Feb 8, 2026

Before you start researching cagrilintide, there is something you need to know. Something the headlines skip over. Something buried in the fine print of clinical trial data that could change how you approach this peptide entirely.
The side effects.
Not the vague, hand-waving kind you find on most websites. The real numbers. The actual percentages. The specific adverse events that researchers reported across multiple phase 2 and phase 3 trials involving hundreds of participants. Because cagrilintide is not semaglutide. It is not tirzepatide. It works through an entirely different mechanism, targeting amylin receptors rather than GLP-1 receptors alone, and that means its side effect profile carries its own unique characteristics that deserve careful examination.
Here is the reality. In the REDEFINE 1 phase 3 trial, cagrilintide 2.4 mg monotherapy produced an average body weight reduction of 11.8% over 68 weeks. Impressive. But gastrointestinal adverse events affected a significant portion of participants. Nausea rates ranged from 20% to 47% depending on dose. Injection site reactions occurred at rates dramatically higher than placebo. And when combined with semaglutide in the CagriSema combination, those GI numbers climbed even further, with 79.6% of participants experiencing some form of gastrointestinal event.
This guide breaks down every side effect documented in clinical trials. It covers the common ones you will almost certainly encounter, the uncommon ones worth watching for, and the rare but serious events that require immediate attention. It explains why these side effects happen, how long they typically last, and what strategies researchers use to manage them. Whether you are comparing cagrilintide to other weight loss peptides or evaluating whether the benefit-to-risk ratio makes sense for your goals, this is the complete picture.
How cagrilintide works and why side effects occur
Understanding why cagrilintide causes specific side effects requires understanding how it works at a fundamental level. And that starts with how peptides work in the body.
Cagrilintide is a long-acting amylin analog. Amylin is a hormone that your pancreatic beta cells naturally co-secrete alongside insulin every time you eat. Under normal conditions, amylin does three critical things. It signals your brain to feel full. It slows gastric emptying so food moves through your stomach more gradually. And it suppresses glucagon release, which helps regulate blood sugar after meals.
Natural amylin breaks down within minutes. Cagrilintide does not.
Through acylation technology, researchers engineered cagrilintide to persist in the body for an entire week. One injection. Seven days of activity. That extended duration is what makes weekly dosing possible, but it also means the body is exposed to sustained amylin receptor activation far beyond what natural physiology produces.
The brain connection
Cagrilintide crosses into the brain and activates amylin receptors in several key regions. The area postrema and nucleus of the solitary tract in the hindbrain regulate appetite and induce satiety. The hypothalamus influences hunger signaling and energy balance. And the ventral tegmental area modulates reward-based eating behavior, essentially reducing the pleasure response to food.
This is fundamentally different from how semaglutide or tirzepatide work. Those compounds primarily target GLP-1 receptors. Cagrilintide targets amylin receptors (AMY1, AMY2, AMY3) and calcitonin receptors (CTR). Different receptor targets mean different downstream effects. And different downstream effects mean a different side effect profile.
Why GI side effects happen
The area postrema sits outside the blood-brain barrier. It is one of the few brain regions directly exposed to circulating blood. This makes it extraordinarily sensitive to changes in hormonal signaling. When cagrilintide activates receptors in this region, the same mechanism that suppresses appetite also triggers nausea in some people. The body interprets sustained amylin signaling as a signal to stop eating, and sometimes that signal overshoots into actual nausea.
Gastric emptying slows because that is exactly what amylin is supposed to do. But when you slow gastric emptying with a compound that lasts seven days instead of seven minutes, you get more pronounced and longer-lasting effects on digestion. Food sits in the stomach longer. Bloating follows. Constipation follows. These are not bugs in the system. They are the expected pharmacological consequences of sustained amylin receptor activation.
Understanding this mechanism matters because it explains why most GI side effects are dose-dependent, temporary, and manageable through titration strategies. Your body adjusts. The receptors adapt. The side effects typically diminish as your system finds its new equilibrium.
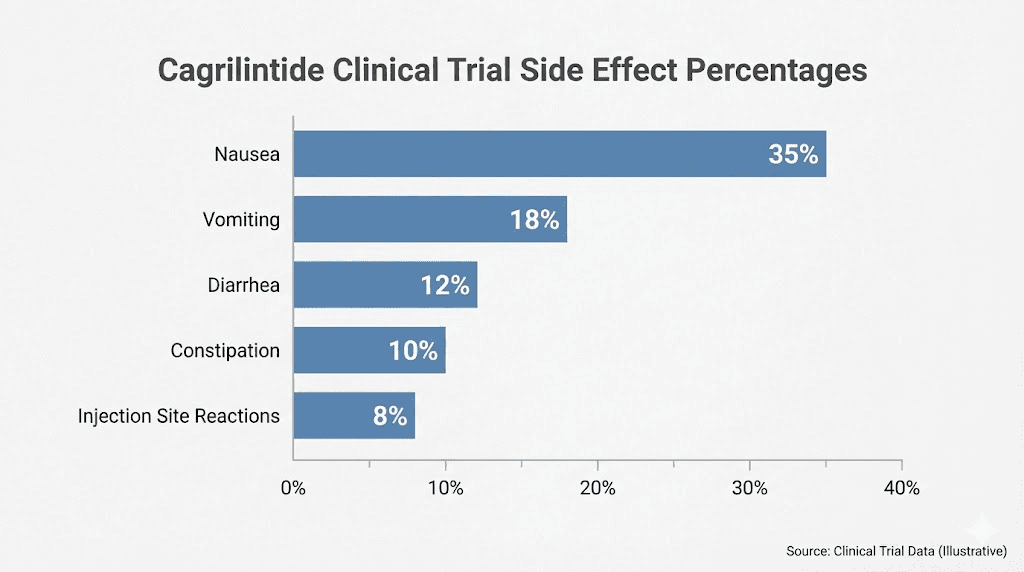
Common gastrointestinal side effects
The GI effects of cagrilintide dominate the side effect conversation, and for good reason. They are the most frequently reported adverse events across every clinical trial conducted to date. But the numbers tell a more nuanced story than simple percentages suggest.
Nausea
Nausea is the single most common side effect of cagrilintide. In the phase 2 dose-finding trial involving 706 participants, nausea rates varied dramatically by dose. At the lowest dose of 0.3 mg weekly, roughly 20% of participants experienced nausea. At the highest dose of 4.5 mg weekly, that number climbed to 47%. The placebo group reported nausea at 18%.
Those numbers need context.
First, the nausea is overwhelmingly mild to moderate. Severe nausea requiring treatment discontinuation was rare in monotherapy trials, with only about 1% of participants in the REDEFINE 1 trial stopping treatment because of adverse events overall. Second, nausea typically peaks during the first few weeks of each dose escalation and then fades as the body adjusts. Most participants who experienced nausea during the initial titration phase reported significant improvement or complete resolution by weeks 4 to 8 at a stable dose.
Compare this to semaglutide, where nausea affects approximately 20% to 44% of users depending on the study. The rates are broadly similar. But here is where cagrilintide has an interesting advantage. In head-to-head comparisons, cagrilintide demonstrated significantly lower rates of vomiting compared to GLP-1 receptor agonists. The odds ratio for vomiting with cagrilintide versus semaglutide or liraglutide was 0.38, meaning cagrilintide caused vomiting at roughly one-third the rate.
For researchers who have struggled with vomiting on GLP-1 medications, this is meaningful data.
Constipation
Constipation ranks as the second most commonly reported GI side effect. It occurs because cagrilintide slows gastric emptying and intestinal transit, the same mechanism that promotes satiety and reduces food intake. When food moves through the digestive system more slowly, the colon absorbs more water from stool, resulting in harder, less frequent bowel movements.
In clinical trials, constipation rates with cagrilintide ranged from approximately 10% to 25% depending on the dose, compared to roughly 5% to 8% with placebo. The good news is that constipation responds well to basic management strategies. Adequate hydration, fiber supplementation, and physical activity address the issue in most cases. Some researchers find that adjusting meal timing and size also helps, eating smaller, more frequent meals rather than large, infrequent ones.
If you are already managing constipation from another peptide or medication, adding cagrilintide could compound the issue. This is particularly relevant for those considering cagrilintide stacked with tirzepatide, as both compounds slow gastric motility.
Diarrhea
Diarrhea might seem contradictory given the constipation discussion, but GI motility disruption can manifest in either direction depending on the individual. In clinical trials, diarrhea occurred in approximately 10% to 15% of participants receiving cagrilintide, compared to roughly 8% with placebo.
Interestingly, the meta-analysis data showed that cagrilintide monotherapy had comparable diarrhea rates to semaglutide. However, the CagriSema combination showed a higher odds ratio for diarrhea at 6.60 compared to semaglutide alone at 2.37. This suggests that combining amylin and GLP-1 receptor agonism creates additive or synergistic effects on gut motility that increase diarrhea risk.
Diarrhea with cagrilintide tends to be episodic rather than chronic. It often appears during dose escalation phases and resolves within days. Persistent diarrhea beyond the titration period warrants evaluation and potential dose adjustment.
Vomiting
This is where cagrilintide actually shines compared to its competitors. Vomiting rates with cagrilintide monotherapy were significantly lower than with GLP-1 receptor agonists. The systematic review and meta-analysis found an odds ratio of 0.38 for vomiting when comparing cagrilintide to semaglutide or liraglutide.
In plain terms, if you take cagrilintide alone, you are roughly 62% less likely to experience vomiting compared to taking semaglutide. This makes cagrilintide a potentially better option for individuals who found vomiting intolerable on GLP-1 medications.
However, this advantage diminishes with combination therapy. When cagrilintide is combined with semaglutide (CagriSema), vomiting rates increase substantially. The REDEFINE 1 trial reported that the odds ratio for vomiting with CagriSema was 9.57 compared to placebo, making it a markedly more common event with the combination.
Abdominal pain and discomfort
Abdominal pain was reported in clinical trials at rates modestly above placebo. The discomfort typically presents as mild cramping or a sensation of fullness in the upper abdomen.
This correlates directly with delayed gastric emptying, as food sitting in the stomach longer creates a sense of pressure and bloating.
Researchers who experience abdominal discomfort often find relief by adjusting their eating patterns. Smaller meals, slower eating, and avoiding high-fat foods that already take longer to digest can reduce the sensation significantly. The pain is not indicative of structural damage. It reflects the pharmacological action of the medication working as designed.
Injection site reactions
Injection site reactions represent the second major category of adverse events with cagrilintide, and this is where the compound differs most dramatically from GLP-1 medications. The clinical data here is striking.
What the trials showed
In the phase 2 trial, injection site reactions with cagrilintide occurred at dramatically higher rates than placebo. The meta-analysis reported an odds ratio of 28.04 for injection site reactions compared to placebo. That is not a typo. Participants receiving cagrilintide were roughly 28 times more likely to experience some form of injection site reaction compared to those receiving placebo injections.
The most commonly reported injection site events included erythema (redness), induration (hardening), pain at the injection site, and itching. These reactions were dose-dependent, with higher doses producing more frequent and sometimes more pronounced reactions.
Why injection site reactions happen
Cagrilintide is formulated at a low pH for subcutaneous injection. This acidic formulation, combined with the acylation modification that extends its half-life, likely contributes to local tissue irritation. The compound essentially creates a depot at the injection site, slowly releasing into circulation over the course of a week. During that time, the surrounding tissue responds to the presence of a foreign protein, producing localized inflammation.
This is fundamentally different from the injection site profile of tirzepatide or semaglutide, which generally produce minimal injection site reactions. For researchers accustomed to peptide injections with clean injection site profiles, the cagrilintide experience may come as a surprise.
Managing injection site reactions
Several practical strategies can minimize injection site issues. Rotating injection sites systematically is essential. Do not inject in the same area consecutively. The abdomen, thigh, and upper arm all serve as viable rotation options. Using proper injection technique matters, including allowing the solution to reach room temperature before injecting and inserting the needle at the correct angle.
Ice applied to the injection site for 2 to 3 minutes before injection can reduce pain. Some researchers report that injecting slowly rather than quickly reduces the burning sensation associated with the low-pH formulation. And while the redness and induration can look alarming, they typically resolve within 24 to 72 hours without intervention.
If injection site reactions persist, worsen, or show signs of infection such as increasing warmth, spreading redness, or pus formation, those warrant immediate medical attention. These signs go beyond normal pharmacological reactions and could indicate a complication.
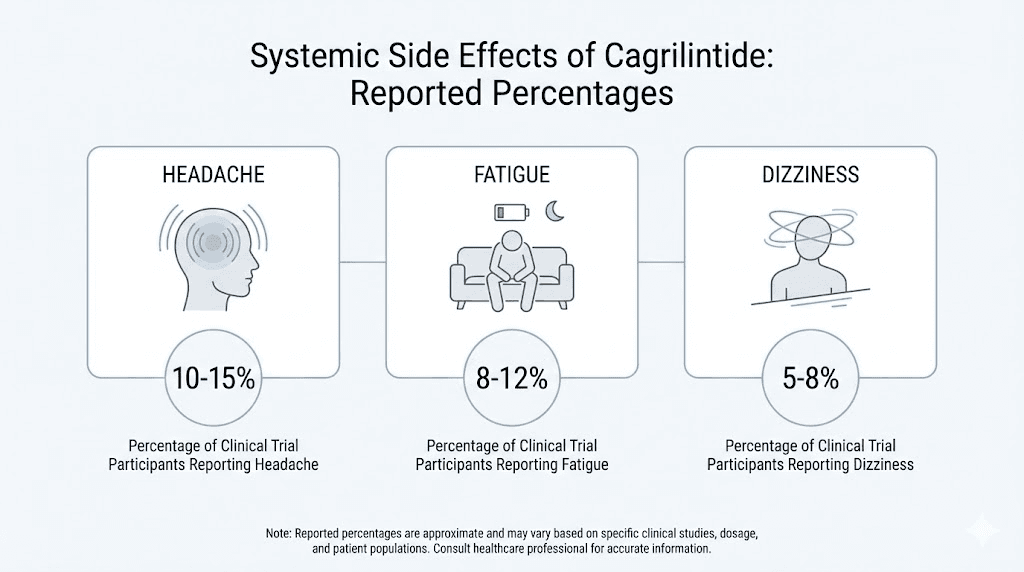
Metabolic and systemic side effects
Beyond the GI tract and injection site, cagrilintide can produce effects that ripple through multiple body systems. These are generally less common than GI events but important to understand.
Headache
Headache was reported in approximately 7% of participants in clinical trials. The mechanism likely relates to changes in appetite regulation and energy metabolism. When caloric intake drops, blood sugar fluctuations can trigger headaches, particularly during the initial adjustment period. Adequate hydration and regular small meals can help mitigate this effect.
For context, headache rates with tirzepatide and semaglutide are similar or slightly higher, suggesting this is more of a general weight-loss medication effect than something specific to cagrilintide pharmacology.
Fatigue
Fatigue affected approximately 20% of participants in the phase 2 trial. This is a substantial number and one that often gets overlooked in discussions focused on GI side effects.
The fatigue likely stems from multiple factors working together. Reduced caloric intake means less available energy. Changes in blood sugar regulation as the body adapts to lower food intake can cause periods of low energy. And the neurological effects of sustained amylin receptor activation in brain regions governing energy homeostasis may contribute directly to feelings of tiredness.
This side effect deserves attention because it can significantly impact quality of life and exercise capacity. Researchers pursuing weight loss with cagrilintide who also maintain intense training protocols should be aware that performance may temporarily decline during the initial weeks. Ensuring adequate protein intake, staying hydrated, and maintaining consistent sleep patterns can help manage fatigue.
If you are comparing options and fatigue is a primary concern, our guide on the best peptides for energy covers compounds that may complement rather than compete with your energy levels.
Dizziness
Dizziness was reported as a mild side effect in clinical trials, though specific incidence rates were less prominently reported than GI events. The mechanism likely parallels headache, relating to shifts in blood sugar, hydration status, and caloric intake rather than a direct pharmacological effect on vestibular function.
Standing up slowly, staying well hydrated, and avoiding skipping meals entirely can help. While cagrilintide suppresses appetite, eating regular small meals maintains more stable blood glucose levels and reduces dizziness episodes.
Decreased appetite (the intended effect that can go too far)
This might sound strange in a list of side effects. After all, appetite suppression is the entire point. But some researchers experience appetite reduction so profound that they struggle to consume adequate nutrition. When you simply do not want to eat anything, ensuring sufficient protein, vitamins, and minerals becomes a challenge.
Inadequate nutrition during weight loss accelerates lean mass loss, impairs recovery, weakens immune function, and can trigger nutrient deficiencies. Researchers using cagrilintide should establish minimum daily protein targets (typically 0.7 to 1.0 grams per pound of body weight) and treat those targets as non-negotiable, regardless of appetite level.
Tools like the SeekPeptides peptide calculator can help researchers plan their protocols around these nutritional requirements. And our guide on peptides for weight loss and muscle gain covers strategies for preserving lean mass during aggressive caloric deficits.
Serious but rare side effects
No discussion of cagrilintide safety would be complete without addressing the serious adverse events. These are rare. But rare does not mean irrelevant, especially when you are making decisions about your own body.
Pancreatitis
Pancreatitis, inflammation of the pancreas, represents a serious potential risk with cagrilintide as it does with other incretin-based therapies. While the mechanism is not fully understood, the theory involves sustained hormonal stimulation of pancreatic cells potentially triggering inflammatory cascades in susceptible individuals.
Clinical trial data showed that serious adverse events, including pancreatitis, were rare and not dose-dependent with cagrilintide. However, the theoretical risk exists, and researchers with a history of pancreatitis or heavy alcohol use should exercise particular caution.
Warning signs include severe, persistent abdominal pain that radiates to the back, nausea and vomiting that do not resolve, and tenderness in the upper abdomen. These symptoms differ from the mild, transient GI discomfort discussed earlier. If they occur, stop the compound and seek medical evaluation immediately.
Gallbladder complications
Acute cholelithiasis (gallstones) was reported in at least one participant receiving 4.5 mg weekly cagrilintide in the phase 2 trial. Gallstone formation during rapid weight loss is well documented across all weight loss interventions, not just pharmaceutical ones. When the body metabolizes fat rapidly, cholesterol concentrations in bile increase, creating conditions favorable for stone formation.
The risk appears more related to the rate and magnitude of weight loss than to cagrilintide specifically. Any intervention producing significant weight loss, whether dietary, surgical, or pharmaceutical, carries elevated gallstone risk. Researchers losing weight rapidly should be aware of symptoms including right upper quadrant pain, nausea after fatty meals, and jaundice.
Thyroid considerations
Cagrilintide carries a theoretical concern regarding thyroid tumors, similar to the boxed warnings on GLP-1 receptor agonists. While this warning is primarily based on rodent studies and has not been confirmed in humans, cagrilintide is contraindicated in individuals with a personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia syndrome type 2 (MEN2).
The relevance of rodent thyroid findings to human physiology remains debated. Rodent thyroid C-cells express high levels of GLP-1 and amylin receptors, while human C-cells express these receptors at much lower levels. Nevertheless, until long-term human data accumulates, the precautionary approach remains appropriate.
If you are researching thyroid-related concerns with weight loss peptides, our peptide safety and risks guide provides broader context for evaluating these considerations.
Heart rate changes
Increased heart rate has been noted as a potential side effect of cagrilintide, consistent with observations across the broader class of weight loss medications. The magnitude of heart rate increase in clinical trials was generally small and not clinically significant for most participants.
However, individuals with pre-existing cardiac conditions should discuss this with their healthcare provider before beginning any weight loss peptide protocol. Heart rate monitoring during the initial titration phase provides useful safety data.
Antibody formation
Greater levels of anti-cagrilintide antibodies were detected with higher doses of cagrilintide in clinical trials. This is the immune system recognizing the modified peptide as foreign and mounting a response against it. However, the data showed that very few of these antibodies were neutralizing against cagrilintide or native amylin.
This means the antibodies generally did not reduce the effectiveness of the medication. And importantly, antibody formation was not associated with any serious allergic reactions in the trials conducted to date. But it is worth monitoring, particularly in researchers using cagrilintide for extended periods, as long-term immunogenicity data remains limited.
Cagrilintide monotherapy vs CagriSema side effects
This distinction matters enormously. The side effect profile of cagrilintide alone versus cagrilintide combined with semaglutide (CagriSema) tells two very different stories.
Monotherapy profile
Cagrilintide as a standalone compound shows a remarkably favorable tolerability profile. In the REDEFINE 1 trial, only 1% of participants discontinued treatment due to adverse events. Compare that to typical discontinuation rates of 5% to 10% with GLP-1 receptor agonists. The GI side effects are present but manageable. The injection site reactions are more prominent than GLP-1s but not treatment-limiting for most people.
The meta-analysis data specifically highlighted that cagrilintide can be considered a promising therapeutic option for weight loss in individuals who may not tolerate GLP-1 receptor agonists or have a lower tolerance for nausea and vomiting. This positions cagrilintide uniquely in the weight loss peptide landscape.
CagriSema combination profile
When cagrilintide is combined with semaglutide, the weight loss results are substantially better, averaging 20.4% body weight reduction versus 11.8% with cagrilintide alone. But the side effect burden increases proportionally.
Gastrointestinal adverse events jumped to 79.6% of participants in the CagriSema group. Nausea hit 55%. Vomiting odds increased dramatically with an odds ratio of 9.57 compared to placebo. Diarrhea showed a higher odds ratio at 6.60 compared to semaglutide alone at 2.37. And the treatment discontinuation rate due to adverse events rose to approximately 6%, still reasonable but six times higher than cagrilintide monotherapy.
The tradeoff is clear. More weight loss. More side effects. The decision depends on individual goals, tolerance, and how much additional weight loss justifies the additional side effect burden.
For researchers evaluating this decision, understanding cagrilintide weight loss outcomes in detail helps contextualize whether the monotherapy or combination approach better suits their situation. And our comparison of mazdutide vs tirzepatide provides perspective on how other dual-agonist approaches handle the efficacy-tolerability balance.

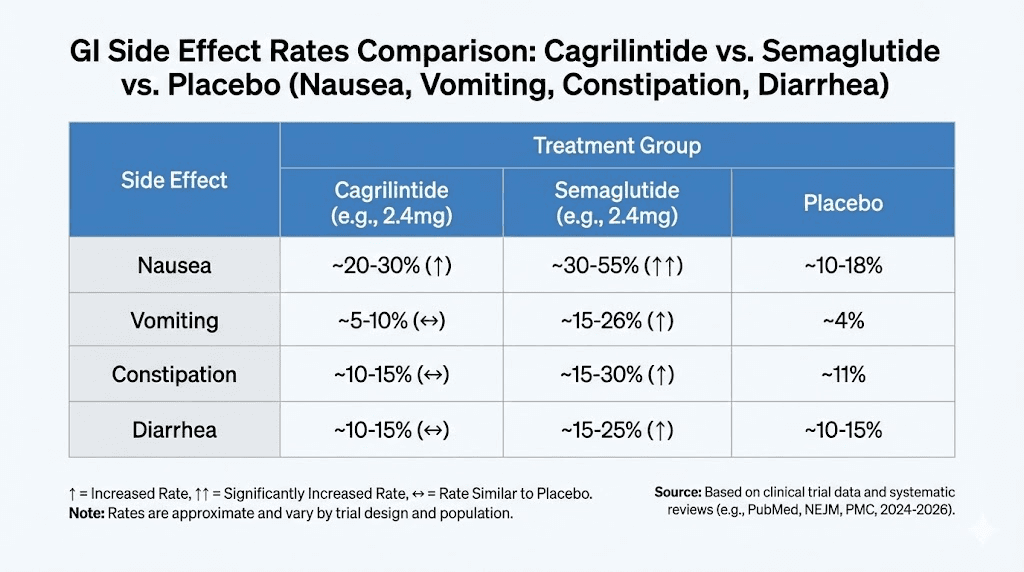
How cagrilintide side effects compare to other weight loss peptides
Putting cagrilintide in context requires comparing it against the compounds researchers are most likely considering as alternatives. Each has its own trade-offs.
Cagrilintide vs semaglutide
Semaglutide (the active ingredient in Ozempic and Wegovy) works primarily through GLP-1 receptor agonism. Semaglutide produces average weight loss of 12% to 15% depending on the trial and dose. Cagrilintide monotherapy produces approximately 11.8% at the 2.4 mg dose.
Side effect comparison:
Nausea: Similar rates between the two compounds
Vomiting: Cagrilintide significantly lower (OR 0.38)
Diarrhea: Comparable rates
Constipation: Similar
Injection site reactions: Cagrilintide substantially higher
Discontinuation due to AEs: Cagrilintide much lower (1% vs approximately 5-7%)
The takeaway: if vomiting was your main problem on semaglutide, cagrilintide may be worth exploring. If injection site reactions bother you, semaglutide has the cleaner profile. For those interested in semaglutide withdrawal symptoms, understanding how switching compounds affects your body is essential.
Cagrilintide vs tirzepatide
Tirzepatide is a dual GIP/GLP-1 receptor agonist that produces weight loss of 15% to 22% depending on the dose and population. It is currently the most effective single-agent weight loss peptide available.
Side effect comparison:
Nausea: Tirzepatide rates range from 12% to 33% depending on dose, comparable to cagrilintide
Vomiting: Tirzepatide higher than cagrilintide
Diarrhea: Similar rates
Injection site reactions: Cagrilintide significantly higher than tirzepatide
Fatigue: Tirzepatide fatigue is commonly reported, comparable to cagrilintide
Some researchers are exploring cagrilintide combined with tirzepatide for potentially enhanced effects. This off-label combination has not been studied in clinical trials and could produce additive GI side effects similar to or exceeding those seen with CagriSema.
Cagrilintide vs retatrutide
Retatrutide is a triple agonist targeting GIP, GLP-1, and glucagon receptors simultaneously. It produced the highest weight loss seen in any phase 2 trial, up to 24% at the highest dose. But its GI side effect profile is also the most aggressive, with nausea rates reaching 40% to 50% and vomiting rates significantly higher than cagrilintide monotherapy.
The retatrutide versus semaglutide comparison and retatrutide dosage chart provide additional context for researchers evaluating the full spectrum of options.
Comparison table
Side Effect | Cagrilintide (mono) | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|---|
Nausea | 20-47% | 20-44% | 12-33% | 40-50% |
Vomiting | Low (OR 0.38 vs GLP-1s) | Moderate | Moderate | High |
Diarrhea | 10-15% | 10-15% | 10-17% | 15-25% |
Constipation | 10-25% | 10-20% | 10-15% | 15-25% |
Injection site reactions | Very high (OR 28 vs placebo) | Low | Low | Low-Moderate |
Fatigue | ~20% | 10-15% | 15-20% | Not well reported |
Discontinuation rate | ~1% | 5-7% | 4-7% | 5-10% |
Weight loss | 11.8% | 12-15% | 15-22% | 17-24% |
This table reinforces a critical point. Cagrilintide monotherapy has the lowest treatment discontinuation rate of any major weight loss peptide in clinical trials. While the weight loss is moderate compared to newer agents, the tolerability profile is genuinely superior for many individuals. SeekPeptides provides detailed protocol comparisons for members evaluating these options.
Managing cagrilintide side effects
Side effects are not inevitable consequences you must simply endure. Smart protocol design and proactive management strategies can significantly reduce their impact.
Dose titration is everything
The single most important factor in managing cagrilintide side effects is proper dose titration. The clinical trial protocol starts at 0.25 mg to 0.3 mg weekly and increases every 4 weeks through escalation steps of 0.6 mg, 1.2 mg, 1.8 mg, and finally 2.4 mg.
This 16 to 20 week titration period exists specifically to allow the body to adapt at each dose level before increasing. Rushing this process, skipping dose levels, or starting at a higher dose to accelerate weight loss dramatically increases side effect severity. Every extra week you spend at each titration step reduces your probability of experiencing intolerable GI effects at the target dose.
For researchers familiar with microdosing approaches, the principle is the same. Slower titration equals better tolerance. And our cagrilintide dosing guide covers the specific steps in detail.
Nausea management strategies
When nausea occurs, these evidence-based approaches help:
Eat smaller, more frequent meals rather than two or three large ones
Avoid high-fat foods that slow gastric emptying further
Stay upright after eating for at least 30 minutes
Ginger supplements or ginger tea have demonstrated anti-nausea effects in clinical studies
Cold foods are often better tolerated than hot foods when nausea is present
Time your dose so that peak nausea coincides with sleep rather than active hours
Stay hydrated with small, frequent sips rather than large volumes
If nausea persists beyond 2 to 3 weeks at a given dose, that is a signal to either extend the titration period or reduce the dose temporarily before attempting re-escalation.
Constipation management
Constipation responds well to proactive management:
Increase water intake to at least 2.5 to 3 liters daily
Add fiber gradually through diet or supplementation (psyllium husk works well)
Maintain physical activity as movement stimulates gut motility
Consider magnesium supplementation (magnesium citrate or oxide), which has natural laxative properties
Avoid foods that worsen constipation such as processed foods, excessive dairy, and refined carbohydrates
These strategies align with general recommendations for anyone experiencing peptide-related digestive changes. Getting the basics right, hydration, fiber, and movement, resolves the majority of constipation cases without additional intervention.
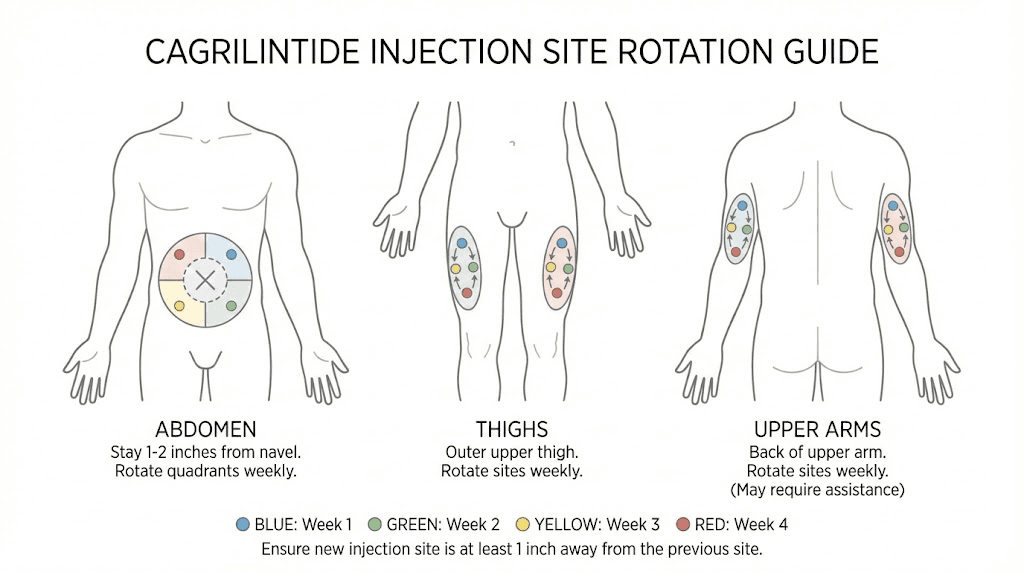
Injection site management
Given the elevated injection site reaction rates with cagrilintide, proactive management is especially important:
Rotate injection sites systematically using a rotation schedule (abdomen, right thigh, left thigh, right arm, left arm)
Allow the solution to reach room temperature before injecting
Inject slowly over 5 to 10 seconds rather than quickly
Apply ice before injection to reduce pain
Do not massage the injection site afterward
Use proper technique as outlined in our peptide injection guide
Keeping a log of injection sites and reactions helps identify whether certain areas are more problematic than others. Some researchers find that abdominal injections produce fewer reactions than thigh injections, though this varies individually.
Fatigue and energy management
Addressing fatigue requires recognizing its root causes:
Ensure adequate caloric intake even though appetite is suppressed, aim for at least 1,200 to 1,500 calories daily minimum
Prioritize protein at every meal to maintain energy and preserve lean mass
Maintain sleep hygiene with consistent bed and wake times
Consider B-vitamin supplementation as reduced food intake may create deficiencies
Time exercise appropriately recognizing that training capacity may temporarily decrease
Our guide on peptides for energy and focus explores complementary options for researchers who need to maintain high performance during weight loss protocols.
Who should be cautious with cagrilintide
While cagrilintide demonstrated a favorable safety profile overall, certain populations should exercise additional caution or avoid the compound entirely.
Individuals with history of pancreatitis
Those with a prior history of pancreatitis face elevated risk with any hormonal weight loss compound that affects pancreatic function. The mechanism of sustained amylin receptor stimulation could theoretically exacerbate underlying pancreatic vulnerability. Medical clearance is essential before considering cagrilintide in this population.
Thyroid cancer history
As discussed, cagrilintide is contraindicated in individuals with personal or family history of medullary thyroid carcinoma or MEN2. This contraindication mirrors the warnings on GLP-1 receptor agonists and reflects precautionary data from animal studies rather than confirmed human risk.
Individuals with severe GI conditions
Gastroparesis, severe inflammatory bowel disease, and other conditions affecting GI motility could be worsened by a compound that further slows gastric emptying. If you already have delayed gastric emptying, adding a long-acting amylin analog on top of that could create problematic levels of GI stasis.
Researchers with gut health concerns should evaluate whether cagrilintide effects would complement or complicate their existing conditions.
Those prone to severe hypoglycemia
While cagrilintide does not directly cause hypoglycemia in most people, its appetite-suppressing effects combined with other glucose-lowering medications could create scenarios where blood sugar drops too low. This is particularly relevant for individuals using cagrilintide alongside insulin or sulfonylureas.
Pregnant or breastfeeding individuals
No safety data exists for cagrilintide use during pregnancy or breastfeeding. Given its effects on appetite, nutrition, and metabolism, use during these periods is not recommended. Weight loss itself is generally contraindicated during pregnancy.
People with eating disorders
Profound appetite suppression in someone with a history of restrictive eating disorders could exacerbate unhealthy patterns. The ability to go extended periods without hunger or eating, while pharmacologically useful for weight loss, can reinforce dangerous behaviors in vulnerable individuals.
The dose-response relationship
One of the most important patterns in cagrilintide clinical data is the clear dose-response relationship for both efficacy and side effects. Understanding this relationship helps researchers make informed decisions about target doses.
Phase 2 dose-finding data
The phase 2 trial tested cagrilintide at 0.3 mg, 0.6 mg, 1.2 mg, 2.4 mg, and 4.5 mg weekly. Here is what emerged:
At 0.3 mg, side effects were minimal. Nausea occurred in about 20% of participants, barely above the 18% placebo rate. Weight loss was modest but present.
At 1.2 mg, the therapeutic sweet spot begins to emerge. Both the 1.2 mg and 2.4 mg doses showed weight loss similar to daily liraglutide 3.0 mg, with manageable side effect profiles.
At 2.4 mg, the target dose for phase 3 trials, the benefit-to-risk ratio appeared optimal. Meaningful weight loss with tolerable side effects and a very low discontinuation rate.
At 4.5 mg, weight loss was notably higher than liraglutide, but GI side effects and injection site reactions increased substantially. This dose provided more weight loss but at the cost of significantly more adverse events.
The practical lesson: higher doses do not always mean better outcomes when you factor in tolerability. A dose that produces 80% of the maximum weight loss with 50% of the side effects is often the smarter choice for sustained use.
Individual variation matters
Clinical trial averages mask enormous individual variation. Some researchers tolerate 2.4 mg from the start with minimal side effects. Others struggle at 0.6 mg. Genetics, gut microbiome composition, baseline GI health, concurrent medications, and individual amylin receptor sensitivity all contribute to this variation.
This is why the titration approach is standard. You cannot predict your individual response from population averages. You discover it through careful, gradual dose escalation with attention to your body's signals at each step.
SeekPeptides members access personalized protocol guidance that accounts for these individual factors, helping researchers navigate titration decisions based on their specific responses rather than generic charts.
What happens when you stop cagrilintide
Understanding discontinuation effects is crucial for anyone considering cagrilintide. Weight regain after stopping weight loss medications is well documented across the entire class of compounds, and cagrilintide is no exception.
Weight regain patterns
The data from similar compounds suggests that most weight lost during treatment is regained within 12 to 18 months of discontinuation. This occurs because the underlying biological drivers of obesity, hormonal setpoints, metabolic adaptation, and appetite regulation, reassert themselves once the pharmacological intervention is removed.
This is not a failure of the medication. It is a reflection of obesity as a chronic condition requiring ongoing management rather than a temporary intervention. The same pattern occurs with semaglutide, tirzepatide, and every other weight loss compound studied long-term.
Rebound appetite
When cagrilintide is discontinued, appetite typically returns. For some, it rebounds beyond pre-treatment levels temporarily before normalizing. This rebound effect occurs because the amylin receptor system, having adapted to sustained agonism, temporarily overcompensates when that stimulation is removed.
Planning for this rebound, maintaining dietary structure, exercise habits, and potentially transitioning to alternative interventions, is essential for anyone discontinuing cagrilintide.
Side effects after stopping
The good news about discontinuation: side effects resolve. GI symptoms typically clear within one to two weeks of the last dose, roughly corresponding to the compound's elimination half-life. Injection site reactions at the final injection site will resolve within their usual 24 to 72 hour timeframe. Fatigue and headache typically improve quickly as eating patterns normalize.
There are no documented withdrawal syndromes specific to cagrilintide, unlike some other compounds where discontinuation produces distinct physiological effects.
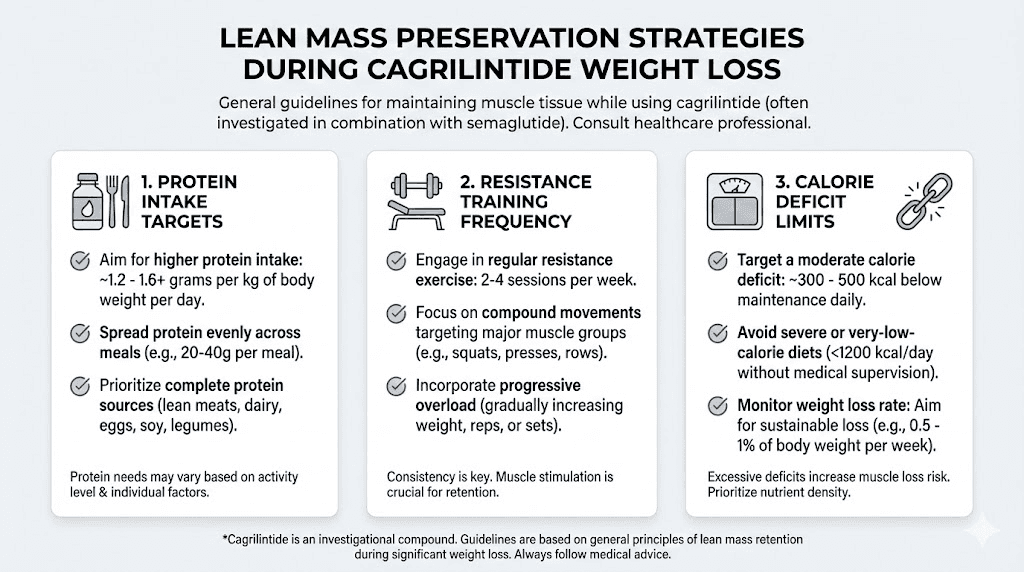
Cagrilintide and lean mass preservation
Every weight loss intervention risks lean mass loss alongside fat loss. This concern is particularly relevant with aggressive caloric deficits, and cagrilintide's appetite-suppressing effects can create substantial caloric deficits.
What the data shows
Clinical trial data suggests that the fat-to-lean mass loss ratio with cagrilintide looks favorable, with most weight lost being body fat rather than lean tissue. However, published phase 3 papers focused primarily on total weight and glycemic control without reporting detailed body composition data comparing lean mass preservation between treatment groups.
This is a gap in the current evidence base. Whether cagrilintide or CagriSema preserves more lean mass than GLP-1 receptor agonists alone remains an open question that future studies need to address.
Protecting lean mass during cagrilintide use
Until definitive data emerges, established principles for lean mass preservation apply:
Resistance training at least 2 to 3 sessions per week provides the strongest stimulus for muscle preservation
Protein intake of 0.7 to 1.0 grams per pound of body weight daily, even when appetite is suppressed
Adequate total calories avoiding deficits greater than 500 to 750 calories below maintenance
Creatine supplementation may provide additional support for muscle maintenance during caloric restriction
For researchers specifically interested in maintaining muscle while losing fat, our guide on the best peptides for muscle growth covers complementary approaches. And the weight loss and muscle gain peptides guide explores how to balance these competing goals.
The safest peptides for muscle growth guide is also relevant for those who want to stack muscle-preserving compounds alongside cagrilintide protocols.
Timeline of side effects: what to expect week by week
Knowing when side effects typically appear and resolve helps researchers prepare mentally and practically. While individual experiences vary, the clinical trial data and pharmacological properties of cagrilintide suggest a general timeline.
Weeks 1 to 4 (initial dose: 0.25-0.3 mg)
At the starting dose, most researchers experience mild or no side effects. Some report slight nausea in the hours following the first injection. Injection site redness may appear and resolve within 24 to 48 hours. Appetite suppression is noticeable but modest. This is the adjustment period where your body begins adapting to exogenous amylin signaling.
Weeks 5 to 8 (dose escalation to 0.6 mg)
Nausea becomes more noticeable in some individuals as the dose doubles. Appetite suppression deepens. Constipation may first appear during this phase. Fatigue can emerge as caloric intake decreases. Injection site reactions become more consistent.
Weeks 9 to 12 (dose escalation to 1.2 mg)
This is often the peak side effect period. The body is adapting to increasingly potent amylin receptor activation. Nausea that appeared at lower doses may intensify temporarily before adapting. GI effects are most pronounced. However, most researchers who tolerate this phase find that subsequent dose increases are easier, suggesting the body develops tolerance over time.
Weeks 13 to 16 (dose escalation to 1.8-2.4 mg)
For many, side effects begin stabilizing. The body has had 12+ weeks to adapt to amylin signaling. Nausea episodes become less frequent and less severe. Injection site reactions follow a predictable pattern that can be managed through rotation. Weight loss accelerates as the full therapeutic dose takes effect.
Weeks 17 and beyond (maintenance at 2.4 mg)
Side effects typically reach their mildest point once a stable maintenance dose is established. GI symptoms that persisted through titration often resolve or become so mild they are barely noticeable. Injection site reactions remain present but predictable and manageable. The main ongoing challenge is maintaining adequate nutrition despite reduced appetite.
This timeline mirrors the general pattern seen with other weight loss peptides. Our guides on semaglutide appetite suppression timelines and tirzepatide effectiveness timelines show similar adaptation patterns.
The regulatory status factor
An important consideration that affects the risk calculus: cagrilintide is not currently FDA-approved as a standalone medication. It remains investigational. Novo Nordisk filed for FDA approval of CagriSema (the cagrilintide plus semaglutide combination) in late 2025, but the standalone compound has not been submitted for approval.
This means several things for researchers:
First, the safety database is smaller than for approved compounds. While phase 2 and phase 3 trials provide substantial data, they involve hundreds to thousands of participants over months. Post-marketing surveillance of approved drugs involves millions of users over years, and often reveals rare side effects that clinical trials miss.
Second, manufacturing standards for research-grade compounds may differ from pharmaceutical-grade products. Purity, consistency, and formulation can vary between sources. This introduces variability in both efficacy and side effects that does not exist with regulated pharmaceuticals.
Third, dosing protocols used in research settings may not perfectly replicate the carefully controlled conditions of clinical trials. Differences in reconstitution, storage, and administration technique can affect both the compound's effectiveness and its side effect profile.
Understanding proper peptide reconstitution and correct storage practices is essential for minimizing variability-related side effects. The peptide reconstitution calculator on SeekPeptides helps ensure accurate preparation.
Long-term safety considerations
The honest answer about long-term cagrilintide safety is that we do not fully know yet. The compound has been studied in trials lasting up to 68 weeks, which provides useful medium-term data but leaves questions about effects beyond 2 years of continuous use.
What we know
Over 68 weeks, the safety profile remained stable. No new safety signals emerged late in the treatment period that were not present early on. Serious adverse events were rare and not dose-dependent. The discontinuation rate due to adverse events remained remarkably low throughout.
What we do not know
Several questions remain unanswered:
Does prolonged amylin receptor agonism produce any adaptive changes in receptor expression or sensitivity that could alter efficacy or safety over years?
What is the long-term impact on gallbladder health with sustained weight loss?
Does antibody formation become clinically relevant with multi-year use?
Are there cardiovascular effects, positive or negative, that only manifest over extended periods?
How does sustained use affect bone mineral density and lean mass over years rather than months?
These unknowns do not invalidate the existing safety data. They contextualize it. The medium-term profile is favorable. The long-term profile requires patience and ongoing monitoring.
Monitoring recommendations
Researchers using cagrilintide should consider regular monitoring including:
Blood work every 3 to 6 months including metabolic panel, lipid panel, liver function, and pancreatic enzymes
Thyroid function tests at baseline and periodically
Body composition assessment to track lean mass versus fat mass changes
Heart rate monitoring particularly during titration
Nutritional status markers including vitamin B12, iron, and vitamin D
The peptide research and studies section provides broader context for evaluating emerging safety data as it becomes available. And SeekPeptides members receive updates on new clinical findings as they are published.
Practical decision framework
Given everything covered in this guide, here is a practical framework for evaluating whether cagrilintide's side effect profile aligns with your goals and tolerance.
Cagrilintide monotherapy may be a good fit if:
You have tried GLP-1 receptor agonists and could not tolerate the vomiting
You prefer a compound with a very low treatment discontinuation rate
You are comfortable with elevated injection site reactions
Your weight loss target is moderate (10-15% body weight)
You want to try a novel mechanism of action (amylin vs GLP-1)
CagriSema may be worth considering if:
You need more aggressive weight loss (15-20%+)
You tolerated GLP-1 receptor agonists reasonably well
You can manage more significant GI side effects
The additional weight loss justifies the additional side effect burden for your situation
You should consider alternatives if:
Injection site reactions are a major concern for you
You have history of pancreatitis or severe GI conditions
You have personal or family history of medullary thyroid carcinoma
You need an FDA-approved option with extensive post-marketing safety data
Fatigue would significantly impact your work or training
Our best peptides for weight loss guide and best fat burning peptide comparison can help researchers who determine that cagrilintide is not the right fit explore other options.
Frequently asked questions
What is the most common side effect of cagrilintide?
Nausea is the most frequently reported side effect, occurring in 20% to 47% of clinical trial participants depending on the dose. It is typically mild to moderate, dose-dependent, and tends to resolve within the first few weeks at each dose level during titration. Proper dose titration significantly reduces nausea severity.
Does cagrilintide cause more side effects than semaglutide?
It depends on the specific side effect. Cagrilintide causes significantly less vomiting than semaglutide (roughly 62% less likely). However, it causes substantially more injection site reactions. Nausea and diarrhea rates are comparable between the two compounds. The overall treatment discontinuation rate is lower with cagrilintide (about 1%) compared to semaglutide (5-7%).
How long do cagrilintide side effects last?
Most GI side effects peak during dose escalation phases and improve within 2 to 4 weeks at a stable dose. Injection site reactions typically resolve within 24 to 72 hours. Fatigue often improves over the first 8 to 12 weeks as the body adapts. By the time researchers reach maintenance dosing, side effects are generally at their mildest.
Can cagrilintide cause hair loss?
Current clinical trial data does not specifically document hair loss as a side effect of cagrilintide. However, significant weight loss from any cause can trigger telogen effluvium, a temporary hair shedding condition related to caloric restriction and nutritional changes. Our GLP-1 hair loss guide covers this phenomenon in detail.
Is cagrilintide safer than tirzepatide?
Both compounds have distinct safety profiles. Cagrilintide has a lower treatment discontinuation rate and causes less vomiting. Tirzepatide has fewer injection site reactions. The "safer" option depends on which specific side effects are most concerning to you and your individual risk factors.
What should I do if I experience severe nausea on cagrilintide?
Severe or persistent nausea should prompt a dose reduction or extension of the titration timeline. Do not push through severe symptoms. Consider reducing to the previous tolerated dose for an additional 4 to 8 weeks before attempting re-escalation. If severe nausea persists at any dose, this compound may not be the right fit.
Does cagrilintide cause pancreatitis?
Pancreatitis was reported as a rare serious adverse event in clinical trials. The risk appears low and was not dose-dependent. However, anyone experiencing severe, persistent abdominal pain radiating to the back should discontinue use and seek immediate medical attention. Individuals with a history of pancreatitis should avoid cagrilintide.
Can I take cagrilintide with other medications?
Because cagrilintide slows gastric emptying, it can affect the absorption of oral medications. This is particularly relevant for medications with narrow therapeutic windows. Any concurrent medication use should be discussed with a healthcare provider who can evaluate potential interactions. Our guide on taking multiple peptides covers stacking considerations.
External resources
Phase 2 clinical trial: Once-weekly cagrilintide for weight management (The Lancet)
REDEFINE 1: Coadministered cagrilintide and semaglutide (NEJM)
Systematic review: Efficacy and safety of cagrilintide alone and in combination (PMC)
For researchers serious about navigating cagrilintide protocols safely, SeekPeptides provides comprehensive, evidence-based guides, dosing tools, and a community of experienced researchers who have worked through these exact questions. Members access personalized protocol support, safety databases, and the latest clinical findings as they emerge.
In case I do not see you, good afternoon, good evening, and good night. May your titration stay gradual, your side effects stay mild, and your protocols stay evidence-based.