Mar 18, 2026

What if shortening your injection schedule could actually make tirzepatide less effective? That question deserves a real answer, not the vague warnings most guides offer. Thousands of people taking tirzepatide notice something frustrating around day five or six. The appetite suppression fades. The food noise creeps back. Hunger returns like it never left. And the natural conclusion feels obvious: just inject sooner. Take it every five days instead of seven. Problem solved.
Except it is not that simple.
The pharmacology behind tirzepatide tells a specific story about half-lives, drug accumulation, and therapeutic windows that makes every-five-day dosing genuinely risky. Not risky in the vague, hand-waving way that medical disclaimers usually mean. Risky in the concrete, measurable way that involves pushing drug concentrations above the levels tested in clinical trials, increasing the severity of gastrointestinal side effects, and potentially triggering complications like pancreatitis or acute kidney issues. The gap between a seven-day schedule and a five-day schedule might look small on a calendar. Inside your body, that gap changes everything about how the medication behaves.
This guide covers the actual pharmacokinetics behind tirzepatide dosing, explains why your appetite returns before day seven, walks through the steady-state concept that most resources skip entirely, and gives you practical strategies for managing end-of-week hunger without touching your injection schedule. If you have been staring at your vial on day five wondering whether to inject early, the answer is here, and it is backed by the same clinical data that earned tirzepatide its FDA approval.
The short answer: no, you should not take tirzepatide every 5 days
The direct answer is straightforward. No. Taking tirzepatide every five days instead of the standard seven-day schedule is not recommended by any clinical guideline, any FDA-approved prescribing information, or any peer-reviewed research. Both Mounjaro (for type 2 diabetes) and Zepbound (for weight management) received approval based exclusively on once-weekly dosing protocols. No clinical trial has tested five-day intervals.
That matters more than most people realize.
When a medication receives FDA approval, the dosing schedule is not a suggestion. It reflects the exact parameters under which safety and efficacy were established. Every side effect profile, every weight loss outcome, every blood sugar reduction number in the clinical data assumes weekly administration. Changing the frequency to every five days means operating completely outside the tested parameters, with no data to predict what happens to side effect rates, drug accumulation levels, or long-term safety. The standard tirzepatide dose chart exists for a reason, and that reason is rooted in pharmacokinetic modeling that accounts for the medication half-life, elimination rate, and accumulation ratio.
The minimum interval between any two tirzepatide injections is 72 hours. That rule exists to prevent acute overdose scenarios. But meeting the 72-hour minimum does not mean that taking the medication every five days is safe as a regular schedule. There is a critical difference between an occasional early dose and a permanently shortened cycle, and understanding that difference requires knowing how tirzepatide works in your body over time.
Understanding tirzepatide half-life and what it means for dosing
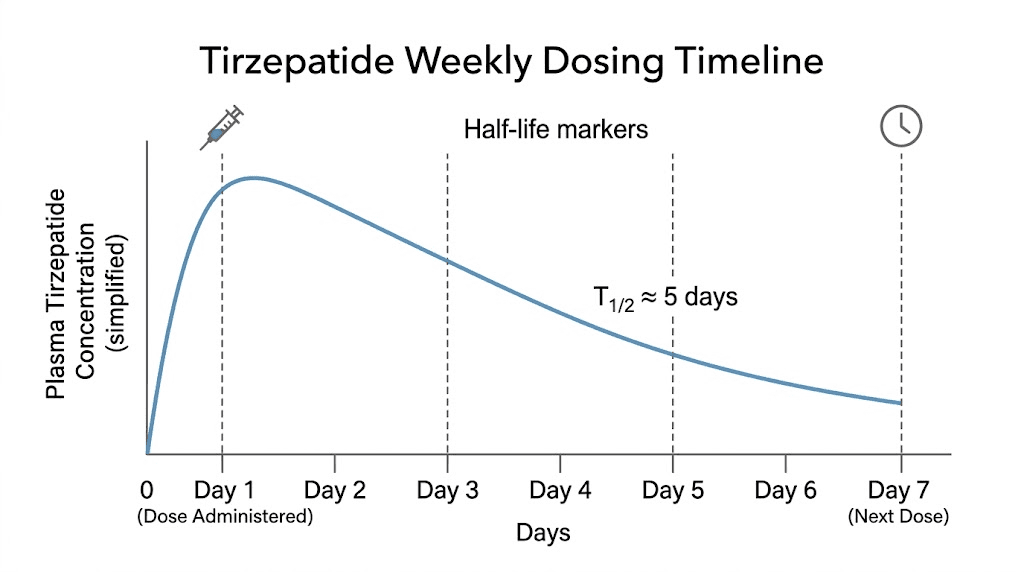
Tirzepatide has a half-life of approximately five days. That number is the foundation for everything about its dosing schedule, and it is also the number that causes the most confusion. People see "five-day half-life" and think the medication is gone after five days. It is not. A half-life means that after five days, half of the original dose remains active in your system. After ten days, one quarter remains. After fifteen days, one eighth.
Here is where the math gets important for the every-five-day question.
When you inject tirzepatide on day one, your body begins absorbing and processing the medication. Peak blood levels typically occur between 8 and 72 hours after injection, depending on the injection site and individual metabolism. From that peak, levels gradually decline according to the 5-day half-life. By day five, roughly half the medication remains. By day seven, when your next dose is scheduled, approximately 37% of the previous dose is still circulating. You inject the new dose on top of that remaining amount, and the cycle continues.
This stacking effect is not a bug. It is the entire design.
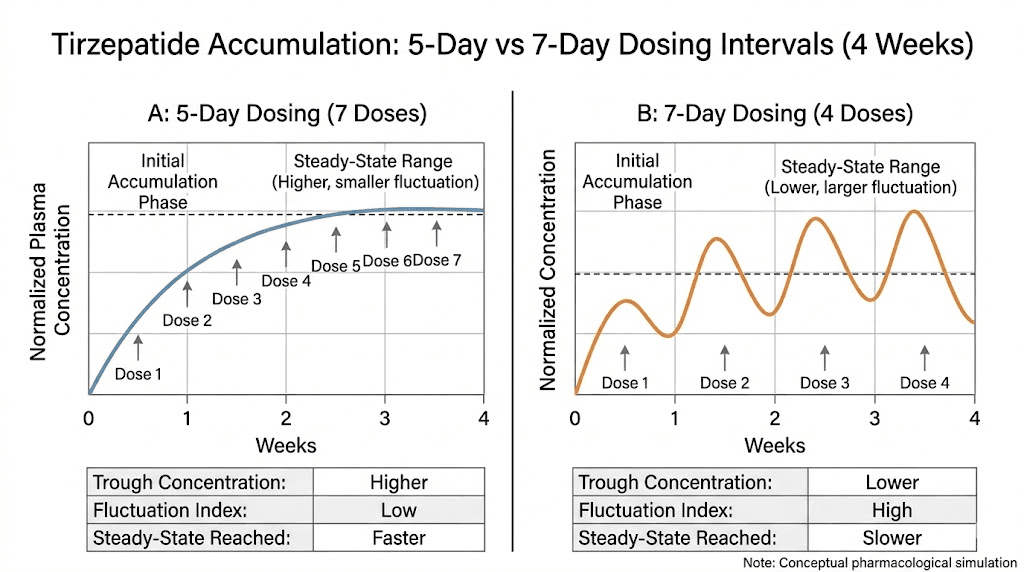
Tirzepatide was engineered to accumulate gradually over multiple weeks until it reaches what pharmacologists call steady state, the point where the amount entering your system each week equals the amount being eliminated. At weekly dosing, steady state occurs at approximately 4 weeks, with an accumulation ratio of about 1.7-fold. That means your steady-state drug levels are roughly 1.7 times the peak level from a single dose.
Now consider what happens at five-day dosing.
At day five, approximately 50% of the previous dose remains, compared to 37% at day seven. That might sound like a small difference. Over multiple cycles, it compounds dramatically. The accumulation ratio at five-day intervals would be significantly higher than 1.7-fold, pushing drug concentrations into territory that has never been studied in humans. Nobody knows the safety profile at those levels because nobody has tested it. The dosage calculations that determine safe therapeutic windows all assume seven-day spacing.
The relationship between half-life and dosing interval is not linear. Shortening the interval by two days does not simply increase drug levels by two-sevenths. It fundamentally changes the accumulation curve, and that change gets amplified with every single injection cycle. By week four of five-day dosing, you could be carrying drug concentrations 20-30% higher than what any clinical trial participant ever experienced. And the side effects of tirzepatide are dose-dependent, which means higher concentrations translate directly to more severe gastrointestinal symptoms, more intense nausea, and a meaningfully higher risk of serious complications.
Why your appetite comes back before day 7
Before explaining what to do about end-of-week hunger, it helps to understand why it happens in the first place. And the reason is surprisingly simple. During the first several weeks of treatment, tirzepatide has not yet reached steady state. Your body is still building up to consistent drug levels, and in the meantime, you experience a predictable weekly pattern that many people find discouraging.
The pattern looks like this for most people starting treatment:
Days 1 through 3: Strong appetite suppression. Minimal food noise. Reduced cravings. This is when tirzepatide blood levels are highest, and the dual GIP and GLP-1 receptor activation is strongest. Most people feel the full effect of their medication during this window, and it can feel almost effortless to eat less.
Days 4 through 5: Gradual return of hunger signals. The suppression effect weakens noticeably. You might find yourself thinking about food more, feeling less satisfied after meals, or noticing that the "I could eat" feeling returns. This is when drug levels have dropped to approximately 50% of peak, and receptor activation becomes inconsistent.
Days 6 through 7: Hunger returns more noticeably. Some people describe this as the "Mounjaro munchies," and it can feel like the medication stopped working entirely. Drug levels are at their lowest point in the cycle, sitting around 37% of peak. For people on lower doses like the starting 2.5 mg dose, this trough can drop below the threshold needed for meaningful appetite suppression.
This weekly fluctuation is completely normal. It does not mean the medication is failing. It does not mean you need a higher dose immediately. And it absolutely does not mean you should inject more frequently. The wearing-off pattern is most pronounced during weeks one through four, before steady state is achieved. Once your body reaches steady state, the difference between peak and trough levels narrows considerably, and the end-of-week hunger effect diminishes or disappears entirely for most people.
Understanding this timeline is essential because it reframes the problem. The question is not "how do I prevent tirzepatide from wearing off?" The question is "how do I manage the first four weeks while steady state builds?" Those are very different questions with very different answers. One leads to dangerous dosing changes. The other leads to practical strategies that work with the medication pharmacology instead of against it.
The steady state concept most guides skip entirely
Steady state is the single most important concept for understanding tirzepatide dosing, and most patient-facing resources barely mention it. Here is what it means in plain language.
When you start any medication with a long half-life, the first several doses are building toward a stable baseline. Think of it like filling a bathtub with a slow drain. Each week, you add water (your dose). Each week, some water drains out (your body eliminates the drug). For the first several weeks, the tub is filling faster than it is draining. Eventually, the inflow and outflow equalize. That is steady state.
For tirzepatide specifically, the pharmacokinetic data shows the following timeline. During weeks one through two, you experience the most dramatic peaks and troughs. Drug levels swing significantly between injection day and day seven. This is when the wearing-off effect is strongest, when hunger fluctuations are most noticeable, and when the temptation to dose more frequently feels most compelling. Weeks three through four bring gradual improvement as drug levels build. The troughs are not as deep. The peaks are slightly higher. The difference between day one and day seven starts narrowing. By weeks five through eight, most people have achieved true steady state. Drug levels at the end of the week are substantially higher than they were during week one, even without any dose change, simply because of accumulation.
This means the medication is literally designed to get more effective over time at the same dose.
The standard tirzepatide dosing protocol accounts for this by keeping patients at each dose level for a minimum of four weeks before considering escalation. That four-week minimum is not arbitrary. It takes approximately four to five half-lives to reach steady state, and with a five-day half-life, that translates to 20-25 days. The four-week mark ensures that the drug has reached its full potential at the current dose before any decision is made about increasing it.
People who take tirzepatide every five days short-circuit this entire process. They push accumulation higher and faster than the pharmacokinetic models predict, and they lose the ability to distinguish between a dose that needs escalation and a dose that simply has not reached steady state yet. That distinction matters enormously for long-term treatment success. If you escalate too quickly because you misread the early wearing-off effect as a sign that the dose is insufficient, you burn through the available dose range faster and potentially reach the maximum dose before your body has fully adapted to lower levels.
SeekPeptides members access detailed pharmacokinetic guides that explain steady-state timelines for every GLP-1 receptor agonist, helping researchers understand exactly when to expect consistent results from their protocols.
What drug accumulation actually does to your body
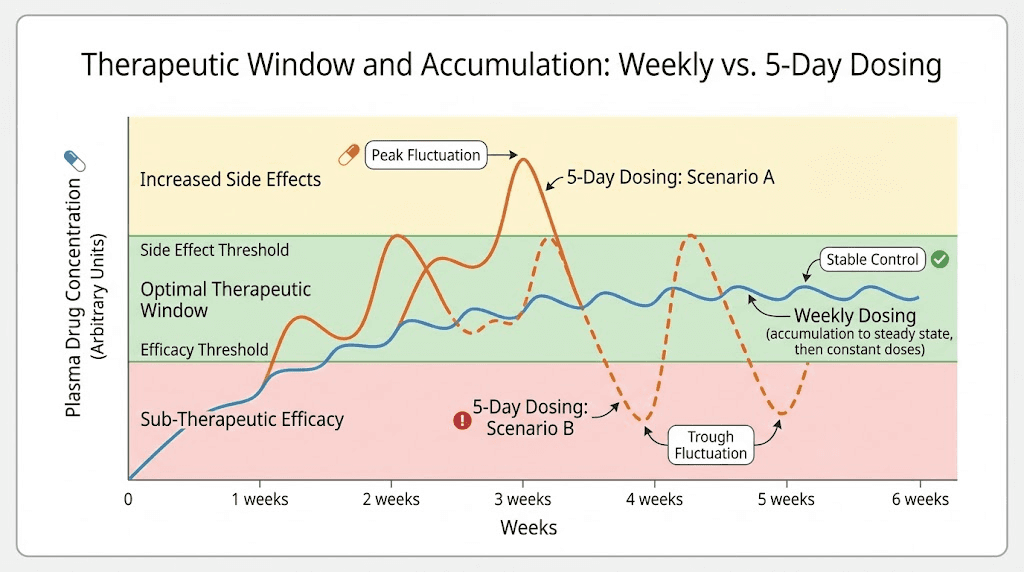
Every medication has a therapeutic window. That window represents the range of drug concentrations where the medication provides its intended benefits without causing excessive harm. Below the window, the drug does not work. Above the window, side effects increase without proportional benefit. The tirzepatide dose escalation schedule was designed to keep patients within that therapeutic window.
Five-day dosing pushes concentrations above the tested therapeutic window.
The consequences of exceeding the therapeutic window are not theoretical. They are predictable, dose-dependent, and well-documented from clinical trial dose-response data. At higher concentrations, tirzepatide GI side effects intensify across every category. Nausea becomes more frequent and more severe. The incidence of vomiting increases. Diarrhea episodes become more common and more prolonged. Constipation affects a larger percentage of patients. Abdominal bloating and discomfort worsen.
These are not minor inconveniences. Severe GI side effects from GLP-1 receptor agonists can lead to dehydration, electrolyte imbalances, and in extreme cases, hospitalization. The clinical trials for tirzepatide reported that GI side effects were the primary reason patients discontinued treatment, and those rates increased at higher doses. By exceeding the tested concentration range through more frequent dosing, you effectively expose yourself to the side effect profile of a dose higher than what you are actually injecting.
Beyond GI effects, elevated drug concentrations raise the risk of more serious complications. Pancreatitis risk increases with higher GLP-1 receptor agonist exposure. The mechanism involves excessive stimulation of pancreatic cells, leading to inflammation that can range from mild to life-threatening. Gallbladder complications including cholecystitis and cholelithiasis also show dose-dependent incidence. Acute kidney injury, often secondary to severe dehydration from persistent nausea and vomiting, becomes more likely when drug levels exceed the tested range.
None of this means tirzepatide is a dangerous medication at its approved doses and schedule. The safety profile at weekly dosing is well-established and favorable for most patients.
But that safety profile was established at weekly intervals, not five-day intervals. Changing the frequency changes the equation entirely, and the risks scale disproportionately with the concentration increase.
Side effects that intensify with more frequent dosing
Understanding exactly which side effects worsen with higher drug concentrations helps illustrate why the every-five-day approach carries real medical risk. The side effects of tirzepatide are well-documented from the SURMOUNT and SURPASS clinical trial programs, and the dose-response relationship is clear across nearly every adverse event category.
Gastrointestinal effects
Nausea is the most common side effect of tirzepatide, affecting 12-33% of patients depending on dose. At 5 mg weekly, nausea rates sit around 12-15%. At 10 mg, they climb to 17-22%. At 15 mg, they reach 24-33%. These percentages come from weekly dosing. Five-day dosing at any given milligram amount would produce effective drug exposure comparable to a higher milligram dose at weekly intervals. Someone taking 5 mg every five days could experience nausea rates closer to the 10 mg weekly profile, without ever intending to take 10 mg.
The same pattern applies to vomiting, diarrhea, and abdominal pain. The SURMOUNT-1 trial specifically noted that GI adverse events were the most common reason for treatment discontinuation at higher doses. Patients who experienced treatment-limiting nausea or vomiting at 15 mg weekly had drug concentrations that, in some cases, overlapped with what five-day dosing at 10 mg would produce. The overlap matters because it means you could trigger the same discontinuation-level side effects at a lower nominal dose simply by injecting more frequently.
Fatigue and energy effects
Tiredness and fatigue are reported by a significant minority of tirzepatide users, and these effects correlate with drug exposure levels. Higher trough concentrations, the kind produced by five-day dosing, can extend and intensify the fatigue effect. Some researchers report that fatigue becomes persistent rather than cyclical at elevated drug levels, affecting daily function and exercise capacity. Given that physical activity plays a key role in weight loss outcomes during the tirzepatide weight loss journey, persistent fatigue could actually undermine the very goal that prompted the frequency increase.
Neurological and psychological effects
Headaches, insomnia, and anxiety have all been reported as side effects with varying frequency. While the exact dose-response relationship for these effects is less clearly established than for GI symptoms, clinical pharmacology suggests that any side effect mediated by GLP-1 and GIP receptor activation could intensify with higher drug exposure. Dizziness is another reported effect that could worsen with elevated concentrations.
Musculoskeletal effects
Muscle pain, body aches, and joint pain are less commonly discussed but documented side effects. At higher effective drug exposure, these musculoskeletal symptoms could become more frequent or more intense, further reducing quality of life and exercise tolerance.
Reproductive and hormonal effects
Tirzepatide can affect menstrual cycles, and higher drug exposure could potentially amplify these disruptions. For women of reproductive age, understanding the interaction between tirzepatide and reproductive health becomes even more important when drug levels exceed tested ranges. The increased absorption of oral contraceptives during GLP-1 use is another factor that demands attention at modified dosing schedules.
What the clinical research actually studied
The evidence base for tirzepatide comes from two major clinical trial programs. SURPASS studied tirzepatide for type 2 diabetes management across multiple trials. SURMOUNT studied tirzepatide for weight management. Both programs exclusively used once-weekly dosing. No trial arm, no sub-study, no exploratory analysis tested five-day dosing intervals.
This absence of data is itself the strongest argument against shorter dosing intervals.
In SURMOUNT-1, which enrolled 2,539 adults with obesity, participants received tirzepatide at 5 mg, 10 mg, or 15 mg once weekly for 72 weeks. The results were remarkable. The 15 mg group achieved mean weight loss of 22.5% from baseline. The 10 mg group achieved 21.4%. Even the 5 mg group achieved 16.0%. These numbers far exceeded what previous GLP-1 receptor agonists had achieved, and they were all produced by weekly dosing.
The SURPASS trials for diabetes showed similar patterns. Tirzepatide at 5, 10, and 15 mg weekly produced superior HbA1c reductions compared to both placebo and active comparators including semaglutide. Again, all weekly. The population pharmacokinetic analysis published by Schneck et al. in CPT: Pharmacometrics and Systems Pharmacology confirmed that the 5-day half-life, combined with weekly dosing, produces the accumulation ratio and steady-state concentrations that support these outcomes.
Some people interpret the strong results as evidence that more frequent dosing would produce even better results. The pharmacology does not support that interpretation. Weight loss outcomes in the clinical trials showed diminishing returns between the 10 mg and 15 mg dose levels, with the jump from 10 to 15 mg producing a much smaller incremental benefit than the jump from 5 to 10 mg. This plateau effect suggests that pushing drug levels higher does not proportionally improve outcomes, while the dose-response data for side effects shows that adverse events continue climbing at higher exposures.
More medication does not equal better results. It equals more side effects.
The 72-hour minimum rule explained
The prescribing information for both Mounjaro and Zepbound states that if a dose is missed, the next dose should be administered as soon as possible within 96 hours (4 days) of the missed dose. If more than 96 hours have passed, skip the missed dose and resume the regular weekly schedule. The information also states that the injection day can be changed, as long as at least 72 hours have passed since the last injection.
Some people misread this as permission to dose every three days if they want. It is not.
The 72-hour rule is a safety floor for occasional schedule adjustments, not a minimum dosing interval for regular use. The scenario it addresses is specific: you normally inject on Wednesday, but next week you have a conflict and want to switch to Friday. As long as 72 hours have passed since Wednesday injection, Friday is acceptable. This is about taking tirzepatide a day or two early as a one-time adjustment, not about permanently shortening your cycle.
The distinction between an occasional early dose and a regular five-day schedule is enormous from a pharmacokinetic perspective. An occasional early dose, followed by returning to the weekly schedule, produces a temporary bump in drug levels that normalizes within one or two cycles. A permanent five-day schedule produces continuously elevated drug levels that compound with every injection, pushing further above the tested range with each cycle until a new, higher steady state is reached.
If your question is "can I take my tirzepatide on day five this one time because of a travel schedule," the answer is likely yes, provided 72 hours have passed, and you return to your regular weekly schedule afterward. Consult your prescriber for confirmation. If your question is "can I permanently switch to every-five-day dosing," the answer is no. These are fundamentally different questions with fundamentally different risk profiles, and conflating them is one of the most common mistakes people make when researching tirzepatide timing flexibility.
Taking tirzepatide early once vs. every 5 days permanently
Because this distinction is so important, it deserves its own section with concrete examples.
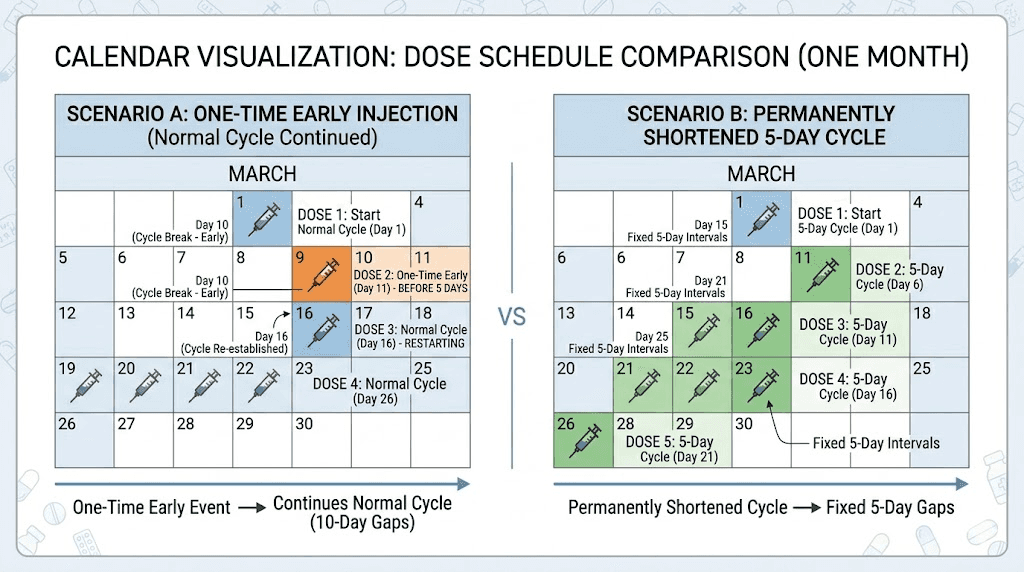
Scenario A: One-time early dose. You normally inject on Mondays. This week, you are traveling Monday and want to inject on Saturday instead. Saturday is day five of your cycle. At least 72 hours have passed since your last Monday injection. You inject on Saturday and return to Monday the following week. The gap between Saturday and the next Monday is nine days, which allows drug levels to normalize. Your steady-state concentrations experience a minor, temporary fluctuation. No meaningful clinical difference in safety or efficacy.
Scenario B: Permanent five-day schedule. You decide to inject every five days because you feel the medication wearing off. Your injection days become Monday, Saturday, Thursday, Tuesday, Sunday, and so on, cycling through the week. Every single dose is administered on top of a higher residual drug concentration than weekly dosing would produce. By week four, your steady-state concentration is approximately 20-30% higher than what clinical trials tested. By week eight, the elevated baseline is fully established, and every side effect is proportionally more likely and more severe.
The difference is not subtle. One is a schedule accommodation with negligible pharmacokinetic impact. The other is a fundamental protocol change with significant safety implications. People who start with Scenario A, making occasional adjustments, sometimes drift into Scenario B without realizing it. They inject early one week, then start doing it every other week, then every week. The gradual shift means they never experience a single dramatic increase in side effects. Instead, they notice a slow worsening of nausea, increased constipation, more headaches, and they attribute it to the medication rather than to their modified schedule.
Tracking your actual injection dates is essential. If you notice that your injection intervals have been creeping shorter over time, that is a signal to return to strict weekly dosing and discuss dose adjustment with your prescriber instead of modifying your injection day.
What to do when tirzepatide wears off before day 7
Now for the practical part. If injecting more frequently is not the answer, what is? The strategies below address the root causes of end-of-week appetite return and work with the medication pharmacology rather than against it.
Wait for steady state
This is the most important and most frequently ignored strategy. If you are in your first four weeks on a new dose, the wearing-off effect is expected and temporary. Your body is still building drug levels. Each week, your trough concentration is higher than the previous week. By week four, the end-of-week experience will be significantly different from week one, even at the same dose.
Patience sounds unsatisfying as medical advice. But in this case, it is the pharmacologically correct answer. The medication was designed to reach its full potential over four weeks. Evaluating its effectiveness before that point is like judging a marathon runner at mile three. The data on how fast tirzepatide works consistently shows that meaningful, consistent appetite suppression takes several weeks to establish.
Optimize protein intake
Protein is the most satiating macronutrient, and its hunger-suppressing effects complement tirzepatide mechanism of action. When drug levels are at their weekly trough, protein becomes your primary tool for managing appetite. Aim for 30-40 grams of protein per meal, with particular emphasis on days five through seven of your injection cycle.
High-protein foods that pair well with tirzepatide include lean meats, fish, eggs, Greek yogurt, cottage cheese, and legumes. The complete guide to eating on tirzepatide covers specific meal strategies in detail, including how to structure meals when appetite is low early in the week and higher later. Meal planning for tirzepatide users can make a significant difference in managing end-of-week hunger without increasing dose frequency.
Increase fiber strategically
Soluble fiber slows gastric emptying and promotes satiety through mechanisms that stack with tirzepatide effects. Adding a fiber supplement or increasing whole grain, vegetable, and legume intake on days five through seven can meaningfully reduce the hunger rebound. The foods to avoid on tirzepatide are just as important to know as the foods to emphasize, since certain high-fat or high-sugar foods can worsen GI symptoms while providing minimal satiety.
Time your meals differently on end-of-week days
Some people find that switching from three meals to four or five smaller meals on days six and seven helps manage the appetite return. Smaller, more frequent meals maintain more consistent blood sugar levels and prevent the extreme hunger that can follow a long gap between meals. This is not about eating more total food. It is about distributing the same calories across more eating occasions.
Stay hydrated
Dehydration can mimic hunger signals. Many tirzepatide users report that drinking 16-20 ounces of water when hunger strikes on end-of-week days reduces the intensity of cravings. The tirzepatide diet planning guide emphasizes hydration as a foundational strategy that supports both appetite management and the prevention of GI side effects like constipation.
Use physical activity as an appetite management tool
Moderate exercise, particularly walking and resistance training, can temporarily suppress appetite through its effects on hunger hormones. Scheduling your most vigorous workout for day six or seven, when drug levels are lowest, can help bridge the gap until your next injection. Exercise also supports lean mass preservation during weight loss, which is critical for long-term metabolic health and a key factor in sustaining tirzepatide weight loss results.
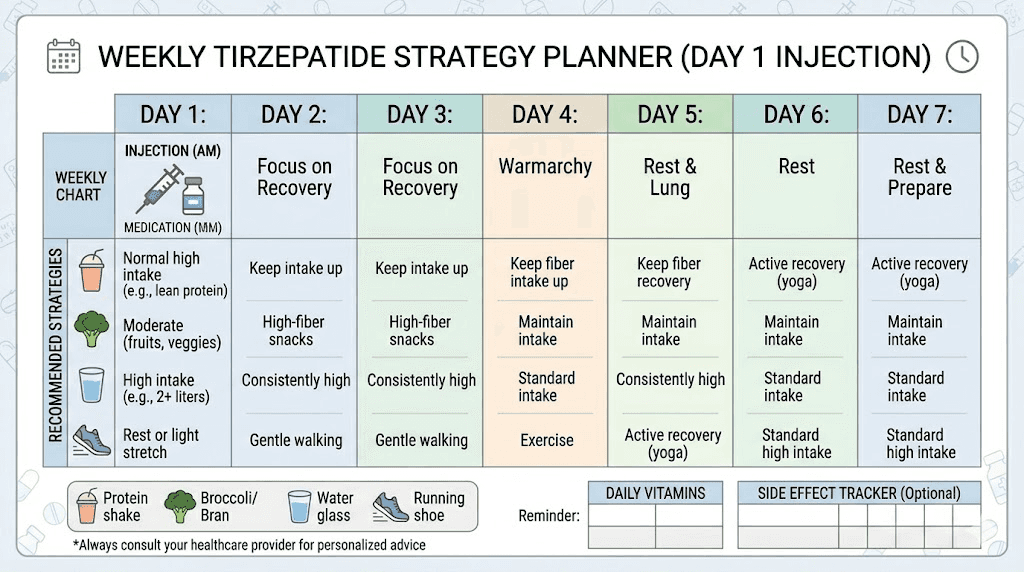
Dose titration: the proper solution for wearing-off effects
If you have been on your current dose for four or more weeks and still experience significant wearing-off before day seven, the appropriate response is dose titration, not frequency modification. The tirzepatide dose escalation protocol provides clear steps for moving from one dose level to the next.
The standard escalation schedule starts at 2.5 mg weekly for four weeks. This starting dose is not intended for meaningful appetite suppression in most people. It is a tolerability dose, designed to let your body adapt to the medication while minimizing GI side effects. After four weeks, you move to 5 mg. If additional appetite suppression is needed after another four weeks, you can escalate to 7.5 mg, then 10 mg, then 12.5 mg, and finally 15 mg as the maximum dose.
Each escalation step should be maintained for a minimum of four weeks before evaluating whether further increases are needed. This four-week window aligns perfectly with the steady-state timeline. By the time you are ready to assess whether a dose is sufficient, you are seeing its full pharmacokinetic potential, not just the first-week impression.
When to escalate versus when to stay depends on several factors. If weight loss has stalled for four or more weeks at steady state, escalation may be appropriate. If appetite suppression is inconsistent throughout the week despite having reached steady state, the current dose may be insufficient. If you are losing 0.5-1 kg per week, however, the current dose is working as intended and escalation is unnecessary even if some end-of-week hunger returns. Not every return of appetite signals a dose problem. Some hunger is normal and healthy, and learning to manage it with food strategies strengthens long-term success after eventually discontinuing the medication.
Understanding your specific unit conversion for your current dose helps you communicate clearly with your prescriber when discussing escalation. Whether you measure in units for 5 mg, 7.5 mg, 10 mg, or 15 mg, precision in dosing conversations ensures you and your provider are aligned on the right next step.
Split dosing: a different conversation entirely
Some prescribers have explored the concept of split dosing, where the total weekly dose is divided into two smaller injections given on different days. For example, instead of 10 mg on Monday, a patient might take 5 mg on Monday and 5 mg on Thursday. This is a fundamentally different approach from taking the full dose every five days, and it is important to understand why.
Split dosing maintains the same total weekly drug exposure. The total amount of tirzepatide entering your system each week is identical. What changes is the distribution. Instead of one large peak followed by a gradual decline, split dosing produces two smaller peaks with shallower troughs. The theoretical benefit is more consistent appetite suppression throughout the week without the end-of-week wearing-off effect.
Some compounding pharmacies and telehealth providers have adopted split dosing protocols for GLP-1 medications, though it is important to note that this approach has not been studied in formal clinical trials for tirzepatide. The distinction between split dosing and five-day dosing is critical. Split dosing keeps total weekly exposure the same. Five-day dosing increases total weekly exposure by approximately 40%. One is a distribution change. The other is a dose increase disguised as a schedule change.
If you are interested in split dosing, this is a conversation to have with your prescriber. It requires adjusting the amount per injection, recalculating your syringe measurements, and monitoring for any changes in side effects or efficacy. It should never be attempted without medical guidance, particularly for compounded formulations where concentration variability is already a consideration. The reconstitution process and mixing calculations must be precise to ensure each split dose delivers the intended amount.
How tirzepatide compares to other GLP-1 medications on dosing flexibility
Understanding how tirzepatide dosing compares to other medications in its class provides useful context for the every-five-day question. Different GLP-1 receptor agonists have different half-lives, and those differences directly affect how much dosing flexibility each medication allows.
Semaglutide, the active ingredient in Ozempic and Wegovy, has a half-life of approximately seven days, which is two days longer than tirzepatide. This longer half-life means semaglutide maintains more consistent blood levels throughout the weekly dosing interval, and the wearing-off effect tends to be less pronounced. The semaglutide versus tirzepatide comparison reveals important differences in how each medication handles the peak-to-trough cycle. People who experienced significant wearing-off on semaglutide often find the effect more noticeable after switching to tirzepatide, not because tirzepatide is less effective, but because its shorter half-life creates a wider gap between peak and trough levels.
The side effect comparison between semaglutide and tirzepatide also shows relevant differences. Tirzepatide tends to produce more GI side effects at equivalent weight loss levels, which is another reason why pushing drug concentrations higher through five-day dosing is particularly risky with this medication compared to its competitors.
Retatrutide, a newer triple agonist targeting GIP, GLP-1, and glucagon receptors, has a half-life of approximately six days. This positions it between semaglutide and tirzepatide in terms of dosing interval sensitivity. For those researching the next generation of weight management peptides, understanding the relationship between half-life and dosing flexibility becomes increasingly important. The retatrutide versus semaglutide comparison and the broader landscape of tirzepatide alternatives provide context for evaluating which medication best suits individual needs.
For people currently on tirzepatide who find the weekly wearing-off effect unmanageable even after dose optimization, switching to semaglutide is sometimes discussed as an option. The longer semaglutide half-life provides more consistent coverage across the seven-day interval. However, tirzepatide generally produces superior weight loss outcomes, so the decision involves weighing consistency against efficacy. The conversion chart between semaglutide and tirzepatide helps patients and providers navigate this transition if it becomes necessary.
Supplements and compounds that may help with end-of-week hunger
Some tirzepatide users find that specific supplements help bridge the gap between the wearing-off effect and their next injection. While these should never replace proper dosing decisions, they can serve as supportive tools in a comprehensive weight management protocol.
Berberine has glucose-modulating properties that may complement tirzepatide effects on blood sugar regulation and appetite. Some researchers report that berberine helps manage the carbohydrate cravings that can intensify during end-of-week hunger. However, combining supplements with prescription medications always warrants a conversation with your prescriber, particularly given potential interactions with glucose-lowering medications.
Vitamin B12 is commonly co-formulated with compounded tirzepatide for good reason. B12 supports energy metabolism and may help address the fatigue that some people experience, particularly toward the end of the dosing week when drug levels are lowest. The tirzepatide with glycine and B12 compound guide covers the rationale behind these combination formulations and how they may support overall treatment outcomes.
Glycine is another compound frequently paired with tirzepatide in compounded formulations. It plays roles in neurotransmitter function, sleep quality, and overall metabolic health. Some users report that tirzepatide formulations containing glycine provide a smoother experience throughout the dosing week, though controlled data specifically comparing glycine-inclusive versus glycine-free formulations is limited.
Niacinamide (vitamin B3) has been added to some compounded tirzepatide formulations as well. Its anti-inflammatory properties and role in cellular energy production make it a theoretically reasonable addition, and some providers include it as standard in their compounding protocols.
The complete guide to supplements with tirzepatide covers additional options including probiotics, magnesium, and fiber supplements. SeekPeptides members access detailed interaction guides and evidence-based supplement stacking protocols designed specifically for people using GLP-1 receptor agonists.
Injection technique and timing optimization
While not directly related to the five-day question, optimizing your injection technique and timing can meaningfully affect how long each dose feels effective. Small changes in how and when you inject can extend the perceived duration of each dose without changing the actual frequency.
The best time of day to inject tirzepatide varies by individual preference and schedule, but many people find that evening injections (between 6-8 PM) provide the strongest appetite suppression during the following day, when willpower tends to be lowest. Morning injectors sometimes report that the peak effect occurs while they are sleeping and they miss out on some of the subjective benefit during waking hours.
Choosing the right day for your tirzepatide injection also matters. If weekends are your hardest days for food management, injecting on Friday evening means peak drug levels coincide with Saturday and Sunday. If weekday lunches with colleagues are your biggest challenge, injecting earlier in the week puts peak suppression where you need it most.
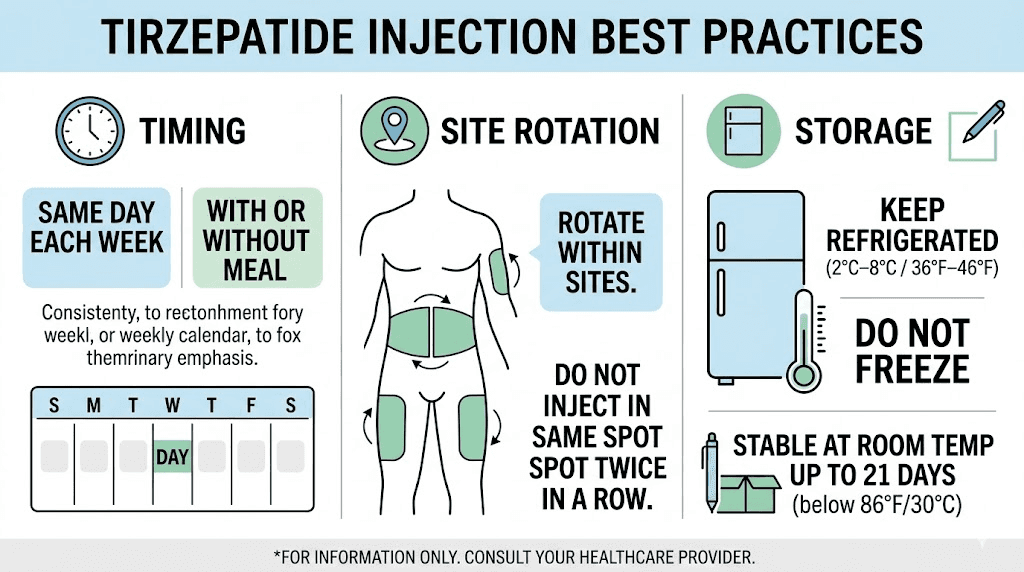
Proper injection technique ensures consistent absorption from dose to dose. Injecting into areas with adequate subcutaneous fat, rotating injection sites to prevent tissue irritation, and avoiding injection site reactions all contribute to more predictable drug delivery. Some people report that inconsistent injection technique, such as accidentally injecting into muscle rather than subcutaneous tissue, can alter how quickly the drug absorbs and how long each dose feels effective.
For those using compounded tirzepatide, proper reconstitution is equally important. Incorrect mixing can result in doses that are weaker or stronger than intended, which directly affects how long appetite suppression lasts. Using the correct amount of bacteriostatic water and measuring doses with the appropriate syringe type and technique ensures that every injection delivers exactly what you intend.
Storage also affects potency. Tirzepatide that has been exposed to heat, left unrefrigerated too long, or stored past its expiration date may deliver reduced effectiveness, making it feel like the medication wears off faster. Checking whether your tirzepatide requires refrigeration, understanding storage duration limits, and knowing how long it can safely be unrefrigerated all protect the potency that keeps each dose working through day seven.
When to talk to your provider about dosing concerns
Certain situations warrant a direct conversation with your prescriber rather than independent troubleshooting. Knowing when to escalate from self-management strategies to medical guidance is an important part of safe tirzepatide use.
Contact your provider if the wearing-off effect persists beyond six to eight weeks at the same dose. By that point, steady state should be well-established, and continued significant appetite return before day seven suggests the dose may genuinely be insufficient for your metabolism and body composition. Your provider can evaluate whether dose escalation is appropriate and determine the right next step within the titration schedule.
Contact your provider if you are experiencing stalled weight loss despite dietary adherence. Sometimes the issue is not the dosing frequency but rather metabolic adaptation, thyroid function changes, or other factors that require clinical evaluation. The troubleshooting guide for tirzepatide weight loss plateaus covers common causes, but a prescriber can run bloodwork and assess factors that self-evaluation cannot.
Contact your provider if you have been tempted to adjust the dosing frequency on your own. This is not about judgment. It is about safety. Providers can discuss options you may not have considered, including split dosing protocols, combination approaches involving metformin or phentermine as adjuncts, dose timing optimization, or even switching to a medication with a longer half-life that provides more consistent weekly coverage.
Contact your provider immediately if you have already been taking tirzepatide more frequently than weekly and are experiencing increased side effects. Honestly reporting your actual dosing pattern allows your provider to assess your current drug levels, adjust your plan, and ensure that no complications are developing. There is no benefit to hiding frequency modifications from the person responsible for your medical safety.
Tracking your tirzepatide experience week by week
One of the most useful things you can do, both for your own understanding and for productive conversations with your provider, is track your tirzepatide experience systematically throughout each dosing week. Simple tracking reveals patterns that subjective memory often distorts.
Record three things daily: hunger level on a 1-10 scale, total food intake (even rough estimates help), and any side effects. After four weeks, patterns emerge. You might discover that the wearing-off effect is less dramatic than it felt in the moment. Or you might find clear evidence that your current dose is insufficient, giving you concrete data to bring to your escalation discussion.
Tracking also helps distinguish between true hunger (a physiological signal that the medication is no longer suppressing appetite) and habitual eating patterns, emotional eating, or environmental triggers. Many people discover that their day-six hunger is less about drug levels and more about returning to social eating situations, stress responses, or conditioned mealtime habits. These factors do not respond to higher drug doses or more frequent injections.
They respond to behavioral strategies that the first month on tirzepatide provides an ideal opportunity to develop.
The before-and-after results from tirzepatide users consistently show that the best outcomes come from people who combine the medication with behavioral changes, not from people who chase maximum drug exposure. The medication creates a window of reduced appetite. What you do inside that window determines your long-term success.
For researchers looking to optimize every aspect of their protocol, SeekPeptides provides comprehensive tracking tools, protocol databases, and a community of experienced users who have navigated these exact dosing questions.
The role of injection day and timing in managing the weekly cycle
Before resorting to any schedule modification, consider whether optimizing your injection timing within the standard weekly framework could address the wearing-off problem. Most people choose their injection day based on convenience, the day their prescription was first filled or the day they happened to start. Few consider the strategic implications of that choice.
Think about which days of the week present the greatest appetite management challenges for you. For many people, weekends involve more social eating, less structured meals, and greater exposure to high-calorie environments. If weekends are your hardest days, injecting on Thursday or Friday evening ensures peak drug levels during Saturday and Sunday, when you need the most support. The wearing-off period then falls on Tuesday or Wednesday, when work routines and structured schedules make appetite management easier even without maximum pharmaceutical support.
Conversely, if your weekday work environment involves frequent lunch meetings, office snacks, and stress-driven eating, a Monday morning injection puts peak suppression right where you need it. The wearing-off period shifts to the weekend, when you might have more control over your food environment and can implement the dietary strategies discussed earlier.
This strategic approach to injection timing can make the difference between feeling like tirzepatide works for five days and feeling like it works for the full seven. The drug levels are identical regardless of which day you inject. What changes is how those levels align with your real-world appetite challenges. For a detailed analysis of timing optimization, the complete guide to tirzepatide injection timing covers morning versus evening injection, the relationship between injection timing and nighttime dosing effects, and how to find the schedule that works best for your lifestyle.
Common misconceptions about tirzepatide dosing frequency
Several myths persist about tirzepatide dosing that deserve direct correction.
"If the half-life is 5 days, the medication is gone after 5 days"
False. The half-life means half the medication remains after five days. After ten days, 25% remains. After fifteen days, 12.5% remains. Complete elimination takes approximately 25-30 days (five to six half-lives). This is why tirzepatide effects on metabolism and appetite persist well beyond the five-day mark.
"More frequent dosing means faster weight loss"
Not supported by evidence. The dose-response relationship for weight loss with tirzepatide shows diminishing returns at higher exposures. The jump from 5 mg to 10 mg weekly produces roughly 5 additional percentage points of weight loss. The jump from 10 mg to 15 mg produces roughly 1-2 additional points. Pushing beyond 15 mg equivalent exposure (which five-day dosing effectively does) would likely produce minimal additional weight loss while substantially increasing side effects.
"My body metabolizes it faster than average"
Population pharmacokinetic analysis shows that tirzepatide half-life is remarkably consistent across different body weights, ages, and demographics. The FDA Clinical Pharmacology Review states that dose adjustment based on demographics or subpopulations is unnecessary. While individual variation exists, it is not large enough to justify a two-day schedule reduction. If you genuinely metabolize the drug faster, the appropriate response is dose escalation, not frequency increase.
"Compounded tirzepatide wears off faster than brand-name"
The active molecule is the same regardless of source. However, compounded formulations can vary in potency if the compounding pharmacy lacks rigorous quality control. If your compounded tirzepatide consistently feels less effective, the issue is more likely potency accuracy than metabolic speed. Choosing a reputable compounding pharmacy and ensuring proper storage and handling addresses this concern directly.
"I can take it every 5 days at a lower dose to keep the same weekly total"
This is essentially split dosing with a different schedule, and it still changes the pharmacokinetic profile. Even if the total weekly milligrams are similar, the peak-to-trough dynamics differ from what was tested in trials. This approach should only be considered under direct medical supervision, never as self-directed modification.
Long-term perspective: building sustainable habits during treatment
The question "can I take tirzepatide every 5 days" often comes from a mindset that prioritizes maximum suppression over sustainable change. And that mindset, however understandable, can undermine long-term success.
Tirzepatide is not a permanent medication for most people. Whether you are using brand-name Mounjaro, Zepbound, or a compounded version, the goal is typically to reach a target weight and eventually taper off or maintain on a lower dose. What happens after you stop depends entirely on the habits you built during treatment.
The end-of-week appetite return is actually a training opportunity. Those days when drug levels are lowest and hunger returns give you a chance to practice the food management skills that will matter most after treatment. Choosing high-protein meals, managing portions without pharmaceutical help, navigating social eating situations, and tolerating mild hunger without reactive overeating are all skills that get stronger with practice. If you eliminate every moment of hunger by dosing more frequently, you lose the practice window that builds post-treatment resilience.
The men who achieve the best tirzepatide results and the women who maintain their weight loss long-term consistently report that behavioral changes, not maximum suppression, determined their lasting success. Using the medication as a tool for building better habits rather than a crutch for avoiding all hunger produces fundamentally different outcomes.
This is where SeekPeptides provides particular value. Members access not just dosing protocols and pharmacokinetic data, but comprehensive lifestyle integration guides, community support from people at every stage of their weight management journey, and evidence-based strategies for maintaining results after treatment concludes.
How dual GIP and GLP-1 receptor activation shapes the dosing timeline
Tirzepatide is not just another GLP-1 receptor agonist. It is a dual agonist that activates both the GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptors simultaneously. This dual mechanism is what makes tirzepatide more effective than semaglutide for weight loss, but it also creates specific pharmacodynamic considerations that make dosing frequency modifications riskier.
GLP-1 receptor activation slows gastric emptying, reduces appetite through hypothalamic signaling, and enhances insulin secretion in response to food. These effects are well-understood from years of research on medications like semaglutide and liraglutide. GIP receptor activation adds complementary effects that include improved fat metabolism, enhanced insulin sensitivity in adipose tissue, and potentially direct effects on energy expenditure. The combination produces superior weight loss outcomes compared to either pathway alone.
Here is where the dosing frequency question gets more complex than most people realize.
GIP and GLP-1 receptors can undergo desensitization when exposed to sustained high concentrations of their agonists. Receptor desensitization is a protective mechanism where cells reduce their responsiveness to a signaling molecule when that molecule is present in excessive amounts. At the drug concentrations produced by weekly dosing, the natural peak-and-trough cycle allows partial receptor resensitization during the trough period. Receptors recover some sensitivity before the next dose restores high concentrations.
Five-day dosing compresses this recovery window. The trough is higher and shorter. Receptors have less time to resensitize between doses. Over weeks and months, this could theoretically reduce the effectiveness of each subsequent dose, a phenomenon sometimes called tachyphylaxis. While this specific scenario has not been studied with tirzepatide (because no one has conducted five-day dosing trials), the underlying receptor biology suggests that more is not always better when it comes to GLP-1 and GIP receptor agonism.
The weekly dosing schedule was not chosen arbitrarily. It reflects a pharmacokinetic and pharmacodynamic balance between maintaining therapeutic drug levels and allowing the receptor cycling that preserves long-term effectiveness. Disrupting that balance by shortening the interval introduces unpredictable changes in both drug concentration and receptor responsiveness. For a medication that people may take for months or years, maintaining receptor sensitivity is not a trivial concern. It may be the difference between sustained weight loss over many months and hitting a premature plateau that no dose increase can overcome.
Real-world dosing patterns and what the data shows
While no clinical trial has tested five-day dosing, real-world prescribing data provides some insight into what happens when patients deviate from the weekly schedule. Post-marketing surveillance and pharmacy dispensing records suggest that a subset of patients do inject more frequently than prescribed, either intentionally or through gradual schedule drift. The available data on these patients is limited but consistent with the pharmacological predictions.
Patients who inject more frequently than weekly report higher rates of GI adverse events, particularly nausea and vomiting. They also report running out of medication before their next refill, which creates periods of forced abstinence that produce their own problems. The stop-start pattern of running out early, going without for several days, and then resuming creates more dramatic concentration swings than consistent weekly dosing, potentially worsening the very wearing-off effect that prompted the frequency increase in the first place.
Insurance and pharmacy considerations add another practical dimension. Both brand-name and compounded tirzepatide are dispensed in quantities calculated for weekly use. Injecting every five days means requiring approximately 40% more medication over any given period. Brand-name prescriptions will not cover the additional supply. Compounded formulations may be available in larger quantities, but the cost increase is proportional. For people already managing the financial burden of tirzepatide costs, a 40% increase in consumption adds meaningful expense without proven benefit.
The compounding landscape adds another variable. Not all compounded tirzepatide is created equal. Variations in potency between compounding pharmacies, and even between batches from the same pharmacy, can create the impression that the medication is wearing off faster than it should. Before concluding that your body metabolizes tirzepatide faster than average, consider whether the issue might be inconsistent potency.
Choosing a reputable compounding pharmacy with rigorous quality testing, whether that is Empower, Orderly Meds, or another established provider, helps ensure that each dose delivers consistent potency.
For researchers navigating the complex landscape of tirzepatide sourcing and dosing, SeekPeptides offers in-depth vendor reviews, potency comparison guides, and member-exclusive insights that help ensure every injection delivers what you expect.
What happens if you already started taking tirzepatide every 5 days
If you have been injecting tirzepatide every five days and are reading this guide, the most important step is to transition back to weekly dosing safely rather than stopping abruptly or making additional sudden changes.
The transition is straightforward in most cases. After your last five-day-interval injection, simply wait seven days before your next dose. Then maintain the seven-day schedule going forward. You may notice the wearing-off effect more acutely during that first extended interval because your body has adapted to higher trough concentrations. This is temporary. Within two to three weekly cycles, your body will readjust to the standard pharmacokinetic profile.
If you have been on a five-day schedule for several weeks or longer, be aware that your effective drug exposure has been higher than your nominal dose suggests. Someone injecting 10 mg every five days has been carrying steady-state concentrations comparable to someone taking a higher dose weekly. Returning to seven-day dosing at the same milligram amount may feel like a step backward in appetite suppression, even though it is actually returning you to the intended therapeutic range.
This is a situation where talking to your prescriber is genuinely valuable. They can help you determine whether your current milligram dose is appropriate for weekly dosing, whether dose escalation is warranted, or whether other modifications to your treatment plan would help address the concerns that led you to shorten the interval in the first place. Being transparent about your actual dosing history allows for the most accurate clinical decision-making.
Monitor for any changes in side effects during the transition. If you were experiencing GI symptoms that you attributed to the medication itself, you may find that they improve at weekly dosing as drug concentrations settle into the tested therapeutic range. Conversely, if you were tolerating the five-day schedule without side effects, that does not retroactively make it safe. Absence of acute symptoms does not guarantee absence of harm at the cellular and metabolic level.
Frequently asked questions
Can I take tirzepatide every 5 days if I lower the dose?
Even with a reduced per-injection dose, five-day dosing changes the pharmacokinetic profile in ways that have not been clinically tested. The peak-to-trough dynamics, accumulation ratio, and steady-state concentration all differ from the studied protocol. Any dosing modification should only occur under direct medical supervision. Discuss microdosing approaches with your prescriber if you are interested in lower, more frequent doses.
What is the minimum time between tirzepatide injections?
The prescribing information states a minimum of 72 hours between injections. This applies to one-time schedule adjustments, not permanent frequency changes. The standard schedule remains once weekly, and the 72-hour minimum is a safety floor for occasional early dosing, not a recommended regular interval.
Why does tirzepatide seem to wear off on day 5 or 6?
With a half-life of approximately five days, drug levels decline to about 50% of peak by day five. During the first four weeks before steady state, this decline produces noticeable reductions in appetite suppression. The effect typically improves significantly once steady state is reached around week four, as trough levels are substantially higher at that point.
Will taking tirzepatide every 5 days help me lose weight faster?
Evidence does not support this. The weight loss timeline shows diminishing returns at higher drug exposures. The additional side effect burden from more frequent dosing, particularly nausea, vomiting, and dehydration, could actually slow progress by reducing your ability to eat adequate protein, exercise consistently, and maintain the behaviors that drive long-term results.
Is the 7-day schedule the same for compounded and brand-name tirzepatide?
Yes. The active molecule is identical, and the pharmacokinetics do not change based on the manufacturer. Whether you are using Empower pharmacy tirzepatide, ProRx tirzepatide, or brand-name Mounjaro, the seven-day interval applies equally.
What should I do if I accidentally took my tirzepatide 2 days early?
If at least 72 hours have passed since your last injection, a single early dose is generally acceptable. Return to your regular weekly schedule afterward. If less than 72 hours have passed, contact your prescriber. The guide to taking tirzepatide 2 days early covers this scenario in detail.
Can my doctor prescribe tirzepatide more often than weekly?
Off-label prescribing is at the discretion of individual providers. However, no clinical data supports more frequent than weekly administration, and most prescribers are reluctant to deviate from the FDA-approved schedule due to the unknown risk profile at elevated drug exposures. If you feel your current dose is insufficient, dose escalation within the approved range (up to 15 mg weekly) is the standard clinical approach.
Does the wearing-off effect mean tirzepatide is not working for me?
No. End-of-week appetite return during the first four to eight weeks is a normal pharmacokinetic phenomenon, not a sign of treatment failure. If you are losing weight at a steady pace and experiencing some appetite suppression in the first half of each week, the medication is working. Complete appetite elimination for all seven days is not the goal, and achieving it would not necessarily improve outcomes.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your dosing schedule stay consistent, your appetite management stay strong, and your steady state arrive sooner than you expect.