide lost up to 28.7% of their body weight in 68 weeks. Tirzepatide, which already redefined what weight loss medications could achieve, delivered approximately 20.9% in similar timeframes.
The numbers are impressive. But numbers alone do not answer the real question researchers keep asking: is retatrutide actually better, or just different? The answer depends on what "better" means for your specific goals, your metabolic profile, and how you weigh factors like availability, safety data, and long-term track record. SeekPeptides members consistently identify this as one of the most asked questions in the peptide research community, and for good reason. These two peptides represent fundamentally different approaches to the same problem.
This guide breaks down every meaningful difference between retatrutide and tirzepatide. Mechanism of action. Weight loss data. Side effect profiles. Dosing protocols. Cost. Availability. Body composition effects. Liver fat reduction. Cardiovascular markers. And most importantly, which situations favor one over the other. By the end, you will have the specific, evidence-based information needed to make an informed decision rather than relying on hype or incomplete comparisons.
What makes retatrutide different from tirzepatide
The distinction starts at the molecular level. Both peptides belong to the incretin family, but they target different combinations of hormone receptors. Understanding these mechanisms is not just academic. It directly predicts which side effects you will experience, how quickly results appear, and what metabolic benefits extend beyond the scale.
Tirzepatide: the dual agonist approach
Tirzepatide activates two receptors simultaneously: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). This dual mechanism earned it FDA approval under the brand names Mounjaro (for type 2 diabetes) and Zepbound (for obesity).
GLP-1 receptor activation slows gastric emptying, reduces appetite signals in the brain, and enhances insulin secretion when blood sugar is elevated. These effects explain why people on tirzepatide feel less hungry and eat significantly fewer calories without white-knuckling through constant cravings.
GIP receptor activation adds a second layer. GIP improves insulin sensitivity, may influence fat tissue metabolism directly, and appears to amplify the appetite-suppressing effects of GLP-1 in ways researchers are still mapping. The combination of these two receptors is what separated tirzepatide from earlier single-agonist medications like semaglutide.
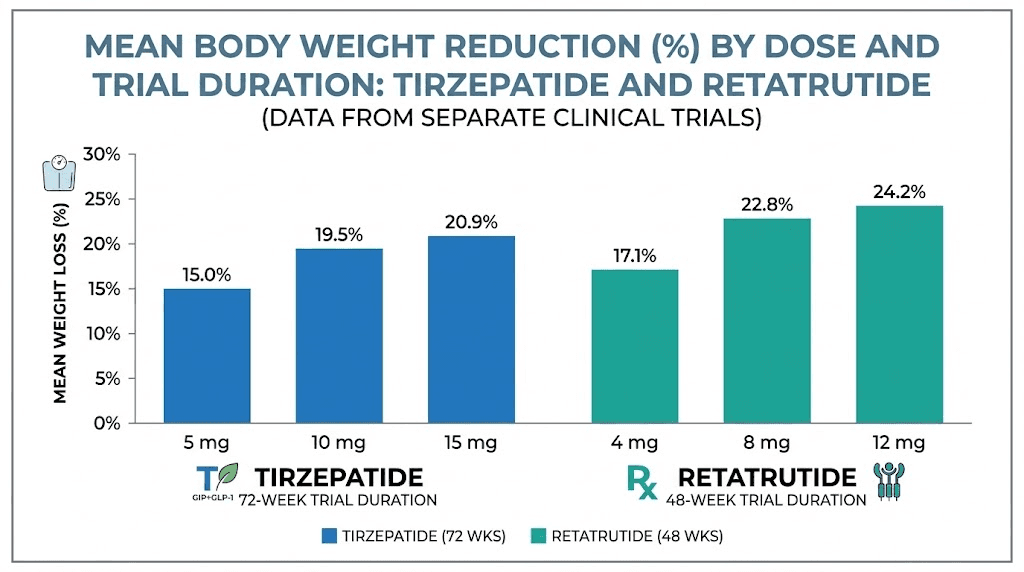
In the landmark SURMOUNT trials, tirzepatide at the 15mg dose produced average body weight reductions of 20.9% over 72 weeks. That translates to roughly 50 pounds for a 240-pound individual. These results were unprecedented at the time and established tirzepatide as the most effective weight loss medication on the market.
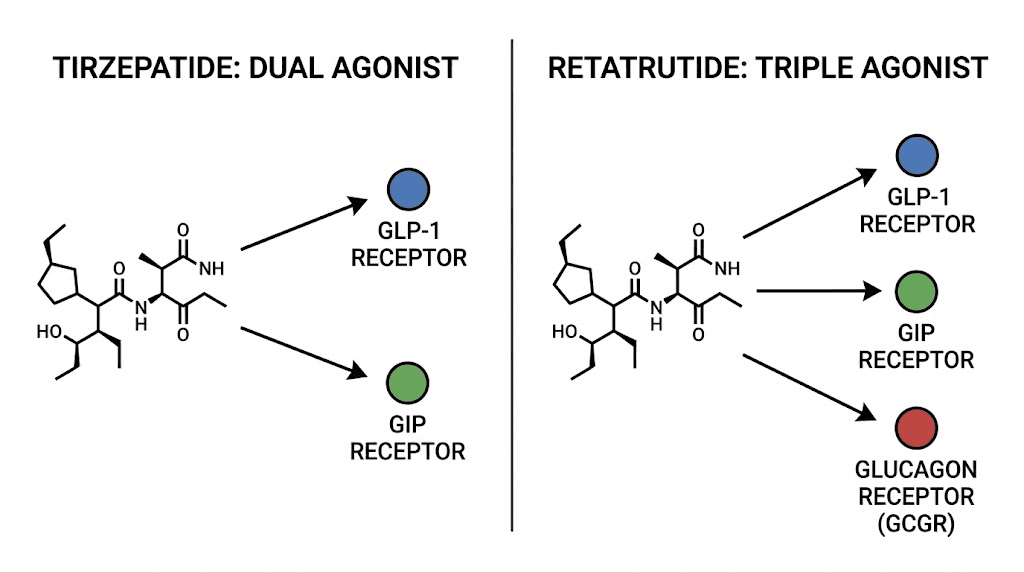
Retatrutide: the triple agonist revolution
Retatrutide takes the tirzepatide formula and adds a third target: the glucagon receptor. This makes it the first triple hormone receptor agonist with published clinical data in humans.
Three receptors. Three distinct metabolic pathways. The result is a compound that attacks obesity from angles that dual agonists simply cannot reach.
The GLP-1 and GIP components function similarly to tirzepatide, providing appetite suppression, improved insulin dynamics, and reduced caloric intake. But the glucagon receptor activation introduces entirely new metabolic effects that researchers find particularly compelling for specific patient populations.
Glucagon receptor activation increases hepatic fat oxidation, meaning the liver burns stored fat more aggressively. It raises resting energy expenditure, so the body burns more calories even at rest. And early data suggests it may help preserve lean muscle mass during rapid weight loss, a problem that plagues every GLP-1 based medication on the market.
The retatrutide Phase 2 obesity trial, published in the New England Journal of Medicine, demonstrated weight loss of up to 24.2% at the 12mg dose after just 48 weeks. That is a shorter treatment period producing greater percentage weight loss than tirzepatide achieved in 72 weeks. The Phase 3 TRIUMPH-4 trial pushed these numbers even further: participants on retatrutide 12mg lost an average of 28.7% of their body weight at 68 weeks.
Why the glucagon receptor changes everything
Of the three receptors retatrutide targets, the glucagon receptor deserves the most attention. It is the differentiator. The X-factor.
Glucagon has traditionally been seen as the "opposite of insulin," a hormone that raises blood sugar by triggering glucose release from the liver. For decades, the pharmaceutical approach was to suppress glucagon activity in diabetes patients. Retatrutide takes the opposite approach, strategically activating the glucagon receptor to unlock metabolic benefits that complement, rather than counteract, the insulin-sensitizing effects of GLP-1 and GIP.
Here is what glucagon receptor activation specifically does in the context of retatrutide:
Increased energy expenditure. Glucagon signals the body to ramp up thermogenesis. In practical terms, this means retatrutide users burn more calories at baseline than those on dual agonists alone. While tirzepatide has modest effects on metabolism, retatrutide appears to meaningfully elevate resting metabolic rate through this glucagon-mediated pathway.
Enhanced hepatic fat oxidation. The liver becomes a more efficient fat-burning organ. This is why retatrutide shows extraordinary results in liver fat reduction, with clinical data showing up to 82% decreases in liver fat content, a result no dual agonist has matched.
Potential muscle preservation. While definitive data is still emerging, glucagon receptor activation appears to shift the ratio of fat loss to lean mass loss more favorably. A Phase 2 substudy found that retatrutide produced up to 26.1% reduction in total body fat mass while limiting lean mass loss to 6.5kg. For researchers concerned about the muscle wasting associated with GLP-1 medications, this mechanism is highly relevant.
Weight loss results compared head to head
Numbers matter. When researchers evaluate whether retatrutide is "better" than tirzepatide, weight loss data is usually the first thing they examine. Here is what the clinical trials actually show.
Tirzepatide clinical trial data
The SURMOUNT program is the most comprehensive clinical trial dataset for tirzepatide in obesity. SURMOUNT-1 enrolled 2,539 adults with obesity (BMI of 30 or higher) or overweight (BMI of 27 or higher) with at least one weight-related condition.
Results at 72 weeks were striking:
5mg dose: 15.0% average weight loss
10mg dose: 19.5% average weight loss
15mg dose: 20.9% average weight loss
Placebo: 3.1% average weight loss
At the highest dose, 36.2% of participants lost 25% or more of their body weight. That is more than one in three people achieving what would have been considered impossible with earlier medications. Real-world tirzepatide results generally align with these trial outcomes, though individual variation is significant.
The more recent SURMOUNT-5 trial directly compared tirzepatide to semaglutide and found tirzepatide superior, with 20.2% weight loss versus 13.7% for semaglutide at 72 weeks. This confirmed that the dual agonist mechanism provides meaningful advantages over single GLP-1 agonism alone.
Retatrutide clinical trial data
Retatrutide data comes primarily from two sources: a Phase 2 trial published in the New England Journal of Medicine and the Phase 3 TRIUMPH program that began reporting results in mid-2025.
Phase 2 results (48 weeks):
1mg dose: 8.7% average weight loss
4mg dose: 17.1% average weight loss
8mg dose: 22.8% average weight loss
12mg dose: 24.2% average weight loss
Placebo: 2.1% average weight loss
These numbers came from a 48-week study period, six months shorter than the 72-week SURMOUNT trials. The rate of weight loss with retatrutide was noticeably faster, with substantial reductions appearing earlier in the treatment course.
Phase 3 TRIUMPH-4 results (68 weeks):
9mg dose: 26.4% average weight loss (29.1 kg)
12mg dose: 28.7% average weight loss (32.3 kg)
Placebo: 2.1% average weight loss
The 12mg retatrutide group lost an average of 71.2 pounds. Let that number sink in for a moment. The average participant lost more than 70 pounds in about 16 months. Analysts expect the TRIUMPH-1 trial, with its longer 80-week treatment period, may show weight loss exceeding 30%, a threshold that approaches what bariatric surgery achieves.
Direct comparison: the numbers side by side
Factor | Tirzepatide (15mg) | Retatrutide (12mg) |
|---|---|---|
Average weight loss | 20.9% | 28.7% |
Trial duration | 72 weeks | 68 weeks |
Absolute weight lost | ~50 lbs avg | ~71 lbs avg |
25%+ weight loss achieved | 36.2% | Data pending |
Mechanism | Dual (GLP-1/GIP) | Triple (GLP-1/GIP/Glucagon) |
FDA status | Approved | Phase 3 trials |
Injection frequency | Once weekly | Once weekly |
The raw percentage difference is approximately 8 percentage points in favor of retatrutide. On a 250-pound individual, that difference represents roughly 20 additional pounds lost. Meaningful? Absolutely. But context matters.
Tirzepatide data comes from larger, more diverse Phase 3 trials with thousands of participants. Retatrutide Phase 3 data is still emerging, and the TRIUMPH-4 population specifically had knee osteoarthritis, which may influence adherence and outcomes. A true apples-to-apples comparison requires the full TRIUMPH program data, which is expected to complete throughout the rest of this year.
What we can say with confidence: retatrutide produces greater absolute weight loss in the timeframes studied. Whether that advantage holds in broader populations, across longer treatment periods, and with varying compliance patterns remains an open question. For researchers tracking these developments, the retatrutide availability timeline is worth monitoring closely.
How the dosing protocols compare
Dosing matters as much as the drug itself. Start too aggressively, and gastrointestinal side effects drive people to quit. Escalate too slowly, and you lose months of potential progress. Both tirzepatide and retatrutide use gradual titration, but the specifics differ in ways that affect real-world outcomes.
Tirzepatide escalation schedule
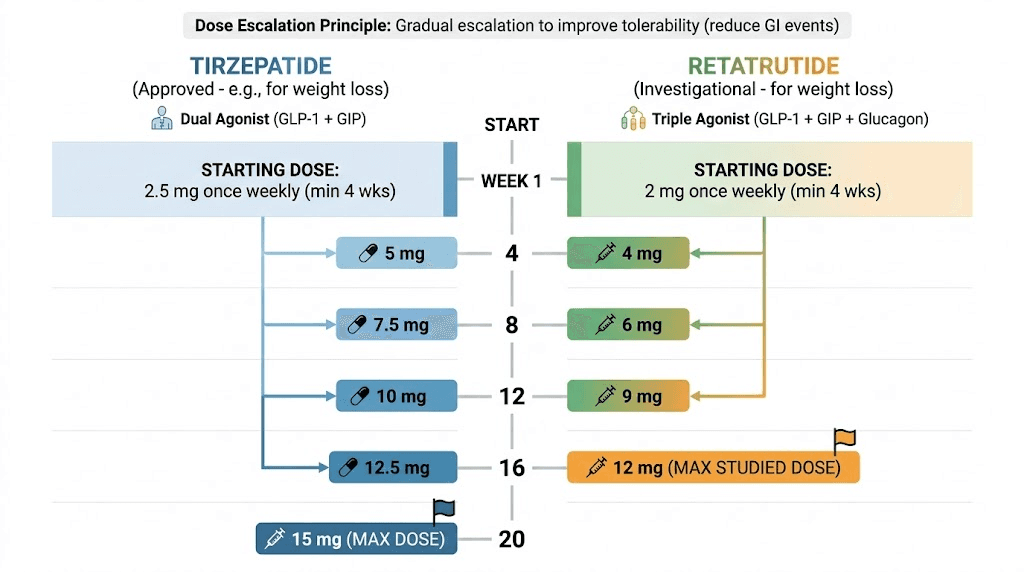
The FDA-approved tirzepatide dosing protocol follows this escalation:
Weeks 1-4: 2.5mg once weekly (initiation dose)
Weeks 5-8: 5mg once weekly
Weeks 9-12: 7.5mg once weekly
Weeks 13-16: 10mg once weekly
Weeks 17-20: 12.5mg once weekly
Week 21+: 15mg once weekly (maintenance)
Each dose increase happens every four weeks, allowing the body to adjust. The starting dose of 2.5mg is sub-therapeutic for weight loss. It exists purely to establish GI tolerance. Most of the meaningful weight loss occurs at 10mg and above.
Full escalation to the maximum 15mg dose takes approximately 20 weeks, or about five months. Many users report that appetite suppression becomes truly noticeable around the 5-7.5mg range, with maximum effects kicking in at 10-15mg. The tirzepatide dosage calculator can help determine exact unit measurements for compounded formulations.
Retatrutide titration protocol
Retatrutide follows a similar monthly escalation pattern, but with different starting points and targets:
Weeks 1-4: 1mg once weekly (initiation)
Weeks 5-8: 2mg once weekly
Weeks 9-12: 4mg once weekly
Weeks 13-16: 8mg once weekly
Week 17+: 12mg once weekly (maintenance)
The retatrutide starting dose is lower at 1mg, reflecting the triple agonist mechanism and the need for careful GI adjustment. Phase 2 trials found that starting at 4mg directly led to significantly more gastrointestinal adverse events compared to the gradual 2mg start. The slower initiation is not optional for tolerability.
Full escalation to 12mg takes approximately 16 weeks, or about four months, slightly faster than tirzepatide reaches its maximum. However, retatrutide doses escalate in larger jumps (particularly the 4mg to 8mg step), which some users find more challenging gastrointestinally. Detailed retatrutide scheduling guides can help plan these transitions.
Time to therapeutic dose: a practical comparison
Milestone | Tirzepatide | Retatrutide |
|---|---|---|
Starting dose | 2.5mg | 1mg |
First therapeutic dose | 5mg (week 5) | 4mg (week 9) |
Mid-range dose | 10mg (week 13) | 8mg (week 13) |
Maximum dose | 15mg (week 21) | 12mg (week 17) |
Injection volume | Varies by concentration | Varies by concentration |
Injection frequency | Once weekly | Once weekly |
Both peptides require patience during titration. Rushing the process leads to nausea, vomiting, and diarrhea severe enough to make people abandon treatment entirely. The retatrutide injection technique and tirzepatide injection method are identical, as both use standard subcutaneous protocols.
For researchers transitioning between these peptides, the retatrutide dosage schedule after tirzepatide provides specific guidance on managing the switch without unnecessary side effects or dose overlap.
Side effects and safety profiles
Every medication comes with tradeoffs. The question is never whether side effects exist, but whether the benefits justify them and whether those side effects are manageable in practice. Here is where retatrutide and tirzepatide diverge in ways that matter.
Common gastrointestinal side effects
Both peptides cause gastrointestinal issues. This is expected with any GLP-1 receptor agonist and is largely a consequence of slowed gastric emptying and altered appetite signaling. The most frequently reported side effects for both include:
Nausea (most common, especially during dose escalation)
Diarrhea
Constipation
Vomiting
Decreased appetite (technically the intended effect)
Abdominal discomfort
For tirzepatide, these side effects are well-characterized from multiple large Phase 3 trials and extensive real-world use. Nausea affects roughly 20-30% of users, typically peaking during dose escalation and subsiding as the body adjusts. Constipation management strategies and diarrhea duration expectations are well documented. Most users find these effects tolerable and transient.
Retatrutide shows a similar GI side effect profile, but with some important nuances. A network meta-analysis found that adverse events were more frequent with retatrutide (relative risk 4.10) compared to tirzepatide (relative risk 2.78). In plain terms: retatrutide causes more side effects per capita than tirzepatide in clinical trial settings.
However, this comparison comes with major caveats. The retatrutide data primarily reflects Phase 2 trials with ongoing dose optimization. The use of a lower 2mg starting dose (versus 4mg) significantly reduced GI events, and Phase 3 protocols have refined the titration schedule further. As more data emerges, the gap may narrow.
Heart rate: the key safety differentiator
This is where the conversation gets serious.
Retatrutide has been associated with greater increases in resting heart rate compared to tirzepatide. This effect appears to be driven by the glucagon receptor agonism, as glucagon naturally increases heart rate through sympathetic nervous system activation.
In clinical trials, retatrutide at higher doses showed mean heart rate increases that exceeded those seen with tirzepatide. While the magnitude was generally modest (single-digit beats per minute in most cases), this cardiac signal is one of the primary reasons retatrutide remains investigational and is undergoing extensive Phase 3 safety monitoring.
For researchers with pre-existing cardiovascular conditions or those taking medications that affect heart rate, this difference is particularly relevant. Tirzepatide, with its established cardiovascular safety profile and completed SURPASS and SURMOUNT programs, provides a more thoroughly characterized risk landscape. Comparing side effect profiles across the GLP-1 class reveals that heart rate effects vary meaningfully between compounds.
Long-term safety data
This is where tirzepatide holds a decisive advantage. It has been on the market since mid-2022 (Mounjaro) and 2023 (Zepbound), with millions of prescriptions filled. The post-marketing safety surveillance provides a level of real-world confidence that no amount of clinical trial data can replicate.
Retatrutide has zero real-world safety data. Everything we know comes from controlled clinical trials involving thousands, not millions, of participants. Phase 3 trials (TRIUMPH-1 through TRIUMPH-7) are specifically designed to address long-term safety questions, but results are still rolling in.
For researchers who prioritize established safety profiles, tirzepatide is the clear choice today. For those willing to accept a less complete safety picture in exchange for potentially greater efficacy, retatrutide represents the calculated risk of an early-stage therapeutic.
Other notable side effects
Beyond GI issues and heart rate, both peptides share several class-wide concerns:
Hair loss. Rapid weight loss from any cause can trigger telogen effluvium, a temporary hair shedding condition. This has been reported with both tirzepatide and retatrutide, though it is likely related to the weight loss itself rather than the medication directly. GLP-1 hair loss is a common concern, and retatrutide hair loss specifically follows similar patterns.
Fatigue. Both compounds can cause tiredness, particularly during early dose escalation when caloric intake drops significantly. Tirzepatide fatigue is generally mild and improves as the body adapts. Adequate protein intake and consistent meal timing help mitigate this effect.
Injection site reactions. Redness, itching, or swelling at the injection site occurs with both peptides but is typically mild and self-resolving. Injection site reaction management is straightforward in most cases.
Headaches. Reported with both compounds, usually during the first few weeks. GLP-1 headaches tend to resolve as the body adjusts to the medication.
Beyond weight loss: metabolic benefits compared
Weight loss is the headline, but the story extends far beyond the number on the scale. Both peptides improve a constellation of metabolic markers, and the differences here may ultimately matter more than the weight loss gap for certain populations.
Liver fat and metabolic dysfunction-associated steatotic liver disease
This is where retatrutide separates itself most dramatically from every other medication in its class.
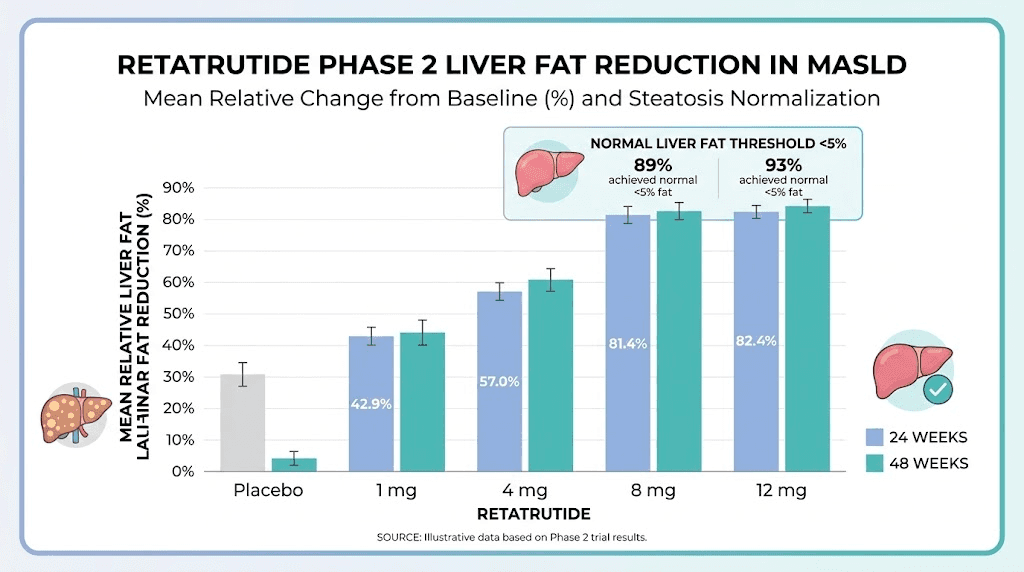
A Phase 2a randomized trial specifically evaluated retatrutide for MASLD (formerly called NAFLD). The results were extraordinary:
Retatrutide 1mg: 42.9% reduction in liver fat at 24 weeks
Retatrutide 4mg: 57.0% reduction
Retatrutide 8mg: 81.4% reduction
Retatrutide 12mg: 82.4% reduction
Placebo: 0.3% increase
By 48 weeks, 89% of participants on 8mg and 93% on 12mg had achieved normal liver fat levels (below 5%). These numbers are staggering. No other GLP-1 based medication has demonstrated this level of liver fat clearance in clinical settings.
Tirzepatide also reduces liver fat, and SURMOUNT trial substudies have shown meaningful improvements in hepatic steatosis. But the magnitude is not comparable. The glucagon receptor in retatrutide directly stimulates hepatic fat oxidation, creating a specific mechanism for liver fat reduction that dual agonists lack.
For individuals with fatty liver disease, this distinction may be the most important factor in choosing between the two compounds. MASLD affects an estimated 25-30% of adults globally and is a leading cause of liver cirrhosis and liver cancer. A medication that can resolve steatosis in 90%+ of patients represents a potential paradigm shift in hepatology.
Cardiovascular markers
Both peptides improve cardiovascular risk factors as a consequence of weight loss and direct metabolic effects. The TRIUMPH-4 trial showed that retatrutide at 12mg reduced:
Systolic blood pressure: 14.0 mmHg reduction
Non-HDL cholesterol: clinically meaningful improvement
High-sensitivity C-reactive protein: significant reduction (a marker of systemic inflammation)
Triglycerides: substantial decrease
Tirzepatide produces similar cardiovascular improvements, with well-documented reductions in blood pressure, triglycerides, and inflammatory markers across the SURMOUNT and SURPASS programs. The magnitude of improvement correlates strongly with the degree of weight loss, meaning that the greater weight loss with retatrutide may translate to greater cardiovascular benefit.
However, the heart rate increase associated with retatrutide complicates this picture. Elevated resting heart rate is itself a cardiovascular risk factor. Whether the metabolic benefits of triple agonism outweigh the heart rate concern is an active area of investigation in the TRIUMPH trials.
Body composition and muscle preservation
One of the most discussed drawbacks of GLP-1 medications is muscle loss. When you lose weight rapidly, a significant portion of that loss comes from lean tissue, not just fat. With both tirzepatide and semaglutide, approximately 33-40% of total weight lost consists of lean mass.
Retatrutide may offer an advantage here. The glucagon receptor activation promotes fat oxidation specifically, which could shift the ratio toward greater fat loss relative to lean mass loss. A Phase 2 substudy analyzing body composition found that retatrutide produced up to 26.1% reduction in total body fat mass while limiting lean mass loss to approximately 6.5kg.
Is this definitively better than tirzepatide for muscle preservation? The honest answer is: we do not know yet. Direct comparisons using identical body composition measurement methods (like DEXA scanning) across both compounds are needed. What we can say is that the theoretical mechanism supports better lean mass retention, and early data is encouraging.
Regardless of which peptide someone uses, the recommendations for nutritional supplementation and resistance training remain critical. Adequate protein intake (1.0-1.2g per kg of body weight daily at minimum), progressive resistance exercise, and proper protein supplementation strategies can significantly reduce lean mass loss with either compound.
Blood sugar and diabetes management
Tirzepatide (Mounjaro) gained its first FDA approval specifically for type 2 diabetes. The SURPASS program demonstrated HbA1c reductions of 2.0-2.3% at the 15mg dose, with many participants achieving HbA1c below 5.7% (the threshold for normal blood sugar). It is one of the most effective diabetes medications ever developed.
Retatrutide also shows potent glucose-lowering effects. Phase 2 data in patients with type 2 diabetes demonstrated HbA1c reductions of up to 2.16% at the 12mg dose after 36 weeks, along with 16.9% body weight reduction. Additional TRIUMPH trials are evaluating retatrutide specifically in diabetes populations.
For researchers managing both obesity and type 2 diabetes simultaneously, both peptides deliver exceptional glycemic control. The choice may ultimately come down to availability, given that tirzepatide is currently the only FDA-approved option.
Availability, approval status, and cost
Efficacy data means nothing if you cannot actually access the medication. This section addresses the practical realities of obtaining each peptide.
Tirzepatide: available now
Tirzepatide is fully FDA-approved and commercially available under two brand names:
Mounjaro: Approved for type 2 diabetes management
Zepbound: Approved for chronic weight management in adults with obesity (BMI 30+) or overweight (BMI 27+) with at least one weight-related condition
Compounded versions of tirzepatide are also available through compounding pharmacies, offering more affordable options for those without insurance coverage. Sources like Empower Pharmacy and various telehealth platforms provide access to compounded tirzepatide at significantly lower costs than brand-name medications.
The availability of compounded tirzepatide has democratized access to this medication, though researchers should verify source quality and testing standards. The peptide cost calculator can help estimate monthly expenses based on dosing requirements.
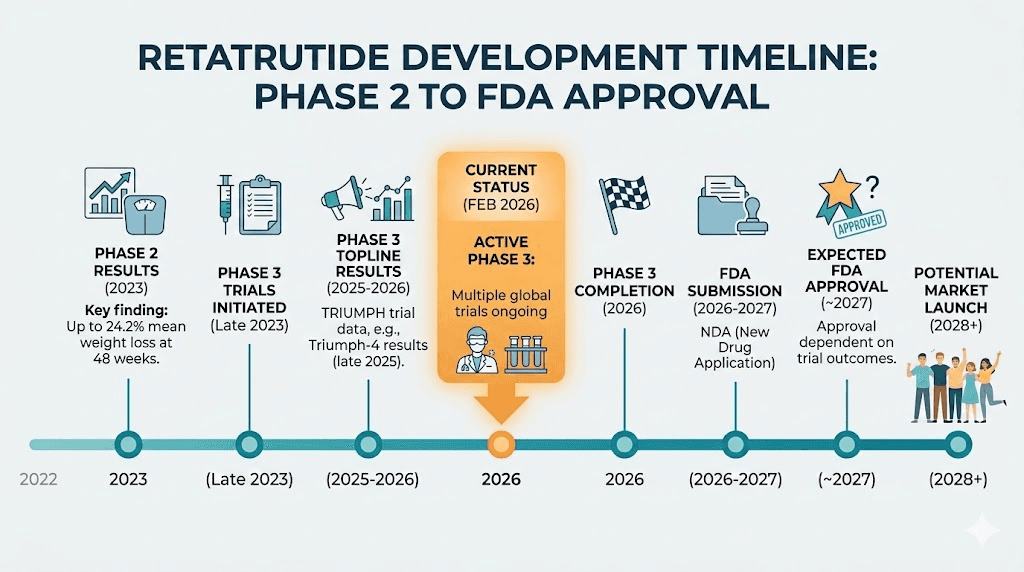
Retatrutide: the timeline to market
Retatrutide is currently in Phase 3 clinical trials. Eli Lilly, the company developing both retatrutide and tirzepatide, is running seven TRIUMPH trials covering obesity, type 2 diabetes, and specific metabolic conditions.
The expected timeline:
Phase 3 trials completing: Results rolling out throughout the rest of this year
FDA submission: Expected sometime after full Phase 3 data is available
Potential FDA approval: Estimated for late 2027 at the earliest
Commercial availability: If approved, launch would follow in 2027-2028
For those tracking retatrutide availability updates, the situation is evolving. Research-grade retatrutide is available through peptide research vendors like Peptide Sciences and Paradigm Peptides for legitimate research purposes. The sourcing landscape for retatrutide requires careful vendor evaluation to ensure purity and authenticity.
Cost comparison
Direct cost comparison is complicated by the fact that retatrutide has no commercial pricing yet. What we can assess:
Factor | Tirzepatide | Retatrutide |
|---|---|---|
Brand-name cost | $950-1,100/month | Not yet priced |
Compounded cost | $150-400/month | N/A (not compounded) |
Research grade cost | Varies | $80-200 per 10mg vial |
Insurance coverage | Variable, improving | None (investigational) |
For a cost-effectiveness perspective, a published analysis found tirzepatide to be the most cost-effective option compared to oral semaglutide, with an incremental cost-effectiveness ratio of $34,212 per QALY gained. Retatrutide cost-effectiveness data will not be available until commercial pricing is established.
Researchers exploring retatrutide pricing for research purposes should account for the reconstitution requirements and bacteriostatic water volumes when calculating total protocol costs.
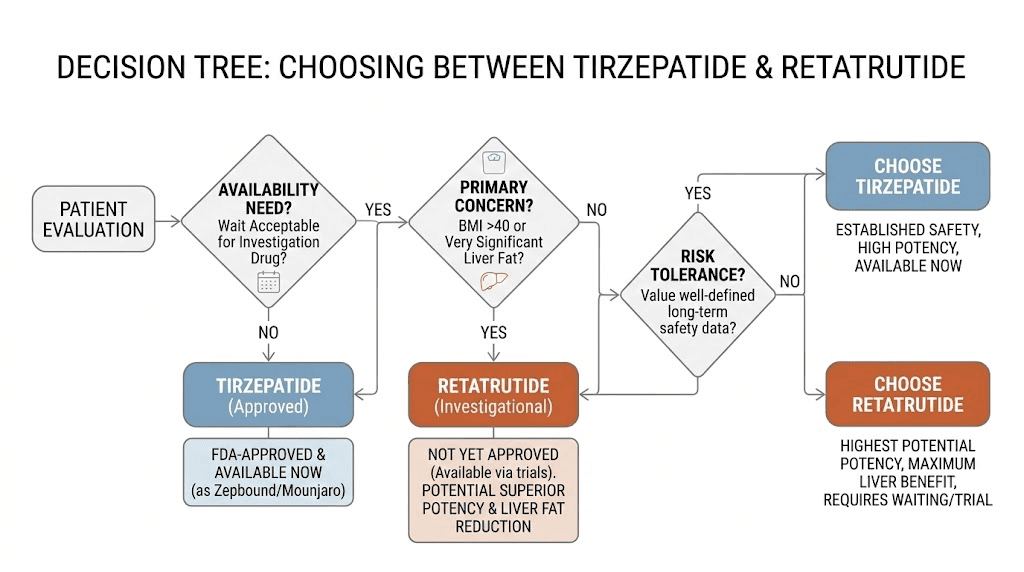
Who should consider retatrutide over tirzepatide
The "better" peptide depends entirely on the individual. Their goals. Their health status. Their risk tolerance. Their access to quality sourcing. Here is how to think about which compound fits which situation.
High BMI and aggressive weight loss goals
For individuals with BMI above 40 or those needing to lose 100+ pounds, the additional weight loss potency of retatrutide may be particularly valuable. The difference between 21% and 29% weight loss becomes clinically significant at higher starting weights.
Consider: a 300-pound individual on tirzepatide might expect to reach approximately 237 pounds (63 pounds lost). The same individual on retatrutide might reach approximately 214 pounds (86 pounds lost). That 23-pound difference represents meaningful additional risk reduction for obesity-related conditions. It could mean the difference between still qualifying as obese and reaching the overweight category.
However, this must be balanced against the less established safety profile. SeekPeptides members often note that the risk-benefit calculation shifts favorably toward retatrutide as starting BMI increases, because the health risks of severe obesity are so substantial that greater weight loss provides proportionally greater benefit.
Fatty liver disease
If MASLD or NASH is present, retatrutide is the standout choice based on current evidence. The 82-93% liver fat resolution rate has no parallel in the GLP-1 medication class. For individuals whose primary concern is liver health rather than (or in addition to) weight loss, this data point may be decisive.
Tirzepatide does improve liver fat, but the magnitude is not comparable to what the glucagon receptor agonism in retatrutide achieves. Any researcher dealing with elevated liver enzymes or diagnosed fatty liver disease should strongly consider the retatrutide data when making their decision.
Switching from tirzepatide
A growing number of researchers are transitioning from tirzepatide to retatrutide, typically because they have hit a tirzepatide weight loss plateau or want to push past the weight loss ceiling of dual agonism.
The transition from tirzepatide to retatrutide requires careful planning. You cannot simply swap one for the other at equivalent doses, as the mechanisms differ. The retatrutide dosing schedule after high-dose tirzepatide accounts for existing GLP-1 and GIP receptor adaptation while introducing glucagon receptor agonism gradually.
Key considerations when switching:
Allow a washout period based on tirzepatide half-life
Start retatrutide at the standard 1mg initiation dose regardless of previous tirzepatide dose
Expect renewed GI side effects as the glucagon receptor is a new stimulus
Monitor heart rate more carefully during the initial escalation period
When tirzepatide is the better choice
Tirzepatide remains the superior option in several specific situations. Do not let the allure of bigger numbers override practical considerations.
You want an FDA-approved medication. If regulatory approval, prescription access, and insurance coverage matter, tirzepatide is the only option. Full stop.
You prioritize long-term safety data. With millions of prescriptions filled and years of post-marketing surveillance, tirzepatide safety is characterized in ways retatrutide cannot match for years to come.
You have cardiovascular concerns. The heart rate signal with retatrutide, while generally modest, warrants caution in individuals with pre-existing heart conditions, arrhythmias, or those taking heart rate-affecting medications.
Moderate weight loss goals. If you need to lose 15-25% of body weight, tirzepatide achieves this reliably at its upper doses. The additional potency of retatrutide may not justify the trade-offs in risk profile.
You want compounded options. Compounded tirzepatide is widely available through legitimate pharmacies. Compounded retatrutide does not exist in this market.
Practical protocols for each peptide
Understanding the theory is one thing. Implementing it is another. Below are evidence-based protocols for each compound, as well as a transition protocol for researchers moving between them.
Protocol 1: standard tirzepatide protocol
Goal: Achieve 15-20% body weight reduction over 6-12 months
Dose escalation:
Weeks 1-4: 2.5mg subcutaneous injection once weekly
Weeks 5-8: 5mg once weekly
Weeks 9-12: 7.5mg once weekly (optional, can skip to 10mg)
Weeks 13-16: 10mg once weekly
Weeks 17-20: 12.5mg once weekly
Week 21+: 15mg once weekly maintenance
Administration:
Inject subcutaneously in abdomen, thigh, or upper arm
Rotate injection sites to prevent injection site reactions
Best administered on the same day each week
Store properly per tirzepatide storage requirements
Nutritional support:
Minimum 100-120g protein daily (see tirzepatide diet planning)
Avoid high-fat, greasy foods during escalation (check foods to avoid list)
Stay hydrated, minimum 64oz water daily
Consider targeted supplementation including multivitamins, omega-3s, and B12
Expected timeline:
Weeks 1-4: Appetite suppression begins, 1-3% weight loss
Weeks 5-12: Noticeable results, 5-8% weight loss
Weeks 13-24: Significant changes, 10-15% weight loss
Weeks 25-52: Approaching maximum benefit, 15-20%+ weight loss
Protocol 2: retatrutide research protocol
Goal: Achieve 20-28% body weight reduction over 6-12 months
Dose escalation:
Weeks 1-4: 1mg subcutaneous injection once weekly
Weeks 5-8: 2mg once weekly
Weeks 9-12: 4mg once weekly
Weeks 13-16: 8mg once weekly
Week 17+: 12mg once weekly maintenance
Administration:
Inject subcutaneously following standard retatrutide injection technique
Optimal injection sites described in the retatrutide injection site guide
Store refrigerated, reconstituted vials have limited stability (see retatrutide shelf life)
Reconstitute with bacteriostatic water per reconstitution chart
Important monitoring:
Track resting heart rate weekly (the glucagon receptor can elevate HR)
Monitor liver enzymes if MASLD is present
Track body composition changes, not just scale weight
Assess GI tolerance at each dose increase, slow escalation if needed
Expected timeline:
Weeks 1-4: Mild appetite effects at initiation dose
Weeks 5-12: Progressive appetite suppression, 5-10% weight loss
Weeks 13-24: Significant metabolic effects, 12-18% weight loss
Weeks 25-48: Approaching maximum benefit, 20-28%+ weight loss
Protocol 3: transitioning from tirzepatide to retatrutide
Goal: Safely switch between compounds while maintaining weight loss momentum
This protocol is specifically for researchers who have been on tirzepatide and want to transition to retatrutide, typically because of a plateau or desire for additional metabolic benefits.
Transition steps:
Step 1: Taper tirzepatide to 10mg or below over 2-4 weeks
Step 2: Take final tirzepatide dose
Step 3: Wait 7-10 days (tirzepatide half-life is approximately 5 days)
Step 4: Begin retatrutide at 1mg, regardless of previous tirzepatide dose
Step 5: Follow standard retatrutide escalation from 1mg
Critical notes:
Do NOT skip the low-dose initiation, glucagon receptor activation is new to your system
Expect temporary appetite rebound during the washout period
GI side effects will likely recur as you introduce the new compound
Full benefit comparison takes 12+ weeks on retatrutide at therapeutic doses
For detailed dose conversion guidance, the retatrutide vs tirzepatide dosage chart provides specific unit calculations for each dose level. The retatrutide dosage calculator can assist with precise reconstitution volumes.
Week by week expectations: what actually happens on each peptide
Clinical trial averages tell you what to expect over months. But researchers want to know what happens week by week. When does the appetite suppression kick in? When do the side effects peak? When do you start seeing real changes? Here is a realistic timeline for each compound based on clinical data and community-reported experiences.
Tirzepatide: the week-by-week journey
Weeks 1-2 (2.5mg): Most researchers describe this as the "nothing" phase. The initiation dose is sub-therapeutic. You might notice very mild appetite reduction, or you might notice absolutely nothing at all. GI side effects are typically minimal or absent. This phase exists purely to prepare the digestive system for higher doses. Some people experience mild nausea, but it tends to be brief and manageable. Use this time to establish your injection technique and routine.
Weeks 3-4 (2.5mg): Still sub-therapeutic, but some users report subtle changes. Slightly smaller portions feel satisfying. Food noise, that constant background hum of thinking about food, begins to quiet down. Scale changes are typically 1-3 pounds, mostly water weight from reduced carbohydrate and sodium intake.
Weeks 5-8 (5mg): This is when things start to shift. Appetite suppression becomes noticeable and consistent. Meals that once seemed normal now feel too large. Cravings for high-calorie foods diminish. Nausea is most commonly reported during this first dose increase. Weight loss accelerates to roughly 1-2 pounds per week. Most users first notice real effects during this period.
Weeks 9-16 (7.5-10mg): The therapeutic range. Appetite suppression is strong and reliable. Many researchers describe feeling genuinely uninterested in food rather than fighting hunger. Weight loss becomes visible to others. Energy levels may dip temporarily as caloric intake drops, which is why fatigue management matters. GI side effects from earlier dose increases have typically resolved. By week 16, most people have lost 8-12% of starting body weight.
Weeks 17-24 (12.5-15mg): Maximum dose territory. Appetite is significantly suppressed. Some researchers struggle to eat enough protein, which makes the meal planning aspect critical. Weight loss continues at a steady pace. Total loss by week 24 typically ranges from 12-16%. Side effects are generally stable at this point, though constipation remains the most persistent GI complaint at higher doses.
Weeks 25-52+: Maintenance and continued slow progress. The rate of weight loss naturally decelerates as you approach a new equilibrium. Most of the total weight loss (about 80%) occurs in the first 36 weeks. The remaining 20% comes gradually over the following months. The full tirzepatide timeline shows that results continue to accumulate even late in the treatment course, just at a slower pace.
Retatrutide: the week-by-week journey
Weeks 1-4 (1mg): Even more subtle than tirzepatide initiation. The 1mg dose activates all three receptors at very low levels. Most researchers report no meaningful appetite changes, though a few notice slight metabolic effects, mild warmth or flushing, that may reflect early glucagon receptor activation. Use this time for baseline monitoring, especially resting heart rate measurements.
Weeks 5-8 (2mg): Appetite effects begin appearing. The triple agonist mechanism means appetite suppression builds differently than with dual agonists alone. Some researchers describe a unique quality to the satiety, not just feeling less hungry, but feeling genuinely indifferent to food. Mild nausea is common during this transition. Weight loss begins in earnest, typically 2-4% of body weight by week 8.
Weeks 9-12 (4mg): A critical transition point. The 4mg dose represents the first truly therapeutic level. Appetite suppression becomes pronounced. Energy expenditure begins to noticeably increase, likely from glucagon receptor effects. Some researchers report feeling warmer than usual or having mildly elevated resting heart rate. GI side effects from the dose increase are typically manageable but present. Weight loss accelerates to 1-3 pounds per week depending on starting weight.
Weeks 13-16 (8mg): This is the most challenging dose transition. Going from 4mg to 8mg represents a doubling, and the GI adjustment can be rougher than earlier increases. Nausea, if it is going to be problematic, usually peaks here. However, the weight loss effects are now dramatic. By week 16, most research subjects in clinical trials had lost 12-16% of body weight. The liver fat reduction is already substantial at this dose level, with 79% of MASLD trial participants achieving normal liver fat at 8mg.
Weeks 17-24 (12mg maintenance): Full maintenance dose. All three receptor pathways are maximally activated. Appetite suppression is powerful. Metabolic rate elevation from glucagon agonism is at full effect. Weight loss continues at an impressive pace that exceeds what most tirzepatide users experience at any dose. Total loss by week 24 typically ranges from 18-22% based on Phase 2 data.
Weeks 25-48+: Continued progress beyond what dual agonists can achieve. The Phase 2 trial showed 24.2% weight loss at 48 weeks, and Phase 3 data suggests 28.7% at 68 weeks. The rate does slow down, but the glucagon-mediated energy expenditure component means the deceleration may be less pronounced than with tirzepatide. Liver fat, cardiovascular markers, and body composition continue to improve throughout this extended treatment period.
Common mistakes researchers make with both peptides
Understanding what goes wrong is as important as understanding what goes right. These are the most frequent errors that undermine results with both tirzepatide and retatrutide.
Rushing dose escalation
Impatience kills protocols. Researchers who jump doses too quickly end up with severe nausea, vomiting, and sometimes enough misery to abandon the peptide entirely. The titration schedule exists for biological reasons, not as a suggestion.
With tirzepatide, some users try to skip the 2.5mg initiation or jump directly to 10mg+. This almost always backfires. With retatrutide, the temptation to skip from 2mg directly to 8mg (bypassing 4mg) can trigger GI events severe enough to require medical attention.
Follow the schedule. Every step serves a purpose. If you cannot tolerate a dose increase, stay at the current level for an additional 2-4 weeks before trying again. There is no prize for reaching maximum dose fastest. The proper starting dose approach prevents the majority of intolerable side effects.
Inadequate protein intake
This is the single biggest mistake affecting long-term outcomes with any GLP-1 medication. When appetite drops dramatically, protein intake drops with it. Protein is the macronutrient most responsible for preserving lean muscle mass during weight loss.
The minimum target is 1.0g of protein per kg of body weight daily. Many experts recommend 1.2-1.6g/kg for people on aggressive weight loss regimens. At a 200-pound body weight, that means 90-145 grams of protein per day, every day, without exception.
When appetite is suppressed, this requires deliberate effort. Protein supplementation strategies become essential. Prioritizing protein at every meal, using shakes or bars as supplements, and tracking intake carefully prevents the excessive lean mass loss that gives these medications a bad reputation for "making you skinny fat."
Ignoring hydration
Reduced food intake means reduced water intake from food sources. Both peptides can cause GI effects that further deplete fluid. Dehydration worsens constipation, headaches, fatigue, and dizziness.
Minimum fluid target: 64 ounces daily, more in hot climates or with regular exercise. Electrolyte supplementation (sodium, potassium, magnesium) helps maintain proper hydration balance, particularly during the first few months when caloric intake drops most sharply.
Skipping resistance training
Weight loss without resistance training means losing both fat and a disproportionate amount of muscle. This is true for all weight loss methods, but particularly relevant with GLP-1 medications because the appetite suppression is so effective that severe caloric deficits can occur.
Progressive resistance training 2-4 times per week signals the body to maintain muscle tissue even in a caloric deficit. Combined with adequate protein, resistance training can shift the ratio of fat-to-lean-mass loss from roughly 60/40 to closer to 80/20 or even 85/15.
This applies equally to both tirzepatide and retatrutide users. While retatrutide may have a slight inherent advantage for muscle preservation through glucagon receptor effects, that advantage does not replace the need for resistance training. Think of the glucagon benefit as an addition to, not a substitute for, proper exercise.
Not monitoring key metrics
Scale weight alone tells an incomplete story. Researchers optimizing their protocols should track:
Body composition: Use DEXA scans, bioimpedance devices, or at minimum waist/hip measurements to distinguish fat loss from lean mass loss
Resting heart rate: Particularly important with retatrutide due to the glucagon-mediated heart rate increase
Blood pressure: Both peptides lower blood pressure, which is beneficial but can cause dizziness if the drop is too rapid
Fasting blood glucose: Especially relevant for those managing pre-diabetes or type 2 diabetes
Liver enzymes: For anyone with suspected or confirmed fatty liver disease
Energy levels and sleep quality: Subjective but important indicators of protocol tolerance
The GLP-1 progress tracking tools available through SeekPeptides help members monitor these metrics systematically rather than relying on memory or sporadic tracking.
Expecting linear progress
Weight loss is never a straight line. Stalls happen. Water retention masks fat loss. Hormonal fluctuations create temporary plateaus. This is normal and expected with both tirzepatide and retatrutide.
Typical patterns include:
Rapid initial loss (first 4-6 weeks), much of which is water and glycogen
Steady loss during mid-protocol (weeks 6-24)
Plateaus lasting 1-3 weeks that resolve spontaneously
Slower deceleration as you approach new equilibrium weight
If you hit a plateau on tirzepatide that persists beyond 4-6 weeks at maximum dose, the tirzepatide troubleshooting guide covers the most common causes and solutions. The same problem-solving framework applies to retatrutide, with the additional consideration that glucagon receptor effects on energy expenditure may help break plateaus that dual agonists cannot.
Real-world results vs clinical trial data
Clinical trials represent ideal conditions. Participants are screened, monitored, counseled, and followed up with regularly. Real-world results often differ, and understanding why helps set appropriate expectations.
Why real-world tirzepatide results sometimes fall short
Post-marketing data and patient surveys suggest that average real-world weight loss with tirzepatide is approximately 15-18% at one year, somewhat below the 20.9% seen in SURMOUNT-1 at 72 weeks. Several factors explain this gap:
Adherence drops. In clinical trials, participants receive regular check-ins, free medication, and professional support. In real life, people miss doses, experience insurance denials, face cost barriers, and stop treatment when side effects become inconvenient. Each missed dose reduces cumulative efficacy.
Dose titration varies. Not everyone reaches the maximum 15mg dose. Some people tolerate only 10mg. Others pause at 7.5mg due to persistent nausea. Proper dosing is essential, but real-world titration is often less systematic than clinical trial protocols.
Lifestyle factors are uncontrolled. Clinical trial participants often receive dietary counseling. Real-world users may not optimize nutrition or avoid problematic foods, reducing the magnitude of benefit.
Real-world tirzepatide results still represent extraordinary outcomes compared to older weight loss medications. A 15-18% average weight loss is life-changing for most individuals. But calibrating expectations to real-world rather than trial-level outcomes prevents disappointment.
What real-world retatrutide results might look like
Since retatrutide is not yet commercially available, we cannot assess real-world performance. However, based on patterns seen with tirzepatide and semaglutide, we can estimate that real-world retatrutide results will likely fall 3-5 percentage points below clinical trial averages.
If the Phase 3 average holds around 28-29%, real-world results might land closer to 23-25%. Still extraordinary. Still better than tirzepatide in most comparisons. But the gap between the two compounds may narrow in real-world settings where adherence and dose optimization are less consistent.
Factors specific to retatrutide that might affect real-world outcomes include:
The higher side effect rate could drive more discontinuations
The heart rate increase might concern prescribers and lead to lower maintenance doses
If pricing is higher than tirzepatide, cost barriers could limit access and adherence
The more complex triple-agonist mechanism may require more careful monitoring, adding burden
Conversely, the additional metabolic benefits (liver fat reduction, possibly better muscle preservation) might improve adherence by providing visible results beyond scale weight, motivating continued use.
The broader landscape of GLP-1 medications
Retatrutide and tirzepatide do not exist in a vacuum. They are part of a rapidly expanding class of incretin-based medications that includes several other notable compounds.
Semaglutide (Ozempic/Wegovy) remains the most prescribed single GLP-1 agonist, with 15-17% weight loss at the 2.4mg dose. It serves as the baseline against which newer medications are measured. For many researchers, semaglutide provides sufficient results and remains the most accessible option.
Survodutide is another dual agonist targeting GLP-1 and glucagon receptors (without GIP), currently in Phase 3 trials. Its mechanism sits between tirzepatide and retatrutide in terms of receptor targets, offering a different balance of metabolic effects.
Mazdutide is a dual GLP-1/glucagon agonist developed by Innovent Biologics, primarily studied in Chinese populations, showing weight loss of 14-18% in clinical trials. It provides another data point for the value of glucagon receptor activation.
CagriSema combines cagrilintide (an amylin analog) with semaglutide, taking a fundamentally different approach by targeting amylin pathways rather than glucagon. Phase 3 data shows approximately 22-25% weight loss, making it competitive with retatrutide through a completely different mechanism.
Orforglipron is an oral non-peptide GLP-1 agonist that could solve the injection barrier entirely. While its weight loss is more modest (approximately 14-15%), the convenience of daily oral dosing represents a fundamentally different value proposition.
The competitive landscape means that by the time retatrutide reaches the market, it will face a crowded field. Its triple agonist mechanism and superior weight loss data will need to justify what will likely be premium pricing. For SeekPeptides members tracking this space, understanding where each compound fits helps inform both current and future protocol decisions.
Stacking and combination considerations
Researchers frequently ask about combining these peptides with other compounds. While adding medications to potent GLP-1 agonists requires caution, several combinations are commonly discussed in the research community.
With cagrilintide. The cagrilintide and retatrutide combination is an area of active interest. Cagrilintide targets the amylin receptor, adding another satiety pathway on top of the three that retatrutide already activates. However, combining multiple potent appetite suppressants increases GI side effect risk significantly.
With tesamorelin. Some researchers explore tesamorelin and retatrutide together for body composition optimization. Tesamorelin is a growth hormone releasing hormone analog that can help preserve lean mass during weight loss, potentially addressing one of the main concerns with rapid GLP-1 mediated weight reduction.
With supplemental compounds. Both tirzepatide and retatrutide are commonly paired with B12, glycine, and other compounds in formulations designed to offset potential nutrient depletion from reduced food intake. Tirzepatide with B12 and tirzepatide glycine B12 compounds are widely available through compounding pharmacies.
For researchers interested in optimizing protocols beyond single-compound use, the peptide stack calculator can help evaluate potential combination effects and dosing considerations.
Reconstitution and practical handling
Research-grade peptides require reconstitution before use. The process is identical for both compounds, but the concentrations and volumes differ.
Both tirzepatide and retatrutide come as lyophilized (freeze-dried) powder that must be reconstituted with bacteriostatic water before injection. The peptide reconstitution calculator simplifies volume calculations for any concentration.
For tirzepatide specifically:
Complete reconstitution guide covers step-by-step preparation
Store reconstituted solution in the refrigerator (see fridge storage duration)
Use insulin syringes for accurate dosing (see syringe dosage guide)
Track dosage in units carefully when using compounded formulations
For retatrutide:
Follow the reconstitution chart for precise water volumes
The bacteriostatic water guide for 10mg vials provides specific calculations
Storage requirements are similar, keep reconstituted retatrutide refrigerated
Check the shelf life guidelines for reconstituted peptide stability
The general peptide storage guide covers best practices for maintaining potency, which applies equally to both compounds. Proper storage is not optional. Degraded peptides deliver unpredictable results and can be unsafe.
What the research community is saying
The peptide research community has been buzzing about retatrutide since the Phase 2 data dropped. The consensus, if you can call it that in a space with diverse opinions, clusters around a few key themes.
First, nearly everyone agrees that the weight loss data is remarkable. A 28.7% average at 68 weeks puts retatrutide in territory that only bariatric surgery has previously occupied. For researchers who have tried tirzepatide and plateaued, the possibility of additional efficacy through a new mechanism is genuinely exciting.
Second, the liver fat data has captured attention beyond the weight loss community. Hepatologists and gastroenterologists are watching the TRIUMPH trials with particular interest, as MASLD treatment options are extremely limited. If retatrutide gains approval with a liver disease indication, it could become a blockbuster on that basis alone.
Third, caution about the heart rate signal is widespread. Experienced researchers recognize that a heart rate increase, even a modest one, requires careful long-term characterization. Nobody wants a repeat of the fen-phen era, where cardiovascular signals were dismissed until catastrophic damage occurred.
SeekPeptides provides detailed protocol databases and community discussions for members exploring both compounds. The shared experiences of thousands of researchers offer practical insights that clinical trial data alone cannot provide, from managing side effects during dose escalation to optimizing nutritional strategies for maximum fat loss with minimum muscle wasting.
Nutrition strategies that differ between the two peptides
While the general nutritional advice applies to both compounds, there are specific considerations that differ based on the mechanism of action. Researchers who optimize nutrition for their specific peptide see better results, fewer side effects, and less muscle loss.
Caloric needs shift differently
Tirzepatide reduces caloric intake primarily through appetite suppression. The calories you burn at rest stay relatively stable (with perhaps a small decrease from lower body weight). This means your caloric deficit comes almost entirely from eating less.
Retatrutide, through glucagon receptor activation, also increases the calories you burn. This creates a dual-sided deficit: eating less AND burning more. The practical implication is that retatrutide users may need to be slightly less aggressive with food restriction to avoid an excessive caloric deficit that accelerates lean mass loss.
For tirzepatide users, the diet plan should focus on calorie-dense protein sources to maintain nutrition despite reduced appetite. For retatrutide users, the elevated metabolic rate means slightly higher caloric targets may be appropriate, especially on training days. The food optimization strategies that apply to GLP-1 medications generally are a starting point, but the higher energy expenditure with retatrutide warrants adjustment.
Micronutrient considerations
Both peptides reduce food volume, which inherently reduces micronutrient intake. But retatrutide elevated metabolic rate may increase demand for certain nutrients, particularly B vitamins involved in energy metabolism and magnesium involved in hundreds of enzymatic reactions.
Recommended supplementation for both peptides includes:
Multivitamin: To cover baseline micronutrient gaps from reduced food intake
Vitamin D3: 2,000-4,000 IU daily, as weight loss releases stored vitamin D from fat tissue in unpredictable patterns
Magnesium glycinate: 200-400mg daily, supports muscle function and helps with constipation
Omega-3 fatty acids: 1,000-2,000mg EPA/DHA daily for anti-inflammatory support
B12/methylcobalamin: Especially important if using formulations without added B12
Tirzepatide formulations with B12 and tirzepatide glycine B12 compounds address the B12 need directly. For retatrutide, supplementation must be handled separately since no commercially compounded formulations exist.
Managing GI symptoms through food choices
The foods that trigger GI distress differ slightly between the two peptides. Tirzepatide users commonly report that high-fat foods cause the worst nausea and bloating, because the slowed gastric emptying means fat sits in the stomach longer. Specific trigger foods include fried items, rich sauces, and heavy dairy.
Retatrutide users report similar fat intolerance but may also experience issues with very large meals of any composition, likely because the triple receptor activation provides more potent gastric slowing. Smaller, more frequent meals (4-5 per day rather than 2-3) tend to be better tolerated with retatrutide than with tirzepatide, where 3 moderate meals often work fine.
For both peptides, the golden rule remains the same: prioritize protein, eat slowly, stop when comfortable rather than full, and stay hydrated. The best foods for GLP-1 users emphasize lean proteins, vegetables, and complex carbohydrates that are easy to digest and nutrient-dense.
Frequently asked questions
Is retatrutide stronger than tirzepatide for weight loss?
Based on current clinical trial data, yes. Retatrutide produced 28.7% average weight loss at 12mg in 68 weeks, compared to 20.9% for tirzepatide at 15mg in 72 weeks. The difference is approximately 8 percentage points, which translates to roughly 20 additional pounds lost for a 250-pound individual. However, retatrutide is still in Phase 3 trials and not yet FDA-approved, so direct real-world comparisons are not yet possible. The full TRIUMPH program data, expected to complete this year, will provide a more comprehensive picture of how these two compounds compare across different populations and treatment durations.
Can I switch from tirzepatide to retatrutide?
Yes, but carefully. Start retatrutide at 1mg regardless of your previous tirzepatide dose, allow a 7-10 day washout period after your last tirzepatide injection, and follow the standard retatrutide escalation schedule. The specific transition protocol accounts for receptor adaptation from prior dual agonist use.
Does retatrutide cause more side effects than tirzepatide?
Clinical trial data suggests slightly higher adverse event rates with retatrutide compared to tirzepatide. The most notable additional concern is a greater increase in resting heart rate, likely related to glucagon receptor activation. GI side effects are similar in nature but may be somewhat more frequent. Detailed incretin medication side effect information helps contextualize these differences.
When will retatrutide be FDA approved?
The earliest expected approval is late 2027, following completion of the Phase 3 TRIUMPH program. Seven trials are running, with results expected throughout the remainder of this year. Commercial availability would follow FDA approval, likely in early 2028. Monitor the retatrutide availability page for ongoing updates.
Is retatrutide better for fatty liver disease?
The data strongly suggests yes. Retatrutide reduced liver fat by up to 82% in 24 weeks and achieved normal liver fat levels in 93% of participants on the 12mg dose by 48 weeks. No other GLP-1 medication has demonstrated comparable liver fat clearance. The glucagon receptor activation specifically promotes hepatic fat oxidation in ways that dual agonists do not.
What is the best dose of retatrutide compared to tirzepatide?
The maximum studied doses are 15mg for tirzepatide and 12mg for retatrutide. At these doses, retatrutide produces greater weight loss. However, "best" depends on individual tolerance and goals. Some researchers achieve optimal results at mid-range doses. Use the retatrutide dosage calculator or tirzepatide dosage calculator to determine exact unit measurements for your protocol.
Can I take retatrutide and tirzepatide together?
No. There is no clinical data supporting concurrent use of two incretin-based medications targeting overlapping receptor pathways. The GI side effects and hormonal disruption from combining a dual and triple agonist would be significant and unpredictable. Choose one compound and optimize the protocol for that compound specifically.
Does retatrutide preserve muscle better than tirzepatide?
Early data is promising but not definitive. The glucagon receptor in retatrutide promotes fat oxidation specifically, which theoretically shifts the fat-to-lean-mass loss ratio more favorably. A Phase 2 substudy showed 26.1% fat mass reduction with limited lean mass loss. However, direct DEXA-based comparisons between the two compounds are needed. Regardless of which peptide you use, adequate protein intake and resistance training remain critical for muscle preservation.
How much does retatrutide cost compared to tirzepatide?
Retatrutide does not have commercial pricing yet, as it is still in clinical trials. Research-grade retatrutide typically costs $80-200 per 10mg vial from reputable vendors. Tirzepatide brand-name costs $950-1,100/month, while compounded tirzepatide ranges from $150-400/month. Use the peptide cost calculator to estimate protocol expenses.
External resources
New England Journal of Medicine: Retatrutide Phase 2 Obesity Trial
PMC: Comparative Efficacy of Tirzepatide vs Retatrutide Network Meta-Analysis
Cell Discovery: Structural Insights into Retatrutide Triple Agonism
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members access detailed comparison tools, protocol databases, and expert recommendations for both tirzepatide and retatrutide research.
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your protocols stay optimized, and your results stay consistent.